| Research Article | ||
Open Vet. J.. 2025; 15(12): 6379-6389 Open Veterinary Journal, (2025), Vol. 15(12): 6379-6389 Research Article Inflammatory effects of titanium dioxide nanoparticles against polymethyl methacrylate on the subcutaneous tissue of rabbitsAli Shakir Hasan1, Jiyar Mohammed Naji1 and Mohammad Ismail Alberfkani2,3*1Dental Technology Department, Technical College of Duhok, Duhok Polytechnic University, Duhok, Iraq 2Department of Medical Laboratory Technology, College of Health and Medical Techniques, Duhok Polytechnic University, Duhok, Iraq 3Department of Medical Laboratory Technology, Zakho Technical Institute, Duhok Polytechnic University, Duhok, Iraq *Corresponding Author: Mohammad Ismail Alberfkani. Department of Medical Laboratory Technology, Zakho Technical Institute, Duhok Polytechnic University, Duhok, Iraq. Email: microbio85 [at] gmail.com Submitted: 22/07/2025 Revised: 20/10/2025 Accepted: 01/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
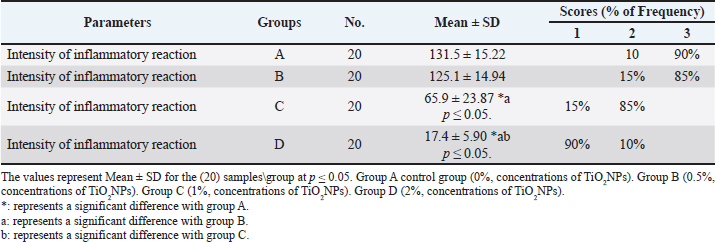
AbstractBackground: Polymethyl methacrylate (PMMA) is widely recognized as a biomaterial in orthopedic and dental applications, particularly in bone cement and prosthetic implants. However, inflammatory responses at the implantation site can affect its long-term performance. Aim: This study aims to investigate the inflammatory outcomes resulting from the incorporation of titanium dioxide nanoparticles (TiO2NPs) into PMMA and to assess the cellular interactions of these composites. Moreover, it explores the potential of TiO2-PMMA composites to enhance implant longevity while reducing adverse tissue reactivity associated with the introduction of TiO2NPs into the body is explored. Methods: A randomized controlled animal study was conducted to evaluate the histopathological tissue response to PMMAs acrylic resin modified with TiO2NPs. Twenty healthy male New Zealand rabbits were used; 80 PMMA implants (3 cm × 3 mm) were prepared and randomly allocated into four groups (n=20 each): Group A (control 0% TiO2NPs), Group B (0.5% TiO2NPs), Group C (1%TiO2NPs), and Group D (2% TiO2NPs). Implants were surgically inserted into the dorsal subcutaneous tissue of rabbits. After 14 days, the implantation sites were excised and histopathological examination was performed to assess inflammatory responses. Results: The study demonstrated lower inflammation and better tissue integration with the TiO2-PMMA composite groups than with pure PMMA. Adverse reactions were noted among the TiO2NPs with 0.5% wt. (severe reaction), whereas the large amounts of TiO2NPs 1% wt. and 2% wt. composites showed greater biocompatibility. Conclusion: TiO2NPs enhance the biological performance of PMMA by reducing inflammatory responses and improving tissue integration. These findings highlight the potential of TiO2-PMMA composites to extend the longevity and success of biomedical implants. Keywords: Polymethyl methacrylates, Titanium dioxide nanoparticles, Inflammation, Histopathology. IntroductionPolymethyl methacrylate (PMMA) is a commonly used biomaterial in orthopedic and dental applications, especially as bone cement and in prosthetic implants, due to its beneficial mechanical properties, easy handling, and biocompatibility (Fatemeh et al., 2023). PMMA can cause an inflammatory process to develop in adjacent tissues, creating significant complications over time (Ramanathan et al., 2024). This must be carefully considered to optimize the longevity of biomedical implants. When implanted, PMMA alone induces the formation of a fibrous capsule, a normal foreign body reaction. The inflammatory profile and tissue integration can be adjusted by the addition of nanoparticles, including TiO2, that can change this response (Asim et al., 2022). While PMMA is generally considered biocompatible due to its non-toxic and non-carcinogenic properties, the addition of titanium dioxide nanoparticles (TiO2 NPs) may alter its biocompatibility (Amirabad et al., 2022). Recent progress in nanotechnology has introduced TiO2 NPs as a new and potentially useful additive to PMMA composites. TiO2 is biocompatible and exhibits some antibacterial effects and the potential to alter inflammatory responses (Ahmed et al., 2020). Nanomaterials may also impact biomedical applications and research following their implementation in both in vivo and in vitro environments (Youssef et al., 2024). Some studies have shown that if engineered properly, TiO2-PMMA composites produce a lesser inflammatory response than pure PMMA and improve tissue integration and implant properties (Świeczko-Żurek et al., 2022). Nhlapo et al. (2020) synthesized the importance of ensuring concentrations of TiO2 NP are optimized to enhance biocompatibility; however, in vivo studies support their claims of biocompatibility. It should be emphasized that the main aim is to achieve a quasi-biological material with TiO2 embedded PMMA when evaluated by animal models that demonstrated very good tissue integration, with mild and minimal inflammatory reactions. Therefore, it is rational to deduce that TiO2 NPs aid in enhancing the biological performance of PMMA with the likelihood of eliciting adverse reactions to the materials (Aligamaleldin and Elsherbini, 2022). The addition of TiO2 nanoparticles into PMMA increases the hardness of the material and decreases its porosity, which can diminish bacterial adhesion and growth to reduce inflammation due to bacterial infection (Asim