| Research Article | ||
Open Vet. J.. 2025; 15(12): 6767-6776 Open Veterinary Journal, (2025), Vol. 15(12): 6767-6776 Research Article Efficacy of a multivalent inactivated Salmonella vaccine against fecal shedding and organ dissemination in chickensWael K. Elfeil1*, Salma Essam2, Esraa Mostafa2, Doaa E. Ismail2, Mohamed Fathy3, Hend Altaib2, Mohamed E. Basuony2,4, Ahmed Sedeek5, Islam Hisham5, Momtaz Wasfy2, Walid H. Kilany5 and Nermeen M. Abu-Elala6,71Avian and Rabbit Medicine Department, Faculty of Veterinary Medicine, Suez Canal University, Ismailia, Egypt 2MEVAC—Middle East for Vaccines, Second Industrial Area, El-Salihya El-Gededa, El-Sharkia, Egypt 3Animal and Poultry Health Department, Desert Research Centre, Cairo, Egypt 4Botany & Microbiology Department, Faculty of Science, Al-Azhar University, Assiut, Egypt 5Reference Laboratory for Veterinary Quality Control on Poultry Production (RLQP), Animal Health Research Institute (AHRI), Agricultural Research Center (ARC), Giza, Egypt 6Aquatic Animal Medicine and Management Department, Faculty of Veterinary Medicine, Cairo University, Giza, Egypt 7Animal Medicine Department, Faculty of Veterinary Medicine, King Salman International University, South Sinai, Egypt *Corresponding Author: Wael K. Elfeil. Avian and Rabbit Medicine Department, Faculty of Veterinary Medicine, Suez Canal University, Ismailia, Egypt. Email: Elfeil [at] vet.suez.edu.eg Submitted: 17/09/2025 Revised: 05/11/2025 Accepted: 15/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
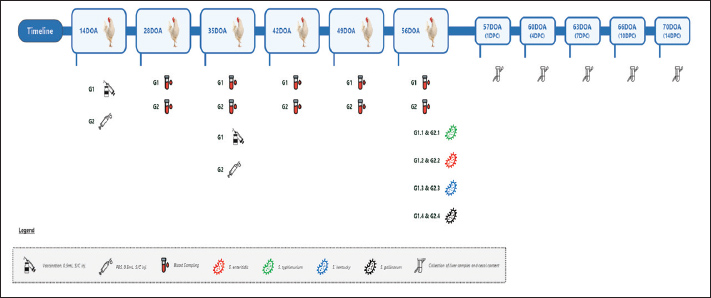
AbstractBackground: Salmonella infection is one of the main zoonotic diseases threatening poultry and food safety. Various serovars, including Salmonella enteritidis, Salmonella typhimurium, Salmonella kentucky, and Salmonella gallinarum, cause economic losses and public health risks. The rise of multidrug-resistant (MDR) Salmonella underscores the need for vaccination. Aim: This study assessed the safety and effectiveness of a tetravalent inactivated Salmonella vaccine (POULMMUNE™ SALMONELLA BCD) in reducing fecal shedding and systemic invasion in chickens. Methods: Antimicrobial susceptibility was tested for Salmonella strains isolated from poultry. The vaccine, expressed as a water-in-oil emulsion with Montanide™ ISA 70 VG, was administered to 250 specific pathogen-free White Leghorn chickens, which were divided into vaccinated and control groups. Safety was assessed through clinical monitoring, while efficacy was evaluated by serological responses, fecal shedding, and post-challenge bacterial translocation to the liver. The most probable number (MPN) method was used to determine bacterial counts, and statistical analysis was performed (p < 0.05). Results: MDR was detected in all isolates, with high resistance to clindamycin, penicillin, ampicillin, tetracycline, and streptomycin. The vaccine was well tolerated with no adverse effects. Vaccinated birds had significantly higher antibody titers, reduced fecal shedding by 14 days post-challenge, and lower liver translocation, with no detectable bacteria by direct plating from day 4 post-challenge. Conclusion: The vaccine demonstrated strong safety, immunogenicity, and efficacy in reducing shedding and systemic dissemination of Salmonella. Given the high prevalence of MDR Salmonella, vaccination is crucial for controlling infection and minimizing antibiotic reliance in poultry. Further studies should be conducted to optimize formulations and assess field efficacy. Keywords: Salmonella, Poultry vaccine, Tetravalent vaccine, Antimicrobial resistance, Bacterial translocation, Fecal shedding, Food safety, Tetravalent vaccine. IntroductionSalmonella infections remain a significant challenge in poultry production, causing substantial economic losses and posing serious public health risks (Brenner et al., 2000). Salmonella species are facultative anaerobic, gram-negative, and flagellated bacteria that are categorized using 114 flagella (H) antigens and 46 LPS (O). Currently, over 2,610 serotypes have been identified using this taxonomy. Salmonella entertidis (SE), Salmonella typhimurium (ST), and Salmonella kentucky (SK) are the most commonly linked to human food poisoning and salmonellosis outbreaks in chicken farms (Dieckmann and Malorny, 2011). Host-specific septic shock infection known as fowl typhoid is caused by SG. It mainly affects adults, growing chickens, turkeys, and other birds. Acute infections are associated with septicaemia. However, during subacute outbreaks, there are dead chicks on hatching trays or dead embryos in shells. Birds with a chronic disease exhibit anemia and necrosis in the