| Research Article | ||
Open Vet. J.. 2026; 16(2): 1222-1229
Open Veterinary Journal, (2026), Vol. 16(2): 1222-1229 Research Article Role of propolis and rosemary extract in improving mitochondrial efficiency and energy metabolism enzymes in late pregnancy Awassi sheep liverFaisal Ali Lattef1*, Mahaba Salah Mahdi Hassan Al_zwelef2, Mohammed T. Jaafar3, Zainab Fadhil Abdulhussein4, and Ghadir Kamil Ghadir51Department of Medical Laboratory Techniques, College of Health and Medical Techniques, Al-Bayan University, Baghdad, Iraq 2AL-Naji University, College of Medical and Health Technologies, Department of Anesthesia Techniques, Baghdad, Iraq 3Department of Physiology, Biochemistry, University of Wasit, Iraq 4Department of Biology, Al-Turath University, Baghdad, Iraq 5College of Pharmacy, Al-Farahidi University, Baghdad, Iraq *Corresponding Author: Faisal Ali Lattef. Department of Medical Laboratory Techniques, College of Health and Medical Techniques, Al-Bayan University, Baghdad, Iraq. Email: Faisal.ali [at] albayan.edu.iq Submitted: 02/11/2025 Revised: 11/01/2026 Accepted: 22/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
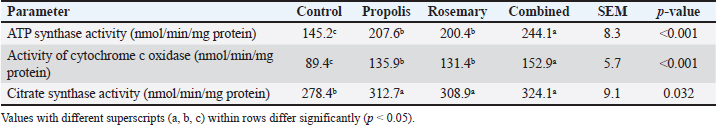
AbstractBackground: The final stages of gestation in ewes are periods with very high energy requirements, with the energy needed to maintain homeostasis rising by 50%–75% compared to non-pregnancy conditions. However, mitochondrial activity can be reduced by oxidative stress induced in late pregnancy. Aim: This study aimed to evaluate the effects of propolis and rosemary extract on mitochondrial enzyme activity, energy metabolism, and oxidative stress in late-pregnant Awassi ewes. Methods: The study sample comprised 24 multiparous Awassi ewes in the late stages of pregnancy (140 ± 5 days of gestation). Animals were divided into four groups (Group I: control, Group II: propolis (5 g/day), Group III: rosemary extract (1.2 g/head/day), and Group IV: combined propolis + rosemary treatment) using a completely randomized design. The activities of Adenosine Triphosphate (ATP) synthase and cytochrome c oxidase were analyzed in liver biopsies using spectrophotometric methods. Malondialdehyde (MDA), glutathione, superoxide dismutase (SOD), Catalase (CAT), and Glutathione Peroxidase (GPx) levels were analyzed using serum samples. Results: Propolis and rosemary extract, as well as their combined use, had a significant effect on the activity of adenosine triphosphate synthase and cytochrome c oxidase enzymes in the mitochondria of liver cells when compared to the control group. The activities of ATP synthase were increased by 43% and 38%, respectively, and the values of cytochrome c oxidase activity in the same groups were increased by 52%, 47%, and 71%, respectively (p < 0.05). The levels of ATP in the liver of pregnant ewes supplemented with propolis, rosemary extract, and both were 34%, 38%, and 41% higher than those in the control group (p < 0.05). The MDA content in the liver of ewes receiving propolis, rosemary extract, and the combination of both was 31%, 35%, and 45% lower, respectively, than that in the control group (p < 0.05). The activities of the antioxidant enzymes SOD, CAT, and GPx in the liver of pregnant ewes in the three groups were higher than those in the control group and increased by 28%–52%. Conclusion: Propolis and rosemary extracts play an important role in regulating oxidative stress and increasing energy metabolism in the liver mitochondria of pregnant ewes during late pregnancy. Keywords: ATP synthase, Bee glue, Oxidative stress, Pregnancy,Saliva rosmarinus extract. IntroductionLate pregnancy is a period of negative energy balance in sheep, which is associated with increased fetal demands and a high energy intake requirement. The fetal liver is the primary site for gluconeogenesis and lipid metabolism in late pregnancy and is vulnerable to oxidative stress that would affect ewe productivity (Al-Khauzai et al., 2023). Liver mitochondrial functions and oxidative stress markers in late pregnant Awassi sheep were determined in response to propolis and rosemary extract supplementation. Awassi ewes were chosen because of their adaptability to hot and dry climates, low productivity, and high milk production potential Talafha and Ababneh, 2011; Yu et al., 2024). During late pregnancy, pregnant ewes undergo mitochondrial stress with dysregulation of respiratory chain enzymes and increased oxidative stress (Simitzis et al., 2008; Calvin-Pereira L Gonz et al., 2015). Fetal growth and development during late pregnancy are adversely affected in dams under energy deficit, resulting in pregnancy toxemia, fatty liver syndrome, and reduced lamb survival rate (Cal-Pereyra et al., 2015; Al-Khauzai et al., 2023). Late pregnancy is characterized by a 50%–75% increase in maternal metabolic rate, and the fetal liver requires 30–40g of glucose supply per fetus per day (Shedeed et al., 2019). The five mitochondrial enzymes of the respiratory chain (Complexes I–V) are required for oxidative phosphorylation, which provides energy for all mitochondrial functions (Franco et al., 2020). The activity of these enzymes, particularly Adenosine Triphosphate (ATP) synthase (Complex V) and cytochrome c oxidase (Complex IV) enzymes, are the rate-limiting factors for ATP synthesis in the cell (Koopman et al., 2012; Brand et al., 2019). The