| Research Article | ||
Open Vet. J.. 2025; 15(12): 6560-6571 Open Veterinary Journal, (2025), Vol. 15(12): 6560-6571 Research Article Phytochemical profiles, chemical composition, molecular docking, and antibacterial activity of the Opuntia cladode from Al Zawia, LibyaAbdalla A. Mohamed1,2,3, Ines Dhouib1, Taher Abdelhameed2,3, Khaled Aburas4, Fatim T. Gammoudi5, Salah M. Azwai5, Ibrahim M. Eldaghayes5, Aboubaker M. Garbaj6, Lazhar Zourgui7 and Bassem Khemakhem1*1Laboratory of Plant Biotechnology, Faculty of Sciences, University of Sfax, Sfax, Tunisia 2Department of Medical Nutrition, Faculty of Medical Technology, University of Zawia, Zawiya, Libya 3Biomedical Research Team, University of Zawia, Zawiya, Libya 4National Medical Research Center, Zawia, Zawiya, Libya 5Department of Microbiology and Parasitology, Faculty of Veterinary Medicine, University of Tripoli, Tripoli, Libya 6Department of Food Hygiene and Control, Faculty of Veterinary Medicine, University of Tripoli, Tripoli, Libya 7University of Gabes, Research Laboratory BMA ‘Biodiversities, Molecules, Applications” Department of Biological Engineering, Higher Institute of Applied Biology of Medenine, Medenine, Tunisia *Corresponding Author: Bassem Khemakhem. University of Gabes, Research Laboratory BMA ‘Biodiversities, Molecules, Applications” Department of Biological Engineering, Higher Institut of Applied Biology of Medenine, Medenine, Tunisia. Email: bassem.khemakhem [at] fss.usf.tn Submitted: 12/09/2025 Revised: 15/11/2025 Accepted: 29/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
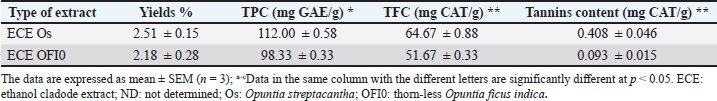
AbstractBackground: Opuntia spp. are prickly pear cactus species that have long been used for medical purposes. Mature cladodes are also used to feed animals during droughts. Aim: The phytochemical and chemical composition of cladode extracts from Opuntia streptacantha and thornless Opuntia ficus indica cultivated in Al-Zawia-Libya and to evaluate their antibacterial activity through both in vitro assay and molecular docking analysis. Methods: Cladode extracts from two Opuntia species cultivated in Libya were analyzed in this study to compare their chemical composition and evaluate their antibacterial properties explained by structure-function relationships studies. Results: The ethanolic extract of O. streptacantha registered the highest total polyphenolic content, total flavonoid content, and tannin contents (112.00 ± 0.58 mg GAE/g, 64.67 ± 0.88 mg CAT/g, and 0.408 ± 0.046 mg CAT/g, respectively). The ethanolic extract of thornless O. ficus-indica exhibited strong antibacterial activity against several bacterial strains, particularly colistin-resistant Escherichia coli. Rutin and luteolin-7-O-glucoside interact with key active-site residues, including the catalytic residues Thr285 and His395. Conclusion: Opuntia spp. cultivated in Libya are a valuable source of biomolecules with significant antibacterial properties and may be exploited as a functional food. Keywords: Phytochemical, Opuntia spp., Molecular docking, Antibacterial. IntroductionPrickly pear cactus has long been used for medicinal purposes. The cladode of Opuntia spp. exhibits antibacterial activity due to its bioactive compound content (Maniaci et al., 2024). Bacterial pathogens, such as S. aureus and Escherichia coli, are major causes of mastitis in dairy cows, leading to milk spoilage, reduced shelf life, and costly veterinary treatments. Cladodes of Opuntia contain bioactive substances (such as polyphenols and flavonoids) that possess antibacterial properties and can exhibit the growth of pathogenic bacteria. Cladodes can help prevent milk spoilage by reducing bacterial infections, thereby improving milk quality and yield (Keba et al., 2020; Liu and Kong, 2021). Healthier cows produce more milk with superior compositional quality, which can result in higher market value, particularly for small-scale farmers without advanced refrigeration facilities (Kousta et al., 2010; Barkema et al., 2015). Cladodes can reduce infection rates and the need for medical interventions and antibiotics as a natural alternative. Exploration of new safe and natural bioactive compounds from plant extracts has become essential due to growing concerns about antibiotic resistance and drug safety (Oumato et al., 2016). Cladodes are often considered agricultural waste, making them an inexpensive and sustainable resource (Auteri et al., 2023). Mature cladodes are also used as animal feed during droughts, emphasizing their potential as a functional feed (Lopez-Garcıa et al., 2001). Although Opuntia cladodes have long been used in traditional Mexican medicine to treat type 2 diabetes mellitus (T2DM), their development as a functional food ingredient for managing various health conditions is more recent (Kashif et al., 2022). Acarbose, a microbial secondary metabolite, is one example of a compound used in treating T2DM. Despite extensive research on the beneficial properties of Opuntia plants, limited data exist regarding the anti–α-amylase and antibacterial effects of ethanolic extracts from Opuntia cladodes, particularly O. streptacantha and thornless O. ficus-indica cultivated in Libya. Therefore, the present study aims to determine the phytochemical and chemical composition of ethanolic cladode extracts (ECEs) from these species and to evaluate their antibacterial and anti–α-amylase activities through in vitro and in silico analyses. Materials and MethodsPlant collectionMature and young cladodes (O. streptacantha and O. ficus-indica) were collected between March and April 2023 in Al-Zawia, Libya (32°36'29.3"N 12°43'06.2"E, 32°46'12.1"N 12°44'52.2"E) and transported