| Case Report | ||
Open Vet. J.. 2026; 16(2): 1394-1401
Open Veterinary Journal, (2026), Vol. 16(2): 1394-1401 Case Report Suspected secondary inflammatory mammary carcinoma developed from pre-existing tumors in a cat: Clinical course from diagnosis to deathMakoto Akiyoshi1,2*, Midori Goto Asakawa3 and Masaharu Hisasue21Akiyoshi Animal Clinic, Yamato City, Japan 2Laboratory of Small Animal Internal Medicine, Azabu University, Sagamihara City, Japan 3Clinical and Anatomic Pathology Unit, Veterinary Specialists Emergency Center, Kawaguchi City, Japan *Corresponding Author: Makoto Akiyoshi. Akiyoshi Animal Clinic, Yamato City, Japan.Email: makotoaoi1109 [at] yahoo.co.jp Submitted: 18/11/2025 Revised: 11/01/2026 Accepted: 18/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
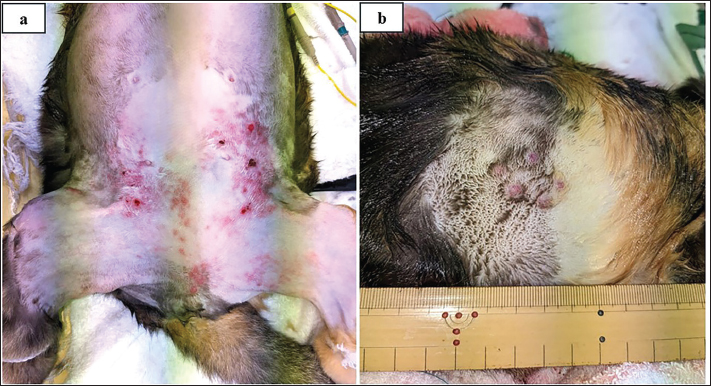
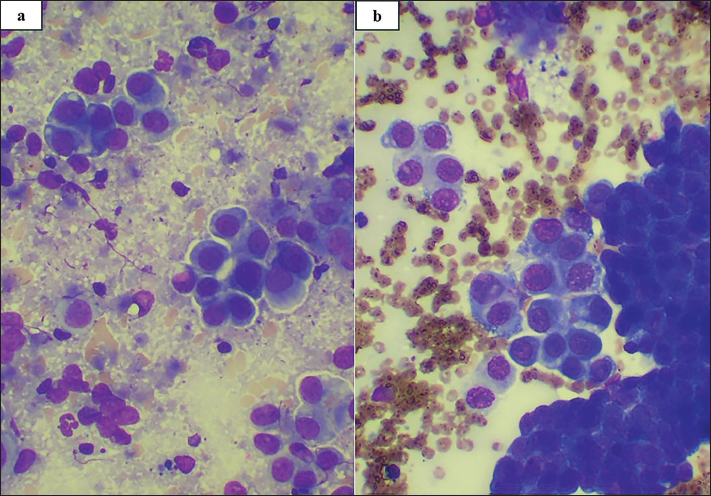
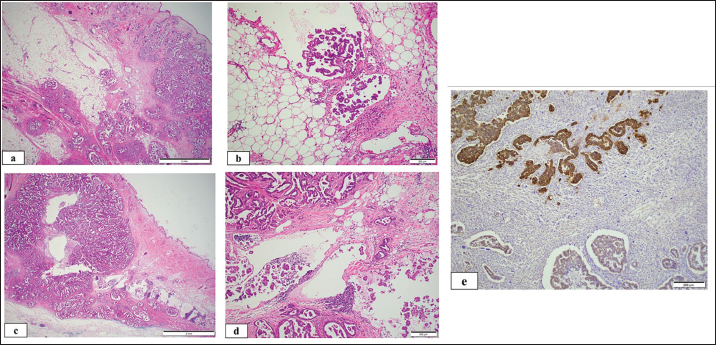
AbstractBackground: Feline inflammatory mammary carcinoma (FIMC) is a rare, aggressive malignancy characterized by rapid progression and a poor prognosis. FIMC is often not clinically suspected before histopathological confirmation because of its low incidence. With its recent inclusion in the World Health Organization classification, awareness of secondary forms of FIMC is becoming increasingly important. Case Description: A 13-year-old spayed female mixed-breed cat with a 1-cm mammary tumor between the right third and fourth glands, previously diagnosed as common mammary carcinoma by cytology 3 months earlier, was referred for severe pain associated with rapid tumor progression and skin erythema. The lesion had enlarged to 1.5 cm with multiple 1–2-mm nodules and diffuse erythema extending from the lower abdomen to the labia. Several distinct 5-mm nodules, clearly separated from the mammary chain, were also identified in the right dorsal lumbar skin. All accessible tumors (bilateral third and fourth mammary glands and right lumbar lesions, each with 2-cm margins) were palliatively resected to alleviate pain and obtain a definitive diagnosis. Histopathology revealed mammary carcinoma with neoplastic emboli in dermal lymphatics; when interpreted in conjunction with the clinical history, this finding was consistent with secondary FIMC. Postoperatively, the cat’s pain score decreased from 18 to 3 within 24 hours, and appetite improved. Sequential treatment with lapatinib, toceranib, and firocoxib, followed by metronomic chlorambucil for pulmonary metastases, achieved local disease stabilization. The cat remained pain-free for 185 days before death from uremia. Conclusion: This case represents one of the first reports of secondary FIMC developing from a pre-existing mammary tumor without prior surgery. Because FIMC has only recently been included in the World Health Organization classification, it is frequently unrecognized before histopathological examination. This case highlights the diagnostic challenges associated with secondary FIMC and demonstrates that multimodal palliative therapy can help cats with advanced FIMC maintain their quality of life. Keywords: Cat, Chlorambucil, Feline inflammatory mammary carcinoma;, Lapatinib, Toceranib. IntroductionFeline inflammatory mammary carcinoma (FIMC) is a rare and highly aggressive malignancy associated with a poor prognosis (Pérez-Alenza et al., 2004; Millanta et al., 2012). It is classified into two forms: primary FIMC, which develops de novo, and secondary FIMC, which typically arises suddenly after mastectomy for mammary tumors (Pérez-Alenza et al., 2004; Millanta et al., 2012). We encountered a suspected case of secondary FIMC that progressed from a pre-existing mammary tumor without any surgical intervention. With the recent inclusion of FIMC in the World Health Organization classification of mammary tumors (Zappulli et al., 