| Case Report | ||
Open Vet. J.. 2026; 16(2): 1369-1379
Open Veterinary Journal, (2026), Vol. 16(2): 1369-1379 Case Report Localized fungal osteomyelitis caused by Aspergillus sp. in the ilium and acetabulum of a dog with disseminated aspergillosis: A case reportJoão Domingos Rocha Júnior1*, Letícia Fornel Mangolin1, Igor Bruno Oliveira da Silva1, Lara Cristina de Melo Lopes1, João Pedro Garcia Luvisoto1, Thaylla Maria Ferreira2, Isadora Pezati Sabino1, Alef Winter Oliveira Alvarenga2, Jessé Ribeiro Rocha1 and Fernanda Gosuen Gonçalves Dias11Small Animal Surgery Department, University of Franca (Unifran), Franca, Brazil 2Clinical Laboratory and Veterinary Pathology Department, University of Franca (Unifran), Franca, Brazil *Corresponding Author: João Domingos Rocha Júnior. Small Animal Surgery Department, University of Franca (Unifran), Franca, Brazil. Email: j.rocha [at] unesp.br. Submitted: 11/10/2025 Revised: 13/01/2026 Accepted: 26/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
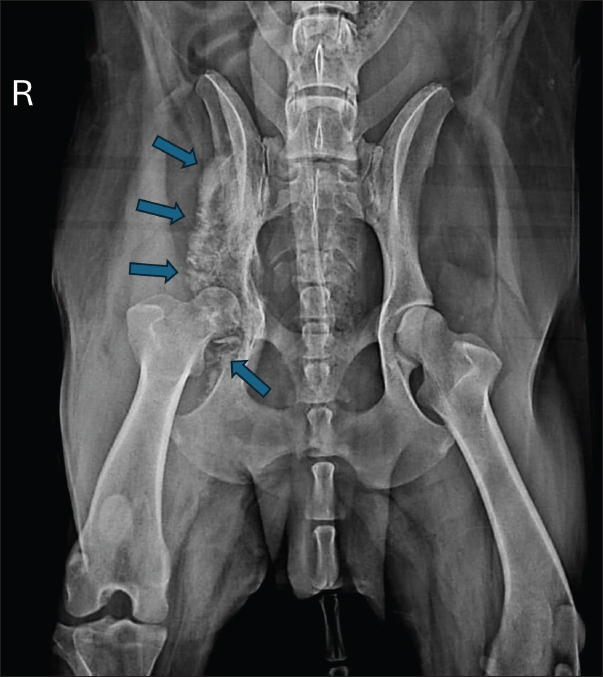
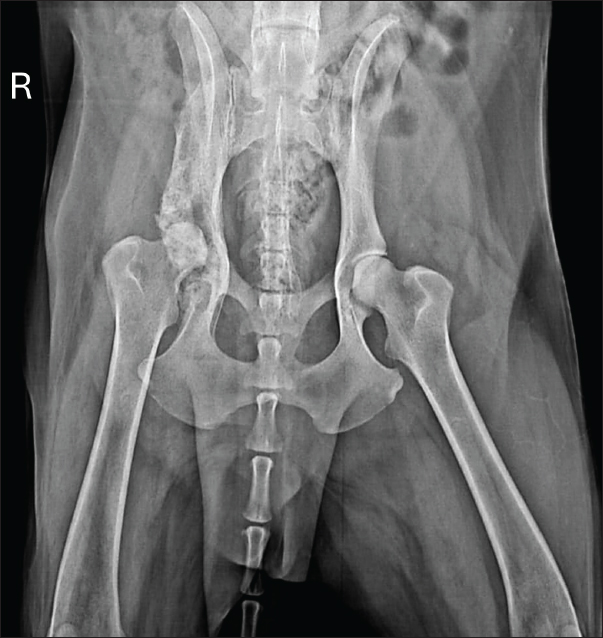
AbstractBackground: Osteomyelitis caused by Aspergillus sp. is uncommon in dogs and cats and may occur in cases of disseminated aspergillosis, a rare and poorly described condition in veterinary medicine. Case Description: Due to the scarcity of reports documenting this disease, particularly with localized involvement of the pelvic bones, the present case report aimed to describe the condition in an 8-year-old mixed-breed dog presenting with severe pain and lameness of the right pelvic limb. Radiographic examination revealed marked periosteal bone proliferation and osteolysis extending from the right wing of the ilium to the acetabulum. Incisional biopsy revealed a dense inflammatory infiltrate, consistent with chronic osteitis and myositis. Samples were submitted for bacterial and fungal cultures, with growth observed only in the fungal culture, identifying Aspergillus species. Treatment with itraconazole at 10 mg/kg every 24 hours was initiated, and the therapeutic response was monitored through periodic radiographs, which showed gradual regression of the initial lesions. Conclusion: Sixteen months after treatment initiation, the patient exhibited continuous clinical improvement without systemic signs of disease, indicating therapeutic success and a longer survival expectancy. Keywords: Aspergillosis, Itraconazole, Osteomyelitis, Systemic mycosis, Veterinary mycology. IntroductionOsteomyelitis is a destructive and progressive condition characterized by inflammation of the bone and bone marrow, occasionally involving the endosteum, periosteum, and vascular channels (Fossum and Hulse, 1992; Piermattei et al., 2009; Mello et al., 2024). Its main clinical features include pain, swelling of surrounding soft tissues, and radiographically detectable bone alterations (Hakamata et al., 2019). In most cases, etiology is bacterial, although viral and fungal causes have also been reported (González-Martín et al., 2022). The primary modes of infection are exogenous and hematogenous routes, with the former occurring mainly through direct contamination following trauma (penetrating wounds or open fractures) or surgical procedures (Gieling et al., 2019). Compared with bacterial forms, fungal osteomyelitis is less common and more frequently observed in young dogs, typically presenting with polyostotic lesions. Infection may occur via direct fungal inoculation through open fractures, surgical procedures, or bite wounds, or via inhalation or ingestion of fungal elements, with subsequent hematogenous dissemination (Johnson et al., 1997; Bubenik and Smith, 2007). The infecting fungal species vary according to geographic distribution (Thrall, 2010; Schulz, 2013); however, Coccidioides immitis, Blastomyces dermatitidis, Histoplasma capsulatum, Cryptococcus neoformans, and Aspergillus spp. are the most frequently reported fungi in osteomyelitis cases (Oxenford and Middleton, 1986; Johnson, 1994). Aspergillosis predominantly represents opportunistic infections, particularly in patients with immunosuppression due to concurrent diseases or administration of corticosteroids and/or other immunosuppressive drugs (Pape and Kirchhoff, 1996; Day, 1998; Smith et al., 1998; Harkin, 2003; Tell, 2005). Clinical manifestations include nasal, bronchopulmonary, and disseminated forms, with the latter being rare in veterinary medicine (Sanches and Coutinho, 2007). German Shepherd dogs are particularly predisposed to both sino-nasal and disseminated aspergillosis, likely due to hereditary abnormalities in humoral and cellular immune responses (Schultz et al., 2008; Seyedmousavi et al., 2015). Aspergillus terreus is the primary species implicated in disseminated aspergillosis, with infection occurring via inhalation of environmental spores, pulmonary penetration, and subsequent entry into the bloodstream, resulting in fungemia (Sharp, 1998; Harkin, 2003). Once