| Research Article | ||
Open Vet. J.. 2026; 16(2): 1329-1337
Open Veterinary Journal, (2026), Vol. 16(2): 1329-1337 Research Article Feline mammary hyperplasia: A comparative study of medical versus surgical treatment approachesNehal Ibrahim1*, Fatma M. Ibrahim2, Maha M. Elgebaly2, Shimaa A. Ezzeldein1, Mahmoud M. Emam3 and Walaa O. M. Youssef11Department of Surgery, Anesthesiology and Radiology, Faculty of Veterinary Medicine, Zagazig University, Zagazig, Egypt 2Department of Theriogenology, Faculty of Veterinary Medicine, Zagazig University, Zagazig, Egypt 3Department of Animal Medicine, Faculty of Veterinary Medicine, Zagazig University, Zagazig, Egypt *Corresponding Author: Nehal Ibrahim. Department of Surgery, Anesthesiology and Radiology, Faculty of Veterinary Medicine, Zagazig University, Zagazig, Egypt, Email: nehalnabil128 [at] gmail.com Submitted: 18/11/2025 Revised: 11/01/2026 Accepted: 19/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
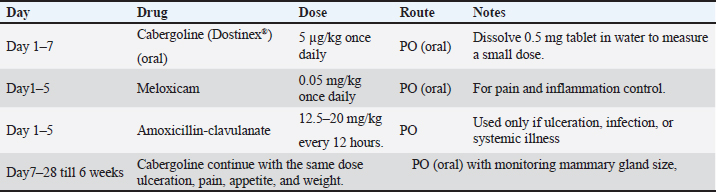
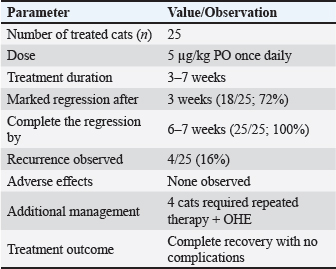
AbstractBackground: Feline mammary fibroepithelial hyperplasia (FMH) is a benign, progesterone-dependent mammary disorder that predominantly occurs in young intact female cats during or shortly after estrus. This condition is characterized by rapid and diffuse mammary enlargement, which often leads to discomfort and local inflammation. Aim: This study aimed to describe the clinical, biochemical, and therapeutic aspects of FMH in cats and compare the outcomes of medical and surgical management approaches. Methods: We evaluated 40 intact female cats diagnosed with FMH between 2023 and 2025. The clinical data, hormone levels, and imaging findings were recorded. Twenty-five cats received cabergoline (5 µg/kg orally once daily), and 15 underwent ovariohysterectomy (OHE). Serum progesterone and estradiol levels were statistically analyzed and measured. Results: The most commonly affected animal breeds were Persian (77.5%), followed by Egyptian Mau (12.5%) and Siamese (10%), with a mean age of 12.4 ± 6.5 months. Clinical examination revealed firm, swollen mammary lobes ranging from 2 to 13 cm in diameter, with 37.5% showing edema or inflammation. Progesterone levels were significantly higher in affected cats (16.27 ± 8.7 ng/ml) than in healthy controls (2.45 ± 1.78 ng/ml; p < 0.01), whereas estradiol levels showed a nonsignificant decrease (p=0.066). Cabergoline therapy led to marked regression within 3-6 weeks in 84% of cats, while complete recovery was achieved after OHE within 2-4 weeks, without recurrence. The combined approach provided the fastest resolution and minimal complications. Conclusion: FMH disorder in cats is closely associated with elevated progesterone levels. Although both medical and surgical management are effective, OHE offers a permanent cure, whereas cabergoline provides a safe, noninvasive alternative suitable for breeding females. Combined therapy may accelerate recovery and prevent relapse. Keywords: Feline mammary hyperplasia, Progesterone, Cabergoline, Ovariohysterectomy, Hormonal imbalance. IntroductionThe mammary glands in cats are organized into four to five pairs of mammary complexes that are symmetrically distributed along the ventral surface, extending from the thoracic to the inguinal regions. These complexes are anatomically categorized as axillary, thoracic, abdominal, and inguinal mammary glands (Görlinger et al., 2002). Because of the wide distribution and high prevalence of mammary pathologies in felines, the systematic examination of mammary tissue is considered an essential part of routine physical assessment in both intact and spayed cats. Approximately 20% of non-neoplastic mammary masses are reported as mammary gland hyperplasia (Görlinger et al., 2002). Feline mammary fibroepithelial hyperplasia (FMH), also known as feline mammary hypertrophy, fibroadenomatous change, or fibroadenoma complex, is a benign, progesterone-dependent proliferation of the ductal epithelium and surrounding stromal tissue of the mammary glands (Allen, 1973; Johnson, 1994; Johnston et al., 2001). This condition can occur in both intact females and males, as well as in spayed females exposed to exogenous progestins for estrus suppression (Johnson, 1994). The disorder is characterized by rapid and diffuse enlargement of one or multiple mammary glands, which can develop within 2–5 weeks (Rutteman and Withrow, 2001; Loretti et al., 2004; Loretti et al., 2005; Sontas et al., 2008). The pathophysiology of FMH is closely associated with progesterone activity in the mammary epithelium. Progesterone stimulates mammary glandular proliferation by binding to specific receptors in ductal and stromal tissues. This hormonal influence can be attributed to endogenous luteal secretion after ovulation or exogenous administration of synthetic progestins, commonly used for contraception or behavioral modification in queens (Hayden et al., 1981; Leidinger et al., 2011). Proliferative changes are reversible and usually regress spontaneously during luteolysis or following the discontinuation of exogenous progesterone sources (Johnson, 1994; Wehrend et al., 2001). Clinically, FMH