| Research Article | ||
Open Vet. J.. 2026; 16(3): 1868-1876 Open Veterinary Journal, (2026), Vol. 16(3): 1868-1876 Research Article Protective effect of young coconut water on MMP -1 levels and collagen fiber thickness in male Wistar rats exposed to UVB light exposureMelinda Angelin1, Siti Thomas Zulaikhah2, Prasetyowati Subchan3, Aswin Rafif Khairullah4, Mustofa Helmi Effendi5, Eka Pramyrtha Hestianah6*, Melda Angelin1, Budiastuti Budiastuti7, Dian Ayu Permatasari5, Bima Putra Pratama8, Saifur Rehman9 and Ainun Ganisia101Master Program of Biomedical Sciences, Faculty of Medicine, Universitas Islam Sultan Agung, Semarang, Indonesia 2Department of Public Health Sciences, Faculty of Medicine, Universitas Islam Sultan Agung, Semarang, Indonesia 3Department of Biomedical Sciences, Faculty of Medicine, Universitas Islam Sultan Agung, Semarang, Indonesia 4Research Center for Veterinary Science, National Research and Innovation Agency (BRIN), Bogor, Indonesia 5Department of Veterinary Public Health, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia 6Department of Veterinary Anatomy, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia 7Study Program of Pharmacy Science, Faculty of Health Science, Universitas Muhammadiyah Surabaya, Surabaya, Indonesia 8Research Center for Process Technology, National Research and Innovation Agency (BRIN), South Tangerang, Indonesia 9Department of Pathobiology, Faculty of Veterinary and Animal Sciences, Gomal University, Indus HWY, Dera Ismail Khan, Pakistan 10Faculty of Health Sciences, Universitas Dr. Soetomo, Surabaya, Indonesia *Corresponding Author: Eka Pramyrtha Hestianah. Division of Veterinary Anatomy, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia. Email:eka-p-h [at] fkh.unair.ac.id Submitted: 13/01/2026 Revised: 15/02/2026 Accepted: 21/02/2026 Published: 31/03/2026 © 2026 Open Veterinary Journal
AbstractBackground: Exposure to ultraviolet B (UVB) light plays a key role in photoaging by increasing oxidative stress, activating inflammatory pathways, and increasing the expression of matrix metalloproteinase-1 (MMP-1), which causes dermal collagen degradation. Young coconut water contains various bioactive compounds with antioxidant activity that can protect the skin from UVB-induced damage. Aim: This study aimed to analyze the effect of the oral administration of young coconut water on MMP-1 levels and collagen fiber thickness in male Wistar rats exposed to UVB light. Methods: A purely experimental study with a post-test only control group design was conducted on 24 male Wistar rats divided into four groups: healthy controls (K1), UVB exposure (K2), UVB + young coconut water 4 ml/200 g BW/day (K3), and UVB + young coconut water 8 ml/200 g BW/day (K4). UVB exposure was administered for 15 days at a cumulative dose of 800 mJ/cm². MMP-1 levels were measured using an enzyme-linked immunosorbent assay method, while collagen fiber thickness was analyzed using Masson’s Trichrome staining. Results: UVB exposure significantly increased MMP-1 levels and decreased collagen fiber thickness (p=0.001). Oral administration of young coconut water significantly reduced MMP-1 levels and increased collagen fiber thickness compared with the untreated UVB group, with the most optimal effect at a dose of 4 ml/200 g body weight/day. Conclusion: Young coconut water has potential as a natural antioxidant agent that can inhibit the increase in MMP-1 and maintain collagen fiber thickness in the skin caused by UVB exposure, thus potentially being used in the prevention of photoaging. Keywords: Young coconut water, UVB, MMP-1, Collagen, Photoaging. IntroductionExposure to ultraviolet radiation, particularly ultraviolet B (UVB) rays with a wavelength of 290–320 nm, is a major environmental factor contributing to skin damage (Al-Sadek and Yusuf, 2024). UVB rays have high photon energy that can penetrate the epidermis to the upper dermis, triggering various biological changes in skin tissue (Gromkowska-Kępka et al., 2021). UVB exposure causes cellular DNA damage, increased reactive oxygen species (ROS) formation, and activation of oxidative stress and inflammatory pathways (Wei et al., 2024). In contrast to UVA exposure, which is more cumulative and chronic, these effects contribute to acute photoaging, characterized by decreased skin elasticity, wrinkles, and extracellular matrix degradation (Brar et al., 2025). Physiologically, the body has a natural defense system against free radicals in the form of endogenous antioxidants that neutralize ROS so that they do not damage cellular components such as membrane lipids, proteins, and DNA (Chandimali et al., 2025). The main endogenous antioxidants include superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx), which synergistically convert superoxide radicals into hydrogen peroxide and then into non-toxic water (Manful et al., 2025). However, excessive UVB exposure can significantly increase ROS production, overwhelming the endogenous antioxidant system’s capacity (Tran et al., 2023). This imbalance is known as oxidative stress and triggers the activation of the Mitogen-Activated Protein Kinase (MAPK) and Nuclear Factor Kappa B (NF-κB) pathways (Guo et al., 2023). MAPK and NF-κB activation play a crucial role in increasing the expression of matrix metalloproteinase-1 (MMP-1), the primary proteolytic enzyme responsible for the degradation of collagen types I and III in the dermis (Feng et al., 2024). Elevated MMP-1 levels decrease the number and thickness of collagen fibers, resulting in brittle skin structure, reduced elasticity, and wrinkle development (Bar and Valiukevičienė, 2025). This condition not only impacts esthetics but also increases the risk of precancerous lesions and skin cancer (Hussein et al., 2025). Chronic exposure to UVB rays is associated with an increased incidence of non-melanoma skin cancers worldwide and decreases skin immune function (immunosuppression) (Anwaar et al., 2025). Efforts to prevent and improve skin aging are generally carried out through the use of topical cosmetics containing chemicals such as retinol or tretinoin (Milosheska and Roškar, 2022). Although effective, the use of these ingredients is often associated with side effects such as skin irritation, peeling, and increased sensitivity to sunlight (Karwal and Mukovozov, 2023). Therefore, the search for alternatives based on natural ingredients that are safe, readily available, and possess antioxidant activity is becoming increasingly important, especially in tropical countries like Indonesia, which are rich in biological resources. Young coconut water, the endosperm fluid from the Cocos nucifera L. fruit, is a potential natural ingredient (Zhang et al., 2024). Young coconut water, particularly from the green wulung coconut variety, is known to contain various bioactive compounds, such as vitamin C, L-arginine, L-methionine, cytokinins, selenium, and