to the Center for Medical Research in Zawia. The spines were stripped (or plucked) off, and the cladodes were cleaned using distilled water before being finely chopped. The pieces were oven-dried at 55°C for 2 days until fully dehydrated, and then ground into a fine powder using an electric grinder (ARSHIA, Flugel Company, Germany). Extraction procedureFor each extraction, 100 g of cladode powder was soaked in 400 ml of absolute ethanol at room temperature for 1 day. The mixture was then filtered through the Whatman No. 2 filter paper to separate the solid residues. The ECE was concentrated using a rotary evaporator set to 55°C. The resulting solid extracts were then collected and kept at −20°C until they were required for analysis. Polyphenol quantificationTotal polyphenolic content (TPC)The Folin–Ciocalteu assay, modified from Lee et al. (2002), was used to detect the total polyphenolic content. Briefly, the mixture was formed by adding 125 μl of both the 0.2 N Folin–Ciocalteu reagent and the cactus extract and 500 μl of distilled water. After 3 minutes, 3 ml of distilled water and 1,250 µl of 7% sodium carbonate were added. The mixture was then incubated for 3 hours in the dark at standard laboratory temperature for 3 hours. Absorbance was quantified at 760 nm using a spectrophotometer. TPC was quantified as milligrams of gallic acid equivalent per gram of dry weight (mg GAE/g DW) based on a gallic acid standard curve (0–100 µg/ml). Total flavonoid content (TFC)The aluminum chloride method was used to assess TFC (Palacios et al., 2011). Briefly, the process involved mixing 75 μl of the 5% NaNO2 solution with 250 μl of the extract. The resulting solution was then reacted for 6 minutes at a standard laboratory temperature. Then, 150 µl of aluminum chloride solution was added and incubated for 5 minutes, followed by the addition of 500 µl of 1 M sodium hydroxide. Distilled water was added until the total volume reached 2,500 μl. Absorbance was calculated at 430 nm. TFC was expressed as milligrams of catechin equivalent per gram of dry weight (mg CAT/g DW), based on a catechin standard curve (0–100 µg/ml). Tannin contentsThe method used to determine the tannin content was modified by Muhammad (2019). In brief, 1.5 ml of concentrated hydrochloric acid (12M) was mixed with 300 µl of the extract and 3 ml of 4% vanillin in methanol. The mixture was incubated for 15 minutes, and the absorbance was measured at 500 nm. Tannin content was quantified as catechin equivalent (mg CAT/g DW) per gram of dry weight (mg CAT/g DW) based on a catechin standard curve (0–200 µg/ml). Liquid chromatography/electrospray ionization/mass spectroscopy (LC-ESI-MS) analysisFor ECE, Ayaz et al. (2005) used a protocol to detect phenolic acid and flavonoids. In brief, 0.5 g of powder was mixed with 10 ml of absolute ethanol and agitated for 24 hours at room temperature for 24 hours. The samples were centrifuged (4,000 rpm, 25 minutes) and filtered through a 0.45 µm Millipore membrane. Aliquots (5 µl) were injected into the Liquid Chromatography-Mass Spectrometry system. Separation and detection were performed according to the protocol of Zourgui et al. (2020). Biological activitiesMicrobial strains and growth conditionsFive bacterial strains were used to assess antibacterial activity. Gram-positive bacteria were Bacillus cereus (BC7) and S. aureus (SA121), and Gram-negative bacteria were Klebsiella pneumonia (Kp243), E. coli (EC321 colistin-resistant), and E. coli (EC47 colistin-sensitive). These isolates were obtained from the Food-borne Libyan type Bacterial Collection at the Department of Food Hygiene, Faculty of Veterinary Medicine, University of Tripoli, Libya. They were identified using standard microbiological techniques and confirmed by 16s mRNA sequencing (Garbaj et al., 2016; Nass et al., 2018; Nass et al., 2019; Azwai et al., 2024). Bacteria were cultured on MHA plates and incubated for 1 day at 37°C. Final inoculum concentrations of 106 CFU/ml were used for Minimum inhibitory concentration and Minimum Bactericidal Concentration determination (Hsouna et al., 2011). The agar well diffusion methodThe agar well diffusion assay (following the procedure of Hsouna et al., 2011) was used to test the antibacterial properties. A fresh bacterial suspension (106 CFU/ml) was inoculated onto an MHA plate using sterile swabs. After drying, 6 mm diameter wells were made using a sterile pasteur pipette, and 50 μl of each extract (150 mg/ml 10% DMSO) was added. DMSO (10%) was used as a negative control. First, the plates were preincubated at 4°C for 2 hours to facilitate diffusion and then incubated at 37°C for 24 hours. The inhibition zone diameter was measured to determine antibacterial activity. Microdilution method for determining the MIC and MBC valuesMIC values were determined using a microdilution assay for strains showing inhibition zones (Emad, 2012). Stock solutions of each extract (150 mg/ml in 10% DMSO) were serially diluted to obtain concentrations ranging from 1.17 to 150 mg/ml. Each well contained 100 µl of extract, 90 µl of Mueller–Hinton broth, and 10 µl of bacterial suspension (106 CFU/ml). The positive controls consisted of the bacterial suspension, whereas the negative controls contained 10% DMSO without extract. 25 µl of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (0.5 mg/ml) was added to each well after incubation at 37°C for 24 hours to assess bacterial growth. No visible growth was observed in MIC at the lowest concentration. The MBC was determined as the lowest concentration showing no bacterial growth after subculturing on agar plates for 48 hours at 37°C (Daoud et al., 2015). Statistical analysisData are expressed as mean ± SEM (n=3). Statistical analyses were performed using the Statistical Package for the Social Sciences version 20. One-way analysis of variance followed by Duncan’s and t-tests were used to determine significant differences (α ≤ 0.05). Ethical approvalNot needed for this study. ResultsPhytochemical studies: extraction yield and total phenol, flavonoid, and tannin contentsTable 1 summarizes the extraction yields, TPC, TFC, and the tannin contents of ethanolic extracts for two species. Table 1. TPC, TFC, and tannin content extracted yields in ECE.
