| Research Article | ||
Open Vet. J.. 2025; 15(12): 6408-6412 Open Veterinary Journal, (2025), Vol. 15(12): 6408-6412 Research Article Serological detection of Ostertagia ostertagi antibodies in cattle in Mosul cityZeena Dhubyan Mohammed Zaki1* and Wasan. A. Alobaidii21Department of Biology, College of Education for Pure Science, University of Mosul, Mosul, Iraq 2Department of Microbiology, College of Veterinary Medicine, University of Mosul, Mosul, Iraq *Corresponding Author: Zeena Dhubyan Mohammed Zaki. Department of Biology, College of Education for Pure Science, University of Mosul, Mosul, Iraq. Email: zeena.dhubyan [at] uomosul.edu.iq Submitted: 16/07/2025 Revised: 27/10/2025 Accepted: 27/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
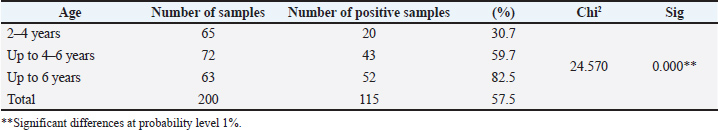
AbstractBackground: Ostertagia ostertagi is considered a great epidemic gastroenteric roundworm that affects cattle in temperate regions and places a central limitation on cattle productivity. This fact is supported by numerous reports. Aim: This study was carried out to detect specific antibodies against O. ostertagi in cow’s milk and investigate the epidemiology of this parasite and its spread in the cows of Mosul city. Methods: Milk samples were separately obtained from 200 cows. The animals suffered from emaciation, weight loss, and watery green diarrhea. Milk was centrifuged at 1,500 rpm for 10 minutes, and the sediment was discarded while the supernatant was stored at −20°C until analysis. An indirect enzyme-linked immunosorbent assay was employed to detect antibodies against O. ostertagi. Results: Among 200 animals, 115 were infected with O. ostertagi, resulting in a total infection rate of 57.5%. The infection rate increased significantly with the age of the animals, with the highest infection rate recorded in animals aged up to 6 years, 82.5% (p < 0.001). The relationship between infection with O. ostertagi showed a significant seropositivity rate of infection in native animals, 69.9% (p < 0.001), compared with imported animals (37.7%). Outdoor animals had a high prevalence of infection with O. ostertagi (70.2%), whereas indoor animals had the lowest prevalence. Some animals were treated with anthelmintic drugs, whereas others were not. Therefore, the animals that were not treated with any anthelmintic drugs showed a high prevalence of infection (64.9%). Conclusion: The infection by O. ostertagi is affected by different parameters, including the age of the animal and breeding management. Moreover, in Mosul city, the infected cows showed more seropositivity to parasites than the unaffected cows. Keywords: Cattle, ELISA, Mosul, Ostertagia ostertagi. IntroductionThe most important cause of parasitic gastroenteritis in cattle is Ostertagia ostertagi infections, the synonym of which is the cow abomasal nematode (Mahmood and Alobaidii, 2022), sometimes referred to as the brown stomach worm. The infection rate of these nematodes depends on the climate and the grazing on pasture that contains the larval stage of this parasite (Tong et al., 2021). After intake and ingestion, larvae develop into adults, become attached to the intestinal lining, and begin to feed on the tissue. Infected animals shed thousands of eggs per day, which are produced by adult worms, contaminating the pasture (Charlier et al., 2023). The disease is mostly chronic in cattle; the affected animals showed clinical signs, including diarrhea, weight loss, malnutrition, decreased reproductive rates, low milk yields, and death. Infected animals with subclinical forms suffer from weight loss. This parasite has a significant effect on the cattle industry (Merlin et al., 2018). According to a recent study, this disease has a negative effect on milk yield, milk fat, and milk protein in high-performance dairy breeds (Kuehne et al., 2025). Cattle helminth contagion is one of the increasing concerns that negatively influence the food industry extensively (Ai et al., 2023). This parasite is a substantial cause of health and economic issues in these animals. The control