| Research Article | ||
Open Vet. J.. 2026; 16(2): 1243-1258 Open Veterinary Journal, (2026), Vol. 16(2): 1243-1258 Research Article Omental pedicle transposition for neuropathic stress urinary incontinence: a urodynamic evaluationRania Kh. Kh. Al-Juboury* and Hameed. A. Al-TimmemiDepartments of Surgery and Obstetrics, College of Veterinary Medicine, University of Baghdad, Baghdad, Iraq *Corresponding Author: Rania Kh. Kh. Al-Juboury. Departments of Surgery and Obstetrics, College of Veterinary Medicine, University of Baghdad, Baghdad, Iraq. Email: rania.khedr2202m [at] covm.uobaghdad.edu.iq Submitted: 03/11/2025 Revised: 25/12/2025 Accepted: 03/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
AbstractBackground: The pudendal nerve (PDN) plays a crucial role in external urethral sphincter (EUS) innervation, contributing to urinary control and coordinating the action of the bladder with the EUS. Aim: This study aimed to assess the therapeutic potential of omental pedicle transposition in treating neuropathic stress urinary incontinence induced by complete transection of the motor branch of the PDN (MBPDN). Methods: Forty healthy adult female New Zealand White rabbits were randomly divided into four groups (n=10). In group A, the MBPDN was completely transected and left untreated. The transected MBPDN was repaired in group B through direct end-to-end coaptation. Group C underwent end-to-end nerve coaptation after neurotmesis, followed by wrapping the coaptation site with an omental pedicle transposition. Group D served as the normal control without nerve injury and was included only for comparative statistical analysis. Results: Cystometric measurements were performed at weeks 4 and 16 to evaluate detrusor pressure (P_det), bladder compliance (BC), and leak point pressure (LPP). These parameters were used to assess bladder dynamics. Surface electromyography of the external urethral sphincter (EUS) was also recorded at the same time points (4th and 16th weeks) to measure the EUS amplitude and assess its neuromuscular integrity. Cytometric analysis revealed a highly significant treatment effect (p < 0.001). Compared with the normal control group (D), group A showed the most severe EUS dysfunction, characterized by increased P_det, minimal BC, and the lowest LPP, indicating persistent detrusor overactivity and poor detrusor-sphincter coordination following untreated MBPDN injury. Group B demonstrated moderate improvement after end-to-end nerve coaptation, indicating partial recovery. By contrast, group C showed the greatest degree of functional recovery, with bladder function parameters approaching those of the normal control group D by week 16. The P_det progressively decreased, whereas BC and LPP increased to near-normal values, indicating almost complete restoration of detrusor stability and sphincteric control. Conclusion: In conclusion, these findings indicate that omental pedicle transposition enhances transected MBPDN regeneration and functional recovery of the EUS, leading to near-normal bladder function restoration compared with direct end-to-end nerve coaptation alone. Keywords: Neuropathic stress urinary incontinence, Motor branch of pudendal nerve, Omental pedicle transposition, Nerve regeneration, Surface electromyography. IntroductionNeuropathic stress urinary incontinence (UI) (NSUI) is a consequence of lumbosacral lesions (El Homsi et al., 2024) that result from injuries to the spinal cord or motor branch of the pudendal nerve (PDN) (MBPDN) that innervates the external urethral sphincter (EUS) (Zapletal et al., 2024). NSUI is the most prevalent condition following transection injury to the MBPDN, which innervates the EUS (Giulioni et al., 2024). Surgical interventions for severed MBPDN: End-to-end tension-free coaptation has historically been the favored regeneration technique for lesions with a gap of 5 mm (Herman and Ilyas, 2020). The omentum is a substantial mesenchymal fibro-fatty tissue known for its exceptional healing properties. The stromal cells of the omentum, which comprise omental adipose cells, fibroblasts, and leukocytes, are a prolific source of growth factors (Shahraki et al., 2024). The omentum regulates inflammation and facilitates revascularization (Morawska-Kozłowska et al., 2024). The omentum is a highly vascularized organ that secretes multiple angiogenic and anti-inflammatory substances and provides several types of stem cells (Goldsmith, 2012). Vasoactive cytokines have been observed in the omentum, resulting in enhanced neurological functions (Fay et al., 2021); the omentum also produces neurotrophins (Kambe et al., 2023). Urodynamics are especially advantageous for accurately diagnosing lower urinary tract functional problems, including UI. The word “urodynamics” has generally become synonymous with pressure/flow studies, with most clinicians referring to cystometry (Chapple et al., 2018). Surface electromyography (sEMG) of the EUS plays a critical role in assessing the neuromuscular activity of this key continence-related muscle in the evaluation of stress UI (SUI) (Shafik, 1992). Injury to the innervation of the EUS at the level of the PDN can result in neurogenic changes that can be identified by electromyography (EMG) (McCloskey et al., 2024). When the EMG is integrated with cystometric testing, synchronized data are provided that reflect the real-time electrical activity of the EUS during the bladder filling phase (Gray, 2011). This simultaneous recording allows for the accurate detection of any delay, weakness, or abnormal firing patterns in the response of the EUS to increases in intra-abdominal pressure (Steward et al., 2010). Materials and MethodsExperimental animalsThis study used 40 healthy adult New Zealand White female rabbits weighing 1.9–2.5 kg in good health as experimental subjects. All animals were housed individually in cages and provided with vegetables, leafy greens, commercial food, and adequate water. A 15-day acclimatization period was provided to allow the rabbits to adjust to their new environment. During this period, the rabbits received a daily intramuscular administration of ceftriaxone, a broad-spectrum antibiotic, at a dosage of 20 mg/kg for five consecutive days (Beskid et al., 1988). Additionally, on the first day and day 14, they were given a subcutaneous injection of ivermectin, an anthelmintic, at a dosage of 0.2 mg/kg (Wang et al., 2023). Calculation of sample sizeA formal sample size calculation was conducted to justify the inclusion of 10 animals per group (n=40). The calculation was based on the detrusor pressure (P_det) as the primary outcome measure. A biologically significant difference of 5 cmH₂O between groups was assumed, with the standard deviation (SD ≈ 0.774) derived from preliminary data. The level of significance was set at α=0.05, and the statistical power was fixed at 80% (1−β=0.80). The required sample size was estimated using the following formula for comparing two independent means: n=2 × (Zα/2 + Zβ/Δ/SD)² as described by Charan and Kantharia (2013). Based on these parameters, the calculated sample size supported the use of 10 animals per group, which was considered sufficient to detect statistically significant differences in P_det between the experimental groups. The calculation indicates a minimal number of animals due to the large effect size relative to SD; 10 animals per group were selected to account for potential variability, dropouts, or unforeseen complications. This choice is consistent with previous studies on PDN neurotmesis in rabbits (Zabbia et al., 2024). This sample size ensures adequate statistical power while minimizing the number of animals used in accordance with ethical guidelines. Experimental designThe experimental design of this study involved 40 New Zealand White female rabbits. Neurotmesis of the left motor branch of the PDN (MBPDN) was performed after general anesthesia to induce NSUI. The surgical procedure was performed only in groups A, B, and C, whereas group D served as a normal control without any surgical intervention. All experimental animals were then divided randomly into four equal groups: group A, group B, group C, and group D. Ingroup A, the left MBPDN neurotmesis was left untreated, in group B, the left MBPDN neurotmesis was treated by end-to-end coaptation, and in group C, the left MBPDN neurotmesis was treated by end-to-end coaptation with wrapping the coaptation site using omental pedicle transposition. Group D included as normal control animals without any surgical