| Research Article | ||
Open Vet. J.. 2026; 16(1): 445-451 Open Veterinary Journal, (2026), Vol. 16(1): 445-451 Research Article Effects of nanocurcumin administration on malondialdehyde and estrogen levels in mice exposed to noise-induced stressViski Fitri Hendrawan1*, Qatrinnada Hazimah2, Tiara Widyaputri3, Galuh Chandra Agustina4, Dwi Rahmawati5 and Ertika Fitri Lisnanti61Department of Veterinary Reproduction, Faculty of Veterinary Medicine, Universitas Brawijaya, Jakarta, Indonesia 2Faculty of Veterinary Medicine, Universitas Brawijaya, Malang, Indonesia 3Veterinary Clinical Pathology and Laboratory Animal Laboratory, Faculty of Veterinary Medicine, Universitas Brawijaya, Malang, Indonesia 4Department of Veterinary Physiology, Faculty of Veterinary Medicine, Universitas Brawijaya, Malang, Indonesia 5Department of Orthodontics, Faculty of Dentistry, Universitas Brawijaya, Malang, Indonesia 6Program of Animal Husbandry, Faculty of Agriculture, Universitas Islam Kediri, Kediri, Indonesia *Corresponding Author: Viski Fitri Hendrawan. Department of Veterinary Reproduction, Faculty of Veterinary Medicine, Universitas Brawijaya, Jakarta, Indonesia. Email: viski [at] ub.ac.id Submitted: 24/09/2025 Revised: 03/12/2025 Accepted: 22/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
AbstractBackground: During pregnancy, noise stress has been associated with the activation of the hypothalamic–pituitary–adrenal axis and increased oxidative stress, which can disrupt hormonal balance and adversely affect pregnancy outcomes. Malondialdehyde (MDA) is a key biomarker of lipid peroxidation, whereas estrogen plays a critical role in maintaining gestational homeostasis. Excess oxidative stress and hormonal imbalance may impair fetal development. Nanocurcumin, a curcumin nanoparticle formulation, exhibits enhanced antioxidant and anti-inflammatory properties compared with conventional curcumin and may offer protective effects against pregnancy-related oxidative damage. Aim: This study aimed to evaluate the effects of nanocurcumin on MDA and estrogen levels in pregnant mice exposed to noise-induced stress, contributing to Sustainable Development Goals (SDG) 3 (Good Health and Well-being) through stress mitigation and maternal health protection, and SDG 9 (Industry, Innovation, and Infrastructure) by applying innovative nanotechnology. Methods: An experimental study was conducted using 25 pregnant BALB/c mice randomly divided into five groups: K(–): untreated control, K(+): exposed to 135 dB noise for 40 minutes, P1, P2, and P3: exposed to noise and treated with nanocurcumin at 14, 21, and 24.5 mg/kg body weight, respectively. An electric siren was placed inside a soundproof chamber to deliver noise, and the 135 dB intensity was calibrated using a precision digital decibel meter (Benetech GM1356). Oral treatment was administered from gestational day (GD) 6 to gestational day 18. Serum MDA and estrogen levels were measured using an enzyme-linked immunosorbent assay. Data were analyzed using one-way analysis of variance followed by Tukey’s honest significant difference post hoc test (p < 0.05). Results: Noise exposure numerically increased both MDA and estrogen levels, although not all differences reached statistical significance. However, nanocurcumin administration significantly reduced both parameters in a dose-dependent manner. The highest dose (24.5 mg/kg BW, P3 group) resulted in a 40.87% reduction in MDA and a 22.88% reduction in estrogen compared to K(+), with values approaching those in the untreated control. Conclusion: Nanocurcumin has potential as a protective agent against noise-induced oxidative stress and hormonal disturbances during pregnancy. To confirm translational relevance, future research should focus on elucidating underlying molecular mechanisms and evaluating fetal outcomes. Keywords: Estrogen, Malondialdehyde, Noise, Pregnant mice, Good Health and Well-being. IntroductionPregnancy represents a complex physiological state in which maternal homeostasis is particularly vulnerable to environmental stressors. Noise exposure is an increasingly recognized disruptor of reproductive health. Noise is defined as an unwanted sound that can interfere with comfort and physiological functions, and its biological effects depend on