| Research Article | ||
Open Vet. J.. 2025; 15(12): 6747-6766 Open Veterinary Journal, (2025), Vol. 15(12): 6747-6766 Research Article Effects of dietary supplementation with fenugreek oil on growth, physiology, and resistance to A. hydrophila infection in Nile tilapia (Oreochromis niloticus) fingerlingsEman M. Moustafa1*, Safaa Abdo2, Seham El-Kassas3,4, Mohamed M. Zayed5, Rasha A. Al Wakeel6, Yasser A. Menesy1, Amira A. Omar11Fish Diseases and Management Department, Faculty of Veterinary Medicine, Kafrelsheikh University, Kafrelsheikh, Egypt 2Genetics and Genetic Engineering, Department of Animal Wealth Development, Faculty of Veterinary Medicine, Kafrelsheikh University, Kafr El-Sheikh, Egypt 3National Committee of Nutritional Sciences Academy for Scientific Research and Technology (ASRT), Cairo, Egypt 4Animal, Poultry and Fish Breeding and Production, Department of Animal Wealth Development, Faculty of Veterinary Medicine, Kafrelsheikh University, Kafrelsheikh, Egypt 5Department of Aquaculture, Faculty of Aquatic and Fisheries Sciences, Kafrelsheikh University, Kafr El-sheikh, Egypt 6Department of Physiology, Faculty of Veterinary Medicine, Kafrelsheikh University, Kafrelsheikh, Egypt *Corresponding Author: Eman M. Moustafa, Fish Diseases and Management Department, Faculty of Veterinary Medicine, Kafrelsheikh University, Kafrelsheikh, Egypt. Email: emantarek2002 [at] yahoo.com Submitted: 23/09/2025 Revised: 08/11/2025 Accepted: 15/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
AbstractBackground: Aeromonas hydrophila is an opportunistic pathogen that causes Motile Aeromonad Septicemia (MAS) with significant losses in Nile tilapia farms. Because of its bioactive compounds that enhance immunity and antioxidant capacity, dietary supplementation with fenugreek oil can effectively mitigate A. hydrophila-associated losses. Aim: This study aimed to explore the hematobiochemical, immune, antioxidant, and histopathological impacts of dietary fenugreek oil (FO) in Nile tilapia under normal culturing plus A. hydrophila infection. Methods: Approximately 300 Nile tilapia fingerlings (15.00 ± 3.00 g) were randomly distributed into five clusters, designated as FO-0 (control), FO-0.5, FO-1.0, FO-2.0, and FO-4.0 in which 0.0, 0.5, 1.0, 2.0, and 4.0 g FO/kg diet were supplemented, respectively. Diets were offered for 8 weeks at a 3% feeding rate. After 8 weeks of FO feeding, blood and tissue samples were collected to evaluate the impact of FO feeding under normal conditions. Then, fish in each treatment were injected (intra-peritoneally) with 0.2 ml of A. hydrophila suspension and monitored for 2 weeks for any mortality. Then, blood and tissue were sampled. Results: Final body weight, gain, feed conversion ratio, specific growth rate, and protein efficiency ratio in all FO-enriched groups displayed numerical increases relative to the control group (P > 0.05), peaking at 4.0 g FO/kg diet. FO dietary supplementation significantly improved antioxidant capacity, reflected by increases in superoxide dismutase and catalase activities with lowered malondialdehyde levels, particularly at 2.0–4.0 g FO/kg diet, both under normal and A. hydrophila challenge. Moreover, the innate immunity biomarkers lysozyme activity, and phagocytic activity and index exhibited marked dose-dependent improvements, both under normal, and following A. hydrophila exposure (P < 0.05). Biochemically, total protein and globulin concentrations were significantly elevated with reduced urea concentration and hepatic enzymes activities in all FO-treated groups (P < 0.05). Moreover, all FO-supplemented groups showed improved histopathological features of the intestine, liver, and spleen with mitigation of the A. hydrophila-degenerative changes. Conclusion: Thus, FO could be used as a promising natural additive in Nile tilapia diet, particularly at 2.0–4.0 g/kg diet, to enhance hemato-biochemical profile, antioxidant defense, innate immunity, tissue integrity, and resistance against A. hydrophila, assuring sustainable and health-oriented aquaculture activities. Keywords: Nile tilapia, Fenugreek oil, Antioxidant status, Innate immunity, Histopathological features, A. hydrophila. IntroductionNile tilapia (Oreochromis niloticus) is a cornerstone fish species in global aquaculture. It is characterized by a steadily increasing production and adaptability to many challenges, such as infection, and suboptimal feed utilization (Khalafalla et al., 2020; Abdo et al., 2024). These challenges cause economic losses in Nile tilapia, particularly with intensified farming and global environmental changes (Hamed et al., 2024). Therefore, it is crucial to explore new natural feed additives that enhance fish’s performance and boost immunity, thereby avoiding the use of antibiotics and alleviating their associated antibiotic resistance challenge (Diab et al., 2023). Medicinal herbs have gained attention for their promising impacts and prebiotics characteristics in improving fish growth, feed utilization, physiology, and susceptibility to diseases (Gabriel, 2019; Kuebutornye and Abarike, 2020; Ahmadifar et al., 2021; Abaho et al., 2022). Several studies have reported the effectiveness of several medicinal plants in improving fish growth performance, biochemical responses, and resistance to diseases compared with antibiotics (Awad and Awaad, 2017; Gabriel, 2019; Kuebutornye and Abarike, 2020; Ahmadifar et al., 2021; Abaho et al., 2022). Fenugreek (Trigonella foenum-graecum) is a medicinal plant that belongs to the family Fabaceae, and its seeds or extracts have many beneficial properties (Bahmani et al., 2016). Its seeds contain various bioactive compounds, including alkaloids, sapogenins, and steroids, which induce valuable pharmacological effects, such as anti-inflammation and antimicrobial effects, and reduce triglyceride and cholesterol levels (Diab et al., 2023). In Nile tilapia, fenugreek seeds and extract have been extensively explored, demonstrating increases in body weight gain, improved in feed conversion ratio, and increased resistance and survival against pathogens such as Aeromonas hydrophila (Moustafa et al., 2020; Diab et al., 2023). Fenugreek oil, particularly Fenugreek essential oil, contains concentrated nutritive and bioactive compounds with potentially higher bioavailability for lipophilic compounds, such as essential fatty acids and volatile compounds, compared with the seed and extract (Akbari et al., 2019). Despite its concentrated content of bioactive compounds, fenugreek oil remains unexplored, offering distinct advantages over seeds and extracts, representing a key research gap. Thus, in this study, we hypothesized that dietary supplementation of Nile tilapia with fenugreek essential oil would add extra nutritional value that would enhance growth indices and hematobiochemical profile, and excel in infection control over dietary Fenugreek seeds or extract. Therefore, this study aimed to explore the impact of dietary supplementation of fenugreek essential oil on Nile tilapia growth performance, physiological and biochemical responses, and resistance to A. hydrophila infection. Materials and methodsPreparation of experimental diets, fish sources, and fish managementIn this study, five experimental diets were formulated to be both isonitrogenous and isocaloric, with 300 g/kg of crude protein and 12.6 MJ/kg of digestible energy according to the guidelines and nutritional requirements set by the National Research Council (Council, 2011). All dietary ingredients are listed in Table 1. All dry ingredients were ground into small particles using a feed processor. Then, fenugreek oil (Lallemond, France and provided by EGA vet co., Egypt) was added to diets at 0.0, 0.5, 1.0, 2.0, and 4.0 g/kg, resulting in five experimental diets. Subsequently, in a combination blender, all ingredients were thoroughly mixed for 20 minutes, pelletized into pellets, allowed to dry for 24 hours at room temperature, and kept in a refrigerator in plastic bags at 4ºC. Table 1. Composition and chemical analysis of experimental diets (on a dry matter basis)
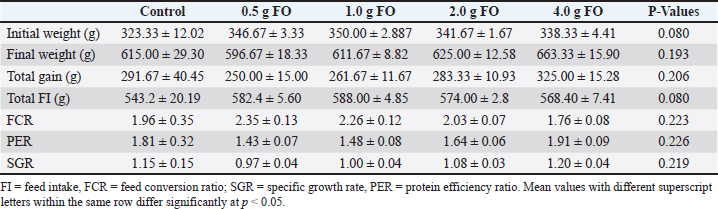