et al., 2022). Rabbit skin is composed of distinct layers, including the epidermis and dermis, which contain keratin, collagen fibers, elastic fibers, and hair follicles. These structural elements are particularly relevant for assessing subcutaneous implantation models in rabbits, as variations in collagen and elastic fibers directly reflect tissue remodeling and fibrosis. Therefore, describing these features helps in understanding the fibrotic and inflammatory responses observed following PMMA and TiO2-PMMA implantation (Zhu et al., 2015). In this context, inflammation in rabbit subcutaneous tissue represents an early response to implanted biomaterials and involves sequential infiltration of immune cells that later contribute to fibrosis. Inflammation is the immune system’s response to harmful stimuli, such as pathogens, damaged cells, toxic compounds, or irradiation (Medzhitov, 2010). Inflammation acts by removing injurious stimuli and initiating the healing process (Ferrero-Miliani et al., 2007). Inflammation is therefore a vital defense mechanism for health (Thongrin et al., 2023). During acute inflammatory responses, cellular and molecular events and interactions efficiently minimize impending injury or infection. This mitigation process contributes to tissue homeostasis restoration and acute inflammation resolution. However, uncontrolled acute inflammation may become chronic, contributing to various chronic inflammatory diseases (Zhou et al., 2016). At the tissue level, inflammation is characterized by redness, swelling, heat, pain, and loss of tissue function, which result from local immune, vascular, and inflammatory cell responses to infection or injury (Takeuchi and Akira, 2010). Inflammation involving CD3 cells indicates T cells (Klinge et al., 2022). The presence and ratio of these cells can indicate the inflammation type and severity (Matsuo and Ichimura, 2012). This study investigated the effects of TiO2 incorporation into PMMA on inflammatory responses, focusing on the mechanisms through which TiO2 influences cellular interactions. Furthermore, the study assessed the potential advantages of TiO2-PMMA composites in biomedical applications, particularly in improving biocompatibility and enhancing the longevity of implants (Jafari et al., 2020). Materials and MethodsExperimental animalsRabbits were used as the experimental model to evaluate the histopathological response of subcutaneous tissue to PMMA acrylic resin, both alone and in combination with TiO2NPs. Rabbits were chosen because of their well-documented suitability in biomedical research, their manageable size, ease of handling, and physiological responses that can reliably model human tissue reactions. The study was conducted as a controlled randomized animal experiment including 20 healthy male New Zealand rabbits, aged 1–2 years and weighing 1–2 kg. Animals were randomly divided into groups and implanted with acrylic resin specimens containing different concentrations of TiO2NPs (0%, 0.5%, 1%, and 2%). This design allowed for a systematic and controlled assessment of biocompatibility and tissue response to the tested materials. Only healthy animals free from systemic or local health issues were included in the study. The Ethics Committee of the College of Veterinary Medicine, University of Duhok, reviewed and approved all experimental procedures. Experimental designThe laboratory-based experimental study was conducted using 80 heat-cured acrylic resin specimens, each measuring 3 cm in length and 3 mm in diameter, allocated into four groups (n=20 per group). Group A (control): acrylic resin without TiO2 NPs. Group B: acrylic resin with 0.5% TiO2NPs. Group C: acrylic resin with 1% TiO2NPs. Group D: acrylic resin with 2% TiO2NPs. Specimen preparationTiO2NPs were obtained from Sigma-Aldrich Chemie GmbH, Eschenstrasse 5D-82024, Taufkirchen, Germany. According to the manufacturer’s datasheet, titanium dioxide nanopowder, 21 nm primary particle size Transmission Electron Microscopy ( TEM) ≥ 99.5% trace metal basis. Molecular weight, 79.87 g. Silanized TiO2NPs powder was mixed with MMA monomer at weight fractions of 0.5%, 1%, and 2% concentration of TiO2NPs, and the mixture was then manually premixed. The suspension, composed of methyl methacrylate monomer and TiO2NPs, was prepared using ultrasonic dual mixing. An ultrasonic pulsed vibration homogenizer (VCX 500, Sonics, USA) was set at 70% amplitude with a pulsed cycle of 2 seconds on and 3 seconds off, serving as a magnetic stirrer (MR Hei-Tech, Heidolph, Germany) with a magnetic fish operating at 1,500 rpm for 10 minutes at 22°C. Through this complementary method, a homogenous suspension of TiO2 nanoparticles was created within the epoxy matrix, ensuring even dispersion and effectively preventing the aggregation of TiO2NPs. The final product was classified as the modified monomer. In accordance with the manufacturer’s instructions, the modified monomer relevant to each group was combined with the PMMA powder. The liquid and powder were combined and covered to make the dough. Specimens measuring 3 cm in length and 3mm in diameter were created by packing this dough into a prepared brass mold containing cavities. The specimens were then cured in hot water following the manufacturer’s instructions. After smoothing and polishing the specimen surfaces. Before implantation, all specimens were sterilized by immersion in 70% ethanol for 30 minutes, air-drying