| Review Article | ||
Open Vet. J.. 2025; 15(12): 6126-6145 Open Veterinary Journal, (2025), Vol. 15(2): 6126-6145 Review Article Unveiling the significance of dog domestication in cognitive dysfunction: Are wolves protected?Rafael Castro-Fuentes1* and Rosy Socas-Pérez21Department of Basic Medical Sciences, School of Health Sciences, Section Medicine, University of La Laguna, San Cristóbal de La Laguna, Spain 2Rehabilitation Service, Canary Islands University Hospital Complex (CHUC), Santa Cruz de Tenerife, Spain *Corresponding Author: Rafael Castro-Fuentes. Department of Basic Medical Sciences, School of Health Sciences, Section Medicine, University of La Laguna, San Cristóbal de La Laguna, Spain. Email: jrcastro [at] ull.edu.es; jrafacastro [at] gmail.com Submitted: 16/09/2025 Revised: 01/11/02025 Accepted: 05/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
AbstractOver the course of their long coexistence with humans, dogs have developed a stronger bond with humans than with any other domestic species. This close relationship has promoted notable parallels in both genetics and lifestyle, thereby facilitating the development of comparable pathological conditions, including several central nervous system disorders. Canine cognitive dysfunction (CCD) is a spontaneous model of neurodegeneration that shares clinical features, neuropathological characteristics, and risk factors with human Alzheimer’s disease (AD). However, the potential role of dog domestication in increasing CCD susceptibility remains poorly explored. In this sense, the gray wolf (Canis lupus), a direct ancestor of the domestic dog (Canis lupus familiaris), represents a comparative model of great interest, as the information available on its neuropathology and behavior associated with aging is very limited. To provide a preliminary framework for assessing how domestication may have shaped vulnerability to cognitive decline in canids, this study employed a database-driven analytical approach to evaluate the degree of impact of various risk factors shared by AD and CCD—including aging, oxidative stress, inflammation, sleep disturbances, and periodontal disease—in domestic dogs and gray wolves kept in captivity or semi-captivity. Our results indicate that domestic dogs have more pronounced key risk factors for CCD than captive or semi-captive gray wolves. This finding suggests that wolves may be less vulnerable to age-related cognitive dysfunction, possibly reflecting differences in evolutionary or domestication processes. Nevertheless, given the scarcity of neuropathological and behavioral data on aged wolves, these conclusions should be interpreted with caution, and further direct investigations are warranted. Domestication may have increased susceptibility to age-related cognitive dysfunction in dogs by enhancing exposure to key risk factors, as oxidative stress, inflammation, and lifestyle-related conditions. Recognizing these domestication-linked vulnerabilities highlights the need for preventive health strategies in dogs and provides a valuable comparative framework for understanding neurodegenerative processes across species within a One Health perspective. Keywords: Alzheimer’s disease, Cognitive dysfunction, Dog, Gray wolf, Risk factors. IntroductionMolecular, morphological, and behavioral analyses have established that domestic dogs (Canis lupus familiaris) are derived from gray wolves (Canis lupus) (Freedman and Wayne, 2017). As the earliest known domesticated species, dogs have dispersed globally alongside humans, occupying diverse habitats and ecological niches. This prolonged coexistence has fostered a uniquely close interspecies relationship compared with other domesticated animals (Freedman and Wayne, 2017). Genomic studies indicate two primary demographic bottlenecks during canine evolution: an initial domestication event involving a limited subset of regional wolf populations, followed by the selective development of modern breeds from these ancestral populations (Freedman and Wayne, 2017; Bergström et al., 2022). The latter process, which has largely occurred within the last few centuries, has yielded roughly 350–400 formally recognized breeds (Parker et al., 2017; Plassais et al., 2019). Demographic and genomic analyses indicate that initial domestication was associated with a marked reduction in effective population size (a strong early bottleneck) and that intensive breed formation over the past ~200–300 years produced a further substantial loss of genetic diversity and increased runs of homozygosity in many modern breeds (Plassais et al., 2019; Axelsson et al., 2021; Meadows et al., 2023). Domestication has also induced a constellation of phenotypic modifications that distinguish domestic dogs from their wild ancestors. These include enhanced tameness, craniofacial shortening, reduced dentition, higher reproductive frequency with attenuated seasonality, diminished sexual dimorphism, alterations in tail and ear morphology (e.g., pendulous ears), coat and skin depigmentation, hair loss, shifts in neuroendocrine parameters, neotenous behavioral traits, and reductions in overall brain volume as well as in discrete brain regions (Wilkins et al., 2014; Pendleton et al., 2018). Traits collectively referred to as the “domestication syndrome” are thought to emerge from selective pressures favoring reduced aggression in mammals, potentially modifying the expression of genes that govern neural crest cell development (Wilkins et al., 2014). Domestic dogs are one of the most compelling examples supporting this framework (Sánchez-Villagra et al., 2016; Pendleton et al., 2018). Accumulating data indicate that the process of domestication in both plant and animal species