microminerals, such as zinc (Zn), manganese (Mn), and copper (Cu) (Shi et al., 2025). These compounds have antioxidant and anti-inflammatory activity by increasing the activity of the enzymes SOD, CAT, and GPx and reducing malondialdehyde (MDA) levels, a marker of lipid peroxidation (Zulaikhah et al., 2022). Furthermore, vitamin C and amino acids in young coconut water play an important role in collagen synthesis by stimulating the expression of collagen-forming genes and increasing the activity of hydroxylase enzymes, which are necessary for the formation of a strong and stable collagen structure (Boo, 2022). Although various benefits of young coconut water have been reported, research specifically examining the effect of oral administration of young coconut water on MMP-1 levels and collagen fiber thickness in skin damaged by UVB exposure remains limited. Therefore, this study aimed to analyze the effect of oral administration of young coconut water on MMP-1 levels and collagen fiber thickness in male Wistar rats exposed to UVB light. The results provide a scientific basis for the use of young coconut water as a natural antioxidant agent in preventing photoaging. Materials and MethodsPlace and time of the researchThis study was conducted at the Integrated Biomedical Laboratories of the Faculty of Medicine, Universitas Islam Sultan Agung (UNISSULA), and the CITO Laboratory. In October 2025, the study lasted for 30 days, covering adaptation, treatment, sampling, and laboratory analysis. Design and type of researchThis was a purely experimental study with a post-test only control group design. This study aimed to analyze the effect of oral administration of young coconut water on MMP-1 levels and collagen fiber thickness in male Wistar rats exposed to UVB light. Experimental animalsThe experimental animals used were 24 male Wistar rats (Rattus norvegicus), approximately 2 months old and weighing 150–200 g, obtained from Semarang City. Rats were housed in standard cages with good ventilation, controlled room temperature, and a 12-hours light–dark cycle. They were fed a standard AD-II diet and drinking water ad libitum (du Sert et al., 2020). All rats underwent a 7-day adaptation period to acclimate to the laboratory environment before treatment. Group division and treatmentAfter the adaptation period, the mice were randomly divided (simple random sampling) into four groups, each consisting of 6 mice, namely:
All treatments were administered for 15 consecutive days. Giving young coconuts waterYoung coconut water was obtained from 5–7-month-old green wulung coconuts (Cocos nucifera var. viridis). Coconut water was collected daily from freshly opened coconuts to maintain nutritional stability. Orally administered using a tube according to the dosage for each treatment group for 15 days (Shi et al., 2025). Based on published literature, young coconut water contains bioactive compounds such as vitamin C (approximately 2.4–5.0 mg/100 ml), L-arginine (approximately 30–50 mg/100 ml), L-methionine in trace amounts, cytokinins, and essential micronutrients including potassium, zinc, manganese, copper, and selenium (Zulaikhah, 2019; Zhang et al., 2024; Shi et al., 2025). No additional laboratory analysis was performed to quantify the exact biochemical composition of the coconut water batch used in the present study. Coconuts from the same cultivar (green wulung) and similar maturity stage (5–7 months) were used to minimize compositional variability. UVB exposureGroups K2, K3, and K4 were exposed to UVB using a 20-watt UVB TL lamp with a wavelength of 312 nm and an irradiation distance of 20 cm. The exposure dose was 1 minimal erythema dose (MED) or 100 mJ/cm² for 10 minutes, carried out every 2 days on days 1, 3, 5, 7, 9, 11, 13, and 15, so that the total cumulative dose reached 800 mJ/cm² (Jin et al., 2010). On non-irradiation days (days 2, 4, 6, 8, 10, 12, and 14), the animals were maintained under standard housing conditions without UVB exposure and handled similarly to irradiation days to minimize handling bias. No sham exposure procedure was performed. The UVB dose was calculated based on the lamp output specifications, irradiation distance (20 cm), and exposure duration, resulting in a cumulative dose of 800 mJ/cm² over the treatment period. SamplingOn day 16 (24 hours after the last UVB exposure), all mice were anesthetized intraperitoneally using 100 mg/kg BW of ketamine until euthanasia was achieved. Approximately 0.5 ml of blood was drawn through the orbital sinus to determine MMP-1 levels. Subsequently, dorsal skin tissue was cleaned of hair and taken in sections measuring approximately 1 × 1 cm² for histological analysis (Li et al., 2025). Examination of MMP-1 levelBlood samples were centrifuged for 15 min to obtain serum. MMP-1 levels were measured using an enzyme-linked immunosorbent assay (ELISA) according to the manufacturer’s protocol (Elabscience). The results are expressed in pg/mL (Aydin et al., 2025). Check the thickness of the collagen fibersSkin tissue was fixed in 10% neutral buffered formalin for 24–48 hours and then processed through dehydration, clearing, infiltration, paraffin embedding, and sectioning using a microtome at a thickness of 4–5 µm. The slides were stained using Masson’s Trichrome to visualize collagen (Baidoo et al., 2023). The thickness of the collagen fiber was measured using a light microscope at 400× magnification, calibrated using an ocular and stage micrometer. Five random fields of view were selected from each dermis slide, and the average collagen fiber thickness was expressed in µm (Woessner et al., 2019). Statistical analysisStatistical analysis was performed using SPSS software version 29 (IBM Corp., Armonk, NY). Data were first tested for normality using the Shapiro–Wilk test and for homogeneity of variance using Levene’s test. If data were normally distributed and homogeneous, comparisons among groups were analyzed using one-way analysis of variance followed by the least significant difference post hoc test. If the assumptions of normality were not met, the non-parametric Kruskal–Wallis test was used, followed by the Mann–Whitney U test for pairwise comparisons. Data are presented as mean ± SD. A p-value < 0.05 was considered statistically significant. Ethical approvalThis research was approved by the Medical/Health Research Bioethics Commission, Faculty of Medicine, Universitas Islam Sultan Agung (Certificate No. 598/XI/2025/Komisi Bioetik). ResultsThis study evaluated the effect of the oral administration of young coconut water on MMP-1 levels and collagen fiber thickness in male Wistar rats exposed to UVB light. Twenty-four rats were divided into four groups (K1–K4), each consisting of six rats. Table 1 presents the mean MMP-1 levels and collagen fiber thickness in each group. Table 1. Mean MMP-1 levels and collagen fiber thickness in each group.
