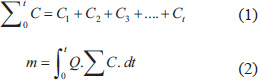Open Veterinary Journal, (2026), Vol. 16(3): -1792
Research Article
10.5455/OVJ.2026.v16.i3.35
A peat-burning smoke exposure model and its effects on rat
pulmonary tissue
Uly Astuti Siregar1,2, Dewi Sukmawati1,3, Agus Dwi Susanto4, Satirah Zainalabidin5,6,
Arif Budianto7 and Sri Widia A. Jusman8,9*
1Doctoral Program of Biomedical Science, Faculty of Medicine, Universitas Indonesia, DKI Jakarta, Indonesia
2Department of Histology, Faculty of Medicine, Universitas Abdurrab, Riau, Indonesia
3Department of Histology, Faculty of Medicine, Universitas Indonesia, DKI Jakarta, Indonesia
4Department of Pulmonology and Respiratory Medicine, Faculty of Medicine,
Universitas Indonesia, DKI Jakarta, Indonesia
5Center for Toxicology and Health Risk Research, Faculty of Health Sciences, Universiti Kebangsaan Malaysia, Kuala Lumpur, Malaysia
6Cardiovascular and Pulmonary (CardioResp) Research Group, Universiti Kebangsaan Malaysia, Bangi, Malaysia
7Physics Study Program, Universitas Mataram, Mataram , Indonesia
8Department of Biochemistry and Molecular Biology, Faculty of Medicine,
Universitas Indonesia, DKI Jakarta, Indonesia
9Center of Hypoxia & Oxidative Stress Studies, Faculty of Medicine, Universitas Indonesia, DKI Jakarta, Indonesia
*Corresponding Author: Sri Widia A. Jusman. Department of Biochemistry and Molecular Biology, Faculty of Medicine, Universitas Indonesia, DKI Jakarta, Indonesia. Email: sriwidiaaj [at] gmail.com
Submitted: 20/08/2025 Revised: 20/01/2026 Accepted: 04/02/2026 Published: 31/03/2026
© 2025 Open Veterinary Journal
This is an Open Access article distributed under the terms of the Creative Commons Attribution-Non Commercial-No Derivatives License (http://creativecommons.org/licenses/by-nc-nd/4.0/), which permits non-commercial re-use, distribution, and reproduction in any medium, provided the original work is properly cited, and is not altered, transformed, or built upon in any way.
Abstract
Background: The incidence of haze from Indonesian forest and peatland fires is a major concern due to its adverse health effects, particularly due to fine particulate matter (PM2.5) exposure, leading to adverse effects on the lungs.
Aim: This study designed a whole-body PM2.5 exposure chamber to develop a PM2.5 exposure model with varying concentrations and durations, and then evaluated its impact on lung tissue.
Methods: Thirty-six male Wistar rats were randomly divided into four groups: a control group and three treatment groups with PM2.5 exposure at concentrations of 300, 500, and 700 µg/m³ for 10, 20, and 30 days. The morphology of PM2.5 was characterized using a scanning electron microscope (SEM) equipped with an energy-dispersive X-ray spectroscopy (EDS) detector. Lung tissue histology was assessed using hematoxylin and eosin for lung injury scoring and Masson’s trichrome for the percentage of fibrotic areas.
Results: SEM-EDS results showed particles deposited within the alveolar cavity. The highest PM2.5 exposure group (700 µg/m³) had significantly higher lung tissue injury scores and fibrotic areas than the control group across all durations (p < 0.05).
Conclusion: Exposure to PM2.5 from peat land-burning smoke causes significant lung tissue injury and fibrosis in a dose-dependent manner in Wistar rats.
Keywords: Biomass-burning smoke, Histopathology, Lung injury, Peatland fire, PM2.5.
Introduction
During the dry season, peatland fires, which mostly occur due to land clearing for farming, are a common cause of haze incidents in Southeast Asia (SEA) (Adam et al., 2021; Nurhidayah et al., 2023). These occurred notably in 1997, 2006, 2015, 2019, and 2023, with hotspots mainly appearing in Sumatra and Kalimantan, Indonesia ( Fujii et al., 2019; Fujii et al., 2021; Kuwata, 2024). These fires release smoke that can widely spread across regions and impair air quality, with high concentrations of particulate matter (total suspended particles [TSP], PM10, and PM2.5) reaching up to 1,000 μg/m3 in source regions such as Riau Province (Fujii et al., 2021; Kuwata, 2024; Lestari et al., 2024). PM2.5, predominantly emitted from peatland burning, is known to carry carcinogenic risks from components such as Cr and Co (Siregar et al., 2022; Lestari et al., 2024). Furthermore, the PM analysis in Southern Thailand between June 2023 and May 2024 revealed that biomass burning tracers, such as levoglucosan and polycyclic aromatic hydrocarbons (PAHs), were most abundant in PM2.5 (Areerob et al., 2025).
Smoke from peatland fires, which mainly contains particulate matter (PM), is positively associated with lung injury from high exposure to toxic components (Fujii et al., 2019; Lan et al., 2021; Carberry et al., 2022). Epidemiological studies have confirmed these health impacts. For instance, haze incidents derived from peatland fires in Pekanbaru were associated with an elevation in respiratory diseases, predominantly presenting as cough and rhinorrhea. Exposure to high PM2.5 levels in Central Kalimantan during peatland fires (reaching an annual average of 26 μg/m³) was estimated to increase premature mortality, causing approximately 26 deaths per 100,000 population from chronic respiratory and cardiovascular events and lung cancer. The effects of PM2.5 exposure depend on various factors, including duration, intensity, chemical composition, and individual susceptibility (Sun et al., 2020; Shamsollahi et al., 2021; Lepistö et al., 2023).
Although epidemiological studies have confirmed an association between haze events and respiratory illness, the detailed cellular and tissue pathophysiological mechanisms remain poorly understood. This is particularly true for responses to specific concentrations of peatland smoke-derived PM. Controlled experimental studies using animal models are crucial for addressing this gap, allowing for tissue damage analysis that is not feasible in human subjects. This study aimed to develop a model of PM2.5 exposure in a laboratory setting using a chamber designed with Wistar rats as the research subjects. The model was designed to simulate the severe field conditions reported in Pekanbaru City during the 2019 haze event (Chaniago and Zahara, 2020). The findings of this study will provide a critical foundation for investigating the effects of PM2.5 exposure on cellular and tissue damage and will inform the development of preventive measures against organ damage, particularly in the lungs.
