| Research Article | ||
Open Vet. J.. 2026; 16(3): 1853-1858 Open Veterinary Journal, (2026), Vol. 16(3): 1853-1858 Research Article Validation of eye occluding device developed from bottle caps for dopamine studies in rabbits: A pilot studyChristina Aritonang1,2,3, Irwanto Irwanto3,4* and Reni Prastyani2,31Doctoral Program of Medical Science, Faculty of Medicine, Universitas Airlangga, Surabaya, Indonesia 2Department of Ophthalmology, Faculty of Medicine, Universitas Airlangga, Surabaya, Indonesia 3Dr. Soetomo General Academic Hospital, Surabaya, Indonesia 4Department of Child Health, Faculty of Medicine, Universitas Airlangga, Surabaya, Indonesia *Corresponding Author: Irwanto Irwanto. Dr. Soetomo General Academic Hospital, Surabaya, Indonesia, and Department of Child Health, Faculty of Medicine, Universitas Airlangga, Surabaya, Indonesia. Email:irwanto [at] fk.unair.ac.id Submitted: 29/11/2025 Revised: 11/02/2026 Accepted: 21/02/2026 Published: 31/03/2026 © 2026 Open Veterinary Journal
AbstractBackground: The designs of custom ocular occluders vary widely and may introduce heterogeneity in biological outcomes. Retinal dopamine D₁ receptor (D₁R) signaling is thought to exert a protective role and reduce dopaminergic activity in the rabbit retina. Aim: The aim of this study was to prove the reduction in D1R concentration in rabbit eyes after the eye occlusion process for 30 days. Methods: A low-cost, opaque monocular occluder was developed by repurposing black plastic bottle caps cushioned with adhesive bandage tape. The occluder was applied to the right eye of 2 to 3-month-old male New Zealand White rabbits for 30 days, with the left eye serving as an unoccluded control. The devices were replaced every 3 days under minimal lighting. At the end of the occlusion period, rabbits were euthanized, their eyes were enucleated, and retinal/vitreal tissues were collected. D₁R concentrations were quantified using an enzyme-linked immunosorbent assay. Results: The bottle-cap occluder was well tolerated; no clinical signs of conjunctivitis, corneal opacity, discharge, eyelid edema, or orbital cellulitis were observed. Retinal/vitreal D₁R levels were consistently lower in occluded eyes than in fellow eyes. In Rabbit 1, D₁R concentrations were 0.41 ng/ml in the left (unoccluded) eye and 0.26 ng/ml in the right (occluded) eye; in Rabbit 2, D1R concentrations were 2.82 and 1.95 ng/ml, respectively. Conclusion: This pilot study suggests that a simple black bottle-cap-based eye occluder can be safely used in rabbits and is associated with reduced retinal D₁R levels, supporting its utility as a low-cost tool for experimental dopamine research. Keywords: Dopamine, Rabbit, Eye occluder, Experimental model, Sustainable laboratory practice. IntroductionDopamine synthesis in the retina entails not only anatomical modifications of ocular structures but also neurochemical alterations within the retina. Dopamine is the main catecholamine in the retina. It is synthesized by dopaminergic amacrine cells from the amino acid tyrosine through the enzyme inhibitor tyrosine hydroxylase and then converted to dopamine by aromatic L-amino-acid decarboxylase (Jackson et al., 2012) etinal dopaminergic system has been identified as a key regulator of ocular growth, acting through its influence on photoreceptor activity, amacrine cell signaling, and intracellular pathways such as cyclic adenosine monophosphate (cAMP) (Huang et al., 2023). Dopamine operates in the mammalian retina through two receptor families, namely D₁-like receptors (D₁R) and D2-like receptors, which are coupled with the enzyme adenylate cyclase, thereby altering intracellular cAMP levels and the cAMP-PKA-DARPP-32 pathway. The dopamine receptor (D₁R) is widely expressed in various retinal cell types, including horizontal cells, ON and OFF bipolar cells, amacrine cells, and ganglion cells (Neve et al., 2004; Farshi et al., 2016). Experimental and epidemiological evidence indicate that dopamine serves as a “stop” signal for ocular growth, with increased retinal dopamine activity (Kim et al., 2025). These results establish dopamine and its downstream signaling pathways, including cAMP-mediated routes, as pivotal elements in the bidirectional interaction between visual stimuli and ocular growth responses. However, the precise temporal dynamics and spatial distribution of dopaminergic changes in relation to structural remodeling of the retina, choroid, and sclera remain incompletely characterized (Zhou et al., 2017). The occluded eye animal models offer a robust experimental