| Research Article | ||
Open Vet. J.. 2026; 16(3): 1578-1589 Open Veterinary Journal, (2026), Vol. 16(3): 1578-1589 Research Article Efficacy of Rosmarinus officinalis nanoemulsion as an anti-inflammatory and antinociceptive agent by targeting the interleukin-1β, substance P, and neurokinin-1 receptor pathways in experimental pulpitisYuli Nugraeni1*, Agustine Hanafi Putri2, Abdur Razaq Kamaruzzaman3, Regina Putri Virigirinia3, Rahmavidyanti Priyanto1 and Wibi Riyawan51Department of Operative Dentistry and Endodontics, Dental Faculty of Brawijaya University, Malang, Indonesia 2Department of Oral Medicine, Dental Faculty of Brawijaya University, Malang, Indonesia 3Department of Oral Biologi, Dental Faculty of Brawijaya University, Malang, Indonesia 4Department of Operative Dentistry and Endodontics, Dental Faculty of Brawijaya University, Malang, Indonesia 5Departement of Biochemistry and Biomoleculer, Faculty of Medicine, Universitas Brawijaya, Malang, 65145, Indonesia *Corresponding Author: Yuli Nugraeni. Department of Operative Dentistry and Endodontics, Dental Faculty of Brawijaya University, Malang, Indonesia. Email: yulinugraenifkgub [at] gmail.com; yulinugraeni [at] ub.ac.id Submitted: 01/10/2025 Revised: 30/01/2026 Accepted: 13/02/2026 Published: 31/03/2026 © 2026 Open Veterinary Journal
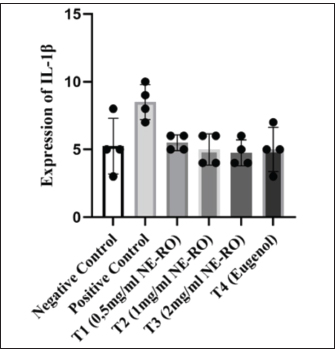
AbstractBackground: Inflammation of the dental pulp is commonly associated with intense nociceptive signaling mediated by interleukin-1β (IL-1β), Substance P, and neurokinin-1 receptors (NK1-R). Natural compounds, such as Rosmarinus officinalis, contain bioactive molecules that may alleviate this response, particularly when expressed in nanoemulsions that enhance tissue penetration. Aim: This study explored the modulatory effect of NE-RO on inflammatory and nociceptive markers in an experimental rat model of acute pulpitis. Methods: Thirty healthy male Wistar rats were randomly assigned to six treatment groups, including negative and positive controls, three NE-RO treatment concentrations (0.5, 1.0, and 2.0 mg/ml), and an eugenol comparison group, after 1 week of acclimatization under controlled housing conditions. Pulpitis was induced by lipopolysaccharide application to the exposed pulp chamber. After 24 hours of treatment, the first maxillary molars were processed for immunohistochemical assessment of IL-1β, Substance P, and NK1-R. Data were analyzed using analysis of variance or Kruskal–Wallis tests with a 95% confidence level. Results: Induction of pulpitis significantly elevated IL-1β, Substance P, and NK1-R expression relative to the untreated control. The administration of 2 mg/ml NE-RO resulted in marked reductions in the levels of Substance P (5.0 ± 0.40) and NK1-R (5.5 ± 0.29) compared with the positive control (p < 0.05), while IL-1β levels showed a downward trend (4.75 ± 0.48; p=0.08). These outcomes were comparable to those of eugenol treatment. Conclusion: Topical application of 2 mg/ml Rosmarinus officinalis nanoemulsion attenuated neuroinflammatory responses in experimental pulpitis, likely through suppression of the IL-1β–Substance P–NK1-R signaling axis. The findings support its potential as a plant-based alternative topical agent for the management of acute pulpitis, pending further safety evaluation. Keywords: Nanoemulsion, IL-1β, Substance P, NK1-R, Acute pulpitis model. IntroductionDental caries remains one of the most widespread oral health challenges globally, affecting individuals in both industrialized and developing regions. The persistent nature of this disease reflects the multifactorial interplay among host factors, microbial activity, and lifestyle-related habits. Epidemiological analyses, such as those reported by Tu et al. (2023), estimate that approximately 2.3 billion people experience caries in permanent teeth, underscoring the magnitude of this condition in public health. Similarly, the Global Burden of Disease study identified caries as a principal cause of tooth loss, which adversely influences nutritional intake, speech, and overall well-being (Bernabe et al., 2020). Dietary behavior and oral hygiene remain the central determinants of caries development. Frequent consumption of refined sugars and insufficient plaque control accelerate demineralization processes and support cariogenic biofilm growth (Qin et al., 2022; Tu et al., 2023). Variations in disease severity are also influenced by host-related factors, including salivary composition, enamel characteristics, and oral microbiota diversity (Pitts et al., 2021). Dental caries can provoke discomfort and impair mastication, facial appearance, and quality of life beyond its local effects. If left unmanaged, the lesion may advance toward the pulp chamber, resulting in irreversible pulpitis and occasionally abscess formation that necessitates invasive therapy or extraction (Warreth, 2023; Abdullah et al., 2024). At the cellular level, pulp inflammation is initiated when bacterial products, particularly lipopolysaccharides (LPS) from gram-negative bacteria, penetrate dentinal tubules and interact with Toll-like receptor 4 on odontoblasts. This interaction activates a downstream signaling cascade involving interleukin-1β (IL-1β) and tumor necrosis factor-alpha (TNF-α) (Brodzikowska et al., 2022; Lan et al., 2022). The resulting cytokine response stimulates arachidonic acid metabolism, generating prostaglandin E₂ (PGE₂) and epoxyeicosatrienoic acids (EETs). These lipid mediators act on transient receptor potential vanilloid 4 (TRPV4) channels, amplifying nociceptive signaling and pain perception (Masotti, 2020; Rodrigues et al., 2022). Transient receptor potential vanilloid 4 channel activation leads to an influx of calcium ions (Ca²⁺) into sensory nerve endings within the dental pulp. This rise in intracellular calcium promotes the secretion of the neuropeptide substance P (SP), which plays a key role in transmitting pain signals from inflamed tissue to the CNS (Rodrigues et al., 2022). Once released, SP interacts with neurokinin-1 receptors (NK1-R) located on adjacent neurons and immune cells, further amplifying local inflammation and nociceptive signaling (Malacarne et al., 2022). Because of their coordinated action in propagating inflammatory pain, IL-1β, SP, and NK1-R are widely recognized as reliable molecular indicators of pulp inflammation and tissue irritation. Recent advances in understanding the management of dental infections have highlighted the importance of targeted antimicrobial and anti-inflammatory strategies in preserving pulp vitality (Abdullah et al., 2024). Emerging evidence suggests that when combined with conventional endodontic therapies, botanical compounds may offer synergistic benefits, particularly in managing inflammatory mediators during the early stages of pulp injury (Abozaid et al., 2025). Additionally, novel therapeutic approaches targeting the host immune response have shown promise in modulating tissue damage while maintaining antimicrobial efficacy (Alsuraifi et al., 2024; Alsuraifi et al., 2025; Ezzat et al., 2025). Pulpal inflammation is not merely a localized process but involves complex interactions between immune mediators and the sensory nervous system (Galler et al., 2021). The activation of proinflammatory cytokines, particularly