in circulation, the pathogen reaches common infection sites such as intervertebral discs, spleen, heart, renal glomeruli, eyes, and long bones (Sharp, 1998; Harkin, 2003). Initially, the clinical signs of disseminated aspergillosis are mild and nonspecific, including fever, anorexia, weight loss, vomiting, and lethargy (Del Magno et al., 2022). As the infection progresses, more specific signs may emerge, such as uveitis, ataxia, lameness, paraparesis, renal insufficiency, and respiratory discomfort, depending on its localization (Bruchim et al., 2006; Schultz et al., 2008). Osteomyelitis is one of the most common clinical manifestations of disseminated aspergillosis, and its symptoms include pain and lameness (Kelly et al., 1995; Day, 1998; Schultz et al., 2008). Aspergillosis diagnosis is challenging and usually requires multiple diagnostic approaches to achieve a definitive result (Day, 1998; Harkin, 2003). In addition to hyperglobulinemia and elevated serum liver enzymes (alanine aminotransferase, aspartate aminotransferase, and alkaline phosphatase), hematologic abnormalities may include neutrophilia, eosinophilia, and/or monocytosis (Day, 1998; Harkin, 2003; Schultz et al., 2008). Biopsy of tissue fragments and body fluids allows for histopathological examination and mycological culture to confirm the diagnosis (Harkin, 2003). With the advancement of molecular pathogen identification tools, the diagnosis of aspergillosis can also be achieved through methods such as polymerase chain reaction (Wengenack and Binnicker, 2009). Radiographic findings in aspergillosis-associated osteomyelitis include soft tissue swelling, periosteal bone proliferation, bone resorption, cortical destruction, and increased medullary density (Day et al., 1986; Lima et al., 2013). The differential diagnoses include primary bone neoplasms (fibrosarcoma, chondrosarcoma, and osteosarcoma), metastases, multiple myeloma, bone lymphoma, bone abscesses, and metabolic bone diseases (Daleck et al., 2002; Dernell et al., 2007). Effective treatment is prolonged and challenging due to poor antifungal penetration into fungal colonies and necrotic tissues, as well as inherent resistance in some Aspergillus species (Kelly et al., 1995; Sutton et al., 1999; Zonderland et al., 2002; Sanches and Coutinho, 2007). Azoles (ketoconazole, itraconazole, fluconazole, clotrimazole, and enilconazole) are the most commonly used antifungal agents (Sanches and Coutinho, 2007). The monitoring of therapeutic response involves the regular evaluation of clinical signs and radiographic changes (Johnson, 1994; Walsh et al., 2008). To date, only one case has been reported in the global scientific literature in Bologna, Italy, in which the wing of the ilium of a dog was affected by Aspergillus species due to disseminated aspergillosis; this case also presented systemic signs such as lymphadenopathy, azotemia, and pyelonephritis (Del Magno et al., 2022). Furthermore, no reports describe acetabular involvement, and only two cases of Aspergillus sp. fungal osteomyelitis in long bones without evidence of disseminated infection have been documented (Oxenford and Middleton, 1986; Abreu et al., 2023). Given the scarcity of scientific reports describing canine aspergillosis with osteomyelitis in Brazil, the present case report aims to describe this condition affecting the pelvic region (ilium and acetabulum) of a mixed-breed dog without prior trauma or surgical history, emphasizing clinical, therapeutic, and diagnostic features essential for ruling out differentials such as primary bone neoplasms. Case DetailsAn 18-year-old, castrated male mixed-breed dog with a 3-month history of hyporexia, severe pelvic pain, and right pelvic limb lameness associated with muscle atrophy was presented to the Veterinary Clinic of the University of Franca. According to the owners, no respiratory, ocular, or renal alterations were observed. The patient had been examined 1 month earlier at another clinic for the same clinical complaints, where hematological testing identified thrombocytopenia and empirical treatment for hemoparasitosis with doxycycline was initiated. On physical examination, the dog presented with fever (39.8ºC) and reactivity of the right popliteal lymph node, pain upon palpation of the right wing of the ilium, reduced mobility of the right coxofemoral joint, and moderate atrophy of the gluteal, medial, and lateral thigh muscles. Hematological analysis revealed normocytic, hypochromic anemia with 318 metarubricytes, thrombocytosis, neutrophilic leukocytosis with a left shift, lymphopenia, hypoalbuminemia, and hyperglobulinemia. The liver and renal enzyme levels were within the reference ranges for the species. Radiographic examination revealed marked periosteal bone proliferation with extensive areas of osteolysis affecting the wing and body of the ilium, acetabulum, and right femur’s head and neck, along with adjacent soft tissue swelling (Fig. 1).
