| Research Article | ||
Open Vet. J.. 2026; 16(1): 147-156 Open Veterinary Journal, (2026), Vol. 16(1): 147-156 Research Article Isolation and phylogenetic characterization of Sarcocystis singaporensis isolates from Surabaya python snakes (Malayopython reticulatus) as a biological control agent for ratsEduardus Bimo Aksono1, Agus Sunarso1, Muchammad Yunus1*, Lucia Tri Suwanti1, Mufasirin1 , Luhur Sediyoadi2, Adam Sediyoadi Putra2, Nadia Tasya Rosalia1 and Nafilah Dzahabiyyah11Department of Veterinary Science, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia 2Centra Biotech Indonesia, Klaten, Indonesia *Corresponding Author: Muchammad Yunus, Department of Veterinary Science, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia. Email: muchammad-y [at] fkh.unair.ac.id Submitted: 10/10/2025 Revised: 30/11/2025 Accepted: 11/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
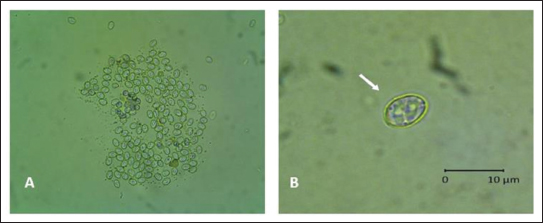
AbstractBackground: Rats are the most difficult problem faced by farmers in every planting season. Various methods, ranging from mechanical, chemical, and biological methods, have been used but have not been efficient and effective. Aim: This study aimed to obtain a biological control agent for rats through the stages of isolation and characterization of Sarcocystis singaporensis derived from the feces of reticulated pythons or pythons (Malayopython reticulatus, M. reticulatus) in Surabaya, both morphologically and molecularly, and testing the potential for rat infection through blood hematology assessment. Methods: The method uses 10 reticulated pythons or pythons, then isolated sporocysts from feces with a float test, to see the morphology using an Optilab microscope, after which molecular characterization is carried out using polymerase chain reaction with specific primers 18S rRNA specific for S. singaporensis with a target of 900–1,000 bp, then the results obtained are continued with sequencing and phylogenetic tree analysis also including reference sequences of S. singaporensis from GenBank. In addition, a potential infection test was conducted on rats in two experiments. Experiment 1 consisted of four treatment groups: control, doses of 4,000 sporocysts, 75,000 sporocysts, and 200,000 sporocysts administered orally for 14 days, followed by blood sampling for a complete blood count. Experiment 2 consisted of three treatment groups: doses of 300,000 sporocysts, 400,000 sporocysts, and 500,000 sporocysts to observe more severe impacts due to treatment, including death without necropsy and blood sampling for a complete blood count. Results: The research results showed that sporocysts from the feces of reticulated pythons, both morphologically and molecularly, were identical to S. singaporensis. Conclusion: The Surabaya isolate was proven to cause death in mice at a dose of 500,000 sporocysts. Keywords: S. singaporensis, Rats, Python snake, Sporocyst. IntroductionIn Indonesia, one of the national food security issues is related to the main pest of rice plants in each planting season, which causes crop failure: rat attacks. Rat pest control is the most difficult problem faced by farmers in every planting season (Van Den Berg and Jiggins, 2007). The rice rat population in rice fields planted with a three-times-a-year cropping index tends to increase each planting season. Rats begin to breed in irrigated rice ecosystems when the rice plants are in the gestation stage and continue until harvest. Rice rats give birth three times in one planting season, with an average of 10 offspring per litter. This is because the increase in the rice cropping index indirectly creates favorable environmental conditions for rats, namely, the availability of abundant food sources, which greatly supports their reproduction process. Rice rats can attack rice plants from the seedling stage to harvest (Sudarmaji and dan Herawati, 2017; Hamdan et al., 2020). Rat infestations can cause up to 80% of rice plants in a single plot to die in a single night (1 day). Rats damage rice plants from the roots, stems, and leaves down to the grains, preventing them from growing (Sarra and Peters, 2003; Xuan et al., 2021). According to the Directorate of Food Crop Protection, cited by Brotodjoyo et al. (2023) 58,443 hectares of rice fields were infested with rats from January to June 2021, resulting in crop failures of approximately 1,842 hectares. The five provinces with the highest rat infestation percentage were Lampung, Central Java, East Java, West Java, and South Sulawesi. Various approaches to rat infestation exist, including mechanical, chemical, and biological methods. In the event of an infestation, the government encourages farmers to adopt safer and more environmentally friendly pest control techniques (Kabir and Rainis, 2015; Halimatunsadiah et al., 2016). Control can be done mechanically, for example, by encouraging the use of rat traps on agricultural land. Control can also be done chemically using compost by spraying it with rat poison, but the poison used is dangerous for users (Xuan et al., 2021). All of these methods are considered ineffective in controlling rat populations; therefore, biological control efforts have been made using S. singaporensis (Jäkel et al., 1999; Ginting and Jakel, 2005). Sarcocystis singaporensis is a parasitic unicellular organism belonging to the phylum Apicomplexa, class Sporozoasida, subclass Coccidiasina, order Eucoccidiorida, and family Sarcocystidae (Bezerra et al., 2023). This parasite has been extensively studied for over 25 years (Khoprasert et al., 2008) and was first discovered in Singapore (Zaman and Coley, 1975; O’Donoghue et al., 1987; Jakel et al., 1997a,b). It is also found in Sulawesi and Java in Indonesia (O’Donoghue et al., 1987; Ginting and Jakel, 2005). The S. singaporensis parasite is not infectious or harmful to various species of birds, reptiles, and mammals, including primates (Khoprasert et al., 2008). Therefore, the absence of epidemiological data that can link this parasite species to human infection does not pose a risk of infection, although the parasite has a high prevalence in nature (O’Donoghue et al., 1987; Jakel et al., 1996; Khoprasert et al., 2008; Aryan et