| Research Article | ||
Open Vet. J.. 2026; 16(2): 834-840 Open Veterinary Journal, (2026), Vol. 16(2): 834-840 Research Article Clinical and sero-diagnosis of feline leptosprosis in the sleman region of Yogyakarta, IndonesiaGuntari Titik Mulyani1, Dyah Ayu Widiasih2* and Roza Azizah Primatika21Department of Internal Medicine, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia 2Department of Veterinary Public Health, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia *Corresponding Author: Dyah Ayu Widiasih. Department of Veterinary Public Health, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia. Email: dyahaw [at] ugm.ac.id Submitted: 15/10/2025 Revised: 30/12/2025 Accepted: 16/01/2026 Published: 28/02/26 © 2026 Open Veterinary Journal
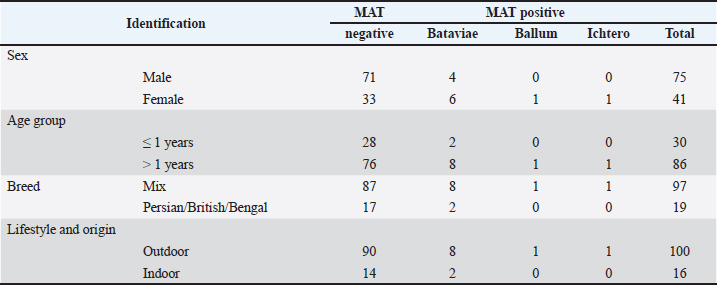
AbstractBackground: Leptospirosis is a zoonotic disease caused by pathogenic Leptospira spp., which can infect a wide range of mammalian hosts. Transmission to humans occurs not only from rodents and wildlife but also from companion animals, such as cats, living in close contact with humans. The occurrence and clinical expression of leptospirosis in animals vary according to species, age, immune status, and serovar infection. In this context, cats may represent an underrecognized component in the epidemiology of leptospirosis in endemic areas. Aim: This study aimed to estimate the seroprevalence of leptospirosis in cats, identify circulating Leptospira serovars, and descriptively characterize clinical signs and selected renal and hepatic laboratory findings in naturally exposed cats in Sleman Regency, Indonesia. Methods: A total of 116 pubertal cats from Sleman Regency, Special Region of Yogyakarta, Indonesia, were clinically examined. Three millilitres of blood were collected for the MAT, routine hematology, and biochemical analyses, including renal biomarkers [blood urea nitrogen (BUN), creatinine, symmetric dimethylarginine, hepatic biomarkers alanine aminotransferase (ALT), aspartate aminotransferase (AST), total bilirubin, albumin, and globulin], and the inflammatory marker feline serum amyloid A (fSAA). Leptospirosis was diagnosed at a MAT titer ≥1:100 against 14 Leptospira serovars. The seropositivity or infection rate was calculated as the proportion of MAT-positive samples, which represents prior exposure or infection rather than clinical disease. Results: Of the 116 cats, 12 (10.34%) were seropositive for leptospirosis. Serovar Bataviae was predominant (83.34%), while Ballum and Icterohaemorrhagiae each accounted for 8.33%. Most seropositive cats (66.67%) were clinically normal at the time of sampling, whereas 25% showed nonspecific signs such as anorexia, fever, icterus, lethargy, vomiting, or diarrhea. One cat (8.33%) presented with clinical signs consistent with renal involvement. The hematological parameters were within the reference ranges in more than half of the seropositive cats (58.33%), whereas leukocytosis indicative of inflammation was observed in 25% and mild anemia in 16.67%. Renal biomarkers (BUN, creatinine) remained within the reference intervals in 91.67% of seropositive cats, indicating minimal renal involvement at the time of sampling. Hepatic alterations were more frequently observed, with elevated ALT and AST levels in 25% of cats and increased total bilirubin or altered albumin-to-globulin ratios in 41.67% of cats. Elevated fSAA concentrations were detected in 33.33% of seropositive cats, indicating subclinical inflammatory responses. Conclusion: This study demonstrates the presence of feline leptospiral exposure in Sleman Regency, with a seroprevalence of 10.34%, predominantly associated with Leptospira interrogans serovar Bataviae. Although most infections were subclinical, variation in clinical and laboratory findings among seropositive cats highlights the heterogeneous nature of feline leptospirosis. The detection of pathogenic serovars in outwardly healthy cats indicates an epidemiological role for cats as subclinical carriers, underscoring the need for surveillance and the integration of feline leptospirosis monitoring within a One Health framework. Keywords: Cats, Indonesia, Leptospira interrogans, One