Open Veterinary Journal, (2025), Vol. 15(12): 6253-6267
Research Article
10.5455/OVJ.2025.v15.i12.10
Biochemical and histopathological insights into different helminth infections in Saurida undosquamis with new host record of Ligula intestinalis
Hadeer Abd El-hak Rashed1, Layla Omran Elmajdoub2*, Raneem Ahmed Khalaf1,
Nourhan Elsayed Elkotby1, Salma Nabil Elfeky1, Nada Ayman Farghaly1, Aya Hossam Ali1,
Tasneem Mohamed Eltaweel1, Raneem Mohamed Mandour1 and Ali H. Abu Almaaty1
1Zoology Department, Faculty of Science, Port Said University, Port Said, Egypt
2Zoology Department, Faculty of Science, Misurata University, Misurata, Libya
*Corresponding Author: Layla Omran Elmajdoub. Zoology Department, Faculty of Science, Misurata University, Misurata, Libya. Email: elmajdoublayla [at] sci.misuratau.edu.ly
Submitted: 19/08/2025 Revised: 20/10/2025 Accepted: 02/11/2025 Published: 31/12/2025
© 2025 Open Veterinary Journal
This is an Open Access article distributed under the terms of the Creative Commons Attribution-Non Commercial-No Derivatives License (http://creativecommons.org/licenses/by-nc-nd/4.0/), which permits non-commercial re-use, distribution, and reproduction in any medium, provided the original work is properly cited, and is not altered, transformed, or built upon in any way.
Abstract
Background: Saurida undosquamis (brushtooth lizardfish) is a species of the Synodontidae family that is widely distributed and commercially significant. Parasites impact fish survival, affecting aquaculture and wild populations.
Aim: This study aimed to investigate the most prevalent helminths infecting different organs of S. undosquamis and to assess their impact on the overall health of the fish.
Methods: To investigate helminth infections, 200 S. undosquamis specimens were collected from local markets. Comparative analysis between uninfected and infected specimens was conducted using organ histopathology, evaluation of digestive tract mucus secretion patterns, and measurement of specific biochemical markers present in the mucus.
Results: Helminth infection affected 75% of the fish, primarily targeting the stomach and intestine, with Contracaecum sp. being the most prevalent and L. intestinal is being the least common, which is the first time that helminth infection has been recorded in this host. Larger fish were more susceptible to infection than smaller ones, with males showing a slightly higher infection rate than females. Histological analysis identified widespread tissue damage in multiple organs of infected fish. Infection was associated with increased mucus secretion and enzymatic changes, including elevated protease and lipase activities, as well as decreased α-amylase activity. Additionally, higher IgD levels were increased in individuals with heavy infection.
Conclusion: The study underscores the high prevalence and pathological effects of helminth infections in S. undosquamis, recommending further investigation into environmental factors influencing these infections. This also highlights the importance of understanding the emergence of L. intestinalis in this newly recorded host.
Keywords: S. undosquamis, Helminths, Dominance, Immunological, Enzymes.
Introduction
Saurida undosquamis (brushtooth lizardfish) are widely distributed. They are one of the essential and main commercial species belonging to the Synodontidae family from the Suez Gulf and Red Sea of Egypt (Abd-Elghany, 2017; Mehanna, 2022).
Fish serve as intermediate hosts for a diverse range of parasites spanning multiple taxonomic groups. These parasites typically complete part of their life cycle within fish, undergoing developmental stages that prepare them to infect their definitive hosts, such as birds, mammals, or other aquatic organisms. Fish harbor parasitic larvae, cysts, or eggs, which can develop into mature parasites and potentially cause zoonotic diseases when consumed by humans (Gabagambi et al., 2019).
Parasites significantly affect the survival of their fish hosts by decreasing growth rates, altering behavioral characteristics, and increasing susceptibility to additional pathogens, impacting both aquaculture and natural populations (Islam et al., 2024). The relationship between fish size, sex, and infection rates of different parasites has been estimated (Jerônimo et al., 2022; El-Fahla et al., 2024; Renner and Duggan, 2024). With the continued expansion of aquaculture, research on fish immune systems has become increasingly critical, given the detrimental effects of fish diseases on the entire production chain (Leung and Bates, 2013; Tavares-Dias and Martins, 2017).
Teleost fish possess an innate immune system comprising physical barriers such as scales, epidermis, and mucus; chemical barriers including epithelial serum lysozymes, complement system molecules, antimicrobial peptides, and lectins within the mucus layer; and microbiological barriers formed by commensal bacteria inhabiting the skin, gills, and intestines (Watts et al., 2001; Gómez and Balcázar, 2008).
Plasma, mucus, and other bodily fluids include various humoral components, such as transferrin, interferon, inhibitory proteins, lysozymes (operating individually or in cascades), antiproteases, type C lectins, pentraxins, natural antibodies, cytokines, and chemokines (Magnadóttir, 2006).
This study aimed to investigate the dominance of parasitic infections in S. undosquamis, with an emphasis on the interplay between host biological traits and infection severity, and to evaluate the subsequent effects of parasitism on enzymatic function, immune response, and histopathological integrity.
Materials and Methods
Chemicals
The protease assay kit (Cat. No: AR4011) was purchased from Boster Biological Technology, USA. Lipase (Cat. No: SL0113FI), alpha-amylase (Cat. No: SL0111FI), and immunoglobulin D (IgD; Cat. No: SL0100FI) kits were procured from SunLong Biotech Co., China. All assays were conducted using a double-beam UV-visible spectrophotometer (Model: 6850 UV/Vis, Jenway, England) and a MicroReader 4 Plus Hyperion system (Florida, USA) for enzyme-linked immunosorbent assay-based techniques.
Sample size
The sample size of 200 fish (Badawi et al., 2021; Rashed et al., 2024) was determined based on one-way ANOVA involving three groups. Assuming a moderate effect size (f=0.25), a significance level of p < 0.05, and a statistical power of 80%, the minimum required sample size was 159. Therefore, 200 fish were included to ensure adequate statistical power and account for potential data variability or sample loss.
Sampling area
Saurida undosquamis samples (Fig. 1) were obtained from commercial fish markets in Port Said, Egypt, from January to March 2024. The fish were transported under appropriate conditions to the Parasitology Laboratory at the Faculty of Science, Port Said University, for detailed examination. Species identification was conducted based on the morphometric characteristics described by Rashed et al. (2024).

Fig. 1. Photograph of S. undosquamis.
Parasitological examination
The liver, spleen, kidneys, and gonads were carefully removed and placed in clean Petri dishes following dissection. The organs were rinsed with 0.9% normal saline solution to remove any debris. For microscopic parasitological analysis, a standardized sample (0.1 g) was taken from each organ and crushed between two glass slides (Ghobashy et al., 2006).
The gastrointestinal tract was separated from the other organs. The stomach and intestine were then opened from one side under a high-intensity flashlight that facilitated the viewing of the internal contents during the cutting process to reduce the probability of slicing the helminths themselves. The mucosa was removed with a scalpel, and the contents were cleaned in a Petri dish with normal saline (Woodland, 2006). Finally, helminths were identified and captured.