al., 2025). Sarcocystis singaporensis is one of the most studied snake species due to its endemicity in Southeast Asia and its ability to cause death in certain rodent species (Bezerra et al., 2023). In its life cycle, this parasite requires two hosts: the reticulated python (Malayopython reticulatus) as the definitive host and rodents of the genera Rattus and Bandiota as intermediate hosts (Jakel et al., 1996; Jäkel et al., 1999; Ginting and Jakel, 2005; Qin et al., 2024). The sporozoite-containing sporocyst is the infectious stage in rats and can be obtained in large numbers from the reticulated python (Python reticulatus) feces (Jakel et al., 1996). The morphology and size of sporocysts found in Langkat Regency, North Sumatra, Indonesia, resemble those of specimens from Thailand and Singapore, which also have four sporozoites, which are characteristic of S. singaporensis (Zaman and Coley, 1975; Jakel et al., 1997a,b). This parasite is initially apathogenic, but when sporocysts are given to mice above a certain threshold, they cause fatal pneumonia and even death, thus providing initial evidence that S. singaporensis has potential as a biological control agent for wild mice in the field (Zaman, 1976; Jakel et al., 1996). Ginting and Jakel (2005)even demonstrated that the S. singaporensis parasite isolated from the feces of the rice paddy snake, P. reticulatus, was capable of infecting laboratory rats (Rattus norvegicus strain Sprague Dawley). Similarly, Jakel et al. (2001)reported that 200,000 sporocyst infection could kill various rat species within 10 to 14 days. A definitive diagnosis of Sarcocystis requires sporocyst identification in the feces. However, sporocysts from different species are similar in size and shape, making species identification under a microscope nearly impossible. Therefore, 18S rRNA gene sequencing has been introduced as an ideal method for species-specific detection (Tenter, 1995). This gene contains hypervariable regions interspersed with highly conserved DNA sequences, making it an ideal candidate for interspecies differentiation (Teaser, 1995). The 18S rRNA gene has been frequently used in the phylogenetic analysis of Apicomplexa species (Lau et al., 2013). Several researchers have used 18S rRNA gene sequencing for Sarcocystis species differentiation and phylogenetic studies (Kan and Pathmanathan, 1991; Daszak and Cunningham, 1995; Jakel et al., 1997a,b; Šlapeta et al., 2002). This study aimed to conduct morphological analysis and phylogenetic analysis based on 18S rRNA gene sequencing to determine the evolutionary relationship of S. singaporensis isolated from the feces of a reticulated python (M. reticulatus) isolated from Surabaya, East Java with previously reported Sarcocystis sp. species as well as aspects of complete blood picture of S. singaporensis infection as a basis for assessing its potential as a candidate biological control agent for rats. Materials and MethodsFecal collection of reticulated pythons (M. reticulatus)Research samples were obtained by collecting feces from 10 reticulated pythons (M. reticulatus) from Surabaya, East Java, Indonesia. The samples were collected and preserved in fecal containers containing a 2.5% potassium dichromate solution to prevent contamination from the environment. Samples treated with 2.5% potassium dichromate were also stored at 4°C until they were used for the isolation and identification of S. singaporensis sporocysts (Lau et al., 2013; Máca et al., 2021; Dzahabiyyah, 2025; Rosalia, 2025). Isolation and identification of Sporocysts of S. singaporensisSarcocystis singaporensis sporocysts were isolated on M. reticulatus feces using the flotation method, which were then dissolved in distilled water at a ratio of 1:10. The resulting mixture was filtered through a sieve and poured into a centrifuge tube. Centrifugation was performed at 1,500 rpm for 5 minutes. The supernatant was discarded, and the process was repeated until the supernatant was clear. The clear supernatant was discarded, and a saturated sugar solution was added to a level 2 cm below the mouth of the tube, followed by centrifugation at 1,500 rpm for 5 minutes. The saturated sugar solution was added until the mouth of the tube appeared convex. Then, a cover glass was placed over the mouth of the tube and allowed to sit for 5 minutes (Dzahabiyyah, 2025; Rosalia, 2025). Identification of the S. singaporensis parasite in the feces of reticulated pythons (M. reticulatus) was performed by observing the parasite’s size and morphology microscopically at 400× magnification. Identification was performed at the Veterinary Parasitology Division Laboratory, Faculty of Veterinary Medicine, Universitas Airlangga, Indonesia. Identification and measurement of the dimensions of Sarcocystis sp. oocytes or sporocysts in the feces of reticulated pythons (M. rericulatus) using Optilab Viewer computer software. Sporocyst counts were performed using a hemocytometer by counting the number of cells in four large boxes (leukocyte counting boxes) located in the corners of the counting chamber. The sporocyst suspension was taken with a pipette and dripped onto the counting chamber area covered with a cover glass. Observations were made under a microscope at 400× magnification. The number of sporocysts per ml was obtained by counting the total number of sporocysts in the four chamber boxes, dividing the result by four, and multiplying by 10,000 (Mahmudah, 2006; Rosalia, 2025). Molecular characterization of S. singaporensisDNA extractionThe sporocyst sample was homogenized by vortex, and DNA extraction was performed on 0.25 g of the fecal sample using the QIAMP DNA Mini Kit (Qiagen) according to the manufacturer’s protocol. DNA was eluted with 50 μl of elution buffer and stored at −20°C until further processing (Aksono et al., 2017). Polymerase chain reaction (PCR)The amplification process was carried out using the nested PCR method with 18s RNA primers (Lau et al., 2013; Mohd Fadil et al., 2019). The primers used were 1H (forward: 5’CCA TGC ATG TCT AAG TAT AAG C 3’) and 1L (reverse: 5’ AAC TGT TAT TGC CTC AAA CTT C 3’) for Nested 1 and 2H (forward: 5’ CTA GTG ATT GGA ATG ATG GG 3’) and 3L (reverse: 