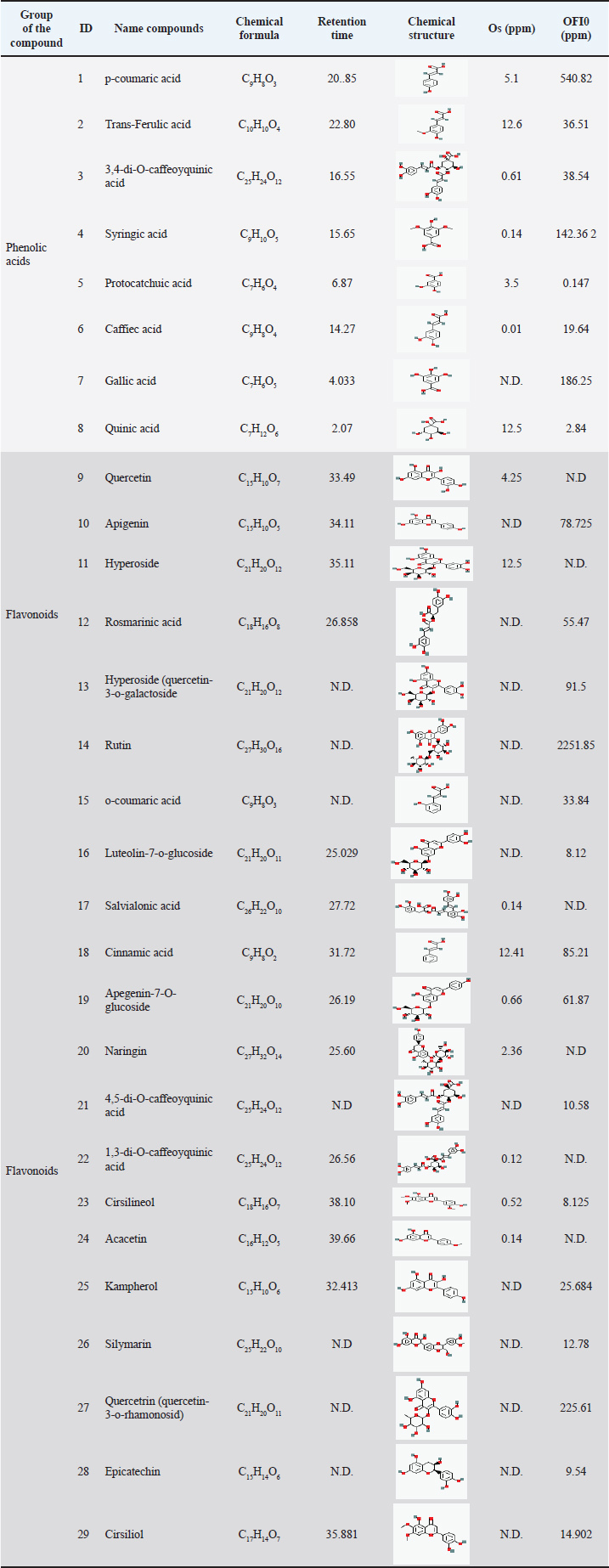
The total phenolic content of ethanolic extracts was expressed as GAE/g (mg), whereas the total flavonoid and tannin contents were expressed as CAT/g (mg). The ECE of O. streptacantha extracts showed the highest phenolic acid, flavonoid, and tannin contents. The obtained results indicate that the ECEs of Os and OFI0 were not significantly different (p ≤ 0.05) in terms of total phenolic content at a concentration of 112.00 mg GAE/g and 98.33 mg CAT/g. The flavonoid contents ranged from 51.67 mg CAT/g in the ECE of OFI0 to 64.67 mg CAT/g in the ECE of Os, with no significant difference observed (P ≤ 0.05). Tannin content ranged from 0.093 mg CAT/g in the ECE of OFI0 to 0.408 mg CAT/g in the ECE of Os. LC-ESI-MS analysis of O. streptacantha and thorn-less O. ficus indicaLC–ESI–MS analysis identified phenolic acids and flavonoids in the ethanolic extracts of O. streptacantha and thorn-less O. ficus-indica (Table 2). A total of 29 compounds were identified, including 8 phenolic acid compounds and 21 flavonoid molecules. Under ESI conditions, all analytes exhibited higher sensitivity in the negative mode. Among these phenolic acids, p-coumaric acid was the most abundant compound in the ethanolic extract, with the highest concentration in OFI0 compared to Os (540.82 ppm and 5.1 ppm, respectively), followed by gallic acid with a concentration of 186.25 ppm registered in OFI0, which was undetectable in Os. In terms of flavonoid contents, rutin exhibited the highest concentration in the ECE of OFI0 (2,251.85 ppm) and was undetectable in the ECE of Os, followed by quercetin-3-o-rhamonosid (225.61 ppm), which was also undetectable in the ECE of Os. Apigenin was not detected in the ECE of Os. Table 2. Chemical composition of ECEs for two types of Opuntia from Libya.
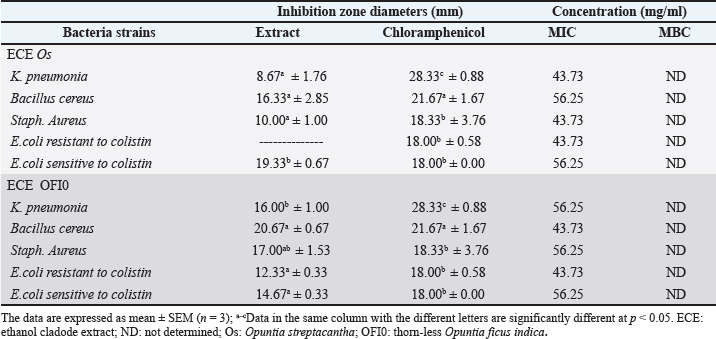
Biological activitiesMIC and MBC antibacterial activities of O. cladode extractsThe antibacterial activity of the O. cladodes extracts was assessed using the agar diffusion method. The microorganisms examined in this study are among the most significant human and animal pathogens known to cause contamination, food spoilage, and foodborne diseases (Eshamah et al., 2020). The inhibition zones ranged from 8.67 to 20.67 mm (Table 3). Significant differences in inhibition zones were observed between the ECEs (p ≤ 0.05). Both extracts exhibited similar antibacterial activity against S. aureus (SA121) and B. cereus (BC7), with inhibition zones of 16.33 ± 2.85 mm and 10.00 ± 1.00 mm for O. streptacantha, and 20.67 ± 0.67 mm and 17.00 ± 1.53 mm for thorn-less O. ficus-indica, respectively. These results were comparable to the positive control (chloramphenicol: 18.33 ± 3.67 mm and 21.67 ± 0.67 mm, respectively). Table 3. Antibacterial activities MIC and MBC for Opuntia species cladode extracts.
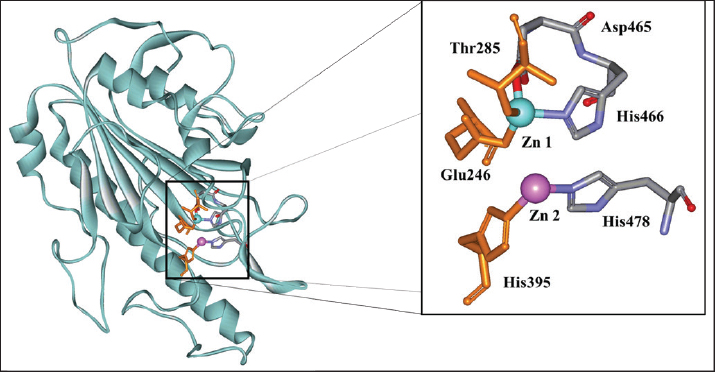
For gram-negative bacteria (K. pneumonia), the ECE of O. streptacantha produced an inhibition zone of 8.67 ± 1.76 mm, whereas the extract of thornless O. ficus-indica produced a markedly higher activity (16.01 ± 1.00 mm). However, both antibiotics were less effective than the reference antibiotic (28.33 ± 0.88 mm). In contrast, the ECE of O. streptacantha showed a strong antibacterial effect on E. coli, with an inhibition zone of 19.33 ± 0.67 mm, which was slightly higher than that of chloramphenicol (18.00 ± 0.00 mm) and greater than that of O. ficus-indica (14.67 ± 0.33 mm). Additionally, the MIC and MBC values were determined to further evaluate the antimicrobial potential of the extracts (Table 3). The ECE of O. streptacantha exhibited the lowest MIC (43.73 mg/mL) against S. aureus (SA121), K. pneumonia (Kp243), and E. coli (EC47 colistin resistant). Similarly, the ECE exhibited the lowest MIC value (43.73 mg/ml) against B. cereus (BC7) and E. coli (EC321 colistin sensitive). Structure-function relationship study of MCR-1In this study, a molecular docking approach was employed to investigate how the compounds identified in thorn-less O. ficus-indica contribute to antibacterial activity against colistin-resistant bacteria, using eugenol and tetrandrine as reference inhibitors. Mutations in chromosomal genes, such as pmrAB, phoPQ, and mgrB, have been linked to resistance to colistin (Luo et al., 2017; Abiola et al., 2014). The MCR-1 enzyme (pdb code: 5GRR) contains two distinct domains. The first is an N-terminal transmembrane domain with five putative α-helices, while the second is a C-terminal α/β/α sandwich domain that faces the periplasmic space, which contains the active site and mobile loop regions (Son et al., 2019). The MCR-1 active site is organized around two zinc ions (Zn1 and Zn2) coordinated by conserved residues (Glu246, Thr285, His395, Asp465, His466, and His478), which are essential for PEA-transferase activity and colistin resistance. The catalytic residues in the active site of MCR-1 include Thr285, which acts as a nucleophile; Glu246, which activates Thr285 and protonates the leaving group; and His395, which protonates the dephosphorylated lipid A (Fig. 1).
