| Research Article | ||
Open Vet. J.. 2025; 15(12): 6442-6460 Open Veterinary Journal, (2025), Vol. 15(12): 6442-6460 Research Article Assessment of deep morphology and cryodamage in spermatozoa of indigenous and exotic pig breeds from the Northeast Indian Himalayas using scanning electron microscopy (SEM)Sunil Kumar1*, Prantik Deka1, Rafiqul Islam1, Pranab Jyoti Das2, Lokesha Eranna3 and Vivek Kumar Gupta41Boar Semen Production Centre, India Council of Agricultural Research (ICAR), ICAR-National Research Centre on Pig, Guwahati, India 2Animal Genetics and Breeding, ICAR- National Research Centre on Pig, Guwahati, India 3Animal Nutrition, ICAR-National Research Centre on Pig, Guwahati, India 4Animal Health, ICAR-National Research Centre on Pig, Guwahati, India *Corresponding Author: Sunil Kumar. Boar Semen Production Centre, India Council of Agricultural Research (ICAR), ICAR-National Research Centre on Pig, Guwahati, India Email: skicar2200 [at] gmail.com Submitted: 11/07/2025 Revised: 02/11/2025 Accepted: 16/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
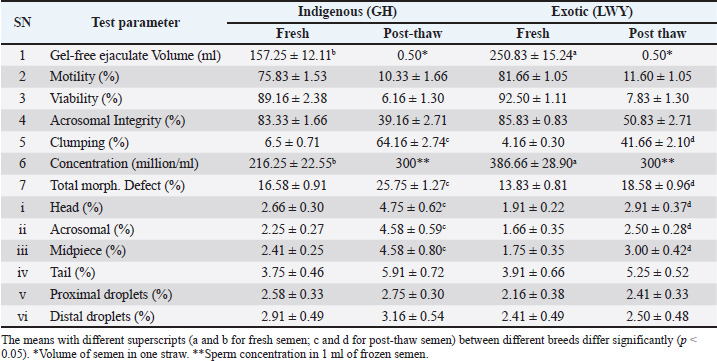
AbstractBackground: Sperm morphology assessment is related to the ejaculate quality, which varies in males and breeds. Routine microscopy does not accurately assess deeper morphological or cryodamage. Comparative quality assessment of indigenous and exotic breed spermatozoa is yet to be characterized under high-resolution microscopy. Aim: This study aimed to identify dimensional, deeper morphological, and cryodamage differences in the fresh and frozen sperm of Indian and exotic pig breeds. Methods: Fresh and frozen-thawed boar spermatozoa of indigenous (Ghungroo) and exotic (Large White Yorkshire) breeds were used to estimate the deep morphological abnormalities, morphometry, and cryodamage using scanning electron microscopy (SEM). Analysis of variance followed by Tukey’s post hoc test was used for data analysis. Results: Under phase contrast microscopy, the quality parameters of fresh semen were found to be within acceptable limits, showing no significant differences (p > 0.05) except for volume and concentration between the two breeds. Furthermore, the quality after thawing was subpar in both breeds. In the examination under SEM, the morphometric dimensions of spermatozoa from the indigenous breed were found to be non-significantly (p > 0.05) lower, with the exception of midpiece width and the perimeter of cytoplasmic droplets, compared to those from the exotic breed. Among the deep morphological abnormalities observed in fresh semen, a significant difference (p < 0.05) was noted between the indigenous and exotic breeds for damaged acrosomal crescent, extruded acrosomal vesicle, cracks on the acrosome, bent tails, fragmented tails, plasma membrane damages (on the head and tail), and multiple vesicles (on the head, midpiece, and tail). Extensive cryodamage was noted after thawing, with significant differences (p < 0.05) in misshapen heads, multiple vesicles on the head, and damage to the acrosomal membrane, crescent, and tail membrane between the indigenous and exotic breeds. Conclusion: SEM is better for assessing deeper sperm morphological abnormalities or cryodamage. Indigenous breed spermatozoa were similar to their exotic counterparts in terms of morphometric dimensions, structural integrity, and cryoresilience. This study opened newer opportunities in semen testing for the early detection of fertility or subfertility for the evaluation of boar breeding soundness. Keywords: Boar, Cryodamage, Electron microscopy, Sperm. IntroductionIndigenous pig breeds in the North East Indian Himalayas are known for their importance in terms of pork preferences, disease resistance, and adaptability to adverse climatic conditions. Indigenous pig breed, namely, Ghungroo, is very important in the backyard to intensive pig farming system (Kumaresan et al., 2007; Patra et al., 2015; Singh et al., 2020). The breed has been identified as a unique indigenous pig breed (Patra et al., 2014; Banik et al., 2024). Exotic pig breeds have also proved their importance in boosting pig production through cross-breeding in different agroclimatic zones (Annual report, 2018) Sperm fertility in fresh or frozen state can differ with respect to morphological abnormalities (Phillips et al., 2004; Kondracki and Wysokińska, 2005). Assessment of sperm morphology is considered a major and routine component of the process as it relates to ejaculate fertility (Saravia et al., 2007). However, the routine methods used to evaluate fresh or frozen sperm morphology under bright-field or phase-contrast microscopy (PCM) allow the investigation of only one characteristic or basic structural property of sperm. This leads to less accuracy in semen quality assessment. Routine semen testing is not useful in predicting or identifying male fertility or subfertility (Wang and Swerdloff, 2014). It is also known that routine sperm morphology by strict criteria does not predict clinical pregnancy rate following intrauterine insemination in humans (Sayegh et al., 2024). For better success, this requires a critical or deep assessment of semen and sperm morphological abnormalities. Moreover, the accuracy in morphological examination becomes or is very valuable during boar selection or breeding soundness examination, so that the positive or negative impacts of sperm morphology can be managed accordingly for future breeding programs in the herd. Hence, deeper morphological studies can provide a better understanding of sperm quality assessment and decision-making for further processing (liquid or frozen) of the collected ejaculate. Morphometry evaluation of spermatozoa can be an indicator of their fertility (Wysokińska et al., 2021). Studies on sperm morphometry in various mammalian species (Peña et al., 2005; Saravia et al., 2007; Hidalgo et al., 2008; Brito et al., 2011) and micro-mini pigs (Kawarasaki et al., 2022) have shown a link between sperm dimensions and fertility. Sperm morphometry is evaluated using traditional microscopy with image analysis software or a computer-assisted semen analyzer (Maroto-Morales et al. 2016; Van Der Horst et al., 2018), and the latter is believed to ensure more reliable and repeatable results (Gago et al., 1998; Yániz et al., 2015). However, comprehensive information on the morphometry of Ghungroo breed spermatozoa is lacking. Frozen semen technology has been important for decades across the world (Yeste, 2016; Lonergan, 2018). However, subfertility and the accurate assessment of semen quality are major hurdles in this technology. In contrast to bovine semen, only 1% of boar semen used for artificial insemination (AI) is cryopreserved (Saravia et al., 2005; Techakumphu et al., 2013). Cryopreservation exposes sperm to physical and chemical stress, and less than 50% of the sperm survive with the ability to fertilize (Hammerstedt et al., 1990). Differences in sperm survival rate after freezing and thawing vary (Waterhouse et al., 2006) between males. A deep understanding of cryodamage at the ultrastructure level is still lacking. Understanding cryodamage can be helpful in developing suitable freezing protocols for the effective cryopreservation of boar sperm (Du et al., 2016; Khophloiklang et al., 2024). For semen quality assessment, the limitations of light microscopy can be overcome using scanning electron microscopy (SEM). SEM approaches go beyond descriptive sperm morphology and can play an essential role in the definition and study of sperm pathology (Chenoweth et al., 2024). SEM provides detailed surface three-dimensional morphological information of the spermatozoa (Bezerra et al., 2018). The same is not possible with PCM. SEM focuses on the surface morphology and external features of sperm and provides a 3D representation of the outer surface of the sperm, showing details such as the head shape, tail, and any abnormalities. Hence, SEM can be useful for assessing sperm morphology and dimensions and detecting surface defects in both fresh and post thaw semen. To date, there is no information available on the morphological dimensions, deep abnormalities, and cryodamage in the spermatozoa of any indigenous pig breed in the country. In the past, the quality of liquid semen in indigenous (Agonda Goan) and Large White Yorkshire (LWY) was assessed using phase contrast microscopy (Chakurkar et al., 2016) and transmission electron microscopy in the Hampshire breed (Khan et al., 2015). No cryodamage or deep morphological changes were reported in these studies. The hypothesis was that SEM can be better than conventional microscopy in assessing differences in the morphological dimensions, abnormalities, and cryoresilence in fresh and post-thaw semen. Moreover, SEM can contribute to a better understanding of the sperm quality assessment of indigenous breeds with exotic counterparts. Hence, this study aimed to assess deep morphological features, dimensional variations, and cryodamage in spermatozoa of indigenous (Ghungroo) and exotic (Large White Yorkshire) pig breeds using field emission scanning electron microscopy (FESEM). Materials and MethodsLocation of the studyThis study was conducted at the Boar Semen Production Centre, within the Animal Reproduction Section of the Indian Council of Agricultural Research (ICAR)-National Research Centre on Pig, Rani 781 131 (Assam). This location is in the lower Brahmaputra valley of the northeast Indian Himalayas, where the average annual rainfall is approximately 1,700 mm. During the months of July and August, the maximum temperature can reach up to 39°C, while it can drop to a minimum of 10°C in January. Selection of animalsHealthy and reproductively viable boars (n=4) from indigenous (Ghungroo; GH, n=2) and exotic (Large White Yorkshire; LWY, n=2) pig breeds (Supplemental Figs. 1 and 2) were selected. These boars were of a similar age range (22–25 months) and body weight (150–170 kg) and were housed in individual pens under standard conditions, unaffected by seasonal variations, at the ICAR-NRC on Pig farm complex in Rani (Assam), India. Semen sample collectionGel-free ejaculates (n=24) were obtained using the double-gloved hand method (Kumar et al., 2024) from healthy, trained, fertile boars of the GH and LWY breeds on a dummy sow (IMV Technologies, France). Six ejaculates (n=6) were collected from each boar, resulting in 12 ejaculates from each breed, resulting in 24 ejaculates. The samples were then transported to the laboratory for initial quality assessment within a timeframe of 30 minutes. Phase contrast microscopy for semen quality assessmentFresh and post-thaw semen samples from both breeds (GH vs. LWY) were assessed for routine tests (motility, viability, acrosomal integrity, morphological abnormalities, and clumping using phase contrast microscopy (DM 3,000 Leica, Germany). Sperm motility, viability, acrosomal integrity, and clumping were evaluated according to standard procedures (Blom, 1950; Zemjanis, 1970; Kovács and Foote, 1992; Rozeboom, 2009). Approximately 200 spermatozoa were counted per sample to determine viability, abnormalities, and acrosomal parameters, and the mean values for each sample were calculated. Sperm concentration was measured using an automated photometer (Accucel, IMV Technologies, France). Ejaculates exhibiting >70% motility, viability, and acrosomal integrity, along with <10 % clumping, were selected for cryopreservation and SEM analysis. Cryopreservation of spermatozoaFollowing the quality assessment, the sperm-rich fraction was maintained at 22°C for 2 hours, subjected to centrifugation (800 × g for 10 minutes), and subsequently reconstituted to a concentration of 3.0 × 109 sperm/ml using Androstar Cryoplus extender, which was enhanced with egg yolk and glycerol. The reconstituted aliquots were then cooled to 5°C, and a second dilution with freezing extender (1.5 × 109 sperm/ml) was performed. After an equilibration period of 1.5 hours, straws (IMV Technologies, France) were promptly filled with 0.5 ml of the extended semen and sealed using polyvinyl alcohol powder. The vapor freezing of the straws was performed by positioning them 5 cm above the liquid nitrogen level for 10 minutes. The straws were then transferred into goblets and immersed in the cryocan. Thawing was performed at 38°C in a water bath for 20 seconds. The post-thaw semen samples were assessed for sperm motility, vitality, acrosomal integrity, and clumping in accordance with the previously outlined procedures. SEM for semen quality assessmentTriplicated semen samples from each boar were processed as fresh (to assess dimensions and deep morphological abnormalities) and cryopreserved post-thaw (to evaluate cryodamage) for SEM microscopy. The sperm samples intended for FESEM were prepared with slight modifications (Nussdorfer et al., 2018). Spermatozoa (both fresh and post-thaw) were washed three times in PBS and fixed in 1% glutaraldehyde (pH 7.4). Following dehydration in graded ethanol and hexamethyldisilazane, the samples were air-dried overnight and mounted on aluminum stubs for gold sputter coating and FESEM imaging (Gemini 300, Zeiss, Germany). Areas of the smear sample (both fresh and post-thaw) were randomly selected under SEM, and spermatozoa were examined at various magnification levels and scale bars. A minimum of 12 spermatozoa per ejaculate were analyzed for morphometry, while 100 spermatozoa per sample were evaluated for deep morphology and cryodamage assessment under SEM. Assessment of sperm morphometric dimensions in spermatozoa of indigenous and exotic breedsIn the SEM images obtained from the fresh sperm of both breeds (GH vs. LWY), various sperm morphometric dimensions were assessed using the J Image Microvision 1.2.7 online open-access tool. These dimensions include total length (µm), head length (µm), head width (µm), sperm head perimeter (µm), sperm head area (µm2), midpiece length (µm), midpiece width (µm), tail length (µm), and the perimeter (µm) of both proximal and distal cytoplasmic droplets. Deep morphological abnormalities and cryodamage assessment in sperm of indigenous and exotic breedSignificant morphological abnormalities in fresh semen and cryodamage in the post-thaw sperm of both breeds were analyzed for (i) head damages (misshapen, detached, with multiple vesicles), (ii) acrosomal damages (acrosomal crescent, acrosomal membrane, extruded acrosomal vesicles, cracks on the acrosome), (iii) midpiece damages (multiple vesicles on midpiece, bent midpiece), (iv) tail damages (bent tail, broken and fragmented tails, multiple vesicles on tail), and (v) plasma membrane damage (over head, midpiece, tail). Although detached heads, bent midpieces, and tails can also be observed under phase contrast microscopy, they were evaluated under SEM as deep morphological abnormalities due to their common occurrence in boar ejaculates. Furthermore, the aforementioned parameters were referred to as deep morphological abnormalities in fresh semen and identified as cryodamage in the post-thaw sperm, as the assessed abnormalities are typically a cause or consequence of cryodamage during semen freezing. The percentage of sperm exhibiting abnormalities or damage to each structural component was calculated individually. Statistical analysisResults were presented as mean ± SEM, and differences were considered significant at p < 0.05. The data were analyzed using two-way analysis of variance followed by Tukey’s post hoc test. Statistical analysis was performed using IBM Statistical Package for the Social Sciences (SPSS) v26 (SPSS Inc.; Chicago, Illinois, USA). Power analysis and assumption checks were performed using open-access statistical tools (clincalc.com). Ethical approvalThe animal study was reviewed and approved by the Institute Animal Ethics Committee of ICAR National Research Centre on Pig, Rani 781 131, Assam, India. The study was approved by the Institutional Animal Ethics Committee as per the approved animal use protocol number NRCP/CPCSEA/1658|IAEC-54 dated 03rd December, 2019. ResultsPhase-contrast microscopy of fresh and post-thaw semen from indigenous and exotic breedsThe quality of semen in both fresh and post-thaw samples was evaluated under phase contrast microscopy for the two breeds (Table 1 and Fig. 1). In the freshly collected ejaculates, the standard semen quality parameters (GH vs. LWY) fell within acceptable ranges. A significant difference (p < 0.05) was noted in the estimated semen volume (ml) (157.25 ± 12.11 vs. 250.83 ± 15.24) and concentration (million/ml) (216.25 ± 22.55 vs. 386.66 ± 28.90) between the two breeds. However, no significant difference (p > 0.05) was found in motility (%) (75.83 ± 1.53 vs. 81.66 ± 1.05), viability (%) (89.16 ± 2.38 vs. 92.50 ± 1.11), acrosomal integrity (%) (83.33 ± 1.66 vs. 85.83 ± 0.83), clumping (%) (6.5 ± 0.71 vs. 4.16 ± 0.30), and total morphological defects (%) (16.58 ± 0.91 vs. 13.83 ± 0.81) between the indigenous and exotic breeds. Additionally, the acrosome (2.25 ± 0.27 vs. 1.66 ± 0.35), midpiece (2.41 ± 0.25 vs. 1.75 ± 0.35), tail (3.75 ± 0.46 vs. 3.91 ± 0.66), proximal droplets (2.58 ± 0.33 vs. 2.16 ± 0.38), and distal droplets (2.91 ± 0.49 vs. 2.41 ± 0.49) were among the routine total sperm morphological abnormalities. Additionally, no significant difference (p > 0.05) was observed in the estimated parameters between the two breeds.
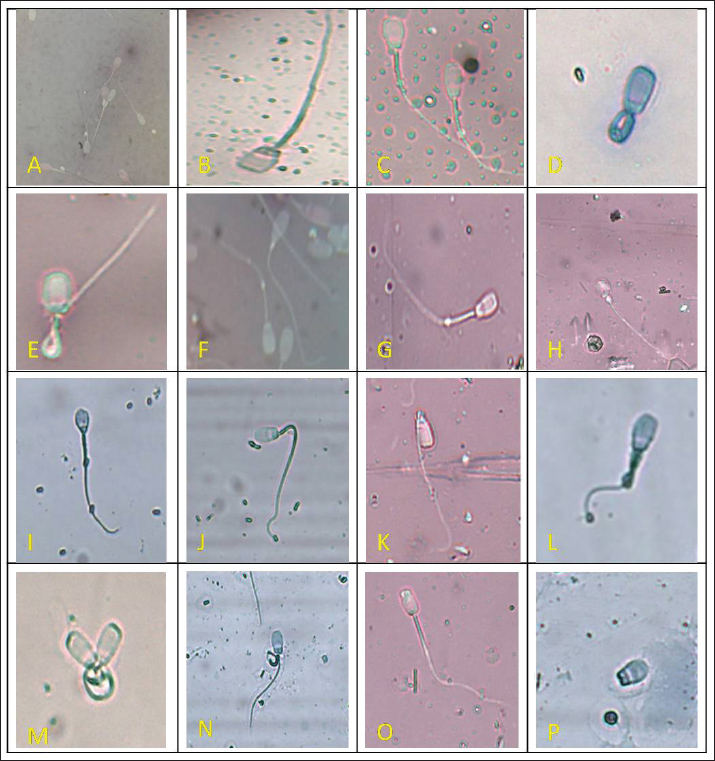
Fig. 1. Phase-contrast microscopic assessment of routine morphological abnormalities in fresh semen of indigenous (GH) and exotic (LWY) pigs. Morphological abnormalities of the head, acrosome, midpiece, and tail are shown (A. double head, B. missing apical crescent, C. globular head, D. stump defect, E. bent tail, F. cytoplasmic droplets, G. distal droplet, H. proximal droplet, I. knobbed acrosome, J. bent mid piece, K. bent neck, L. tail coiled on mid piece, M. abnormal heads, N. coiled mid piece, O. damaged apical ridge, P. detached head). Table 1. Sperm quality of fresh ejaculates and post-thaw semen samples of the LWY and GH breeds.