IL-1β, has been shown to trigger the release of nociceptive neuropeptides such as Substance P (Skoff et al., 2009), which bind to NK1-R on neuronal and immune cells within the pulp tissue (Zhu and Bhatia, 2023). This interaction amplifies the inflammatory response and enhances peripheral pain transmission. Emerging evidence further indicates that this neuroinflammatory pathway represents a promising therapeutic target for the management of pulpitis and orofacial pain. Recent reports suggest that modulation of IL-1β and Substance P expression correlates with reduced NK1-R activation and improved pulpal inflammatory responses in both animal models and translational studies (Mashaghi et al., 2016; Symmank et al., 2025). Therapeutic approaches capable of suppressing the IL-1β–Substance P–NK1-R axis are considered highly relevant for developing mechanism-based anti-inflammatory and antinociceptive agents (Lundy and Linden, 2004). Eugenol remains a conventional topical agent for managing pulpitis in clinical dentistry owing to its antibacterial and analgesic potential (Ulanowska and Olas, 2021; Nandlal et al., 2024). However, its clinical use is constrained by reports of dose-dependent cytotoxicity on dental pulp fibroblasts (Nandlal et al., 2024) and by its tendency to cause local irritation and chemical instability in restorative formulations (Kamal et al., 2025). Therefore, there is a critical need to explore safer and more effective alternative therapeutic agents derived from natural sources. Given these limitations, there is growing interest in discovering plant-based alternatives with comparable efficacy but lower toxicity. Rosmarinus officinalis L. (rosemary) is an aromatic herb abundant in biologically active phenolic compounds such as rosmarinic acid, carnosic acid, carnosol, and ursolic acid. These molecules exert antioxidant, anti-inflammatory, and analgesic effects (Andrade et al., 2018). Previous studies have indicated that extracts of R. officinalis can suppress the synthesis of major proinflammatory cytokines, such as TNF-α and IL-1β, leading to a reduction in the local inflammatory response (Borrás-Linares et al., 2014). Moreover, rosemary extract has been reported to modulate pain signaling pathways, potentially involving TRPV4 channels and NO/cGMP/PKG-dependent mechanisms, which may reduce the release of neuropeptide substance P and subsequent pain transmission(Borrás-Linares et al., 2014; Hajhashemi and Najibzadeh, 2025). The nanoemulsion system improves both the pharmacokinetic profile and the absorption depth of active constituents in the tissue. The formulation increases the surface area and enhances bioavailability within target tissues by dispersing hydrophobic compounds into nanosized droplets. Previous research has reported that nanoemulsions prepared from R. officinalis extracts exert beneficial effects in several inflammatory models, including osteoarthritis, and have shown promise for both topical and systemic anti-inflammatory applications (Mohammadifar et al., 2021) and both local and systemic anti-inflammatory applications (Rocha et al., 2014). Based on these findings, the present study was designed to evaluate whether a rosemary nanoemulsion could modulate the expression of IL-1β, Substance P, and NK1-R in a rat model of LPS-induced pulpitis. This formulation may provide a safer and more effective alternative to conventional agents used for the management of pulp inflammation. Materials and MethodsResearch designThis laboratory-based experimental study was conducted at the Oral Biology Laboratory, Faculty of Dentistry, and the Anatomical Pathology Laboratory, Faculty of Medicine, Universitas Brawijaya, Malang, Indonesia. Thirty healthy male Wistar rats (Rattus norvegicus) aged between 8 and 10 weeks and weighing 220–300 g were used in the experiment. Animals were acclimatized for 7 days in individual polypropylene cages lined with sterilized rice husks. Room conditions were maintained at 22°C ± 2°C with a relative humidity of 55% ± 5% under a 12-hour light/dark cycle (Castelhano-Carlos and Baumans, 2009). The standard pellet feed and tap water were provided ad libitum. Only active and healthy rats were included; animals showing >10% body weight loss during acclimatization were excluded. The sample size was determined based on a preliminary power analysis using G*Power 3.1 software (Kang, 2021). Assuming an effect size (f) of 0.50, α error probability of 0.05, and desired power (1-β) of 0.80 for detecting differences among six groups using one-way Analysis of variance (ANOVA), a minimum of four animals per group was required. We employed five animals per group (n=5) to account for potential technical variability and ensure robust statistical analysis, consistent with sample sizes reported in similar pulpitis studies. Animals were randomly allocated into six experimental groups (n=5 per group) using a computer-generated randomization sequence following acclimatization. Group assignments were concealed in sealed, opaque envelopes that were opened sequentially by an investigator who was not involved in the experimental procedures or data analysis. The experimental groups were as follows: Negative control (no intervention); Positive control (pulpitis induction only); T1–pulpitis + 0.5 mg/ml rosemary nanoemulsion; T2–pulpitis + 1.0 mg/ml rosemary nanoemulsion; T3–pulpitis + 2.0 mg/ml rosemary nanoemulsion; and T4–pulpitis + eugenol treatment. The selected concentrations (0.5, 1.0, and 2.0 mg/ml) were based on preliminary dose-ranging studies (unpublished data) and previous reports demonstrating the effective anti-inflammatory activity of rosemary extracts in other inflammatory models. The nanoemulsion formulation was designed to enhance tissue penetration through dentinal tubules, thereby improving the bioavailability of lipophilic bioactive compounds at the pulp inflammation site. Histological slide preparation was coded, and immunohistochemical evaluation was performed by two independent, blinded observers who were unaware of group assignments to minimize bias. Any discrepancies in cell counting were resolved through discussion and re-evaluation until a consensus was reached. Preparation of rosemary extract and nanoemulsionFreshly dried rosemary (Rosmarinus officinalis) leaves were rinsed to remove surface debris, cut into smaller pieces, and oven-dried at 40°C–50°C until a constant low-moisture level was reached. The dried leaves were ground into fine powder (40-mesh) and subjected to maceration using 96% ethanol at a ratio of 1:10 (w/v) for 48 hours under gentle agitation (150 rpm). Subsequently, the filtrate was clarified by repeated filtration and concentrated at 45°C under reduced pressure using a rotary evaporator. The resulting extract was stored in airtight, light-protected vials at 4°C until further use (Salamatullah et al., 2021). NE-RO was formulated using the spontaneous emulsification method, employing a mixture composed of soybean oil (16.4%), PEG-40 hydrogenated castor oil (32.1%), Span-80 (11.6%), and PEG-400 (40%). This mixture was heated to 60°C, thoroughly homogenized, and then combined with 100 mg/ml of rosemary extract stock solution, followed by sonication at 40 kHz for 10 minutes. The droplet size (Z-average approximately 190 