| Research Article | ||
Open Vet. J.. 2026; 16(2): 944-952 Open Veterinary Journal, (2026), Vol. 16(2): 944-952 Research Article Metabolic adaptive role of interleukin-15 in postpartum dairy cows with negative energy balanceAli Naer Aljumaili and Miaad Jabbar Alhilali*Department of Physiology, Biochemistry and Pharmacology, College of Veterinary Medicine, AlQadisiyiah University, Al-Diwanyiah City, Iraq *Corresponding Author: Miaad Jabbar Alhilali. Department of Physiology, Biochemistry and Pharmacology, College of Veterinary Medicine, AlQadisiyiah University, Al-Diwanyiah City, Iraq. Email: miaad.sahib [at] qu.edu.iq Submitted: 24/09/2025 Revised: 10/01/2026 Accepted: 25/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
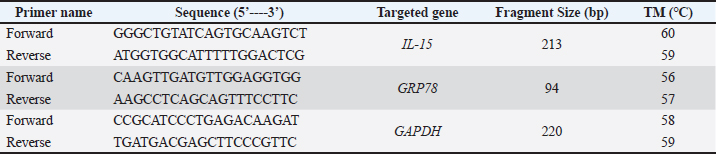
AbstractBackground: Dairy cows exhibit a specific physiological state during the transition from late pregnancy to early lactation. This period is characterized by metabolic and immunological alterations to sustain increased energy needs. Interleukin-15 (IL-15) is a well-known anabolic cytokine that regulates energy expenditure in muscle tissue. Aim: This study aimed to investigate the role of IL-15 in the metabolic adaptation of dairy cows during the transition period. Methods: Thirty-four Holstein–Friesian cows were included in this study. Blood samples were collected twice: three weeks prepartum and three weeks postpartum from all animals to estimate β-HB and non-esterified fatty acids (NEFA) concentrations using ELISA, in addition to IL-15 and Glucose-regulated protein 78 (GRP78) gene expression. Cows are considered to be at normal status or in negative energy balance (NEB) when β-HB is <1.4 mmol/L or β-HB >1.4 mmol/L, respectively. Results: The results of our study revealed that β-HB concentrations are higher significantly (p=0.0001) in NEB than in normal-status cows, both prepartum and postpartum. Similar results were observed regarding NEFA concentration in NEB compared with that in normal-status cows. However, this increase in NEFA concentration did not reach a significant level both prepartum (p=0.100) and postpartum (p=0.141). In addition, NEB cows showed significant downregulation of IL-15 expression levels (p=0.003) compared with normal-status cows postpartum. There were no significant (p=0.934) differences in IL-15 expression levels between the two groups prepartum. Furthermore, the results showed that GRP78 expression levels were significantly upregulated in NEB compared to normal-status cows both prepartum (p=0.010) and postpartum (p=0.034). Conclusion: The body may downregulate IL-15 as a cost-saving immune adaptation. These findings indicate the pivotal physiological role of IL-15 in the regulation of postpartum energy expenditure and metabolic adaptation. Furthermore, the results showed that GRP78 expression levels were upregulated in NEB cows both pre- and postpartum as a part of metabolic adaptation in the transition period. Keywords: Cows, GRP78, IL-15, NEB, Postpartum. IntroductionOne of the most important physiological phases in dairy cows is the transition period, which occurs between 3 weeks before and 3 weeks after parturition (Horst et al., 2021). Since most metabolic and infectious disturbances occur during this period, management practices should be focused on reducing its impact on animal welfare (Van Saun, 2016). The discrepancy between energy intake and energy usage for production (milk, meat, reproduction, etc.) and maintenance is referred to as energy balance. As a result, when energy expenditure exceeds intake energy availability, the animal enters a state of NEB, which frequently results in restricted production of milk or milk components, loss of physical condition, and reduced reproductive performance (Folnožić I Samard et al., 2019; Đuričić et al., 2020). Cows under NEB struggle to maintain glucose homeostasis and experience lipid metabolism dysregulation in the liver (Wathes et al., 2021). During the transition period, the rate of lipolysis exceeds the rate of lipogenesis, leading to rising serum levels of non-esterified fatty acids (NEFA) to satisfy energy demand. If NEFA could not be completely oxidized to carbon dioxide, it would be partially oxidized to ketone bodies or be stored in the liver as triglycerides (Lomander et al., 2012). NEB has the potential to cause ketosis due to increased ketone body levels including β-hydroxybutyric acid (β-HB), acetone, and acetoacetate in different body fluids (Ho et al., 2013). In the early stages of NEB, the liver is activated to regulate lipogenesis and fatty acid oxidation, maintain metabolic homeostasis, and regulate the expression of genes involved in endoplasmic reticulum (ER) stress prepartum (Yonekura, 2024). ER stress initiates a protein response, often referred to as the unfolded protein response (Islam et al., 2022). Glucose-regulated protein 78 (GRP78) is an ER membrane protein triggered by ER stress (Gessner et al., 2010). Due to physiological stress, GRP78 is overexpressed to protect cells from misfolded proteins (Ibrahim et al., 2019). The signaling pathways of interleukins and cytokines play a critical role in metabolic status and energy homeostasis (García et al., 2018). Most cytokines produced