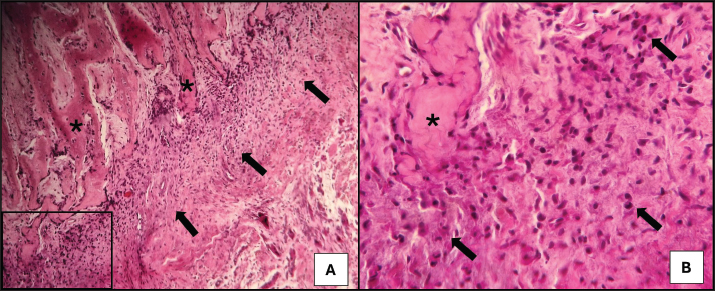
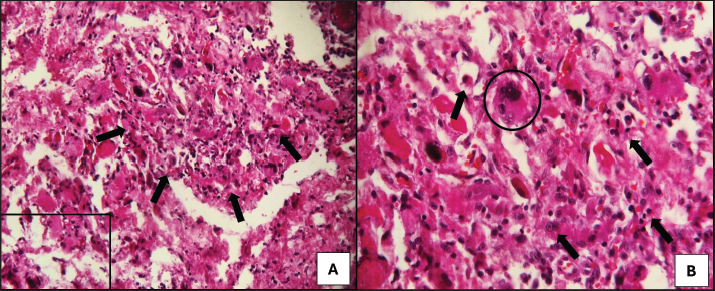
Fig. 1. Radiographic examination of the dog showing periosteal bone proliferation and areas of osteolysis (arrows) in the right wing and body of the ilium and acetabulum, accompanied by adjacent soft tissue swelling. Fine-needle aspiration cytology (FNAC) revealed a prominent population of mesenchymal cells arranged in clusters supported by connective tissue, as well as scattered cells, with moderate anisocytosis and anisokaryosis, mild karyomegaly, and variable cytoplasm (from scant to abundant, fusiform to round, basophilic, and occasionally vacuolated). Nuclei were eccentric to central, round to oval, with loose chromatin and either single or multiple prominent nucleoli. A dense neutrophilic inflammatory infiltrate with intracellular bacteria within macrophages or scattered in the background was also observed. The cytological findings were compatible with suppurative osteomyelitis, although osteosarcoma could not be definitively excluded. Owing to the inability to differentiate lesions based solely on radiographic and cytological findings, an incisional biopsy of the right wing of the ilium was performed under general inhalation anesthesia. Macroscopically marked osteophytic proliferation was observed extending to the acetabulum, with fibrous adhesion of the adjacent musculature. The bone and muscle fragments were submitted for histopathological analysis. Sterile swabs from the surgical field were collected for bacterial and fungal cultures, along with small bone fragments. Postoperative medications included dipyrone (25 mg/kg PO q12 hours for 7 days), tramadol hydrochloride (5 mg/kg PO q12 hours for 7 days), meloxicam (0.1 mg/kg PO q24 hours for 5 days), and cephalexin (30 mg/kg PO q12 hours for 10 days). Thirteen fragments (nine bone, four muscle) were submitted for histopathology. Bone fragment microscopy revealed multiple foci of neutrophilic inflammatory infiltrate and cellular debris (Fig. 2). Muscle tissue showed multifocal dense inflammatory infiltrates composed of macrophages, lymphocytes, plasma cells, neutrophils, and multinucleated giant cells (Fig. 3), with necrotic muscle fibers and cellular debris. No neoplastic cells were identified. The findings were compatible with chronic active osteitis and myositis.
Fig. 2. Photomicrograph of a dog with chronic active osteitis. (A) Fibroblastic reaction with neutrophilic infiltrate (arrows) at the osteoid matrix periphery (*). Objective 10×, H&E. (B) Higher magnification (40×) showing neutrophils (arrows) interspersed among reactive fibroblasts, permeating the osteoid matrix (*). H&E.
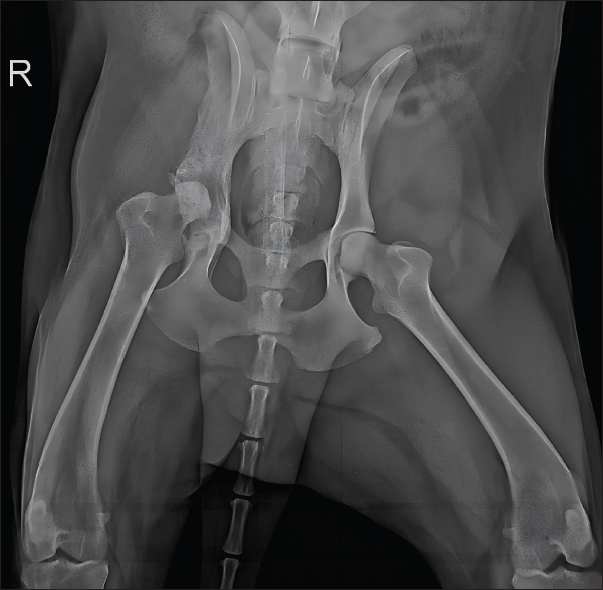
Fig. 3. Photomicrograph of a dog with chronic active myositis. (A) Intense inflammatory infiltrate (arrows) among muscle fibers. Objective 10×, H&E. (B) Higher magnification (40×) showing an infiltrate composed of a mixture of macrophages, neutrophils, and plasma cells (arrows), along with a multinucleated giant cell (circled). H&E. Microbiological culture showed no bacterial growth after 48 hours, but fungal culture yielded Aspergillus spp. colonies after 30 days. Species-level identification was not performed. Based on these results, a diagnosis of fungal osteomyelitis secondary to aspergillosis was established. Thoracic radiographs and abdominal ultrasound findings were unremarkable, ruling out systemic involvement. Treatment, based on the current literature, consisted of itraconazole (10 mg/kg PO q24 hours for 30 days). Although bacterial culture was negative, prophylactic cephalexin (25 mg/kg PO q12 hours for 30 days) was administered concurrently. Chronic osteoarticular pain was managed with gabapentin (10 mg/kg PO q12 hours for 30 days). The dog showed significant clinical improvement, including reduced lameness and restored appetite, after 30 days of therapy. Pelvic radiographs revealed slight regression of periosteal proliferation and osteolytic areas (Fig. 4). Hematology and serum biochemistry remained within normal limits.
