| Research Article | ||
Open Vet. J.. 2025; 15(12): 6390-6400 Open Veterinary Journal, (2025), Vol. 15(12): 6390-6400 Research Article Placental histomorphology and fetal growth impairment in mice prenatally exposed to cell phone radiationDesy Armalina1,2*, Neni Susilaningsih2, Heri Sutanto3 and Sunarno Sunarno41Doctoral Study Program of Medical and Health Science, Universitas Diponegoro, Semarang, Indonesia 2Anatomy-Histology Department, Faculty of Medicine, Universitas Diponegoro, Semarang, Indonesia 3Department of Physics, Faculty of Science and Mathematics, Universitas Diponegoro, Semarang, Indonesia 4Department of Biology, Faculty of Science and Mathematics, Universitas Diponegoro, Semarang, Indonesia *Corresponding Author: Desy Armalina. Doctoral Study Program of Medical and Health Science, Diponegoro University, Diponegoro, Indonesia. Email: desyarmalina [at] lecturer.undip.ac.id Submitted: 22/07/2025 Revised: 30/10/2025 Accepted: 16/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
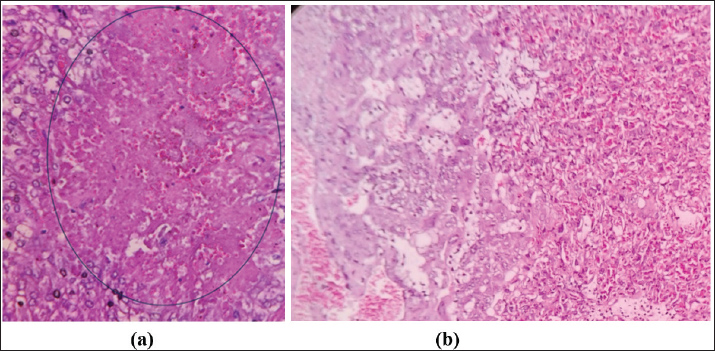
AbstractBackground: Electromagnetic field (EMF) exposure has recently increased significantly. Its influence on pregnancy outcomes, such as small for gestational age, low birth weight, miscarriage, stillbirth, and preterm delivery, remains ambiguous. Extensive research has demonstrated that such exposure increases the risk of adverse pregnancy outcomes, although the mechanisms underlying changes in embryonic development are poorly understood. Aim: This study investigated the impact of EMFs on the placenta and offspring of BALB/c mice subjected to cell phone radiation. Methods: Fourteen pregnant mice were divided into two groups and placed in separate cages. The control group (CG) remained unexposed, whereas the experimental group (EG) was subjected to mobile phone radiation for 24 hours daily between gestational days 1 and 19 at a whole-body specific absorption rate of 1.75 W/kg. On day 20 of gestation, the mice were euthanized, the placenta was excised, and the fetuses were carefully detached. The body dimensions and weights of the pups were then measured. Results: Placental thickness in the decidua basalis of EG mice was reduced compared with that of CG mice, although not statistically significant, whereas the labyrinth zone showed significant thinning. Microscopic damage scoring revealed significantly higher degeneration in the decidua basalis and labyrinth layers of EG. Pup crown–rump length, tail length, and foot size were significantly longer in the CG than in the EG (*p* < 0.05), and body weight was significantly higher in the CG than in the EG. Conclusion: Continuous cell phone radiation during gestation resulted in labyrinth zone thinning, increased microscopic damage scores, and reduced fetal growth parameters. Keywords: Cell phone radiation, Electromagnetic field, Placenta, Mice, Offspring. IntroductionExposure to electromagnetic fields (EMFs) has significantly increased in recent years. Reliance on electronic equipment is a significant component of this heightened exposure (İkinci et al., 2019) As part of a current initiative to evaluate the potential health effects of Radiofrequency Electromagnetic Field exposure in both broad and job-related populations, the International EMF Project of the World Health Organization (WHO) delineated six priority themes for investigation. Oncology, harmful reproductive results, cognitive dysfunction, self-reported manifestations, oxidative stress, and heat-induced consequences are critical for understanding the interplay between environmental factors and health (Verbeek et al., 2021). The WHO has initiated methodical qualitative and experimental research assessments to examine and integrate current scientific knowledge on these topics. Negative reproductive results encompass implications for fertility and conception in men (Cordelliin men and conception (Cordelli et al., 2024). The International Commission on Non-Ionizing Radiation Protection (ICNIRP) suggests an upper limit allowable threshold of 2 W/kg for specific absorption rate (SAR) values in mobile devices [ICNIRP(ICNIRP, 1999]. The European Union sets this conformity. The US Federal Communications Commission (FCC) has instituted a SAR limit of 1.6 W/kg [FCC (The Federal Communications Commission, 2019)]. Not all mobile phone models adhere to the SAR standard of less than 1.6 W/kg, raising concerns about the health effects of radiation. The World Health Organization has recognized electromagnetic radiation as one of the most ubiquitous and rapidly proliferating environmental dangers. (Davies et al., 1995). Microwave radiation is electromagnetic, characterized by frequencies between 300 MHz and 300 GHz. It is extensively used in many fields, such as communications, industry, healthcare, and the military. Previous research has demonstrated that microwave radiation can negatively impact the structure and function of neurological, cardiovascular, reproductive, and hormonal systems, jeopardizing human health (Cameron 1998; Yakymenko et al., 2011; Pall, 2016; Panagopoulo et al., 2021). A study involving pregnant mice revealed heightened oxidative stress and morphological changes at the embryo implantation site in the placenta after exposure to 2.45 GHz EMW, with a specific absorption rate of 0.023 W/kg, for 2 hours daily over 45 days. The result was reproductive dysfunction, as evidenced by implantation failure or embryo resorption, possibly due to increased reactive oxygen species (ROS) generation. This study examined the early stages of gestation. (Shahin et al., 2013). A case–control study observed that women reporting computer or cell phone use exceeding six hours daily had higher odds of abortion than those with lower usage (Zhang et al., 2011). Similarly, a meta-analytic