framework for investigating these mechanisms under regulated conditions (McGlinn et al., 2007). In the study of retinal dopamine, the monocular deprivation model is widely used to modulate visual input and retinal dopaminergic activity. However, the current methods of occluders, such as using conventional patches, eyelid suturing, or commercial occlusion devices, often face limitations such as position instability, animal discomfort, risk of periocular tissue injury, and lack of standardization (Ozkan et al., 2025). The rabbit model is very important to ensure that the retinal circuitry, dopaminergic distribution, and eye growth patterns are similar enough to those of the human eye to make the research more useful (Mcglinn et al., 2007; Troilo et al., 2019). Rabbits are biologically significant as an experimental model for dopamine research owing to their comparatively large ocular dimensions, which enable accurate biometric, imaging, and histological evaluations, and the presence and distribution pattern of dopaminergic amacrine cells that closely mimic those of the human retina (Kim et al., 2025). Their heightened sensitivity to alterations in the visual environment further substantiates the choice of New Zealand White rabbits (Oryctolagus cuniculus) as a suitable model for investigating dopaminergic effects (Hu et al., 2025; Kim et al., 2025; Miyagishima et al., 2025). Furthermore, rabbits are optimal for longitudinal studies that look at how neurochemical changes occur along with structural changes in the eyes because they are easy to handle, have well-established husbandry protocols, and can be measured repeatedly in vivo (Kim et al., 2025). Nevertheless, reports regarding eye occluding devices that are stable, easy to apply, minimally invasive, and cost-effective are still limited. The development of occluder devices on bottle caps offers a low-cost alternative approach that can provide consistent visual occlusion while enhancing animal welfare during observation periods. Considering these scientific underpinnings and the existing knowledge gaps, it is imperative to examine retinal dopamine activity using innovative eye occlude developed from bottle caps, considering the feasibility of use, device stability, and suitability as a model for monocular deprivation. This study focuses on retinal neurotransmitter pathways and their subsequent signaling cascades using a descriptive analytic experimental design. Materials and MethodsEye occluder fabricationOpaque monocular eye occluders were constructed by repurposing black plastic bottle caps with a diameter of approximately 42 mm and a depth of 16 mm. Black caps were selected to minimize residual light exposure to the occluded eye. Three small holes were made by soldering the inferior aspect of each cap to allow air circulation without allowing light entry. To reduce the risk of pressure-related injury to the periocular region, the rim of each cap was cushioned with bandage tape (Leukoplast®, OneMed, Sidoarjo, Indonesia). The caps were used as eye occluders after cushioning. Figure 1 shows the final occluder design.
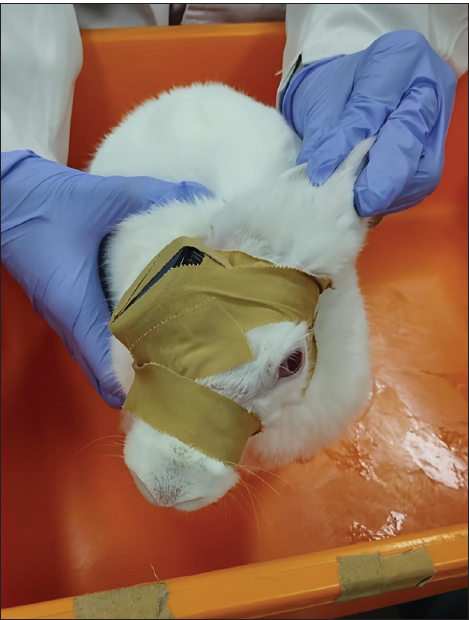
Fig. 1. Occluding device design with measurement. Animals used and experimental designTo validate the effectiveness of the occluder design for inducing dopamine, the device was tested in 2 to 3-month-old male New Zealand White rabbits (Oryctolagus cuniculus). Before the dopamine studies, the rabbits underwent a 7-day period of acclimatization in a controlled setting (temperature maintained at 22℃ and a 12-hour light/dark cycle) where they were provided with commercial feed, vegetables, and unlimited access to water. The health and ocular status of the rabbits were confirmed before they were included in the study. After acclimatization, dopaminergic activity was induced by occluding the eye with a black plastic cap for 30 consecutive days. For each rabbit, the right eye was assigned as the occluded eye, whereas the left eye served as the non-occluded fellow eye. Occlusion procedureThe cushioned occluder was placed over the right eye and secured in position using adhesive bandage tape. The tape was circumferentially wrapped around the head and neck, posterior to the ears, to prevent occluder dislodgement due to scratching or grooming behavior. Eye occlusion was maintained continuously for 30 days. The occluder and bandage tape were replaced every 3 days under minimal lighting conditions to avoid unintended light exposure to the occluded eye. The rabbits wearing the occluder are shown in Figure 2.