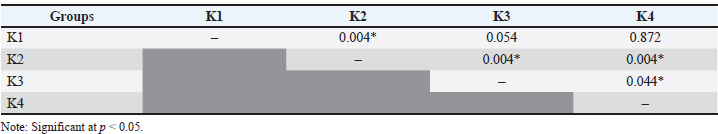
The results showed that the group of rats exposed only to UVB light (K2) had the highest mean MMP-1 levels, at (6.615 ± 0.162 pg/ml). Meanwhile, the lowest mean MMP-1 levels were found in the treatment group receiving young coconut water at a dose of 4 ml/200 g body weight/day (K3), at 4.435 ± 0.278 pg/ml. The healthy control group (K1) and the 8 ml/200 g BW/day treatment group (K4) each showed mean MMP-1 levels of 4.926 ± 0.072 pg/ml and 4.955 ± 0.028 pg/ml, respectively. The Shapiro–Wilk normality test showed that MMP-1 levels in groups K1 and K4 were normally distributed (p > 0.05), whereas those in groups K2 and K3 were not normally distributed (p < 0.05). A homogeneity of the variance test using Levene’s test showed data homogeneity (p=0.081). Therefore, the analysis was continued using the nonparametric Kruskal–Wallis test. The Kruskal–Wallis test revealed a significant difference in MMP-1 levels between the four groups (p=0.001). A follow-up Mann–Whitney test revealed significant differences between K1 and K2, K2 and K3, K2 and K4, and between K3 and K4 (p < 0.05). However, no significant differences were found between K1 and K3 or between K1 and K4 (p > 0.05). Table 2 presents a summary of the comparisons between groups. Table 2. Comparison of MMP-1 Levels between Groups (Mann–Whitney U test).
The results of the collagen fiber thickness analysis showed that the K3 treatment group had the highest mean collagen fiber thickness, at 35.866 ± 6.549 µm, followed by the K4 group (30.700 ± 8.295 µm), healthy control group K1 (27.700 ± 5.370 µm), and untreated UVB-exposed group K2, which showed the lowest mean thickness (18.100 ± 4.684 µm). Representative Masson’s Trichrome-stained skin sections from each experimental group (K1–K4) are presented in Figure 1. Consistent with the quantitative findings, the UVB-only group (K2) exhibited visibly reduced collagen density and disrupted fiber organization, whereas the treatment groups, particularly K3, exhibited thicker and more densely arranged collagen fibers.
Fig. 1. Representative dermal sections stained with Masson’s Trichrome from the experimental groups (K1–K4). The Shapiro–Wilk normality test showed that all groups had a normal distribution of data (p > 0.05). The homogeneity of the variance test using Levene’s test showed data homogeneity (p=0.402). Based on these results, one-way ANOVA was used for statistical analysis. The results of one-way ANOVA showed a statistically significant difference between the four groups (p=0.001). Further post hoc LSD tests revealed significant differences between K1 and K2, K1 and K3, K2 and K3, and K2 and K4 (p < 0.05). However, no significant differences were found between K1 and K4 or between K3 and K4 (p > 0.05). Table 3 presents the results of the intergroup comparison. Table 3. Comparison of collagen fiber thickness between groups (post hoc LSD).