activity of respiratory chain complexes in the mitochondria of ruminants is known to be adversely affected by late pregnancy and poor nutritional conditions (Paradies et al., 2014; Zhang et al., 2020). Propolis (bee glue) is a resinous mixture and metabolic byproducts collected by honeybees from plant sources. Propolis is a rich source of various polyphenols, including flavonoids (chrysin, pinocembrin, and quercetin), phenolic acids (caffeic acid phenethyl ester), stilbenes, coumarins, and lignans (Habtemariam, 2020; Kurek-Górecka et al., 2022). It improves mitochondrial function through direct stimulation of respiratory chain complexes, mitochondrial membrane stabilization, and antioxidant protection (Cedikova et al., 2014; Tvrdá et al., 2023; Fawaz et al., 2025). Propolis treatment was reported to increase mitochondrial succinate dehydrogenase activity and respiratory control ratio, and it was also found to reduce mitochondrial production of superoxide by 37%–48% (Tvrdá et al., 2023). Rosemary extract (Rosmarinus officinalis) is a rich source of carnosic acid, carnosol, and rosmarinic acid (Naimi et al., 2017; Liu et al., 2022). Carnosic acid reduces the mitochondrial unfolded protein response, a stress response associated with mitochondrial cristae disruption and mitochondrial membrane potential loss (Liu et al., 2022). Rosmarinic acid activates AMP-Activated Protein Kinase (AMPK), resulting in improved glucose consumption in cells (6%–21%) and increased fatty acid oxidation, a mechanism essential to pregnancy energy metabolism (Wang et al., 2013; Naimi et al., 2017). Propolis and rosemary extract have comparable mechanisms of action in the mitochondria; therefore, their combined effects may be enhanced. Both propolis and rosemary extract are antioxidants that may work together to reduce oxidative stress during pregnancy (Shedeed et al., 2019; Belardinelli et al., 2020). Therefore, this study aimed to determine the effects of propolis and rosemary extract supplementation on liver mitochondrial respiratory chain enzymes and oxidative stress markers in late pregnant Awassi sheep. Materials and MethodsAnimals and experimental designThe inclusion criteria were as follows: clinically healthy multiparous Awassi ewes aged 3–5 years, at late gestation (140 ± 5 days), with a body condition score of 2.5–3.5, and free from reproductive or systemic diseases based on clinical examination and veterinary records. The exclusion criteria comprised primiparous ewes. Animals with signs of metabolic disorders (including toxemia or ketosis during pregnancy). The experiment was conducted at the Livestock Research Station under institutional animal care guidelines. Animals were kept individually in pens at the animal house of the college and were provided with ad libitum water and fed a basal diet that supplied 2.1 Mcal/kg ME throughout the experiment. Ewes were randomly assigned to four groups (n=6/treatment) as follows: control group (basal diet), propolis group (basal diet + 5 g/day propolis extract), rosemary group (basal diet + 1.2 g/head/day rosemary extract), and combined group (basal diet + both propolis 5 g/day and rosemary 1.2 g/day extracts). The treatment period was 14 days before parturition (Naimi et al., 2017; Shedeed et al., 2019 Preparation and administration of supplementsThe standardization of propolis extract was performed using high-performance liquid chromatography for the quantification of major phenolic and flavonoid compounds, including chrysin, pinocembrin, and Caffeic Acid Phenethyl Ester. The propolis extract was obtained from Slovak propolis samples (Tvrdá et al., 2023). Concentrations of active compounds were standardized: chrysin (30.56 ± 0.60 mg/g), pinocembrin (30.96 ± 0.34 mg/g), caffeic acid phenethyl ester (15.2 mg/g), and total phenolic content (156.2 mg GAE/g) (Tvrdá et al., 2023). The extraction solvent was 70% ethanol, after which the solvent was removed, and the extract was standardized to 75% active compounds. Rosemary extract was standardized to 10.3% carnosic acid, 10.3% carnosol, and 24.5 mg/g rosmarinic acid (Naimi et al., 2017; Liu et al., 2022). Commercial standardized extract (Kemin Industries) was used to standardize the concentrations of the bioactive compounds. Both supplements were added to 100 g of concentrate feed and given once per day during the morning feeding. Sample collection and processingLiver biopsies were obtained on day 10 of supplementation using an ultrasound-guided percutaneous needle biopsy technique under local anesthesia (2% lidocaine). The samples were immediately processed for mitochondrial isolation or snap-frozen in liquid nitrogen and stored at −80°C until analysis (Bouwman et al., 2019). The biological matrix for measuring oxidative stress and metabolic markers was serum by jugular venipuncture into ethylenediaminetetraacetic acid tubes. The spectrophotometric method was used to assess malondialdehyde (MDA), and Ellman’s method was used to calculate reduced glutathione (GSH). Antioxidant enzyme activities [superoxide dismutase (SOD), Catalase (CAT), and Glutathione Peroxidase (GPx)] were measured using commercial assay kits (Randox Laboratories, UK) according to the manufacturer’s instructions. Mitochondrial isolationFollowing previously established protocols (Brand and Nicholls, 2011): liver tissue (500 mg) was