Fig. 2. One of the rabbits during occlusion. The device (black) can be seen in the picture and was eventually completely covered with bandage tape. Tissue collection and D₁R assayRetinal dopamine D₁R expression was selected as the primary outcome measure for validating the occluder design, given its documented role in experimental dopamine research models. After 30 days of occlusion, the rabbits were euthanized before enucleation via intramuscular administration of ketamine (40 mg/kg) and xylazine (5 mg/kg). Both eyes were then enucleated, and retinal and vitreous tissues were carefully dissected and collected for biochemical analysis. Each tissue sample of the rabbit retina and vitreous have 10–20 mg. The tissue was homogenized by added 600 µl/10 mg tissue in 1.5 ml of lysis buffer (PRO-PREP™, iNtRON Biotechnology, Inc., Seongnam, Gyeonggi, Republic of Korea) and a protease inhibitor. After incubation on ice, the homogenates were centrifuged at 13,000 rpm (4°C) for 5 minutes, and the resulting supernatants were collected for ELISA. D₁R levels were quantified using a commercial ELISA kit (RE1689b, Reed Biotech Ltd., Wuhan, Hebei, People’s Republic of China), following the manufacturer’s instructions. Both eyes from each rabbit were analyzed, and the measured retinal D₁R expression was compared with values reported in previous experimental studies to support the validity of the occluder design for dopaminergic changes. Ethical approvalAll procedures were approved by the Animal Care and Use Committee of the Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia (No. 2.KEH.160.10.2023). ResultsThe two rabbits tolerated the bottle-cap occluding device well throughout the 30-day deprivation period. No localized or systemic adverse effects were observed, and no clinical signs of conjunctivitis, corneal opacity, discharge, eyelid edema, or orbital cellulitis were observed in either animal. Biochemical analysis of retinal and vitreal samples showed a consistent reduction of dopamine D₁R concentration in the occluded eyes compared with the unoccluded eyes. In Rabbit 1, D₁R levels were 0.41 ng/ml in the left (unoccluded) eye and 0.26 ng/ml in the right (occluded) eye. In rabbit 2, D₁R concentrations were 2.82 ng/ml in the left eye and 1.95 ng/ml in the right eye. The effect size was small for each rabbit (36,6% in Rabbit 1 and 30,9% in Rabbit 2) because the aim of this study was not to test a hypothesis but as a pilot study to prove the reduction in D1R concentration in rabbit eyes after the eye occlusion process for 30 days, and because D1R has a low affinity for dopamine. However, this study proves that a black bottle cap occluder can reduce D1R in the occluded eye in both rabbits. Thus, in both rabbits, monocular form deprivation with the bottle-cap occluder was associated with a lower retinal and vitreal D₁R concentration in the occluded eye (right eye of rabbits 1 and 2). DiscussionAnimal models of dopamine studies have historically used a wide variety of occluding or diffusing devices, including translucent hoods, contact lenses, and plastic diffusers (McGlinn et al., 2007; Troilo et al., 2019). This diversity of designs contributes to heterogeneity in experimental conditions, which can produce variable outcomes even within the same species and age group. Such variability complicates the comparison across studies and may limit the translational relevance of preclinical findings. In this context, simple, low-cost, and reproducible occluding devices are valuable, particularly if they produce consistent biological signals that align with established mechanisms of dopamine receptors (Huang et al., 2023; Zhou et al., 2017). In the present pilot study, we repurposed black plastic bottle caps into monocular occluders and demonstrated that 30 days of form deprivation was associated with reduced retinal and vitreal D₁R concentration in the occluded eyes compared with those in the unoccluded eyes. This pattern is consistent with the prevailing concept that retinal dopamine and D₁R signaling act as dopamine inhibitors (Huang et al., 2023). D₁R activity is a key link. In the retinal dopamine biosynthesis pathway and the regulation of retinal neuron function and homeostasis within the vitreous environment (Chen et al., 2017; Junior et al., 2024). Under normal visual conditions, patterned light