| Research Article | ||
Open Vet. J.. 2026; 16(1): 189-200 Open Veterinary Journal, (2026), Vol. 16(1): 189-200 Research Article Albumin/Globulin ratio as a valuable marker for monitoring remdesivir therapy in feline infectious peritonitisRebar N. Mohammed1,2*, Ramyar M. S Ako3, Hanar A. Abdulrahman3, Rhawa H. Gharib2, Lava M. Sabir2, Khoshy Osman2 and Pzho Kamil21Department of Medical Laboratory Technology, Faculty of Health Science, Qaiwan International University, Sulaimani, Iraq 2College of Veterinary Medicine, University of Sulaimani, Sulaymaniyah, Iraq 3Department of Medical Laboratory Technology, Kurdistan Technical Institute, Sulaymaniyah, Iraq *Corresponding Author: Rebar N. Mohammed. College of Veterinary Medicine, University of Sulaimani,Sulaymaniyah, Iraq. Email: rebar.mohammed [at] univsul.edu.iq Submitted: 04/11/2025 Revised: 07/12/2025 Accepted: 13/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
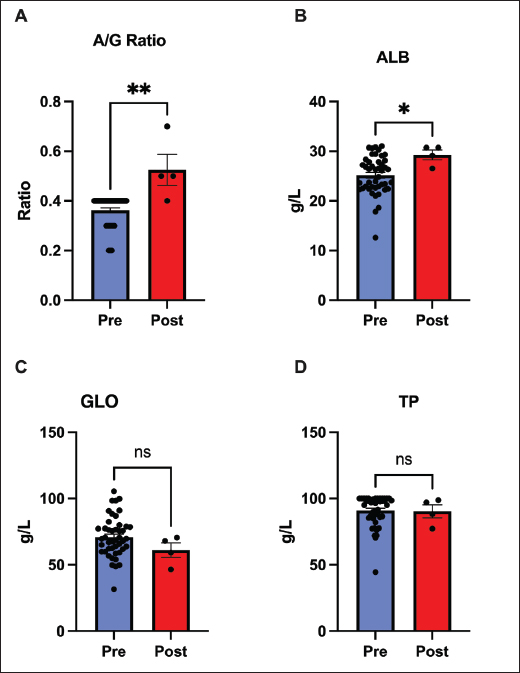
AbstractBackground: R Feline infectious peritonitis (FIP) is a viral infection in cats that develops when a coronavirus mutates and causes an immune reaction throughout. The disease is usually fatal and requires extreme care to save the animal Aim: This study examined the albumin-to-globulin (A/G) ratio as an important marker for monitoring the therapy effect. Simultaneously, the hematological and biochemical profiles of cats suffering from FIP were explored following intravenous treatment with remdesivir. Methods: Intravenous remdesivir was administered to 45 confirmed cases of FIP pets for 60 days. The cats were aged 3 months to 2 years because this is the most susceptible age in feline animals. Clinical data were collected at both the start and end of the 60-day period to examine the A/G ratio and monitor the hematological and biochemical profiles. Results: A significant alteration in the albumin concentration, A/G ratio, and lymphocyte counts was found after remdesivir administration for 60 days. The creatinine-to-Blood Urea Nitrogen ratio was also altered, indicating an improvement in renal metabolism or protein turnover. No significant changes were found in protein and globulin levels, Red Blood Cell indices, platelet counts, electrolytes, and minerals. We also did not find evidence of hepatic injury. Liver enzymes, including Alanine Aminotransferase, Aspartate Aminotransferase, and Gamma-Glutamyl Transferase, the ALT/AST ratio, and total bilirubin did not change, indicating no evidence of remdesivir-induced hepatic injury. Conclusion: The A/G ratio is useful to indicate recovery in FIP-affected cats. Keywords: Albumin, Globulin, A/G ratio, FIP, Coronavirus. IntroductionFeline infectious peritonitis (FIP) is a mutated form of coronavirus infection that allows the enteric, harmless virus to invade macrophages and spread throughout the body. Following the development of FIP, the disease results in a systemic inflammatory reaction that develops into a severe, progressive disease that requires medical intervention to ensure feline survival (Pedersen, 2014; Kipar and Meli, 2014). Feline Infectious Peritonitis affects domestic and wild felids, primarily cats aged 2 years. Data also indicate that it may be more common in male felines (Riemer, et al., 2016; Olarte-Castillo, et al., 2024). Clinically, FIP manifests in two major forms: the effusive or “wet” type and the non-effusive or “dry” type. The former includes fluid accumulation in body cavities, whereas the latter presents as granulomatous lesions in organs. Many cats show an overlap of both forms of FIP with time and after the disease has developed (Addie, et al., 2009). Currently, there is no clear diagnosis for this infection because no single test can confirm the same in a living cat. Diagnosis of FIP with confidence can be performed by clinicians only by assimilating information from history, physical examination, and various laboratory findings (Felten and Hartmann, 2019). Feline Infectious Peritonitis manifests with various signs, including weight loss, lethargy, inflammation, and fever that persists even with antibiotic treatment. Clinically, the disease often causes lymphopenia, mild nonregenerative anemia, and polyclonal hyperglobulinemia along with reduced albumin levels (Kennedy, 2020). Since total protein or globulin concentration alone is not specific enough to diagnose FIP, these protein imbalances lead veterinarians to use the albumin-to-globulin (A/G) ratio as a key supportive indicator. Generally, an A/G ratio below 0.4 suggests FIP, while one above 0.6–0.8 makes the diagnosis highly unlikely (Jeffery, et al., 2012). However, the combination of the A/G ratio with total protein and globulin concentration alone cannot confidently make the diagnosis, as similar changes may appear in other inflammatory diseases (Tasker, 2018; Tasker, et al., 2023). Hence, to fully support a diagnosis, veterinarians often rely on additional tests that detect coronavirus antibodies or Ribonucleic Acid (Longstaff, et al., 2017). When an effusion is present, its analysis can contribute to the diagnosis. FIP effusions are straw-colored, highly proteinaceous, and exhibit low cellularity with a low A/G ratio. A positive Rivalta’s test has high sensitivity for FIP, whereas a negative test practically eliminates it from consideration (Fischer et al., 2012). For many years, FIP was an incurable disease, and its management was designed around symptomatic treatment and corticosteroids, which provided only transient relief. In 2018, the development of GS-441524 and its prodrug remdesivir offered new hope for treating FIP (Moyadee et al., 2024). Since this discovery, studies have confirmed that both drugs exhibit high antiviral activity and can achieve survival rates of up to 80% (Pedersen et al., 2019; Goto et al., 2025). However, cats should be monitored throughout the treatment course. Clinicians must monitor felines for relapse and treatment failures, which are more common in