| Research Article | ||
Open Vet. J.. 2026; 16(3): 1954-1960 Open Veterinary Journal, (2026), Vol. 16(3): 1954-1960 Research Article Evaluation of dorper sheep semen quality preserved in milk egg yolk extender at room and cold temperaturesTri Wahyu Suprayogi1*, Suherni Susilowati1, Tatik Hernawati1, Supriyadi Supriyadi2 and Satriawan Wedniyanto Putra31Division of Veterinary Reproduction, Faculty of Veterinary Medicine, Universitas Airlangga, Campus C UNAIR, Mulyorejo, Surabaya 60115, East Java, Indonesia 2Master Program in Reproductive Biology, Faculty of Veterinary Medicine, Universitas Airlangga, Campus C UNAIR, Mulyorejo, Surabaya 60115, East Java, Indonesia 3Mayapada Hospital, Jl. Mayjend Sungkono No. 20, Surabaya 60225, East Java, Indonesia *Corresponding Author: Tri Wahyu Suprayogi. 1Division of Veterinary Reproduction, Faculty of Veterinary Medicine, Universitas Airlangga, Campus C UNAIR, Mulyorejo, Surabaya 60115, East Java, Indonesia. Email: tri-w-s [at] fkh.unair.ac.id Submitted: 22/11/2025 Revised: 11/02/2026 Accepted: 23/02/2026 Published: 31/03/2026 © 2026 Open Veterinary Journal
AbstractBackground: Artificial insemination (AI) is widely used to improve genetic progress and reproductive efficiency in sheep production systems, including Dorper sheep. The success of AI programs for liquid semen depends on the ability to maintain semen quality during storage. Although milk–egg yolk extenders are commonly used for ram semen preservation, the optimal egg yolk concentration for maintaining sperm quality under different storage conditions remains unclear. Aim: This study evaluated the effects of 5% and 10% egg yolk extenders on the quality of Dorper ram semen stored at room temperature (≈25°C) and 5°C. Methods: Semen was collected from healthy Dorper rams using an artificial vagina and divided into four treatment groups based on the extender concentration and storage temperature. Semen stored at room temperature was evaluated at 0, 2, 4, 6, and 8 hours, whereas semen stored at 5°C was evaluated daily for 5 days. Sperm quality was assessed based on progressive motility, sperm vitality, and plasma membrane integrity. Oxidative stress was evaluated by measuring malondialdehyde concentration and catalase activity. Data were analyzed using one-way analysis of variance followed by Tukey’s test (p < 0.05). Results: Within each storage condition, semen diluted with the 5% milk–egg yolk extender consistently maintained higher sperm motility, vitality, membrane integrity, and oxidative stability compared with the 10% extender. Conclusion: These findings indicate that a 5% milk–egg yolk extender is a practical and effective option for the short-term preservation of Dorper ram semen for AI. Keywords: Dorper sheep, Liquid semen preservation, Milk–egg yolk extender, Sperm quality, Artificial insemination. IntroductionArtificial insemination (AI) is a fundamental reproductive biotechnology for accelerating genetic improvement and enhancing reproductive efficiency in sheep production systems, particularly in high-value meat breeds such as Dorper sheep. The success of AI programs is highly dependent on the maintenance of semen quality during storage before insemination. Effective semen preservation facilitates semen distribution between farms, maximizes the use of each ejaculate, and provides greater breeding management flexibility throughout the year (Galarza et al., 2020; Godfrey et al., 2020). Milk–egg yolk extenders have been widely used for preserving ram semen due to their ability to protect spermatozoa from physical and biochemical damage during storage. Egg yolk contains low-density lipoproteins, phospholipids, and cholesterol, which interact with the sperm plasma membrane and improve its stability, thereby reducing cold shock and oxidative stress-related damage. Milk-based extenders supplemented with low concentrations of egg yolk (approximately 5%) maintain sperm motility, membrane integrity, and viability during liquid storage at 4°C–5°C (Gil et al., 2011; Galarza et al., 2020). Although refrigerated storage has been extensively investigated, information on the effectiveness of milk–egg yolk extenders for the short-term preservation of ram semen at room