Parasitological identification
Helminths were identified based on their morphological characteristics, as described in previous studies. Plerocercoids were classified by distinct morphological features, including overall body shape, coloration, presence of clefts, and bothria structure, when present (Blazhekovikj-Dimovska et al., 2021; Morsy et al., 2022; Elmajdoub et al., 2023a). Nematode larvae were identified according to worm coloration, ventricular appendix morphology, and pointed tooth position (Carvalho et al., 2020; Vergara-Flórez and Consuegra, 2021). Trematodes were differentiated based on the coloration of external and internal organs, sucker morphology, and the presence and form of the ecosoma, when applicable (Carreras-Aubets et al., 2011; EL-Garhy et al., 2017).
Parasitological dominance
The formula described by Bush et al. (1997) was used to compute prevalence (%), mean intensity, and abundance as follows:



where: Pr=Prevalence, MA=Mean abundance, MI=mean intensity, N=total number of examined fish, n=total number of infected fish, and Rp=recovered parasites.
The parasitological indices of helminths in S. undosquamis were analyzed in terms of fish weight, length, and sex. The fish ranged in weight from 68 to 250 g and in length from 22 to 35 cm. Fish were categorized into two weight classes (<113 and >113 g) and two length classes (<25 and >25 cm). Sex determination followed the method of Guerrero and Shelton (1974).
One criterion was applied with modification according to Rashed et al. (2025) to categorize infection severity for further analysis. Samples with no detectable parasites were labeled as uninfected, those with fewer than 25 parasites were considered low-infected, and samples with infection varied between 25 and 50 were described as moderate. Finally, those with more than 50 parasites were classified as highly infected.
Histological analysis
Internal organ samples were collected immediately after fish dissection, preserved in 10% buffered formalin, fixed, embedded in paraffin, and sectioned into 4 μm-thick slices. They were then stained with hematoxylin and eosin following the protocol of Humason (1979).
Mucus isolation
Mucus was collected from the isolated intestine using a rubber spatula to scrape the inside of the intestine. Afterward, the mucus was homogenized in phosphate-buffered saline. To remove particles and cellular material, all mucus preparations underwent two centrifugations at 27,000 rpm for 15 minutes at 4°C for further investigation (Chabrillón et al., 2005). Mucus was graded into four categories as follows:
1→Mild
2→Moderate
3→ High
4→Severe
Detection of digestive enzymes in mucus samples
The activities of protease, lipase, and α-amylase were measured according to the methods described by Folin and Ciocalteu (1927), Lott et al. (1986), and Garber and Wulff (1987), respectively.
IgD detection in mucus
IgD levels were determined according to the method of Mortensen et al. (1989).
Statistical analysis
Data were provided as mean ± SE. Version 20 of the statistical program Sigma Stat was used to analyze all data. Analysis of variance was used to assess significant differences between groups. A statistically significant value was defined as p < 0.05.
Ethical approval
The research animal care ethical committee of the Faculty of Science, Port Said University, approved all studies under protocol ERN: PSU. Sci. 75.
Results
The helminths recovered from the examined S. undosquamis were the Plerocercoid of Tetraphyllidean species, Ligula intestinalis, and Callitetrarhynchus sp. Two different third-stage nematode larvae were also observed: Contracaecum sp. and Anisakis sp. Finally, Aponurus sp. and Lecithochirium sp. represented the trematodes.
Plerocercoids
Tetraphyllidean larvae
At this stage, the overall body form was characterized by a conical shell appearance with a dark brown color. The anterior end, represented by the scolex, was small and rounded, whereas the posterior end tapered to a point. Axial movement was observed through successive alternating contractions and extensions (Fig. 2A–D). The contracted body measured 98.7–101 µm in length, whereas the expanded form ranged from 117.4 to 142.5 µm.
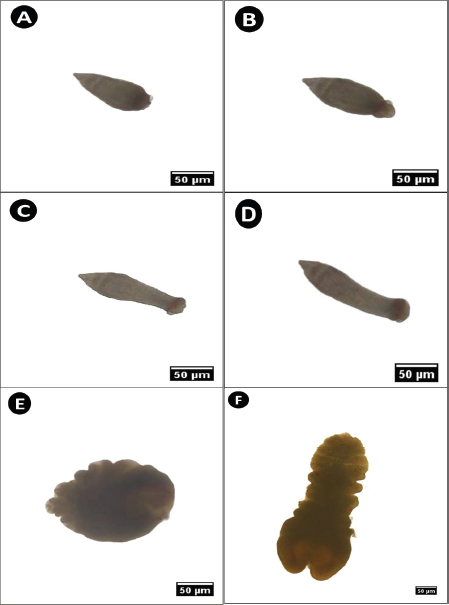
Fig. 2. Microphotograph of parasitic plerocercoids isolated from the digestive tract of S. undosquamis. (A–D) Various stretching forms of tetraphyllidean larvae during movement. (E) Contracted form of L. intestinalis larvae. (F) Expanded form of L. intestinalis. (×40).
Ligula intestinalis (Family: Diphyllobothriidae)
This stage exhibited a dark brown coloration and a leaf-like appearance. The anterior end was rounded with a central cleft, whereas the posterior end was slightly tapered. Similar to tetraphyllidean larvae, L. intestinalis moved comparably. The contracted body measured between 104 and 143 µm, whereas the stretched phase ranged from 259 to 296 µm (Fig. 2E and F).
Callitetrarhynchus sp. (Family: Lacistorhynchidae)
The plerocercoid was encapsulated within a small white cyst (Fig. 3A–C). These cysts of varying sizes were found rolled within the internal cavity of S. undosquamis (Fig. 3B). Under microscopic examination, the parasite inside the cysts consisted of two distinct regions (Fig. 3C). Each lateral side of the scolex displayed two short, crescent-shaped bothria (Fig. 3D). Four long black tentacles were enclosed within a tentacle sheath between the bothrial regions (Fig. 3D). The plerocercoid posterior end was characterized by four identical bulbs (Fig. 3E).
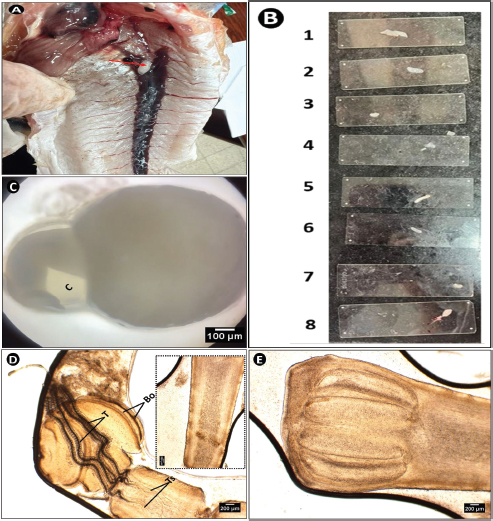
Fig. 3. Microphotograph of Callitetrarhynchus sp. plerocercoid collected from the peritoneum of S. undosquamis. (A) A white parasitic cyst inside the fish’s body (indicated by the red arrow). (B) Variations in the size of the isolated plerocercoids. (C) The shape of the parasite within its cyst under a microscope (×4). (D) Scolex of the parasite (×40), showing four bothria (Bo) and central tentacles (T). The inset (D window) highlights the tentacle sheath extensions (Ts). (E) The posterior end of the larvae (×40) with four identical bulbs.
