| Research Article | ||
Open Vet. J.. 2026; 16(2): 824-833 Open Veterinary Journal, (2026), Vol. 16(2): 824-833 Research Article Evaluation of acute phase protein, cardiac marker, and antioxidant enzymes in Madura Cattle infected with foot-and-mouth diseaseDwi Kristanto1*, Nabila Tsabitsa Ramadhani1, Brigitta Sekar Ayu1, Rif Anna1, Ovia Mustika Kusumawardhani1, Aprilia Rizky Riadini1, Tiara Widyaputri1, Dodik Prasetyo1, Siti Khairunissa1 and Wike Andre Septian21Faculty of Veterinary Medicine, Universitas Brawijaya Jalan Puncak Dieng, Malang, Indonesia 2Faculty of Animal Science, Universitas Brawijaya Jalan Veteran, Ketawanggede, Lowokwaru, Malang City, Indonesia *Corresponding Author: Dwi Kristanto. Faculty of Veterinary Medicine, Universitas Brawijaya Jalan Puncak Dieng, Malang, Indonesia. Email: dkristanto [at] ub.ac.id Submitted: 17/10/2025 Revised: 11/01/2026 Accepted: 25/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
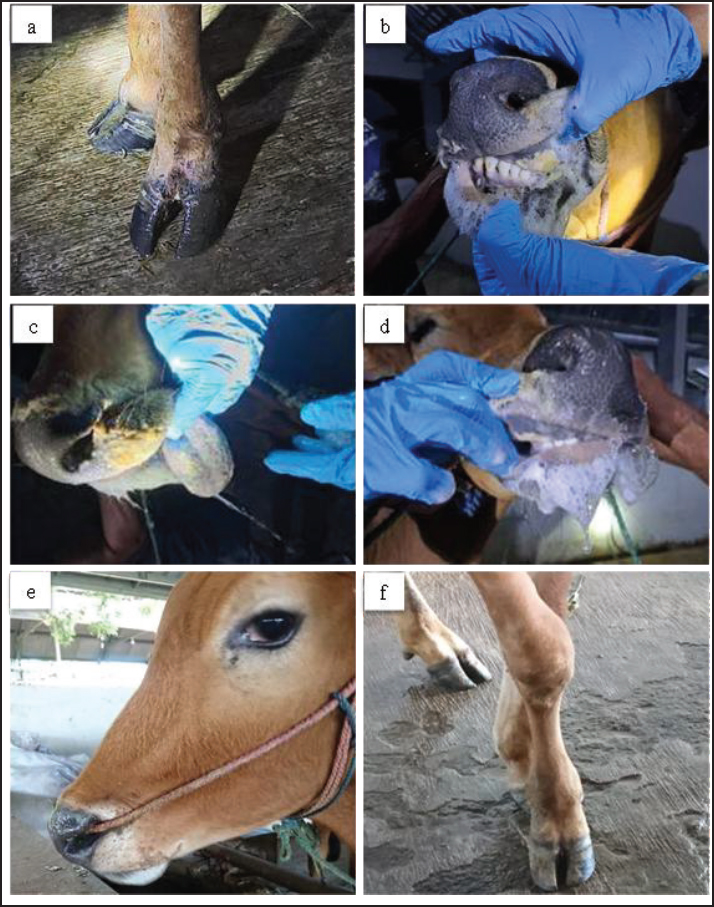
AbstractBackground: Foot-and-mouth disease (FMD) is a highly contagious viral disease in cloven-hoofed animals and remains a significant cause of economic loss in the livestock sector. In addition to causing characteristic clinical symptoms, FMD infection triggers a systemic inflammatory response and oxidative stress that can alter various biochemical and cardiac parameters. However, information on the biochemical and antioxidant profiles of local Indonesian cattle, particularly Madura cattle, remains limited. Aim: This study aimed to evaluate changes in acute phase proteins (APPs), cardiac markers, and antioxidant enzyme activity in Madura cattle naturally infected with FMD and to analyze the relationships between these biochemical parameters during the infection and recovery phases. Methods: Ten 2.5 ± 0.5-year-old male Madura cattle clinically diagnosed with FMD had blood samples taken twice during the infection phase (days 3–7) and the recovery phase (days 28–30). The biochemical parameters analyzed included creatine kinase (CK), CK–myocardial band (CK-MB), lactate dehydrogenase (LDH), cardiac troponin I (cTn-I), haptoglobin (Hp), C-reactive protein (CrP), superoxide dismutase (SOD), glutathione peroxidase (GPx), and malondialdehyde (MDA) using enzyme-linked immunosorbent assay and spectrophotometry methods. Data were analyzed using paired t-tests and Spearman's correlation with a significance level of p < 0.05. Results: There were significant differences (p < 0.05) between the infection and recovery phases in CK and CK-MB parameters, while LDH, Hp, CrP, GPx, MDA, and SOD showed no significant differences. A strong positive correlation was found between CK and CK-MB in the infection phase (r=0.85; p < 0.01), indicating simultaneous damage to skeletal muscle and myocardium. In the recovery phase, there was a positive relationship between Hp and GPx (r=0.80; p < 0.01), indicating recovery of antioxidant function as the inflammatory response decreased. Conclusion: FMD infection in Madura cattle causes physiological changes characterized by increased cardiac biomarkers and APPs and decreased antioxidant enzyme activity. The positive relationship between Hp and GPx reflects the link between the inflammatory process and oxidative stress during recovery. The combination of APP parameters and antioxidant enzymes can be used as biochemical indicators to assess the severity and health status of FMD-infected cattle. Keywords: Acute phase protein, Cardiac marker, Foot-and-mouth disease, Madura, Oxidative stress. IntroductionFoot-and-mouth disease (FMD) is an acute infectious disease that affects cloven-hoofed animals (OIE, 2015). Aphthovirus causes this disease from the Picornaviridae family with seven strains (A, O, C, SAT1, SAT2, SAT3, and Asia 1) that are endemic in various countries around the world, including parts of Asia and most of Africa and the Middle East (Khoshvaghti et al., 2014). This virus is non-enveloped, with a single-stranded RNA genome that is positive-sense, non-segmented, and enclosed in an icosahedral capsid (Eid et al., 2022). This virus spreads rapidly through various direct and indirect contacts and air (Soltani et al., 2020). FMD in cattle is clinically characterized by high fever, loss of appetite, hypersalivation, and vesicles in the interdigital clefts, oral cavity, and mammary glands (Adjid, 2020; Soltani et al., 2020). The transmission of FMD occurs very rapidly, resulting in a high morbidity rate of nearly 100% and a low mortality rate of 5% in adult animals (Mahmoud and Galbat, 2017). Based on the report by Sutawi et al. (2023), FMD in Indonesia in 2022 spread to 19 provinces across 207 