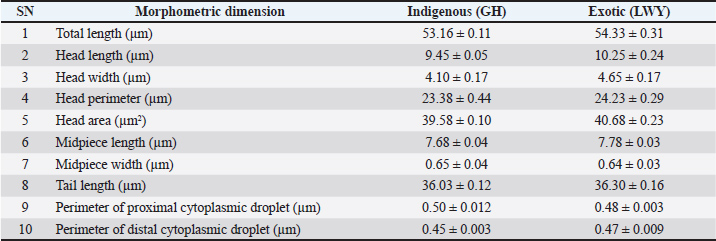
The semen evaluation parameters (GH vs. LWY) in the post-thaw semen samples were not within acceptable bounds, and the percentages of post-thaw motility (10.33 ± 1.66 vs. 11.60 ± 1.05), viability (6.16 ± 1.30 vs. 7.83 ± 1.30), and acrosomal integrity (39.16 ± 2.71 vs. 50.83 ± 2.71) were not significantly different (p > 0.05), with the exception of total morphological defects (%) (25.75 ± 1.27 vs. 18.58 ± 0.96) and clumping (%) (p < 0.05) (64.16 ± 2.74 vs. 41.66 ± 2.10). The percentage of routine sperm morphological abnormalities (GH vs. LWY) in post-thaw semen samples included proximal droplets (2.75 ± 0.30 vs. 2.41 ± 0.33), distal droplets (3.16 ± 0.54 vs. 2.50 ± 0.48), head (4.75 ± 0.62 vs. 2.91 ± 0.37), acrosomal (4.58 ± 0.59 vs. 2.50 ± 0.28), midpiece (4.58 ± 0.80 vs. 3.00 ± 0.42), and tail (5.91 ± 0.72 vs. 5.25 ± 0.52). The head, acrosome, and midpiece abnormalities differed significantly (p < 0.05) between GH and LWY, whereas the tail, proximal droplets, and distal droplets abnormalities did not differ significantly (p > 0.05). In post-thaw semen samples, there was no significant difference (p > 0.05) in abnormalities of the tail, proximal droplets, or distal droplets, but there was a significant difference (p < 0.05) between GH and LWY in abnormalities of the head, acrosome, and midpiece. Morphometric dimensions, deep morphological abnormalities, and cryodamage using SEMTable 2 shows the morphometric dimensions of boar spermatozoa (GH vs. LWY) estimated in fresh semen samples. The overall length of spermatozoa (53.16 ± 0.11 vs. 54.33 ± 0.31), which includes the head (9.45 ± 0.05 vs. 10.25 ± 0.24), midpiece (7.68 ± 0.04 vs. 7.78 ± 0.03), and tail (36.03 ± 0.12 vs. 36.30 ± 0.16), was greater in LWY than in GH but did not show a significant difference (p > 0.05). The dimensions of the head, such as head length (9.45 ± 0.05 vs. 10.25 ± 0.24), head width (4.10 ± 0.17 vs. 4.65 ± 0.17), head perimeter (23.38 ± 0.44 vs. 24.23 ± 0.29), and head area (39.58 ± 0.10 vs. 40.68 ± 0.23) in GH were not significantly lower (p > 0.05) than those in Large White Yorkshire. The width of the midpiece was not significantly wider in the indigenous breed (0.65 ± 0.04) compared to its exotic counterpart (0.64 ± 0.03). The measurements (size) of the proximal droplets (0.50 ± 0.012 vs. 0.48 ± 0.003) and distal droplets (0.45 ± 0.003 vs. 0.47 ± 0.009) were not significantly (p > 0.05) greater in the spermatozoa of the Ghungroo breed than in the LWY breed. Table 2. Dimensions (Mean ± SE) of fresh spermatozoa of pigs of GH and LWY breeds.
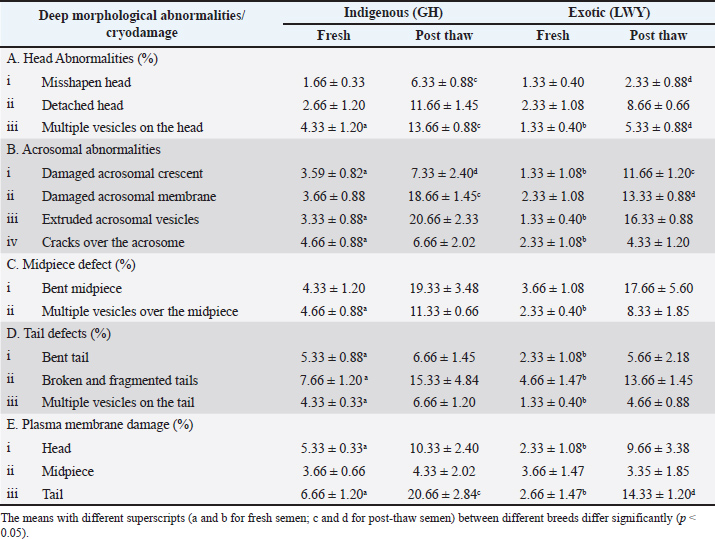
Deep morphological abnormalities and estimation of cryodamageIn fresh semen samples (GH vs. LWY) observed under SEM microscopy (Fig. 2, Fig. 3, Table 3), there was no significant difference (p > 0.05) in the percentage of head abnormalities, such as misshapen head (1.66 ± 0.33 vs. 1.33 ± 0.40) and detached head (2.66 ± 1.20 vs. 2.33 ± 1.08). However, a significant difference (p < 0.05) was found in the presence of multiple vesicles on the head (4.33 ± 1.20 vs. 1.33 ± 0.40) between the two breeds. Additionally, significant differences (p < 0.05) were noted in acrosomal abnormalities, including damaged acrosomal crescent (3.59 ± 0.82 vs. 1.33 ± 1.08), extruded acrosomal vesicles (3.33 ± 0.88 vs. 1.33 ± 0.40), and cracks over the acrosome (4.66 ± 0.88 vs. 2.33 ± 1.08), with the exception of damaged acrosomal membrane, which showed no significant difference (p > 0.05) (3.66 ± 0.88 vs. 2.33 ± 1.08). The percentage of midpiece abnormalities included bent (p > 0.05) with values of 4.33 ± 1.20 compared to 3.66 ± 1.08, and multiple vesicles on the midpiece (p < 0.05) at 4.66 ± 0.88 versus 2.33 ± 0.40. Tail abnormalities showed bent tails (5.33 ± 0.88 vs. 2.33 ± 1.08), along with broken and fragmented tails (7.66 ± 1.20 vs. 4.66 ± 1.47) and multiple vesicles on the tail (4.33 ± 0.33 vs. 1.33 ± 0.40), all exhibiting significant differences (p < 0.05) between the two breeds. There was a notable difference (p < 0.05) in plasma membrane abnormalities (%) in the head (5.33 ± 0.33 vs. 2.33 ± 1.08) and tail (p < 0.05) (6.66 ± 1.20 vs. 2.66 ± 1.47), but no significant difference in the midpiece (p > 0.05) (3.66 ± 0.66 vs. 3.66 ± 1.47) between the indigenous and exotic breeds. In post thaw semen samples (GH vs. LWY) observed under SEM Fig. 4 and Fig. 5), the percentage of head abnormalities, such as misshapen heads (6.33 ± 0.88 vs. 2.33 ± 0.88) and the presence of multiple vesicles on the head (13.66 ± 0.88 vs. 5.33 ± 0.88), showed significant differences, whereas detached heads (11.66 ± 1.45 vs. 8.66 ± 0.66) did not exhibit a significant variation in both breeds. A significant difference (p < 0.05) was noted in acrosomal abnormalities, including damaged acrosomal crescent (7.33 ± 2.40 vs. 11.66 ± 1.20) and compromised acrosomal membrane (18.66 ± 1.45 vs. 13.33 ± 0.88), however, no significant difference (p > 0.05) was noted in the number of extruded acrosomal vesicles (20.66 ± 2.33 vs. 16.33 ± 0.88) and cracks on the acrosome (6.66 ± 2.02 vs. 4.33 ± 1.20).
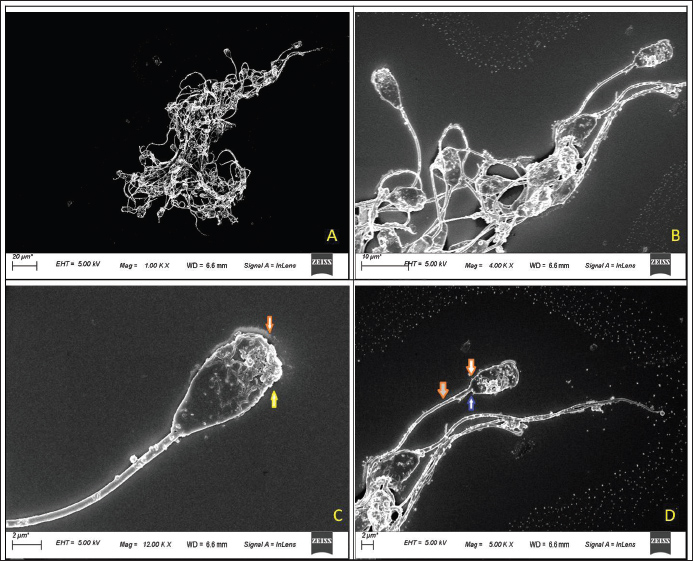
Fig. 2. Scanning Electron Microscopy Assessment of Deep Morphological Abnormalities in Fresh Semen of Exotic (LWY) Pig Breeds The deep morphological abnormalities of the head, acrosome, midpiece, and tail are shown (A. SEM microscopic view of fresh semen sample at 1.00 KX magnification, scale bar=20 m; B. magnified view of fresh semen sample at 4.00 KX magnification, scale bar=10 m; C. fresh spermatozoa showing extrusion of acrosomal vesicle or vesiculated acrosome (yellow arrow0 and discontinued plasma membrane (red arrow) at 12.00 KX magnification, scale bar=2 m) ; D. fresh spermatozoa showing damage to the plasma membrane (red arrow) on the head along with loose attachment of the tail (blue arrow) in the capitultum at 5.00 KX magnification, scale bar=2 m).
