| Research Article | ||
Open Vet. J.. 2025; 15(12): 6367-6372 Open Veterinary Journal, (2025), Vol. 15(12): 6367-6372 Research Article Ex vivo validation of colonoscopy-guided colon biopsy in dromedary camelsGhaliah Al-Shehab and Abdulaziz H. Almuhanna*Department of Clinical Science, College of Veterinary Medicine, King Faisal University, Al-Ahsa, Saudi Arabia *Corresponding Author: Abdulaziz H. Almuhanna. Department of Clinical Science, College of Veterinary Medicine, King Faisal University, Al-Ahsa, Saudi Arabia. Email: aalmohana [at] kfu.edu.sa Submitted: 28/08/2025 Revised: 02/11/2025 Accepted: 11/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
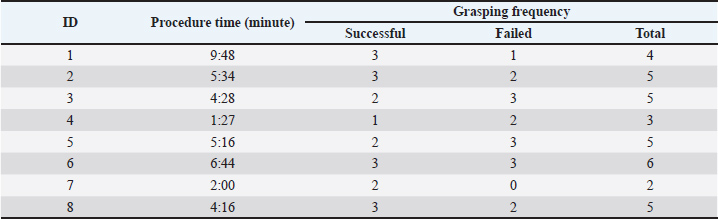
AbstractBackground: The histological evaluation of colon tissues offers valuable information for assessing the health and disease process in the intestine of camels. Currently, colon samples are routinely collected during postmortem examination, whereas antemortem sampling is not a common practice. Colonoscopy-guided colon biopsy can be used as a minimally invasive alternative. However, preclinical validation of the technique is essential before clinical translation. Aim: This study aimed to evaluate the applicability of colonoscopy-guided biopsy and microscopically assess the quality of the collected tissue. Methods: Colon biopsies (n=8) were obtained by colonoscopy using intestinal samples collected from the abattoir. A blunt uterine biopsy punch was used to grasp the tissue. The collected tissues were fixed in formalin and processed in paraffin. Sections were then cut and stained with H&E for histological architecture evaluation. Results: Endoscopy provided clear visualization of the colon lumen and enabled appropriate tissue collection. The procedure was performed quickly and easily, and no evidence of full intestinal perforation was found in any samples. The quality of the histological sections was good, allowing a satisfactory assessment of the mucosal and submucosal layers. Conclusion: Colonoscopy-guided colon biopsy is feasible in practice and a promising clinical technique for live camels. Keywords: Camel, Colonoscopy, Colon, Endoscope, Diagnostics. IntroductionInterest in camel raising in the Gulf Region, especially in the Kingdom of Saudi Arabia, has been growing exponentially for many decades (Ali et al., 2018). Camels are one of the most preferred animal species kept by rural and suburban residents because they are a great source of milk and meat and provide a means of transportation for nomadic people in the desert (Abrhaley and Leta, 2018). Although camels have a great resilience for coping with the harsh environment in the desert, they remain susceptible to various digestive disturbances (Niehaus and Mora, 2022). Currently, there is a wealth of scientific publications in the veterinary literature discussing the innovative diagnostic approaches for assessing equine gastrointestinal diseases, such as endoscopy (Vokes et al., 2023), ultrasonography (Le Jeune and Whitcomb, 2014), radiography (Kelleher et al., 2014), and computed tomography (Manso-Díaz et al., 2015), whereas diagnostic studies for gastrointestinal disorders in camels are underrepresented. Digestive endoscopy provides a direct, safe, and quick evaluation of the upper and lower digestive tracts, and it is routinely employed in equine practice settings; however, it is less frequently used in ruminants (Franz et al., 2024). In addition, the role of endoscopy in camel medicine, especially for assessing the digestive tract, is rarely documented. Recently, a group of researchers validated and optimized the use of endoscopy to visualize the interior of the rumen in clinically healthy camel calves, which is deemed as a direct method to assess the content and mucosal ruminal appearance and also enables the evaluation of ruminal motility (Almuhanna et al., 2025). Such advancement in the diagnostic imaging of the digestive tract of camels would significantly enhance their welfare. The improvement of camel welfare implies the application of minimally invasive clinical techniques instead of those involving invasive procedures, such as exploratory laparotomy. The majority of published work related to the use of minimally invasive clinical techniques involved the utilization of ultrasound-guided clinical procedures, which have been frequently used for the assessment of the abdominal viscera in apparently healthy camels (Tharwat, 2020), to identify the