pancreas, liver, heart, and intestines (Berhanu and Fulasa, 2020). The rise in antibiotic resistance in Salmonella is a significant global public health issue. Therefore, chicken vaccination is a key tactic in reducing the incidence of Salmonella in poultry flocks, which will reduce food-borne Salmonella infections and create a safer food supply. It should be used in conjunction with other control techniques as part of a comprehensive Salmonella control program. Therefore, vaccination of chickens, along with other control measures as part of a comprehensive Salmonella control program, is an important strategy in lowering the prevalence of Salmonella in poultry flocks, leading to a reduction in foodborne Salmonella infections and a safer food supply (Berhanu and Fulasa, 2020). Despite the availability of commercial Salmonella vaccines, their effectiveness across different serovars remains uncertain. Live-attenuated vaccines estimate both cell-mediated and humoral immunity but carry risks of environmental shedding and potential virulence reversion. Inactivated vaccines are safer but rely on antigenic similarity to confer protection, necessitating the development of multivalent formulations to address diverse serovars (Desin et al., 2013). However, Atterbury et al. (2009) reported that the inactivated vaccine provided better protection against Salmonella colonization when layer hens were vaccinated with either live or inactivated vaccines. The efficacy of inactivated Salmonella vaccines appears to be dependent on the homology of the O antigens present in the vaccine and those present in the infecting strain. A major gap in current Salmonella vaccination strategies is the lack of comprehensive studies evaluating the effectiveness of multivalent inactivated vaccines in poultry. Existing research has primarily focused on monovalent or bivalent vaccines, leaving uncertainty about the level of protection achievable with broader antigenic coverage. Additionally, there is limited information on how vaccination influences fecal shedding and systemic bacterial translocation, both of which are critical for controlling Salmonella dissemination in poultry production systems (Van Immerseel et al., 2004; Okamura et al., 2007; Deguchi et al., 2009). Given these gaps, this study aimed to assess the efficacy of a tetravalent inactivated Salmonella vaccine developed with SE, ST, SK, and SG. The primary objectives of this study were to assess the ability of the vaccine to reduce fecal shedding, minimize bacterial translocation to internal organs, and elicit a strong humoral immune response in chickens. This study provides valuable insights into the role of inactivated multivalent vaccines in integrated Salmonella control programs, contributing to poultry health management and public health safety. Materials and MethodsSeeds cultivationSalmonella strains resembling SE, ST, SK, and SG were obtained from the Reference Laboratory for Veterinary Quality Control on Poultry Production, Animal Health Research Institute, Egypt. Salmonella spp. isolation was performed according to the FDA guidelines (Andrews et al., 2018). A 16–20-hour pre-enrichment in buffered peptone water (Merck, Germany) at 35°C–37°C was used to maximize the viability of the samples. A 0.1 ml pre-enriched inoculum was subcultured in 10 ml Rappaport-Vassiliadis broth (Difco, France) and cultured for 24 hours at 42°C to achieve selective enrichment. After each enrichment broth was thoroughly mixed, a 10 µl loopful was removed and streaked onto Xylose Lysine Deoxycholate (XLD) agar (Merck, Germany). The colonies of Salmonella spp. (i.e., SE, ST, SK, and SG) were examined after 24 hours of incubation at 37°C. Purified colonies that showed a slightly translucent red halo and a black center surrounded by a pink-red zone on XLD agar were collected and subcultured on tryptic soya broth (TSB) at 37˚C for 24 hours. The grown bacteria were preserved at −80˚C in 15:20% glycerol (Merck, Germany) (Tarabees et al., 2017; Marin et al., 2021) Antimicrobial susceptibility test resultsSalmonella isolates were cultivated for 18 hours at 37°C in nutrient broth before being subcultured on Mueller-Hinton agar (Oxoid Ltd, UK) at a concentration equal to 0.5 McFarland standard (1.5 × 108 colony-forming units or CFU/ml). The disc diffusion method was used to test susceptibility against 12 commonly used antibiotics. The antibiotic discs used were clindamycin (2 µg), penicillin (10 µg), ampicillin (10 µg), tetracycline (30 µg), cefotaxime (30 µg), gentamycin (10 µg), streptomycin (10 µg), levofloxacin (5 µg), ciprofloxacin (5 µg), amoxicillin clavulanic acid (30 µg), amikacin (30 µg), and