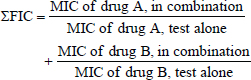Open Veterinary Journal, (2026), Vol. 16(1): 157-164
Research Article
10.5455/OVJ.2026.v16.i1.15
Gallic acid improves the bactericidal efficacy of tylosin against Staphylococcus aureus isolated from broilers in Babylon Province, Iraq
Zahraa M. Ayad1*, Ali I. Al-ameedi1, Walaa Farhan Obaid2, and Bareq A. Al-lateef3
1Department of Physiology and Biochemistry and Pharmacology, College of Veterinary Medicine, Al-Qasim Green University, Babylon, Iraq
2Department of Microbiology, College of Veterinary Medicine, Al-Qasim Green University, Babylon, Iraq
*Corresponding Author: Zahraa Mohammed Ayad. Department of Physiology and Biochemistry and Pharmacology, College of Veterinary Medicine, Al-Qasim Green University, Babylon, Iraq. Email: zahraamohammed [at] vet.uoqasim.edu.iq
Submitted: 13/10/2025 Revised: 02/12/2025 Accepted: 17/12/2025 Published: 31/01/2026
© 2025 Open Veterinary Journal
This is an Open Access article distributed under the terms of the Creative Commons Attribution-Non Commercial-No Derivatives License (http://creativecommons.org/licenses/by-nc-nd/4.0/), which permits non-commercial re-use, distribution, and reproduction in any medium, provided the original work is properly cited, and is not altered, transformed, or built upon in any way.
Abstract
Background: The use and abuse of antimicrobials in poultry products, dairy products, and beef cattle is a growing global concern. Furthermore, with the rise of bacterial resistance to these drugs, an innovative approach is needed to reduce antimicrobial doses and eradicate resistance.
Aim: This study aimed to investigate the antibacterial properties of gallic acid and its potential to enhance the efficacy of tylosin.
Methods: This study morphologically, biochemically, and molecularly identified Staphylococcus aureus. The molecular diagnostic approach relied upon the polymerase chain reaction (PCR) test to identify the Staphylococcus genus by determining the main specific gene, 16s rRNA. Of the 30 isolates, 20 were positively identified by biochemical and traditional methods. Furthermore, to ensure that all isolates were positively identified as S. aureus species, 10 random samples were identified by PCR. The tube dilution method will then be used to determine the antibacterial minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) of gallic acid and tylosin. The second step assessed gallic acid and tylosin antibacterial synergy using checkerboard and Chou–Talalay techniques.
Results: The morphologically, biochemically, and molecularly PCR-identified S. aureus isolates were approved for investigation. The MIC of gallic acid was determined to be 4 μg/ml, with a MBC of 8 μg/ml. The range of MIC and MBC for tylosin was found to be between 8 and 16 μg/ml. In addition, synergistic interactions were reported when gallic acid was coupled with tylosin, resulting in a record , fractional inhibitory concentration value of 0.357 against the tested isolate.
Conclusion: Gallic acid synergises the antibacterial action of tyrosine against S. aureus in a sub-MIC concentration.
Keywords: Gallic acid, Tylosin, Antibiotic resistance, Synergistic, S. aureus.
Introduction
At present, the growing resistance to antibiotics and the failure of treatments result in rising economic and therapeutic expenses. In today’s world, the veterinary and human health care industries are extremely concerned about antimicrobial resistance (AMR) (Murray et al., 2022). Numerous possible consequences for animals, people, and our surroundings emerge from the complicated concerns surrounding the development and dissemination of antibiotic resistance because of antibiotic usage in livestock. The use of antibiotics in livestock necessitates a worldwide strategy to ensure that measures implemented to mitigate the onset and dissemination of AMR are sustainable (Vidovic and Vidovic, 2020).
Antibiotics and their alternatives influence the dynamics of AMR through direct and indirect impacts on microbial populations (Brown et al., 2017). Due to their association with bacterial resistance, antibiotics used for growth promotion in the past, including avilamycin and bambermycin, have co-selected with antibiotics that are vital for human therapy (Kerek et al., 2025). Evidently, the manner in which antimicrobials are employed in chicken farming profoundly affects public health concerns, particularly related to the spread of antibiotic-resistant diseases and animal welfare.
Understanding how resistance develops requires examining chicken commensal bacterial communities. The environmental prevalence of Staphylococcus species is extremely high, and chicken flocks contain both coagulase-positive and coagulase-negative isolates of this bacterium (Becker et al., 2015). Joint inflammation (arthritis), respiratory sickness (pneumonia), and infections of the yolk sac (omphalitis) are some of the poultry illnesses attributed to Staphylococcus aureus (Al-Ameedi and Nahi, 2019; Park and Ronholm, 2021). Several studies have shown that multidrug-resistant strains of Methicillin resistant staph. aureus (MRSA) may spread from humans to poultry (Leonard and Markey, 2008; Kerek et al., 2025).
