| Research Article | ||
Open Vet. J.. 2026; 16(3): 1511-1522 Open Veterinary Journal, (2026), Vol. 16(3): 1511-1522 Research Article Descriptive histopathological assessment of kidney and gill tissues in Lagocephalus sceleratus from the Libyan coastSana A. A. Mohammed1, Ismail M. Hdud2, Samia M. Efkeren3 and Eda M. A. Alshailabi3*1Department of Pathology and Clinical Pathology, Faculty of Veterinary Medicine, Omar Al-Mukhtar University, Al-Bayda, Libya 2Department of Pathology and Clinical Pathology, Faculty of Veterinary Medicine, University of Tripoli, Tripoli, Libya 3Department of Zoology, Faculty of Science, Omar Al-Mukhtar University, Al-Bayda, Libya *Corresponding Author: Eda M. A. Alshailabi. Department of Zoology, Faculty of Sciences, Omar Al Mukhtar University, El-Beyda, Libya. Email: eda.muftah [at] omu.edu.ly Submitted: 22/12/2025 Revised: 15/02/2026 Accepted: 25/02/2026 Published: 31/03/2026 © 2026 Open Veterinary Journal
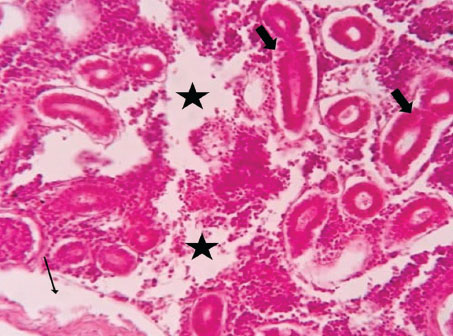
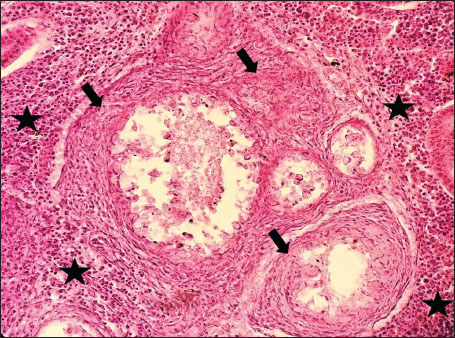
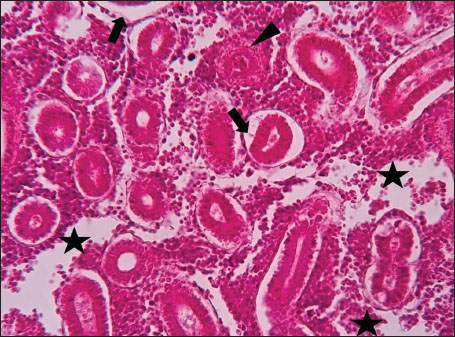
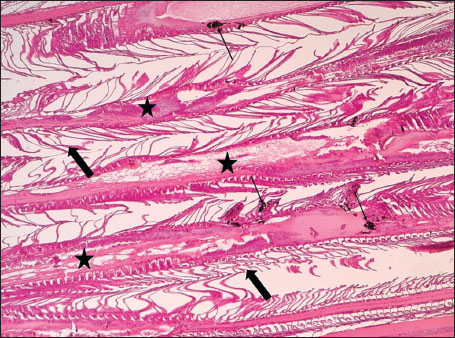
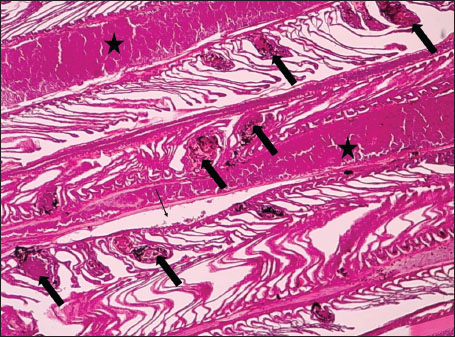
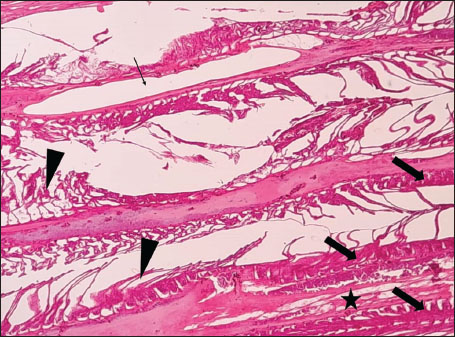
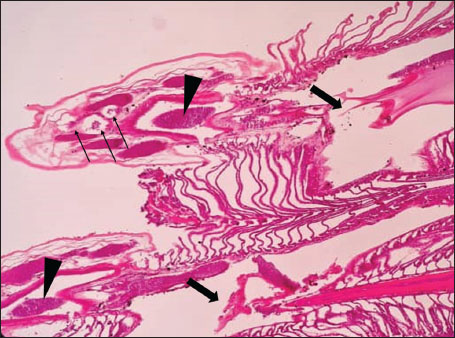
ABSTRACTBackground: The Mediterranean Sea has undergone significant ecological changes in recent decades, partly due to the introduction of non-native species. Lagocephalus sceleratus is an invasive Lessepsian species widely distributed in the Mediterranean, increasingly abundant along the Libyan coast, and potentially exposed to chronic environmental stressors. This species represents an important model for baseline pathological assessment in newly colonized marine environments. Aim: This study provides a descriptive histopathological assessment of kidney and gill tissues in adult L. sceleratus to document baseline tissue alterations without inferring direct environmental or pollution-related causation. Methods: A total of 150 adult specimens were collected from four Libyan coastal locations. Tissues were processed using standard histological techniques and evaluated semi-quantitatively (+, ++, +++) for lesion severity. Lesion severity grades were converted into ordinal numerical data and summarized using descriptive statistical approaches only. Results: Kidney tissues exhibited tubular degeneration, Bowman’s capsule dilatation, vascular congestion, extensive melanomacrophage centers (MMCs), and inflammatory lesions. Gill tissues showed disorganization of secondary lamellae, epithelial hyperplasia, edema, aneurysmal dilatations, vascular congestion, and MMC proliferation, some of which were associated with histologically observed parasitic structures. Conclusion: The observed alterations represent descriptive baseline tissue changes and should not be interpreted as direct evidence of pollution or specific environmental stressors. These findings provide reference data for future comparative pathological and ecological studies. Keywords: Lagocephalus sceleratus, Kidney, Gills, Histopathology, Melanomacrophage centers, Invasive species, Mediterranean Sea. IntroductionIn recent decades, the Mediterranean Sea has undergone profound ecological changes driven by the arrival and establishment of non-native marine species. Among these, Lagocephalus sceleratus has emerged as one of the most impactful Lessepsian migrants, due to its rapid geographic expansion, high ecological adaptability, and the presence of the potent neurotoxin tetrodotoxin in its tissues. This species has been widely documented to alter local fish communities, affect fisheries, and pose public health risks, making it an important target for biological and environmental assessment across the region (Katikou et al., 2022; Christidis et al., 2024). Fish are widely recognized as sensitive biological organisms in aquatic ecosystems, particularly in coastal habitats that are exposed to