temperature remains limited. Previous studies have demonstrated that milk–egg yolk extenders can preserve ram semen viability for up to 96 hours under refrigerated conditions (Godfrey et al., 2020), whereas skim milk-based extenders have been shown to improve sperm motility and fertility outcomes in Dorper rams during cold storage (Bai et al., 2020). Storage temperature is a critical factor affecting sperm survival during liquid preservation. Refrigeration reduces sperm metabolic activity and bacterial proliferation, thereby slowing cellular degradation. However, exposure to low temperatures may induce cold shock, alter membrane phospholipid phase transitions, and increase oxidative stress by excessively producing reactive oxygen species (ROS) (El Shereif et al., 2022). In contrast, storage at room temperature (approximately 25°C) avoids cold shock but accelerates sperm metabolism, leading to rapid depletion of intracellular energy reserves and increased susceptibility to lipid peroxidation. Lipid peroxidation is commonly evaluated by measuring malondialdehyde (MDA), a widely accepted biomarker of oxidative damage to sperm membrane lipids (Aitken and Roman, 2008; Agarwal et al., 2014). Elevated MDA levels are associated with impaired sperm motility, membrane integrity, and fertilizing capacity. Antioxidant defense systems, including enzymatic antioxidants such as catalase, play a crucial role in protecting spermatozoa by decomposing hydrogen peroxide and limiting oxidative injury during semen storage. Breed-specific differences in semen preservation response have also been reported. Dorper rams are more susceptible to storage-induced sperm damage than other sheep breeds, emphasizing the importance of optimizing extender composition and storage conditions for this genotype (Goshme S Banerjee et al., 2020). Therefore, systematic evaluation of extender formulations under different storage temperatures is necessary to improve semen preservation strategies tailored to Dorper sheep. Considering these physiological and practical aspects, evaluating milk–egg yolk extender formulations under both refrigerated and room temperature conditions is relevant for identifying effective, low-cost, and field-applicable semen preservation methods. This study aimed to evaluate the quality of Dorper ram semen diluted in milk–egg yolk extenders containing 5% and 10% egg yolk during storage at room temperature (25°C) and at 5°C. Semen quality was assessed based on sperm motility, vitality, plasma membrane integrity, and oxidative stress indicators (Arando et al., 2023; Rahman et al., 2023; Menon et al., 2024; Sevilla et al., 2024). Materials and MethodsAnimal and semen collectionSemen was collected from eight clinically healthy adult Dorper rams (n=8 ejaculates) maintained under uniform management and feeding conditions at a local breeding facility. Each ejaculate was obtained from a different ram to avoid bias. The rams were aged 2–3 years, had an average body weight of 65–75 kg, and had no history of reproductive disorders. Rams were sexually rested for 3–5 days before semen collection to ensure consistent semen quality. Semen collection was performed using an artificial vagina pre-warmed to 42°C–45°C. All collection tubes and laboratory equipment in direct contact with semen were pre-warmed to 37°C to prevent thermal shock during handling. Semen samples were placed in a thermos container immediately after collection and transported to the laboratory for evaluation. Evaluation of semen and ejaculate selectionMacroscopic evaluation included the assessment of semen volume (ml), color, consistency, and odor. Semen pH was measured using a digital pH meter. Microscopic evaluation was conducted immediately after collection using a phase-contrast microscope (Nikon Eclipse E100, Japan) at 400× magnification. Progressive sperm motility was subjectively estimated by examining at least five microscopic fields per sample. Sperm vitality and plasma membrane integrity were evaluated using eosin–nigrosin staining following standard procedures. A minimum of 200 spermatozoa were evaluated per slide. Only ejaculates with progressive motility ≥70% and vitality ≥70% were included in the experiment. Preparation