defects. The inclusion of a normal control group D without surgical intervention was considered essential to establish baseline physiological values for cystometric and electromyographic (EMG) parameters. This group provided a reference for normal lower urinary tract function, allowing for the accurate interpretation of functional recovery in the experimental groups. Anesthetic protocol and preoperative managementAnimals were randomly allocated to the experimental groups using a simple randomization method prior to the surgical procedure. Food was withheld from the rabbits for 2 hours. As a pre-anesthetic drug, a dose of 1 mg/kg body weight of acepromazine maleate was injected intramuscularly (IM), followed by injection of a mixture of 50 mg/kg of ketamine hydrochloride and 4 mg/kg of xylazine hydrochloride IM (Flecknell et al., 2015). During the surgical procedures, all animals were monitored perioperatively. The depth of anesthesia was assessed by monitoring the respiratory rate, heart rate, pedal withdrawal reflex, and palpebral reflex. Body temperature was maintained within the physiological range, and any signs of distress were continuously observed throughout the procedure. Surgical protocolThe test subject was positioned in ventral recumbency, and the surgical aperture was crafted on the dorsal midline just above the area from the acetabulum and downward under the tuber ischi through the fenestrated surgical drape at the designated surgical locale. A skin incision was made 1 cm left lateral to the dorsal midline and extended downward from the greater trochanter to the tuber ischi. Both the subcutaneous layer and fascia were sectioned, thereby separating the gluteus maximus muscle and the muscle underneath it, the gluteus medius muscle. The muscle section was achieved using the blunt dissection approach, facilitated by Mayo scissors, revealing the underlying neurovascular structures, including the pudendal artery and vein lateral to the vertebral column and the MBPDN medial to the vertebral column. In group A, the MBPDN was laid bare at the operation site using a self-retaining wound dilator, and neurotmesis was executed using microsurgical scissors to cut the nerve from the midportion. For group B, after MBPDN neurotmesis, the proximal and distal segments of the nerve were coaptated by two interrupted epineurial sutures using 8-0 nylon sutures. The coaptated nerve was then carefully repositioned, flushed with physiological saline, and the excess saline was swabbed with sterile gauze. In group C, the hair on the midline abdominal wall was clipped off, and an antiseptic was applied from the xiphoid cartilage to the pubic region. Following coaptation of the transected left MBPDN, the position of the animal was changed to dorsal recumbency to create an omental pedicle. A laparotomy was performed with a 5-cm midline incision from the umbilical to the pubis. The omentum was extended by releasing the dorsal leaf of the omentum by blunt dissection from the duodenum and descending colon. The omentum was then incised in an inversion L-shape to provide a double length of the omental pedicle (Fig. 1A). The omental pedicle was extended caudally on the peritoneal surfaces of the abdominal wall at the pelvic bone level. The lower part of the abdominal wall was perforated, and the omental pedicle was gently retracted using straight Kelly forceps (Fig. 1B). The omental pedicle was extended to the left MBPDN coaptation site. The animal’s position was changed to ventral recumbency. The omentum was wrapped around the coaptated nerve area and fixed with the gluteus medius muscle by two stitches of 4.0 PDS simple interrupted suturing (Fig. 1C). A simple continuous suture was applied to close the gluteus medius muscle. The gluteus maximus muscle was closed using 4-0 PDS continuous sutures, and the skin was closed using a 4-0 PDS subcuticular suture pattern. The abdominal wall was closed with routine work by closing the peritoneum and fascia using 4-0 PDS continuous sutures, closing the subcutaneous tissue using 4-0 PDS continuous sutures, and then closing the skin using a 4-0 PDS subcuticular suture pattern. Postoperative careAll rabbits received routine postoperative care after surgery. Ceftriaxone was administered intramuscularly at a dose of 20 mg/kg (Beskid et al., 1988) twice daily for 5 days to prevent postoperative infection. To control postoperative pain, acepromazine was administered intramuscularly at a dose of 1 mg/kg (Chen and Quesenberry, 2006) once daily for 3 days. During the recovery period, the animals were observed daily for general activity, feeding behavior, wound condition, and any signs of pain, distress, or complications. Cystometry and EMG testsBladder and urethral function were assessed after the 4th and 16th postoperative weeks, representing the early and late phases of peripheral nerve regeneration, respectively. The 4-week time point was selected to detect early functional changes associated with initial axonal regeneration, whereas the 16-week time point was chosen to evaluate more advanced functional recovery and maturation of neural reinnervation (Mohammadi et al., 2013). Evaluations were conducted using cystometric parameters, including P_det, BC, and leak point pressure (LPP), as well as EMG amplitude. An investigator blinded to group allocation performed all assessments. Statistical analysisCystometry and EMG amplitude data subjected to two-way analysis of variance using the standard least squares procedure of JMP Pro 16.0.0 software (SAS Institute Inc., Cary, NC). The model included the fixed factors of treatment (A, B, C, and D), (W4 vs. W16), and their interaction (Treatment × Week). Significant main effects and interaction means were identified using the least significant difference post-hoc test. Results with p ≤ 0.05 were considered statistically significant (SAS Institute Inc., 2021).
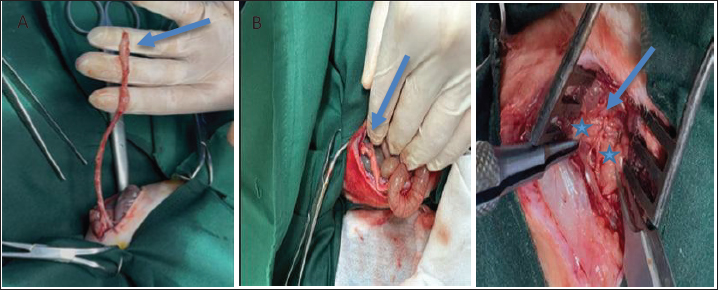
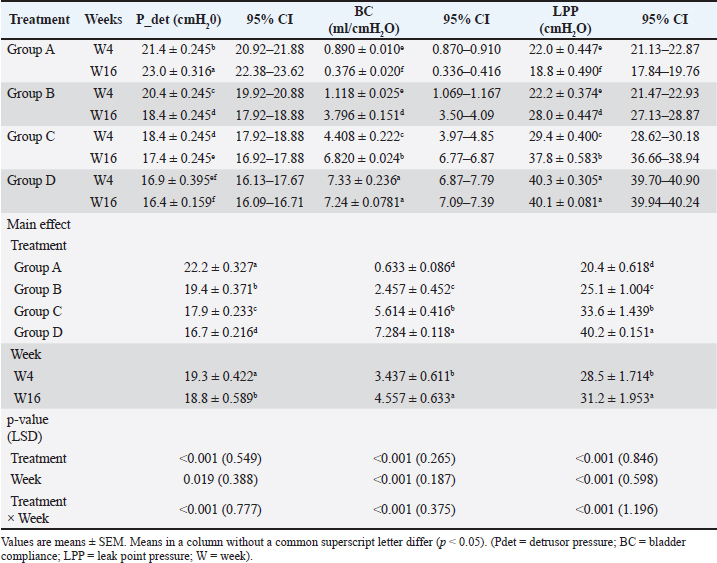
Fig. 1. Surgical procedure for omental pedicle transposition. (A) Laparotomy: 5-cm paramedian incision from the umbilicus to the pubis. (B) Straight Kelly forceps were used to extend the omental pedicle to the coaptation site of the motor branch of the pudendal nerve. C. Omental pedicle wrapped around the pudendal nerve’s coaptated motor branch. Ethical approvalAll experimental animals used in this study were approved by the Scientific Committee of the College of Veterinary Medicine, University of Baghdad, in compliance with the ethical principles’ guidelines on the care and use of animals in research (approval no: 5871 P.G, date 13/3/2024) of animal welfare. ResultsCystometryP_detThe treatment had a high effect on p-values (p < 0.001). At the 4th and 16th postoperative weeks, group A recorded the highest P_det values (21.4 ± 0.245 cmH₂O at week 4 and 23.0 ± 0.316 cm H₂O at week 16), which were higher than those of the normal control group D (see Table 1), and persistent detrusor overactivity in group A (see Figs. 3 and 4). Group D maintained a stable detrusor function throughout the study period (Fig. 2). Group B showed a moderate but consistent enhancement in P_det values over time (Figs. 5 and 6), with P_det decreasing from 20.4 ± 0.245 cmH₂O at week 4 to 18.4 ± 0.245 cmH₂O at week 16 (p < 0.05) (Table 1). In contrast, group C demonstrated a steady and greater reduction in P_det from 18.4 ± 0.245 cmH₂O at week 4 to 17.4 ± 0.245 cmH₂O at week 16, approaching the normal physiological values of group D (Table 1). Detrusor stability was clearly observed in group C at both evaluation points, mirroring the stable pattern of the normal control group D (Figs. 7 and 8). The main effect of time (p=0.019) indicated a general improvement in bladder function over the postoperative period, particularly in groups B and C (Table 1). The interaction between treatment and time was high (p < 0.001), confirming that the type of intervention directly influenced the extent of P_det reduction across the study period. Table 1. Comparison of cystometry parameters between groups A, B, C, and D in the 4th and 16th weeks of PO
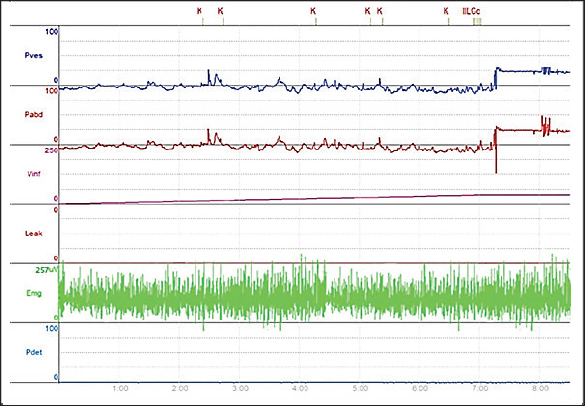
Fig. 2. Cystometry and EMG examination in group D. The chart shows preserved continence during the six episodes of increased intra-abdominal pressure (P abd), followed by increased intra-vesical pressure (P ves); detrusor activity remained stable (P_det), and leakage was only observed once the bladder reached its maximum capacity of 111 ml. Periurethral EMG tracing showed an amplitude of 277 μV.