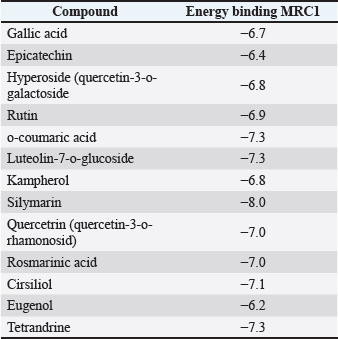
Fig. 1. MRC1 3D structure (pdb code: 5GRR) and Zn 1 and Zn 2 binding residues (catalytic residues shown in orange). The docking analysis of the compounds identified in the ethanolic extract of thornless O. ficus indica was compared with the docking profile of two standard inhibitors (eugenol and tetrandrine) reported in a previous study as MRC-1 inhibitors (Wang et al., 2018; Yi et al., 2022). These two molecules were docked using the same parameters as the docking of our compounds. The results showed that the binding led to the affinity of the corresponding compounds in the active sites of MRC-1. The binding energies of the identified compounds in (OFI0), eugenol, and tetrandrine are presented in Table 4. Table 4. The binding energies of specific compound from the ECE of OFI0, Eugenol and Tetrandrine with MRC1.
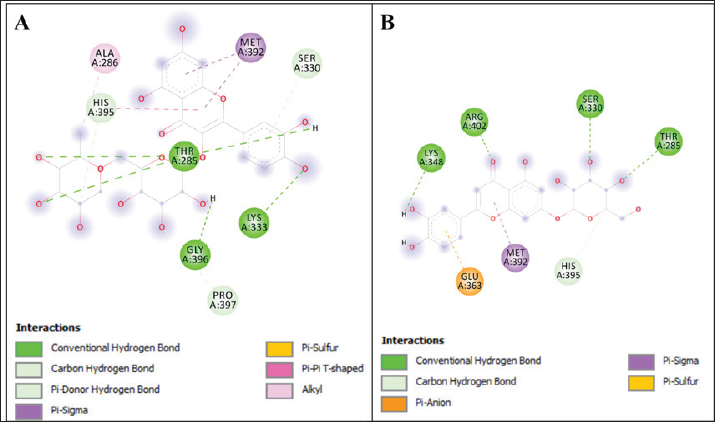
In summary, the binding affinities of the identified compounds in thorn-less O. ficus-indica (ranged from −6.4 to −8.0 kcal/mol) were comparable to those of the reference compounds eugenol and tetrandrine (binding energy −6.2 and −7.3 kcal/mol, respectively). To further analyze these results, the binding interactions of all the identified compounds in thornless O. ficus-indica against MRC-1 were compared. The analysis highlighted two compounds with particularly interesting interaction profiles: rutin and luteolin-7-O-glucoside (Fig. 2). Rutin and Luteolin-7-o-glucoside were anchored in the active site of MRC-1 through electrostatic, hydrophobic, and hydrogen interactions. These compounds also interact with the catalytic residue Thr285. Specifically, rutin forms three hydrogen bonds with Thr285 (catalytic amino acid) and two additional hydrogen bonds with Lys 333 and Gly 396. Two hydrophobic bonds Pi-Pi were found with Ala 286, Met 392, and His 395 (catalytic residue). Carbone hydrogen bonds with His 395 (catalytic residue) and Pro 397 were observed. One pi-donner hydrogen bond was established with Ser 330. On the other hand, the luteolin-7-o-glucoside MRC1 complex is stabilized by 4 hydrogens interaction with Lys 348, Arg 402, Ser 330, and Thr 285 (catalytic residue). The electrostatic bond-type pi-anion was formed by Glu 363. Pi-sigma hydrophobic interactions were observed with Met 392, and carbon–hydrogen bonds were observed with His 395.
Fig. 2. Interaction profile of rutin (A) and luteolin-7-o-glucoside (B) against MRC1 (pdb code: 5GRR). DiscussionPhenolic compounds are small molecules characterized by the presence of one or more phenol groups in their chemical structure. This group of compounds can be divided into smaller groups, including flavonoids, tannins, and phenolic acids, which are found in plant extracts (Ponnusamy et al., 2011; Ćavar Zeljković et al., 2021). Plants produce a wide variety of polyphenols and many other compounds that are used to protect against various pathogens and UV radiation. Numerous factors, including physiological-biochemical, molecular-genetic, and environmental factors, influence polyphenol biosynthesis and accumulation in plants. The solubility of these compounds also depends on the type of solvent used (Ksouri et al., 2008; Del Socorro Sánchez Correa et al., 2023). The ECEs of Os and OFI0, collected from Libya, exhibited high polyphenol levels (112 and 98.33 mg GAE/g, respectively), and prior research on O. ficus-indica fruit extract also documented its total phenolic content, which showed a value of 59.48 GAE/100 g dry weight (Medina-Pérez et al., 2019). The flavonoid concentrations of the examined samples were higher than those reported by Dib et al. (2013) (11.86 mg CAT/g of DW) (Dib et al., 2013). The total tannin contents of the two ECEs of Os and OFI0 were 0.093 and 0.408 mg CAT/g, respectively. The tannin contents in Os extracts were significantly higher than those in the OFI0 ECE. These findings are similar to the results found by Haile et al. (2016) in the same species grown in Ethiopia. In fact, the flavonoid and tannin contents from the ECE showed the same pattern, with the Os extract having the highest contents and the OFI0 extract having the lowest contents. The tannin content was the lowest, with a value of 1 mg CAT/g. LCMSMS analysis for phenolic and flavonoid compounds in the ethanolic extracts for Os and OFI0 showed 29 compounds. The 29 compounds in the ethanolic extracts included 8 phenolic acids and 21 flavonoids. The concentrations of the identified compounds ranged from 0.01 to 2251.85 ppm. Caffeic acid had the lowest concentration (0.01 ppm) in the ECE of Os. Rutin in the ECE of OFI0 had the highest concentration (2251.85 ppm), as reported previously (Rosiak et al., 2023; Wang et al., 2023). Regarding the flavonoid compounds present in the ECE of Os, hyperoside was the most abundant component in the extracts, with a concentration of 12.5 ppm. Similar results were achieved by Dib et al. (2013), Boutakiout et al. (2018), and Wit et al. (2013). On the other hand, the OFI0 extract showed different results, where rutin was the most abundant flavonoid compound, with a concentration of 2251.85 ppm. These results agree with those of Belhadj Slimen and his group in 2019, who found that rutin had the highest concentration in the ECEs (31.927 mg/kg) (Belhadj Slimen et al., 2019). Transferulic acid was the main phenolic acid identified in the ECE of Os, while p-coumaric acid was the predominant phenolic acid in the ethanolic extract of OFI0. The differences in the phenolic and flavonoid contents and concentrations between the two species agree with the results reported by De Santiago et al. (2018a, b). They stated that differences in environmental conditions, the original, maturity stage, harvesting season, species type, and post-harvest