districts/cities, with 492,894 infected cattle, 334,838 recovered, and 6,435 dead. This outbreak caused economic losses because most livestock are a source of animal-based food consumption, affecting the balance of animal-based food in Indonesia, such as meat and milk, which ultimately impacts national food security and the farmer's exchange rate (Zainuddin et al., 2023). Techniques for detecting FMD viruses, such as enzyme-linked immunosorbent assay (ELISA), complement fixation test, and reverse transcriptase-polymerase chain reaction, are effective gold standard methods for FMD diagnosis that have been widely reviewed and implemented by various parties (Adjid, 2020). However, blood chemical biomarker tests correlating with FMD disease, such as antioxidant enzyme activity, acute phase proteins (APPs), and musculoskeletal and cardiac functions, have not been widely reviewed (Adjid, 2020). According to Sobhy et al. (2018), using cardiac biomarkers in FMD diagnosis is essential because it relates to myocarditis. Myocarditis caused an increase in cardiac troponin I (cTn-I) and lactate dehydrogenase (LDH) enzyme levels (Soltani et al., 2020). Studies indicate that cTn-I has been used as a diagnostic biomarker because this enzyme is released into the blood immediately after myocardial injury (Sobhy et al., 2018). In addition, research by Soltani et al. (2020) states that other biomarkers that undergo significant changes in the diagnosis of myocardial infarction are the enzymes LDH, creatine kinase myocardial band (CK-MB), and CK. Other important markers to examine, especially in cases of FMD, are haptoglobin and C-reactive protein (CrP). Lee et al. (2003) stated that CrP is a serum APP that responds most rapidly to infection. This protein recognizes phosphocholine on the cytoplasmic membrane of cells, activates the complement cascade, and stimulates macrophages to destroy infectious agents. CrP can regulate the immune system during the early stages of infection. The CrP protein plays a role in destroying infectious agents, minimizing tissue damage, and facilitating tissue repair and regeneration. Viral infections can activate the immune system by causing the release of free radicals such as reactive oxygen species (ROS) and reactive nitrogen species (RNS), which can potentially induce oxidative stress (Mousa and Galal, 2013). Antioxidants serve as biochemical parameters for monitoring oxidative stress to determine the pathogenesis and severity of infection, evaluate the metabolic status of animals, and improve supportive treatment procedures for diseases (Tufarelli et al., 2023). Important antioxidant enzymes that prevent free radicals and the onset of lipid peroxidation include superoxide dismutase (SOD), glutathione peroxidase (GPx), and catalase (CAT) (Yurdakul and Yildirim, 2018). SOD and GPx enzymes are antioxidant enzymes that serve as the first line of defense against free radicals (Soltani et al., 2020). Soltani et al. (2020) also emphasized that information related to the biochemical data of livestock naturally infected with the FMD virus is still limited. Research on the biochemical analysis of blood from native Indonesian cattle with FMD has never been conducted. Given the important relationship described earlier, it is necessary to measure blood biochemical parameters, including antioxidant enzyme activity, cardiac marker activity, and APP in Madura cattle naturally infected with FMD, in order to support diagnosis and provide further insight to improve FMD prevention and mitigation in Madura cattle. Materials and MethodsAnimals, experimental design, and blood collectionThe study was conducted from January to March 2023, collecting blood samples from Madura cattle naturally infected with FMD at the Agro Techno Park of Brawijaya University, Kepanjen District, Malang Regency, East Java. The animals used in this study were 10 male Madura cattle with an average age of 2.5 ± 0.5-years and a body weight of 242 ± 68 kg, which were intensively raised in group pens. The animals used had been diagnosed with FMD based on clinical symptoms and physical examinations, such as decreased appetite, hypersalivation, pyrexia, tachycardia, tachypnea, enteritis, lameness, and vesicular lesions in the interdigital area and oral cavity (Fig. 1). Serological detection of FMD also was performed using a non-structural protein (NSP) ELISA to identify antibodies against FMD virus non-structural protein, enabling differentiation between naturally infected and vaccinated. Serum samples were analyzed according to the manufacturer’s protocol, and results were interpreted based on the recommended cutoff values.