2008), an increasing number of cats are expected to be definitively diagnosed with secondary FIMC only after surgery. The present report describes the clinical course of a case in which FIMC was not suspected preoperatively, and a definitive diagnosis was established histopathologically after palliative surgery and subsequent multimodal postoperative management, including toceranib. Case DetailsA 13-year-old spayed female mixed-breed cat was referred to the author’s private veterinary hospital, Akiyoshi Animal Clinic (Kanagawa, Japan), with progressive enlargement of a mammary tumor causing extreme pain. 3 months earlier, a 1-cm mass was detected between the right third and fourth mammary glands and was cytologically diagnosed as common mammary carcinoma. The patient declined surgery at that time, and the lesion was monitored. The cat was in extreme pain; its appetite had decreased, and it had lost 1 kg of body weight within a week. At presentation, the original tumor had enlarged to approximately 1.5 cm, with newly developed firm, erythematous thickening of the overlying skin. Multiple small nodules, 1–2 mm in diameter, were palpated within the surrounding subcutaneous tissue, and diffuse erythema and induration extended from the caudal abdomen to the inguinal region and labia (Fig. 1a). Several distinct nodules measuring approximately 5 mm were identified in the dorsal skin of the right lumbar region (Fig. 1b). These lesions were clearly isolated from the mammary chain and showed no anatomical continuity with the primary tumor. Their presence suggested multifocal cutaneous involvement rather than local mammary lesion extension. The affected skin was warm, edematous, and painful on palpation, and handling was resisted. No other systemic abnormalities were noted on physical examination. Cytological examination of the mammary and right lumbar dorsal cutaneous masses was performed. The cytological findings were suggestive of a highly malignant mammary carcinoma arising from the ventral abdomen, with metastasis to the right dorsal lumbar dermis (Fig. 2). The cytological features were more pleomorphic and aggressive than those observed in the previous cytological diagnosis of common mammary carcinoma performed 3 months earlier, suggesting tumor progression and increased malignancy. Thoracic and abdominal radiography and ultrasonography revealed no other metastases. A routine plasma biochemistry panel revealed moderately elevated blood urea nitrogen and creatinine concentrations and severely elevated serum amyloid A concentrations (Table 1). The patient requested rapid pain control and histopathological examination for a definitive diagnosis. As the mammary tumor had spread over a wide area and the cat was also in stage II of the international renal interest society (IRIS) staging staging classification for kidney disease (Relford et al., 2016), palliative resection was performed on day 3 instead of curative surgery to remove all mammary glands, including both third and fourth mammary glands bilaterally with 2 cm lateral margins and deep margins including the fascia. A tumor in the right lumbar region was also resected with the same margin. All visibly affected tissues, including ulcerated and highly inflamed lesions, were removed to the extent feasible. The aim was to reduce tension, edema, and inflammatory cytokine release in the skin. No analgesic drugs were administered postoperatively because of the cat’s chronic kidney disease and the unavailability of fentanyl patches at the clinic at that time. Pain relief was achieved immediately after surgery; the cat no longer resisted handling, and its appetite and activity returned to normal within 24 hours. Histopathological examination (Fig. 3a–d) revealed an infiltrative, poorly demarcated, and unencapsulated neoplasm extending