Fig. 4. Radiographic examination of the dog 30 days after initiation of itraconazole treatment, showing slight regression of periosteal bone proliferation and reduction of osteolytic areas. Based on clinical and radiographic improvement, itraconazole and gabapentin were continued for an additional 30 days, while cephalexin was discontinued to minimize potential adverse effects. After 60 days of treatment, follow-up radiographs demonstrated continued gradual reduction in osteolysis (Fig. 5). Therapy was extended for an additional 3 months, with ongoing clinical improvement.
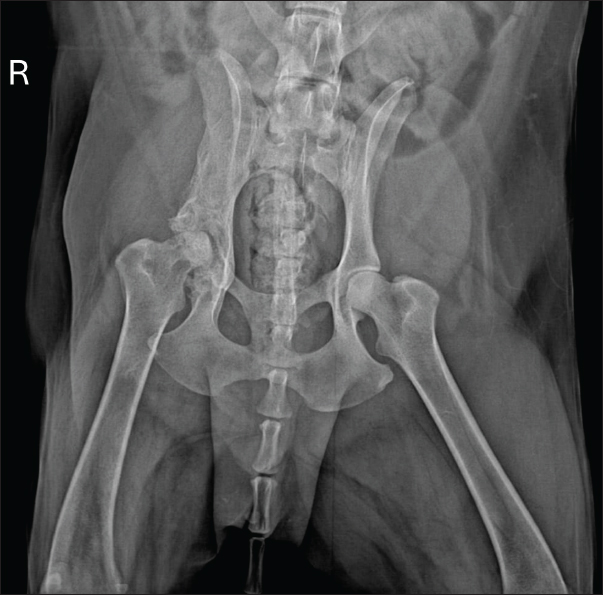
Fig. 5. Radiographic examination of the dog 60 days after initiation of itraconazole treatment, showing ongoing, albeit slight, reduction of osteolytic areas. At 150 days, reevaluation included radiography and hematology, which remained within normal limits, including hepatic and renal parameters. Radiographs showed marked reduction of periosteal proliferation and osteolytic areas, primarily concentrated in the acetabular region, likely due to joint degeneration from the infection (Fig. 6).
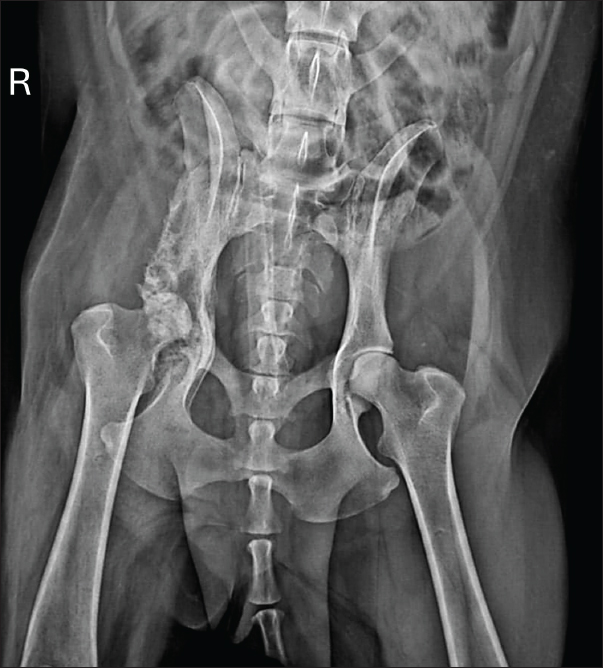
Fig. 6. Radiographic examination of the dog 150 days after initiation of itraconazole treatment, showing marked reduction of periosteal proliferation and osteolytic areas. The patient was reexamined 16 months (480 days) after treatment initiation. The owners reported the patient to be in good general condition, with normal appetite and no clinical alterations. Hematology remained normal. Radiographs revealed slight residual osteolysis, particularly in the acetabular region (Fig. 7). At the time of this report, the dog was on itraconazole and gabapentin with periodic renal and hepatic monitoring and has shown no progression or evidence of systemic aspergillosis.