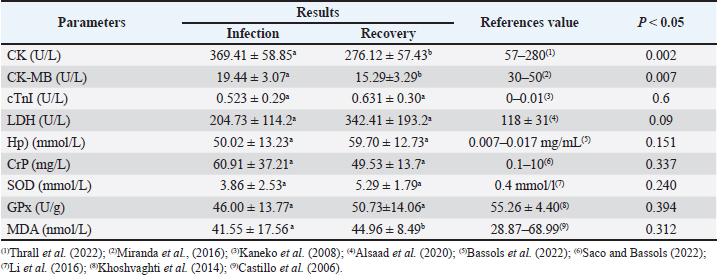
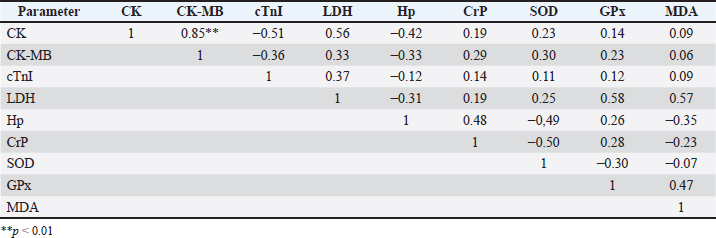
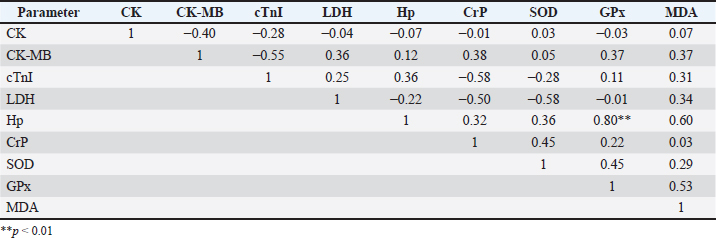
Fig. 1. Clinical symptoms during the infection phase include interdigital lesions, ulcers on the oral mucosa, and hypersalivation (a–d); Madura cattle in the recovery phase showed no symptoms of FMD (e,f). Blood sampling was performed twice, during the infection and recovery phases. Blood sampling during the infection phase was performed between days 3 and 7, when the animals showed clinical symptoms. The second sampling was performed during the recovery phase, between 28 and 30 days post-infection, when the animals were declared healthy. It showed no clinical symptoms of FMD through a series of physical and clinical examinations. Blood samples were collected in 3 mL volumes from the coccygeal vein using plain tubes (Becton Dickinson®, US). The blood samples were then centrifuged at 3500 rpm at room temperature for 10 minutes to obtain serum samples, which were stored in 1.5 mL microtubes (Safe-Lock 3810X; Eppendorf®) and stored in a freezer at (−20) °C. Serum biochemical preparation and analysisSerum sample analysis for CK, CK-MB, and LDH parameters was performed using a Semi-auto Blood Chemistry Analyzer ICHEM-I UBIO 535 with a spectrophotometry method, which measures absorbance by passing light with a specific wavelength through a glass object or cuvette with a wavelength of 340 nm (Brune et al., 2022). Measurements of MDA, SOD, GPx, haptoglobin (Hp), CrP, and cTnI activities were performed using the Sandwich ELISA method with the following catalog products: BT Laboratory, Shanghai, China, Cat. No. E0198Bo for MDA test; BT Laboratory, Shanghai, China, Cat. No. E0003Bo for SOD test; BT Laboratory, Shanghai, China, Cat. No. E0006Bo for GPx test; BT Laboratory, Shanghai, China, Cat. No. E2263Bo for haptoglobin test; LifeSpan BioSciences, USA, Cat. No. LS-F38924 for CrP test, and LifeSpan BioSciences, USA, Cat. No. LS-F14626 for cTnI. The microtiter plates provided in this kit were pre-coated with specific antibodies. Standards or samples were added to the appropriate microtiter plate wells, followed by specific biotin-conjugated antibodies. Next, avidin conjugated with horseradish peroxidase (HRP) is added to each well of the microplate and incubated. After the TMB substrate solution is added, only the wells containing antigen, biotin-conjugated antibody, and enzyme-conjugated avidin showed a color change. The enzyme-substrate reaction is stopped by adding sulfuric acid solution, and the color change is measured using spectrophotometry at a wavelength of 450 ± 10 nm. The concentration of the parameter in the sample is then determined by comparing the sample's optical density (OD) with the standard curve (Santosa, 2020). Statistical analysisThe variables observed in this study included antioxidant enzyme activity, cardiac markers, and APPs in Madura cattle infected with FMD. The data obtained from this study were analyzed statistically using software. The data results were analyzed using IBM SPSS Statistics for Windows, version 26.0. Statistical analysis uses a paired t-test with a significance level of 0.05 (p < 0.05). Meanwhile, Pearson correlation analysis is used to measure the relationship between blood chemistry parameters in the infection and recovery phases with a significance level of p < 0.05. The results of the analysis were presented in the form of means, SD, SE, correlation directions, and significance levels. Ethical approvalAll procedures involving animals in this study were approved by the Animal Ethics Committee of the Institute of Biosciences, Universitas Brawijaya, Indonesia, and were conducted in accordance with national and international guidelines on the treatment of laboratory animals (No. 039-KEP-UB-2023). ResultsIn the present study, APPs, cardiac biomarkers, and antioxidant enzyme activities were evaluated in Madura cattle naturally infected with FMD during the infection and recovery phase. Data analysis showed that the parameters CK and CK-MB in Madura cattle (Table 1) showed significant differences between the infection phase and the recovery phase (p < 0.05). At the same time, parameters such as CrP, Hp, GPx, cTnI, MDA, LDH, and SOD in the infection and recovery phases showed no differences (p > 0.05). The results of the correlation analysis between blood chemistry parameters showed a positive relationship (r=0.85; p < 0.05) between CK and CK-MB in the infection phase (Table 2). In contrast, in the recovery phase, there was a positive relationship (r=0.80; p < 0.05) between Hp and GPx (Table 3). Meanwhile, no relationship was found in other parameters. Table 1. Results of the analysis of acute phase protein values, cardiac enzyme activity, and antioxidant enzyme activity in Madura cattle naturally infected with FMD.
Table 2. The relationship between acute phase protein parameters, cardiac markers, and antioxidant enzymes during the infection period.
Table 3. The relationship between acute phase protein parameters, cardiac markers, and antioxidant enzymes during the recovery period.