Health, Serovar Bataviae. IntroductionLeptospirosis is a zoonotic disease caused by Leptospira Gram-negative bacteria. It is transmitted through contact with the urine of infected animals or with Leptospira-contaminated environments (Murillo et al., 2020). Leptospirosis has a wide geographic distribution; warm air, moist/wet soil, and alkaline pH are suitable climates for Leptospira development. The incidence of leptospirosis is 100 times higher in tropical countries than in subtropical countries (Adler and De La Peña Moctezuma, 2010). Leptospirosis can be classified as pathogenic Leptospira based on the structural heterogeneity of lipopolysaccharide surface molecules; more than 300 serovars grouped into 24 antigenically related serogroups have been identified (Davignon et al., 2023). This means that animals previously infected with only one serovar have immunity against that specific serovar. The dominant Leptospira serovar in one region may differ from that in other regions (Chadsuthi et al., 2017). Cats are susceptible to Leptospira spp. infection; however, many seropositive cats show no clinical signs and are therefore not recognized as reservoir hosts. The clinical signs of feline leptospirosis include anorexia, lethargy, dehydration, weight loss, polyuria and polydipsia, vomiting, hematuria, uveitis, lameness, ascites, and hepatomegaly (Arbor et al., 2012). Some cats with antibodies against Leptospira species have kidney disease-related signs and histopathological evidence of renal inflammation (Schuller et al., 2015). Despite increasing reports of feline leptospirosis worldwide, data from Southeast Asia remain scarce, and no previous studies have documented the serovar-specific pathogenicity of Leptospira interrogans in cats in Indonesia. Leptospira spreads through the bloodstream after penetrating through the skin/mucosa (leptospiremia phase). Primary lesions develop in the endothelium of small blood vessels; in the liver, they attach to the endothelial lining of hepatic sinusoids and invade hepatocytes, resulting in jaundice and liver dysfunction, with varying degrees of severity among species (more severe in dogs and humans, relatively mild in cats). Leptospira causes local ischemia in the kidneys, leading to tubular necrosis (Adler and De La Peña Moctezuma, 2010). Renal colonization occurs in most infected animals because the bacteria replicate and persist in the renal tubular epithelial cells. This process causes cytokine release and inflammatory cell recruitment, which triggers nephritis (Craig et al., 2015; Murillo, et al., 2020). Chronic interstitial nephritis, which can lead to chronic kidney damage, has been reported in cats infected with Leptospira (Samrot et al., 2021). After 10 days of infection, Leptospira enters the tubular lumen and is excreted in the urine for several days to months. Cats can shed Leptospira in their urine and act as reservoirs or incidental hosts during transmission (Hartmann et al., 2013; Schuller et al., 2015; Murillo et al., 2020). Several studies have described the specific pathogenicity of serovar and its impact on domestic cats in the tropics (Miotto et al., 2024). Despite the evidence of the spread of Leptospira in cats, data on the specific pathogenicity of serovar and subclinical organ damage in domestic cats in Indonesia are still inadequate. This study aimed to determine the seroprevalence and identify the Leptospira serovars infecting domestic cats in Sleman Regency, Indonesia, and to evaluate the associated clinical signs and renal and hepatic alterations in naturally exposed cats. Materials and MethodsStudy design and locationThis study used an analytical observational design with cross-sectional sampling. Sampling was conducted from June to August 2025 in Sleman District, Yogyakarta, Indonesia. Laboratory analyses were performed at the Faculty of Veterinary Medicine, Prof. Soeparwi Veterinary Hospital, Universitas Gadjah Mada; accredited private diagnostic laboratories; and the Veterinary Assembly and Modernization Center. Animals used and samplingA total of 116 domestic cats (>6 months old) were included in this study, comprising 87 clinically healthy cats