Inventive strategies are required to impede the advancement of bacteria that are resistance to antibiotics and other microbes. Researchers are also working to extend the efficacy of traditional antibiotics (Minarini et al., 2020). Researchers have recently begun considering the use of naturally occurring substances to combat microorganisms’ resistance (Álvarez-Martínez et al., 2020; Keyvani-Ghamsari et al., 2020). Phenolic compounds (PCs) are one of the secondary metabolites found in plants. Their unique characteristics are the presence of one or more aromatic rings that contain hydroxyl groups. Currently, there is knowledge of over 8,000 phenolic substances, including tannins, flavonoids, ligands, and coumarins. A significant number of PCs possess antimicrobial properties (Muniyandi et al., 2019; Khorsandi et al., 2020).
Gallic acid is one of the most significant natural polyphenols. Carl Wilhelm Scheele first extracted gallic acid from plants in 1786. Subsequently, research began to look into the role and properties of this polyphenol and the accompanying compounds (Yang et al., 2020). It is a naturally occurring compound produced by plants that has a wide range of biological effects, including reducing inflammation, antimicrobial action, and preventing ulcers, in addition to inhibiting cancer cell growth (Wianowska and Olszowy-Tomczyk, 2023). In recent years, researchers have confirmed the positive in vivo safety profile of gallic acid (Elmileegy et al., 2023; Sabbagh et al., 2024; Dnan et al., 2025). The polyphenol gallic acid and its associated compounds, either alone or in conjunction with different medications, can inhibit the proliferation of numerous bacterial biofilm and plankton forms by several mechanisms (Subramanian et al., 2016). Combining antimicrobial agents with phytochemicals has shown promise in eliminating highly resistant bacteria (Gould, 2008; Rajamanickam et al., 2019).
Combining phytochemicals with antimicrobials has been proven to increase their effectiveness and slow down the emergence of AMR. Tylosin, often known as Ty, is a macrolide antibiotic that works by blocking the protein synthesis pathway in Gram-positive bacteria and Mycoplasma species. Veterinary primarily employ it for the treatment of respiratory diseases in livestock (Dinos, 2017). Consequently, this study aims to assess whether gallic acid possesses antibacterial properties, enhances the antibacterial effectiveness of tylosin, and decreases antimicrobial resistance to tylosin against S. aureus pathogen.
Materials and Methods
Chemicals
The gallic acid and NaOH were obtained from Sigma–Aldrich Canada. Tylosin purchased from Vapco, Jordan. Himedia, based in India, supplied all the cultural media used in the identification and characterization of bacteria.
Bacterial identification
Identification of S. aureus by morphological and cultural characteristics
Thirty specimens were isolated from the nasal swabs of symptomatic broilers in two farms in the Al-Nill area of Babylon province at age 22–30 days. Morphological characteristics and biochemical properties examination performed for strain identification. These tests were performed according to the methods of Benson (2002), Harley and Prescott (2002), Kumar (2007), and Nagoba (2019). Bacterial isolate activation and maintenance: Slants containing 10 ml of brain infusion agar were used to grow bacterial cultures in glass tubes with screw caps for 24 hours at 37°C. Sub culturing nutrition and brain heart agar once every 2 weeks and storing at 4°C is performed for maintenance of isolate viability (Quinn et al., 2004).
Molecular characterization
DNA extraction
In accordance with the instructions provided by the Wizard® Genomic DNA Purification Kit ABIO pure, USA, 10 bacterial growth samples were used to isolate genomic DNA (Ahmed and Al-Daraghi, 2022). A Quantus Fluorometer was employed to measure the concentration and purity of the extracted DNA to ensure that the samples were suitable for further usage. The mixture contained 199 μl of diluted Quanty Flour Dye for every 1 μl of DNA. The results for DNA concentration were obtained after 5 minutes of incubation at room temperature.
16S rRNA polymerase chain reaction (PCR) sequencing
The primers created to target the 16S rRNA and medA genes in the DNA of the isolates are shown in Table 1 (Alves et al., 2018; Shamkhi, 2019). The reaction mixture, which had a total volume of 20 l, consisted of 13 l of DNA, 1 l of each primer, 10 l of master mix, and 5 l of nuclease-free water. As indicated in Table 2, the experiment was conducted according to the following program: a 5-minutes denaturation at 95°C, 30 cycles at 55°C and a final 7-minutes extension at 72°C. The PCR results were examined by electrophoresing on a 1.5% agarose gel and then seeing the bands stained with ethidium bromide using gel-imaging equipment (Liuyi Gel Document Imaging System, China).
Table 1. PCR primer sequence.