Materials and Methods
Animals
This study used male Wistar rats (N=36), aged 2–3 months and weighing 150–200 g. Before the experiments, the rats were acclimated for 7 days in cages at room temperature, maintained at 25°C ± 2°C and humidity 50% ± 5% with a 12-hours light/12-hours dark cycle. The rats were given pellets (Red Bio) and water ad libitum. For the experiments, the rats were randomly divided into four groups using a simple lottery method: a control group (A; n=3) and three treatment groups exposed to PM2.5 at concentrations of 300, 500, and 700 μg/m³ (B, C, and D; n=3 per group) (Fig. 1). Lung tissue assessments were performed after 10, 20, and 30 days of exposure (n=3 per group at each time point).
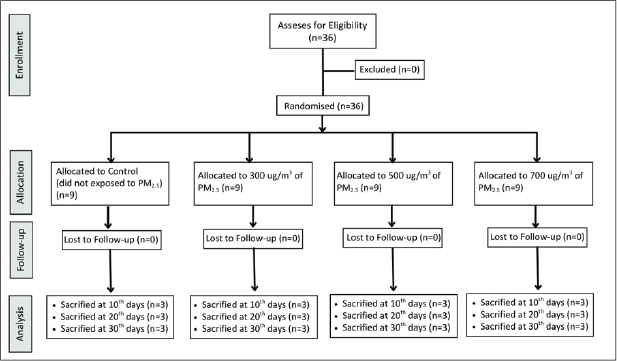
Fig. 1. The research design and timeline of PM2.5 exposure.
Peat biomass and exposure chamber
The peat biomass used in this study was obtained from a peatland in Pelalawan Regency, Riau Province, Sumatra, Indonesia, with a water content of ≤ approximately 15%. The water content of the peat biomass was measured using the gravimetric method (Wilkinson et al., 2019). The designed exposure chambers and sensors were installed in the Department of Physics, Faculty of Mathematics and Natural Sciences, Brawijaya University’s Air Quality and Health Impact Laboratory. This setup includes a combustion chamber, a smoke reservoir chamber (made of acrylic, 30 × 20 × 20 cm) equipped with a mini fan to ensures the smoke is homogenized before injection, a cyclone box, and an inhalation chamber (made of acrylic, 40 × 30 × 35 cm) (Wardoyo et al., 2018; Uly Astuti Siregar et al., 2023) (Fig. 2).
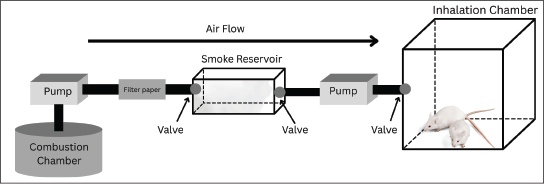
Fig. 2. The designed PM2.5 exposure chamber as a model for peat smoke exposure in a laboratory setting.
Measurement of PM2.5 and gas concentrations
PM2.5 and gas concentrations were measured using a system consisting of several sensors assembled with an Arduino Uno board. This study utilized a ZH03 sensor (Zhengzhou Winsen Electronics Technology Co., Ltd, China), an MiCS 4515 (SGX Sensortech, Switzerland), an MG-811 (GitHub, Inc.) sensor, an MQ-136 (Hanwei Electronics Co., Ltd, China) sensor, an ME-3 (Zhengzhou Winsen Electronics Technology Co., Ltd, China) sensor, and an MICS-4514 (SGX Sensortech, Switzerland). These sensors were used to identify and measure the concentrations of PM2.5, CO, CO2, SO2, O3, and NO2, respectively. All sensors were calibrated (average accuracy > 91%) using a Piezobalance Dust Monitor (Kanomax, model 3522), an Ozone Detector, and a Gas Analyzer (Sauermann, model SI-CA 130) (Amalia et al., 2021; Widhowati et al., 2021) (Fig. 3). The PM2.5 concentration in the exposure chamber was determined by summing the PM2.5 concentrations (Ct) based on the time of emission injection (t). The gas concentration was calculated as the total of the injected concentrations.
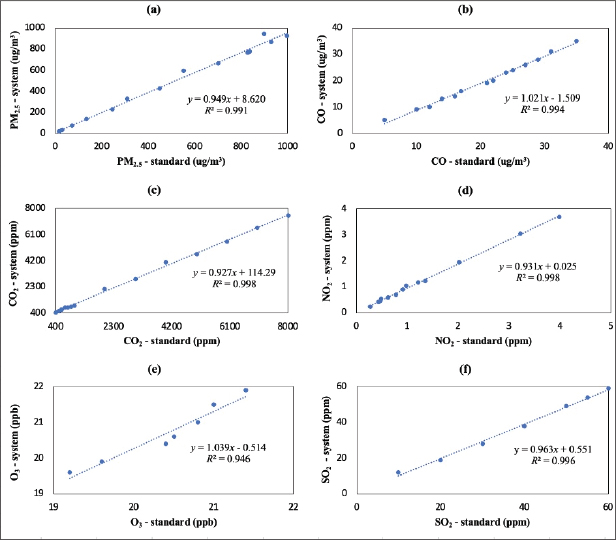
Fig. 3. Calibration curves of the used sensors: (a) PM2.5, (b) CO, (c) CO2, (d) NO2, (e) O3, and (f) SO2.