of materials and extenderWhole milk, fresh chicken egg yolk, fructose, sodium citrate, sodium chloride, distilled water, penicillin (3,000,000 IU/l extender), and streptomycin sulfate (1 g/l extender) were used for extender preparation. Sperm vitality was assessed using eosin–nigrosin staining solution. The milk–egg yolk extenders were prepared aseptically on the day of semen collection. The egg yolk was carefully separated from the albumen and filtered through sterile gauze to remove impurities. Egg yolk was then added to whole milk to obtain extender formulations containing 5% and 10% egg yolk (v/v). Fructose (0.5 g/100 ml extender) was added as an energy source, and antibiotics were added to prevent bacterial growth. The pH of the extenders was adjusted to 6.8–7.0 using sodium citrate buffer. The prepared extenders were equilibrated at 37°C before semen dilution. Semen dilution and storageSemen samples were diluted with the respective extenders at a semen:extender ratio of 1:4 to obtain a final sperm concentration of approximately 200 × 10⁶ spermatozoa/ml. Dilution was performed gradually at 37°C to minimize osmotic and thermal shock. For refrigerated storage, diluted semen samples were gradually cooled from room temperature to 5°C over approximately 2 hours. Samples assigned to room temperature storage were maintained at approximately 25°C. All samples were stored in sterile tubes in the dark to minimize light-induced oxidative damage. Experimental designWe employed a factorial experimental design with two factors: egg yolk concentration (5% and 10%) and storage temperature (room temperature ≈25°C, and refrigerated temperature 5°C). Four treatment groups were established as follows: Group I: 5% egg yolk extender, stored at room temperature; Group II: 10% egg yolk extender, stored at room temperature; Group III: 5% egg yolk extender, stored at 5°C; Group IV: 10% egg yolk extender, stored at 5°C. Each treatment group consisted of eight replicates (n=8). Semen stored at room temperature was evaluated at 0, 2, 4, 6, and 8 hours, whereas semen stored at 5°C was evaluated daily for up to 5 days. Semen quality assessmentSemen quality parameters included progressive sperm motility (%), sperm vitality (%), and plasma membrane integrity (%). Lipid peroxidation was assessed by measuring MDA concentration (nmol/ml) using the thiobarbituric acid reactive substances method. Antioxidant activity was evaluated by measuring catalase activity (U/mg protein). Oxidative stress parameters were assessed only in semen samples stored at 5°C. Statistical analysisData were analyzed using one-way analysis of variance within each storage temperature to evaluate the effect of egg yolk concentration. Tukey’s post hoc test was applied when significant differences were detected. Statistical significance was set at p < 0.05. Results are presented as mean ± standard deviation (SD). All analyses were performed using SPSS version 25.0 (IBM Corp., Armonk, NY). Ethical approvalThis study was approved by the Animal Ethics Committee of the Faculty of Veterinary Medicine, Universitas Airlangga (Approval Number: 1.KEH.108.07.2024). All procedures involving animals were conducted in accordance with internationally accepted animal welfare guidelines to minimize stress and ensure animal welfare. ResultsMacroscopic and initial microscopic quality of semenTable 1 summarizes the macroscopic characteristics of fresh Dorper ram semen (n=8 ejaculates). All ejaculates exhibited normal physical characteristics, including typical odor, yellowish-white color, and lumpy consistency, indicating good semen quality suitable for further processing. The semen pH was consistent among samples. Table 1. Macroscopic characteristics of Dorper ram semen (n=8).
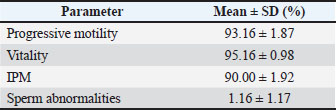
The initial microscopic evaluation (Table 2) demonstrated high semen quality prior to dilution and storage. Progressive motility, sperm vitality, and intact plasma membrane (IPM) integrity were consistently high, whereas sperm abnormalities were minimal. All ejaculates met the predefined inclusion criteria and were used in subsequent storage experiments. Table 2. Initial microscopic semen quality of Dorper rams (n=8).