Fig. 7. Radiographic examination of the dog 480 days after initiation of itraconazole treatment, showing a slight reduction in the osteolytic areas, particularly in the acetabular region. DiscussionSystemic mycoses, particularly in domestic animals, are still not included in the list of notifiable diseases in Brazil, and thus, epidemiological data on these infections are unavailable at a national level. Few reports of disseminated aspergillosis in dogs have been described in the country, with only two cases affecting bone structures in German Shepherd dogs in the state of Rio Grande do Sul (Galiza et al., 2014). Globally, reports of aspergillosis affecting bone tissue are primarily described in Australia (Oxenford and Middleton, 1986; Lim et al., 2022) and the United States (Zhang et al., 2012), with additional cases documented in Israel (Bruchim et al., 2006), Portugal (Abreu et al., 2023), and Italy (Del Magno et al., 2022). Only one report described fungal osteomyelitis caused by Aspergillus sp. in the wing of the ilium (Del Magno et al., 2022), and only one case presented osteomyelitis as the sole clinical sign without associated systemic alterations (Oxenford and Middleton, 1986), highlighting the uniqueness of the present case. The patient described in this report (male, early senior stage, mixed breed) differs from the most commonly affected population in disseminated aspergillosis, which generally comprises young females, with a mean age of 4–5 years, of the German Shepherd breed (Schultz et al., 2008). However, due to the unknown lineage of the patient, potential genetic predisposition from ancestral German Shepherd lineage cannot be excluded, as observed in the case reported by Del Magno et al. (2022) in which the dog was a mixed-breed female with German Shepherd ancestry. Aspergillus species are opportunistic fungi that typically infect animals with immunosuppression due to concurrent diseases (Harkin, 2003). According to the patient’s history, empirical treatment for tick-borne hemoparasitosis with doxycycline was initiated 1 month prior. Although no confirmatory diagnostic testing was performed, the possibility of an underlying infection contributing to susceptibility to aspergillosis should be considered. Nonetheless, as Schultz et al. (2008) noted, most reported cases lack documented immunosuppression, which may also apply to the present patient. The presence of nonspecific clinical signs, such as fever, anorexia, and weight loss, associated with reactivity of the lymph node of the affected limb and the hematological abnormalities—neutrophilia and hyperglobulinemia—suggests systemic alterations of infectious origin, consistent with disseminated aspergillosis (Kelly et al., 1995; Day, 1998; Schultz et al., 2008). Based on the clinical history, hematologic evaluation, and imaging examinations (radiography and ultrasonography), the absence of other signs indicative of involvement of additional organs, such as uveitis, ataxia, renal insufficiency, and respiratory alterations, strongly suggests that the infection site in the pelvic bones was localized. Radiographic findings, including periosteal bone proliferation, osteolysis, and soft tissue swelling in the ilium wing, acetabulum, and femur, align with observations reported by Lima et al. (2013) in aspergillosis-associated osteomyelitis. Primary infection of the ilium wing has only been reported once (Del Magno et al., 2022) in patients with disseminated aspergillosis. Pelvic bone involvement in fungal infections is rare in both animals and humans (Schultz et al., 2008; Koehler et al., 2014), with discospondylitis being the most commonly reported skeletal lesion in dogs, followed by osteomyelitis of long bones such as the humerus, tibia, femur, and scapula. Acetabular involvement has never been documented in dogs. The infection route in this case remains to be elucidated. The likelihood of entry via common pathways, including inhalation of spores, ingestion of fungal elements, or direct inoculation through penetrating trauma, open fracture, or surgical procedures (Johnson et al., 1997; Bubenik and Smith, 2007; Gieling et al., 2019), raises doubts regarding the primary site of infection. This is corroborated by the absence of abnormalities in other commonly affected organs (such as the lungs, kidneys, eyes, spleen, and brain) in cases of hematogenous dissemination, as well as by the lack of any history of trauma, fracture, or previous surgery in the affected region. The most plausible hypotheses include inhalation or ingestion of fungal elements with subclinical respiratory or gastrointestinal involvement, followed by hematogenous dissemination to the pelvic bones, or direct inoculation via a previously healed wound. Definitive diagnosis was achieved exclusively through fungal culture, as no fungal structures were observed in cytology or histopathology, consistent with previous reports emphasizing that aspergillosis diagnosis is challenging and usually requires multiple diagnostic methods (Day, 1998; Harkin, 2003). Albeit difficulties in the culture and isolation of the pathogen, recent studies indicate that advances in the molecular diagnostic settings—such as the use of LAMP machine learning, algorithmic overlap extension PCR, and other and innovative PCR simulation methods—could be applied with relative ease in the currently challenging settings (Camer et al., 2018; Kayama et al., 2020; Endoh et al., 2024). Histopathological findings, characterized by neutrophil-predominant infiltrates with macrophages and multinucleated giant cells, corroborate previous descriptions in canine aspergillosis (Day, 1998). Although