treatments influence the detection and amounts of these compounds (Park et al., 2004; Djeridane et al., 2006). In the investigation of antibacterial activity, Opuntia species extracts from Libya showed efficacy against gram-positive and gram-negative germs. The antibacterial activity of Opuntia extracts may be attributed to their high polyphenol content, particularly trans ferulic acid, which has been studied for its bacteriostatic activity against E. coli, Bacillus subtilis, and S. aureus. Ferulic acid also inhibited S. aureus and E. coli (Bhattacharya et al., 2016; Pinheiro et al., 2021). Another previous study on the antibacterial activity of ethanolic extracts of O. stricta cladodes (Affi et al., 2021) reported an inhibition zone against gram-positive and gram-negative bacteria. Similar results were reported for the ethanolic extract of O. ficus indica L. mill cladode, the inhibition zone, which produces against gram-positive (S. aureus) and gram-negative bacteria (E. coli) (Blando et al., 2019). Furthermore, the ECE of OFI0 exhibited antibacterial activity against E. coli colistin resistance. Molecular docking is a powerful computational approach for predicting the binding configurations and affinities of potential drug candidates. (Elkady et al., 2022). The molecular docking approach was used in our investigation to provide deeper insights into the antibacterial potential of the compounds identified exclusively in the ethanolic cladodes extract of OFI0. These compounds were docked as ligands into key enzymes implicated in colistin resistance mechanisms. To the best of our knowledge, recent studies have reported the synergistic effect of eugenol (Wang et al., 2018) and tetrandrine (Yi et al., 2022) with colistin against clinically isolated colistin-resistant E. coli strains. Interestingly, the docking result shows that eugenol and tetrandrine interact with several active site amino acids but not with the catalytic residue, whereas Rutin and Luteoline-7-o-glucoside interact with the catalytic residues Trp 286 and His 395. The results of this study also agreed with those of Selma, W. B and colleagues tested the ability of essential oil from Tunisian Thymus algeriensis to kill gram-negative bacterial isolates that had previously shown resistance to colistin in vitro. Additionally, they confirmed the enhanced effect (synergism) achieved when this essential oil and colistin were administered together against the colistin-resistant E. coli strains carrying the mcr-1 gene. Furthermore, a docking study showed that carvacrol exhibited high binding free energies against MCR-1, a phosphoethanolamine transferase catalytic domain (PDB ID: 5GRR). These findings allowed them to confirm the therapeutic strategy to use carvacrol alone or as an antibiotic adjuvant to colistin against gram-negative bacteria resistant to colistin (Ben Selma et al., 2024). Similarly, our findings support the potential use of rutin and luteolin-7-o-glucoside alone or synergistically with colistin as an effective strategy against gram-negative bacteria exhibiting resistance to colistin. This suggestion is supported by Yi et al. (2024), who found that rutin is effective against multidrug-resistant E. coli (Yi et al., 2024). Wu et al. (2023) also found that Luteoline-7-o-glucoside is effective against drug-resistant intestinal microbiota, including E. coli (Wu et al., 2023). ConclusionThe study’s findings demonstrated that the O. streptacantha extract contains higher levels of polyphenols than the thornless O. ficus indica extract. Both extracts exhibited antibacterial activity against gram-positive and gram-negative bacteria; however, only the ECE of thornless O. ficus indica showed antibacterial activity against E. coli resistant to colistin. The structure-function relationship analysis showed that the bioactive compounds responsible for this antibacterial activity against colistin-resistant E. coli are rutin and luteolin-7-o-glucoside. These findings highlight the potential role of Opuntia species as valuable natural resources for treating some bacterial infections and other animal health issues. Nevertheless, further in-depth studies on the isolation and individual characterization of the active compounds are required to support their potential application in the development of agrifeed and/or pharmaceutical preservatives. AcknowledgmentsThe authors would like to thank the Tunisian Ministry of Higher Education and Scientific Research for their support in this project. The authors would also like to thank the Research Laboratory BMA “Biodiversity, Molecules, Application” at the Higher Institute of Applied Biology of Medenine, University of Gabes (Tunisia) for their involvement in supporting this project. The authors would also like to thank the Faculty of Veterinary Medicine at the University of Tripoli (Libya), the National Medical Research Center, Zawia (Libya), and the Laboratory of Plant Biotechnology at the Faculty of Sciences, University of Sfax, Tunisia for their support. Conflict of interestThe authors have no conflicts of interest to disclose. FundingThis study received no external funding. Authors’ contributionsB.K. drafted the manuscript. LZ, SMA, and AAM revised and edited the first draft of the manuscript. TA, SMA, AMG, and IME participated in preparing and critically checking this manuscript. ID, AAM, FTG, and KA performed the methodology. All authors have read, revised, and approved the published version of the manuscript. Data availabilityData are available upon request from the corresponding author. ReferencesAbiola, O.O., Serge, M. and Rolain, J.-M. 2014. Mechanisms of polymyxin resistance: acquired and intrinsic resistance in bacteria. Front. Microbiol. 