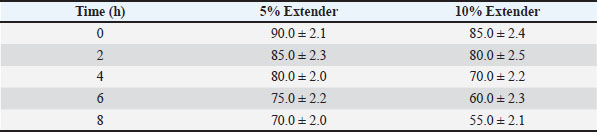
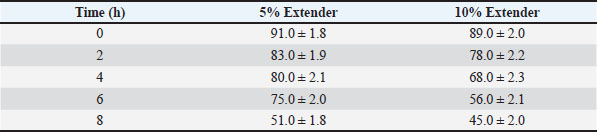
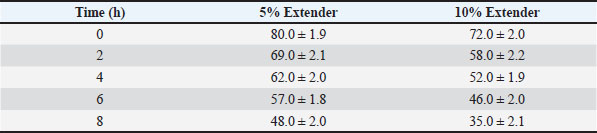
Sperm quality during storage at room temperature (≈25°C)Tables 3–5 present the changes in sperm quality parameters during storage at room temperature. Progressive motility, sperm vitality, and IPM integrity decreased significantly over storage time in both extender groups (p < 0.05). Table 3. Progressive motility (%) of Dorper ram semen stored at room temperature (≈25°C) (n=8).
Table 4. Sperm vitality (%) of Dorper ram semen stored at room temperature (≈25°C) (n=8).
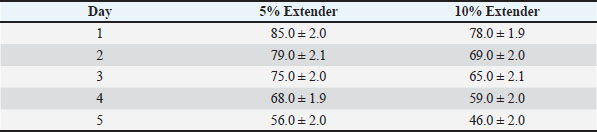
Table 5. IPM integrity (%) of Dorper ram semen stored at room temperature (≈25°C) (n=8).
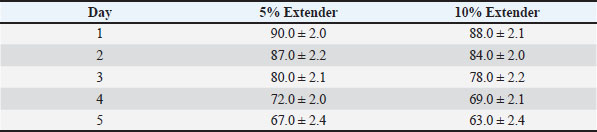
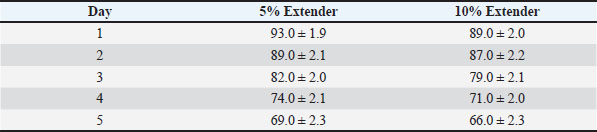
At all evaluated time points, semen diluted with the 5% egg yolk extender maintained significantly higher progressive motility, vitality, and IPM integrity compared with the 10% egg yolk extender (p < 0.05). Semen stored in the 5% egg yolk extender maintained progressive motility above the minimum threshold recommended for AI for a longer duration than semen extended with 10% egg yolk. Sperm quality during cold storage at 5°CThe sperm quality parameters during refrigerated storage are shown in Tables 6–8. Progressive motility, sperm vitality, and IPM integrity declined gradually throughout the 5-day storage period in both extender groups (p < 0.05). Table 6. Progressive motility (%) of Dorper ram semen stored at 5°C (n=8).
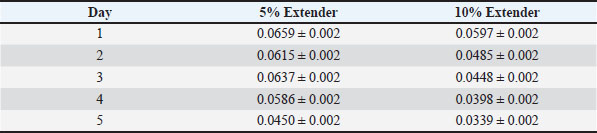
Table 7. Sperm vitality (%) of Dorper ram semen stored at 5°C (n=8).
Table 8. IPM integrity (%) of Dorper ram semen stored at 5°C (n=8).
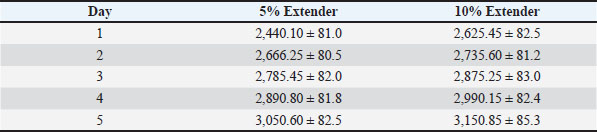
Semen samples extended with 5% egg yolk consistently exhibited significantly higher motility, vitality, and membrane integrity than those extended with 10% egg yolk at corresponding storage times (p < 0.05). Semen preserved with the 5% egg yolk extender maintained progressive motility within acceptable limits for AI until the final day of storage. Oxidative stress indicators during cold storageTables 9 and 10 present the effects of extender concentration on oxidative stress parameters during cold storage. The MDA concentration increased significantly over time in both extender groups (p < 0.05). Semen samples diluted with 10% egg yolk extender showed significantly higher MDA levels than those diluted with 5% extender at corresponding storage times (p < 0.05). Table 9. MDA concentration (nmol/ml) of Dorper ram semen stored at 5°C (n=8).
Table 10. Catalase activity (U/mg protein) of Dorper ram semen stored at 5°C (n=8).
