| Research Article | ||
Open Vet. J.. 2025; 15(12): 6423-6433 Open Veterinary Journal, (2025), Vol. 15(12): 6423-6433 Research Article Functional recovery from malnutrition via extracts of Glycine max L. Merr and Brassica oleracea L in rats: Impacts on bodyweight, hematology, and PEPT1 expressionTri Widyawati1*, Dwi R. Anggraini2, Siti Syarifah1, Sri Melinda Kaban1 and Nor Adlin Md. Yusoff31Department of Pharmacology and Therapeutic, Faculty of Medicine, Universitas Sumatera Utara, Medan, Indonesia 2Department of Anatomy, Faculty of Medicine, Universitas Sumatera Utara, Medan, Indonesia 3Department of Toxicology, Advanced Medical and Dental Institute, Universiti Sains Malaysia, Penang, Malaysia *Corresponding Author: Tri Widyawati. Department of Pharmacology and Therapeutic, Faculty of Medicine, Universitas Sumatera Utara, Medan, Indonesia. Email: tri.widyawati [at] usu.ac.id Submitted: 14/07/2025 Revised: 22/10/2025 Accepted: 03/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
AbstractBackground: Malnutrition, particularly protein-energy malnutrition (PEM), remains a global health concern. Functional foods sourced from crops such as Glycine max and Brassica oleracea may offer sustainable solutions. Aim: This study evaluated the effects of G. max ethanol extract (EEG), Brassica oleracea methanol extract (MEB), and their combinations on bodyweight, hematologic status, and molecular outcomes in a rat model of malnutrition. Methods: Male Wistar rats were fed a low-protein diet for 14 days to induce PEM, followed by a 28-day oral treatment phase using EEG, MEB, or their combinations at graded doses. Bodyweight, hematological profiles, and serum iron levels were measured. Small intestine peptide transporter 1 (PEPT1) gene expression was analyzed by immunohistochemistry. Phytochemical content and antioxidant capacity (IC50) of the extracts were also assessed. Results: All treatment groups showed improved weight gain, with the most significant effect observed in the high-dose combination group. Hematological parameters remained stable, and serum iron levels increased in all treated rats. PEPT1 expression was significantly upregulated, especially in the MEB and combination groups. MEB exhibited higher phenolic and flavonoid content and stronger antioxidant activity (IC50=127.58 ppm), while EEG contained notable levels of saponins. Conclusion: EEG and MEB, individually or in combination, improved nutritional markers and enhanced nutrient transporter expression in malnourished rats. These results support their potential as functional food candidates for managing PEM. Keywords: Brassica oleracea, Extract, Glycine max, Malnutrition, PEPT1. IntroductionMalnutrition, encompassing both undernutrition and micronutrient deficiencies, remains a pervasive global health problem, particularly in low- and middle-income countries. It is responsible for a substantial proportion of morbidity and mortality, especially among children under five years of age (Riaz et al., 2022; Bradley et al., 2023). Protein-energy malnutrition (PEM) is a severe condition, particularly in developing nations, where access to sufficient protein and energy sources is limited (Olson et al., 2016). It not only impairs physical growth and development but also compromises cognitive function and immune response (Spieldenner 2010). The need for nutrient-dense food sources to combat malnutrition is now more urgent than ever. Glycine max L. Merr (Fig. 1) is a well-recognized source of protein, dietary fiber, and essential micronutrients, rendering it a potential candidate for addressing malnutrition (Dissanayaka et al., 2021). It has been widely cultivated across different agro-climatic zones, demonstrating its adaptability to diverse environmental conditions. Its ability to fix nitrogen from the atmosphere contributes to improved soil fertility, making it a sustainable crop choice (Robinson et al., 2019). Brassica oleracea (Fig. 1) also boasts a rich nutritional profile, containing vitamins, minerals, and bioactive compounds with antioxidant and anti-inflammatory properties (Key and Drake, 2008).
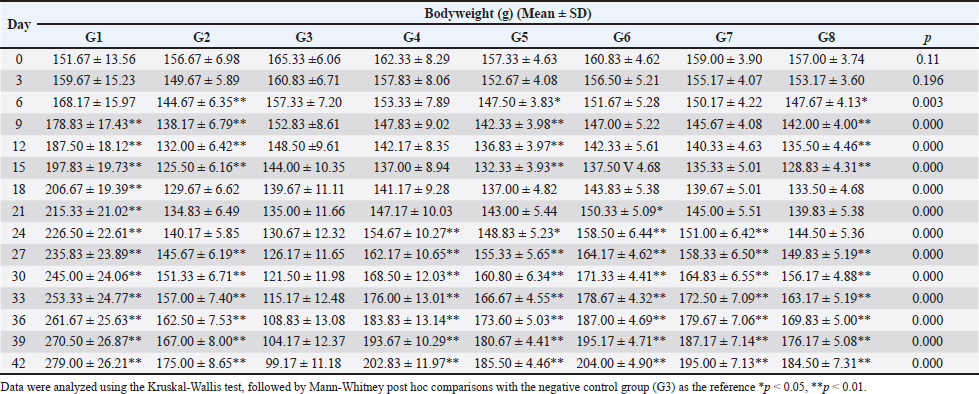
Fig. 1. Glycine max L. Merr and Brassica oleracea. (A: Fresh pods of Glycine max; B: Shelled Glycine max seeds; C: Fresh Brassica oleracea). The functional food concept, which emphasizes the use of food components to promote health and prevent disease, offers a promising avenue for addressing malnutrition. Exploring the potential of G. max and B. oleracea extracts as functional food ingredients could provide innovative strategies for enhancing the nutritional value of diets and mitigating the adverse effects of malnutrition. The integration of nutrient-dense crops like soybean into food blends represents a pragmatic strategy to augment caloric and protein intake, particularly in regions grappling with food insecurity (Amoakoah Twum et al., 2021). However, empirical evidence supporting the functional benefits of these crops in the context of malnutrition is still limited, particularly regarding their effects on growth recovery, hematologic profiles, and molecular pathways involved in nutrient absorption. The peptide transporter 1 (PEPT1) is a proton-dependent oligopeptide transporter that mediates the absorption of di- and tripeptides across the intestinal brush border