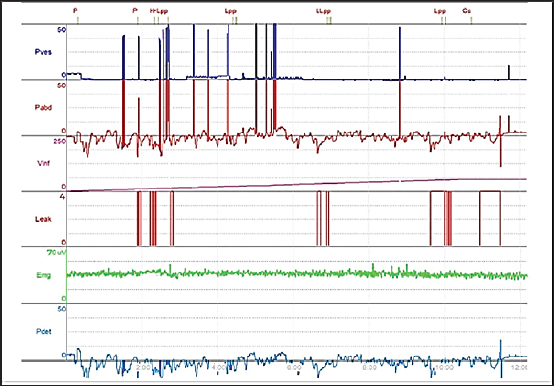
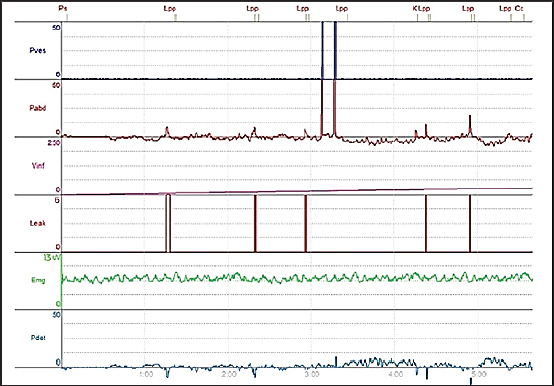
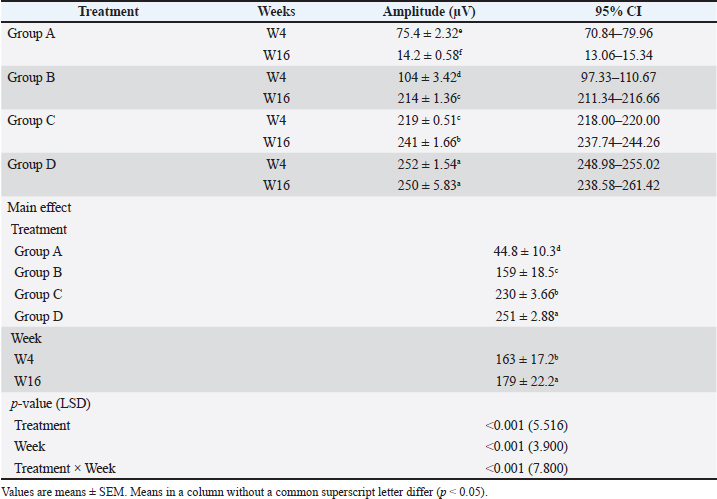
Fig. 3. Cystometry and electromyography examination in group A at week 4 postoperatively. The chart shows four leakage events occurring during the six episodes of increased intra-abdominal pressure (P abd), which were followed by increased intra-bladder vesical (P ves), detrusor overactivity (P_det) is evident throughout the tracing, and terminal continuous leakage was observed once the bladder reached its maximum capacity of 17 ml. Periurethral EMG tracing shows an amplitude of 70 μV. BCA high main effect of the treatment on BC was observed (p < 0.001). Group A exhibited severely reduced BC throughout the postoperative period. Group B showed gradual improvement, whereas group C demonstrated pronounced recovery. Over time, the BC increased in both treated groups (B and C), from 1.118 ± 0.025 ml/cmH₂O at week 4 to 3.796 ± 0.151 ml/cmH₂O at week 16 (group B) and from 4.408 ± 0.222 ml/cmH₂O at week 4 to 6.820 ± 0.024 ml/cmH₂O at week 16 (group C). The normal control group D maintained the highest and most stable values throughout the experiment period (≈7.3 ml/cmH₂O) (Table 1). The main effect of time was also significant (p < 0.001), indicating progressive improvement in BC as the postoperative period progressed (Table 1). The treatment × time interaction (p < 0.001) confirmed that the degree of improvement depended on the intervention applied. By week 16, the values recorded in group C were comparable to those of group D (Table 1). LPPThe main effect of treatment on LPP was high (p < 0.001). Group A exhibited the lowest LPP values at both evaluation points (weeks 4 and 16). Group B showed moderate improvement over time, while group C demonstrated values approaching those of the normal control group D, particularly by the 16th postoperative week (37.8 ± 0.583 cmH₂O vs. 40.1 ± 0.081 cmH₂O) (Table 1). The main effect of time (p < 0.001) reflects the overall enhancement in sphincteric function across the treated groups (B and C) as the postoperative period progressed (Table 1). The interaction between treatment and time (p < 0.001) confirms that omental pedicle transposition strongly influenced the extent of LPP improvement (see Table 1). sEMGAmplitudeThe EMG amplitude showed differences among the experimental groups and across the study period (p < 0.001). Group A exhibited the lowest amplitude, starting at 75.4 ± 2.32 µV at week 4 and declining sharply to 14.2 ± 0.58 µV by week 16, compared with the stable normal values of group D (252 ± 1.54 µV) (Table 2) (Figs. 2–4). Group B showed partial recovery, with the EMG amplitude rising from 104 ± 3.42 µV at week 4 to 214 ± 1.36 µV at week 16 (Table 2; Figs. 5 and 6). Group C exhibited a higher EMG amplitude at week 4 (219 ± 0.51 µV), which was further improved to 241 ± 1.66 µV by week 16, approaching the normal values of group D and indicating near-complete functional recovery of the EUS (Figs. 2, 7, and 8). The main effect of treatment was high (p < 0.001), with mean EMG amplitudes increasing progressively from group A (44.8 ± 10.3 µV) to group C (230 ± 3.66 µV), while group D maintained the highest and most stable values (251 ± 2.88 µV). This pattern highlights a strong treatment-dependent recovery in groups B and C relative to an untreated group A (Table 2). The main effect of time was also high (p < 0.001), showing an overall increase in the EMG amplitude from 163 ± 17.2 µV at week 4 to 179 ± 22.2 µV at week 16 postoperatively (PO) (Table 2). The interaction between treatment and time was high (p < 0.001), indicating that the type of intervention strongly influenced the rate and pattern of EMG-amplitude recovery. Group C displayed a steady and time-dependent enhancement closely resembling the normal profile of group D. By week 16 PO, group B showed a delayed but notable increase in EMG amplitude. In contrast, group A exhibited a progressive decline in the EMG amplitude over time (Table 2).
Fig. 4. Cystometry and EMG examination in group A at week 16 postoperative week. The chart shows six leakage events occurring during the six episodes of increased intra-abdominal pressure (P abd), which were followed by increased intra-bladder vesical (P ves) and detrusor overactivity (P_det) evident throughout the tracing, and terminal continuous leakage was observed once the bladder reached its maximum capacity of 6 ml. Periurethral EMG tracing shows an amplitude of 13 μV. Table 2. Comparison of amplitude between groups A, B, C, and D in 4th and 16th weeks PO.