homogenized in ice-cold isolation buffer (250 mM sucrose, 10 mM Tris-HCl, 1 mM EDTA, pH 7.4) using a Potter–Elvehjem homogenizer. Differential centrifugation was then carried out at 800×g for 10 minutes (to remove the nuclear pellet) and at 8,000×g for 15 minutes (to collect the mitochondrial pellet). Then, the mitochondrial pellet was washed twice and resuspended in isolation buffer. The protein concentration was assessed using the Bradford assay. Enzyme activity assaysATP synthase activity was determined using a coupled enzymatic assay by measuring Nicotinamide Adenine Dinucleotide (NADH) oxidation at 340 nm (Berry et al., 2018; Ley-Ngardigal et al., 2022). Reaction medium (100 μl) was composed of 50 mM Tris-HCl (pH 8.0), 5 mM MgCl2, 2.5 mM KCN, 0.25 mM NADH, 2.5 mM ADP, 5 mM glucose, 5 U/ml hexokinase, and 5 U/ml glucose-6-phosphate dehydrogenase. The oligomycin-sensitive activity was calculated from the differences in rates in the presence and absence of oligomycin (2.5 μg/ml). Activity was calculated as nmol ATP synthesized/min/mg protein using the extinction coefficient for NADH (6.22 mM−¹cm−¹). Cytochrome c oxidase activity was measured spectrophotometrically by recording the oxidation of cytochrome c at 550 nm (Brand and Nicholls, 2011; Spinazzi et al., 2012). The reaction medium comprised 50 mM potassium phosphate buffer, pH 7.4, 0.1 mM EDTA, and 50 μM reduced cytochrome C. Reaction rates were measured at 37°C, and activity was calculated using the extinction coefficient of reduced cytochrome c (19.1 mM−¹cm−¹). Cyanide-sensitive activity was measured to confirm enzyme specificity. ATP was quantified using the luciferin-luciferase bioluminescence assay (Sigma-Aldrich) (Büttner et al., 2019; Ley-Ngardigal et al., 2022). Liver tissue was extracted in perchloric acid and neutralized with potassium carbonate. Neutralized extracts were transferred to a 96-well plate, and luminescence was measured using a luminometer. ATP concentrations were determined using standard curves and expressed as μmol/g tissue wet weight. Analysis of oxidative stress markersMDA levels were determined using the thiobarbituric acid reactive substances assay, as previously described (Patlevič et al., 2016; Al-Khauzai et al., 2023). Serum samples were mixed with a thiobarbituric acid reagent and heated at 95°C for 60 minutes. The absorbance was measured at 532 nm. Results are expressed as nmol MDA/ml serum. Reduced GSH concentration was measured using Ellman’s reagent (5,5′-dithiobis-2-nitrobenzoic acid; DTNB) according to the method of Calvin–Pereira L Gonz et al. (2015). Serum samples were deproteinized with 5% sulfosalicylic acid, neutralized with KOH, and reacted with DTNB. Absorbance was recorded at 412 nm. The GSH concentrations were calculated using standard calibration curves and expressed as μmol/l. The antioxidant enzyme activities were determined using commercial assay kits (Randox Laboratories, UK). SOD activity was measured using the xanthine/xanthine oxidase method, with absorbance read at 470 nm. CAT activity was determined by monitoring the rate of hydrogen peroxide (H2O2) decomposition at 240 nm. GPx activity was assessed using a coupled assay based on NADPH oxidation, with absorbance measured at 340 nm (Pasciu et al., 2024). Enzyme activity was expressed as U/mg protein or U/ml serum, as appropriate. Analysis of the liver enzymesLiver enzyme activity was measured using serum. Jugular venipuncture was used to draw blood samples, which were then allowed to clot before being centrifuged at 3,000×g for 15 minutes at 4°C to extract serum. The activities of alkaline phosphatase (ALP), aspartate aminotransferase (AST), and alanine aminotransferase (ALT) were measured in accordance with the manufacturer’s instructions using commercially available diagnostic kits and standard kinetic spectrophotometric techniques. U/l was used to express the enzyme activity. Statistical analysisSAS 9.4 (SAS Institute Inc., Cary, NC) was used for data analysis. Before analysis, the data were examined for homogeneity of variance (Levene’s test) and normality (Shapiro–Wilk test). One-way analysis of variance (ANOVA) was used to assess treatment effects. Tukey’s honestly significant difference test was used to separate means when ANOVA showed a significant impact. p < 0.05 was statistically significant, while p < 0.10 was a tendency. Least-squares mean ± SEM was used to display all data. Ethical approvalThe Animal Ethics Committee of the Livestock Research Station approved the experimental protocol (Protocol #2024). Twenty-four multiparous Awassi ewes (3–5 years old, 52 ± 4.2 kg body weight) were used at 140 ± 5 days of gestation. ResultsMitochondrial enzyme activitiesThe most pronounced results were related to the significant (p < 0.001) increases in the activities of ATP synthase (Complex V) by 43% and 38% in the propolis and rosemary groups, respectively, and of cytochrome c oxidase (Complex IV) by 52% and 47% in the propolis and rosemary groups, respectively, when compared to the control group. In addition, the combined treatment group showed higher ATP synthase and cytochrome c oxidase activities than the individual supplementation groups (p < 0.001) (Table 1). Table 1. Effects of propolis and rosemary extract supplementation on mitochondrial enzyme activities in the liver of Awassi ewes during late pregnancy.