nm; PDI < 0.3) and ζ-potential (approximately 13 mV) were confirmed by dynamic light scattering analysis (Rocha-Filho and Maruno, 2025). The nanoemulsion was then diluted with sterile distilled water to final concentrations of 0.5, 1, and 2 mg/ml for biological testing. Preparation, treatment, and observation of pulpitis animal modelTikus male Wistar rats were acclimated under standard laboratory conditions. General anesthesia was administered via intramuscular injection of ketamine hydrochloride (Hengrui Inc., Nanjing, China) at a dose of 100 mg/kg body weight. Pulpitis induction was performed by preparing a cavity approximately 0.5 mm deep on the occlusal surface of the maxillary first molar using a low-speed dental bur. Subsequently, 1 µl of Escherichia coli LPS (10 mg/ml) was applied into the cavity (Miron et al., 2024). After approximately 20 minutes of exposure to E. coli LPS, the treated cavities received 1 µl of either NE-RO or eugenol. Each cavity was immediately sealed with a thin layer of phosphate cement to prevent pulp tissue contamination and desiccation. Twenty-four hours later, the animals were humanely euthanized by gradual CO₂ inhalation in accordance with institutional ethical guidelines (Valentim et al., 2016). The maxillae were dissected and fixed in 10% neutral buffered formalin for fixation. Decalcification was performed using 10% Ethylenediaminetetraacetic acid (EDTA) for approximately 30 days, with the solution replaced every few days to maintain the decalcifying efficiency. Following decalcification, the samples were dehydrated using graded ethanol, cleared in xylene, and embedded in paraffin blocks (Savi et al., 2017). Sections of approximately 4 µm thickness were cut and mounted on glass slides. The slides were immersed in EDTA buffer (pH 8.0) and heated at 95°C for 20 minutes for antigen retrieval. The endogenous peroxidase activity was then quenched using 3% hydrogen peroxide. Tissue sections were incubated overnight at 4°C with the following primary antibodies at a dilution of 1:200: IL-1β (mouse monoclonal, catalog no. sc-32294, Santa Cruz Biotechnology), Substance P (mouse monoclonal, catalog no. sc-58591, Santa Cruz Biotechnology), or NK1-R (mouse monoclonal, catalog no. sc-365091, Santa Cruz Biotechnology) at a dilution of 1:200. The DAB-HRP detection system was used to visualize the bound antibodies, and cell nuclei were counterstained with hematoxylin to aid microscopic evaluation. For quantitative analysis, immunopositive cells within the odontoblast layer were counted in five randomly selected microscopic fields per slide (400 × magnification). Each treatment group consisted of five animals (n=5), and one representative histological section was evaluated per animal. The mean cell count from the five microscopic fields per section was calculated, yielding one data point per animal. Therefore, statistical analysis was based on five biological replicates per group. Data analysisData were analyzed using GraphPad version 10. Normality was assessed using the Shapiro-Wilk test. For normally distributed data, one-way analysis of variance followed by post hoc Tukey HSD comparisons was applied. For non-normally distributed data, the Kruskal–Wallis test was used, with Mann–Whitney U tests for pairwise comparisons. Statistical significance was set at p < 0.05. Ethical approvalEthical approval was granted on March 15, 2023 (Approval No. 043-KEP-UB-2023). ResultsExpression of IL-1βThe average number of IL-1β-positive cells tended to decrease as the NE-RO concentration increased. Mean values were recorded at 5.5 ± 0.29 for T1 (0.5 mg/ml), 5.0 ± 0.58 for T2 (1 mg/ml), and 4.75 ± 0.48 for T3 (2 mg/ml), respectively. These findings were similar to those of the eugenol-treated group (5.0 ± 0.82 cells/field). Although a downward trend was evident, statistical analysis (Kruskal–Wallis test) did not reveal significant differences among groups (p > 0.05) (Figs. 1 and 2).
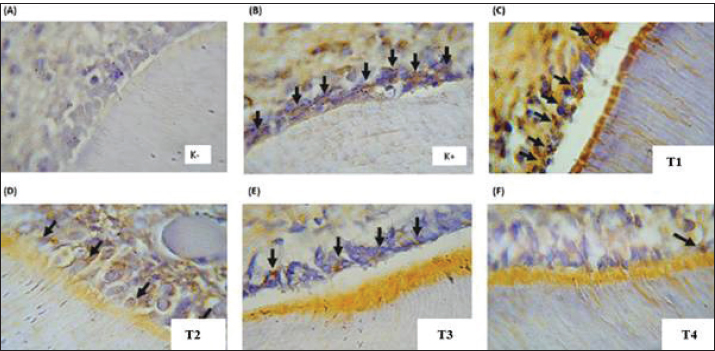
Fig. 1. Expression of IL-1β in Odontoblast cells of rat dental pulp tissue (Immunohistochemistry, 400×). IL-1β expression is indicated by brown staining in the cytoplasm of odontoblast cells (black arrows). Odontoblast cells are arranged along the pulp chamber periphery in the maxillary first molar of rats. Representative micrographs show the following: (A) Negative control—minimal IL-1β expression, with only a few positively stained cells visible; (B) Positive control (pulpitis)—the strongest immunoreactivity, indicated by intense brown coloration in most odontoblasts; (C) NE-RO 0.5 mg/ml—moderate staining intensity; (D) NE-RO 1 mg/ml—mild expression pattern; (E) NE-RO 2 mg/ml—very faint staining, almost comparable with the negative control; (F) Eugenol treatment—mild to moderate IL-1β positivity. Micrographs are original images obtained from the authors' own experimental study.
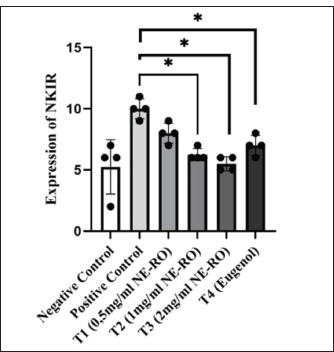
Fig. 2. Quantitative analysis of IL-1β expression in the odontoblast layers. The bar chart (mean ± SD) indicates that IL-1β-positive cells were most abundant in the positive control group (8.5 ± 2.65 cells/field). Treatment with R. officinalis nanoemulsion at all tested concentrations resulted in a gradual reduction in IL-1β expression, but the overall differences among groups were not statistically significant (Kruskal–Wallis test, p > 0.05). Expression of NK1-RA significant overall treatment effect was detected for NK1-R expression (p < 0.05, Kruskal–Wallis test). The untreated pulpitis group displayed the highest number of NK1-R-positive cells (10 ± 0.41 cells/field). In contrast, the administration of NE-RO at 2 mg/ml (T3) led to a marked decline in NK1-R-positive cells, reaching 5.5 ± 0.29 cells/field. Treatments with 1 mg/ml NE-RO (T2) and eugenol produced comparable results, both averaging around cells/field (6.25 ± 0.25 and 7.0 ± 0.41, respectively). The lowest concentration group, T1 (0.5 mg/ml), showed a moderate reduction to 8.0 ± 0.41 cells/field; however, this change did not differ significantly from the positive control (Figs. 3 and 4).
Fig. 3. Fig. 3. NK1-R expression in odontoblast cells of rat dental pulp tissue (Immunohistochemistry, 400×). NK1-R expression is identified by brown staining in the cytoplasm of odontoblast cells (black arrows). Odontoblast cells are arranged along the margin of the pulp chamber of the maxillary first molar in rats. (a) Negative control group (K–), showing minimal NK1-R expression with only a few positively stained cells compared to the positive control group. (b) Positive control group (K+), demonstrating the highest NK1-R expression with prominent brown staining. (c) NE-RO 0.5 mg/ml treatment group, showing moderate NK1-R expression. (d) NE-RO 1 mg/ml treatment group, showing mild NK1-R expression. (e) NE-RO 2 mg/ml treatment group, exhibiting significantly reduced NK1-R expression, comparable to the negative control group. (f) Eugenol treatment group, presenting mild NK1-R expression, similar to NE-RO at 1 mg/ml. Immunohistochemical staining using DAB-HRP with hematoxylin counterstain, original magnification 400×. Statistical analysis revealed significant differences among the groups (Kruskal–Wallis, p < 0.05). Micrographs are original images obtained from the authors' own experimental study.
