| Research Article | ||
Open Vet. J.. 2026; 16(2): 870-876 Open Veterinary Journal, (2026), Vol. 16(2): 870-876 Research Article Role of the small intestinal submucosa-extracellular matrix grafts powder on regeneration of full-thickness wound in skin of rabbitsAbd-Alhadi Jaithom Marzok1, Methaq Faisal Yousf1, Mushtaq A. Alabodi1 and Murtadha abbas2*1Department of Surgery and Obstetrics, College of Veterinary Medicine, University of Kufa, Kufa, Iraq 2Department of Public Health, Faculty of Veterinary Medicine, University of Kufa, Kufa, Iraq *Corresponding Author: Murtadha abbas. Department of Public Health, Faculty of Veterinary Medicine, University of Kufa, Kufa, Iraq. Email: murtadhaa.alghazali [at] uokufa.edu.iq Submitted: 06/10/2025 Revised: 11/01/2026 Accepted: 25/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
AbstractBackground: Wound healing continues to pose a considerable clinical challenge, especially in the context of surgical intervention, despite numerous treatment options; an ideal solution for wound injuries has yet to be established. Small intestinal submucosa-extracellular matrix (SIS-ECM) has shown potential in regenerative medicine due to its properties that may facilitate tissue repair and wound healing. Aim: This study aims to assess the efficacy of SIS-ECM powder in promoting the healing of full-thickness wounds in the rabbit model, with particular emphasis on focusing on epithelialization rate and inflammatory response reduction. Methods: Twenty clinically healthy adult male rabbits were allocated into two experimental groups. A full-thickness excisional wounds measuring 2 × 2 cm was surgically induced on the dorsal skin of each animal. Group A (control group) received phosphate-buffered saline, whereas Group B (treatment group) was administered SIS-ECM powder. Histopathological evaluation was conducted via biopsy on days 7th, 14th, and 35th post wounding to monitor healing progression. Results: Topical application SIS-ECM powder-significantly accelerated epithelialization by day 21st of treatment (p < 0.05) and markedly reduced inflammation within the week signs within the first week (p < 0. 01). Compared to the control group These statistically significant findings suggest that SIS-ECM powder fosters a supportive microenvironment that conductive to cellular proliferation, and differentiation, thereby enhancing the repair of full-thickness wounds. Conclusion: SIS-ECM powder creates a favorable microenviroment that supports cell growth and differentiation, thereby enhancing the healing process of full-thickness wounds. Keywords: Healing, Intestine, Rabbit, SIS-ECM powder, Wounds. IntroductionThe need to treat difficult wounds has led to more studies to discover substances and drugs that help overcome this problem. It has become important to pay attention to wound cleanliness and care, using several therapeutic methods to recover wound tissue (Badylak et al., 2002). The management of wound healing aims to expedite repair while alleviating pain, distress, and minimizing scar formation (Brown et al., 2010). A suitable physiological environment can aid in the repair, regeneration, and renewal of tissues. The phenomenon by which substances impact cellular behavior during the healing process is known as biomodulation (Callanan et al., 2011). Xenografts originating from collagen are of particular significance in wound healing, primarily attributed to their therapeutic impact on wounds, especially those with elevated inflammation (Cao et al., 2019), such as natural biomaterials of the urinary bladder matrix (UBM), equine pericardium (EP), and small intestinal submucosa (SIS), which have been proposed for tissue repair procedures (Freytes et al., 2004; Field, 2013; Cao et al., 2019). Extracellular matrix (ECM) proteins may be used as scaffolds (natural biomaterials) for cell attachment, migration, and proliferation (Guo et al., 2016). The SIS has a vital role in enabling natural