by mammalian adipose tissue, such as leptin, adiponectin, and tumor necrosis factor-α, are involved in maintaining homeostasis, regulating appetite and metabolism, and balancing energy (Clemente-Suárez et al., 2023). Changes in the expression of cytokines and their receptors in NEB cows during the transition period have been reported (Noori Sabzikar et al., 2023; Serbetci et al., 2024). Reactive oxygen species (ROS) are produced in NEB due to lipolysis and elevated lipopolysaccharides, with reduced glucose availability, especially in the postpartum period (Contreras and Sordillo, 2011). The production of proinflammatory cytokines by macrophages is stimulated by ROS production, which is a key signal transduction molecule (Morris et al., 2009). IL-15 is a macrophage-derived cytokine that is known to play a role in activating and proliferating natural killer cells, stimulating T cells, promoting B-cell growth, and producing antibodies (Cohen and Cohen, 1996). The IL-15 gene codes for the inflammatory cytokine IL-15, which has structural similarities to interleukin-2 (IL-2) and is produced by mononuclear phagocytes and a few other cells (Lee et al., 2024). In both in vivo and in vitro studies, IL-15 plays a crucial role in controlling glucose metabolism via increased glucose transporter-4 mRNA expression (Busquets et al., 2006). In response to metabolic signals, IL-15 acts through the Janus kinase-signal transducer and activator of the transcription pathway and as a mediator for glucose to enhance glucose oxidation, metabolism, and uptake in skeletal muscle (Krolopp et al., 2016). It promotes lipid oxidation (Busato et al., 2022), maintains skeletal muscle homeostasis (Nadeau and Aguer, 2019). Additionally, it inhibits fibro-adipogenic progenitor differentiation and promotes myofibril regeneration (Duan et al., 2024). To the best of our knowledge, previous researchers have not explored the potential function of IL-15 in dairy cows during NEB, although it has a significant role in glucose and lipid oxidation in regulating energy expenditure. The only study in this field that investigated pro-inflammatory cytokines and discovered that IL-15 is among the most dramatically changed cytokines in follicular granulosa cells in postpartum cows (Warma et al., 2020). Therefore, the objective of this research is to determine whether IL-15 plays a role in the metabolic adaptation of dairy cows during the pre- and postpartum transition period. Materials and MethodsExperimental design and collection of samplesA total of sixty-eight blood samples (two samples from each cow, collected 3 weeks before and 3 weeks after parturition) were collected from the Taj Al-Nahrain for Plant Production and Animal Wealth in Al-Diwaniyah Governorate. The animal age was 50.34 ± 3.05 months, weighed 511.3 ± 12.26 kg prepartum and 489.58 ± 11.23 kg postpartum, and the parity was 2.21 ± 0.23. Healthy animals without any diseases before or after parturition were included in this study, and all cows were fed a balanced diet. Sample collection continued from December 2024 until April 2025. Five milliliters of blood were drawn from the jugular vein of cows and distributed into two tubes, one containing gel and the other containing ethylenediaminetetraacetic acid. Gel tube (Afco, Jordan) samples were centrifuged to collect serum for the Sandwich ELISA test for β-BH and NEFA. For the EDTA tubes (Afco, Jordan), 200 L were withdrawn and preserved in Trizol (TriPure, China) until the PCR test was conducted for IL-15 and GRP78. Whole blood serum was separated by centrifugation (Ohaus, Germany) at 5,000 rpm for 3 minutes and stored at −20°C until analysis. All reagents were stored at room temperature (25℃) for 30 minutes before use. Determination of β-HB and NEFA concentrationsβ-HB concentrations are used to determine the energy status of the dairy cows. Cows were considered at normal status or in NEB when β-HB is <1.4 mmol/L or β-HB >1.4mmol/L, respectively (Warma et al., 2020). The number of normal-status and NEB cows were 12 and 22, respectively, prepartum. Postpartum, the number of normal-status cows was 10 and that of NEB cows was 24. Bovine beta-hydroxybutyric acid (β-HB) ELISA kits (SunLong, China) and bovine non-esterified fatty acid (NEFA) ELISA Kit (SunLong, China) were used. The preparation of β-HB and NEFA standard solutions and assay procedures were performed following the manufacturer’s instructions. IL-15 and GRP78 gene expression in negative energy balance pre and postpartumA TRIzol reagent kit (ADDBio, Korea) was used for total RNA extraction and the determination of extracted RNA quality by employing Quantus™ Fluorometer (Promega, USA). cDNA synthesis and RT-qPCR were performed according to the manufacturer’s instructions using Promega kits (USA) and AddScript RT-qPCR SYBR master mix (ADDBio, Korea). IL-15 amplification was performed using primer sets according to Shi et al. (2019) (Table 1). For the GRP78 gene, primers were designed using the National Center for Biotechnology Information database (Table 1) with an accession number (NM_001075148). GAPDH was used as the reference gene for normalization. Table 1. Primers of the IL-15, GRP78, and GAPDH genes explored in this study.