and 29 clinically ill cats. Clinically ill cats presented with one or more of the following signs: fever, anorexia, lethargy, vomiting, diarrhea, icterus, dehydration, or polyuria and polydipsia. All cats underwent a complete physical examination. Approximately 3 ml of blood was aseptically collected from the cephalic vein. Ethylenediaminetetraacetic acid-anticoagulated blood was used for hematological analysis, while serum samples were used for serological and biochemical examinations. Serological testingThe MAT was performed using 14 pathogenic Leptospira serovars, and results were interpreted using a cutoff titer of ≥1:100 following OIE/WOAH (2021) guidelines. We prepared and standardized commercial live antigen panels according to the manufacturer’s instructions and tested them following the OIE/WOAH guidelines (2021). A reciprocal MAT titer ≥1:100 was interpreted as positive. Hematology and biochemistry data analysisThe infecting Leptospira serovars, observed clinical effects, and organ disorders in seropositive cats were descriptively analyzed, following standard epidemiological analytical approaches (Thrusfield, 2018). Data analysisDescriptive statistics, including frequency distributions, percentages, means, and standard deviations, were used to determine the infection rate and summarize clinical, hematological, and biochemical findings. Inferential statistical tests were not applied because the study aimed to describe patterns rather than assess associations. All analyses were performed using SPSS version 25.0 (SPSS Inc., Chicago, IL, USA). Ethical approvalAll ethical considerations were considered in this study; however, the study was approved by the Animal Ethics Committee, Faculty of Veterinary Medicine, Universitas Gadjah Mada (No. 69/EC-FKH/Int/2025). ResultsA total of 116 samples were collected and tested by MAT against 14 Leptospira serovars: MAT results showed that 12 (10.34%) of 116 samples were positive for leptospirosis with titers ranging from 1/100 to 1/1600. Table 1 presents the identification of serovars in the 12 seropositive samples. Table 1. Identification of sample and MAT results of 12 leptospirosis seropositive cats in Sleman Regency, Indonesia.
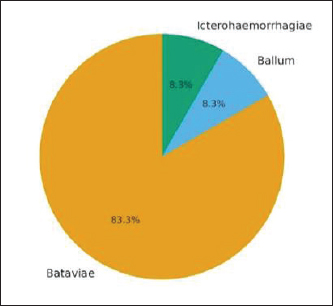
Leptospira infection was dominated by serovar Bataviae (83.34%) with an antibody titer of 1/1,600, Ballum (8.33%) with a titer of 1/100, and Ichterohaemorrhagiae (8.33%) with a titer of 1/1,600, 1/100, and 1/100, respectively. Most cats with leptospirosis were adults (88.33%) and female (66.67%). All cats were domestic or mixed domestic cats with outdoor or semi-outdoor access. Figure 1 shows the distribution of L. interrogans serovars in 12 seropositive catsIn Sleman Regency, Indonesia. Serovar Bataviae predominated (83.3%), followed by the ballum (8.3%) and the ichterohaemorrhagiae (8.3%).
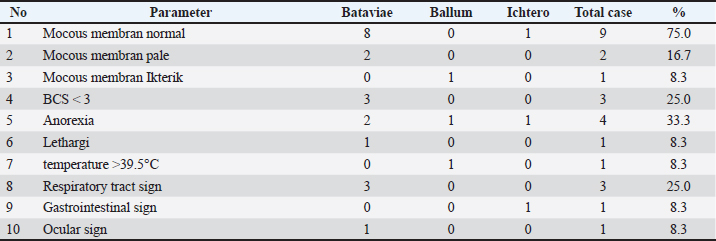
Fig. 1. Distribution of Leptospira interrogans serovars in 12 seropositive catsIn Sleman Regency, Indonesia. Serovar Bataviae predominated (83.3%), Followed by the ballum (8.3%) and the ichterohaemorrhagiae (8.3%). Most Leptospira-positive cats were asymptomatic. Three cats (25%) exhibited clinical signs of leptospirosis, including anorexia, lethargy, icterus, fever, and vomiting; mild diarrhea was also observed in a few cases. Hematological examination of seropositive cats showed that seven cats (58.33%) were normal, three cats (25%) had leukocytosis, and two cats (16.67%) had hypochromic anemia. Differential leukocyte evaluation showed that neutrophilia accompanied by relative lymphopenia primarily characterized leukocytosis in the affected cats. Mild monocytosis was observed in some cats, whereas eosinopenia was observed in one individual. No basophil abnormalities were detected. Kidney function tests for blood urea nitrogen (BUN), creatinine, and symmetric dimethylarginine (SDMA) values were within normal limits, except for one cat that also suffered from feline infectious peritonitis (FIP). Table 2 presents the hematology and blood chemistry examination results of leptospirosis-positive cats. Table 2. Clinical condition of leptospirosis-positive cats in Sleman Regency, Indonesia.