Using 300 fingerlings of O. niloticus, weighing on average 15.00 ± 3.00 g, the five experimental diets were distributed into five treatments, designated as FO-0, FO-0.5, FO-1.0, FO-2.0, and FO-4.0, with 0.0, 0.5, 1.0, 2.0, and 4.0 g of fenugreek oil/kg diet, respectively. Each group contained 60 Nile tilapia fingerlings and was replicated 3 times with 20 Nile tilapia fingerlings per replicate. Nile tilapia fingerlings were obtained from a private fish farm located in Al Reyad, Tolompate 7, Kafr El-Sheikh Governorate, Egypt, where they were exposed to a commercial diet containing 30% dietary protein for 2 weeks at the Laboratory of Fish Diseases and Management Department, Faculty of Veterinary Medicine, Kafrelsheikh University, Egypt. Throughout the experiment, fish were stocked in glass aquariums of 60 × 35 ×40 cm, with a water capacity of 70 l and supplied with a functional aeration system. The experiment was continued for eight weeks, feeding fish at 3% of their biomass per aquarium twice per day at 8:00 am and 14:00 pm. Throughout this experiment, the water in each aquarium was exchanged daily, and approximately half of the water in each aquarium was replenished with dechlorinated water. The water quality attributes were periodically assessed using a Lamotte device (USA) with a temperature range of 24°C–27°C, dissolved oxygen with a concentration of 6.5 ± 0.5 mg L−1, a pH of 7.1 ± 0.8, an EC of 219 ± 2 μ mho/cm, ammonia amended to the standard allowable limits (0.1 mg total ammonia), and day-to-night photoperiods of 12:12 hours. Evaluation of the growth performance indicesThe Nile tilapia fingerlings’ starting and final body weights were recorded using a digital balance (PW Balance, ADAM Equipment Co., USA), with 6 hours fasting and thorough drying before weighing. Total body weight gain was calculated by subtracting initial weights from the final weight according to the following equation, BWG=final weight − initail weight (Siddiqui et al., 1988). The specific growth rate (SGR) was calculated using the following formula: Specific growth rate equation: Blood and tissue samplesAfter 8 weeks of dietary Fenugreek oil supplementation, 45 fish (9 fish/treatment) were randomly selected for blood and tissue sampling after 24 hour fasting and under mild anesthesia using tricaine methane sulfonate at a concentration of 100 mg/l (Dawood et al., 2020). Blood samples were collected from the caudal vein using 5-ml gauge syringes. Each collected blood sample was subsequently allocated into two equal halves, one half of which was used immediately for hematological analysis and differential leukocyte count after being collected in EDTA-heparinized tubes. The other half was used for serum separation by centrifugation at 3000 rpm and 4 °C for 10 minutes (Hawk, 1965). Subsequently, fish were dissected, and tissue samples from the intestine, hepatopancreas, and spleen were collected for histopathological examination. Tissue samples were immediately fixed in 10% formalin solution for 24–48 hours, then dehydrated using 70% ethyl alcohol. Dehydrated samples were cleared in xylene and embedded in paraffin wax. For histopathological and morphometric analysis, sections of 4–5 µm thickness were stained with haematoxylin and eosin dye according to the method described by (Bancroft and Gamble, 2008). Finally, the stained sections were photomicrographed using a digital camera (Leica EC3, Leica, Germany) attached to a microscope (Leica DM500). The length, width, crypt depth, villi length/crypt depth, and villi surface area were measured using imaging analysis software (NIH, Bethesda, MD). Briefly, 10 random villi and villus-associated crypts from 5 intestinal cross-sections were selected from each sample. Assessment of hematobiochemical response to dietary supplementation with fenugreek oilFrom whole blood samples, the counts of erythrocytes (RBC, × 106/mm) and leucocytes (WBC, ×103/mm) were performed using a hemocytometer (Houston, 1990). Based on the method described by (Thrall et al., 2012), the hemoglobin concentration (Hb g/dl) was calculated. The microhematocrit method developed by (Bain et al., 2016) was applied to calculate packed cell volume (PCV%). The calculation of the mean corpuscular hemoglobin concentration (MCHC) was performed in accordance with the methods described by (Wintrobe, 1934). As soon as possible, the analyzer of automated Coulter LH 750 hematology (Beckman Coulter, Fullerton, CA) was utilized to ascertain the mean corpuscular volume (MCV) (Bain et al., 2016). Differential leucocytic (WBC) counts were executed complying to the methodology explained by (Houston, 1990). The concentrations of albumin and total protein were measured in serum samples using commercial kits (Biodiagnostics, Egypt) according to the manufacturer’s instructions. Globulin content was determined by subtracting the albumen concentration from the total protein concentration. Aspartate aminotransferase (AST) and serum alanine aminotransferase (ALT) were also measured in serum samples using commercial kits (Biodiagnostics, Egypt) following the kit’s instructions. Innate immune parameters and antioxidant activity EvaluationPhagocytosis biomarkers, phagocytic activity (PA) and index (PI), were assessed following the method described by (Kawahara et al., 1991). Lysozyme activity was tested using the method outlined by (Demers and Bayne, 1997), which relied on the capacity of lysozyme to lyse Gram-positive lysozyme-delicate bacteria; Micrococcus lysodeikticus. Enzyme activity of supper oxide dismutase and catalase, and lipid peroxide levels (Malondialdehyde, MDA) were assessed in serum samples using commercial kits according to the manufacturer’s guidelines (Biodiagnostic Co., Egypt). Measuring the activity of digestive enzymesLipase, total protease, and amylase activities were determined colorimetrically (580 nm for lipase and 660 nm for amylase) using commercial kits from Spectrum Diagnostic Co., Egypt. Challenge with A. hydrophilaTo evaluate the impact of dietary supplementation of fenugreek oil on the resistance of Oreochromis niloticus fingerlings to Aeromonas hydrophila bacterial infection, an intra-peritoneal injection of 0.2 ml of fresh A. hydrophila culture suspension (2.7×106 CFU) was performed according to (Li et al., 2011), followed by 15 days of monitoring to report mortalities. Briefly, at the end of this feeding experiment and after blood and tissue sampling, fish in each treatment group were rested for 24 hours to eliminate sampling stress. Then, A. hydrophila infection was performed by intra-peritoneal injection of 0.2 ml of A. hydrophila suspension for each fish. Nile tilapia fingerlings in the control group (no fenugreek oil) were injected with 0.2 ml of phosphate-buffered saline (PBS). The culture suspension of A. hydrophila strain was prepared, and the concentration of A. hydrophila for the experiments was determined based on the drop plate technique. Briefly, A. hydrophila strain was cultured for 24 hours on tryptic soy agar (TSA) plates at 37°C for 24 h with constant shaking at 250 rpm for 24 hours. Colonies were determined using standard dilution and plating methods. Each group received an intraperitoneal injection of every bacterial dilution (0.2 ml/fish). According to (Reed and Muench, 1938), LD50, which refers to the lethal dose that results in the mortality of 50% of the administered fish, was computed to contain 1.3 x 108 CFU/ml (Reed and Muench, 1938; Moustafa et al., 2020). All fish were maintained for 2 weeks following the injection for observation. Dead fish were transported for additional PM inspection. After the two-week inspection and monitoring of fish following the A. hydrophila challenge, blood and tissue samples were collected for hematobiochemical, innate immunity, and histopathological examination. Histopathological examinationFor morphometric and histopathological investigations, tissue samples from the intestine, hepatopancreas, and spleen of both pre- and post-infection groups were collected immediately from five randomly selected fish from each treatment group. The samples were first fixed in 10% formaldehyde solution for 24–48 hours, then dehydrated in ethyl alcohol (70% to absolute alcohol). Dehydrated samples were cleared in xylene and embedded in paraffin wax. For histopathological and morphometric analysis, sections of 4–5 µm thickness were stained with haematoxylin and eosin according to the method of (Moustafa et al., 2020). Morphometric parameters, including the length, width, crypt depth, villi length/crypt depth of intestinal villi and villi surface area, were measured using image analysis software (NIH, Bethesda, MD). A total of 10 random villi and villus-associated crypts from 5 intestinal cross-sections were randomly selected for each treatment group. Statistical analysisTwo-way ANOVA and Tukey’s multiple comparisons were used to statistically assess the effect of dietary supplementation of fenugreek oil, A. hydrophila infection, and their interaction using SPSS (SPSS version 22, SPSS Inc., Chicago, Il, USA). Results were expressed as means ± standard error (SE) with statistical significance set at p < 0.05. GraphPad Prism 9.0 (GraphPad® Software Inc., San Diego, CA, USA) was used to create figures. Ethical ApprovalThis experiment was conducted at the Laboratory of Fish Diseases and Management Department, Faculty of Veterinary Medicine, Kafrelsheikh University, Egypt. All experimental methodology, fish management, and protocols were adhered to all pertinent legislation and the guidelines set by the Animal Ethics Committee of Kafrelsheikh University, with ethical approval ID KFS-IACUC/137/2023. ResultsEffect of dietary supplementation with fenugreek oil on growth performance of Nile tilapia fingerlingsTable 2 shows the growth-related indices of Nile tilapia fingerlings fed diets supplemented with different levels of FO. Supplementation of the diet with FO did not significantly alter the growth of Nile tilapia fingerlings (P > 0.05). However, the dietary supplementation of FO at 4.00 g/kg diet induced numerical increases in final body weight (663.33 ± 15.90 g) and body weight gain (325.00 ± 15.28 g) compared with the control ( no FO in diet) and other FO-supplementing levels. Similarly, feed conversion, protein efficiency ratios, and specific growth rate showed improved responses with increasing FO supplementation level, peaking at 4.00 g FO/kg diet. Although the reported differences in growth-related indices did not show statistical significance, the numerical increases in their values reflect the positive impact of FO dietary supplementation on Nile tilapia growth and feed utilization efficiency. Table 2. Growth performance of Nile tilapia fingerlings in response to FO dietary supplementation.