of this parasite relies on anthelmintic drugs, although the widespread continued exploitation of these drugs caused the selection of resistant parasites (Bartley et al., 2021). An understanding of suitable and correct identification procedures is required to improve herd parasite control and minimize the dispensable use of anthelmintics (Vercruysse et al., 2018). The classical parasitology methods for detecting this parasite are coproparasitological analysis. However, it has not been demonstrated that either of these approaches can quantify the worm load, and they frequently miss the disease’s prepatent period phases (Constancis et al., 2022). Host immunoglobulins specific to nematodes are regarded as one of the most trustworthy indicators of bovine gastrointestinal nematode infections. The important characteristic of this method is early and fast diagnosis, especially in the first grazing season (Opsal et al., 2021). The development of immunity against gastrointestinal parasites in cattle depends on host-related factors, parasite characteristics, and environmental or management conditions that shape the host–parasite interaction. Relevant conditions include grazing practices, herd management, and anthelmintic treatment use. Host-related factors include genetic background, age, sex, and nutritional status (Charlier et al., 2020). The enzyme-linked immunosorbent assay (ELISA) is one of the most effective serological tests to detect the specific antibodies of parasites, which has proven to be a reliable method for investigating or estimating the status of cattle health and production management (Kelly et al., 2019). This test is easy to apply, reasonably priced, provides quick diagnostic procedures to evaluate different infections, and is used to indicate the level of antibodies in milk and plasma samples. For mature cows, ELISA can be used to examine both individual milk samples and bulk tank milk (BTM) samples (Sekiya et al., 2013). Therefore, the objective of this study was to explore for particular antibodies against O. ostertagi in cow’s milk, hence investigating the epidemiology of this parasite and its spread in the cows of Mosul city. Materials and MethodsAnimalsA total of 200 cows were included in this study. Data on age, origin (native or imported), breeding management (indoor or outdoor), and antihelminthic treatment history were recorded for each animal. The animals included in this study presented with clinical signs such as emaciation, weight loss, and watery green diarrhea. SamplesMilk samples were collected from 200 cattle. The samples were centrifuged at 1,500 rpm for 10 minutes. The sediment was discarded, and the supernatant was stored at −20°C until further analysis. Indirect ELISA testThis test was performed according to the manufacturer’s instructions (SVANOVIR O. ostertagi-Ab, Cat. No.: SV-104897). The sensitivity of the test is 84%, and the specificity is 78%. Statistical analysisStatistical analysis was performed using chi-square tests to evaluate the association between infection status and epidemiological variables, such as age, source, breeding management, anthelmintic treatment, and clinical signs. Ethical approvalThe scientific board of the College of Veterinary Medicine at the University of Mosul approved this study; UM.VET.2024.068. ResultsThe results showed that 115 animals were infected with O. ostertagi, with a total infection rate of 57.5%. The infection rate increased with the age of the animals, with the highest percentage of infections recorded in animals aged up to 6 years (82.5%) (Table 1). There was a statistically significant variance between the age groups of the animals. Table 1. Age relationship between infection of O. ostertagi.
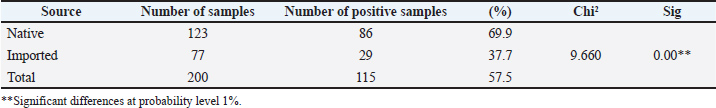
The relationship between O. ostertagi infection and cattle origin showed a high prevalence of infection in native (local) animals (69.9%) compared with imported animals, with a significant variance (37.7%) (Table 2). Table 2. Relationship between infection of O. ostertagi with the source of animals.