PM2.5 Smoke exposure procedures
Peat biomass (m=3 g) was ignited in the combustion chamber using a lighter for 30 seconds. Following the activation of the combustion pump, the reservoir chamber’s input valve was opened, while the valve connecting the reservoir chamber and the inhalation chamber was closed. The smoke collected in the reservoir chamber was filtered using Whatman filter paper No. 42 to isolate particles with a diameter of less than 2.5 μm (PM2.5). The reservoir chamber was completed with a cyclone system consisting of a fan (for dilution) and a microorifice (for filtering particles with a diameter > 2.5 μm) (Budianto et al., 2023). Subsequently, the inlet valve of the reservoir chamber was shut, and the outlet valves of both the reservoir chamber and inhalation chamber were opened to inject PM2.5 into the inhalation chamber, where the Wistar rats were housed. The chamber was purged with filtered air for 5 minutes after each daily exposure session to remove residual aerosols. The concentration of PM2.5 in the exposure chamber was adjusted to the expected levels of 300 μg/m³, 500 μg/m³, and 700 μg/m³ by controlling the emission injection time (t). The Wistar rats were kept in the sealed inhalation chamber for 100 s per day. All exposure treatments were conducted using a gravimetric principle at a constant flow rate (Q) of 80 cm3/s and a surface area (A) of 0.50 cm2 (Budianto et al., 2023).
Histopathological and SEM examination of lung tissue
At the end of the treatment procedures, lung tissue samples were collected after 10, 20, and 30 days of exposure to PM2.5. The rats were euthanized using the exsanguination method following sedation with intraperitoneal injection of ketamine (0.1 cc) and Xylazine (0.05 cc). The lung tissue was fixed in a 10% NBF solution, left at room temperature for 48 hours, and then processed for scanning electron microscope/energy-dispersive X-ray spectroscopy (SEM-EDS) analysis and histopathological examination with hematoxylin and eosin (H&E) and Masson’s trichrome (MT) staining (Ji et al., 2023).
To prepare the lung tissue samples, the fixed tissue was dehydrated using ethanol solutions in increasing percentages (70%–80%–90%–100%–100%, each for 1 hour), cleared with xylene, and embedded in liquid paraffin. Subsequently, the paraffin-embedded tissue was sectioned at 4 µm thickness using a microtome, followed by deparaffinization in xylene (twice, each for 2 minutes) and rehydration using ethanol solutions in decreasing percentages (100%–95%–80%–70%, each for 5 minutes). The slides were air-dried at room temperature, mounted on carbon tape, and coated with a conductive layer using an ion-sputtering machine.
The morphology of lung tissue and particles was examined using a JEOL JSM 6510-LA SEM equipped with EDS detector (Adhika et al., 2018). The SEM/EDS analysis is preliminary to illustrate the general elemental nature of the particles deposited from one sample per group (n=1). The quantification of the deposited particle area was performed on SEM images using a standardized ImageJ (NIH) pipeline. In brief, the images analyzed in ImageJ were converted to a grayscale (8-bit) image. The alveolar spaces were manually outlined and isolated to define the ROI. To create a binary image, an automated global brightness threshold was applied within the ROI, and the “Analyze Particles" command was used to measure the total particle area (Sinkhonde et al., 2025). This particle area was then normalized to the total area of the ROI using the following formula: percentage=(Total Particle Area/Total Alveolar ROI Area) × 100%. The final value was reported as the ″Percentage of particle area in the alveolar cavity″.
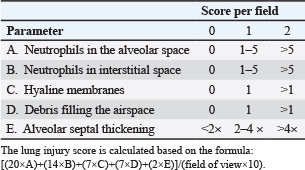
The severity of lung injury in the histopathology slides stained with hematoxylin and eosin was determined based on the lung injury scoring system by the American Thoracic Society (Table 1). For lung injury scoring, five random fields from each slide were selected at 400x magnification under a light microscope (Olympus CX23). The lung injury score was calculated using the following formula: [(20×A)+(14×B)+(7×C)+(7×D)+(2×E)]/(field of view×10) (Song et al., 2019). Furthermore, quantification of the fibrosis area on MT-stained slides was performed using an ImageJ (NIH) pipeline. In brief, five random areas of each slide were captured at 400× magnification using a digital camera (Optilab Advance) on a light microscope (Olympus CX23). The images were first analyzed using the ″Color Deconvolution″ to isolate the blue channel (collagen) from the tissue. Then, the automated global threshold was applied to create a binary image of the fibrotic area. The ″Analyze Measure″ was commanded to measure the total blue-positive area. The final value was reported as the percentage of the fibrotic area (Song et al., 2019). Pathologists performed histopathological analysis in a blinded manner (group allocation and time point).
Table 1. Lung injury scoring system of the American Thoracic Society (2011).
Data analysis
Data analysis was carried out using the Kruskal–Wallis test, followed by Dunn’s post hoc test. The results were presented as medians in graphs using GraphPad Prism 10.3.0. A significant difference between groups was defined as p < 0.05.
Ethical approval
The procedures for animal care were approved by the Ethics Committee of the Faculty of Medicine, Universitas Indonesia (No. KET-1754/UN2.F1/ETIK/PPM.00.02/2023).
Results
PM2.5 and gas concentrations
Measurements of PM2.5 and gas concentrations were measured every 2 seconds using the developed system, relying on the emission injection time. The obtained concentrations of PM2.5 and gas are shown in Figure 4. The total exposed concentrations of PM2.5 (Ct) were calculated using Equation (1) in units of μg/m3. The total concentration is interpreted as a continuous summation. When applied to a mass function, the integral calculates the total amount of the exposed mass (m) over a specific exposure time (Δt) and flow rate (Q) (Eq. 2). In line with this, this study has three PM2.5 concentration variations: C1 (68 s, 300 µg/m3), C2 (96 s, 500 µg/m3), and C3 (124 s, 700 µg/m3).
Fig. 4. Resulting data of PM2.5 concentration measurements at different times: (a) C1, (b) C2, (c) C3, and (d) total exposed PM2.5 concentrations.
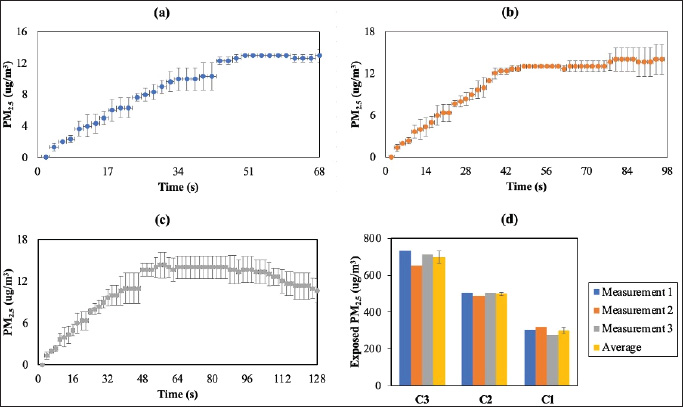
The gas concentration in the inhalation chamber was determined by calculating the average values of CO2 concentration at emission injection times of 68, 96, and 124 seconds, namely 423, 449, and 427 ppm, respectively. Meanwhile, throughout the exposure period, the ozone (O3) concentration remained constant at 20 ppb, whereas the concentrations of CO, SO2, and NO2 remained constant. These results indicate that the gas concentrations were very steady (no fluctuation or significant change detected). The exposed smoke might contain several gases with concentrations below the sensor detection limitations (below the sensor’s LOD) and the sensor’s resolution.