cats with neurological complications or a late therapeutic response (Kim, et al., 2016; Taylor et al., 2023). Given that certain biomarkers indicate recovery, this study was conducted to assess their use in cats with FIP undergoing remdesivir treatment. We focused on the A/G ratio and the associated clinical pathology parameters. Past research has demonstrated that antivirals such as GS-441524 and remdesivir can reverse characteristic hyperalbuminemia and hyperglobulinemia, thereby improving the A/G ratio (Goto, et al., 2025). This increasing ratio likely reflects a decrease in inflammation. Moreover, successful therapy should normalize lymphocytes, which are frequently depressed in FIP due to virus-induced T-cell apoptosis (Katayama et al., 2024). Therefore, we postulated that successful therapy would increase albumin and the A/G ratio, increase total lymphocyte count, and demonstrate stable organ function. Materials and MethodsAnimals and study designWe enrolled 45 domestic cats (21 males and 24 females) who presented at the referral veterinary clinic from June to September 2024. The cats were aged between 3 and 24 months, with a median age of 12.6 months. The diagnosis was based on the typical clinical presentation of abdominal pain, abdominal effusion, and laboratory abnormalities, including globulin elevation, low albumin concentration, and low A/G ratio, along with positive immunofluorescent feline coronavirus test results. The subjects contained both effusive and non-effusive forms, while ocular or neurological involvement did not exclude cats, but none showed severe neurological signs at the beginning of this study. All cats were treated with intravenous remdesivir (GS-5734) from a licensed veterinary compounding pharmacy. The drug was administered once daily at a dose of 5 mg/kg, with mild adjustments made according to the clinical response, ranging from 4 to 6 mg/kg, for 60 days, following previously described protocols (Dietz , 2022). Supportive management, including fluid therapy and nutritional care, was provided based on individual needs, but corticosteroids and other antiviral agents were intentionally excluded. Each cat acted as its own control, enabling a direct comparison between baseline and post-treatment parameters. Data and sample collectionData for each cat were collected at two time points, before and after 60 days of treatment. Data recorded for each cat included clinical examination, body weight, and hematological and biochemical analyses. For biochemical analyses, 1 ml of blood was collected in lithium-heparin tubes and immediately centrifuged at 2,500 rpm for 3 minutes to separate the plasma. Blood samples were collected from the jugular vein in kittens and from the cephalic or saphenous veins in larger cats (Taylor, 2015). The MNCHIP Celercare V5 veterinary chemistry analyzer (Tianjin, China) was used for biochemical analysis. The measured components included total protein (TP), albumin (ALB), globulin (GLO-calculated as TP-ALB), and the A/G ratio. Other parameters were measured in relation to hepatic and renal functions. Finally, the creatinine-to-BUN ratio was calculated to compare the balance of protein metabolism with renal excretion. Whole blood samples were collected in ethylenediaminetetraacetic acid tubes for complete blood count. All samples were analyzed within 1 h of collection to prevent cellular artifacts. Statistical analysisData analysis was performed using the GraphPad Prism software (version 9.3.1). For each parameter, the values of each cat were compared before and after treatment. Because the data were normally distributed, mean comparisons were performed using paired two-tailed t-tests. The results are presented as the mean ± SEM. The error bars in the figures represent the standard error of the mean (SEM), which is used to visualize the key findings. Ethical approvalThe procedure performed in this study was approved by the College of Veterinary Medicine Research Committee, University of Sulaimani, Kurdistan Regional Government, Kurdistan/Iraq, under registration no. VET0266 on 27/05/2022. ResultsIn these feline patients, significant improvements in serum protein markers were observed following the 60-day remdesivir treatment: both serum albumin (p < 0.05) and the A/G ratio (p < 0.01) increased. The mean A/G ratio increased from approximately 0.4–0.8. In contrast, total protein and globulin levels did not significantly change (p > 0.05). The improvement in the A/G ratio appears to be due to an increase in albumin, accompanied by stability or a slight decrease in globulin. Normalization of the A/G ratio to such an extent usually indicates recovery or remission. These data selectively emphasize that remdesivir therapy tends to restore protein homeostasis, likely by improving hepatic albumin synthesis and concomitantly reducing inflammation-related hyperglobulinemia. Thus, our study indicates that the A/G ratio is a more sensitive marker of treatment response than total protein concentration alone (Fig. 1).
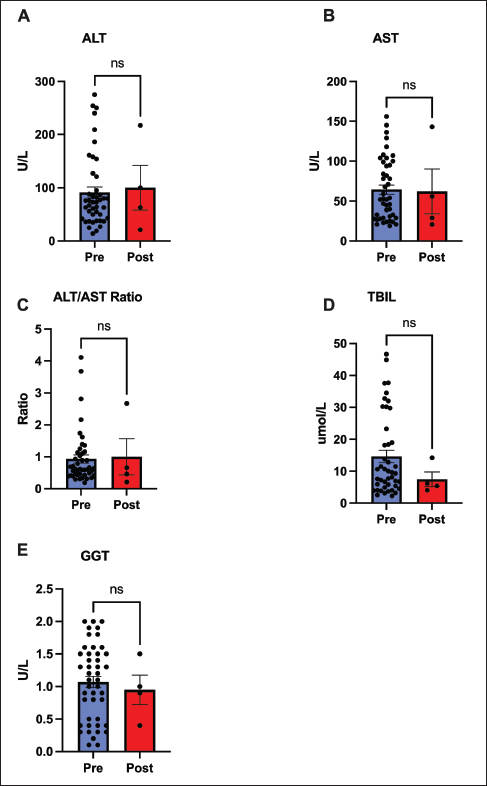
Fig. 1. Changes in serum protein markers before and after treatment with remdesivir for 60 days. Comparison of (A) albumin/globulin (A/G) ratio, (B) albumin, (C) globulin, and (D) total protein. Both the A/G ratio and albumin levels significantly increased (p < 0.01 and p < 0.05, respectively), reflecting normalization of protein homeostasis. Globulin and total protein levels remained unchanged. Data are presented as mean ±SEM. (*p < 0.05, **p < 0.01, ns=not significant). No hepatotoxicity or significant hepatic recovery occurred within 60 days after remdesivir administration, with largely stable liver parameters. No significant changes were observed in ALT, AST, GGT, or bilirubin levels before and after treatment (p > 0.05). The ALT/AST ratio also remained unchanged. Only a few cats developed mild elevation of baseline bilirubin levels; no overt cases of jaundice were noted (Fig. 2).