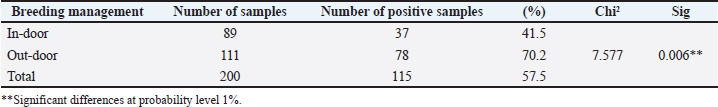
There is a significant variance in the breeding management of animals; the outdoor animals recorded a high prevalence of infection with O. ostertagi (70.2%), while the indoor animals recorded the lowest prevalence of infection (41.5%) (Table 3). Table 3. Relationship between infection of O. ostertagi with Breeding management of animals.
Some animals were treated with anthelmintic drugs, whereas others were not. Therefore, the animals that were not treated with any anthelmintic drugs showed a high prevalence of infection with O. ostertagi (64.9%), whereas the animals treated with anthelmintic drugs recorded the lowest prevalence of infection (50.4%) (Table 4). A statistically significant variance between the age groups of the animals with watery diarrhea had a high prevalence of infection with O. ostertagi. Table 4. Relationship between infection of O. ostertagi with status of anthelmintic of animals.
DiscussionThe high seropositivity result (82.5%) for O. ostertagi, which was clearly observed in this study, reflects the efficiency of the ELISA method in ostertagiosis detection. This method helps in investigating and controlling this parasite (Opsal et al., 2023). It was applied in various studies, and its efficacy was confirmed (Vanderstichel et al., 2010 ; Charlier et al., 2023). Ostertagiosis disease may cause a serious threat to young cattle during their first grazing season, as it may reduce their productivity, especially in dairy cows, through subclinical infections (Opsal et al., 2023). The correlation between the age of cattle and infection with O. ostertagi revealed an enhancement in the infection rate with increasing age of the cattle, with a significant variance; other researchers showed the same results (Ravinet et al., 2017), which demonstrated the appropriateness of treating heifers with targeted selectivity. This allowed for the development of a worldwide strategy, in which GIN management in heifers is linked to GIN management in adult cows (Rose Vineer et al., 2020). The age of cows increases the chance of exposure to parasites. Another reason is that it is due to the method of raising cows, which is milk production, especially since the majority of cow breeds in Iraq are for milk production, which requires raising them for many years. Others, such as Verschave et al. (2014), showed that the establishment rate was lower and decreased with increasing infection dose in younger animals. Animals with concurrent infections had a higher proportion of swallowed larvae that entered hypobiosis (mixed infections). Data from 28 trials (70 experiments) were analyzed. The amount of infection was positively linked with mortality in adults. Based on nine studies (10 experiments), the mean female ratio of O. ostertagi was measured based on individual animal data. We believe that information is currently available on the primary characteristics of the O. ostertagi stage (Santos et al., 2020). The results showed a high infection rate in outdoor cattle with significant variance with indoor animals, and this was described previously by Bosco et al. (2018), who studied the correlation with pasture access and concluded that it was positively correlated, as animals with outdoor access had considerably higher titers of antibodies in their milk than those housed. Other researchers (Forbes et al., 2008) agreed with them and compared the infection rate in some European countries. The differences across nations seemed to reflect various husbandry techniques, especially those pertaining to pasture access. The ELISA was designed to evaluate the serostatus of O. ostertagi and its potential effects