DiscussionThe results of this study indicate that UVB exposure significantly increased MMP-1 levels and decreased collagen fiber thickness in the skin of male Wistar rats. Conversely, the oral administration of young coconut water reduced MMP-1 levels and increased collagen fiber thickness in the treatment group, with the most optimal effect observed at a dose of 4 ml/200 g body weight/day. These findings confirm the role of UVB light as a primary trigger of photoaging and the potential of young coconut water as a natural photoprotective agent. The increase in MMP-1 levels in the UVB-only group (K2) is consistent with the widely reported pathophysiological mechanisms of photoaging (Moon et al., 2008). UVB radiation triggers the formation of large amounts of ROS in skin tissue, which exceeds the capacity of endogenous antioxidant systems such as SOD, CAT, and GPx (Dunaway et al., 2018). ROS accumulation acts as a second messenger that activates intracellular signaling pathways, particularly mitogen-activated protein kinase (MAPK) and nuclear factor kappa B (NF-κB) (Gunaseelan et al., 2017). Activation of this pathway increases the expression of the transcription factor Activator Protein-1 (AP-1), which plays a direct role in the induction of MMP-1 gene transcription (Kim et al., 2018). MMP-1 is the primary collagenase responsible for the degradation of collagen types I and III, so its increased levels contribute to decreased collagen fiber thickness and weakened dermal structure (Brennan et al., 2003). In addition to increasing collagen degradation, UVB exposure also inhibits new collagen synthesis by suppressing the Transforming Growth Factor-β (TGF-β)/Smad pathway (Geng et al., 2021). AP-1 activation due to oxidative stress is known to decrease the expression of the TGF-β receptor type II (TβRII) and increase the expression of Smad7, which functions as an inhibitor of Smad2/3 phosphorylation (He et al., 2014). Consequently, the transcription of collagen-constituting genes, such as COL1A1 and COL3A1, is significantly reduced. This mechanism explains why group K2 had the highest MMP-1 levels and the lowest collagen fiber thickness compared to the other groups (Zhu et al., 2025). Oral administration of young coconut water to groups K3 and K4 suppressed the increase in MMP-1 levels caused by UVB exposure. This effect is thought to be related to the bioactive compounds in young coconut water, particularly vitamin C, L-arginine, L-methionine, cytokinins, selenium, and trace minerals such as Zn, Mn, and Cu (Zulaikhah, 2019). Vitamin C is a powerful antioxidant that plays a role in neutralizing ROS by donating electrons, thereby reducing oxidative stress and inhibiting the activation of the MAPK and NF-κB pathways (Oudemans-van Straaten et al., 2014). By reducing AP-1 and NF-κB activation, MMP-1 expression can be suppressed, as reflected in the lower MMP-1 levels in the treated group compared to the untreated UVB group (Lee et al., 2024). In addition to its antioxidant role, vitamin C is also an essential cofactor for the prolyl and lysyl hydroxylase enzymes, which are involved in the proline and lysine hydroxylation stages of collagen synthesis (Alberts et al., 2025). This process is essential for the formation of a strong and stable triple helix collagen structure. Therefore, the increased collagen fiber thickness in the K3 and K4 groups reflects not only reduced collagen degradation but also increased new collagen synthesis (Singh et al., 2023). L-arginine in young coconut water also contributes to the protective effects observed. This amino acid is a precursor of nitric oxide (NO), which can increase the activity of endogenous antioxidant enzymes such as SOD and GPx at physiological concentrations (Wu et al., 2021). ROS accumulation can be suppressed by increasing endogenous antioxidant capacity, thereby reducing the activation of the MAPK–AP-1 and NF-κB pathways (Behl et al., 2021). Furthermore, L-arginine stimulates the TGF-β pathway, which plays a role in regulating collagen synthesis by dermal fibroblasts (Hamanaka et al., 2025). These findings are consistent with those of previous studies that reported increased skin elasticity and the number of collagen and elastin fibers after L-arginine supplementation (Miler et al., 2024). The L-methionine and selenium contained in young coconut water also support antioxidant mechanisms by increasing GSH synthesis and GPx activity (Li et al., 2024). GSH is a major intracellular antioxidant that plays a crucial role in detoxifying reactive oxygen species (ROS) and lipid peroxides (Dawi et al., 2024). Reducing oxidative stress can suppress inflammatory pathway activation and MMP-1 expression (Lee et al., 2024). Micronutrients such as Zn, Mn, and Cu function as cofactors for antioxidant enzymes (Cu/Zn-SOD and Mn-SOD), thereby strengthening the overall antioxidant defense system of the skin (Darenskaya et al., 2025). Interestingly, the results of this study demonstrated that a dose of 4 mL/200 g BW/day (K3) produced a more pronounced protective effect than 8 mL/200 g BW/day (K4), both in reducing MMP-1 levels and increasing collagen fiber thickness. This indicates a biphasic dose–response relationship rather than a simple linear correlation between dose and biological