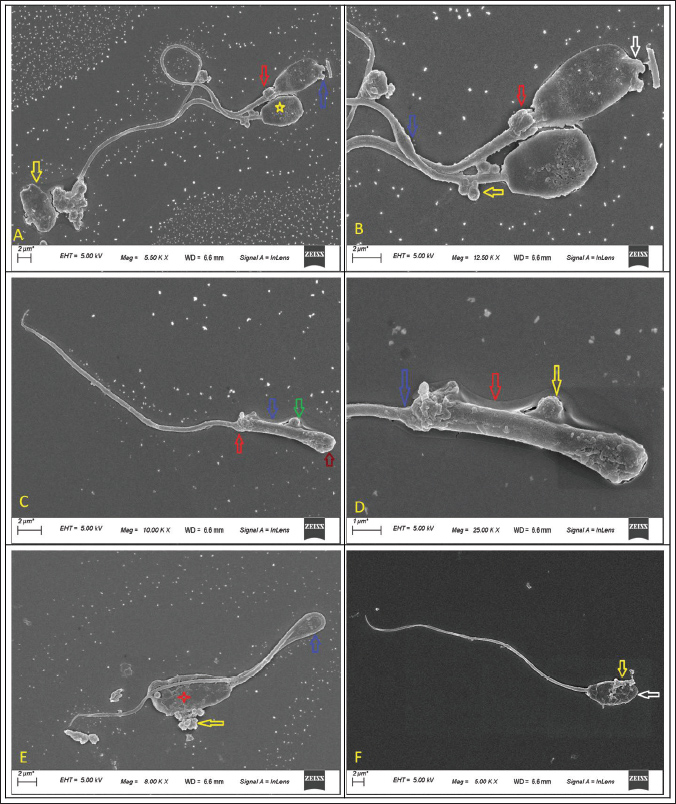
Fig. 3. Scanning electron microscopic assessment of deep morphological abnormalities in fresh semen of GH pigs The deep morphological abnormalities of the head, acrosome, midpiece and tail are shown (A. spermatozoa showing extrusion of acrosomal vesicle (blue arrow), proximal cytoplasmic droplet (red arrow), detached (yellow arrow) and misshaped head (yellow star) at magnification 5.50 KX and scale bar=2 um.; B. spermatozoa showing extrusion of acrosomal vesicle (white arrow) and swollen midpiece (blue arrow), proximal cytoplasmic droplet (red arrow), multiple vesicles on midpiece (yellow arrow) at magnification 12.50 KX and scale bar=2 um.; C. head less spermatozoa showing midpiece (arrow colour- green showing a round vesicle, blue- plasmalemma, red-abaxial tail attachment, red- no head) at magnification 10.00 KX and scale bar=2 um; D. enlarged view of C. at magnification 25.00 KX; and scale bar=1 um; E. spermatozoa showing bent tail (blue arrow), vesicle extrusions (yellow arrow) and cracks over acrosome (red star) at magnification 8.00 KX and scale bar=2 um; F. spermatozoa showing damages acrosome (yellow arrow) and microscopic thinner head(white arrow) at magnification 5.00 KX and scale bar=2 um.
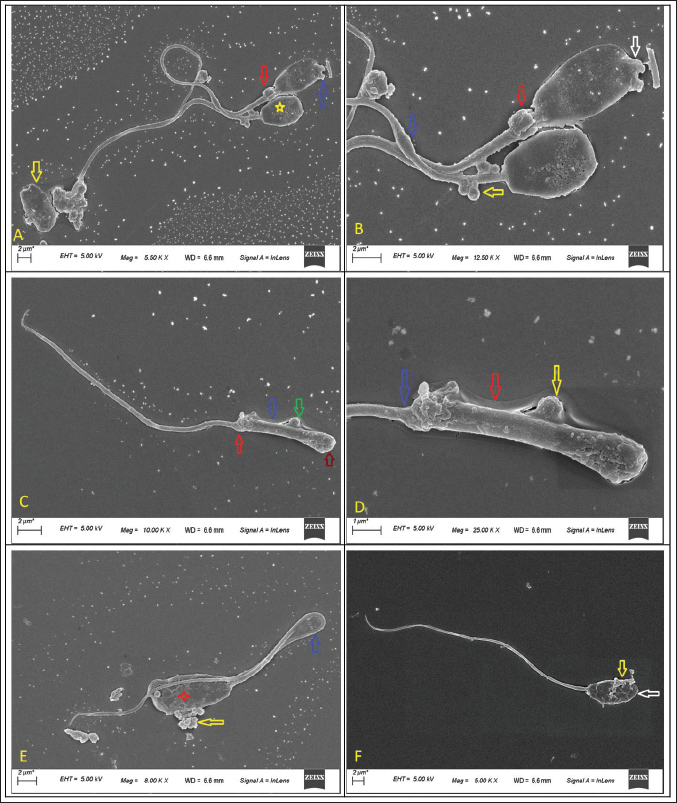
Fig. 4. Scanning electron microscopic assessment of deep morphological abnormalities in fresh semen of GH pigs The deep morphological abnormalities of the head, acrosome, midpiece and tail are shown (A. spermatozoa showing extrusion of acrosomal vesicle (blue arrow), proximal cytoplasmic droplet (red arrow), detached (yellow arrow) and misshaped head (yellow star) at magnification 5.50 KX and scale bar=2 um.; B. spermatozoa showing extrusion of acrosomal vesicle (white arrow) and swollen midpiece (blue arrow), proximal cytoplasmic droplet (red arrow), multiple vesicles on midpiece (yellow arrow) at magnification 12.50 KX and scale bar=2 um.; C. head less spermatozoa showing midpiece (arrow colour- green showing a round vesicle, blue- plasmalemma, red-abaxial tail attachment, red- no head) at magnification 10.00 KX and scale bar=2 um; D. enlarged view of C. at magnification 25.00 KX; and scale bar=1 um; E. spermatozoa showing bent tail (blue arrow), vesicle extrusions (yellow arrow) and cracks over acrosome (red star) at magnification 8.00 KX and scale bar=2 um; F. spermatozoa showing damages acrosome (yellow arrow) and microscopic thinner head(white arrow) at magnification 5.00 KX and scale bar=2 um.
Fig. 5. Scanning electron microscopic assessment of cryodamage in post-thaw semen of GH pigs The cryodamage of the head, acrosome, midpiece, and tail are shown (A. post-thaw spermatozoa at a magnification of 1.00 KX and scale bar=20 m; B. spermatozoa showing detached heads (red arrow), abnormal midpiece (yellow arrow), and damage to the plasma membrane (yellow line) in the head, midpiece, and tail region at a magnification of 2.00 KX and scale bar=10 m; C. spermatozoa showing loss of acrosomal crescent (yellow bar), thinner midpiece (blue arrow) at a magnification of 3.00 KX and scale bar=10 m; D. showing loss of acrosomal crescent (yellow bar), plasmalemma damage (green arrow) at a magnification of 3.00 KX and scale bar=10 m; E. spermatozoa showing damage to the plasma membrane (yellow line) over the sperm at magnification 4.78 KX and scale bar=5 um; F. spermatozoa showing damages to plasma membrane over the sperm(yellow line), loss of acrosomal cap (yellow arrow), vesiculations in the head region (green arrow) at magnification 5.00KX and scale bar=5 um. Table 3. Deep morphological abnormalities (in fresh) and cryodamage (post-thaw) spermatozoa of indigenous and exotic pig breeds.
Similarly, no significant difference (p > 0.05) was noted in the midpiece defect percentages, which encompassed bent midpieces (19.33 ± 3.48 vs. 17.66 ± 5.60) and multiple vesicles on the midpiece (11.33 ± 0.66 vs. 8.33 ± 1.85). No significant differences in tail defects (%) were observed (p > 0.05) between the two breeds, including bent tails (6.66 ± 1.45 vs. 5.66 ± 2.18), broken and fragmented tails (15.33 ± 4.84 vs. 13.66 ± 1.45), and multiple vesicles on tails (6.66 ± 1.20 vs. 4.66 ± 0.88). Regarding plasma membrane damages (%), no significant difference (p > 0.05) was observed over the head (10.33 ± 2.40 vs. 9.66 ± 3.38) or midpiece (4.33 ± 2.02 vs. 3.35 ± 1.85), except for the tail where a significant difference (p < 0.05) was identified between both breeds (20.66 ± 2.84 vs. 14.33 ± 1.20). DiscussionThe quality of ejaculate is influenced by numerous factors, such as breed (Smital et al., 2004; Smital, 2009), seasonal variations (Wysokińska et al., 2009), age of the boar (De Vos et al., 2003), testicular dimensions (Oyeyemi et al., 2002; Oyeyemi and Okediran, 2007), and collection frequency (Frangež et al., 2005). The estimated volume and sperm concentration for both breeds aligned with previous findings (Larsson, 1986; Wiseman et al., 1998; Kondracki et al., 2012). The sperm concentration in indigenous breed semen was lower at (216.25 ± 22.55) compared to exotic breed semen (386.66 ± 28.90). Notably, the sperm concentration in the exotic breed was lower than that previously reported (559.46 × 106) (Chakurkar et al., 2016). These variations in semen volume and concentration may be due to differences in testis size among these breeds or disparities in sperm production per gram of testis or daily sperm output rate. In phase contrast microscopy, the motility (%), viability (%), acrosomal integrity (%), morphological abnormalities (%), and clumping (%) in fresh semen from both breeds were within acceptable limits. While most of the measured parameters showed no significant difference (p > 0.05) between the two breeds, superior values indicative of enhanced semen quality were observed in the exotic breed compared with the indigenous breed. This indicated that there is no significant difference in fertility between spermatozoa from the indigenous and exotic LWY breeds. In the current investigation, the total morphological abnormalities (head, acrosomal, midpiece, and tail) observed in fresh semen were (16.58 ± 0.91 vs. 13.83 ± 0.81) greater in GH than in LWY. The values for total morphological abnormalities were consistent with previous findings (Chakurkar et al., 2016) in indigenous (16.38 ± 1.66) and exotic (10.64 ± 0.88) breeds. Additionally, another study reported that for exotic pigs, the percentages of normal spermatozoa for Pietrain and Duroc (Kondracki et al., 2012) were 94.02 ± 5.64 and 93.53 ± 4.28, respectively. Semen samples from the Pietrain breed exhibited 1.39% ± 1.94% and 4.59% ± 4.49% of primary and secondary abnormalities, respectively, whereas Duroc breed samples showed 1.06% ± 2.02% and 5.41% ± 4.03% for the same categories. This indicated that the Ghungroo and LWY breeds exhibit a higher percentage of total morphological abnormalities than the Pietrain and Duroc pig breeds. The number of abnormalities affects the fertilizing capability of sperm, which is directly related to boar prolificacy (Colenbrander and Kemp, 1990). This may explain the lower fertility observed in GH sperm compared with LWY and in LWY compared with Pietrain and Duroc breeds. No notable difference was found in the quality parameters of fresh semen when examined under phase contrast microscopy. Additionally, no significant difference (p > 0.05) was observed in the quality parameters of post-thaw semen, except for morphological abnormalities, between the indigenous and exotic breeds. This suggested that phase contrast microscopy is not effective at a significant level in distinguishing or determining the routine morphological differences in fresh sperm from both boar breeds. However, such distinctions were possible in post thaw semen samples, likely because of the extensive morphological disturbances caused by the freezing process. Only routine morphological parameters, such as bent tails or double or detached heads, which are easily identifiable, can be evaluated using PCM. Therefore, routine morphological abnormalities assessed under phase contrast microscopy do not provide a reliable and significant measure of semen quality, particularly in fresh semen samples from boars. Morphometric dimensions, along with spermatozoa viability and motility, are crucial factors in semen post-copulation selection. Spermatozoa dimensions may correlate with sperm counts (Pinart et al., 1998; Rijsselaere et al., 2004; Wysokińska et al., 2009). The sperm morphometric dimensions assessed through FESEM revealed no significant differences (p > 0.05) between the two breeds. Exotic breed sperm exhibited higher dimensional values than their indigenous counterparts, although this difference was not statistically significant. In the current investigation, the total length (53.16 ± 0.11 vs. 54.33 ± 0.31), head length (9.45 ± 0.05 vs. 10.25 ± 0.24), head width (4.10 ± 0.17 vs. 4.65 ± 0.17), sperm head perimeter (23.38 ± 0.44 vs. 24.23 ± 0.29), sperm head area (39.58 ± 0.10 vs. 40.68 ± 0.23), and tail length (36.03 ± 0.12 vs. 36.30 ± 0.16) of sperm from the LWY boar were found to be greater than those of GH sperm, although no significant difference was observed (p > 0.05). Additionally, the LWY breed exhibited higher sperm motility in both fresh (75.83 ± 1.53 vs. 81.66 ± 1.05) and post-thaw (10.33 ± 1.66 vs. 11.6 ± 1.05) semen compared to the GH breed, which may