intensity, frequency, exposure duration, and species-specific auditory sensitivity (Brouček, 2014). In mice, which detect sound frequencies between 0.25 and 80 kHz, exposure to high-intensity noise (≥90 dB) has been shown to activate the hypothalamic–pituitary–adrenal (HPA) axis, triggering stress responses that may affect pregnancy outcomes (Jafari et al., 2017). Therefore, noise stress was selected as a relevant and measurable model of environmental stress during pregnancy. Physiological stress during gestation is closely associated with elevated oxidative stress, a condition arising from an imbalance between reactive oxygen species (ROS) production and endogenous antioxidant defense capacity. This imbalance can disrupt redox signaling, damage cellular components, and impair maternal–fetal homeostasis (Alizadeh and Kheirouri, 2019). Oxidative stress during pregnancy is of particular concern because it can trigger placental insufficiency, inflammation, and endothelial dysfunction, contributing to gestational disorders such as preeclampsia and Intrauterine growth restriction (Subandi et al., 2025). Malondialdehyde, a reactive aldehyde that serves as a biomarker of cellular damage caused by ROS accumulation, is one of the principal products of lipid peroxidation under oxidative stress (Ayala et al., 2014; Jakubczyk, et al., 2020a). Elevated MDA levels have been linked to pathological changes in gestational tissues, especially when exposure occurs during critical stages of fetal development, such as organogenesis and placentation (Parisi et al., 2023). Moreover, estrogen homeostasis is vulnerable to oxidative insult. During pregnancy, estrogen plays an essential role in uterine vascular remodeling, decidualization, angiogenesis, and immune modulation (Supriyanto et al., 2019; Parisi et al., 2023). However, dysregulation of estrogen levels, particularly excessive elevations, has been associated with impaired trophoblastic invasion and placental pathologies (Hajar et al., 2023; Kokkorachedu et al., 2024). Noise-induced oxidative stress may disrupt estrogen biosynthesis and metabolism by affecting aromatase expression and redox-sensitive signaling pathways, compromising the hormonal balance critical to maintaining gestational health. Curcumin, a polyphenolic compound extracted from Curcuma longa, has been widely studied for its antioxidant and anti-inflammatory activities. These effects are primarily mediated by ROS scavenging, lipid peroxidation inhibition, and redox-sensitive transcription factor modulation, such as Nuclear factor erythroid 2–related factor 2 (Alizadeh and Kheirouri, 2019; Bateni et al., 2022). However, the clinical potential of curcumin is limited due to its poor aqueous solubility, rapid metabolism, and low systemic bioavailability (Baradaran et al., 2020). To overcome these limitations, a nanoparticle formulation of nanocurcumin has been developed to enhance its solubility, stability, and systemic absorption. This nanoformulation, typically encapsulated using biodegradable carriers such as poly (lactic-co-glycolic acid), improves pharmacokinetic profiles and facilitates enhanced cellular uptake (Bateni et al., 2022). Unlike conventional curcumin, nanocurcumin demonstrates significantly increased bioavailability and efficacy in modulating oxidative and inflammatory pathways in vivo (Baradaran et al., 2020). Despite its therapeutic promise, limited data exist regarding the application of nanocurcumin during pregnancy, especially under oxidative stress conditions such as those induced by environmental noise. Therefore, this study aimed to evaluate the effects of nanocurcumin administration on serum MDA and estrogen levels in pregnant mice exposed to noise stress, addressing a specific gap in the literature regarding advanced curcumin delivery systems for the mitigation of maternal stress. This research aligns with the global health and innovation agenda outlined in the Sustainable Development Goals (SDGs). Specifically, it supports SDG 3 (Good Health and Well-being) by exploring preventive strategies for reducing oxidative and hormonal disruptions during pregnancy, which may compromise maternal and fetal health. Furthermore, by utilizing nanocurcumin, an advanced nanoformulated herbal compound, the study contributes to SDG 9 (Industry, Innovation, and Infrastructure) through the development and application of innovative biomedical interventions to address reproductive health challenges. Materials and MethodsAnimalsThis experimental study used 25 pregnant female BALB/c mice, aged 3 months, with an average body weight of 36.5 g. The animals were obtained from a certified breeding facility and acclimatized for 7 days at the Embryology Laboratory, Faculty of Veterinary Medicine, Universitas Airlangga. Mice were housed in polypropylene cages with husk bedding and maintained under standard laboratory conditions (25°C ± 2°C, 12 hours light/dark cycle). All animals were fed a standard commercial rodent pellet diet (Betagro® Rodent Maintenance Feed, Indonesia) containing approximately 18% crude protein, 5% fat, 4% fiber, 1% calcium, and essential vitamins for reproductive stability. Feed was provided once daily at a fixed amount of 5 g/mouse/day, which is sufficient to meet the nutritional requirements of pregnant mice. The type of feed and total daily quantity were kept identical across all groups to ensure dietary uniformity and eliminate nutrition-related bias. Drinking water was provided ad libitum, and bedding was replaced every 3 days. Experimental designMice were randomly divided into five groups (n=5 per group):
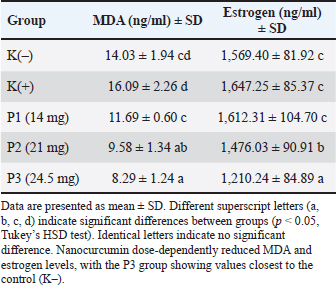
The sample size (n=5 per group) was selected based on previously published studies that evaluated oxidative and hormonal changes in murine models using similar group sizes. Although relatively small, this number was deemed sufficient for detecting statistically significant differences in MDA and estrogen levels, as confirmed by our pilot trial. Nonetheless, we acknowledge the limitations regarding statistical power and recommend future studies to perform formal power analysis with larger cohorts. The selected nanocurcumin doses (14, 21, and 24.5 mg/kg BW) were based on previous in vivo studies demonstrating the effective antioxidant and anti-inflammatory activities of nanocurcumin formulations within this range (Baradaran et al., 2020; Bateni et al., 2022). Additionally, a preliminary pilot study conducted in our laboratory on pregnant BALB/c mice confirmed that these doses were well tolerated and produced measurable reductions in oxidative stress markers. Preparation and characterization of nanocurcuminNanocurcumin was obtained in a 50 mg/ml suspension from NanolabTech Bioresearch, Indonesia. The formulation consisted of curcumin encapsulated in PLGA nanoparticles with an average size of 87 ± 6 nm, which was verified by dynamic light scattering. As supported by prior studies, this nanoformulation enhances systemic bioavailability and cellular uptake (Bateni et al., 2022). The administered doses (14, 21, and 24.5 mg/kg BW) were selected based on previous in vivo reports and pilot dose optimization trials (Baradaran et al., 2020). Pregnancy inductionEstrous synchronization was conducted through the administration of PMSG (5 IU, intraperitoneally), followed by HCG (5 IU, intraperitoneally) 48 hours later. The females were mated using the polydatin method, and the appearance of a vaginal plug the next morning was designated as gestational day 0. Stress exposure and treatment protocolFrom gestational day 6 to gestational day 18, pregnant mice in the K(+), P1, P2, and P3 groups were subjected to noise stress by exposure to sound pressure levels of 135 dB for 40 minutes daily. The noise was generated using an electric siren placed inside a soundproof chamber and calibrated using a digital sound level meter (Benetech GM1356). This high-intensity noise was selected based on prior experimental protocols to reliably induce physiological stress responses in rodents (Jafari et al., 2017; Shafiei et al., 2017). Immediately after each daily noise exposure, animals in groups P1, P2, and P3 received nanocurcumin treatment via oral gavage using a sterile feeding needle and a water-based suspension containing the respective doses (14, 21, and 24.5 mg/kg BW). The negative control group [K(–)] was not exposed to noise and did not receive any treatment. Although the noise intensity used was considerably high, the exposure duration was limited to 40 minutes per day to minimize distress and potential harm. All animals were monitored daily