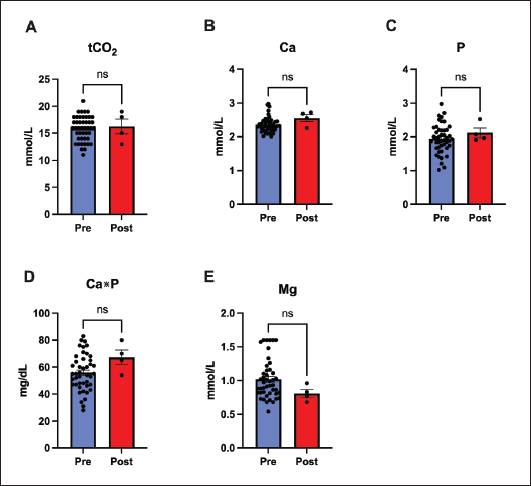
Fig. 2. Assessment of hepatic function markers before and after treatment with remdesivir for 60 days. Key hepatic markers remained stable, supporting the safety of remdesivir treatment. Panels show (A) ALT, (B) AST, (C) ALT/AST ratio, (D) total bilirubin (TBIL), and (E) gamma-glutamyl transferase (GGT), none of which changed significantly. Data represent mean ± SEM. (ns=not significant). Systemic electrolyte and mineral concentrations did not show any evidence of metabolic or organ-related disturbances and remained within the referent ranges throughout the treatment. The values for Carbon Dioxide, calcium, phosphorus, magnesium, and the calculated Ca×P product before and after treatment did not differ significantly (p > 0.05 for all comparisons) (Fig. 3).
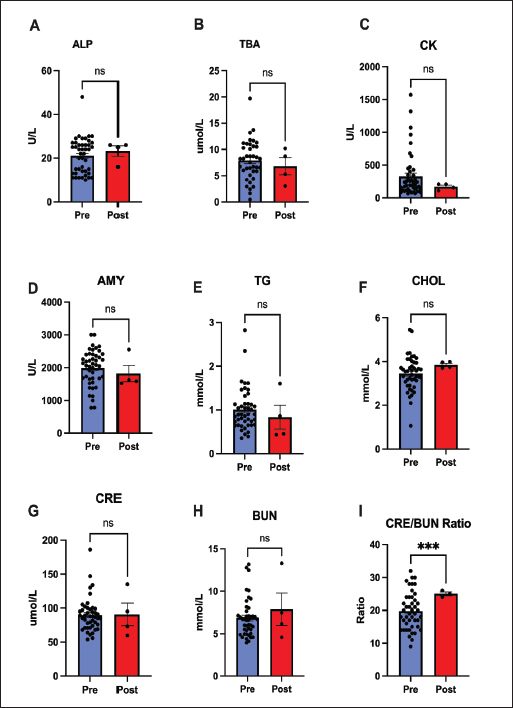
Fig. 3. Stability of systemic electrolytes and minerals after 60 days of remdesivir therapy. Panels show (A) total carbon dioxide (tCO2), (B) calcium (Ca), (C) phosphorus (P), (D) Calcium-Phosphorus Product (Ca*P), and (E) magnesium (Mg). All parameters remained unaffected and within normal ranges, indicating no metabolic or renal disturbances. Values are mean ± SEM. (ns=not significant). Most biochemical markers remained unchanged after treatment for 60 days. Hepatic and muscular markers, such as ALP, bile acids, Creatine Kinase, and amylase, showed no significant change (p > 0.05). Similarly, metabolic and renal markers, such as cholesterol and triglycerides, did not significantly change. The exception was the creatinine-to-BUN ratio, which changed significantly (p < 0.001) (Fig. 4).
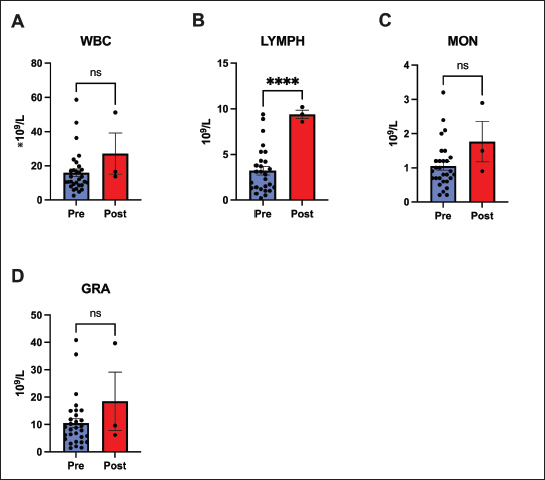
Fig. 4. Biochemical, metabolic, and renal marker analysis after 60 days of remdesivir. Most parameters, including (A) ALP, (B) TBA, (C) CK, (D) AMY, (E) TG, (F) CHOL, (G) CRE, and (H) BUN, remained stable. The sole exception was a significant increase in the (I) CRE/BUN ratio (p < 0.001). Values are presented as mean ± SEM. (*** p < 0.001, ns=not significant). Our analysis of the hematological response after 60 days of remdesivir therapy indicates a significant recovery of immune competence. Throughout the treatment, total WBC, granulocyte, and monocyte cell lines remained stable and within the reference limits. Notably, there was a significant increase in the lymphocyte count (p < 0.0001). These counts rose from low or low-normal baseline values to within or above the referent range after treatment, suggesting a rebound from FIP-induced immunosuppression (Fig. 5).