be linked to the reduced total and tail length dimensions. The findings of this study align with previous reports (Kondracki et al., 2012; Chakurkar et al., 2016), where the total length of the Pietrain breed spermatozoa was recorded at 53.72 ± 1.79 µm, and that of the Duroc breed was 53.20 ± 1.35 µm, both of which fall within the range observed in the current study for GH (53.16 ± 0.11) and LWY (54.33 ± 0.31). Furthermore, Pietrain spermatozoa (44.52 ± 1.72) possess longer tails than Duroc spermatozoa (43.72 ± 1.09), indicating a higher motility, as spermatozoon velocity is influenced by length (Gomendio and Roldan, 1991). Tail length that remains within acceptable limits may be associated with varying energy levels, which could be lower in GH compared to Large White Yorkshire (Larsson, 1986; Wiseman et al., 1998). The sperm head serves as the carrier of genetic information during the fertilization process; thus, its size and shape may significantly influence this process (Kondracki et al., 2012). The sperm head is linked to chromatin structure and integrity, which, in turn, affects male prolificacy (Ostermeier et al., 2001). In the current study, the head dimensions in the indigenous breed were found to be smaller than those of the exotic breed. Previous studies reported that the head length of Pietrain pig sperm was 9.20 ± 0.39, while for Duroc it measured 9.41 ± 0.31. The head width, perimeter, and area of the sperm head were recorded as 4.65 ± 0.25 µm, 23.82 ± 1.25 µm, and 40.47 ± 2.72 µm², respectively, for the Pietrain, and 4.80 ± 0.18 µm, 24.17 ± 0.88 µm, and 40.73 ± 1.18 µm², respectively, for the Duroc breed (Kondracki et al., 2012). The spermatozoa of highly fertile boars possess smaller and shorter heads compared to those of less fertile boars (Hirai et al., 2001). This indicated that GH spermatozoa may exhibit greater fertility than LWY spermatozoa, particularly in the tropical conditions of the Northeast Himalayas. Furthermore, as previously reported (Chakurkar et al., 2016), the head dimensions of indigenous Agonda goan sperm were smaller than those of the exotic LWY counterparts. This confirms that indigenous breeds have shorter tail and head dimensions than exotic breeds, with no significant difference observed (p > 0.05). The length of the midpiece was shorter in the indigenous GH breed (7.68 ± 0.04) compared to the exotic LWY breed (7.78 ± 0.03). However, the width was greater in the indigenous breed (0.65 ± 0.04) than in the exotic breed (0.64 ± 0.03). This indicated that while the number of mitochondria may be higher in the exotic breed, the mitochondrial width is greater in the indigenous breed. This could be related to the spermatozoa’s energy levels from both breeds, which appear to have similar energy levels. The perimeter measurements of the proximal (0.50 ± 0.012 vs. 0.48 ± 0.003) and distal (0.45 ± 0.003 vs. 0.47 ± 0.009) droplets were greater in indigenous sperm than in exotic sperm, with no significant difference (p > 0.05) observed between the two breeds. The larger size of the droplets suggested a higher level of immaturity in the spermatozoa of the GH breed compared to the spermatozoa of the LWY breed. This may have led to a reduced viability of the indigenous GH spermatozoa in both fresh (89.16 ± 2.38 vs. 92.50 ± 1.11) and post-thaw (6.16 ± 1.30 vs. 7.83 ± 1.30) conditions when compared to the LWY exotic breed, as indicated by the findings of the current study. A similar pattern was noted in the acrosomal integrity for both fresh (83.33 ± 1.66 vs. 85.83 ± 0.83) and post-thaw (39.16 ± 2.71 vs. 50.83 ± 2.71) spermatozoa between the indigenous and exotic breeds. To the best of our knowledge, this is the first report on the estimation of deep morphological characteristics in indigenous pigs using SEM. Routine assessment of morphological abnormalities using phase contrast microscopy can only identify superficial or distinctly visible morphological irregularities or structural cryodamage. In contrast, SEM can reveal or differentiate deeper and even subtle morphological variations in the structural elements of spermatozoa. For instance, phase contrast microscopy cannot detect cracks, fragmentations, extrusions, and vesicles within the spermatozoa structure. This issue becomes increasingly challenging in post thaw spermatozoa, where identifying such morphological damages is more complex. Therefore, in the current study, SEM was utilized to examine deeper morphological abnormalities in the spermatozoa of both breeds, focusing on multiple vesicles, compromised plasma membranes, damaged acrosomal crescents, impaired acrosomal membranes, extruded vesicles, and cracks, which are typically not discernible under phase contrast microscopy. Consequently, these abnormalities remain unaccounted for in semen quality assessments, potentially affecting quality evaluations and leading to productivity loss. Understanding and assessing deep morphological abnormalities are crucial during breeding soundness evaluations and subfertility investigations. Investigating deep morphological abnormalities may be particularly significant when combined with marker-based selection and fluorescence-based sperm function tests, ensuring the selection and evaluation of boars in AI programs. In the fresh semen of indigenous and exotic breeds, there was no significant difference (p > 0.05) in misshapen heads or detached heads, except for the presence of multiple vesicles on the head (p < 0.05) among the head abnormalities. This suggests that the occurrence of multiple vesicles on the sperm heads was higher in the indigenous breed than in the exotic pig breeds, which may be correlated with a lower fertilization rate in the Ghungroo breed than in its LYW counterpart. A significant difference (p < 0.05) was noted in the estimated acrosomal abnormalities (excluding damaged acrosomal membranes), including damaged acrosomal crescents, extruded acrosomal vesicles, and cracks on the acrosome, with a greater number of abnormalities observed in the indigenous breed compared with the exotic breed. This finding indicates that the indigenous breed’s acrosome is weaker and more fragile than that of the exotic breed. While no significant difference was found in the bent midpiece, a significant difference (p < 0.05) was observed in the presence of multiple vesicles on the midpiece between the two breeds. This suggested that the midpiece of the spermatozoa of the Ghungroo breed may be weaker than that of the LWY breed. Significant differences (p < 0.05) were also noted in tail abnormalities, including bent, broken, and fragmented tails, as well as multiple vesicles on the tail between the two breeds, indicating that the indigenous breed exhibits more tail abnormalities, which may contribute to lower motility compared with the exotic breed. There was a significant difference (p < 0.05) in the percentage of plasma membrane damage over the head and tail, excluding the midpiece (p > 0.05), indicating greater plasma membrane vulnerability in the indigenous breed than in the exotic breed. The data presented above suggested that the fresh semen spermatozoa of the indigenous breed may possess a structural integrity similar to that of the exotic breed. The findings of the current study (intact acrosome: 83.33 ± 1.66; damaged acrosomal membrane: 3.66 ± 0.88; plasma membrane damaged: 6.66 ± 1.20) closely align with the results reported by Akiko et al. (2001) which indicated that over 90% of fresh spermatozoa in cynomolgus monkeys possessed an intact acrosome, intact plasma membrane, and outer acrosomal membrane. In addition, Prinosilová et al. (2012) found that 98.1% ± 2.95% of spermatozoa had intact acrosomal membranes, and 82.25% had intact plasma membranes in the head and midpiece of fresh semen. In pigs, freezing results in significant cryodamage to post-thaw spermatozoa (Vicente-Carrillo et al., 2016; Gillis et al., 2022). The average motility of frozen-thawed semen was 36.3% and 19.6% for Norwegian Landrace and Duroc boars, respectively (Hofmo and Grevle, 1999). When frozen-thawed boar semen is utilized for AI, rates decrease by 10%–25%, and the litter sizes diminish by 1–3 piglets compared with AI with fresh semen (Johnson et al., 1981; Almlid and Hofmo, 1996). This reduction is attributed to the more severe damage inflicted on boar spermatozoa by cryopreservation compared to other animal species. The use of frozen boar semen has been explored under Indian conditions (Baishya et al., 2016), resulting in moderate fertility rates. The plasma membrane of boar sperm is more susceptible to cold shock than that of other mammals due to a lower cholesterol-to-phospholipids ratio, which causes a transition from a liquid to a gel phase in the membrane during cooling. These biophysical alterations (Yeste, 2015) are triggered by the conversion of liquid water into ice (Rodriguez-Martinez and Wallgren, 2011) and osmotic changes (Hammerstedt et al., 1990). Additionally, during the thawing process, the fluidity and functional characteristics of the sperm membranes are altered (Maxwell and Johnson, 1997), which diminishes their selective permeability and ultimately results in a significant influx of calcium (Bailey et al., 2003). Consequently, the plasma membrane is regarded as the primary site for damage induced by cold exposure. Significant cryodamage was noted in the post-thaw semen of both breeds in the