throughout the treatment period for signs of pain, stress, or abnormal behavior. The institutional animal ethics committee reviewed and approved the entire experimental protocol, and all procedures were conducted in accordance with internationally accepted guidelines for the humane use of animals in research. Anesthesia, euthanasia, and sample collectionOn GD19, anesthesia was achieved via intraperitoneal administration of ketamine (10%, 0.1 ml/10 g BW) and xylazine (2%, 0.08 ml/10 g BW). Euthanasia was performed via intracardiac exsanguination. Blood samples (1.5 ml) were collected in plain tubes and centrifuged at 3,500 rpm for 15 minutes at room temperature to isolate the serum. The samples were stored at 20°C. Measurement of MDA and estrogenSerum levels of MDA and estrogen were quantified using commercial enzyme-linked immunosorbent assay kits (BT-LAB, China) according to the manufacturer’s instructions. All assays were performed in triplicate to ensure the reproducibility of the results. Optical density was measured at 450 nm using an enzyme-linked immunosorbent assay reader (Diatek DR-200Bc). Outliers were assessed using the Grubbs’ test (α=0.05) and excluded when statistically justified. The ELISA kits used were from BT-LAB (Cat. No. E0032Mo for MDA and E0086Mo for Estrogen, BT-LAB, China), with sensitivity ranges of 0.1–10 ng/ml and intra-/inter-assay CVs <10%. Standard curves were prepared for each plate, and all samples were measured in triplicate. Data analysisData were tested for normality using the Shapiro–Wilk test and for homogeneity using Levene’s test. Normally distributed and homogeneous data were analyzed using one-way analysis of variance, followed by Tukey’s honest significant difference test for multiple comparisons. A significance threshold of p < 0.05 was used. Statistical analyses were conducted using the Statistical Package for the Social Sciences version 25 (). Ethical approvalThis study was conducted in strict compliance with the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines and institutional policies on animal welfare. All animal procedures were reviewed and approved by the Animal Ethics Committee of Universitas Airlangga, Indonesia (Approval No. E.5.a/038/KEPKUMM/III/2025), approved on March 25, 2025.. Animal handling and experimental procedures were designed to minimize pain, distress, and fetal injury. ResultsLevels of malondialdehydeExposure to 135 dB noise for 40 minutes resulted in a numerical increase in serum MDA levels in the K(+) group compared with that in the K() group, although this increase was not statistically significant. The administration of nanocurcumin resulted in a dose-dependent reduction in MDA levels across all treatment groups.
These trends demonstrate the antioxidant potential of nanocurcumin in mitigating noise-induced oxidative stress. The full numerical data are presented in Table 1. Table 1. Serum malondialdehyde (MDA) and estrogen levels in pregnant mice exposed to noise and nanocurcumin.
Estrogen levelsSerum estrogen levels (ng/ml) were significantly elevated in the noise-exposed K(+)group compared to KQ–) (p < 0.01), which reflects oxidative stress-induced hormonal imbalance. Serum estrogen levels were elevated in mice exposed to 135 dB noise [K(+)] compared with the untreated control [K()], indicating that hormonal dysregulation was induced by noise. Administration of nanocurcumin attenuated this increase in a dose-dependent manner: the P1 group showed a mild reduction, P2 exhibited a moderate decrease, and P3 demonstrated a significant reduction of approximately 23% compared to K(+). Importantly, the P3 group’s estrogen levels approached those of the K(–) group, which reflects the physiological baseline level for pregnant mice under non-stress conditions (approximately 9.6–10.2 ng/ml, as reported in Supriyanto et al., 2019). Among all groups, P3 achieved the closest normalization of estrogen levels and was significantly lower than K(+). The detailed results are presented in Table 1. Nanocurcumin administration attenuated the following changes:
These results indicate that nanocurcumin not only modulates oxidative stress markers but also helps restore estrogen balance under stress conditions. DiscussionExposure to 135 dB noise during pregnancy significantly increased oxidative stress and disrupted hormonal balance in mice, as evidenced by elevated serum MDA and estrogen levels. These findings reinforce previous reports that high-intensity noise acts as a potent environmental stressor that activates the HPA axis. Activation of the HPA axis stimulates glucocorticoid secretion, which enhances the generation of ROS and contributes to oxidative imbalance. Excessive ROS can impair mitochondrial activity, disrupt redox homeostasis, and interfere with the regulation of key reproductive hormones, including estrogen. This pattern of biochemical disturbance is consistent with earlier studies showing that environmental noise exposure during gestation alters oxidative markers and endocrine profiles in laboratory animals (Jafari et al., 2017; Al-Shammary et al., 2023). Administration of nanocurcumin attenuated these adverse changes in a dose-dependent manner. Notably, the highest dose group (24.5 mg/kg BW; P3) showed the most significant reductions in both MDA and estrogen levels. Nanocurcumin, as a nanoencapsulated formulation of curcumin, provides enhanced solubility, stability, and systemic bioavailability compared to conventional curcumin (Bateni et al., 2022). Its efficacy is attributed to its potent antioxidant capacity, including ROS scavenging, inhibition of lipid peroxidation, and upregulation of endogenous antioxidant enzymes such as superoxide dismutase, Catalase, and Glutathione peroxidase (Alizadeh and Kheirouri, 2019). These mechanisms contribute to the reduction of oxidative damage biomarkers, such as MDA. Importantly, estrogen levels were significantly elevated in noise-stressed pregnant mice, which is indicative of an endocrine disturbance associated with oxidative stress. Although estrogen is essential for pregnancy maintenance, including supporting endometrial receptivity and placental development, excessive levels of estrogen may pose risks such as placental dysfunction and abnormal fetal growth (Rohmah et al., 2017; Parisi et al., 2023). Nanocurcumin treatment restored estrogen levels toward the physiological range, particularly in the P3 group. This normalization may be mediated by inhibition of aromatase, the enzyme that converts androgens into estrogens, consistent with previous findings (Filardi et al., 2020). However, it is critical to emphasize that balanced estrogen levels, not merely reduced ones, are vital for successful pregnancy maintenance. Excessive estrogen suppression could also pose risks to fetal development, highlighting the need for precise dose calibration and physiological monitoring in future studies. Although this study provides mechanistic insight into the protective role of nanocurcumin, several limitations must be acknowledged:
Recent studies have increasingly highlighted the role of maternal oxidative stress in disrupting fetal development and endocrine regulation. Mendoza et al. (2023) demonstrated that elevated MDA levels impair placental function and fetal growth, supporting the significance of oxidative damage observed in our study. Furthermore, sustained exposure to high-intensity noise has been shown to dysregulate Hypothalamic–pituitary–gonadal axis axis signaling, resulting in hormonal imbalance and compromised reproductive performance in rodents (Tanaka et al., 2025). These data reinforce the rationale for using noise stress as a relevant model in gestational studies. Emerging evidence indicates that nanocurcumin may modulate hormonal biosynthesis pathways beyond its antioxidant activity. Chen et al. (2024) reported that nanocurcumin downregulates estrogen synthesis by suppressing aromatase expression via PI3K/Akt and NF-κB signaling in reproductive tissues under stress. Although further molecular validation is required, these mechanisms are likely involved in the hormonal normalization observed in our study. Collectively, integrating nanotechnology-based antioxidants into pregnancy stress models may provide new avenues for therapeutic intervention. Although noise exposure produced numerical increases in both MDA and estrogen levels, the differences were not statistically significant. This may be due to the short exposure period (13 days), limited daily exposure (40 minutes), or sample size (n=5), which may have constrained the power to detect subtle