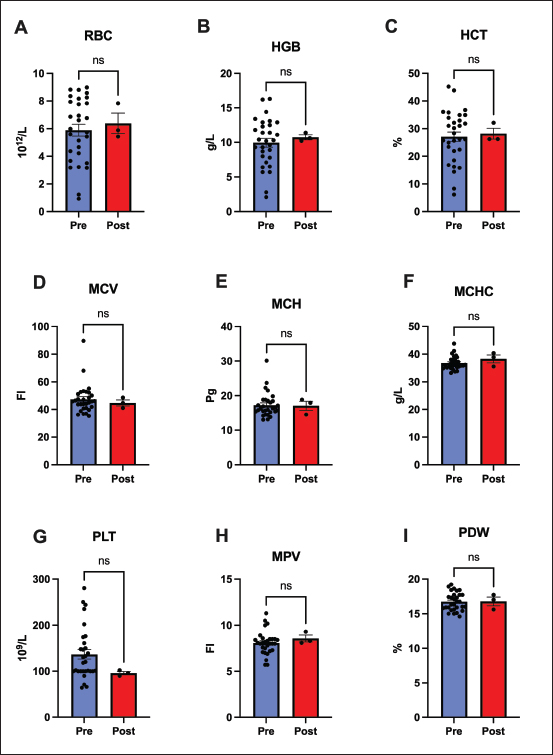
Fig. 5. White blood cell populations. This figure compares white blood cell populations before and after 60 days of remdesivir administration. (A) Total WBC, (C) monocytes, and (D) granulocytes remained stable, and (B) lymphocyte counts increased significantly (p < 0.0001). This lymphocyte recovery indicates a rebound from FIP-induced immunosuppression. Data represent mean ± SEM. (**** p < 0.0001, ns=not significant). Unlike the lymphocyte response, no significant changes were observed in the RBC count or platelet count after treatment. Mean RBC count, Hemoglobin, and Hematocrit remained stable (p > 0.05), with no notable changes in Mean Corpuscular Volume, Mean Corpuscular Hemoglobin, or Mean Corpuscular Hemoglobin Concentration . Some cats had mild anemia before treatment, which generally persisted after 60 days (Fig. 6).
Fig. 6. Erythrocyte and platelet parameters stability after 60 days of remdesivir. No significant alterations were found in (A) red blood cell (RBC) count, (B) hemoglobin (HGB), (C) hematocrit (HCT), (D-F) red cell indices (MCV, MCH, and MCHC), (G) platelet (PLT) count, or (H-I) platelet indices (MPV and PDW). Data represent mean ± SEM. (ns=not significant). DiscussionIn this study, the clinicopathological responses of FIP-affected cats following 60 days of antiviral therapy were evaluated, with a particular focus on the diagnostic and prognostic utility of the A/G ratio. Our cohort included cats, aged 3–24 months. This age range is consistent with established FIP epidemiology, which shows that the disease primarily affects cats aged 2 years (Thayer et al., 2022; Murphy et al., 2024). While some studies have suggested a higher risk of FIP in male felines (Pesteanu-Somogyi et al., 2006; Gaonkar et al., 2019), our study enrolled a relatively balanced group (21 males vs. 25 females). All cats exhibited FIP-related clinical signs, including persistent fever, weight loss, lethargy, and abdominal distension in effusive cases. These consistent demographics and clinical signs confirm that our study cohort is representative of typical, naturally occurring FIP cases seen in veterinary practice (Abbas, et al., 2014; Hamin, 2023). The most striking observation in our study was the dramatic improvement in the A/G ratio within 60 days following remdesivir therapy. Most of the cats enrolled in this study initially presented with A/G ratios close to or below 0.4, an indicator of active FIP due to hypoalbuminemia and hyperglobulinemia (Nururrozi, et al., 2022; Moyadee, et al., 2024). After therapy, however, most cats had an A/G ratio greater than 0.6–0.8, indicating normalization of their serum protein balance. This shift was the result of two changes: first, a remarkable increase in albumin. Albumin is a negative acute-phase reactant, and its rebound suggests decreased systemic inflammation and enhanced hepatic protein synthesis (Barle, et al., 2006). Second, globulins remained stable or slightly decreased. In FIP, high globulin levels are a consequence of chronic antigenic stimulation (Goitsuka et al., 1990; Kipar and Meli, 2014); therefore, its stabilization indicates decreased immune activation after viral replication control. Thus, the rise in the A/G ratio appears to be an undisputed hallmark of both clinical and immunological recovery, further supporting its utility as an inexpensive and informative biomarker of therapeutic response. Although protein parameters improved, hepatic markers such as ALT, AST, and bilirubin did not change. This stability of liver values has two major implications. First, from the standpoint of disease, it suggests that there was neither significant deterioration nor marked recovery in hepatic function over the 60-day period. FIP-related liver enzyme abnormalities are usually mild unless granulomatous lesions are directly present within the liver (Attipa, et al., 2023). Hyperbilirubinemia without high ALT may occur in FIP, possibly due to cytokine-mediated cholestasis independent of direct liver cell damage (Tsai et al., 2011; Torres et al., 2024). The stability of bilirubin in our cohort could indicate that the cats either did not have significant cholestasis at baseline or that it takes longer than 60 days to resolve. Second, the liver panel stability strongly supports the safety of remdesivir and indicates that it is not notably hepatotoxic in cats at this dosage. These results indicate that liver function was mild at the start of treatment and that continuous therapy did not worsen or resolve hepatobiliary activity during the 60-day period. Even in the absence of significant ALT elevations, hyperbilirubinemia is commonly seen in 20%–60% of the effusive form of FIP cases. The lack of significant change in bilirubin or hepatic enzyme alteration supports the view that remdesivir does not exhibit adverse action on liver function, consistent with prior safety evaluations (Force et al., 2023). This finding is particularly significant, given that remdesivir is known to elevate liver enzymes in other species, including humans (Kaka and Wilt, 2022). Our data align with other large field studies in cats, which have found that the hepatic side effects of remdesivir are rare (Rasmussen et al., 2022; Coggins et al., 2023). Furthermore, a number of metabolic and renal markers did not show any significant