Table 2. Bacterial morphological investigations used to identify S. aureus.

Gallic acid and tylosin solution
A stock solution of each antibacterial medicine was generated by combining 0.1 mg of gallic acid and tylosin with 10 µl of DW (for gallic acid, warmed DW at 50°C with a tiny amount of NaOH to make a slightly alkaline condition, which enhanced the solubility), resulting in a concentration of 10 mg/ml (Pant et al., 2019). Several different concentrations were subsequently produced by mixing a known volume of the stock solution with distilled water.
Estimation of the antibacterial activity (MIC, MBC) for tylosin and gallic acid
The broth dilution method is used to estimate the MIC and then the MBC (NCCLS, 2000; Asghari et al., 2006). Approximately 1.5 × 105 (CFU/ml) of standard bacterial suspension was added to the previously prepared 9 ml of nutrient broth tubes. Various concentrations of gallic acid (64, 32, 16, 8, 4, 2, 1, and 0.5 μg/ml) and tylosin (64, 32, 16, 8, 4, 2, 1, and 0.5 μg/ml) were prepared. One tube of antibiotic and one tube of nutritious broth were used as the negative control, and the other tube was designated as the positive control (broth and bacteria). Following a 24-hours incubation period at 37°C, the minimum inhibitory concentration (MIC) of gallic acid was defined as the concentration at which bacterial growth was inhibited. By spreading 10 µl of gallic acid-treated bacterial cultures over the surfaces of Muller–Hinton agar plates at a concentration equal to or greater than the MIC, the minimum bactericidal concentration (MBC) was obtained. After a 24-hour incubation period, colony-forming unit (CFU)/ml counts were taken, and the MBC, which is the concentration at which 99.9% of bacteria are killed, was recorded. Additionally, the same technique used for gallic acid (MIC) and MBC was used for tylosin determination.
Combination of gallic acid and tylosin to combat
S. aureus
The efficacy of combining gallic acid with tylosin to combat S. aureus was evaluated using the checkerboard method. The combination response was assessed by calculating the fractional inhibitory index (Σ FIC) as follows:
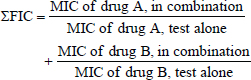
When the FIC value is less than 0.5, the interaction is considered synergistic. When the FIC index exceeds 0.5 but is less than four, it is considered neutral. Finally, it is considered antagonistic when the FIC index exceeds four (Hall et al., 1983). Subsequently, the data resulting from the checkerboard were analyzed using the software isobolographic program for analysis of combinations of gallic acid and tylosin of the experiment and represented as synergism if the combination result < 1, additives if=1, and antagonism if > 1, and this method is called the Chou–Talalay method (Chou, 2010).
Ethical approval
The Research Ethics Committee of Al-Qasim Green University/Biotechnology College building, Al-Qasim, Babylon, Iraq, 51013, approved the microbiological experiment with the number QGEC/10/2024.
Results
This study was conducted on S. aureus isolate, producing yellowish colonies on mannitol salt agar (MSA) and hemolysis on blood agar Tables 2 and 3.
Table 3. Biochemical examinations for the identification of S. aureus.

Identification of S. aureus 16S rRNA gene
Figure 1 demonstrates that all 10 S. aureus strains have a 133-bp 16S rRNA gene. Figure 1 was recognized by prior methods, confirming the reliability of this study’s test and genus identification procedure.
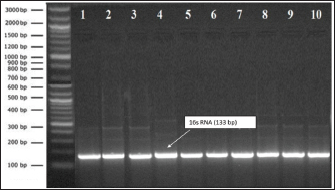
Fig. 1. S. aureus isolate 16S rRNA PCR product gel electrophoresis. Lane (M): l00-bp ladder, Lane (c): negative control, Lane (4): positive result with 133-bp S. aureus bands.
Determination of the MIC and MBC of gallic acid and tylosin against S. aureus
The tube dilution technique determined that the MIC of gallic acid was 4 μg/ml, with a MBC of 2 μg/ml. The Tylosin antibiotic demonstrated efficacy against the tested isolate, with a MIC90 (minimum inhibitory concentration appropriate to stop the growth of 90% of the isolate) value of 8 μg/ml and an MBC90 (MBC required to kill 90% of the isolate) value of 16 μg/ml; Figures 2 and 3.

Fig. 2. The minimum inhibitory concentration of gallic acid.

Fig. 3. Tylosin minimum inhibitory concentration.
Combination of gallic acid and tylosin to combat S. aureus
The checkerboard technique was used to evaluate the efficacy of the gallic acid and tylosin combination against the selected S. aureus isolate. The combination of gallic acid and tylosin resulted in synergistic activity against the S. aureus isolate (Fig. 4). The results of the Chou–Talalay analysis are plotted in Figure 5, which reveals the synergistic effect of the combinations of gallic acid and tylosin.