attachment and movement of host cells, promoting biocompatibility and tissue adherence to improve function (Hodde et al., 1996; Ji et al., 2019). The efficacy and fate of decellularized SIS-ECM for the acceleration and reconstruction of rabbit skin defects were examined in this study. Materials and MethodsExperimental animalsThe present investigation adhered to the ethical principles outlined by the Animal Experimentation Ethics Committee of the Faculty of Veterinary Medicine, University of Kufa. Twenty healthy adult male rabbits of local breed, weighing between 1 and 2 kg, were selected for this study. The study was conducted on twenty clinically healthy male rabbits. The animals were kept in metal cages (30 × 70 × 60 cm) in the animal house with healthy conditions of temperature and humidity in the College of Veterinary Medicine, University of Kufa. Water and food (pellets) were freely available to them. Animals were treated with oral anticoccidial drug (Amprolium 50% - Kypro - Netherlands) for four weeks to adjust to the experimental environment. The animals were divided into two groups of ten rabbits each. Group A (control group) were administered with phosphate buffer saline. Group B (treatment group) treated with 0.08 mg SIS-ECM powder (Vince et al., 2020). Fabrication of powder derived from SISThe fabrication of the SIS-extracellular matrix (SIS-ECM) was depicted in recent studies (Koob et al., 2014). Following slaughter, the small intestine in cattle was obtained of Kufa abattoir. The small intestine transported to the laboratory in medium of phosphate buffered saline (PBS). the tissues were washed with PBS to remove blood and other debris, excluding most of the mucosa and muscularis externa by gentle rubbing. Then the remnants of the subcutaneous mucosa and the basal part of the mucosa of the tunica were cleaned for 15 minutes with PBS and deionized disteld water after being disinfected and decellularized for 2 hours in a mixture of 4% ethanol and 0.1% peracetic acid (PAA). After SIS-ECM was decellularized, associated tissue matrices were kept in PBS with 1% Gentamycin at 4°C. Finally, tissue fragments were lyophilized for 24 hours (Bulk Freeze Dryer Model). Powdered (Rotary Cutting Mills) and frozen (FTS Systems Bulk Freeze Dryer Model 8–54). The powdered SIS-ECM was sterilized in ethylene oxide overnight at 37°C prior to use for the in vivo application (Gessner et al., 2010)). In vivo study on the injuryThe animal was sedated with diazepam 1 mg/kg, followed by intramuscular injection of a mixture of xylazine hydrochloride (5 mg/kg) and ketamine hydrochloride (35 mg/kg). After that the skin sterilized by 10% povidone iodine solution for aseptic surgical procedure. Two incisions were created (2 × 2 cm) on the same side of the dorsal area. (crainal and caudal), with a distance of 5–7 cm between each wound. The caudal wound was treated with SIS-ECM powder, while the cranial wound left untreated (control). The animals were divided into two groups of ten rabbits each. Group A (control group) were administered with phosphate buffer saline. and Group B (treatment group) treated with 0.08 mg SIS-ECM powder (Fig. 1). All of the animals underwent a complete clinical examination, including measuring the body temperature, respiratory activity and behaviour. Throughout the entirety of the experiment, a daily gross checking for the wounds was performed. The animals were given penicillin (10,000 IU/kg.B.W) and streptomycin (10 mg/kg B.W) daily intramuscular injection till day five after the implant.
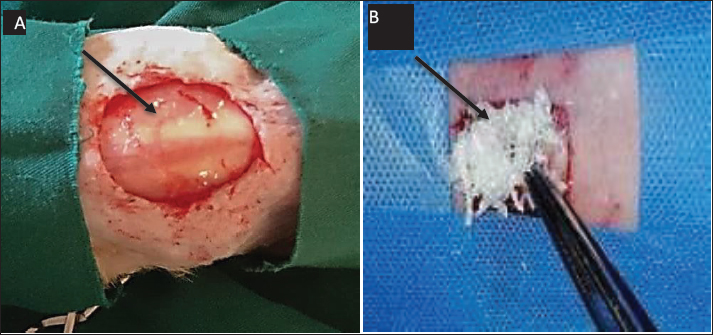