changes, but the ratio of creatinine-BUN showed a significant change, which represents a significant improvement in the renal protein handling in the cats following remedsivir therapy (Maharani et al., 2025). The most critical immunological finding was the significant rebound of lymphocyte counts (Dascalu, et al., 2024). FIP characteristically results in T-cell depletion and lymphopenia (Knotek et al., 2000; Vermeulen et al., 2013), suggesting a restitution of immune competence. We attribute this to reduced viral replication and the subsequent decrease in systemic inflammation (Mustaffa-Kamal et al., 2019). Our observation is consistent with other reports that equate lymphocyte recovery with clinical remission, positioning adaptive immunity restoration as a vital marker of therapeutic success (Addie, et al., 2022; Tršar, et al., 2019). No significant changes in erythrocyte parameters were observed following the treatment regimen. This lack of fluctuation is consistent with the type of anemia associated with FIP (Olson and Hohenhaus, 2019). Chronic anemia results from cytokine-mediated suppression of erythropoiesis and impaired iron metabolism; resolution typically occurs only after full remission and reduction of systemic inflammation (Chikazawa and Dunning, 2016; Ottenjann et al., 2006). The persistence of mild anemia in some cats likely reflects ongoing recovery or the short monitoring interval, as hematologic normalization may require several months post-therapy (Černá, et al., 2015). FIP cats have lower RBC counts and Packed Cell Volume than healthy cats (Moyadee, et al., 2019). The fact that hemoglobin and hematocrit did not significantly rise suggests that while the cats were clinically improving, their bone marrow was likely still in recovery mode. The stability of RBC parameters might have been affected by fluid shifts. Rehydrated cats following therapy showed lower hematocrit count, masking a simultaneous increase in RBCs caused by erythropoiesis during recovery. The lack of significant statistical changes is likely due to the two opposing factors of rehydration and improved erythropoiesis; there was no evidence of treatment-induced hemolysis or hemorrhage. We hypothesize that a significant increase in RBC mass (resolution of anemia) may be observed following a longer follow-up period (e.g., 3–6 months) if the cats remain disease-free. These results demonstrate that remdesivir therapy does not cause renal failure or systemic electrolyte imbalances. Calcium and phosphorus levels remained stable during the entire experiment. Furthermore, no evidence of metabolic acidosis, alkalosis, or any abnormal mineral disposition was observed. The findings are consistent with those of previous studies (Daka et al., 2021; Green et al., 2023). Our findings are complementary and confirm existing evidence about the effectiveness of remdesivir in treating cats affected by FIP, as evidenced in Cosaro et al. (2023) and Krentz et al. (2022). This is supported by a normal A/G ratio, a return to normal lymphocyte counts, and unchanging hepatic and renal profiles. Taken together, these findings point to the pivotal role of remdesivir in biochemical recovery and immune reconstitution in cats with FIP. Our data indicate that the A/G ratio and lymphocyte counts are affordable and practical FIP monitoring markers. Cats demonstrating a continuously rising A/G ratio and normalized lymphocytes probably respond appropriately to treatment. Persistent low values might indicate incomplete suppression of the virus or the need for dose adjustment. The inclusion of these markers in follow-up care would add an objective element to the monitoring of recovery, augmenting the usual clinical assessments. The major limitation of this study was the lack of a placebo or untreated control group. Remedsevir is a lifesaving therapy for FIP, a disease that is otherwise progressive and invariably fatal (Goodson et al., 2009; Nekoui et al., 2022). Positive outcomes we have encountered—such as an increase in albumin, A/G ratio, and lymphocytes—are not typical for spontaneous remission. In fact, all these markers are expected to deteriorate during the disease’s natural, untreated course (Bohm, 2022). Therefore, we believe that the observed positive trends are a direct result of the therapeutic benefits of remdesivir. ConclusionRemdesivir therapy is highly effective in reversing the fatal trajectory of FIP and restoring normal physiological and biochemical profiles. Our findings confirm that the A/G ratio provides a reliable and inexpensive tool for monitoring the recovery process. Therefore, we recommend that regular monitoring of the A/G ratio be incorporated into the standard clinical management of FIP as a useful adjunct to routine clinical assessment. In our investigation, we confirmed that remdesivir is both safe and effective. This was based on the significant normalization of key hematological and biochemical parameters and the clear absence of any hepatic or renal toxicity. Further clinical follow-up in addition to multicenter studies about long-term survival outcome, relapse rates, and any possible delayed adverse effects associated with these antiviral therapies are recommended. The advent of remdesivir-based therapy has fundamentally improved the outlook for FIP. When coupled with vigilant clinical