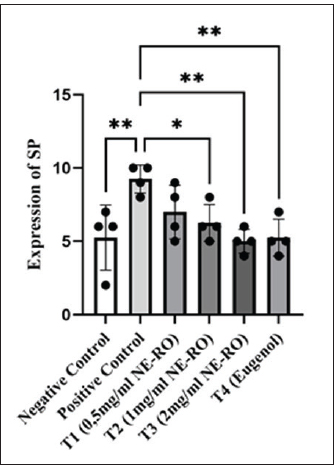
Fig. 4. NK1-R expression after NE-RO treatment. The bar graph presents the mean number of NK1-R–positive cells across the experimental groups. The positive control group showed the highest NK1-R expression (10.00 ± 0.41 cells/field). In contrast, treatment with NE-RO at a concentration of 2 mg/ml (T3) resulted in a marked reduction in NK1-R–positive cells (5.50 ± 0.29 cells/field), reaching values comparable to those observed in the eugenol-treated and other treatment groups. Statistical analysis using the Kruskal–Wallis test demonstrated significant differences among groups (p < 0.05). Asterisks indicate statistically significant differences relative to the positive control. Expression of the substance PAnalysis of variance (one-way) revealed a clear difference in the expression of substance P among the groups (p < 0.001). The positive control exhibited the highest mean value (9.25 ± 0.48 cells/field), whereas the negative control had a considerably lower count (5.25 ± 1.10 cells/field). Treatment with 2 mg/ml rosemary nanoemulsion (T3) resulted in the greatest reduction in Substance P expression (5.00 ± 0.41 cells/field), exceeding the effect observed with eugenol and other treatment groups. Eugenol treatment (T4) resulted in Substance P expression of 5.25 ± 0.63 cells/field, whereas intermediate doses of rosemary nanoemulsion (T1: 0.5 mg/ml and T2: 1.0 mg/ml) reduced the expression of Substance P to 7.0 ± 0.91 and 6.25 ± 0.63 cells/field, respectively, compared with the positive control group (Figs. 5 and 6).
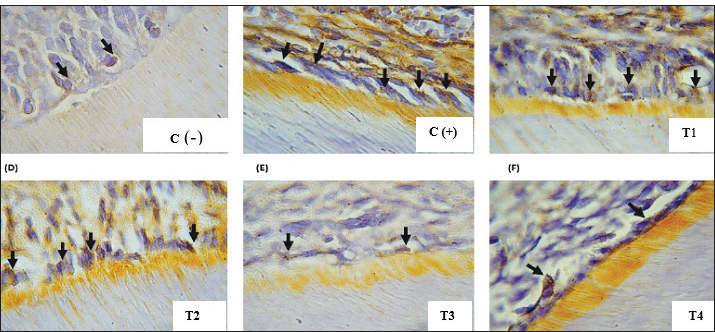
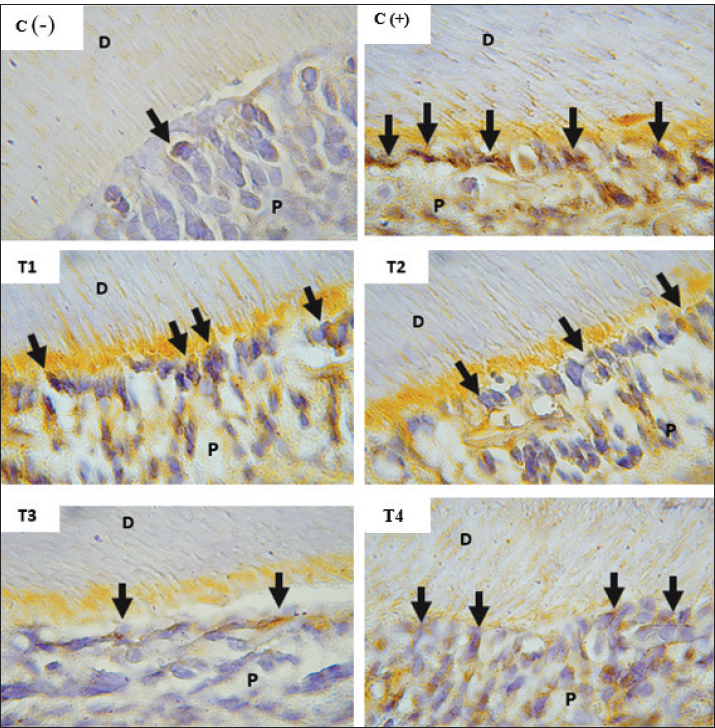
Fig. 5. Fig. 5. Substance P expression in odontoblast cells of rat dental pulp tissue (Immunohistochemistry, 400×). Substance P (SP) expression is indicated by brown staining in the cytoplasm of odontoblast cells (arrows). The odontoblast layer is observed along the pulp chamber edge (P) adjacent to the dentin (D). (a) Negative control group [C(–)] showing minimal SP expression with fewer stained cells. (b) Positive control group [C(+)] (pulpitis) demonstrating the highest SP expression, indicated by strong brown staining intensity. (c) Group treated with rosemary nanoemulsion [T1] (NE-RO) 0.5 mg/ml, showing moderate SP expression. (d) Group treated with NE-RO [T2] 1 mg/ml, displaying mild SP expression. (e) Group treated with NE-RO [T3] 2 mg/ml, exhibiting the lowest SP expression, comparable to the negative control group. (f) Group treated with eugenol [T4], showing mild-to-moderate SP expression. Immunohistochemical staining using DAB-HRP with hematoxylin counterstain, original magnification 400×. One-way ANOVA revealed significant differences among groups (p < 0.001). Micrographs are original images obtained from the authors' own experimental study.