histopathology alone was insufficient for a definitive diagnosis, primary or metastatic bone neoplasms, multiple myeloma, bone lymphoma, bone abscesses, and metabolic bone diseases, which constitute important differentials for the observed lesions, should be ruled out (Daleck et al., 2002; Dernell et al., 2007). Putro et al. (2024) reported the use of bone tumor markers for the identification of primary and metastatic bone neoplasms in humans, which could support differential diagnosis in companion animals in the future. Treatment with itraconazole, the most commonly used antifungal for this condition, resulted in significant clinical improvement (Abreu et al., 2023). Gradual but notable reduction of periosteal proliferation was observed radiographically, although complete remission was not achieved, consistent with the literature indicating that treatment is prolonged and challenging due to poor antifungal penetration into fungal colonies and necrotic tissues, as well as inherent resistance in some Aspergillus species (Kelly et al., 1995; Sutton et al., 1999; Zonderland et al., 2002; Sanches and Coutinho, 2007). According to Kelly et al. (1995), itraconazole therapy may last up to 3 years, potentially eradicating infection or prolonging patient survival. Discontinuation of prophylactic cephalexin treatment after 30 days of use was primarily intended to minimize associated adverse effects, including gastrointestinal disturbances, drug-induced dermatologic reactions, and nephrotoxicity (Rang et al., 2016). The survival time of approximately 480 days observed in this patient differs markedly from that reported by Lim et al. (2022), who observed a mean survival of 63 days in dogs treated with itraconazole monotherapy compared with 830 days in dogs receiving combination therapy. This discrepancy may be related to differences in disease stage and Aspergillus species, which exhibit variable antifungal susceptibility. The predominantly localized manifestation of the disease may also be involved in the observed longer life expectancy compared with other studies. ConclusionBased on the case described, disseminated aspergillosis may present in dogs as a localized infection affecting only bone structures, resulting in severe osteomyelitis, although rare. Clinical and radiographic signs resemble other bone lesions, such as neoplasms, abscesses, and metabolic bone diseases, making diagnostic methods such as cytology, histopathology, and microbiological culture crucial for differentiation. Moreover, more than 1 diagnostic method may be necessary to establish a definitive diagnosis due to the challenging identification of Aspergillus species. Itraconazole is one of the most commonly used antifungals in veterinary medicine, and although its isolated efficacy is debated—particularly because of limited penetration into fungal colonies and bone tissue—marked clinical improvement was observed in the present patient, as monitored through clinical and radiographic follow-up, in combination with neuropathic analgesia. Although the prognosis for disseminated aspergillosis is generally poor, the observed survival time of 480 days in this case exceeds that reported in previous studies. The scarcity of disseminated aspergillosis case reports in Brazil highlights the need for additional studies and data on this disease. As previously noted, this report is among the few describing isolated fungal osteomyelitis caused by Aspergillus sp. without involvement of other organs and is only the second to describe pelvic bone involvement, emphasizing the importance of thorough diagnostic investigation in cases with compatible lesions. Epidemiological studies focusing on geographic distribution and pathogenesis are essential for a better understanding of the true impact of aspergillosis at a national level. With early diagnosis and further therapeutic studies, the quality of life of affected animals can be more effectively ensured. AcknowledgmentsWe thank the entire team at the Veterinary Hospital, who directly or indirectly contributed to the patient’s diagnosis and therapeutic management. We also extend our gratitude to the owners for trusting our work and for their commitment and dedication, which were essential to achieving a satisfactory therapeutic outcome and ensuring the quality of life of the patient. FundingThis case report received no specific grant. Author’s contributionsJDRJ: Conceptualization, Data curation, Investigation, Methodology, Writing – original draft, Writing – review & editing. LFM: Data curation, Investigation, Methodology. IBOS: Data curation, Investigation. LCML: Data curation, Investigation. JPGL: Data curation, Investigation. TMF: Data curation, Investigation. IPS: Data curation, Investigation. AWOA: Data curation, Investigation, Methodology. JRR: Conceptualization, Data curation, Investigation, Methodology, Supervision. FGGD: Conceptualization, Project administration, Resources, Supervision, Writing – original draft, Writing – review & editing. All authors have read and approved the published version of the manuscript. Conflict of interestThe authors declare that there is no conflict of interest. Data availabilityAll data supporting this study’s findings are available within the manuscript. ReferencesAbreu, R., Martinho, A., Noiva, R., Pissarra, H., Cota, J., Cunha, E., Tavares, L. and Oliveira, M. 2023. Osteomyelitis caused by Aspergillus terreus complex in a dog: a case report. BMC. Vet. Res. 19(1), 1. Bruchim, Y., Elad, D. and Klainbart, S. 2006. Disseminated aspergillosis in two dogs in Israel. Mycoses 49(2), 130–133. Bubenik, L.J. and Smith, M.M. 2007. Infecções