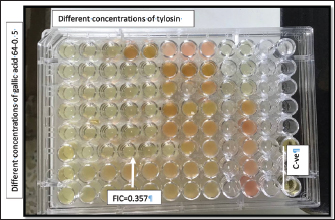
Fig. 4. Checkerboard assay showing the concentrations of gallic acid and tylosin.
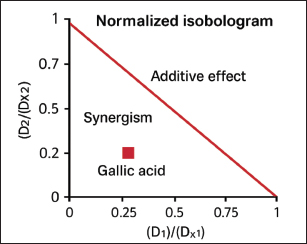
Fig. 5. The Chou-Chou plot for the non-constant ratio combination design, which normalizes the isobologram.
Discussion
The current investigation included the identification of the S. aureus isolate, as accurately identifying the targeted pathogen is essential for microbiological diagnosis. Microbiologists have used multiple culture media (selective and differential) to identify S. aureus. MSA is a frequently used selective and differential medium. MSA can detect the metabolic activity of S. aureus, namely, its ability to ferment mannitol.
Additional secondary bacteriological tests are necessary to identify the S. aureus isolate accurately. Coagulase determination and β-hemolysis activities are reliable methods for identifying S. aureus. Employing both of these confirmatory tests in conjunction with a selective medium significantly enhances the S. aureus identification accuracy. Culturing S. aureus on blood agar for β-hemolysis identification, along with a secondary coagulase test, improved the identity of selected isolates. Furthermore, MSA culture exhibited significant specificity when combined with other secondary identification tests. Our findings complemented the conclusions of Bautista-Trujillo et al. (2013) and Omwenga et al. (2021).
Conducting a thorough examination to differentiate between organisms that cause disease and those that do not is crucial. The staphylococci subjected to a coagulase test using the slide technique. The findings indicated agglutination particles after 90 seconds, indicating a positive result. This test is used to estimate the presence of cell wall-associated coagulase, also known as clumping factor (Cheng et al., 2010).
The oxidase test results were completely negative for the tested isolate, and this negative outcome for S. aureus is consistent with the findings of Subhankari et al. (2011). On the other hand, the urease test recorded a positive result due to the urease enzyme hydrolyzing urea to carbon dioxide and ammonia, and giving a positive result due to pH increases and the phenol red indicator changing from yellow–orange to pink color. This result agrees with that of Brink (2010).
The powerful technique utilized in the identification and detection of bacterial strains by targeting conserved regions of the 16S rRNA when amplifying DNAs from phylogenetically divergent bacteria (Greisen et al., 1994; Malik et al., 2022). From the findings of the present study, the PCR technique using 16S rRNA is considered an excellent method for the detection of S. aureus isolates. Therefore, the current results corroborate those of previous research that demonstrated that this gene might be effectively used to identify clinical isolates of S. aureus by detection and sequencing (Shah et al., 2007; Ahmed and Al-Daraghi, 2022). These are the findings of Al-Alak and Qassim (2016). This method demonstrated the capacity to molecularly identify Staphylococcus clinical isolates using 16S rRNA and 23S rRNA genes; this method was quick, precise, and had a high genomic identification rate of 100%. Our findings corroborate those of previous research showing that this gene may be effectively used to identify clinical isolates of S. aureus by sequencing and detection in poultry (Lu et al., 2003; Jafarzadeh et al., 2023).
Bacterial antibiotic resistance is a persistent and important global concern. The improper and excessive use of antibacterial treatment of diseases and animal nutrition has led to the development and prevalent distribution of antibiotic-resistant bacteria. This has led to the development of cross-resistance and Multi drug resistance in bacterial pathogens (Rather et al., 2021; Upadhayay et al., 2023). This has hindered the treatment of bacterial diseases, particularly those affecting cattle and poultry. In addition, using untreated animal and poultry manure as fertilizer might lead to the spread of drug-resistant bacteria and their genes to the soil, which is already a major problem (Larsson and Flach, 2022).
The MIC and MBC of tylosin toward the S. aureus isolate were 8 and 16 μg/ml, respectively. Similarly, the MIC and MBC of gallic acid were four and eight μg/ml, respectively. Tylosin is commonly used as a prophylactic measure against various microorganisms (Mycoplasma, S. aureus, Pseudomonas aeruginosa, and S. suis) that cause respiratory infections (Qiu et al., 2018). Our study agrees with Entorf et al. (2014), who reported different MICs of tylosin against S. aureus. While disagreeing with Bahraminia et al. (2017), who reported a 64 μg/ml MIC of tylosin against S. aureus (Bahraminia et al., 2017). However, the MIC result of this investigation is incompatible with that of Liu et al. (2017).