sulfamethoxazole/trimethoprim (25 µg) (Oxoid Ltd, UK). The plates were incubated at 37°C for 18 hours. The susceptibility patterns of the isolates to the different antibiotics were noted as “Sensitive” (S) or “Resistant” (R) as per the CLSI standards (Clinical and Laboratory Standards Institute, 2020). The data were expressed as a heatmap using GraphPad Prism 9.1.1 (GraphPad, San Diego, CA, USA). Intermediate resistance was regarded as full-blown resistance. Multidrug resistance (MDR) is defined as resistance to four or more antibiotics tested (Adenaike et al., 2016). The multiple antibiotic resistance index was calculated as the number of antibiotics to which the isolate is resistant divided by the total number of antibiotics against which the isolate was tested (Apun et al., 2008; Ezekiel et al., 2011). Vaccine formulationSalmonella isolates were propagated in TSB for 24 hours at 37°C. The harvested antigen bulks were then concentrated to contain 3 × 1010 CFU/ml. Formaldehyde solution 0.25% (v/v) was added to the harvested cultures and incubated at 37°C for 24 hours for inactivation. Inactivation completion was estimated by culturing samples derived from each antigen bulk on XLD. The inactivated antigen bulks and MONTANIDE™ ISA 70 VG (SEPPIC, France) were mixed in a volume ratio of 30:70 to obtain a water-in-oil (W/O) emulsion using an L5M-A high-speed high shear mixer (Silverson Machines, Inc.). The developed vaccine used in the studies was identified as POULMMUNE™ SALMONELLA BCD, an inactivated tetravalent bacterial vaccine comprising SE, ST, SK, and SG (Middle East for Vaccines). Evaluation of the physicochemical properties of the vaccineThe JENWAY® Model 4520 conductivity meter (Cole-Parmer®, France) was used to measure the conductivity of the vaccine, and the Brookfield viscometer (AMETEK® Brookfield, USA) was used to determine its viscosity. The vaccination samples were centrifuged for 15 minutes at 6,000 rpm using a CYAN Tabletop centrifuge (Cypress Diagnostics, Belgium) to ascertain the emulsion stability. The JENWAY® Model 3510 Standard Digital pH Meter (Cole-Parmer®, France) was used to measure the pH of the vaccine. Experimental designTwo-week-old specific pathogen-free (SPF) White Leghorn chickens (n=250) were obtained from Nile SPF Farm, Fayoum, Egypt. The experimental birds of all the considered trials were kept inside BioFlex® B50 poultry isolators (Bell Isolation Systems, UK) under fully controlled conditions for the study period. Feed and freshwater were offered ad libitum. Experiment 1 was conducted to assess the safety of the vaccine. Twenty-two-week-old SPF chickens were injected subcutaneously (S/C) with 0.5 ml of the vaccine in the lower third of the neck. Similarly, another 20 chickens received 1 ml (double dose) of the vaccine via subcutaneous injection. A booster dose (single repeated dose) was administered to the first group 3 weeks after primary vaccination. An additional 10 birds were injected S/C with 0.5 ml of PBS and considered the control group. The vaccinated chickens were monitored daily by the responsible experimenter, who noted and recorded any abnormal clinical signs that could be interpreted as an effect of the vaccine. During the observation period, the appetite and behavior of the vaccinated chickens, as well as the local reactions (e.g., redness and swelling at the injection site), were checked every day. Four weeks after vaccination, five vaccinated chickens from each group were humanely euthanized and submitted to necropsy. The tissues surrounding the injection sites were examined organoleptically. Experiment 2 Assessed the efficacy of the vaccine against challenge. Two hundred two-week-old SPF hens were split into two groups of 100 (Fig. 1). G1 chickens were injected with 0.5 ml of vaccine in the lower region of the neck. The unvaccinated (G2) birds received 0.5 ml of PBS via S/C injection. G1 received a booster (0.5 ml/bird, S/C) 3 weeks after immunization. Three weeks after booster dosage delivery, the experimental groups were split into 5 subgroups. The vaccine groups were G1.1–G1.5, whereas the unvaccinated groups were G2.1–G2.5. Counterpart subgroups from each treatment were orally challenged with 109 CFU/ml from SE, ST, SK, and SG. G1.5 was left as vaccinated unchallenged; however, G2.5 was allocated as an unvaccinated unchallenged “sham” after receiving 1.0 ml of PBS orally (Supplementary Table 1).
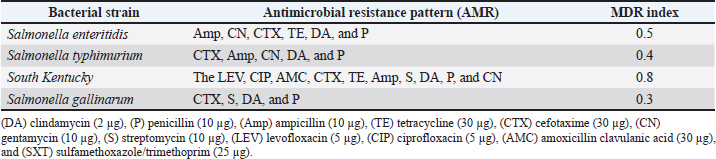
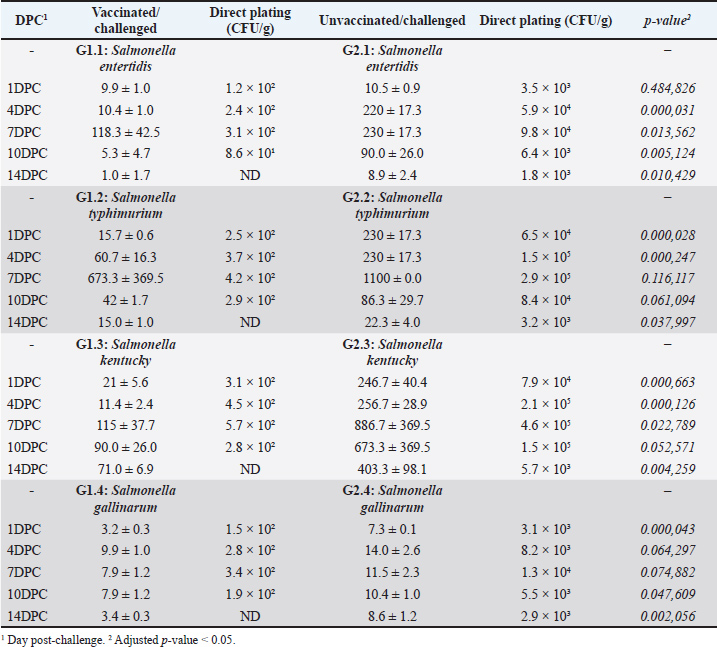
Fig. 1. Diagrammatic explanation of vaccine potency and efficacy evaluation in chicken. Table 1. Antimicrobial resistance pattern and MDR indices of Salmonella isolates.