under sterile conditions, and then exposure to ultraviolet light for 30 min on each side. Implantation and surgical procedureDorsal subcutaneous implantation was performed on 20 rabbits. Each animal received four implant specimens, representing one from each group: control group PMMA, and three experimental groups containing TiO2NPs-modified resin at concentrations of 0.5%, 1%, and 2%. Before surgery, the animals underwent a 7-day acclimatization period with ad libitum access to food and water. General anesthesia was induced by intramuscular injection of ketamine (35 mg/kg) and xylazine (5 mg/kg) (Hassan et al., 2024). The dorsal surgical region was shaved and disinfected with an antiseptic solution, and a subcutaneous incision was made to create implant pockets. Implants were positioned to avoid overlapping tissue responses between groups. Postoperatively, the animals were monitored for 14 days. On day 15, the implants and surrounding tissue were excised, fixed in 10% neutral buffered formalin for 24 hours, dehydrated in graded alcohols, and embedded in paraffin (Sánchez-Hernández et al., 2023). Histological sections of 4 μm thickness were prepared and stained with hematoxylin and eosin for general evaluation and Masson’s trichrome staining for fibrosis assessment. Immunohistochemistry was performed using the CD3 T-cell marker. Inflammatory response intensity was scored as follows: 0=absent (<5 cells), 1=mild (5–25 cells), 2=moderate (25–125 cells), and 3=severe (>125 cells) (Al-Jubori et al., 2017). To ensure consistency and reproducibility, the scoring and histological evaluations were performed using digital pathology and slide viewer software. CD3 membranous expression was classified using the combined intensity/percentage scoring system described by Blondy et al. (2021): + (Mild, <10% weak staining), ++ (Moderate, 10–50% moderate staining), and +++ (strong, >50% strong staining). To minimize bias, all histological and immunohistochemical evaluations were performed by an experienced histologist blinded to group allocation. Statistical analysisData obtained from histopathological evaluations were analyzed using the Statistical Package for the Social Sciences software (version 12.5). All results are expressed as mean ± SD (mean ± SD). For numerical data, one-way analysis of variance was used to assess significant differences among the four groups, followed by Duncan’s multiple range test for pairwise comparisons (Holzer and Precht, 1992). For ordinal data, such as inflammation scores (0–3), the non-parametric Kruskal–Wallis test was applied, followed by Dunn’s post hoc test to identify differences between groups. A significance level of p ≤ 0.05 was considered statistically significant. Ethical approvalThis study was conducted in accordance with the ethics guidelines of the Faculty of Veterinary Medicine, Duhok University, Iraq (ethics approval number: 10/01/2025, Reference No: CVM2025/1001UoD). ResultsHistopathological featuresAssessment of inflammatory reaction intensity in subcutaneous tissue using hematoxylin and eosin stainingThe anti-inflammatory effects of TiO2NPs were evaluated by assessing inflammatory cell infiltration across four experimental groups. In the positive control group (Group A), a very high inflammatory cell count was observed (131.5 ± 15.22), with 90% of samples showing severe inflammation (Score 3) and 10% showing moderate inflammation (Score 2). Group B, treated with 0.5% TiO2NPs, exhibited a comparable inflammatory response (125.1 ± 14.94), with 85% of the samples scoring severe (Score 3) and 15% scoring moderate (Score 2), showing no statistically significant difference compared with Group A. In contrast, Group C (1% TiO2NPs) demonstrated a significant reduction in inflammatory cell infiltration (65.9 ± 23.87), with 85% of the samples showing moderate inflammation (Score 2). The most pronounced anti-inflammatory effect was recorded in Group D (2% TiO2NPs), which showed a highly significant decrease in inflammatory cells compared with all other groups. In this group, 90% of the samples scored low (Score 1) and 10% scored moderate (Score 2), with the lowest mean inflammatory cell count (17.4 ± 5.90) (Table 1). Table 1. Determining the inflammatory reaction intensity of subcutaneous tissues by using different concentrations of TiO2NPs.
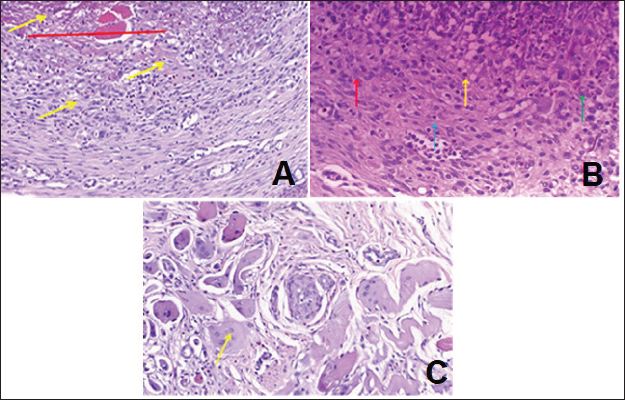
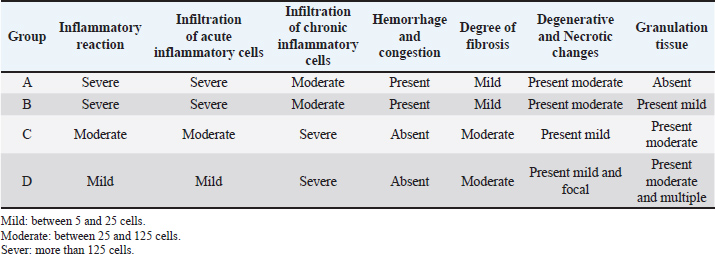
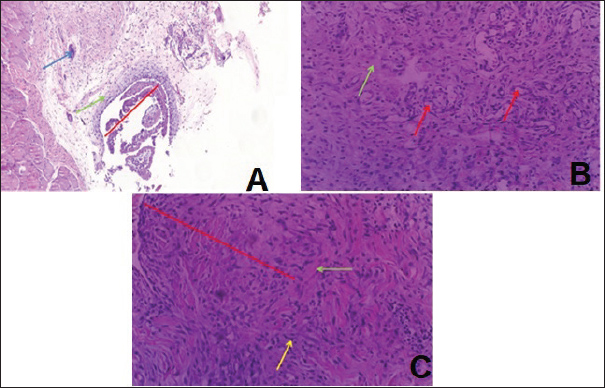
Granulomatous inflammation is a distinctive form of chronic inflammation induced by subcutaneous injection of PMMAs in rabbits (Table 2). Histopathological examination of Group A revealed a severe inflammatory reaction characterized by extensive subcutaneous tissue damage, marked hemorrhage, pus formation, and dense infiltration of acute inflammatory cells (mainly neutrophils) along with chronic inflammatory cells such as monocytes, plasma cells, lymphocytes, and macrophages (Fig. 1A and B). Mononuclear leukocytes, particularly histiocytes (macrophages), were observed to respond to PMMA-induced injury, with many exhibiting the typical features of epithelioid cells, including round-to-oval nuclei, irregular nuclear contours, and abundant granular eosinophilic cytoplasm with indistinct borders. Some of these epithelioid cells fused to form multinucleated giant cells, highlighting the granulomatous pattern of inflammation, which is important in narrowing clinical differential diagnoses (Fig. 1C).