Fig. 6. Comparative expression of substance P among the experimental groups. The bar chart illustrates clear group variations in Substance P–positive cell counts, confirmed by one-way ANOVA (p < 0.001). The positive control group exhibited the highest level of Substance P expression, whereas the treatment with NE-RO at 2 mg/ml produced the lowest mean value. Post-hoc Dunnett’s analysis identified significant (p < 0.05) and highly significant (*p < 0.001) reductions relative to the positive control. DiscussionThe induction of acute pulpitis with LPS successfully reproduced a local inflammatory condition characterized by a significant rise in IL-1β, SP, and NK1-R expression compared with healthy controls (Brodzikowska et al., 2022; Lan et al., 2022). This outcome aligns with the well-established mechanism by which bacterial endotoxins activate odontoblasts and resident immune cells within the dental pulp. Once activated, these cells release a range of pro-inflammatory mediators, including IL-1β, TNF-α, and various prostaglandins, which collectively sustain the inflammatory response. The resulting vascular dilation, permeability changes, and leukocyte migration explain the classical clinical signs of pulpitis, such as pain, tenderness, and heightened sensitivity (Cooper et al., 2017). Microscopic observations from the current study (unpublished data) reinforced these findings. Inflamed tissues showed odontoblast layer disruption, small blood vessel congestion, and abundant neutrophil infiltration. Similar histopathological patterns were described by Wang et al. (2024), who reported that vascular engorgement and cellular damage typify acute pulpitis, in contrast to chronic cases, where fibrotic remodeling and persistent mononuclear infiltration dominate. The cytokines IL-1β and TNF-α act as early mediators that amplify both inflammatory and nociceptive responses. They activate phospholipase A₂, which releases AA from membrane phospholipids. The liberated substrate is then metabolized through two main enzymatic routes: the cyclooxygenase (COX) pathway, yielding PGE₂, and the epoxygenase pathway, which produces EETs (Soares et al., 2023; Wautier and Wautier, 2023). PGE₂ increases the excitability of nociceptors, whereas EETs act on TRPV4 channels, promoting calcium entry into sensory neurons. Elevated intracellular calcium triggers the release of substance P from nerve endings, which then binds to NK1-R receptors and stimulates the p38-MAPK pathway, thereby extending peripheral sensitization and pain transmission (Masotti, 2020; Mehboob et al., 2021). These established mechanisms provide a plausible framework for understanding how NE-RO may alleviate pulp pain. The observed reductions in SP and NK1-R expression following NE-RO administration are consistent with the downstream modulation of inflammatory signaling cascades. However, the direct demonstration of TRPV4 channel activity or upstream cytokine inhibition would be required to establish the specific molecular targets of NE-RO in this model. Histopathological data further supported this biochemical evidence. At lower concentrations (0.5–1 mg/ml), NE-RO application was associated with moderate tissue edema, fewer infiltrating leukocytes, and early signs of odontoblast realignment. Treatment at 2 mg/ml, as well as eugenol exposure, restored the pulp structure to nearly normal architecture, showing minimal inflammatory infiltration, closely resembling the negative control (unpublished data). This dose-dependent trend mirrors typical phytopharmacological behavior, where increasing bioactive compound concentrations enhance therapeutic potency up to an optimal threshold. However, beyond that point, further dose escalation may not improve outcomes and could even increase cytotoxic risk. These findings highlight the relevance of optimizing herbal nanoformulations, such as NE-RO, to achieve maximal biological benefit while maintaining tissue safety. Nevertheless, careful consideration must be given to dosing because exceeding certain thresholds may lead to cytotoxic effects due to the inherent toxicity potential of some phytochemicals at high concentrations (European Medicines Health, 2022; Yadav et al., 2023). In this study, the lower efficacy observed at doses of 0.5 and 1 mg/ml likely resulted from insufficient concentrations of bioactive compounds to reach therapeutic levels within pulp tissues. Furthermore, developing RO extracts as nanoemulsions with droplet sizes below 200 nm significantly enhances the solubility and penetration of lipophilic compounds into dentinal tubules, thereby improving therapeutic effectiveness (Vieira et al., 2023). Based on molecular docking analyses (unpublished data), bioactive compounds from R. officinalis L. extract interact effectively with molecular targets involved in inflammatory and pain pathways associated with pulpitis. Carnosic acid and carnosol exhibited low binding energies, indicating strong and stable interactions with the active sites of TRPV4 and Nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB). Additionally, rosmarinic acid demonstrated a high binding affinity toward the IL-1β binding site, indicating its ability to inhibit proinflammatory signaling pathways. Rosemary leaves have significant anti-inflammatory, analgesic, and antioxidant effects. These compounds inhibit transcription factors such as NF-κB and MAPK, consequently reducing the synthesis of proinflammatory cytokines, including IL-1β. Reduced cytokine expression diminishes nociceptive sensitization, subsequently decreasing the release of the neuropeptide Substance P and NK1-R receptor activation. Ultimately, this cascade reduces the pain and inflammation associated with pulpitis (Maione et al., 2017; Habtemariam, 2023). Ursolic acid also interacts with critical residues of molecular targets implicated in pulpitis, exerting immunomodulatory effects by inhibiting leukocyte adhesion (unpublished data). This triterpenoid compound decreases the expression of key leukocyte adhesion molecules, such as intercellular adhesion molecule-1 and vascular cell adhesion molecule-1, thereby reducing the migration and infiltration of proinflammatory immune cells into the affected tissue. The modulatory effects of R. officinalis compounds appear to extend to the cellular immune response. Our data show that NE-RO reduces leukocyte accumulation within the inflamed pulp. This observation is consistent with prior evidence showing that rosemary-derived triterpenes limit polymorphonuclear cell migration and promote tissue repair (Choi and Lee, 2019; Renda et al., 2022). Such modulation of immune cell recruitment likely contributes to a more controlled inflammatory environment that favors pulp architecture recovery. Notably, the response pattern observed in the NE-RO group was comparable to that of eugenol-treated animals. Eugenol, a conventional agent for direct pulp capping, exerts its therapeutic action through phenolic constituents capable of modulating the NF-κB signaling cascade, thereby limiting the synthesis of pro-inflammatory cytokines such as IL-1β. It also interferes with the COX pathway, reducing PGE₂ production. This reduction indirectly dampens the activity of the TRPV4 channel and, in turn, curtails the release of neuropeptides such as SP. When