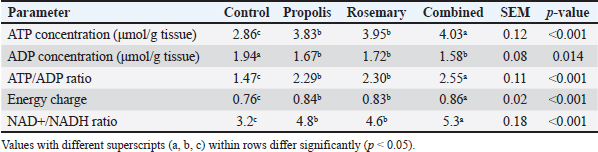
Cellular ATP levels and energy statusHepatic ATP levels increased in all treatment groups by 34%, 38%, and 41% for propolis, rosemary, and the combination treatment, respectively (p < 0.001). Hepatic ATP levels were significantly higher in all groups than in the control group (p < 0.001) (Table 2). Table 2. Effects of propolis and rosemary extract supplementation on hepatic adenosine triphosphate levels and energy status markers in Awassi sheep during late pregnancy.
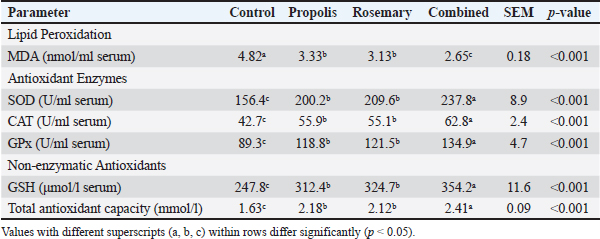
Oxidative stress markersSerum MDA levels were significantly lower in all groups than in the control group (p < 0.001). MDA (primary indicator of lipid peroxidation of cellular membranes) levels were significantly decreased by 31%, 35%, and 45% in the propolis, rosemary, and combined treatment groups, respectively (p < 0.001). The antioxidant enzyme activities were significantly improved in all treatment groups. The activity of superoxide dismutase, the first antioxidant enzyme barrier against reactive oxygen species, was enhanced by 28%, 34%, and 52% for propolis, rosemary, and combined treatments, respectively (Table 3). Table 3. Effects of propolis and rosemary extract supplementation on oxidative stress markers in late pregnancy Awassi sheep.
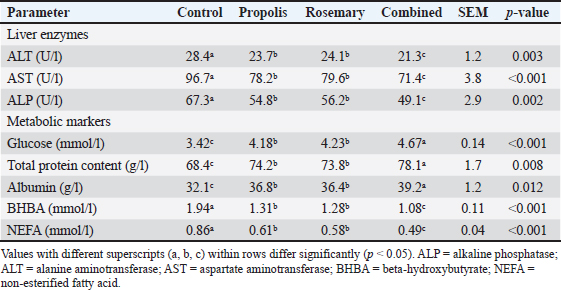
Liver function and metabolic parametersEvaluation of liver function parameters revealed that both supplements exert strong protective effects against pregnancy-induced hepatic stress. Liver enzyme activities (ALT, AST, and ALP) were significantly lower in all treatment groups, suggesting better hepatic health and less cellular damage (Table 4). Table 4. Effects of propolis and rosemary extract supplementation on liver function parameters in late-pregnancy Awassi sheep.