effect. Such responses are consistent with the concept of hormesis, in which low-to-moderate doses of bioactive compounds exert beneficial adaptive effects, whereas higher doses may attenuate or even counteract these effects (Jodynis-Liebert and Kujawska, 2020). At moderate concentrations, the antioxidant components of young coconut water, particularly vitamin C, amino acids, and trace minerals, may effectively enhance endogenous antioxidant defense systems, suppress ROS-mediated MAPK/AP-1 signaling activation, and reduce MMP-1 expression. However, several biological mechanisms may limit this protective effect at higher doses. First, the antioxidant pathways may become functionally saturated, reducing the additional benefit despite increased intake. Second, cellular transport systems, such as sodium-dependent vitamin C transporters (SVCTs), exhibit saturation kinetics, potentially limiting the intracellular accumulation of ascorbic acid at higher concentrations. As a result, increasing the administered dose may not proportionally increase cellular antioxidant capacity. Moreover, high concentrations of certain antioxidants, including vitamin C, exert mild pro-oxidant effects under specific conditions, particularly in the presence of transition metals, potentially leading to redox imbalance rather than protection (Peper, 2009). Excessive antioxidant supplementation may also trigger feedback regulatory mechanisms that downregulate endogenous antioxidant enzyme activity, thereby diminishing overall redox homeostasis. Collectively, these mechanisms may explain why the higher dose did not provide superior protection compared with the moderate dose observed in this study. This study demonstrates that young coconut water has potential as a natural anti-photoaging agent through mechanisms such as reducing oxidative stress, inhibiting the MAPK and NF-κB pathways, suppressing MMP-1 expression, and increasing dermal collagen synthesis and thickness. However, this study has several limitations that should be acknowledged. First, MMP-1 levels were measured only in serum using an enzyme-linked immunosorbent assay, which may not fully reflect local dermal MMP-1 expression or activity within skin tissue. Previous studies have shown that UVB-induced MMP-1 expression occurs predominantly in dermal fibroblasts and keratinocytes at the tissue level (Brennan et al., 2003; Choi et al., 2020). Therefore, the absence of immunohistochemical (IHC) analysis limits our ability to directly evaluate the localization and intensity of MMP-1 expression within the dermis. Second, not only MMP-1 but also its endogenous inhibitors, particularly TIM-1, regulate extracellular matrix turnover. The balance between MMPs and TIMPs plays a critical role in collagen homeostasis (Cabral-Pacheco et al., 2020). The lack of TIMP-1 measurement in this study prevents a comprehensive assessment of collagen degradation–inhibition dynamics. Third, this study did not quantitatively analyze the composition of young coconut water. Natural products such as coconut water exhibit variability in bioactive compounds depending on cultivar, maturity stage, and environmental factors (Zhang et al., 2024; Shi et al., 2025). Therefore, the absence of compositional standardization may limit reproducibility and comparability with other studies. Fourth, the healthy control group (K1) did not undergo sham gavage procedures, which may introduce potential procedural stress bias compared with treatment groups receiving repeated oral administration. Repeated handling and gavage reportedly influence physiological stress responses in rodents (du Sert et al., 2020; Li et al., 2025). Finally, the relatively short UVB exposure duration represents an acute photodamage model and may not fully mimic chronic photoaging conditions. Chronic UV exposure induces cumulative structural and molecular alterations in dermal collagen (Gromkowska-Kępka et al., 2021; Feng et al., 2024). To strengthen the mechanistic understanding, future studies should include longer exposure periods, tissue-level molecular analyses, and broader oxidative stress and inflammatory markers. Previous veterinary studies in Indonesia have also emphasized the importance of molecular and morphological investigations to better understand animal health problems and support the development of effective disease prevention strategies in livestock and experimental animal models (Kurniawan et al., 2025). ConclusionExposure to UVB light significantly increases MMP-1 levels and decreases collagen fiber thickness in the skin of male Wistar rats. Oral administration of young coconut water can suppress the increase in MMP-1 and significantly increase collagen fiber thickness, with the most optimal effect at a dose of 4 ml/200 g body weight per day. These findings indicate that young coconut water has the potential to be a natural antioxidant agent in preventing skin damage due to UVB exposure and photoaging processes, although further research is still needed to strengthen its clinical application. AcknowledgmentsThe authors would like to thank Universitas Islam Sultan Agung and Universitas Airlangga. Conflict of interestThe authors declare no conflict of interest. FundingThe author funded this research. Authors' contributionsMA, PS, and