Relative gene quantification was performed using the 2-ΔΔCT Livak approach to analyze the qRT–PCR data obtained for the IL-15, GRP78, and GAPDH genes (Schmittgen and Livak, 2008). Statistical analysisStatistical differences between pre- and postpartum samples from NEB and normal status cows were analyzed. We used an unpaired Student’s t-test to statistically analyze these data with GraphPad Prism (Version 8.4.3) (GraphPad Software Inc., La Jolla, CA). Data were expressed as mean ± SEM. Values were considered statistically significant when p < 0.05. Ethical approvalThe study was conducted according to the Ethics of the University of Al-Qadisyiah/College of Veterinary Medicine with ethics approval No. VMCP 547/2025. ResultsConcentrations of β-HB and NEFA prepartum versus postpartumThe concentrations of β-HB were significantly higher (p=0.0001) in NEB than in normal-status cows prepartum (Fig. 1A). The values of β-HB are 1.18 ± 0.04 in normal-status group and 2.01 ± 0.11 in NEB group. Likewise, β-HB concentrations in NEB cows were significantly higher prepartum (Fig. 1B). The values of β-HB were 1.25 ± 0.03 and 1.74 ± 0.06 in normal-status and NEB groups, respectively.
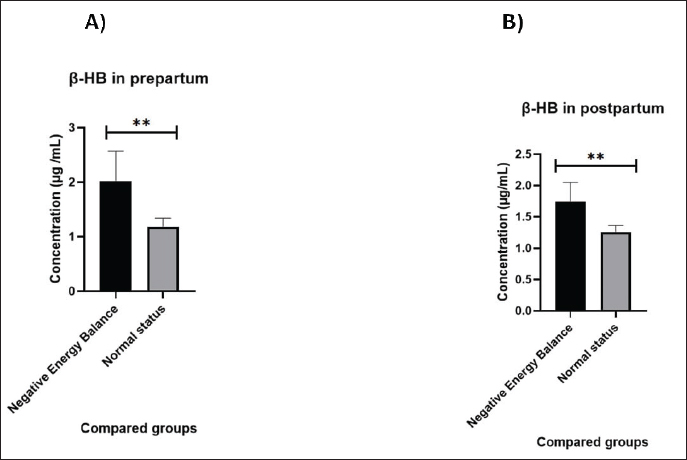
Fig. 1. Concentrations of β-HB (μg/ml) in NEB and normal-status cows in A) Prepartum and B) Postpartum. NEFA concentrations were higher in prepartum NEB than in normal-status cows, but this increase did not reach significance (p=0.100) (Fig. 2A). The values of NEFA are 3.36 ± 0.13 in normal status group and 3.93 ± 0.23 in NEB group. Similarly, the concentrations of NEFA were higher in postpartum NEB cows than in normal status cows, but this increase did not reach a significant level (p=0.141) (Fig. 2B). The values of NEFA are 4.28 ± 0.25 and 5.06 ± 0.31 in normal-status and NEB groups, respectively.
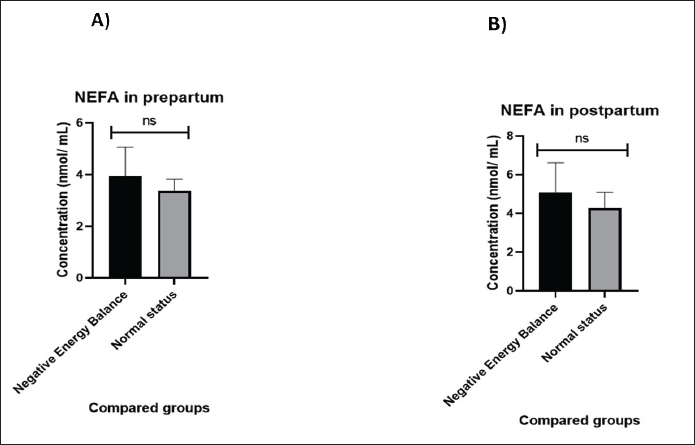
Fig. 2. NEFA concentrations (nmol/mL) in NEB and normal-status cows in A) Prepartum and B) Postpartum. IL15 and GRP78 expression prepartum versus postpartum No significant differences (p=0.934) were observed in the gene expression of IL-15 in cows both in NEB and normal status prepartum (Fig. 3A). The values of IL-15 are 1.36 ± 1.48 in the NEB group and 1.40 ± 1.21 in the normal status group. In addition, IL-15 was significantly downregulated (p=0.003) in the NEB group compared with the normal status group postpartum (Fig. 3B). The values of IL-15 are 0.59 ± 0.27 and 1.30 ± 1.02 in NEB and normal status groups, respectively.