monitoring and laboratory tools, such as the A/G ratio, veterinarians now have a realistic means to guide treatment, evaluate prognosis, and sustain recovery in a disease that was once universally fatal. AcknowledgmentI would like to thank the College of Veterinary Medicine, University of Sulaimani, Suleimanyah, Iraq, for supporting the research and providing us with the ethical consideration and help and ethical approval of the study. Conflict of interestThe authors declare no conflict of interest. FundingNone. Authors’ contributionsRNM and RS designed and supervised the study. KO and PK performed the hematological and biochemical tests for the animals. RHG and HAA supervised and helped with the laboratory work. Later, RNM, RS, HAA, and RHG analyzed all the data and created the graphs with the statistical analysis. RNM, RS, KO, and PK wrote the manuscript and prepared all the aspects of the manuscript. Data availabilityThe datasets used and analyzed in this study are available from the corresponding author upon reasonable request. ReferencesAbbas, G., Mughal, M.N., Asi, M.N. and Muhammad, G. 2014. Feline Infectious Peritonitis in a Domestic Cat in Pakistan: a Case Report. Int. J. Innov. Appl. Res. 2(6), 21–25. Addie, D.D., Belak, S., Boucraut, C. and Egberink, H.F. 2009. Feline Infectious Peritonitis: aBCD Prevention and Management Guidelines. J. Feline Med. Surg. 11(7), 594–604. Addie, D. D., Silveira, C., Aston, C., Brauckmann, P., Covell-Ritchie, J., Felstead, C., Fosbery, M., Gibbins, C., Macaulay, K., McMurrough, J., Pattison, E., and Robertson, E. 2022. Alpha-1 Acid Glycoprotein Reduction Differentiated Recovery from Remission in a Small Cat Cohort Treated for Feline Infectious Peritonitis Viruses. 14(4) 14(4), 744. Attipa, C., Gunn-Moore, D., Mazeri, S., Lyraki, M., Hardas, A. and Gentil, M. 2023. Feline infectious peritonitis outbreak in Cyprus. Vet. Rec. 192(11), 449–450. Barle, H., Hammarqvist, F., Westman, B., Klaude, M., Rooyackers, O., Garlick, P. J., and Wernerman, J. 2006. The synthesis rates of total liver protein and albumin are both increased in patients with an acute inflammatory response. Clin. Sci. 110(1), 93–99. Bohm. and M. 2022. Successful treatment of effusive feline infectious peritonitis with remdesivir in a South African cat. J. South Afr. Vet. Med. Assoc. 93(2), 112–115. Černá, P., Ayoob, A., Baylor, C., Champagne, E., Hazanow, S., Heidel, R.E., Wirth, K., Legendre, A.M. and Gunn-Moore, D.A. 2015. Retrospective Survival Analysis of Cats with Feline Infectious Peritonitis Treated with Polyprenyl Immunostimulant That Survived over 365 Days. Pathogens 11(8), 1–14. Chikazawa, S. and Dunning, M.D. 2016. A review of inflammatory disease anemia in dogs and cats. J. Small Anim. Pract. 57, 348–353. Coggins, S.J., Norris, J.M., Malik, R., Govendir, M., Hall, E.J., Kimble, B. and Thompson, M.F. 2023. Outcomes of treatment of cats with feline infectious peritonitis using parenterally administered remdesivir, with or without transition to orally administered GS-441524. J. Vet. Internal. Med. 37(5), 1772–1783. Cosaro, E., Pires, J., Castillo, D., Murphy, B. G., and Reagan, K. L. 2023. Efficacy of Oral Remdesivir Compared with GS-441524 for Cats with Naturally Occurring Effusive Feline Infectious Peritonitis: a Blinded, Non-Inferiority Study. Viruses 15(8), 1680. Daka, V. N., Bandi, L. S., Alla, S., Cheedella, V. S., and Galaba, S. R 2021. A brief review of remdesivir. Int. J. Health. Care. Biol. Sci. 2(3), 57–62. Dascalu, M.A., Daraban Bocaneti, F., Soreanu, O., Tutu, P., Cozma, A., Morosan, S. and Tanase, O. 2024. Filgrastim efficiency in cats naturally infected with feline panleukopenia virus. Animals 14(24), 3582. Dietz, B. 2022. Successful treatment of a South African cat with effusive feline infectious peritonitis with remdesivir. J. S. Afr. Vet. Assoc. 93(2), 112–115. Felten, S. and Hartmann, K. 2019. Diagnosis of Feline Infectious Peritonitis: a Review of the Current Literature. Viruses 11(11), 1068. Fischer, Y., Sauter-Louis, C. and Hartmann, K. 2012. Diagnostic accuracy of the Rivalta test for infectious peritonitis in felines. Vet. Clin. Pathol. 41(4), 558–567. Force, L., Blair, C., Duckworth, J., Abdelghany, M., Nguyen, N.-Q., Hyland, R. H., Davies, S., Chen, S., Zhao, Y., and Fu, O. 2023. Hepatic Safety of Remdesivir Across Phase 3 Placebo-Controlled Study. Open Forum Infect. Dis. 10(Supplement 2). Gaonkar, P., Halmare, N., Jamdade, S., Sonkusale, P. and Kurkure, N. 2019. Feline Infectious Peritonitis in a Male Persian Cat. Int. J. Curr. Microbiol. Appl. Sci. 8(1), 1446–1453. Goitsuka, R., Ohashi, T., Ono, K., Yasukawa, K., Koishibara, Y., Fukui, H., Ohsugi, Y. and Hasegawa, A. 1990. IL-6 activity in feline infectious peritonitis. J. Immunol. 144(7), 2599–2603. Goodson, T.I., Randell, S.C. and Moore, L. 2009. Feline infectious peritonitis. Compend. Contin. Educ. Vet. 31(10), 31. Goto T Kamiyoshi. and Iwasaki, R. 2025. Predictive factors associated with short-term mortality in cats with feline infectious peritonitis treated with remdesivir, GS-441524, or both. J. Vet. Internal. Med. 39(1), e17249. Green, J.M., Syme, H.M. and Tayler, S. 2023. Thirty-two cats with effusive or noneffusive feline infectious peritonitis treated with Remdesivir and GS-441524. J. Vet. Internal Med. 37(5), 1784–1793. Hamin, A.W. 2023. STUDI KASUS: feline infectious peritonitis (FIP) TIPE EFUSIF PADA KUCING TINA. Vitek. Bidang. Kedokteran. Hewan. 