anthropogenic pressures such as industrial effluents, agricultural runoff, and untreated wastewater (Pinna et al., 2023). Among fish organs, the gills and kidneys are commonly regarded as sensitive to environmental stress, as they respond to physiological and pathological changes under environmentally suboptimal conditions. The gills, due to their large surface area and continuous exposure to surrounding water, are often the first tissues to exhibit structural alterations when fish encounter pollutants (Shahid et al., 2022). Histopathological changes in gills observed in polluted environments include epithelial lifting, lamellar fusion, hyperplasia, increased mucous cell density, and focal necrosis, which collectively reflect early physiological stress and potential compromise of respiratory function (Osman, 2010; Shahid et al., 2022). Similarly, the kidneys play crucial roles in osmoregulation, excretion of metabolic waste, and detoxification, making them highly vulnerable to chronic or systemic toxic effects (Wahidi et al., 2025). Documented renal lesions associated with chemical exposure include tubular deterioration, glomerular shrinkage, interstitial edema, and infiltration of inflammatory cells. These structural alterations often coincide with underlying biochemical disruptions, such as oxidative stress, impaired detoxification pathways, and activation of apoptotic mechanisms (Bernet et al., 1999; Authman, 2015). Despite the growing body of research on the ecological and toxicological implications of L. sceleratus, detailed histopathological assessments of this species remain scarce, especially in the southern Mediterranean basin, where environmental conditions and pollution profiles may differ from those in other regions. To date, detailed tissue-level studies of L. sceleratus from the southern Mediterranean are lacking, which limits the understanding of how this invasive species responds physiologically to local environmental stressors (Shakman et al., 2019; Ulman et al., 2021; Mohmmed et al., 2023). Accordingly, the present study aims to provide a comprehensive descriptive evaluation of histopathological changes in the kidneys and gills of L. sceleratus. The study is designed as a baseline histopathological assessment, focusing on tissue-level alterations without establishing causal links to environmental pollution or physicochemical stressors. By documenting structural aberrations and pathological features, this study contributes reference data for future monitoring and comparative studies in Mediterranean coastal ecosystems. Materials and MethodsSample designA total of 150 adult specimens of L. sceleratus, including both sexes, were collected from four Libyan coastal locations: Talamitha (n=39), Susah (n=34), Ain El-Ghazala (n=30), and Khalij Al-Bambah (n=47). Fish ranged in total length from 51 to 66 cm and in weight from 1.5 to 3.5 kg. Only apparently healthy adults were included, while juveniles were excluded to avoid age-related histopathological variation. Sampling was conducted opportunistically with assistance from local fishermen. All specimens were collected within a comparable seasonal window to minimize seasonal histopathological variability. Sex was recorded when possible; however, sex-based histopathological comparisons were not performed, as this was beyond the descriptive scope of the study. Fish were transported on ice at +4°C and examined in the Pathology Laboratory, Faculty of Veterinary Medicine, Omar Al-Mukhtar University (Mohmmed et al., 2023). Specimens were collected from a range of depths (0.5–70 m), with the majority from shallow waters (<10 m) and a subset (approximately 50 fish) from deeper locations (>30 m). Environmental physicochemical parameters such as temperature, salinity, and dissolved oxygen were not measured; therefore, no direct associations between histopathological findings and environmental variables were