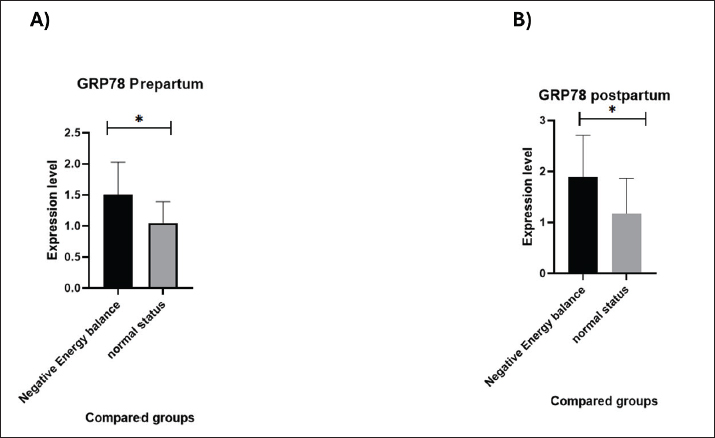
Fig. 3. IL-15 concentrations compared between NEB and normal status in: A) Prepartum and B) Postpartum. GRP78 was significantly upregulated (p=0.010) in prepartum NEB compared with normal status cows (Fig. 4A). The values of GRP78 are 1.50 ± 0.11 in the NEB group and 1.04 ± 0.09 in the normal status group. In addition, GRP78 was significantly upregulated (p=0.034) in NEB compared with normal status cows postpartum (Fig. 4B). The values of GRP78 are 1.88 ± 0.16 and 1.23 ± 0.22 in the NEB and normal status groups, respectively.
Fig. 4. GRP78 concentrations in NEB and normal-status cows in A) Prepartum and B) Postpartum. DiscussionThe elevated β-HB levels prepartum are caused by lipid mobilization linked to NEB and elevated lipolysis with incomplete NEFA oxidation by the liver, resulting in the accumulation of excess NEFA that is largely transformed into ketone bodies as β-HB (McArt et al., 2012). This lipolysis in NEB cows may lead to an acute remodeling process of adipose tissue that exceeds its capacity to buffer the elevated circulating NEFA concentrations (Contreras et al., 2018). However, in our research, NEFA levels were higher in NEB cows prepartum and postpartum but insignificant. The major finding of our study is the notable downregulation of IL-15 levels in NEB cows postpartum versus prepartum. The downregulation of IL-15 levels could be attributed to several factors. The first potential factor may be linked to the important role of IL-15 in energy metabolism, since it acts in an autocrine and paracrine manner to enhance muscle glucose homeostasis and oxidative metabolism (Nadeau and Aguer, 2019). IL-15 is a member of the myokines that improve muscle energy metabolism, including insulin sensitivity, glucose uptake, mitochondrial function, and fatty acid oxidation (Ewendt et al., 2025). It mediates glucose uptake in skeletal muscle cells in vitro in a mouse model (Krolopp et al., 2016). In addition, IL-15 plays an adaptive role in fat metabolism in NEB, where increased fat metabolism is required (Yan et al., 2022). The expression levels of IL-15 and fat content in human muscle and circulatory system were negatively correlated, suggesting direct secretion of IL-15 by skeletal muscle and action of this cytokine on fat cells to inhibit adipocyte differentiation (Pierce et al., 2015). IL-15 expression is downregulated during fatty infiltration, and it inhibits adipogenesis in mouse model, both in vitro and in vivo (Kang et al., 2018). Furthermore, IL-15 inhibits the adipogenic differentiation of stem cells derived from cattle bone marrow in vitro (Shi et al., 2019). The drop in energy availability during NEB triggers AMP-activated protein kinase, a critical cellular energy balance regulator that functions as a cellular energy sensor and promotes catabolic processes (Steinberg and Hardie, 2023; Langer et al., 2024). Other causes of IL-15 downregulation include inflammation and immune suppression. During the postpartum period, the immune system is suppressed in conjunction with NEB (Contreras and Sordillo, 2011; Ingvartsen and Moyes, 2013; Horst et al., 2021). Immune system explores oxidative phosphorylation as an energy source to ensure glucose availability (Wculek et al., 2023). Increased ketone bodies and reduced glucose availability during NEB impair immune system cells, especially macrophages (Morris and Hynes, 2014). A similar downregulation in IL-15 expression, a macrophage-specific cytokine, was proposed by Morris et al. (2009) in cow spleen within severe NEB (Morris et al., 2009). Mitochondrial dysfunction and ROS production due to NEB-associated lipolysis and elevated NEFA levels are another potential cause of IL-15 downregulation, which causes inflammatory dysregulation (Khan et al., 2014). This elevation triggers oxidative stress and disrupts the activities of immune cells and may alter IL-15 transcription (Contreras and Sordillo, 2011; Ingvartsen and Moyes, 2013). Reduced muscular mass and/or activity may explain IL-15 downregulation. IL-15, which is highly expressed in skeletal muscle, stimulates immune cell development and affects myogenesis and muscle mass (Ewendt et al., 2025). In addition, IL-15 is expressed and secreted by primary culture of bovine satellite cells, supporting the involvement of this myokine in primary muscle cell growth (Shira et al., 2024). Muscle activity and/or muscle wasting reduction in