review identified residing within 100 m of a mobile phone base station as an independent risk factor for spontaneous abortion (Ghazanfarpour et al., 2021). Maternal exposure to electromagnetic waves has been linked to pregnancy difficulties, including miscarriage, stillbirth, preterm labor, sex ratio alterations, and congenital impairments (Morvarid et al., 2023). A previous study demonstrated that electromagnetic wave exposure among 1,063 pregnant women in San Francisco resulted in a significant rise in abortion rates associated with elevated peak magnetic field exposure during normal activities (World Health Organization, 2023). Further research indicates that fetal contact with higher electromagnetic waves may decrease the weight at birth, circumference of the head, circumference of the upper arm, circumference of the abdomen, and thickness of the skinfold of the dorsal region, triceps, and abdominal area (Abdul-Al, 2022). Despite extensive research on EMW and pregnancy outcomes, there remains insufficient evidence specifically addressing its effects on the placenta—an essential organ for fetal growth and survival. This knowledge gap, compounded by the impossibility of direct in vivo measurements in humans, underscores the relevance of experimental models to clarify how cell phone radiation may influence placental structure and function. Women undergo substantial geometric and morphological adaptations during pregnancy, including uterine enlargement, placental development, and alterations in maternal body composition, which influence tissue architecture and perfusion (Fang 2024). These dynamic changes can modify the properties of dielectric and biophysical tissues, potentially affecting the absorption and distribution of electromagnetic energy (Sai et al., 2023; Farrugia et al., 2024). Recent experimental and review studies further indicate that prenatal or perinatal exposure to electromagnetic fields may induce oxidative stress, alter growth trajectories, and affect reproductive tissue morphology. However, the findings remain heterogeneous (Cordelli et al., 2023; Kashani et al., 2023) and suggest that pregnancy-related morphological adaptations could interact with EMW exposure, highlighting the need for more targeted investigations with well-defined exposure metrics and morphological endpoints. This study investigated the effects of electromagnetic energy emitted from cell phones on the placenta and offspring of mice with BALB/c. Materials and MethodsAnimal experimentalA total of 21 BALB/c mice were obtained for experimental breeding, maintaining a 2:1 female-to-male ratio of 2:1 (14 females, 7 males). After 7 days of acclimatization, the animals were housed together at night. The presence of a vaginal plug was recorded as GD0. Pregnancy was confirmed on GD10–GD13 by a 3–4 g increase in female body weight, as previously described (Neres et al., 2008). Following pregnancy confirmation, the 14 gestating females were randomly divided into control [control group (CG), n=7] and electromagnetic wave–exposed [experimental group (EG), n=7]. The remaining seven males were only used for mating and were excluded from the experiments. The group size (n=7 per group) was determined based on previous murine pregnancy studies reporting strong effect sizes in placental endpoints (Neres et al., 2008; Kim et al., 2021). An a priori power analysis (G*Power v3.1, two-group comparison, α=0.05, power=0.8) indicated that ~25 animals per group were required to detect medium effects (Cohen’s d=0.8). Considering ethical principles (3R) and resource limitations, the present study was conducted as a pilot with n=7 per group, sufficient to detect large effects. All animals were maintained at 24°C ± 2°C, 50%–60% humidity, with a 12 hours light/dark cycle and ad libitum access to food and water. Temperature and environmental monitoringThe ambient temperature and relative humidity were monitored throughout the exposure period using a digital thermo-hygrometer (HTC-2, Shenzhen, China; range -10°C to 50°C, accuracy ±1°C; humidity range 10%–99% RH, accuracy ±5% RH) placed adjacent to the cages. Measurements were recorded at baseline and every 6 hours during gestational days 0–19. The mean ambient temperature remained at 24°C ± 2°C and the relative humidity was 50%–60%, with no significant differences between EG and CG. These data confirm that the exposure regimen was nonthermal and that the environmental conditions were stable across the groups. The electromagnetic wave exposure systemThe exposure system was designed to simulate radiation from mobile phones. Mice in the EG were continuously exposed 24 hours/day from GD0 to GD19 at a whole-body SAR of 1.75 W/kg. The animals were housed in polypropylene cages (45 × 45 × 25 cm) positioned 20 cm from a commercially available smartphone operating primarily at a GSM carrier frequency of 900 MHz with pulsed modulation (217 Hz). In addition, the device emitted a Wi-Fi signal at 2.45 GHz, reflecting typical dual-mode smartphone operation. The SAR values were standardized to the 900 MHz GSM signal. The output power was calibrated daily using a spectrum analyzer (Agilent Technologies, USA) to ensure uniformity. SAR values were estimated from power density measurements with an isotropic probe and compared with dosimetric data from validated rodent EMW exposure models (Ozguner et al., 2005; Kim et al., 2021). Control animals were kept in a separate room, >3 m from the exposure source. Environmental EMF monitoringBackground EMF levels were monitored using a handheld electromagnetic radiation tester (GM320, Shenzhen, China). The device measures electric fields (1–1999 V/m; resolution 1 V/m) and magnetic fields (0.01–19.99 µT; resolution 0.01 µT) over a bandwidth of 50–3500 MHz with a 0.4-second