assessed. Tissue processing and histopathological examinationTissue specimens were fixed in 10% neutral buffered formalin for 24 hours. Following fixation, the samples were transferred to 70% ethanol for storage at room temperature. Subsequently, the tissues were processed for routine histopathological examination following standard procedures (Paul and Chanda, 2017). Paraffin embedding was performed, and 5 µm-thick sections were prepared using a microtome. Sections were stained with hematoxylin and eosin (H&E) and examined under a light microscope. Photomicrographs of representative lesions were captured using a high-resolution digital camera. Histopathological alterations were evaluated using a semi-quantitative scoring system adapted from Hose et al. (1996), Moshaie-Nezhad et al. (2021), and Alshailabi et al. (2023), where lesion severity was graded as mild (+), moderate (++), or severe (+++). Lesion severity grades (+, ++, +++) were converted into ordinal numerical values (1–3) for descriptive summarization of lesion severity. Due to the descriptive baseline nature of the study and the absence of a reference control group, inferential statistical comparisons were not emphasized or applied. Lesion severity scores were therefore summarized descriptively to avoid overinterpretation of the findings. Scoring was performed independently by two experienced observers, and representative lesions were confirmed across three sections per organ to ensure consistency. Tissues with visible parasitic structures were described separately from non-parasitized tissues to avoid conflating parasite-associated lesions with non-specific tissue alterations. The functional implications of the observed lesions were interpreted in accordance with Flores-Lopes and Thomaz (2011). Ethical approvalAll animal experiments conducted in this study were approved by the Ministry of Higher Education & Scientific Research and the Libyan National Committee for Biosafety & Bioethics, Libya. All procedures were performed in accordance with the relevant ethical guidelines, with session number 21/CH/25, dated 26/05/2021. ResultsHistopathological examination of the kidneyHistopathological examination of the kidney tissues of L. sceleratus revealed multiple alterations. Prominent melanomacrophage centers (MMCs), necrotic areas, dilatation of Bowman’s capsules, and vacuolar degeneration were observed (Fig. 1), associated with interstitial lymphohematopoietic tissue (Fig. 2). Dilated and congested blood vessels and extensive MMCs were also noted (Fig. 3). Renal degeneration surrounding lymphohematopoietic tissue, large clusters of MMCs, and thickened, congested vessel walls within fibrotic areas were evident (Fig. 4). Atrophic renal degeneration with necrotic and vacuolar changes was observed (Fig. 5). Severe inflammatory infiltration of lymphatic cells and granulomas with necrotic centers, surrounded by fibrous tissue, was detected (Figs. 6–7).
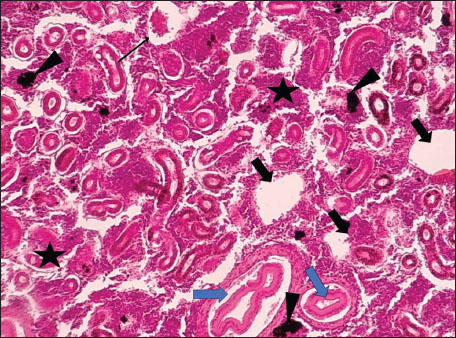
Fig. 1. Histopathology of the kidney in an adult L. sceleratus showing kidney parenchyma (stars), MMCs (head arrows), necrotic tissue (thick arrows), dilatation of Bowman’s capsule (thin arrow), and vacuolar degeneration (blue arrows). ×100 H&E.
Fig. 2. Histopathology of the kidney in an adult L. sceleratus showing renal degeneration with necrotic areas (stars), severe MMCs (head arrows), and interstitial lymphohematopoietic tissue (thick arrows). ×40 H&E.
Fig. 3. Histopathology of the kidney in an adult L. sceleratus showing dilated and congested blood vessels (star) and severe MMC proliferation (head arrows). ×40 H&E.
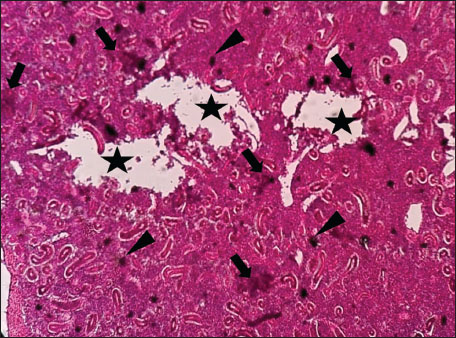
Fig. 4. Histopathology of the kidney in an adult L. sceleratus showing renal degeneration surrounding lymphohematopoietic tissue (stars), large MMC clusters (head arrows), and congested, dilated, thickened vessel walls within fibrotic areas (thin arrows). ×400 H&E.