DiscussionBiochemical analysis of serum samples can be a valuable tool for assessing animal health and helping to understand the pathogenesis of a disease (Soltani et al., 2020). The results of creatine kinase (CK) parameter testing in Madura cattle showed a significant difference between the infection phase and the recovery phase (p < 0.05) (Table 1). These results were consistent with the reports by Soltani et al. (2020) on cattle in Egypt and Tunca et al. (2008) on crossbred cattle in Turkey, which showed significant differences in CK levels between the infection phase and healthy cattle. FMD in the infection phase causes damage to various cells, including skeletal muscle and heart muscle cells, as indicated by increased levels of the enzyme CK in the blood. According to Soltani et al. (2020), CK is a biomarker for degeneration and necrosis in muscle cells. Increased CK activity also stems from increased muscle cell membrane permeability due to disease or trauma (Pathipati et al., 2020). Meanwhile, during the recovery phase, the healing process in skeletal and cardiac muscles affects CK concentration levels. Skeletal muscle cell regeneration, which accounts for 94%–100% of total CK, begins 4–5 days after the infection phase and peaks after 2 weeks. The results of CK-MB parameter testing showed a significant difference between the infection phase and the recovery phase (p < 0.05). These results were consistent with the reports by Alsaad et al. (2020) on local breed calves in Iraq, Tunca et al. (2008) on cross-breed calves in Turkey, Nikvand et al. (2018) on cattle in Iran, Nasr El-Deen et al. (2017) on Holstein cattle, and Hashem et al. (2018) and Soltani et al. (2020) on Egyptian cattle, which showed significant differences between the infection phase and healthy cattle. According to Soltani et al. (2020), after the virus replicates in the oropharynx, it then replicates in secondary sites, including the heart muscle, causing cardiac degeneration and necrosis, so that the CK-MB enzyme is released into the blood and enzyme levels increase. This study also found a strong positive correlation between increased CK and CK-MB levels. It is important to note that cattle infected with FMD showed increased CK as an indication of skeletal muscle damage, while increased CK-MB is caused by myocardial damage (Tunca et al., 2008; Soltani et al., 2020). There were no significant differences in cTnI levels during the infection and recovery phases (p < 0.05). Several studies have reported that cTnI levels during the infection and recovery phases of FMD in cattle or calves do not show statistically significant differences. For example, a study in Egypt found that mean cTnI levels in fatal and recovered calves were 2.794 ± 0.502 ng/mL and 1.196 ± 0.443 ng/mL, respectively, both significantly higher than healthy controls, but the difference between the infection and recovery phases was not prognostically significant (Sobhy et al., 2018). This suggests that the myocardial damage that occurs during FMD infection remains long-lasting, resulting in high cTnI levels during the recovery phase (Tunca et al., 2008; Hashem et al., 2018). The cTnI level in the recovery phase increases compared to the infection phase and can also be caused by a long and limited period of heart cell regeneration. Regeneration of heart muscle tissue is dominated by the formation of scar tissue or fibrosis after damage. After myocardial cell necrosis, tissue formation and granulation occur on days 3 to 7 (Tunca et al., 2008; Mahadappa et al., 2021; Deka et al., 2024). Fibrosis or scar tissue formation begins approximately 2 weeks after infection. Myocardial muscle cells undergo refinement and maturation within 2 months (Ghafoor et al., 2020). Another study also showed that elevated cTnI correlated with myocardial injury and loss of cTnI expression in cardiac tissue, with no significant differences between the acute and recovery phases (Tunca et al., 2008; Hashem et al., 2018). Thus, high cTnI levels in both phases reflect ongoing cardiac damage and limited regeneration in FMD-infected cattle. Therefore, cTnI is more useful as an indicator of cardiac damage than as a marker of the difference between the infection and recovery phases in FMD cases (Tunca et al., 2008; Hashem et al., 2018; Sobhy et al., 2018; Aly et al., 2020). The results of the study showed that LDH levels in the infection and recovery phases showed no significant differences (p > 0.05). This is because LDH is a widely distributed enzyme in various tissues and organs of the body, and its increased activity usually results from generalized cell damage, not specific to a particular phase of the disease. In FMD infection, tissue damage may not be severe or specific enough to cause significant changes in LDH levels between the infection and recovery phases (Klein et al., 2009). During the infection phase, the trend or tendency showed higher LDH concentrations compared to normal reference levels. This trend is consistent with studies by Aktas et al. (2015), Salim et al. (2019), and Soltani et al. (2020) on Fresian Holstein cattle and Alsaad et al. (2020) on local breed cattle, which stated that there was an increase in LDH concentration in calves infected with FMD virus compared to healthy animals. During the recovery phase, the trend showed an increase in LDH concentration compared to the infection phase, with levels higher than usual. On the other hand, physical examination results showed that the cattle no longer exhibited clinical symptoms. Lesions in the oral cavity and interdigital area were no longer visible, and the lymph glands were normal and not swollen. According to Sobrino et al. (2011), in ruminants, infection can persist and be