membrane. Upregulation of PEPT1 is associated with improved protein absorption and utilization, which can be particularly important in states of malnutrition (Spanier 2013; Markovic et al., 2020). It is crucial to emphasize the value of animal-based models in functional food research (Wang et al., 2022) in order to support the rationale of this experimental design in a veterinary setting. Preclinical mouse models are crucial for assessing the physiological and molecular effects of nutritional interventions before they are used in clinical or field settings, as several studies have shown (Vandamme, 2014; Huang et al., 2019; Ferreira-Paes et al., 2021; Whalen et al., 2024) Researchers can evaluate functional recovery, micronutrient status, and gut transporter modulation in a controlled environment by using malnutrition-induced rat models (Sosrosumihardjo et al., 2006; Ferreira-Paes et al., 2021; Pinho et al., 2022), which are specifically well accepted to replicate the pathophysiological conditions of protein-energy malnutrition. Furthermore, preclinical research has demonstrated encouraging outcomes for functional food strategies that target malnutrition, such as those that employ plant-based extracts high in polyphenols, saponins, or essential amino acids (Vignesh et al., 2024). The necessity for such interventions would be further contextualized by including data on the incidence of protein-energy malnutrition worldwide in both human and veterinary contexts, particularly in areas with a high burden of undernutrition or food insecurity. Thus, using this rat model to evaluate the bioactivity of extracts from G. max and B. oleracea offers a pertinent and transferable method for creating new nutraceuticals or dietary supplements that could eventually improve the health of both people and animals. This model not only reflects human malnutrition physiology but also has potential translational value in veterinary contexts. Functional food interventions are increasingly being evaluated for use in livestock, companion animals, and zoo animals to enhance recovery from nutritional deficits. This study aims to investigate the impact of G. max and B. oleracea extracts on body weight, hematologic parameters, and molecular markers related to nutrient absorption in a rat model of malnutrition. By examining these specific outcomes, the study seeks to provide insights into the potential of these plant extracts as functional food ingredients for combating malnutrition and improving overall health outcomes. Next, we conduct phytochemical analyses of the extracts to characterize their bioactive profiles and antioxidant capacities. Such analyses are crucial for linking observed physiological effects to specific phytochemical constituents, providing a mechanistic understanding of how these extracts may exert their functional benefits. Materials and MethodsPreparation of extractsSimplicia of G. max seeds (560 g) obtained from a traditional herbal market in Medan, Indonesia, were macerated in 96% ethanol (4.2 l) for three days at room temperature with occasional stirring. The resulting macerate was concentrated using a rotary evaporator to produce a viscous ethanolic extract of edamame (EEG). Fresh B. oleracea sprouts from Penang, Malaysia, were chopped and macerated in methanol (ratio 2:1 w/v) overnight at 65°C–85°C. The extract was concentrated under low pressure, frozen, and freeze-dried to yield a dry methanolic extract of broccoli (MEB). Both extracts were authenticated at the Medanense Herbal Identification Laboratory (number 2681/MEDA/2024 and 2682/MEDA/2024) and reconstituted in Na CMC before administration. Animal models and induction of malnutritionMale Wistar rats (150–200 g) were acclimatized for 1 week under standard laboratory conditions (12-hour light/dark cycle, temperature 22°C–25°C, relative humidity 50%–60%) with ad libitum access to water and a standard pellet diet. Malnutrition was caused by administering a low-protein diet (LPD) to the rats for 14 days. The LPD consists of 104.4 kcal of calories, 20 g of granulated sugar, 5 g of wheat flour, 1 g of maize oil, and 19 g of cornstarch, as shown by the prior study (Anggraeny et al., 2016). Treatment procedure and groupingFollowing the 14-day malnutrition induction, the rats were randomly assigned to eight experimental groups (n=6 per group) for a subsequent 28-day treatment period: G1: Normal control group (standard diet, no treatment) G2: Diet recovery group (standard diet only) G3: LPD control (malnutrition control, no treatment) G4: EEG 40 mg/kg body weight (BW) G5: MEB 12 mg/kg BW G6: Combination of EEG 40 mg/kg + MEB 12 mg/kg BW (high dose) G7: Combination of EEG 20 mg/kg + MEB 6 mg/kg BW (medium dose) G8: Combination of EEG 10 mg/kg + MEB 3 mg/kg BW (low dose) Treatments were administered once daily by oral gavage. Bodyweight was measured every three days to monitor growth recovery. Hematologic profile assessmentAt the end of the treatment period (day 42), rats were anesthetized, and blood samples were collected via cardiac puncture. Hematological parameters, including hemoglobin concentration, hematocrit levels, total iron (Fe), and Total Iron Binding Capacity (TIBC), were analyzed. Intestinal PEPT1 expression by immunohistochemistry (IHC)After euthanasia, small intestine segments were collected, fixed