head, acrosome, midpiece, tail, and plasma membrane. The cryodamage observed in the head was significantly (p < 0.05) greater in the indigenous breed, particularly for misshapen heads and the presence of multiple vesicles; however, no significant (p > 0.05) difference in detached heads was observed between the two breeds. This suggested that the tail attachment strength to the head capitulum may be comparable in both indigenous and exotic breeds. A previous study reported head abnormalities, including vacuole-like structures on the heads of spermatozoa (Khan et al., 2015). However, no such abnormalities were observed in either the indigenous or exotic breeds in the current study. A notable distinction (p < 0.05) was identified in the acrosomal damage between the two breeds, including acrosomal crescent, acrosomal membrane, and cracks on the acrosome. Nevertheless, a higher incidence of acrosomal damage was observed in the indigenous breed than in the exotic one. Earlier studies (Khan et al., 2015) indicated that spermatozoa in the Hampshire exotic breed exhibited intact acrosomes (37.88%), whereas the current study revealed the corresponding values for acrosomal damages, such as damaged acrosomal apex (7.33 ± 2.40 vs. 11.66 ± 1.20), compromised acrosomal membrane (18.66 ± 1.45 vs. 13.33 ± 0.88), extruded acrosomal vesicles (20.66 ± 2.33 vs. 16.33 ± 0.88), and cracks on the acrosome (6.66 ± 2.02 vs. 4.33 ± 1.20) for indigenous GH versus exotic LWY breeds. In addition, it was reported that most cells exhibited swollen acrosomes, while a minority displayed loss of acrosomal contents and dissolution of the acrosomal apical ridge. Larsson et al. (1976) documented comparable acrosomal alterations in frozen-thawed spermatozoa. Furthermore, Akiko et al. (2001) reported spermatozoa with normal acrosomes (21.2%), vesiculated acrosomes (36.4%), and swollen acrosomes (28.8%) in post-thaw semen samples of cynomolgus monkey spermatozoa. Based on these findings, the spermatozoa from both indigenous and exotic breeds exhibit similar acrosomal integrity, even under post thaw conditions. In a previous report, the majority of cells exhibited swollen, separating, and ruptured plasma membranes in the head region, with only 31.42% of spermatozoa displaying an intact plasma membrane (Khan et al., 2015). In the current study, the damaged plasma membranes in the head (9.66 ± 3.38 vs. 10.33 ± 2.40), midpiece (4.33 ± 2.02 vs. 3.33 ± 1.85), and tail (20.66 ± 2.84 vs. 14.33 ± 1.20) were documented, indicating a greater integrity of the plasma membrane in both breeds compared to the previous report. This improvement may be attributed to the cryopreservation and thawing protocols employed, as well as the breed of semen samples used (Hampshire/LWY/GH) Moreover, no notable differences were detected in the cryodamage at the midpiece (bent midpiece, multiple vesicles over midpiece), tail (bent tail, broken and fragmented tails, and multiple vesicles on tail), and plasma membrane (head, midpiece) between the two breeds. This suggests that the spermatozoa of the indigenous Ghungoo breed may be equally or insignificantly less resistant than those of the exotic LWY breed. A thinned mitochondrial matrix was noted in frozen-thawed spermatozoa (Watson and Plummer, 1985) in the boar semen of the exotic breed, and similar findings were also reported in the present study. Based on the aforementioned findings, the quality of post thaw semen did not meet acceptable standards in both breeds. The sperm plasma membrane was the primary structure affected (Loomis and Graham, 2008), likely due to changes in lipid-protein complexes during the freezing and thawing processes (Mazur et al., 1972). As it transitioned from a liquid to a gel state, the sperm plasmalemma became more rigid and fragile (De Leeuw et al., 1990), resulting in increased plasmalemma permeability and reduced sperm metabolism (Hammerstedt and Graham, 1992). Consequently, this contributed to the poor viability of post-thaw sperm in both breeds. SEM of post thaw semen samples from both breeds indicated that while some sperm appeared normal, there were various degrees of cellular damage. The post-thaw spermatozoal acrosomes of both breeds were significantly damaged. The extent and percentage of cellular damage varied depending on whether the spermatozoa were fresh or frozen. The damaged acrosomal crescent may have resulted from cryocapacitation during the freezing process. A multivesicular defect was noted in the heads of post thaw spermatozoa in both breeds. Although Waterhouse et al. (2006) suggested that individual male rather than breed differences account for boar sperm quality, the variations in sperm survival among males after freezing and thawing appear to be associated with the levels of long-chain PUFAs present in the plasma membrane following these processes. The differences in membrane lipid composition across species (bull, ram, dog, human) cannot adequately account for the significant disparities in post-thaw survival and fertility observed between species such as bulls and boars, nor can they explain the variations seen between breeds and the male-to-male differences within those breeds (White, 1993; Holt et al., 2005; Gu et al., 2019). Nevertheless, this study found no notable differences in the post-thaw quality of the two breeds. In summary, the current study clearly indicates that the spermatozoa from both breeds may exhibit similarities in their morphometric dimensions and cryoresilience. Comprehensive morphological analyses are essential for evaluating semen quality and should be considered in assessing male fertility. SEM proved to be superior to PCM for assessing semen quality, especially in fresh ejaculates. Therefore, SEM could serve as an innovative tool for evaluating the semen quality of new or younger boars intended for inclusion in AI programs. This approach will facilitate a more effective evaluation of breeding soundness and the early identification of fertility or sub-fertility under field conditions. ConclusionConventional microscopy failed to detect significant differences in fresh semen quality, whereas FESEM enabled the identification of subtle morphological abnormalities and cryodamage, particularly in post-thaw samples. Incorporating SEM-based evaluation into breeding soundness examinations may improve the early detection of subfertility and enhance the selection of suitable sires for AI programs. This may require further validation of the SEM on a larger number of semen samples and animals, along with field or in vivo fertility correlation. The present study also revealed that the indigenous Ghungroo and exotic Large White Yorkshire boars exhibit broadly similar sperm morphometry, structural integrity, and cryoresilience despite some differences in semen volume and concentration. Hence, indigenous breed spermatozoa are at par with their exotic counterparts. In conclusion, the findings of the present study highlight the added value of high-resolution imaging over conventional microscopy for a more accurate assessment of boar semen quality in both indigenous and exotic breeds. AcknowledgmentsThe authors are thankful to the Director, ICAR-NRC on Pig, (Guwahati), Dr. Manish Kumar (Professor), and Md. Sadam Hussian (Research Scholar) and CIF, Indian Institute of Technology, Guwahati, for providing the necessary support for the electron microscopy required for this study. Conflict of interestThe authors have no conflict of interest. FundingThis study was financially supported by NECBH, IIT (Guwahati) Campus, Department of Biotechnology, Govt. of India. A part of the present study was also supported by the National Livestock Mission, DAHD, Govt. of India funded project at ICAR-National Research Centre on Pig, Rani 781 131, Assam. Authors' contributionsConceived of and designed the experiments and revised the manuscript: SK, RI, VKG; analyzed the data and wrote the manuscript: SK, RI, PJD; performed the experiments and collected the samples: SK, LE, PD. All authors have reviewed and approved the manuscript. Data availability statementThe original contributions presented in the study are included in the article/supplementary material; further inquiries can be directed to the corresponding author. ReferencesAkiko, O., Hiroaki, I., Masaru, K., Keiji, T., Yasuhiro, Y. and Tadashi, S. 2001. Cryopreservation induced acrosomal vesiculation in live spermatozoa from cynomolgus monkeys (Macaca fasicularis). Human Reprod. 16(10):2139–47. doi: 10.1093/humrep/16.10.2139 Almlid, T. and Hofmo, P.O. 1996. A brief review of frozen semen application under Norwegian AI service conditions. Reprod. Domestic. Animals. 31(1), 169–173; doi:10.1111/j.1439-0531.1995.tb00021.x Annual Report. 2018. Editors: Banik, S., Mohan N.H., Barman, K., Das, P.J. and Kumar, S. Annual Report (2017-18) for AICRP on Pig and Mega Seed Project on Pig. Published by Director, ICAR-National Research Centre on Pig, Rani, Guwahati, Assam. Bailey, J.L., Bilodeau, J.F. and Cormier, N. 2003. Semen cryopreservation in domestic animals: a damaging and capacitating phenomenon. J. Andrology. 