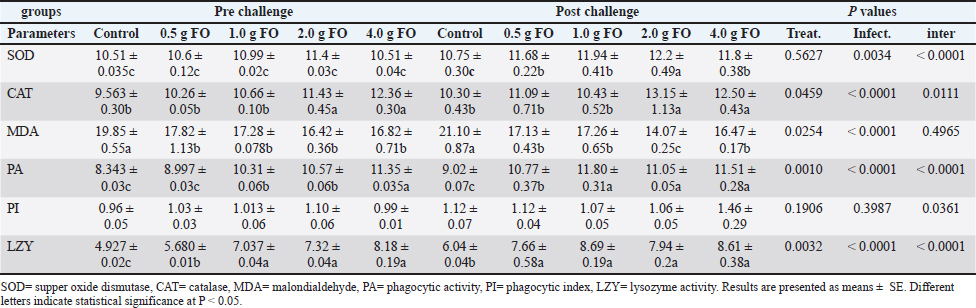
Effect of dietary fenugreek oil supplementation on antioxidant and immune-related biomarkers of Nile tilapia fingerlingsThe assessment of antioxidant enzyme activity and immune-related indices in Nile tilapia fingerlings following FO supplementation is illustrated in Table 3. Superoxide dismutase activity was significantly enhanced in the FO-supplemented groups, particularly after A. hydrophila challenge, with the highest activity reported in the 2.00 g FO/kg diet (12.20 ± 0.49) and 4.00 g FO/kg diet (11.80 ± 0.38), and exposed to A. hydrophila challenge compared to the other groups (P < 0.05). Catalase activity also increased in a dose-dependent pattern, with the highest activity reported at the 2.00 and 4.00 g of FO/kg diet supplementation levels in both normal and following the A. hydrophila challenge (P < 0.05). The improved activities of antioxidant enzymes were correlated with a significant reduction in the lipid peroxidation indicator, malondialdehyde (MDA), in all FO-supplemented groups, particularly at 2.00 and 4.00 g of FO/kg diet under normal culturing conditions. The MDA level significantly increased in the tilapia fingerlings exposed to A. hydrophila infection without FO dietary supplementation (control), which interestingly reduced in the FO-supplemented groups and exposed to A. hydrophila infection, particularly at 2.00 g of FO/diet (P < 0.05). Table 3. Antioxidant enzyme activity and innate immune response biomarkers in response to FO dietary supplementation.
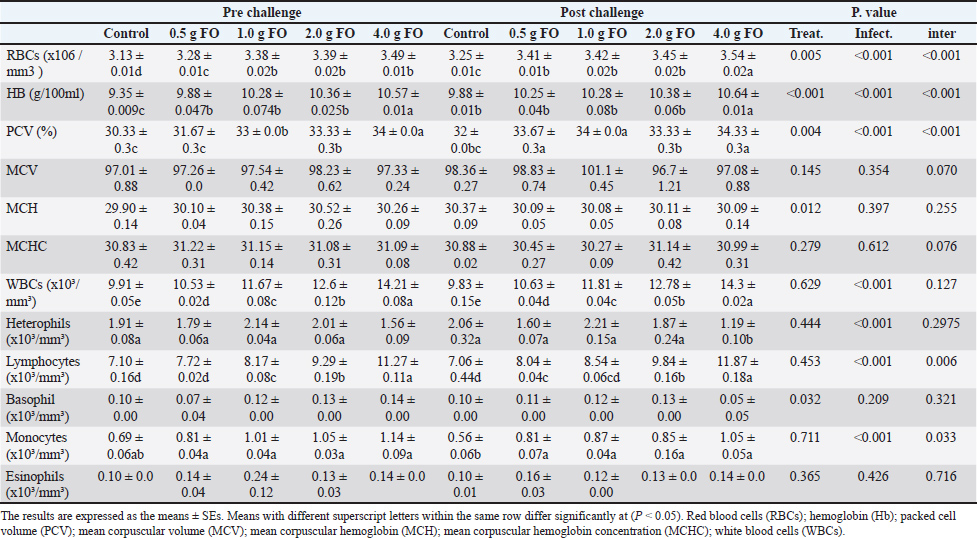
For phagocytic activity (PA) and index (PI), there were obvious increases in their levels with the higher FO-supplementation dose (4.00 g FO/kg diet) in both normal and following A. hydrophila infection compared to the other groups (P < 0.05). Lysozyme also displayed a dose-dependent increase in its activity. Under normal culturing conditions (pre-challenge), the supplementation doses 1–4.00 g of FO/kg diet showed the highest activity (compared with the control and 0.5g FO (P < 0.05). After A. hydrophila exposure, all FO-supplemented groups (0.5–4.00 g FO/kg diet) exhibited higher lysozyme activity than the non-supplemented group (control). Collectively, FO supplementation, particularly at 2 and 4.00 g FO/kg diet, enhanced the antioxidant response by increasing SOD and CAT activities, reduced stress response by reducing MDA levels, and improved innate immunity-related biomarker (PA, PI, and LZ activity). These effects were pronounced with A. hydrophila infection. Hematological parameters and differential leucocytic countDietary supplementation with graded FO levels significantly influenced several hematological parameters under normal culturing (pre-challenge) and following A. hydrophila exposure (post-challenge) (Table 4). FO-dietary supplementation induced clear dose-dependent increases in the red blood cell count (RBCs), hemoglobin (Hb), and PCV (P < 0.05). Under normal culturing, the RBC count, Hb, and PCV peaked at 4.00 g FO/kg, while following A. hydrophila, all FO-supplemented groups showed higher values of these parameters compared with the control (non-supplemented) (P < 0.05). However, no marked alteration was observed in MCV, MCH, or MCHC values across different groups (P > 0.05). Table 4. Hematology and differential leukocytic counts of Nile tilapia fingerlings following FO dietary enrichment.
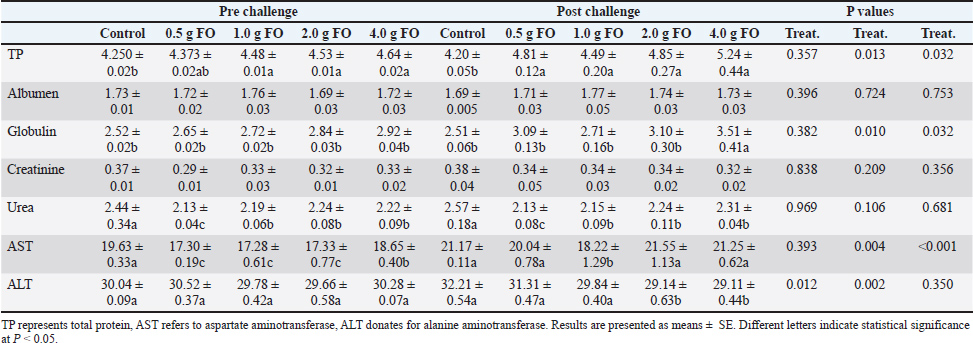
For leukocytic parameters, the total white blood cell (WBC) count markedly increased with increasing FO supplementation levels both pre- and post-A. hydrophila challenge. The highest WBC count was reported in Nile tilapia fingerlings fed with FO supplementation at 4.00 g/kg diet. The count of lymphocytes and monocytes, particularly post-A. hydrophila infection, increased as the dietary FO supplementation level increased, with the highest values at 4.00 g FO/kg diet. However, the counts of heterophils, basophils, and eosinophils did not display a specific pattern either under normal culturing or following A. hydrophila exposure (P > 0.05). In general, dietary FO supplementation up to 4.00 g/kg diet obviously improved the hematological profile and increased the RBC count, Hb concentration, PCV, and total leukocyte count, mainly the counts of lymphocytes and monocytes. These outcomes underscore the role of FO as an effective feed additive that promotes hematopoietic and immune responses. Blood biochemical profile of Nile tilapia fingerlings following fenugreek oil dietary supplementation under normal and A. hydrophila infection conditionsAs shown in Table 5, under normal culturing conditions, dietary FO feeding significantly induced a dose-dependent increase in the total protein levels, peaking at the highest supplementation dose (4.00 g FO/kg diet) compared to the control (P < 0.05). This effect was more pronounced following A. hydrophila challenge, where the highest supplementation dose (4.00 g of FO/kg diet) showed the highest total protein level (5.24 ± 0.44 g/dL) compared to the other groups. Albumen levels displayed minor numerical changes among different supplementation levels, either under normal culturing conditions or following A. hydrophila infection (P > 0.05). However, globulin concentration showed dose-related increases, particularly following A. hydrophila exposure (P < 0.05). Table 5. Blood biochemical profile and liver and kidney function indicators.