Fig. 1. Histopathological features of the subcutaneous tissue in rabbits that received only PMMA. A: Severe inflammatory reaction red line, represented by severe infiltration of inflammatory cells (yellow arrow). B: Severe infiltration of histocytes (red arrow), lymphocytes (blue arrow), neutrophils (green arrow), and eosinophils (yellow arrow). C: Formation of foreign body giant cells (yellow arrow) in the area of inflammatory reaction (H&E, magnif. 20×). Table 2. Overall histological hallmarks of subcutaneous tissue in rabbits.
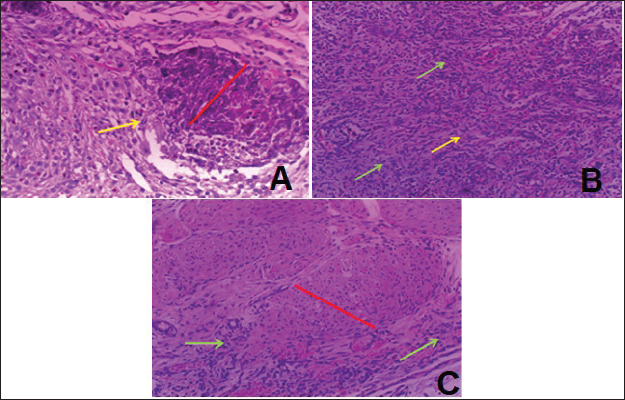
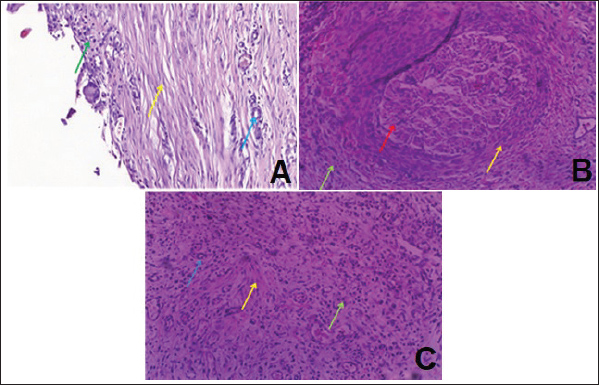
Microscopical examination of the subcutaneous tissue of rabbits exposed to PMMA combined with 0.5% TiO2NPs revealed granulomatous inflammation similar to that observed in Group A, but with less severe inflammatory changes (Fig. 2A). The reaction was characterized by severe hemorrhage, vascular congestion, focal granuloma formation, moderate pus accumulation, and marked infiltration of acute inflammatory cells together with mononuclear inflammatory cells, including monocytes, plasma cells, lymphocytes, and macrophages. Numerous histiocytes were also present, many of which exhibited epithelioid cell morphology with round-to-oval nuclei, irregular nuclear contours, and abundant granular eosinophilic cytoplasm. Some of these epithelioid cells fused to form multinucleated giant cells, although they were less abundant than those in Group A (Fig. 2B). Moderate degenerative and necrotic tissue changes were also evident (Fig. 2C).
Fig. 2. Histopathological features of subcutaneous tissue in rabbits treated with PMMA with 0.5 % TiO2NPs. A: shows the beginning of granuloma formation (red line) with mild fibrosis formation (yellow arrow). B: Infiltration of acute and chronic inflammatory cells (green arrow) with mild fibrosis (yellow arrow). C: A severe inflammatory reaction (granulomatous red line) associated with severe infiltration of acute and chronic inflammatory cells (green arrow) (H&E, magnif. 20×). In Group C, rabbits that received a subcutaneous injection of PMMA with 1% TiO2NPs showed no inflammatory exudate, hemorrhage, and vascular congestion, but displayed degenerative and necrotic changes in the subcutaneous muscle tissue, along with marked proliferation of newly formed blood vessels (neovascularization) compared with the previous groups (Fig. 3A–C). In contrast, rabbits in Group D, which received PMMA with 2% TiO2NPs, exhibited a markedly reduced inflammatory reaction characterized by mild infiltration of acute inflammatory cells and moderate to severe infiltration of chronic inflammatory cells. This was accompanied by abundant histiocytic cells and foreign body giant cells, without evidence of degenerative or necrotic changes, but with prominent angiogenesis, as indicated by extensive proliferation of newly formed blood vessels (Fig. 4A–C).
Fig. 3. Histopathological features of subcutaneous tissue in rabbits treated with PMMA with 1 % TiO2NPs. A: Localized inflammatory reaction as granuloma red line surrounded by inflammatory cells, fibrous connective tissue zone (green arrow), and foreign body giant cells (blue arrow). B: shows obvious proliferation of newly blood vessels neovascularization red arrow and fibrosis green arrow. C: shows localization inflammatory reaction as a granuloma red line surrounded by a zone of fibrous connective tissue green area and severe infiltration of mononuclear inflammatory cells yellow arrow (hematoxylin and eosin, magnif. 20×).
