| Research Article | ||
Open Vet. J.. 2026; 16(2): 907-919 Open Veterinary Journal, (2026), Vol. 16(2): 907-919 Research Article Growth performance, antioxidant capacity, and microbial modulation in broilers supplemented with Arabic gum under a small-scale production systemSaif Ali Mahmoud Alkassar1, Ali Mahmood Amer Alkassar2, Ahmed Khalid Ahmed3, Hanan Al-Khalaifah4, Moustafa Amin Osman5, Reda El-Sayed Hamouda6 , and Mai Ahmed Maher Youssif6*1Animal Production Department, College of Agriculture, Kufa University, Najaf, Iraq 2Public Health Branch, Faculty of Veterinary Medicine, Kufa University, Najaf, Iraq 3Department of Animal Production, College of Agriculture, Tikrit University, Tikrit, Iraq 4Environment and Life Sciences Research Center, Kuwait Institute for Scientific Research, Kuwait City, Kuwait 5Regional Center for Food and Feed, Agricultural Research Center, Ministry of Agriculture and Land Reclamation, Giza, Egypt 6Department of Animal Production Systems Research, Animal Production Research Institute, Agricultural Research Center, Ministry of Agriculture and Land Reclamation, Giza, Egypt *Corresponding Author: Mai Ahmed Maher Youssif. Department of Animal Production Systems Research, Animal Production Research Institute, Agricultural Research Center, Ministry of Agriculture and Land Reclamation, Giza, Egypt. Email: maiahmed25.24.23 [at] gmail.com Submitted: 22/10/2025 Revised: 20/12/2026 Accepted: 05/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
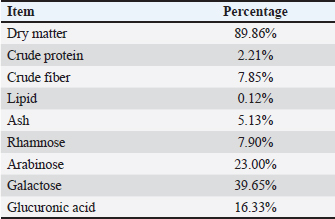
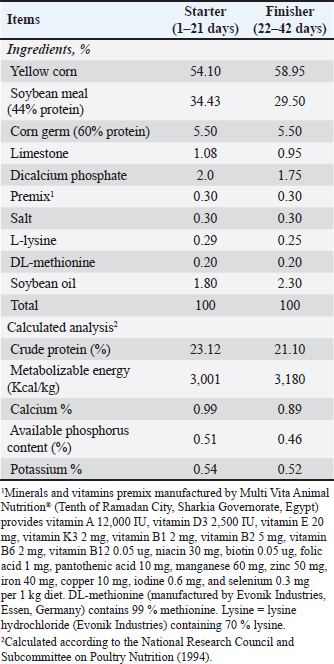
AbstractBackground: Arabic gum is a soluble fiber that acts as a natural prebiotic and can be fermented by intestinal bacteria. This characteristic can enhance broiler growth, blood biomarkers, and carcass traits. Aim: This study aimed to evaluate the effects of Arabic gum (AG) on growth performance traits, carcass characteristics, blood biochemistry, cecal microbial load, and economic efficiency in Ross 308 broilers raised under a small-scale production system. Methods: Five treatment groups were randomly assigned, with each group consisting of 6 replicates of 15 one-day-old Ross 308 broiler chicks, totaling 450 chicks. The first group served as the control (CON) and received only the basal diet, whereas the second, third, fourth, and fifth groups were fed the basal diet supplemented with 0.25%, 0.50%, 0.75%, and 1.0% AG, respectively. The experiment was continued until 42 days of age. Results: Supplementation with AG at all levels, especially a level of 1.0% AG, significantly (p < 0.05) improved live body weight, body weight gain, feed intake, feed conversion ratio and performance index. Carcass traits were also significantly enhanced (p < 0.05), and carcass, breast, and thigh yields were significantly increased, whereas bursa percentage decreased. Blood biochemical parameters were markedly improved; the AG-treated groups exhibited significantly higher (p < 0.05) levels of total protein, albumin, globulin, immunoglobulins (IgM, IgA, and IgY), antioxidant enzymes, and liver enzymes compared with the CON group, along with reduced urea, creatinine, and malondialdehyde levels. AG also significantly (p < 0.05) altered cecal microbiota; the 1.0% group showed the highest total bacterial counts and Lactobacillus populations. Conclusion: AG supplementation in broiler diets may improve intestinal flora, immunity, antioxidant activity, and overall productivity. Keywords: Antioxidant enzymes, Blood biochemistry, Carcass traits, Cecal microbiota, Feed conversion ratio. IntroductionThe demand for meat, particularly chicken, has increased significantly over the past few decades. In 2020, poultry became the most popular meat consumed globally in 2020 (Bist et al., 2024; Kleyn and Ciacciariello, 2025). Consequently, efforts have been made to enhance the production efficiency of traditional broiler chicken systems (Gharib et al., 2023). The poultry industry, especially small-scale production systems, has focused on increasing body weight and growth rate while reducing feed intake (FI) to improve broiler growth performance (Gadzama, 2024; Hamouda et al., 2025). Additionally, antibiotics have been used in chicken production for decades to promote growth; however, this practice has led to the development of bacterial resistance and residues in chicken eggs and meat (Atay, 2023; Elbaz et al., 2025). Therefore, instead of relying on conventional antibiotics to enhance animal growth, natural additives, such as powders and extracts derived from aromatic and medicinal herbs, have been increasingly used (Ashour et al., 2025a; Gabriel et al., 2025; El-Abbasy et al., 2025). Arabic gum (AG), a natural exudate from Acacia senegal, has gained attention as a chicken feed additive due to its prebiotic properties and potential advantages for growth efficiency, carcass quality, and physiological parameters (Al-Baadani et al., 2023a). Furthermore, AG consists mostly of soluble dietary fibers (80%–85%), which are indigestible but fermentable by intestinal bacteria (Al-Baadani et al., 2024). Additionally, short-chain fatty acids (SCFAs) generated during AG fermentation promote intestinal integrity, regulate immunological responses, and prevent the colonization of harmful bacteria, thereby improving the growth and overall health of chickens (Al-Baadani et al., 2022). Additionally, Khan et al. (2022) found that supplementing broiler chickens with AG at 0.12%–1.5% enhances body weight gain (BWG) and feed efficiency. Specifically, 1.5% AG significantly increased FI and BWG while reducing harmful intestinal bacteria, such as Escherichia coli and Salmonella, indicating improved microbial balance and overall health. Similarly, the administration of AG at concentrations ranging from 0.12% to 0.75% boosted development performance and augmented the dimensions of immune organs, as indicated by an increased thymus weight, implying enhanced immune function (Tabidi and Ekram, 2015; Al-Baadani et al., 2022). Moreover, supplementation with AG has been associated with reduced mortality rates and an elevated European production efficiency factor, indicating enhanced survival rates and economic advantages in chicken production (Hassan et al., 2024). In the present study, blood biochemical parameters responded positively to AG supplementation, as evidenced by reduced blood cholesterol and glucose levels, indicating improved metabolic health and potentially decreased stress in avian species (Khan et al., 2022). The shift away from antibiotic growth promoters (AGPs), driven by concerns over resistance and consumer