DiscussionEnhanced mitochondrial function through supplementation with natural compoundsThe results of this investigation conclusively establish that propolis and rosemary extract supplementation exerts a markedly positive impact on mitochondrial activity in Awassi sheep during the late stages of pregnancy. The findings of this study highlight the significant enhancement observed in mitochondrial ATP synthase (34%–68% increase) and cytochrome c oxidase (47%–71% increase), which can be primarily attributed to the underlying mitochondrial adaptations that transpire in response to the heightened energy demands during the late stages of pregnancy. This period is characterized by a surge in energy requirements by 50%–75%, mandating a state of heightened mitochondrial efficiency, especially within the hepatic tissues, to accommodate an upsurge in gluconeogenesis required to fulfill the elevated fetal glucose demand, typically ranging between 30 and 40g per fetus per day (Franco et al., 2020; Al-Khauzai et al., 2023). The mechanistic underpinnings of this enhancement are multifaceted. The flavonoids present in propolis, specifically chrysin and pinocembrin, stabilize the mitochondrial membrane and augment electron transport efficiency (Cedikova et al., 2014; Tvrdá et al., 2023). The maintenance of 80%–90% thermodynamic efficiency during oxidative phosphorylation, a well-established finding in the context of propolis, is likely the pivotal contributor to the observed improvement in ATP synthase activity. Similarly, the presence of rosmarinic acid in rosemary extract activates the AMPK signaling pathways, resulting in enhanced PGC-1α expression, thereby contributing to improved mitochondrial biogenesis. The observed improvement in mitochondrial function is consistent with the mechanism reported in previous studies, which may involve the activation of pathways such as AMPK and the regulation of PGC-1alpha (Naimi et al., 2017; Liu et al., 2022). The idea that natural polyphenolic substances can directly improve mitochondrial respiratory efficiency is supported by the reported increase in ATP synthase and cytochrome c oxidase activities, especially during physiologically demanding conditions such as late pregnancy. Similar improvements in respiratory control ratios and mitochondrial enzyme activities have been documented after propolis supplementation, which has been linked to improved electron transport chain efficiency and mitochondrial inner membrane stabilization (Tvrdá et al., 2023). Comprehensive oxidative stress protectionThe results of oxidative stress are astonishing, where both natural compound treatments offered significant protective actions. The decrease in MDA levels by 31%–45% and the increase in antioxidant enzyme activities by 28%–52% can offer all-around protection against oxidative stress during pregnancy. Pregnancy-induced oxidative stress is most prominent in the third trimester, in line with the peak fetal growth period (Simitzis et al., 2008; Calvin-Pereira L Gonz et al., 2015). This study is in accordance with the molecular mechanism of direct ROS scavenging by PPCs, along with Nrf2-dependent antioxidant pathway activation (Belardinelli et al., 2020). In the same regard, carnosic acid in rosemary extract has been shown to have a superior free radical quenching ability, with iron-chelation capacity greater than 75% at low concentrations (Nieto-Bona et al., 2020). Propolis’s antioxidant effects are largely attributed to its rich content of flavonoids and phenolic acids, which directly scavenge reactive oxygen species and activate endogenous antioxidant defense systems through Nrf2-dependent pathways (Kurek-Górecka et al., 2022). The liver is a primary regulator of systemic redox homeostasis, especially during late pregnancy and metabolic load. Therefore, this approach is still biologically relevant even though oxidative stress indicators were detected in serum. Hepatic oxidative changes in ruminants and other mammals under metabolic stress are reflected in changes in circulating MDA and antioxidant enzyme activities (Pasciu et al., 2023; Yu et al., 2024). Metabolic integration and physiological significanceThese results suggest a coordinated upregulation of several pathways that contribute to successful pregnancy. The concomitant increase in the activities of mitochondrial enzymes, improved cellular energy status, and enhanced antioxidant capacity support the notion that supplementation with natural compounds may help overcome different physiological barriers faced during late pregnancy in ewes. In this context, the observed increases in the activities of ATP synthase and cytochrome c oxidase and the resulting improvements in the respiratory control ratio are indicative of improved coupling