5, 643; doi: 10.3389/fmicb.2014.00643 Affi, W., Zourgui, M.N., Lataief, S.B., Agil, A. and Zourgui, L. 2021. Comparative study of phenolic compound antioxidant and antimicrobial activities of fruits peel and cladodes from Tunisian Opuntia Stricta. J. Altern. Complement. Integr. Med. 7, 1–7. doi: 10.24966/ACIM-7562/100202 Auteri, N., Scalenghe, R. and Saiano, F. 2023. Phosphorus recovery from agricultural waste via cactus pear biomass. Heliyon 9, e19996; doi:10.1016/j.heliyon.2023.e19996 Ayaz, F.A., Hayirlioglu-Ayaz, S., Gruz, J., Novak, O. and Strnad, M. 2005. Separation, characterization, and quantitation of phenolic acids in a little-known blueberry (Vaccinium arctostaphylos L.) Fruit by HPLC-MS. J. Agric. Food. Chem. 53, 8116–8122; doi:10.1021/jf058057y Azwai, S.M., Lawila, A.F., Eshamah, H.L., Sherif, J.A., Farag, S.A., Naas, H.T., Garbaj, A.M., Salabi, A.A.E., Gammoudi, F.T. and Eldaghayes, I.M. 2024. Antimicrobial susceptibility profile of Klebsiella pneumoniae isolated from some dairy products in Libya as a foodborne pathogen. Vet. World. 1168, 1168–1176; doi:10.14202/vetworld.2024.1168-1176 Barkema, H.W., Von Keyserlingk, M.A.G., Kastelic, J.P., Lam, T.J.G.M., Luby, C., Roy, J.P., Leblanc, S.J., Keefe, G.P. and Kelton, D.F. 2015. Invited review: changes in the dairy industry affecting dairy cattle health and welfare. J. Dairy. Sci. 98, 7426–7445; doi:10.3168/jds.2015-9377 Belhadj Slimen, I., Chabaane, H., Chniter, M., Mabrouk, M., Ghram, A., Miled, K., Behi, I., Abderrabba, M. and Najar, T. 2019. Thermoprotective properties of Opuntia ficus-indica f. inermis cladodes and mesocarps on sheep lymphocytes. J. Therm. Biol. 81, 73–81; doi:10.1016/j.jtherbio.2019.02.018 Ben Selma, W., Farouk, A., Ban, Z., Ferjeni, M., Alsulami, T., Ali, H. and Boukadida, J. 2024. Thymus algeriensis essential oil: phytochemical investigation, bactericidal activity, synergistic effect with colistin, molecular docking, and dynamics analysis against gram-negative bacteria resistant to colistin. Heliyon 10, e38281; doi:10.1016/j.heliyon.2024.e38281 Bhattacharya, D., Bhattacharya, S., Patra, M.M., Chakravorty, S., Sarkar, S., Chakraborty, W., Koley, H. and Gachhui, R. 2016. Antibacterial activity of polyphenolic fraction of kombucha against enteric bacterial pathogens. Curr. Microbiol. 73, 885–896; doi:10.1007/s00284-016-1136-3 Blando, F., Russo, R., Negro, C., De Bellis, L. and Frassinetti, S. 2019. Antimicrobial and antibiofilm activity against Staphylococcus aureus of Opuntia ficus-indica (L.) Mill. cladode polyphenolic extracts. Antioxidants 8, 117; doi:10.3390/antiox8050117 Boutakiout, A., Elothmani, D., Hanine, H., Mahrouz, M., Le Meurlay, D., Hmid, I. and Ennahli, S. 2018. Effects of different harvesting seasons on antioxidant activity and phenolic content of prickly pear cladode juice. J. Saudi. Soc. Agric. Sci. 17, 471–480; doi:10.1016/j.jssas.2016.11.005 Ćavar Zeljković, S., Šišková, J., Komzáková, K., De Diego, N., Kaffková, K. and Tarkowski, P. 2021. Phenolic Compounds and Biological Activity of Selected Mentha Species. Plants 10, 550; doi:10.3390/plants10030550 Daoud, A., Malika, D., Bakari, S., Hfaiedh, N., Mnafgui, K., Kadri, A. and Gharsallah, N. 2015. Assessment of polyphenol composition, antioxidant and antimicrobial properties of various extracts of Date Palm Pollen (DPP) from two Tunisian cultivars. Arab. J. Chem. 12, 3075–3086; doi:10.1016/j.arabjc.2015.07.014 Del Socorro Sánchez Correa, M., El Rocío Reyero Saavedra, M., Estrella Parra, E.A., Ontiveros, E.N., del Carmen Benítez Flores, J., Ortiz Montiel, J.G., Campos Contreras, J.E., López Urrutia, E., Guillermo Ávila Acevedo, J., Edith Jiménez Nopala, G. and Espinosa González, A.M. 2023. Ultraviolet radiation and its effects on plants. Abiotic stress in plants-adaptations to climate change. IntechOpen, London. doi: 10.5772/intechopen.109474 De Santiago, E., Domínguez-Fernández, M., Cid, C. and De Peña, M.P. 2018. Impact of cooking process on nutritional composition and antioxidants of cactus cladodes (Opuntia ficus-indica). Food. Chem. 240, 1055–1062; doi:10.1016/j.foodchem.2017.08.039 De Santiago, E., Pereira-Caro, G., Moreno-Rojas, J.M., Cid, C. and De Peña, M.P. 2018. Digestibility of (Poly)phenols and antioxidant activity in raw and cooked cactus cladodes (Opuntia ficus-indica). J. Agric. Food Chem. 66, 5832–5844; doi:10.1021/acs.jafc.8b01167 Dib, H., Beghdad, M.C., Belarbi, M., Seladji, M. and Ghalem, M. 2013. Antioxidant activity of phenolic compounds of the cladodes of Opuntia ficus-indica mill. from northwest Algeria. Djeridane, A., Yousfi, M., Nadjemi, B., Boutassouna, D., Stocker, P. and Vidal, N. 2006. Antioxidant activity of some algerian medicinal plants extracts containing phenolic compounds. Food. Chem. 97, 654–660; doi:10.1016/j.foodchem.2005.04.028 Elkady, W.M., Raafat, M.M., Abdel-Aziz, M.M., Al-Huqail, A.A., Ashour, M.L. and Fathallah, N. 2022. Endophytic fungus from Opuntia ficus-indica: a source of potential bioactive antimicrobial compounds against multidrug-resistant bacteria. Plants 11(8), 1070. Emad, M.A. 2012. Antimicrobial, antioxidant and phytochemical investigation of Balanites aegyptiaca (L.) Del. edible fruit from Sudan. Afr. J. Biotechnol. 11, 11535–11542; doi: 10.5897/AJB12.1102 Eshamah, H.L., Naas, H.T., Garbaj, A.M., Azwai, S.M., Gammoudi, FT., Barbieri., I., Eldaghayes. and IM. 2020. Extant of pathogenic and spoilage microorganisms in whole muscle meat, meat products and seafood sold in Libyan market. Open Vet. J. 10(3), 276–288. Garbaj, A.M., Awad, E.M., Azwai, S.M., Abolghait, S.K., Naas, H.T., Moawad, A.A., Gammoudi, F.T., Barbieri, I. and Eldaghayes, I.M. 2016. Enterohemorrhagic Escherichia coli O157 in milk and dairy products from Libya: isolation and molecular identification by partial sequencing of 16S rDNA. Vet. World 9, 1184–1189; doi:10.14202/vetworld.2016.1184-1189 Haile, K., Mehari, B., Atlabachew, M. and Chandravanshi, B. 2016. Phenolic composition and antioxidant activities of cladodes of the two varieties of cactus pear (Opuntia ficus-indica) grown in Ethiopia. Bull. Chem. Soc. Ethiop. 