demand for antibiotic-free chicken, has heightened interest in natural alternatives such as AG (Al-Baadani et al., 2023b). The ability of AG to enhance intestinal architecture, strengthen immune responses, and inhibit harmful microbes makes it a promising candidate for sustainable broiler production (Al-Baadani et al., 2022). Based on the documented prebiotic, immunomodulatory, and antioxidative features of AG, the study hypothesizes that supplementing broilers’ diet with AG will boost their growth performance traits, carcass quality, immunity, and antioxidant capacity, and beneficially modulate the cecal microbiota of broiler chickens raised under small-scale production systems. Most previous studies have evaluated the effects of AG, focusing on commercial-scale broiler production systems in Egypt. However, none have focused on small-scale production systems, and none have combined growth performance, carcass traits, blood biochemistry, antioxidant capacity, immune indicators, cecal microbiota, and economic efficiency in a single experiment. Therefore, this study aimed to evaluate the effect of AG in the diet of broiler chickens on growth performance, carcass traits, blood biochemical parameters, cecal microbiota, and economic efficiency under small-scale production systems. It was hypothesized that using AG as a safe and healthy feed additive could improve broiler growth performance, carcass traits, antioxidant capacity, immune response, and economic efficiency. Additionally, the study seeks to provide evidence-based recommendations to improve the sustainability and resilience of small-scale broiler production systems. Materials and MethodsEthical approvalThis investigation was conducted on a private small-scale household broiler producer farm under the supervision of the Animal Production Research Institute, Agriculture Research Center. The tested additives, experimental samples, and diets were chemically analyzed in the Animal Production Research Institute Labs, Agriculture Research Center. The Agricultural Research Center-Institutional Animal Care and Use Committee (ARC-IACUC) assessed and approved all experimental protocols (ARC-CLAR-5129). The approvals ensured that all research activities complied with ethical standards for the treatment and utilization of animals. Birds and managementA small-scale broiler production system consists of producers who raise broiler chickens for profit in smaller batches, often 2,000 or fewer birds. These systems can be either intensive or semi-intensive, with reduced investment, labor costs, and direct sales to local markets or customers (FAO, 2004; Gilbert et al., 2015). A small-scale household broiler producer farm in El-Qalyubia Province, Egypt, served as the site of the current investigation. A total of 450-day-old, unsexed Ross308 broiler chicks with an average starting body weight of 42.60 g were obtained from a commercial hatchery that follows standard health and quality protocols. The chicks were randomly distributed in 5 treatments, with 6 replicates for each treatment (15 chicks/replicate). Chicks were reared on floor pens covered with sawdust, with unlimited access to feed and water. The artificial light program was 24 hours during the brooding period (first week of age) and 23 hours for the rest of the experiment period (42 days). The temperature was 34°C–35°C during the brooding period, then gradually reduced by 2°C–3°C every week up to the end of the experiment. Electric fans were used for ventilation. The vaccination program was as follows: at 7 days of age, chicks received the Hitchner vaccine, at 21 and 35 days of age, chicks received the Lasota vaccine, and at 14 and 28 days of age, chicks received the Gumboro vaccine. The experiment was conducted under controlled and standardized broiler management conditions. Diets and experimental designIn a randomized design with five treatments (90 chicks per treatment), all experimental chicks were randomly separated into six replicates (15 chicks/replicate). The treatments consisted of five levels of AG supplementation: 0% basal diet, 0.25%, 0.50%, 0.75%, and 1.0 %. For each treatment, AG powder was blended into the basal diet by first pre-blending it with a small amount of the diet to ensure an even distribution, then mechanically mixing it into the entire batch until it was fully homogenous before feeding it to the birds. AG was obtained from an Egyptian private company, and the technique described by Al-Baadani et al. (2022) was employed to evaluate the chemical analysis of the AG powder (Table 1). The basal diets, in a mash form, were prepared according to the guidelines of the National Research Council and Subcommittee on Poultry Nutrition (1994). The birds were fed two types of diets: the starter diet from 1 to 21 days (23.12% CP and 3,001 Kcal ME/kg diet) and the finisher diet from 22 to 42 days (21.10% CP and 3,180 Kcal ME/kg diet), as shown in Table 2. The experimental samples and diets were chemically analyzed in the Animal Production Research Institute Labs, Agriculture Research Center, according to the procedures outlined by A.O.A.C. (1990). Table 1. Chemical analysis of Arabic gum (% on a dry matter basis).
Table 2. Chemical composition of experimental diets (% dry matter).
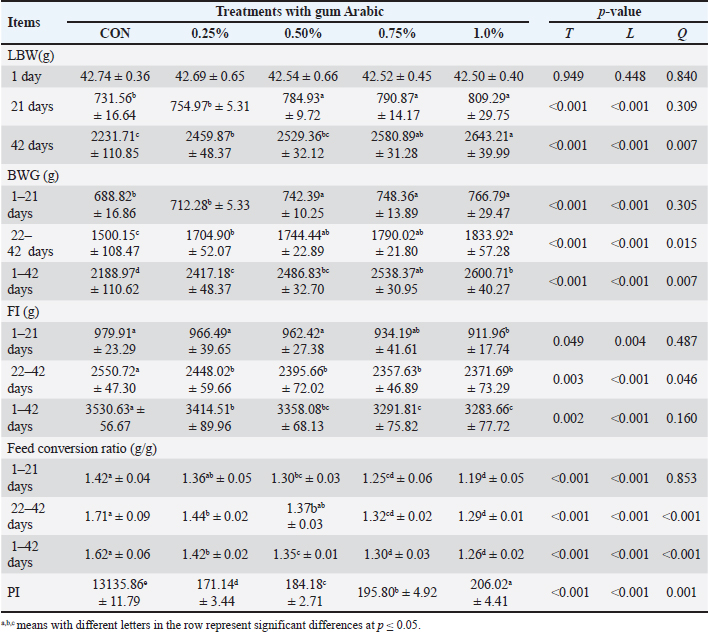
Growth performanceAt the beginning of the experiment, all chicks were individually weighed to the nearest gram (initial live body weight), and then at 21 and 42 days of age, chicks of each experimental group were weighed individually to obtain live body weight (LBW). FI was also estimated weekly for all replicates. The average BWG and feed conversion ratio (FCR) were calculated weekly. Furthermore, performance index (PI)=BWG (kg)/FCR × 100 was estimated during the experiment period (Siham et al., 2015). Carcass and blood parametersAt 42 days, 12 birds were randomly selected from each treatment (2 birds/replicate), fasted overnight with free access to water, weighed individually (pre-slaughter weight), and slaughtered to measure the carcass’ physical characteristics. Blood samples were collected during the slaughtering process in clean, sterilized, and non-heparinized tubes and then centrifuged at 3,000 r/min for 15 minutes at 4°C to separate the serum. Then, the samples were frozen at −20°C until chemical analysis. Serum was analyzed for alanine aminotransferase (ALT) and aspartate aminotransferase (AST) activities, and total protein (TP), albumin (Alb), urea, and creatinine levels were evaluated according to previous studies (Klin, 1980; Burtis and Ashwood, 1999; Wainstein