from the dermis to the subcutis, with prominent dermal lymphatic invasion and dilatation. The neoplasm comprised polygonal epithelial cells arranged in tubules, papillae, and islands, with a moderate amount of stroma and normal tissue architecture effacement. The neoplastic cells had moderate to large oval nuclei with coarse chromatin and a prominent nucleolus with moderate amounts of eosinophilic to amphophilic cytoplasm. Mitotic figures were observed in 13 of the 10 high-power fields (2.37 mm2). Multifocal necrosis and hemorrhage were also observed. Neoplastic cells were identified at multiple lateral surgical margins. Metastatic foci were observed in the adjacent inguinal lymph nodes. Histological features within the right lumbar dorsal dermis and subcutis were similar to those in the ventral abdomen. The cat was histologically diagnosed with IMC with regional (inguinal) lymph node and distant metastasis (DM). Suspected secondary FIMC that had developed from preexisting mammary tumors was considered based on the clinical history. Immunohistochemical staining showed that 20% of the neoplastic cells showed strong membranous and cytoplasmic positivity for the anti-HER2 antibody, whereas the remaining neoplastic cells were weakly to moderately positive (Fig. 3e). The anti-HER2 antibody used was a commercially available rabbit monoclonal antibody originally developed for human tissue and has been previously applied in studies of feline mammary carcinoma, in which tissue HER2 status has been shown to be clinically relevant (Soares et al., 2016).
Fig. 1. Gross findings of the (a) lower abdominal mammary tumor and (b) right lumbar dorsal skin tumor on day 1.
Fig. 2. Cytological findings of the lower abdominal mammary tumor (a) and the right lumbar dorsal tumor (b) on day 1 after surgery. Several cohesive clusters of epithelial cells with multiple cytological features of malignancy were observed. The epithelial cells had large oval nuclei with a single prominent nucleolus, clumped chromatin, and small amounts of light-to-mid-blue cytoplasm with a high Nuclear-to-cytoplasmic (N/C) ratio. Moderate to marked anisocytosis and anisokaryosis were observed. Some individualized cells with cell adhesion loss were also noted. (Wright Giemsa stain, ×400). The extreme pain disappeared on day 4 without analgesic drugs, indicating that the UNESP-Botucatu Feline Pain Scale (UFEPS) score decreased from 18 preoperatively to 3 postoperatively; the appetite and vigor also improved (Belli et al., 2021). However, on day 25, new papular nodules appeared on the inner thigh and perineal region, representing local progression rather than true recurrence, as residual tumor tissue likely remained postoperatively. On day 25, lapatinib (22 mg/kg, PO, q24h) was administered to prevent further enlargement. However, the cat showed a loss of appetite and vomiting, making it difficult to continue lapatinib administration, and the chemotherapy was changed to firocoxib (1 mg/kg, PO, q48h) and toceranib (2.6 mg/kg, PO, q48h) on day 35. No side effects were observed, and recurrent lesions from the inner thigh to the labia remained stable. However, thoracic radiography was performed on day 125 because the cat developed mild dyspnea and decreased activity. This revealed pulmonary metastasis and carcinomatous pleuritis, and renal failure worsened. Toceranib was changed to chlorambucil (0.24 mg/kg, PO, q48h) as a metronomic chemotherapeutic agent, and a subcutaneous infusion of 150 ml/day was administered daily. The pulmonary metastatic lesions remained unchanged in size; however, on day 185, the cat died of uremia. Extreme pain was consistently absent, indicating that the UFEPS score remained consistent at 3 from days 4 to 185. Therefore, the owner’s satisfaction levels were very high. No autopsy was performed.