stimulation promotes dopamine release from amacrine cells, activates D₁Rs, and enhances cAMP-mediated pathways that help maintain physiological ocular growth. Dopaminergic activity decreases when form is degraded or visual input is severely reduced, leading to a relative loss of this “stop” signal (Zhou et al., 2017; Veruki et al., 2025). Molecular pharmacology studies of D₁R emphasize that changes in D₁R expression or sensitivity affect the retina’s response to both endogenous and exogenous dopaminergic stimuli. The lower D₁R levels observed in our occluded eyes likely reflect a dysregulated dopaminergic environment, either as a direct consequence of reduced receptor expression or as a downstream effect of chronic reduction in dopamine release and receptor activation (Zhou et al., 2017; Gan et al., 2025; Sahlholm et al., 2025). Although we did not directly administer dopaminergic agents in our study, the observed reduction in D₁R concentration in the form-deprived occluded rabbit’s eye is compatible with these earlier reports, suggesting that our black bottle-cap occluder produces a retinal environment in a rabbit eye model (Zhou et al., 2017; Huang et al., 2023). This study is the first experimental validation of an eye occluder device developed from bottle caps for use in mammalian retinal studies related to dopamine. No previous studies have reported the use or systematic evaluation of similar bottle cap–based occluder devices in rabbits, highlighting the methodological approach’s novelty. Compared with studies using non-mammalian species, such as chicks or fish, our use of New Zealand White rabbits offers closer anatomical and neurochemical similarity to the human visual system (Troilo et al., 2019; Miyagishima et al., 2025). For example, chicks have a very different retinal architecture and scleral composition, which can limit the extrapolation of dopaminergic findings to humans (McGlinn et al., 2007). Although mammalian models better represent human retinal circuitry and scleral remodeling processes, they also introduce challenges related to device design, animal handling, and long-term occlusion tolerability (Miyagishima et al., 2025). Previous mammalian occluded studies have employed white plastic diffusers or translucent caps that allow light transmission while degrading image quality. For example, Kim et al. (2025) used white diffusers in a rabbit form deprivation model. In contrast, our black, opaque bottle-cap occluder was designed to minimize residual light input while permitting limited airflow through inferior ventilation holes. Despite these design differences, our biochemical endpoint—reduced D₁R concentration in occluded eyes—aligns with the general direction of dopaminergic changes reported in prior work, supporting our device’s validity as a paradigm for monocular deprivation and an inducer of neurochemical alterations (Huang et al., 2023; Kim et al., 2025). This pilot study also suggests that the bottle-cap occluder is well tolerated because no clinically evident ocular surface disease or orbital infection occurred during the 30-day occlusion period. This is an important practical consideration because some occluder designs can predispose patients to conjunctivitis, corneal damage, or skin irritation. The use of cushioning bandage tape and the secure head-and-neck wrapping likely contributed to both comfort and device stability, reducing the risk of dislodgement due to scratching. These features, combined with the low cost and wide availability of bottle caps, make the device a particularly attractive and strong candidate as a tool for laboratory research that primarily investigates retinal dopamine neurotransmitters operating in resource-limited settings or for studies requiring multiple animals. Nevertheless, several limitations must be acknowledged. The sample size was restricted to two rabbits, which is appropriate for a pilot feasibility and validation study but insufficient to support inferential statistics or detailed dose–response analyses. Only one biochemical marker—D₁R concentration—was assessed at a single time point, without concurrent measurements of refractive error, axial length, or other structural parameters. The initial D₁R on each rabbit is not the same because each rabbit has a different sensitivity to D1 dopamine, and the D₁R has a low