on performance and treatment response (Charlier et al., 2023). This is an important advancement in the field of evidence-based care for dairy veterinarians, advisers, and farmers (Singh et al., 2019). Other factors contributing to these differences include greater exposure of outdoor livestock to the infectious stages of parasites compared with indoor animals. This increased risk has been previously described (Pablos-Tanarro et al., 2013) and is associated with grazing management practices and prolonged access to pastures, with higher optical density ratio (ODR) values observed in herds where cows had more than 1 year of exposure to pasture. The comparison between cows treated with anthelmintics and those not treated showed a high percentage of not-treated cows (Ravinet et al, 2017 ). While 115 (27.8%) of the 414 treated animals were deemed "cows to treat." The parity and output level were the markers used to profile these cows and herds because they were significantly more specific than using single indicator profiles and caused lower treatment rates. In particular, the profile "high BTM Ostertagia ODR and low-TEC" improved the specificity more than just considering the BTM ODR score when targeting a herd. Another study (Charlier et al., 2005), which recorded higher ODRs, was linked to the cows' increased pasture exposure (p < 0.001). The purpose of the second model was to assess the impact of the anthelmintic treatment approach and pasture management parameters. Mowing (p=0.002) and later turn-out on pasture (p < 0.001) were both substantially linked to reduced ODRs. The ODR of cows with limited daily grazing time was lower than that of cows with 24 hours of grazing per day (p=0.07). Higher ODRs were substantially correlated with the heifers’ increasing pasture exposure (p=0.001). ODR did not correlate with calf-related management parameters, anthelmintic treatment approach, turn-in time, rotational grazing type, or stocking rate. ConclusionIt becomes obvious that O. ostertagi has a significant impact on infected cows compared to uninfected ones in Mosul city. In addition, different parameters, such as the age of the animals, source, and breeding management, played a significant role in the seropositivity to parasites. AcknowledgmentThe researchers would like to thank the University of Mosul’s College of Veterinary Medicine and veterinary teaching hospital for their support and assistance. Conflict of interestThe authors declare that there is no conflict of interest. Conflict of interestThe authors declare that there is no conflict of interest. FundingThis research was funded by the authors. Authors’ contributionsBoth researchers equally contributed to the design of the experiments, sample preparation, data collection, and manuscript writing. Data availabilityAll data were provided in the manuscript. ReferencesAi, S., Zhang, Z., Wang, J., Wang, X., Liu, C. and Duan, Z. 2023. Prevalence and molecular identification of gastrointestinal nematodes in Qinghai-Tibetan Plateau of China. Vet. Med. Sci. 9(6), 2693–2702. Bartley, D.J., Jewell, N.J., Andrews, L.M., Mitchell, S. and Morrison, A.A. 2021. Molecular and phenotypic characterisation of fenbendazole resistance in a field-derived isolate of Ostertagia ostertagi. Vet. Parasitol. 289, 109319. Bosco, A., Amadesi, A., Morandi, N., Pepe, P., Maurelli, M.P., Cringoli, G. and Rinaldi, L. 2018. Ostertagia ostertiagi antibodies in bulk tank milk from dairy cattle in Italy: a nation-wide survey. Vet. Parasitol. Reg. Stud. Rep. 13, 166–170. Charlier, J., Claerebout, E., Mûelenaere, E.D. and Vercruysse, J. 2005. Associations between dairy herd management factors and bulk tank milk antibody levels against Ostertagia ostertagi. Vet. Parasitol. 