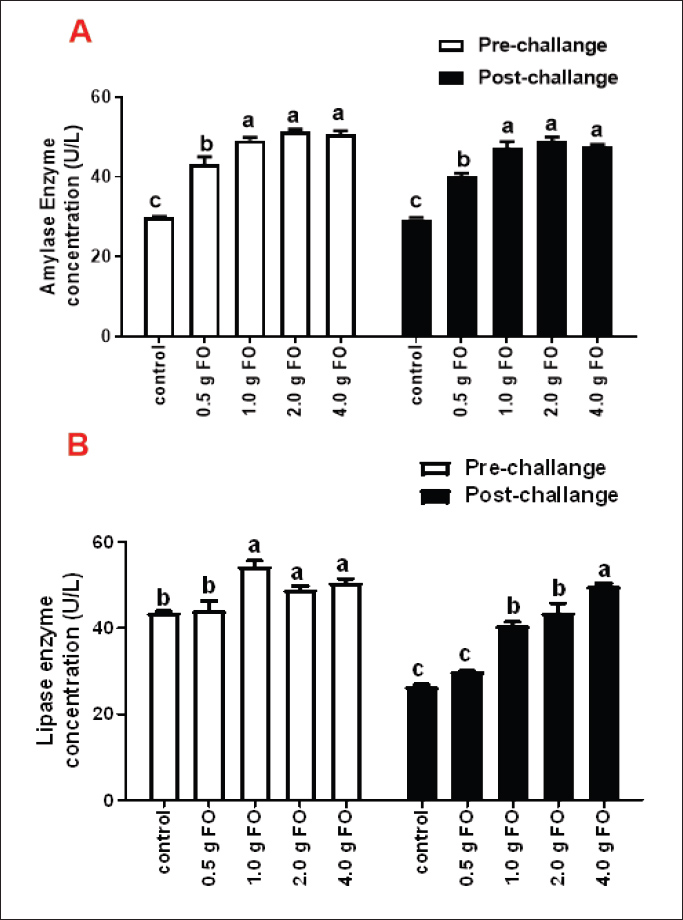
For kidney function markers, creatinine showed nearly stable concentrations across different FO-supplemented groups and control either under normal or following A. hydrophila infection conditions (P > 0.05). However, blood urea levels exhibited significant reductions in all FO-fed groups compared with the control, both under normal culturing conditions and following A. hydrophila challenge. The lowest urea levels were recorded for Nile tilapia fingerlings fed a diet containing 0.5 g FO/kg diet. For liver enzymes, under normal culturing conditions, AST levels were reduced in all FO-supplemented groups compared with the control (P < 0.05). However, A. hydrophila infection significantly elevated its values, which were not influenced by FO supplementation, with little changes compared to the non-supplemented group and those exposed to A. hydrophila (P < 0.05). Under normal conditions, ALT levels displayed little numerical change among the different FO-treated groups and the control (P > 0.05). However, ALT values following A. hydrophila infection significantly decreased across FO-supplemented groups with increasing FO supplementation dose. The lowest values were reported for the 2.00 and 4.00-g FO/kg diet. Concentration of amylase and lipase enzymes in Nile tilapia fingerlings following fenugreek oil supplementation under normal and A. hydrophila infection conditionsThe effect of FO-dietary supplementation on the activity of amylase and lipase enzymes under normal culturing and following A. hydrophila infection is shown in Fig. 1. Under normal culturing conditions, dietary enrichment with different FO levels markedly increased amylase enzyme activity (Fig. 1A) in all FO-containing groups compared with the control (0.0 g FO/kg diet), without significant differences among different FO concentrations (P < 0.05). Amylase activity exhibited a similar pattern following infection with A. hydrophila.
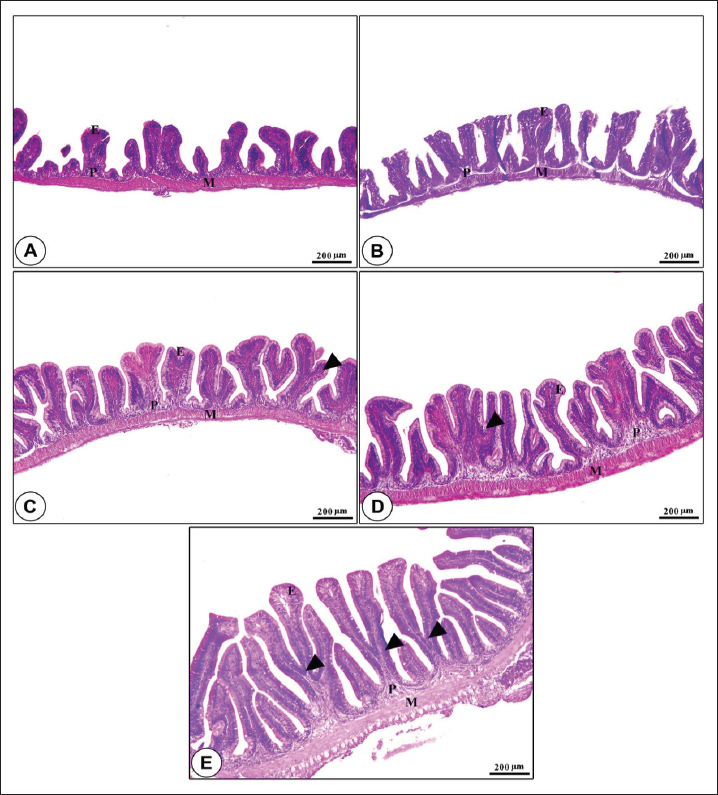
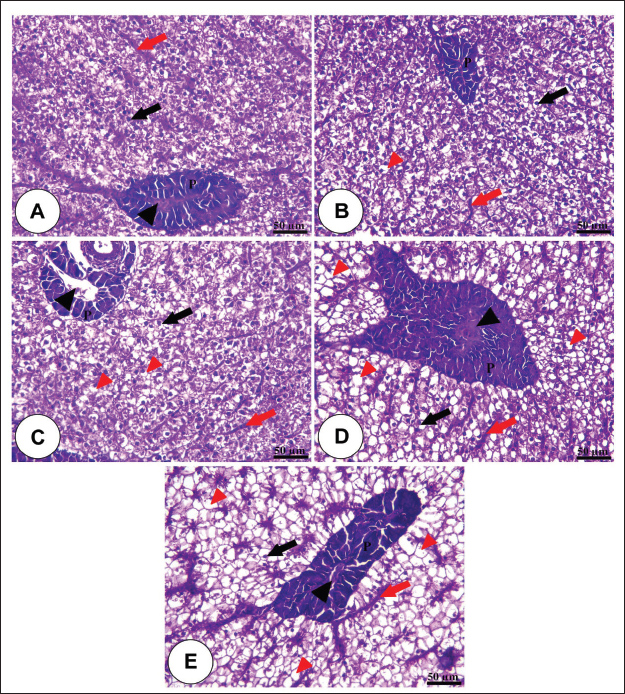
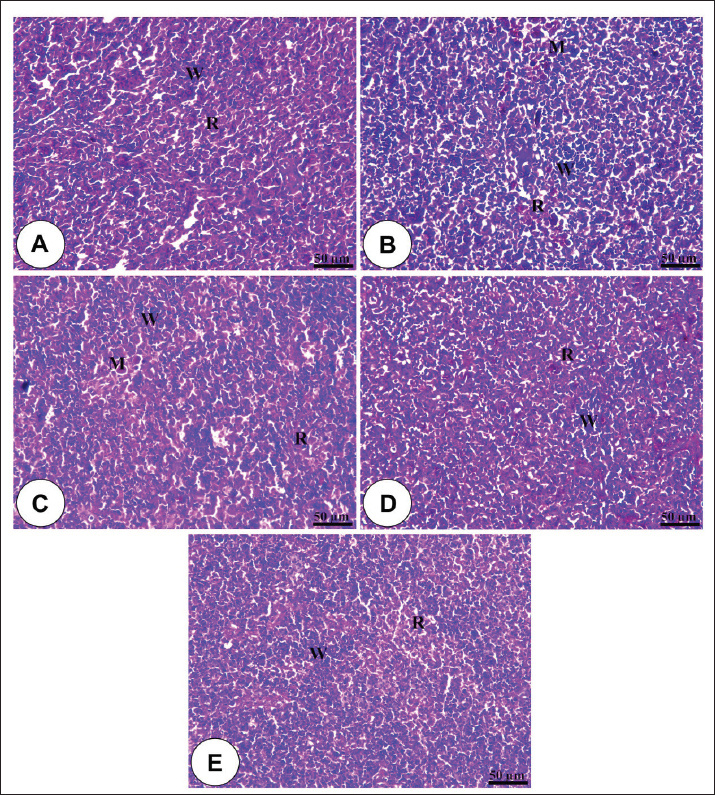
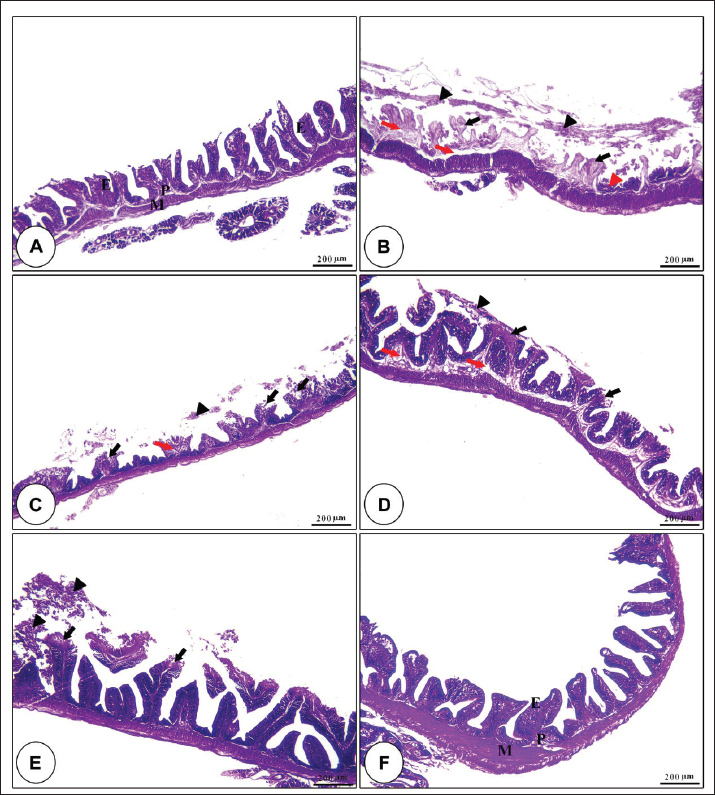
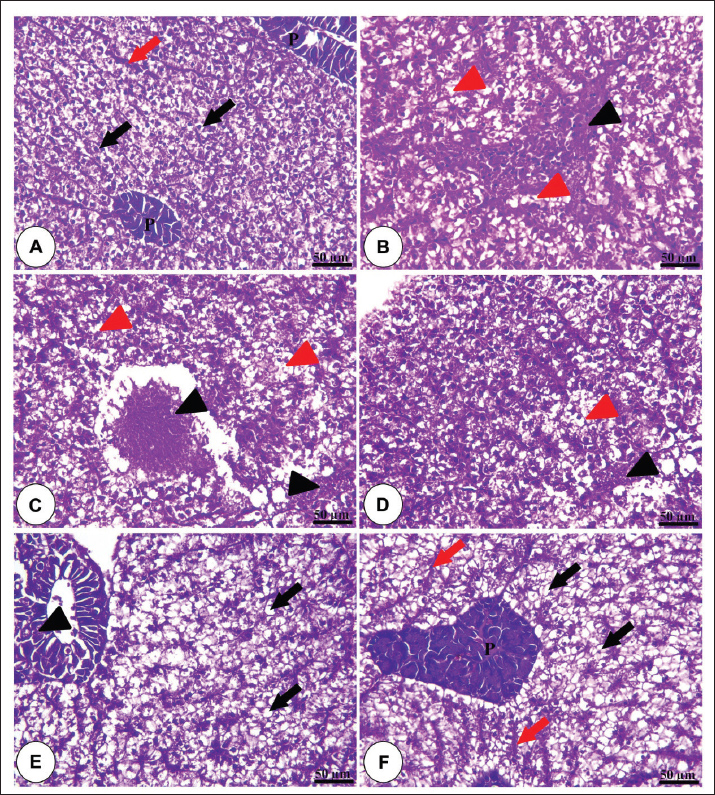
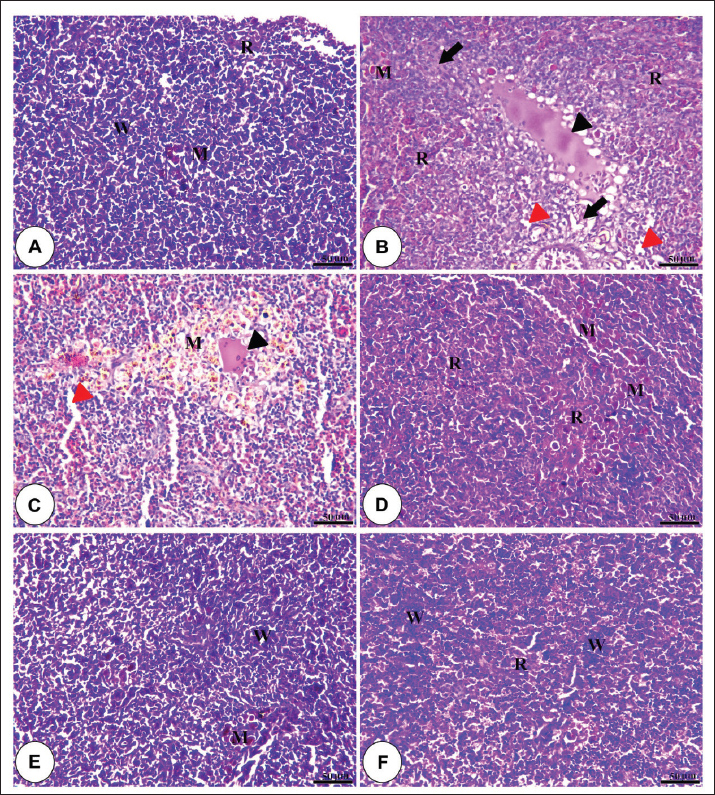
Fig. 1. Amylase and lipase enzyme concentrations following fenugreek oil dietary enrichment at 0.0, 0.5, 1.0, 2.0, and 4.0 g/kg diet under normal and post-A. hydrophila challenge. Results are expressed as means ± SE and different letters indicate statistical significances at p < 0.05. The lipase enzyme activity (Fig. 1B) demonstrated a variable pattern among different groups. The highest lipase activity was measured in the 1.0-g FO/kg diet compared with the control (P < 0.05). Following A. hydrophila infection, lipase enzyme activity did not differ from that under normal culturing conditions (P > 0.05). Histopathological features of the intestine, hepatopancreas, and spleen of Nile tilapia fingerlings in response to FO-dietary supplementation under normal and A. hydrophila infection conditionsUnder normal culturing conditions (before the A. hydrophila challenge)The histological structure of the intestine of all experimental groups did not show any deviation from the normal histological structure; intestinal villi lined by simple columnar epithelium with goblet cells rested on lamina propria of loose connective tissue and lamina muscularis bounded externally with serosal covering (Fig. 2). The morphometric analysis of data obtained from measurement of intestinal villi (Table 6) revealed marked increases in villi length, surface area, and villi length/crypt depth in a dose-dependent manner, especially in the fenugreek oil-containing groups at 2.0 and 4.0 g/kg diet compared with fenugreek oil at 0.5 and 1.0 g. The histological structure of the hepatopancreas of all groups revealed the presence of macrosteatosis in a dose-dependent pattern in all FO-treated groups compared with the non-supplemented group (0.0 g FO/kg diet) (Fig. 3). The microscopic structure of the splenic parenchyma revealed the presence of interconnected cords of red and white pulp in addition to melanomacrophage centers with no evidence of pathological changes (Fig. 4).