pathological abnormalities associated with John’s disease in camels (Tharwat et al., 2012), to characterize the normal ultrasonographic appearance of liver and kidneys (Tharwat et al. 2012; Tharwat et al., 2025), and to assess the sonographic characteristics of lung and pleura in normal camels (Tharwat, 2013). On the other hand, endoscopy-guided clinical techniques are lacking, possibly due to procedural standardization. Therefore, there is an urgent need to employ endoscopy in camel practice as it can be performed rapidly with no harmful effects on the examined animals. Camels are affected by a wide range of diseases, including parasitosis (Tharwat et al., 2025), chronic parasitism (Locklear et al., 2021), intestinal lymphoma (Simmons et al., 2005), and adenocarcinoma (Tharwat et al., 2018). Some of these diseases are only detected during postmortem examination, and early diagnosis can be challenging. Tissue biopsy could be a valuable alternative for assessing normal histological appearance and could subsequently enable the detection of any possible histopathological abnormalities related to the underlying disorders. Therefore, a colon biopsy would be an additional diagnostic modality for detecting some intestinal disorders in camels. This study aimed to standardize and optimize a colonoscopy-guided colon tissue biopsy technique in camels. The current study was conducted using postmortem samples as an initial simulation. We expect that the validation of such experiments will pave the way toward the clinical application of colon biopsy. Materials and MethodsThe study was conducted on cadaver samples collected from eight adult dromedary camels. The animals were initially examined clinically prior to slaughter to exclude any underlying systemic disease and to ensure that all animals were healthy. Following that, each camel was slaughtered and subsequently hoisted. The skin surrounding the anal sphincter was carefully dissected to facilitate its detachment with the carcass being hung on the rail. Subsequently, the abdominal wall was opened and the abdominal viscera exposed. Then, the caudal intestinal segment, including the anal sphincter, rectum, and part of the colon, was carefully dissected from the surrounding viscera. The length of the harvested samples, extending from the anal sphincter to the point where the colon was cut, was approximately 1.2–1.5 m. The intestinal lumen of each sample was flushed with tap water to enable the removal of fecal contents and to enhance the cleanliness of the intestinal mucosa. The sample collection process was completed within 2 hours. Subsequently, the samples were kept refrigerated until use. Each sample was placed horizontally on a table upon arrival at the laboratory. Initially, the cranial end of the colon was firmly ligated using a non-absorbable suture, while the anal sphincter was maintained closed through the application of a rubber band placed around the exterior of the rectum, immediately cranial to the anal sphincter. This step was performed to prevent air leakage during sample inflation. Once both ends were secured, a 1.5 m flexible equine endoscopy, with 8 mm outer diameter, was used (VET-3313; GUANGZHOU TUESHEN MEDICAL EQUIPMENT CO., LTD. CHINA). The endoscopy is powered with irrigation and insufflation systems and provided with an LED light source. The procedure required two personnel: one handling the scope and the other performing the tissue collection. Once the sample was entirely prepared for collection, the endoscope was inserted through the anal sphincter, advanced through the rectum, and eventually placed in the colon. Subsequently, a 90 cm long equine uterine biopsy punch was used to allow colon biopsy sampling. With the aid of endoscopy, the mucosa of the colon was firmly grasped by the biopsy punch. Care was also taken not to grasp a deep chunk of the colon tissue to prevent wall perforation. Several tissue sites were biopsied from each sample, enabling the collection of 19 biopsy samples. Each harvested tissue was labeled and then fixed in 10% formalin buffer. Each sample was then processed, embedded in paraffin, and stained with hematoxylin and eosin. The procedure time and grasping frequency (involving successful and failed trials) for each sample were calculated and reported (Table 1). Table 1. Details of the colonoscopy-guided colon biopsy, including the procedure interval and the grasping frequency, including the successful and failed trials.