With the extensive prevalence of bacterial resistance to current antibiotics, combination treatment is a very successful approach to promoting the therapeutic effect of existing antibiotics. Furthermore, the use of two or more combination antibiotics leads to a decrease in the proliferation of drug resistance by virtue of the diminished administration of antibiotic dosages (Buchmann et al., 2022). In the present study, the combination of gallic acid and tylosin revealed obvious synergistic antibacterial activity against S. aureus using the checkerboard method. There are several mechanisms of antibacterial action manifested by gallic acid to disrupt the bacterial cell membrane, increase permeability, and release internal components (Aldulaimi, 2017). Particularly, gallic acid can block efflux pumps, which inhibit S. aureus’s TetK and MepA transporters and decrease the bacteria’s ability to evacuate antimicrobial chemicals, leading to an increase in intracellular drug accumulation (Macêdo et al., 2022). In addition, gallic acid inhibits biofilm development by lowering the levels of two important regulators of polysaccharide intercellular adhesion, SarA and IcaAD (Sang et al., 2024). Nevertheless, the macrolide antibiotic tylosin binds to the 50S subunit of the ribosome in bacteria (more precisely, to 23S rRNA) and prevents peptide translocation, which in turn stops protein synthesis (Lenz et al., 2021). In combination, gallic acid and tylosin probably work better because gallic acid increases tylosin uptake via membrane disruption and reduces efflux via pump inhibition. Both membrane damage and efflux inhibition are stressful enough without adding tylosin’s protein synthesis blockage. When administered together, the two substances have a multiplicative effect that makes them more effective at lower concentrations compared to when they are administered separately. Another study observed a synergistic impact of gallic acid when used with norfloxacin and gentamicin against S. aureus (Lima et al., 2016). Another study demonstrated that the combination of macrolide azithromycin and gallic acid has a significant effect against methicillin-resistant S. aureus and can be considered an effective treatment option (Khoshi et al., 2025). Research has demonstrated that gallic acid, a type of phenolic acid, can impede the growth of different bacterial strains. Additionally, it enhances the effectiveness of antimicrobial drugs (Fu et al., 2016). Prior research has also shown that gallic acid effectively suppresses efflux pump activity, which is an essential factor contributing to the development of antibiotic-resistant S. aureus and MDR E. coli (Abreu et al., 2012).
Conclusion
From the outcomes of this study, we can conclude that gallic acid potentiates the antibacterial effect of tylosin in a sub-MIC concentration against S. aureus bacteria. Therefore, we recommend further in vivo studies to evaluate the efficiency of the gallic acid and tylosin combination. Furthermore, the addition of phyto-constituents, such as phenols, flavonoids, and alkaloids, in combination with antibiotics broadens the antibiotic spectrum and reduces bacterial resistance.
Acknowledgment
The authors express their immense gratitude to the Physiology and Pharmacology Department/College of Veterinary Medicine for their support and facilities provided for this research.
Conflict of interest
The authors have no conflicts of interest to declare regarding this paper.
Funding
None.
Authors’ contributions
Zahraa Mohammed Ayad: Conceptualization and research design. Ali Ibrahim Al-ameedi: Sample collection, bacteriology laboratory analysis, data interpretation, and manuscript drafting. Bareq A. Al-lateef: PCR laboratory analysis Walla Farhan obaid: is involved in manuscript revision, editing, and approving the final version of the manuscript for publication.
Data availability
All data are available in the revised manuscript.
References
Abreu, A.C., McBain, A.J. and Simões, M. 2012. Sources of new antimicrobials and resistance-modifying agents. Natural. Product. Rep. 29(9), 1007–1021.
Ahmed, Z.F. and Al-Daraghi. 2022. Molecular detection of the medA virulence gene in S. aureus isolated from Iraqi patients. Iraqi J. Biotechnol. 21(1).
AL–Alak, S.K. and Qassim, K. 2016. Molecular identification of the 16S rRNA gene in Staphylococcus aureus isolated from wounds and burns by polymerase chain reaction and study of fusidic acid resistance. Iraqi J. Cancer Med. Genet. 9, 25–30.
Al-Ameedi, A.I. and Nahi, H. 2019. Therapeutic effects of hydro-alcoholic extract of clove buds and E. erectus on healing of infected wounds in dogs. Adv. Anim. Vet. Sci. 7(5), 389–396.
Aldulaimi, O.A. 2017. General overview of phenolics from plant to laboratory, whether they are good or not. Pharmacognosy Rev. 11, 123.