133(1), 91–100. Charlier, J., Höglund, J., Morgan, E.R., Geldhof, P., Vercruysse, J. and Claerebout, E. 2020. Biology and epidemiology of gastrointestinal nematodes in cattle. Vet. Clin. North. Am. Food. Anim. Pract. 36(1), 1–15. Charlier, J., Wang, T., Verschave, S.H., Höglund, J. and Claerebout, E. 2023. Review and evaluation of Ostertagia ostertagi antibody ELISA for application on serum samples in first season grazing calves. Animals 13(13), 2226. Constancis, C., Chartier, C., Leligois, M., Brisseau, N., Bareille, N., Strube, C. and Ravinet, N. 2022. Gastrointestinal nematode and lungworm infections in organic dairy calves reared with nurse cows during their first grazing season in western France. Vet. Parasitol. 302, 109659. Forbes, A.B., Vercruysse, J. and Charlier, J. 2008. A survey of the exposure to Ostertagia ostertagi in dairy cow herds in Europe through the measurement of antibodies in milk samples from the bulk tank. Vet. Parasitology 157(1-2), 100–107. Kelly, R.F., Mazeri, S., Hartley, C., Hamman, S.M., Ngu Ngwa, V., Nkongho, E.F., Tanya, V., Sander, M., Ndip, L., Morgan, K.L., Muwonge, A., Handel, I., De Bronsvoort, B.M.C. and Williams, D.J.L. 2019. Assessing the performance of a Fasciola gigantica serum antibody ELISA to estimate prevalence in cattle in Cameroon. BMC. Vet. Res. 15(1), 8. Kuehne, L., Hoedemaker, M., Strube, C., Knubben-Schweizer, G., Springer, A. and Oehm, A.W. 2025. Breed-dependent associations of production characteristics with on-farm seropositivity for Ostertagia ostertagi in dairy cows. Parasit. Vectors. 18(1), 91. Mahmood, M.S. and Alobaidii, W.A. 2022. Molecular detection of Trypanosoma species in sheep and goats in Mosul city. Iraqi J. Vet. Sci. 36(2), 445–449. Merlin, A., Ravinet, N., Madouasse, A., Bareille, N., Chauvin, A. and Chartier, C. 2018. Mid-season targeted selective anthelmintic treatment based on flexible weight gain threshold for nematode infection control in dairy calves. Animal 12(5), 1030–1040. Opsal, T., Toftaker, I., Nødtvedt, A., Robertson, L.J., Tysnes, K.R., Woolsey, I. and Hektoen, L. 2021. Gastrointestinal nematodes and Fasciola hepatica in Norwegian cattle herds: a questionnaire to investigate farmers’ perceptions and control strategies. Acta Vet. Scand. 63(1), 52. Opsal, T., Toftaker, I., Robertson, L., Woolsey, I. and Hektoen, L. 2023. Comparison of diagnostic methods for assessment of Ostertagia ostertagi exposure in Norwegian dairy herds. Acta Vet. Scand. 65(1), 52. Pablos-Tanarro, A., Pérez-Cabal, M.A., Ortega-Mora, L.M. and Ferre, I. 2013. Presence of Ostertagia ostertagi antibodies in bulk tank milk from cattle herds in northern Spain. Vet. Parasitol. 197(1-2), 388–392. Ravinet, N., Lehebel, A., Bareille, N., Lopez, C., Chartier, C., Chauvin, A. and Madouasse, A. 2017. Design and evaluation of multi-indicator profiles for targeted-selective treatment against gastrointestinal nematodes at housing in adult dairy cows. Vet. Parasitol. 237, 17–29. Rose Vineer, H., Morgan, E.R., Hertzberg, H., Bartley, D.J., Bosco, A., Charlier, J., Chartier, C., Claerebout, E., De Waal, T., Hendrickx, G., Hinney, B., Höglund, J., Ježek, J., Kašný, M., Keane, O.M., Martínez-Valladares, M., Mateus, T.L., Mcintyre, J., Mickiewicz, M., Munoz, A.M., Phythian, C.J., Ploeger, H.W., Rataj, A.V., Skuce, P.J., Simin, S., Sotiraki, S., Spinu, M., Stuen, S., Thamsborg, S.M., Vadlejch, J., Varady, M., Von Samson-himmelstjerna, G. and Rinaldi, L. 2020. Increasing importance of anthelmintic resistance in European livestock: creation and meta-analysis of an open database. Parasite 27, 