NEB may result in reduced IL-15 production (Busato et al., 2022; Tanaka et al., 2024). Hormonal fluctuations during NEB can also contribute to the decrease in IL-15 levels. The body in the NEB state experiences metabolic stress, which results in an increase in cortisol, glucocorticoids, and other stress hormones (Sapolsky et al., 2000). Cortisol reduces the expression and transcription of pro-inflammatory cytokines, including IL-15, in immune cells to preserve energy consumption during metabolic stress (Sapolsky et al., 2000; Bradford et al., 2015). Additionally, insulin-like growth factor-1 (IGF-1) and insulin have critical functions in T-cell survival (Kiernan et al., 2024). In turn, IL-15 promotes the production and activation of IGF-1 from dendritic epidermal T-cells in diabetic mice (Wang et al., 2017). According to the findings of this study, GRP78 expression levels were increased in NEB compared to normal status cows both pre- and postpartum. NEB-associated lipolysis and the increased β-HB and NEFA levels can cause ER stress and dysfunction in bovine mammary epithelial cells (Sharmin et al., 2020; Yan et al., 2022). ER stress induces the unfolded protein response in bovine mammary tissue biopsies both pre- and postpartum (Yonekura et al., 2018) and increases the expression of heat shock proteins in metabolically imbalanced dairy cows (Wathes et al., 2021). When energy deficiency occurs both pre- and postpartum, this ER and metabolic stress activate GRP78 transcription as a component of the adaptive unfolded protein response in the liver and muscles (Gessner et al., 2014; Islam et al., 2022) to regulate lipid metabolism in the periparturient period (Almanza et al., 2019) and to maintain protein quality and homeostasis (Chen et al., 2023). GRP78 regulates the proliferation of bovine mammary epithelial cells to accommodate the increased milk biosynthesis via the mammalian target of the rapamycin signaling pathway (Liu et al., 2019). LimitationsThe observational nature of this study and the small sample size may be the most significant limitations. In addition, we did not investigate the types and numbers of immune cells that produce this interleukin. ConclusionAccording to our study, the incidence of NEB is higher postpartum than prepartum due to prolonged stress and physiologic imbalance. Thus, we can observe the body’s ability to adapt to NEB postpartum rather than prepartum. This conclusion is based on our finding of a greater increase in β-HB prepartum, contrasted by a decreased conversion. In addition, our findings revealed that prepartum IL-15 levels did not differ between NEB and normal status cows. During the postpartum period, animals under NEB showed downregulation of IL-15 levels compared with normal status cows. These findings may indicate the pivotal physiological role of IL-15 in different mechanisms to regulate postpartum energy expenditure and metabolic adaptation. In other words, the body may downregulate IL-15 as an energy-conserving immune adaptation. Furthermore, the results showed that GRP78 expression levels were upregulated in NEB cows both pre- and postpartum as part of metabolic adaptation in this transition period. List of AbbreviationsER, endoplasmic reticulum; GRP78, glucose regulated protein 78; IL, interleukin; IGF-1, insulin-like growth factor-1; NEB, negative energy balance; NEFA, non-esterified fatty acids; ROS, reactive oxygen species; β-HB, beta hydroxy butyric acid. AcknowledgmentsThe authors appreciate the efforts of the dean of Veterinary Medicine at AlQadisiyiah University in supporting this experiment. FundingNo specific grant was provided for this study. Authors’ contributionsAli Naer Aljumaili and Miaad Jabbar Alhilali: Conceptualized the study, developed the methodology, conducted the formal analysis, and collected the data. Miaad Jabbar Alhilali: Served as the corresponding author and played a key role in the final review and editing of the manuscript. Conflict of interestThe authors declare no conflicts of interest. Data availabilityAll data supporting the findings of this study can be found in the manuscript. ReferencesAlmanza, A., Carlesso, A., Chintha, C., Creedican, S., Doultsinos, D., Leuzzi, B., Luís, A., McCarthy, N., Montibeller, L., More, S., Papaioannou, A., Püschel, F., Sassano, M.L., Skoko, J., Agostinis, P., De Belleroche, J., Eriksson, L.A., Fulda, S., Gorman, A.M., Healy, S., Kozlov, A., Munoz-Pinedo, C., Chevet, E., Rehm, M. and