sampling rate. During the study, the control rooms consistently recorded background E-fields < 1 V/m and H-fields < 0.01 µT, confirming negligible incidental exposure. Sample collectionOn GD20, pregnant mice were euthanized via ketamine injection. All placentas (n=7) and fetuses (n=7) were collected from the 14 pregnant females. Fetal body, tail, and foot lengths (foot length was defined as the distance from the calcaneus to the tip of the third digit) were measured with a digital micrometer (precision 0.01 mm). Histological analysisPlacental tissues were fixed in 4% paraformaldehyde, dehydrated in graded ethanol, cleared in xylene, and embedded in paraffin. Sections (4 µm) were cut with a rotary microtome, deparaffinized, rehydrated, and stained with hematoxylin and eosin (H&E) following standard protocols. (Kiernan, 2015; Fischer et al., 2008; Shah et al., 2025). Microscopic evaluation was performed using an Olympus CX23 microscope. We assessed placental damage in the decidua basalis and labyrinthine layers using a semi-quantitative scoring system adapted with modification from previously published placental histopathology scales (Bos et al., 2023). Placental damage was evaluated in the decidua basalis and labyrinthine layers using a semi-quantitative scoring system: score 0=no damage (0%), score 1=mild damage (<25%), score 2=moderate damage (26%–50%), score 3=severe damage (51%–75%), and score 5=extensive damage (>76%). Damage to the decidua basalis was defined as necrotic cell presence, inflammatory cell infiltration, vascular congestion, and disorganization of maternal tissue architecture. Damage to the labyrinthine zone was defined as syncytiotrophoblast layer thinning, reduced villous branching, capillary collapse, and erythrocyte infiltration within the intervillous space. Figure 1 shows representative photomicrographs illustrating each score category.
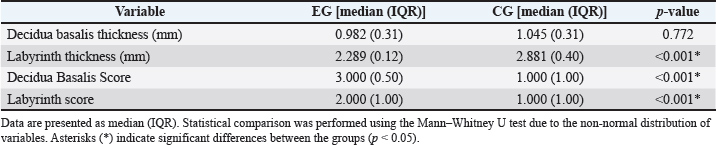
Fig. 1. Histological features of mouse placenta damage score in (a) electromagnetic wave–experimental group (EG) and (b) CG. Placental sections were stained with hematoxylin–eosin (H&E). The blue circle indicates necrosis in the decidual basalis layer. Images captured using an Olympus CX23 microscope at 400 × magnification. Scale bar=50 μm. Statistical analysisThe data distribution was assessed using the Shapiro–Wilk test. Because most variables were nonnormally distributed, non-parametric tests, such as the Mann–Whitney U test, were used for two-group comparisons. For each comparison, we report the Mann–Whitney U, standardized Z, two-tailed p-value, the effect size r (calculated as r=Z / √N, where N is the total sample size), and a 95% confidence interval for the location shift (Hodges–Lehmann estimator). Cohen’s thresholds are used to interpret the effect-size magnitude: small (r ≈ 0.1), medium (r ≈ 0.3), large (r ≥ 0.5). P-values are reported to three decimals, with values below 0.001 reported as p < 0.001. All analyses were performed using SPSS v26.0 (IBM Corp., Armonk, NY, USA) and R v4.x (Wilcox.test with conf.int=TRUE for Hodges–Lehmann CIs). Significance was set at α=0.05. Ethical approvalEthical approval was obtained from the Medical and Health Research Ethics Commission, Diponegoro University (No. 116/EC-H/KEPK/FK-UNDIP/IX/2023). Date: 19 September 2023. ResultsPlacental histology findingsThe analysis of placental parameters revealed the following results (Table 1). The median decidua basalis thickness in the EG group was 0.982 mm (IQR 0.31), compared to 1.045 mm (IQR 0.31) in the CG group, with no significant difference (p=0.772). The labyrinth thickness was lower in the EG group with a median of 2.289 mm (IQR 0.12), whereas that in the CG group was 2.881 mm (IQR 0.40), and the difference was statistically significant (p < 0.001). For the decidua basalis score, the EG group had a median of 3.000 (IQR 0.50) compared to 1.000 (IQR 1.00) in the CG group, showing a significant difference (p < 0.001). Similarly, the labyrinth score in the EG group (median 2.000, IQR 1.00) was significantly higher than that in the CG group (median 1.000, IQR 1.00), with p < 0.001. Table 1. Placental morphometric and histological scores in experimental (EG) and control groups (CG).
Microscopic analysis supported these findings, with Figures 1 and 2 demonstrating clear histological differences. Inflammation characterized by erythrocyte infiltration (blue arrows) was more apparent in the labyrinth layer of the EG, whereas necrotic cell presence (green arrows) was identified in the decidua basalis layer, suggesting that placental integrity may be disrupted by EMW exposure during gestation. Additionally, the tissue structure in the EG appeared more irregular and degenerated than the well-organized CG cellular architecture.
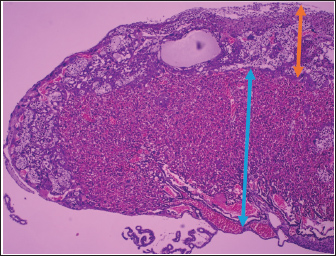
Fig. 2. Histological section of the placenta of a pregnant mouse at gestational age. (H&E staining, 100 ×, scale bar ≈ 100 µm). The orange arrow indicates the decidua basalis, which is the maternal component of the placenta and consists of large decidual cells with eosinophilic cytoplasm and supporting connective tissue. The blue arrow indicates the labyrinth zone, the fetal component of the placenta characterized by abundant vascular spaces and thin trophoblastic septa, which is the main site of maternal–fetal exchange. Findings of fetal anthropometryThe comparison of morphometric parameters between the EG and the CG is presented in Table 2. Representative images of pup and placenta weighing for both groups are shown in Figure 3. The median crown–rump length was significantly lower in the EG [126.66 mm (126.23–126.93)] than in the CG [131.96 mm (131.89–132.20), p < 0.001]. Examples of crown–rump length measurement procedures in both groups are illustrated in Figure 4.