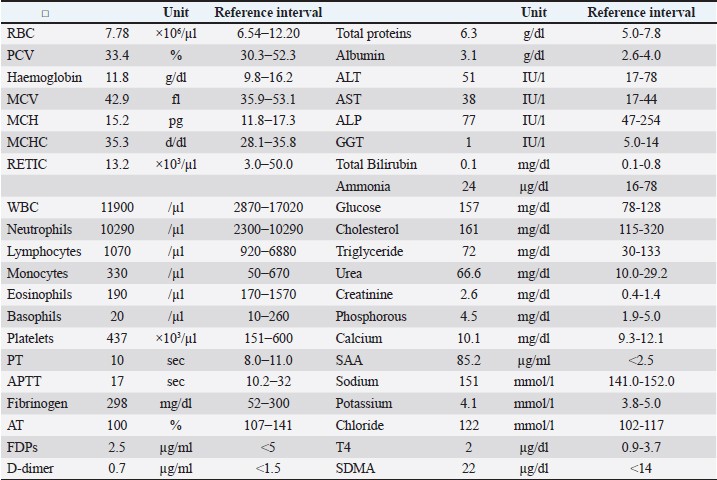
Fig. 3. Photomicrographs of the mammary carcinoma of the ventral abdomen (a and b, bar=2 and 200 µm, respectively, H&E stain) and dorsal lumbar region (c and d, bar=2 and 200µm, respectively, H&E stain). An infiltrative, poorly-demarcated, unencapsulated neoplasm with prominent dermal lymphatic invasion and dilatation was noted within the dermis and subcutis. The neoplasm comprised polygonal epithelial cells arranged in tubules, papillae, and islands. (e) Immunohistochemical staining demonstrated that the neoplastic cells were membranous and cytoplasmic positive for the anti-HER2 antibody. (Bar=200 µm). Ethical approvalNo ethical approval was required for this study. Informed consent was obtained from the patient for all procedures described. DiscussionTo the best of our knowledge, this case represents one of the first suspected cases of secondary FIMC arising from a pre-existing mammary tumor without prior surgery. In this case, FIMC was diagnosed based on histopathological findings showing neoplastic emboli within dermal lymphatic vessels. Clinically, the disease showed several atypical features, including the absence of prior surgical manipulation and the presence of discrete, non-contiguous cutaneous nodules distant from the primary lesion. Although a histological examination was not performed at the time of first detection, 3 months earlier, cytological findings supported the diagnosis of malignancy, suggesting the presence of a pre-existing mammary carcinoma. In addition, the cytological features on day 1 were more pleomorphic and aggressive than those observed in the previous cytological diagnosis of common mammary carcinoma performed 3 months earlier, suggesting tumor progression and increased malignancy. Therefore, we considered this case to represent a secondary form of FIMC that developed through the progression of a pre-existing mammary tumor, rather than a classical primary or post-surgical secondary form. Due to the limited number of reports on FIMC, we were unable to suspect inflammatory mammary carcinoma based solely on the clinical history and cytological findings, but the histopathological evidence supported its inclusion within the FIMC spectrum. FIMC is a rare disease that has been described in only two previous reports (Pérez-Alenza et al., 2004; Millanta et al., 2012). Secondary canine IMC (CIMC) appears to have two patterns: the first is a postoperative IMC that develops at the surgical site after non-inflammatory mammary carcinoma is removed, and the second is atypical, indicating a preexisting mammary tumor that changes and progresses to IMC (Pérez-Alenza et al., 2001; Queiroga et al., 2005; Marconato et al., 2009; Dawood et al., 2011). However, there have been no case reports of atypical patterns in dogs. Two reports on FIMC have described postoperative FIMC and primary FIMC (Pérez-Alenza et al., 2004; Millanta et al., 2012). However, no reports have reported the development of secondary FIMC from pre-existing mammary tumors without surgery (Pérez-Alenza et al., 2001). In our case, the mammary tumor had been identified 3 months earlier, and no surgery had been performed. Although distinguishing primary from secondary FIMC is sometimes difficult, the presence of a cytologically confirmed mammary tumor 3 months before the onset of visible inflammation suggested a diagnosis of secondary FIMC. Primary FIMC typically arises de novo without pre-existing nodules, whereas this case showed gradual evolution from a previously identified 1 cm-sized mammary mass without visible inflammation. Therefore, our case was suspected to be a secondary FIMC that had developed from a preexisting condition. Inflammatory mammary carcinoma has been reported more frequently in dogs and humans than in cats. Across species, this disease is histopathologically characterized by neoplastic emboli within dermal lymphatic vessels and clinically by rapid progression and severe inflammatory changes. However, FIMC remains poorly characterized due to the limited number of reported cases, highlighting the importance of detailed clinical case reports. The key clinical implications of this case include the possibility of secondary FIMC arising from a pre-existing mammary tumor without surgical intervention, the critical role of histopathology in establishing a definitive diagnosis, and the benefit of palliative surgery combined with targeted therapy for effective pain control and temporary disease stabilization. Table 1. Day 1 results of complete blood cell count, coagulation test and blood chemistry.