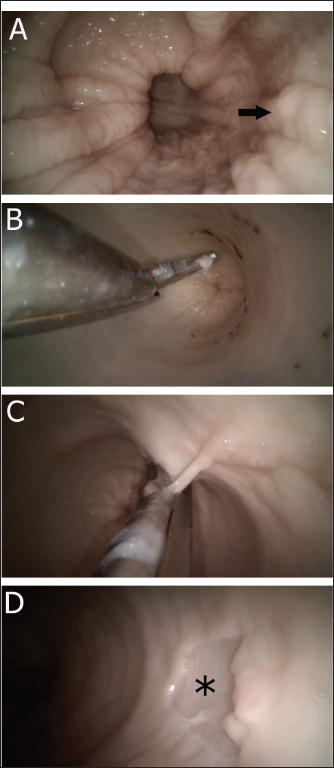
Ethical approvalNot needed for this study. ResultsThe procedure was performed without difficulty. None of the samples exhibited evidence of complete colon perforation. The intestinal segment was easily inflatable, and the lumen was clearly visualized. The rectal mucosa appeared rosy pink, shining, and had a longitudinally folded appearance. On the other hand, the colon lumen appeared cylindrical-shaped, glistening, and had a smooth surface (Fig. 1). The mean procedure time (including both successful and failed trials) was 4:56 minutes. A total of 19 tissue specimens were successfully collected from the colon samples. In each sampling procedure, the colon mucosa tended to slip out from the biopsy punch, especially when a small chunk of tissue was grasped.
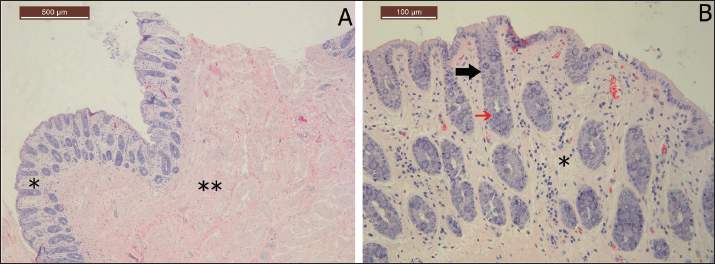
Fig. 1. Colonoscopy-guided colon biopsy technique. Figure A shows the endoscopic appearance of the rectum revealing longitudinal rectal folds (arrow). Figure B reveals the appearance of the colon before tissue grasping with the colon lumen being inflated and smooth and glistening mucosa. Figure C shows the biopsy punch while the tissue is grasped. Figure D illustrates the colon mucosa site after tissue collection (asterisk). Microscopic examination of the histology sections revealed that all grasped tissues, including even small-sized biopsies, yielded identifiable mucosal and submucosal architectures. Complete smearing or smashing of the colon tissue was not observed in any sample. The sections were mainly composed of the mucosal layer, with numerous crypts of Lieberkuhn glands and goblet cells within the lamina propria and loose connective tissue in the submucosa (Fig. 2).
Fig. 2. Histological morphology of colon tissue stained with H&E. Figure A shows the general appearance of the collected tissue from the colon illustrating the mucosal layer (asterisk) and the submucosal layer (double asterisk). The colon crypt is indicated by a black arrow in Figure B, while the red arrow indicates goblet cells. The lamina propria in Figure B is labeled with an asterisk. DiscussionPrioritizing the application of less invasive clinical methods over those that require major surgery has a huge advantage for the welfare of camels. Endoscopy-guided colon biopsy is a minimally invasive procedure that has the potential to expand the understanding of some unexplored diseases in camel medicine. The technique is not time-consuming and is likely to be safe when performed by experienced clinicians. The biopsy technique of the colon tissue is feasible, repeatable, and provides a satisfactory histological assessment. Although this study was conducted on cadaver samples, the impact of our findings would support the possible translation of this technique into its clinical application. The initial validation of cadaver samples aligns with the application of the 3Rs in animal research, which involves replacement, reduction, and refinement (Cheluvappa et al., 2017). This study represented an effective replacement of live animals with cadaveric colon samples, thus avoiding unnecessary pain, suffering, stress, and mucosal hemorrhage. Traditionally, colon tissue samples have been collected from diseased camels during postmortem examination (Alharbi et al., 2012). However, this method is associated with tissue autolysis, especially when sampling is not performed immediately after slaughter (Cocariu et al., 2016). Furthermore, if samples are not handled properly during transportation, they are likely to be subjected to tissue damage (Brooks, 2016). In contrast, sampling via laparotomy has been successfully achieved in horses and enabled full-thickness biopsy of the intestine for the diagnosis of inflammatory