et al., 2022). Immunoglobulin (IgM, IgA, and IgY) levels were also measured using commercially available enzyme-linked immunosorbent assay kits following the manufacturer’s protocols. Oxidative status parameters, including glutathione peroxidase (GPx), glutathione-S-transferase (GST), malondialdehyde (MDA), and glutathione (GSH), were determined using the ELISA Kit-Elabscience biotechnology (Elabscience, China) following the manufacturer’s instructions. Microbiological analysisAfter the birds were slaughtered, the cecal samples (10 g) from each bird were collected aseptically into sterile tubes and immediately transported on ice to the laboratory. For each sample, 1 g of cecal content was homogenized in 9 ml of sterile buffered peptone water (1:10 dilution) using a vortex mixer for 2 minutes. Serial 10-fold dilutions (10−1–10−7) were prepared in sterile saline solution. Total bacterial count (TBC) was estimated by spreading 0.1 ml of the appropriate dilution onto plate count agar and incubating the plates at 37°C for 24–48 hours. Lactobacillus spp. were enumerated using MRS agar under anaerobic conditions at 37°C for 48–72 hours Coliforms and E. coli were quantified by plating on MacConkey agar and EMB agar, followed by incubation at 37°C for 24 hours. Salmonella spp. counts were obtained by pre-enriching samples in buffered peptone water for 24 hours at 37°C, then streaking onto Xylose Lysine Deoxycholate agar and incubating at 37°C for 24 hours. Clostridium was enumerated by plating on tryptose sulfite cycloserine agar under anaerobic conditions at 37°C for 24–48 hours. Colony counts were taken only from plates containing 30–300 colonies, and the results were expressed as log10 CFU/g of cecal content, following the methodology described by Abou-Kassem et al. (2021). Economic efficiencyThe economic efficiency was assessed using the gross margin and benefit cost ratio (BCR). First, the price of the chicks and the prices of the vaccine, medicine, energy, and feed were added together to calculate the total variable cost/chick/experiment period (42 days). Second, the benefits/chick/period were calculated using the meat revenues for the LBW of the chicken at the end of the experiment. Third, the formula gross margin=total variable cost minus benefit was used to calculate the gross margin. Finally, the formula BCR=benefit/cost was used to determine the BCR (Gittinger, 1982). Statistical analysisThe general linear model method was used to examine the data procedure of the Statistical Package for the Social Sciences (2019). Before analysis, the assumption of normality was verified using the Shapiro–Wilk test, and Levene’s test was used to assess the homogeneity of variances. Duncan’s test was used to estimate differences between means (Duncan, 1955). The following model was used: Yij=μ + Ti + eij, where Yij=observed values of the concerned treatment, μ=observed means for the concerned treatments, Ti is the treatment effect, and eij=the error related to individual observations. ResultsGrowth efficiencyTo assess the effects of AG on LBW, BWG, FI, and FCR, values were recorded weekly during the experimental period (Table 3). Increasing AG levels significantly (p < 0.05) improved the overall growth performance of Ross 308 broiler chicks. Elevated AG levels increased LBW, BWG, and PI, and lowered FI and FCR values. The 1.0% AG treatment showed the highest LBW (2643.21), BWG (2600.71), and PI (206.02), accompanied by the lowest FI (3283.66) and FCR (1.26) when compared to the control (CON) and other treatment groups. Table 3. The impact of AG supplementation on broilers’ performance (mean ± SD).
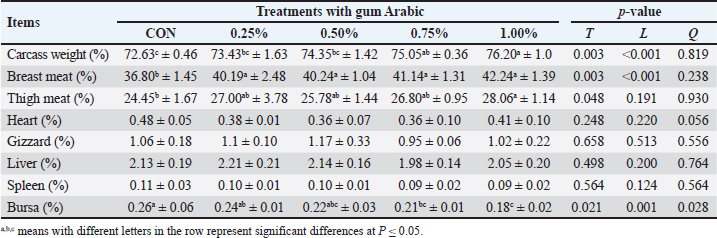
Carcass featuresTable 4 displays the effects of AG on carcass characteristics. The additives tested had a significant (p < 0.05) impact on the percentage of thigh meat, carcass weight, breast meat, and bursa. The other carcass characteristics were not significantly affected. In comparison to broilers fed different levels of AG and those in the CON group, broilers fed 1.0% AG had the largest percentages of carcass weight, breast meat, and thigh meat, with the lowest bursa percentage. Conversely, the CON, 0.25%, and 0.50% treatments yielded the highest percentage values for the heart, spleen, liver, and gizzard, respectively. Table 4. The impact of AG supplementation on broilers’ carcass traits (mean ± SD).
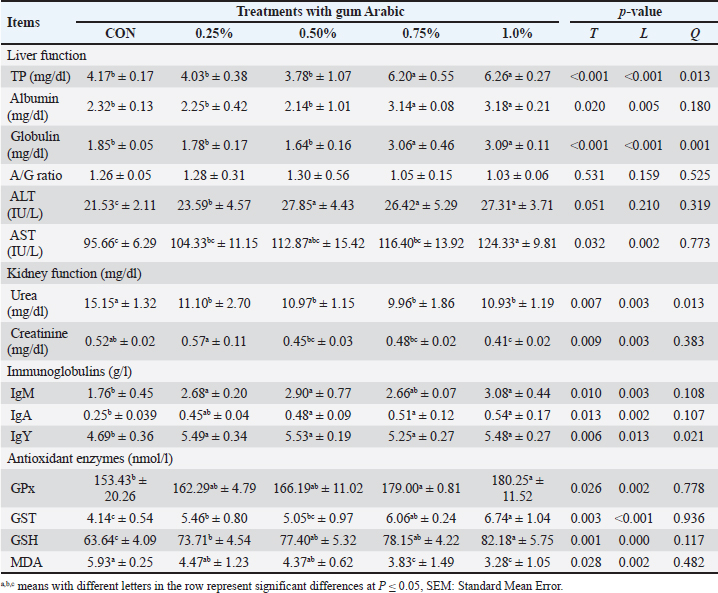
Blood biochemistryBlood serum analysis and antioxidant enzyme activity are indicators of the general health and nutritional status of birds. Table 5 presents the liver and kidney function parameters and antioxidant enzyme levels. Significant changes (p < 0.05) were observed between the CON group and AG-fed chicks, except for the albumin-to-globulin (A/G) ratio. Regarding liver function, the 1.0% AG group showed the highest TP, AL, globulin, and AST values compared with the CON group, whereas the 0.50% treatment resulted in the highest ALT level. Conversely, the lowest levels of urea and creatinine were found in the groups receiving 0.75% and 1.0%, respectively. Table 5. The impact of AG supplementation on broilers’ blood biochemicals (mean ± SD).
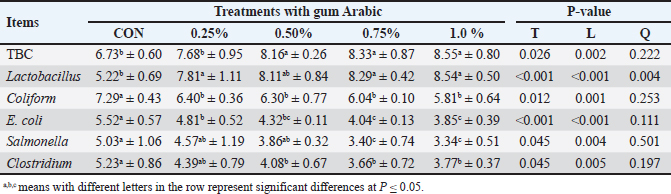
The addition of AG caused immunoglobulins (IgM, IgA, and IgY) to significantly increase (p < 0.05). Our investigation demonstrated that the 1.0% group had the highest IgM and IgA values, while the 0.50% group had the highest IgY values. The addition of AG at varying concentrations significantly improved the antioxidant enzyme activity (p < 0.05), indicating the capacity of the broilers’ bodies to eliminate free radicals. In addition, broilers fed diets supplemented with 1.0% AG exhibited the highest levels of GPx, GST, and GSH, along with the lowest MDA level when compared to other treatments, whereas the CON group exhibited the lowest levels of antioxidant enzyme activity. Microbial analysis of the broiler cecumTable 6 presents the impact of different AG levels as feed additives for broiler diets on the microbial load in their cecal samples. Increasing the AG levels significantly decreased the number of harmful bacteria, such as Salmonella, Clostridium perfringens, E. coli, and Coliform, and a significant rise (p < 0.05) in the number of TBC and Lactobacillus. Compared with the other AG levels and the CON group, the 1.0% treatment had the highest TBC and Lactobacillus counts and the lowest Coliform, E. coli, Salmonella, and C. perfringens counts. Table 6. The impact of AG supplementation on the cecal microbial load of broilers (Log10CFU/g) (mean ± SD).