Fig. 1. Represents the Created (2 × 2 cm) on the dorsal area (A) the wound left untreated (as control group). While, the wound was treated with SIS-ECM powder (as treatment group). Histopathological evaluationRabbits were euthanized with ether on days 7, 14, and 35 post-injury. Biopsies of full-thickness incisional samples (5–6 mm) and adjacent normal skin (3–4 mm) were fixed in 10% formalin, dehydrated, cleared, and embedded in paraffin. Transverse sections (5 μm) were stained with hematoxylin and eosin (H&E) (Liu et al., 2011). Wound contraction measurementFor assessment of wound healing rate wound area were measurement daily from day 0 to 35. A standardized digital imaging was employed. Each wound was photographic daily by camera mounted. A calibration ruler was placed adjacent to the wound in each image, the wound perimeter was traced manually, and wound area was calculated in cm2 (Rao et al., 2007). The wound contraction percentage for each wound on day (7,14,21,35) was calculated by following formula % wound contraction=initial wound area-wound area on day n /initial wound area x 100 Statistical analysis of data was performed using t-test to compare between the two group with a p value (p < 0.05) considered statistically significant. Ethical approvalThis study was conducted in conformity with the rules of the Animal Ethics Committee of the Faculty of Veterinary Medicine at the University of Kufa (license number: 27865 in 13/10/2023). ResultsClinical evaluationDaily clinical observations of wounds refer to a rapid reduction in size in both treated and control groups over the study period. However, a marked improvement was observed in the treated wounds starting from day 7, demonstrating a faster clinical resolution compared to the non- treated wounds while a little significant variation in day 3 was observed . These distinct differences between the two groups persisted until day 35. The centers of the treated wounds had transitioned into mature scar tissue , while the control wounds remained visibly larger and less healed . The appearance of scar tissue in treated group commenced earlier , beginning from day 7 post-wounding, and this differentiation continued until the experiments conclusion (Fig. 2).
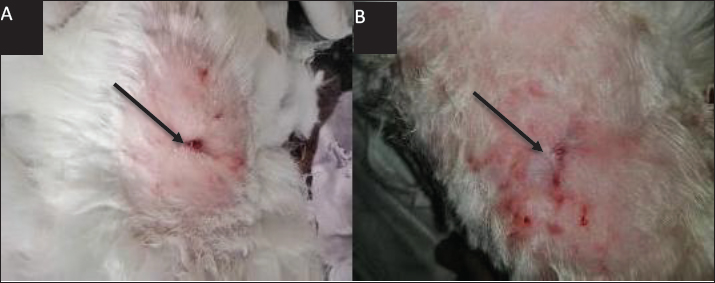
Fig. 2. Represents the macroscopic features of wounds between groups at day 35th post-operation. (A) control (B) treatment groups, showed the granulation, scar formation and healing were more rapid in rats of the treatment group, the area of wound was lower in rats of the treatment group (black arrow). Histopathological evaluationThe histopathological assessment revealed fundamental between the groups which became distinctly evident by day 7 post-wounding on day 3 demonstrated, epithelialization on the inner wall of the wound margins in the between groups, well-marked in treated with SIS-ECM powder. As well as, in control group showed no epithelialization was showed and inflammatory cells infiltration (Khan et al., 2014). On day 7 Advanced epithelialization was observed on the inner wound margins in the SIS-ECM powder-treated group, with the epithelium beginning to cover the entire exposed wound edge (Fig. 3A). In contrast, the control group exhibited incomplete epithelization, with a persistent and notable infiltration of inflammatory cells (Fig. 3B).
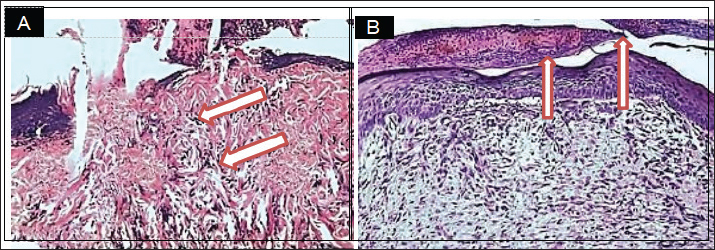
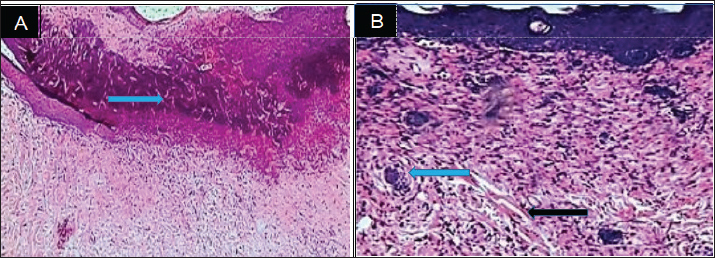
Fig. 3. Histopathological section, at 7 days PO, (A) in control group showed neutrophils and mononuclear cells infiltration in the immature granulation tissue (Red arrow). (B) in treatment group, showed granulation tissue in the dermis with complete epidermal layer (Red arrow). (H& E stain 100X). On 14 day epithelialization was complete in treated wounds, with the wound site densely filled by cohesive tissues and new collagen bundles (Fig. 4B). conversely, in control group displayed a persistence of delayed epithelial reorganization and fibrous tissue deposition (Fig. 4A). On day 35 Histological examination revealed near-complete recovery in the treated wound characterized by collagen reformation,skin keratinization, and the reappearance of hair follicles in the wound site (Fig. 5B). In contrast, the control wounds exhibited delayed healing, with advanced fibrosis and prominent scarring (Fig. 5A).