less Substance P is available to engage NK1-R receptors in sensory neurons, the propagation of pain impulses becomes markedly attenuated, explaining the analgesic response observed in pulpitis. Eugenol demonstrates a modest local anesthetic effect by blocking neuronal sodium channels, which further helps reduce discomfort at the site of application (Ulanowska and Olas, 2021; Nandlal et al., 2024). The therapeutic outcomes achieved with NE-RO were closely aligned with those observed in the eugenol-treated group, a standard agent widely applied in pulpitis management. The pharmacological activity of eugenol is largely attributed to its phenolic constituents, which can downregulate inflammatory signaling by suppressing the activation of the NF-κB pathway and consequently reducing the expression of key cytokines, including IL-1β. Eugenol also interferes with the COX pathway, limiting the production of PGE₂. The decline in PGE₂ levels indirectly decreases TRPV4 channel activity and reduces the subsequent release of neuropeptides, such as SP. Together, these interactions result in lower stimulation of NK1-R receptors within sensory neurons, effectively dampening pain transmission during pulp inflammation. Eugenol also exerts a mild local anesthetic effect by blocking sodium channels in peripheral nerves, further reinforcing its analgesic action (Ulanowska and Olas, 2021; Nandlal et al., 2024). The similar outcomes observed between eugenol and rosemary nanoemulsion treatments indicate that NE-RO offers comparable therapeutic effects at the 24-hour endpoint. Several limitations of this study warrant consideration. First, the assessment was limited to a single 24-hour post-treatment time point, which only captures the acute inflammatory response. Conclusions regarding long-term pulp healing, tissue repair, or sustained therapeutic efficacy are not permitted in this timeframe. Future investigations should incorporate multiple time points extending to 7, 14, and 28 days to evaluate inflammation resolution and reparative dentinogenesis progression. Second, although immunohistochemical markers provided insights into inflammatory and nociceptive signaling, direct behavioral or electrophysiological assessment of pain responses was not performed. The term "antinociceptive" in the title reflects the downregulation of molecular pain mediators rather than the direct measurement of nociceptive behavior. Subsequent studies incorporating established pain assessment methods, such as thermal sensitivity testing or operant behavioral paradigms, would strengthen analgesic efficacy claims. Third, systemic toxicity and local tissue tolerance were not formally evaluated. Although no overt signs of adverse effects were observed during the experimental period, comprehensive safety profiling of human dental pulp cells, including serum biochemistry, organ histopathology, and cytotoxicity assays, would be necessary before clinical translation. Long-term studies are still required to confirm these findings and to assess the continued effectiveness and safety of NE-RO during the pulp-healing phase extending beyond 7 days. Further formulation research, particularly on physicochemical stability during storage and clinical application, would also be beneficial. On a molecular level, exploring additional inflammatory signaling pathways, such as the NLRP3 inflammasome and oxidative Nrf2 pathways, may help clarify the broader mechanisms through which NE-RO exerts its anti-inflammatory and cytoprotective actions. ConclusionAt 2 mg/ml, Rosemary nanoemulsion effectively reduced Substance P and NK1-R expression in experimental pulpitis, with effects comparable to those of eugenol. These findings support its potential as a plant-based alternative for managing acute pulp inflammation. However, this study was limited to a 24-hour endpoint, precluding conclusions about long-term healing or pulp repair. No direct behavioral assessment of pain was performed, and no formal toxicity testing was conducted. Future work should evaluate extended time points (7–28 days), incorporate nociceptive behavioral assays, perform cytotoxicity testing on human dental pulp cells, and clarify the specific molecular mechanisms underlying the anti-inflammatory activity of NE-RO. Such investigations are necessary to validate safety and therapeutic potential before clinical application. AcknowledgmentsNone. Conflict of interestThe authors declare no conflicts of interest related to this article’s research, authorship, or publication. FundingThis work received financial support from the 2024 Non-Tax State Revenue allocation and an internal research grant from the Faculty of Dentistry, Universitas Brawijaya, Indonesia. Author’s contributionsConceptualization: Y.N.; Methodology: Y.N.; Validation: Y.N.; Formal Analysis: Y.N, R.P.V, A.H.P, A.R.K, R.P, W.R.; Investigation: Y.N.; Data Curation: Y.N, R.P.V, A.H.P, A.R.K, R.P, W.R.; Writing—Original Draft Preparation: Y.N, R.P.V, A.H.P, A.R.K, R.P, W.R.; Writing—Review and Editing: Y.N, R.P.V, A.H.P, A.R.K, R.P, W.R.; Supervision: Y.N, R.P.V, A.H.P, A.R.K, R.P, W.R. Data availabilityAll data supporting the results of this research are presented within the manuscript. Additional information can be made available from the corresponding author upon reasonable request. ReferencesAbdullah, F.M., Hatim, Q.Y., Oraibi, A.I., Alsafar, T.H., Alsandook, T.A., Lutfi, W. and Al-Hussaniy, H.A. 2024. Antimicrobial management of dental infections: an updated review. Medicine 103(27), e38630; doi:10.1097/md.0000000000038630 Abozaid, A., Diouchi, A., El Fattah, A.A., El-Sayed, H., Mahmoud, M., Abdelrahman, R. and Hassan, N. 2025. Anti-inflammatory and regenerative potential of plant-derived compounds in dental pulp therapy. J. Inflamm. Res. 18, 1123–1138; doi:10.2147/JIR.SXXXXX Alsuraifi , A., Mouzan, M.M., Ali, A.A.A., Algzaare, A., Aqeel, Z., Ezzat, D. and Ayad, A. 2025. Revolutionizing tooth regeneration: innovations from stem cells to tissue Engineering. Ahmed, M.S. and Mahdee, A.F. 2025. Role of neurogenic inflammation in pulp repair and assessment techniques (narrative review). Front. Dent. Med. 6, 1686734; doi:10.3389/fdmed.2025.1686734 Alsuraifi, A., Sulaiman, Z.M., Mohammed, N.A.R., Mohammed, J., Ali, S.K., Abdualihamaid, Y.H., Husam, F. and Ayad, A. 2024. Explore the most recent developments and upcoming developments in the field of dental nanomaterials. Beni-Suef. Univ. J. Basic. Appl. Sci. 13(1), 82; doi:10.1186/s43088-024-00540-7 Andrade, J.M., Faustino, C., Garcia, C., Ladeiras, D., Reis, C.P. and Rijo, P. 2018. Rosmarinus officinalis L.: an update review of its phytochemistry and biological activity. Future Sci. OA. 4(4), FSO283; doi: 10.4155/fsoa-2017-0124 Bernabe, E., Marcenes, W., Hernandez, C.R., Bailey, J., Abreu, L.G., Alipour, V., Amini, S., Arabloo, J., Arefi, Z., Arora, A., Ayanore, M.A., Bärnighausen, T.W., Bijani, A., Cho, D.Y., Chu, D.T., Crowe, C.S., Demoz, G.T., Demsie, D.G., Dibaji Forooshani, Z.S., Du, M., El Tantawi, M., Fischer, F., Folayan, M.O., Futran, N.D., Geramo, Y.C.D., Haj-Mirzaian, A., Hariyani, N., Hasanzadeh, A., Hassanipour, S., Hay, S.I., Hole, M.K., Hostiuc, S., Ilic, M.D., James, S.L., Kalhor, R., Kemmer, L., Keramati, M., Khader, Y.S., Kisa, S., Kisa, A., Koyanagi, A., Lalloo, R., Le