Fig. 3. (A) The image shows a pup weighing from the EG along with its placenta using a digital balance (OHAUS Scout). The sample was placed on aluminum foil to maintain cleanliness and prevent direct contact with the balance surface. The recorded weight was 0.83 g, representing the combined weight of the pup and the attached placental tissue. (B) The image shows a pup weighing its placenta from the CG using a digital balance (OHAUS Scout). The specimen was placed on aluminum foil to prevent direct contact with the balance surface and to maintain hygiene. The measurement recorded a weight of 1.55 g, representing the combined weight of the pup and its placenta. <AQ13>
Fig. 4. (A) The image shows the CRL measurement in an EG mouse fetus using a digital caliper (Mitutoyo, Japan). Measurements were taken from the crown (top of the fetal head) to the rump (buttocks). The caliper display shows a representative length of 12.73 mm. (B) Another crown–rump length (CRL) measurement in a CG mouse fetus. The specimen was positioned on a clean laboratory surface, and the digital caliper reading indicated a representative CRL of 13.18 mm. Table 2. Comparison of crown–rump length, tail length, foot length, and body weight between the control (CG) and experimental (EG) groups.
A similar trend was observed in tail length, with the EG exhibiting shorter values [115.65 mm (114.96–116.36)] than the CG [119.00 mm (118.70–119.20), p < 0.001]. The measurement method for tail length is demonstrated in Figure 5. The EG also demonstrated significantly shorter foot length [112.31 mm (112.18–112.45)] than the CG [112.66 mm (112.64–112.82), p < 0.001]. Representative images of foot (hind limb) length measurement are provided in Figure 6.
Fig. 5. (A) The image demonstrates the measurement of tail length in an EG mouse fetus using a digital caliper (Mitutoyo, Japan). Measurements were taken from the base of the tail to the distal tip. The caliper display shows a representative tail length of 11.69 mm. (B) Using a digital caliper, the image demonstrates the tail length measurement in a CG mouse fetus (Mitutoyo, Japan). Measurements were taken from the base of the tail to the distal tip. The caliper display shows a representative tail length of 11.82 mm.
Fig. 6. (A) The image shows the measurement of hind limb (foot) length in a mouse fetus using a digital caliper (Mitutoyo, Japan). Measurements were taken from the proximal base of the limb to the distal tip of the digits. The caliper reading demonstrates a representative length of 11.12 mm. (B) The image shows the measurement of hind limb (foot) length in a mouse fetus using a digital caliper (Mitutoyo, Japan). Measurements were taken from the proximal base of the limb to the distal tip of the digits. The caliper reading demonstrates a representative length of 11.25 mm. Moreover, body weight in the EG [0.65 g (0.62–0.72)] was markedly reduced compared with that in the CG [1.38 g (1.32–1.41), p < 0.001]. Overall, all measured parameters (crown–rump length, tail length, foot length, and body weight) were significantly lower in the experimental group than in the control group. DiscussionThis study investigated the impact of continuous EMW exposure at 900 MHz during gestation on placental morphology and fetal anthropometry in BALB/c mice. Our findings demonstrate that EMW exposure induced morphological alterations in placental tissue, particularly in the labyrinthine zone, and increased histological damage scores in both the decidua basalis and labyrinth regions. In addition, fetal anthropometric parameters, including crown–rump length, tail length, foot size, and body weight, were significantly reduced in the exposed group than in the control group. These findings suggest that prenatal exposure to EMW may compromise placental development and impair fetal growth. Placental histology and functional implicationsIn rodents, the placenta comprises distinct zones with specific anatomical and physiological roles. The decidua basalis is the maternal component directly beneath the implantation site and is rich in maternal blood vessels, immune cells, and trophoblast invasion. It establishes the maternal-fetal interface and supports structural anchoring, nutrient supply, and immunological tolerance (Kusuma et al., 2018; Vornic et al., 2024). In contrast, the labyrinth zone is the fetal component specialized for exchange: it contains maternal sinusoids and fetal capillaries separated by syncytiotrophoblast layers, creating a large surface area where gas, nutrient, and waste exchange occurs (Furukawa et al., 2011; Woods et al., 2018). Microscopic assessment revealed labyrinth layer thinning, which is responsible for maternal–fetal exchange, and necrosis and inflammatory infiltration. Such histopathological alterations indicate compromised structural integrity and may impair nutrient and oxygen transfer. Previous studies have shown that electromagnetic field exposure can induce marked structural alterations in placental tissue, including nuclear and cytoplasmic degeneration. Other investigations have demonstrated that early placental villi exhibit reduced expression of key antioxidant enzymes under similar conditions. These findings reinforce the view that placental tissue is highly