Samali, A. 2019. Endoplasmic reticulum stress signaling - from basic mechanisms to clinical applications. FEBS. J. 286(2), 241–278. Bradford, B.J., Yuan, K., Farney, J.K., Mamedova, L.K. and Carpenter, A.J. 2015. Invited review: inflammation during the transition to lactation: new adventures with an old flame. J. Dairy Sci. 98(10), 6631–6650. Busato, S., Ford, H.R., Abdelatty, A.M., Estill, C.T. and Bionaz, M. 2022. Peroxisome Proliferator-Activated Receptor Activation in Precision-Cut Bovine Liver Slices Reveals Novel Putative Peroxisome Proliferator-Activated Receptor Targets in Periparturient Dairy Cows. Front. Vet. Sci. 9, 931264. Busquets, S., Figueras, M., Almendro, V., López-Soriano, F.J. and Argilés, J.M. 2006. Interleukin-15 increases glucose uptake in the skeletal muscle Anti-diabetogenic effect of the cytokine. Biochimica. Et. Biophysical. Acta. 1760, 1613–1617. Chen, X., Shi, C., He, M., Xiong, S. and Xia, X. 2023. Endoplasmic reticulum stress: molecular mechanism and therapeutic targets. Signal Transduct. Target. Ther. 8(1), 352. Clemente-Suárez, V.J., Redondo-Flórez, L., Beltrán-Velasco, A.I., Martín-Rodríguez, A., Martínez-Guardado, I., Navarro-Jiménez, E., Laborde-Cárdenas, C.C. and Tornero-Aguilera, J.F. 2023. The role of adipokines in health and disease. Biomedicines 11(5), 1290. Cohen, M.C. and Cohen, S. 1996. Cytokine function: a study in biologic diversity. Am. J. Clin. Pathol. 105(5), 589–598. Contreras, G.A. and Sordillo, L.M. 2011. Lipid mobilization and inflammatory responses in dairy cows during transition. Comparative. Immunol. Microbiol. Infect. Dis. 34(3), 281–289; doi:10.1016/j.cihd.2012.01.012 Contreras, G.A., Strieder-Barboza, C. and De Koster, J. 2018. Symposium review: modulating adipose tissue lipolysis and remodeling to improve immune function during the transition period and early lactation of dairy cows. J. Dairy. Sci. 101(3), 2737–2752. Duan, Z., Yang, Y., Qin, M. and Yi, X. 2024. Interleukin 15: a new intermediary in the effects of exercise and training on skeletal muscle and bone function. J. Cell. Mol. Med. 28, e70136. Đuričić, D., Vince, S., Lojkić, M., Jelušić, S., Turk, R., Valpotić, H., Gračner, D., Maćešić, N., Folnožić, I., Šostar, Z. and Samardžija, M. 2020. Effects of dietary clinoptilolite on reproductive performance, serum progesterone and insulin-like growth factor-1 concentrations in dairy cows during pregnancy and lactation. Polish. J Vet. Sci. 23(1), 69–75. Ewendt, F., Drewitz, F., Althammer, M., Eichler, C., Brandsch, C., Brey, S., Winkler, T. H., Wilkens, M. R., St-Arnaud, R., Kreutz, M. and Stangl, G. I. 2025. Vitamin D stimulates Il-15 synthesis in rodent muscle. Biochem. Biophys. Rep. 41, 101925. Folnožić I Samard., žija, M., Đuričić D Vince., Perkov S Jelu., šić S Valpoti., ć H Ljubi., ć BB Lojki., ć M Gra., čner, D., Žaja IŽ Ma., ćešić N Grizelj., Dobrani, J., ć T Red., žepi, G. and Šostar Z Turk. 2019. Effects of in-feed clinoptilolite treatment on serum metabolic and antioxidative biomarkers and APR in dairy cows during pregnancy and early lactation. Res. Vet. Sci. 127, 57–64. García, M.D.C., Pazos, P., Lima, L. and Diéguez, C. 2018. Energy Expenditure and Brown/Beige Thermogenic Activity Regulation by Interleukins: new Roles for Old Actors. Int. J. Mol. Sci. 19(9), 2569. Gessner, D.K., Schlegel, G., Ringseis, R., Schwarz, F.J. and Eder, K. 2014. Up-regulation of endoplasmic reticulum stress induced genes of the unfolded protein response in the liver of periparturient dairy cows. BMC Vet. Res. 10, 46. Ho, C.F., Chan, K.W., Yeh, H.I., Kuo, J., Liu, H.J. and Wang, C.Y. 2013. Ketone bodies upregulate endothelial Cx43 gap junctions. Vet. J. 198, 696–701. Horst, E.A., Kvidera, S.K. and Baumgard, L.H. 2021. Invited review: influence of immune activation on transition cow health and performance: A critical evaluation of traditional dogmas. J. Dairy. Sci. 104(8), 8380–8410. Ibrahim, I.M., Abdelmalek, D.H. and Elfiky, A.A. 2019. GRP78: a cell's response to stress. Life Sci. 226, 156–163. Ingvartsen, K.L. and Moyes. 2013. Nutrition, immune function, and health. Animal 7, 112–122. Islam, M.A., Adachi, S., Shiiba, Y., Takeda, K.I., Haga, S. and Yonekura, S. 2022. Effects of starvation-induced negative energy balance on endoplasmic reticulum stress in cow liver. Anim. Bioscience. 