ALP, alkaline phosphatase; ALT, alanine aminotransferase; APTT, activated partial thromboplastin time; AST, asparate aminotransferase; AT, anti-thrombin; FDPs, fibrin degradation products; GGT, gammma ghutamyl transferase; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; MCV, mean cell volume; PCV, packed cell volume; PT, prothrombin time; RBC, red blood cell; SAA, serum amyloid A; SDMA, symmetric dimethylarginine; WBC, white blood cell. FIMC has recently been incorporated into the World Health Organization classification (Zappulli et al., 2008). The diagnostic criterion is marked invasion of neoplastic cells into the dermal lymphatics, which is confirmed via histopathology. Therefore, a definitive diagnosis of FIMC can only be made histopathologically (Zappulli et al., 2008). In this case, we were able to infer that the cat had an aggressive mammary carcinoma based on the rapid progression, extent of the tumor, and cytology at the preoperative stage, but we could not confirm secondary FIMC. Therefore, both the lower abdominal and right lumbar dorsal skin tumors could only be histopathologically diagnosed as FIMC after palliative surgical excision. As FIMC has now been incorporated into the WHO classification, more cases will likely be definitively diagnosed using postoperative histopathology. Secondary FIMC occurrence should be a defining factor. Treatment options for FIMC have not yet been published because of the scarcity of data and experience with IMC, even in dogs. Surgery is generally not recommended for CIMC and is considered contraindicated (Pérez-Alenza et al., 2001; Queiroga et al., 2005; Marconato et al., 2009; Dawood et al., 2011). Although it has not been determined whether surgery is contraindicated or indicated for FIMC, it may be a treatment option for pain relief, although the current case is merely one example. Further accumulation of secondary FIMC cases is warranted. Tyrosine kinase inhibitors, including lapatinib and toceranib, firocoxib, and chlorambucil, were used for metronomic chemotherapy instead of maximum tolerated dose (MTD) chemotherapy in the hope that it would be an effective anti-angiogenic and anti-inflammatory strategy (Petrucci et al., 2024). An association between HER2 expression and malignancy has been suggested in feline mammary carcinomas (Millanta et al., 2005; Soares et al., 2021). In humans, lapatinib has some efficacy against HER2-positive breast cancer (Collins et al., 2021; Abdelgalil and Alkahtani, 2023). In vitro studies have suggested that lapatinib-containing anti-HER2-targeted drugs may also be effective in treating feline mammary tumors (Gameiro et al., 2021). The in vivo efficacy of lapatinib in feline mammary carcinoma is unclear, but the owner requested its use. However, we could not continue using lapatinib due to its side effects (reduced appetite and vomiting); therefore, toceranib was mainly used as a tyrosine kinase inhibitor. In CIMC, MTD chemotherapy with single agents, such as doxorubicin and carboplatin (Marconato et al., 2009), and MTD chemotherapy with multiple agents, such as mitoxantrone, vincristine, and cyclophosphamide (Clemente et al., 2009), have not shown favorable antitumor effects, and metronomic chemotherapy with toceranib, piroxicam, and thalidomide has reportedly shown better antitumor efficacy (Rossi et al., 2018). In FIMC, as in CIMC, toceranib, firocoxib, and chlorambucil, used as metronomic chemotherapy, aimed at reducing angiogenesis, may be considered more effective than MTD chemotherapy using cytotoxic chemotherapeutic agents (Petrucci et al., 2021a; Petrucci et al., 2024). However, Petrucci et al. (2021b) reported that in feline mammary carcinoma, the use of tyrosine kinase inhibitors, such as toceranib and chlorambucil, as metronomic chemotherapy alone