30, 347; doi:10.4314/bcse.v30i3.3 Hsouna, A.B., Trigui, M., Mansour, R.B., Jarraya, R.M., Damak, M. and Jaoua, S. 2011. Chemical composition, cytotoxicity effect and antimicrobial activity of Ceratonia siliqua essential oil with preservative effects against Listeria inoculated in minced beef meat. Int. J. Food. Microbiol. 148, 66–72; doi:10.1016/j.ijfoodmicro.2011.04.028 Kashif, R.R., D’Cunha, N.M., Mellor, D.D., Alexopoulos, N.I., Sergi, D. and Naumovski, N. 2022. Prickly Pear Cacti (Opuntia spp.) Cladodes as a Functional Ingredient for Hyperglycemia Management: a Brief Narrative Review. Medicina. (Mex). 58, 300; doi:10.3390/medicina58020300 Keba, A., Rolon, M.L., Tamene, A., Dessie, K., Vipham, J., Kovac, J. and Zewdu, A. 2020. Review of the prevalence of foodborne pathogens in milk and dairy products in Ethiopia. Int. Dairy J. 109, 104762; doi:10.1016/j.idairyj.2020.104762 Kousta, M., Mataragas, M., Skandamis, P. and Drosinos, E.H. 2010. Prevalence and sources of cheese contamination with pathogens at farm and processing levels. Food. Control. 21, 805–815; doi:10.1016/j.foodcont.2009.11.015 Ksouri, R., Megdiche, W., Falleh, H., Trabelsi, N., Boulaaba, M., Smaoui, A. and Abdelly, C. 2008. Influence of biological, environmental and technical factors on phenolic content and antioxidant activities of Tunisian halophytes. C. R. Biol. 331, 865–873; doi:10.1016/j.crvi.2008.07.024 Lee, J.C., Kim, H.R., Kim, J. and Jang, Y.S. 2002. Antioxidant property of an ethanol extract of the stem of Opuntia ficus-indica var. Saboten. J. Agric. Food Chem. 50, 6490–6496; doi: 10.1021/jf020388c Liu, C. and Kong, D. 2021. Business strategy and sustainable development: evidence from China. Bus. Strategy. Environ. 30, 657–670; doi:10.1002/bse.2645 Lopez-Garcıa J, Fuentes-Rodr´ıguez J, and Rodr´ıguez R. 2001. Production and use of Opuntia as forage in northern Mexico. In: Mongragon-Jacobo C, P ´ erez-Gonz ´ alez S, editors. Cactus ´(Opuntia spp.) as forage. Rome: FAO Plant Production and Protection Paper. pp. 29–36. Luo, Q., Yu, W., Zhou, K., Guo, L., Shen, P., Lu, H., Huang, C., Xu, H., Xu, S., Xiao, Y. and Li, L. 2017. Molecular epidemiology and colistin resistant mechanism of mcr-positive and mcr-negative clinical isolated Escherichia coli. Front. Microbiol. 8, 2262; doi:10.3389/fmicb.2017.02262 Maniaci, G., Ponte, M., Giosuè, C., Gannuscio, R., Pipi, M., Gaglio, R., Busetta, G., Di Grigoli, A., Bonanno, A. and Alabiso, M. 2024. Cladodes of Opuntia ficus-indica (L.) as a source of bioactive compounds in dairy products. J. Dairy Sci. 107, 1887–1902; doi:10.3168/jds.2023-23847 Medina-Pérez, G., Zaldívar-Ortega, A.K., Cenobio-Galindo, A.D.J., Afanador-Barajas, L.N., Vieyra-Alberto, R., Estefes-Duarte, J.A. and Campos-Montiel, R.G. 2019. Antidiabetic activity of cactus acid fruit extracts: simulated intestinal conditions of the inhibitory effects on α-amylase and α-glucosidase. Appl. Sci. 9, 4066; doi:10.3390/app9194066 Muhammad, K. 2019. Assessment of phytochemical content and antioxidant activity of the extracts collected from the leaves and flowers of Hypericum scabrum. Tikrit. J. Agric. Sci. 19, 93–102; doi:10.25130/tjas.19.1.10 Naas, H.T., Zurghani, M.M., Garbaj, A.M., Azwai, S.M., Eshamah, H.L., Gammoudi, F.T., Abolghait, S.K., Moawad, A.A., Barbieri, I. and Eldaghayes, I.M. 2018. Bacillus cereus as an emerging public health concern in Libya: isolation and antibiogram from food of animal origin. Libyan J. M. Sci. 2, 56–61. Naas, H.T., Edarhoby, R.A., Garbaj, A.M., Azwai, S.M., Abolghait, S.K., Gammoudi, F.T., Moawad, A.A., Barbieri, I. and Eldaghayes, I.M. 2019. Occurrence, characterization, and antibiogram of Staphylococcus aureus in meat, meat products and some seafood from Libyan retail markets. Vet. World 12, 925–931. Oumato, J., Zrira, S., Petretto, G.L., Saidi, B., Salaris, M. and Pintore, G. 2016. Volatile constituents and polyphenol composition of Opuntia ficus- indica (L.) Mill from Morocco. Rev. Mar. Sci. Agron. Vét. 4(3), 5–11. Palacios, I., Lozano, M., Moro, C., D’Arrigo, M., Rostagno, M.A., Martínez, J.A., García-Lafuente, A., Guillamón, E. and Villares, A. 2011. Antioxidant properties of phenolic compounds occurring in edible mushrooms. Food. Chem. 128, 674–678; doi:10.1016/j.foodchem.2011.03.085 Park, K., -Y.., Jung, G., -O.., Lee, K., -T.., Choi, J., Choi, M., -Y.., Kim, G., -T.., Jung, H., -J.., Park, H. and -J. 2004. Antimutagenic activity of flavonoids from the heartwood of Rhus verniciflua. J. Ethnopharmacol. 90, 73–79; doi:10.1016/j.jep.2003.09.043 Pinheiro, P.G., Santiago, G.M.P., Da Silva, F.E.F., De Araújo, A.C.J., De Oliveira, C.R.T. and Freitas, P.R. 2021. Antibacterial activity and inhibition against Staphylococcus NorA efflux pump by ferulic acid and its esterified derivatives. Asian Pacific J. Trop. Biomed. (Asian. Pac. J. Trop. Biomed). 11(9), 405–413; doi:10.4103/2221-1691.321130 Ponnusamy, S., Ravindran, R., Zinjarde, S., Bhargava, S. and Ravi Kumar, A. 2011. Evaluation of traditional Indian antidiabetic medicinal plants for human pancreatic amylase inhibitory effect in vitro. Evid. Based. Complement. Alternat. Med. 2011, 515647; doi:10.1155/2011/515647 Rosiak, N., Cielecka-Piontek, J., Skibiński, R., Lewandowska, K., Bednarski, W. and Zalewski, P. 2023. Do Rutin and Quercetin Retain Their Structure and Radical Scavenging Activity after Exposure to Radiation?. Molecules 28, 2713; doi:10.3390/molecules28062713 Son, S.J., Huang, R., Squire, C.J. and Leung, I.K.H. 2019. MCR-1: a promising target for structure-based design of inhibitors to tackle polymyxin resistance. Drug Discovery Today 24, 206–216; doi:10.1016/j.drudis.2018.07.004 Wang, J., Rani, N., Jakhar, S., Redhu, R., Kumar, S., Kumar, S., Kumar, S., Devi, B., Simal-Gandara, J., Shen, B. and Singla, R.K. 2023. Opuntia ficus-indica (L.) Mill. - anticancer properties and phytochemicals: current trends and future perspectives. Front. Plant. Sci. 14, 1236123; doi:10.3389/fpls.2023.1236123 Wang, Y.M., Kong, L.C., Liu, J. and Ma, H.X. 2018. Synergistic effect of eugenol with Colistin against clinical isolated Colistin-resistant Escherichia coli strains. Antimicrob. Resist. Infect Control. 