often presents as a sudden, symmetrical enlargement of the mammary glands, which may simultaneously affect the thoracic, abdominal, and inguinal complexes. On palpation, the affected glands are typically firm, warm, and non-painful. However, ulceration, necrosis, and secondary bacterial infection may develop in severe or neglected cases, leading to systemic illness (Loretti et al., 2005). Although FMH is non-neoplastic, its macroscopic appearance can mimic mammary carcinoma or adenocarcinoma, particularly when ulcerated, thus requiring careful differentiation through clinical and histopathological evaluation (Sontas et al., 2008; Murphy 2009; Mayayo et al., 2018). The diagnosis of FMH is primarily clinical and is supported by a detailed reproductive history and hormonal assessment. Rapid enlargement of multiple mammary glands in a young or recently treated cat strongly suggests FMH. Serum progesterone concentration measurement or history of progesterone-based medication further substantiates the diagnosis (Johnston et al., 2001;Vitasek and Dendisova, 2006; Mayayo et al., 2018). Imaging tools, such as ultrasound, may aid in evaluating glandular architecture. Fine-needle aspiration or biopsy can confirm the benign proliferative nature of the tissue in ambiguous cases (Mayayo et al., 2018). FMH management can be medical or surgical, depending on severity, underlying hormonal status, and the animal’s breeding value. Medical therapy typically involves the administration of antiprogestins, such as aglepristone, which competitively inhibit progesterone receptors and induce regression of glandular hyperplasia (Wehrend et al., 2001; Nak et al., 2004). Alternatively, luteolytic agents may be used to accelerate luteal regression and reduce endogenous progesterone levels. Ovariohysterectomy (OHE) remains a definitive option to eliminate the hormonal stimulus and prevent recurrence in intact females not intended for breeding (Keskin et al., 2008). Mastectomy has been used in the past to manage severe cases with ulceration or necrosis; however, it is now largely discouraged due to its invasiveness and the availability of effective pharmacologic treatments (Görlinger et al., 2002). FMH recurrence is relatively uncommon but has been reported, particularly in animals re-exposed to progesterone or in incomplete medical management cases (Loretti et al., 2004; Jelinek et al., 2007; Jurka and Max, 2009; Leidinger et al., 2011). It is crucial to inform cat owners about the benign nature of FMH, its hormonal basis, and the importance of preventing future exposure to progesterone from a clinical standpoint. Early recognition and appropriate therapy can prevent complications such as necrosis, secondary infection, or misdiagnosis as a malignant disease. Therefore, the present study aims to comprehensively describe the etiology, pathophysiology, and clinical characteristics of feline mammary fibroepithelial hyperplasia, highlighting the diagnostic approaches that distinguish it from mammary neoplasia. Additionally, it seeks to outline the current treatment protocols, management strategies, prognosis, and preventive measures relevant to this condition. Materials and MethodsAnimal and study designMedical case records of 40 female cats with confirmed FMH between 2023 and the end of 2025 were selected and reviewed at the Faculty of Veterinary Medicine Clinic in Zagazig University, Egypt. Diagnosis and confirmation depended on anamnesis, recent estrus, typical clinical findings, ultrasonographic imaging, elevated serum progesterone concentration, and histopathologic evaluation of the punch biopsy specimen. Diseased animals were divided into groups according to treatment protocol: Group A: 25 cats were subjected to medical treatment. Group (B): 15 cats were subjected to OVH based on the owner’s desire, as there was no further interest in breeding or recurrence of affection. Four cats from Group A that showed incomplete response to cabergoline were subsequently shifted to combined therapy (cabergoline + ovariohysterectomy). These cases were analyzed separately in the results. Healthy intact female cats (n=10) were recruited from the same population as a control group. Controls were age- and breed-matched to the FMH cases, clinically healthy on physical examination, had no history of recent progestin administration, and showed no mammary enlargement. Blood samples for hormonal assays were collected, when possible, at comparable stages relative to estrus. Clinical and physical examination resultsBased on the patient’s history data, age, breed, time of previous heat, beginning of mammary gland swelling, appetite, feeding, and vaccination data were recorded. All animals were subjected to clinical and physical examinations to record data regarding body temperature, respiratory rate, and pulse, in addition to all macroscopical features of the affected glands. Biochemical analysisBlood samples were collected from the cephalic vein in plain sterile tubes without anticoagulant, centrifuged, and harvested. Serum estrogen and progesterone levels were measured using standard procedures. Radiographic examinationBefore