Catalase activity progressively decreased during refrigerated storage (p < 0.05). However, semen preserved in the 5% egg yolk extender maintained significantly higher catalase activity compared with the 10% egg yolk extender throughout the storage period (p < 0.05). DiscussionThe fresh Dorper ram semen used in this study exhibited high baseline quality prior to dilution and storage, as evidenced by elevated progressive motility, sperm vitality, and IPM integrity, along with a low proportion of abnormal spermatozoa. These findings confirm that all ejaculates met the minimum quality requirements for semen preservation studies. High initial semen quality is widely recognized as a prerequisite for successful liquid semen storage, as ejaculates with suboptimal baseline characteristics are more susceptible to accelerated deterioration during preservation (Bustani and Baiee, 2021). A reduction in sperm quality parameters was observed immediately after dilution and equilibration compared with fresh semen. This initial decline is commonly attributed to handling procedures, osmotic adjustment, and temperature equilibration, which may transiently affect sperm membrane stability and metabolic homeostasis. Similar post-dilution reductions in sperm motility and membrane integrity have been consistently reported in ram semen preservation studies and are considered a normal physiological response rather than an indication of extender inadequacy (Gil et al., 2011). During storage at room temperature (≈25°C), sperm quality progressively declined in both extender groups, reflecting the increased metabolic activity and accelerated Adenosine Triphosphate consumption characteristic of warm storage conditions. Under these circumstances, excessive production of ROS can overwhelm the antioxidant defense mechanisms of spermatozoa, leading to lipid peroxidation and membrane damage (Fadl et al., 2022). The consistently superior performance of the 5% milk–egg yolk extender in maintaining motility, vitality, and IPM integrity suggests that the composition of the extender plays a critical role in moderating metabolic and oxidative stress during short-term storage at ambient temperature. Similarly, during refrigerated storage at 5°C, sperm quality parameters gradually declined over the 5-day storage period, consistent with previous reports on chilled ram semen preservation. Although refrigeration effectively slows sperm metabolism and bacterial proliferation, it also exposes spermatozoa to cold shock and membrane phase transitions, which may compromise membrane integrity if not adequately protected (Gil et al., 2011; Fadl et al., 2022). The superior preservation observed with the 5% egg yolk extender indicates that this formulation provided sufficient membrane stabilization while minimizing the chilling-associated negative effects (Arando et al., 2023; Rahman et al., 2023; Menon et al., 2024; Sevilla et al., 2024). The evaluation of oxidative stress markers further elucidates the mechanisms underlying the observed differences between extender concentrations. The progressive increase in MDA levels during cold storage reflects ongoing lipid peroxidation, while the concomitant decline in catalase activity indicates a reduction in antioxidant capacity. Semen diluted with 10% egg yolk extender exhibited higher MDA concentrations and lower catalase activity than semen diluted with 5% extender, suggesting an imbalance between ROS production and antioxidant defense. Excessive egg yolk content may increase the availability of lipid substrates for peroxidation or alter extender physicochemical properties of extenders, such as viscosity, thereby negatively affecting sperm metabolism and oxidative stability (Neila Montero et al., 2024). Therefore, the superior efficacy of the 5% milk–egg yolk extender can be attributed to an optimal concentration of low-density lipoproteins and phospholipids that stabilize the sperm plasma membrane without imposing excessive metabolic or osmotic stress. Conversely, higher egg yolk concentrations may hinder sperm motility, disrupt