asymptomatic. Both vaccinated and unvaccinated animals can experience continuous infection after the acute infection phase. This is due to the dynamic equilibrium between the host's immune response and the selection of viral antigenic variation in the upper respiratory tract mucosa (Sobrino et al., 2011). This statement is consistent with Stenfeldt et al. (2011), where several studies explain the mechanism of the inability of animals to clear the virus from the host's body, including due to kinetic variations in the response of FMD-infected hosts. These viral mutations cause antigenic variation, or differences in innate immune responses. The results of Hp concentration testing showed no significant difference between the infection phase and the recovery phase (p > 0.05). These results were consistent with the report by Badiei et al. (2010) that serum haptoglobin concentrations in FMD-infected livestock did not show significant results. Insignificant haptoglobin concentrations were also reported by Eid et al. (2022) in Egyptian cattle and Lee et al. (2019). Haptoglobin concentrations can increase up to 50-100 times during inflammatory reactions (Brady et al., 2019). This report is consistent with the results of this study, where concentrations in infected cows increased to 50 mmol/L compared to the normal physiological value (Pegolo et al., 2023). Haptoglobin is a positive APP whose concentration increases during infection or inflammation. This is because Hp is produced by hepatocytes in response to proinflammatory cytokines such as IL-6, IL-1, and TNF. Haptoglobin acts as an anti-inflammatory agent by binding to free hemoglobin to form Hp-Hb complexes, which are then removed from the body by the reticuloendothelial system (Jain et al., 2011). Haptoglobin concentration in the recovery phase was found to be higher than in the infection phase. In addition, a strong positive correlation was found between Hp and GPx in the recovery phase (Table 3). This may occur due to the suspected persistent infection of the FMD virus, where previous studies reported a prevalence of cattle carrying the FMD virus of 50% regardless of vaccination status, and cattle can remain carriers for up to 2 years (Moonen and Schrijver, 2000). This is supported by the statement made by Singh et al. (2019) that FMD can become a persistent infection in animals that have entered the recovery phase, and it is mentioned that to detect persistent infection, samples can be collected after >28 days post-infection. Meanwhile, a different report by Nikvand et al. (2018) indicates a negative correlation between Hp and GPx, showing a negative relationship between inflammation (Hp) and antioxidant status (GPx). CrP belongs to the APP-positive group because its concentration increases during an inflammatory response. In a previous study by Stenfeldt et al. (2011), it was mentioned that APP is used as a marker of health status and the acute level of FMD infection in cattle. The results of CRP concentration testing in Madura cattle infected with FMD showed no significant difference (p > 0.05). This was supported by Upadhaya et al. (2020), who found that FMD infection in pigs caused insignificant changes in serum CrP concentration. Lee et al. (2019) also reported that animals infected with FMD experienced changes in serum CrP concentration, but not significantly. Meanwhile, in this study, CrP concentration was reported to be insignificant because cattle can tolerate stressful conditions. This is supported by another statement by Semra et al. (2016), who mentioned that insignificant changes in CRP concentration can be influenced by good animal care management. The results of this study showed that CrP concentrations in the infection phase were higher than normal references in healthy animals and decreased in the recovery phase compared to the infection phase. This occurs because CrP is a type of APP that rises during an inflammatory reaction and returns to normal concentrations when the inflammatory reaction no longer occurs (Yogeshpriya and Selvaraj, 2020). Cray (2012) also mentions that CrP concentrations decrease from the infection phase when animals enter the recovery phase. Another study on pigs infected with FMD obtained data showing that CRP increases during the infection phase and decreases again when entering the recovery phase (Lee et al., 2019). Monitoring oxidative stress and antioxidant enzyme profiles were important tool for reducing the impact of oxidative stress on animals (Hussain et al., 2022). One antioxidant enzyme profile that needs to be known is the SOD enzyme. The SOD enzyme was the first detoxification enzyme and the strongest antioxidant in cells. The results of SOD enzyme level tests in Madura cattle showed no significant difference between the infection phase and the recovery phase (p > 0.05). These results were consistent with the reports by Souza et al. (2011) on Holstein cattle and Ali et al. (2019)on local cattle in Egypt. The insignificant results were supported by Ali et al. (2019), who stated that viral infection can alter the oxidative status by increasing nitric oxide (NO) formation and inhibiting the synthesis of enzymes involved in oxidative defense within the body's cells. The inhibition of enzyme synthesis may be the reason why SOD enzyme levels did not increase significantly in the recovery phase. The results of GPx enzyme level tests in Madura cattle showed no significant difference between the infection phase and the recovery phase (p > 0.05). The results of this study showed that GPx enzyme levels during the infection phase were lower than normal levels. These results were consistent with the reports by Khoshvaghti et al. (2014) on local cattle in Iran, Soltani et