vulnerable to EMW-induced injury and that oxidative imbalance may be one of the critical pathways that mediate these effects (Wdowiak et al., 2017; Kashani et al., 2023). Although oxidative stress is often proposed as the primary mechanism mediating EMW-related placental alterations, our study did not directly measure biomarkers such as malondialdehyde (MDA), superoxide dismutase (SOD), or catalase (CAT). Therefore, our mechanistic interpretation remains speculative. Based on existing literature, the placenta is a major source of ROS due to its high mitochondrial activity and rapid cellular proliferation. (Grzeszczak et al., 2023). Under physiological conditions, antioxidant defenses, including Cu/Zn-SOD, CAT, and glutathione peroxidase, maintain redox balance. However, excessive ROS generation can overwhelm these defenses, leading to lipid peroxidation, protein damage, and Deoxyribonucleic Acid instability. Previous studies support this hypothesis: Bektas et al. (2020) reported elevated oxidative stress markers (MDA, 8-OHdG) in placental and umbilical cord samples from women with high cell phone use. (Bektas et al., 2020), while Luo et al. (2013) and Kashani et al. (2023) found that EMW exposure decreased the antioxidant enzyme TXNL-2 in early pregnancy (Luo et al., 2013; Kashani et al., 2023). Thus, although our histological findings are consistent with oxidative injury, the absence of direct biochemical assays limits definitive mechanistic conclusions. To validate the proposed pathway and distinguish oxidative from non-oxidative mechanisms, such as altered calcium signaling (Wdowiak et al., 2017), or endocrine modulation (Kim et al., 2021), future studies should incorporate quantification of oxidative stress biomarkers alongside morphological analyses (Wdowiak et al., 2017; Kim et al., 2021). Stress response and endocrine modulationIn addition to oxidative imbalance, exposure to EMW may influence the maternal hypothalamic–pituitary–adrenal axis, thereby altering glucocorticoid levels and placental hormone regulation. The placenta plays a pivotal role at the maternal–fetal interface in cortisol transfer and metabolism (Kim et al., 2021). Elevated maternal cortisol levels during stress can remodel placental structures and disrupt hormone transport. Although we did not measure cortisol or related endocrine markers in this study, the histological evidence of tissue damage suggests potential endocrine dysregulation as an additional contributor to placental compromise. Changes in fetal anthropometricAnthropometric evaluation revealed significantly lower crown–rump length, tail length, foot size, and body weight in fetuses from the EMW group compared with controls. These results are consistent with previous reports of reduced fetal weight and implantation success following prenatal RF-EMF exposure (Shahin et al., 2013; Cordelli et al., 2024). Keleş (2020) similarly demonstrated that exposure to 900 MHz during the perinatal period affected vertebral development, leading to degenerative alterations in offspring (Keleş, 2020). Conversely, Odaci et al. (2008) found no significant difference in body weight across groups, underscoring the heterogeneity of findings in this field. Such variability likely reflects differences in exposure frequency, SAR values, duration, and animal models (Odaci et al., 2008). In this study, the smaller and lighter fetuses were linked to impaired placental morphology, particularly the reduction of labyrinth zone thickness identified in histological scoring. Because the labyrinth is the principal site of maternal–fetal exchange, its structural compromise may limit the transfer of oxygen and nutrients to the fetus, thereby restricting growth potential. This mechanistic link is supported by evidence that RF-EMF exposure can induce oxidative stress, alter trophoblast function, and disrupt vascularization, all of which may converge to impair labyrinth development and function (Kim et al., 2021; Cordelli et al., 2024). Thus, the coherence between placental alterations and fetal anthropometric deficits strengthens the interpretation that EMW exposure compromises maternal-fetal exchange. Nevertheless, further work is needed to delineate the precise molecular mechanisms involved, whether mediated by oxidative imbalance, endocrine disruption, or altered calcium signaling. Strengths and limitationsThis study provides in vivo evidence of the histological and developmental effects of continuous 900 MHz EMW exposure during pregnancy, employing standardized exposure parameters (SAR 1.75 W/kg) and placental layer histological scoring. Nevertheless, several limitations must be acknowledged. First, the sample size was limited, and larger cohorts would increase the robustness of statistical inference, although supported by power analysis. Second, oxidative stress biomarkers and endocrine markers were not measured, thereby precluding direct mechanistic conclusions. Third, the study relied on a single exposure frequency and intensity, limiting generalizability to real-world scenarios where multiple bands (GSM 1800) are simultaneously active. Finally, the absence of long-term postnatal follow-up restricts the interpretation of EMW exposure’s enduring developmental consequences. Future