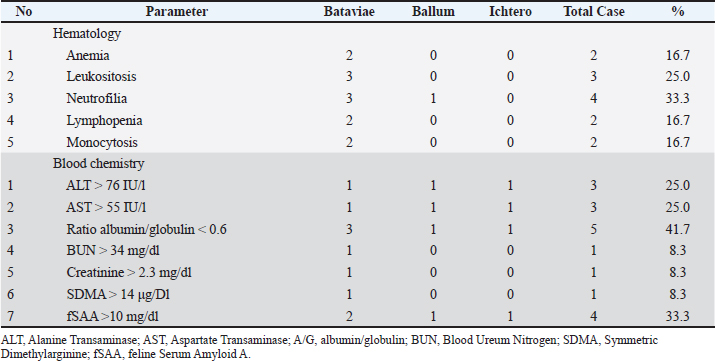
Among the seropositive cats, 25% showed mild to moderate increases in alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels, indicating hepatocellular involvement. Additionally, 41.7% of patients had decreased albumin–globulin ratios or elevated total bilirubin, indicating impaired hepatic function or cholestatic changes. Increased feline serum amyloid A (fSAA) levels in 33.3% of cats reflected ongoing systemic inflammation. In contrast, renal biomarkers (BUN, creatinine) remained within normal limits in most cats, indicating limited renal impairment (Table 3). Table 3. Hematology and blood chemistry in leptospirosis-seropositive cats in Sleman Regency, Indonesia.
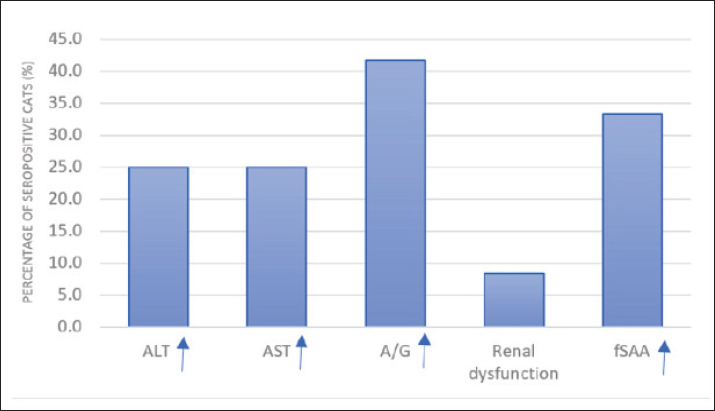
Figure 2 shows the percentage of leptospirosis-seropositive cats (n=12) with abnormalities. In liver and kidney function parameters. Elevated A/G ratio and total BMI were the most frequent (41.7%), while renal dysfunction was the least frequent (8.3%).