Fig. 5. Histopathology of the kidney in an adult L. sceleratus showing atrophic renal degeneration (thick arrows), necrotic tissue (stars), and vacuolar degeneration (thin arrow). ×400 H&E.
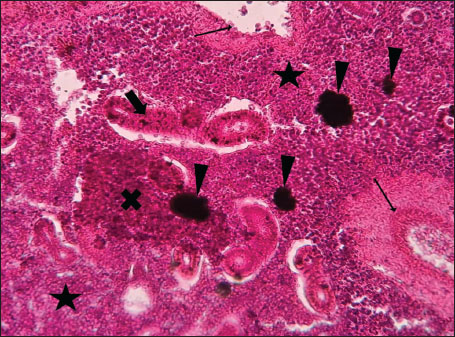
Fig. 6. Histopathology of the kidney in an adult L. sceleratus showing atrophic renal degeneration with severe inflammatory infiltration of lymphatic cells (stars) and granulomas with necrotic content, surrounded by fibrous tissue (thick arrows). ×400 H&E.
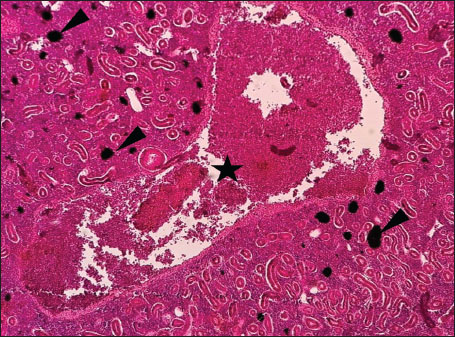
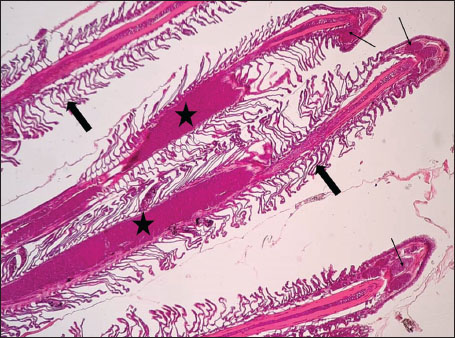
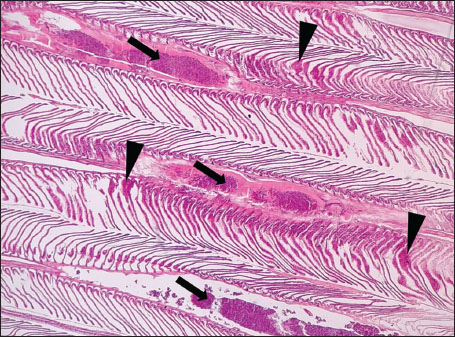
Fig. 7. Histopathology of the kidney in an adult L. sceleratus showed the atrophic renal degeneration (stars), dilatation of Bowman’s capsules (thick arrows), and a granuloma (head arrow). ×400 H&E. Histopathological examination of the gillsThe gill sections of adult L. sceleratus exhibited multiple structural alterations. Secondary lamellae showed marked disorganization, while gill filaments displayed club-shaped deformities and occasional aneurysmal dilatations (Fig. 8). Vascular congestion with telangiectatic changes and proliferation of MMCs were also observed (Figs. 9–10). Edematous changes in primary lamellae, separation of the epithelial layer, lamellar aneurysms, and epithelial hyperplasia were documented (Figs. 10–11). Focal damage to secondary lamellae, disruption of lamellar architecture, presence of lamellar aneurysms, and unidentified parasitic structures were observed (Fig. 12). Pronounced vascular congestion and dilatation of lamellae due to red blood cell accumulation were noted (Fig. 13).
Fig. 8. Histopathology of the gills in an adult L. sceleratus showing disorganization of secondary lamellae (thick arrows), club-shaped filament deformities with aneurysms (thin arrows), and congestion/telangiectasia in filament vessels (stars). ×40 H&E.
Fig. 9. Histopathology of the gills in an adult L. sceleratus showing the disorganized secondary lamellae (thick arrows), MMCs (thin arrows), and vascular congestion/telangiectasia in filaments (stars). ×40 H&E.
Fig. 10. Histopathology of the gills in an adult L. sceleratus showing the edema in primary lamellae and epithelial detachment (thin arrows), lamellar aneurysm with MMCs (thick arrows), and congestion/telangiectasia (stars). ×40 H&E.
Fig. 11. Histopathology of the gills in an adult L. sceleratus showing edema in primary lamellae and epithelial detachment (thin arrow), curling of secondary lamellae (head arrows), congestion/telangiectasia (star), and epithelial hyperplasia (thick arrows). ×40 H&E.
Fig. 12. Histopathology of the gills in an adult L. sceleratus showing the damage to secondary lamellae and lamellar disorganization (thick arrows), lamellar aneurysm (head arrows), and unidentified parasites (thin arrows). ×40 H&E.