radiography, the animals were sedated with 2% xylazine hydrochloride. Radiography was performed using Toshiba Rotanode X-ray machine (POX-300BT, Japan). All images were taken during the inspiration–expiration pause. Radiographs of the lateral (LA) and ventrodorsal (VD) projections were taken. The radiographic exposure factors used to obtain X-ray photos were adjusted on kilovoltage: 58.33 ± 4.79 (for LA view) and 56.66 ± 3.79 (for VD view); milliampere second: 5.02 ± 0.55. The X-ray images were evaluated to detect the definite site and volume of the FMH swelling in all body parts. Ultrasonographic examinationAll animals were examined ultrasonographically to exclude pregnancy, and a general examination was performed using an ultrasound machine (Sonoscape A5V, China) connected with a 5-9 MHz transducer for transabdominal examination. Histopathological examinationTissue specimens of the affected mammary gland were dissected and fixed by immersion in 10% buffered formalin. The formalin-fixed tissue was dehydrated in gradual ascending ethanol, cleared in xylene, embedded in paraffin wax, and sectioned using a microtome (Leica RM 2155, England). Sections were stained with hematoxylin and eosin and evaluated under a light microscope. Medical treatmentTwenty-five cats were treated with cabergoline, anti-inflammatory drugs, and antibiotics according to the following protocol (Table 1). Surgical managementFifteen intact female cats were examined ultrasonographically to rule out pregnancy, and ovariohysterectomy was performed under general anesthesia. Every animal was fasted for at least 8–12 hours before surgery, and water was withheld for at least 6 hours before surgery. The cats were subcutaneously premedicated with atropine sulfate (0.04 mg/kg body weight). Anesthesia was induced by a combination of xylazine hydrochloride (1 mg/kg body weight) and ketamine hydrochloride (10–15 mg/kg body weight). Both drugs were loaded into the same syringe and intramuscularly administered. The incision was centered at the midpoint (0.5–1.5 cm long) between the umbilicus and the cranial brim of the pubis. The surgical procedures were performed using a spay hook after midline celiotomy. The surgical wound was closed routinely after the completion of the spaying procedure (Concannon, 2023). Antibiotic (containing 140 mg amoxicillin trihydrate and 35 mg clavulanic acid per ml, GlaxoSmithKline (GSK) was administered for 5 days after the operation. One day after the operation, cabergoline (Dostinex®, Pfizer, Egypt) was administered orally at a dose of 5 µg/kg/day once a day for 5 days. Statistical analysisResults were reported as mean ± SD and analyzed using Welch’s t-test for comparison between affected and control groups because the variances were unequal. Statistical significance was set at p < 0.05. All analyses were performed using SPSS version 24.0 (IBM Corp., Armonk, NY, USA). Ethical approvalThe ethical committee of Zagazig University approved the study in accordance with the guidelines of Zagazig University’s animal ethics committee (ZU-IACUC/2/F/294/2024) (25/12/2024 to 25/12/2026). ResultsForty intact female cats were referred for FMH treatment between 2023 and 2025. All affected cats were intact females aged 6 months to 7 years (mean ± SD=12.4 ± 6.5 months). The most commonly affected breeds were Persian (31/40) (77.5%), Egyptian Mau (5/40) (12.5%), and Siamese (4/40) (10%) with a history of mammary mass that gradually swelled in the last 15 days. Anamnesis taken from the patient’s owner revealed that mammary mass enlargement was observed within 1–3 weeks after estrus in 31 cases, and in the other nine cases, the mass was observed within 2–4 weeks after progesterone hormone administration. Their appetite and general condition were normal. Table 1. Medical treatment protocol in this investigation.
Twenty-eight animals depended on a commercially available dry diet, and the other 12 animals depended on mixed food for feeding. All cats were regularly vaccinated. Physical examination revealed that all parameters, including body temperature ranged from (38.1 to 38.9C), pulse (130–160/minute), and respiration (14–25 /minute), were within normal limits. The clinical findings of cats with FMH are summarized in Table 2. Table 2. Summary of the clinical findings of cats with FMH.
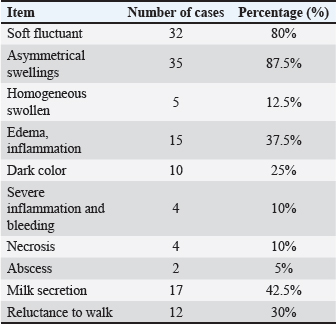
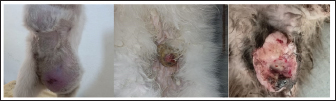
Fig. 1. The mammary lobes were extremely tight with asymmetrical swellings in two cases accompanied with ulcer in the 3rd one.. It was observed that the mammary lobes were extremely tight, and the size of the swollen mammary lobes ranged from 2 to 13 cm. Furthermore, the swellings were freely movable and did not adhere to the abdominal wall. Cats with FMH showed soft fluctuant jelly swelling in 32 cases (80%), asymmetrical swellings in 35 cases (87%) (Fig. 1), homogeneous swelling in 5 cats (12.5%), inflammation and edema in 15 cases (37.5%), dark color in 10 cases (25%), severe inflammation and bleeding in 4 cases (10%), necrosis in 4 cases (10%), abscess in 2 cases (5%), milk secretion in 17 cases (42.5%), and unwillingness to walk in 12 cases (30%) (Fig. 2).