69. Santos, L.L., Salgado, J.A., Drummond, M.G., Bastianetto, E., Santos, C.P., Brasil, B.S.A.F., Taconeli, C.A. and Oliveira, D.A.A. 2020. Molecular method for the semiquantitative identification of gastrointestinal nematodes in domestic ruminants. Parasitol. Res. 119(2), 529–543. Sekiya, M., Zintl, A. and Doherty, M.L. 2013. Bulk milk ELISA and the diagnosis of parasite infections in dairy herds: a review. Ir. Vet. J. 66(1), 14. Singh, B., Flampouri, E. and Dempsey, E. 2019. Electrochemical enzyme-linked immunosorbent assay (e-ELISA) for parasitic nematode Ostertagia ostertagi (brown stomach worm) infections in dairy cattle. Analyst 144(19), 5748–5754. Tong, W., Redman, E.M., Morosetti, A., Chen, R., Kulle, S., Morden, N. and Gilleard, J.S. 2021. Seasonal epidemiology of gastrointestinal nematodes of cattle in the northern continental climate zone of western Canada as revealed by internal transcribed spacer-2 ribosomal DNA nemabiome barcoding. Parasit. Vectors 14(1), 604. Vanderstichel, R., Dohoo, I. and Stryhn, H. 2010. The impact of milk handling procedures on Ostertagia ostertagi antibody ELISA test results. Vet. Parasitol. 169(1-2), 204–208. Vercruysse, J., Charlier, J., Van Dijk, J., Morgan, E.R., Geary, T., Von Samson-himmelstjerna, G. and Claerebout, E. 2018. Control of helminth ruminant infections by 2030. Parasitology 145(13), 1655–1664. Verschave, S.H., Vercruysse, J., Claerebout, E., Rose, H., Morgan, E.R. and Charlier, J. 2014. The parasitic phase of Ostertagia ostertagi: quantification of the main life history traits through systematic review and meta-analysis. Int. J. Parasitol. 44(14), 1091–1104. | ||
| How to Cite this Article |
| Pubmed Style Zaki ZDM, Alobaidii WA. Serological detection of Ostertagia ostertagi antibodies in cattle in Mosul city. Open Vet. J.. 2025; 15(12): 6408-6412. doi:10.5455/OVJ.2025.v15.i12.25 Web Style Zaki ZDM, Alobaidii WA. Serological detection of Ostertagia ostertagi antibodies in cattle in Mosul city. https://www.openveterinaryjournal.com/?mno=271282 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.25 AMA (American Medical Association) Style Zaki ZDM, Alobaidii WA. Serological detection of Ostertagia ostertagi antibodies in cattle in Mosul city. Open Vet. J.. 2025; 15(12): 6408-6412. doi:10.5455/OVJ.2025.v15.i12.25 Vancouver/ICMJE Style Zaki ZDM, Alobaidii WA. Serological detection of Ostertagia ostertagi antibodies in cattle in Mosul city. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6408-6412. doi:10.5455/OVJ.2025.v15.i12.25 Harvard Style Zaki, Z. D. M. & Alobaidii, . W. A. (2025) Serological detection of Ostertagia ostertagi antibodies in cattle in Mosul city. Open Vet. J., 15 (12), 6408-6412. doi:10.5455/OVJ.2025.v15.i12.25 Turabian Style Zaki, Zeena Dhubyan Mohammed, and Wasan. A. Alobaidii. 2025. Serological detection of Ostertagia ostertagi antibodies in cattle in Mosul city. Open Veterinary Journal, 15 (12), 6408-6412. doi:10.5455/OVJ.2025.v15.i12.25 Chicago Style Zaki, Zeena Dhubyan Mohammed, and Wasan. A. Alobaidii. "Serological detection of Ostertagia ostertagi antibodies in cattle in Mosul city." Open Veterinary Journal 15 (2025), 6408-6412. doi:10.5455/OVJ.2025.v15.i12.25 MLA (The Modern Language Association) Style Zaki, Zeena Dhubyan Mohammed, and Wasan. A. Alobaidii. "Serological detection of Ostertagia ostertagi antibodies in cattle in Mosul city." Open Veterinary Journal 15.12 (2025), 6408-6412. Print. doi:10.5455/OVJ.2025.v15.i12.25 APA (American Psychological Association) Style Zaki, Z. D. M. & Alobaidii, . W. A. (2025) Serological detection of Ostertagia ostertagi antibodies in cattle in Mosul city. Open Veterinary Journal, 15 (12), 6408-6412. doi:10.5455/OVJ.2025.v15.i12.25 |