Particle morphology in the alveolar cavity
The SEM images at 3,000× magnification are presented in Figure 5, depicting the rats’ lung tissue across different groups with particles in the alveolar cavity. The presence of PM2.5 particles in the alveolar cavity was quantified in percentages. All treatment groups exhibited a higher percentage of PM2.5 particles in the alveolar cavity than the control group. Specifically, the treatment group exposed to PM2.5 at a concentration of 700 µg/m3 had the highest percentage of particle area (18.026%), followed by the 500 µg/m3 and 300 µg/m3 groups at 14.996% and 12.269%, respectively (Fig. 6).

Fig. 5. SEM images of rat lung tissue from different exposure groups at 3,000× magnification with a scale bar of 5 μm. A. Control group, B10. 300 µg/m3 exposure for 10 days, B20. 300 µg/m3 exposure for 20 days, B30. 300 µg/m3 exposure for 30 days, C10. 500 µg/m3 exposure for 10 days, C20. 500 µg/m3 exposure for 20 days, C30. 500 µg/m3 exposure for 30 days, D10. 700 µg/m3 exposure for 10 days, D20. 700 µg/m3 exposure for 20 days, D30. 700 µg/m3 exposure for 30 days.
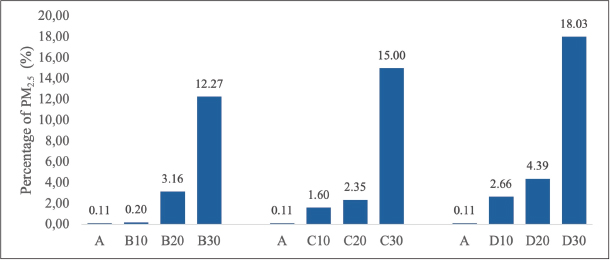
Fig. 6. Quantification of the presence (percentage) of PM2.5 in the alveolar cavity. A. Control group, B10. 300 µg/m3 exposure for 10 days, B20. 300 µg/m3 exposure for 20 days, B30. 300 µg/m3 exposure for 30 days, C10. 500 µg/m3 exposure for 10 days, C20. 500 µg/m3 exposure for 20 days, C30. 500 µg/m3 exposure for 30 days, D10. 700 µg/m3 exposure for 10 days, D20. 700 µg/m3 exposure for 20 days, D30. 700 µg/m3 exposure for 30 days.
EDS results
The elemental components of the smoke were analyzed using an EDS detector. The results are presented in Table 2, which shows that carbon (C) and oxygen (O) are the predominant elements in all samples. Some samples also contain Na, Ca, and Co. Other elements, namely, arsenic (As), selenium (Se), and bromine (Br), are present in varying amounts.
Table 2. EDS results on SEM images of 3,000x magnification.
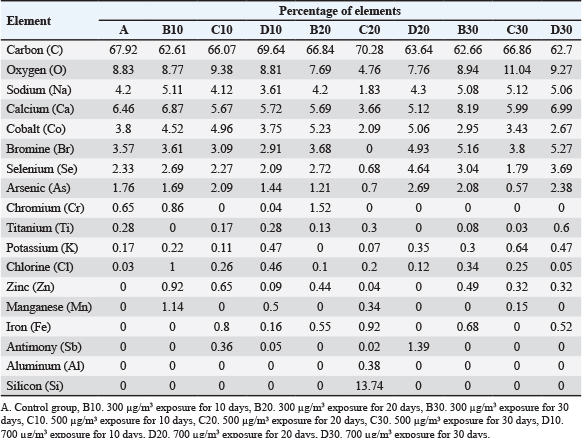
Histopathological analysis
Based on the results of the lung injury assessment (Fig. 7), a Kruskal-Wallis test was performed to compare the lung injury scores across the groups at each time point. On day 10, the test revealed a statistically significant difference between the groups (H(3)=9.585, p=0.0008). On day 20, a significant difference was observed (H(3)=9.186, p=0.0027). At day 30, the difference remained significant, H(3)=10.01, p=0.0003. Post-hoc analysis using Dunn’s multiple comparisons test showed that the 700 µg/m³ group had significantly increased the severity of lung injury in all exposure durations (p < 0.05). Furthermore, collagen fiber deposition analysis (Fig. 8) also revealed a significant difference in the percentage of fibrotic area between groups at day 10 (H(3)=9.667. p=0.0006), day 20 (H(3)=9.462. p=0.0014), and day 30 (H(3)=9.154. p=0.0028). Dunn’s post-hoc test also confirmed that the 700 µg/m³ group significantly increased the fibrotic area compared to the control group at all timepoints (p < 0.05).
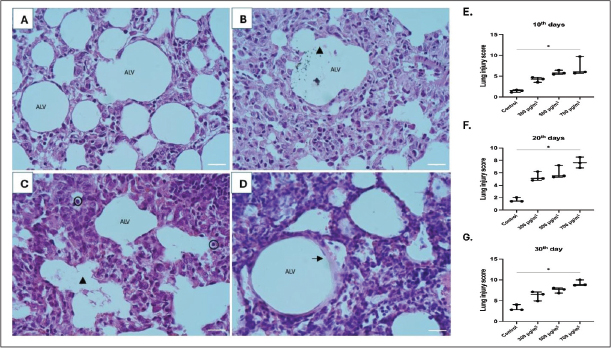
Fig. 7. Histopathological images of the lung with hematoxylin and eosin staining at 400x magnification, scale bar 50 μm. A. Control group, B–D. PM2.5 exposure groups, E–G. Comparison of lung injury scores in varying exposure durations (Kruskal-Wallis test, *p < 0.05, n=3). ALV: alveolus; triangle: debris in the alveolar cavity; circle: neutrophils; arrow: hyaline membrane.