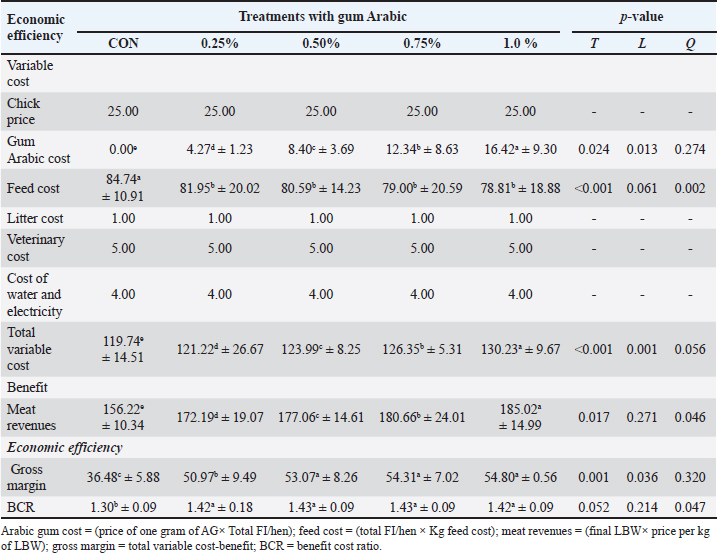
Economic efficiencyTable 7 presents the economic efficiency of Ross 308 broilers fed diets supplemented with AG at different levels. Although increasing AG levels resulted in a higher total variable cost, the benefit and economic efficiency offset the AG cost. Compared with the CON group, the Ross 308 broilers fed with diets supplemented with AG at a level of 1.0% yielded the highest meat revenues (185.02 LE) and gross margin (54.80 LE). Conversely, the highest BCR (1.43) was detected in broilers in the 0.50% and 0.75% groups, followed by the 1.0% group (1.42). Table 7. Economic efficiency (calculated in LE) for broilers fed AG-supplemented diets (mean ± SD).
DiscussionThe prospective growth-promoting, antibacterial, and additional health benefits of herbs, extracts, and diverse natural phytochemicals are presently under examination as alternatives to antibiotics (Ashour et al., 2025b; Elsherbeni et al., 2025). In this study, AG supplementation improved LBW and BWG, decreased FI, and improved FCR. These findings are consistent with those of Ahmad and Essien (2023), who reported that supplementing chicken diets with 5% AG significantly increased daily BWG and LBW while enhancing FCR. Birds fed 5% AG reached a weight of 1,710 g, which was higher than that of the control group. Moreover, AG promotes the proliferation of beneficial bacteria (e.g., Lactobacillus spp.), which subsequently suppresses pathogenic bacteria, including Salmonella, C. perfringens, and E. coli, thereby improving the gut health and overall physiological condition of broilers (Hassan et al., 2024; Sikiru et al., 2024). Improved ileal digestibility and improved apparent metabolizable energy could be the cause of this improved growth performance (Wealleans et al., 2017a,b). Similarly, Abdel-Moneim et al. (2020) reported that improved LBW was due to greater feed digestibility and increased lipolytic, proteolytic, and amylolytic activity in the duodenum. A previous study by Al-Baadani et al. (2023a) showed that broilers fed 0.12%–0.75% AG exhibited significant increases in BW and BWG, along with decreases in FI, compared with controls. These improvements may be related to the ability of AG to modulate feed passage time within the digestive system, thereby improving nutrient absorption, as evidenced by the enhanced FCR. Moreover, the reduction in FI values may be attributed to increased satiety by AG (Ahmed et al., 2021). It is proposed that incorporating AG into poultry diets reduces FI values, which may be related to an increase in digesta viscosity and a prolonged digestion period's gastrointestinal tract retention period, which ultimately influences voluntary FI (Saad et al., 2022). In addition, the type and amount of dietary fiber influence how chickens respond to FI; an earlier study by Wim Calame et al. (2008) found that AG can be regarded as a prebiotic fiber with functionality like inulin because it yields more beneficial bacteria. They noticed that AG increased the number of Bacteroides by 10 logs. Similarly, Hassan et al. (2024) tested the effects of various AG levels on broilers from day 1 to day 35 and found that AG improves growth performance, increases weight gain, and notably reduces FI values. This improvement is attributed to the soluble fiber and natural prebiotic properties of AG, which promote the fermentation of beneficial gut microbiota and the production of short-chain fatty acids, which in turn boost the height of the intestinal villi and enhance growth and nutrient absorption. The evaluation of carcass traits and slaughter value in broiler chickens, including dressing yield and carcass characteristics, is a critical consideration (Shewita and Taha, 2018). The results of the current study demonstrated that dietary modifications influenced chicken carcass weight, breast meat yield, and bursa, as well as certain organs such as the liver, gizzard, and spleen. These findings align with those of Falaki et al. (2011), who reported that prebiotic-supplemented chickens had greater breast and carcass meat yields. Furthermore, Sato et al. (2009) observed that the relative weights of the bursa and spleen (lymphoid organs) were higher in AG-fed broilers. Prebiotic supplementation improves carcass characteristics by enhancing nutrient absorption in broilers (Toghyani et al., 2011). The present study suggests that adding AG to broilers’ diets may enhance carcass traits. Saad et al. (2022) indicated that these improvements may be attributed to the soluble fiber content of AG, which enhances digesta viscosity and promotes nutrient absorption. Furthermore, Nasir et al. (2008) reported that dietary fiber improves mineral bioavailability and exerts positive physiological effects. Conversely, Ahmad and Essien (2023) found that the weights of the giblets (heart, liver, and gizzard) were significantly lower in the group that received the AG-supplemented diets than in the group that did not. Moreover, many studies have noted that AG supplementation at different levels often does not significantly affect carcass traits (Al-Baadani et al., 2023a,b). The FI values in the current study reflect actual consumption under controlled conditions. Furthermore, the improved FCR of the 1.0% AG group (1.26) is biologically plausible due to enhanced nutrient digestibility, increased metabolizable energy, improved gut morphology, and the prebiotic effects of AG, as reported previously by Al-Baadani et al. (2022) and Wealleans et al. (2017a,b), reducing FI while improving weight gain. Furthermore, from previous studies, small-scale broiler producers can enhance their economic efficiency. By lowering the birds’ mortality rates, FCR, and production duration, both their technical and financial efficiency could be improved. Thus, under small-scale production systems, performance should not be directly compared with commercial-scale reference ranges due to different conditions (Ramukhithi et al., 2023; Fotou et al., 2024). Blood chemistry and liver and kidney function biochemical indicators provide crucial information about the overall health and condition of chicks (Xu et al., 2022; Kamal et al., 2023). AG supplementation decreased urea and creatinine levels while increasing TP, AL, globulin, and AST levels. Additionally, AG increases serum TP levels, suggesting improved health or enhanced protein anabolism (Siham et al., 2015; Khan et al., 2022; Al-Baadani et al., 2023b). Blood tests also revealed variations in blood parameters that may indicate the health benefits of AG consumption. Although AST and ALT enzymes are commonly used as