affinity to dopamine. Furthermore, we did not evaluate other components of the dopaminergic pathway, such as overall dopamine levels, D₂-like receptor expression, or downstream cAMP signaling, which could provide a more comprehensive picture of the molecular changes induced by the rabbit eye model after 30 days. Despite these limitations, the present pilot data support the concept that a simple, black bottle cap–based occluder can induce dopaminergic alterations in rabbits, consistent with the established pathophysiology of the dopamine receptor mechanism. Future studies with larger sample sizes should integrate longitudinal biometric measurements, detailed retinal and scleral histology, and a broader panel of molecular markers to fully characterize the dopaminergic changes produced by this device. Comparative experiments against existing diffuser designs (e.g., white or translucent occluders) would also be valuable to determine whether the degree of light deprivation versus image degradation differentially affects dopaminergic signaling. ConclusionIn conclusion, this pilot study demonstrates that a simple eye patch made from a black bottle cap can be safely used in rabbits without visible adverse effects and is associated with a reduction in retinal D₁R levels, supporting its validity as a low-cost tool for future experimental myopia research with a larger sample size. AcknowledgmentsThe authors express sincere gratitude to the animal laboratory of Airlangga University Stem Cell Research and Development Center for providing facilities in the successful completion of this project. Conflict of interestThe authors declare no conflicts of interest. FundingThis study received no funding. Data availabilityData are available upon reasonable request to the first author. Authors' contributionsAll authors confirm their responsibility for the writing and content of the article, and each author approved the final version of the manuscript. Author contributions are as follows: Christina A., Reni P., and Irwanto drafted the original manuscript, developed the experimental protocol, and performed data analysis. Christina A. also conducted the experiments and evaluated the resulting data. ReferencesChen, S., Zhi, Z., Ruan, Q., Liu, Q., Li, F., Wan, F., Reinach, P.S., Chen, J., Qu, J. and Zhou, X. 2017. Bright light suppresses form-deprivation myopia development with activation of dopamine D1 receptor signaling in the ON pathway in retina. Invest. Ophthalmol. Vis. Sci. 58(4), 2306–2316; doi:10.1167/iovs.16-20402 Farshi, P., Fyk‐Kolodziej, B., Krolewski, D.M., Walker, P.D. and Ichinose, T. 2016. Dopamine D1 receptor expression is bipolar cell type-specific in the mouse retina. J. Comp. Neurol. 524(10), 2059–2079; doi:10.1002/cne.23932 Gan, J.H., Wang, M.J., Li, C.Y., Huang, Y., Liu, Z.H., Bai, W.L., Xu, W.J., Sun, M.H., Kang, M.T., Morgan, I., Wang, N. and Li, S.M. 2025. The interplay between form deprivation and dopamine signaling in regulating the expression of circadian rhythm-related genes in the retina of mice. Graefe's. Arch. For. Clin. Exp. Ophthalmol. 2025, 1–9; doi:10.1007/S00417-025-06950-2 Hu, R., Wu, K., Shi, J., Yu, J. and Yao, X.L. 2025. Glaucoma animal models in rabbits: state of the art and perspectives—a review. Anim. Model. Exp. Med. 8, 429–440; doi:10.1002/ame2.12565 Huang, Y., Chen, X., Zhuang, J. and Yu, K. 2023. The role of retinal dysfunction in myopia development. Cell. Mol. Neurobiol. 43; doi: 10.1007/s10571-022-01309-1 Jackson, C.R., Ruan, G.X., Aseem, F., Abey, J., Gamble, K., Stanwood, G., Palmiter, R.D., Iuvone, P.M. and McMahon, D.G. 2012. Retinal dopamine mediates multiple dimensions of light-adapted vision. J. Neurosci. 32(27), 9359–9368; doi:10.1523/JNEUROSCI.0711-12.2012 Junior, P.S., Wakeham, C.M. and von Gersdorff, H. 2024. Dopamine regulates the membrane potential and glycine release of AII amacrine cells via D1-like receptor modulation of gap junction coupling. bioRxiv; doi:10.1101/2024.12.11.625486 Kim, D.H., Hwang, J.M. and Yang, H.K. 2025. Topical dopamine application on form-deprivation myopia in rabbits. Life 15, 461; doi:10.3390/life15030461 Mcglinn, A.M., Baldwin, D.A., Tobias, J.W., Budak, M.T., Khurana, T.S. and Stone, R.A. 2007. Form-deprivation myopia in chick induces limited changes in retinal gene expression. Invest. Ophthalmol. Vis. Sci. 48, doi: 