ortopédicas. In Manual de cirurgia de pequenos animais. Slatter, D. 3rd ed., Manole, São Paulo, Brazil, pp: 1862–1875. Camer, G.A., Oikawa, Y., Omaki, H. and Endoh, D. 2018. Novel synthetic nucleotides of notifiable Dengue (1–4), Japanese encephalitis, yellow fever and Zika flaviviruses. Future Sci. OA. 5(1). Daleck, C.R., Fonseca, C.S. and Canola, J.L. 2002. Osteossarcoma canino – revisão. Rev. Educ. Contin. CRMV-SP. 5(3), 233–242. Day, M.J. 1998. Canine disseminated aspergillosis. In Infectious diseases of the dog and the cat. Greene, C.E. 2nd ed., Saunders, Philadelphia, PA, USA, pp: 409–412. Day, M.J., Penhale, W.J., Eger, C.E., Shaw, S.E., Kabay, M.J., Robinson, W.F., Huxtable, C.R., Mills, J.N. and Wyburn, R.S. 1986. Disseminated aspergillosis in dogs. Aust. Vet. J. 63(2), 55–59. Del Magno, S., Gruarin, M., Foglia, A., Cola, V., Agnoli, C., Galuppi, R., Dondi, F. and Pisoni, L. 2022. Disseminated aspergillosis in a German shepherd mixed breed dog with unusual initial localization to the iliac wing. Vet. Ital. 58(3), 353–360. Dernell, W.S., Ehrhart, N.P., Straw, R.C. and Vail, D.M. 2007. Tumors of the skeletal system. In Small animal oncology. Withrow, S.J. and MacEwen, E.G. 4th ed., Elsevier, St. Louis, MO, USA, pp: 540–561. Endoh, T., Sanekata, Y., Kayama, K., Endoh, D. and Camer, G. 2024. Development of machine learning algorithm for loop-mediated isothermal amplification including influence of temperature. SciEngg. J. 17(2), 202–244. Fossum, T.W. and Hulse, D.A. 1992. Osteomyelitis. Seminar Vet. Med. Surg. 7, 85–91. Galiza, G.J.N., Silva, T.M.D., Caprioli, R.A., Barros, C.S.L., Irigoyen, L.F., Fighera, R.A., Lovato, M. and Kommers, G.D. 2014. Ocorrência de micoses e pitiose em animais domésticos: 230 casos. Pesq. Vet. Bras. 34(3), 224–232. Gieling, F., Peters, S., Erichsen, C., Richards, R.G., Zeiter, S. and Moriarty, T.F. 2019. Bacterial osteomyelitis in veterinary orthopaedics: pathophysiology, clinical presentation and advances in treatment across multiple species. Vet. J. 250, 44–54. González-Martín, M., Silva, V., Poeta, P., Corbera, J.A. and Tejedor-Junco, M.T. 2022. Microbiological aspects of osteomyelitis in veterinary medicine: drawing parallels to the infection in human medicine. Vet. Q. 42(1), 1–11. Hakamata, M., Kano, R., Kondo, H. and Watari, T. 2019. Canine fungal osteomyelitis. Mycopathologia 184(5), 707–708. Harkin, K.R. 2003. Aspergillosis: an overview in dogs and cats. Vet. Med. 98(7), 602–618. Johnson, K.A. 1994. Osteomyelitis in dogs and cats. J. Am. Vet. Med. Assoc. 204(12), 1882–1887. Johnson, K.A., Watson, A.D.J. and Page, R.L. 1997. Afecções esqueléticas. In Tratado de medicina interna veterinária – moléstias do cão e do gato. Ettinger, S.J. and Feldman, E.C. 4th ed., Manole, São Paulo, Brazil, pp: 2867–2900. Kayama, K., Hashizume, H., Camer, G.A. and Endoh, D. 2020. An improved gene synthesis method with asymmetric directions of oligonucleotides designed using a simulation program. BioTechniques 69(3), 211–219. Kelly, S., Shaw, S. and Clark, W. 1995. Long-term survival of four dogs with disseminated Aspergillus terreus infection treated with itraconazole. Aust. Vet. J. 72(8), 311–313. Koehler, P., Tacke, D. and Cornely, O.A. 2014. Aspergillosis of bones and joints – a review from 2002 until today. Mycoses 57, 323–335. Lim, Y.Y., Mansfield, C., Stevenson, M., Thompson, M., Davies, D., Whitney, J., James, F., Tebb, A., Fry, D., Buob, S., Hambrook, L., Boo, G. and Dandrieux, J.R.S. 2022. A retrospective multi-center study of treatment, outcome, and prognostic factors in 34 dogs with disseminated aspergillosis in Australia. J. Vet. Intern. Med. 36(2), 580–590. Lima, T.B., Leal, L.M., Morato, G.D.O., Marinho, P.V.T., Moares, P.C. and Minto, B.W. 2013. Osteomielite fúngica em fratura de tíbia de cão: relato de caso. Rev. Bras. Ciênc. Vet. 20(3), 132–136. Mello, S.S., Vasconcelos, L.M.A., Milken, V.M.F., Mota, F.C.D. and Medeiros-Ronchi, A.A. 2024. Osteomielite fúngica causada por Penicillium sp. e Acremonium sp. em um cão. Med. Vet. 18(1), 15–21. Oxenford, C.J. and Middleton, D.J. 1986. Osteomyelitis and arthritis associated with Aspergillus fumigatus in a dog. Aust. Vet. J. 63(2), 59–60. Pape, S. and Kirchhoff, A. 1996. Aspergillosis in a German Shepherd dog. Kleintierpraxis 41(5), 380. Piermattei, D.L., Flo, G.L. and Decamp, C.E. 2009. Tratamento das infecções ósseas agudas e crônicas. In Ortopedia e tratamento de fraturas de pequenos animais. Piermattei, D.L., Flo, G.L. & Decamp, C.E. (eds.), 4th ed., Manole, São Paulo, Brazil, pp: 200–208. Putro, Y.A.P., Aryandono, T., Widodo, I., Magetsari, R., Pramono, D., Johan, M.P., Abidin, M.A., Wikantyasa, A., Saraswati, P.A. and Huwaidi, A.F. 2024. Diagnostic approach to bone metastasis of unknown origin: a systematic review. Orthop. Rev. 16, 123441. Rang, H.P., Ritter, J.M., Flower, R.J. and Henderson, G. 2016. Rang & Dale Pharmacology. 8th ed., Elsevier, Edinburgh, UK, pp: 1482–1490. Sanches, P.P. and Coutinho, S.D.A. 2007. Aspergilose em cães – revisão. Rev. Inst. Ciênc. Saúde 25(4), 391–397. Schulz, K.S. 2013. Other diseases of bones and joints. In Small animal surgery. Fossum, T.W. 3rd ed., Elsevier, St. Louis, MO, USA, pp: 1391–1410. Schulz, K.S. 2013. Other diseases of bones and joints. In Small animal surgery. Fossum, T.W. 3rd, Elsevier, 36, pp: 1391–410. Seyedmousavi, S., Guillot, J., Arné, P., De Hoog, G.S., Mouton, J.W., Melchers, W.J.G. and Verweij, P.E. 2015. Aspergillus and aspergilloses in wild and domestic animals: a global health concern with parallels to human disease. Med. Mycol. 