Measurement of the antibody responseHumoral antibody titers were measured using a microtiter plate agglutination test as described (Williams and Whittemore, 1971). Briefly, blood samples were collected from the wing veins of birds, left to clot, centrifuged, and the serum samples were separated. The test was performed in 96-well, rounded (U-shaped) microplates. Serum samples were individually diluted in PBS on a twofold scale. The dilution was prepared by adding 50 µl of PBS into all wells included in the titration series. Serum (50 µl) was placed into the first well of the microplate, and a twofold serial dilution of the serum was performed. Salmonella were diluted with PBS to a density of 0.5 McFarland scale. Then, 50 l of the stained Salmonella antigens were added to each well. Each plate was tightly sealed with an adhesive plate sealer and incubated for 18 has at 37°C for 18 hours. The results were recorded as log2 (x) of the reciprocal of the highest serum dilution, indicating agglutination. Re-isolation of the challenge strainsRe-isolation was evaluated at 1, 4, 7, 10, and 14 days post-challenge (DPC) through direct plating on XLD agar to assess the persistence and dissemination of the challenge strains. Samples that exhibited no visible Salmonella growth on direct plating underwent an indirect enrichment process to confirm the presence or absence of the pathogen. One gram of cecal content or liver tissue was inoculated into 10-ml Rappaport-Vassiliadis (RV) broth for enrichment. The enrichment cultures were serially diluted to 0.1, 0.01, and 0.001, with three replicate tubes allocated per dilution. The tubes were incubated overnight at 42°C ± 2°C in a shaking water bath to enhance the selective growth of Salmonella spp. The enriched samples were streaked onto XLD agar and incubated at 35°C ± 2°C for 24 ± 2 hours for bacterial isolation and confirmation. Enumeration of bacterial load using the most probable number MPN methodTo quantify the bacterial load, the MPN method was applied following established protocols (Blodgett, 2005; Blodgett, 2006). MPN was calculated as follows (Bansal et al., 2010)
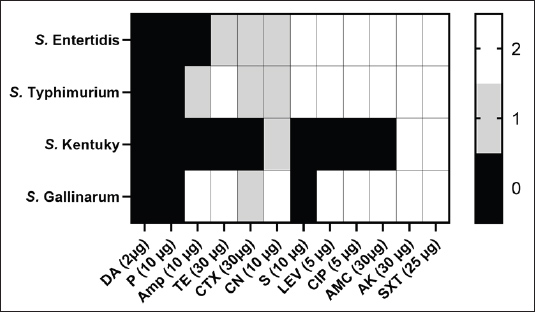
Here, P represents the number of positive tubes, N is the total quantity of samples (in grams) in all negative tubes, and T is the total quantity of samples (in grams) in all tubes. Cecal droppings and liver samples were used to determine bacterial shedding and translocation. The procedures were implemented as follows. Bacterial shedding in the cecal contentCecal content (1 g) was collected from euthanized or freshly deceased birds. The samples were suspended in Buffered Peptone Water (HiMedia, India) at a 1:10 ratio and incubated for 24 hours at 37°C to promote microbial growth. Subsequently, 10 ml of RV broth was mixed with 100 μl of the enhanced broth culture and cultured for 24 hours at 42°C. After enrichment, each RV broth culture was streaked onto XLD agar and incubated for 24 hours at 37°C to detect Salmonella. Bacterial translocation to the liverTo assess bacterial translocation from the gastrointestinal tract to the systemic circulation, 1 g of liver samples were collected, weighed, and homogenized in sterile BPW using a mortar and pestle under aseptic conditions. The homogenized suspension was serially diluted in 10-fold increments up to 105. Aliquots from each dilution were plated onto XLD agar and incubated at 37°C for 24 hours. Bacterial colonies obtained from direct culture were counted, and the bacterial load was recorded to determine the extent of systemic infection. Ethical approvalThe animal phase of the study was conducted in accordance with the Good Laboratory Practice (GLP) provisions. The Animal Care and Use Committee (ACUC), Middle East for Vaccines (MEVAC) (n°ME-BP20-2403) reviewed and approved all the experimental procedures (Dated: 15 January 2024). Statistical analysisAn unpaired t-test was used to detect differences in antibody responses and recovered bacteria between vaccinated and control groups. Statistical significance was defined as a p-value < 0.05. ResultsCultivation and antimicrobial susceptibility testingIn this study, the colonial morphology of Salmonella isolates was investigated using MacConkey agar and XLD. Smooth, colorless colonies were identified on MacConkey agar. Red colonies with black centers appear on XLD agar. In vitro sensitivity tests revealed that the four tested Salmonella strains were highly sensitive to amikacin and sulfamethoxazole/trimethoprim, followed by levofloxacin, ciprofloxacin, amoxicillin, and clavulanic acid. The isolates were highly clindamycin-resistant and penicillin, followed by ampicillin, tetracycline, and streptomycin. Intermediate sensitivity was noted for cefotaxime and gentamicin (Fig. 2). MDR was detected among the four Salmonella isolates, indicating the resistance for ≥3 antibiotic classes (Table 1).