STZ: Conceived, designed, and coordinated the study. ARK and MHE: designed data collection tools, supervised field sample and data collection, and performed laboratory work and data entry. EPH and BB: Validation, supervision, and formal analysis of data. DAP and SR: Contributed reagents, materials, and analysis tools. MA, AG, and BPP: Carried out the statistical analysis and interpretation and participated in manuscript preparation. All authors have read, reviewed, and approved the final version of the manuscript. Data availabilityAll data are available in the revised manuscript. ReferencesAlberts, A., Moldoveanu, E.T., Niculescu, A.G. and Grumezescu, A.M. 2025. Vitamin C: a comprehensive review of its role in health, disease prevention, and therapeutic potential. Molecules 30(3), 748. Al-Sadek, T. and Yusuf, N. 2024. Ultraviolet radiation: biological and medical implications. Curr. Issues Mol. Biol. 46(3), 1924–1942. Anwaar, S., Ashraf, A., Jahfali, S., Yunis, J., Cruz, J.L.G. and Wells, J.W. 2025. Immunomodulation of UVB-induced regulatory T cells prevents the establishment of squamous cell carcinoma. J. Immunother. Cancer 13(12), 13118. Aydin, S., Emre, E., Ugur, K., Aydin, M.A., Sahin, I., Cinar, V. and Akbulut, T. 2025. An overview of ELISA: a review and update on best laboratory practices for quantifying peptides and proteins in biological fluids. J. Int. Med. Res. 53(2), 3000605251315913. Baidoo, N., Sanger, G.J. and Belai, A. 2023. Histochemical and biochemical analysis of collagen content in formalin-fixed, paraffin embedded colonic samples. MethodsX 11(1), 102416. Bar, O. and Valiukevičienė, S. 2025. Skin aging and type I collagen: a systematic review of interventions with potential collagen-related effects. Cosmetics 12(4), 129. Behl, T., Upadhyay, T., Singh, S., Chigurupati, S., Alsubayiel, A.M., Mani, V., Vargas-De-La-Cruz, C., Uivarosan, D., Bustea, C., Sava, C., Stoicescu, M., Radu, A.F. and Bungau, S.G. 2021. Polyphenols targeting MAPK mediated oxidative stress and inflammation in rheumatoid arthritis. Molecules 26(21), 6570. Boo, Y.C. 2022. Ascorbic acid (vitamin C) as a cosmeceutical to increase dermal collagen for skin antiaging purposes: emerging combination therapies. Antioxidants 11(9), 1663. Brar, G., Dhaliwal, A., Brar, A.S., Sreedevi, M., Ahmadi, Y., Irfan, M., Golbari, R., Zumárraga, D., Yateem, D., Lysak, Y. and Abarca-Pineda, Y.A. 2025. A comprehensive review of the role of UV radiation in photoaging processes between different types of skin. Cureus 17(3), e81109. Brennan, M., Bhatti, H., Nerusu, K.C., Bhagavathula, N., Kang, S., Fisher, G.J., Varani, J. and Voorhees, J.J. 2003. Matrix metalloproteinase-1 is the major collagenolytic enzyme responsible for collagen damage in UV-irradiated human skin. Photochem. Photobiol. 78(1), 43–48. Cabral-Pacheco, G.A., Garza-Veloz, I., Castruita-de La Rosa, C., Ramirez-Acuña, J.M., Perez-Romero, B.A., Guerrero-Rodriguez, J.F., Martinez-Avila, N. and Martinez-Fierro, M.L. 2020. The roles of matrix metalloproteinases and their inhibitors in human diseases. Int. J. Mol. Sci. 21(24), 9739. Chandimali, N., Bak, S.G., Park, E.H., Lim, H.J., Won, Y.S., Kim, E.K., Park, S.I. and Lee, S.J. 2025. Free radicals and their impact on health and antioxidant defenses: a review. Cell Death Discovery 11(1), 19. Choi, H.J., Alam, M.B., Baek, M.E., Kwon, Y.G., Lim, J.Y. and Lee, S.H. 2020. Protection against UVB-induced photoaging by Nypa fruticans via inhibition of MAPK/AP-1/MMP-1 signaling. Oxid. Med. Cell. Longev. 2020(1), 2905362. Darenskaya, M., Cloete, K.J., Rychkova, L., Kolesnikov, S., Prokhorova, Z., Semenova, N., Yuzvak, N. and Kolesnikova, L. 2025. Oxidative stress, antioxidant cofactor micronutrients, and cognitive outcomes in childhood obesity: mechanisms, evidence, and therapeutic opportunities. Int. J. Mol. Sci. 26(24), 12012. Dawi, J., Misakyan, Y., Affa, S., Kades, S., Narasimhan, A., Hajjar, F., Besser, M., Tumanyan, K. and Venketaraman, V. 2024. Oxidative stress, glutathione insufficiency, and inflammatory pathways in type 2 diabetes mellitus: implications for therapeutic interventions. Biomedicines 13(1), 18. Du Sert, N.P., Hurst, V., Ahluwalia, A., Alam, S., Avey, M.T., Baker, M., Browne, W.J., Clark, A., Cuthill, I.C., Dirnagl, U., Emerson, M., Garner, P., Holgate, S.T., Howells, D.W., Karp, N.A., Lazic, S.E., Lidster, K., MacCallum, C.J., Macleod, M., Pearl, E.J., Petersen, O.H., Rawle, F., Reynolds, P., Rooney, K., Sena, E.S., Silberberg, S.D., Steckler, T. and Würbel, H. 2020. The ARRIVE guidelines 2.0: updated guidelines for reporting animal research. PLos Biol. 18(7), e3000410. Dunaway, S., Odin, R., Zhou, L., Ji, L., Zhang, Y. and Kadekaro, A.L. 2018. Natural antioxidants: multiple mechanisms to protect skin from solar radiation. Front. Pharmacol. 9(1), 392. Feng, C., Chen, X., Yin, X., Jiang, Y. and Zhao, C. 2024. Matrix metalloproteinases on skin photoaging. J. Cosmet. Dermatol. 23(12), 3847–3862. Geng, R., Kang, S.G., Huang, K. and Tong, T. 2021. Boosting the photoaged skin: the potential role of dietary components. Nutrients 13(5), 1691. Gromkowska‐Kępka, K.J., Puścion‐Jakubik, A., Markiewicz‐Żukowska, R. and Socha, K. 2021. The impact of ultraviolet radiation on skin photoaging: review of in vitro studies. J. Cosmet. Dermatol. 