did not achieve longer survival than surgical therapy and toceranib (Petrucci et al., 2024). This may be because toceranib and chlorambucil have antiangiogenic and immunosuppressive effects in small lesions at an early stage, but are ineffective against larger masses. Therefore, the use of tyrosine kinase inhibitors (toceranib and chlorambucil) as metronomic chemotherapy alone may not be effective against FIMC, and the combination of palliative tumor excision and postoperative tyrosine kinase inhibitor administration may be considered a more reasonable approach. This patient also had a local recurrence of IMC on day 25 with a small papule-like lesion, but a further increase in size was avoided using toceranib and firocoxib. Toceranib and firocoxib have shown some efficacy in inhibiting tumor progression. Although no postmortem examination was performed, the cat showed progressive azotemia and clinical signs of uremia consistent with chronic kidney disease rather than widespread metastatic FIMC, suggesting that renal failure was the most likely cause of death. Although the efficacy of palliative resection combined with tyrosine kinase inhibitors, such as toceranib, cannot be established in single cases, this report describes that palliative resection combined with tyrosine kinase inhibitors, such as toceranib, may provide meaningful pain relief and temporary disease stabilization in FIMC. This clinical outcome provides practical evidence to guide the management of similarly advanced cases. This study has some limitations. First, this was a single case report, and further case studies are needed to investigate pain relief using palliative surgery, the effects of toceranib and firocoxib, and the pathogenesis of secondary FIMC. Second, idiopathic FIMC cannot be completely ruled out. Third, the interpretation of HER2 staining results and the required dosage of lapatinib in cats are currently unclear (Yu et al., 2024; Holanda et al., 2025). Caution is required when interpreting lapatinib use in cats and HER2 staining results. Finally, no autopsy was performed; therefore, the degree of FIMC systemic metastasis could not be determined. ConclusionReports of FIMC are limited, and this case may provide an initial description of a suspected secondary form. There are concerns that the number of cases diagnosed with FIMC in postoperative pathology will increase in the future. However, palliative surgery, toceranib, and firocoxib maintained pain control and QoL for several months. Further case accumulation will clarify the optimal approach to this rare and aggressive disease. AcknowledgmentsThe authors would like to thank Masami Akiyoshi and the animal care staff at the Akiyoshi Animal Clinic for their dedicated assistance. Conflict of interestThe authors declare no potential conflicts of interest with respect to this article’s research, authorship, and/or publication. FundingThe authors received no financial support for the research, authorship, and publication of this article. Authors’ contributionsMA: conceived and designed the clinical study, analyzed and interpreted the data, and wrote the manuscript. MGA: analyzed and interpreted the data. MH: analyzed and interpreted the data. Data availabilityAll data were provided in the manuscript. ReferencesAbdelgalil, A.A. and Alkahtani, H.M. 2023. Lapatinib: a comprehensive profile. Profiles. Drug. Subst. Excip. Relat. Methodol. 