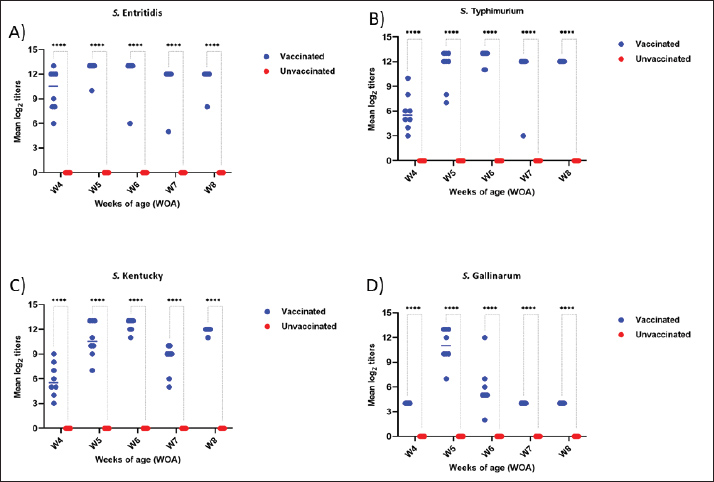
Fig. 2. Antimicrobial resistance of selected Salmonella isolates recovered from chickens. Evaluation of the physicochemical properties of the vaccinePOULMMUNE™ SALMONELLA BCD appeared as a whitish W/O emulsion. The conductivity of the vaccine was 0.01 µS × cm-1, and the viscosity was 48 mPa.s. The emulsion exhibited stability under centrifugation conditions of 6,000 rpm for 15 minutes. The pH of the final product was 6.4. Vaccine safetyNo local response, general symptoms, or mortality were recorded either after primary or booster vaccination. The results of gross pathological examinations suggest that the subcutaneous administration of the vaccine to chickens does not induce any adverse effects. Examination of the tissue surrounding the injection site revealed no abnormalities. The vaccine exhibited full absorption after 21 days of repeated administration. Humoral antibody response to SE, SG, SK, and STHumoral antibodies against SE, SG, SK, and ST were significantly different between vaccinated and unvaccinated birds (p < 0.00001) (p < 0.00001) (Fig. 3). After primary immunization, antibody levels increased for all strains except SG, which showed a notable decrease before the challenge. Seroconversion patterns varied, with strong and consistent antibody responses against ST, SE, and SK over time, whereas the response to SG was weaker and declined by week 5. In contrast, unvaccinated birds showed minimal antibody responses, underscoring the vaccine’s effectiveness in stimulating immunity, particularly against ST, SE, and SK (Fig. 3).
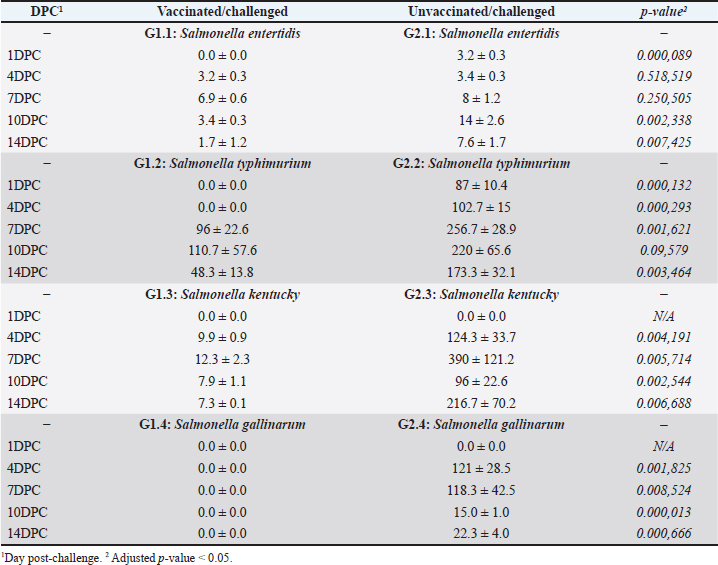
Fig. 3. Serological monitoring results of the vaccinated and unvaccinated groups. Cecal shedding and liver translocation in SE-, ST-, SK-, and SG-challenged chickensIn the control groups, Salmonella shedding increased starting from the fourth day after the challenge (4DPC), as determined through direct plating. The ST group had the highest rate of shedding, followed by the SK and SE groups. The SG group exhibited the minimal rate of shedding. Vaccinated birds showed a notable decrease in fecal shedding compared with the control groups. The shedding decreased consistently for all serovars in 14 DPC (Table 2). Bacterial translocation was detected by direct plating from the liver. The degree of invasiveness exhibited by the Salmonella serovars was diverse in the control group. SK exhibited the highest level of invasiveness, followed by ST and SG. The SE exhibited the lowest level of invasiveness. No Salmonella colonies were recovered from the liver samples of vaccinated birds by direct plating, indicating that systemic dissemination was effectively prevented by vaccination. The degree of liver invasion in vaccinated birds was significantly lower than that in the control group (p < 0.05). Additionally, enrichment of liver and cecal samples revealed a substantial reduction in Salmonella MPN/g in vaccinated birds (Tables 2 and 3). In the liver, vaccinated birds showed significantly lower bacterial loads for all strains, especially from DPC 4 days onward. The reduction was most pronounced in ST and SE in 1DPC, whereas significant differences appeared for SK and SG in 4DPC. In the cecum, vaccination effectively reduced bacterial loads for SE, ST, and SK, starting at 4DPC, with reductions for SG observed in 10DPC and 14DPC (Table 3). These findings demonstrate the broad efficacy of the vaccine in controlling bacterial colonization and dissemination in challenged chickens. Table 2. Most probable number (CFU/g) of cecal contents.
Table 3. Most probable number (CFU/g) of liver translocations.