13(2), 114–120. Jeffery, U., Deitz, K.L. and Hostetter, S.J. 2012. Positive predictive value of albumin: globulin ratio for feline infectious peritonitis in a mid-western referral hospital population. J. Feline Med. Surg. (SAGE. Publications). 14(12), 903–905. Kaka, A.S. and Wilt, T.J. 2022. Major Update 2: remdesivir for Adults with COVID-19. Venereal. Dis. Infection. 175, W81. Katayama, M., Uemura, Y. and Katori, D. 2024. Effect of nucleic acid analog administration on fluctuations in the albumin-to-globulin ratio in cats with feline infectious peritonitis. Animals 14(9), 1322. Kennedy, M.A. 2020. Feline Infectious Peritonitis: update on Pathogenesis, Diagnostics, and Treatment. Vet. Clinics North Amer. Small Anim. Pract. 50(5), 1001–1011. Kim, Y., Liu, H., Galasiti Kankanamalage, A.C., Weerasekara, S., Hua, D.H., Groutas, W.C., Chang, K.O. and Pedersen, N.C. 2016. Reversal of the progression of fatal coronavirus infection in cats by a broad-spectrum coronavirus protease inhibitor. PLos Pathogens. 12(5), e1005650. Kipar, A. and Meli, M.L. 2014. Feline infectious peritonitis: still an enigma?. Vet. Pathol. 51(2), 505–526. Knotek, Z., Toman, M. and Faldyna, M. 2000. Clinical and immunological characteristics of cats with feline infectious peritonitis. Acta. Veterinaria. Brno. 69(1), 51–60. Krentz, D H. et al., 2022. Clinical Follow-Up and Postmortem Findings in a Cat Cured of Feline Infectious Peritonitis with GS-441524 Viruses, vol. 14, no. 9, pp. 2040-2040. Longstaff, L., Porter, E., Crossley, V. J., Hayhow, S. E., Helps, C. R., and Tasker, S. 2017. Quantitative reverse-transcriptase polymerase chain reaction of feline coronavirus on effusion samples in cats with and without feline infectious peritonitis. J. Feline. Med. Surg. (SAGE. Publications). 19(2), 240–245. Maharani S, Dani, R.N. and Sutra R, et al (2025) Feline infectious peritonitis and feline leukemia virus co-infection : a case study on diagnostic and clinical pathological findings. 9:15–16 Moyadee, W., Sunpongsri, S., Choowongkomon, K., Roytrakul, S., Rattanasrisomporn, A., Tansakul, N. and Rattanasrisomporn, J. 2024. Feline infectious peritonitis: a comprehensive evaluation of clinical manifestations, laboratory diagnosis, and therapeutic approaches. J. Adv. Vet. Anim. Res. 11(1), 19–26. Moyadee, W., Jaroensong, T., Roytrakul, S., Boonkaewwan, C., and Rattanasrisomporn, J. 2019. Characteristic clinical signs and blood parameters in cats with FIP. Agriculture. Natural. Resour. 53(4), 433–438. Murphy, B.G., Castillo, D., Neely, N.E., Kol, A., Brostoff, T., Grant, C.K. and Reagan, K.L. 2024. Serologic, Virologic, and Pathologic Features of Cats with Naturally Occurring Feline Infectious Peritonitis Enrolled in Antiviral Clinical Trials. Viruses 16(3), 462. Mustaffa-Kamal, F., Liu, H., Pedersen, N.C. and Sparger, E.E. 2019. Characterization of antiviral T cell responses during primary and secondary challenge of laboratory cats with feline infectious peritonitis virus (FIPV). BMC Vet. Res. 15(1), 99–115. Nekouei, O., St-Hilaire, S., Hui, P. C., Chan, K., Chan, I. S., Ngan, S. Y. L., Chan, Y., Chung, K. P., Hong, S., Chan, H. M. I., Or, H. L. I., Chan, F. Y., Yim, H. T., and Barrs, V. R. 2022. Potential therapeutic effects of GS-441524 and GC376 in cats with infectious peritonitis. Vet. Evidence 7(1), 1–12. Nururrozi, A., Ramandani, D., Wasissa, M., Yanuartono, and Indarjulianto, S. 2022. Serum Biochemistry Profiles in Cats with Confirmed Effusive Feline Infectious Peritonitis. Adv. Anim. Vet. Sci. 10(1), 126–130. Olarte-Castillo, X.A., Goodman, L.B., Whittaker, G. 2024. Molecular detection using hybridization capture and next-generation sequencing reveals cross-species transmission of feline coronaviru type-1 between a domestic cat and a captive wild felid. Microb. Spectr. 12(10), e0006124; doi: 10.1128/spectrum.00061-24 Ottenjann C Weingart. and Arndt Kohn, B. 2006. Characterization of inflammatory disease anemia in cats with abscesses, pyothorax, or fat necrosis. J. Vet. Internal Med. 20(5), 1143–1150. Pedersen, N.C. 2014. Update on feline infectious peritonitis: diagnostics and therapeutics. Vet. J. 201(2), 133–141. Pedersen, N.C. and et al. 2019. Efficacy and safety of the nucleoside analog GS-441524 for treating naturally occurring feline infectious peritonitis in cats. J. Feline Med. Surg. 21(4), 271–281. Pesteanu-Somogyi, L.D. and Radzai C Pressler. 2006. Prevalence of infectious peritonitis in specific cat breeds. J. Feline Med. Surg. 8(1), 1–5. Rasmussen, H.B., Thomsen, R. and Hansen, P.R. 2022. Nucleoside analog GS-441524: pharmacokinetics in different species, safety, and potential effectiveness against Covid-19. Pharmacol. Res. &. Perspect. 10(2), 1–10. doi:10.1016/j.prpert.2010.09.010 Riemer, F., Kuehner, K. A., Ritz, S., Sauter-Louis, C., and Hartmann, K. 2016. Clinical and laboratory features of feline infectious peritonitis: a retrospective study of 231 confirmed cases (2000-2010). J. Feline Med. Surg. 18(4), 348–356. Salord Torres, X., Jeevaratnam, K., Schofield, I., Taylor, S., Stallwood, J., Heyes, M., Hughes, D. and Defauw, P. 2024. Diagnostic and predictive ability of hyperbilirubinemia severity in cats: a multicenter retrospective study. J. Vet. Internal Med. 38(2), 1043–1050. Sarker, D. et al., 2025. Molecular detection using hybridization capture and next-generation sequencing reveals cross-species transmission of feline coronavirus type-1 between a domestic cat and a captive wild felid. BioRxiv. 