between electron transport and ATP synthesis. This improvement in efficiency is particularly important during pregnancy when the liver’s energy requirements are increased to support gluconeogenesis, Fatty Acids synthesis, and protein production for fetal growth and maternal metabolic adaptations (Brand and Nicholls, 2011; Zhang et al., 2020). Clinical and practical implicationsIn practice, these outcomes have significant implications for sheep production systems, specifically in the management of late pregnancy nutrition to prevent pregnancy complications. The dosages used in this experiment (5 g/day propolis and 1,200 mg/day rosemary extract) are consistent with previous research on livestock and offer potential for practical, cost-effective integration into commercial sheep farming operations (Kandiel et al., 2019; Sheedeed et al., 2019). Preventing pregnancy-related metabolic disorders, such as pregnancy toxemia and fatty liver syndrome, can lead to economic benefits. These conditions, which affect 2%–10% of pregnant ewes, result in substantial maternal mortality losses, reduced lamb survival, and decreased subsequent reproductive performance (Cal-Pereyra et al., 2015; Al-Khauzai et al., 2023). The positive metabolic outcomes demonstrated in the current study provide a rationale for the use of natural compound supplementation as a means to reduce the incidence of such complications. The findings of this study highlight the potential application of natural antioxidant supplementation as a nutritional strategy to mitigate pregnancy-related metabolic disorders in sheep, such as pregnancy toxemia and fatty liver syndrome, which are associated with significant economic losses and reduced lamb survival (Al-Khauzai et al., 2023). ConclusionThe results confirm the original idea that supplementing Awassi sheep with propolis and rosemary extract, either separately or in combination, improves mitochondrial function and lowers oxidative stress in late pregnancy. The positive function of these supplements in promoting pregnancy-associated metabolic adaptations is highlighted by increased ATP synthase and cytochrome c oxidase activities, higher ATP levels, and decreased oxidative stress indicators. The stronger benefits of the combined treatment point to a possible synergistic interaction between rosemary extract and propolis, which is probably mediated by complementing mechanisms of antioxidant action and mitochondrial protection. The applied dosages are compatible with sustainable and animal-friendly management practices and are suitable for use in commercial sheep production systems. Future research should look into long-term reproductive outcomes, underlying biological mechanisms, and optimal supplementing techniques to further improve animal welfare and production. AcknowledgmentThe authors would like to thank the Department of Medical Laboratory Techniques, College of Health and Medical Techniques, Al-Bayan University, Iraq, for their assistance. Conflict of interestThe authors declare no conflicts of interest. FundingNo funding was received for this study. Authors’ contributionsMahaba salah Mahdi hassan Al_zwelef and Faisal Ali Lattef compiled the manuscripts, conducted the research, and ensured that the descriptions were accurate. The samples were gathered, and the research data were processed by Mohammed T. Jaafar and Zainab Fadhil Abdulhussein, who are the main authors. The study strategy, content organization, statistical analysis, and manuscript editing were all performed by Ghadir Kamil Ghadir. Data availabilityThe datasets created and investigated in this study are available upon reasonable request from the corresponding author. ReferencesAl-Khauzai, M.H.M., Al-Thuwaini, T.M. and Al-Anbari, N.N. 2023. Clinical and biochemical study of pregnancy-related toxemia in Iraqi ewes. Vet. World 16(6), 1186–1192; doi:10.14202/vetworld.2023.1186-1192 Belardinelli, C., Bogdanov, S., Celli, N., Ferioli, M., Fini, M., Galli, A. and Vannini, L. 2020. Molecular and cellular mechanisms of the effects of propolis on inflammation, oxidative stress, and glycemic control in patients with chronic diseases. Nutr. Metab. 17, 65; doi:10.1186/s12986-020-00485-5 Berry, B.J., Trewin, A.J., Amitrano, A.M., Kim, M. and Wojtovich, A.P. 2018. The protonmotive force: mitochondrial uncoupling and reactive oxygen species. J. Mol. Biol. 430(21), 3873–3891; doi:10.1016/j.jmb.2018.03.025 Bouwman, R.A., Musters, R.J., Van Beek-harmsen, B.J., De Lange, J.J. and Boer, C. 2019. During cardiac surgery, reactive oxygen species precede protein disulfide isomerase release from the endoplasmic reticulum. PLos One. 14(6), 218724; doi:10.1371/journal.pone.0218724 Brand, M.D. and Nicholls, D.G. 2011. Assessing mitochondrial dysfunction in cells. Biochem. J. 435(2), 297–312; doi: 10.1042/BJ20110162 Brand, M.D., Orr, A.L., Perevoshchikova, I.V. and Quinlan, C.L. 2019. The role of mitochondrial function and cellular bioenergetics in aging and disease. Br. J. Dermatol. 