al. (2020) on Holstein Friesian cattle in Egypt, and Yurdakul and Yildirim (2018) on cloven-hoofed animals, which showed that GPx enzyme levels in sick animals were lower than in healthy animals. During the FMD virus infection phase, the cow's body experienced an increase in ROS production, and antioxidant enzymes such as GPx reacted with ROS to protect the animal from severe tissue damage. Soltani et al. (2020) stated that lower antioxidant enzyme activity is caused by a reduction in the antioxidant defense system as a result of free radical production due to FMD virus infection. Excessive ROS production plays an important role in the pathogenesis of inflammatory diseases and contributes to FMD virus replication and the activation of pro-inflammatory cytokines during FMD virus infection. During the recovery phase, GPx enzyme levels tend to increase compared to the infection phase, but are not yet within the normal range in healthy cattle. These results were consistent with the report by Abou-Zeina et al. (2019) on cloven-hoofed animals, which showed a decrease in GPx enzyme levels during the infection phase and an increase during the recovery phase. ConclusionThis study showed that FMD infection in Madura cattle causes a clear physiological response in the form of increased APPs (haptoglobin and CrP), cardiac markers (CK, CK-MB, and cTnI), and decreased antioxidant enzyme activity (GPx, SOD, and MDA). Overall, the results of this study confirm that the combination of APP and antioxidant enzyme parameters can be used as effective biochemical indicators to evaluate the severity and health status of cattle infected with FMD, particularly in the Madura cattle population. AcknowledgmentWe want to express our gratitude to the staff of Agro Techno Park, Brawijaya University, for their support in conducting this research. FundingThis research received no specific grant. Authors' contributionsDK and WAS designed the research and analyzed the data; ARR, DP, and TW evaluated the manuscript and translation; and RA, OM, BSA, and NSR conducted the research and prepared the manuscript. Conflict of interestThe authors declare that there is no conflict of interest. Data availabilityAll data supporting the findings of this study are available within the manuscript. ReferencesAbou-Zeina, H.A.A., Nasr, S.M., Nassar, S.A., Farag, T.K., El-Bayoumy, M.K., Ata, E.B., Hassan, N.M.F. and Abdel-Aziem, S.H. 2019. Beneficial effects of antioxidants in improving health conditions of sheep infected with Foot- and-Mouth disease. Trop. Anim. Health. Prod. 51, 2379–2386. Adjid, A. 2020. Penyakit Mulut dan Kuku: penyakit hewan eksotik yang harus diwaspadai masuknya ke Indonesia. Wartazoa 30(2), 61–70. Aktas, M.S., Ozkanlar, Y., Oruc, E., Sozdutmaz, I. and Kirbas, A. 2015. Myocarditis associated with foot-and-mouth disease in suckling calves. Veterinarski. Arhiv. 85(3), 273–282. Ali, A.F., Selim, A., Manaa, E.A., Abdelrahman, A. and Sakr, A. 2019. Oxidative state markers and clinicopathological findings associated with Bovine Leukemia Virus infection in cattle. Microbial. Pathogenesis. 136, 103662. Alsaad, K.M., Al-Autaish, H.N. and Ahmed, J.A. 2020. Evaluation of cardiac enzymes and acute phase response as biomarkers for rapid diagnosis of myocarditis in calves with FMD. Iraqi. J. Vet. Sci. 34(1), 31–37. Aly, M., Nayel, M., Salama, A., Ghazy, E. and Elshahawy, I. 2020. Cardiac troponin I as a cardiac biomarker has prognostic and predictive value for poor survival in Egyptian buffalo calves with foot-and-mouth disease. Vet. World. 13, 890–895. Badiei, K., Pourjafar, M., and Ghane, M. 2010. Changes in acute phase proteins in naturally occurring FMD affected cattle in Shiraz Suburbs. Fars, Iran: World Buiatrics Congress. Bassols, A., Robles‐Guirado, J.A., Arroyo, L., Soler, L., García, N., Pato, R., Peña, R., Saco, Y., Armengol, R., Lampreave, F., Alava, M.A., Canalias, F. and Piñeiro, M. 2022. Validation of new automated turbidimetric immunoassays for the measurement of haptoglobin and inter-α-trypsin inhibitor heavy chain H4 specific for the bovine species. Vet. Clin. Pathol. 52(1), 64–74. Brady, N., O’Reilly, E.L., Mccomb, C., Macrae, A.I. and Eckersall, P.D. 2019. An immunoturbidimetric assay for bovine haptoglobin. Comparative. Clinic. Pathol. 28, 21–27. Brune, J., Chen, A. and Coffey, T. 2022. Determination of the effect of iatrogenic blood contamination on lactate dehydrogenase and creatine kinase activity in canine cerebrospinal fluid. Vet. Clin. Pathol. 52(1), 64–70. Castillo, C., Hernández, J., Valverde, I., Pereira, V., Sotillo, J., Alonso, M.L. and Benedito, J.L. 2006. Plasma malonaldehyde (MDA) and total antioxidant status (TAS) during lactation in dairy cows. Res. Vet. Sci. 80(2), 133–139. Cray, C. 2012. Acute phase proteins in animals. Prog. Mol. Biol. Translational. Sci. 105, 113–150. Deka, P., Das, S., Hazarika, R., Kayaga, R., Dutta, B., Deka, A., Barman, U., Ahmed, R., Islam, N., Sarma, M., Deka, I., Rout, M., Sharma, K. and Sharma, R.K. 2024. Foot-and-mouth disease-associated myocarditis is age dependent in suckling calves. Sci. Rep. 14, 10289. Eid, S.A., Alagmy, G.N., Hamed, T.A., Abdelwahed, D.A. and Salah, S. 2022. Immunological studies on cattle naturally infected with FMD compared with the vaccinated cattle in Sharkia Governorate Egypt. J. Adv. Vet. Res. 12(6), 694–705. Ghafoor, M., Kamal, M., Nadeem, U. and Husain, A. 2020. Educational case: myocardial infarction: histopathology and timing of changes. Academic Pathol. Sage J. 7(1), 1–6. Hashem, M., El-Mandrawy, S., El- Araby, I. and El- Sayed, A. 2018. Molecular diagnosis of Foot and Mouth Disease virus in cattle with reference to hematological and biochemical changes. Zagazig. Vet. J. 46(2), 105–116. Hussain, R., Khan, I., Jamal, A., Mohamed, B.B. and Khan, A. 2022. Evaluation of hematological, oxidative stress, and antioxidant profile in cattle infected with brucellosis in Southern Punjab, Pakistan. BioMed. Res. Int. 2022, 7140909. Jain, S., Gautam, V. and Naseem, S. 2011. Acute-phase proteins: as diagnostic tool. J. Pharm. Bioallied. Sci. 3(1), 118–127. Kaneko, J.J., Harvey, J.W. and Bruss, M.L. 2008. Clinical biochemistry of domestic animals. Khoshvaghti, A., Askari, A., Nazifi, S. and Ghane, M. 2014. Evaluation of some antioxidant enzymes in cattle infected with Foot and Mouth virus. İstanbul. Üniversitesi. Veteriner. Fakültesi. Dergisi. 