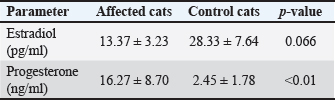
Fig. 2. Showing severe swelling in the mammary lobes with edema in one case and sever ulcer and necrosis in the other case. Biochemical analysisThe biochemical findings are summarized in Table 3. Serum hormonal assays revealed a significant increase in progesterone levels in cats affected with FMH compared with healthy controls, whereas a non-significant decrease in estradiol levels was observed. Table 3. Mean values (± SD) of progesterone and estradiol in control and affected cats.
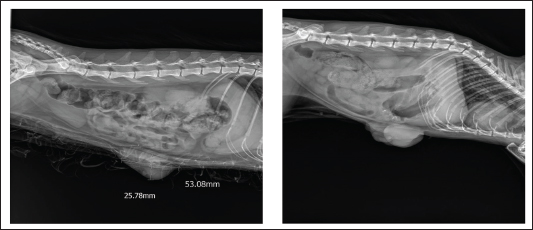
Serum progesterone and estradiol concentrations were compared between the affected and healthy control cats. Progesterone levels were markedly elevated in cats with FMH (16.27 ± 8.70 ng/ml) compared with normal controls (2.45 ± 1.78 ng/ml). Welch’s t-test (two-tailed) indicated that this difference was statistically significant (p=0.009, p < 0.01), demonstrating that the disease was associated with excessive luteal activity and pathological overproduction of progesterone. The mean serum estradiol concentration in affected cats (13.37 ± 3.23 pg/ml) was lower than that of healthy controls (28.33 ± 7.64 pg/ml). However, Welch’s t-test showed that this difference was not statistically significant (p=0.066). Therefore, estradiol cannot be confirmed or excluded as a contributing factor to FMH, and further studies with larger sample sizes are recommended. RadiographyOn radiography images, the mammary gland tissue shows enlargement of the mammary glands with intact body wall (Fig. 3).
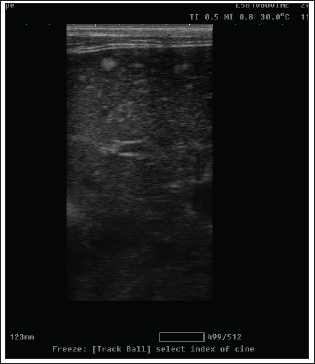
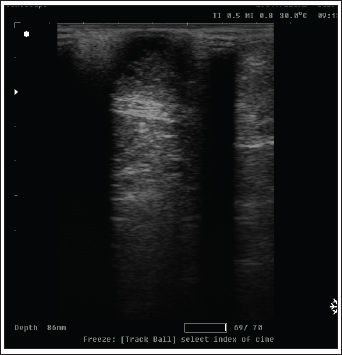
Fig. 3. Radiography images of mammary gland tissue showing enlargement of the mammary glands with intact body wall in different affected female cats. UltrasonographyOn ultrasound images, mammary gland tissue showed higher echogenicity than normal tissue. The FMH lesions were well-defined, slightly hyperechoic, lobulated masses containing multiple small anechoic clefts within the mammary parenchyma. The intraductal pattern is dominant, with anechoic areas corresponding to clefts of different shapes within the mammary gland parenchyma. The presence of clefts in mammary fibroepithelial lesions provided a more heterogeneous appearance to the ultrasound images (Fig. 4). In the solid lesions, the ultrasound pattern is more homogeneous (Fig. 5).
Fig. 4. Ultrasonographic image of lesions presenting cleft-like anechoic areas and characteristics of the intraductal pattern.
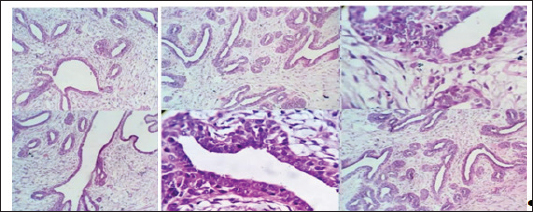
Fig. 5. Ultrasonographic image of a solid pattern lesion. Histopathological examinationThe microscopic appearance of feline mammary hyperplasia is described as fibroadenomatous changes with glandular and fibrous proliferation and dilated ducts. Hyperplasia of both acini (secretory units) and interlobular ducts surrounded by fibrous stroma. The mammary ducts are dilated and often filled with proteinaceous fluid or secretions (Fig. 6).