Fig. 2. Photomicrograph of H&E-stained panel of the anterior part of intestine in fenugreek oil dietary enrichment at 0 g (A), 0.5 g (B), 1.0 g (C), 2.0 g (D) and 4.0 g (E) groups showing intestinal villi lined by simple columnar epithelium of lamina epithelialis (E), lamina propria (P), and lamina muscularis (M) in addition to the presence of long branched intestinal villi in all fenugreek oil-supplemented groups.
Fig. 3. Photomicrograph of the H&E-stained panel of the hepatopancreas of the fenugreek oil-enriched groups at 0 g (A), 0.5 g (B), 1.0 g (C), 2.0 g (D), and 4.0 g (E) showing polyhedral-shaped hepatocytes (black arrows), hepatic sinusoids (red arrows), pancreatic acini (P), and pancreatic blood vessels (black arrow heads) in addition to the presence of microsteatosis (red arrows) in the fenugreek oil-containing groups.
Fig. 4. Photomicrograph of the H&E-stained panel of the spleen of fenugreek oil dietary enrichment at 0.0 g (A), 0.5 g (B), 1.0 g (C), 2.0 g (D), and 4.0 g (E) groups showing interconnecting cords of red pulp (R) and white pulp (W) in addition to melanomacrophage (M) with an increase in the density of white pulp in fenugreek oil-containing groups. Table 6. Morphometric analysis of the intestine of O. niloticus of different groups.
Postchallenge infection with A. hydrophilaThe microscopic structure of the intestinal villi of the control group (0.0 g fenugreek oil/kg diet) and those challenged with A. hydrophila revealed severe degenerative effects of the epithelial lining along the entire length of the villi, with exposure of the lamina propria and interstitial edema, in addition to the accumulation of necrotic epithelial cells inside the intestinal lumen. These harmful effects were diminished with dietary enrichment with different fenugreek oil concentrations, especially at higher concentrations (Fig. 5).
Fig. 5. H&E-stained panel of the intestine in the control group (0.0 g fenugreek oil [A] group) showing intact lamina epithelialis of intestinal villi (E), lamina propria (P), and lamina muscularis (M). Group (B) represents the 0.0 g FO group challenged with A. hydrophilla showing severe degeneration and sloughing of the lamina epithelialis (black arrowheads), loss of the entire epithelial covering of the alveoli (black arrows), necrosis of the crypt epithelial covering (red arrowheads), and edema in the lamina propria (red arrows). Groups C, D, and E represent the FO supplementation at 0.5, 1, and 2 g/kg diet and challenged with A. hydrophilla showing sloughing of the lamina epithelialis apical part of intestinal villi (black arrow heads), exposure of the apical part of the lamina propria (black arrows), and edema in the lamina propria (red arrows). Group F represents 4.0 g FO and challenged with A. hydrophilla showing intact lamina epithelialis of intestinal villi (E), lamina propria (P), and lamina muscularis (M). The hepatopancreas of Nile tilapia fingerlings following A. hydrophila infection showed severe vacuolar degeneration and necrotic changes in both hepatic and pancreatic cells in the non-supplemented group. These harmful effects were gradually diminished with dietary fenugreek oil enrichment in a dose-dependent pattern. However, the hepatic cells showed microsteatosis in dietary supplementation with 2.0 and 4.0 g of fenugreek oil (Fig. 6).