Fig. 2. Percentage of leptospirosis-seropositive cats (n=12) with abnormalities In liver and kidney function parameters. Elevated A/G ratio and total BMI Were the most frequent (41.7%), while renal dysfunction was the least frequent (8.3%). DiscussionLeptospirosis is an endemic zoonotic disease in Indonesia, with an increasing number of human cases. In Sleman Regency, Indonesia, 46 human cases of leptospirosis were recorded between January and June 2025, including eight fatalities. In this context, the present study, conducted from June to August 2025, identified a 10.34% seroprevalence of leptospirosis in cats in the same region. This finding is comparable to the global seroprevalence of 11.7% reported by Ricardo et al. (2023) and the prevalence of 9.95% reported in Asia (Andityas et al., 2024). Pathogenic Leptospira spp. can be detected in feline urine, with prevalence ranging from 0.8% to 67.8% (Rodriguez et al., 2014). These findings indicate that feline exposure to Leptospira spp. in endemic regions warrants greater attention. However, seropositivity and urinary shedding reflect exposure and possible carriage, rather than direct evidence of active transmission to other hosts. The majority of seropositive cats (83.34%) reacted to L. interrogans serovar Bataviae, with antibody titers reaching 1:1600. This finding is consistent with previous reports from Asia, where Bataviae is the predominant serovar in feline leptospirosis (Alashraf et al., 2020). In contrast, studies from other regions have reported different dominant serovars, including Pomona, Grippotyphosa, and Canicola (Azocar-Aedo, 2022). Serovar Bataviae has also been reported as a pathogenic serovar in dogs in Indonesia (Mulyani et al., 2019), indicating that environmental exposure is shared across species rather than reflecting species-specific infection patterns. These variations highlight the influence of local ecological and epidemiological factors on circulating Leptospira serovars. Feline serum amyloid A has been recognized as a sensitive biomarker of inflammation in cats (Waugh et al., 2022; Ricardo et al., 2023). Leptospirosis remains an emerging zoonosis of global concern (Kakita et al., 2021; Haake and Levett, 2024). Most of the seropositive cats in this study (66.67%) were clinically healthy. Only two young cats infected with the Icterohaemorrhagiae and Ballum serovars exhibited non-specific clinical signs, including anorexia, fever, icterus, vomiting, and diarrhea. Although the number of clinically affected cats was limited and no statistical inference can be made, these observations remain clinically relevant because these serovars are associated with higher pathogenic potential. One seropositive cat was concurrently diagnosed with FIP, and its clinical manifestations could not be attributed solely to leptospirosis due to overlapping clinical features between the two diseases. Previous studies have reported that the clinical signs of feline leptospirosis are often nonspecific and include weakness, anorexia, lethargy, and fever, whereas icterus occurs less frequently in cats than in dogs (Rodriguez et al., 2014; Murillo et al., 2020). The hematological findings in most cats were within the reference intervals. A small proportion of cats showed anemia or leukocytosis with neutrophilia and monocytosis; however, these findings are reported descriptively and should be interpreted cautiously due to the limited sample size. Similar hematological alterations have been described in cats exposed to Leptospira spp. (Donato et al., 2022). However, their clinical relevance in subclinical infections remains unclear. Notably, the seropositive cats in this study did not exhibit elevations in SDMA, BUN, or creatinine concentrations. As SDMA is considered an early biomarker of reduced glomerular filtration rate in cats (Hall et al., 2014), the absence of renal biomarker elevation suggests that most infections were subclinical at the time of sampling. This finding is consistent with the descriptive, field-based epidemiological nature of this study, which is not a clinical disease investigation. Although Leptospira spp. colonize renal tubules, feline infections often remain clinically unapparent, with limited renal pathology (Alashraf et al., 2020). The proposed explanations include an effective feline immune response and reduced inflammatory activation within renal tissues (Adler and De La Peña Moctezuma, 2010). In contrast to renal findings, mild hepatic alterations, including increased Serum glutamic pyruvic transaminase (alanine aminotransferase, ALT), Serum glutamic oxaloacetic transaminase (aspartate aminotransferase, AST), and bilirubin levels, were observed in a subset of seropositive cats. Similar hepatic involvement without concurrent renal dysfunction has been reported in other animal species, including dogs (McCallum et al., 2019) and cats (Rodriguez et al., 2014; Murillo et al., 2020). These findings indicate that hepatic involvement, particularly in subclinical or early-stage infections, can occur independently of detectable renal impairment. Serum amyloid A, an acute-phase protein produced by the liver in response to inflammatory stimuli, was elevated in four seropositive cats. Increased fSAA levels have been recognized as a sensitive inflammation marker in feline patients (Yuki et al., 2020; Rossi, 2023). In this study, elevated fSAA levels frequently coincided with mild hepatic alterations, further supporting its utility as an adjunctive biomarker in the evaluation of feline inflammatory conditions. Overall, these findings underscore the importance of considering feline leptospirosis from an epidemiological perspective within endemic settings. Although this study does not establish causal relationships or transmission dynamics, it highlights the presence of subclinical exposure in domestic cats and supports the integration of feline surveillance into broader One Health frameworks. ConclusionThis study indicates that leptospiral exposure among domestic cats in Sleman Regency is predominantly subclinical and is most associated with L. interrogans serovar Bataviae. Although most seropositive cats appeared clinically healthy and showed no evidence of renal impairment, a proportion exhibited mild hepatic alterations, emphasizing the heterogeneous clinical presentation of feline leptospirosis. The detection of pathogenic Leptospira serovars in outwardly healthy cats indicates an epidemiological role for cats as subclinical carriers rather than overtly diseased hosts. These findings highlight the need for increased feline leptospirosis awareness and surveillance, particularly in endemic regions where human cases continue to occur. Routine monitoring of domestic cats and integration of veterinary diagnostics into local One Health strategies may improve the understanding of environmental exposure patterns. Future studies with larger sample sizes, molecular confirmation, and the inclusion of appropriate control groups are recommended to further clarify the epidemiological significance of leptospiral ecology in cats. AcknowledgmentsThe authors would like to express their deepest gratitude to the Directorate of Research and Community Service, Directorate General of Research and Development, Ministry of Higher Education, Science, and Technology, Republic of Indonesia, for providing funding for this research through the BIMA program for Fiscal Year 2025. The authors also express gratitude to veterinary practitioners in Sleman Regency, Indonesia, and the staff of ProLab Veterinary for their assistance in sampling and laboratory analysis. Conflict of interestThe author declares no conflict of interest during the study. FundingThis research is fully supported by the Directorate of Research and Community Service, Directorate General of Research and Development, Ministry of Higher Education, Science and Technology, through decree number 067/C3/DT.005.00/PL/2025 and Gadjah Mada University through agreement/contract number 2446/UN1/DITLIT/Dit-Lit/PT 01.03/2025. Authors’ contributionGTM contributes to sample collection, clinical examination, hematological analysis, and blood chemistry. DAW contributes to MAT analysis and publications; RAP contributes to data collection, report preparation, and publication. All authors have read and approved the final version of the manuscript. Data availabilityThe original data and contributions from this study are available in the article or supplementary materials. Further inquiries should be directed to the corresponding author. ReferencesAdler, B. and De La Peña Moctezuma, A. 2010. Leptospira and leptospirosis. Microbiol 140, 287–296; doi:10.1016/j.vetmic.2009.03.012 Alashraf SF Lau., Khairani-Bejo KH Khor., Ajat R Radzi. and Roslan MSA Rahman. 