5’ AAC TGT TAT TGC CTC AAA CTTC 3’) for Nested 2.6 PCR amplification was performed in a 25 μl mixture containing 35 mM Tris–HCl, pH 9.0, 25 mM KCl, 3.5 mM MgCl2, 5 pmol of each primer, 1 mM dNTP, and 1 U of Taq polymerase (Promega) and 4 μl of DNA template. The mixture was subjected to the following PCR conditions: 94°C for 2 minutes and 35 cycles of denaturation for 40 seconds at 94°C, annealing for 30 seconds at 50°C, and extension for 1.5 minutes at 72°C. A final extension for 10 minutes at 72°C was added in the last cycle. The expected amplicon size was approximately 1,000 base pairs. PCR products were viewed on 1% (w/v) agarose gel (Lau et al., 2013; Mohd Fadil et al., 2019). DNA sequencing and phylogenetic analysisSarcocystis singaporensis, showing a positive PCR result, was then purified using QIAquick DNA Purification Kit (Qiagen). Each 18S rRNA gene sequence was searched for matches in the GenBank nucleotide database using BLASTn. The best sequence was selected to represent each amplicon in the phylogenetic analysis. These sequences were compared and aligned with the 18S rRNA gene sequences of Sarcocystis species available in GenBank. Multiple sequence alignments were performed using Clustal W software. Based on the 18S rRNA gene sequences, a phylogenetic tree was constructed using the neighbor-joining method (bootstrap=1,000) available in MEGA4. The 18S rRNA gene sequence of Eimeria tenella was used as the outgroup (Lau et al., 2013; Aksono et al., 2017). Experimental animalsExperiment 1: Twenty-four male white rats (Rattus novergicus), approximately 9 weeks old and weighing approximately 80 g, were housed in 18 × 24 × 15 cm cages according to the treatment groups. Rats were acclimatized for 7 days prior to the study. Pellet feed and water were provided ad libitum (Rosalia, 2025). White rats with uniform body weight were then divided into four groups as follows: P0 (control group not given S. singaporensis sporocysts); P1 (S. singaporensis sporocyst infection group with a dose of 4,000 sporocysts orally); P2 (S. singaporensis sporocyst infection group with a dose of 75,000 sporocysts orally); P3 (S. singaporensis sporocyst infection group with a dose of 200,000 sporocysts orally). Observations were conducted for 21 days and sacrificed on the 22nd day after treatment using an intramuscular injection of ketamine at a dose of 300 mg/kg BW and xylazine at a dose of 30 mg/kg BW (Derakhshanfar et al., 2022). Histopathological examination was performed on the heart muscle as one of the development sites of S. singaporensis to observe the changes and impacts resulting from them. Experiment 2: Eighteen male white rats were divided into three groups: P1 (infected orally with 300,000 sporocysts), P2 (400,000 sporocysts), and P3 (500,000 sporocysts) to observe more severe impacts due to treatment, including death without necropsy and blood sampling for a complete blood count. Hematological examinationFollowing euthanasia, the rats were dissected from the lower abdomen to the thoracic cavity. Blood was collected directly from the heart in vacutainer tubes containing EDTA at a ratio of 1 mg of EDTA per 1 ml of blood. The blood collected in the EDTA tube was immediately shaken gently using a figure-of-eight motion to mix the blood and anticoagulant. Blood samples were stored at 4°C in a refrigerator until ready for use. The hematological parameters assessed included: leukocytes, erythrocytes, hemoglobin, hematocrit, and platelets, as well as leukocyte differential counts (lymphocytes, monocytes, and granulocytes) (Fayer and Prase, 1981; Mahaffey et al., 1986; Putri et al., 2023). Data analysisHematological data were statistically analyzed using SPSS. Differences among groups were first assessed using the Kruskal–Wallis test. When significant differences were observed (p < 0.05), pairwise comparisons were performed using the Mann–Whitney U test. Ethical approvalThis research was approved by the Animal Care and Use Committee (ACUC) Faculty of Veterinary Medicine, Universitas Airlangga (No.1.KEH.34.03.2025; March 3, 2025). ResultsIdentification of S. singaporensis oocysts or sporocystsBased on the results of an Optilab light microscope examination, sporocyst-containing Sarcocystis sp. oocysts were found in the feces and intestines of 2 out of 10 reticulated pythons (M. reticulatus) in Surabaya. From the research data microscopically at 400× and 1,000× magnification, it shows that the oocytes or sporocytes of Sarcocystis sp. isolated from reticulated pythons or pythons (M. reticulatus) in Surabaya, Indonesia have an average size of 8.2 (± 0.5) × 6.4 (± 0.2) μm, oval in shape, with a sporocyst wall that appears quite thick (Fig. 1).
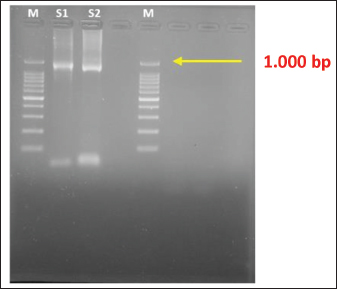
Fig. 1. A. Sarcocystis spp., 400× magnification; B. Sarcocystis singaporensis, 1,000× magnification. Molecular characterization of S. singaporensisThe sample found in the feces of the reticulated pythons or python (M. reticulatus) after PCR with specific 18S rRNA primers from S. singaporensis shows a positive band at approximately 900–1,000 bp (Fig. 2).
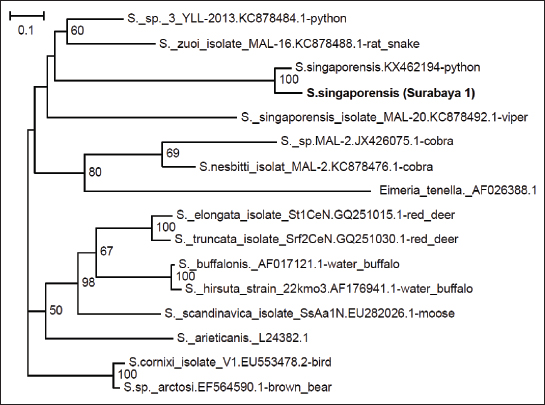
Fig. 2. PCR results of S. singaporensis using 1% agarose gel. M: marker (1.000 bp); S1: isolate of S. singaporensis 1 (1.000 bp); S2: isolate of S. singaporensis 2 (1.000 bp). In this research, for phylogenetic analysis of samples found in reticulated pythons or pythons in Surabaya against S. singaporensis, comparative references from isolates that have been reported in GenBank are also used, namely JX426075.1 cobra (Sarcocystis sp.), KC878476.1 cobra (S. nesbitti), EU553478.2 bird (S.cornixie), EF564590.1 brown bear (S. arctosi), KX462194 python (S. singaporensis), KC878484.1 python (Sarcocystis sp.), AF017121.1 water buffalo (S. buffalonis), AF176941.1 water buffalo (S. hirsute), EU282026.1 moose (S. scandinavica), GQ251015.1 red deer (S. tarandi), GQ251030.1 red deer (S. rangiferi), L24382.1 (S. arieticanis), KC878492.1 viper (S. singaporensis), and KC878488.1 rat snake (S. zuoi) from outgroup used AF026388.1 (Eimeria tenella). The phylogenetic tree shows that sample-1 (isolate Surabaya) has 100% homology with KX462194 python (S. singaporensis) (Figs. 3 and 4).