membrane dynamics, and intensify oxidative damage, ultimately accelerating sperm quality deterioration under both storage conditions. Although the present study did not include fertility trials or intermediate storage temperatures, such as 17°C, the findings provide valuable insight into the optimization of extender composition for preservation of Dorper ram semen. Dorper rams exhibit increased sensitivity to storage-induced sperm damage than other breeds, underscoring the importance of breed-specific preservation strategies. The results of this study demonstrate that a milk–egg yolk extender containing 5% egg yolk is more effective than a 10% formulation in preserving sperm motility, vitality, membrane integrity, and oxidative stability within each storage temperature evaluated. ConclusionSemen quality progressively decreased during liquid storage under all conditions. Within each storage temperature, the extender composition significantly influenced semen preservation. The milk–egg yolk extender containing 5% egg yolk consistently maintained higher sperm motility, vitality, plasma membrane integrity, and oxidative stability than the 10% formulation. These findings indicate that the 5% milk–egg yolk extender is a reliable, practical, and cost-effective option for preserving Dorper ram semen for AI programs. AcknowledgmentsThe authors gratefully acknowledge the Dean of the Faculty of Veterinary Medicine, Universitas Airlangga, for the institutional support that facilitated the completion of this study. Funding: This research was funded under the Superior Basic Research (PDU) scheme, contract number 1515/UN3.FKH/PT.01.03/2024. This research was conducted in the absence of any commercial or financial relationships that could be understood as a potential conflict of interest. Conflict of interestAll authors declare no conflicts of interest related to this study. Authors’ contributionTri Wahyu Suprayogi contributed to semen collection, data acquisition, and manuscript drafting. Suherni Susilowati supervised the research and provided critical revisions to the manuscript. Tatik Hernawati performed the laboratory analyses and assisted with data interpretation. Supriyadi performed the statistical analysis and contributed to the research methodology. Satriawan Wedniyanto Putra assisted with manuscript formatting and final proofreading. All authors have read and approved the final version of the manuscript. Data availabilityAll data generated or analyzed during this study are included in this article and its accompanying tables. ReferencesAgarwal, A., Virk, G., Ong, C. and du Plessis, S.S. 2014. Effect of oxidative stress on male reproduction. World J. Men’s Health 32(1), 1–17. Aitken, R.J. and Roman, S.D. 2008. Antioxidant systems and oxidative stress in the testes. Oxid. Med. Cell. Longev. 1(1), 15–24. Arando , A., Delgado, J.V., Pérez-Marín, C.C. and Barón, P. 2023. Effects of storage temperature and antioxidant supplementation on ram semen preserved in liquid form. Theriogenology 205, 52–59. Bai, Y.Y., Xu, X., Yu, X.J., Guo, J., Dong, X.X., Wang, X.Y., Zhao, Z.A. and Wang, J. 2020. Skimmed milk diluent promotes the sperm motility and conception rate of Dorper sheep compared to vitamin B12 diluent. CryoLetters 41, 365–370. Bustani, G.S. and Baiee, H. 2021. Semen extenders: an evaluative overview of preservative media for livestock semen. Vet. World. 14, 1449–1458. El Shereif, A., Fath El Bab, A.Z., Nour El Din, A.N.M. and Salem, M.H. 2022. Trehalose as an alternative to egg yolk in ram semen extender. J. Adv. Vet. Res. 12, 221–226. Fadl, A.M., El Sherbiny, H.R. and Abdelnaby, E.A. 2022. INRA82 extender enhances semen quality in ram under cooled and frozen conditions. Asian-Aust. J. Anim. Sci. 35, 1505–1514. Galarza, D.A., Sebastián, L. and Santiago, J. 2020. Supplementing a skimmed milk egg yolk-based extender with L-carnitine helps maintain motility, membrane integrity and fertilizing capacity of chilled ram sperm. Reprod. Domest. Anim. 55, 805–813. Gil, J., Fierro, S., Bentancur, O. and Olivera‐Muzante, J. 2011. Chilled