indicators of hepatic integrity, mild elevations within physiological limits may also reflect increased metabolic activity or adaptive enzyme induction rather than hepatocellular damage. Mousa et al. (2025) suggested that AG supplementation lowers creatinine levels, which could be related to better kidney function and less intestinal fluid absorption. In addition, dietary AG supplementation at different levels lowers uric acid levels in broilers. It may benefit the microbiota by producing SCFAs and ammonia, leading to increased fecal nitrogen secretion and lower serum urea nitrogen concentration. In addition, SCFAs are absorbed and metabolized by the liver, potentially increasing hepatic enzymatic activity. This may be linked to improved gut bacterial growth and activity (Al-fadil et al., 2013). AG supplementation enhanced antioxidant and immunological properties, indicating improved immune function and metabolic balance. AG can scavenge reactive oxygen metabolites or promote antioxidant production. These antioxidant qualities can be linked to its flavonoid and polyphenol content. These phytocomponents have antioxidant and anti-lipoperoxidative properties (Kassem and Abdullah, 2015; Sadeek, 2018). Previous studies suggest that AG may boost antioxidant capacity by containing amino acid residues such as lysine, tyrosine, and histidine, which are known to be antioxidant molecules (Park et al., 2005). Furthermore, the enhanced immune response observed in birds fed AG-enriched diets may help them resist diseases and improve overall health, as demonstrated by increased immunological markers in these birds. Further investigation into these mechanisms is warranted, as differences in hematocrit and platelet counts between treatment groups suggest potential modulatory effects of AG on blood composition, coagulation processes, and possible links to immune regulation (Ahmed et al., 2023; Zahid et al., 2023). The elevated immunoglobulin (IgM, IgA, and IgY) levels in our study suggest enhanced systemic and mucosal immunity. IgM is crucial for primary immune responses, IgA protects mucosal surfaces by blocking pathogen adhesion, and IgY provides sustained humoral immunity in birds (Eriksson and Larsson, 2025). This improvement suggests greater disease resistance, upgraded immunological readiness, and greater resilience to enteric and systemic infections (Megha and Mohanan, 2021; Ibeagha-Awemu et al., 2025). Such immune enhancement is especially beneficial in antibiotic-free systems, where natural immunomodulators, such as AG, can reinforce flock health and decrease susceptibility to common bacterial infections. These combined mechanisms explain why 1.0% AG yielded the best physiological, microbial, and economic benefits. Adding AG dramatically decreased harmful bacteria and increased TBC and Lactobacillus populations; the 1.0% AG treatment yielded the best results. The shift away from AGPs due to resistance concerns and consumer demand for antibiotic-free poultry has heightened interest in natural alternatives such as AG (Al-Baadani et al., 2023b). The primary components of AG are soluble fermentable fibers, which positively influence intestinal histomorphology and function as a natural prebiotic. They increase villus height and the villus height-to-crypt depth ratio while reducing crypt depth in several intestinal regions, thereby enhancing nutrient absorption (Al-Baadani et al., 2022). Additionally, Wang et al. (2016) observed that supplementing broilers with prebiotics, probiotics, and synbiotics reduced the levels of E. coli, C. perfringens, and coccidiosis. Prebiotics such as AG may prevent the adhesion of harmful bacteria to the intestinal epithelium by promoting the proliferation of beneficial Bifidobacteria and Lactobacilli and stimulating goblet cell secretion (Khan et al., 2022). Moreover, Alvarez-Sieiro et al. (2016) reported that intestinal microbiota improvement may be attributed to the fact that prebiotics lower pH levels by increasing lactic acid through fermentation and the expansion of bacterial populations, particularly lactobacilli, in the cecum. Lactic acid is one of the main Lactobacillus byproducts. Gum Arabic, like other well-known prebiotics such as fructooligosaccharides, mannan-oligosaccharides, and inulin, exerts its effects by enhancing beneficial gut microbiota, increasing SCFA production, and improving nutrient absorption. Compared with many commercial prebiotics, GA is highly soluble, highly fermentable, and economically advantageous, making it particularly suitable for small-scale poultry systems (Bevilacqua et al., 2024; Kezer et al., 2025). According to our results, the addition of AG to the diet of Ross 308 broilers significantly improved their economic efficiency, particularly at the 1.0% inclusion level. Saad et al. (2022) recommended incorporating AG at concentrations of 1%–3% in the diets of local poultry strains aged 3–19 weeks to optimize economic efficiency. Furthermore, broilers fed diets supplemented with 0.12% and 0.25% AG per kilogram showed an increase in the production efficiency index, a parameter of economic value (Al-Baadani et al., 2023b). Finally, several limitations of the current study should be noted. This study provides insightful information about how AG supplementation affects broilers raised in a small-scale production system. First, the results may not apply to other production systems with differing managerial, environmental, or biosecurity conditions because the experiment was conducted on a single small-scale farm. Second, the study did not assess probable interactions with other feed additives or different diet ingredients; moreover, only 5 AG levels were tested. Third, cecal microbial load analysis relied on culture-based methods, which may not fully capture the microbial community’s diversity or functional composition. Lastly, although important biochemical, immunological, and antioxidant markers were examined, more advanced molecular analyses (such as gut histomorphology and gene expression) were not performed. A more thorough understanding of the action of AG in chickens would be possible with future research that incorporates many farms, wider environmental contexts, and sophisticated molecular tools. LimitationsOne of the limitations of this study is the use of unsexed broiler chicks. Growth performance, FI, and FCR were influenced by sex, with males typically displaying better growth and feed efficiency values than females. Therefore, the increased variability observed in some performance parameters may be a result of raising birds as unsexed batches. Nevertheless, this