Fig. 6. H&E-stained panel of hepatopancreas of the control group (0.0 g fenugreek oil [A] group) showing intact hepatocytes (black arrows), blood sinusoids (red arrows), and pancreatic acini (P). Group (B) represents the 0.0 g FO group challenged with A. hydrophilla showing severe necrosis of hepatic and pancreatic tissue (black arrowheads) and vacuolar degeneration of hepatocytes (black arrowheads). Groups C, D, and E represent the FO supplementation at 0.5, 1, and 2 g/kg diet and challenged with A. hydrophilla showing necrosis of hepatic and pancreatic tissue (black arrow heads) and vacuolar degeneration of hepatocytes (black arrow heads). Groups E and F were administered 2.0 and 4.0 g FO, respectively, and were challenged with Aeromonas hydrophila showing mild degenerative changes in pancreatic acini (black arrow head) in 2 g fenugreek oil, in addition to the presence of marked steatosis of hepatocytes (black arrows) and intact blood sinusoids (red arrows). In the non-supplemented group (0.0 g FO/kg diet), the splenic parenchyma showed severe congestion and hemolysis inside splenic blood vessels, degenerative changes of splenic cells, perivascular edema, and depletion of white pulp in response to A. hydrophila infection. These pathological lesions were markedly diminished in a dose-dependent concentration in all FE-enriched groups (Fig. 7).
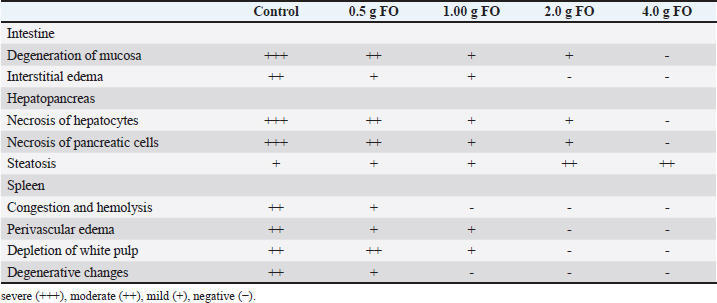
Fig. 7. Photomicrograph of the H&E-stained panel of the spleen of the control group (0.0 g fenugreek oil [A] group) showing interconnecting cords of red pulp (R) and white pulp (W) beside the presence of a melanomacrophage (M). The presence of fenugreek oil at 0.0 g (B), 0.5 g (C), 1.0 g (D), 2.0 g (E), and 4.0 g (F) and A. hydrophilla challenge showing hemolysis inside splenic blood vessels (black arrowhead), degeneration of splenic tissue (black arrows), perivascular edema (red arrowheads), and increased melanomacrophage (M). Table 7 displays the histopathological scoring of detected lesions in the intestine, hepatopancreas, and spleen tissue following A. hydrophila infection. The nonsupplemented (control) group recorded the highest scores for all reported lesions in all examined tissues. These scores decreased, in a dose-dependent manner, with the highest fenugreek oil supplementation level (4.0 g/kg diet) nearly had negative scores for all assessed tissues. Table 7. Histopathological scoring of intestinal, hepatopancreatic and splenic lesions challenged by A. hydrophila.
DiscussionIn aquaculture, research on fenugreek (Trigonella foenum-graecum) oil as a dietary supplement for Nile tilapia fingerlings is limited compared with studies on fenugreek seeds, meal, or extracts. Fenugreek oil, which is extracted from fenugreek’s seeds, is rich in many effective active compounds, such as unsaturated fatty acids (e.g., linoleic and oleic acids), flavonoids, alkaloids, and other volatile compounds, which improve feed palatability, lipid metabolism, antioxidant, and immunity with potential growth-improving effects (Gu et al., 2017; Akbari et al., 2019). However, no direct studies have been conducted on Nile tilapia, with most applications in aquaculture focusing on using fenugreek seed, meal, or extracts, and only a few studies have documented its effect in poultry (Hasanain N. Ezzat, 2023). Thus, in the current study, dietary FO supplementation has been proven to exert marked benefits on Nile tilapia’ physio-biochemical, antioxidant, and immunological responses, particularly under A. hydrophila exposure. These impacts were correlated with numerical improvement in growth and feed utilization indices. Most of these findings peaked at 4.0-g FO/kg diet. The growth outcomes showed consistent numerical improvements in all assessed growth parameters, including final body weight, total weight gain, FCR, PER, and SGR. These findings are in line with those of (Abbas et al., 2019; Moustafa et al., 2020; Diab et al., 2023) who reported improvements in Nile tilapia growth performance in response to dietary supplementation of fenugreek seeds. (Awad et al., 2015) stated that dietary supplementation of fenugreek enhanced the growth performance of gilthead seabream (Sparus aurata). All of these studies argued the improved growth performance to the bioactive compounds in fenugreek products, including its seeds, extracts, and oily form, such as flavonoids, saponins, alkaloids, polyphenols, and tannins which enhance feed palatability, nutrient digestibility and absorption, feed utilization, and the metabolic status (Olaiya and Soetan, 2014; Yu et al., 2019). Improving nutrient absorption in response to FO dietary supplementation might be correlated with improving the intestinal architecture, as it caused marked increases in villi length, surface area, and villi length/crypt depth, especially at the 2–4 g of FO/kg diet. The improved effect of FO on intestinal morphometric indices is probably correlated with its flavonoids contents, which have a magic antioxidant ability to reduce the formation of reactive oxygen species (ROS) and stress-related biomarkers (Brunetti et al., 2013; Moustafa et al., 2020). It may also be linked to the improvement of the structure enterocytes (Moustafa et al., 2020). Moreover, the reported improvement in Nile tilapia’s growth performance in response to FO-dietary supplementation might be correlated with enhanced lipase and amylase activities, which are possibly associated with the improved intestinal structure and would enhance nutrient digestion, metabolism, and absorption (Sherif et al., 2024). However, there are mixed growth benefits in most fenugreek-related studies: some studies reported significant enhancement of growth performance, while others reported only nonsignificant numerical responses or no effect on growth. These contradictory outcomes might be associated with differences in the supplementation or inclusion rate, form of fenugreek (whole seed, oil, methanolic or ethanolic extract, and fraction of bioactive compounds), feeding trial duration, fish development stage (fingerlings, juveniles, or market size), and sample size. The FO also exerted notable physiological, antioxidant, and immunological benefits in Nile tilapia fingerlings, particularly following A. hydrophila exposure. It enhanced the antioxidant defense of Nile tilapia fingerlings by increasing the enzyme activity of SOD and CAT with an obvious reduction in MDA content, particularly at 2–4 g FO/kg diet and A. hydrophila challenge. These effects are probably correlated with the content of FO of steroidal saponins, such as diosgenin, flavonoids, galactomannans, and phenolic compounds (Bahubali and Bornare, 2021; Gavahian et al., 2024). All these bioactive compounds are characterized by their ability to scavenge free radicals and enhance the synthesis of antioxidant enzymes (Moustafa et al., 2020). Alike our findings, feeding gilthead seabream (Sparus aurata) a diet containing fenugreek markedly enhanced the SOD and GPx levels (Awad et al., 2015). The authors also argued that this enhancement to the fenugreek’s high content of numerous bioactive compounds might improve the antioxidant status and alleviate the excessive ROS production associated with microbial infection (Awad et al., 2015; Awad and Awaad, 2017). For example, the presence of phenolic acid, gallic acid, and quercetin, as potent antioxidants, in fenugreek would increase the antioxidant properties by creating hybrids with other phenolics, such as caffeic acid (Badhani et al., 2015; Bahubali and Bornare, 2021; Gavahian et al., 2024). Moreover, it has been shown that bioactive compounds of fenugreek could improve the antioxidant capacity by activating several pathways, such as the antioxidant transcriptional pathway. The KEAP1/Nrf2 axis and AMPK pathway in mammalian cells and the Nrf2-mediated pathway in fish upregulate antioxidant enzyme activities (Alsuliam et al., 2022). Therefore, further studies are recommended to deeply elucidate the mechanism by which FO regulates growth, physiological, immunity, and antioxidant properties. The improved antioxidant capacity in response to dietary FO supplementation was also linked, with consistent dose-dependent increases in immune-related parameters, such as PA, PI, lysozyme concentration, total leukocyte count, and lymphocyte and monocyte count, especially under A. hydrophila challenge. These findings might confirm the immunostimulant properties of FO to stimulate innate immunity and enhance resistance to A. hydrophila infection in Nile tilapia, as non-specific immunity is the first line of defense against infection (Van Doan et al., 2017). Similar studies have reported the improvement of non-specific immunity in various fish species in response to long-term dietary enrichment with medicinal plants, including fenugreek (Awad and Awaad, 2017). Accordingly, dietary administration of fenugreek seed powder significantly increased lysozyme activity and IgM and NBT concentrations in Nile tilapia (Moustafa et al., 2020). The reported increases