48, 135–166. Belli, M., De Oliveira, A.R., De Lima, M.T., Trindade, P.H.E., Steagall, P.V. and Luna, S.P.L. 2021. Clinical validation of the short and long UNESP-Botucatu scales for feline pain assessment. PeerJ 9, e11225. Clemente, M., De Andrés, P.J., Peña, L. and Pérez‐Alenza, M.D. 2009. Survival time of dogs with inflammatory mammary cancer treated with palliative therapy alone or palliative therapy plus chemotherapy. Vet. Rec. 165(3), 78–81. Collins, D.M., Madden, S.F., Gaynor, N., AlSultan, D., Le Gal, M., Eustace, A.J., Gately, K.A., Hughes, C., Davies, A.M., Mahgoub, T., Ballot, J., Toomey, S., O'Connor, D.P., Gallagher, W.M., Holmes, F.A., Espina, V., Liotta, L., Hennessy, B.T., O'Byrne, K.J., Hasmann, M., Bossenmaier, B., O'Donovan, N. and Crown, J. 2021. Effects of HER family-targeting tyrosine kinase inhibitors on antibody-dependent cell-mediated cytotoxicity in HER2-expressing breast cancer. Clin. Cancer Res. 27(3), 807–818. Dawood, S., Merajver, S.D., Viens, P., Vermeulen, P.B., Swain, S.M., Buchholz, T.A., Dirix, L.Y., Levine, P.H., Lucci, A., Krishnamurthy, S., Robertson, F.M., Woodward, W.A., Yang, W.T., Ueno, N.T. and Cristofanilli, M. 2011. International expert panel on inflammatory breast cancer: consensus statement for standardized diagnosis and treatment. Ann. Oncol. 22(3), 515–523. Gameiro, A., Nascimento, C., Correia, J. and Ferreira, F. 2021. HER2-targeted immunotherapy and combined protocols showed promising antiproliferative effects in feline mammary carcinoma cell-based models. Cancers (Basel). 13(9), 2007. Holanda, A.G.A., Civa, P.A.S., Fonseca-Alves, C.E. and Dos Anjos, D.S. 2025. Safety and feasibility of lapatinib for the treatment of a EGFR1/HER-2-positive advanced gastrointestinal stromal tumor in a cat. Braz. J. Vet. 47, e001825. Marconato, L., Romanelli, G., Stefanello, D., Giacoboni, C., Bonfanti, U., Bettini, G., Finotello, R., Verganti, S., Valenti, P., Ciaramella, L. and Zini, E. 2009. Prognostic factors for dogs with mammary inflammatory carcinoma: 43 cases (2003-2008). J. Am. Vet. Med. Assoc. 235(8), 967–972. Millanta, F., Calandrella, M., Citi, S., Della Santa, D. and Poli, A. 2005. Overexpression of HER-2 in feline invasive mammary carcinomas: an immunohistochemical survey and evaluation of its prognostic potential. Vet. Pathol. 42(1), 30–34. Millanta, F., Verin, R., Asproni, P., Giannetti, G. and Poli, A. 2012. A case of feline primary inflammatory mammary carcinoma: clinicopathological and immunohistochemical findings. J. Feline. Med. Surg. 14(6), 420–423. Pérez Alenza, M.D., Tabanera, E. and Peña, L. 2001. Inflammatory mammary carcinoma in dogs: 33 cases (1995-1999). J. Am. Vet. Med. Assoc. 219(8), 1110–1104. Pérez-Alenza, M.D., Jiménez, A., Nieto, A.I. and Peña, L. 2004. First description of feline inflammatory mammary carcinoma: clinicopathological and immunohistochemical characteristics of three cases. Breast Cancer Res. 6(4), 300–307. Petrucci, G., Henriques, J., Gregório, H., Vicente, G., Prada, J., Pires, I., Lobo, L., Medeiros, R. and Queiroga, F. 2021b. Metastatic feline mammary cancer: prognostic factors, outcome and comparison of different treatment modalities - a retrospective multicentre study. J. Feline. Med. Surg. 23(6), 549–556. Petrucci, G.N., Henriques, J., Lobo, L., Vilhena, H., Figueira, A.C., Canadas‐Sousa, A., Dias‐Pereira, P., Prada, J., Pires, I. and Queiroga, F.L. 2021a. Adjuvant doxorubicin vs metronomic cyclophosphamide and meloxicam vs surgery alone for cats with mammary carcinomas: a retrospective study of 137 cases. Vet. Comp. Oncol. 19(4), 714–723. Petrucci, G.N., Magalhães, T.R., Dias, M. and Queiroga, F.L. 2024. Metronomic chemotherapy: bridging theory to clinical application in canine and felian oncology. Front. Vet. Sci. 11, 1397376. Queiroga, F.L., Perez-Alenza, M.D., Silvan, G., Peña, L., Lopes, C. and Illera, J.C. 2005. Cox-2 levels in canine mammary tumors, including inflammatory mammary carcinoma: clinicopathological features and prognostic significance. Anticancer. Res. 25(6B), 4269–4275. Relford, R., Robertson, J. and Clements, C. 2016. Symmetric dimethylarginine: improving the diagnosis and staging of chronic kidney disease in small animals. Vet. Clin. North Am. Small Anim. Pract. 