Fig. 6. Microscopic appearance of feline mammary hyperplasia, acinar and interlobular ductular fibroadenoma with ductectasia.. Medical treatment outcome using cabergolineAll affected cats (n=25) were medically treated with cabergoline (Dostinex®) at a dose of 5 µg/kg once daily per os (PO). A marked regression of the mammary glands was observed in 18 cats (72%) after 3 weeks. Complete regression occurred between weeks 6 and 7 in all cases. Four cats (16%) showed signs of recurrent estrus at the end of the third week and required an additional 3-week treatment course combined with ovariohysterectomy. No systemic adverse effects were detected during the treatment period, and all cats recovered completely (Table 4). Table 4. Clinical response of cats with FMH treated with cabergoline.
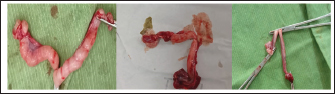
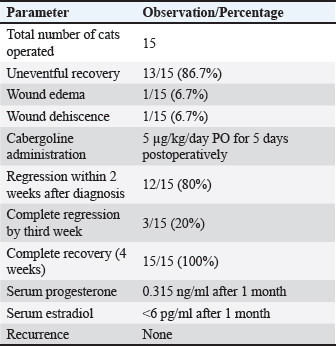
Surgical approachOvariohysterectomy was performed in 15 (37.5%) cats through a median line incision (Fig. 7). The postoperative recovery of cats was smooth and uneventful in 13 of 15 cases (86.7%). One cat (6.7%) developed mild wound edema, whereas another (6.7%) experienced wound dehiscence. Cabergoline was orally administered at a dose of 5 µg/kg/day for 5 consecutive days starting 1 day after surgery. During follow-up examinations conducted 2 weeks after surgery, a marked regression of the mammary gland size and cessation of milk secretion were observed in 12 cats, while the remaining 3 cats showed subsequent evaluations performed 3–4 weeks later revealed total resolution of mammary enlargement without recurrence.
Fig. 7. Showing ovariohysterectomy operation in different female cats with different ages. At the 1-month follow-up, the mammary glands had recovered completely, with serum progesterone levels reduced to 0.315 ng/ml and estradiol levels below 6 pg/ml. No postoperative recurrences or complications were reported during or after treatment (Table 5). Table 5. Postoperative outcomes in surgically treated cats (n=15)
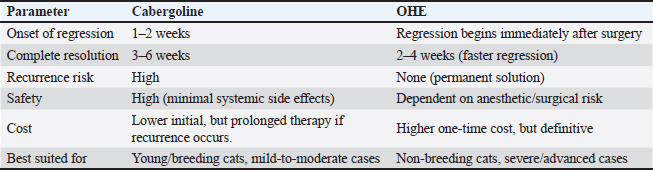
In our investigation, both medical and surgical management are effective, but OHE offers a permanent treatment, whereas cabergoline provides a safe, noninvasive alternative suitable for breeding females. Combined therapy may accelerate recovery and prevent relapse (Table 6). Table 6. Comparative outcome between medical and surgical treatment.
DiscussionFeline mammary fibroepithelial hyperplasia is a hormonally driven benign enlargement of the mammary glands that occurs due to an exaggerated response of mammary tissue to progesterone during the estrous phase (Loretti et al., 2004). The disorder primarily affects intact young female cats and is increasingly recognized worldwide. These results are similar to those reported by Johnston (1994) and Johnston et al. (2001). Feline mammary fibroepithelial hyperplasia is a worldwide disorder that affects cats and is extensively reported in female cats. In the present investigation, most of the affected animals were young, with an average age of approximately 1 year, although the range extended from early juvenile to adult stages (Allen, 1973; Hayden et al., 1989; Wehrend et al., 2001; Enginler and Şenünver, 2011; Uçmak et al., 2011). The disease is most frequently observed in Persian cats, followed by Egyptian Mau and Siamese breeds. This pattern likely reflects breed distribution among the examined population rather than a specific genetic predisposition (Enginler and Şenünver, 2011). In most cases, mammary enlargement developed within 3 weeks after estrus, while in a smaller proportion, it followed the administration of exogenous progesterone. These findings support the concept that FMH is a progesterone-dependent disorder that can occur spontaneously or iatrogenically. This result was previously confirmed (Jurka and Max, 2009; Enginler and Şenünver, 2011). The mammary lobes were extremely tight, soft, fluctuant, jelly, asymmetrical, homogeneous, swollen, inflammatory, edema, dark color, severe inflammation, ulcerations with bleeding, necrosis, abscess, milk secretion, and unwilling to walk. Previous studies reported similar findings (Moulton, 1990; Rutteman and Withrow, 2001; Ginn et al., 2007; Payan, 2013). Most of these signs were attributed to the enlargement of the mammary lobe and exposure to trauma, causing ulceration and necrosis (Loretti et al., 2004; Voorwald et al., 2021). Fibroepithelial mammary hyperplasia was attributed to increased tissue volume, which led to perfusion problems, erythema, pain, ulceration, and dark and necrotic areas on the