Third-stage larvae
Anisakis sp. (Family: Anisakidae)
The newly hatched larvae were considerably smaller than the larger third-stage larvae (Fig. 4A), with a size ratio of 1:6. A small, dark pointed tooth was visible at the tip of the blunt anterior end, accompanied by a lateral excretory pore (Fig. 4B). The border along the intestine clearly defined the ventricle (Fig. 4C). Small, green circular rectal glands were scattered around the rectum, which opened to the exterior through a lateral anus (Fig. 4D).
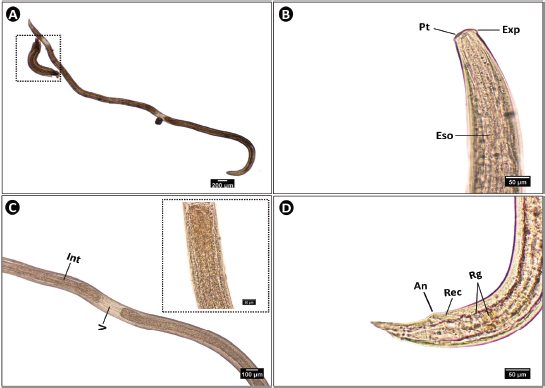
Fig. 4. Microphotograph of Anisakis sp. larvae collected from different organs of S. undosquamis. (A) Whole mount of the third-stage larva (×4), with the square indicating a newly hatched first-stage larva. (B) Lateral view of the anterior end (×40), showing a pointed tooth (PT), terminal lateral excretory pore (Exp), and esophagus (Eso). (C) Middle portion of the worm (×10), showing the tubular intestine (Int) and a lighter ventricle (V), enlarged in the inset. (D) Lateral view of the posterior end (×40), characterized by several rectal glands (Rg) and the rectum (Rec), which terminates laterally at the anus (An).
Contracaecum sp. (Family: Anisakidae)
The third-stage larvae of this worm were elongated and dark brown in color. The eggs were oval with a double-layered structure and were embryonated (Fig. 5A and B). The anterior third of the larva contained the upper digestive tract, including the mouth and esophagus (Fig. 5C). The intestine expanded and became more pronounced in the mid-body region (Fig. 5D) and then gradually narrowed near the ventricular appendix (Fig. 5E). The digestive tract terminated posteriorly at the rectum, which opened externally via an anus enclosed within a protective sheath (Fig. 5F).
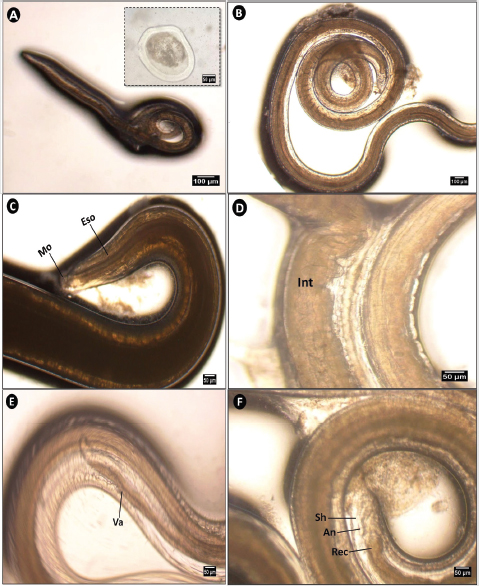
Fig. 5. Microphotograph of Contracaecum sp. larva collected from different organs of S. undosquamis. (A, B) Whole mount of the third-stage larva (A, ×4 & B, x10), with an inset showing an embryonated egg. (C) Lateral view of the anterior end (×40), showing the mouth (Mo) and esophagus (Eso). (D, E) Middle portion of the worm (×40), showing the intestine (Int) and ventricular appendix (Va). (F) Lateral view of the posterior end (×40), showing the external sheath (Sh) and rectum (Rec), which terminates laterally at the anus (An).
Adult trematodes
Aponurus sp. (Family: Lecithasteridae)
This specimen exhibited a brownish–orange coloration and semi-translucent appearance. The uterus, filled with eggs, occupied most of the middle region (Fig. 6A and B). The oral sucker could expand to a length ranging from 20 to 57 µm. The ventral sucker was notably large and concave (Fig. 6B and C). The posterior end was distinctly hemispherical (Fig. 6D).
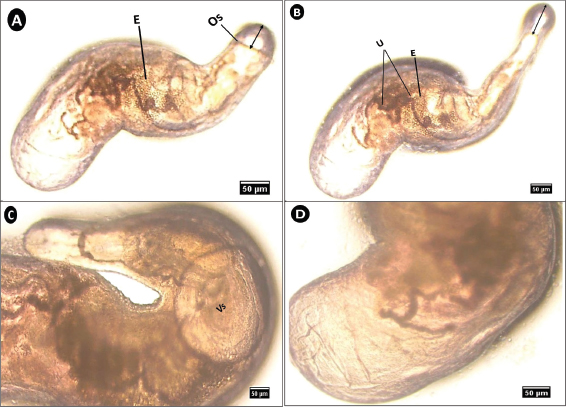
Fig. 6. Microphotograph of Aponurus sp. isolated from the digestive tract of S. undosquamis. (A & B) Lateral view of the parasite (×10) showing some internal details. (B) Enlarged view of the anterior end (×40) showing the size of the ventral sucker (Vs). (C) Enlarged view of the posterior end (×40) (Os; oral sucker, E; eggs, U; uterus).
Lecithochirium sp. (Family: Hemiuridae)
This worm was observed in both a contracted state, displaying a C-shaped appearance, and an extended form (Fig. 7A and B). Anteriorly, a small oral sucker was followed by the pharynx, two lateral intestinal ceca, and a sinus sac located just anterior to the ventral sucker. A distinct excretory vesicle (ecosoma) was noted at the posterior end (Fig. 7B). A lateral view (Fig. 7C) illustrates the details of the reproductive system. The seminal vesicle opened into a dark, circular testis, whereas the uterus was highly curved, wider, and more branched than that of Aponurus sp. Toward the posterior end, the ovary was positioned in front of the lobulated vitelline glands.
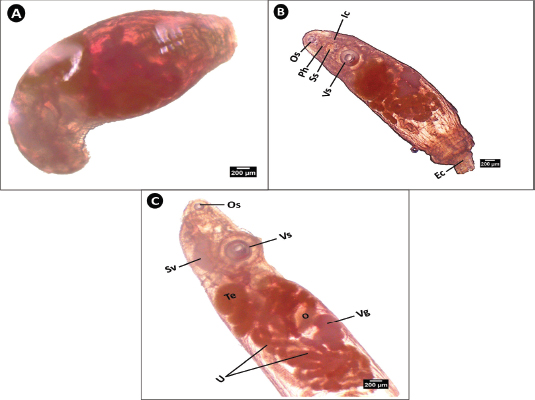
Fig. 7. Microphotograph of Lecithochirium sp. isolated from the digestive tract of S. undosquamis. (×40) (A) Contracted form of the worm. (B) Expanded ventral view. (C) Lateral view showing details of the reproductive organs. Os; oral sucker, Ph; pharynx, Ss; sinus sac, Vs; ventral sucker, Ic; intestinal caeca, Ec; ecosoma, Sv; seminal vesicle, Te: Testis, O; ovary Vg; vitelline glands, U; uterus.