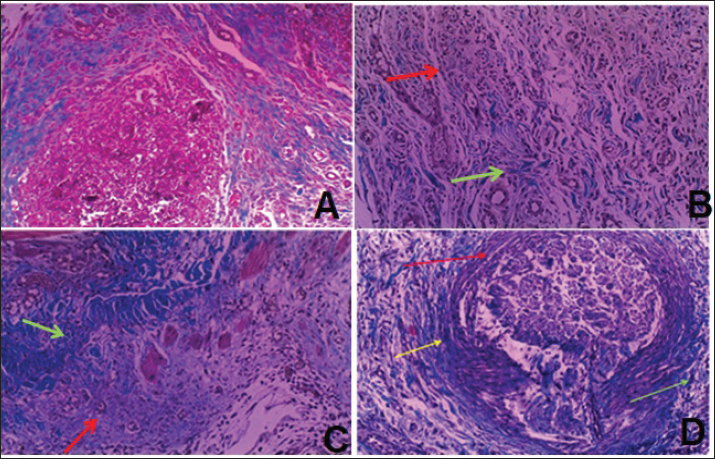
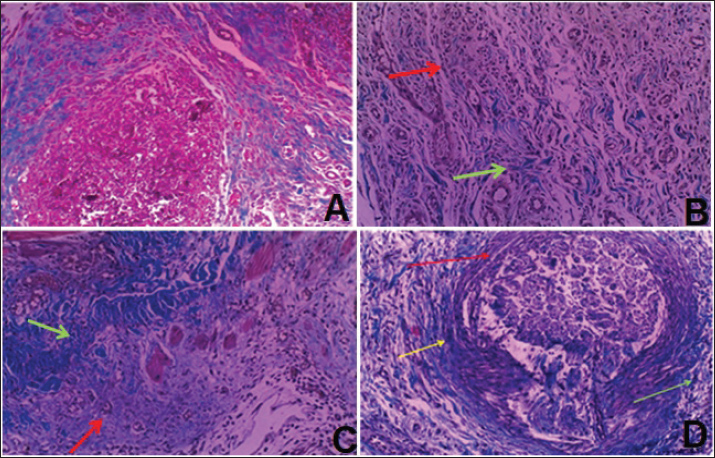
Fig. 4. Histopathological features of subcutaneous tissue in rabbits treated with PMMA with 2 % TiO2NPs. A: Granulomatous reaction surrounded by excessive fibrous connective tissue (yellow arrow) with clear infiltration of chronic inflammatory cells (green arrow) and neovascularization (blue arrow). B: granulomatous reaction red arrow surrounded by excessive fibrous connective formation yellow arrow with clear infiltration of chronic inflammatory cells green arrow. C: excessive formation of fibrosis yellow arrow with clear infiltration of chronic inflammatory cells green and neovascularization blue arrow (H&E, magnif. 20×). Evaluation of subcutaneous tissue fibrosis using Masson’s trichrome stainingMasson’s trichrome staining was used to assess the degree of fibrosis in the subcutaneous tissue at the implantation sites. Mild fibrosis was detected surrounding the granulomatous reaction in the group exposed to PMMA alone, without evidence of degenerative or necrotic changes or neovascularization (Fig. 5A). In the group treated with PMMA and 0.5% TiO2NPs, mild fibrosis was also observed, associated with a severe inflammatory response (Fig. 5B). In contrast, the group treated with 1% TiO2NPs exhibited a localized foreign body granulomatous reaction, which was microscopically identified as a granulomatous lesion encircled by a zone of chronic inflammatory cells and fibrotic tissue (Fig. 5C). The most pronounced fibrotic response was observed in the group treated with 2% TiO2NPs, which demonstrated marked fibroblast proliferation and extensive fibrosis surrounding multiple localized granulomatous areas (Fig. 5D).
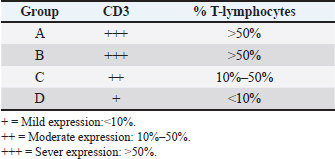
Fig. 5. Histopathological features of the subcutaneous tissue in rabbits receiving only PMMA (A), PMMA with 0.5% TiO2NPs (B), PMMA with 1% TiO2NPs (C), and PMMA with 2% TiO2NPs (D). PMMA, polymethyl methacrylate; PMMA, polyacrylamide. A: An inflammatory reaction that appears as a granuloma surrounded by different types of inflammatory cells and fibrosis (blue area). B: An inflammatory reaction that appears as a granulomatous reaction (red arrow) with infiltration of both types of inflammatory cells and fibrosis (green arrow). C: Localization of the inflammatory reaction as a granuloma (red arrow) surrounded by a zone of fibrous connective tissue (green arrow). D: The granulomatous reaction (red arrow) is surrounded by excessive fibrous connective formation (yellow arrow) with clear infiltration of chronic inflammatory cells (green arrow) (MT, magnif. 20×). Immunohistochemical evaluation of CD3 expression in subcutaneous tissuesImmunohistochemical analysis was performed to evaluate CD3 expression in the subcutaneous tissue of rabbits, and the results are summarized in Table 3. CD3-positive T lymphocytes were detected in all experimental groups, and their distribution was correlated with the degree of immune cell infiltration in areas of inflammation. Immunostaining appeared as distinct membranous staining, confirming the presence of T cells, while also reflecting the activation of histiocytes, which originate from bone marrow-derived myeloid precursors that mature into circulating monocytes and subsequently differentiate into histiocytes upon tissue recruitment. Strong CD3 immunostaining was observed in Group A (Table 3; Fig. 6A). Groups B and C exhibited severe to moderate CD3 expression (Fig. 6B and C), whereas Group D showed only mild CD3 staining intensity, indicating reduced T-cell infiltration compared with the other groups (Fig. 6D).
Fig. 6. Immunohistochemical staining of subcutaneous tissue showing membranous CD3 expression in all experimental groups (A, B, C, and D) and in the positive control (E). Red circles indicate CD3+ T lymphocytes (magnification. 20×). Table 3. Results of IHC membranous expression of CD3.