35(1), 22–28. Kang, X., Yang, M.Y., Shi, Y.X., Xie, M.M., Zhu, M., Zheng, X.L., Zhang, C.K., Ge, Z.L., Bian, X.T., Lv, J.T., Wang, Y.J., Zhou, B.H. and Tang, K.L. 2018. Interleukin-15 facilitates muscle regeneration through modulation of fibro/adipogenic progenitors. Cell Communication Signaling CCS. 16(1), 42. Khan, M.Z., Huang, B., Kou, X., Chen, Y., Liang, H., Ullah, Q., Khan, I.M., Khan, A., Chai, W. and Wang, C. 2014. Enhancing bovine immune, antioxidant and anti-inflammatory responses with vitamins, rumen-protected amino acids, and trace minerals to prevent periparturient mastitis. Front. Immunol. 14, 1290044. Kiernan, K., Alwarawrah, Y., Nichols, A. G., Danzaki, K. and MacIver, N.J. 2024. Insulin and IGF-1 have both overlapping and distinct effects on CD4+ T cell mitochondria, metabolism, and function. Sci. Rep. 14(1), 4331. Krolopp, J.E., Thornton, S.M. and Abbott, M.J. 2016. IL-15 activates the Jak3/STAT3 signaling pathway to mediate glucose uptake in skeletal muscle cells. Front. Physiol. 7, 626. Langer, H.T., Rohm, M., Goncalves, M.D. and Sylow, L. 2024. AMPK as a mediator of tissue preservation: time for a shift in dogma?. Nature Rev. Endocrinol. 20, 526–540. Lee, H., Park, S.H. and Shin, E.C. 2024. IL-15 in T-cell responses and immunopathogenesis. Immune. Netw. 24(1), e11. Liu, Y., Wang, X., Zhen, Z., Yu, Y., Qiu, Y. and Xiang, W. 2019. GRP78 regulates milk biosynthesis and the proliferation of bovine mammary epithelial cells through the mTOR signaling pathway. Cell. Mol. Biol. Lett. 24, 57. Lomander, H., Frössling, J., Ingvartsen, K.L., Gustafsson, H. and Svensson, C. 2012. Supplemental feeding with glycerol or propylene glycol of dairy cows in early lactation--effects on metabolic status, body condition, and milk yield. J. Dairy Sci. 95(5), 2397–2408. McArt, J.A., Nydam. and Oetzel. 2012. Epidemiology of subclinical ketosis in early lactation dairy cattle. Dairy Sci. 95, 5056–5066. Morris, D. and Hynes, A. 2014. Negative energy balance and immune function in cows Technology updates: animal and grassland research and innovation. Springer, Berlin. Morris, D.G., Waters, S.M., McCarthy, S.D., Patton, J., Earley, B., Fitzpatrick, R., Murphy, J.J., 2009. Pleiotropic effects of negative energy balance in postpartum dairy cows on splenic gene expression: repercussions for innate and adaptive immunity. Washington, DC: American Physiology Society. https://doi.org/10.1152/physiolgenomics.90394.2008 Nadeau, L. and Aguer, C. 2019. Interleukin-15 as a myokine: mechanistic insight into its effect on skeletal muscle metabolism. Appl. Physiol. Nutr. Metab. 44, 229–238. Noori Sabzikar, Z., Mohri, M. and Seifi, H.A. 2023. Variations in adipokines, proinflammatory cytokines, oxidative stress biomarkers, and energy characteristics during the transition period in dairy cows. Vet. Res. Forum. 14(2), 87–95. Pierce, J.R., Maples, J.M. and Hickner, R.C. 2015. IL-15 concentrations in skeletal muscle and subcutaneous adipose tissue in lean and obese humans: local effects of IL-15 on adipose tissue lipolysis. Am. J. Physiol. Endocrinol. Metab. 308(12), E1131–E1139. Sapolsky, R.M., Sapolsky LM Romero. and Munck. 2000. How do glucocorticoids influence stress responses?. Endocrine Rev. 21(1), 55–89. Schmittgen, T.D. and Livak, K.J. 2008. Analyzing real-time PCR data by the comparative CT method. Nat. Protoc. 3, 1101–1108; doi:10.1038/nprot.2008.73 Serbetci, I., González-Grajales, L.A., Herrera, C., Ibanescu, I., Tekin, M., Melean, M., Magata, F., Malama, E., Bollwein, H. and Scarlet, D. 2024. Impact of negative energy balance and postpartum diseases during the transition period on oocyte quality and embryonic development in dairy cows. Front. Vet. Sci. 10, 1328700. Sharmin, M.M., Mizusawa, M., Hayashi, S., Arai, W., Sakata, S. and Yonekura, S. 2020. Effects of fatty acids on inducing endoplasmic reticulum stress in bovine mammary epithelial cells. J. Dairy. Sci. 103(9), 8643–8654. Shi, M., Li, Z., Miao, Z., Guo, Y. and Yi, L. 2019. Interleukin-15 inhibits adipogenic differentiation of cattle bone marrow-derived mesenchymal stem cells via regulating the crosstalk between signal transducer and activator of transcription 5A and Akt signaling. Biochem. Biophysical Res. Commun. 