method is predictable for small-scale broiler production systems. Future studies should examine AG supplementation employing sexed flocks or involve sex-specific analyses to decrease variability and better understand dietary effects. ConclusionIn conclusion, the use of AG as a natural prebiotic feed additive in broilers’ diets, particularly at a 1.0% inclusion level, improves broilers’ overall productivity by enhancing gut health, strengthening immunological responses, increasing antioxidant capacity, and promoting carcass features. Importantly, the combination of physiological, biochemical, microbiological, and economic outcomes demonstrates its potential to increase the sustainability and profitability of various broiler production systems. These results underscore the potential of Arabic gum as a safe and effective feed additive, aligning with the goals of the modern poultry industry to provide high-quality and healthy poultry products. AcknowledgmentThe authors would like to express their sincere gratitude to the Faculty of Veterinary Medicine, Kufa University, Najaf, Iraq, for funding this research under the project KU2024R12769. Conflict of interestAll authors declare no conflicts of interest that could inappropriately influence this manuscript. FundingFaculty of Veterinary Medicine, Kufa University, Najaf, Iraq, for funding this research under the project KU2024R12769. Author contributionsSAMA, AMAA, AKA, HA, MAO, REH, and MAMY drafted, revised, and edited the manuscript. All authors have read and approved the final version of the manuscript. Data availabilityThe data supporting the findings of this study are available upon reasonable request from the corresponding author. ReferencesAbdel-Moneim, A.M.E., Selim, D.A., Basuony, H.A., Sabic, E.M., Saleh, A.A. and Ebeid, T.A. 2020. Effect of dietary supplementation of Bacillus subtilis spores on growth performance, oxidative status, and digestive enzyme activities in Japanese quail birds. Trop. Anim. Health Prod. 52(2), 671–680. Abou-Kassem, D.E., Elsadek, M.F., Abdel-Moneim, A.E., Mahgoub, S.A., Elaraby, G.M., Taha, A.E., Elshafie, M.M., Alkhawtani, D.M., Abd El-hack, M.E. and Ashour, E.A. 2021. Growth, carcass characteristics, meat quality, and microbial aspects of growing quail fed diets enriched with two different types of probiotics (Bacillus toyonensis and Bifidobacterium bifidum). Poultry. Sci. 100(1), 84–93. Ahmad, B.M. and Essien, E.N. 2023. Impact of Arabic gum powder on the growth performance and health status of Chickens. AAJARR 3(2). Ahmed, A.A., Adam Essa, M.E., Mollica, A., Stefanucci, A., Zengin, G. and Ahmed, H. 2021. Gum Arabic modifies anti-inflammatory cytokine in mice fed with high fat diet induced obesity. Bioact. Carboh. Diet. Fiber. 25, 100258. Ahmed, E.G.M., Basher, A.W., Ahmed, M., Seddek, A.L., Morad, S.A. and Abdelmageed, N. 2023. The evaluation of the safety and toxicological characteristics of Acacia nilotica in broiler. SVU-IJVS 6(2), 124–135. Al-Baadani, H.H., Alhotan, R.A. and Azzam, M.M. 2023b. Evaluation of the interaction between gum Arabic addition and stocking density on growth performance, carcass characteristics, and general health parameters of broiler chickens. Animals 13(19), 3024. Al-Baadani, H.H., Alhotan, R.A., Al-Abdullatif, A.A., Alhidary, I.A., Alharthi, A.S., Al-Mufarrej, S.I., Al-Garadi, M.A., Qaid, M.M., Al-Sagan, A.A., Ibrahim, K.E. and Azzam, M.M. 2022. The effect of gum arabic supplementation on growth performance, blood indicators, immune response, cecal microbiota, and the duodenal morphology of broiler chickens. Animals 12(20), 2809. Al-Baadani, H.H., Alhotan, R.A., Azzam, M.M., Alhidary, I.A., Alharthi, A.S. and Al-Abdullatif, A.A. 2024. Effect of gum Arabic as natural prebiotic on intestinal ecosystem of post-hatched broiler chicks. J. Anim. Sci. Technol. 66(6), 1203. Al-Baadani, H.H., Alhotan, R.A., Azzam, M.M., Suliman, G.M., Alharthi, A.S., Fazea, E.H. and Alhidary, I.A. 2023a. Effects of gum Arabic (Acacia senegal) powder on characteristics of carcass and breast meat quality parameters in male broiler chickens. Foods 12(13), 2526. Al-Fadil, S., Mukhtar, M.A. and Tabidi, M.H. 2013. Response of broiler chicks to diets containing gum arabic as a natural prebiotic. J. Curr. Res. Sci. 1(4), 247–253. Alvarez-Sieiro, P., Montalbán-López, M., Mu, D. and Kuipers, O.P. 2016. Bacteriocins of lactic acid bacteria: extending the family. Appl. Microbiol. Biotechnol. 100(7), 2939–2951. AOAC. Official Methods of Analysis. 1995. Vol. I. 15th Ed. Association of Official Analytical Chemists, Arlington, VA, 1995. Available viahttps:// www. scirp. org/reference /References Papers?ReferenceID=1929875 Ashour, E.A., Aldhalmi, A.K., Elolimy, A.A., Madkour, M., Elsherbeni, A.I., Alqhtani, A.H., Khan, I.M. and Swelum, A.A. 2025a. Optimizing broiler performance, carcass traits, and health: evaluating thyme and/or garlic powders as natural growth promoters in antibiotic-free diets. Poult. Sci. 104(2), 104689. Ashour, E.A., Aldhalmi, A.K., Kamal, M., Salem, S.S., Mahgoub, S.A., Alqhtani, A.H., Madkour, M., Elolimy, A.A., Abd El-hack, M.E. and Swelum, A.A. 2025b. The efficacy of Artichoke leaf extract conjugated with organic zinc nanoparticles on growth, carcass traits and blood biochemical parameters of broilers. Poult. Sci. 104(1), 104521. Atay, A. 2023. The effect medicinal plants on performance, carcass parameters and meat quality in broiler chickens. JIST 13(2), 1418–1428. Bevilacqua, A., Campaniello, D., Speranza, B., Racioppo, A., Sinigaglia, M. and Corbo, M.R. 2024. An update on prebiotics and on their health effects. Foods 13(3), 446. Bist, R.B., Bist, K., Poudel, S., Subedi, D., Yang, X., Paneru, B., Mani, S., Wang, D. and Chai, L. 2024. Sustainable poultry farming practices: a critical review of current strategies and future prospects. Poult. Sci. 103(12), 104295. Burtis, C.A. and Ashwood, E.R.1999. Tietz textbook of clinical chemistry. Philadelphia, PA: WB Saunders, pp: 1654–1655. Calame, W., Weseler, A.R., Viebke, C., Flynn, C., Siemensma, A.D. 2008. Gum Arabic establishes prebiotic functionality in healthy human volunteers in a dose-dependent manner. Br. J. Nutr. 100, 1269–1275. Duncan B.D. 1955. Multiple range and multiple F-test: Biometrics 1142. El-Abbasy, M.M., Aldhalmi, A.K., Ashour, E.A., Bassiony, S.S., Kamal, M., Alqhtani, A.H., Abou-Kassem, D.E., Elolimy, A.A., Abd El-hack, M.E. and Swelum, A.A. 2025. Enhancing broiler growth and carcass quality: impact of diets enriched with Moringa oleifera leaf powder conjugated with zinc nanoparticles. Poult. Sci. 104(1), 