DiscussionThis study demonstrates that TiO2NPs elicit dose-dependent anti-inflammatory and immunomodulatory effects in a rabbit model of subcutaneous inflammation. The TiO2NPs appear to exert anti-inflammatory activity primarily by attenuating oxidative stress. Their interaction with endogenous and exogenous antioxidants helps to scavenge excessive reactive oxygen species, thereby protecting tissues from oxidative injury and downregulating key pro-inflammatory mediators. Additionally, TiO2 may promote the production of regulatory cytokines and suppress macrophage activation, potentially inducing immune cell exhaustion and reducing inflammatory responses (Huang et al., 2021). The control group exhibited acute inflammatory responses characterized by prominent infiltration of polymorphonuclear and mononuclear cells, with no evidence of necrosis or granuloma formation (Milhomem et al., 2023). This is consistent with the tendency of PMMA to enhance microbial biofilm growth and surface roughness, facilitating microbial adhesion (Marra et al., 2012; Arab et al., 2021). While 0.5% exhibited no significant decrease compared with the control group, it did not reach the biological threshold required to activate anti-inflammatory regulatory pathways. In contrast, increasing concentrations of TiO2 group with NP in PMMA (1% and 2%, respectively) significantly reduced acute inflammatory indicators, such as hemorrhage, congestion, inflammatory exudates, and edema. Meanwhile, markers of chronic inflammation, including lymphocytic infiltration, multinucleated giant cells, fibrosis, and neovascularization, increased progressively. These outcomes reflect the immunomodulatory and antimicrobial properties of TiO2NPs, which are recognized as broad-spectrum antibacterial and antifungal agents with minimal adverse effects (Planchon et al., 2017; López de Dicastillo et al., 2021). Granulomatous reactions were observed at 0.5% TiO2 NPs and became more pronounced at 1% and 2%, histologically defined by aggregates of epithelioid histiocytes surrounded by lymphocytes, plasma cells, and fibrous connective tissue, consistent with foreign body granulomas (El-Daly, 2017; Shah et al., 2017). Masson’s trichrome staining revealed a significant increase in collagen fiber area in treated groups compared to controls, with loosely arranged bundles associated with granulomatous tissue, suggesting that TiO2NPs promote collagen deposition and enhance wound healing (Mack, 2018; Cruz et al., 2021). Mechanistically, these effects are likely mediated by immune pathways involving CD3+ T lymphocytes that release cytokines such as IL-2, IFN-γ, TNF, and lymphotoxin to drive macrophage transformation into epithelioid and multinucleated giant cells, resulting in granuloma formation as part of a chronic healing process and linking innate and adaptive immunity (Harish et al., 2012; Ohtani, 2013). Overall, these results indicate that TiO2NPs, when incorporated into biomaterials like PMMA, not only attenuate acute inflammation but also promote chronic reparative processes, granulomatous reaction, fibrosis, and collagen remodeling, highlighting their potential as therapeutic agents. However, the observed increase in fibrosis at higher TiO2NPs concentrations suggests potential alterations in tissue mechanical properties that may be detrimental if excessive, thus requiring careful dose optimization to balance anti-inflammatory benefits with fibrotic effects. ConclusionThis study demonstrates that the incorporation of TiO2NPs into PMMA induces granulomatous inflammation in a dose-dependent manner, with histopathological features reflecting progressive immune modulation. Granulomatous reactions were closely associated with lymphocyte activity, indicating a strong interplay between the material and host immune response. Importantly, the controlled nature of these reactions and the enhancement of collagen deposition suggest favorable biocompatibility of TiO2-PMMA composites for potential biomedical applications, particularly in the oral mucosa and skin. AcknowledgmentsNot applicable. Conflict of interestThe authors declare no conflict of interest. FundingThis study did not receive any funding support. Authors' contributionsAli Shakir Hasan conducted the majority of the work, including data collection, analysis, interpretation, and manuscript writing. Jiyar Mohammed Naji and Mohammad Ismail Alberfkani supervised the study, provided guidance throughout the research process, and reviewed the final manuscript. All authors read and approved the final version of the manuscript. Data availabilityAll data were provided in the manuscript. ReferencesAhmed, F.Y., Farghaly Aly, U., Abd El-baky, R.M. and Waly, N.G.F.M. 2020. Comparative study of antibacterial effects of titanium dioxide nanoparticles alone and in combination with antibiotics on MDR Pseudomonas Aeruginosa Strains. Int. J. Nanomed. 