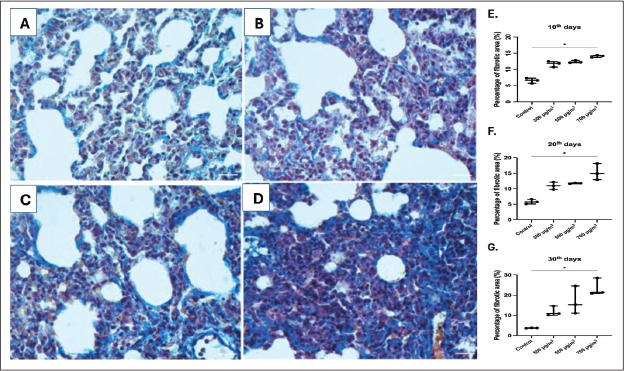
Fig. 8. Histopathological image of the lung with MT staining at 400× magnification, scale bar 50 μm. A. Control group, B–D. PM2.5 exposure groups, E–G. Comparison of lung injury scores in varying exposure durations (Kruskal-Wallis test, *p < 0.05, n=3).
Discussion
Laboratory research on PM2.5 exposure provides valuable data and insights, particularly regarding its emissions and health impacts. Previous studies have developed two methods for PM2.5 exposure models: the nose-only and whole-body exposure methods (Peng et al., 2019; Park et al., 2021; Lee et al., 2025). This study used the Sumatran peatland in Riau Province with a water content of ≤ 15%. This accurately reflects the peak dry season field conditions, as the topsoil water content in Sumatran peat can drop to as low as 8.7-12.4% during simulated droughts. (Taufik et al., 2022 ). Watson et al. (2019) developed a comparable peat smoke model using peat with a water content of 25% from various regions, including boreal (western Russia and Siberia), temperate (northern Alaska, USA), subtropical (northern and southern Florida), and tropical (Kalimantan, Malaysia) areas, to generate PM2.5 concentrations ranging from 328 to 2277 µg/m3 (Watson et al., 2019). Previous toxicology studies on the respiratory effects of peat smoke have used other peat sources, such as pocosin and Irish peat, which contain different chemical compounds than those found in Southeast Asian (SEA) peat. A study by Fujii et al. revealed that SEA peat smoke contains a higher proportion of organic carbon (70% of PM2.5 mass) than Irish or pocosin peat (20%–43%) (Martin et al., 2020; Fujii et al., 2021; Carberry et al., 2022; Lestari et al., 2024) and contains hazardous levels of polycyclic aromatic hydrocarbons (PAHs) ( Areerob et al., 2025). Therefore, the present study provides an essential assessment of the pulmonary health risks associated with the local phenomenon using Sumatran peat.
In the present study, a short duration of PM2.5 exposure (100s static period) daily was designed to investigate the repeated, acute, high-concentration effects of PM2.5 on the lungs. This method is adapted to haze conditions, where severe human exposure often involves intermittent exposure to high smoke concentrations in ambient air. The short duration of daily exposure does not imply a short biological effect. Aerosol (1–5 μm in diameter) penetrated the deep region of the lung, both using nasal and oral breathing (Corley et al., 2021; Herrmann et al., 2026). A study by Pan et al. (2023) using 89Zr-labeled PM2.5 mimic particles (89Zr-PM2.5) in rats confirmed this, demonstrating that >50% of the deposited particles remained in the lungs for >384 hours (16 days) after administration (Pan et al., 2023). Therefore, each daily dose of PM2.5 could cause chronic and sustained irritation and inflammation, resulting in pulmonary damage and fibrotic progression.
The elemental analysis of PM2.5 using an EDS detector showed that carbon (62.61%–70.28%) and oxygen (4.76%–11.04%) were the dominant components in all samples, confirming that the smoke originated from the combustion of organic material or biological tissues. These findings were followed by the presence of minerals and various toxic trace elements, such as Co, Mn, and Cr. The presence of these components strongly aligned with a previous study in Pekanbaru City (Sumatra, Indonesia), which identified biomass burning as the primary source of PM2.5, characterized by the presence of SO42–, K, Cl, Na, and Ca (Siregar et al., 2022). The dominant carbon and oxygen compounds can also form highly toxic secondary organic and inorganic aerosols (Longhin et al., 2016; Bora et al., 2021; Ma et al., 2025).
Many of the toxic elements detected in this study (Co, Mn, and Cr) harm organ health, particularly the respiratory system, and can cause systemic effects if they enter the body through the pulmonary capillaries (Siregar et al., 2022). The main mechanism by which these metal components, along with PAHs contained in PM2.5, result in damage is oxidative stress. This leads to intra- and extracellular redox imbalance, damaging lipid, protein, and DNA structures (Li et al., 2023b; Hou et al., 2024; Vilas-Boas et al., 2024). Metals such as iron (Fe) and copper (Cu) produce reactive radicals through the Fenton reaction, while the oxidation of trivalent arsenic [As (III)] to pentavalent arsenic [As (V)] contributes to the formation of H2O2 (Sulong et al., 2019; Hou et al., 2024).
This study revealed that higher concentrations of PM2.5 exposure are associated with elevated levels of lung damage, as indicated by higher lung injury scores. This damage is characterized by the infiltration of inflammatory cells in the interstitial and alveolar spaces, the presence of protein residue in the alveolar space, hyaline membranes, and the thickening of the interalveolar septum in the exposure groups compared to the control. Our findings are consistent with the existing literature showing that even short-term exposure to high concentrations of PM2.5 can cause tissue damage and changes in innate immunity mechanisms (Yang et al., 2019; Siregar et al., 2022; Li et al., 2023a). The proposed mechanism involves macrophages and neutrophils clearing inhaled particles, which then release proinflammatory cytokines (IL-1, IL-6, and TNF-α). In cases of chronic tissue injury, this sustained inflammation can lead to abnormal tissue repair responses, involving fibroblasts and myofibroblasts, and potentially drive EMT (Liu et al., 2018; Niu et al., 2022). Exposure to high concentrations of PM2.5 from peat smoke significantly enhanced collagen deposition in a dose-dependent manner. Although this animal model demonstrates fibrotic progression, supporting data on the correlation between haze incidents and lung fibrosis in the human population are currently lacking. Therefore, future studies are needed in two key areas: human cohort studies to investigate the long-term effects of haze on lung fibrosis and molecular research to identify the potential underlying mechanisms and pathways driving the fibrotic response to peat smoke-derived PM2.5.