53, 765–797. Sharp, N.J.H. 1998. Canine nasal aspergillosis – penicilliosis. In Infectious diseases of the dog and the cat. Greene, C.E. 2nd ed., Saunders, Philadelphia, PA, USA, pp: 404–409. Smith, S.A., Andrews, G. and Biller, D.S. 1998. Management of nasal aspergillosis in a dog with a single, noninvasive intranasal infusion of clotrimazole. J. Am. Anim. Hosp. Assoc. 34(6), 487–492. Sutton, D.A., Sanche, S.E., Revankar, S.G., Fothergill, A.W. and Rinaldi, M.G. 1999. In vitro amphotericin B resistance in clinical isolates of Aspergillus terreus with a head-to-head comparison to voriconazole. J. Clin. Microbiol. 37(7), 2343–2345. Tell, L.A. 2005. Aspergillosis in mammals and birds: impact in veterinary medicine. Med. Mycol. 43(1), 71–73. Thrall, D.E. 2010. Radiographic features of bone tumors and bone infections. In Diagnostic veterinary radiology. Thrall, D.E. 5th ed., Elsevier, St. Louis, MO, pp: 390–402 Walsh, T.J., Anaissie, E.J., Denning, D.W., Herbrecht, R., Kontoyiannis, D.P., Marr, K.A., Morrison, V.A., Segal, B.H., Steinbach, W.J., Stevens, D.A., Van Burik, J.A., Wingard, J.R. and Patterson, T.F. 2008. Treatment of aspergillosis: clinical practice guidelines of the infectious diseases Society of America. Clin. Infect. Dis. 46(3), 327–360. Wengenack, N.L. and Binnicker, M.J. 2009. Fungal molecular diagnostics. Clin. Chest Med. 30, 391–408. Zhang, S., Corapi, W., Quist, E., Griffin, S. and Zhang, M. 2012. Aspergillus versicolor, a new causative agent of canine disseminated aspergillosis. J. Clin. Microbiol. 50(1), 187–191. Zonderland, J.L., Stork, C.K., Saunders, J.H., Hamaide, A.J., Balligand, M.H. and Clerex, C.M. 2002. Intranasal infusion of enilconazole for treatment of sinonasal aspergillosis in dogs. J. Am. Vet. Med. Assoc. 221(10), 1421–1425. | ||
| How to Cite this Article |
| Pubmed Style Júnior JDR, Mangolin LF, Silva IBOD, Lopes LCDM, Luvisoto JPG, Ferreira TM, Sabino IP, Alvarenga AWO, Rocha JR, Dias FGG. Localized fungal osteomyelitis caused by Aspergillus sp. in the ilium and acetabulum of a dog with disseminated aspergillosis: A case report. Open Vet. J.. 2026; 16(2): 1369-1379. doi:10.5455/OVJ.2026.v16.i2.53 Web Style Júnior JDR, Mangolin LF, Silva IBOD, Lopes LCDM, Luvisoto JPG, Ferreira TM, Sabino IP, Alvarenga AWO, Rocha JR, Dias FGG. Localized fungal osteomyelitis caused by Aspergillus sp. in the ilium and acetabulum of a dog with disseminated aspergillosis: A case report. https://www.openveterinaryjournal.com/?mno=289713 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.53 AMA (American Medical Association) Style Júnior JDR, Mangolin LF, Silva IBOD, Lopes LCDM, Luvisoto JPG, Ferreira TM, Sabino IP, Alvarenga AWO, Rocha JR, Dias FGG. Localized fungal osteomyelitis caused by Aspergillus sp. in the ilium and acetabulum of a dog with disseminated aspergillosis: A case report. Open Vet. J.. 2026; 16(2): 1369-1379. doi:10.5455/OVJ.2026.v16.i2.53 Vancouver/ICMJE Style Júnior JDR, Mangolin LF, Silva IBOD, Lopes LCDM, Luvisoto JPG, Ferreira TM, Sabino IP, Alvarenga AWO, Rocha JR, Dias FGG. Localized fungal osteomyelitis caused by Aspergillus sp. in the ilium and acetabulum of a dog with disseminated aspergillosis: A case report. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 1369-1379. doi:10.5455/OVJ.2026.v16.i2.53 Harvard Style Júnior, J. D. R., Mangolin, . L. F., Silva, . I. B. O. D., Lopes, . L. C. D. M., Luvisoto, . J. P. G., Ferreira, . T. M., Sabino, . I. P., Alvarenga, . A. W. O., Rocha, . J. R. & Dias, . F. G. G. (2026) Localized fungal osteomyelitis caused by Aspergillus sp. in the ilium and acetabulum of a dog with disseminated aspergillosis: A case report. Open Vet. J., 16 (2), 1369-1379. doi:10.5455/OVJ.2026.v16.i2.53 Turabian Style Júnior, João Domingos Rocha, Letícia Fornel Mangolin, Igor Bruno Oliveira Da Silva, Lara Cristina De Melo Lopes, João Pedro Garcia Luvisoto, Thaylla Maria Ferreira, Isadora Pezati Sabino, Alef Winter Oliveira Alvarenga, Jessé Ribeiro Rocha, and Fernanda Gosuen Goncalves Dias. 2026. Localized fungal osteomyelitis caused by Aspergillus sp. in the ilium and acetabulum of a dog with disseminated aspergillosis: A case report. Open Veterinary Journal, 16 (2), 1369-1379. doi:10.5455/OVJ.2026.v16.i2.53 Chicago Style Júnior, João Domingos Rocha, Letícia Fornel Mangolin, Igor Bruno Oliveira Da Silva, Lara Cristina De Melo Lopes, João Pedro Garcia Luvisoto, Thaylla Maria Ferreira, Isadora Pezati Sabino, Alef Winter Oliveira Alvarenga, Jessé Ribeiro Rocha, and Fernanda Gosuen Goncalves Dias. "Localized fungal osteomyelitis caused by Aspergillus sp. in the ilium and acetabulum of a dog with disseminated aspergillosis: A case report." Open Veterinary Journal 16 (2026), 1369-1379. doi:10.5455/OVJ.2026.v16.i2.53 MLA (The Modern Language Association) Style Júnior, João Domingos Rocha, Letícia Fornel Mangolin, Igor Bruno Oliveira Da Silva, Lara Cristina De Melo Lopes, João Pedro Garcia Luvisoto, Thaylla Maria Ferreira, Isadora Pezati Sabino, Alef Winter Oliveira Alvarenga, Jessé Ribeiro Rocha, and Fernanda Gosuen Goncalves Dias. "Localized fungal osteomyelitis caused by Aspergillus sp. in the ilium and acetabulum of a dog with disseminated aspergillosis: A case report." Open Veterinary Journal 16.2 (2026), 1369-1379. Print. doi:10.5455/OVJ.2026.v16.i2.53 APA (American Psychological Association) Style Júnior, J. D. R., Mangolin, . L. F., Silva, . I. B. O. D., Lopes, . L. C. D. M., Luvisoto, . J. P. G., Ferreira, . T. M., Sabino, . I. P., Alvarenga, . A. W. O., Rocha, . J. R. & Dias, . F. G. G. (2026) Localized fungal osteomyelitis caused by Aspergillus sp. in the ilium and acetabulum of a dog with disseminated aspergillosis: A case report. Open Veterinary Journal, 16 (2), 1369-1379. doi:10.5455/OVJ.2026.v16.i2.53 |