DiscussionP_detCystometric analysis of the P_det revealed clear distinctions among the experimental groups, reflecting how each surgical method influenced bladder performance after complete transection of the MBPDN. P_det increased gradually in group A over 16 weeks PO (Table 1). This increase indicates ongoing detrusor overactivity, which is likely due to the absence of neural regeneration and the persistent effects of denervation. Previous studies have reported similar results, where untreated PDN injury produced heightened detrusor activity and maladaptive contractility (Hannestad et al., 2000; Furuta et al., 2008). The mild nature of the rise observed in the current study might be explained by species-specific compensatory mechanisms or the partial involvement of alternative neural inputs (Kerns et al., 2000). Overall, these data reinforce the hypothesis that the PDN contributes to the vesico-inhibitory reflex, suppressing the detrusor contractions during the bladder filling phase by stimulating the EUS contraction (Herroelen et al., 2024). P_det decreased gradually in group B over 16 weeks PO (Table 1). This decline indicates that direct end-to-end nerve coaptation helps to restore part of the neural pathway and moderate detrusor activity. These findings align with the earlier work showing that PDN regeneration can reestablish both sensory and motor communication, leading to improved bladder regulation (Sakamoto et al., 2000). The limited degree of recovery might be attributed to incomplete axonal regeneration, scar formation at the repair site, and misdirection of regenerating axons (Tannemaat et al., 2009). Nevertheless, the partial restoration observed here supports the findings of a previous study demonstrating that end-to-end nerve repair enhances nerve conduction and improves urinary control (Portillo et al., 2004). The P_det values dropped in group C over 16 weeks PO, reaching the values of normal control group D (Table 1). This improvement is likely due to the beneficial influence of the omental pedicle transposition, which provides the neurotrophic and vascular environment that encourages the axonal regeneration and functional recovery of the EUS (Yang and Chen, 2024). The omentum is well recognized for its trophic capacity and its ability to promote nerve regeneration when wrapped around the coaptated nerve (Castañeda and Kinne, 2002; Siemionow and Brzezicki, 2009). It secretes a diverse range of growth-promoting molecules, including nerve growth factor (NGF), brain-derived neurotrophic factor (BDNF), ciliary neurotrophic factor (CNTF), and glial cell line-derived neurotrophic factor (GDNF), all of which play distinct roles in neuronal survival, axonal elongation, and Schwann cell activation (Dujovny et al., 2004; Gama et al., 2020). NGF supports sensory neuron maintenance and axonal sprouting (Zorman et al., 2007), whereas BDNF enhances neuronal plasticity and functional reconnection (Leal et al., 2017; Solinas et al., 2019). Additionally, CNTF and GDNF preserve motor neurons and reduce apoptosis during the early stages following nerve injury (Kotliarova and Sidorova, 2021; Guo et al., 2022; Ma et al., 2022).
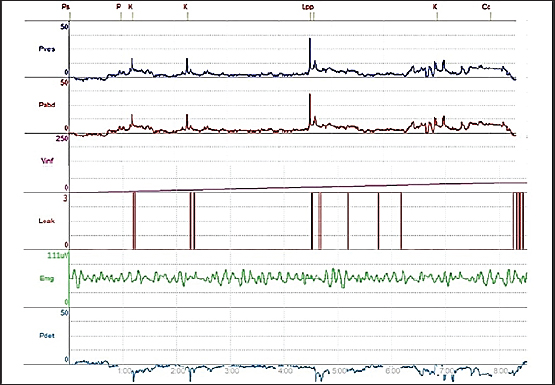
Fig. 5. Cystometry and EMG examination in group B at week 4 postoperative. The chart shows three leakage events occurring during the six episodes of increased intra-abdominal pressure (P abd), followed by increased intra-bladder vesical (P ves), detrusor overactivity (P_det) is evident throughout the tracing, and terminal continuous leakage was observed once the bladder reached its maximum capacity of 23 ml. Periurethral EMG tracing showed an amplitude of 111 μV.
Fig. 6. Cystometry and EMG examination in group B at week 16 postoperative The chart shows preserved continence during the six episodes of increased intra-abdominal pressure (P abd), followed by increased intra-vesical pressure (P ves), detrusor activity remained stable (P_det), and leakage was only observed once the bladder reached its maximum capacity of 68 ml. Periurethral EMG tracing showed an amplitude of 218 μV.
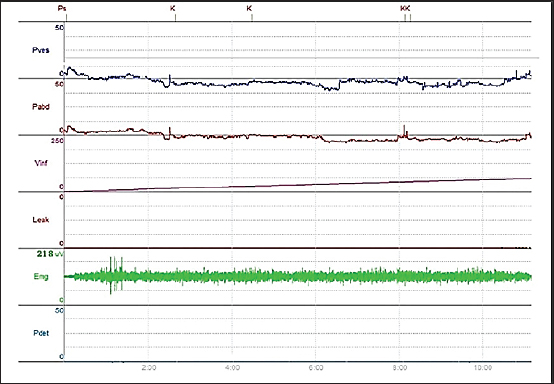
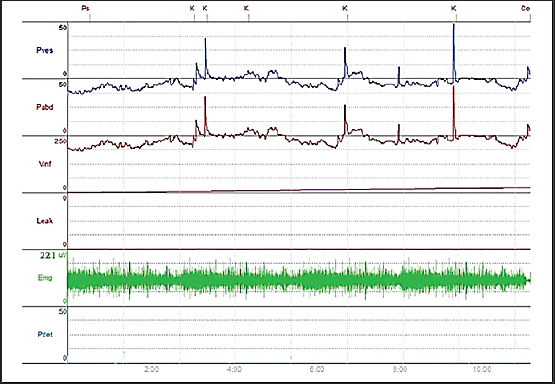
Fig. 7. Cystometry and EMG examination in group C at week 4 postoperatively. The chart shows preserved continence during the six episodes of increased intra-abdominal pressure (P abd), followed by increased intra-vesical pressure (P ves), detrusor activity remained stable (P_det), and leakage was only observed once the bladder reached its maximum capacity of 69 ml. Periurethral EMG tracing showed an amplitude of 221 μV.
Fig. 8. Cystometry and EMG examination in group C at week 16 postoperatively. The chart shows preserved continence during the six episodes of increased intra-abdominal pressure (P abd), followed by increased intra-vesical pressure (P ves); detrusor activity remained stable (P_det), and leakage was only observed once the bladder reached its maximum capacity of 96 ml. Periurethral EMG tracing showed an amplitude of 245 μV. BCThe BC analysis revealed pronounced variations among the experimental groups, highlighting the influence of the different repair approaches on bladder wall adaptability following the MBPDN transection. BC is a critical indicator of the bladder’s ability to expand during the filling phase while maintaining low intravesical pressure, and its impairment is a hallmark of neurogenic bladder dysfunction. The BC declined significantly in group A compared with the normal control group D (Table 1). This progressive deterioration indicates marked bladder rigidity and poor detrusor–sphincter coordination resulting from chronic denervation. These results are consistent with previous studies showing that untreated transection of the PDN leads to fibrotic remodeling, loss of elasticity, and structural alteration of the bladder wall (Yang et al., 2022). The extremely low BC observed in this model may also be influenced by species-specific differences in neural regeneration and the degree of compensatory parasympathetic innervation, which could explain the higher BC residual reported in other experimental models (Macneil et al., 1992). The findings of the present study further support the hypothesis that PDN activity is essential for modulating detrusor contraction and EUS function, both of which are vital for maintaining BC and coordinated voiding (McGee and Grill, 2016). Loss of the MBPDN input disrupts EUS control, resulting in increased bladder stiffness and pressure (Hannan et al., 2017). The BC showed substantial improvement that increased significantly in group B, reaching the values of the normal control group D at the 16th week PO (see Table 1). This improvement indicates that direct end-to-end nerve coaptation effectively restores partial bladder–sphincter coordination and improves detrusor adaptability during bladder filling. These outcomes are consistent with earlier studies reporting that the re-establishing of peripheral nerve continuity reinstates afferent signaling and suppresses maladaptive detrusor overactivity, ultimately improving BC (Seif et al., 2005; Jiang et al., 2018). The BC showed remarkable recovery in group C, which matched the values of the normal control group D (Table 1), indicating near-complete restoration of the detrusor–sphincter coordination. The superior recovery in this group can be attributed to the strong anti-inflammatory and neuroprotective effects of the omental pedicle, which play a pivotal role in promoting peripheral nerve regeneration and functional target muscle restoration (Castañeda and Kinne, 2002; Siemionow and Brzezicki, 2009). Following nerve injury, an acute inflammatory response is required to clear the debris and initiate regeneration; if the inflammation persists, perineural edema, fibrosis, and neuronal apoptosis may occur, thereby hindering axonal regrowth and neuromuscular recovery (Mietto et al., 2015; Gu et al., 2024). The omentum mitigates this pathological process by releasing a wide array of bioactive mediators that suppress inflammation and create