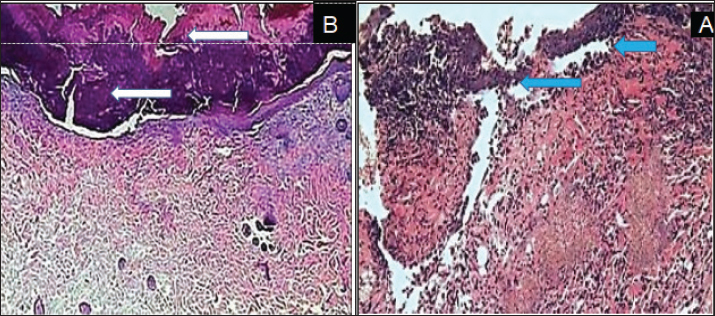
Fig. 4. Histopathological section, at 14 days PO, (A) in control group showed the reorganization of the collagen high and deposition of fibrous tissue (blue arrow). (B) in treatment group, showed thickened layer of epithelial cells extended over mature granulation tissue and under cellular debris (white arrow). (H and E X100).
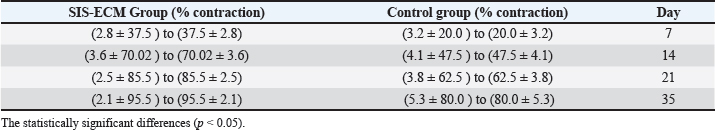
Fig. 5. Histopathological section, at 35 days PO (A) in control group showed presence of dense collagen fibers in the wound site with mononuclear cells aggregation around blood vessels, in the dermal layer (blue arrow). (B) In treatment group, showed newly formed hair follicle (blue arrow) and sebaceous gland in the injury site (black arrow). (H and E X40). Wound contraction evaluationThe measurement of wound contraction yielded data supporting the clinical and Histopathological evaluation. Wound treated with SIS-ECM powder exhibited a significantly higher percentage of wound Contraction (p < 0.05) at day (7, 14, 21, and 35) compared to the control group. In treated group the contraction percentage reached 95% ± 2.1% by day 35 while in control group did not exceed 80% ± 5.3% in same period. The rapid contraction rate in treated group occurred during first two weeks, that indicating a critical acceleration in the early phase of the healing process (Table 1) (Shi et al., 2012). Table 1. The qouantatve assessment of wound contraction provided cpmpelling evidence for the acceleratory effect of SIS-ECM powder on wound healing.