Study limitations
This study has several limitations. As this was a preliminary study, a minimal sample size of n=3 per group was used, adhering to the 3Rs principles of reduction. Although this design meets the minimum statistical threshold, the low sample size limits the statistical power. Therefore, the findings should be interpreted as foundational. Second, SEM/EDS analysis was performed on only one sample per group (n=1). This was intended only as a preliminary assessment to confirm the presence of smoke-derived elements; consequently, these data cannot be used for statistical comparisons between groups. Finally, the scope of this study was limited to histopathological analysis and did not include detailed chemical speciation of the aerosol (e.g., PAH) or the measurement of molecular biomarkers (e.g., inflammatory cytokines, oxidative stress markers). Future research is crucial to validate and expand upon these findings, using larger animal cohorts, detailed molecular analysis, and also investigating the effects of long-term exposure to low doses of PM2.5.
Conclusion
A peat smoke exposure chamber was successfully developed as a model for PM2.5 exposure in a laboratory setting. Exposure to PM2.5 from peatland biomass-burning smoke causes significant lung tissue injury in Wistar rats in a dose-dependent manner. This study showed that all exposure durations to high concentrations of PM2.5 are critical factors in the progression to PF.
Acknowledgments
The authors would like to express their deepest gratitude to Prof. Arinto Yudi Ponco Wardoyo, M.Sc., Ph.D., for his expert technical assistance.
Funding
This study was supported by the Indonesian Education Scholarship (BPI), the Center for Higher Education Funding and Assessment, the Ministry of Higher Education, Science, and Technology of the Republic of Indonesia, and the Indonesia Endowment Fund for Education Agency (LPDP), Ministry of Finance of the Republic of Indonesia.
Authors’ contributions
U.A. conceptualization, project administration, writing and editing; D.S. conceptualization, writing-review and editing, supervision; A.D.S. review and supervision; S.W.A. conceptualization, writing-review and editing, supervision; S.Z. review and supervision; A.B. methodology, review and editing.
Conflict of interest
The authors declare no conflict of interest in this study.
Data availability
All data supporting this study’s findings are available within the manuscript.
References
Adam, M.G., Tran, P.T.M., Bolan, N. and Balasubramanian, R. 2021. Biomass burning-derived airborne particulate matter in Southeast Asia: a critical review. J. Hazardous Mater. 407, 1–20; doi: 10.1016/j.jhazmat.2020.124760
Adhika, R., Anindya, A. L., Tanuwijaya, V. V. and Rachmawati, H. 2018. Teknik Pengamatan Sampel Biologi dan Non-konduktif Menggunakan Scanning Electron Microscopy. 1–5.
Amalia, R., Wardoyo, A. Y. P., Dharmawan, H. A., Nurhuda, M. and Budianto, A. 2021. Development of a measurement system for volcanic CO and CO2 concentrations. IEEE Xplore, 1–6. doi: 10.1109/ISESD53023.2021.9501595
Areerob, T., Kleangklao, B. and Wongniramaikul, W. 2025. Environmental Technology & Innovation Spatiotemporal distribution of PAHs and levoglucosan in fine and ultrafine PM across Southern Thailand : insights from a novel GC-MS approach. Environ. Technol. &. Innov. 40, 104550; doi:10.1016/j.eti.2025.104550
Bora, J., Deka, P., Bhuyan, P., Sarma, K.P. and Hoque, R.R. 2021. Morphology and mineralogy of ambient particulate matter over mid-Brahmaputra Valley: application of SEM–EDX, XRD, and FTIR techniques. SN. Appl. Sci. 3(1), 1–15; doi:10.1007/s42452-020-04117-8
Budianto, A., Wirawan, R., Ramadian Ridho Illahi., Dian Wijaya Kurniawidi., Rahayu, S. and A.A. Ngurah Nara Kusuma. 2023. A Gravimetry-Based Fine Particle Concentration Measurement System for Humid Environment Using Graphene Oxide Layer. Evergreen 10(3), 1414–1421.