changes. Recent findings further support the central role of oxidative stress and endocrine disruption in pregnancy complications induced by environmental stressors. Hendrawan et al. (2025) demonstrated that exposure to noise during gestation elevated oxidative markers, such as SOD, and altered progesterone levels, reinforcing the vulnerability of hormonal regulation under stress. Curcumin and its nanoformulations have shown promising outcomes in modulating antioxidant responses and hormonal balance. Jakubczyk et al. (2020b) confirmed the systemic antioxidant effects of curcumin across various clinical conditions, while Tossetta et al. (2021) highlighted its multifaceted benefits in pregnancy outcomes. The nanocurcumin formulation offers, a therapeutic edge in mitigating oxidative damage and endocrine dysregulation, potentially via suppression of aromatase and redox-sensitive signaling pathways (Hajar et al., 2023; Kokkarachedu et al., 2024). Furthermore, maternal oxidative stress is increasingly recognized as a key driver of impaired fetal development and placental dysfunction (Grzeszczak et al., 2023), underscoring the need for targeted antioxidant strategies, such as nanocurcumin, to preserve gestational homeostasis. In conclusion, the findings support the protective role of nanocurcumin against pregnancy-related oxidative stress and hormonal imbalance, likely through redox and steroidogenic pathway modulation. Future research should focus on confirming these mechanisms, assessing maternal–fetal safety, and evaluating long-term reproductive outcomes to support the therapeutic potential of nanocurcumin during gestational stress. To the best of our knowledge, this study is among the first to investigate the protective effects of nanocurcumin on both oxidative stress and estrogen regulation in a noise-induced pregnancy stress model. While several studies have explored the antioxidant roles of curcumin, few have examined nanoparticle formulations in the context of prenatal environmental stress. Thus, our findings contribute to the growing interest in the use of targeted antioxidant therapies for maternal-fetal health. ConclusionExposure to 135 dB noise during pregnancy induced oxidative and hormonal disturbances, as reflected by increased serum MDA and estrogen levels. Nanocurcumin treatment dose-dependently attenuated these effects, with the highest dose providing the strongest protective response. These findings support the potential role of nanocurcumin as an antioxidant candidate for mitigating stress-related biochemical alterations during pregnancy. However, further research is needed to elucidate the underlying molecular pathways, evaluate their interaction with steroidogenic and oxidative regulatory mechanisms, and assess maternal–fetal safety and pregnancy outcomes. Such studies are essential before nanocurcumin can be considered for broader therapeutic application. AcknowledgmentsThe authors would like to express their gratitude to the Faculty of Veterinary Medicine, Brawijaya University, for providing laboratory facilities and technical support. Special thanks are extended to Dr. Viski Fitri Hendrawan, M.Vet, for his supervision and valuable guidance, as well as Drh. Tiara Widyaputri, M.Si., and Drh. Galuh Chandra Agustina, M.Si., for their constructive feedback during the preparation of this study. Conflict of interestThe authors declare no conflict of interest regarding the publication of this paper. FundingThe Faculty of Veterinary Medicine, Brawijaya University (FKH UB), Indonesia, funded this research through the DPP SPP internal grant. Authors’ contributionsQatrinnada Hazimah performed the experimental work, collected the data, and prepared the first draft of the manuscript. Viski Fitri Hendrawan supervised the study design, guided the research process, and critically revised the manuscript. Tiara Widyaputri contributed to the laboratory experiments and assisted in data interpretation. Galuh Chandra Agustina was involved in the statistical analysis and provided academic input for manuscript improvement. All authors have read and approved the final version of the manuscript. Data availabilityAll data supporting this study’s findings are included within the manuscript. Additional datasets generated and analyzed during the current