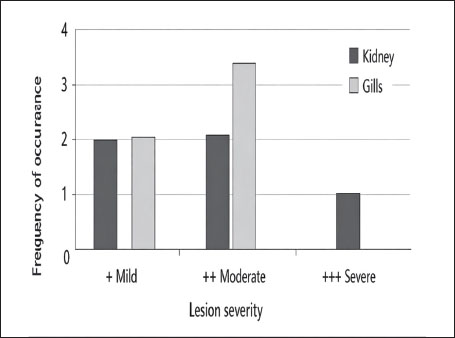
Fig. 13. Histopathology of the gills in an adult L. sceleratus showing congestion and telangiectasia of gill filament vessels (thick arrows) and lamellar dilation due to RBC accumulation (head arrows). ×40 H&E. Gill lesions were described separately for tissues with visible parasites and those without parasitic structures to improve interpretative clarity. Summary of lesionsHistopathological alterations in both kidney and gill tissues were classified using a semi-quantitative scoring system (+, ++, +++) (Fig. 14).
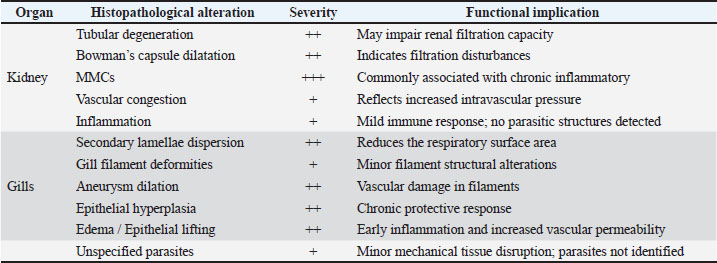
Fig. 14. Frequency distribution of lesion severity in the kidney and gills (+, ++, +++). Descriptive baseline data; no statistical comparisons due to lack of control. Kidney tissuesModerate tubular degeneration and Bowman’s capsule dilatation were recorded, whereas MMC proliferation was severe (+++). Vascular congestion and mild inflammation were observed. No parasitic structures were detected in kidney tissues. Observed alterations represent descriptive baseline histopathological changes. Gill tissuesModerate secondary lamellae dispersion, aneurysm dilation, epithelial hyperplasia, and edema/epithelial lifting were observed, whereas filament deformities and unidentified parasites were mild (+). Lesions were described separately for tissues with visible parasites and non-parasitized tissues to improve interpretative clarity. It should be noted that no unpolluted control group was included, and these observations represent descriptive baseline data; no causal inference regarding environmental stressors can be drawn (Table 1). Table 1. Semi-quantitative scoring of histopathological alterations in kidney and gill tissues of L. sceleratus along the Libyan coast (n=150 adult specimens).
DiscussionThe present study revealed pronounced histopathological alterations in both the kidneys and gills of L. sceleratus collected along the Libyan coast. Kidney lesions, including tubular degeneration, Bowman’s capsule dilatation, proliferation of MMCs, vascular congestion, and inflammatory infiltration, were associated with histologically detected structures, but no parasitic structures were detected in kidney tissues. These patterns are commonly reported in marine teleosts exposed to chronic environmental stressors (Ali et al., 2014; Steinel and Bolnick, 2017; Carreras-Colom et al., 2022). These lesions should be interpreted as descriptive indicators of physiological stress rather than direct evidence of chemical pollution or causality, consistent with Flores-Lopes and Thomaz (2011). Vascular abnormalities, including congestion and thickened vessel walls, were evident, which may reflect chronic circulatory disturbances or inflammatory responses rather than acute toxicity (Bernet et al., 1999; Thophon et al., 2003). Interstitial lymphohematopoietic tissue, inflammatory cell infiltration, and granulomatous formations suggest that parasitic or microbial challenges could contribute to renal lesions. Despite these histopathological changes, the sampled fish appeared healthy at capture, demonstrating the potential physiological resilience of marine teleosts. Fish kidneys possess compensatory mechanisms, and gills serve critical osmoregulatory and excretory functions, allowing survival despite structural alterations (Evans et al., 2005; Hwang et al., 2011; Hinton et al., 2018; Leone et al., 2018). Gradual development of renal lesions likely enables adaptation without acute organ failure. Gill tissues also exhibited pronounced alterations, including disorganization of secondary lamellae, club-shaped filament deformities, lamellar aneurysms, epithelial hyperplasia, and edema. These changes are consistent with chronic, sublethal tissue responses observed descriptively, without inferring specific environmental stressors or chemical toxicity (Sinha et al., 2014; Carvalho et al., 2020; Pramanik and Biswas, 2024). Unidentified parasitic structures, morphologically consistent with metazoan parasites, were observed based on histological appearance. Lesions were described separately for tissues with visible parasites and non-parasitized tissues to improve interpretative clarity (Sitjà-Bobadilla, 2008; Biller and Chagas, 2022). The proliferation of MMCs in gills reflects an immune response to tissue damage and cellular debris, reinforcing the descriptive nature of the findings. Vascular congestion and lamellar dilation due