DiscussionThis study evaluated the safety, immunogenicity, and protective efficacy of a novel tetravalent inactivated Salmonella vaccine, POULMMUNE™ SALMONELLA BCD, in specific pathogen-free chickens. The vaccine, targeting SE, ST, SK, and SG, demonstrated a favorable safety profile, elicited robust humoral immune responses, and significantly reduced shedding and translocation of Salmonella. These findings contribute to the growing body of evidence supporting the use of vaccination as a critical strategy for controlling Salmonella in poultry and mitigating the risk of human exposure. POULMMUNE™ SALMONELLA BCD induced significant increases in antibody titers against SE, ST, and SK. However, a decline in antibody titers against SG was observed over time. This differential serotype-specific immune response is consistent with previous studies highlighting the influence of serovar-specific antigenic composition on CII (Ijaz et al., 2021). Variations in host adaptation and virulence factors among Salmonella serovars can also contribute to these differences (Köster et al., 2013). SG, being highly host-adapted to chickens, may elicit a distinct immune response compared to the more zoonotic serovars. Future vaccine optimization may involve enhancing SG antigenicity to improve long-term protection. The observed strong antibody responses against SE and ST agree with previous research demonstrating the essential role of humoral immunity in reducing the colonization and systemic spread of Salmonella (Barrow et al., 2012). Inactivated Salmonella vaccines, particularly those employing oil adjuvants like MONTANIDE™ ISA 70 VG, have been shown to effectively stimulate antibody production and reduce fecal shedding (Gast et al., 1993; Enbaawy et al., 2013). This suggests that POULMMUNE™ SALMONELLA BCD formulation is effective in inducing a protective humoral response. The assessment of immune responses and bacterial shedding at various time points revealed fluctuating shedding rates before day 7 post-challenge, followed by a significant reduction by day 14. The initial fluctuations may reflect the dynamics of the host-pathogen interaction, including the initial colonization and immune response development. The subsequent decline in shedding likely reflects the vaccine-induced immune response’s increasing effectiveness. The complete cessation of SG shedding by day 14 aligns with previous reports demonstrating the efficacy of vaccination in controlling SG infections in poultry (De Paiva et al., 2009). However, the observed fluctuating shedding pattern for SK warrants further investigation. Although SK is less frequently studied than SE and ST, its potential role as a foodborne pathogen and its increasing prevalence in some regions necessitates a better understanding of its epidemiology and control strategies (Le Hello et al., 2011; Chen et al., 2021; Siddique et al., 2024). The observation of ST persistence and systemic dissemination in this study is consistent with its known intracellular survival mechanisms (Wang et al., 2021; Roy Chowdhury et al., 2025). The ability of ST to evade phagocytic killing within macrophages contributes to its prolonged shedding and potential for systemic spread. Although inactivated vaccines are effective in stimulating humoral immunity, they may not fully address intracellular Salmonella, highlighting the importance of booster vaccinations to maintain protective immunity. Live-attenuated Salmonella vaccines, which induce both humoral and cell-mediated immunity, have shown promise in providing robust protection against intracellular Salmonella (Curtiss 2024; Wang et al., 2024). However, the use of live vaccines in poultry is often limited by concerns about shedding and the potential for reversion to virulence. POULMMUNE™ SALMONELLA BCD demonstrated excellent safety in SPF chickens, with no observed local or systemic adverse reactions. This is consistent with the safety profile of other oil-adjuvanted Salmonella vaccines used in poultry (Stone et al., 1993; Ahmed et al., 2013). The absence of injection site reactions and complete absorption of the vaccine formulation suggest that the vaccine is well-tolerated and unlikely to cause local tissue damage. The robust antibody responses observed against SE and ST corroborate previous findings on inactivated Salmonella vaccines (Stone et al., 1993; Curtiss III 2024; Wang et al., 2024). The observed decline in SG-specific antibody titers suggests that optimizing the SG antigen component of the vaccine may be necessary to improve long-term protection against this serovar. While live-attenuated vaccines induce both humoral and cell-mediated immunity, they pose risks such as environmental shedding and reversion to virulence. In contrast, inactivated vaccines like POULMMUNE™ SALMONELLA BCD, provide a safer alternative with a well-defined antigenic composition. However, their reliance on humoral immunity underscores the need for periodic booster doses to maintain protective efficacy. Future research directions include optimizing inactivated vaccine formulations to enhance cross-protection against different serovars of Salmonella, exploring novel adjuvants that stimulate both humoral and cell-mediated immune responses, and conducting field trials in commercial poultry settings to evaluate vaccine efficacy under real-world conditions. Further research on the epidemiology and control of SK and SG in