mammary gland skin (Bonatto et al., 2021; Voorwald et al., 2021). Serum hormone analysis revealed a marked elevation in progesterone concentrations among affected cats compared with healthy controls, while estradiol levels were moderately reduced. This hormonal pattern indicates overstimulation of the mammary epithelium by progesterone and supports its central role in the pathogenesis of the disease. These findings were consistent with those of previous studies (Görlinger et al., 2002; Akkuş and Yaprakcı,2024). This marked elevation in progesterone level indicates an abnormal luteal activity or pathological condition associated with progesterone overproduction. In contrast, progesterone concentrations in normal cats were within the expected preovulatory/early luteal baseline range. These findings suggest that progesterone may play an important role as a biomarker for the reproductive disorder under investigation. Radiology was of little interest in cases of FMH, as lateral abdominal surveys only reveal the enlargement of the mammary glands with an intact body wall (Burstyn, 2010). In comparison, ultrasonography provides more valuable information about the assessment of lesion pattern and echogenicity. Similar findings have been reported (Payan, 2013). In the present study, medical treatment was administered to 25 cats using cabergoline drug administration (5 µg/kg once daily PO), which proved effective in the majority of treated cats, leading to substantial regression of mammary size within three to 6 weeks, as previously reported (Giménez et al., 2010; Akkuş and Yaprakcı, 2024). Eighteen cats revealed a marked regression of the mammary glands parallel to the disappearance of the erythema, tightness of the mammary skin, and cessation of milk secretion. After 6 weeks, the mammary glands were completely regressed, which was consistent with other study data (Keskin et al., 2008; Uçmak et al., 2011; Akkuş and Yaprakcı, 2024). Ovariohysterectomy was performed on 15 (37.5%) cats. Significant regression and decrease in the volume of the mammary glands and end of milk production were observed in 12 cases and in the third week in the other 3 cats. A follow-up examination performed 1 month later revealed that the mammary glands had completely recovered. This finding was similar to that reported in previous studies (Keskin et al., 2008; Uçmak et al., 2011; Akkuş and Yaprakcı, 2024). Ovariohysterectomy is one of the most effective treatment methods (Kutzler and Wood, 2006). When making the decision to undergo an ovariohysterectomy, it should be considered that this operation will prevent the patient from having offspring in the future (De Melo et al., 2021). In our study, the combination of OHE and cabergoline was administered to the mammary glands in two different ways, as previously reported (Nak et al., 2004; Uçmak et al., 2011; Küçükbekir et al., 2020). A significant regression was noted within a week, and no complications related to both the operation and regression process in the mammary gland were observed. The dose of cabergoline administered in our study was 5 µg/kg once daily PO, especially in cases of milk secretion, which was consistent with other study data (Keskin et al., 2008; Uçmak et al., 2011), and cabergoline administration at the specified dose did not cause any adverse effects. Contrary to some studies (Keskin et al., 2008; Uçmak et al., 2011). In these cases, the mammary lobes become ulcerated, and relapses occur shortly after the application (Keskin et al., 2008; Uçmak et al., 2011). Therefore, combined treatment, including surgery, is an effective treatment method in MFH cases that do not respond to treatment. Comparative outcomes between medical and surgical treatment indicate that cabergoline is a safe, non-invasive, and effective treatment option for FMH, particularly in young or breeding queens. However, treatment requires several weeks, and recurrence is possible upon future progesterone exposure. Ovariohysterectomy offers a definitive and permanent cure, prevents recurrence, and should be considered in severe cases or when breeding is not intended. These results were consistent with those of other studies (De Melo et al., 2021). Therefore, combined treatment, including surgery, is an effective treatment method in MFH cases. ConclusionCabergoline is a safe, non-invasive, and effective treatment option for FMH, particularly in young or breeding queens. However, treatment requires several weeks, and recurrence is possible upon future progesterone exposure. Ovariohysterectomy, while invasive, offers a definitive