a pro-regenerative microenvironment. Among these bioactive mediators, interleukin-10 (IL-10), transforming growth factor-β (TGF-β), and adiponectin are particularly important because they inhibit the release of proinflammatory cytokines such as tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), and IL-6 (Ishigaki et al., 2021). In addition, the stromal compartment of the omentum contains immunomodulatory macrophages and mesenchymal-like stem cells that drive macrophage polarization toward the M2 phenotype (Ferrero et al., 2024). This shift is essential because M2 macrophages secrete anti-inflammatory factors and growth mediators that promote axonal sprouting, enhance Schwann cell survival, and limit fibroblast-driven scarring at the repair site (Sezginer and Unver, 2024). As a result, the omental pedicle not only attenuates inflammation but also prevents secondary fibrosis and provides a favorable scaffold for regenerating axons (Pinheiro et al., 2019). Interestingly, the early improvement observed in group C contrasts with the findings of Damaser et al. (2007), who reported that the BC recovery following the PDN crush injury required up to 6 weeks, highlighting the superior regenerative effect of the omental pedicle transposition model used in the present study. LPPLPP is a key indicator of the competence and ability of the EUS to maintain continence during the bladder filling phase. The LPP gradually declined in group A (Table 1). This continuous decline reflects a worsening of the EUS resulting from complete motor branch transection of the PDN and left without treatment. The reduced resistance of the EUS can be attributed to persistent denervation, atrophy of the EUS fibers, and loss of coordinated neuromuscular control. These results are consistent with previous studies that reported decreased external sphincteric pressure and stress incontinence following untreated PDN injury (Weber, 2001; Khorramirouz et al., 2016). The LPP increased gradually in group B (Table 1). This improvement indicates partial restoration of the EUS function after direct end-to-end nerve coaptation. This gradual recovery supports the concept that axonal regeneration can reestablish communication with the EUS, thereby improving continence (Zhu et al., 2019). Similar outcomes have been reported in studies demonstrating the functional recovery of the EUS tone following surgical repair of the damaged PDN (Jiang et al., 2018). The LPP showed pronounced improvement in group C, reaching the values of the normal control group D (Table 1). This marked recovery indicates that omental pedicle wrapping enhances not only axonal regeneration but also functional integration of regenerating nerve fibers at the target muscle level (Valerio et al., 2020). The omentum supports this process through several mechanisms. First, it promotes synaptic maturation via the secretion of growth factors, such as BDNF and NGF, which facilitate presynaptic terminal differentiation, vesicle recycling, and acetylcholine release processes that are essential for efficient neuromuscular transmission (Boyd and Gordon, 2003; Mantilla et al., 2023). Second, the omentum improves blood supply to the neuromuscular junctions at the target muscle by stimulating angiogenesis, ensuring adequate oxygenation and energy for active synaptic function (Goldsmith, 1994; Huang et al., 2022; Longden et al., 2023). Third, its antifibrotic cytokines, the HGF and the TGF-β modulators, reduce the scarring at the repair site, creating a favorable environment for axonal growth and synaptic reconnection (Sulaiman et al., 2018; DiNicolantonio et al., 2021). These results align with previous findings highlighting the role of the omentum in accelerating functional recovery following peripheral nerve injuries (Li et al., 2024). ElectromyographyAmplitudeThe EMG amplitude directly reflects the neuromuscular activity level and the degree of functional reinnervation of the EUS. The EMG amplitude declined sharply in group A (Table 2). This pronounced reduction indicates progressive EUS denervation, ongoing motor unit degeneration, and lack of spontaneous reinnervation. These results are consistent with earlier reports demonstrating that untreated PDN neurotmesis leads to irreversible motor unit loss, impaired EUS contractility, and severe UI (Maher et al., 2024). Although some studies have indicated the possibility of collateral sprouting from nearby nerves (Jeon et al., 2024), the minimal residual EMG amplitude in this group provides little evidence of such compensatory mechanisms. The results at the 16th postoperative week further support the outcomes of a study by Rosato and Oliveira (2020), who reported that EMG amplitude remains reduced for weeks following PDN injury, indicating persistent neuromuscular disruption. The EMG amplitude increased gradually in group B, reaching the values of normal control group D at 16th week PO (see Table 2). This improvement reflects the partial functional recovery achieved through direct end-to-end nerve coaptation, which facilitates axonal regrowth and reconnection with EUS muscle fibers. These results are consistent with those of previous studies demonstrating that mechanical approximation through end-to-end nerve coaptation provides a stable pathway for axonal regeneration and promotes the progressive restoration of motor unit activity (Liao et al., 2009). The EMG amplitude showed the highest improvement in group C, matching the values of normal control group D (Table 2). This strong and early recovery indicates the successful structural reinnervation of the EUS and the effective functional integration between the regenerated nerve and the EUS. The omentum appears to enhance peripheral nerve regeneration and target muscle recovery through several mechanisms. Its natural adhesiveness and angiogenic properties accelerate axonal growth, minimize denervation-related damage, and help preserve the neuromuscular junction structure (Goldsmith, 2012; Studenova, 2021). Additionally, it maintains target muscle readiness for reinnervation by improving blood flow, reducing fibrosis, and providing the anti-inflammatory cytokines (IL-10, TGF-β) along with the trophic factors such as the insulin-like growth factor-1 (IGF-1), the HGF, the CNTF, and the neuregulin-1 from the omentum’s mesenchymal stem cells (MSCs), all of which reduce atrophy and support the satellite cell activity (Gama et al., 2020; Waters et al., 2024). Omental MSCs also enhance Schwann cell activity, guiding axonal regrowth and improving myelination, while their extracellular matrix promotes proper synaptic alignment (Muppirala et al., 2021; Qu et al., 2022). This synergistic environment enhances conduction, prevents aberrant sprouting, and refines motor unit coordination, leading to more efficient target muscle activation (Goitz and Steichen, 2005). Furthermore, the omental pedicle establishes a local trophic feedback loop between the peripheral nerve, target muscle, and itself, thereby stabilizing the synapses and sustaining functional recovery. Mechanically, the omentum’s thin, living tissue cuff protects the regenerating nerve fibers from scar tissue formation and traction-related injuries, thereby supporting long-term nerve regeneration (Zhang et al., 2011; Fay et al., 2021). Some researchers have raised concerns that omental pedicle wrapping could lead to excessive angiogenesis or unpredictable tissue remodeling (Kuniyoshi et al., 2004; Zhou et al., 2006; Shao et al., 2008). In this study, the EMG amplitude remained within the normal physiological ranges at both 4 and 16 weeks PO, indicating that omental pedicle transposition did not lead to pathological remodeling. Instead, it effectively supported functional recovery and preserved neuromuscular integrity. LimitationsThis study has several limitations that should be acknowledged. First, although the histopathological examination of the nerve and muscle tissues was performed using hematoxylin and eosin and Luxol Fast Blue stains, these findings were not included in the present