in 10% neutral buffered formalin, and embedded in paraffin. Sections (5 μm thick) were prepared and mounted on glass slides. Immunohistochemical staining was performed using anti-PEPT1 primary antibody (BT LAB Cat: BT-AP07059) to assess the expression of the peptide transporter 1. Visualization was achieved using a DAB chromogen detection system. Staining intensity and distribution were semi-quantitatively scored by a pathologist. The total IHC score was calculated using the formula: Total Score=Staining Area × Staining Intensity. The percentage of tissue-stained brown was used to determine the staining area, with the following scoring system: 0%–4%=1; 5%–20%=2; 21%–40%=3; 41%–60%=4; 61%–80%=5; 81%–100%=6. The staining intensity was graded based on the shade of brown observed in the cells: light brown=1; moderate brown=2; dark brown=3. The resulting total scores were classified as follows: Mild (1–6), Moderate (7–12), and Strong (13–18). Phytochemical screening and quantificationQualitative phytochemical screening was conducted on the extracts to detect the presence of major secondary metabolites, including alkaloids, saponins, tannins, flavonoids, and terpenoids, following standard phytochemical tests. Total phenolic content was measured using the Folin-Ciocalteu method with gallic acid as the reference standard at a wavelength of 745 nm, expressed in milligrams of gallic acid equivalents (mg GAE/g extract). Meanwhile, total flavonoid content was determined through the aluminum chloride colorimetric assay using quercetin as the reference at 740 nm, expressed in milligrams of quercetin equivalents (mg QE/g extract). In vitro antioxidant activityThe antioxidant potential of the extract was assessed using the DPPH radical scavenging method, which determines the extract's ability to reduce DPPH radicals by measuring the percentage of radical scavenging activity and calculating the IC50 value. The absorbance of the samples was then measured at 515 nm with a UV-Vis spectrophotometer. The IC50 value, reflecting the concentration of extract required to achieve 50% inhibition of DPPH radicals, was used to evaluate antioxidant capacity. For interpreting the antioxidant activity based on the IC50 value, the following classification was applied: IC50 < 50 ppm: Very strong; 50–100 ppm: Strong; 100–150 ppm: Moderate; 150–200 ppm: Weak antioxidant activity. This classification provides a clear reference for comparing the antioxidant strength of the tested extract. Data analysisAll data analyses were performed using SPSS version 23. The Kruskal-Wallis test was used for variables with non-normal distributions, and post hoc comparisons were conducted with the Mann-Whitney U test. For within-group comparisons, the Wilcoxon signed ranks test and paired samples t-test were employed. A p-value of less than 0.05 was considered statistically significant. Ethical approvalAll experimental procedures were conducted following ethical standards for animal research and approved by the Institutional Animal Ethics Committee, Universitas Sumatera Utara (approval number 0655/KEPH-FMIPA/2024, Approved on July 26, 2024.). ResultsEffect of extracts on bodyweight changesTable 1 showed that all experimental groups began the study with relatively similar baseline weights (151–165 g). During the initial 15-day malnutrition induction phase, the normal group consistently gained weight, while the LPD (G3) and control groups (G2) exhibited a steady decline. All treatment groups, whether receiving EEG, EMB, or their combinations, also experienced progressive weight loss, confirming effective induction of malnutrition. By day 15, all treatment groups reached their lowest body weights, with the LPD group showing the most severe decline. Upon entering the 28-day intervention phase, recovery trends began to emerge. The normal group continued its upward trajectory, while the LPD group showed further deterioration. In contrast, all extract-treated groups gradually gained weight, with the high-dose combination group (EEG 40 mg/kg + MEB 12 mg/kg BW) demonstrating the earliest and most substantial recovery, followed by medium- and low-dose combinations. From day 21 onward, all treatment groups consistently outperformed the DRP group, and by day 30, the high-dose combination surpassed 170 g. By day 42, the normal group had reached 279.00 ± 26.21 g, while the LPD group declined to a critical low of 99.17 ± 11.18 g. All extract-treated groups achieved significant recovery, with the high-dose combination group peaking at 204.00 ± 4.90 g, indicating its superior efficacy in reversing weight loss induced by protein-energy malnutrition. Table 1. Effect of extract on bodyweight changes.
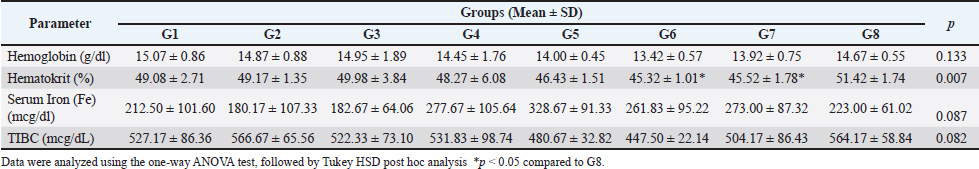
Effect of extracts on hematological profilesAs shown in Table 2, hematological and iron profile assessments on day 42 revealed varying responses across treatment groups. Hemoglobin levels in the normal control group were highest (15.07 ± 0.86 g/dl), whereas the lowest value was recorded in the high-dose combination group (13.42 ± 0.57 g/dl). Other treatment groups, including single and lower combination doses, showed hemoglobin levels ranging between 13.92 and 14.67 g/dl, which were slightly lower than both normal and recovery controls. Table 2. Effect of extracts on hematological profile.