and permanent cure, prevents recurrence, and should be considered in severe cases or when breeding is not intended. Clinical decision-making should be individualized, considering the reproductive value, clinical severity, owner preference, and surgical risk of the cat. AcknowledgmentsNone. Conflict of interestThe authors have no conflicts of interest to declare. FundingNot applicable. Authors’ contributionsAll authors contributed equally to this study. Data availabilityAll data were included in this manuscript. ReferencesAkkuş, T. and Yaprakcı, O. 2024. Cabergoline administration after ovariohysterectomy in a queen with fibroepithelial hyperplasia. Vet. J. Kastamonu Univ. 3(1), 17–21. Allen, H.L. 1973. Feline mammary hypertrophy. Vet. Pathol. 410, 501–508. Bonatto, G.L., Gariba, V., Favero, L.J., Kano, N.N., De Sousa, R.S. and Albernaz, V.G.P. 2021. Mammary fibroepithelial hyperplasia in a male cat model. Acta Scientiae Veterinariae, 49, 1–5. Burstyn, U. 2010. Management of mastitis and abscessation of mammary glands secondary to fibroadenomatous hyperplasia in a primiparous cat. J. Am. Vet. Med. Assoc. 236(3), 326–329. Concannon, P. 2023. Reproductive sterilization of cats and dogs: techniques and long-term effects. J. Feline. Med. Surg. 25(4), 287–300. De Melo, E.H., Câmara, D.R., Notomi, M.K., Jabour, F.F., Garrido, R.A., Nogueira, A.C., Júnior, J.C. and De Souza, F.W. 2021. Effectiveness of ovariohysterectomy on feline mammary fibroepithelial hyperplasia treatment. J. Feline Med. Surg. 23(4), 351–356. Enginler, S. and Şenünver, A. 2011. The effects of progesterone hormone applications used for suppression of estrus on mammary glands in queens. Kafkas Üniversitesi. Veteriner. Fakültesi. Dergisi. 17(2), 277–284. Giménez, F., Hecht, S., Craig, L.E. and Legendre, A.M. 2010. Early detection, aggressive therapy: optimizing the management of feline mammary masses. J. Feline Med. Surg. 12(3), 214–224. Ginn, P., Mansell, J. and Rakich, P. 2007. Tumors of the mammary gland.In Jubb, Kennedy, and Palmer’s Pathology of Domestic Animals. Maxie, M New York, USA: Saunders, Elsevier, pp: 777–80. Görlinger, S., Kooistra, H.S., Van Den Broek, A. and Okkens, A.C. 2002. Treatment of fibroadenomatous hyperplasia with aglepristone in cats. J. Vet. Internal Med. 16(6), 710–713. Hayden, D.W., Barnes, D.M. and Johnson, K.H. 1989. Morphologic changes in the mammary gland of cats treated and untreated with megestrol acetate: a retrospective study. Vet. Pathol. 26(2), 104–113. Hayden, D.W., Johnston, S.D., Kiang, D.T., Johnson, K.H. and Barnes, D.M. 1981. Feline mammary hypertrophy/fibroadenoma complex: clinical and hormonal aspects. Am. J. Vet. Res. 42(10), 1699–1703. Jelinek, F., Barton, R., Posekana, J. and Hasonova, L. 2007. Gynecomastia in a tom-cat caused by cyproterone acetate: a case report. Veterinarni Medicina 52, 521–525. Johnson, C. 1994. Diseases of the mammary glands. In The Cat: diseases and Clinical Management. Sherding, R 2nd, New York, NY: Churchill Livingstone, pp: 1874–1875. Johnston , S., Root Kustritz, M. and Olson, P. 2001. Disorders of the mammary gland of the queen. In Canine and Feline Theriogenology. Philadelphia, USA: W.B. Saunders Co., pp: 474–485. Jurka, P. and Max, A. 2009. Treatment of fibroadenomatosis within 14 cats with aglepristone: changes in blood parameters and follow-up. Vet. Rec. 165, 657–660. Keskin, A., Yilmazbas, G., Simsek, G. and Seyrek-Intas, K. 2008. Mammary hyperplasia in a pregnant queen. Austral. Vet. Practitioner 38(2), 75–77. Küçükbekir, C., Günay Uçmak, Z., Kirşan, I. and Tek, C. 2020. A case of feline fibroepithelial hyperplasia in a male cat. J. Istanbul. Vet. Sci. 4(1), 8–12. Kutzler, M. and Wood, A. 2006. Non-surgical methods of contraception and sterilization. Theriogenology 66(3), 514–525. Leidinger, E., Sick, K., Reinelt, B., Kirtz, G. and Hooijberg, E. 2011. Fibroepithelial hyperplasia in an entire male cat: cytologic and histopathological features. Tierarztliche Praxis Ausgabe Kleintiere Heimtiere 39(3), 198–202. Loretti, A., Ilha, M., Breitsameter, I. and Faraco, C. 2004. Clinical and pathological study of feline mammary fibroadenomatous changes associated with depot medroxyprogesterone acetate therapy. Arq. Bras. Med. Vet. Zootec. 