poultry is warranted. ConclusionPOULMMUNE™ SALMONELLA BCD exhibits promising potential as an effective and safe tool for restricting Salmonella infections in poultry. Its ability to reduce bacterial shedding and translocation, coupled with its capacity to induce robust humoral immune responses, makes it a valuable candidate for inclusion in integrated Salmonella control programs. Further research is warranted to optimize vaccine formulations and evaluate their performance in commercial settings. The development of effective vaccines against MDR Salmonella is crucial for protecting both animal and public health. Conflict of interestThe authors have no competing interests to declare. Consent for publicationNot applicable. Authors’ contributionAll authors contributed equally. Availability of data and materialsThe datasets used and analyzed in this study are available from the corresponding author upon reasonable request. ReferencesAdenaike, O., Olonitola J Ameh. and Whong. 2016. Multidrug resistance and multiple antibiotic resistance index of Escherichia coli strains isolated from retailed smoked fish. J. Natural Sci. Res. 6(9), 18–27. Andrews, W.H., Wang, H., Jacobson, A., Hammack, T. and Food and Drug Administration 2018. Bacteriological analytical manual (BAM) chapter 5: Salmonella. Bacteriological Analytical Manual 110, 1-25. Apun, K., Chong, Y. L., Abdullah, M. and Micky, V. 2008. Antimicrobial susceptibilities of Escherichia coli isolates from food animals and wildlife animals in Sarawak, East Malaysia. Atterbury, R., Allen, J.C. and Mas, R.D. 2009. Salmonella colonization of laying hens following vaccination with killed and live attenuated commercial Salmonella vaccines. Vet. Rec. 165(17), 493–496. Bansal, A., Jones, T.M., Abd, S.J., Danyluk, M.D. and Harris, L.J. 2010. Most-probable-number determination of Salmonella levels in naturally contaminated raw almonds using two sample preparation methods. J. Food Prot. 73(11), 1986–1992. Barrow, P.A., Jones, M.A., Smith, A.L. and Wigley, P. 2012. The long view: salmonella–the last forty years. Avian Pathol. 41(5), 413–420. Berhanu, G. and Fulasa. 2020. Pullorum disease and fowl typhoid in poultry: a review. Br. J. Poult. Sci. 9, 48–56. Blodgett, R.J. 2005. Serial dilution with a confirmation step. Food. Microbiol. 22(6), 547–552. Blodgett, R.J. 2006. Testing deviation for a set of serial dilution most probable numbers from a poisson-binomial model. J. AOAC. Int. 89(1), 166–171. Brenner, F.W., Villar, R.G., Angulo, F.J., Tauxe, R. and Swaminathan, B. 2000. Salmonella nomenclature. J. Clin. Microbiol. 38(7), 2465–2467. Chen, H., Song, J., Zeng, X., Chen, D., Chen, R., Qiu, C. and Zhou, K. 2021. National prevalence of Salmonella enterica serotype Kentucky ST198 with high-level resistance to ciprofloxacin and extended-spectrum cephalosporins in China, 2013 to 2017. Msystems 6(1), 10. Clinical and Laboratory Standards Institute. 2020. Performance standards for antimicrobial susceptibility testing, Clinical and Laboratory Standards Institute, Wayne, PA. Curtiss, R. 2024. Vaccines to control Salmonella in poultry. Avian Dis. 67(4), 427–440. De Paiva, J., Penha Filho, Y. Arg., üello M Da., Silva Y Gardin., Resende A Berchieri. and Junior, L. Sesti. 2009. Efficacy of several Salmonella vaccination programs against experimental challenge with Salmonella gallinarum in commercial brown layer and broiler breeder hens. Braz. J. Poultry Sci. 11, 65–72. Deguchi, K., Yokoyama, E., Honda, T. and Mizuno, K. 2009. Efficacy of a novel trivalent inactivated vaccine against the shedding of Salmonella in a chicken challenge model. Avian Dis. 53(2), 281–286. Desin, T.S., Köster, W. and Potter, A.A. 2013. Salmonella vaccines in poultry: past, present and future. Expert Rev. Vaccines 12(1), 87–96. Dieckmann, R. and Malorny, B. 2011. Rapid screening of epidemiologically important Salmonella enterica subsp. enterica serovars by whole-cell matrix-assisted laser desorption ionization-time of flight mass spectrometry. Appl. Environ. Microbiol. 77(12), 4136–4146. El-Enbaawy, M.I., Ahmed M Sadek. and Ibrahim. 2013. Protective efficacy of Salmonella local strains representing groups B, C, D and E in a prepared polyvalent formalin inactivated oil adjuvant vaccine in layers. Int. J. Microbiol. Res. 4, 288–295. Ezekiel, C., Olarinmoye J Oyinloye. and Olaoye , A. Edun. 2011. Distribution, antibiogram and multidrug resistance in Enterobacteriaceae from commercial poultry feeds in Nigeria. Afr. J. Microbiol. Res. 5(3), 294–301. Gast, R.K., Stone, H.D. and Holt, P.S. 1993. Evaluation of the efficacy of oil-emulsion bacterins for reducing fecal shedding of Salmonella enteritidis by laying hens. Avian. Dis. 1085, 1085–1091. Ijaz, A., Veldhuizen, E.J.A., Broere, F., Rutten, V.P.M.G. and Jansen, C.A. 2021. The interplay between Salmonella and intestinal innate immune cells in chickens. Pathogens 10(11), 1512. Jiang, L., Wang, P., Song, X., Zhang, H., Ma, S., Wang, J., Li, W., Lv, R., Liu, X., Ma, S., Yan, J., Zhou, H., Huang, D., Cheng, Z., Yang, C., Feng, L. and Wang, L. 2021. Salmonella Typhimurium reprograms macrophage metabolism via T3SS effector SopE2 to promote intracellular replication and virulence. Nature. Commun. 