Álvarez-Martínez, F.J., Barrajón-Catalán, E. and Micol, V. 2020. Tackling antibiotic resistance with compounds of natural origin: a comprehensive review. Biomedicines 8(10), 405; doi:10.1016/j.biomedicines.2010.04.015
Alves, D.F., Magalhães, A.P., Neubauer, D., Bauer, M., Kamysz, W. and Pereira, M.O. 2018. Unveiling the fate of adhering bacteria to antimicrobial surfaces: resistance-associated gene expression and macrophage-mediated phagocytosis. Acta Biomaterialia 78, 189–197.
Asghari, G., Nourallahi, H., Havaie, S.A. and Issa, L. 2007. Antimicrobial activity of Otostegia persica Boiss. extracts. Res. Pharm. Sci. 1(1), 53–58.
Bahraminia, F., Emadi, S.R., Emaneini, M., Farzaneh, N., Rad, M. and Khoramian, B. 2017. A high prevalence of tylosin resistance among S. aureus strains isolated from bovine mastitis. Vet. Res. Forum. 8(2), 121.
Bautista-Trujillo, G.U., Solorio-Rivera, J.L., Renteria-Solorzano, I., Carranza-German, S.I., Bustos-Martinez, J.A., Arteaga-Garibay, R.I., Baizabal-Aguirre, V.M., Cajero-Juarez, M., Bravo-Patino, A. and Valdez-Alarcon, J.J. 2013. Performance of culture media for the isolation and identification of S. aureus from bovine mastitis. J. Med. Microbiol. 62(3), 369–376.
Becker, K., Skov, R.L. and von Eiff, C. (2015) ‘Staphylococcus, Micrococcus, and other catalase-positive cocci’, in Jorgensen, J.H., Pfaller, M.A., Carroll, K.C., Funke, G., Landry, M.L., Richter, S.S. and Warnock, D.W. (eds.) Manual of Clinical Microbiology. 11th edn. Hoboken, NJ: John Wiley & Sons, pp. 354–382.
Benson. 2002. Microbiological applications: a general laboratory manual. McGraw-Hill.
Brink, B. 2010. Urease test protocol. Washington, DC: American Society for Microbiology (ASM), pp. 1–7
Brown, K., Uwiera, R.R., Kalmokoff, M.L., Brooks, S.P. and Inglis, G.D. 2017. Use of antimicrobial growth promoters in livestock: a requirement to understand their modes of action to develop effective alternatives. Int. J. Antimicrob. Agents. 49(1), 12–24.
Buchmann, D., Schultze, N., Borchardt, J., Böttcher, I., Schaufler, K. and Guenther, S. 2022. Synergistic antimicrobial activities of epigallocatechin gallate, myricetin, daidzein, gallic acid, epicatechin, 3-hydroxy-6-methoxyflavone and genistein against ESKAPE pathogens. J. Appl. Microbiol. 132(2), 949–963.
Cheng, A.G., McAdow, M., Kim, H.K., Bae, T., Missiakas, D.M. and Schneewind, O. 2010. Contribution of coagulases toward Staphylococcus aureus disease and protective immunity. PLos Pathogens 6(8), e1001036.
Chou, T.C. 2010. Drug combination studies and synergy quantification using the Chou-Talalay method. Cancer Res. 70(2), 440–446.
Dinos, G.P. 2017. The renaissance of macrolide antibiotics. Br. J. Pharmacol. 174(18), 2967–2983.
Dnan, G.D., Al-Awadi, H.K. and Al-AMeedi, A.I., 2025. Gallic acid and olive oil ameliorate atherosclerosis in experimentally induced in male rats. J. Anim. Health Prod, 13(s1), 422–427.
Elmileegy, I.M.H., Waly, H.S.A., Alghriany, A.A.I., Abou Khalil, N.S., Mahmoud, S.M.M. and Negm, E.A. 2023. Gallic acid rescues uranyl acetate induced-hepatic dysfunction in rats by its antioxidant and cytoprotective potentials. BMC Complement. Med. Ther. 23(1), 423.
Entorf, M., Feßler, A.T., Kadlec, K., Kaspar, H., Mankertz, J., Peters, T. and Schwarz, S. 2014. Tylosin susceptibility of bovine mastitis staphylococci. Vet. Microbiol. 171(3-4), 368–373.
Fu, L., Lu, W. and Zhou, X. 2016. Phenolic compounds and in vitro antibacterial and antioxidant activities of three tropic fruits: persimmon, guava, and sweetsop. BioMed Res. Int. 58, 6.
Gould. 2008. Antibiotic policies to control HAI. J. Antimicrob. Chemother. 61(4), 763–765.
Greisen, K., Loeffelholz, M., Purohit, A. and Leong, D. 1994. PCR primers and probes for the 16S rRNA gene of most pathogenic bacteria species, including bacteria found in CSF. J. Clin. Microbiol. 32(2), 335–351.
Hall, M.J., Middleton, R.F. and Westmacott, D. 1983. The FIC index as a measure of synergy. J. Antimicrob. Chemother. 11(5), 427–433.