DiscussionSubmucosa of Small Intestine has been expansively employed for tissue regeneration as a totally decellularized matrix; skin replacement, vascular repair, and renewal of other soft tissues are among these applications (Luna, 1992). In the present study, it was observed that there was a swift reduction in the size of the wounds in both the treatment and control groups. However, according to histopathological section, the rate of wound closure was noticeably greater in the SIS-ECM treated wounds than in untreated. Wound healing has been evaluated by using a xenogeneic, collagen-rich membrane scaffold derived from the umbilical cord of cow. The examination of wound sections post-treatment through histopathology demonstrated a significant presence of well-developed granulation tissue, myofibroblasts, and newly formed blood vessels. Statistical analysis , performed using Students t-test with significance (p < 0.05) (Wasserstein et al., 2016). in contraction percentages at all measured time points (Table 1) demonstrate a consistent and sustained enhancement of the healing process. The most remarkable acceleration occurred during the initial two weeks, where the SIS-ECM group achieved nearly double the contraction rate of the control group by day 7 (37.5% vs. 20%). The early boost aligns with the critical inflammatory and proliferative phases of healing, suggesting that SIS-ECM actively modulates these stage rather than merely providing a passive scaffold. The progressive nature of wound closure, culminating in 95% contraction by day 35 in the treatment group compared 80% in controls (Field et al., 2013) however, the sections of wounds in the control group showed myo fibroblasts dispersed throughout fibrous connective tissue containing congested blood vessels. This finding can be connected to the impact of implanted SIS-ECM, which can contribute significantly to the acceleration and enhancement of cutaneous wound healing by helping infiltration, adhesion, as well as progenitor cell proliferation, and acceleration of formation of blood vessels at the wound site. Implantation of ECM has been demonstrated to facilitate tissue regeneration by stimulating infiltration, adhesion, and proliferation of progenitor cells, hastening angiogenesis at the wound site, augmenting formation of granulation tissue and encouraging the host-derived neomatrix to be deposited (collagen matters), thus minimizing scar tissue formation this was quantitavely confirmed by wound contraction measurement, which revealed a statistically significant (p < 0.05) (Luna et al., 1992; Luo et al., 2011). The research determined that cells engaged with regard to regenerative medicine and tissue engineering necessitate guidance for proliferation, morphogenesis, and differentiation. This guidance emanates from the microenvironment of cells and the surfaces of scaffolds or matrices which these cells interact. Rashtbar et al., 2018 and Schultz et al., 2009 mentioned that the surfaces of the scaffold or matrix that these cells contact with and the cellular microenvironment is the sources of this "instruction". SIS-ECM contained approximately 461 protein biomolecules, the majority of which were growth factors or cytokines involved in physiological procedures that involve homeostasis and tissue remodeling (Sheikh et al., 2014). It is important in the subject of both angiogenesis and cell proliferation, these include factors including platelet-derived growth factor AA and transforming growth factor beta 1 (TGF-1) (PDFG-AA) (Massee, et al., 2016). Chemokines, inflammatory modulators, and proteases and inhibitors, have also been identified (Shi et al., 2013). Serine and cysteine proteases, involved in the activation of proteins, together with matrix proteases and inhibitors including matrix metalloproteinase-1 (MMP-1) and MMP-3, as well as tissue inhibitor of metalloproteinases (TIMPs 1-2), were identified as key regulators of ECM changeover in the period of remodeling (Ramsay et al., 2012; Singh et al., 2022). Curcumin in decellularized goat small intestine submucosa for wound healing and skin tissue engineering. Journal of Biomedical Materials Research Part