Dominance of helminths
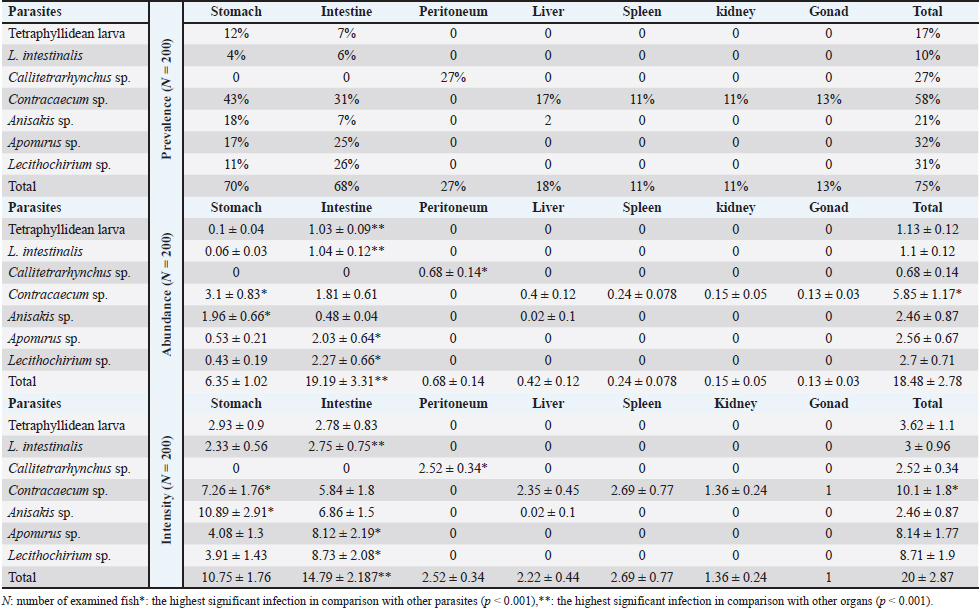
The total prevalence in all examined fish was 75%. The highest prevalence was recorded in the stomach (70%), followed by the intestine (68%). Among the recovered helminths, L. intestinalis had the lowest infection rate, recorded for the first time in this fish. Conversely, Contracaecum sp. exhibited the highest prevalence, abundance, and intensity at 58%, 5.85 ± 1.17, and 10.1 ± 1.8, respectively (Table 1).
Table 1. Parasites incidence (±SE) infecting different organs of S. undosquamis.
Effect of fish biological factors on helminth dominance
The prevalence was the highest (100%) in larger fish within class 2 (>113 g) than in smaller ones; 82.26% belong to class 1 (< 113 g). Contracaecum sp., Aponurus sp., and Lecithochirium sp. were the most prevalent worms in class 2, and the former only in class 1. The total abundance and intensity were significantly higher (p < 0.001) in larger S. undosquamis than in smaller fish (Table 2).
Table 2. Parasites incidence (±SE) infecting different organs of S. undosquamis according to host biological factors.
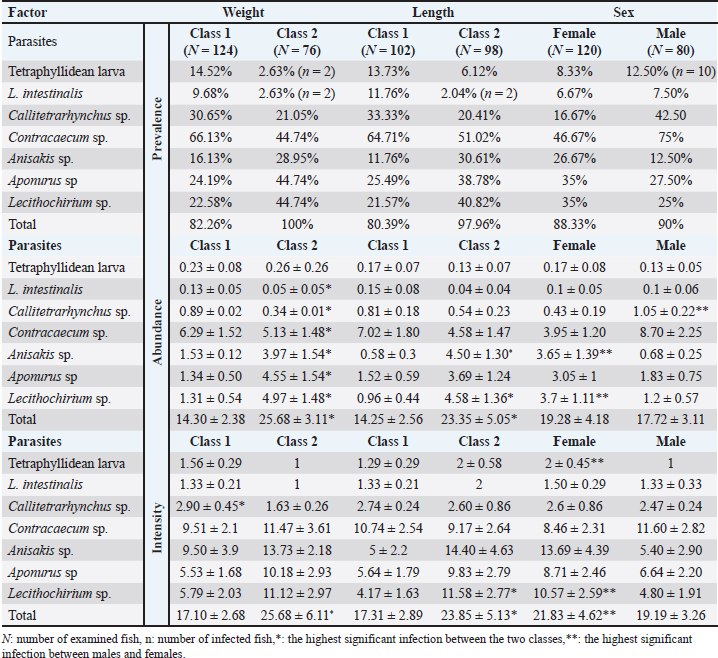
The incidence of S. undosquamis varied by fish length, with a higher prevalence (97.96%) in longer fish (class 2, >25 cm) than in shorter fish (80.39%). Contracaecum sp. had the highest prevalence in both classes (64.71% in class 1 and 51.02% in class 2), while L. intestinalis showed the lowest prevalence. The prevalence of Lecithochirium sp. was nearly twice as high in class 2 (40.82%) as in class 1 (21.57%). Abundance and intensity also increased significantly in longer class 2 fish (Table 2).
The infection rate was slightly higher in males (90%) than in females (88.33%). Contracaecum sp. had the highest prevalence, while L. intestinalis had the lowest in both sexes. Aponurus sp. and Lecithochirium sp. both had a 35% prevalence in females. Although there was no significant difference in abundance between sexes, the intensity was significantly higher in females (21.83 ± 4.62) than in males (19.19 ± 3.26) (Table 2).
Histopathological impacts of infection on various organs
Histological analysis of the stomach revealed helminths embedded within the submucosal layer. The surrounding tissue exhibited significant cellular infiltration by immune cells. The serosal layer and circular muscle fibers also showed structural deformation. In contrast, the longitudinal muscle fibers were unaffected (Fig. 8A). The worms adhered to the intestinal villi, causing damage to the villous region, which was accompanied by cellular infiltration (Fig. 8B). The infection severely affected the hepatocytes of infected livers, exhibiting karyolysis and significant vasodilation (Fig. 8C). Histological sections of the infected spleen revealed normal white pulp. In contrast, the red pulp, similar to the liver, exhibited vasodilation (Fig. 8D). Certain tubular regions in the kidney sections were markedly malformed, along with considerable hemorrhagic areas (Fig. 8E). While the testes presented with hemorrhage (Fig. 8F). The ovaries exhibited cellular infiltration and deformed oocytes (Fig. 8G).