to RBC accumulation may indicate sublethal circulatory stress, but these observations are descriptive and not confirmed by physiological measurements (Triebskorn et al., 2007; Hasan et al., 2022). Moderate lesions (++), as summarized in Table 1 and illustrated in Fig. 14, were predominant, reflecting chronic, sublethal tissue responses rather than lethal injury. Overall, the kidney and gill lesions detected in L. sceleratus reflect chronic, sublethal tissue responses observed descriptively. No causal relationships with environmental stressors or chemical pollution can be inferred. Future studies incorporating water and sediment analyses, parasitological identification, and reference/control populations are recommended to strengthen the interpretation of these histopathological responses as potential biomarkers. LimitationsThis study is limited by the absence of environmental physicochemical data, the lack of a reference or control site, and the descriptive nature of the histopathological assessment. In addition, parasitic structures were not identified to species level, and quantitative prevalence metrics were not calculated. Accordingly, the findings should be interpreted as baseline descriptive observations rather than indicators of specific environmental stressors or pollution sources. ConclusionHistopathological examination of the kidneys and gills of L. sceleratus revealed tissue-level changes that serve as descriptive baseline data. Moderate to severe alterations were observed, including tubular degeneration, MMC proliferation, vascular congestion, secondary lamellae disorganization, and epithelial hyperplasia. No parasitic structures were detected in kidney tissues, while parasitic structures were observed only in some gill tissues and described separately. Despite these alterations, the species exhibits physiological compensation mechanisms, including renal adaptive responses and gill-mediated osmoregulation, supporting resilience under sublethal stress conditions. These findings are descriptive and should not be interpreted as direct evidence of environmental contamination or causation. All observations represent baseline histopathological data, intended to support future comparative and environmentally integrated studies rather than establish causal relationships. AcknowledgmentsThe authors sincerely thank the staff of the Pathology and Clinical Pathology Department (Faculty of Veterinary Medicine, Omar Al-Mukhtar University) and the Zoology Department (Faculty of Science, Omar Al-Mukhtar University) for their valuable support. Conflict of interestThe authors declare that there is no conflict of interest. FundingThis research received no specific grant. Authors' contributionsEda M. A. Alshailabi and Sana A. A. Mohammed contributed to the project idea and design, and Sana A. A. Mohammed and Ismail M. Hdud executed the study. Eda M. A. Alshailabi drafted and wrote the manuscript. Sana A. A. Mohammed and Samia M. Efkeren reviewed the manuscript critically. All authors have read and approved the final manuscript. Data availabilityAll data supporting the findings of this study are available within the manuscript. ReferencesAli, A.O., Hohn, C., Allen, P.J., Ford, L., Dail, M.B., Pruett, S. and Petrie-Hanson, L. 2014. The effects of oil exposure on peripheral blood leukocytes and splenic melano-macrophage centers of Gulf of Mexico fishes. Mar. Pollut. Bull. 79(1–2), 87–93; doi:10.1016/j.marpolbul.2013.12.036 Alshailabi, E., I.Al-Zail, N. and Abraheem, R. 2023. The effects of cigarette smoke on the epididymal tissues in adult male albino rats and the ameliorative effect of the Sidr honey. Libyan. J. Med. Res. 16(2 B), 79–92; doi:10.54361/LJMR.16.2B.08 Authman, M.M. 2015. Use of fish as bio-indicator of the effects of heavy metals pollution. J. Aquac. Res. Develop. 6, 328; doi:10.4172/2155-9546.1000328 Bernet, D., Schmidt, H., Meier, W., Burkhardt‐Holm, P. and Wahli, T. 1999. Histopathology in fish: proposal for a protocol to assess aquatic pollution. J. Fish Dis. 22(1), 25–34; doi:10.1046/j.1365-2761.1999.00134.x Biller, J.D. and Chagas, E.C. 2022. Mechanisms of resistance and tolerance against parasites in fish: the impairments caused by Neoechinorhynchus buttnerae in Colossoma macropomum. Anais. Da. Academia. Brasileira. De. Ciências. 