15, 3393–3404; doi: 10.2147/IJN.S246310 Aligamaleldin, N. and Elsherbini, N. 2022. Biocompatibility of conventional acrylic resin versus modified with titanium dioxide nanoparticles in maxillofacial prosthesis. A parallel randomized control trial. Egypt. Dental. J. 68(1), 807–815. Al-Jubori, S.H., Alsalman, T.H. and Taqa, A.A. 2017. Evaluation of the biocompatibility of a newly prepared endodontic biosealer using subcutaneous implant on rabbits. Int. J. Enhanced. Res. Medicines. &. Dental. Care. (IJERMDC). 4(5), 2349–1590; https://www.researchgate.net/publication/325806580 Amirabad, L.M., Tahriri, M., Zarrintaj, P., Ghaffari, R. and Tayebi, L. 2022. Preparation and characterization of TiO2-coated polymerization of methyl methacrylates (PMMA) for biomedical applications: in vitro study. Asia. Pac. J. Chem. Eng. 17(3), 2761; doi:10.1002/apj.2761 Arab, S., Bahador, A., Sodagar, A., Pourhajibagher, M., Akhavan, A., Niema Hafith, A. and Pornamazeh, T. 2021. Antimicrobial properties of acrylic resin incorporated with propolis nanoparticles. Front. Dent. 18, 29; doi: 10.18502/fid.v18i29.6939 Asim, F.A., Al-Mosaweb, E.H.A. and Hussain, W.A. 2022. Studying the physical and biological characteristics of denture base resin PMMA reinforced with ZrO2 and TiO2 nanoparticles. Karbala. Int. J. Modern. Sci. 8(3), 503–513; doi:10.33640/2405-609X.3251 Blondy, S., Durand, S., Lacroix, A., Christou, N., Bouchaud, C., Peyny, M., Battu, S., Chauvane, A., Carré, V., Jauberteau, M.O., Lalloué, F. and Mathonnet, M. 2021. ColoSTEM Dx kit: innovative alliance of CSCs and glycosylation for earlier prediction of colon cancer aggressiveness and prognosis. Oncology 1, 918702. Cruz, M.A., Araujo, T.A., Avanzi, I.R., Parisi, J.R., De Andrade, A.L.M. and Rennó, A.C.M. 2021. Collagen from marine sources and skin wound healing in experimental animals: a systematic review. Mar. Biotechnol. 23(1), s1–11. El-Daly, A.A. 2017. Histopathological, Ultrastructural, and Immunohistochemical Effects of Intraperitoneal Injection with Titanium Dioxide Nanoparticles and Titanium Dioxide Bulk on the Liver of Albino Mice. J. Anim. Health Behav. Sci. 1, 104; https://api.semanticscholar.org/CorpusID:67767779 Fatemeh, G., Afsaneh, J., Ali, M., Mohammad, H. and Nafiseh, J. 2023. Different Modification Methods of Poly Methyl Methacrylate (PMMA) Bone Cement for Orthopedic Surgery Applications. Bone. Jt. Surg. 11(8), 485–492. Ferrero-Miliani, L., Nielsen, O.H., Andersen, P.S. and Girardin, S.E. 2007. Chronic inflammation: importance of NOD2 and NALP3 in IL-1 generation. Clin. Exp. Immunol. 147(2), 227–235. Harish, S.P., Jayaprakash, S.K., Shetty, K.P., Teerthanath, S., Michelle, M., Sunil, K.Y., Kishan, P.H.L. and Chandrika. 2012. A histopathological study of granulomatous inflammation. nhs 2(1), 2249; doi:10.1055/s-0040-1703548 Hassan, M., Abdalla, M., Saber, M., Senosy, W., Nassif, M. and Elsherif, M. 2024. Evaluation of Xylazine Ketamine Anesthesia in Rabbits Undergoing Tendon Surgery: a Prospective Randomized Controlled Study. New Valley Vet. J. 4(1), 1–7. doi: 10.21608/nvvj.2023.247402.1038 Holzer, C. and Precht, M. 1992. Multiple comparison procedures for normally distributed analysis of variance models in SAS, SPSS, BMDP, and MINITAB. Comput. Statist. Data. Anal. 13(3), 351–358; doi:10.1016/0167-9473(92)90141-2 Huang, X., He, D., Pan, Z., Luo, G. and Deng, J. 2021. Reactive-oxygen-scavenging nanomaterials for resolving inflammation. Mater. Today. Bio. 11, 100124; doi:10.1016/j.mtbio.2021.100124 Jafari, S., Mahyad, B., Hashemzadeh, H., Janfaza, S., Gholikhani, T. and Tayebi, L. 2020. Biomedical applications of TiO2 nanostructures: recent advances. Int. J. Nanomed. 14(15), 3447–3470; doi:10.2147/IJN.S249441 Klinge, U., Dievernich, A. and Stegmaier, J. 2022. Quantitative characterization of macrophage, lymphocyte, and neutrophil subtypes within the foreign body granuloma of human mesh explants by 5-marker multiplex fluorescence microscopy. Front. Med. 9, 777439; doi:10.3389/fmed.2022.777439 López de Dicastillo, C., Guerrero Correa, M.B., Martínez, F., Streitt, C. and José Galotto, M. 2021. Antimicrobial effect of titanium dioxide nanoparticles. Antimicrobial resistance: a one health perspective. London: IntechOpen. Available at: http://dx.doi.org/10.5772/intechopen.90891. Mack, M. 2018. Inflammation and fibrosis. Matrix. Biol. 2017, 106–121; doi:10.1016/j.matbio.2017.11.010 Marra, J., Paleari, A.G., Rodriguez, L.S., Leite, A.R.P., Pero, A.C. and Compagnoni, M.A. 2012. Effect of an acrylic resin combined with an antimicrobial polymer on biofilm formation. J. Appl. Oral. Sci. 20(6), 643–648. Matsuo, T. and Ichimura, K. 2012. Immunocytochemical diagnosis as inflammation by vitrectomy cell blocks in patients with vitreous opacity. Ophthalmology 119(4), 827–837; doi:10.1016/j.ophtha.2011.10.020 Medzhitov, R. 2010. Inflammation 2010: new adventures of an old flame. Cell 140(6), 771–776; doi:10.1016/j.cell.2010.03.006 Milhomem, A.C., De Souza Jorge, I.M., Dos Santos Arruda, F., Tomé, F.D., Da Costa, E.L., Vinaud, M.C., Pereira, J.X. and De Souza Lino Júnior, R. 2023. Tissue Remodeling After Implantation with Polymethylmethacrylate: an Experimental Study in Mice. Esthetic. Plast. Surg. 47(3), 1205–1216; doi:10.1007/s00266-022-03179-w Nhlapo, N., Dzogbewu, T.C. and De Smidt, O.A. 