storage of ram semen improves with the addition of egg yolk and glycerol to milk-based extenders. Reprod. Domestic. Anim. 46(3), 503–507. Godfrey, R.W., Keiper, S. and Lakos, S. 2020. Evaluation of extended hair sheep ram semen stored as liquid at 5 °C. J. Anim. Sci. 98, 74–75. Goshme, S., Banerjee, S., Rekik, M., Haile, A., Yitagesu, E. and Getachew, T. 2020. Evaluation and characterization of semen quality in rams of Menz, Dorper and Awassi crosses in different seasons in Ethiopia. Livest. Res. Rural Dev. 32, 1–10. Menon, A.G., Chandra, V. and Kumaresan, A. 2024. Advances in semen extender formulations for small ruminants: focus on oxidative stress management. Small Ruminant Res. 230, 106123. Neila Montero, M., Alvarez, M., Riesco, M.F., Soriano Úbeda, C., Montes Garrido, R., Palacin Martinez, C., De Paz, P. and Anel, L. 2024. The adaptation time to the extender as a crucial step for an accurate evaluation of ram sperm quality during liquid storage. Vet. Sci. 11, 132. Rahman, S.U., Qamar, A.Y., Fang, X., Shang, Q., Wu, Z. and Fang, X. 2023. Antioxidant strategies to improve ram semen quality during liquid storage: a review. Anim. Reprod. Sci. 255, 107113. Sevilla, F., Muça, G., Turmalaj, L., Silvestre, M.A., Araya-Zúñiga, I. and Valverde, A. 2024. An overview on extenders used in ram sperm cryopreservation. Agron. Mesoam. 35, 59591. | ||
| How to Cite this Article |
| Pubmed Style Suprayogi TW, Susilowati S, Hernawati T, Supriyadi S, Putra SW. Evaluation of dorper sheep semen quality preserved in milk egg yolk extender at room and cold temperatures. Open Vet. J.. 2026; 16(3): 1954-1960. doi:10.5455/OVJ.2026.v16.i3.51 Web Style Suprayogi TW, Susilowati S, Hernawati T, Supriyadi S, Putra SW. Evaluation of dorper sheep semen quality preserved in milk egg yolk extender at room and cold temperatures. https://www.openveterinaryjournal.com/?mno=298557 [Access: April 02, 2026]. doi:10.5455/OVJ.2026.v16.i3.51 AMA (American Medical Association) Style Suprayogi TW, Susilowati S, Hernawati T, Supriyadi S, Putra SW. Evaluation of dorper sheep semen quality preserved in milk egg yolk extender at room and cold temperatures. Open Vet. J.. 2026; 16(3): 1954-1960. doi:10.5455/OVJ.2026.v16.i3.51 Vancouver/ICMJE Style Suprayogi TW, Susilowati S, Hernawati T, Supriyadi S, Putra SW. Evaluation of dorper sheep semen quality preserved in milk egg yolk extender at room and cold temperatures. Open Vet. J.. (2026), [cited April 02, 2026]; 16(3): 1954-1960. doi:10.5455/OVJ.2026.v16.i3.51 Harvard Style Suprayogi, T. W., Susilowati, . S., Hernawati, . T., Supriyadi, . S. & Putra, . S. W. (2026) Evaluation of dorper sheep semen quality preserved in milk egg yolk extender at room and cold temperatures. Open Vet. J., 16 (3), 1954-1960. doi:10.5455/OVJ.2026.v16.i3.51 Turabian Style Suprayogi, Tri Wahyu, Suherni Susilowati, Tatik Hernawati, Supriyadi Supriyadi, and Satriawan Wedniyanto Putra. 2026. Evaluation of dorper sheep semen quality preserved in milk egg yolk extender at room and cold temperatures. Open Veterinary Journal, 16 (3), 1954-1960. doi:10.5455/OVJ.2026.v16.i3.51 Chicago Style Suprayogi, Tri Wahyu, Suherni Susilowati, Tatik Hernawati, Supriyadi Supriyadi, and Satriawan Wedniyanto Putra. "Evaluation of dorper sheep semen quality preserved in milk egg yolk extender at room and cold temperatures." Open Veterinary Journal 16 (2026), 1954-1960. doi:10.5455/OVJ.2026.v16.i3.51 MLA (The Modern Language Association) Style Suprayogi, Tri Wahyu, Suherni Susilowati, Tatik Hernawati, Supriyadi Supriyadi, and Satriawan Wedniyanto Putra. "Evaluation of dorper sheep semen quality preserved in milk egg yolk extender at room and cold temperatures." Open Veterinary Journal 16.3 (2026), 1954-1960. Print. doi:10.5455/OVJ.2026.v16.i3.51 APA (American Psychological Association) Style Suprayogi, T. W., Susilowati, . S., Hernawati, . T., Supriyadi, . S. & Putra, . S. W. (2026) Evaluation of dorper sheep semen quality preserved in milk egg yolk extender at room and cold temperatures. Open Veterinary Journal, 16 (3), 1954-1960. doi:10.5455/OVJ.2026.v16.i3.51 |