bowel disease (Boshuizen et al., 2018). However, this method requires the use of general anesthesia, is invasive, has a risk of wound contamination post-surgery, and requires intensive postoperative care. In contrast, colon biopsy with the aid of endoscopy can be performed when the animal is slightly sedated and in sternal recumbency (or) when the animal is standing.Moreover, the aforementioned complications are likely to be avoided when a colonoscopy-guided colon biopsy is performed. The possible futuristic clinical application of this technique would aid in the diagnosis of many diseases affecting the intestine of camel, especially those with a chronic nature, such as paratuberculosis or neoplasia. In a previous study by Hereba et al. (2015), the pathological examination of the camels affected with paratuberculosis revealed that the mucosa had obvious corrugation, which is often severe enough to be manually palpated during rectal examination. Similarly, in horses, gastrointestinal biopsy is useful for identifying the exact cause of chronic undifferentiated diarrhea by detecting the type of leukocytic infiltration in histopathological sections (Hostetter and Uzal, 2022). The application of colon biopsy, with the aid of endoscopy, would also allow better detection of diseases affecting camel’s intestine, such as paratuberculosis, and coccidiosis. Additionally, it would pave the way for the identification of unknown disorders. Our results indicated that the histopathological architecture was preserved in all samples, and no histological artifacts were found. In normal camels, the normal mucosal layer of the colon is composed of columnar epithelium with prominent Lieberkuhn crypts, which appear as inward invagination of the intestinal mucosa down into the lamina propria, as observed in our samples (Mohamed et al., 2018). Goblet cells, specialized glands responsible for mucus secretion, were abundantly found in the histological sections (Al-Samawy et al., 2019). It is expected that when this method is incorporated into practice in the future, it would facilitate the identification of histopathological lesions associated with the disease process. The possible limitations associated with our study include that the application of biopsy on cadaver samples does not allow for the assessment of the effect of peristalsis on grasping efficacy, intestinal inflation challenges, mucosal hemorrhage extent, and patient tolerance. These factors should be considered when the procedure is repeated on live animals. However, a blind colon biopsy has been reported with great success in horses. The clinician manually holds the rectal mucosa to direct it into the arms of the biopsy punch and eventually collects the required tissue (Mair et al., 2011). Therefore, the clinical application of this technique in camels can be practically replicated and refined. In conclusion, the validation and optimization of colonoscopy-guided colon biopsy on cadaver samples have the potential to replicate the technique in clinical cases. Endoscopy enables a clear visualization of the intestinal lumen and allows proper grasping of the colon tissue. The histological assessment of the mucosa holds the promise of advancing our understanding of enteric abnormalities affecting dromedary camels. AcknowledgmentsThe authors would like to thank Professor Saeed Al-Ramadan and Faten Alrasheed for their assistance in evaluating the histology sections. Conflict of interestThe authors have no conflicts of interest to declare. FundingThis work was supported by the Deanship of Scientific Research, Vice Presidency for Graduate Studies and Scientific Research, King Faisal University, Saudi Arabia, grant number (KFU254054). Authors' contributionsAbdulaziz Almuhanna