recommendationFuture studies should include the direct measurement of oxidative stress biomarkers (MDA, SOD, CAT, and Glutathione Peroxidase) and inflammatory mediators to confirm the mechanisms underlying EMW-induced placental damage. Endocrine profiling, particularly cortisol and placental hormones, would further clarify maternal–fetal interactions. Experiments using multiple exposure frequencies and intermittent regimens, along with long-term postnatal follow-up, are recommended to improve translational relevance and better reflect real-world conditions. ConclusionContinuous prenatal EMW exposure at 900 MHz induces histological alterations in the placenta and restricts fetal growth in Bagg Albino/c mice. While oxidative stress is a plausible underlying mechanism, this interpretation is based on existing literature rather than direct measurement of biomarkers. Therefore, the proposed role of oxidative imbalance should be considered as a hypothetical pending biochemical confirmation. These results highlight the need for comprehensive mechanistic studies to determine how EMW exposure impacts maternal–fetal health, with implications for the safety of mobile phone use during pregnancy. AcknowledgmentThe authors express gratitude for the financial support from the International Publication Research, LPPM Universitas Diponegoro, and Endowment Fund for Education Agency. Special thanks are also extended to the laboratory technicians at the Faculty of Medicine, Universitas Diponegoro, for their valuable assistance and technical support during the experimental procedures. Conflict of interestThe authors(s) declare no potential conflicts of interest. FundingThis research was financially supported by the International Publication Research, LPPM Universitas Diponegoro, and Endowment Fund for Education Agency, Indonesia. Authors' contributionDesy Armalina: Conceived and designed the study, conducted the animal handling and exposure procedures, performed data collection and measurements, analyzed the results, and drafted the manuscript. Neni Susilaningsih: Supervised the animal ethics protocol, provided clinical insights into reproductive physiology and placental evaluation, and contributed to data interpretation and manuscript revision. Heri Sutanto: Responsible for designing and validating the electromagnetic wave exposure system, ensuring SAR calibration, and contributing to interpreting physical parameters related to electromagnetic wave effects. Sunarno: Performed histological tissue preparation and microscopic examination, evaluated placental structural changes, and contributed to interpreting biological outcomes and editing the final manuscript. All authors have reviewed and approved the final version of the manuscript. Data availabilityAll data generated or analyzed during this study are included in this article. ReferencesAbdul-Al, M., Amar, A. S. I., Elfergani, I., Littlehales, R., Ojaroudi Parchin, N., Al-Yasir, Y., See, C. H., Zhou, D., Zainal Abidin, Z., Alibakhshikenari, M., Zebiri, C., Elmegri, F., Abusitta, M., Ullah, A., Abdussalam, F. M. A., Rodriguez, J., McEwan, N. J., Noras, J. M., Hodgetts, R. and Abd-Alhameed, R. A. 2022. Wireless Electromagnetic Radiation Assessment Based on the Specific Absorption Rate (SAR): a Review Case Study. Electron. (Switzerland). 11(4), 1–31; doi: 10.3390/electronics11040511 Bektas, H., Dasdag, S. and Bektas, M.S. 2020. Comparison of effects of 2.4 GHz Wi-Fi and mobile phone exposure on human placenta and cord blood. Biotechnol. Biotechnological. Equip. 34(1), 154–162; doi:10.1080/13102818.2020.1725639 Bos, M., Koenders, M.J.M., Dijkstra, K.L., Van Der Meeren, L.E., Nikkels, P.G.J., Bloemenkamp, K.W.M., Eikmans, M., Baelde, H.J. and Van Der Hoorn, M.L.P. 2023. The severity of chronic histiocytic intervillositis is associated with gestational age and fetal weight. Placenta 2022, 28–35; doi:10.1016/j.placenta.2022.11.014 Cameron, I. 1998. Nitric oxide in the endometrium. Hum. Reprod. Update. 4(5), 565–569 Cherukuri, A.S.S., Modi, V.K., Baraskar, B., Sood, S., Reguram, R., Palvia, D., Gopalakrishnan, K., Damani, D.N., Gaddam, S., Samaddar, P., Katukuri, N., Shivaram, S., Dey, S., Mitra, D., Roy, S., Linden, D.R., Beyder, A., Kulkarni, K. and Arunachalam, S.P. 2023. Microwave-Based Dielectric Properties as an Electrophysiological Biomarker : future Perspectives. Electronics 12(3276), 1–25; doi:10.3390/electronics12153276 Cordelli, E., Ardoino, L., Benassi, B., Consales, C., Eleuteri, P., Marino, C., Sciortino, M., Villani, P., Brinkworth, M.H., Chen, G., Mcnamee, J.P., Wood, A.W., Belackova, L., Verbeek, J. and Pacchierotti, F. 2023. Effects of Radiofrequency Electromagnetic Field (RF-EMF) exposure on pregnancy and birth outcomes: a systematic review of experimental studies on non-human mammals. Environ. Int. 180, 108178; doi:10.1016/j.envint.2023.108178 Cordelli, E., Ardoino, L., Benassi, B., Consales, C., Eleuteri, P., Marino, C., Sciortino, M., Villani, P., H. Brinkworth, M., Chen, G., P. Mcnamee, J., Wood, A.W., Belackova, L., Verbeek, J. and Pacchierotti, F. 2024. Effects of radiofrequency electromagnetic field (RF-EMF) exposure on male fertility: a systematic review of experimental studies on non-human mammals and human sperm in vitro. Environ. Int. 185 , 108509; doi: 10.1016/j.envint.2024.108509 Davies, M.G., Fulton, G.J. and Hagen, P.O. 1995. Clinical biology of nitric oxide. Br. J. Surg. 82, 1598–1610. Dimitris, P., Andreas, K., Igor, Y. and Chrousos, G. 2021. Human-made electromagnetic fields: ion forced-oscillation and voltage-gated ion channel dysfunction, oxidative stress and DNA damage. Int. J. Oncol. 