169(2), 1–8; doi:10.1111/bjd.12208 Buettner, R., Nguyen, L.X.T., Morales, C., Chen, M.H., Wu, X., Chen, L.S. and Rosen, S.T. 2021. Targeting the metabolic vulnerability of acute myeloid leukemia blasts with a combination of venetoclax and 8-chloro-adenosine. J. Hematol. Oncol. 14(1), 70; doi: 10.1186/s13045-021-01076-4 Cal-Pereyra, L., González-Montaña, J.R., Benech, A., Acosta-Dibarrat, J., Martín, A., Perini, S. and Rodríguez, P. 2015. Evaluation of three therapeutic alternatives for the early treatment of ovine pregnancy toxemia. Irish Vet. J. 68(1), 25; doi:10.1186/s13620-015-0053-2 Calvin-Pereira L Gonz., ález-Montaña, J.R., Benech A Acosta., -Dibarrat, J., Martín, A., Perini, S. and Rodríguez, P. 2015. Changes in some blood indicators of pregnant ewes with toxemia treated with propylene glycol or glycerol. J. Dairy Res. 82(4), 394–399; doi:10.1017/S0022029915000436 Cedikova, M., Pitule, P., Kripnerova, M., Markova, M. and Kuncova, J. 2014. Effects of Czech propolis on mitochondrial function in sperm. Evid. Based Complement. Alternat. Med. 2014, 248768; doi:10.1155/2014/248768 Fawaz, M.A., Ali, M.A. and Khalaphallah, R. 2025. Impacts of propolis and Spirulina platensis supplementation on Japanese quail growth, nutrient digestibility, and gut microbiota under heat stress. Sci. Rep. 15, 31688; doi:10.1038/s41598-025-17082-2 Franco, L.V.R., Su, C.H. and Tzagoloff, A. 2020. Modular assembly of ATP synthase and cytochrome oxidase in yeast mitochondria. Biol. Chem. 401(6-7), 835–853; doi:10.1515/hsz-2020-0112 Habtemariam. and S. 2020. Exploring the prospective role of propolis in modifying aging hallmarks. Biomed. Pharmacother. 128, 110262; doi:10.1016/j.biopha.2020.110262 Kandiel, M.M.M., El-Khaiat, H.M. and Karima, M.M. 2019. Changes in some hematobiochemical and hormonal profiles of Barki sheep with various reproductive statuses. Small. Ruminant. Res. 136(3), 87–95; doi:10.1007/s11250-011-9966-2 Koopman, W.J.H., Nijtmans, L.G.J., Dieteren, C.E.J., Roestenberg, P., Valsecchi, F., Smeitink, J.A.M. and Willems, P.H.G.M. 2012. Mammalian mitochondrial complex I: biogenesis, regulation, and reactive oxygen species generation. Antioxid. Redox Signal. 12(12), 1431–1470; doi:10.1089/ars.2009.2743 Kurek-Górecka, A., Walczyńska-Dragon, K., Felitti, R., Baron, S. and Olczyk, P. 2022. Propolis and diet rich in polyphenols as cariostatic agents reducing accumulation of dental plaque. Molecules 27(1), 271; doi: 10.3390/molecules27010271 Ley-Ngardigal, S.A., Rodríguez-Nuevo A Sch., äffner I Adrover., Hakimi, A., Brunet, M. and Mitter, A. 2022. Monitoring ATP levels in living cells: where do we stand?. FEBS J. 289(2), 397–426; doi:10.1111/febs.16169 Liu, M., Dai, Y., Yao, X., Li, Y., Luo, Q., Xia, Y. and Chen, Z. 2022. Rosmarinic acid inhibits mitochondrial damage by alleviating the unfolded protein response. Front. Pharmacol. 13, 859978; doi:10.3389/fphar.2022.859978 Naimi, M., Vlavcheski, F., Shamshoum, H. and Tsiani, E. 2017. Rosemary extract as a potential anti-hyperglycemic agent: current evidence and future perspectives. Nutrients 9(9), 968; doi:10.3390/nu9090968 Nieto-Bona, A., García, V., Fernández-García, A., Olmo R Aguirre., Gonz, L., ález-Paramás AM Santos. and -Buelga, C. 2020. Antioxidant and anti-inflammatory properties of a flavonoid-rich extract from R. officinalis L. leaves. Antioxidants 9(11), 1073; doi:10.3390/antiox9111073 Paradies, G., Paradies, V., Ruggiero, F.M. and Petrosillo, G. 2014. Oxidative stress, cardiolipin, and mitochondrial dysfunction in NAFLD. World. J. Gastroenterol. 20(39), 14205–14218; doi:10.3748/wjg.v20.i39.14205 Patlevič, P., Vašková, J., Švorc Jr, P., Vaško, L. and Švorc, P. 2016. Reactive oxygen species and antioxidant defense in human gastrointestinal diseases. Integrative Med. Res. 5(4), 250–258; doi: 10.1016/j.imr.2016.07.004 Pasciu, V., Nieddu, M., Sotgiu, F.D., Baralla, E. and Berlinguer, F. 2023. An overview on assay methods to quantify ROS and enzymatic antioxidants in erythrocytes and spermatozoa of small domestic ruminants. Animals 13(14), 2300; doi: 10.3390/ani13142300 Pasciu, V., Nieddu, M., Sotgiu, F.D., Baralla, E. and Berlinguer, F. 2023. An overview of assay methods to quantify ROS and enzymatic antioxidants in small domestic ruminant erythrocytes and spermatozoa. Animals (Basel). 