21(1), 1–7. Baishya, S.K., Biswas, R.K., Kadirvel, G., Deka, B.C., Kumar, S. and Ngachan, S.V. 2016. First report on in vivo fertility trial of frozen thawed boar semen in India. Indian J. Anim. Res. 50(2), 181–184; doi:10.18805/ijar.8426 Banik, S., Kumar, S., Das, P.J., Barman, K., Thomas, R., Islam, R., Kumar, S. and Gupta, V.K. 2024. Generation-wise performance evaluation of Ghoongroo pig: an effort to improve the productivity. Indian. J. Anim. Sci. 94(11), 960–965; doi:10.56093/ijans.v94i11.138253 Bezerra, L.G.P., Souza, A.L.P., Silva, H.V.R., Vasconcelos, F.R., Moura, A.D.A.A., Pereira, A.F., Oliveira, M.F.D. and Silva, A.R. 2018. Ultrastructural description of fresh and frozen/thawed sperm derived from collared peccaries (Pecaritajacu Linnaeus, 1,758). Microsc. Res. Tech. 81(11), 1301–1309; doi:10.1002/jemt.23138 Blom, E. 1950. A simple rapid staining method for the differentiation between live and dead sperm cells by means of eosin and nigrosin. Nord. Vet. Med. 2, 58. Brito, L.F.C., Greene, L.M., Kelleman, A., Knobbe, M. and Turner, R. 2011. Effect of method and clinician on stallion sperm morphology evaluation. Theriogenology 76(4), 745–750; doi:10.1016/j.theriogenology.2011.04.007 Chakurkar, E.B., Naik, S.S., Barbuddhe, S.B., Karunakaran, M., Naik, P.K. and Singh, N.P. 2016. Seminal attributes and sperm morphology of AgondaGoan pigs. J. Appl. Anim. Res. 44(1), 130–134; doi:10.1080/09712119.2015.1021807 Chenoweth, P., Brito, L., Peter, A., Waberski, D., Althouse, G., Aurich, C., Luvoni, G., Turner, R., Fraser, N. and Lopate, C. 2024. Andrology laboratory review: evaluation of sperm morphology. Clin. Theriogenology. 16, 10600; doi: 10.58292/CT.v16.10600 Colenbrander, B. and Kemp, B. 1990. Factors influencing semen quality in pigs. J. Reprod. Fertil. Suppl., 40, 105–115. De Leeuw, F.E., Chen, H.C., Colenbrander, B. and Verkleij, A.J. 1990. Cold-induced ultrastructural changes in bull and boar sperm plasma membranes. Cryobiology 27(2), 171–183; doi:10.1016/0011-2240(90)90009-s De Vos, A., Van De Velde, H., Joris, H., Verheyen, G., Devroey, P. and Van Steirteghem, A. 2003. Influence of individual sperm morphology on fertilization, embryo morphology, and pregnancy outcome of intracytoplasmic sperm injection. Fertility Sterility 79(1), 42–48; doi:10.1016/S0015-0282(02)04571-5 Du, J., Shen, J., Wang, Y., Pan, C., Pang, W., Diao, H. and Dong, W. 2016. Boar seminal plasma exosomes maintain sperm function by infiltrating into the sperm membrane. Oncotarget 7(37), 58832; doi:10.18632/oncotarget.11315 Frangež, R., Gider, T. and Kosec, M. 2005. Frequency of boar ejaculate collection and its influence on semen quality, pregnancy rate and litter size. Acta Veterinaria Brno 74(2), 265–273; doi:10.2754/avb200574020265 Gago., Perez-Sanchez., Yeung., Tablado., Cooper. and Soler. 1998. Standardization of sampling and staining methods for the morphometric evaluation of sperm heads in the Cynomolgus monkey (Macaca fascicularis) using computer-assisted image analysis. Int. J. Andrology 21(3), 169–176; doi:10.1111/j.1365-2605.1998.00113.x Gillis, J.D., Holt, W.V., Penfold, L.M., Woad, K.J., Graham, J.K., Watts, J.A., Gardner, D.S. and Yon, L. 2022. Cryo-scanning electron microscopy demonstrates that ice morphology is not associated with the post-thaw survival of domestic boar (Susdomesticus) spermatozoa: a comparison of directional and conventional freezing methods. Cryobiology 108, 10–18; doi:10.1016/j.cryobiol.2022.08.006 Gomendio, M. and Roldan, E.R. 1991. Sperm competition influences sperm size in mammals. Proc. Roy. Soc. London. Ser. B. Biol. Sci. 243(1308), 181–185; doi:10.1098/rspb.1991.0029 Gu, N.H., Zhao, W.L., Wang, G.S. and Sun, F. 2019. Comparative analysis of mammalian sperm ultrastructure reveals relationships between sperm morphology, mitochondrial functions and motility. Reproductive Biol. Endocrinol. 17(1), 66. Hammerstedt, R.H. and Graham, J.K. 1992. Cryopreservation of poultry sperm: the enigma of glycerol. Cryobiology 29(1), 26–38; doi:10.1016/0011-2240(92)90004-l Hammerstedt, R.H., Graham, J.K. and Nolan, J.P. 1990. Cryopreservation of mammalian sperm: what we ask them to survive. J. Andrology. 11(1), 73–88; doi:10.1002/j.1939-4640.1990.tb01583.x Hidalgo, M., Rodríguez, I., Dorado, J. and Soler, C. 2008. Morphometric classification of Spanish thoroughbred stallion sperm heads. Anim. Reprod. Sci. 103(3-4), 374–378; doi:10.1016/j.anireprosci.2007.06.001 Hirai, M., Boersma, A., Hoeflich, A., Wolf, E., Föll, J., Aumüller, R. and Braun, J. 2001. Objectively measured sperm motility and sperm head morphometry in boars (Sus scrofa): relation to fertility and seminal plasma growth factors. J. Andrology. 22(1), 104–110; doi:10.1002/j.1939-4640.2001.tb02159.x Hofmo, P.O. and Grevle, I.S. 1999. Development and commercial utilization of frozen boar semen in Norway. In IV International Conference on Boar Semen Preservation, Beltsville, Maryland. Holt, W.V., Medrano, A., Thurston, L.M. and Watson, P.F. 2005. The significance of cooling rates and animal variability for boar sperm cryopreservation: insights from the cryomicroscope. Theriogenology 63(2), 370–382; doi:10.1016/j.theriogenology.2004.09.018 Johnson, L.A., Aalbers, J.G., Willems, C.M.T. and Sybesma, W. 1981. Use of boar spermatozoa for artificial insemination. I. Fertilizing capacity of fresh and frozen spermatozoa in sows on 36 farms. J. Anim. Sci. 52(5), 1130–1136; doi:10.2527/jas1981.5251130x Kawarasaki, T., Enya, S. and Otake, M. 2022. Sperm Characteristics in Microminipigs. Vivo 36(2), 731–737; doi:10.21873/invivo.12759 Khan, M.H., Nath, K.C., Naskar, S., Deka, B.C. and Kumar, S. 2015. Electron microscopic studies of porcine sperm: changes during freezing and post-thawing. Indian. J. Anim. Sci. 85(7), 723–728; doi:10.56093/ijans.v85i7.50262 Khophloiklang, V., Chanapiwat, P. and Kaeoket, K. 2024. Crude Garden Cress Seed Oil (Lepidium sativum Linn.) Enhances Post-Thawed Boar Sperm Quality. Animals 14(22), 3178; doi:10.3390/ani14223178 Kondracki, S. and Wysokińska, A. 2005. Characterization of sperm abnormalities of boars with regard of age and breed (in Polish). Folia UnivAgricStetinZootech. 243, 97–104. Kondracki, S., Iwanina, M., Wysokińska, A. and Huszno, M. 2012. Comparative analysis of Duroc and Pietrain boar sperm morphology. Acta Veterinaria Brno 81(2), 195–199; doi:10.2754/avb201281020195 Kovács, A. and Foote, R.H. 1992. Viability and acrosome staining of bull, boar and rabbit spermatozoa. Biotech. Histochem. 67(3), 119–124; doi:10.3109/10520299209110020 Kumar, S., Islam, R., Chanda, A., Das, P.J., Banik, S., Barman, K., Pegu, S.R., Rajkhowa, S. and Gupta, V.K. 2024. Biostimulatory effects of boar seminal gel, saliva and semen on sexual behavior of young boars, gilts and sows. Syst. Biol. Reproductive Med. 70(1), 59–72; doi:10.1080/19396368.2024.2314548 Kumaresan, A., Bujarbaruah, K.M., Pathak, K.A., Chhetri, B., Das, S.K., Das, A. and Ahmed, S.K. 2007. Performance of pigs reared under traditional tribal low input production system and chemical composition of non-conventional tropical plants used as pig feed. Livestock. Sci. 107(2-3), 294–298; doi:10.1016/j.livsci.2006.12.007 Larsson, K. 1986. Evaluation of boar semen. Larsson, K., Einarsson, S. and Bane, A. The fertility of boarspermatozoa by two different methods. In VIII International Congress on Animal Reproduction and A.I.,1976 Karkov, Nottingham, United Kingdom, pp 1024–1025. Lonergan. and P. 2018. Historical and futuristic developments in bovine semen technology. Animal 12(s1), s4–s18; doi:10.1017/S175173111800071X Loomis, P.R. and Graham, J.K. 2008. Commercial semen freezing: individual male variation in cryosurvival and the response of stallion sperm to customized freezing protocols. Anim. Reprod. Sci. 105(1-2), 119–128; doi:10.1016/j.anireprosci.2007.11.010 Maroto-Morales, A., García-Álvarez, O., Ramón, M., Martínez-Pastor, F., Fernández-Santos, M.R., Soler, A.J. and Garde, J.J., 2016. Current status and potential of morphometric sperm analysis. Asian J. Androl. 18(6), 863–870. doi: 10.4103/1008-682X.187581 Maxwell, W.M.C. and Johnson, L.A. 1997. Chlortetracycline analysis of boar spermatozoa after incubation, flow cytometric sorting, cooling, or cryopreservation. Mol. Reprod. Develop. Incorporating. Gamete. Res. 46(3), 408–418. Mazur, P., Leibo, S.P. and Chu, E.H. 1972. A two-factor hypothesis of freezing injury: evidence from Chinese hamster tissue-culture cells. Exp. Cell Res. 71(2), 345–355; doi:10.1016/0014-4827(72)90303-5 Nussdorfer, P., Cilenšek, I., Zorn, B. and Petrovič, D. 2018. Adapted methods for scanning electron microscopy (SEM) in assessment of human sperm morphology. Bosnian J. Basic Med. Sci. 18(1), 43; doi:10.17305/bjbms.2017.2173 Ostermeier, G.C., Sargeant, G.A., Yandell, B.S., Evenson, D.P. and Parrish, J.J. 2001. Relationship of bull fertility to sperm nuclear shape. J. Androl. 22(4), 595–603; doi:10.1002/j.1939-4640.2001.tb02219.x Oyeyemi, M.O. and Okediran, B.S. 2007. Testicular parameters and sperm morphology of chinchilla rabbit fed with different planes of soymeal. Int. J. Morphol. 25(1), 139–144; doi:10.4067/S0717-95022007000100021 Oyeyemi, M.O., Oke, A.O., Ajala, O.O. and Idehen, C.O. 2002. Differences in testicular parameters and morphological characteristics of spermatozoa as related to age of West African Dwarf bucks. Trop. J. Anim. Sci. 5(1), 99–107; doi:10.4314/tjas.v5i1.49986 Patra, M.K., Kent, Y., Ngullie, L., Das, R.K. and Deka, B.C. 2015. Comparative performance of Ghungroo and Large Black pig at organized institutional farm conditions. Indian J. Anim. Res. 50(5), 776–781; doi:10.18805/ijar.5714. Patra, M.K., Kent, Y., Rungsung, S., Ngullie, L., Nakhro, R. and Deka, B.C. 2014. Performance appraisal of artificial insemination technique in pig under organized farm and field condition in Nagaland. Indian. Res. J. Extension. Educ. 14(4), 55–60. Peña, F.J., Saravia, F., García-Herreros, M., Núñezmartínez, I., Tapia, J.A., Johannisson, A., Wallgren, M. and Rodríguez-Martínez, H. 2005. Identification of sperm morphometric subpopulations in two different portions of the boar ejaculate and its relation to postthaw quality. J. Andrology. 