B: Applied Biomaterials, 110(1), 210–219]. The soluble protein analysis demonstrates that SIS-ECM is a physiologically active tissue that has a wide variety of signaling components that are able to stimulate responses that take place during tissue regeneration. Placental-derived graft, which has been shown to be effective in treating leg ulcers caused by veins and non-healing refractory wounds, also contains similar factors (Tan et al., 2009; Voytik‐Harbin et al., 1997). As a result, it is anticipated that SIS grafts may also have the therapeutic potential to promote wound closure in chronic wounds. Since SIS-ECM is a naturally occurring scaffold that encourages cellular attachment and proliferation and is both biocompatible and biodegradable, we believe that it has desirable characteristics in place of the advantages and disadvantages of normal and artificial scaffolds. When compared to autologous one, allogeneic and xenogeneic grafts have a lower threat of infection or immune rejection (Ji et al., 2019). Clinical and histopathological observations indicated that human umbilical cord (HUC) usually connected with tissue acceptance, and in spite of implant's xenogeneic character, no indications of immunological rejection were seen. The implant's composition, which primarily consisted of collagen-based biomaterial that is resorbable, non-immunogenic, and acellular, may be responsible for this outcome. The statistical significant of these histological improvement reinforce the biological relevance of our finding. The introduction of the small intestine submucosa scaffold triggers an immune response characterized by predominantly T- helper lymphocyte-2-like activity, that promotes secretion of interleukins (IL-4, IL-5, IL-6, and IL-10) (Yeum et al., 2013). In previous investigation the capacity of decellularized SIS-ECM to regenerate skin in full- thickness wounds in mice showed that the scaffolds were well-integrated into the tissue after a week. Furthermore, the scab had disappeared from the wound, and re-epithelialization process was complete after 12 days (Shi et al., 2012). In a study conducted on mouse model of full-thickness skin excisional wounds, the findings revealed paracrine signaling was responsible for the positive effects of SIS- ECM conditioned-medium on that the human in vivo wound healing: enhancing the proliferation and migration of human skin fibroblasts and up regulating the gene expression of wound healing factors like TGF-ß2, HIF-1a, and PAI-1. (Singh et al., 2022; Wasserstein et al., 2016; Field et al., 2013). ConclusionThe qualified healing process demonstrated that using of a powder scaffold promote and enhance skin wound healing by promoting cell migration, allowing fibroblasts to differentiate earlier into myofibroblasts, and allowing for earlier wound contraction. These potential findings indicate that the treatment with SIS-ECM have significant benefits in wound healing. Beside that SIS-ECM is a novel and organic three- dimensional scaffold can be used for practices in regenerative medicine and tissue engineering. AcknowledgmentsThe authors extend their sincere thanks to the Faculty of Veterinary Medicine / the University of Kufa for their unwavering support. We also intensely appreciate the contributions of the veterinarians and staff at the Veterinary Hospital Laboratory and Wasit Abattoir for their technical support and collaboration in sample collection and analysis. FundingNone. Authors' contributionsAll listed authors have made significant intellectual and academic contributions to this study. Each author has reviewed and approved the final manuscript for publication. Conflict of interestThere are no conflicts of interest numerous of the authors concerning the publishing this manuscript. Data availabilityAll the datasets formed or examined during this work are included in this publication form ReferencesBadylak, S., Kokini, K., Tullius, B., Simmons-Byrd, A. and Morff, R. 2002. Morphologic study of small intestinal submucosa as a body wall repair device. J. Surgical Res. 103(2), 190–202. Brown, B.N., Barnes, C.A., Kasick, R.T., Michel, R., Gilbert, T.W., Beer-Stolz, D., Castner, D.G., Ratner, B.D. and