Fig. 3. Results of sample and reference multisequence.
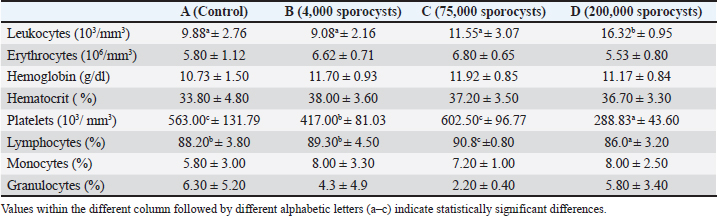
Fig. 4. Phylogenetic analysis results. Hematological parametersAs shown in Table 1, hematological analysis of white rats (Rattus norvegicus) revealed significant differences across several parameters, specifically leukocytes, platelets, and lymphocytes, among the experimental groups. The leukocyte count differed significantly between the groups (p=0.005), with group D having the highest average value (16.32 × 10³/mm³), which was significantly higher than the other groups, especially compared to groups A (p=0.002), B (p=0.002), and C (p=0.009). The platelet count also showed a highly significant difference (p=0.001), with group D having the lowest platelet count (288.83 × 10³/mm³), significantly lower than groups A (p=0.002), B (p=0.009), and C (p=0.002). The percentage of lymphocytes also differed significantly between groups (p=0.031), with group C having the highest value (90.8%) and group D the lowest (86.0%), and a significant difference was found between groups C and D (p=0.002). Table 1. Effects of S. singaporensis infection on hematological parameters.
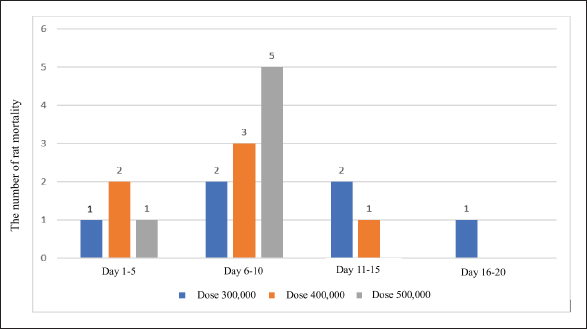
Mortality in ratsThe results demonstrated that the oral administration of sporocyst doses at 500,000 caused mortality in rats within 1–10 days, whereas doses of 400,000 and 300,000 caused mortality within 1–15 days, and doses of 300,000 led to mortality within 1–20 days (Fig. 5).
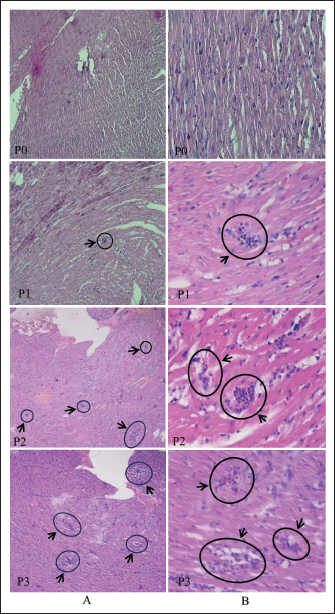
Fig. 5. Number of rat deaths at each dose and corresponding time. Histopathological findingRats observed after infection with S. singaporensis sporocysts showed decreased food and water consumption. Behavioral changes, including impaired motor coordination, such as unsteady walking and a tilted head position, also occurred in group P3 (200,000 sporocysts). Groups P1 (4,000 sporocysts) and P2 (75,000 sporocysts) generally showed no significant clinical symptoms during the observation period. Macroscopic examination of the hearts of rats from all treatment groups revealed a normal appearance, with no changes in shape, size, or color. Microscopic examination of the hearts of white rats revealed histopathological changes in the form of inflammatory cell infiltration in the heart muscle tissue surrounding the S. singaporensis cysts. Inflammatory cell infiltration appeared as clusters of inflammatory cells scattered among the heart muscle fibers, indicating parasite development and the body’s immune response to S. singaporensis sporocyst administration. Following the administration of S. singaporensis sporocysts, inflammatory cell distribution varied between treatment groups, with a tendency for increased infiltration as the dose increased in the treatment group. Histopathological examination of the inflammatory cell infiltration category in the control group showed no inflammatory cell accumulation (Fig. 6).