2020. Pathogenic Leptospira spp. isolated from the urine and kidneys of naturally infected cats. PLos One. 15(3), e0230048; doi:10.1371/journal.pone.0230048 Andityas, M., Nuraini, D.M., Sota, P., Loong, S.K., Sripa, B., Sukon, P., Tangkawattana, P. and Tangkawattana, S. 2024. Worldwide prevalence of feline leptospirosis: a systematic review and meta-analysis of diagnostic approaches. Vet. World. 17(2), 255–272; doi:10.14202/vetworld.2024.255-272 Arbor, J., Blais, M.C., Carioto, L. and Sylvestre, D. 2012. Clinical Leptospirosis in Three Cats (2001–2009). J. Am. Anim. Hosp. Assoc. 48, 256–260; doi:10.5326/jaaha-ms-5748 Azocar-Aedo, L. 2022. Global prevalence and epidemiology of leptospirosis in domestic cats: a systematic review and meta-analysis. Veterinaria Mex. OA. 9, 1–20; doi:10.22201/fmvz.24486760e.2022.1129 Chadsuthi, S., Bicout, D.J., Wiratsudakul, A., Suwancharoen, S., Petkanchanapong, W., Modchang, C., Triampo, W. and Chalvet-Monfray, K. 2017. Predominant Leptospira serovars and their distribution in humans and livestock in Thailand, 2010–2015. PLos Negl. Trop. Dis. 11, 5228; doi:10.1371/journal.pntd.0005228 Craig, E.G. and Sykes, J.E. 2015. Leptospirosis.In Infectious Diseases of the Dog and Cat (4th ed.). Greene, C.E Davignon , G., Cagliero, J., Guentas, L., Bierque, E., Genthon, P., Gunkel-Grillon, P., Juillot, F., Kainiu, M., Laporte-Magoni, C., Picardeau, M., Selmaoui-Folcher, N., Soupé-Gilbert, M.E., Tramier, C., Vilanova, J., Wijesuriya, K., Thibeaux, R. and Goarant, C. 2023. Leptospirosis: toward a better understanding of the environmental lifestyle of Leptospira. Front. Water. 5, 1–15; doi:10.3389/frwa.2023.1195094 Donato, G., Masucci, M., Hartmann, K., Goris, M.G.A., Ahmed, A.A., Archer, J., Alibrandi, A. and Pennisi, M.G. 2022. Prevalence of Leptospira spp. in cats from southern Italy with evaluation of risk factors for exposure and clinical findings in infected cats. Pathogens 11(10), 1129; doi:10.3390/pathogens11101129 Haake, D.A. and Levett, P.N. 2024. Leptospirosis—an emerging zoonosis. Nature. Rev. Microbiol. 22(3), 221–235; doi:10.1038/s41579-023-00923-1 Hall, J.A., Yerramilli, M., Obare, E., Yerramilli, M. and Jewell, D.E. 2014. Serum concentrations of symmetric dimethylarginine and creatinine as kidney function biomarkers in cats with chronic kidney disease. J. Vet. Intern. Med. 28(6), 1676–1683; doi:10.1111/jvim.12445 Hartmann, K., Egberink, H., Pennisi, M.G., Lloret, A., Addie, D., Belák, S., Boucraut-Baralon, C., Frymus, T., Gruffydd-Jones, T. and Hosie, M.J. 2013. Leptospira species infection in cats: aBCD guidelines on prevention and management. Feline. Med. Surg. 15, 576–581; doi:10.1177/1098612X13489217 Kakita, T., Okano, S., Kyan, H., Miyahira, M., Taira, K., Kitashoji, E. and Koizumi, N. 2021. Diagnostic, epidemiological, and clinical laboratory characteristics of human leptospirosis in Okinawa Prefecture, Japan, 2003–2020. PLos Negl. Trop. Dis. 15(12), e0009993; doi:10.1371/journal.pntd.0009993 McCallum, K.E., Constantino-Casas, F., Cullen, J.M., Warland, J.H., Swales, H., Linghley, N., Kortum, A.J., Sterritt, A.J., Cogan, T. and Watson, P.J. 2019. Hepatic leptospiral infections in dogs without obvious renal involvement: a case report. J. Vet. Intern. Med. 33(1), 141–150; doi:10.1111/jvim.15340 Miotto, B.A., Camelo, Q.C., Grolla, A.C.M.M., De Oliveira, A.B.M., Silva, M.M.B., Hagiwara, M.K. and Esteves, S.B. 2024. Current knowledge on leptospirosis in cats: a systematic review of direct detection, serological response, and clinical data. Res. Vet. Sci. 174, 105292; doi:10.1016/j.rvsc.2024.105292 Mulyani, G.T., Hartati, S., Santoso, Y., Pramono, K. and Wirapratiwi, D.K. 2019. Kejadian Leptospirosis Pada Anjing di Daerah Istimewa Yogyakarta. Jurnal. Veteriner. 18(3), 403–408; doi:10.22146/jsv.39201 Murillo, A., Goris A Ahmed., Cuenca. and Pastor. 