in lysozyme activity might be linked with the fenugreek’s high content of flavonoids, which stimulate the lysozyme activity, phagocytosis, and leukocytes (Awad et al., 2015; Moustafa et al., 2020). At the hematological level, the reported dose-dependent increases in the RBC count, Hb concentration, and PCV values without changes in the MCV, MCH, and MCHC values possibly signify the stimulatory effect of FO dietary enrichment on erythropoiesis while maintaining the shape of blood cells. These findings may be correlated with the improved nutritional status and reduced oxidative damage to blood cells of Nile tilapia fingerlings following FO dietary supplementation, particularly under A. hydrophila challenge. These improved hematological indices are possibly mediated by the antioxidant and anti-inflammatory effects of bioactive compounds such as flavonoids and phenolics (Bahmani et al., 2016; Awad and Awaad, 2017). Besides, the improved hematological picture would suggest improved hepatic functions, as confirmed by the concurrent reduction in the levels of ALT and AST enzymatic activities and alleviation of hepatopancreatic necrosis linked with the A. hydrophila challenge. Similar supporting hematological impacts were reported in response to dietary enrichment with fenugreek seed powder in Nile tilapia, and in common carp (Awad et al., 2015; Moustafa et al., 2020). Moreover, increasing the total leukocytes, lymphocytes, and monocytes counts with FO supplementation may reflect the enhanced non-specific innate immunity of Nile tilapia fingerlings and a stronger resistance to A. hydrophila. These impacts may be related to fenugreek-derived flavonoids and saponins, which are well-known to improve lymphocyte and macrophage functions via cytokine signaling and membrane receptors stimulation (Pham et al., 2020), suggesting an improved innate immunity and immunological balance with FO-dietary enrichment during A. hydrophila infection. At the biochemical level, FO supplementation improved total protein, albumen, and globulin concentrations, which correlated with improved kidney function, as confirmed by the reduced urea and creatinine levels, especially under A. hydrophila infection. Again, all these findings probably reflect the tissue protective effects of FO. This tissue protective effect of FO in the spleen, intestinal, and hepatopancreatic tissues, particularly in A. hydrophila infection. Dietary FO supplementation caused dose-dependent improvement in the histological architectures of these tissues and mitigated the histopathological lesions and the tissue-damaged effects of A. hydrophila infection. These influences may also be, attributed to the enriched content of FO of bioactive compound with anti-inflammatory, antioxidant, and immunomodulatory properties (Bahubali and Bornare, 2021). In a similar context, an improvement in the liver’s histological structure was reported in silver bream (Vimba vimba) supplemented with natural feed additives (Ostaszewska et al., 2008). Sylimarin, a natural additive, has been shown to lower the hepatic and splenic pathological alteration of Nile tilapia infected with Streptococcus agalactiae (Owatari et al., 2018). The presence of pathological lesions in tissue reflects oxidative damage caused by microbial infection, e.g., A. hydrophila infection. Thus, the reported alleviation of these lesions in the FO-supplemented Nile tilapia fingerlings may be associated with the enriched content of bioactive compounds, such as flavonoids, that could lower A. hydrophila-associated stress by reducing ROS synthesis and its linked harmful effects (Brunetti et al., 2013). ConclusionIn summary, the current study investigated the impact of 8 weeks of dietary supplementation with different FO levels on Nile tilapia performance and resistance to A. hydrophila challenge. The results revealed that dietary supplementation of FO at 2–4 g/kg diet enhanced the physio-biochemical status, antioxidant capacity, innate immunity, and tissue resilience of Nile tilapia fingerlings to A. hydrophila challenge. These benefits were linked with numerical improvement in growth performance. Therefore, FO dietary supplementation at 2–4 g/kg diet is strongly recommended as a natural health-promoting feed additive in tilapia diet. Future studies that explore the molecular mechanisms of antioxidant redox, immune regulators, and growth performance are highly recommended. AcknowledgmentThe authors would like to thank the participants and staff involved in this study for their invaluable contributions. FundingNo funding sources. Authors' contributionsEman M. Moustafa: performed the experiment, collected data, and wrote the original draft of the manuscript. Safaa Abdo: carried out the experiment, collected data, and performed formal analysis. Seham El-Kassas : collected data, performed formal analysis, conceived and planned the experiments, and wrote and reviewed the final paper and manuscript. Mohamed M. Zayed: carried out the experiment, collected data, and cultured A. hydrophila. Rasha A. Al Wakeel: biochemical and hematological analysis. Yasser A. Menesy: performed the experiment and collected data. Amira A. Omar: performed the experiment and collected data. All authors provided critical feedback and helped shape the research, analysis, and manuscript. Conflict of interestThe authors have no potential conflicts of interest to report. Data availabilityAll data supporting the results or analyses are presented in the paper and its supplementary files. ReferencesAbaho, I., Masembe, C., Akoll, P. and Jones, C.L. 2022. The use of plant extracts to control tilapia reproduction: current status and future perspectives. J. World. Aquac. Soc. 53(3), 593–619. Abbas, W.T., Abumourad, I.M., Mohamed, L.A., Abbas, H.H., Authman, M., Soliman, W.S. and Elgendy, M.Y. 2019. The Role of the Dietary Supplementation of Fenugreek Seeds in Growth and Immunity in Nile Tilapia with or without Cadmium Contamination. Jordan. J. Biol. Sci. 12(5), 649. Abdo, S.E., El-Nahas, A.F., Abdellatif, R.E., Mohamed, R., Helal, M.A., Azzam, M.M., Di Cerbo, A. and El-Kassas, S. 2024. Combined dietary Spirulina platensis and Citrus limon essential oil enhances the growth, immunity, antioxidant capacity and intestinal health of Nile Tilapia. Vet. Sci. 11(10), 474. Ahmadifar, E., Pourmohammadi Fallah, H., Yousefi, M., Dawood, M.A.O., Hoseinifar, S.H., Adineh, H., Yilmaz, S., Paolucci, M. and Doan, H.V. 2021. The gene regulatory roles of herbal extracts on the growth, immune system, and reproduction of fish. Animals 11(8), 2167. Akbari, S., Abdurahman, N.H., Yunus, R.M., Alara, O.R. and Abayomi, O.O. 2019. Extraction, characterization and antioxidant activity of fenugreek (Trigonella-Foenum Graecum) seed oil. Mater. Sci. Energy. Technol. 2(2), 349–355. Alsuliam, S.M., Albadr, N.A., Almaiman, S.A., Al-Khalifah, A.S., Alkhaldy, N.S. and Alshammari, G.M. 2022. Fenugreek seed galactomannan aqueous and extract protects against diabetic nephropathy and liver damage by targeting NF-κB and Keap1/Nrf2 Axis. Toxics 10(7), 362. Awad, E. and Awaad, A. 2017. Role of medicinal plants on growth performance and immune status in fish. Fish. &. Shellfish. Immunol. 67, 40–54. Awad, E., Cerezuela, R. and Esteban, M.A. 2015. Effects of fenugreek (Trigonella foenum graecum) on gilthead seabream (Sparus aurata L.) immune status and growth performance. Fish Shellfish Immunol. 45(2), 454–464. Badhani, B., Sharma, N. and Kakkar, R. 2015. Gallic acid: a versatile antioxidant with promising therapeutic and industrial applications. Rsc. Adv. 5(35), 27540–27557. Bahmani, M., Shirzad, H., Mirhosseini, M., Mesripour, A. and Rafieian-Kopaei, M. 2016. A review on ethnobotanical and therapeutic uses of fenugreek (Trigonella foenum-graceum L). J. Evid. Based. Complementary. Altern. Med. 21(1), 53–62. Bahubali, M.B.S. and Bornare, D. 2021. Study of Bioactive Components Present in Oil Extracted from Fenugreek Seed. Int. J. Agric. Food Sci. 5(5), 14–17. Bain, B.J., Bates, I. and Laffan, M.A. 2016. Dacie and Lewis Practical Haematology E-Book. London, UK: Elsevier Health Sciences. Bancroft, J.D. and Gamble, M. 2008. Theory and practice of histological techniques. London, UK: Elsevier health sciences. Elsevier health sciences. Brunetti, C., Di Ferdinando, M., Fini, A., Pollastri, S. and Tattini, M. 2013. Flavonoids as antioxidants and developmental regulators: relative significance in plants and humans. Int. J. Mol. Sci. 14(2), 3540–3555. Council, N.R., 2011. Nutrient requirements of fish and shrimp. NRC, Oslo. Dawood, M.A.O., Abdel-Kader, M.F., Moustafa, E.M., Gewaily, M.S. and Abdo, S.E. 2020. Growth performance and hemato-immunological responses of Nile tilapia (Oreochromis niloticus) exposed to deltamethrin and fed immunobiotics. Environ. Sci. Pollut. Res. 27(11), 11608–11617. Degani, G., Viola, S. and Levanon, D. 1986. Effects of dietary carbohydrate source on growth and body composition of the European eel (Anguilla anguilla L.). Aquaculture 52(2), 97–104. Demers, N.E. and Bayne, C.J. 1997. The immediate effects of stress on hormones and plasma lysozyme in rainbow trout. Dev. Comp. Immunol. 