Fig. 6. Microscopic image of cardiac muscle tissue of white rats in group P0 with hematoxylin and eosin staining. (A) 100× magnification and (B) 400× magnification. Histopathology of the heart muscle tissue of white rats in groups P1, P2, and P3 with hematoxylin and eosin staining. (A) 100× magnification shows multifocal inflammatory cell infiltration surrounding S. singaporensis cysts as indicated by the black arrow. (B) 400× magnification show inflammatory cells consisting of mononuclear inflammatory cells (mn) and polymorphonuclear inflammatory cells (pmn). DiscussionDifferentiation between species is often demonstrated through morphological characterization of Sarcocystis using a light or transmission electron microscope. However, in this study, an Optilab light microscope was used at magnifications of 400× and 1,000×. This analytical method is not entirely accurate because the appearance of Sarcocystis sp. in the feces and intestines is not distinct, and some literature suggests that it can change depending on the location and stage of cyst development. In this study, the differentiation between Sarcocystis sp. species was based on their walls, which vary significantly in thickness (Mohammed et al., 2023). Morphologically identical and having the same size as that reported (Dzahabiyyah, 2025), namely having a size of 8.19 × 6.14 μm and a size of 7.61 × 5.71 μm. According to Wassermann et al. (2017)based on the measurement of sporocyst dimensions found in pythons there are 4 classes, but in Southeast Asia there are only 2 classes, namely class 1 (S. singaporensis) has a size of 10.2 (± 0.8) × 7.9 (± 0.7) μm and class 2 (S. samani) has a size of 10.4 (± 0.4) × 8.1 (± 0.4) μm. This means that the number of sporocysts found in reticulated pythons or pythons (M. reticulatus) in Surabaya is smaller. In addition, most sporocysts of the Sarcocystis species found in reticulated pythons or pythons (M. reticulatus) in Surabaya have an oval shape with a size of 10 × 15 μm, with a special characteristic of sporocysts that have four sporozoites, which is a characteristic of S. singaporensis. Intact oocysts can be excreted together with sporocysts appearing as two adjacent sporocysts with a thin oocyst wall that often ruptures to release the sporocyst, the most common structure found in feces (Zaman and Coley, 1975; Jakel et al., 1997a,b; Mufa et al., 2020; Rathish, 2022; Dzahabiyyah, 2025). Therefore, a molecular approach is needed to strengthen the results of species identification based on morphology, as it allows for the rapid and accurate identification of Sarcocystis species. A molecular approach to identify S. singaporensis from samples found in reticulated pythons (M. reticulatus) in Surabaya involved PCR using 18S rRNA primers, as these primers are capable of identifying taxonomic systems primarily based on phenotypic characteristics (Dolezel et al., 1999; Watthanakaiwan et al., 2017). Therefore, the use of 18S rRNA primers is very appropriate to ensure that the isolate found in the reticulated python (M. reticulastus) is a S. singaporensis isolate with a target of 1,000 bp (Fig. 2) (Lau et al, 2013; Mohd Fadil et al., 2019; Swar and Shnawa, 2021). Phylogenetic analysis of members of the Sarcocystis group is largely based on nuclear ribosomal DNA (rDNA), such as 18S rDNA and 28S rDNA nucleotide sequences, to reconstruct evolutionary relationships between organisms (Zhu et al., 2000; Lau et al., 2013; Watthanakaiwan et al., 2017; Jäkel et al., 2023; Dameshghi et al., 2024). Sarcocystis cysts in cardiac muscle tissue often trigger inflammatory cell infiltration, resulting in visible inflammation (Hong et al., 2016; Faghiri et al., 2019). Sarcocystis singaporensis infection before cyst formation has been shown to stimulate the production of proinflammatory cytokines, such as interleukin-6 (IL-6), which plays a key role in the recruitment of immune cells to target tissues. IL-6 activation induces endothelial cell and chemokine release, leading to immune cell migration to infected target tissues (Gabay, 2006). This infiltration can be focal or multifocal, depending on the extent of tissue infection or damage (Portella et al., 2021). Cardiac histopathology showing inflammatory cell infiltration also shows patterns that can be used to identify the predominant cell types, such as polymorphonuclear cells (neutrophils and eosinophils) in the acute phase and mononuclear cells (macrophages and lymphocytes) in the chronic phase (Afifi et al., 2016; Dubey et al., 2016). Myocardial granulomas with necrotic centers surrounded by mixed inflammatory cells and massive fibrosis, often leading to death, have also been reported in cases of severe sarcocystosis, with the presence of multinucleated giant cells and mineralization in the inflamed area (Aráoz et al., 2019). This condition is thought to occur in groups of rats given doses of 300,000 sporocysts. The severity of these pathological changes is strongly influenced by two main factors: the host’s immune status and the infecting sporocyst dose (Moré, 2024). ConclusionThe Surabaya isolate identified in the feces of the reticulated python (M. reticulatus), although smaller in size than previously reported S. singaporensis, exhibited morphological, molecular, and hematological characteristics consistent with S. singaporensis. Oral administration of 500,000 sporocysts caused mortality in rats within 1–10 days. AcknowledgmentWe are indebted to the PT. Centra Biotech Indonesia for their assistance in providing samples and laboratory members. We thank the anonymous reviewers and also Veterinary Biomedical Science Group of Faculty of Veterinary Medine Universitas Airlangga members for their careful reading