59(92), 1–16; doi:10.3892/ijo.2021.5272 Fang, S., Duarte, C.A., Fodera, D.M., Shi, L., Chen, X., Advincula, A.P., Vink, J., Hendon, C.P., Myers, K.M. 2024. Equilibrium tension and compression mechanical properties of the human uterus. Acta Biomater. Doi: 10.1101/2024.04.25.591208 Fang, S. et al. 2025. Acta Biomaterialia Equilibrium mechanical properties of the human uterus in tension and compression. Acta Biomater. 194, 219–232. Farrugia, L., Porter, E., Conceição, R.C., Meo, S.D., Godinho, D.M., Bonello, J., Ragulskis, M., Ocket, I., Farina, L., Cavagnaro, M. and Peyman, A. 2024. The Complex Permittivity of Biological Tissues : a Practical Measurement Guideline. IEEE Access 12, 10296–10314; doi:10.1109/ACCESS.2024.3352728 Fischer, A.H. et al. 2008. Hematoxylin and eosin staining of tissue and cell sections.’, CSH protocols, 2008, p. pdb.prot4986. Available at: https://doi.org/10.1101/pdb.prot4986. Furukawa, S., Hayashi, S., Usuda, K., Abe, M., Hagio, S. and Ogawa, I. 2011. ‘Toxicological Pathology in the Rat Placenta’. J. Toxicol. Pathol. 24, 95–111. Ghazanfarpour, M., Kashani, Z.A., Pakzad, R., Abdi, F., Rahnemaei, F.A., Akbari, P.A. and Roozbeh, N. 2021. Effect of electromagnetic field on abortion: a systematic review and meta-analysis. Open Med. (Poland). 16(1), 1628–1641; doi:10.1515/med-2021-0384 Grzeszczak, K., Łanocha-Arendarczyk, N., Malinowski, W., Ziętek, P. and Kosik-Bogacka, D. 2023. ‘Oxidative Stress in Pregnancy’. Biomolecules 13(1768), 1–23. ICNIRP. 1999. Guidelines for limiting exposure to time-varying electric, magnetic, and electromagnetic fields. Health. Phys. 74(4), 494–522. Kashani, Z.A., Pakzad, R., Fakari, F.R., Haghparast, M.S., Abdi, F., Kiani, Z., Talebi, A. and Haghgoo, S.M. 2023. Electromagnetic fields exposure on fetal and childhood abnormalities: systematic review and meta-analysis. Open Med. (Poland). 18(1), doi: 10.1515/med-2023-0697 Keleş, A.I. 2020. Morphological changes in the vertebrae and central canal of rat pups born after exposure to the electromagnetic field of pregnant rats. Acta. Histochemica. 122, 1–7; doi:10.1016/j.acthis.2020.151652 İkinci, A., Randel, J. and Odacı, E. 2019. Changes in pyramidal and granular neuron numbers in the rat hippocampus 7 days after exposure to a continuous 900-MHz electromagnetic field during early and mid-adolescence. J. Chem. Neuroanatomy 101, 101681. Keleş, A.I., Yıldırım, M., Gedikli, O., Çolakoğlu, S., Kaya, H., Baş, O., Sönmez, O.F. and Odacı, E. 2018. The effects of a continuous 1-h a day 900-MHz electromagnetic fi eld applied throughout early and mid-adolescence on hippocampus morphology and learning behavior in late adolescent male rats. J. Chem. Neuroanatomy 94, 46–53. Kiernan. 2015. Histological and histochemical methods. Kim, H.S., Choi, H.D., Pack, J.K., Kim, N. and Ahn, Y.H. 2021. Biological Effects of Exposure to a Radiofrequency Electromagnetic Field on the Placental Barrier in Pregnant Rats. Bioelectromagnetics 199(2), 191–199; doi:10.1002/bem.22322 Abumaree, M.H., Pertile, M.D. and Kalionis, B. 2018. Isolation and Characterization of Mesenchymal Stem/Stromal Cells Derived from Human Third Trimester Placental Chorionic Villi and Decidua Basalis.In Preeclampsia: methods and Protocols, Methods in Molecular Biology. New York, NY: Humana Press; pp: 247–66. https://doi.org/ 10.1007/978-1-4939-7498-6_19 Li, Z., Yao, F., Cheng, L., Cheng, W., Qi, L., Yu, S., Zhang, L., Zha, X. and Jing, J. 2018. Low Frequency Pulsed Electromagnetic Field Promotes the Recovery of Neurological Function After Spinal Cord Injury in Rats. J. Orthopaedic Res. 80(80), 449–456; doi:10.1002/jor.24172 Luo, Q., Jiang, Y., Jin, M., Xu, J. and Huang, H.F. 2013. Proteomic Analysis on the Alteration of Protein Expression in the Early-Stage Placental Villous Tissue of Electromagnetic Fields Associated With Cell Phone Exposure. Reproductive Sci. 20(9), 1055–1061; doi:10.1177/1933719112473660 Mehmet, Y. et al. 2018. The effects of a continuous 1-h a day 900-MHz electromagnetic fi eld applied throughout early and mid-adolescence on hippocampus morphology and learning behavior in late adolescent male rats. J. Chem. Neuroanat. 94, 46–53. Morvarid, I., Maryam, A. and Mohammad, G. 2023. Electromagnetic field exposure and abortion in pregnant women : a systematic review and meta-analysis. 30(5), 70–80. Neres, R., Romero, C. and Marinho, F. 2008. Pregnancy Outcome and Placenta Pathology in Plasmodium berghei ANKA Infected Mice Reproduce the Pathogenesis of Severe Malaria in Pregnant. PLos One. 3(2), doi:10.1371/journal.pone.0001608 Odaci, E., Bas, O. and Kaplan, S. 2008. Effects of prenatal exposure to a 900 MHz electromagnetic field on the dentate gyrus of rats : a stereological and histopathological study i. Brain Res. 8, 0–5; doi:10.1016/j.brainres.2008.08.013 Ozguner, M., Koyu, A., Cesur, G., Ural, M., Ozguner, F., Gokcimen, A. and Delibas, N. 2005. Biological and morphological effects on the reproductive organ of rats after exposure to electromagnetic field. Saudi. Med. J. 26(3), 405–410. Pall, M. 2016. Electromagnetic Fields Act Similarly in Plants as in Animals: probable Activation of Calcium Channels via Their Voltage Sensor. Curr. Chem. Biol. 10, 74–82; doi:10.2174/22127968106661604191604 Sai, A. et al. 2023. Microwave-based dielectric properties as an electrophysiological biomarker : future perspectives. Electronics. 