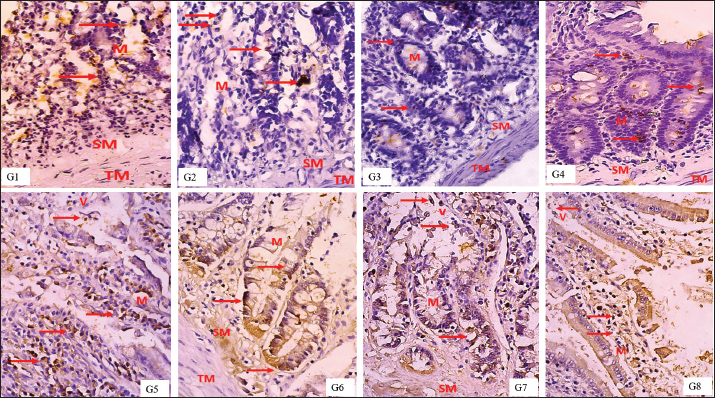
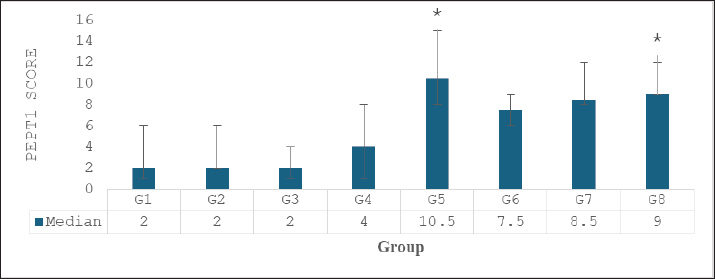
Hematocrit values were relatively stable across all groups, with the lowest observed in the high-dose combination group (45.32% ± 1.01%) and the highest in the low-dose combination group (51.42% ± 1.74%), indicating variability in erythropoietic response. While DRP-fed groups did not show sharp reductions in hematocrit, there was a modest downward trend in groups receiving single extracts compared to normal. Interestingly, serum iron (Fe) concentrations increased substantially in all extract-treated groups. The highest level was observed in the MEB group (328.67 ± 91.33 μg/dl), followed by EEG (277.67 ± 105.64 μg/dl) and EEG 20 mg/kgBW + MEB 6 mg/kgBW (273.00 ± 87.32 μg/dl). These values were markedly higher than the LPD group (182.67 ± 64.06 μg/dl) and normal control (212.50 ± 101.60 μg/dl), suggesting an enhancement in iron absorption or mobilization following extract administration. Total iron-binding capacity was slightly increased in most groups, with the highest value in the LPD + diet normal group (566.67 ± 65.56 μg/dl). A notable decrease was seen in the high-dose combination group (447.50 ± 22.14 μg/dl), possibly reflecting improved iron utilization and reduced iron-deficiency signals. These findings indicate that both EEG and EMB extracts, alone or in combination, positively influence iron metabolism, though dose-dependent effects on hemoglobin restoration may require further elucidation. Effect of extracts on intestinal PEPT1 expressionThe IHC analysis of PepT1 expression in the small intestine revealed distinct staining patterns across the experimental groups. In the normal group (G1), mild PepT1 expression was observed, with weak brown staining in the mucosa (M) and sparse expression in the tunica muscularis (TM) and submucosa (SM). The positive control group (G2) showed similar mild expression, while the negative control group (G3) had minimal to absent PepT1 staining. In the treatment groups, EEG40 (G4) and MEB12 (G5) displayed moderate PepT1 expression, indicated by brown staining in the mucosa and extending to the TM and SM. The most intense PepT1 expression was seen in the high-dose combination group (G6), where dense brown staining localized predominantly in the mucosal villi (V) and mucosa (M), with notable extension into the TM and SM layers. This suggests robust transporter activity in response to the combined extract. The medium-dose (G7, EEG20+MEB6) and low-dose (G8, EEG10+MEB3) combination groups also showed pronounced PepT1 expression, although less intense than in G6. In these groups, staining was consistently observed in the villi and mucosa, indicating enhanced nutrient absorption capacity. Overall, these histological observations confirm that the combined extracts, particularly at high doses, effectively upregulated PepT1 expression in the small intestine. This enhanced transporter activity is consistent with the observed bodyweight recovery in these groups, highlighting the therapeutic potential of these functional food extracts in addressing protein-energy malnutrition. The results in Figure 2 showed varying median scores for different treatment groups. The normal, positive control, and negative control groups had comparable median scores of 2, with narrow minimum-maximum ranges. Among the treatment groups, EMB (10.5; 8–15) and EEG10+MEB3 (9; 9–12) exhibited the highest median values, suggesting notable effects of the MEB and its low-dose combination with EEG. The high-dose combination group had a median score of 7.5 (6–9), while EEG alone was lower at 4 (1–8). The progressive increase in median scores in combination groups suggests a potential synergistic effect between EEG and MEB, particularly at lower or moderate doses, which may reflect enhanced biological activity or improved nutrient uptake. Overall, these findings support the role of these functional foods in modulating key outcomes in malnutrition recovery, balanced synergistic effect without oversaturation or suppression.
Fig. 2. PEPT1 expression in the rat intestine by immunohistochemistry. PepT1 protein expression (brown colour in nucleus, red arrow in the figure), Tunica Muscularis (TM), Mucosa (M), villous (V). IHC, 400x.
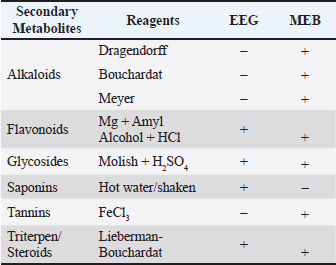
Fig. 3. PEPT1 score. Data were analyzed using the Kruskal-Wallis test, *p < 0.05 compared to Normal Control Group (G1). Overall, these results indicate that both EEG and MEB extracts can upregulate PEPT-1 expression in the small intestine of malnourished rats, with broccoli extract demonstrating superior potency. The observed expression patterns support the role of these functional foods in enhancing intestinal peptide absorption, which may contribute to improved recovery outcomes in protein-energy malnutrition. Phytochemical screening and quantificationPhytochemical screening (Table 3) showed that EEG contained saponins, flavonoids, glycosides, and triterpenes/steroids, while MEB contained alkaloids, tannins, flavonoids, glycosides, and triterpenes/steroids. These bioactive compounds suggest complementary antioxidant and immunomodulatory effects, aligning with the observed benefits in weight recovery and intestinal PEPT1 expression, supporting their potential as functional foods. Table 3. Phytochemical screening.