of our manuscript and their insightful comments and suggestions, which have helped improve the quality of our manuscript. Conflicts of interestThe authors have no conflicts of interest to declare. FundingWe would like to thank the Rector of Universitas Airlangga for financial support through the Airlangga Research Fund (ARF) Batch 1 Program, Airlangga University’s Excellent Basic Research Scheme in 2025 (Number 1768/UN3.LPPM/PT.01.03/2025). Authors’ contributionAll authors contributed to the conception and design of the study. Material preparation, data collection, and analysis were performed by all authors: laboratory processing of samples (EBA, AS, MY, LTS, M, DL, LS, ASP, NTR, ND, S, and YPD). The first draft of the manuscript was written by EBA and MY and subsequently revised by MY and EBA. All authors have read and approved the final version of the manuscript. Data availabilityAll data were provided in the manuscript. ReferencesAfifi, A.F., East, B., West, E., Aswad, N.M.A., Hussein, N.M.A. and Kersh, W.M.E. 2016. A histopathological and ultrastructural study of experimental murine sarcocystosis. Menoufia. Med. J. 29(4), 862–867. Aksono, E.B., Farahdiba, A.A. and Hestianah, E.P. 2017. Bakteri Legionella pneumophila Terdeteksi pada Air Kolam Renang di Kota Surabaya dengan nested polymerase chain reaction. Jurnal. Veteriner. 18(2), 221–225; doi:10.19087/jveteriner.2017.18.2.221 Aráoz, V., Da Silva Silveira, C., Moré, G., Banchero, G., Riet-Correa, F. and Giannitti, F. 2019. Fatal Sarcocystis cruzi–induced eosinophilic myocarditis in a heifer in Uruguay. J. Vet. Diagn. Invest. 31(4), 656–660. Aryan, F.A.M., El-Azazy, O.M.E., Juozaitytė-Ngugu, E., Šneideris, D., Tahrani, L.M.A., Butkauskas, D. and Prakas, P. 2025. Morphological and molecular description of Sarcocystis meriones n. sp. from the Libyan Jird (Meriones libycus) in Kuwait. Animals 15(17), 2575; doi:10.3390/ani15172575 Bezerra, T.L., Soares, R.M. and Gondim, L.F.P. 2023. Sarcocystis species (Apicomplexa, Eucoccidiorida) parasitize snakes. Parasitologia 3, 327–347; doi:10.3390/parasitologia3040032 Brotodjojo, R.R.R., Lazuardi, I. and Suprihanti, A. 2023. Population distribution of rats (Rattus argentiventer) and the damage intensity on rice and other crops in Pleret, Yogyakarta, Indonesia. J. Proteksi. Tanaman. 7(2), 115–126; doi: 10.25077/jpt.7.2.115-126.2023. Dameshghi, F., Shirali, S., Shayan, P. and Shemshadi, B. 2024. Molecular identification of Sarcocystis cruzi isolated from Iranian buffaloes in Guilan province. Bulg. J. Vet. Med. 27, 468–474. Daszak, P. and Cunningham, A. 1995. A report of intestinal sarcocystosis in the bullsnake (Pituophis melanoleucus sayi) and a re-evaluation of Sarcocystis sp. from snakes of the genus Pituophis. J. Wildl. Dis. 31, 400–403; doi:10.7589/0090-3558-31.3.400 Derakhshanfar, A., Kian, M., Dehghan, Z., Valizadeh, A. and Moayedi, J. 2022. Comparison of the effects of two methods of euthanasia on post mortem changes in rats: histopathological and molecular findings. Comp. Clin. Pathol. 31, 815–826; doi:10.1007/s00580-022-03385-7 Dolezel, D., Koudela, B., Jirků, M., Hypsa, V., Oborník, M., Votýpka, J., Modrý, D., Slapeta, J.R. and Lukes, J. 1999. Phylogenetic analysis of Sarcocystis spp. of mammals and reptiles supports the co-evolution of Sarcocystis spp. with their final hosts. Int. J. Parasitol. 29, 795–798; doi:10.1016/j.ijpara.2012.09.020 Dubey, J.P., Calero-Bernal, R., Rosenthal, B.M., Speer, C.A. and Fayer, R. 2016. Sarcocystosis of animals and human, 2nd ed. Boca Raton, FL: CRC Press. Inc., pp: 243–8. Dzahabiyyah, N. 2025. Deteksi Sarcocystis sp. pada Feses Ular Sanca Kembang (Malayopython reticulatus) Dengan Metode Pemeriksaan Feses. Universitas Airlangga (Unpublised). Faghiri, E., Davari, A. and Nabavi, R. 2019. Histopathological survey on Sarcocystis species infection in slaughtered cattle of Zabol, Iran. Turkiye. Parazitol. Derg. 43(4), 182–186. Fayer, R. and Prase, K.W. 1981. Hematology of experimental Sarcocystis bovicanis infection in calves. I. cellular and serologic changes. Vet. Pathol. 18, 351–357. Gabay, C. 2006. Interleukin-6 and chronic inflammation. Arthritis. Res. Therapy. 8(Suppl 2), S3. Ginting, S. and Jakel, T. 2005. Discovery of Sarcocystis singaporensis, a protozoan parasite, for rat control in North Sumatera, Indonesia. Jurnal. Penelitian. Pertanian. 25(1), 57–61. Available via https://www.researchgate.net/publication/262726109 Halimatunsadiah, A.B., Norida, M., Omar, D. and Kamarulzaman, N.H. 2016. Application of in pest management: the case of lowland vegetable growers. Int. Food Res. J. 23(1), 85–94. Available via https://www.researchgate.net/publication/291287587 Hamdan, M.S., Priyambodo, S. and Hindayana, D. 2020. Preferensi serangan tikus sawah (Rattus argentiventer) terhadap tanaman padi. Agrovigor 13(1), 16–21; doi:10.21107/agrovigor.v13i1.6249 Hong, E.J., Sim JS Chae., Kim J Park., Choi DH Yu., Park JG Yoo. and Park, B.K. 2016. Ultrastructural and molecular identification of Sarcocystis tenella (Protozoa, Apicomplexa) in naturally infected Korean natice goats. Vet. Med. 61(7), 374–381. Jakel, T., Burgstaller, H. and Frank, W. 1996. Sarcocystis singaporensis: studies on host specificity, pathogenicity, and potential use as a biocontrol agent in wild rats. J. Parasitol. 82(2), 280–287. Jakel, T., Burgstaller, H. and Frank, W. 1997. Sarcocystis singaporensis: studies on host specificity, pathogenicity, and potential use as biocontrol agent of wild rats. J. Parasitol. 82, 280–287; doi:10.2307/3284161 Jäkel, T., Khoprasert, Y., Endepols, S., Archer-Baumann, C., Suasa-Ard, K., Promkerd, P., Kliemt, D., Boonsong, P. and Hongnark, S. 1999. Biological control of rodents using Sarcocystis singaporensis. Intern. J. Parasitol. 