12(1), 879. Le Hello, S., Hendriksen, R.S., Doublet, B., Fisher, I., Nielsen, E.M., Whichard, J.M., Bouchrif, B., Fashae, K., Granier, S.A., Jourdan-da Silva, N., Cloeckaert, A., Threlfall, E.J., Angulo, F.J., Aarestrup, F.M., Wain, J. and Weill, F.X. 2011. International spread of an epidemic population of Salmonella enterica serotype Kentucky ST198 resistant to ciprofloxacin. J. Infect. Dis. 204(5), 675–684. Marin, C., Lorenzo-Rebenaque, L., Laso, O., Villora-Gonzalez, J. and Vega, S. 2021. Pet reptiles: a potential source of transmission of multidrug-resistant Salmonella. Front. Vet. Sci. 7, 613718. Okamura, M., Tachizaki, H., Kubo, T., Kikuchi, S., Suzuki, A., Takehara, K. and Nakamura, M. 2007. Comparative evaluation of a bivalent killed Salmonella vaccine to prevent egg contamination with Salmonella enterica serovars Enteritidis, Typhimurium, and Gallinarum biovar Pullorum, using 4 different challenge models. Vaccine 25(25), 4837–4844. Roy Chowdhury, A., Hajra, D., Mukherjee, D., Nair, A.V. and Chakravortty, D. 2025. Functional OmpA of Salmonella Typhimurium provides protection from lysosomal degradation and inhibits autophagic processes in macrophages. J. Infect. Dis. 231(3), 716–728. Siddique, A., Wang, Z., Zhou, H., Huang, L., Jia, C., Wang, B., Ed-Dra, A., Teng, L., Li, Y. and Yue, M. 2024. The evolution of vaccines development across Salmonella Serovars among animal hosts: a systematic review. Vaccines 12(9), 1067. Tarabees, R., Elsayed, M.S.A., Shawish, R., Basiouni, S. and Shehata, A.A. 2017. Isolation and characterization of Salmonella Enteritidis and Salmonella Typhimurium from chicken meat in Egypt. J. Infectdeveloping. Countries. 11(04), 314–319. Van Immerseel, F., De Buck, J., Pasmans, F., Bohez, L., Boyen, F., Haesebrouck, F. and Ducatelle, R. 2004. Intermittent long-term shedding and induction of carrier birds after infection of chickens early posthatch with a low or high dose of Salmonella Enteritidis. Poultry. Sci. 83(11), 1911–1916. Williams, J.E. and Whittemore, A.D. 1971. Serological diagnosis of pullorum disease with the microagglutination system. Appl. Microbiol. 21(3), 394–399. | ||
| How to Cite this Article |
| Pubmed Style Elfeil WK, Essam S, Mostafa E, Ismail DE, Fathy M, Altaib H, Basuony ME, Sedeek A, Hisham I, Wasfy M, Kilany WH, Abu-elala NM. Efficacy of a multivalent inactivated Salmonella vaccine against fecal shedding and organ dissemination in chickens. Open Vet. J.. 2025; 15(12): 6767-6776. doi:10.5455/OVJ.2025.v15.i12.55 Web Style Elfeil WK, Essam S, Mostafa E, Ismail DE, Fathy M, Altaib H, Basuony ME, Sedeek A, Hisham I, Wasfy M, Kilany WH, Abu-elala NM. Efficacy of a multivalent inactivated Salmonella vaccine against fecal shedding and organ dissemination in chickens. https://www.openveterinaryjournal.com/?mno=291007 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.55 AMA (American Medical Association) Style Elfeil WK, Essam S, Mostafa E, Ismail DE, Fathy M, Altaib H, Basuony ME, Sedeek A, Hisham I, Wasfy M, Kilany WH, Abu-elala NM. Efficacy of a multivalent inactivated Salmonella vaccine against fecal shedding and organ dissemination in chickens. Open Vet. J.. 2025; 15(12): 6767-6776. doi:10.5455/OVJ.2025.v15.i12.55 Vancouver/ICMJE Style Elfeil WK, Essam S, Mostafa E, Ismail DE, Fathy M, Altaib H, Basuony ME, Sedeek A, Hisham I, Wasfy M, Kilany WH, Abu-elala NM. Efficacy of a multivalent inactivated Salmonella vaccine against fecal shedding and organ dissemination in chickens. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6767-6776. doi:10.5455/OVJ.2025.v15.i12.55 Harvard Style Elfeil, W. K., Essam, . S., Mostafa, . E., Ismail, . D. E., Fathy, . M., Altaib, . H., Basuony, . M. E., Sedeek, . A., Hisham, . I., Wasfy, . M., Kilany, . W. H. & Abu-elala, . N. M. (2025) Efficacy of a multivalent inactivated Salmonella vaccine against fecal shedding and organ dissemination in chickens. Open Vet. J., 15 (12), 6767-6776. doi:10.5455/OVJ.2025.v15.i12.55 Turabian Style Elfeil, Wael K., Salma Essam, Esraa Mostafa, Doaa E. Ismail, Mohamed Fathy, Hend Altaib, Mohamed E. Basuony, Ahmed Sedeek, Islam Hisham, Momtaz Wasfy, Walid H. Kilany, and Nermeen M. Abu-elala. 2025. Efficacy of a multivalent inactivated Salmonella vaccine against fecal shedding and organ dissemination in chickens. Open Veterinary Journal, 15 (12), 6767-6776. doi:10.5455/OVJ.2025.v15.i12.55 Chicago Style Elfeil, Wael K., Salma Essam, Esraa Mostafa, Doaa E. Ismail, Mohamed Fathy, Hend Altaib, Mohamed E. Basuony, Ahmed Sedeek, Islam Hisham, Momtaz Wasfy, Walid H. Kilany, and Nermeen M. Abu-elala. "Efficacy of a multivalent inactivated Salmonella vaccine against fecal shedding and organ dissemination in chickens." Open Veterinary Journal 15 (2025), 6767-6776. doi:10.5455/OVJ.2025.v15.i12.55 MLA (The Modern Language Association) Style Elfeil, Wael K., Salma Essam, Esraa Mostafa, Doaa E. Ismail, Mohamed Fathy, Hend Altaib, Mohamed E. Basuony, Ahmed Sedeek, Islam Hisham, Momtaz Wasfy, Walid H. Kilany, and Nermeen M. Abu-elala. "Efficacy of a multivalent inactivated Salmonella vaccine against fecal shedding and organ dissemination in chickens." Open Veterinary Journal 15.12 (2025), 6767-6776. Print. doi:10.5455/OVJ.2025.v15.i12.55 APA (American Psychological Association) Style Elfeil, W. K., Essam, . S., Mostafa, . E., Ismail, . D. E., Fathy, . M., Altaib, . H., Basuony, . M. E., Sedeek, . A., Hisham, . I., Wasfy, . M., Kilany, . W. H. & Abu-elala, . N. M. (2025) Efficacy of a multivalent inactivated Salmonella vaccine against fecal shedding and organ dissemination in chickens. Open Veterinary Journal, 15 (12), 6767-6776. doi:10.5455/OVJ.2025.v15.i12.55 |