designed the study and collected the intestinal samples. Both authors equally participated in the biopsy sampling procedure using colonoscopy. Abdulaziz Almuhanna wrote the manuscript, and Ghaliah Al_Shehab revised it. Both authors have agreed to the final version of the manuscript. Data availabilityAll data are available within the manuscript. ReferencesAbrhaley, A. and Leta, S. 2018. Medicinal value of camel milk and meat. J. Appl. Anim. Res. 46, 552–558. Alharbi, K.B., Al-Swailem, A., Al-Dubaib, M.A., Al-Yamani, E., Al-Naeem, A., Shehata, M. and Mahmoud, O.M. 2012. Pathology and molecular diagnosis of paratuberculosis of camels. Trop. Anim. Health Prod. 44, 173–177. Ali, R.M.E., Ahmed, S.H. and Al-Mahish, M.A. 2018. Camel production in Kingdom of Saudi Arabia: Economic and environmental impacts. Fundam. Appl. Agric. 3, 602–608. Almuhanna, A.H., Elnahas, A., Zabady, M.K., El-Hawari, S., Marzok, M., Eldeeb, W. and Khalaphallah, A. 2025. Ruminoscopy in apparently healthy camel calves (Camelus dromedarius): A technique description and ruminoscopic observations. Open Vet. J. 15, 2122–2126. Boshuizen, B., Ploeg, M., Dewulf, J., Klooster, S., de Bruijn, M., Picavet, M.-T. and Theelen, M. 2018. Inflammatory bowel disease (IBD) in horses: a retrospective study exploring the value of different diagnostic approaches. BMC Vet. Res. 14, 21. Brooks, J.W. 2016. Postmortem changes in animal carcasses and estimation of the postmortem interval. Vet. Pathol. 53, 929-940. Cheluvappa, R., Scowen, P. and Eri, R. 2017. Ethics of animal research in human disease remediation, its institutional teaching; and alternatives to animal experimentation. Pharmacol. Res. Perspect. 5, 332. Cocariu, E.A., Mageriu, V., Stăniceanu, F., Bastian, A., Socoliuc, C. and Zurac, S. 2016. Correlations between the autolytic changes and postmortem interval in refrigerated cadavers. Rom. J. Intern. Med. 54, 105–112. Franz, S., Hofer, L. and Dadak, A.M. 2024. The role of endoscopy in bovine internal medicine – a review of current indication fields. Vet. J. 302, 106093. Hereba, A.M., Hamouda, M.A. and Al-Hizab, F.A. 2015. Pathological and molecular diagnosis of paratuberculosis among dromedary camels in Saudi Arabia. J. Anim. Plant Sci. 25, 997–1002. Hostetter, J.M. and Uzal, F.A. 2022. Gastrointestinal biopsy in the horse: overview of collection, interpretation, and applications. J. Vet. Diagn. Invest. 34, 376–388. Kelleher, M.E., Puchalski, S.M., Drake, C. and Le Jeune, S.S. 2014. Use of digital abdominal radiography for the diagnosis of enterolithiasis in equids: 238 cases (2008–2011). J. Am. Vet. Med. Assoc. 245, 126–129. Le Jeune, S. and Whitcomb, M.B. 2014. Ultrasound of the equine acute abdomen. Vet. Clin. North Am. Equine Pract. 30, 353–381. Locklear, T.R., Videla, R., Breuer, R.M., Mulon, P.-Y., Passmore, M., Mochel, J.P. and Smith, J.S. 2021. Presentation, clinical pathology abnormalities, and identification of gastrointestinal parasites in camels (Camelus bactrianus and Camelus dromedarius) presenting to two North American veterinary teaching hospitals: A retrospective study (1980–2020). Front. Vet. Sci. 8, 651672. Mair, T.S., Kelley, A.M. and Pearson, G.R. 2011. Comparison of ileal and rectal biopsies in the diagnosis of equine grass sickness. Vet. Rec. 168, 266. Manso-Díaz, G., García-López, J.M., Maranda, L. and Taeymans, O. 2015. The role of head computed tomography in equine practice. Equine Vet. Educ. 27, 136–145. Mohamed, A.A., Kadhim, K.H. and Hussein, D.M. 2018. Morphological and histological study of the cecum and colon in adult local Camelus dromedarius. Adv. Anim. Vet. Sci. 6, 286–291. Niehaus, A.J. and Mora, D.P. 2022. Digestive system and abdomen. In: Medicine and Surgery of Camelids (3rd Ed.), John Wiley & Sons, Hoboken, NJ, USA, pp. 