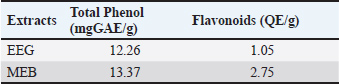
Furthermore, quantitative analysis of the extracts revealed that MEB contained higher total phenolic content (13.37 mg GAE/g extract) compared to the EEG, which contained 12.26 mg GAE/g extract. MEB also exhibited superior total flavonoid content (2.75 mg QE/g extract), significantly higher than that of EEG (1.05 mg QE/g extract). The elevated phenolic and flavonoid contents in MEB suggest enhanced antioxidant potential, aligning with its observed biological activities and supporting its role as a potent functional food component (Table 4). Table 4. Total phenol and flavonoids of extracts.
In vitro antioxidant activityThe antioxidant activities of the extracts were quantified using the DPPH radical scavenging assay, as indicated by their IC550 values. The MEB exhibited an IC50 of 127.58 ppm, suggesting moderate antioxidant strength. In contrast, the EEG has an IC50 of 180.67 ppm, indicating weaker antioxidant capacity (Table 5). Table 5. In vitro antioxidant activity by DPPH assay.
These differences align with the phytochemical profiles of the extracts, as MEB demonstrated higher total phenolic and flavonoid content compared to EEG. The enhanced antioxidant activity of MEB underscores the importance of these polyphenolic compounds in neutralizing free radicals and mitigating oxidative stress, which is crucial in addressing the pathophysiological consequences of malnutrition. The moderate antioxidant activity of EEG, despite its saponin-rich composition, suggests that while saponins contribute to some protective effects, they may be less potent in direct radical scavenging compared to phenolic-rich MEB. Overall, these results reinforce the complementary role of these extracts in functional food interventions, with MEB offering notable antioxidant protection that could support recovery in protein-energy malnutrition models. DiscussionThese results align with previous studies highlighting the nutritional and functional benefits of Glycine max and B. oleracea. For example, the improvement of bodyweight recovery in malnourished rats receiving soybean extract, linked to its high-quality protein and bioactive peptides (Dang et al., 2023). Similarly, previous research has indicated that a combined eel and soy-based tempeh flour can improve nutritional status (Sabrina et al., 2022). Consumption of Brassica oleracea and bodyweight changes are associated with the rich array of phytonutrients, including glucosinolates and flavonoids, which could contribute to the observed weight recovery through enhanced metabolic function and reduced oxidative stress (Castelão-Baptista et al., 2021). Although an overall increase in serum iron levels was observed across the treatment groups, this did not consistently translate into improvements in hemoglobin concentration or hematocrit values. Several factors may account for this discrepancy. First, the 28-day treatment duration, although sufficient to demonstrate weight recovery and molecular changes, may have been insufficient to reverse anemia or stimulate erythropoiesis fully. Second, the high saponin content observed in the G. max extract EEG could have interfered with iron absorption by forming insoluble complexes with dietary iron, potentially counteracting the positive effects of Brassica oleracea MEB, which exhibited higher phenolic and flavonoid content. This antagonistic interaction may explain the lack of additive effects on hematological indices in the combination groups. Third, the current study did not assess hepcidin levels, a central regulator of systemic iron homeostasis. As an iron-regulatory hormone, hepcidin plays a pivotal role in modulating intestinal iron absorption and systemic iron homeostasis. The absence of hepcidin data in this study limits the interpretation of how phytochemical constituents may have influenced iron utilization and erythropoiesis (D’Angelo, 2013). The upregulation of PEPT1 expression observed in this study may be partially explained by the presence of bioactive compounds such as flavonoids, polyphenols, and saponins found in the administered plant extracts. Flavonoids and polyphenols (Widyawati et al., 2023) are known to exert regulatory effects on intestinal transporters through their antioxidant and anti-inflammatory properties (Cueva et al., 2017; Wang et al., 2022; Singh et al., 2025). Specifically, these compounds can modulate signaling pathways such as nuclear factor kappa B (NF-κB) and p38 mitogen-activated protein kinase (MAPK), both of which are implicated in the transcriptional regulation of PEPT1 in enterocytes (Saklatvala, 2004; Spanier 2013; Roudsari et al., 2019; Teng et al., 2021). Antioxidants may protect intestinal cells from oxidative stress-induced downregulation of PEPT1 (Warsi et al., 2014) and restore transporter function under inflammatory or malnourished conditions. Furthermore, data indicate that certain polyphenols can improve food absorption by preserving membrane integrity and stimulating cellular stress response pathways that elevate transporter production (Riaz Rajoka et al., 2021). For example, catechins and quercetin have been shown to influence gene expression of intestinal transporters, including PEPT1, in vitro and in vivo (Yu et al., 2016; Hai et al., 2020; Yu et al., 2023). On the other hand, saponins—predominantly present in G. max—may exert a dual role. While they exhibit immunomodulatory properties, excessive saponin exposure could disrupt membrane fluidity or interact with membrane proteins, potentially modulating transporter availability or function (Melzig et al., 2021). The precise molecular mechanisms