Harley. 2008. Laboratory exercises in microbiology The McGraw−Hill Companies. 5th ed., New York.
Harley, J.P. and L.M. Prescott., 2002. Laboratory Exercises in microbiology. The McGraw−Hill Companies. 5th ed. New York, USA.
Jafarzadeh, M., Meamar, N., Mohsenzadeh, M. and Razmyar, J. 2023. Identification of methicillin-resistant Staphylococcus aureus in poultry meat portions using multiplex polymerase chain reaction. J. Poultry Sci. Avian Dis. 1(4), 13–19.
Kerek., Szab., Barnácz, F., Csirmaz, B., Kovács, L. and Jerzsele. 2025. Antimicrobial resistance in commensal bacteria from large-scale chicken flocks in Hungary’s Dél-Alföld Region. Vet. Sci. 12(8), 691; doi:10.3390/vetsci12080691
Keyvani-Ghamsari, S., Khorsandi, K. and Gul, A. 2020. Curcumin effect on multidrug resistance in cancer cells: an update. Phytotherapy Res. 34(10), 2534–2556.
Khorsandi, K., Kianmehr, Z., Hosseinmardi, Z. and Hosseinzadeh, R. 2020. Anticancer effect of gallic acid in the presence of low-level laser irradiation: ROS production and apoptosis and ferroptosis induction. Cancer. Cell. Int. 20, 1–14.
Khoshi, M.A., Keyvani-Ghamsari, S. and Khorsandi, K. 2025. Gallic acid synergistically enhances the antibacterial activity of azithromycin against MRSA. Int. Microbiol. 28(4), 829–836.
Kumar, D.R. 2007. Diagnostic microbiology for students with DMLT J.P. New Delhi: Medical Publishers (P). Ltd.
Larsson, D.G.J. and Flach, C.F. 2022. Antibiotic resistance in the environment. Nature Rev. Microbiol. 20(5), 257–269.
Lenz, K.D., Klosterman, K.E., Mukundan, H. and Kubicek-Sutherland, J.Z. 2021. Macrolides: from toxins to therapeutics. Toxins 13(5), 347; doi:10.1016/j.toxins.2021.01.07
Leonard, F.C. and Markey, B.K. 2008. Meticillin-resistant Staphylococcus aureus in animals: a review. Vet. J. 175, 27–36.
Lima, V.N., Oliveira-Tintino, C.D., Santos, E.S., Morais, L.P., Tintino, S.R., Freitas, T.S., Geraldo, Y.S., Pereira, R.L., Cruz, R.P., Menezes, I.R. and Coutinho, H.D. 2016. Antimicrobial activity and enhancement of antibiotic activity by phenolic compounds: gallic acid, caffeic acid, and pyrogallol. Microbial. Pathogenesis. 99, 56–61.
Liu, M., Wu, X., Li, J., Liu, L., Zhang, R., Shao, D. and Du, X. 2017. The specific anti-biofilm effect of gallic acid on S. aureus by regulating the expression of the ica operon. Food. Control. 73, 613–618.
Lu, J., Sanchez, S., Hofacre, C., Maurer, J.J., Harmon, B.G. and Lee, M.D. 2003. Evaluation of broiler litter with reference to microbial composition as assessed using 16S rRNA and functional gene markers. Appl. Environ. Microbiol. 69(2), 901–908.
Macêdo, N.S., Barbosa, C.R., Bezerra, A.H., Silveira, Z.D., Da Silva, L., Coutinho, H.D., Dashti, S., Kim, B., Da Cunha, F.A. and Da Silva, M.V. 2022. Evaluation of ellagic acid and gallic acid as efflux pump inhibitors in S. aureus strains. Biol. Open. 11(10), 59434.
Malik, Z.J., Alameedi, A.I. and Abdulah, M. 2022. Effects of common carp, Cyprinus carpio skin collagen extract on infected wounds. Iranian J. Ichthyology 9, 104–109.
Minarini, L.A.D.R., Andrade, L.N.D., De Gregorio, E., Grosso, F., Naas, T., Zarrilli, R. and Camargo, I.L. 2020. Antimicrobial resistance as a global public health problem: how can it be addressed?. Front. Public. Health. 8, 612844.
Muniyandi, K., George, E., Sathyanarayanan, S., George, B.P., Abrahamse, H., Thamburaj, S. and Thangaraj, P. 2019. Phenolics, tannins, flavonoids, and anthocyanin contents influence antioxidant and anticancer activities of Rubus fruits from the Western Ghats, India. Food. Sci. Hum. Wellness. 8(1), 73–81.
Murray, C.J., Ikuta, K.S., Sharara, F., Swetschinski, L., Aguilar, G.R., Gray, A. and Tasak, N. 2022. The global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. Lancet 399(10325), 629–655.