Badylak, S.F. 2010. Surface characterization of extracellular matrix scaffolds. Biomaterials 31(3), 428–437. Callanan, A., Davis, N.F., Walsh, M.T. and McGloughlin, T.M. 2011. Tissue-engineered extracellular matrices (ECMs) as adjuvant scaffolds for endovascular aneurysmal repair (EVAR).In Regenerative medicine and tissue engineering-cells and biomaterials. Ed., Eberli D. Rijeka, Croatia: IntechOpen. Cao, G., Huang, Y., Li, K., Fan, Y., Xie, H. and Li, X. 2019. Small intestinal submucosa: superiority, limitations and solutions, and its potential to address bottlenecks in tissue repair. J. Mater. Chem. B 7(33), 5038–5055. Field, A. 2013. Discovering statistics using IBM SPSS statistics. Freytes, D.O., Badylak, S.F., Webster, T.J., Geddes, L.A. and Rundell, A.E. 2004. Biaxial strength of multilaminated extracellular matrix scaffolds. Biomaterials 25(12), 2353–2361. Guo, X., Xia, B., Lu, X.B., Zhang, Z.J., Li, Z., Li, W.L., Xiong, A.B., Deng, L., Tan, M.Y. and Huang, Y.C. 2016. Grafting of mesenchymal stem cell-seeded small intestinal submucosa to repair the deep partial-thickness burns. Connective Tissue Res. 57(5), 388–397. Hodde, J.P., Badylak, S.F., Brightman, A.O. and Voytik-Harbin, S.L. 1996. Glycosaminoglycan content of small intestinal submucosa: a bioscaffold for tissue replacement. Tissue Eng. 2(3), 209–217. Ji, Y., Zhou, J., Sun, T., Tang, K., Xiong, Z., Ren, Z., Yao, S., Chen, K., Yang, F., Zhu, F. and Guo, X. 2019. Diverse preparation methods for small intestinal submucosa (SIS): decellularization, components, and structure. J. Biomed. Mater. Res. Part A 107(3), 689–697. Koob, T.J., Lim, J.J., Massee, M., Zabek, N., Rennert, R., Gurtner, G. and Li, W.W. 2014. Angiogenic properties of dehydrated human amnion/chorion allografts: therapeutic potential for soft tissue repair and regeneration. Vascular. Cell. 6(1), 10–22. Liu, Z., Tang, R., Zhou, Z., Song, Z., Wang, H. and Gu, Y. 2011. Comparison of two porcine-derived materials for repairing abdominal wall defects in rats. PLos One. 6(5), e2. Luna, L.G. 1992. Histopathologic methods and color atlas of special stains and tissue artifacts. Maryland: American Histolabs. Luo, J.C., Chen, W., Chen, X.H., Qin, T.W., Huang, Y.C., Xie, H.Q., Li, X.Q., Qian, Z.Y. and Yang, Z.M. 2011. A multi-step method for preparation of porcine small intestinal submucosa (SIS). Biomaterials 32(3), 706–713. Massee, M., Chinn, K., Lim, J. J., Godwin, L., Young, C. S. and Koob, T. J. 2016. Type I and II diabetic adipose-derived stem cells respond in vitro to dehydrated human amnion/chorion membrane allograft treatment by increasing proliferation, migration, and altering cytokine secretion. Adv. Wound Care 5(2), 43–54. Rao, K.S., Patil, P.A. and Malur, P.R. 2007. Promotion of cutaneous wound healing by famotidine in Wistar rats. Indian J. Med. Res. 125(2), 149–154. Rashtbar, M., Hadjati, J., Ai, J., Jahanzad, I., Azami, M., Shirian, S., Ebrahimi‐Barough, S. and Sadroddiny, E. 2018. Characterization of decellularized ovine small intestine submucosal layer as extracellular matrix‐based scaffold for tissue engineering. J. Biomed. Mater. Res. Part. B. Appl. BioMater. 106(3), 933–944. Rose, W., Wood, J.D., Simmons-Byrd, A. and Spievack, A.R. 2009. Effect of a xenogeneic urinary bladder injectable bioscaffold on lameness in dogs with osteoarthritis of the coxofemoral joint (hip): a randomized, double blinded controlled trial. Int. J. Appl. Res. Vet. Med. 7(1/2), 13–12. Schallberger, S.P., Stanley, B.J., Hauptman, J.G. and Steficek, B.A. 2008. Effect of porcine small intestinal submucosa on acute full‐thickness wounds in dogs. Vet. Surg. 37(6), 515–524. Schultz, G.S. and Wysocki, A. 2009. Interactions between extracellular matrix and growth factors in wound healing. Wound Repair Regeneration 17(2), 153–166. Sheikh, E.S., Sheikh, E.S. and Fetterolf, D.E. 2014. Use of dehydrated human amniotic membrane allografts to promote healing in patients with refractory wounds. Int. Wound J. 11(6), 711–717. Shi, L. and Ronfard, V. 2013. Biochemical and biomechanical characterization of porcine small intestinal submucosa (SIS): a mini review. Int. J. Burns. Trauma. 