2020. Systematic review of nanoparticles for improving the biocompatibility of titanium implants. Manuf. Rev. 7, 31; doi: 10.1051/mfreview/2020030 Ohtani, H. 2013. In chronic inflammation, granuloma cells express CD205 (DEC205) antigen and harbor proliferating T lymphocytes: similarity to antigen-presenting cells. Pathol. Int. 63(2), 85–93. Planchon, M., Léger, T., Spalla, O., Huber, G. and Ferrari, R. 2017. Metabolomic and proteomic investigations of impacts of titanium dioxide nanoparticles on Escherichia coli. PLos One. 12(6), e0178437; doi:10.1371/journal.pone.0178437 Ramanathan, S., Lin, Y.C., Thirumurugan, S., Hu, C.C., Duann, Y.F. and Chung, R.J. 2024. Polymethyl methacrylate in Orthopedics: strategies, Challenges, and Prospects in Bone Tissue Engineering. Polymers 16(3), 367. Sánchez-Hernández, A., Polleys, C.M. and Georgakoudi, I. 2023. Formalin fixation and paraffin embedding interfere with the preservation of endogenous NAD(P)H and FAD two-photon excited fluorescence-based optical metabolic assessments. Biomed. Opt. Express 14(10), 5238–5253; doi:10.1364/BOE.498297 Shah, K.K., Pritt, B.S. and Alexander, M.P. 2017. Histopathologic review of granulomatous inflammation (GI). J. Clin. Tuberculosis. Other. Mycobacterial. Dis. 7(1), 1–12; doi:10.1016/j.jctube.2017.02.001 Świeczko-Żurek, B., Zieliński, A., Bociąga, D., Rosińska, K. and Gajowiec, G. 2022. Influence of Nanometals Implemented in PMMA Bone Cement on Biological and Mechanical Properties. Nanomaterials 12(5), 732. Takeuchi, O. and Akira, S. 2010. Pattern recognition receptors and inflammation. Cell 140(6), 805–820. Thongrin, T., Suyapoh, W., Wendo, W., Tangkawattana, P., Sukon, P., Salao, K., Suttiprapa, S., Saichua, P. and Tangkawatana, S. 2023. Inflammatory cell responses in biliary mucosa during Opisthorchis viverrini infection: insights into susceptibility differences among hosts. Open. Vet. J. 13(9), 1150–1166. Youssef, F.S., Fouad, O.A., Ismail, S.H. and Mohammed, G.G. 2024. Therapeutic and toxicological aspects of some metal nanoparticles on the central nervous system. Egypt. J. Vet. Sci. 55(33), 733–745. Zhou, Y., Hong, Y. and Huang, H. 2016. Triptolide attenuates the inflammatory response in membranous glomerulo-nephritis rats via downregulation of NF-κB Signaling Pathway. Kidney Blood Press. Res. 41(6), 901–910; doi:10.1159/000452591 Zhu, X., Xu, Y., Hong, Z., Chen, J., Zhuo, S. and Chen, J. 2015. Multiphoton microscopic imaging of rabbit dorsal skin. Scanning 37(2), 95–100; doi:10.1002/SCA.21184 | ||
| How to Cite this Article |
| Pubmed Style Hasan AS, Naji JM, Alberfkani MI. Inflammatory effects of titanium dioxide nanoparticles against polymethyl methacrylate on the subcutaneous tissue of rabbits. Open Vet. J.. 2025; 15(12): 6379-6389. doi:10.5455/OVJ.2025.v15.i12.22 Web Style Hasan AS, Naji JM, Alberfkani MI. Inflammatory effects of titanium dioxide nanoparticles against polymethyl methacrylate on the subcutaneous tissue of rabbits. https://www.openveterinaryjournal.com/?mno=272597 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.22 AMA (American Medical Association) Style Hasan AS, Naji JM, Alberfkani MI. Inflammatory effects of titanium dioxide nanoparticles against polymethyl methacrylate on the subcutaneous tissue of rabbits. Open Vet. J.. 2025; 15(12): 6379-6389. doi:10.5455/OVJ.2025.v15.i12.22 Vancouver/ICMJE Style Hasan AS, Naji JM, Alberfkani MI. Inflammatory effects of titanium dioxide nanoparticles against polymethyl methacrylate on the subcutaneous tissue of rabbits. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6379-6389. doi:10.5455/OVJ.2025.v15.i12.22 Harvard Style Hasan, A. S., Naji, . J. M. & Alberfkani, . M. I. (2025) Inflammatory effects of titanium dioxide nanoparticles against polymethyl methacrylate on the subcutaneous tissue of rabbits. Open Vet. J., 15 (12), 6379-6389. doi:10.5455/OVJ.2025.v15.i12.22 Turabian Style Hasan, Ali Shakir, Jiyar Mohammed Naji, and Mohammad Ismail Alberfkani. 2025. Inflammatory effects of titanium dioxide nanoparticles against polymethyl methacrylate on the subcutaneous tissue of rabbits. Open Veterinary Journal, 15 (12), 6379-6389. doi:10.5455/OVJ.2025.v15.i12.22 Chicago Style Hasan, Ali Shakir, Jiyar Mohammed Naji, and Mohammad Ismail Alberfkani. "Inflammatory effects of titanium dioxide nanoparticles against polymethyl methacrylate on the subcutaneous tissue of rabbits." Open Veterinary Journal 15 (2025), 6379-6389. doi:10.5455/OVJ.2025.v15.i12.22 MLA (The Modern Language Association) Style Hasan, Ali Shakir, Jiyar Mohammed Naji, and Mohammad Ismail Alberfkani. "Inflammatory effects of titanium dioxide nanoparticles against polymethyl methacrylate on the subcutaneous tissue of rabbits." Open Veterinary Journal 15.12 (2025), 6379-6389. Print. doi:10.5455/OVJ.2025.v15.i12.22 APA (American Psychological Association) Style Hasan, A. S., Naji, . J. M. & Alberfkani, . M. I. (2025) Inflammatory effects of titanium dioxide nanoparticles against polymethyl methacrylate on the subcutaneous tissue of rabbits. Open Veterinary Journal, 15 (12), 6379-6389. doi:10.5455/OVJ.2025.v15.i12.22 |