104519. Elbaz, N., Ashour, E.A., Bassiony, S.S., Elsherbeni, A.I., Mahgoub, S.A., Osman, A.O., Sabike, I., Al-Gabri, N.A., Moustafa, M., Al-Shehri, M. and Abd El-hack, M.E. 2025. Dietary probiotic supplementation with Lactococcus lactis and Bacillus velezensis enhances growth performance, meat quality, blood profiles, and cecal and feed microbiota in growing rabbits. Bio 36(1), 2577947. Elsherbeni, A.I., Aldhalmi, A.K., Youssef, I.M., Kamal, M., Ashour, E.A., Moustafa, M., Al-Kahtani, M.A., Al-Shehri, M. and Abd El-hack, M.E. 2025. The role of trimethylglycine (betaine) as an anti-heat stress agent in sustainable poultry production: enhancing growth, stress resilience, and nutrient utilization. J. Therm. Biol. 130, 104152. Eriksson, M. and Larsson, A. 2025. Avian antibodies as potential therapeutic tools. Antibodies 14(1), 18. Falaki, M., Shargh, M.S., Dastar, B. and Zrehdaran, S. 2011. Effects of different levels of probiotic and prebiotic on performance and carcass characteristics of broiler chickens. Int. J. Recent. Trends. Eng. Res. 3(7), 82–90. FAO. 2004. Small-scale poultry production: technical guide. Food and Agriculture Organization of the United Nations, Rome, Italy. Available viahttp://www.fao.org/3/a-y5169e.pdf. Fotou, E., Moulasioti, V., Papadopoulos, G.A., Kyriakou, D., Boti, M.E., Moussis, V., Papadami, M., Tellis, C., Patsias, A., Sarrigeorgiou, I., Theodoridis, A., Lymberi, P., Tsiouris, V., Tsikaris, V. and Tsoukatos, D. 2024. Effect of farming system type on broilers’ antioxidant status, performance, and carcass traits: an industrial-scale production study. Sustainability 16(11), 4782. Gabriel, G.O., Chukwudi, P., Sodipe, O.G., Folayan, T.A., Tella, A., Kehinde, O.A., Odumboni, A.A., Ogunlade, J.T. and Babalola, T.O. 2025. Effects of Crinum glaucum bulb extract on growth performance, carcass and organ traits, haemato-biochemistry parameters, and oxidative enzyme markers in broiler chickens. Iran. J. Appl. Anim. Sci. 15(1), 119–128. Gadzama, I. 2024. Strategies to improve chicken welfare through enhanced free-range systems. Strategies 2, 10. Gharib, H.B., El-Menawey, M.A. and Hamouda, R.E. 2023. Factors affecting small-scale broiler chicken farm profitability and challenges faced by farmers in Egyptian rural. Trop. Anim. Sci. J. 46(2), 261–268. Gilbert, M., Conchedda, G., Van Boeckel, T.P., Cinardi, G., Linard, C., Nicolas, G., Thanapongtharm, W., D'Aietti, L., Wint, W., Newman, S.H. and Robinson, T.P. 2015. Income disparities and the global distribution of intensively farmed chicken and pigs. PLos One. 10(7), 133381. Gittinger, J.P. 1982. Economics analysis of agricultural projects. 2nd ed. Baltimore, London: Johns Hopkins University, pp: 505. Hamouda, R.E.S., Gharib, H.B., El-Menawey, M.A.R., Abdel-Azeez, Y.A. and Youssif, M.A.M. 2025. Increasing meat viability for food security through rural household broiler family business models. Egypt. J. Vet. Sci. 56(9), 2255–2264. Hassan, R.A., Bayoumi, A.A.E., Mousa, M.A.E., Khosht, A.R., Salim, I.H., Shamseldeen, A.E., Arafa, A.S., El-Deeb, M.A.E., Elkomy, H.M. and Mostafa, S.M. 2024. Effect of dietary inclusion of gum arabic (Acacia senegal) and Lactobacillus acidophilus or their combination on broiler chickens. J. Desert. Environ. Agric. 4(2), 325–345. Ibeagha-Awemu, E.M., Omonijo, F.A., Piché, L.C. and Vincent, A.T. 2025. Alternatives to antibiotics for sustainable livestock production in the context of the One-Health approach: tackling a common foe. Front. Vet. Sci. 12, 1605215. Kamal, M., Kishk, W.H., Khalil, H.A., Abdel-Khalek, A.M., Ayoub, M.A., Swelum, A.A., Alqhtani, A.H., Ba-Awadh, H.A. and Abd El-hack, M.E. 2023. Effect of dietary chitosan supplementation on productive and physiological performance parameters of growing New Zealand white rabbits. Int. J. Biol. Macromol. 230, 123166. Kassem, A. and Abdullah, A. 2015. Dietary gum Arabic supplementation alter plasma and tissue antioxidant and free radical scavenging activities in Sprague Dawley male rats. J. Biol. Life Sci. 6(1), 129. Kezer, G., Paramithiotis, S., Khwaldia, K., Harahap, I.A., Čagalj, M., Šimat, V., Smaoui, S., Elfalleh, W., Ozogul, F. and Esatbeyoglu, T. 2025. A comprehensive overview of the effects of probiotics, prebiotics and synbiotics on the gutbrain axis. Front. Microbiol. 16, 1651965. Khan, S., Chand, N., Hafeez, A. and Ahmad, N. 2022. Effect of gum Arabic on overall growth performance, visceral and lymphoid organs along with intestinal histomorphology and selected pathogenic bacteria of broiler chickens. JAHP 10(1), 73–80. Kleyn, R. and Ciacciariello, M. 2025. Putting sustainable poultry production into perspective. W. Poult. Sci. J. 81(2), 343–357. Klin, B.J. 1980. Cumulative effect of X-ray radiation and inflammatory reaction on the circadian rhythm of tyrosine aminotransferase in the liver of mice. Acta Physiol. Pol. 2(2), 131–139. Megha, K.B. and Mohanan, P.V. 2021. Role of immunoglobulin and antibodies in disease management. Int. J. Biol. Macromol. 169, 28–38. Mousa, M.A., Khosht, A.R., Salim, I.H., Shamseldeen, A., Arafa, A.S., El-Deeb, M.A. and Mobarez, S.M. 2025. The effect of gum arabic (Acasia senegal) as a natural prebiotic supplementation for broiler chickens. J. King Abdulaziz Univ. Meteorol. 34(2), 128–147. NRC, National Research Council. 1994. Nutrient Requirements of Poultry: Ninth Revised Edition, 1994. Washington, DC: The National Academies Press;doi: 10.17226/2114. Nasir, O., Artunc, F., Saeed, A., Kambal, M.A., Kalbacher, H., Sandulache, D. and Lang, F. 2008. Effects of gum arabic (Acacia senegal) on water and electrolyte balance in healthy mice. JREN 18(2), 230–238. Park, E.Y., Murakami, H. and Matsumura, Y. 2005. Effects of the addition of amino acids and peptides on lipid oxidation in a powdery model system. J. Agric. Food. Chem. 53(21), 8334–8341. Ramukhithi, T.F., Nephawe, K.A., Mpofu, T.J., Raphulu, T., Munhuweyi, K., Ramukhithi, F.V. and Mtileni, B. 2023. An assessment of economic sustainability and efficiency in small-scale broiler farms in Limpopo province: a review. Sustainability 15(3), 2030. Saad, M., Saad, M.F., Abd Alla, E.A., Tawfeek, F.A., Abd Elhady, D., Eshraa, A. and Badawy, Y.K. 2022. Effect of feeding different levels of Arabic gum (AG) on physiological and productive performance of a local breed of chicken. J. Anim. Poult. Produc. 