3(4), 173–179. Shi, L., Ramsay, S., Ermis, R. and Carson, D. 2012. In vitro and in vivo studies on matrix metalloproteinases interacting with small intestine submucosa wound matrix. Int. Wound. J. 9(1), 44–53. Singh, H., Purohit, S.D., Bhaskar, R., Yadav, I., Bhushan, S., Gupta, M.K. and Mishra, N.C. 2022. Curcumin in decellularized goat small intestine submucosa for wound healing and skin tissue engineering. J. Biomed. Mater. Res. Part B. Appl. BioMater. 110(1), 210–219. Tan, M.Y., Zhi, W., Wei, R.Q., Huang, Y.C., Zhou, K.P., Tan, B., Deng, L., Luo, J.C., Li, X.Q., Xie, H.Q. and Yang, Z.M. 2009. Repair of infarcted myocardium using mesenchymal stem cell seeded small intestinal submucosa in rabbits. Biomaterials 30(19), 3234–3240. Voytik‐Harbin, S.L., Brightman, A.O., Kraine, M.R., Waisner, B. and Badylak, S.F. 1997. Identification of extractable growth factors from small intestinal submucosa. J. Cellular. Biochem. 67(4), 478–491. Wasserstein, R.L. and Lazar, N.A. 2016. The ASA statement on p-values: context, process, and purpose. Am. Statistician 70(2), 129–133. Wassertein, R.L. and Lazar, N.A. 2016. The ASA statement on p-values: context, purpose. Am. Statistician 70(2), 129–133. Yeum, C.E., Park, E.Y., Lee, S.B., Chun, H.J. and Chae, G.T. 2013. Quantification of MSCs involved in wound healing: use of SIS to transfer MSCs to wound site and quantificatioof MSCs involved in skin wound healing. J. Tissue Eng. Regenerative Med. 7(4), 279–291. | ||
| How to Cite this Article |
| Pubmed Style Marzok AJ, Yousf MF, Alabodi MA, Abbas M. Role of the small intestinal submucosa-extracellular matrix grafts powder on regeneration of full-thickness wound in skin of rabbits. Open Vet. J.. 2026; 16(2): 870-876. doi:10.5455/OVJ.2026.v16.i2.11 Web Style Marzok AJ, Yousf MF, Alabodi MA, Abbas M. Role of the small intestinal submucosa-extracellular matrix grafts powder on regeneration of full-thickness wound in skin of rabbits. https://www.openveterinaryjournal.com/?mno=288473 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.11 AMA (American Medical Association) Style Marzok AJ, Yousf MF, Alabodi MA, Abbas M. Role of the small intestinal submucosa-extracellular matrix grafts powder on regeneration of full-thickness wound in skin of rabbits. Open Vet. J.. 2026; 16(2): 870-876. doi:10.5455/OVJ.2026.v16.i2.11 Vancouver/ICMJE Style Marzok AJ, Yousf MF, Alabodi MA, Abbas M. Role of the small intestinal submucosa-extracellular matrix grafts powder on regeneration of full-thickness wound in skin of rabbits. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 870-876. doi:10.5455/OVJ.2026.v16.i2.11 Harvard Style Marzok, A. J., Yousf, . M. F., Alabodi, . M. A. & Abbas, . M. (2026) Role of the small intestinal submucosa-extracellular matrix grafts powder on regeneration of full-thickness wound in skin of rabbits. Open Vet. J., 16 (2), 870-876. doi:10.5455/OVJ.2026.v16.i2.11 Turabian Style Marzok, Abd-alhadi Jaithom, Methaq Faisal Yousf, Mushtaq A. Alabodi, and Murtadha Abbas. 2026. Role of the small intestinal submucosa-extracellular matrix grafts powder on regeneration of full-thickness wound in skin of rabbits. Open Veterinary Journal, 16 (2), 870-876. doi:10.5455/OVJ.2026.v16.i2.11 Chicago Style Marzok, Abd-alhadi Jaithom, Methaq Faisal Yousf, Mushtaq A. Alabodi, and Murtadha Abbas. "Role of the small intestinal submucosa-extracellular matrix grafts powder on regeneration of full-thickness wound in skin of rabbits." Open Veterinary Journal 16 (2026), 870-876. doi:10.5455/OVJ.2026.v16.i2.11 MLA (The Modern Language Association) Style Marzok, Abd-alhadi Jaithom, Methaq Faisal Yousf, Mushtaq A. Alabodi, and Murtadha Abbas. "Role of the small intestinal submucosa-extracellular matrix grafts powder on regeneration of full-thickness wound in skin of rabbits." Open Veterinary Journal 16.2 (2026), 870-876. Print. doi:10.5455/OVJ.2026.v16.i2.11 APA (American Psychological Association) Style Marzok, A. J., Yousf, . M. F., Alabodi, . M. A. & Abbas, . M. (2026) Role of the small intestinal submucosa-extracellular matrix grafts powder on regeneration of full-thickness wound in skin of rabbits. Open Veterinary Journal, 16 (2), 870-876. doi:10.5455/OVJ.2026.v16.i2.11 |