Carberry, C.K., Koval, L.E., Payton, A., Hartwell, H., Ho Kim, Y., Smith, G.J., Reif, D.M., Jaspers, I., Ian Gilmour, M. and Rager, J.E. 2022. Wildfires and extracellular vesicles: exosomal MicroRNAs as mediators of cross-tissue cardiopulmonary responses to biomass smoke. Environ. Int. 167, 1–36; doi:10.1016/j.envint.2022.107419
Chaniago, D. and Zahara, A. 2020. Kondisi Kualitas Udara di Beberapa Kota Besar Tahun 2019. Portal Direktorat Pengendalian Pencemaran Udara Ditjen Ppkl Klhk, 2020. Available via https://ditppu.menlhk.go.id/portal/read/kondisi-kualitas-udara-di-beberapa-kota-besar-tahun-2019
Corley, R.A., Kuprat, A.P., Suffield, S.R., Kabilan, S., Hinderliter, P.M., Yugulis, K. and Ramanarayanan, T.S. 2021. New approach methodology for assessing inhalation risks of a contact respiratory cytotoxicant: computational fluid dynamics-based aerosol dosimetry modeling for cross-species and in vitro comparisons. Toxicological Sci. 182(2), 243–259; doi:10.1093/toxsci/kfab062
Fujii, Y., Huboyo, H.S., Tohno, S., Okuda, T. and Syafrudin. 2019. Chemical speciation of water-soluble ionic components in PM2.5 derived from peatland fires in Sumatra Island. Atmos. Pollut. Res. 10(4), 1260–1266; doi:10.1016/j.apr.2019.02.009
Fujii, Y., Tohno, S., Kurita, H., Huboyo, H.S. and Zaman, B. 2021. Characteristics of organic components in PM2.5 emitted from peatland fires on Sumatra in 2015: Significance of humic-like substances. Atmospheric Environ. X, 11, 1–7; doi.org/10.1016/j.aeaoa.2021.100116
Herrmann, A., Puente, L., Ma-Hock, L. and Stratmann, H. 2026. Comparative assessment of 24 pigment dusts reveals analogue inhalation effects. Regulatory Toxicol. Pharmacol. 164, 1–7; doi.org/10.1016/j.yrtph.2025.105934
Hou, T., Zhu, L., Wang, Y. and Peng, L. 2024. Oxidative stress is the pivot for PM2.5-induced lung injury. Food. Chem. Toxicol. 184, 1–11; doi:10.1016/j.fct.2023.114362
Ji, D., Hu, C., Ning, J., Ying, X., Zhang, H., Zhang, B., Liu, B., Liu, Q., Ji, W. and Zhang, R. 2023. N6-methyladenosine mediates Nrf2 protein expression involved in PM2.5-induced pulmonary fibrosis. Ecotoxicol. Environ. Saf. 254, 114755; doi:10.1016/j.ecoenv.2023.114755
Kuwata, M. 2024. Atmospheric emissions, processes, and impacts of tropical peatland fire haze in Equatorial Asia: a review. Atmos. Environ. 331, 1–19; doi:10.1016/j.atmosenv.2024.120575
Lan, Y., Tham, J., Jia, S., Sarkar, S., Fan, W.H., Reid, J.S., Ong, C.N. and Yu, L.E. 2021. Peat-forest burning smoke in Maritime Continent: impacts on receptor PM2.5 and implications at emission sources. Environ. Pollut. 275, 1–9; doi:10.1016/j.envpol.2021.116626
Lee, J.U., Nam, Y., Baek, J., Park, E., Hong, J., Choi, Y.M., Kim, S.H., Jeong, S.H., Kim, W.J. and Park, S.W. 2025. Progressive lung fibrosis in aged mice induced by repetitive bleomycin: exacerbation by long-term PM2.5 inhalation. Ecotoxicol. Environ. Saf. 304, 119146; doi:10.1016/j.ecoenv.2025.119146
Lepistö, T., Lintusaari, H., Oudin, A., Barreira, L.M.F., Niemi, J.V., Karjalainen, P., Salo, L., Silvonen, V., Markkula, L., Hoivala, J., Marjanen, P., Martikainen, S., Aurela, M., Reyes, F.R., Oyola, P., Kuuluvainen, H., Manninen, H.E., Schins, R.P.F., Vojtisek-Lom, M., Ondracek, J., Topinka, J., Timonen, H., Jalava, P., Saarikoski, S. and Rönkkö, T. 2023. Particle lung deposited surface area (LDSAal) size distributions in different urban environments and geographical regions: towards understanding of the PM2.5 dose–response. Environ. Int. 180, 1–16; doi:10.1016/j.envint.2023.108224
Lestari, P., Tasrifani, A.R., Suri, W.I., Wooster, M.J., Grosvenor, M.J., Fujii, Y., Ardiyani, V., Carboni, E. and Thomas, G. 2024. Gaseous, particulate matter, carbonaceous compound, water-soluble ion, and trace metal emissions measured from 2019 peatland fires in Palangka Raya, Central Kalimantan. Atmospheric Environ. 316, 1–8; doi.org/10.1016/j.atmosenv.2023.120171
Li, Y., Fan, Z., Lu, W., Xu, R., Liu, T., Liu, L., Chen, G., Lv, Z., Huang, S., Zhou, Y., Liu, Y. and Sun, H. 2023a. Chemosphere Long-term exposure to ambient fine particulate matter-bound polycyclic aromatic hydrocarbons and cancer mortality : a difference-in-differences approach. Chemosphere 340, 1–10; doi:10.1016/j.chemosphere.2023.139800
Li, Y., Lin, B., Hao, D., Du, Z., Wang, Q., Song, Z., Li, X., Li, K., Wang, J., Zhang, Q., Wu, J., Xi, Z. and Chen, H. 2023b. Short-term PM2.5 exposure induces transient lung injury and repair. J. Hazardous. Mater. 459, 1–17; doi:10.1016/j.jhazmat.2023.132227
Liu, C.W., Lee, T.L., Chen, Y.C., Liang, C.J., Wang, S.H., Lue, J.H., Tsai, J.S., Lee, S.W., Chen, S.H., Yang, Y.F., Chuang, T.Y. and Chen, Y.L. 2018. PM2.5-induced oxidative stress increases intercellular adhesion molecule-1 expression in lung epithelial cells through the IL-6/AKT/STAT3/NF-ΚB-dependent pathway. Part. Fibre Toxicol. 15(1), 1–16; doi:10.1186/s12989-018-0240-x
Longhin, E., Gualtieri, M., Capasso, L., Bengalli, R., Mollerup, S., Holme, J.A., Øvrevik, J., Casadei, S., Di Benedetto, C., Parenti, P. and Camatini, M. 2016. Physico-chemical properties and biological effects of diesel and biomass particles *. Environ. Pollut. 215, 366–375; doi:10.1016/j.envpol.2016.05.015
Ma, J., Liu, J., Chen, J., Pang, X., Jia, B., Zhao, G. and Cheng, P. 2025. Laboratory investigations into secondary organic aerosol derived from polycyclic aromatic hydrocarbons: formation processes, underlying mechanisms, and implications for health. Atmos. Environ. 360, 121415; doi:10.1016/j.atmosenv.2025.121415
Martin, B.L., Thompson, L.C., Kim, Y.H., King, C., Snow, S., Schladweiler, M., Haykal-Coates, N., George, I., Gilmour, M.I., Kodavanti, U.P., Hazari, M.S. and Farraj, A.K. 2020. Peat Smoke Inhalation Alters Blood Pressure, Baroreflex Sensitivity and Cardiac Arrhythmia Risk in Rats. J. Toxicol. Environ. Health A 83(23–24), 1–24; doi:10.1080/15287394.2020.1826375
Niu, R., Cheng, J., Sun, J., Li, F., Fang, H., Lei, R., Shen, Z., Hu, H. and Li, J. 2022. Alveolar Type II Cell Damage and Nrf2-SOD1 Pathway Downregulation Are Involved in PM2.5-Induced Lung Injury in Rats. Int. J. Environ. Res. Public Health 19, D1–14; doi:10.3390/ijerph191912893
Nurhidayah, L., Astuti, R., Hidayat, H. and Siburian, R. 2023. Community-Based Fire Management and Peatland Restoration in Indonesia. doi: 10.1007/978-3-031-15904-6_8
Pan, D., Xu, Y., Wang, X., Wang, L., Yan, J., Shi, D., Yang, M. and Chen, M. 2023. Evaluation the in vivo behaviors of PM2.5 in rats using noninvasive PET imaging with mimic particles. Chemosphere 339, 139663; doi:10.1016/j.chemosphere.2023.139663
Park, S.Y., An, K.S., Lee, B., Kang, J.H., Jung, H.J., Kim, M.W., Ryu, H.Y., Shim, K.S., Nam, K.T., Yoon, Y.S. and Oh, S.H. 2021. Establishment of particulate matter-induced lung injury model in mouse. Lab. Anim. Res. 37(20), 1–12.