manuscript. The primary objective of this study was to evaluate functional recovery following PDN injury. Except in selected cases such as suspected neoplasia, rare inflammatory conditions, or atypical neuromuscular disorders, histopathological assessment is not routinely required for the diagnosis or classification of UI. Functional investigations, particularly cystometry and electromyography, are the most clinically relevant tools for assessing UI and determining its underlying mechanism (Aanestad and Flink, 1999; Krhut et al., 2018; Yao and Simoes, 2020). The follow-up period was limited to 16 weeks. Although longer observation periods may provide additional information on long-term neural remodeling, this duration was sufficient to capture the major functional changes associated with nerve regeneration. Notably, animals in the treatment group C demonstrated measurable functional improvement as early as week 4, with near-normal functional parameters observed by week 16, comparable to those in the normal control group D. Third, nerve repair was performed immediately after transection, which may not fully replicate delayed or chronic clinical presentations. However, immediate repair allowed for standardized injury conditions and controlled evaluation of regenerative outcomes within the practical and temporal constraints of a master’s thesis project. Despite these limitations, this study provides valid and reliable functional evidence regarding the effectiveness of the applied nerve repair strategies. ConclusionOmental pedicle transposition may contribute to functional recovery after peripheral nerve injury, a finding that has been attributed to its rich vascularization and the presence of trophic and angiogenic factors. In this study, the cystometric and EMG assessments of group C at the 4th postoperative week revealed clear functional improvement, with values approaching the normal ranges. At 16th weeks PO, these functional parameters were comparable to those recorded in healthy control animals, indicating a substantial recovery of lower urinary tract function and a return toward near-physiological performance. AcknowledgmentsThe authors would like to express sincere thanks to the College of Veterinary Medicine/University of Baghdad for their assistance in conducting this research. Conflicts of interestThe authors that they have no conflicts of interest to declare. FundingThe authors received no financial support for the research, authorship, and/or publication of this article. Authors’ contributionsProf. Dr. Hameed. A. Al-Timmemi conceived and designed the study. Rania Kh. Kh. Al-Juboury performed the experiments, collected the data, conducted the statistical analysis, and wrote the initial draft of the manuscript. Both authors critically revised the manuscript and approved the final version. Data availabilityThe data that support the findings of this study are available from the corresponding author upon reasonable request. ReferencesAanestad, Ø. and Flink, R. 1999. Urinary stress incontinence. A urodynamic and quantitative electromyographic study of the perineal muscles. Acta Obstet. Gynecol. Scand. 78(3), 245–253. Beskid, G., Unowsky, J., Behl, C.R., Siebelist, J.A., Tossounian, J.L., Mcgarry, C.M., Shah, N.H. and Cleeland, R. 1988. Enteral, oral, and rectal absorption of ceftriaxone using glyceride enhancers. Chemotherapy 34(2), 77–84; doi:10.1159/000238551 Boyd, J.G. and Gordon, T. 2003. Neurotrophic factors and their receptors in axonal regeneration and functional recovery after peripheral nerve injury. J. Neurobiol. 58(1), 23–46; doi:10.1385/MN:27:3:277 Castañeda, F. and Kinne, R.K. 2002. Omental graft improves functional recovery of transected peripheral nerve. Muscle Nerve 26(4), 527–532; doi:10.1002/mus.10229 Chapple, C.R., Hillary, C.J., Patel, A. and MacDiarmid, S.A. 2018. Urodynamics made easy e-book. Amsterdam: Elsevier Health Sciences. Charan, J. and Kantharia, N. 2013. How to calculate sample size in animal studies. J. Pharmacol. Pharmacother. 4(4), 303–306; doi:10.4103/0976-500X.119726 Chen, S. and Quesenberry, K.E. 2006. Rabbits. In Birchard, S.J. and Sherding, R.G. (eds), Saunders Manual of Small Animal Practice, Philadelphia: Elsevier, pp: 1858–80. HYPERLINK “https://doi.org/10.1111/rda.14466” doi: 10.1111/rda.14466. Damaser, M.S., Samplaski, M.K., Parikh, M., Lin, D.L., Rao, S. and Kerns, J.M. 2007. Time course of neuroanatomical and functional recovery after bilateral pudendal nerve injury in female rats. Am. J. Physiol. Renal. Physiol. 293(5), F1614–F1621; doi:10.1152/ajprenal.00176.2007 Dinicolantonio, J.J., Mccarty, M.F., Barroso-Aranda, J., Assanga, S., Lujan, L.M.L. and O'Keefe, J.H. 2021. A nutraceutical strategy for downregulating TGFβ signalling: prospects for prevention of fibrotic disorders, including post-COVID-19 pulmonary fibrosis. Open. Heart. 8(1), 1663; doi:10.1136/openhrt-2021-001663 Dujovny, M., Ding, Y.H., Ding, Y., Agner, C. and Perez-Arjona, E. 2004. Current concepts on the expression of neurotrophins in the greater omentum. Neurol. Res. 26(2), 226–229; doi:10.1179/016164104225013879 El Homsi, M., Zadeh, C., Charbel, C., Alsheikh Deeb, I., Gharzeddine, K., Rebeiz, K., Hourani, R., Khoury, N. and Moukaddam, H. 2024. Neurologic pathologies of the vertebral spine. Skelet. Radiol. 53(3), 419–436; doi:10.1007/s00256-023-04428-y Fay, L.Y., Lin, Y.R., Liou, D.Y., Chiu, C.W., Yeh, M.Y., Huang, W.C., Wu, J.C., Tsai, M.J. and Cheng, H. 2021. The application of an omentum graft or flap in spinal cord injury. Int. J. Mol. Sci. 22(15), 7930; doi:10.3390/ijms22157930 Ferrero, R., Rainer, P.Y., Rumpler, M., Russeil, J., Zachara, M., Pezoldt, J., Van Mierlo, G., Gardeux, V., Saelens, W., Alpern, D., Favre, L., Vionnet, N., Mantziari, S., Zingg, T., Pitteloud, N., Suter, M., Matter, M., Schlaudraff, K.U., Canto, C. and Deplancke, B. 2024. A human omentum-specific mesothelial-like stromal population inhibits adipogenesis through IGFBP2 secretion. Cell. Metab. 36(7), 1566–1585; doi:10.1016/j.cmet.2024.04.017 Flecknell, P., Lofgren, J.L., Dyson, M.C., Marini, R.R., Swindle, M.M. and Wilson, R.P. 2015. Preanesthesia, anesthesia, analgesia, and euthanasia. In Fox, J.G., Anderson, L.C., Otto, G.M., Pritchett-Corning, K.R. and Whary, M.T. (eds), Laboratory Animal Medicine, London, UK: Academic Press, pp: 1135–200. Furuta, A., Kita, M., Suzuki, Y., Egawa, S., Chancellor, M.B., De Groat, W.C. and Yoshimura, N. 2008. Association of overactive bladder and stress urinary incontinence in rats with pudendal nerve ligation injury. Am. J. Physiol. Regul. Integr. Comp. Physiol. 294(5), R1510–R1516; doi:10.1152/ajpregu.00838.2007 Gama, J.F.G., Pinheiro, D.F., Da Silva, R.F., Quirico-Santos, T. and Lagrota-Candido, J. 2020. The omentum harbors unique conditions in the peritoneal cavity to promote healing and regeneration for diaphragm muscle repair in mdx mice. Cell. Tissue. Res. 382, 447–455; doi:10.1007/s00441-020-03238-1 Giulioni, C., Pitoni, L., Fuligni, D., Beltrami, M., Passarella, V., Palantrani, V., De Stefano, V., Castellani, D. and Galosi, A.B. 2024. Pudendal nerve neurolysis outcomes for urogenital and rectal disorders in patients suffering from pudendal nerve entrapment: a systematic review. Investig. Clin. Urol. 65(3), 230; doi:10.4111/icu.20230402 Goitz, R.J. and Steichen, J.B. 2005. Microvascular omental transfer for the treatment of severe recurrent median neuritis of the wrist: a long-term follow-up. Plast. Reconstr. Surg. 115(1), 163–171. Goldsmith HS, ed. 2012. The omentum: research and clinical applications. New York: Springer Science & Business Media. doi:10.22541/au.172805072.21222449/v1 Goldsmith, H.S. 1994. Brain and spinal cord revascularization by omental transposition. Neurol. Res. 16(3), 159–162; doi:10.1080/01616412.1994.11740218 Gray, M. 2011. Traces: making sense of urodynamics testing—part 3: electromyography of the pelvic floor muscles. Urol. Nurs. 31(1), 31–38. Gu, D., Xia, Y., Ding, Z., Qian, J., Gu, X., Bai, H., Jiang, M. and Yao, D. 2024. Inflammation in the peripheral nervous system after injury. Biomedicines 12(6), 1256; doi:10.3390/biomedicines12061256 Guo, H., Chen, P., Luo, R., Zhang, Y., Xu, X. and Gou, X. 2022. The roles of ciliary neurotrophic factor—from neuronutrition to energy metabolism. Protein. Pept. Lett. 29(10), 815–828; doi:10.2174/0929866529666220905105800 Hannan, J.L., Powers, S.A., Wang, V.M., Castiglione, F., Hedlund, P. and Bivalacqua, T.J. 2017. Impaired contraction and decreased detrusor innervation in a female rat model of pelvic neuropraxia. Int. Urogynecol. J. 28(7), 1049–1056; doi:10.1007/s00192-016-3223-1 Hannestad, Y.S., Rortveit, G., Sandvik, H. and Hunskaar, S. 2000. A community-based epidemiological survey of female urinary incontinence: the Norwegian EPINCONT study. J. Clin. Epidemiol. 