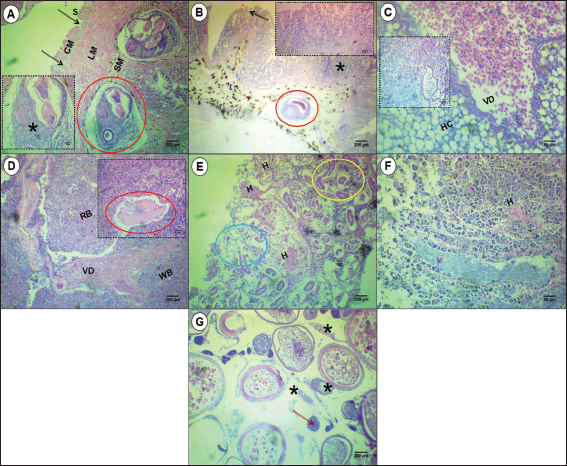
Fig. 8. Microphotographs of different organs of S. undosquamis infected with helminths. (A-B) Stomach and intestine sections, respectively (×10), Damage to the serosa (S) and circular muscle fibers (CM) is indicated by black arrows, whereas the longitudinal muscle fibers (LM) remain normal. Worms (red circles) were located in the submucosa (SM) of the stomach and within the intestinal villi, causing damage to the infected area (black arrow) and surrounded by cellular infiltration (*) as enlarged in the A and B insets (×40). (C) Liver section (×40) showing significant vasodilation (VD) and abnormal hepatocytes (Hc) compared to the control uninfected liver (C window). (D) Spleen section (×10) showing the two main compartments: red pulp (RP) and white pulp (WP). Vasodilation (VD) was the main lesion in the infected spleen. The D inset (×40) highlights a parasite (red circle) near the vasodilated area. (E) Kidney sections (×10) reveal hemorrhage (H), with some deformed tubular areas (blue circle) and others normal (yellow circle). (F) Testis sections (×40) with hemorrhage (H) in some areas. (G) Ovary sections (×10) exhibit malformations in the oocysts (red arrow) and cellular infiltration (*).
Variation in mucus flow relative to the rates of helminth infection
Overall, an increase in the infection rate corresponded with an increase in the quantity of mucus collected. The most significant increase (p < 0.002) was observed in S. undosquamis, where higher infection levels were associated with a greater mucus output compared to uninfected samples (Fig. 9).
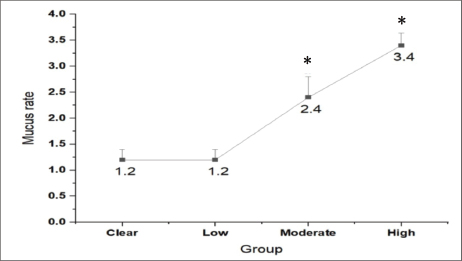
Fig. 9. Mucus rate in relation to the severity of infection.*: significant difference compared with clear fish.
Enzymatic activity and IgD levels in S. undosquamis mucus in relation to helminth infection intensity
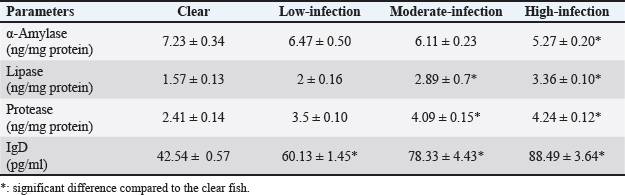
Lipase and protease levels were significantly higher in moderately and highly infected samples than in uninfected ones (p < 0.02 and p < 0.001, respectively). In contrast, α-amylase activity was highest in uninfected fish (7.23 ± 0.34) and lowest in infected fish (5.27 ± 0.20). Similar to protease and lipase, IgD levels were significantly elevated in heavily infected fish compared to uninfected ones (Table 3).
Table 3. Mean (±SE) activities of different digestive enzymes and IgD levels in S. undosquamis intestinal mucus.
Discussion
This study revealed the presence of various helminth infections in S. undosquamis sourced from Port Said markets, indicating that the parasites circulate continuously and survive beyond harvest. Additionally, the observed association between helminth infection, mucus production, and biochemical alterations highlights the adaptive physiological and immune responses of the host to parasitic stress.
Among the 200 examined fish, seven different helminths were recovered from the organs. These included the plerocercoid larvae of Tetraphyllidean species, L. intestinalis, and Callitetrarhynchus sp. Two distinct third-stage nematode larvae, namely, Contracaecum sp. and Anisakis sp. were identified. Finally, Aponurus sp. and Lecithochirium sp. represented the trematodes.
Tetraphyllidean plerocercoids were primarily distinguished by their conus shell shape. The body displayed two configurations: stretched and contracted, with varying lengths. These variations corroborate the findings of Elmajdoub et al. (2023a), who observed several forms of this stage with similar appearances but differing lengths.
The middle anterior cleft of L. intestinalis was a key feature used to identify this parasite, consistent with the description provided by Blazhekovikj-Dimovska et al. (2021). The observations of Morsy et al. (2022) for Callitetrarhynchus sp. align with the findings of this study. These similarities include the white coloration of the external cysts surrounding the worms, the circularity of the short bothria, and the number of bulbs.
The third-stage larvae of Contracaecum sp. are characterized by brown coloration and the shape of the ventricular appendix, aligning with the previously reported data by Vergara-Flórez and Consuegra (2021). The characteristics of Anisakis sp. closely matched those described by Carvalho et al. (2020). These morphometric features included the position of the pointed tooth, excretory pore, and ventricle, the presence of green rectal glands, and the overall shape of the worm.
The oral sucker extensions of Aponurus sp. exhibit a unique characteristic: their ability to expand, a feature observed for the first time in this study. The remaining morphological details were determined based on the description provided by Carreras-Aubets et al. (2011). The details and shapes of the reproductive organs, along with the presence of the posterior ecosoma, were key features used for the identification of Lecithochirium sp. These observations closely matched those of El-Garhy et al. (2017).
The overall infection rate of the identified helminths in S. undosquamis was recorded at 75%, which is approximately 1.67 times higher than the prevalence reported by Abd-Elghany (2017) in specimens collected from fish markets in the Zagazig Governorate, Egypt.
Tetraphyllidean plerocercoids were recorded only in the stomach and intestine, with prevalence rates of 12% and 7%, respectively. These values were lower than the 59.6% recorded by El-Ekiaby (2019). Despite L. intestinalis plerocercoids being recorded in different fish as documented in previous studies (Tizie et al., 2014; Arslan et al., 2015), the present study recorded their presence in S. undosquamis for the first time, confirming it as an acceptable host. Callitetrarhynchus sp. was observed exclusively encysted in the peritoneum of S. undosquamis, with no infestations noted in internal organs. The infection rate of Callitetrarhynchus sp. was 27%, which is lower than the infection rate of 46% reported by Abdel-Mawla et al. (2017), who documented the presence of Trypanorhyncha larvae, including Pseudogrillotia sp., Callitetrarhynchus gracilis, Callitetrarhynchus speciosus, and Floriceps sp.
Contracaecum sp. was the only parasite recovered from all internal organs and was the dominant species. Its mean abundance and intensity were recorded at 5.85 ± 1.17 and 10.1 ± 1.8, respectively. These values were higher than the mean abundance (0.076) and intensity (0.15) of the same fish species reported by El-Asely et al. (2015).