20(11), 3427–3431. Gunaseelan, S., Balupillai, A., Govindasamy, K., Ramasamy, K., Muthusamy, G., Shanmugam, M., Thangaiyan, R., Robert, B.M., Prasad Nagarajan, R., Ponniresan, V.K. and Rathinaraj, P. 2017. Linalool prevents oxidative stress activated protein kinases in single UVB-exposed human skin cells. PLos One 12(5), e0176699. Guo, C., Ning, X., Zhang, J., Zhang, C., Wang, J., Su, L., Han, J. and Ma, N. 2023. Ultraviolet B radiation induces oxidative stress and apoptosis in human lens epithelium cells by activating NF-κB signaling to down-regulate sodium vitamin C transporter 2 (SVCT2) expression. Cell. Cycle. 22(12), 1450–1462. Hamanaka, R.B., Shin, K.W.D., Atalay, M.V., Cetin-Atalay, R., Shah, H., Houpy Szafran, J.C., Woods, P.S., Meliton, A.Y., Shamaa, O.R., Tian, Y., Cho, T. and Mutlu, G.M. 2025. Arginine promotes the activation of human lung fibroblasts independent of its metabolism. Biochem. J. 482(12), 823–838. He, T., Quan, T., Shao, Y., Voorhees, J.J. and Fisher, G.J. 2014. Oxidative exposure impairs TGF-β pathway via reduction of type II receptor and SMAD3 in human skin fibroblasts. Age 36(3), 9623. Hussein, R.S., Bin Dayel, S., Abahussein, O. and El‐Sherbiny, A.A. 2025. Influences on skin and intrinsic aging: biological, environmental, and therapeutic insights. J. Cosmet. Dermatol. 24(2), e16688. Jin, X.J., Kim, E.J., Oh, I.K., Kim, Y.K., Park, C.H. and Chung, J.H. 2010. Prevention of UV-induced skin damages by 11,14,17-eicosatrienoic acid in hairless mice in vivo. J. Korean Med. Sci. 25(6), 930–937. Jodynis-Liebert, J. and Kujawska, M. 2020. Biphasic dose-response induced by phytochemicals: experimental evidence. J. Clin. Med. 9(3), 718. Karwal, K. and Mukovozov, I. 2023. Topical AHA in dermatology: formulations, mechanisms of action, efficacy, and future perspectives. Cosmetics 10(5), 131. Kim, J.M., Kim, S.Y., Noh, E.M., Song, H.K., Lee, G.S., Kwon, K.B. and Lee, Y.R. 2018. Reversine inhibits MMP-1 and MMP-3 expressions by suppressing ROS/MAPK/AP-1 activation in UV-stimulated human keratinocytes and dermal fibroblasts. Exp. Dermatol. 27(3), 298–301. Kurniawan, M.A., Suwanti, L.T., Mufasirin, M., Suprihati, E., Hastutiek, P., Kusnoto, K., Ansori, A.N.M., Puspitasari, Y., Khairullah, A.R., Moses, I.B., Pratama, B.P. and Riwu, K.H.P. 2025. Morphometric and molecular identification of Eimeria bovis and Eimeria zuernii on beef cattle in Lamongan, East Java, Indonesia. J. Medik Vet. 8(1), 153–166. Lee, J.S., Min, J.W., Gye, S.B., Kim, Y.W., Kang, H.C., Choi, Y.S., Seo, W.S. and Lee, B.Y. 2024. Suppression of UVB-induced MMP-1 expression in human skin fibroblasts using lysate of Lactobacillus iners derived from Korean women's skin in their twenties. Curr. Issues. Mol. Biol. 46(1), 513–526. Li, X., Li, J., Lu, K., Li, X., Song, K., Wang, L. and Zhang, C. 2024. Effect of dietary supplementation of selenium-L-methionine on growth, antioxidant capacity and resistance to nitrite stress of spotted seabass (Lateolabrax maculatus) under two rearing water temperatures. Anim. Nutr. 19(1), 166–179. Li, Z., Lobue, A., Heuser, S.K., Li, J., Engelhardt, E., Papapetropoulos, A., Patel, H.H., Lilley, E., Ferdinandy, P., Schulz, R. and Cortese‐Krott, M.M. 2025. Best practices for blood collection and anaesthesia in mice: selection, application and reporting. Br. J. Pharmacol. 182(11), 2337–2353. Manful, C.F., Fordjour, E., Subramaniam, D., Sey, A.A., Abbey, L. and Thomas, R. 2025. Antioxidants and reactive oxygen species: shaping human health and disease outcomes. Int. J. Mol. Sci. 26(15), 7520. Miler, I., Rabasovic, M.D., Askrabic, S., Stylianou, A., Korac, B. and Korac, A. 2024. Short-term L-arginine treatment mitigates early damage of dermal collagen induced by diabetes. Bioengineering 11(4), 407. Milosheska, D. and Roškar, R. 2022. Use of retinoids in topical antiaging treatments: a focused review of clinical evidence for conventional and nanoformulations. Adv. Ther. 39(12), 5351–5375. Moon, H.J., Lee, S.R., Shim, S.N., Jeong, S.H., Stonik, V.A., Rasskazov, V.A., Zvyagintseva, T. and Lee, Y.H. 2008. Fucoidan inhibits UVB-induced MMP-1 expression in human skin fibroblasts. Biol. Pharm. Bull. 31(2), 284–289. Oudemans-van Straaten, H.M., Man, A.M.S.D. and De Waard, M.C. 2014. Vitamin C revisited. Crit. Care. 18(1), 460. Peper, A. 2009. Aspects of the relationship between drug dose and drug effect. Dose. Response. 7(2), 172–192. Shi, S., Wang, W., Wang, F., Yang, P., Yang, H., He, X. and Liao, X. 2025. Research progress in coconut water: a review of nutritional composition, biological activities, and novel processing technologies. Foods 14(9), 1503. Singh, D., Rai, V. and Agrawal, D.K. 2023. Regulation of Collagen I and Collagen III in tissue injury and regeneration. Cardiol. Cardiovasc. Med. 7(1), 5–16. Tran, J.T., Diaz, M.J., Rodriguez, D., Kleinberg, G., Aflatooni, S., Palreddy, S., Abdi, P., Taneja, K., Batchu, S. and Forouzandeh, M. 2023. Evidence-based utility of adjunct antioxidant supplementation for the prevention and treatment of dermatologic diseases: a comprehensive systematic review. Antioxidants 12(8), 1503. Wei, M., He, X., Liu, N. and Deng, H. 2024. Role of reactive oxygen species in ultraviolet-induced photodamage of the skin. Cell. Div. 19(1), 1. Woessner, A.E., McGee, J.D., Jones, J.D. and Quinn, K.P. 2019. Characterizing differences in the collagen fiber organization of skin wounds using quantitative polarized light imaging. Wound. Repair. Regen. 