Nguyen, Q., London, S.D., Manohar, N.D., Massenburg, B.B., Mathur, M.R., Meles, H.G., Mestrovic, T., Mohammadian-Hafshejani, A., Mohammadpourhodki, R., Mokdad, A.H., Morrison, S.D., Nazari, J., Nguyen, T.H., Nguyen, C.T., Nixon, M.R., Olagunju, T.O., Pakshir, K., Pathak, M., Rabiee, N., Rafiei, A., Ramezanzadeh, K., Rios-Blancas, M.J., Roro, E.M., Sabour, S., Samy, A.M., Sawhney, M., Schwendicke, F., Shaahmadi, F., Shaikh, M.A., Stein, C., Tovani-Palone, M.R., Tran, B.X., Unnikrishnan, B., Vu, G.T., Vukovic, A., Warouw, T.S.S., Zaidi, Z., Zhang, Z.J. and Kassebaum, N.J. 2020. Global, Regional, and National Levels and Trends in the Burden of Oral Conditions from 1990 to 2017: a Systematic Analysis of the Global Burden of Disease 2017 Study. J. Dent. Res. 99(4), 362–373; doi:10.1177/0022034520908533 Borrás-Linares, I., Stojanović, Z., Quirantes-Piné, R., Arráez-Román, D., Švarc-Gajić, J., Fernández-Gutiérrez, A. and Segura-Carretero, A. 2014. Rosmarinus officinalis leaves as a natural source of bioactive compounds. IJMS 15(11), 20585–20606; doi:10.3390/ijms151120585 Brodzikowska, A., Ciechanowska, M., Kopka, M., Stachura, A. and Włodarski, P.K. 2022. Role of Lipopolysaccharides derived from various bacterial species, Pulpitis—a. systematic. Rev. Biomolecules 12(1), 138; doi:10.3390/biom12010138 Castelhano-Carlos, M.J. and Baumans, V. 2009. Impact of light, noise, cage cleaning, and in-house transport on laboratory rats’ welfare and stress. Lab. Anim. 43(4), 311–327; doi:10.1258/la.2009.0080098 Choi, W.H. and Lee, I.A. 2019. Mechanism of action of ursolic acid as a potential anti-toxoplasmosis agent and its immunomodulatory effects. Pathogens 8(2), 61; doi:10.3390/pathogens8020061 Cooper, P.R., Chicca, I.J., Holder, M.J. and Milward, M.R. 2017. Inflammation and regeneration in the dentin-pulp complex: net gain or net loss?. J. Endodontics 43(9), S87–S94; doi:10.1016/j.joen.2017.06.011 European Medicines Agency (EMA). 2022. Guidelines on the quality of herbal medicinal products/traditional herbal medicinal products (EMA/HMPC/CHMP/CVMP/201116/2005 Rev. 3). Amsterdam, The Netherlands: European Medicines Agency. Ezzat, D., Azab, A., Kamel, I.S., Abdelmonem, M., Ibrahim, M.A., Ayad, A., Soomro, R., Wagdy, M. and Eldebawy, M. 2025. Phytomedicine and green nanotechnology: enhancing glass ionomer cements for sustainable dental restorations: a comprehensive review. Beni-Suef Univ. J. Basic Appl. Sci. 14(1), 48; doi:10.1186/s43088-025-00633-x Galler, K.M., Weber, M., Korkmaz, Y., Widbiller, M. and Feuerer, M. 2021. Inflammatory response mechanisms of the dentin–pulp complex and the periapical tissues. IJMS 22(3), 1480; doi:10.3390/ijms22031480 Habtemariam, S. 2023. Anti-inflammatory therapeutic mechanisms of natural products: insights from rosemary diterpenes, carnosic acid, and carnosol. Biomedicines 11(2), 545; doi:10.3390/biomedicines11020545 Hajhashemi, V. and Najibzadeh, E. 2024. Investigating the antinociceptive mechanism of Cinnamaldehyde in the formalin test in mice. Res. J. Pharmacogn. 12(2), 15–21; doi: 10.22127/rjp.2024.483814.2631 Hajhashemi, V. and Najibzadeh, E. 2025. Antinociceptive mechanisms of cinnamaldehyde in inflammatory pain models. Res. J. Pharmacogn. 12(2), 45–56; doi:10.22127/rjp.2025.xxxxxx Kamal, F.Z., Ciobica, A., Dascalescu, G., Rammali, S., Aalaoui, M.E., Lefter, R., Vata, I., Burlui, V. and Novac, B. 2025. Eugenol nanoparticles in dental composites: literature review of antimicrobial, anti-inflammatory, and clinical applications. Microorganisms 13(5), 1148; doi:10.3390/microorganisms13051148 Kang. and H. 2021. Determination of sample size and power analysis using the G*Power software. J. Educ. Eval. Health Prof. 18, 17; doi:10.3352/jeehp.2021.18.17 Lan, C., Chen, S., Jiang, S., Lei, H., Cai, Z. and Huang, X. 2022. Different expression patterns of inflammatory cytokines induced by lipopolysaccharides from Escherichia coli or Porphyromonas gingivalis in human dental pulp stem cells. BMC. Oral Health. 22(1), 121; doi:10.1186/s12903-022-02161-x Lundy, F.T. and Linden, G.J. 2004. Neuropeptides and neurogenic mechanisms in oral and periodontal inflammation. Crit. Rev. Oral. Biol. Med. 15(2), 82–98; doi:10.1177/154411130401500203 Maione, F., Cantone, V., Pace, S., Chini, M.G., Bisio, A., Romussi, G., Pieretti, S., Werz, O., Koeberle, A., Mascolo, N. and Bifulco, G. 2017. Anti‐inflammatory and analgesic activities of carnosol and carnosic acid in vivo, in vitro, and in silico analysis of their target interactions. Br. J. Pharmacol. 174(11), 1497–1508; doi:10.1111/bph.13545 Malacarne, P.F., Bezzenberger, J., Lopez, M., Warwick, T., Müller, N., Brandes, R.P. and Rezende, F. 2022. Epoxyeicosatrienoic acid and prostanoid crosstalk at the receptor and intracellular signaling levels to maintain vascular tone. IJMS 23(11), 5939; doi:10.3390/ijms23115939 Mashaghi, A., Marmalidou, A., Tehrani, M., Grace, P.M., Pothoulakis, C. and Dana, R. 2016. Neuropeptide substance P and the immune response. Cell. Mol. Life Sci. 73(22), 4249–4264; doi:10.1007/s00018-016-2293-z Masotti, M. 2020. Pathophysiological mechanisms of visceral pain in the murine model of Fabry disease. [Dissertation thesis]. Alma Mater Studiorum Università di Bologna, Bologna, Italy; doi: 10.48676/unibo/amsdottorato/9469 Mehboob, R., Hassan, S., Gilani, S.A., Hassan, A., Tanvir, I., Waseem, H. and Hanif, A. 2021. Enhanced neurokinin‐1 receptor expression is associated with human dental pulp inflammation and pain severity. BioMed Res. Int. 2021(1), 5593520; doi:10.1155/2021/5593520 Miron, R.J., Estrin, N.E., Sculean, A. and Zhang, Y. 2024. Understanding exosomes: part 3—therapeutic + diagnostic potential in dentistry. Periodontology 2000 94(1), 415–482; doi:10.1111/prd.12557 Mohammadifar, M., Aarabi, M.H., Aghighi, F., Kazemi, M., Vakili, Z., Memarzadeh, M.R. and Talaei, S.A. 2021. Anti-osteoarthritis potential of peppermint and rosemary essential oils in a nanoemulsion form: behavioral, biochemical, and histopathological evidence. BMC Complement. Med. Ther. 21(1), 57; doi:10.1186/s12906-021-03236-y MSh, A. and Mahdee, A.F. 2025. Role of neurogenic inflammation in pulp repair and assessment techniques (narrative review). Front. Dent. Med. 6, 1686734; doi:10.3389/fdmed.2025.1686734 Nandlal, B., Prem, K.S., Paras Mull, G., Vinutha, M. and Manjunath, M. 2024. Effectiveness of an ayurvedic gel for tooth pain relief due to dental caries: a randomized controlled trial. J. Clin. Adv. Dent. 8(1), 13–19; doi:10.29328/journal.jcad.1001041 Pitts, N.B., Twetman, S., Fisher, J. and Marsh, P.D. 2021. Understanding dental caries as a non-communicable disease. Br. Dent. J. 231(12), 749–753; doi:10.1038/s41415-021-3775-4 Qin, X., Zi, H. and Zeng, X. 2022. Changes in the Global Burden of Untreated Dental Caries from 1990 to 2019: a Systematic Analysis for the Global Burden of Disease Study. Heliyon 8(9), e10714; doi:10.1016/j.heliyon.2022.e10714 Renda, G., Gökkaya, I. and Şöhretoğlu, D. 2022. Immunomodulatory properties of terpenes. Phytochem. Rev. 21(2), 537–563; doi:10.1007/s11101-021-09785-x Rocha, J., Figueira, M.E., Barateiro, A. and Fernandes, A. 2014. Anti-inflammatory effect of Rosmarinic acid and Rosmarinus officinalis extract in rat models of local and systemic inflammation. Basic. Clin. Pharmacol. Toxicol. 