Anisakis sp. was recovered from the stomach, intestine, and liver, with a total prevalence of 21% and a mean intensity of 2.46 ± 0.87. Regional differences were observed in S. undosquamis collected from the Indian Ocean, specifically between East Java and West Java. In West Java, the prevalence (1.45%) and mean intensity (3.33) were lower than those reported in the current study, while East Java recorded significantly higher values (prevalence: 75.44%, mean intensity: 10.38), as documented by Syarifah et al. (2023). In Egypt, the total prevalence of Anisakis sp. in S. undosquamis collected from the Sharkia Governorate (15.25%) was much closer to the values reported in this study (El-Atabany et al., 2019).
Lecithochirium sp. is the most common genus within the Hemiuridae family of parasitic digenean that infects the digestive tracts of marine fishes (Shih et al., 2004). In the current study, Lecithochirium sp. was found in the stomach and intestine, with infection rates of 11% and 26%, respectively. In contrast, this parasite was reported to be specific to the stomach in samples collected from fish markets in Zagazig Governorate, Egypt, with an incidence of 16% (Abd-Elghany, 2017). In samples from fish markets in Misurata, Libya, the infection rate increased significantly, reaching 94.2%, as documented by Elmajdoub et al. (2023b).
Similar to Lecithochirium sp., Aponurus sp. was found in the intestine at a higher rate of 25%, compared with 17% in the stomach, resulting in an overall infection rate of 32%. Few studies have confirmed the presence of Aponurus sp. in S. undosquamis, with one such study by Elmajdoub et al. (2023b), in which Aponurus sp. recorded a prevalence rate of 4.7%. The variations in parasitic dominance observed in the current study compared to previous reports may be attributed to differences in sampling locations. Since environmental factors can limit the spread of infections, the infection rates and transmission of these agents are expected to vary among habitats (Patz et al., 2000; Wells et al., 2015).
Limited data focuses on the relationship between length/weight and parasite dominance in S. undosquamis. Cipriani et al. (2022) found a significantly positive correlation between the abundance of A. typica and the total length and weight of S. undosquamis. In the current study, the biological factors of the host, represented by its length, weight, and sex, affect the dominance of parasitic infection. The incidence increased in taller and larger fish compared with shorter and smaller fish. This may be because larger fish consume more food, which comprises Mollusca, Arthropoda, and smaller fish (Rashed et al., 2024). These food types act as pathogen carriers for the fish; therefore, increased food consumption leads to a higher infection rate. Barber (2005) clarified that the closest predictor of parasite mass was the body size-corrected host growth rate, indicating that the fastest-growing fish developed the largest parasites.
The effect of host sex has a lower impact on the incidence of parasites than either length or weight. In general, males had a higher prevalence rate than females. Adjei et al. (1986) found that in S. undosquamis, the estimated mortality rate due to Callitetrarhynchus gracilis was 3% for females and 5% for males.
Histopathological analysis of S. undosquamis reveals pronounced organ-specific pathologies associated with helminth infections, attributable to both direct parasitic damage and host-mediated immune responses. Within the digestive tract, helminth embedding and villous atrophy were observed, indicating compromised tissue integrity and impaired nutrient absorption, consistent with the findings of Lafferty and Kuris (2002). Hepatic tissue exhibited hepatocellular karyolysis—a necrotic process likely induced by helminth-derived toxins and inflammatory stress—similar to pathological changes reported in infected teleosts (Kundu et al., 2016). Furthermore, vasodilation in both the spleen and liver, as well as tubular deformities in renal tissue, suggest systemic circulatory disturbances and functional impairment associated with helminthiasis, in line with observations in other marine fish hosts (Williams and Jones, 1994). Reproductive tissues, including malformed oocytes and hemorrhagic testes, also showed notable damage, posing a significant threat to population fecundity. These findings corroborate previous reports of helminth-induced reproductive dysfunction in aquatic species (Marcogliese, 2004; Mir et al., 2012).
This study presents the first evidence linking helminth invasion in the gastrointestinal tract of S. undosquamis to changes in intestinal mucus characteristics. Infected individuals exhibited significantly increased mucus production (p < 0.002) compared with their uninfected counterparts. Beyond its roles in lubrication, digestion, absorption, and microbial colonization, mucin is critical for managing infectious pathogens (Domeneghini et al., 2005), explaining the observed correlation between infection severity and heightened mucus production.
Infected samples showed significantly higher lipase and protease activities than uninfected S. undosquamis samples. In contrast, the non-infected fish exhibited higher α-amylase activity than the infected ones. Sitjà-Bobadilla et al. (2006) reported that the elevated protease activities in the infected samples may be due to either the interference of host and parasite proteases or the release of proteases from the host’s damaged intestinal cells. These results contrast with those of Izvekova et al. (2011), who found that bream fish infected only with Caryophyllaeus laticeps (Cestoda, Caryophyllidea) exhibited a decrease in digestive enzyme activity. The discrepancy between the current study and previous findings may be due to variations in the parasitic helminth species, their dominance, and differences in the fish host. Highly infected fish had significantly higher IgD values than uninfected fish. Compared to uninfected fish, highly infected fish had significantly higher IgD values. Bacterial, viral, and parasitic infections have been shown to transcriptionally promote IgD expression (Basu et al., 2016; Zhang et al., 2018; Xu et al., 2019).
Conclusion
This study identified a diverse range of helminths infecting the organs of 200 S. undosquamis, including the plerocercoid of Tetraphyllidean species, L. intestinalis, Callitetrarhynchus sp., third-stage larvae of Contracaecum sp. and Anisakis sp., and the trematodes Aponurus sp. and Lecithochirium sp. The findings demonstrate a high prevalence of helminth infections, particularly within the stomach and intestines, with larger fish exhibiting significantly higher infection levels. Contracaecum sp. was identified as the most prevalent helminth, while L. intestinalis showed the lowest infection rate. Histological analysis revealed significant tissue damage across multiple organs, immune responses, increased mucus secretion, and changes in enzyme activity, all of which indicate substantial physiological stress in the infected fish. These results underscore the importance of further research into seasonal, environmental, and zoonotic factors affecting helminth infections to inform effective marine conservation and public health strategies.
Acknowledgment
The authors express their gratitude to Port Said University.
Conflicts of interest
The authors declare no conflicts of interest.
Funding
The authors self-funded this study without external support.
Author’s contributions
All authors have made equal contributions to this work. They reviewed, revised, and approved the final version of the manuscript.
Data availability
All data supporting this study’s findings the findings of this study are available in the manuscript.
References
Abd-Elghany, A. 2017. Parasitic fauna of the marine greater lizard fish Saurida undosquamis (Richardson, 1848). Egypt. Vet. Med. Soc. Parasitol. J. 13, 108–121.
Abdel-Mawla, H.I., Salem, S.M. and El-ghayaty, H.A. 2017. Metacestodiasis in some marine fishes with biochemical changes and public health significance. Anim. Health. Res. J. 5, 147–159.