13(9), 125–130. Sadeek, F. 2018. Effect of Arabic Gum as prebiotics and Lactobacillus casei Shirota (LcS) as probiotic on oxidative stress and renal function in adenine–induced chronic renal failure in rats. Eur. J. Nutr. Food Saf. 8(1), 29–46. Sato, K., Takahashi, K., Tohno, M., Miura, Y., Kamada, T., Ikegami, S. and Kitazawa, H. 2009. Immunomodulation in gut-associated lymphoid tissue of neonatal chicks by immunobiotic diets. Poult. Sci. 88, 2532–2538. Shewita, R.S. and Taha, A.E. 2018. Influence of dietary supplementation of ginger powder at different levels on growth performance, haematological profiles, slaughter traits and gut morphometry of broiler chickens. S. Afr. J. Anim. Sci. 48(6). Siham, A., Khadiga, A., Huwaida, E., Bakheit, M.D. and Khalid, M.E. 2015. Effect of dietary inclusion of gum Arabic (Acacia senegal) on performance and blood chemistry of broiler chicks. WJAS 3(2), 305–310. Sikiru, A.B., Harande, I.S., Hassan, Y., Hassan, A.U., Ibrahim, N.T. and Adam, M.N. 2024. Evaluation of the impact of Acacia nilotica and Annona senegalensis as potential sources of phytogenic compounds on performances of commercial broiler chickens. ADAN J. Agric. 5(1), 122–131. SPSS. 2019. SPSS Statistics for Windows, Version 26.0. Chicago, IL: SPSS Inc. Tabidi, M.H. and Ekram, K.A. 2015. Effect of feeding gum Arabic with or without commercial xylem enzyme 500 on the performance of broiler chicks. World J. Pharm. Pharm. Sci. 9(9), 1863–1872. Toghyani, M., Toghyani, M. and Tabeidian, S.A. 2011. Effect of probiotic and prebiotic as antibiotic growth promoter substitutions on productive and carcass traits of broiler chicks. In International Conference on Food Engineering and Biotechnology, IACSIT Press, Singapore, Vol. 9, pp 82–86. Wainstein, T., Marshall, S.K., Ross, C.J.D., Virani, A.K., Austin, J.C., Elliott, A.M., Knoppers, B., Lynd, L.D., Dey, A., Adam, S., Bansback, N., Birch, P., Clarke, L., Dragojlovic, N., Friedman, J., Lambert, D., Pullman, D., Wasserman, W. and Zawati, M. 2022. Experiences with genetic counseling, testing, and diagnosis among adolescents with a genetic condition: a scoping review. JAMA. Pediatr. 176, 185–195. Wang, X., Farnell, Y.Z., Peebles, E.D., Kiess, A.S., Wamsley, K.G.S. and Zhai, W. 2016. Effects of prebiotics, probiotics, and their combination on growth performance, small intestine morphology, and resident Lactobacillus of male broilers. Poult. Sci. 95(6), 1332–1340. Wealleans, A.L., Sirukhi, M. and Egorov, I.A. 2017. Performance, gut morphology and microbiology effects of a Bacillus probiotic, avilamycin and their combination in mixed grain broiler diets. Br. Poult. Sci. 58(5), 523–529. Wealleans, A.L., Walsh, M.C., Romero, L.F. and Ravindran, V. 2017. Comparative effects of two multi-enzyme combinations and a Bacillus probiotic on growth performance, digestibility of energy and nutrients, disappearance of non-starch polysaccharides, and gut microflora in broiler chickens. Poult. Sci. 96(12), 4287–4297. Xu, H.L., Li, H., Bao, R.K., Tang, Y.X., Elsherbeni, A.I.A., Gharib, H.B.A. and Li, J.L. 2022. Transport stress induced cardiac NO-NOS disorder is mitigated by activating Nrf2/HO-1/NQO1 antioxidant defense response in newly hatched chicks. Front. Vet. Sci. 9, 938826. Zahid, M.U., Khalique, A., Qaisrani, S.N., Ashraf, M., Sheikh, A.A. and Yaqoob, M.U. 2023. The effect of Acacia nilotica bark extract on growth performance, carcass characteristics, immune response, and intestinal morphology in broilers as an alternative to antibiotic growth promoter. Anim. Biosci. 36(7), 1059. | ||
| How to Cite this Article |
| Pubmed Style Alkassar SAM, Alkassar AMA, Ahmed AK, Al-khalaifah H, Osman MA, Hamouda RE, Youssif MAM. Growth performance, antioxidant capacity, and microbial modulation in broilers supplemented with Arabic gum under a small-scale production system. Open Vet. J.. 2026; 16(2): 907-919. doi:10.5455/OVJ.2026.v16.i2.13 Web Style Alkassar SAM, Alkassar AMA, Ahmed AK, Al-khalaifah H, Osman MA, Hamouda RE, Youssif MAM. Growth performance, antioxidant capacity, and microbial modulation in broilers supplemented with Arabic gum under a small-scale production system. https://www.openveterinaryjournal.com/?mno=292018 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.13 AMA (American Medical Association) Style Alkassar SAM, Alkassar AMA, Ahmed AK, Al-khalaifah H, Osman MA, Hamouda RE, Youssif MAM. Growth performance, antioxidant capacity, and microbial modulation in broilers supplemented with Arabic gum under a small-scale production system. Open Vet. J.. 2026; 16(2): 907-919. doi:10.5455/OVJ.2026.v16.i2.13 Vancouver/ICMJE Style Alkassar SAM, Alkassar AMA, Ahmed AK, Al-khalaifah H, Osman MA, Hamouda RE, Youssif MAM. Growth performance, antioxidant capacity, and microbial modulation in broilers supplemented with Arabic gum under a small-scale production system. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 907-919. doi:10.5455/OVJ.2026.v16.i2.13 Harvard Style Alkassar, S. A. M., Alkassar, . A. M. A., Ahmed, . A. K., Al-khalaifah, . H., Osman, . M. A., Hamouda, . R. E. & Youssif, . M. A. M. (2026) Growth performance, antioxidant capacity, and microbial modulation in broilers supplemented with Arabic gum under a small-scale production system. Open Vet. J., 16 (2), 907-919. doi:10.5455/OVJ.2026.v16.i2.13 Turabian Style Alkassar, Saif Ali Mahmoud, Ali Mahmood Amer Alkassar, Ahmed Khalid Ahmed, Hanan Al-khalaifah, Moustafa Amin Osman, Reda El-sayed Hamouda, and Mai Ahmed Maher Youssif. 2026. Growth performance, antioxidant capacity, and microbial modulation in broilers supplemented with Arabic gum under a small-scale production system. Open Veterinary Journal, 16 (2), 907-919. doi:10.5455/OVJ.2026.v16.i2.13 Chicago Style Alkassar, Saif Ali Mahmoud, Ali Mahmood Amer Alkassar, Ahmed Khalid Ahmed, Hanan Al-khalaifah, Moustafa Amin Osman, Reda El-sayed Hamouda, and Mai Ahmed Maher Youssif. "Growth performance, antioxidant capacity, and microbial modulation in broilers supplemented with Arabic gum under a small-scale production system." Open Veterinary Journal 16 (2026), 907-919. doi:10.5455/OVJ.2026.v16.i2.13 MLA (The Modern Language Association) Style Alkassar, Saif Ali Mahmoud, Ali Mahmood Amer Alkassar, Ahmed Khalid Ahmed, Hanan Al-khalaifah, Moustafa Amin Osman, Reda El-sayed Hamouda, and Mai Ahmed Maher Youssif. "Growth performance, antioxidant capacity, and microbial modulation in broilers supplemented with Arabic gum under a small-scale production system." Open Veterinary Journal 16.2 (2026), 907-919. Print. doi:10.5455/OVJ.2026.v16.i2.13 APA (American Psychological Association) Style Alkassar, S. A. M., Alkassar, . A. M. A., Ahmed, . A. K., Al-khalaifah, . H., Osman, . M. A., Hamouda, . R. E. & Youssif, . M. A. M. (2026) Growth performance, antioxidant capacity, and microbial modulation in broilers supplemented with Arabic gum under a small-scale production system. Open Veterinary Journal, 16 (2), 907-919. doi:10.5455/OVJ.2026.v16.i2.13 |