10.1167/iovs.06-1538 Miyagishima, K.J., Lai, X., Nath, A., Grimes, W.N., Ping, X., Diamond, J.S., Cameron, M.A., Li, W. and Nadal-Nicolás, F.M. 2025. Comparative analysis of tyrosine hydroxylase amacrine cells in the mammalian retina: distribution and quantification in mouse, rat, ground squirrel and macaque retinas. Int. J. Mol. Sci. 26, 6972; doi:10.3390/ijms26146972 Neve, K.A., Seamans, J.K. and Trantham-Davidson, H. 2004. Dopamine receptor signaling. J. Recept. Signal Transduct. Res. 24(3), 165–205; doi:10.1081/rrs-200029981 Ozkan, K., Aydın, B., Ucgul, A.Y., Bayrakceken, K., Ozmen, M.C. and Akata, R.F. 2025. Intrastromal suturing technique compared with interrupted corneal suturing technique, loose suture, and knot exposure: a comparative rabbit study. Surg. Innov. 2025, doi:10.1177/15533506251328456 Sahlholm, K., Svensson, P., Malo, M., Andersson, D.R. and Betari, N. 2025. Estimation of dopamine D1 receptor agonist binding kinetics using time-resolved functional assays: relation to agonist-induced receptor internalization by investigational antiparkinsonian therapeutics. ACS Chem. Neurosci. 16(13), 2502–2512; doi:10.1021/acschemneuro.5c00270 Troilo, D., Smith, E.L., Nickla, D.L., Ashby, R., Tkatchenko, A.V., Ostrin, L.A., Gawne, T.J., Pardue, M.T., Summers, J.A., Kee, C.S., Schroedl, F., Wahl, S. and Jones, L. 2019. IMI – report on experimental models of emmetropization and myopia. Invest. Ophthalmol. Vis. Sci. 60, M31; doi:10.1167/iovs.18-25967 Veruki, M.L., Liu, J.H., Singh, J.B., Luppi, M.S. and Hartveit, E. 2025. Activation of dopamine D1 receptors at the axon initial segment-like process of retinal AII amacrine cells modulates action potential firing. J. Neurosci. 45(33), 736252025; doi:10.1523/JNEUROSCI.0736-25.2025 Zhou, X., Pardue, M.T., Iuvone, P.M. and Qu, J. 2017. Dopaminee signaling and myopia development: what are the key challenges. Prog. Retin. Eye Res. 61, 60–71; doi:10.1016/j.preteyeres.2017.06.003 | ||
| How to Cite this Article |
| Pubmed Style Aritonang C, Irwanto I, Prastyani R. Validation of eye occluding device developed from bottle caps for dopamine studies in rabbits: A pilot study. Open Vet. J.. 2026; 16(3): 1853-1858. doi:10.5455/OVJ.2026.v16.i3.40 Web Style Aritonang C, Irwanto I, Prastyani R. Validation of eye occluding device developed from bottle caps for dopamine studies in rabbits: A pilot study. https://www.openveterinaryjournal.com/?mno=300129 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.40 AMA (American Medical Association) Style Aritonang C, Irwanto I, Prastyani R. Validation of eye occluding device developed from bottle caps for dopamine studies in rabbits: A pilot study. Open Vet. J.. 2026; 16(3): 1853-1858. doi:10.5455/OVJ.2026.v16.i3.40 Vancouver/ICMJE Style Aritonang C, Irwanto I, Prastyani R. Validation of eye occluding device developed from bottle caps for dopamine studies in rabbits: A pilot study. Open Vet. J.. (2026), [cited March 31, 2026]; 16(3): 1853-1858. doi:10.5455/OVJ.2026.v16.i3.40 Harvard Style Aritonang, C., Irwanto, . I. & Prastyani, . R. (2026) Validation of eye occluding device developed from bottle caps for dopamine studies in rabbits: A pilot study. Open Vet. J., 16 (3), 1853-1858. doi:10.5455/OVJ.2026.v16.i3.40 Turabian Style Aritonang, Christina, Irwanto Irwanto, and Reni Prastyani. 2026. Validation of eye occluding device developed from bottle caps for dopamine studies in rabbits: A pilot study. Open Veterinary Journal, 16 (3), 1853-1858. doi:10.5455/OVJ.2026.v16.i3.40 Chicago Style Aritonang, Christina, Irwanto Irwanto, and Reni Prastyani. "Validation of eye occluding device developed from bottle caps for dopamine studies in rabbits: A pilot study." Open Veterinary Journal 16 (2026), 1853-1858. doi:10.5455/OVJ.2026.v16.i3.40 MLA (The Modern Language Association) Style Aritonang, Christina, Irwanto Irwanto, and Reni Prastyani. "Validation of eye occluding device developed from bottle caps for dopamine studies in rabbits: A pilot study." Open Veterinary Journal 16.3 (2026), 1853-1858. Print. doi:10.5455/OVJ.2026.v16.i3.40 APA (American Psychological Association) Style Aritonang, C., Irwanto, . I. & Prastyani, . R. (2026) Validation of eye occluding device developed from bottle caps for dopamine studies in rabbits: A pilot study. Open Veterinary Journal, 16 (3), 1853-1858. doi:10.5455/OVJ.2026.v16.i3.40 |