Adjei, E.L., Barnes, A. and Lester, R.J.G. 1986. A method for estimating possible parasite-related host mortality, illustrated using data from Callitetrarhynchus gracilis (Cestoda: trypanorhyncha) in lizardfish (Saurida spp.). Parasitol 92, 227–243.
Arslan, M.O., Yılmaz, M. and Taşçı, G.T. 2015. Infections of Ligula intestinalis on freshwater fish in Kars plateau of north-eastern Anatolia, Turkey. Turkiye. Parazitol. Derg. 39, 218–221.
Badawi, L.A., Ibrahim, G.D., Ali, A.M. and Omar, A.A. 2021. Effect of using moringa (Moringa oleifera lam.) leaves as feed additives on growth performance, feed utilization and immune response of red tilapia (Oreochromis sp.) fingerlings. Sinai J. Appl. Sci. 10, 365–378.
Barber, I. 2005. Parasites grow larger in faster growing fish hosts. Int. J. Parasitol. 35, 137–143.
Basu, M., Lenka, S.S., Paichha, M., Swain, B., Patel, B., Banerjee, R., Jayasankar, P., Das, S. and Samanta, M. 2016. Immunoglobulin (Ig) D in Labeo rohita is widely expressed and differentially modulated in viral, bacterial and parasitic antigenic challenges. Vet. Immunol. Immunopathol. 179, 77–84.
Blazhekovikj-Dimovska, D., Stojanovski, S. and Smiljkov, S. 2021. Distribution of cestodes in farmed common carp (Cyprinus carpio l., 1758) from cyprinid aquaculture in macedonia. Curr. Trends Nat. Sci. 10, 427–439.
Bush, A.O., Lafferty, K.D., Lotz, J.M. and Shostak, A.W. 1997. Parasitology meets ecology on its own terms: margolis et al. revisited. J. Parasitology. 83, 575–583.
Carreras-Aubets, M., Repullés-Albelda, A., Kostadinova, A. and Carrassón, M. 2011. A new cryptic species of Aponurus Looss, 1907 (Digenea: lecithasteridae) from Mediterranean goatfish (Teleostei: Mullidae). Syst. Parasitol. 79, 145–159.
Carvalho, E.L.D., Santana, R.L.S., Gonçalves, E.C., Pinheiro, R.H.D.S. and Giese, E.G. 2020. First report of Anisakis sp. (Nematoda: anisakidae) parasitizing Muscovy duck in Marajó Island, state of Pará, Brazil. Rev. Bras. Parasitol. Vet. 29, e020319.
Chabrillón, M., Rico, R.M., Balebona, M.C. and Moriñigo, M.A. 2005. Adhesion to sole, Solea senegalensis Kaup, mucus of microorganisms isolated from farmed fish, and their interaction with Photobacterium damselae subsp. piscicida. J. Fish Dis. 28, 229–237.
Cipriani, P., Giulietti, L., Shayo, S.D., Storesund, J., Bao, M., Palomba, M., Mattiucci, S. and Levsen, A. 2022. Anisakid nematodes in Trichiurus lepturus and Saurida undosquamis (Teleostea) from the South-West Indian Ocean: genetic evidence for the existence of sister species within Anisakis typica (sl), and food-safety considerations. Foodborne Parasitology 28, e00177.
Domeneghini, C., Arrighi, S., Radaelli, G., Bosi, G. and Veggetti, A. 2005. Histochemical analysis of glycoconjugate secretion in the alimentary canal of Anguilla anguilla L. Acta. Histochem. 106, 477–487.
El-Asely, A.M., El Madawy, R.S., El Tanany, M.A. and Afify, G.S. 2015. Prevalence and molecular characterization of anisakidosis in both European (Merluccius merluccius) and lizard head (Saurida undosquamis) hakes. GSTF J. Vet. Sci. 1, 1–10.
El-Atabany, A.I., Mahmoud, A.F.A. and Abd, A.M. Prevalence and public health importance of anisakid nematode in lizardfish collected from Sharkia Governorate, Egypt. In the Proceedings of the 2019 Fifth International Food Safety Conference, Damanhour, 2019, pp 576–582.
El-Ekiaby, W.T. 2019. Morphological studies on prevailing parasitic infections in the greater lizardfish Saurida undosquamis from the Red Sea. Egypt. J. Aquat. Res. 9, 47–69.
El-Fahla, N.A., Saad El-din, M.I., El Mageed, Y.S.M.A., Gad El-hak, H.N., El-Shenawy, N.S. and Rashed, H.A.E.H. 2024. Seasonal fluctuations of heavy metal accumulation and gastrointestinal helminth induce oxidative stress and histological lesions in resident catfish Clarias gariepinus. Environ. Pollut. 362, e124989.
EL-Garhy, M., Mostafa, N., Al Ghamdi, A., Hamdi, S.H. and Morsy, K. 2017. Description of two digenean parasites lecithochirium priacanthi yamaguti, 1953 (hemiuridae) and pseudopecoeloides artherinomori aken’ova et al, 2009 (opecoelidae) infecting anguillidae and serranidae marine hosts in Egypt. J. Egypt. Soc. Parasitol. 47, 341–348.
Elmajdoub, L., Emshiheet, K., Abushiba, F., Elzwawy, S. and Abusahal, M. 2023. Prevalence and identification of cestode species recovered from infected organs of lizardfish (Saurida undosquamis). Int. J. Sci. Res. 2, 43–55.
Elmajdoub, L.O., Emshihee, K.A., Abushiba, F.M., Elzwawy, S.E., Abusahal, M.M. and Aagme, R.S. 2023. Prevalence and morphological identification of digenean trematode species recovered from infected organs of lizardfish (Saurida undosquamis). Eur. J. Zool. 2(1), 1–6.
Folin, O. and Ciocalteu, V. 1927. On tyrosine and tryptophane determinations in proteins. J. Biol. Chem. 73, 627–650.
Gabagambi, N.P., Salvanes, A.G.V., Midtøy, F. and Skorping, A. 2019. The tapeworm Ligula intestinalis alters the behavior of the fish intermediate host Engraulicypris sardella, but only after it has become infective to the final host. Behav. Processes. 158, 47–52.
Garber, M. and Wulff, K. 1987. A new pancreas specific α-Amylase assay using the synergistic action of two different monoclonal antibodies. Clinic. Chemist. 33, 997–1002.
Ghobashy, M.A., Soliman, M.F.M. and Hassan, E.A. 2006. Heterophyid metacercariae in Liza auratus (Grey mullet) from lake Manzala, Egypt: occurrence and impact. J. Egyp. German. Soc. Zool. 50, 109.
Gómez, G.D. and Balcázar, J.L. 2008. A review on the interactions between gut microbiota and innate immunity of fish. FEMS. Immunol. Med. Microbiol. 52, 145–154.
Guerrero, R.D. and Shelton, W.L. 1974. An aceto-carmine squash method for sexing juvenile fishes. Prog. Fish. Cult. 36, 56.
Humason, G.L. 1979. Animal tissue techniques. W.H. & Co., San Francisco. P. 641.