53, 1150–1157; doi:10.1016/s0895-4356(00)00232-8 Herman, Z.J. and Ilyas, A.M. 2020. Sensory outcomes in digital nerve repair techniques: an updated meta-analysis and systematic review. Hand 15(2), 157–164; doi:10.1177/1558944719844346 Herroelen, S., Knowles, C., Kerrigan-Smith, J., Noone, T., Denison, T. and De Wachter, S. 2024. Pudendal nerve stimulation for treatment of lower urinary tract symptoms: a systematic review of safety, technical feasibility and clinical efficacy. Continence 11, 101685; doi:10.1016/j.cont.2024.101685 Huang, X., Jiang, J. and Xu, J. 2022. Denervation-related neuromuscular junction changes: from degeneration to regeneration. Front. Mol. Neurosci. 14, 810919; doi:10.3389/fnmol.2021.810919 Ishigaki, K., Kumano, K., Fujita, K. and Ueno, H. 2021. Cellular basis of omentum activation and expansion revealed by single-cell RNA sequencing using a parabiosis model. Sci. Rep. 11(1), 13958; doi:10.1038/s41598-021-93330-5 Jeon, S.M., Pradeep, A., Chang, D., McDonough, L., Chen, Y., Latremoliere, A., Crawford, L.K. and Caterina, M.J. 2024. Skin reinnervation by collateral sprouting following spared nerve injury in mice. J. Neurosci. 44(15), e1494232024; doi:10.1523/JNEUROSCI.1494-23.2024 Jiang, H.H., Song, Q.X., Gill, B.C., Balog, B.M., Juarez, R., Cruz, Y. and Damaser, M.S. 2018. Electrical stimulation of the pudendal nerve promotes neuroregeneration and functional recovery from stress urinary incontinence in a rat model. Am. J. Physiol. Renal. Physiol. 315(6), F1555–F1564; doi:10.1152/ajprenal.00431.2017 Kambe, M., Hashikawa, K., Takanari, K., Yagi, S.,Toriyama, K., Ebisawa, K., Nishio, N., Maruo, T., Mukoyama, N., Fujimoto, Y., Fujii, M., Saito, K.,Takahashi, M. and Kamei, Y. 2023. Long-term outcomes of lateral skull base reconstruction with a free omental flap and facial nerve reconstruction. Nagoya J. Med. Sci. 85(2), 255–264, doi:10.18999/nagjms.85.2.255 Kerns, J.M., Damaser, M.S., Kane, J.M., Sakamoto, K., Benson, J.T., Shott, S. and Brubaker, L. 2000. Effects of pudendal nerve injury in the female rat. Neurourol. Urodyn. 19(1), 53–69. Khorramirouz, R., Mozafarpour, S., Kameli, S.M., LadiSeyedian, S.S., Oveisi, N., Rahimi, Z., Alijani, M. and Kajbafzadeh, A.-M. 2016. A novel method of urinary sphincter deficiency: serial histopathology evaluation in a rat model of urinary incontinence. Anat. Rec. 299(2), 173–180; doi:10.1002/ar.23291 Kotliarova, A. and Sidorova, Y.A. 2021. Glial cell line-derived neurotrophic factor family ligands, players at the interface of neuroinflammation and neuroprotection: focus onto the glia. Front. Cell. Neurosci. 15, 679034; doi:10.3389/fncel.2021.679034 Krhut, J., Zachoval, R., Rosier, P.F., Shelly, B. and Zvara, P. 2018. ICS educational module: electromyography in the assessment and therapy of lower urinary tract dysfunction in adults. Neurourol. Urodyn. 37(1), 27–32; doi:10.1002/nau.23278 Kuniyoshi, Y., Koja, K., Miyagi, K., Arakaki, K. and Nagano, T. 2004. Aortic arch graft wrapping with pedicled omentum: angiographic assessment of the omentum. EJVES. Extra. 8(1), 7–9; doi:10.1016/j.ejvsextra.2004.05.007 Leal, G., Bramham, C.R. and Duarte, C.B. 2017. BDNF and hippocampal synaptic plasticity. Vitamins. Horm. 104, 153–195; doi:10.1016/bs.vh.2016.10.004 Li, X., Ding, Y., Haddad, Y.W. and Geng, X. 2024. Greater omentum: multifaceted interactions in neurological recovery and disease progression. Aging Dis. 15(6), 2381; doi:10.14336/AD.2024.0213 Liao, W.C., Chen, J.R., Wang, Y.J. and Tseng, G.F. 2009. The efficacy of end-to-end and end-to-side nerve repair (neurorrhaphy) in the rat brachial plexus. J. Anat. 215(5), 506–521; doi:10.1111/j.1469-7580.2009.01135.x Longden, T., Weir, N., Xiang, L., Garcia, D., Qadir, H., Patton, M., Mathur, B. and Dabertrand, F. 2023. Vascular signaling plasticity reprograms neurovascular coupling pathways to precisely match energy delivery to neuronal metabolic needs. Physiology 38(S1), 5731521; doi:10.1016/bs.pbr.2016.02.002 Ma, F., Wang, H., Yang, X., Wu, Y., Liao, C., Xie, B., Li, Y. and Zhang, W. 2022. Controlled release of ciliary neurotrophic factor from bioactive nerve grafts promotes nerve regeneration in rats with facial nerve injuries. J. Biomed. Mater. Res. A 110(4), 788–796; doi:10.1002/jbm.a.37327 Macneil, H.F., Brading, A.F. and Williams, J.H. 1992. Cause of low compliance in a guinea-pig model of instability and low compliance. Neurourol. Urodyn. 11(1), 47–52; doi:10.1002/nau.1930110106 Maher, S., Gerber, D., Balog, B., Wang, L., Kuang, M., Hanzlicek, B., Malakalapalli, T., Van Etten, C., Khouri, R. and Damaser, M.S. 2024. Contribution of pudendal nerve injury to stress urinary incontinence in a male rat model. Sci. Rep. 14(1), 7444; doi:10.1038/s41598-024-57493-1 Mantilla, C.B., Ermilov, L.G., Greising, S.M., Gransee, H.M., Zhan, W.Z. and Sieck, G.C. 2023. Electrophysiological effects of BDNF and TrkB signaling at type-identified diaphragm neuromuscular junctions. J. Neurophysiol. 129(4), 781–792; doi:10.1152/jn.00015.2023 Mccloskey, K.D., Kanai, A., Panicker, J.N., Hashitani, H. and Fry, C.H. 2024. What do we really know about the external urethral sphincter. Continence 10, 101223; doi:10.1016/j.cont.2024.101223 Mcgee, M.J. and Grill, W.M. 2016. Modeling the spinal pudendo-vesical reflex for bladder control by pudendal afferent stimulation. J. Comput. Neurosci. 40(3), 283–296; doi:10.1007/s10827-016-0597-5 Mietto, B.S., Kroner, A., Girolami, E.I., Santos-Nogueira, E., Zhang, J. and David, S. 2015. Role of IL-10 in resolution of inflammation and functional recovery after peripheral nerve injury. J. Neurosci. 35(50), 16431–16442; doi:10.1523/JNEUROSCI.2119-15.2015 Mohammadi, R., Hirsaee, M.A. and Amini, K. 2013. Improvement of functional recovery of transected peripheral nerve by means of artery grafts filled with diclofenac. Int. J. Surg. 11(3), 259–264; doi:10.1016/j.ijsu.2013.01.008 Morawska-Kozłowska, M., Wilkosz, A. and Zhalniarovich, Y. 2024. The omentum—A forgotten structure in veterinary surgery in small animals’ surgery. Animals 14(13), 1848; doi:10.3390/ani14131848 Muppirala, A.N., Limbach, L.E., Bradford, E.F. and Petersen, S.C. 2021. Schwann cell development: from neural crest to myelin sheath. Wiley. Interdiscip. Rev. Dev. Biol. 10(5), e398; doi:10.1002/wdev.398 Pinheiro, D.F., Da Silva, R.F., Barbosa, T.M., Gama, J.F.G., Gomes, A.C., Quirico-Santos, T. and Lagrota-Candido, J. 2019. Omentum acts as a regulatory organ controlling skeletal muscle repair of mdx mice diaphragm. Cell Tissue Res. 377, 269–279; doi:10.1007/s00441-019-03012-y Portillo, F.J.M., Osmonov, D.K., Seif, C., Braun, P.M., Boehler, G., Alken, P. and Juenemann, K.P. 2004. Restoration of external urethral sphincter function after pudendal nerve end-to-end anastomosis in the male rabbit. J. Urol. 171(4), 1715–1719; doi:10.1097/01.ju.0000116124.58457.b4 Qu, Q., Liu, L., Cui, Y., Chen, Y., Wang, Y. and Wang, Y. 2022. Exosomes from human omental adipose-derived mesenchymal stem cells secreted into ascites promote peritoneal metastasis of epithelial ovarian cancer. Cells 11(21), 3392; doi:10.3390/cells11213392 Rosato, G.O. and Oliveira, L.C.C. 2020. Electromyography and pudendal nerve terminal motor latency. In Oliveira, L.C.C. (ed.). Anorectal physiology: a clinical and surgical perspective. Springer International Publishing, 2457, pp: 165–74. https://doi.org/ 10.1016/s1388-2457(01)00464-3 Sakamoto, K., Smith, G.M., Storer, P.D., Jones, K.J. and Damaser, M.S. 2000. Neuroregeneration and voiding behavior patterns after pudendal nerve crush in female rats. Neurourol. Urodyn. 