through which these compounds act on PEPT1 remain an active area of investigation, but current findings support their role in nutrient absorption recovery under malnourished conditions. Further studies employing transcriptomic or proteomic approaches are needed to confirm the pathway-level interactions involved. The combined administration of G. max (EEG) and B. oleracea (MEB) in this study suggests a potential synergistic interaction in promoting nutritional recovery, as reflected by improved bodyweight and enhanced PEPT1 expression in the intestinal mucosa. This finding may be partly explained by the modulatory effects of polyphenols and flavonoids on intestinal transporters, as reported in studies implicating p38 MAPK and NF-κB pathways (Saklatvala, 2004; Spanier 2013). Saponins may further enhance this activity through interactions with membrane cholesterol, thereby promoting transporter function (del Hierro et al., 2018). This may also be attributed to the complementary phytochemical profiles of the two extracts. MEB is rich in polyphenols and flavonoids, which possess potent antioxidant properties, while EEG is notable for its high saponin content. The co-administration of flavonoid-rich and saponin-containing extracts may enhance nutrient absorption through distinct but potentially cooperative mechanisms at the intestinal level. Flavonoids have been shown to stabilize epithelial tight junctions (Suzuki and Hara, 2011) and modulate nutrient transporter expression (Murugan et al., 2022), while in low concentrations, saponins may facilitate intestinal absorption by increasing membrane permeability through interaction with cholesterol and modulation of membrane fluidity, as previously attributed to their amphiphilic and surfactant-like properties (del Hierro et al., 2018). At the molecular level, flavonoids may upregulate transporter genes via activation of signaling pathways such as PI3K/Akt or MAPK, while saponins might increase bioavailability by interacting with membrane cholesterol, altering fluidity, and promoting nutrient uptake (Mansuri et al., 2014; Liao et al., 2021). These complementary mechanisms may underlie the improved PEPT1 expression observed in combination groups. However, high concentrations of saponins may exert antagonistic effects by forming insoluble complexes with minerals or disrupting membrane proteins (Sharma et al., 2023), which might explain the non-linear or plateaued effects in higher-dose combinations. Study limitationsThis study has several limitations that warrant consideration. First, while IHC analysis provided evidence of increased PEPT1 protein expression, the absence of complementary mRNA expression data (e.g., via RT-qPCR) limits the ability to confirm transcriptional regulation. Assessing mRNA levels would provide a more comprehensive understanding of gene-level modulation by the extracts. Second, the study did not include inflammatory markers such as tumor necrosis factor-alpha or interleukin-6, which could have helped elucidate systemic recovery mechanisms, particularly given the known immunomodulatory role of saponins and polyphenols. Evaluating such parameters would offer greater insight into the anti-inflammatory and homeostatic effects of the interventions in the context of malnutrition recovery. Clinical and veterinary relevanceBeyond human health applications, the findings of this study have potential implications for veterinary practice and animal nutrition. The observed improvements in bodyweight and nutrient transporter expression suggest that G. max and B. oleracea extracts could be formulated as functional feed additives or nutraceutical supplements for animals recovering from malnutrition. Such interventions may be particularly beneficial for production animals (e.g., poultry, ruminants) under intensive farming conditions, shelter animals recovering from neglect, or zoological species requiring tailored nutritional support. The bioactive properties of these plant extracts—particularly their antioxidant and nutrient absorption-enhancing effects—offer a promising approach to improving health outcomes in various veterinary contexts. ConclusionCombining G. max and B. oleracea extracts enhances weight recovery, hematologic stability, and intestinal PepT1 expression in malnourished rats. MEB's higher phenolic and flavonoid content underpins its superior antioxidant capacity. These findings support the synergistic potential of extracts as functional foods to address protein-energy malnutrition. AcknowledgmentThanks to Sasniwati Sari Hasibuan, S.Farm for assistance as laboratory assistant. Conflicts of interestThe authors declare that there is no conflict of interest. FundingThis research was supported by the Talenta Grant Lembaga Penelitian Universitas Sumatera Utara No: 13/UN5.4.10.S/PPM/KP-TALENTA/RB1/2024, 31 Mei 2024. Author contributionsTri Widyawati and Nor Adlin Md Yusoff carried out the concept, design, and drafting of the article. The acquisition of data, interpretation of data, statistical analysis, critical revision, supervision, and final approval were carried out by Tri Widyawati, Siti Syarifah, Sri Melinda Kaban, and Dwi Rita Anggraini. Data availabilityAll data generated and analyzed are included in this research article. ReferencesAmoakoah Twum, L., Ocloo, F.C.K., Duah-Bisiw, D. and Odai, B.T. 2021. Determining the effect of heat treatment on iron-fortified soybean gari blend and its potential bioavailability. Sci. Afr. 12, e00763. Anggraeny, O., Dianovita, C., Putri, E.P., Sastrina, M. and Dewi, R.S. 2016. Korelasi pemberian diet rendah protein terhadap status protein, imunitas, hemoglobin, dan