27(6), 711–714. Wu, G., Meininger, C.J., Mcneal, C.J., Bazer, F.W. and Rhoads, J.M. 2021. Role of L-arginine in nitric oxide synthesis and health in humans. Adv. Exp. Med. Biol. 1332(1), 167–187. Zhang, Y., Kan, J., Liu, X., Song, F., Zhu, K., Li, N. and Zhang, Y. 2024. Chemical components, nutritional value, volatile organic compounds and biological activities in vitro of coconut (Cocos nucifera L.) water with different maturities. Foods 13(6), 863. Zhu, F., Qu, L., Xu, R., Yuan, Y., Zhang, S. and Chen, Y. 2025. Synergistic anti-photoaging and anti-inflammatory effects of Eucommia ulmoides, Styphnolobium japonicum, and Portulaca oleracea extracts via TGF-β/Smad/IL-17 pathway. Chem. Biol. Technol. Agric. 12(1), 139. Zulaikhah, S.T. 2019. Health benefits of tender coconut water (TCW). Int. J. Pharm. Sci. Res. 10(2), 474–480. Zulaikhah, S.T., Nurul Aini, H.F., Rini, A.S., Abiyyu, B.H., Dewi, E.A.T. and Pratama, A.A. 2022. Tender coconut water (Cocos nucifera L.) can increase antioxidant enzymes and decrease MDA levels: experimental study on cigarette smoke-exposed rats. Pharmacognosy J. 14(5), 469–476. | ||
| How to Cite this Article |
| Pubmed Style Angelin M, Zulaikhah ST, Subchan P, Khairullah AR, Effendi MH, Hestianah EP, Angelin M, Budiastuti B, Permatasari DA, Pratama BP, Rehman S, Ganisia A. Protective effect of young coconut water on MMP -1 levels and collagen fiber thickness in male Wistar rats exposed to UVB light exposure. Open Vet. J.. 2026; 16(3): 1868-1876. doi:10.5455/OVJ.2026.v16.i3.42 Web Style Angelin M, Zulaikhah ST, Subchan P, Khairullah AR, Effendi MH, Hestianah EP, Angelin M, Budiastuti B, Permatasari DA, Pratama BP, Rehman S, Ganisia A. Protective effect of young coconut water on MMP -1 levels and collagen fiber thickness in male Wistar rats exposed to UVB light exposure. https://www.openveterinaryjournal.com/?mno=306671 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.42 AMA (American Medical Association) Style Angelin M, Zulaikhah ST, Subchan P, Khairullah AR, Effendi MH, Hestianah EP, Angelin M, Budiastuti B, Permatasari DA, Pratama BP, Rehman S, Ganisia A. Protective effect of young coconut water on MMP -1 levels and collagen fiber thickness in male Wistar rats exposed to UVB light exposure. Open Vet. J.. 2026; 16(3): 1868-1876. doi:10.5455/OVJ.2026.v16.i3.42 Vancouver/ICMJE Style Angelin M, Zulaikhah ST, Subchan P, Khairullah AR, Effendi MH, Hestianah EP, Angelin M, Budiastuti B, Permatasari DA, Pratama BP, Rehman S, Ganisia A. Protective effect of young coconut water on MMP -1 levels and collagen fiber thickness in male Wistar rats exposed to UVB light exposure. Open Vet. J.. (2026), [cited March 31, 2026]; 16(3): 1868-1876. doi:10.5455/OVJ.2026.v16.i3.42 Harvard Style Angelin, M., Zulaikhah, . S. T., Subchan, . P., Khairullah, . A. R., Effendi, . M. H., Hestianah, . E. P., Angelin, . M., Budiastuti, . B., Permatasari, . D. A., Pratama, . B. P., Rehman, . S. & Ganisia, . A. (2026) Protective effect of young coconut water on MMP -1 levels and collagen fiber thickness in male Wistar rats exposed to UVB light exposure. Open Vet. J., 16 (3), 1868-1876. doi:10.5455/OVJ.2026.v16.i3.42 Turabian Style Angelin, Melinda, Siti Thomas Zulaikhah, Prasetyowati Subchan, Aswin Rafif Khairullah, Mustofa Helmi Effendi, Eka Pramyrtha Hestianah, Melda Angelin, Budiastuti Budiastuti, Dian Ayu Permatasari, Bima Putra Pratama, Saifur Rehman, and Ainun Ganisia. 2026. Protective effect of young coconut water on MMP -1 levels and collagen fiber thickness in male Wistar rats exposed to UVB light exposure. Open Veterinary Journal, 16 (3), 1868-1876. doi:10.5455/OVJ.2026.v16.i3.42 Chicago Style Angelin, Melinda, Siti Thomas Zulaikhah, Prasetyowati Subchan, Aswin Rafif Khairullah, Mustofa Helmi Effendi, Eka Pramyrtha Hestianah, Melda Angelin, Budiastuti Budiastuti, Dian Ayu Permatasari, Bima Putra Pratama, Saifur Rehman, and Ainun Ganisia. "Protective effect of young coconut water on MMP -1 levels and collagen fiber thickness in male Wistar rats exposed to UVB light exposure." Open Veterinary Journal 16 (2026), 1868-1876. doi:10.5455/OVJ.2026.v16.i3.42 MLA (The Modern Language Association) Style Angelin, Melinda, Siti Thomas Zulaikhah, Prasetyowati Subchan, Aswin Rafif Khairullah, Mustofa Helmi Effendi, Eka Pramyrtha Hestianah, Melda Angelin, Budiastuti Budiastuti, Dian Ayu Permatasari, Bima Putra Pratama, Saifur Rehman, and Ainun Ganisia. "Protective effect of young coconut water on MMP -1 levels and collagen fiber thickness in male Wistar rats exposed to UVB light exposure." Open Veterinary Journal 16.3 (2026), 1868-1876. Print. doi:10.5455/OVJ.2026.v16.i3.42 APA (American Psychological Association) Style Angelin, M., Zulaikhah, . S. T., Subchan, . P., Khairullah, . A. R., Effendi, . M. H., Hestianah, . E. P., Angelin, . M., Budiastuti, . B., Permatasari, . D. A., Pratama, . B. P., Rehman, . S. & Ganisia, . A. (2026) Protective effect of young coconut water on MMP -1 levels and collagen fiber thickness in male Wistar rats exposed to UVB light exposure. Open Veterinary Journal, 16 (3), 1868-1876. doi:10.5455/OVJ.2026.v16.i3.42 |