56(2), 270–274. Loretti, A., Ilha, M., Ordás, J. and De Las Mulas, J.M. 2005. Clinical, pathological and immunohistochemical study of feline mammary fibroepithelial hyperplasia following a single injection of depot medroxyprogesterone acetate. J. Feline Med. Surg. 7(1), 43–52. Mayayo, S.L., Bo, S. and Pisu, M.C. 2018. Mammary fibroadenomatous hyperplasia in a male cat. J. Feline. Med. Surg. Open. Rep. 4(1), 2055116918760155. Moulton, J. 1990. Mammary tumors of the cat.In Tumours in Domestic Animals. Moulton 3rd, Berkeley, CA, USA: University of California Press, pp: 547–8. Murphy , S. Mammary tumors in cats—causes and prac-tical management. In: Proceedings of the ESFM Feline Symposium; 2009; Birmingham, UK. European Society of Feline Medicine. Harvard Nak, D., Nak, Y., Seyrek-Intas, K. and Kumru, I.H. 2004. Treatment of feline mammary fibroadenomatous hyperplasia with aglepristone. Austral. Vet. Practitioner 34(4), 161–162. Payan, R. 2013. Feline mammary fibroepithelial hyperplasia: a clinical approach. In Insights from veterinary medicine. Ed., Payan-Carreira, R. Rijeka, Croatia: InTechOpen. Rutteman , G. and Withrow, S. 2001. Tumours of the mammary gland.In Small Animal Clinical Oncology. Withrow, S. and MacEwen, E. Philadelphia, USA: W.B. Saunders Co., pp: 455–477. Sontas, B., Turna, O., Ucmak, M. and Ekici, H. 2008. What is your diagnosis? Feline mammary fibroepithelial hyperplasia. J. Small Anim. Pract. 49(10), 545–547. Uçmak, M., Enginler, S., Gündüz, M., Kirşan, I. and Sönmez, K. 2011. Treatment of feline mammary fibroepithelial hyperplasia with the combination of aglepristone and cabergoline. J. Fac. Vet. Med. 37(1), 69–73. Vitasek , R. and Dendisova, H. 2006. Treatment of feline mammary fibroepithelial hyperplasia following a single injection of proligestone. Surg 135(11), 135. Voorwald , F.A., Lopes, C., Silveira, G.C., Lima, D.T., Silva, M.D.F.C.D., Andreão, N.B. and Toniollo, G.H. 2021. Severe mammary fibroepithelial hyperplasia due to single injection of medroxyprogesterone acetate in two male cats. Ciência Rural, 51(4), e20200347 Wehrend, A., Hospes, R. and Gruber, A.D. 2001. Treatment of feline mammary fibroadenomatous hyperplasia with a progesterone antagonist. Vet. Rec. 148(11), 46–47. | ||
| How to Cite this Article |
| Pubmed Style Ibrahim N, Ibrahim FM, Elgebaly MM, Ezzeldein SA, Emam MM, Youssef WOM. Feline mammary hyperplasia: A comparative study of medical versus surgical treatment approaches. Open Vet. J.. 2026; 16(2): 1329-1337. doi:10.5455/OVJ.2026.v16.i2.49 Web Style Ibrahim N, Ibrahim FM, Elgebaly MM, Ezzeldein SA, Emam MM, Youssef WOM. Feline mammary hyperplasia: A comparative study of medical versus surgical treatment approaches. https://www.openveterinaryjournal.com/?mno=297732 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.49 AMA (American Medical Association) Style Ibrahim N, Ibrahim FM, Elgebaly MM, Ezzeldein SA, Emam MM, Youssef WOM. Feline mammary hyperplasia: A comparative study of medical versus surgical treatment approaches. Open Vet. J.. 2026; 16(2): 1329-1337. doi:10.5455/OVJ.2026.v16.i2.49 Vancouver/ICMJE Style Ibrahim N, Ibrahim FM, Elgebaly MM, Ezzeldein SA, Emam MM, Youssef WOM. Feline mammary hyperplasia: A comparative study of medical versus surgical treatment approaches. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 1329-1337. doi:10.5455/OVJ.2026.v16.i2.49 Harvard Style Ibrahim, N., Ibrahim, . F. M., Elgebaly, . M. M., Ezzeldein, . S. A., Emam, . M. M. & Youssef, . W. O. M. (2026) Feline mammary hyperplasia: A comparative study of medical versus surgical treatment approaches. Open Vet. J., 16 (2), 1329-1337. doi:10.5455/OVJ.2026.v16.i2.49 Turabian Style Ibrahim, Nehal, Fatma M. Ibrahim, Maha M. Elgebaly, Shimaa A. Ezzeldein, Mahmoud M. Emam, and Walaa O. M. Youssef. 2026. Feline mammary hyperplasia: A comparative study of medical versus surgical treatment approaches. Open Veterinary Journal, 16 (2), 1329-1337. doi:10.5455/OVJ.2026.v16.i2.49 Chicago Style Ibrahim, Nehal, Fatma M. Ibrahim, Maha M. Elgebaly, Shimaa A. Ezzeldein, Mahmoud M. Emam, and Walaa O. M. Youssef. "Feline mammary hyperplasia: A comparative study of medical versus surgical treatment approaches." Open Veterinary Journal 16 (2026), 1329-1337. doi:10.5455/OVJ.2026.v16.i2.49 MLA (The Modern Language Association) Style Ibrahim, Nehal, Fatma M. Ibrahim, Maha M. Elgebaly, Shimaa A. Ezzeldein, Mahmoud M. Emam, and Walaa O. M. Youssef. "Feline mammary hyperplasia: A comparative study of medical versus surgical treatment approaches." Open Veterinary Journal 16.2 (2026), 1329-1337. Print. doi:10.5455/OVJ.2026.v16.i2.49 APA (American Psychological Association) Style Ibrahim, N., Ibrahim, . F. M., Elgebaly, . M. M., Ezzeldein, . S. A., Emam, . M. M. & Youssef, . W. O. M. (2026) Feline mammary hyperplasia: A comparative study of medical versus surgical treatment approaches. Open Veterinary Journal, 16 (2), 1329-1337. doi:10.5455/OVJ.2026.v16.i2.49 |