2020. Leptospirosis in cats: current literature review to guide diagnosis and management. Feline Med. Surg. 22, 216–228; doi:10.1177/1098612x20903601 Ricardo, T., Azócar-Aedo, L., Signorini, M. and Previtali, M.A. 2023. Leptospiral infection in domestic cats: systematic review and meta-analysis. Prev. Vet. Med. 212, 105851; doi:10.1016/j.prevetmed.2023.105851 Rodriguez, J., Blais, M.C., Lapointe, C., Arsenault, J., Carioto, L. and Harel, J. 2014. Serologic and urinary polymerase chain reaction survey of leptospirosis in healthy cats and in cats with kidney disease. J. Vet. Intern. Med. 28, 284–293; doi:10.1111/jvim.12287 Rossi, G. 2023. Acute phase proteins in cats: diagnostic and prognostic role, future directions, and analytical challenges. Vet. Clin. Pathol. 52(Suppl 1), 37–49; doi:10.1111/vcp.13238 Samrot, A.V., Sean, T.C., Bhavya, K.S., Sahithya, C.S., Chan-Drasekaran, S., Palanisamy, R., Robinson, E.R., Subbiah, S.K. and Mok, P.L. 2021. Leptospiral infection, pathogenesis and its diagnosis—A review. Pathogens 10, 145–130; doi:10.3390/pathogens10020145 Schuller, S., Francey, T., Hartmann, K., Hugonnard, M., Kohn, B., Nally, J.E. and Sykes, J. 2015. European Consensus Statement on Leptospirosis in Dogs and Cats. Small Anim. Pract. 56, 159–179; doi:10.1111/jsap.12328 Thrusfield , M. and Christley, R. 2018. Veterinary Epidemiology, 4th ed. Oxford, UK: Wiley-Blackwell. Waugh , E.M., Haining, H., Harvie, J., Ridyard, A.E. and Eckersall, P.D. 2022. Validation of an automated immunoturbidimetric assay for feline serum amyloid A. BMC. Vet. Res. 18, 359; doi:10.1186/s12917-022-03456-5 World Organisation for Animal Health (WOAH). 2021. Leptospirosis. In: Manual of Diagnostic Tests and Vaccines for Terrestrial Animals. Paris, France: WOAH. Available viahttps://www.woah.org Yuki, M., Aoyama, R., Nakagawa, M., Hirano, T., Naitoh, E. and Kainuma, D. 2020. A clinical investigation on serum amyloid A concentration in client-owned healthy and diseased cats in a primary care animal hospital. Vet. Sci. 7(2), 45;doi:10.3390/vetsci7020045 | ||
| How to Cite this Article |
| Pubmed Style Mulyani GT, Widiasih DA, Primatika RA. Clinical and sero-diagnosis of feline leptosprosis in the sleman region of Yogyakarta, Indonesia. Open Vet. J.. 2026; 16(2): 834-840. doi:10.5455/OVJ.2026.v16.i2.7 Web Style Mulyani GT, Widiasih DA, Primatika RA. Clinical and sero-diagnosis of feline leptosprosis in the sleman region of Yogyakarta, Indonesia. https://www.openveterinaryjournal.com/?mno=290581 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.7 AMA (American Medical Association) Style Mulyani GT, Widiasih DA, Primatika RA. Clinical and sero-diagnosis of feline leptosprosis in the sleman region of Yogyakarta, Indonesia. Open Vet. J.. 2026; 16(2): 834-840. doi:10.5455/OVJ.2026.v16.i2.7 Vancouver/ICMJE Style Mulyani GT, Widiasih DA, Primatika RA. Clinical and sero-diagnosis of feline leptosprosis in the sleman region of Yogyakarta, Indonesia. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 834-840. doi:10.5455/OVJ.2026.v16.i2.7 Harvard Style Mulyani, G. T., Widiasih, . D. A. & Primatika, . R. A. (2026) Clinical and sero-diagnosis of feline leptosprosis in the sleman region of Yogyakarta, Indonesia. Open Vet. J., 16 (2), 834-840. doi:10.5455/OVJ.2026.v16.i2.7 Turabian Style Mulyani, Guntari Titik, Dyah Ayu Widiasih, and Roza Azizah Primatika. 2026. Clinical and sero-diagnosis of feline leptosprosis in the sleman region of Yogyakarta, Indonesia. Open Veterinary Journal, 16 (2), 834-840. doi:10.5455/OVJ.2026.v16.i2.7 Chicago Style Mulyani, Guntari Titik, Dyah Ayu Widiasih, and Roza Azizah Primatika. "Clinical and sero-diagnosis of feline leptosprosis in the sleman region of Yogyakarta, Indonesia." Open Veterinary Journal 16 (2026), 834-840. doi:10.5455/OVJ.2026.v16.i2.7 MLA (The Modern Language Association) Style Mulyani, Guntari Titik, Dyah Ayu Widiasih, and Roza Azizah Primatika. "Clinical and sero-diagnosis of feline leptosprosis in the sleman region of Yogyakarta, Indonesia." Open Veterinary Journal 16.2 (2026), 834-840. Print. doi:10.5455/OVJ.2026.v16.i2.7 APA (American Psychological Association) Style Mulyani, G. T., Widiasih, . D. A. & Primatika, . R. A. (2026) Clinical and sero-diagnosis of feline leptosprosis in the sleman region of Yogyakarta, Indonesia. Open Veterinary Journal, 16 (2), 834-840. doi:10.5455/OVJ.2026.v16.i2.7 |