336–390. Al-Samawy, R.M., Al-Saffar, F.J. and Kadhim, D.M. 2019. Histological and histochemical study on the large intestine of one-humped camel in Iraq. Asian J. Agric. Biol. 7, 373–380. Simmons, H.A., Fitzgerald, S.D., Kiupel, M., Rost, D.R. and Emery, R.W. 2005. Multicentric T-cell lymphoma in a dromedary camel (Camelus dromedarius). J. Zoo Wildl. Med. 36, 727–729. Tharwat, M. 2013. Ultrasonography of the lungs and pleura in healthy camels (Camelus dromedarius). Acta Vet. Hung. 61, 309–318. Tharwat, M. 2020. Ultrasonography of the abdomen in healthy and diseased camels (Camelus dromedarius): A review. J. Appl. Anim. Res. 48, 300–312. Tharwat, M., Al-Sobayil, F., Ali, A., Hashad, M. and Buczinski, S. 2012. Clinical, ultrasonographic, and pathologic findings in 70 camels (Camelus dromedarius) with Johne’s disease. Can. Vet. J. 53, 543–549. Tharwat, M., Al-Sobayil, F. and Buczinski, S. 2012. Ultrasound-guided hepatic and renal biopsy in camels (Camelus dromedarius): Technique development and assessment of safety. Small Rumin. Res. 103, 211–219. Tharwat, M., Ali, H. and Alkheraif, A.A. 2025. Clinical insights on paratuberculosis in Arabian camels (Camelus dromedarius): A review. Open Vet. J. 15, 8–15. Tharwat, M., El-Shafaey, E.-S., Sadan, M., Ali, A., Al-Sobayil, F. and Al-Hawas, A. 2018. Omaso-abomasal adenocarcinoma in a female Arabian camel (Camelus dromedarius). J. Appl. Anim. Res. 46, 1268–1271. Tharwat, M. and Elmoghazy, H.M.M. 2025. Ongoing evolution of urinary tract disorders in dromedary camels (Camelus dromedarius): A comprehensive illustrated sonographic overview. Front. Vet. Sci. 12, 1638275. Vokes, J., Lovett, A. and Sykes, B. 2023. Equine gastric ulcer syndrome: an update on current knowledge. Animals 13, 1261. | ||
| How to Cite this Article |
| Pubmed Style Al-shehab G, Almuhanna AH. Ex vivo validation of colonoscopy-guided colon biopsy in dromedary camels. Open Vet. J.. 2025; 15(12): 6367-6372. doi:10.5455/OVJ.2025.v15.i12.20 Web Style Al-shehab G, Almuhanna AH. Ex vivo validation of colonoscopy-guided colon biopsy in dromedary camels. https://www.openveterinaryjournal.com/?mno=280401 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.20 AMA (American Medical Association) Style Al-shehab G, Almuhanna AH. Ex vivo validation of colonoscopy-guided colon biopsy in dromedary camels. Open Vet. J.. 2025; 15(12): 6367-6372. doi:10.5455/OVJ.2025.v15.i12.20 Vancouver/ICMJE Style Al-shehab G, Almuhanna AH. Ex vivo validation of colonoscopy-guided colon biopsy in dromedary camels. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6367-6372. doi:10.5455/OVJ.2025.v15.i12.20 Harvard Style Al-shehab, G. & Almuhanna, . A. H. (2025) Ex vivo validation of colonoscopy-guided colon biopsy in dromedary camels. Open Vet. J., 15 (12), 6367-6372. doi:10.5455/OVJ.2025.v15.i12.20 Turabian Style Al-shehab, Ghaliah, and Abdulaziz H. Almuhanna. 2025. Ex vivo validation of colonoscopy-guided colon biopsy in dromedary camels. Open Veterinary Journal, 15 (12), 6367-6372. doi:10.5455/OVJ.2025.v15.i12.20 Chicago Style Al-shehab, Ghaliah, and Abdulaziz H. Almuhanna. "Ex vivo validation of colonoscopy-guided colon biopsy in dromedary camels." Open Veterinary Journal 15 (2025), 6367-6372. doi:10.5455/OVJ.2025.v15.i12.20 MLA (The Modern Language Association) Style Al-shehab, Ghaliah, and Abdulaziz H. Almuhanna. "Ex vivo validation of colonoscopy-guided colon biopsy in dromedary camels." Open Veterinary Journal 15.12 (2025), 6367-6372. Print. doi:10.5455/OVJ.2025.v15.i12.20 APA (American Psychological Association) Style Al-shehab, G. & Almuhanna, . A. H. (2025) Ex vivo validation of colonoscopy-guided colon biopsy in dromedary camels. Open Veterinary Journal, 15 (12), 6367-6372. doi:10.5455/OVJ.2025.v15.i12.20 |