40(1), 70–75. Klein, J. 2009. Understanding the molecular epidemiology of foot-and-mouth-disease virus. Infection, genetics and evolution. J. Infect. Genet. Evol. 9(2), 153–161. Lee, K.W., Lee, K.N., Lillehoj, H.S. and Park, J.H. 2019. Serum concentration of acute phase proteins and cytokines in vaccinated pigs challenged with Foot and Mouth Disease virus serotype o. Revista Brasileira De Zootecnia 48, 1–7. Lee, W.C., Hsiao, H.C., Wu, Y.L., Lin, J.H., Lee, Y.P., Fung, H.P. and Chu, R.M. 2003. Serum C-reactive protein in dairy herds. Can. J. Vet. Res. 67(2), 102–107. Li, Y., Ding, H.Y., Wang, X.C., Feng, S.B., Li, X.B., Wang, Z., Liu, G.W. and Li, X.W. 2016. An association between the level of oxidative stress and the concentrations of NEFA and BHBA in the plasma of ketotic dairy cows. J. Anim. Physiol. Anim. Nutr. 100(5), 844–851. Mahadappa, P., Mahendran, K., Winter, R.L., Umapathi, V., Krishnaswamy, N., Gopalakrishnan, A., Rao, S., Gangaiah, M., Kumar, S., Patel, B.H.M., Gautam, N., Hegde, R., Dechamma, H.J. and Sanyal, A. 2021. Characterization of arrhythmias evaluation of cardiac biomarkers and their association with survival in calves suffering from Foot and Mouth Disease. J. Vet. Cardiol. Elsevier. 36(1), 64–76. Mahmoud, M.A. and Galbat, S.A. 2017. Outbreak of Foot and Mouth Disease and peste des petits ruminants in sheep flock imported for immediate slaughter in Riyadh. Vet. World 10(2), 238–243. Miranda, A., Argentina, I., Harry, H., Francés, O., Laudecina, V., and Gelid, L., 2016. Evaluation of cardiac insult in steers with different dietary sodic monensin concentration measured thought miocardic kreatin kinaza. 18, 59–66. Moonen, P. and Schrijver, R. 2000. Carrier of Foot and Mouth Disease virus: a review. Vet. Quart. 22(4), 193–197. Mousa, S.A. and Galal, M.K.H. 2013. Alteration in clinical, hemobiochemical and oxidative stress parameters in Egyptian Cattle infected with Foot and Mouth Disease. J. Anim. Sci. Adv. 3, 485–491. Nasr El-deen, N.A.M., Neamat-Allah, A.N.F., Rizk, L.G. and Fareed, R.S.G. 2017. Serological, hematological, biochemical and oxidative markers during Foot and Mouth Disease serotype 'O' infection, Egypt. Bull. Univ. Agricult. Sci. Vet. Med. Cluj-Napoca. 74(2), 218. Nikvand, A.A., Jalali, S.M., Nouri, M., Ghadrdan Mashhadi, A. and Hassanpour Amirabadi, S. 2018. Electrocardiogram and serum cardiac biomarkers changes in FMD in cattle. Turkish J. Vet. Anim. Sci. 43(2), 285–291. Office International des Epizooties (OIE). 2015. Terrestrial animal health code volume ii: recommendations applicable to OIE listed disease and other diseases of importance to international trade. 24th ed. Paris: World Organization for Animal Health (OIE). 455–477. Pacheco, J.M., Smoliga, G., O’Donnell, V., Brito, B., Stenfeldt, C., Rodriguez, L.L. and Arzt, J. 2015. Persistent Foot-and-Mouth Disease virus infection in the nasopharynx of cattle; tissue-specific distribution and local cytokine expression. PLos One J. 10(5), 1–20. Pegolo, S., Giannuzzi, D., Piccioli-Cappelli, F., Cattaneo, L., Gianesella, M., Ruegg, P.L., Trevisi, E. and Cecchinato, A. 2023. Blood biochemical changes upon subclinical intramammary infection and inflammation in Holstein cattle. J. Dairy Sci. 106(9), 6539–6550. Saco, Y. and Bassols, A. 2022. Acute phase proteins in cattle and swine: a review. Vet. Clin. Pathol. , doi:10.1111/vcp.13220 Salim, S.A.S., Talb, A.O.Q., Saad, A.M.S. and Yousif, A.S. 2019. Clinico-pathological and biochemical aspects of Foot and Mouth Disease in calves. Adv. Anim. Vet. Sci. 7(10), 835–843. Santosa, B. 2020. Teknik ELISA: Metode Elisa Untuk Pengukuran Protein Metallothionein Pada Daun Padi Ir Bagendit. Semarang: Unimus Press. Semra, K., Kacar, C., Ogun, M., Kuru, M., Hasan, O., Demir, M.C. and Zonturlu, K. 2016. Evaluation of serum c-reactive protein and natural antibodies in cows with endometritis. Kafkas Universitesi. Veteriner. Fakultesi. Dergisi. 22(5), 709–715. Singh, R.K., Sharma, G.K., Mahajan, S., Dhama, K., Basagoudanavar, S.H., Hosamani, M., Sreenivasa, B.P., Chaicumpa, W., Gupta, V.K. and Sanyal, A. 2019. Foot-and-Mouth Disease virus: immunobiology, advances in vaccines and vaccination strategies addressing vaccine failures an indian perspective. Vaccines 7(3), 1–28. Sobhy, N.M., Bayoumi, Y.H., Mor, S.K., El-Zahar, H.I. and Goyal, S.M. 2018. Outbreaks of foot and mouth disease in Egypt: molecular epidemiology, evolution and cardiac biomarkers prognostic significance. Int. J. Vet. Sci. Med. 6(1), 22–30. Sobrino, F., SIz, M., JimNez-Clavero, M.A., NEz, J.I., Rosas, M.F., Baranowski, E. and Ley, V. 2011. Foot-and-mouth disease virus: a long-known virus, but a current threat. Vet. Res. 32(1), 1–30. Soltani, H., Aslani, M.R., Mohebbi, A. and Mokhtari, A. 2020. Serum biochemical and oxidative status in Holstein cattle affected with foot and mouth disease. Iranian J. Vet. Sci. Technol. 12(2), 19–24. Souza, F.N., Monteiro, A.M., Dos Santos, P.R., Sanchez, E.M.R., Blagitz, M.G., Latorre, A.O., Neto, A.M.F., Gidlund, M. and Libera, A.M.M.P.D. 2011. Antioxidant Status and Biomarkers of Oxidative Stress in Bovine Leukemia Virus-Infected Dairy Cows. Vet. Immunol. Immunopathology. 