Peng, X., Maltz, M.R., Botthoff, J.K., Aronson, E.L., Nordgren, T.M., Lo, D.D. and Cocker, D.R. 2019. Establishment and characterization of a multi-purpose large animal exposure chamber for investigating health effects. Rev. Scientific Instrum. 90, 1–10; doi:10.1063/1.5042097
Shamsollahi, H.R., Jahanbin, B., Rafieian, S. and Yunesian, M. 2021. Particulates induced lung inflammation and its consequences in the development of restrictive and obstructive lung diseases: a systematic review. Environ. Sci. Pollut. Res. 28(20), 25035–25050; doi:10.1007/s11356-021-13559-5
Sinkhonde, D., Bezabih, T. and Mirindi, D. 2025. Particle size distributions and shape identification of pozzolanic materials via various dimensional representations. Hybrid. Adv. 8, 100384; doi:10.1016/j.hybadv.2025.100384
Siregar, S., Idiawati, N., Lestari, P., Berekute, A.K., Pan, W.C. and Yu, K.P. 2022. Chemical Composition, Source Appointment and Health Risk of PM2.5 and PM2.5-10 during Forest and Peatland Fires in Riau, Indonesia. Aerosol Air Qual. Res. 22(9), 1–19; doi:10.4209/aaqr.220015
Song, L.C., Chen, X.X., Meng, J.G., Hu, M., Huan, J.B., Wu, J., Xiao, K., Han, Z.H. and Xie, L.X. 2019. Effects of different corticosteroid doses and durations on smoke inhalation-induced acute lung injury and pulmonary fibrosis in the rat. Int. Immunopharmacology 71, 392–403; doi:10.1016/j.intimp.2019.03.051
Sulong, N.A., Latif, M.T., Sahani, M., Khan, M.F., Fadzil, M.F., Tahir, N.M., Mohamad, N., Sakai, N., Fujii, Y., Othman, M. and Tohno, S. 2019. Distribution, sources and potential health risks of polycyclic aromatic hydrocarbons (PAHs) in PM2.5 collected during different monsoon seasons and haze episode in Kuala Lumpur. Chemosphere 219, 1–14; doi:10.1016/j.chemosphere.2018.11.195
Sun, B., Shi, Y., Li, Y., Jiang, J., Liang, S., Duan, J. and Sun, Z. 2020. Short-term PM2.5 exposure induces sustained pulmonary fibrosis development during post-exposure period in rats. J. Hazardous Mater. 385, 1–11; doi:10.1016/j.jhazmat.2019.121566
Taufik, M., Widyastuti, M. T., Sulaiman, A., Murdiyarso, D., Santikayasa, I. P. and Minasny, B. 2022. An improved drought-fire assessment for
managing fire risks in tropical peatlands. Agricult. Forest Meteorol. 312, 1–13; doi.org/10.1016/j.agrformet.2021.108738
Uly Astuti Siregar, U.A.S., May Valzon., Fitrianti. and Budianto, A. 2023. Effect of peat biomass smoke exposure on oxidative stress in Wistar rats. Jurnal. Kedokteran. Dan. Kesehatan. Indonesia. 2, 121–127; doi:10.20885/jkki.vol14.iss2.art2
Vilas-Boas, V., Chatterjee, N., Carvalho, A. and Alfaro-Moreno, E. 2024. Particulate matter-induced oxidative stress – Mechanistic insights and antioxidant approaches reported in in vitro studies. Environ. Toxicol. Pharmacol. 110, 1–17; doi:10.1016/j.etap.2024.104529
Wardoyo, A.Y.P., Juswono, U.P. and Noor, J.A.E. 2018. Varied dose exposures to ultrafine particles in the motorcycle smoke cause kidney cell damages in male mice. Toxicol. Rep. 5, 383–389; doi:10.1016/j.toxrep.2018.02.014
Watson, J.G., Cao, J., Chen, L.W.A., Wang, Q., Tian, J., Wang, X., Gronstal, S., Ho, S.S.H., Watts, A.C. and Chow, J.C. 2019. Gaseous, PM2.5 mass, and speciated emission factors from laboratory chamber peat combustion. Atmos. Chem. Phys. 19, 14173–14193; doi:10.5194/acp-19-14173-2019
Widhowati, A. A., Wardoyo, A. Y. P., Dharmawan, H. A., Nurhuda, M. and Budianto, A. 2021. Development of a portable volatile organic compounds concentration measurement system using a CCS811 air quality sensor. IEEE Xplore, 1–5. https://doi.org/10.1109/ISESD53023.2021.9501642
Wilkinson, S.L., Moore, P.A. and Waddington, J.M. 2019. Assessing drivers of cross-scale variability in peat smoldering combustion vulnerability in forested boreal peatlands. Front. For. Glob. Change 2, 1–11; doi.org/10.3389/ffgc.2019.00084
Yang, J., Chen, Y., Yu, Z., Ding, H. and Ma, Z. 2019. The influence of PM2.5 on lung injury and cytokines in mice. Exp. Therapeutic. Med. 18, 2503–2511; doi:10.3892/etm.2019.7839