Nagoba, B.S. 2019. Clinical Microbiology. New Delhi, India: Wolters Kluwer India Pvt. Ltd.
NCCLS, 2000.National Committee For Clinical Laboratory Standards. Approved Standard M7-A5: Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically. 5th ed.
Omwenga, I., Aboge, G.O., Mitema, E.S., Obiero, G., Ngaywa, C., Ngwili, N., Wamwere, G., Wainaina, M. and Bett, B. 2021. Antimicrobial usage and detection of multidrug-resistant Staphylococcus aureus, including methicillin-resistant strains, in raw milk of livestock from Northern Kenya. Microbial Drug Resist. 27(6), 843–854.
Pant, A.F., Özkasikci, D., Fürtauer, S. and Reinelt, M. 2019. Effect of deprotonation on the reaction kinetics of gallic acid-based oxygen scavenger. Front. Chem. 7, 680.
Park, S. and Ronholm, J. 2021. Staphylococcus aureus in agriculture: lessons in evolution from a multispecies pathogen. Clin. Microbiol. Rev. 34, 182.
Qiu, G., Rui, Y., Zhang, J., Zhang, L., Huang, S., Wu, Q., Li, K., Han, Z., Liu, S. and Li, J. 2018. Macrolide resistance selection in Tibetan pigs with a high Mycoplasma hyopneumoniae load. Microbial Drug Resist. 24(7), 1043–1049.
Quinn , P.J., Carter, M.E., Markey, B., and Carter, G.R. 2004. Clinical and veterinary microbiology osby. St. Louis, MO, USA: Mosby (Elsevier). Pp: 26-63.
Rajamanickam, K., Yang, J. and Sakharkar, M.K. 2019. Gallic acid potentiates the antimicrobial activity of tulathromycin against two key pathogens causing BRD. Front. Pharmacol. 9, 415123.
Rather, M.A., Gupta, K. and Mandal, M. 2021. Microbial biofilm: formation, architecture, antibiotic resistance, and control strategies. Braz. J. Microbiol. 52(4), 1701–1718.
Sabbagh, S., Eslamifar, Z., Goudarzi, M. and Ebrahimzadeh, A. 2023. Gallic acid protects against methotrexate-induced intestinal mucositis: oxidative stress, histopathology and inflammatory status. Jundishapur J. Natl. Pharm. Prod. 18(4), e138661.
Sang, H., Jin, H., Song, P., Xu, W. and Wang, F. 2024. Gallic acid exerts antibiofilm activity by inhibiting adhesion of methicillin-resistant Staphylococcus aureus. Sci. Rep. 14(1), 17220.
Shah, M.M., Iihara, H., Noda, M., Song, S.X., Nhung, P.H., Ohkusu, K., Kawamura, Y. and Ezaki, T. 2007. dnaJ gene sequence-based assay for species identification and phylogenetic grouping in the genus Staphylococcus. Int. J. Syst. Evol. Microbiol. 57(1), 25–30.
Shamkhi, G.J. 2019. Effect of natural bioactive inhibitors on efflux pump in methicillin-resistant Staphylococcus aureus. Doctoral dissertation, PhD. Thesis, Genetic Engineering & Biotechnology for Postgraduate Studies, University of Baghdad, Iraq.
Subhankari, P.C., Santanu, K.M. and Somenath, R. 2011. Biochemical characteristics and antibiotic susceptibility of S. aureus isolates. Asian Pacific J. Trop. Biomed. 1(3), 212–216.
Subramanian, A.P., Jaganathan, S.K., Mandal, M., Supriyanto, E. and Muhamad, I.I. 2016. Gallic acid-induced apoptotic events in HCT-15 colon cancer cells. World J. Gastroenterol. 22(15), 3952.
Upadhayay, A., Ling, J., Pal, D., Xie, Y., Ping, F.F. and Kumar, A. 2023. Resistance-proof antimicrobial drug discovery to combat the global threat of antimicrobial resistance. Drug Resist. Updat. 66, 100890.
Vidovic, N. and Vidovic, S. 2020. Antimicrobial resistance and food animals: influence of livestock environment on the emergence and dissemination of antimicrobial resistance. Antibiotics 9, 52.
Wianowska, D. and Olszowy-Tomczyk, M. 2023. A concise profile of gallic acid—from its natural sources through biological properties and chemical methods of determination. Molecules 28(3), 1186.
Yang, K., Zhang, L., Liao, P., Xiao, Z., Zhang, F., Sindaye, D., Xin, Z., Tan, C., Deng, J., Yin, Y. and Deng, B. 2020. Impact of gallic acid on gut health: focus on the gut microbiome, immune response, and mechanisms of action. Front. Immunol. 11, 580208.