nafsu makan tikus wistar jantan. Indones. J. Hum. Nutr. 3(2), 107–113. Bradley, M., Melchor, J., Carr, R. and Karjoo, S. 2023. Obesity and malnutrition in children and adults: a clinical review. Obesity Pillars 8, 100087. Castelão-Baptista, J., Barros, A., Martins, T., Rosa, E. and Sardão, V. 2021. Three in one: the potential of Brassica by-products against economic waste, environmental hazard, and metabolic disruption in obesity. Nutrients 13(12), 4194. Cueva, C., Gil-Sánchez, I., Ayuda-Durán, B., González-Manzano, S., González-Paramás, A., Santos-Buelga, C., Bartolomé, B. and Moreno-Arribas, M. 2017. An integrated view of the effects of wine polyphenols and their relevant metabolites on gut and host health. Molecules 22(1), 99. Dang, Y., Ren, J., Guo, Y., Yang, Q., Liang, J., Li, R., Zhang, R., Yang, P., Gao, X. and Du, S.K. 2023. Structural, functional properties of protein and characteristics of tofu from small-seeded soybeans grown in the Loess Plateau of China. Food. Chem. X. 18, 100689. D'Angelo, G. 2013. Role of hepcidin in the pathophysiology and diagnosis of anemia. Blood Res. 48(1), 10. Del Hierro, J.N., Herrera, T., Fornari, T., Reglero, G. and Martin, D. 2018. The gastrointestinal behavior of saponins and its significance for their bioavailability and bioactivities. J. Funct. Foods 40, 484–497. Dissanayaka, D.M.S.B., Rankoth, L.M., Gunathilaka, W.M.N.D., Prasantha, B.D.R. and Marambe, B. 2021. Utilizing food legumes to achieve iron and zinc nutritional security under changing climate. J. Crop Improv. 35(5), 700. Ferreira-Paes, T., Seixas-Costa, P. and Almeida-Amaral, E.E. 2021. Validation of a feed protocol in a mouse model that mimics marasmic malnutrition. Front. Vet. Sci. 8, 757136. Hai, Y., Zhang, Y., Liang, Y., Ma, X., Qi, X., Xiao, J., Xue, W., Luo, Y. and Yue, T. 2020. Advance on the absorption, metabolism, and efficacy exertion of quercetin and its important derivatives: absorption, metabolism and function of quercetin. Food. Front. 1(4), 420–434. Huang, W., Percie du Sert, N., Vollert, J., and Rice, A.S.C. 2019. General Principles of Preclinical Study Design. In A. Bespalov et al. (Eds.), Good Research Practice in Non-Clinical Pharmacology and Biomedicine (Handbook of Experimental Pharmacology, Vol. 257). Springer Nature, Berlin, Germany, pp. 55–71. Key, S.K.C. and Drake, P.M.W. 2008. Genetically modified plants and human health. J. R. Soc. Med. 101(6), 290–298. Liao, Y., Li, Z., Zhou, Q., Sheng, M., Qu, Q., Shi, Y., Yang, J., Lv, L., Dai, X. and Shi, X. 2021. Saponin surfactants used in drug delivery systems: a new application for natural medicine components. Int. J. Pharm. 603, 120709. Mansuri, M.L., Parihar, P., Solanki, I. and Parihar, M.S. 2014. Flavonoids in modulation of cell survival signalling pathways. Genes Nutr. 9(3), 400. Markovic, M., Ben-Shabat, S. and Dahan, A. 2020. Prodrugs for improved drug delivery: lessons learned from recently developed and marketed products. Pharmaceutics 12(11), 1031. Melzig, M.F., Bader, G. and Loose, R. 2001. Investigations of the mechanism of membrane activity of selected triterpenoid saponins. Planta. Med. 67(1), 43–48. Murugan, S.K., Bethapudi, B., Raghunandhakumar, S., Purusothaman, D., Nithyanantham, M., Mundkinajeddu, D. and Talkad, M.S. 2022. A flavonoid-rich standardized extract of Glycyrrhiza glabra protects intestinal epithelial barrier function and regulates the tight-junction proteins expression. BMC. Complement. Med. Ther. 22(1), 38. Olson, M.E., Sankaran, R.P., Fahey, J.W., Grusak, M.A., Odee, D. and Nouman, W. 2016. Leaf protein and mineral concentrations across the “miracle tree” genus Moringa. PLos One 11(7), 7. Pinho, R.M., Garas, L.C., Huang, B.C., Weimer, B.C. and Maga, E.A. 2022. Malnourishment affects gene expression along the length of the small intestine. Front. Nutr. 9, 787351. Riaz Rajoka, M.S., Thirumdas, R., Mehwish, H.M., Umair, M., Khurshid, M., Hayat, H.F., Phimolsiripol, Y., Pallarés, N., Martí-Quijal, F.J. and Barba, F.J. 2021. Role of food antioxidants in modulating gut microbial communities: novel understandings in intestinal oxidative stress damage and their impact on host health. Antioxidants 10(10), 1563. Riaz, N., Yousaf, Z., Yasmin, Z., Munawar, M., Younas, A., Rashid, M., Aftab, A., Shamsheer, B., Yasin, H., Najeebullah, M. and Simon, P.W. 2022. Development of carrot nutraceutical products as an alternative supplement for the prevention of nutritional diseases. Front. Nutr. 8. Robinson, G.H.J., Balk, J. and Domoney, C. 2019. Improving pulse crops as a source of protein, starch and micronutrients. Nutr. Bull. 44(3), 202. Roudsari, N.M., Lashgari, N.A., Momtaz, S., Farzaei, M.H., Marques, A.M. and Abdolghaffari, A.H. 2019. Natural polyphenols for the prevention of irritable bowel syndrome: molecular mechanisms and targets; a comprehensive review. DARU. J. Pharm. Sci. 27(2), 755. Sabrina, N., Rizal, M., Nurkolis, F., Hardinsyah, H., Tanner, M.J., Gunawan, W.B., Handoko, M.N., Mayulu, N., Taslim, N.A., Puspaningtyas, D.S., Noor, S.L., Yusuf, V.M., Permatasari, H.K. and Radu, S. 2022. Bioactive peptides identification and nutritional status ameliorating properties on malnourished rats of combined eel and soy-based tempe flour. Front. Nutr. 9. Saklatvala, J. 2004. The p38 MAP kinase pathway as a therapeutic target in inflammatory disease. Curr. Opin. Pharmacol. 4(4), 372. Sharma, K., Kaur, R., Kumar, S., Saini, R.K., Sharma, S., Pawde, S.V. and Kumar, V. 2023. Saponins: a concise review on food-related aspects, applications and health implications. Food Chem. Adv. 2, 100191. Singh, A., Kaur, P., Kumar, M., Shafi, S., Upadhyay, P. K., Tiwari, A., Tiwari, V., Rangra, N. K., Thirunavukkarasu, V., Kumari, S., Roy, D., Ghosh, M., Arora, N., Sharma, N. and Garg, Y. 2025. The role of phytochemicals in modulating the gut microbiota: Implications for health and disease. Med. Microecol. 