46(6), 941–960. Rossi, F., Sabattini, S., Vascellari, M. and Marconato, L. 2018. The impact of toceranib, piroxicam and thalidomide with or without hypofractionated radiation therapy on clinical outcome in dogs with inflammatory mammary carcinoma. Vet. Comp. Oncol. 16(4), 497–504. Soares, M., Correia, J., Nascimento, C. and Ferreira, F. 2021. Anaplastic mammary carcinoma in cat. Vet. Sci. 8(5), 77. Soares, M., Ribeiro, R., Najmudin, S., Gameiro, A., Rodrigues, R., Cardoso, F. and Ferreira, F. 2016. Serum HER2 levels are increased in cats with mammary carcinomas and predict tissue HER2 status. Oncotarget 7(14), 17314–17326. Yu, T.W., Yamamoto, H., Morita, S., Fukushima, R., Elbadawy, M., Usui, T. and Sasaki, K. 2024. Comparative pharmacokinetics of tyrosine kinase inhibitor, lapatinib, in dogs and cats following single oral administration. J. Vet. Med. Sci. 86(3), 317–321. Zappulli, V., Peña, L., Rasotto, R., Goldschmidt, M., Gama, A., Scruggs, J., Kiupel, M.M. and Tumors. 2008. Mammary tumors. In Surgical pathology of tumors of domestic animals Volume 2. Kiupel, M Washington DC: Thompson DVM Foundation, 2, pp: 1–97. | ||
| How to Cite this Article |
| Pubmed Style Akiyoshi M, Asakawa MG, Hisasue M. Suspected secondary inflammatory mammary carcinoma developed from pre-existing tumors in a cat: Clinical course from diagnosis to death. Open Vet. J.. 2026; 16(2): 1394-1401. doi:10.5455/OVJ.2026.v16.i2.56 Web Style Akiyoshi M, Asakawa MG, Hisasue M. Suspected secondary inflammatory mammary carcinoma developed from pre-existing tumors in a cat: Clinical course from diagnosis to death. https://www.openveterinaryjournal.com/?mno=297520 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.56 AMA (American Medical Association) Style Akiyoshi M, Asakawa MG, Hisasue M. Suspected secondary inflammatory mammary carcinoma developed from pre-existing tumors in a cat: Clinical course from diagnosis to death. Open Vet. J.. 2026; 16(2): 1394-1401. doi:10.5455/OVJ.2026.v16.i2.56 Vancouver/ICMJE Style Akiyoshi M, Asakawa MG, Hisasue M. Suspected secondary inflammatory mammary carcinoma developed from pre-existing tumors in a cat: Clinical course from diagnosis to death. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 1394-1401. doi:10.5455/OVJ.2026.v16.i2.56 Harvard Style Akiyoshi, M., Asakawa, . M. G. & Hisasue, . M. (2026) Suspected secondary inflammatory mammary carcinoma developed from pre-existing tumors in a cat: Clinical course from diagnosis to death. Open Vet. J., 16 (2), 1394-1401. doi:10.5455/OVJ.2026.v16.i2.56 Turabian Style Akiyoshi, Makoto, Midori Goto Asakawa, and Masaharu Hisasue. 2026. Suspected secondary inflammatory mammary carcinoma developed from pre-existing tumors in a cat: Clinical course from diagnosis to death. Open Veterinary Journal, 16 (2), 1394-1401. doi:10.5455/OVJ.2026.v16.i2.56 Chicago Style Akiyoshi, Makoto, Midori Goto Asakawa, and Masaharu Hisasue. "Suspected secondary inflammatory mammary carcinoma developed from pre-existing tumors in a cat: Clinical course from diagnosis to death." Open Veterinary Journal 16 (2026), 1394-1401. doi:10.5455/OVJ.2026.v16.i2.56 MLA (The Modern Language Association) Style Akiyoshi, Makoto, Midori Goto Asakawa, and Masaharu Hisasue. "Suspected secondary inflammatory mammary carcinoma developed from pre-existing tumors in a cat: Clinical course from diagnosis to death." Open Veterinary Journal 16.2 (2026), 1394-1401. Print. doi:10.5455/OVJ.2026.v16.i2.56 APA (American Psychological Association) Style Akiyoshi, M., Asakawa, . M. G. & Hisasue, . M. (2026) Suspected secondary inflammatory mammary carcinoma developed from pre-existing tumors in a cat: Clinical course from diagnosis to death. Open Veterinary Journal, 16 (2), 1394-1401. doi:10.5455/OVJ.2026.v16.i2.56 |