26(6), 716–723; doi:10.2164/jandrol.05030 Phillips, N.J., Mcgowan, M.R., Johnston, S.D. and Mayer, D.G. 2004. Relationship between thirty post-thaw spermatozoal characteristics and the field fertility of 11 high-use Australian dairy AI sires. Anim. Reprod. Sci. 81(1-2), 47–61; doi:10.1016/j.anireprosci.2003.10.003 Pinart, E., Camps, R., Briz, M.D., Bonet, S. and Egozcue, J. 1998. Unilateral spontaneous abdominal cryptorchidism: structural and ultrastructural study of sperm morphology. Anim. Reprod. Sci. 49(4), 247–268; doi:10.1016/S0378-4320(97)00074-2 Prinosilová, P., Sedlackova, M., Kopecká, V. and Hlavicová, J. 2012. Boar sperm head membrane damage during cryopreservation evaluated by electron microscopy. Res. Pig Breeding 6(2), 58–61. Rijsselaere, T., Van Soom, A., Hoflack, G., Maes, D. and De Kruif, A. 2004. Automated sperm morphometry and morphology analysis of canine semen by the Hamilton-Thorne analyser. Theriogenology 62(7), 1292–1306; doi:10.1016/j.theriogenology.2004.01.005 Rodriguez-Martinez, H. and Wallgren, M. 2011. Advances in boar semen cryopreservation. Vet. Med. Int. 2011(1), 396181; doi:10.4061/2011/396181 Rozeboom, K.J. 2009. Evaluating Boar Semen Quality. In Guidelines for Boar Semen Processing Reviewed by W.L. Flowers, Department of Animal Science, North Carolina St. University and W. Singleton, Department of Animal Science, Purdue University. pp. 1–8. Saravia, F., Núñez-Martínez, I., Morán, J.M., Soler, C., Muriel, A., Rodríguez-Martínez, H. and Peña, F.J. 2007. Differences in boar sperm head shape and dimensions recorded by computer-assisted sperm morphometry are not related to chromatin integrity. Theriogenology 68(2), 196–203; doi:10.1016/j.theriogenology.2007.04.052 Saravia, F., Wallgren, M., Nagy, S., Johannisson, A. and Rodríguez-Martínez, H. 2005. Deep freezing of concentrated boar semen for intra-uterine insemination: effects on sperm viability. Theriogenology 63(5), 1320–1333; doi:10.1016/j.theriogenology.2004.06.012 Sayegh, L., Shah, R., Shmoury, M., Depret-Bixio, L. and Fakih, M. 2024. Sperm morphology by strict criteria does not predict clinical pregnancy rate following intra-uterine insemination. Arab. J. Urology. 22(4), 195–205; doi:10.1080/20905998.2024.2327194 Singh, M., Mollier, R.T. and Sharma R. 2020. Reproductive attributes of Hampshire, Gunghroo, Large Black and Tenyi (local Naga Pig) under intensive management system in sub-tropical condition of Nagaland. Indian J. Anim. Sci. 90(6), 934–936; doi:10.56093/ijans.v90i6.105008 Smital, J. 2009. Effects influencing boar semen. AnimReprod. Sci. 110(3-4), 335–346; doi:10.1016/j.anireprosci.2008.01.024 Smital, J., De Sousa, L.L. and Mohsen, A. 2004. Differences among breeds and manifestation of heterosis in AI boar spermoutput. Anim. Reprod. Sci. 80(1-2), 121–130; doi:10.1016/S0378-4320(03)00142-8 Techakumphu, M., Buranaamnuay, K., Tantasuparuk, W. and Am-In, N., 2013. Improvement of Semen Quality by Feed Supplement and. Success in Artificial Insemination: Quality of Semen and Diagnostics Employed, p.17.DOI: 10.5772/51737 Van Der Horst, G., Maree, L. and Du Plessis, S.S. 2018. Current perspectives of CASA applications in diverse mammalian spermatozoa. Reprod. Fertility. Develop. 30(6), 875–888; doi:10.1071/RD17468 Vicente-Carrillo, A., Ekwall, H., Álvarez-Rodríguez, M. and Rodríguez-Martínez, H. 2016. Membrane stress during thawing elicits redistribution of aquaporin 7 but not of aquaporin 9 in boar spermatozoa. Reprod. Domestic Animals 51(5), 665–679; doi:10.1111/rda.12728 Wang, C. and Swerdloff, R.S. 2014. Limitations of semen analysis as a test of male fertility and anticipated needs from newer tests. Fertility Sterility 102(6), 1502–1507; doi:10.1016/j.fertnstert.2014.10.021 Waterhouse, K.E., Hofmo, P.O., Tverdal, A. and Miller, R.R. 2006. Within and between breed differences in freezing tolerance and plasma membrane fatty acid composition of boar sperm. Reproduction 131(5), 887–894; doi:10.1530/rep.1.01049 Watson P F and Plummer J M. 1985. The responses of boar spermmembranes to cold shock and cooling. Deep Freezing of BoarSemen. Vol. 1, 113–27. (Eds) Johnson L A and Larsson K.Swedish University of Agricultural Sciences, Uppsala,Sweden White, I. 1993. Lipids and calcium uptake of sperm in relation to cold shock and preservation: a review. Reprod. Fertility. Develop. 5(6), 639–658; doi:10.1071/RD9930639 Wiseman, J., Varley, M.A. and Chadwick, J.P. 1998. Progress in pig science. Nottingham, UK: Nottingham University Press. Wysokińska, A., Kondracki, S. and Banaszewska, D. 2009. Morphometrical characteristics of spermatozoa in Polish Landrace boars with regard to the number of spermatozoa in an ejaculate. Reproductive. Biol. 9(3), 271–282. Wysokińska, A., Wójcik, E. and Chłopik, A. 2021. Evaluation of the morphometry of sperm from the epididymides of dogs using different staining methods. Animals 11(1), 227; doi:10.3390/ani11010227 Yániz, J.L., Soler, C. and Santolaria, P. 2015. Computer assisted sperm morphometry in mammals: a review. Anim. Reprod. Sci. 156(1-2), 1–12. doi: 10.1016/j.anireprosci.2015.03.002 Yeste M. 2015. Recent Advances in Boar Sperm Cryopreservation: State of the Art and Current Perspectives. Reprod Domest Anim. 50 (Suppl 2):71-9. doi: 10.1111/rda.12569. PMID: 26174922. Yeste, M. 2016. Sperm cryopreservation update: cryodamage, markers, and factors affecting the sperm freezability in pigs. Theriogenology 85(1), 47–64; doi:10.1016/j.theriogenology.2015.09.047 Zemjanis R. 1970. Collection and Evaluation of Semen. In: Diagnostics and Therapeutic Techniques in Animal Reproduction, 2nd Edition, Williams and Wilson Co., Baltimore, MD, 139–156. Supplementary Materias
Supplemental Fig. 1. Indigenous Ghungroo breed boar.
Supplemental Fig. 2. Exotic large white Yorkshire breed boar. | ||
| How to Cite this Article |
| Pubmed Style Kumar S, Deka P, Islam R, Das PJ, Eranna L, Gupta VK. Assessment of deep morphology and cryodamage in spermatozoa of indigenous and exotic pig breeds from the Northeast Indian Himalayas using scanning electron microscopy (SEM). Open Vet. J.. 2025; 15(12): 6442-6460. doi:10.5455/OVJ.2025.v15.i12.29 Web Style Kumar S, Deka P, Islam R, Das PJ, Eranna L, Gupta VK. Assessment of deep morphology and cryodamage in spermatozoa of indigenous and exotic pig breeds from the Northeast Indian Himalayas using scanning electron microscopy (SEM). https://www.openveterinaryjournal.com/?mno=270321 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.29 AMA (American Medical Association) Style Kumar S, Deka P, Islam R, Das PJ, Eranna L, Gupta VK. Assessment of deep morphology and cryodamage in spermatozoa of indigenous and exotic pig breeds from the Northeast Indian Himalayas using scanning electron microscopy (SEM). Open Vet. J.. 2025; 15(12): 6442-6460. doi:10.5455/OVJ.2025.v15.i12.29 Vancouver/ICMJE Style Kumar S, Deka P, Islam R, Das PJ, Eranna L, Gupta VK. Assessment of deep morphology and cryodamage in spermatozoa of indigenous and exotic pig breeds from the Northeast Indian Himalayas using scanning electron microscopy (SEM). Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6442-6460. doi:10.5455/OVJ.2025.v15.i12.29 Harvard Style Kumar, S., Deka, . P., Islam, . R., Das, . P. J., Eranna, . L. & Gupta, . V. K. (2025) Assessment of deep morphology and cryodamage in spermatozoa of indigenous and exotic pig breeds from the Northeast Indian Himalayas using scanning electron microscopy (SEM). Open Vet. J., 15 (12), 6442-6460. doi:10.5455/OVJ.2025.v15.i12.29 Turabian Style Kumar, Sunil, Prantik Deka, Rafiqul Islam, Pranab Jyoti Das, Lokesha Eranna, and Vivek Kumar Gupta. 2025. Assessment of deep morphology and cryodamage in spermatozoa of indigenous and exotic pig breeds from the Northeast Indian Himalayas using scanning electron microscopy (SEM). Open Veterinary Journal, 15 (12), 6442-6460. doi:10.5455/OVJ.2025.v15.i12.29 Chicago Style Kumar, Sunil, Prantik Deka, Rafiqul Islam, Pranab Jyoti Das, Lokesha Eranna, and Vivek Kumar Gupta. "Assessment of deep morphology and cryodamage in spermatozoa of indigenous and exotic pig breeds from the Northeast Indian Himalayas using scanning electron microscopy (SEM)." Open Veterinary Journal 15 (2025), 6442-6460. doi:10.5455/OVJ.2025.v15.i12.29 MLA (The Modern Language Association) Style Kumar, Sunil, Prantik Deka, Rafiqul Islam, Pranab Jyoti Das, Lokesha Eranna, and Vivek Kumar Gupta. "Assessment of deep morphology and cryodamage in spermatozoa of indigenous and exotic pig breeds from the Northeast Indian Himalayas using scanning electron microscopy (SEM)." Open Veterinary Journal 15.12 (2025), 6442-6460. Print. doi:10.5455/OVJ.2025.v15.i12.29 APA (American Psychological Association) Style Kumar, S., Deka, . P., Islam, . R., Das, . P. J., Eranna, . L. & Gupta, . V. K. (2025) Assessment of deep morphology and cryodamage in spermatozoa of indigenous and exotic pig breeds from the Northeast Indian Himalayas using scanning electron microscopy (SEM). Open Veterinary Journal, 15 (12), 6442-6460. doi:10.5455/OVJ.2025.v15.i12.29 |