21(4), 363–373. Diab, A.M., Al-Khefa, B.T., Khalafallah, M.M., Salah, A.S., Farrag, F.A. and Dawood, M.A.O. 2023. Dietary methanolic extract of fenugreek enhanced the growth, haematobiochemical, immune responses, and resistance against Aeromonas hydrophila in Nile Tilapia, Oreochromis niloticus. Aquac. Res. 1(1), 3055476. Gabriel, N.N. 2019. Review on the progress in the role of herbal extracts in tilapia culture. Cogent Food Agric. 5(1), 1619651. Gavahian, M., Bannikoppa, A.M., Majzoobi, M., Hsieh, C.W., Lin, J. and Farahnaky, A. 2024. Fenugreek bioactive compounds: a review of applications and extraction based on emerging technologies. Crit. Rev. Food. Sci. Nutr. 64(28), 10187–10203. Gu, L.B., Liu, X.N., Liu, H.M., Pang, H.L. and Qin, G.Y. 2017. Extraction of fenugreek (Trigonella foenum-graceum L.) seed oil using subcritical butane: characterization and process optimization. Molecules 22(2), 228. Hamed, S., El-Kassas, S., Abo-Al-Ela, H.G., Abdo, S.E., Al Wakeel, R.A., Abou-Ismail, U.A. and Mohamed, R.A. 2024. Interactive effects of water temperature and dietary protein on Nile tilapia: growth, immunity, and physiological health. BMC Vet. Res. 20(1), 349. Hasanain, N.E. 2023. Effect of supplementation fenugreek oil to the diet on the physiological, anatomical and histological traits of broilers. Iraqi J. Agric. Sci. 54(1), 106–113. Hawk, P.B., 1965. Hawk's physiological chemistry. (No Title). Houston, A., 1990. Blood and circulation. Methods for fish biology. 415–488. Jauncey, K. and Ross, B. 1982. A guide to tilapia feeds and feedings. University of Stirling, Institute of Aquaculture. Kawahara, E., Ueda, T. and Nomura, S. 1991. In vitro Phagocytic-activity of white-spotted char blood-cells after injection with aeromonas-salmonicida extracellular products. Japan Soc Fish Pathol Dept Fisheries-Fac Agr Academy Center 358, 213–214. Khalafalla, M.M., Ibrahim, S.A., Zayed, M.M., Awad, M.N. and Mohamed, R.A. 2020. Effect of a dietary mixture of beneficial bacteria on growth performance, health condition, chemical composition, and water quality of Nile Tilapia, Oreochromis niloticus fingerlings. J. Aquat. Food Prod. Technol. 29(8), 823–835. Kuebutornye, F.K.A. and Abarike, E.D. 2020. The contribution of medicinal plants to tilapia aquaculture: a review. Aquac. Int. 28(3), 965–983. Li, J., Ni, X.D., Liu, Y.J. and Lu, C.P. 2011. Detection of three virulence genes alt, ahp and aerA in Aeromonas hydrophila and their relationship with actual virulence to zebrafish. J. Appl. Microbiol. 110(3), 823–830. Moustafa, E.M., Dawood, M.A.O., Assar, D.H., Omar, A.A., Elbialy, Z.I., Farrag, F.A., Shukry, M. and Zayed, M.M. 2020. Modulatory effects of fenugreek seeds powder on the histopathology, oxidative status, and immune related gene expression in Nile tilapia (Oreochromis niloticus) infected with Aeromonas hydrophila. Aquaculture 515, 734589. Olaiya, C.O. and Soetan, K.O. 2014. A review of the health benefits of fenugreek (Trigonella foenum-graecum L.): nutritional, Biochemical and pharmaceutical perspectives. Am. J. Soc. Issues Humanit. 4, 3–12. Ostaszewska, T., Dabrowski, K., Hliwa, P., Gomółka, P. and Kwasek, K. 2008. Nutritional regulation of intestine morphology in larval cyprinid fish, silver bream (Vimba vimba). Aquac. Res. 39(12), 1268–1278. Owatari, M.S., Alves Jesus, G.F., Brum, A., Pereira, S.A., Lehmann, N.B., De Pádua Pereira, U., Martins, M.L. and Pedreira Mouriño, J.L. 2018. Sylimarin as hepatic protector and immunomodulator in Nile tilapia during Streptococcus agalactiae infection. Fish Shellfish Immunol. 82, 565–572. Pham, D.C., Shibu, M.A., Mahalakshmi, B. and Velmurugan, B.K. 2020. Effects of phytochemicals on cellular signaling: reviewing their recent usage approaches. Crit. Rev. Food Sci. Nutr. 60(20), 3522–3546. Reed, L.J. and Muench, H. 1938. A simple method of estimating 50 per cent end points. Am. J. Hyg. 27, 493–497. Sherif, E.M., Abd El-razek, I.M., El-Sharawy, M.E., Amer, A.A., Zaineldin, A.I., Gewaily, M.S., Ashry, A.M., Younis, N.A., Ahmed, H.A. and Dawood, M.A.O. 2024. Growth performance, antioxidative status, and immune response of Nile tilapia (Oreochromis niloticus) fed dietary fermented Spirulina platensis. Aquac. Rep. 39, 102324. Siddiqui, A.Q., Howlader, M.S. and Adam, A.A. 1988. Effects of dietary protein levels on growth, feed conversion and protein utilization in fry and young Nile tilapia, Oreochromis niloticus. Aquaculture 70(1-2), 63–73. Thrall, M.A., Weiser, G., Allison, R.W. and Campbell, T.W. 2012. Veterinary hematology and clinical chemistry. London, UK: John Wiley & Sons. Van Doan, H., Hoseinifar, S.H., Dawood, M.A.O., Chitmanat, C. and Tayyamath, K. 2017. Effects of Cordyceps militaris spent mushroom substrate and Lactobacillus plantarum on mucosal, serum immunology and growth performance of Nile tilapia (Oreochromis niloticus). Fish Shellfish Immunol. 70, 87–94. Wintrobe, M.M. 1934. Variations in the size and hemoglobin content of erythrocytes in the blood of various vertebrates. Folia. Haematol. 51(32), 32–49. Yu, H., Liang, H., Ren, M., Ji, K., Yang, Q., Ge, X., Xi, B. and Pan, L. 2019. Effects of dietary fenugreek seed extracts on growth performance, plasma biochemical parameters, lipid metabolism, Nrf2 antioxidant capacity and immune response of juvenile blunt snout bream (Megalobrama amblycephala). Fish. Shellfish. Immunol. 94, 211–219. | ||
| How to Cite this Article |
| Pubmed Style Moustafa EM, Abdo S, El-kassas S, Zayed MM, Wakeel RAA, Menesy YA, Omar AA. Effects of dietary supplementation with fenugreek oil on growth, physiology, and resistance to A. hydrophila infection in Nile tilapia (Oreochromis niloticus) fingerlings. Open Vet. J.. 2025; 15(12): 6747-6766. doi:10.5455/OVJ.2025.v15.i12.54 Web Style Moustafa EM, Abdo S, El-kassas S, Zayed MM, Wakeel RAA, Menesy YA, Omar AA. Effects of dietary supplementation with fenugreek oil on growth, physiology, and resistance to A. hydrophila infection in Nile tilapia (Oreochromis niloticus) fingerlings. https://www.openveterinaryjournal.com/?mno=292349 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.54 AMA (American Medical Association) Style Moustafa EM, Abdo S, El-kassas S, Zayed MM, Wakeel RAA, Menesy YA, Omar AA. Effects of dietary supplementation with fenugreek oil on growth, physiology, and resistance to A. hydrophila infection in Nile tilapia (Oreochromis niloticus) fingerlings. Open Vet. J.. 2025; 15(12): 6747-6766. doi:10.5455/OVJ.2025.v15.i12.54 Vancouver/ICMJE Style Moustafa EM, Abdo S, El-kassas S, Zayed MM, Wakeel RAA, Menesy YA, Omar AA. Effects of dietary supplementation with fenugreek oil on growth, physiology, and resistance to A. hydrophila infection in Nile tilapia (Oreochromis niloticus) fingerlings. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6747-6766. doi:10.5455/OVJ.2025.v15.i12.54 Harvard Style Moustafa, E. M., Abdo, . S., El-kassas, . S., Zayed, . M. M., Wakeel, . R. A. A., Menesy, . Y. A. & Omar, . A. A. (2025) Effects of dietary supplementation with fenugreek oil on growth, physiology, and resistance to A. hydrophila infection in Nile tilapia (Oreochromis niloticus) fingerlings. Open Vet. J., 15 (12), 6747-6766. doi:10.5455/OVJ.2025.v15.i12.54 Turabian Style Moustafa, Eman M., Safaa Abdo, Seham El-kassas, Mohamed M. Zayed, Rasha A. Al Wakeel, Yasser A. Menesy, and Amira A. Omar. 2025. Effects of dietary supplementation with fenugreek oil on growth, physiology, and resistance to A. hydrophila infection in Nile tilapia (Oreochromis niloticus) fingerlings. Open Veterinary Journal, 15 (12), 6747-6766. doi:10.5455/OVJ.2025.v15.i12.54 Chicago Style Moustafa, Eman M., Safaa Abdo, Seham El-kassas, Mohamed M. Zayed, Rasha A. Al Wakeel, Yasser A. Menesy, and Amira A. Omar. "Effects of dietary supplementation with fenugreek oil on growth, physiology, and resistance to A. hydrophila infection in Nile tilapia (Oreochromis niloticus) fingerlings." Open Veterinary Journal 15 (2025), 6747-6766. doi:10.5455/OVJ.2025.v15.i12.54 MLA (The Modern Language Association) Style Moustafa, Eman M., Safaa Abdo, Seham El-kassas, Mohamed M. Zayed, Rasha A. Al Wakeel, Yasser A. Menesy, and Amira A. Omar. "Effects of dietary supplementation with fenugreek oil on growth, physiology, and resistance to A. hydrophila infection in Nile tilapia (Oreochromis niloticus) fingerlings." Open Veterinary Journal 15.12 (2025), 6747-6766. Print. doi:10.5455/OVJ.2025.v15.i12.54 APA (American Psychological Association) Style Moustafa, E. M., Abdo, . S., El-kassas, . S., Zayed, . M. M., Wakeel, . R. A. A., Menesy, . Y. A. & Omar, . A. A. (2025) Effects of dietary supplementation with fenugreek oil on growth, physiology, and resistance to A. hydrophila infection in Nile tilapia (Oreochromis niloticus) fingerlings. Open Veterinary Journal, 15 (12), 6747-6766. doi:10.5455/OVJ.2025.v15.i12.54 |






 , where W1=final body weight, W0=initial body weight (
, where W1=final body weight, W0=initial body weight ( (
(