13(14), 2300; doi:10.3390/ani13142300 Shedeed, H.A., Farrag, B., Elwakeel, E.A., Abd El-Hamid, I.S. and El-Rayes, M.A.H. 2019. Propolis supplementation improved productivity, oxidative status, and immune response of Barki ewes and lambs. Vet. World 12(6), 834; doi: 10.14202/vetworld.2019.834-843 Simitzis, P.E., Deligeorgis, S.G., Bizelis, J.A., Dardamani, A., Theodosiou, I. and Fegeros, K. 2008. Effect of dietary oregano oil supplementation on lamb meat characteristics. Meat. Sci. 79(2), 217–223; doi:10.1016/j.meatsci.2007.09.005 Spinazzi, M., Casarin, A., Pertegato, V., Salviati, L. and Angelini, C. 2012. Assessment of enzymatic activities of mitochondrial respiratory chains in tissues and cultured cells. Nature Protocols 7(6), 1235–1246; doi:10.1038/nprot.2012.058 Talafha, A.Q. and Ababneh, M.M. 2011. Awassi sheep reproduction and milk production. Trop. Anim. Health Prod. 43(7), 1319–1326; doi: 10.1007/s11250-011-9858-5 Tvrdá, E., Árvay, J., Ďuračka, M. and Kačániová, M. 2023. Mitochondria-stimulating and antioxidant effects of slovak propolis on bovine spermatozoa. Oxygen 3(2), 179–189; doi:10.3390/oxygen3020013 Wang, T., Takikawa, Y. and Tabuchi, T. 2013. Rosemary (Rosmarinus officinalis L.) extract regulates glucose and lipid metabolism by activating the AMPK and PPAR pathways in HepG2 cells. J. Agricult. Food Chem. 61(11), 2803–2810; doi:10.1016/j.jafc.2012.08.010 Yu, C., Luo, Y. and Shen, C. 2024. Effects of microbe-derived antioxidants on growth performance, hepatic oxidative stress, mitochondrial function and cell apoptosis in weaning piglets. Nat. Biol. Chem. Biol. J. Anim. Sci. Biotechnol. 15, 128; doi:10.1186/s40104-024-01088-3 Zhang, H., Wu, L., Xu, C., Xia, C., Sun, L. and Shu, S. 2020. Plasma metabolomics reveals metabolic disturbance and tissue-specific metabolic reprogramming in bovine ketosis. Scientific Rep. 10(1), 8691; doi:10.1038/s41598-023-35383-2 | ||
| How to Cite this Article |
| Pubmed Style Lattef FA, Al_zwelef MSMH, Jaafar MT, Abdulhussein ZF, Ghadir GK. Role of propolis and rosemary extract in improving mitochondrial efficiency and energy metabolism enzymes in late pregnancy Awassi sheep liver. Open Vet. J.. 2026; 16(2): 1222-1229. doi:10.5455/OVJ.2026.v16.i2.40 Web Style Lattef FA, Al_zwelef MSMH, Jaafar MT, Abdulhussein ZF, Ghadir GK. Role of propolis and rosemary extract in improving mitochondrial efficiency and energy metabolism enzymes in late pregnancy Awassi sheep liver. https://www.openveterinaryjournal.com/?mno=294161 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.40 AMA (American Medical Association) Style Lattef FA, Al_zwelef MSMH, Jaafar MT, Abdulhussein ZF, Ghadir GK. Role of propolis and rosemary extract in improving mitochondrial efficiency and energy metabolism enzymes in late pregnancy Awassi sheep liver. Open Vet. J.. 2026; 16(2): 1222-1229. doi:10.5455/OVJ.2026.v16.i2.40 Vancouver/ICMJE Style Lattef FA, Al_zwelef MSMH, Jaafar MT, Abdulhussein ZF, Ghadir GK. Role of propolis and rosemary extract in improving mitochondrial efficiency and energy metabolism enzymes in late pregnancy Awassi sheep liver. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 1222-1229. doi:10.5455/OVJ.2026.v16.i2.40 Harvard Style Lattef, F. A., Al_zwelef, . M. S. M. H., Jaafar, . M. T., Abdulhussein, . Z. F. & Ghadir, . G. K. (2026) Role of propolis and rosemary extract in improving mitochondrial efficiency and energy metabolism enzymes in late pregnancy Awassi sheep liver. Open Vet. J., 16 (2), 1222-1229. doi:10.5455/OVJ.2026.v16.i2.40 Turabian Style Lattef, Faisal Ali, Mahaba Salah Mahdi Hassan Al_zwelef, Mohammed T. Jaafar, Zainab Fadhil Abdulhussein, and Ghadir Kamil Ghadir. 2026. Role of propolis and rosemary extract in improving mitochondrial efficiency and energy metabolism enzymes in late pregnancy Awassi sheep liver. Open Veterinary Journal, 16 (2), 1222-1229. doi:10.5455/OVJ.2026.v16.i2.40 Chicago Style Lattef, Faisal Ali, Mahaba Salah Mahdi Hassan Al_zwelef, Mohammed T. Jaafar, Zainab Fadhil Abdulhussein, and Ghadir Kamil Ghadir. "Role of propolis and rosemary extract in improving mitochondrial efficiency and energy metabolism enzymes in late pregnancy Awassi sheep liver." Open Veterinary Journal 16 (2026), 1222-1229. doi:10.5455/OVJ.2026.v16.i2.40 MLA (The Modern Language Association) Style Lattef, Faisal Ali, Mahaba Salah Mahdi Hassan Al_zwelef, Mohammed T. Jaafar, Zainab Fadhil Abdulhussein, and Ghadir Kamil Ghadir. "Role of propolis and rosemary extract in improving mitochondrial efficiency and energy metabolism enzymes in late pregnancy Awassi sheep liver." Open Veterinary Journal 16.2 (2026), 1222-1229. Print. doi:10.5455/OVJ.2026.v16.i2.40 APA (American Psychological Association) Style Lattef, F. A., Al_zwelef, . M. S. M. H., Jaafar, . M. T., Abdulhussein, . Z. F. & Ghadir, . G. K. (2026) Role of propolis and rosemary extract in improving mitochondrial efficiency and energy metabolism enzymes in late pregnancy Awassi sheep liver. Open Veterinary Journal, 16 (2), 1222-1229. doi:10.5455/OVJ.2026.v16.i2.40 |