12(3276), 1–25; doi: 10.3390/electronics12153276. Shah, H., Dan, N., Salunke, A., Ramachandran, A.V. and Pandya, P. 2025. Different dietary compositions alter pubertal onset in Wistar rats. Nutritional. Neurosci. 185, 1–18; doi:10.1080/1028415X.2025.2522454 Shahin, S., Singh, V.P., Shukla, R.K., Dhawan, A., Gangwar, R.K., Singh, S.P. and Chaturvedi, C.M. 2013. 2.45 GHz Microwave Irradiation-Induced Oxidative Stress Affects Implantation or Pregnancy in Mice, Mus musculus. Appl. Biochem. Biotechnol. 169, 1727–1751; doi:10.1007/s12010-012-0079-9 The Federal Communications Commission. 2019. Specific Absorption Rate (SAR) for Cellular Telephones. Available via https://www.fcc.gov/general/specific-absorption-rate-sar-cellular-telephones (accessed 8 September, 2023). The World Health Organization. 2023. Electromagnetic fields. Available via https://www.who.int/health-topics/electromagnetic-fields#tab=tab_1 (accessed 5 September, 2023). Verbeek, J., Oftedal, G., Feychting, M., Van Rongen, E., Rosaria Scarfì, M., Mann, S., Wong, R. and Van Deventer, E. 2021. ‘Prioritizing health outcomes when assessing the effects of exposure to radiofrequency electromagnetic fields : a survey among experts’. Environ. Int. , 146. Vornic, I., Buciu, V., Furau, C.G., Zara, F., Novacescu, D., Barb, A.C., Cumpanas, A.A., Latcu, S.C., Sas, I., Serban, D., Cut, T.G. and Dumitru, C.S. 2024. The Interplay of Molecular Factors and Morphology in Human Placental Development and Implantation. Biomedicines 2908(2908), 12. Wdowiak, A., Mazurek, P.A., Wdowiak, A. and Bojar, I. 2017. Effect of electromagnetic waves on human reproduction. Ann. Agricult. Environ. Med. 24(1), 13–18; doi:10.5604/12321966.1228394 Woods, L., Perez-Garcia, V. and Hemberger, M. 2018. Regulation of Placental Development and Its Impact on Fetal Growth — New Insights From Mouse Models. Front. Endocrinol. 9, 1–18; doi:10.3389/fendo.2018.00570 Yakymenko, I., Sidorik, E., Kyrylenko, S. and Chekhun, V. 2011. Long-term exposure to microwave radiation provokes cancer growth: evidences from radars and mobile communication systems. Exp. Oncol. 33(2), 62–70. Zhang, X., Li, J., Gu, Y., Zhao, Y., Wang, Z. and Jia, G. 2011. A pilot study on environmental and behavioral factors related to missed abortion. Environ. Health. Prev. Med. 16, 273–278; doi:10.1007/s12199-010-0196-4 | ||
| How to Cite this Article |
| Pubmed Style Armalina D, Susilaningsih N, Sutanto H, Sunarno S. Placental histomorphology and fetal growth impairment in mice prenatally exposed to cell phone radiation. Open Vet. J.. 2025; 15(12): 6390-6400. doi:10.5455/OVJ.2025.v15.i12.23 Web Style Armalina D, Susilaningsih N, Sutanto H, Sunarno S. Placental histomorphology and fetal growth impairment in mice prenatally exposed to cell phone radiation. https://www.openveterinaryjournal.com/?mno=272419 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.23 AMA (American Medical Association) Style Armalina D, Susilaningsih N, Sutanto H, Sunarno S. Placental histomorphology and fetal growth impairment in mice prenatally exposed to cell phone radiation. Open Vet. J.. 2025; 15(12): 6390-6400. doi:10.5455/OVJ.2025.v15.i12.23 Vancouver/ICMJE Style Armalina D, Susilaningsih N, Sutanto H, Sunarno S. Placental histomorphology and fetal growth impairment in mice prenatally exposed to cell phone radiation. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6390-6400. doi:10.5455/OVJ.2025.v15.i12.23 Harvard Style Armalina, D., Susilaningsih, . N., Sutanto, . H. & Sunarno, . S. (2025) Placental histomorphology and fetal growth impairment in mice prenatally exposed to cell phone radiation. Open Vet. J., 15 (12), 6390-6400. doi:10.5455/OVJ.2025.v15.i12.23 Turabian Style Armalina, Desy, Neni Susilaningsih, Heri Sutanto, and Sunarno Sunarno. 2025. Placental histomorphology and fetal growth impairment in mice prenatally exposed to cell phone radiation. Open Veterinary Journal, 15 (12), 6390-6400. doi:10.5455/OVJ.2025.v15.i12.23 Chicago Style Armalina, Desy, Neni Susilaningsih, Heri Sutanto, and Sunarno Sunarno. "Placental histomorphology and fetal growth impairment in mice prenatally exposed to cell phone radiation." Open Veterinary Journal 15 (2025), 6390-6400. doi:10.5455/OVJ.2025.v15.i12.23 MLA (The Modern Language Association) Style Armalina, Desy, Neni Susilaningsih, Heri Sutanto, and Sunarno Sunarno. "Placental histomorphology and fetal growth impairment in mice prenatally exposed to cell phone radiation." Open Veterinary Journal 15.12 (2025), 6390-6400. Print. doi:10.5455/OVJ.2025.v15.i12.23 APA (American Psychological Association) Style Armalina, D., Susilaningsih, . N., Sutanto, . H. & Sunarno, . S. (2025) Placental histomorphology and fetal growth impairment in mice prenatally exposed to cell phone radiation. Open Veterinary Journal, 15 (12), 6390-6400. doi:10.5455/OVJ.2025.v15.i12.23 |