study are available upon reasonable request from the corresponding author. ReferencesAl Shammary, Z.A.A., Hameid, S.A. and Jouda, J. 2023. Relationship between noise stress effects and neurotransmitters and sex hormone levels in male and female Balb/c mice. Al. Mustansiriyah. J. Sci. 33(5), 88–94; doi: 10.23851/mjs.v33i5.1321 Alizadeh, M. and Kheirouri, S. 2019. Curcumin reduces malondialdehyde and improves antioxidants in humans with diseased conditions: a comprehensive meta-analysis of randomized controlled trials. BioMedicine 9(4), 23; doi:10.1051/bmdcn/2019090423 Ayala, A., Muñoz, M.F. and Argüelles, S. 2014. Lipid peroxidation: malondialdehyde production, metabolism, and signaling mechanisms of malondialdehyde and 4 hydroxy nonenal. Oxidative Med Cell Longev 2014, 360438; doi:10.1155/2014/360438 Baradaran, S., Moghaddam, A., Khanjani Jelodar, S. and Moradi Kor, N. 2020. Protective effects of curcumin and its nanophytosome on carrageenan-induced inflammation in a mouse model: behavioral and biochemical responses. J. Inflammation. Res. 13, 45–51; doi:10.2147/JIR.S232462 Bateni, Z., Behrouz, V., Rahimi, H.R., Hedayati, M., Afsharian, S. and Sohrab, G. 2022. Effects of nanocurcumin supplementation on oxidative stress, systemic inflammation, adiponectin, and NF κB in patients with metabolic syndrome: a randomized, double-blind clinical trial. J. Herbal. Med. 31, 100531; doi:10.1016/j.hermed.2021.100531 Brouček, J. 2014. Effect of noise on animal performance, stress, and behavior. Slovak J. Anim. Sci. 47(2), 111–123. Chen, Y., Zhao, Y., Lin, H. and Wang, J. 2024. Nanocurcumin regulates estrogen synthesis by modulating the PI3K/Akt and NF-κB pathways in stressed reproductive tissues. J. Reproductive Biochem. 18(2), 112–121; doi:10.1016/j.jrb.2024.02.008 Filardi, T., Varì, R., Ferretti, E., Zicari, A., Morano, S. and Santangelo, C. 2020. Curcumin: could this compound be useful in pregnancy and pregnancy-related complications?. Nutrients 12, 3179; doi:10.3390/nu12103179 Grzeszczak, R., Słowińska, M. and Baranowska, A. 2023. Maternal oxidative stress and placental dysfunction: a review of the literature. Placenta 141, 1–8; doi:10.1016/j.placenta.2023.01.005 Hajar, H., Shojaei, M., Askari, G., Majeed, M., Bagherniya, M., Barreto, G.E. and Sahebkar, A. 2023. Impact of curcumin on migraine: a comprehensive review. Biomed. Pharmacotherapy. 164, 114910; doi:10.1016/j.biopha.2023.114910 Hendrawan, V.F., Mariyam, D. and Al Ichsan, I.F.O.N. 2025. Effect of nanocurcumin administration on superoxide dismutase and progesterone level exposure to noise stress during pregnancy in mice: a meta-analysis. Open Vet. J. 15(3), 1239–1243; doi:10.5455/OVJ.2025.v15.i3.15 Jafari, Z., Mehla, J., Kolb, B.E. and Mohajerani, M.H. 2017. Prenatal noise stress impairs the HPA axis and cognitive performance in mice. Scientific. Rep. 7(1), 10560; doi:10.1038/s41598-017-09799-6 Jakubczyk, K., Kochman, J., and Skonieczna-Żydecka, K. 2020a. Curcumin and its potential therapeutic properties in inflammatory diseases. Antioxidants (Basel), 9(5), 454; doi:10.3390/antiox9050454 Jakubczyk, K., Drużga, A., Katarzyna, J. and Skonieczna-Żydecka, K. 2020b. Antioxidant potential of curcumin—a meta-analysis in randomized clinical trials. Antioxidants. (Basel). 9(11), 1092; doi:10.3390/antiox9111092 Kokkorachedu, A., Varaprasad, K., Jayaramudu, T. and Sadiku, R. 2024. Emerging trends in nanocurcumin: a novel therapeutic against inflammation and oxidative stress. Pharmacol. Rep. 76(1), 112–126; doi:10.5603/PHARMREP.a2024.0012 Mendoza, N., Salvatore, D., Li, M. and Choi, H. 2023. Maternal oxidative imbalance impairs fetal development: role of malondialdehyde and antioxidant defense in rodent models. Placenta. Develop. Biol. 42, 64–72; doi:10.1016/j.placdevbio.2023.05.005 Parisi, F., Fenizia, C., Introini, A., Zavatta, A., Scaccabarozzi, C., Biasin, M. and Savasi, V. 2023. Role of Estrogens in the Initial Stages of Pregnancy: molecular Mechanisms and Clinical Implications. Hum. Reprod. Update. 29, 699–720. Rohmah, N., Ondho, Y.S. and Samsudewa, D. 2017. Pengaruh weberian pakan flushing dan non-flushing terada intensity Ibrahim dan angka kebuntingan induk sapi potong. Jurnal. Sain. Peternakan. Indonesia. 