94(4), e20210258; doi:10.1590/0001-3765202220210258 Carreras-Colom, E., Constenla, M., Dallarés, S. and Carrassón, M. 2022. Natural variability and potential use of melanomacrophage centres as indicators of pollution in fish species from the NW Mediterranean Sea. Mar. Pollut. Bull. 176, 113441; doi:10.1016/j.marpolbul.2022.113441 Carvalho, T.L.A.D.B., Do Nascimento, A.A., Gonçalves, C.F.D.S., Dos Santos, M.A.J. and Sales, A. 2020. Assessing the histological changes in fish gills as environmental bioindicators in Paraty and Sepetiba bays in Rio de Janeiro, Brazil. Latin. Am. J. Aquatic. Res. 48(4), 590–601. Christidis, G., Batziakas, S., Peristeraki, P., Tzanatos, E., Somarakis, S. and Tserpes, G. 2024. Another one bites the net: assessing the economic impacts of Lagocephalus sceleratus on small-scale fisheries in Greece. Fishes 9(3), 104; doi:10.3390/fishes9030104 Evans, D.H., Piermarini, P.M. and Choe, K.P. 2005. The multifunctional fish gill: dominant site of gas exchange, osmoregulation, acid-base regulation, and excretion of nitrogenous waste. Physiological Rev. 85(1), 97–177; doi:10.1152/physrev.00050.2003 Flores-Lopes, F. and Thomaz, A. 2011. Histopathologic alterations observed in fish gills as a tool in environmental monitoring. Braz. J. Biol. 71(1), 179–188; doi:10.1590/s1519-69842011000100026 Hasan, J., Ferdous, S.R., Rabiya, S.B.A., Hossain, M.F., Hasan, A.M. and Shahjahan, M. 2022. Histopathological responses and recovery in gills and liver of Nile tilapia (Oreochromis niloticus) exposed to diesel oil. Toxicol. Rep. 9, 1863–1868; doi:10.1016/j.toxrep.2022.10.005 Hinton, D.E., Baumann, P.C., Gardner, G.R., Hawkins, W.E., Hendricks, J.D., Murchelano, R.A. and Okihiro, M.S. 2018. Histopathologic biomarkers. In Biomarkers: biochemical, physiological, and histological markers of anthropogenic stress. Eds., Huggett, R.A. Kimerle, P.M. Mehrle, Jr.,Bergman, H. Chelsea, MI: CRC Press, pp: 155–209; doi: 10.1201/9781351070270. Hose, J.E., Mcgurk, M.D., Marty, G.D., Hinton, D.E., Brown, E.D. and Baker, T.T. 1996. Sublethal effects of the Exxon Valdez oil spill on herring embryos and larvae: morphological, cytogenetic, and histopathological assessments, 1989–1991. Can. J. Fisheries Aquatic Sci. 53, 2355–2365. Hwang, P.P., Lee, T.H. and Lin, L.Y. 2011. Ion regulation in fish gills: recent progress in the cellular and molecular mechanisms. Am. J. Physiol–Regulatory. Integr. Comparative. Physiol. 301, R28–R47; doi:10.1152/ajpregu.00047.2011 Katikou, P., Gokbulut, C., Kosker, A.R., Campàs, M. and Ozogul, F. 2022. An updated review of tetrodotoxin and its peculiarities. Mar. Drugs 20(1), 47; doi:10.3390/md20010047 Leone, O., Valdecantos, S. and Martínez, V. 2018. Histopathological markers of environmental stress in the fish Odonthestes bonariensis (Atheriniformes, Atherinopsidae) in two reservoirs of Argentina. UNED Res. J. 10(2), 273–282; doi: 10.22458/urj.v10i2.1909 Mohmmed, S., Ekhnefer, A., Fadel, A. and Sharif, M. 2023. Prevalence of some parasitic infestations in Lagocephalus sceleratus in Eastern Libya. AlQalam J. Med. Appl. Sci. 6(2), 608–616. Moshaie-Nezhad, P., Bahari, Z., Jangravi, Z., Zarei, S.M. and Iman, M. 2021. The effect of Descurainia sophia seed extract on nephrotoxicity markers induced by acetaminophen in mice. J. Adv. Med. Biomed. Res. 29(134), 139–144; doi:10.30699/jambs.29.134.139 Osman, A.G.M. 2010. Enzymatic and histopathologic biomarkers as indicators of aquatic pollution in fishes. Natural. Sci. 2(11), 1302–1311; doi:10.4236/ns.2010.211158 Paul, M. and Chanda, M. 2017. Histological slide preparation of fish tissues (paraffin method). Asutosh College Kolkata India 1, 1–5; doi: 10.13140/RG.2.2.15130.34243 Pinna, M., Zangaro, F., Saccomanno, B., Scalone, C., Bozzeda, F., Fanini, L. and Specchia, V. 2023. An overview of ecological indicators of fish to evaluate the anthropogenic pressures in aquatic ecosystems: from traditional to innovative DNA-based approaches. Water 15(5), 949; doi:10.3390/w15050949 Pramanik, S. and Biswas, J.K. 2024. Histopathological fingerprints and biochemical changes as multi-stress biomarkers in fish confronting concurrent pollution and parasitization. iScience 27(12), 111432; doi:10.1016/j.isci.2024.111432 Shahid, S., Sultana, T., Sultana, S., Hussain, B., Al-Ghanim, K.A., Al-Bashir, F., Riaz, M.N. and Mahboob, S. 2022. Detecting aquatic pollution using histological investigations of the gills, liver, kidney, and muscles of Oreochromis niloticus. Toxics 10(10), 564; doi:10.3390/toxics10100564 Shakman, E., Eteayb, K., Taboni, I. and Ben Abdalha, A. 2019. Status of marine alien species along the Libyan coast. J. Black Sea / Medit. Environ. 