143(1-2), 162–166. Stenfeldt, C., Heegaard, P.M., Stockmarr, A., Tjornehoj, K. and Belshan, G.J. 2011. Analysis of the acute phase responses of serum amyloid Amylase, haptoglobin and type 1 interferon in cattle experimentally infected with Foot and Mouth Disease virus serotype O. Vet. Reserch. 42(1), 1–10. Sutawi, S., Wahyudi, A., Malik, A., Suyatno, S., Hidayati, A., Rahayu, I.D. and Hartatie, E.S. 2023. Re-emergence of Foot and Mouth Disease outbreak in Indonesia: a review. Adv. Anim. Vet. Sci. 11(2), 263–270. Thrall, M.A., Weiser, G., Allison, R.W. and Campbell, T.W. 2022. Veterinary hematology and clinical chemistry (3rd ed.). Hoboken, NY: Wiley Blackwell. Tufarelli, V., Colonna, M.A., Losacco, C. and Puvača, N. 2023. Biological health markers associated with oxidative stress in dairy cows during lactation period. Metabolites 13(3), 405. Tunca, R., Sozmen, M., Erdogan, H., Citil, M., Uzlu, E., Ozen, H. and Gokçe, E. 2008. Determination of cardiac troponin I in the blood and heart of calves with Foot-and-Mouth Disease. J. Vet. Diagnostic Invest. 20(5), 598–605. Upadhaya, S.D., Kim, Y.M., Shi, H., Le Cour Grandmaison, J., Blanchard, A. and Kim, I.H. 2020. Standardized plant extract alleviates the negative effects of FMD vaccination on animal performance. Animals 10(3), 1–11. Wang, D., Li, T., Cui, H. and Zhang, Y. 2016. Analysis of the indicating value of cardiac troponin I, tumor necrosis factor-α, interleukin-18, miR-1 and miR-146b for viral myocarditis among children. Cellular. Physiol. Biochem. 40(1), 1325–1333. Yogeshpriya, S., and Selvaraj, P. 2020. C-reactive protein: is early prognostic marker? Springer, Berlin. Yurdakul, I. and Yildirim, B.A. 2018. Assessment of oxidative status in foot diseases of sheep. Acta. Scientiae. Veterinariae. 46, 6. Zainuddin, N., Susila, E.B., Wibawa, H., Daulay, R.S.D., Wijayanti, P.E., Fitriani, D., Hidayati, D.N., dris, S., Wadsworth, J., Polo, N., Hicks, H.M., Mioulet, V., Knowles, N.J. and King, D.P. 2023. Genome Sequence of a Foot-and-Mouth Disease Virus Detected in Indonesia in 2022. Microbiol. Resour. Announc. 12(2), e0108122. | ||
| How to Cite this Article |
| Pubmed Style Kristanto D, Ramadhani NT, Ayu BS, Anna R, Kusumawardhani OM, Riadini AR, Widyaputri T, Prasetyo D, Khairunissa S, Septian WA. Evaluation of acute phase protein, cardiac marker, and antioxidant enzymes in Madura Cattle infected with foot-and-mouth disease. Open Vet. J.. 2026; 16(2): 824-833. doi:10.5455/OVJ.2026.v16.i2.6 Web Style Kristanto D, Ramadhani NT, Ayu BS, Anna R, Kusumawardhani OM, Riadini AR, Widyaputri T, Prasetyo D, Khairunissa S, Septian WA. Evaluation of acute phase protein, cardiac marker, and antioxidant enzymes in Madura Cattle infected with foot-and-mouth disease. https://www.openveterinaryjournal.com/?mno=290987 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.6 AMA (American Medical Association) Style Kristanto D, Ramadhani NT, Ayu BS, Anna R, Kusumawardhani OM, Riadini AR, Widyaputri T, Prasetyo D, Khairunissa S, Septian WA. Evaluation of acute phase protein, cardiac marker, and antioxidant enzymes in Madura Cattle infected with foot-and-mouth disease. Open Vet. J.. 2026; 16(2): 824-833. doi:10.5455/OVJ.2026.v16.i2.6 Vancouver/ICMJE Style Kristanto D, Ramadhani NT, Ayu BS, Anna R, Kusumawardhani OM, Riadini AR, Widyaputri T, Prasetyo D, Khairunissa S, Septian WA. Evaluation of acute phase protein, cardiac marker, and antioxidant enzymes in Madura Cattle infected with foot-and-mouth disease. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 824-833. doi:10.5455/OVJ.2026.v16.i2.6 Harvard Style Kristanto, D., Ramadhani, . N. T., Ayu, . B. S., Anna, . R., Kusumawardhani, . O. M., Riadini, . A. R., Widyaputri, . T., Prasetyo, . D., Khairunissa, . S. & Septian, . W. A. (2026) Evaluation of acute phase protein, cardiac marker, and antioxidant enzymes in Madura Cattle infected with foot-and-mouth disease. Open Vet. J., 16 (2), 824-833. doi:10.5455/OVJ.2026.v16.i2.6 Turabian Style Kristanto, Dwi, Nabila Tsabitsa Ramadhani, Brigitta Sekar Ayu, Rif Anna, Ovia Mustika Kusumawardhani, Aprilia Rizky Riadini, Tiara Widyaputri, Dodik Prasetyo, Siti Khairunissa, and Wike Andre Septian. 2026. Evaluation of acute phase protein, cardiac marker, and antioxidant enzymes in Madura Cattle infected with foot-and-mouth disease. Open Veterinary Journal, 16 (2), 824-833. doi:10.5455/OVJ.2026.v16.i2.6 Chicago Style Kristanto, Dwi, Nabila Tsabitsa Ramadhani, Brigitta Sekar Ayu, Rif Anna, Ovia Mustika Kusumawardhani, Aprilia Rizky Riadini, Tiara Widyaputri, Dodik Prasetyo, Siti Khairunissa, and Wike Andre Septian. "Evaluation of acute phase protein, cardiac marker, and antioxidant enzymes in Madura Cattle infected with foot-and-mouth disease." Open Veterinary Journal 16 (2026), 824-833. doi:10.5455/OVJ.2026.v16.i2.6 MLA (The Modern Language Association) Style Kristanto, Dwi, Nabila Tsabitsa Ramadhani, Brigitta Sekar Ayu, Rif Anna, Ovia Mustika Kusumawardhani, Aprilia Rizky Riadini, Tiara Widyaputri, Dodik Prasetyo, Siti Khairunissa, and Wike Andre Septian. "Evaluation of acute phase protein, cardiac marker, and antioxidant enzymes in Madura Cattle infected with foot-and-mouth disease." Open Veterinary Journal 16.2 (2026), 824-833. Print. doi:10.5455/OVJ.2026.v16.i2.6 APA (American Psychological Association) Style Kristanto, D., Ramadhani, . N. T., Ayu, . B. S., Anna, . R., Kusumawardhani, . O. M., Riadini, . A. R., Widyaputri, . T., Prasetyo, . D., Khairunissa, . S. & Septian, . W. A. (2026) Evaluation of acute phase protein, cardiac marker, and antioxidant enzymes in Madura Cattle infected with foot-and-mouth disease. Open Veterinary Journal, 16 (2), 824-833. doi:10.5455/OVJ.2026.v16.i2.6 |