517(2), 346–352. Shira, K.A., Murdoch, B.M., Thornton, K.J., Reichhardt, C.C., Becker, G.M., Chibisa, G.E. and Murdoch, G.K. 2024. Myokines produced by cultured bovine satellite cells harvested from 3- and 11-month-old Angus Steers. Animals 14, 709. Steinberg, G.R. and Hardie, D.G. 2023. New insights into AMPK activation and function. Nature Rev. Mol. Cell Biol. 24(4), 255–272. Tanaka, M., Sugimoto, K., Akasaka, H., Yoshida, S., Takahashi, T., Fujimoto, T., Xie, K., Yasunobe, Y., Yamamoto, K., Hirabayashi, T., Nakanishi, R., Fujino, H. and Rakugi, H. 2024. Effects of interleukin-15 on autophagy regulation in the skeletal muscle of mice. Am. J. Physiol. Endocrinol. Metab. 326(3), E326–E340. Van Saun, R.J. 2016. Indicators of dairy cow transition risks: metabolic profiling revisited. Tierarztliche Praxis Ausgabe. G. Grosstiere/Nutztiere. 44(2), 118–127. Wang, Y., Bai, Y., Li, Y., Liang, G., Jiang, Y., Liu, Z., Liu, M., Hao, J., Zhang, X., Hu, X., Chen, J., Wang, R., Yin, Z., Wu, J., Luo, G. and He, W. 2017. IL-15 Enhances Activation and IGF-1 Production of Dendritic Epidermal T Cells to Promote Wound Healing in Diabetic Mice. Front. Immunol. 8, 1557. Warma, A., Marianne, D., Younes, C., Raynald, D., Roxane, R. and Kalidou, N. 2020. Changes in the expression profile of interleukins in granulosa cells of preovulatory follicles during the postpartum period in dairy cows. Cytokine X 2(1), 100022. Wathes, D.C., Cheng, Z., Salavati, M., Buggiotti, L., Takeda, H., Tang, L., Becker, F., Ingvartsen, K.I., Ferris, C., Hostens, M. and Crowe, M.A. 2021. Gplus E. Relationships between metabolic profiles and gene expression in the liver and leukocytes of early lactating dairy cows. J. Dairy Sci. 104(3), 3596–3616. Wculek, S.K., Heras-Murillo, I., Mastrangelo, A., Mañanes, D., Galán, M., Miguel, V., Curtabbi, A., Barbas, C., Chandel, N.S., Enríquez, J.A., Lamas, S. and Sancho, D. 2023. Oxidative phosphorylation orchestrates tissue macrophages homeostasis. Immunity 56, 516–530. Yan, Y., Huang, J., Huan, C., Li, L. and Li, C. 2022. Non-esterified fatty acid induces ER Stress-Mediated Apoptosis via ROS/MAPK Signaling in the Bovine Mammary Epithelial Cells. Metabolites 12(9), 803. Yonekura M Tsuchiya., Tokutake M Mizusawa., Nakano M Miyaji., Ishizaki, H. and Haga. 2018. The unfolded protein response is involved in differentiation and apoptosis of bovine mammary epithelial cells. J. Dairy Sci. 101(4), 3568–3578. Yonekura, S. 2024. Role of endoplasmic reticulum stress in metabolic diseases and mammary epithelial cell homeostasis in dairy cows. Anim. Sci. J. 95(1), e13935. | ||
| How to Cite this Article |
| Pubmed Style Aljumaili AN, Alhilali MJ. Metabolic adaptive role of interleukin-15 in postpartum dairy cows with negative energy balance. Open Vet. J.. 2026; 16(2): 944-952. doi:10.5455/OVJ.2026.v16.i2.16 Web Style Aljumaili AN, Alhilali MJ. Metabolic adaptive role of interleukin-15 in postpartum dairy cows with negative energy balance. https://www.openveterinaryjournal.com/?mno=286271 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.16 AMA (American Medical Association) Style Aljumaili AN, Alhilali MJ. Metabolic adaptive role of interleukin-15 in postpartum dairy cows with negative energy balance. Open Vet. J.. 2026; 16(2): 944-952. doi:10.5455/OVJ.2026.v16.i2.16 Vancouver/ICMJE Style Aljumaili AN, Alhilali MJ. Metabolic adaptive role of interleukin-15 in postpartum dairy cows with negative energy balance. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 944-952. doi:10.5455/OVJ.2026.v16.i2.16 Harvard Style Aljumaili, A. N. & Alhilali, . M. J. (2026) Metabolic adaptive role of interleukin-15 in postpartum dairy cows with negative energy balance. Open Vet. J., 16 (2), 944-952. doi:10.5455/OVJ.2026.v16.i2.16 Turabian Style Aljumaili, Ali Naer, and Miaad Jabbar Alhilali. 2026. Metabolic adaptive role of interleukin-15 in postpartum dairy cows with negative energy balance. Open Veterinary Journal, 16 (2), 944-952. doi:10.5455/OVJ.2026.v16.i2.16 Chicago Style Aljumaili, Ali Naer, and Miaad Jabbar Alhilali. "Metabolic adaptive role of interleukin-15 in postpartum dairy cows with negative energy balance." Open Veterinary Journal 16 (2026), 944-952. doi:10.5455/OVJ.2026.v16.i2.16 MLA (The Modern Language Association) Style Aljumaili, Ali Naer, and Miaad Jabbar Alhilali. "Metabolic adaptive role of interleukin-15 in postpartum dairy cows with negative energy balance." Open Veterinary Journal 16.2 (2026), 944-952. Print. doi:10.5455/OVJ.2026.v16.i2.16 APA (American Psychological Association) Style Aljumaili, A. N. & Alhilali, . M. J. (2026) Metabolic adaptive role of interleukin-15 in postpartum dairy cows with negative energy balance. Open Veterinary Journal, 16 (2), 944-952. doi:10.5455/OVJ.2026.v16.i2.16 |