Islam, S.I., Rodkhum, C. and Taweethavonsawat, P. 2024. An overview of parasitic co-infections in tilapia culture. Aquac. Int. 32, e899–e927.
Izvekova, G.I., Solovyov, M.M. and Izvekov, E.I. 2011. Effect of Caryophyllaeus laticeps (Cestoda, Caryophyllidea) upon activity of digestive enzymes in bream. Biol. Bull. Russ. Acad. Sci. 38, 50–56.
Jerônimo, G.T., Da Cruz, M.G., Ede, B.-A.., Furtado, W.E. and Martins, M.L. 2022. Fish parasites can reflect environmental quality in fish farms. Rev. Aquac. 14, 1558–1571.
Kundu, I., Bandyopadhyay, P.K., Mandal, D.R. and Gürelli, G. 2016. Study of pathophysiological effects of the nematode parasite Eustrongylides sp. on freshwater fish Channa punctatus by hematology, serum biochemical, and histological studies. Turkiye. Parazitol. Derg. 40, 42–47.
Lafferty, K.D. and Kuris, A.M. 2002. Trophic strategies, animal diversity and body size. Trends Ecol. &. Evol. 17, 507–513.
Leung, T.L.F. and Bates, A.E. 2013. More rapid and severe disease outbreaks for aquaculture at the tropics: implications for food security. J. Appl. Ecol. 50, 215–222.
Lott, J.A., Patel, S.T., Sawhney, A.K., Kazmierczak, S.C. and Love, J.E. 1986. Assays of serum lipase: analytical and clinical considerations. Clin. Chem. 32, 1290–1302.
Magnadóttir, B. 2006. Innate immunity of fish (overview). Fish. Shellfish. Immunol. 20, 137–151.
Marcogliese, D.J. 2004. Parasites: small players with crucial roles in the ecological theater. Eco. Health 1, 151–164.
Mehanna, S.F. 2022. Population dynamics and stock assessment of two Lizardfish species in the southern Red Sea coasts, Foul Bay, Egypt. Egypt. J. Aquat. Biol. Fish. 26, 299–311.
Mir, T.A., Kaur, P. and Manohar, S. 2012. Pathogenic effects of nematode parasite Eustrongylides sp. larvae on serum LH level and histology of gonads of freshwater fish, Clarias gariepinus. Recent Res. Sci. Technol. 4, 24–26.
Morsy, K., Dajem, S.B., Al-Kahtani, M., El-Kott, A., Ibrahim, E., Hamdi, H., Al-Doaiss, A., Abumandour, M., El-Mekkawy, H., Massoud, D., Adel, A. and El-Kareem, S.A. 2022. Larval cestodes infecting commercial fish of Alexandria coast along the Mediterranean Sea: morphology and phylogeny. Rev. Bras. Parasitol. Vet. 31, e003022.
Mortensen, J., Nielsen, S.L., Sørensen, I. and Andersen, H.K. 1989. Specific serum immunoglobulin D, detected by antibody capture enzyme-linked immunosorbent assay (ELISA), in cytomegalovirus infection. Clin. Exp. Immunol. 7, 234–238.
Patz, J.A., Graczyk, T.K., Geller, N. and Vittor, A.Y. 2000. Effects of environmental change on emerging parasitic diseases. Int. J. Parasitol. 30, 1395–1405.
Rashed Et Al., H.A.E.H. 2024. Some of the biological aspects of the brushtooth lizardfish Saurida undosquamis (Richardson, 1848) collected from the commercial markets of Port Said Governorate, Egypt. Egypt. J. Aquat. Biol. Fish. 28, 383–401.
Rashed, H.A.E.H., El-Shenawy, N.S. and El-Fahla, N.A. 2025. Influences of parasitic stress on the health condition of African Catfish (Clarias gariepinus): biochemical and histopathological alterations. Mol. Biochem. Parasitol. 262, 111677.
Renner, E.D. and Duggan, I.C. 2024. Season, size, and sex: factors influencing monogenean prevalence and intensity on Gambusia affinis in New Zealand. Parasitol. Res. 123(1), 1–10.
Shih, H.H., Liu, W. and Zhao, Z. 2004. Digenean fauna in marine fishes from Taiwanese water with the description of a new species. Lecithochirium tetraorchis sp. nov. Zool. Stud. 43, 671–676.
Sitjà-Bobadilla, A., Redondo, M.J., Bermúdez, R., Palenzuela, O., Ferreiro, I., Riaza, A., Quiroga, I., Nieto, J.M. and Alvarez-Pellitero, P. 2006. Innate and adaptive immune responses of turbot, Scophthalmus maximus (L.), following experimental infection with Enteromyxum scophthalmi (Myxosporea: myxozoa). Fish Shellfish Immunol. 21, 485–500.
Syarifah, R.F., Murwantoko, M. and Setyobudi, E. 2023. Prevalence and intensity of larvae of the genus Anisakis sensu lato (Nematoda, Anisakidae) in Bigeye Scad, Selar crumenophthalmus (Bloch 1793), from the Indian Ocean off Java, Indonesia. Asian. Fish. Sci. 36, 192–202.
Tavares-Dias, M. and Martins, M.L. 2017. An overall estimation of losses caused by diseases in the Brazilian fish farms. J. Parasit. Dis. 41, 913–918.
Tizie., Baye, D. and Mohamed, A. 2014. Prevalence of Ligula intestinalis larvae in Barbus fish genera at Lake Tana, Ethiopia. World J. Fish. Mar. Sci. 6, 408–416.
Vergara-Flórez, V. and Consuegra, A. 2021. Contracaecum sp. (Nematode: anisakidae) in fish of commercial interest in the Gulf of Morrosquillo, Sucre - Colombia. Gestión YAmbiente. 24, e97356.
Watts, M., Munday, B. and Burke, C. 2001. Immune responses of teleost fish. Aust. Vet. J. 79, 570–574.
Wells, K., O’Hara, R.B., Morand, S., Lessard, J., -P.. and Ribas, A. 2015. The importance of parasite geography and spillover effects for global patterns of host–parasite associations in two invasive species. Divers. Distrib. 21, 477–486.
Williams, A.J. 1994. Parasitic Worms of Fish (1st ed.). London: CRC Press.
Woodland, J. 2006. National wild fish health survey–laboratory procedures manual. US Fish and Wildlife Service, Washington, DC, USA.
Xu, J., Yu, Y., Huang, Z., Dong, S., Luo, Y., Yu, W., Yin, Y., Li, H., Liu, Y., Zhou, X. and Xu, Z. 2019. Immunoglobulin (Ig) heavy chain gene locus and immune responses upon parasitic, bacterial and fungal infection in loach, Misgurnus anguillicaudatus. Fish &. Shellfish Immunol. 86, 1139–1150.
Zhang, X., Ding, L., Yu, Y., Kong, W., Yin, Y., Huang, Z., Zhang, X. and Xu, Z. 2018. The change of teleost skin commensal microbiota is associated with skin mucosal transcriptomic responses during parasitic infection by Ichthyophthirius multifillis. Front. Immunol. 9, 2972.