25(2), 188–209. Sinha, A.K., Matey, V., Giblen, T., Blust, R. and De Boeck, G. 2014. Gill remodeling in three freshwater teleosts in response to high environmental ammonia. Aquatic Toxicol. 155, 166–180; doi:10.1016/j.aquatox.2014.05.016 Sitjà-Bobadilla, A. 2008. Living off a fish: a trade-off between parasites and the immune system. Fish Shellfish Immunol. 25(4), 358–372; doi:10.1016/j.fsi.2008.03.018 Steinel, N.C. and Bolnick, D.I. 2017. Melanomacrophage centers as a histological indicator of immune function in fish and other poikilotherms. Front. Immunol. 8, 827; doi:10.3389/fimmu.2017.00827 Thophon, S., Kruatrachue, M., Upatham, E.S., Pokethitiyook, P., Sahaphong, S. and Jaritkhuan, S. 2003. Histopathological alterations of white seabass (Lates calcarifer) in acute and subchronic cadmium exposure. Environ. Pollut. 121(3), 307–320; doi:10.1016/S0269-7491(02)00270-1 Triebskorn, R., Casper, H., Scheil, V. and Schwaiger, J. 2007. Ultrastructural effects of pharmaceuticals (carbamazepine, clofibric acid, metoprolol, diclofenac) in rainbow trout (Oncorhynchus mykiss) and common carp (Cyprinus carpio). Anal. BioAnal. Chem. 387(4), 1405–1416; doi:10.1007/s00216-006-1033-x Ulman, A., Yildiz, T., Demirel, N., Canak, O., Yemişken, E. and Pauly, D. 2021. The biology and ecology of the invasive silver-cheeked toadfish (Lagocephalus sceleratus), with emphasis on the Eastern Mediterranean. NeoBiota 68, 145–175; doi:10.3897/neobiota.68.71767 Wahidi, B.R., Hakimah, N., Suhermanto, A., Triyastuti, M.S. and Utami, D.A.S. 2025. Histopathological analysis of gills, liver, and kidneys of Nile tilapia (Oreochromis niloticus) affected by feverish Lapindo mud in Porong River, Sidoarjo-East Java. J. Aquac. Fish. Health. 14(1), 114–121; doi:10.20473/jafh.v14i1.61592 | ||
| How to Cite this Article |
| Pubmed Style Mohammed SA, Hdud IM, Efkeren SM, Alshailabi EM. Descriptive histopathological assessment of kidney and gill tissues in Lagocephalus sceleratus from the Libyan coast. Open Vet. J.. 2026; 16(3): 1511-1522. doi:10.5455/OVJ.2026.v16.i3.10 Web Style Mohammed SA, Hdud IM, Efkeren SM, Alshailabi EM. Descriptive histopathological assessment of kidney and gill tissues in Lagocephalus sceleratus from the Libyan coast. https://www.openveterinaryjournal.com/?mno=304162 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.10 AMA (American Medical Association) Style Mohammed SA, Hdud IM, Efkeren SM, Alshailabi EM. Descriptive histopathological assessment of kidney and gill tissues in Lagocephalus sceleratus from the Libyan coast. Open Vet. J.. 2026; 16(3): 1511-1522. doi:10.5455/OVJ.2026.v16.i3.10 Vancouver/ICMJE Style Mohammed SA, Hdud IM, Efkeren SM, Alshailabi EM. Descriptive histopathological assessment of kidney and gill tissues in Lagocephalus sceleratus from the Libyan coast. Open Vet. J.. (2026), [cited March 31, 2026]; 16(3): 1511-1522. doi:10.5455/OVJ.2026.v16.i3.10 Harvard Style Mohammed, S. A., Hdud, . I. M., Efkeren, . S. M. & Alshailabi, . E. M. (2026) Descriptive histopathological assessment of kidney and gill tissues in Lagocephalus sceleratus from the Libyan coast. Open Vet. J., 16 (3), 1511-1522. doi:10.5455/OVJ.2026.v16.i3.10 Turabian Style Mohammed, Sana A., Ismail M. Hdud, Samia M. Efkeren, and Eda M. Alshailabi. 2026. Descriptive histopathological assessment of kidney and gill tissues in Lagocephalus sceleratus from the Libyan coast. Open Veterinary Journal, 16 (3), 1511-1522. doi:10.5455/OVJ.2026.v16.i3.10 Chicago Style Mohammed, Sana A., Ismail M. Hdud, Samia M. Efkeren, and Eda M. Alshailabi. "Descriptive histopathological assessment of kidney and gill tissues in Lagocephalus sceleratus from the Libyan coast." Open Veterinary Journal 16 (2026), 1511-1522. doi:10.5455/OVJ.2026.v16.i3.10 MLA (The Modern Language Association) Style Mohammed, Sana A., Ismail M. Hdud, Samia M. Efkeren, and Eda M. Alshailabi. "Descriptive histopathological assessment of kidney and gill tissues in Lagocephalus sceleratus from the Libyan coast." Open Veterinary Journal 16.3 (2026), 1511-1522. Print. doi:10.5455/OVJ.2026.v16.i3.10 APA (American Psychological Association) Style Mohammed, S. A., Hdud, . I. M., Efkeren, . S. M. & Alshailabi, . E. M. (2026) Descriptive histopathological assessment of kidney and gill tissues in Lagocephalus sceleratus from the Libyan coast. Open Veterinary Journal, 16 (3), 1511-1522. doi:10.5455/OVJ.2026.v16.i3.10 |