2012;2(1), pp. 14-18. Tasker, S., Addie, D.D., Egberink, H., Hofmann-Lehmann, R., Hosie, M.J., Truyen, U., Belák, S., Boucraut-Baralon, C., Frymus, T., Lloret, A., Marsilio, F., Pennisi, M.G., Thiry, E., Möstl, K. and Hartmann, K. 2023. Feline Infectious Peritonitis : european Advisory Board on Cat Diseases Guidelines. Viruses 15(1), 103. Tasker. and S. 2018. Diagnosis of feline infectious peritonitis: update on evidence supporting available tests. J. Feline. Med. Surg. (SAGE. Publications. Ltd). 20(3), 228–243. Taylor, S. M. (2015). Small-Animal Clinical Techniques 2nd ed. St. Louis: Saunders. Taylor, S. S., Coggins, S., Barker, E. N., Gunn-Moore, D., Jeevaratnam, K., Norris, J. M., Hughes, D., Stacey, E., MacFarlane, L., O’Brien, C., Korman, R., McLauchlan, G., Salord Torres, X., Taylor, A., Bongers, J., Espada Castro, L., Foreman, M., McMurrough, J., Thomas, B., Royaux, E., Calvo Saiz, I., Bertoldi, G., Harlos, C., Work, M., Prior, C., Sorrell, S., Malik, R., and Tasker, S. 2023. Retrospective study and outcome of 307 cats with feline infectious peritonitis treated with remdesivir and GS-441524 (2020-2022). J. Feline Med. Surg. 25(9) 1–12. Thayer, V., Gogolski, S., Felten, S., Hartmann, K., Kennedy, M. and Olah, G.A. 2022. 2022 AAFP/EveryCat Feline Infectious Peritonitis Diagnosis Guidelines. J. Feline Med. Surg. 24(9), 905–933. Tršar, L., Štrljič, M., Svete, A.N., Koprivec, S., Tozon, N., Žel, M.K. and Pavlin, D. 2019. Evaluation of selected inflammatory markers in cats with feline infectious peritonitis before and after therapy. BMC. Vet. Res. 21, 330. Tsai, H., -Y.., Chueh, L., -L.., Lin, C., -N.. and Su, B.L. 2011. Clinicopathological findings and disease staging of feline infectious peritonitis: 51 cases in Taiwan from 2003 to 2009. J. Feline. Med. Surg. 13(2), 74–80. Vermeulen, B. L., Devriendt, B., Olyslaegers, D. A., Dedeurwaerder, A., Desmarets, L. M., Favoreel, H. W., Dewerchin, H. L., and Nauwynck, H. J. 2013. Suppression of natural killer cells and regulatory T lymphocytes in cats naturally infected with feline infectious peritonitis virus. Vet. Microbiol. 164(1), 46–59. Winzelberg Olson, S. and Hohenhaus, A.E. 2019. Feline nonregenerative anemia: diagnostic and treatment recommendations. J. Feline Med. Surg. 21(7), 615–631. | ||
| How to Cite this Article |
| Pubmed Style Mohammed RN, Ako RMS, Abdulrahman HA, Gharib RH, Sabir LM, Osman K, Kamil P. Albumin/Globulin ratio as a valuable marker for monitoring remdesivir therapy in feline infectious peritonitis. Open Vet. J.. 2026; 16(1): 189-200. doi:10.5455/OVJ.2026.v16.i1.18 Web Style Mohammed RN, Ako RMS, Abdulrahman HA, Gharib RH, Sabir LM, Osman K, Kamil P. Albumin/Globulin ratio as a valuable marker for monitoring remdesivir therapy in feline infectious peritonitis. https://www.openveterinaryjournal.com/?mno=294486 [Access: January 31, 2026]. doi:10.5455/OVJ.2026.v16.i1.18 AMA (American Medical Association) Style Mohammed RN, Ako RMS, Abdulrahman HA, Gharib RH, Sabir LM, Osman K, Kamil P. Albumin/Globulin ratio as a valuable marker for monitoring remdesivir therapy in feline infectious peritonitis. Open Vet. J.. 2026; 16(1): 189-200. doi:10.5455/OVJ.2026.v16.i1.18 Vancouver/ICMJE Style Mohammed RN, Ako RMS, Abdulrahman HA, Gharib RH, Sabir LM, Osman K, Kamil P. Albumin/Globulin ratio as a valuable marker for monitoring remdesivir therapy in feline infectious peritonitis. Open Vet. J.. (2026), [cited January 31, 2026]; 16(1): 189-200. doi:10.5455/OVJ.2026.v16.i1.18 Harvard Style Mohammed, R. N., Ako, . R. M. S., Abdulrahman, . H. A., Gharib, . R. H., Sabir, . L. M., Osman, . K. & Kamil, . P. (2026) Albumin/Globulin ratio as a valuable marker for monitoring remdesivir therapy in feline infectious peritonitis. Open Vet. J., 16 (1), 189-200. doi:10.5455/OVJ.2026.v16.i1.18 Turabian Style Mohammed, Rebar N., Ramyar M. S Ako, Hanar A. Abdulrahman, Rhawa H. Gharib, Lava M. Sabir, Khoshy Osman, and Pzho Kamil. 2026. Albumin/Globulin ratio as a valuable marker for monitoring remdesivir therapy in feline infectious peritonitis. Open Veterinary Journal, 16 (1), 189-200. doi:10.5455/OVJ.2026.v16.i1.18 Chicago Style Mohammed, Rebar N., Ramyar M. S Ako, Hanar A. Abdulrahman, Rhawa H. Gharib, Lava M. Sabir, Khoshy Osman, and Pzho Kamil. "Albumin/Globulin ratio as a valuable marker for monitoring remdesivir therapy in feline infectious peritonitis." Open Veterinary Journal 16 (2026), 189-200. doi:10.5455/OVJ.2026.v16.i1.18 MLA (The Modern Language Association) Style Mohammed, Rebar N., Ramyar M. S Ako, Hanar A. Abdulrahman, Rhawa H. Gharib, Lava M. Sabir, Khoshy Osman, and Pzho Kamil. "Albumin/Globulin ratio as a valuable marker for monitoring remdesivir therapy in feline infectious peritonitis." Open Veterinary Journal 16.1 (2026), 189-200. Print. doi:10.5455/OVJ.2026.v16.i1.18 APA (American Psychological Association) Style Mohammed, R. N., Ako, . R. M. S., Abdulrahman, . H. A., Gharib, . R. H., Sabir, . L. M., Osman, . K. & Kamil, . P. (2026) Albumin/Globulin ratio as a valuable marker for monitoring remdesivir therapy in feline infectious peritonitis. Open Veterinary Journal, 16 (1), 189-200. doi:10.5455/OVJ.2026.v16.i1.18 |