12(3), 290–298; doi:10.31186/jspi.id.12.3.290-298 Shafiei, A., Ehteram, H., Akbari, H., Kashani, M., Beigi, M., Mahabadi, J.A. and Mazoochi, T. 2017. Effect of chronic noise stress on serum cortisol, gonadotropins, and sexual hormones in mice at the time of implantation. Comparative. Clin. Pathol. 26(4), 823–829; doi: 10.1007/s00580-017-2446-y Subandi., Sari, L.I. and Nurdiana. 2025. Effect of nanocurcumin administration on plasma malondialdehyde (MDA) levels in pregnant Wistar rats as a model of preeclampsia. Majalah Kesehatan Indonesia 6(1), 35–42. Supriyanto, S., Pramu, P. and Ahadiati, N. 2019. Ultrasonografi perkembangan folikel ovaria selama siklus estrus dan kebuntingan awal pada sapi Peranakan Ongole. Jurnal Pengembangan Penyuluhan Pertanian 13(23), 82; doi:10.36626/jppp.v13i23.116 Tanaka, K., Fujimoto, R. and Yamazaki, T. 2025. Chronic exposure to noise disrupts endocrine signaling and increases reproductive risk in murine pregnancy models. Environ. Reproductive Toxicol. 9(1), 21–33; doi:10.1016/j.envretox.2025.01.003 Tossetta, G., Fantone, S., Giannubilo, S.R. and Marzioni, D. 2021. The Multifaced Actions of Curcumin in Pregnancy Outcomes. Antioxidants 10(126), 126; doi:10.3390/antiox10010126 Varaprasad, K., Sisubalan, N., Jayaramudu, T. and Yallapu, M.M. 2024. Nanocurcumin: a new and improved way to fight cancer and infections. Nano-Structures & Nano-Objects 40, 101352; doi:10.1016/j.nanoso.2024.101352 | ||
| How to Cite this Article |
| Pubmed Style Hendrawan VF, Hazimah Q, Widyaputri T, Agustina GC, Rahmawati D, Lisnanti EF. Effects of nanocurcumin administration on malondialdehyde and estrogen levels in mice exposed to noise-induced stress. Open Vet. J.. 2026; 16(1): 445-451. doi:10.5455/OVJ.2026.v16.i1.41 Web Style Hendrawan VF, Hazimah Q, Widyaputri T, Agustina GC, Rahmawati D, Lisnanti EF. Effects of nanocurcumin administration on malondialdehyde and estrogen levels in mice exposed to noise-induced stress. https://www.openveterinaryjournal.com/?mno=286107 [Access: January 31, 2026]. doi:10.5455/OVJ.2026.v16.i1.41 AMA (American Medical Association) Style Hendrawan VF, Hazimah Q, Widyaputri T, Agustina GC, Rahmawati D, Lisnanti EF. Effects of nanocurcumin administration on malondialdehyde and estrogen levels in mice exposed to noise-induced stress. Open Vet. J.. 2026; 16(1): 445-451. doi:10.5455/OVJ.2026.v16.i1.41 Vancouver/ICMJE Style Hendrawan VF, Hazimah Q, Widyaputri T, Agustina GC, Rahmawati D, Lisnanti EF. Effects of nanocurcumin administration on malondialdehyde and estrogen levels in mice exposed to noise-induced stress. Open Vet. J.. (2026), [cited January 31, 2026]; 16(1): 445-451. doi:10.5455/OVJ.2026.v16.i1.41 Harvard Style Hendrawan, V. F., Hazimah, . Q., Widyaputri, . T., Agustina, . G. C., Rahmawati, . D. & Lisnanti, . E. F. (2026) Effects of nanocurcumin administration on malondialdehyde and estrogen levels in mice exposed to noise-induced stress. Open Vet. J., 16 (1), 445-451. doi:10.5455/OVJ.2026.v16.i1.41 Turabian Style Hendrawan, Viski Fitri, Qatrinnada Hazimah, Tiara Widyaputri, Galuh Chandra Agustina, Dwi Rahmawati, and Ertika Fitri Lisnanti. 2026. Effects of nanocurcumin administration on malondialdehyde and estrogen levels in mice exposed to noise-induced stress. Open Veterinary Journal, 16 (1), 445-451. doi:10.5455/OVJ.2026.v16.i1.41 Chicago Style Hendrawan, Viski Fitri, Qatrinnada Hazimah, Tiara Widyaputri, Galuh Chandra Agustina, Dwi Rahmawati, and Ertika Fitri Lisnanti. "Effects of nanocurcumin administration on malondialdehyde and estrogen levels in mice exposed to noise-induced stress." Open Veterinary Journal 16 (2026), 445-451. doi:10.5455/OVJ.2026.v16.i1.41 MLA (The Modern Language Association) Style Hendrawan, Viski Fitri, Qatrinnada Hazimah, Tiara Widyaputri, Galuh Chandra Agustina, Dwi Rahmawati, and Ertika Fitri Lisnanti. "Effects of nanocurcumin administration on malondialdehyde and estrogen levels in mice exposed to noise-induced stress." Open Veterinary Journal 16.1 (2026), 445-451. Print. doi:10.5455/OVJ.2026.v16.i1.41 APA (American Psychological Association) Style Hendrawan, V. F., Hazimah, . Q., Widyaputri, . T., Agustina, . G. C., Rahmawati, . D. & Lisnanti, . E. F. (2026) Effects of nanocurcumin administration on malondialdehyde and estrogen levels in mice exposed to noise-induced stress. Open Veterinary Journal, 16 (1), 445-451. doi:10.5455/OVJ.2026.v16.i1.41 |