29(8), 1321–1330; doi:10.1016/S0020-7519(99)00081-8 Jäkel, T., Khoprasert, Y., Sorger, I., Kliemt, D., Seehabutr, V., Suasa-Ard, K. and Hongnark, S. 1997. Sarcosporidiasis in rodents from Thailand. J. Wildl. Dis. 33, 860–867; doi:10.7589/0090-3558-33.4.860 Jäkel, T., Raisch, L., Richter, S., Wirth, M., Birenbaum, D., Ginting, S., Khoprasert, Y., Mackenstedt, U. and Wassermann, M. 2023. Morphological and molecular phylogenetic characterization of Sarcocystis kani sp. nov. and other novel, closely related Sarcocystis spp. infecting small mammals and colubrid snakes in Asia. Int. J. For. Parasitol. Parasites. Wildlife. 22, 184–198; doi:10.1016/j.ijppaw.2023.10.005 Jakel, T., Scharpfenecker, M., Jitrawang, P., Ruckle, J., Kliemt, D., Mackenstedt, U., Hongnark, S. and Khoprasert, Y. 2001. Reduction of transmission stages concomitant with increased host immune responses to hyper virulent S. singaporensis and natural selection for intermediate virulence. Int. J. For Parasitol. 31(14), 1639–1647; doi:10.1016/S0020-7519(01)00289-2 Kabir, M.H. and Rainis, R. 2015. Adoption and intensity of integrated pest management (IPM) vegetable farming in Bangladesh: an approach to sustainable agricultural development. Environ. Dev. Sustain. 17(6), 1413–1429; doi:10.1007/s10668-014-9613-y Kan, S.P. and Pathmanathan, R. 1991. Review of sarcocystosis in Malaysia. Southeast. Asian. J. Trop. Med. Public. Health. 22, 129–134. Khoprasert, Y., Promkerd, P., Ginting, S. and Jakel, T. 2008. Combination of biological rodent control using Sarcocystis singaporensis anticoagulant rodenticides. In Crop protection research advances. New York, NY: Nova Science Publishers, Inc., pp: 151–67. Available from: https://www.researchgate.net/publication/262726139. Lau, Y.L., Chang, P.Y., Subramaniam, V., Han Ng, Y., Mahmud, R., Ahmad, A.F. and Fong, M.Y. 2013. Genetic assemblage of Sarcocystis spp. in Malaysian snakes. Parasit. Vectors 6(257), 1–6; doi:10.1186/1756-3305-6-257 Máca, O., Kouba, M., Korpimäki, E. and González-Solís, D. 2021. Molecular identification of Sarcocystis sp. (Apicomplexa, Sarcocystidae) in offspring of Tengmalm’s Owls, Aegolius funereus (Aves, Strigidae). Front. Vet. Sci. 8(804096), 1–8; doi:10.3389/fvets.2021.804096 Mahaffey, E.A., George, J.W., Duncan, J.R., Prasse, K.W. and Fayer, R. 1986. Hematologic values in calves infected with S. cruzi. Vet. Parasitol. 19, 75–280; doi:10.1016/0304-4017(86)90074-9 Mahmudah, S.N. 2006. Pengaruh Infeksi Ookista Eimeria tenella Hasil Penyinaran Ultraviolet Terhadap Gambaran Patologis Sekum dan Jumlah Produksi Ookista pada Ayam Pedaging [Skripsi]. University of Airlangga (unpublished). Mohammed, B.M.E., Rabie, S.A., Hassanine, R.M.E., Hassan, A.A., Elhussien, O.F.A., Ghoneum, M. and El-Gerbed, M.S.A. 2023. Molecular identification, pathogenesis, and life cycle of Sarcocystis cruzi isolated from cattle (Bos taurus) in New Valley Governorate, Egypt. J. Parasitol. Res. 2023, 1–16; doi:10.1155/2023/7829290 Mohd Fadil, N.F., Tengku-Idris, T.I.N., Shahari, S., Fong, M.Y. and Lau, Y.L. 2019. Molecular evidence of Sarcocystis species infecting reptiles in Peninsular Malaysia. Iran. J. Parasitol. 14, 623–630. Moré, G.A. 2024. Sarcocystosis in animals. Rahway, NJ: Merck & Co., Inc. Available via https://www.merckvetmanual/com/musculoskeletalsystem/sarcocystosis/sarcocyst-in-animals (Accessed 14 June 2025). Mufa, R.M.D., Lastuti, N.D.R., Rantam, F.A., Suwanti, L.T., Suprihati, E. and Handijatno, D. 2020. Deteksi Cryptosporidium canis pada Anjing di Kota Surabaya. Jurnal Veteriner 21(2), 176–182. O’Donoghue, P.J., Watts, C.H.S. and Dixon, B.R. 1987. Ultrastructure of Sarcocystis spp. (Protozoa: apicomplexa) in rodents from North Sulawesi and West Java, Indonesia. J. Wildl. Dis. 23, 225–232; doi:10.7589/0090-3558-23.2.225 Portella, L.P., Fernandes, F.D., Rodrigues, F.D.S., Minuzzi, C.E., Sangioni, L.A., Flores, M.M. and Vogel, F.S.F. 2021. Macroscopic, histological, and molecular aspects of Sarcocystis spp. infection in tissues of cattle and sheep. Rev. Bras. Parasitol. Vet. 30(3), e003621; doi:10.1590/S1984-29612021050 Putri, F.M., Sitasiwi, A.J., Isdadiyanto, S. and Mardati, S.M. 2023. Profil Leukosit Tikus Jantan (Rattus novergicus L) Galur Sprague Dawley setelah Paparan Nanokitosan Ekstrak Etanol Daun Mimba (Azadirachta indica A. Juss.). Jurnal. Sain. Veteriner. 41, 29–36; doi:10.22146/jsv.75946 Qin, T., Ortega-Perez, P., Wibbelt, G., Lakim, M.B., Ginting, S., Khoprasert, Y., Wells, K., Hu, J. and Jäkel, T. 2024. A cyst forming coccidian with large geographical range infecting forest and commensal rodents: Sarcocystis muricoelognathis sp. nov. Parasites. Vectors. 17, 135; doi:10.1186/s13071-024-06230-8 Rathish, B.K.R. 2022. Sarcocystis. Treasure Island, FL: StatPearls Publishing; 2025. Available via https://www.ncbi.nlm.nih.gov/books/NBK574577/ (Accessed 12 September 2022) Rosalia, N.T. 2025. Gambaran histopatologi organ Jantung Tikus Putih (Rattus norvegicus) yang Terinfeksi Sarcocystis singaporensis. Universitas Airlangga. (unpublished). Sarra, S. and Peters, D. 2003. The rice yellow mottle virus is transmitted by cows, donkeys, and grass rats in irrigated rice crops. Plant. Dis. 87(7), 804–808; doi:10.1094/PDIS.2003.87.7.804 Šlapeta, J., Kyselová, I., Richardson, A., Modrý, D. and Lukeš, J. 2002. Phylogeny and sequence variability of the Sarcocystis singaporensis Zaman and Colley (1975) 1976 ssrDNA. Parasitol. Res. 88, 810–815; doi:10.1007/s00436-002-0657-9 Sudarmaji. and dan Herawati, N.A. 2017. Perkembangan populasi tikus sawah pada lahan sawah irigasi dalam pola indeks pertanaman padi 300. Penelitian Pertanian Tanaman Pangan 1(2), 125–131; doi:10.21082/jpptp.v1n2.2017.p125-13 Swar, S.O. and Shnawa, B.H. 2021. Recent advances in the molecular characterization of Sarcocystis species in some meat-producing animals: an updated review. Asian J. Agric. Biol. 1(1), 2021; doi:10.35495/ajab.2020.09.502 Tenter, A.M. 1995. Current research on Sarcocystis species of domestic animals. Int. J. Parasitol. 