100125. Sosrosumihardjo, R., Firmansyah, A., Rasad, A., Harjodisastro, D., Ridwan, E., Wanandi, S.I. and Retno, D. 2006. Effects of realimentation on small intestinal morphology and disaccharidase activity in malnutrition Sprague-Dawley rats. Med. J. Indones. 208–216. Spanier, B. 2013. Transcriptional and functional regulation of the intestinal peptide transporter PEPT1. J. Physiol. 592(5), 871. Spieldenner, J. 2010. Health economic perspectives of pediatric malnutrition: Determinants of innovative progress. Nestlé Nutr. Inst. Workshop Ser. 97. Suzuki, T. and Hara, H. 2011. Role of flavonoids in intestinal tight junction regulation. J. Nutr. Biochem. 22(5), 401–408. Teng, H., Deng, H., He, Y., Lv, Q. and Chen, L. 2021. The role of dietary flavonoids for modulation of ATP binding cassette transporter mediated multidrug resistance. eFood 2(5), 234. Vandamme, T. 2014. Use of rodents as models of human diseases. J. Pharm. Bioallied Sci. 6(1), 2. Vignesh, A., Amal, T.C., Sarvalingam, A. and Vasanth, K. 2024. A review on the influence of nutraceuticals and functional foods on health. Food. Chem. Adv. 5, 100749. Wang, X., Qi, Y. and Zheng, H. 2022. Dietary polyphenol, gut microbiota, and health benefits. Antioxidants 11(6), 1212. Warsi, J., Elvira, B., Bissinger, R., Shumilina, E., Hosseinzadeh, Z. and Lang, F. 2014. Downregulation of peptide transporters PEPT1 and PEPT2 by oxidative stress responsive kinase OSR1. Kidney. Blood. Press. Res. 39(6), 591. Whalen, C., Verma, A., Kurashima, K., Carter, J., Nazzal, H. and Jain, A. 2024. Novel models for assessing and pathophysiology of hepatic ischemia–reperfusion injury mechanisms. Medicina 60(9), 1507. Widyawati, T., Sari, D.K., Sumantri, I.B., Mustanti, L.F. and Yusoff, N.A. 2023. Phytochemical screening and DPPH assay of Talas Padang (Colocasia gigantea) ethanol leaf extract. IOP Conf. Ser. Earth Environ. Sci.,1241(1), 12016. Yu, B., Jiang, Y., Jin, L., Ma, T. and Yang, H. 2016. Role of quercetin in modulating chloride transport in the intestine. Front. Physiol. 7, 549. Yu, J., Hu, G., Guo, X., Cao, H. and Zhang, C. 2023. Quercetin alleviates inflammation and energy deficiency induced by lipopolysaccharide in chicken embryos. Animals 13(13), 2051. | ||
| How to Cite this Article |
| Pubmed Style Widyawati T, Anggraini DR, Syarifah S, Kaban SM, Yusoff NAM. Functional recovery from malnutrition via extracts of Glycine max L. Merr and Brassica oleracea L in rats: Impacts on bodyweight, hematology, and PEPT1 expression. Open Vet. J.. 2025; 15(12): 6423-6433. doi:10.5455/OVJ.2025.v15.i12.27 Web Style Widyawati T, Anggraini DR, Syarifah S, Kaban SM, Yusoff NAM. Functional recovery from malnutrition via extracts of Glycine max L. Merr and Brassica oleracea L in rats: Impacts on bodyweight, hematology, and PEPT1 expression. https://www.openveterinaryjournal.com/?mno=270852 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.27 AMA (American Medical Association) Style Widyawati T, Anggraini DR, Syarifah S, Kaban SM, Yusoff NAM. Functional recovery from malnutrition via extracts of Glycine max L. Merr and Brassica oleracea L in rats: Impacts on bodyweight, hematology, and PEPT1 expression. Open Vet. J.. 2025; 15(12): 6423-6433. doi:10.5455/OVJ.2025.v15.i12.27 Vancouver/ICMJE Style Widyawati T, Anggraini DR, Syarifah S, Kaban SM, Yusoff NAM. Functional recovery from malnutrition via extracts of Glycine max L. Merr and Brassica oleracea L in rats: Impacts on bodyweight, hematology, and PEPT1 expression. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6423-6433. doi:10.5455/OVJ.2025.v15.i12.27 Harvard Style Widyawati, T., Anggraini, . D. R., Syarifah, . S., Kaban, . S. M. & Yusoff, . N. A. M. (2025) Functional recovery from malnutrition via extracts of Glycine max L. Merr and Brassica oleracea L in rats: Impacts on bodyweight, hematology, and PEPT1 expression. Open Vet. J., 15 (12), 6423-6433. doi:10.5455/OVJ.2025.v15.i12.27 Turabian Style Widyawati, Tri, Dwi R. Anggraini, Siti Syarifah, Sri Melinda Kaban, and Nor Adlin Md. Yusoff. 2025. Functional recovery from malnutrition via extracts of Glycine max L. Merr and Brassica oleracea L in rats: Impacts on bodyweight, hematology, and PEPT1 expression. Open Veterinary Journal, 15 (12), 6423-6433. doi:10.5455/OVJ.2025.v15.i12.27 Chicago Style Widyawati, Tri, Dwi R. Anggraini, Siti Syarifah, Sri Melinda Kaban, and Nor Adlin Md. Yusoff. "Functional recovery from malnutrition via extracts of Glycine max L. Merr and Brassica oleracea L in rats: Impacts on bodyweight, hematology, and PEPT1 expression." Open Veterinary Journal 15 (2025), 6423-6433. doi:10.5455/OVJ.2025.v15.i12.27 MLA (The Modern Language Association) Style Widyawati, Tri, Dwi R. Anggraini, Siti Syarifah, Sri Melinda Kaban, and Nor Adlin Md. Yusoff. "Functional recovery from malnutrition via extracts of Glycine max L. Merr and Brassica oleracea L in rats: Impacts on bodyweight, hematology, and PEPT1 expression." Open Veterinary Journal 15.12 (2025), 6423-6433. Print. doi:10.5455/OVJ.2025.v15.i12.27 APA (American Psychological Association) Style Widyawati, T., Anggraini, . D. R., Syarifah, . S., Kaban, . S. M. & Yusoff, . N. A. M. (2025) Functional recovery from malnutrition via extracts of Glycine max L. Merr and Brassica oleracea L in rats: Impacts on bodyweight, hematology, and PEPT1 expression. Open Veterinary Journal, 15 (12), 6423-6433. doi:10.5455/OVJ.2025.v15.i12.27 |