19(3), 311–321. SAS Institute Inc. 2021. JMP Pro, Version 16 [Computer software]. Cary, NC: SAS Institute Inc. Seif, C., Van Der Horst, C., Naumann, C.M., Jünemann, K.P., Bosch, R., Buller, J. and Braun, P.M. 2005. Chronische Pudendale Neurostimulation zur Therapie der überaktiven Blase—eine Alternative zur sakralen Neuromodulation?. Aktuelle. Urol. 36(03), 234–238; doi:10.1055/s-2004-830204 Sezginer, O. and Unver, N. 2024. Dissection of pro-tumoral macrophage subtypes and immunosuppressive cells participating in M2 polarization. Inflamm. Res. 73(9), 1411–1423; doi:10.1007/s00011-024-01907-3 Shafik, A. 1992. The cutaneo-urethral reflex. Description of the reflex and its role in stress urinary incontinence. Urologia. J. 59(2), 50–55; doi:10.1038/sc.1992.57 Shahraki, A.R., Abaee, R. and Shahraki, E. 2024. Omentum a powerful viable organ in patient with liver crash trauma: a case series. East. Afr. Sch. J. Med. Surg. 6(2), 64–66; doi:10.36349/easjms.2024.v06i02.010 Shao, Z.Q., Kawasuji, M., Takaji, K., Katayama, Y. and Matsukawa, M. 2008. Therapeutic angiogenesis with autologous hepatic tissue implantation and omental wrapping. Circ. J. 72(11), 1894–1899; doi:10.1253/circj.cj-08-0167 Siemionow, M. and Brzezicki, G. 2009. Current techniques and concepts in peripheral nerve repair. Int. Rev. Neurobiol. 87, 141–172; doi:10.1016/S0074-7742(09)87008-6 Solinas, S.M.G., Edelmann, E., Leßmann, V. and Migliore, M. 2019. A kinetic model for brain-derived neurotrophic factor mediated spike timing-dependent LTP. PLos Comput. Biol. 15(4), e1006975; doi:10.1371/journal.pcbi.1006975 Steward, J.E., Clemons, J.D., Zaszczurynski, P.J., Butler, R.S., Damaser, M.S. and Jiang, H.H. 2010. Quantitative evaluation of electrodes for external urethral sphincter electromyography during bladder-to-urethral guarding reflex. World. J. Urol. 28(3), 365–371; doi:10.1007/s00345-009-0463-4 Studenova, E.A. 2021. Is a big omentum the surgeon’s best friend or worst enemy? Review. Surg. Pract. (Rus). 4, 15–22; doi:10.38181/2223-2427-2021-4-15-22 Sulaiman, W., Dreesen, T. and Nguyen, D. 2018. Single local application of TGF-β promotes a proregenerative state throughout a chronically injured nerve. Neurosurgery 82(6), 894–902; doi:10.1093/neuros/nyx362 Tannemaat, M.R., Boer, G.J., Eggers, R., Malessy, M.J. and Verhaagen, J. 2009. From microsurgery to nanosurgery: how viral vectors may help repair the peripheral nerve. Prog. Brain. Res. 175, 173–186; doi:10.1016/S0079-6123(09)17512-X Valerio, I., Schulz, S.A., West, J., Westenberg, R.F. and Eberlin, K.R. 2020. Targeted muscle reinnervation combined with a vascularized pedicled regenerative peripheral nerve interface. Plast. Reconstr. Surg. Glob. Open. 8(3), 2689; doi:10.1097/GOX.0000000000002689 Wang, C., Chen, Y., Chen, X., Hu, C., Chen, J. and Guo, A. 2023. Evaluation of antiviral activity of ivermectin against infectious bovine rhinotracheitis virus in rabbit model. Animals 13(20), 3164; doi:10.3390/ani13203164 Waters, J.A., Robinson, M., Lujano-Olazaba, O., Lucht, C., Gilbert, S.F. and House, C.D. 2024. Omental preadipocytes stimulate matrix remodeling and IGF signaling to support ovarian cancer metastasis. Cancer Res. 84(13), 2073–2089; doi:10.1158/0008-5472.CAN-23-2613 Weber, A.M. 2001. Leak point pressure measurement and stress urinary incontinence. Curr. Womens. Health Rep. 1(1), 45–52. Yang, X., Wang, X., Gao, Z., Li, L., Lin, H., Wang, H., Zhou, H., Tian, D., Zhang, Q. and Shen, J. 2022. The anatomical pathogenesis of stress urinary incontinence in women. Medicina 59(1), 5; doi:10.3390/medicina59010005 Yang, Y. and Chen, L. 2024. Omentin-1 attenuates lipopolysaccharide-induced inflammation and osteogenic differentiation in periodontal ligament stem cells and reduces M1 macrophages polarization through repressing endoplasmic reticulum stress. Prostaglandins Other Lipid Mediat. 174, 106882; doi:10.1016/j.prostaglandins.2024.106882 Yao, M. and Simoes, A. 2020. Urodynamic testing and interpretation. Treasure Island (FL), USA: StatPearls Publishing. Zabbia, G., Toia, F., Coppola, F., Cassata, G., Cicero, L., Giglia, G., Puleio, R. and Cordova, A. 2024. Nerve regeneration after a nerve graft in a rat model: the effectiveness of fibrin glue. J. Pers. Med. 14(5), 445; doi:10.3390/jpm14050445 Zapletal, J., Nanka, O., Halaska, M.J., Maxova, K., Hajkova Hympanova, L., Krofta, L. and Rob, L. 2024. Anatomy of the pudendal nerve in clinically important areas: a pictorial essay and narrative review. Surg. Radiol. Anat. 46(2), 211–222; doi:10.1007/s00276-023-03285-7 Zhang, Y.G., Huang, J.H., Hu, X.Y., Sheng, Q.S., Zhao, W. and Luo, Z.J. 2011. Omentum-wrapped scaffold with longitudinally oriented micro-channels promotes axonal regeneration and motor functional recovery in rats. PLos One 6(12), e29184; doi:10.1371/journal.pone.0029184 Zhou, J., Tang, Z. and Xiao, Y. 2006. An effect of the omental wrapping on revascularization and nerve regeneration of the artificial nerve graft. Zhongguo. Xiufu. Chongjian. Waike. Zazhi. 20(8), 797–800. Zhu, L., Zhou, Z.B., Shen, D. and Chen, A.M. 2019. Ipsilateral S2 nerve root transfer to pudendal nerve for restoration of external anal and urethral sphincter function: an anatomical study. Sci. Rep. 9(1), 13993; doi:10.1038/s41598-019-50484-7 Zorman, P., Kovačič, U., Sketelj, J. and Bajrović, F.F. 2007. Ingrowth of sensory axons into an end-to-side coapted nerve stump after donor nerve crush in the rat. In Millesi, H. and Schmidhammer, R. (eds), How to Improve the Results of Peripheral Nerve Surgery, Vienna & New York, NY: Springer, pp: 85–87. doi: 10.1007/978-3-211-72958-8_18 | ||
| How to Cite this Article |
| Pubmed Style Al-juboury RKK, Al-timmemi HA. Omental pedicle transposition for neuropathic stress urinary incontinence: A urodynamic evaluation. Open Vet. J.. 2026; 16(2): 1243-1258. doi:10.5455/OVJ.2026.v16.i2.42 Web Style Al-juboury RKK, Al-timmemi HA. Omental pedicle transposition for neuropathic stress urinary incontinence: A urodynamic evaluation. https://www.openveterinaryjournal.com/?mno=294246 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.42 AMA (American Medical Association) Style Al-juboury RKK, Al-timmemi HA. Omental pedicle transposition for neuropathic stress urinary incontinence: A urodynamic evaluation. Open Vet. J.. 2026; 16(2): 1243-1258. doi:10.5455/OVJ.2026.v16.i2.42 Vancouver/ICMJE Style Al-juboury RKK, Al-timmemi HA. Omental pedicle transposition for neuropathic stress urinary incontinence: A urodynamic evaluation. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 1243-1258. doi:10.5455/OVJ.2026.v16.i2.42 Harvard Style Al-juboury, R. K. K. & Al-timmemi, . H. A. (2026) Omental pedicle transposition for neuropathic stress urinary incontinence: A urodynamic evaluation. Open Vet. J., 16 (2), 1243-1258. doi:10.5455/OVJ.2026.v16.i2.42 Turabian Style Al-juboury, Rania Kh. Kh., and Hameed. A. Al-timmemi. 2026. Omental pedicle transposition for neuropathic stress urinary incontinence: A urodynamic evaluation. Open Veterinary Journal, 16 (2), 1243-1258. doi:10.5455/OVJ.2026.v16.i2.42 Chicago Style Al-juboury, Rania Kh. Kh., and Hameed. A. Al-timmemi. "Omental pedicle transposition for neuropathic stress urinary incontinence: A urodynamic evaluation." Open Veterinary Journal 16 (2026), 1243-1258. doi:10.5455/OVJ.2026.v16.i2.42 MLA (The Modern Language Association) Style Al-juboury, Rania Kh. Kh., and Hameed. A. Al-timmemi. "Omental pedicle transposition for neuropathic stress urinary incontinence: A urodynamic evaluation." Open Veterinary Journal 16.2 (2026), 1243-1258. Print. doi:10.5455/OVJ.2026.v16.i2.42 APA (American Psychological Association) Style Al-juboury, R. K. K. & Al-timmemi, . H. A. (2026) Omental pedicle transposition for neuropathic stress urinary incontinence: A urodynamic evaluation. Open Veterinary Journal, 16 (2), 1243-1258. doi:10.5455/OVJ.2026.v16.i2.42 |