116(5), 85–94; doi:10.1111/bcpt.12335 Rocha-Filho, P.A. and Maruno, M. 2025. Formulation, physical properties, and instability of transparent nanoemulsions containing sesame seed oil (SO) and raspberry seed oil (RO) and in vitro evaluation. Cosmetics 12(2), 32; doi:10.3390/cosmetics12020032 Rodrigues, P., Ruviaro, N.A. and Trevisan, G. 2022. Role of TRPV4 in neuropathic pain mechanisms in rats. Antioxidants 12(1), 24; doi:10.3390/antiox12010024 Salamatullah, A.M., Hayat, K., Arzoo, S., Alzahrani, A., Ahmed, M.A., Yehia, H.M., Alsulami, T., Al-Badr, N., Al-Zaied, B.A.M. and Althbiti, M.M. 2021. Effects of boiling technique-based food processing on the bioactive and antimicrobial properties of basil and rosemary. Molecules 26(23), 7373; doi:10.3390/molecules26237373 Savi, F.M., Brierly, G.I., Baldwin, J., Theodoropoulos, C. and Woodruff, M.A. 2017. Comparison of different decalcification methods using rat mandibles as a model. J. Histochem. Cytochem. 65(12), 705–722; doi:10.1369/0022155417733708 Skoff, A.M., Zhao, C. and Adler, J.E. 2009. Interleukin-1α regulates substance P expression and release in adult sensory neurons. Exp. Neurol. 217(2), 395–400; doi:10.1016/j.expneurol.2009.03.022 Soares, C.L.R., Wilairatana, P., Silva, L.R., Moreira, P.S., Vilar Barbosa, N.M.M., Da Silva, P.R., Coutinho, H.D.M., De Menezes, I.R.A. and Felipe, C.F.B. 2023. Biochemical aspects of the inflammatory process: a narrative review. Biomed. Pharmacotherapy 168, 115764; doi:10.1016/j.biopha.2023.115764 Symmank, J., Löffler, L., Schulze-Späte, U. and Jacobs, C. 2025. Bidirectional substance P signaling between PL fibroblasts and sensory neurons under mechanical stress. Front. Mol. Neurosci. 18, 1583908; doi:10.3389/fnmol.2025.1583908 Tu, C., Wang, G., Hu, Z., Wang, S., Yan, Q. and Liu, X. 2023. The burden of oral disorders, 1990–2019: estimates from the Global Burden of Disease Study 2019. Arch. Med. Sci. 19(4), 930–940; doi:10.5114/aoms/165962 Ulanowska, M. and Olas, B. 2021. Biological properties and prospects for the application of Eugenol—a review. Int. J. Med. Sci. 22(7), 3671; doi:10.3390/ijms22073671 Valentim, A.M., Guedes, S.R., Pereira, A.M. and Antunes, L.M. 2016. Euthanasia using gaseous agents in rodents. Lab. Anim. 50(4), 241–253; doi:10.1177/0023677215618618 Vieira, I.R.S., Silva, A.C.L.N., Castro, N.R., Pinto, C.D.S.C., Freitas, Z.M.F.D., Ricci-Júnior, E., Santos, E.P.D., Camara, A.L., Costa, M.C.P. and Adam Conte-junior, C. 2023. Development and characterization of photoprotective nanoemulsions containing Babassu (Orbignya phalerata Mart.) lipophilic extract. Braz. J. Pharm. Sci. 59, e23011; doi:10.1590/s2175-97902023e23011 Wang, X., Wu, Z., Zhang, Y., Lian, B., Ma, L. and Zhao, J. 2024. Hypoxia-induced autophagy in pulpitis is mediated by HIF-1α/BNIP3. Arch. Oral. Biol. 159, 105881; doi:10.1016/j.archoralbio.2024.105881 Warreth. and A. 2023. Dental caries and management. Int. J. Dentistry 1, 1–15; doi:10.1155/2023/9365845 Wautier, J.L., Wautier, M.P. 2023. Pro- and anti-inflammatory prostaglandins and cytokines in humans: a mini review. IJMS 24(11), 9647; doi:10.3390/ijms24119647 Yadav, N., Singh Chandel, S., Venkatachalam, T. and Fathima, S.N. 2023. Herbal medicine formulation, standardization, and commercialization challenges and sustainable strategies for improvement. In: Herbal medicine phytochemistry. Reference series in phytochemistry. Eds., Izah, S.C., Ogwu, M.C., Akram, M. Cham, Switzerland: Springer. Zhu, Z. and Bhatia, M. 2023. Inflammation and organ injury the role of substance P and its receptors. IJMS 24(7), 6140; doi:10.3390/ijms24076140 | ||
| How to Cite this Article |
| Pubmed Style Nugraeni Y, Putri AH, Kamaruzzaman AR, Virigirinia RP, Priyanto R, Riyawan W. Efficacy of Rosmarinus officinalis nanoemulsion as an anti-inflammatory and antinociceptive agent by targeting the interleukin-1β, substance P, and neurokinin-1 receptor pathways in experimental pulpitis. Open Vet. J.. 2026; 16(3): 1578-1589. doi:10.5455/OVJ.2026.v16.i3.15 Web Style Nugraeni Y, Putri AH, Kamaruzzaman AR, Virigirinia RP, Priyanto R, Riyawan W. Efficacy of Rosmarinus officinalis nanoemulsion as an anti-inflammatory and antinociceptive agent by targeting the interleukin-1β, substance P, and neurokinin-1 receptor pathways in experimental pulpitis. https://www.openveterinaryjournal.com/?mno=287765 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.15 AMA (American Medical Association) Style Nugraeni Y, Putri AH, Kamaruzzaman AR, Virigirinia RP, Priyanto R, Riyawan W. Efficacy of Rosmarinus officinalis nanoemulsion as an anti-inflammatory and antinociceptive agent by targeting the interleukin-1β, substance P, and neurokinin-1 receptor pathways in experimental pulpitis. Open Vet. J.. 2026; 16(3): 1578-1589. doi:10.5455/OVJ.2026.v16.i3.15 Vancouver/ICMJE Style Nugraeni Y, Putri AH, Kamaruzzaman AR, Virigirinia RP, Priyanto R, Riyawan W. Efficacy of Rosmarinus officinalis nanoemulsion as an anti-inflammatory and antinociceptive agent by targeting the interleukin-1β, substance P, and neurokinin-1 receptor pathways in experimental pulpitis. Open Vet. J.. (2026), [cited March 31, 2026]; 16(3): 1578-1589. doi:10.5455/OVJ.2026.v16.i3.15 Harvard Style Nugraeni, Y., Putri, . A. H., Kamaruzzaman, . A. R., Virigirinia, . R. P., Priyanto, . R. & Riyawan, . W. (2026) Efficacy of Rosmarinus officinalis nanoemulsion as an anti-inflammatory and antinociceptive agent by targeting the interleukin-1β, substance P, and neurokinin-1 receptor pathways in experimental pulpitis. Open Vet. J., 16 (3), 1578-1589. doi:10.5455/OVJ.2026.v16.i3.15 Turabian Style Nugraeni, Yuli, Agustine Hanafi Putri, Abdur Razaq Kamaruzzaman, Regina Putri Virigirinia, Rahmavidyanti Priyanto, and Wibi Riyawan. 2026. Efficacy of Rosmarinus officinalis nanoemulsion as an anti-inflammatory and antinociceptive agent by targeting the interleukin-1β, substance P, and neurokinin-1 receptor pathways in experimental pulpitis. Open Veterinary Journal, 16 (3), 1578-1589. doi:10.5455/OVJ.2026.v16.i3.15 Chicago Style Nugraeni, Yuli, Agustine Hanafi Putri, Abdur Razaq Kamaruzzaman, Regina Putri Virigirinia, Rahmavidyanti Priyanto, and Wibi Riyawan. "Efficacy of Rosmarinus officinalis nanoemulsion as an anti-inflammatory and antinociceptive agent by targeting the interleukin-1β, substance P, and neurokinin-1 receptor pathways in experimental pulpitis." Open Veterinary Journal 16 (2026), 1578-1589. doi:10.5455/OVJ.2026.v16.i3.15 MLA (The Modern Language Association) Style Nugraeni, Yuli, Agustine Hanafi Putri, Abdur Razaq Kamaruzzaman, Regina Putri Virigirinia, Rahmavidyanti Priyanto, and Wibi Riyawan. "Efficacy of Rosmarinus officinalis nanoemulsion as an anti-inflammatory and antinociceptive agent by targeting the interleukin-1β, substance P, and neurokinin-1 receptor pathways in experimental pulpitis." Open Veterinary Journal 16.3 (2026), 1578-1589. Print. doi:10.5455/OVJ.2026.v16.i3.15 APA (American Psychological Association) Style Nugraeni, Y., Putri, . A. H., Kamaruzzaman, . A. R., Virigirinia, . R. P., Priyanto, . R. & Riyawan, . W. (2026) Efficacy of Rosmarinus officinalis nanoemulsion as an anti-inflammatory and antinociceptive agent by targeting the interleukin-1β, substance P, and neurokinin-1 receptor pathways in experimental pulpitis. Open Veterinary Journal, 16 (3), 1578-1589. doi:10.5455/OVJ.2026.v16.i3.15 |