25(11), 1311–1330; doi:10.1016/0020-7519(95)00068-d Van Den Berg, H. and Jiggins, J. 2007. Investing in farmers: the impacts of farmer field schools in relation to integrated pest management. World Develop. 35(4), 663–686; doi:10.1016/j.worlddev.2006.05.004 Wassermann, M., Raisch, L., Lyons, J.A., Natusch, D.J.D., Richter, S., Wirth, M., Preeprem, P., Khoprasert, Y., Ginting, S., Mackenstedt, U. and Jäkel, T. 2017. Examination of Sarcocystis spp. of giant snakes from Australia and Southeast Asia confirms presence of a known pathogen-Sarcocystis nesbitti. PLos One 12(11), 1–16; doi:10.1371/journal.pone.0187984 Watthanakaiwan, V., Sukmak, M., Hamarity, K., Kaolom, N., Wajjwalku, W. and Muangkram, Y. 2017. Molecular characterization of the ribosomal DNA unit of S.singaporensis, S. zamani, and S. zuoi from rodents in Thailand. J. Vet. Med. Sci. 79(8), 1412–1418; doi:10.1292/jvms.16-0086 Xuan, T., Yen, T. and Caugh, T. 2021. Understanding farmer and government measure in rat pests control in rice fields. J. La. Lifesci. 2(2), 24–30; doi:10.37899/journallalifesci.v2i2.373 Zaman, V. and Coley, F.C. 1975. Light and electron microscopic observations of the life cycle of Sarcocystis orientalis sp. in the rat (Rattus norvegicus) and the Malaysian reticulated python (Python reticulatus). Zeitschrift Fuer. ParasitenKunde. 47, 169–185; doi:10.1007/BF00380518 Zaman, V. 1976. Host range of S. orientalis. Southeast. Asian. J. Trop. Med. Public. Health. 1, 112. Zhu, G., Keithly, J.S. and Philippe, H. 2000. What is the phylogenetic position of Cryptosporidium?. Int. J. Syst. Evol. Microbiol. 50(4), 1673–1681; doi:10.1099/00207713-50-4-1673 | ||
| How to Cite this Article |
| Pubmed Style Aksono EB, Sunarso A, Yunus M, Suwanti LT, Mufasirin , Sediyoadi L, Putra AS, Rosalia NT, Dzahabiyyah N. Isolation and phylogenetic characterization of Sarcocystis singaporensis isolates from Surabaya python snakes (Malayopython reticulatus) as a biological control agent for rats. Open Vet. J.. 2026; 16(1): 147-156. doi:10.5455/OVJ.2026.v16.i1.14 Web Style Aksono EB, Sunarso A, Yunus M, Suwanti LT, Mufasirin , Sediyoadi L, Putra AS, Rosalia NT, Dzahabiyyah N. Isolation and phylogenetic characterization of Sarcocystis singaporensis isolates from Surabaya python snakes (Malayopython reticulatus) as a biological control agent for rats. https://www.openveterinaryjournal.com/?mno=289507 [Access: January 31, 2026]. doi:10.5455/OVJ.2026.v16.i1.14 AMA (American Medical Association) Style Aksono EB, Sunarso A, Yunus M, Suwanti LT, Mufasirin , Sediyoadi L, Putra AS, Rosalia NT, Dzahabiyyah N. Isolation and phylogenetic characterization of Sarcocystis singaporensis isolates from Surabaya python snakes (Malayopython reticulatus) as a biological control agent for rats. Open Vet. J.. 2026; 16(1): 147-156. doi:10.5455/OVJ.2026.v16.i1.14 Vancouver/ICMJE Style Aksono EB, Sunarso A, Yunus M, Suwanti LT, Mufasirin , Sediyoadi L, Putra AS, Rosalia NT, Dzahabiyyah N. Isolation and phylogenetic characterization of Sarcocystis singaporensis isolates from Surabaya python snakes (Malayopython reticulatus) as a biological control agent for rats. Open Vet. J.. (2026), [cited January 31, 2026]; 16(1): 147-156. doi:10.5455/OVJ.2026.v16.i1.14 Harvard Style Aksono, E. B., Sunarso, . A., Yunus, . M., Suwanti, . L. T., Mufasirin, ., Sediyoadi, . L., Putra, . A. S., Rosalia, . N. T. & Dzahabiyyah, . N. (2026) Isolation and phylogenetic characterization of Sarcocystis singaporensis isolates from Surabaya python snakes (Malayopython reticulatus) as a biological control agent for rats. Open Vet. J., 16 (1), 147-156. doi:10.5455/OVJ.2026.v16.i1.14 Turabian Style Aksono, Eduardus Bimo, Agus Sunarso, Muchammad Yunus, Lucia Tri Suwanti, Mufasirin, Luhur Sediyoadi, Adam Sediyoadi Putra, Nadia Tasya Rosalia, and Nafilah Dzahabiyyah. 2026. Isolation and phylogenetic characterization of Sarcocystis singaporensis isolates from Surabaya python snakes (Malayopython reticulatus) as a biological control agent for rats. Open Veterinary Journal, 16 (1), 147-156. doi:10.5455/OVJ.2026.v16.i1.14 Chicago Style Aksono, Eduardus Bimo, Agus Sunarso, Muchammad Yunus, Lucia Tri Suwanti, Mufasirin, Luhur Sediyoadi, Adam Sediyoadi Putra, Nadia Tasya Rosalia, and Nafilah Dzahabiyyah. "Isolation and phylogenetic characterization of Sarcocystis singaporensis isolates from Surabaya python snakes (Malayopython reticulatus) as a biological control agent for rats." Open Veterinary Journal 16 (2026), 147-156. doi:10.5455/OVJ.2026.v16.i1.14 MLA (The Modern Language Association) Style Aksono, Eduardus Bimo, Agus Sunarso, Muchammad Yunus, Lucia Tri Suwanti, Mufasirin, Luhur Sediyoadi, Adam Sediyoadi Putra, Nadia Tasya Rosalia, and Nafilah Dzahabiyyah. "Isolation and phylogenetic characterization of Sarcocystis singaporensis isolates from Surabaya python snakes (Malayopython reticulatus) as a biological control agent for rats." Open Veterinary Journal 16.1 (2026), 147-156. Print. doi:10.5455/OVJ.2026.v16.i1.14 APA (American Psychological Association) Style Aksono, E. B., Sunarso, . A., Yunus, . M., Suwanti, . L. T., Mufasirin, ., Sediyoadi, . L., Putra, . A. S., Rosalia, . N. T. & Dzahabiyyah, . N. (2026) Isolation and phylogenetic characterization of Sarcocystis singaporensis isolates from Surabaya python snakes (Malayopython reticulatus) as a biological control agent for rats. Open Veterinary Journal, 16 (1), 147-156. doi:10.5455/OVJ.2026.v16.i1.14 |