7, 7–17; doi:10.1186/s13756-018-0303-7 Wit, A.D., Toit, O. and Hugo. 2013. Cactus pear antioxidants: a comparison between fruit pulp, fruit peel, fruit seeds and cladodes of eight different cactus pear cultivars (Opuntia ficus indica and Opuntia robusta). J. Food. Meas. Charact. 7(3), 101–114. Yi, K., Liu, S., Liu, P., Luo, X., Zhao, J., Yan, F., Pan, Y., Liu, J., Zhai, Y. and Hu, G. 2022. Synergistic antibacterial activity of tetrandrine combined with colistin against MCR-mediated colistin-resistant Salmonella. Biomed. Pharmacother. 149, 112873; doi:10.1016/j.biopha.2022.112873 Yi, L., Bai, Y., Chen, X., Wang, W., Zhang, C., Shang, Z., Zhang, Z., Li, J., Cao, M., Zhu, Z. and Zhang, J. 2024. Synergistic effects and mechanisms of action of rutin with conventional antibiotics against Escherichia coli. Int. J. Mol. Sci. 25, 13684; doi:10.3390/ijms252413684 Wu, Z., Gou, R., Sha, L., Yu, C., Meng, L. and Jin, X. 2023. Effects of luteolin-7-O-glucoside on intestinal microbiota dysbiosis and drug resistance transmission caused by Raoultella ornithinolytica B1645-1: modulating the composition of intestinal microbiota and promoting the transfer of blaNDM-1 gene from genus enterococcus to lactobacillus in mice. Microorganisms 11(10), 2477. Zourgui, M.N., Hfaiedh, M., Brahmi, D., Affi, W., Gharsallah, N., Zourgui, L. and Amri, M. 2020. Phytochemical screening, antioxidant and antimicrobial activities of Opuntia streptacantha fruit skin. J. Food Meas. Charact. 14, 2721–2733; doi:10.1007/s11694-020-00518-w | ||
| How to Cite this Article |
| Pubmed Style Mohamed AA, Dhouib I, Abdelhameed T, Aburas K, Gammoudi FT, Azwai SM, Eldaghayes IM, Garbaj AM, Zourgui L, Khemakhem B. Phytochemical profiles, chemical composition, molecular docking, and antibacterial activity of the Opuntia cladode from Al Zawia, Libya. Open Vet. J.. 2025; 15(12): 6560-6571. doi:10.5455/OVJ.2025.v15.i12.40 Web Style Mohamed AA, Dhouib I, Abdelhameed T, Aburas K, Gammoudi FT, Azwai SM, Eldaghayes IM, Garbaj AM, Zourgui L, Khemakhem B. Phytochemical profiles, chemical composition, molecular docking, and antibacterial activity of the Opuntia cladode from Al Zawia, Libya. https://www.openveterinaryjournal.com/?mno=289815 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.40 AMA (American Medical Association) Style Mohamed AA, Dhouib I, Abdelhameed T, Aburas K, Gammoudi FT, Azwai SM, Eldaghayes IM, Garbaj AM, Zourgui L, Khemakhem B. Phytochemical profiles, chemical composition, molecular docking, and antibacterial activity of the Opuntia cladode from Al Zawia, Libya. Open Vet. J.. 2025; 15(12): 6560-6571. doi:10.5455/OVJ.2025.v15.i12.40 Vancouver/ICMJE Style Mohamed AA, Dhouib I, Abdelhameed T, Aburas K, Gammoudi FT, Azwai SM, Eldaghayes IM, Garbaj AM, Zourgui L, Khemakhem B. Phytochemical profiles, chemical composition, molecular docking, and antibacterial activity of the Opuntia cladode from Al Zawia, Libya. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6560-6571. doi:10.5455/OVJ.2025.v15.i12.40 Harvard Style Mohamed, A. A., Dhouib, . I., Abdelhameed, . T., Aburas, . K., Gammoudi, . F. T., Azwai, . S. M., Eldaghayes, . I. M., Garbaj, . A. M., Zourgui, . L. & Khemakhem, . B. (2025) Phytochemical profiles, chemical composition, molecular docking, and antibacterial activity of the Opuntia cladode from Al Zawia, Libya. Open Vet. J., 15 (12), 6560-6571. doi:10.5455/OVJ.2025.v15.i12.40 Turabian Style Mohamed, Abdalla A., Ines Dhouib, Taher Abdelhameed, Khaled Aburas, Fatim T. Gammoudi, Salah M. Azwai, Ibrahim M. Eldaghayes, Aboubaker M. Garbaj, Lazhar Zourgui, and Bassem Khemakhem. 2025. Phytochemical profiles, chemical composition, molecular docking, and antibacterial activity of the Opuntia cladode from Al Zawia, Libya. Open Veterinary Journal, 15 (12), 6560-6571. doi:10.5455/OVJ.2025.v15.i12.40 Chicago Style Mohamed, Abdalla A., Ines Dhouib, Taher Abdelhameed, Khaled Aburas, Fatim T. Gammoudi, Salah M. Azwai, Ibrahim M. Eldaghayes, Aboubaker M. Garbaj, Lazhar Zourgui, and Bassem Khemakhem. "Phytochemical profiles, chemical composition, molecular docking, and antibacterial activity of the Opuntia cladode from Al Zawia, Libya." Open Veterinary Journal 15 (2025), 6560-6571. doi:10.5455/OVJ.2025.v15.i12.40 MLA (The Modern Language Association) Style Mohamed, Abdalla A., Ines Dhouib, Taher Abdelhameed, Khaled Aburas, Fatim T. Gammoudi, Salah M. Azwai, Ibrahim M. Eldaghayes, Aboubaker M. Garbaj, Lazhar Zourgui, and Bassem Khemakhem. "Phytochemical profiles, chemical composition, molecular docking, and antibacterial activity of the Opuntia cladode from Al Zawia, Libya." Open Veterinary Journal 15.12 (2025), 6560-6571. Print. doi:10.5455/OVJ.2025.v15.i12.40 APA (American Psychological Association) Style Mohamed, A. A., Dhouib, . I., Abdelhameed, . T., Aburas, . K., Gammoudi, . F. T., Azwai, . S. M., Eldaghayes, . I. M., Garbaj, . A. M., Zourgui, . L. & Khemakhem, . B. (2025) Phytochemical profiles, chemical composition, molecular docking, and antibacterial activity of the Opuntia cladode from Al Zawia, Libya. Open Veterinary Journal, 15 (12), 6560-6571. doi:10.5455/OVJ.2025.v15.i12.40 |