results in significant genomic modifications compared with their wild ancestors (Cruz et al., 2008; Freedman et al., 2016). Furthermore, domesticated populations often harbor a greater load of harmful alleles than their wild relatives. Notably, the nuclear DNA of domesticated dogs contains a higher proportion of non-synonymous mutations near functionally important regions than that of gray wolves (Marsden et al., 2016; Cagan and Blass, 2016; Bosse et al., 2018). In fact, 25.3% of nonsynonymous mutations are predicted to be functionally harmful for dogs (Henn et al., 2015). Breed dogs carry an estimated 320 additional homozygous genotypes that result in amino acid changes, which corresponds to roughly a 22% increase compared with wolves (Marsden et al., 2016). These variants illustrate the genetic costs associated with domestication. In domestic populations, the occurrence of deleterious mutations is strongly positively correlated with linkage disequilibrium and negatively correlated with nucleotide diversity, highlighting the reduced effectiveness of purifying selection (Gray et al., 2009; Hou et al., 2020). In addition to nonsynonymous protein alterations, other forms of genetic variation have been identified in dogs (Makino et al., 2018; Plassais et al., 2019). The accumulation of harmful variants has also been observed in the mitochondrial genome of domestic dogs (Tkaczyk-Wlizlo et al., 2022). Interestingly, positive selection acted on genes involved in neurological processes in both humans and canines. Genes involved in canine neurological processes extensively overlap with these genes in humans (Wang et al., 2013). Dogs coexist with humans, inhabit the same spaces, breathe the same air, eat similar foods, and are exposed to the same environmental conditions, including air pollutants, household chemicals, pesticides, heavy metals, flame retardants, and other toxicants. This shared environment strengthens the species’ translational relevance, as dogs serve as sentinels reflecting human exposure and its biological consequences (Craun et al., 2020; Wise et al., 2020). Chronic exposure to environmental contaminants in urban settings has been associated with oxidative stress, neuroinflammation, DNA damage, and early neuropathological changes in canine brains, resembling those seen in human neurodegenerative disorders (Calderon-Garciduenas et al., 2003). Thus, the close genetic, physiological, and environmental parallels between dogs and humans make the dog an exceptional model for exploring how gene–environment interactions contribute to neurological diseases. The close interactions between humans and canines and the drastic changes in the living environments of canines may have caused some of the striking parallelism between human and canine genetic evolution (Huang et al., 2020). Aggressive dog breeding practices involve high levels of inbreeding and have intensified minor morphological differences among breeds. Many purebred dogs exhibit elevated rates of breed-specific inherited disorders. Remarkably, many of these diseases and pathological conditions are also common in humans, particularly age-related disorders affecting organs such as the brain, kidneys, and cardiovascular system. The clinical manifestations observed in dogs often mirror those in humans, making dogs a valuable translational model for studying complex and age-associated diseases. Comparative genomic analyses further demonstrate that dogs and humans share a highly similar gene repertoire, with many orthologous genes implicated in analogous diseases (Hytönen and Lohi, 2016; Marsden et al., 2016; Hoffman et al., 2018; Plassais et al., 2019; Axelsson et al., 2021; Ruple et al., 2022; Donner et al., 2018). When wolves began their transformation into the first dogs, artificial selection acted strongly on genes expressed in the brain, promoting the evolution of behavior patterns characteristic of domestic dogs (Li et al., 2013). To gain broader insight into how domestication drives behavioral changes, researchers analyzed genomic and transcriptomic data from chickens and six other domesticated mammals, including dogs (Hou et al., 2020). These studies revealed overlaps between the molecular foundations of animal behavior and human mental health conditions (Shpigler et al., 2017; Vaz et al., 2018). The results further showed that many of the genes recurrently shaped by selection were deeply conserved across species and shared links with human neuropsychiatric disorders, highlighting a compromise: adaptation to human environments was achieved at the expense of neural alterations that may reduce fitness in natural habitats (Hou et al., 2020). Genetic studies suggest that during domestication, neurobiological processes have been targeted by selection (O'Rourke and Boeckx, 2020; Benítez-Burraco et al., 2023). This selection has involved genes related to neurological processes, behavior, and synapse activity. Using brain samples, several differentially metilated regions have been found in genes (epigenetic factors) related to neurotransmission between domestic dogs and their ancestors, providing further evidence that domestication has targeted neurological functions (Sundman et al., 2020). Natural selection does not always efficiently eliminate harmful variants; therefore, pleiotropic genes can remain in the genome, increasing the likelihood of disease. A classic example is the strong association between coat color and sensorineural deafness, which has been reported in many dog breeds (Webb and Cullen, 2010). Brachycephaly, characterized by shortened facial features, is another widespread condition that appears in various breeds (Dupré and Heidenreich, 2016). Beyond external traits, humans and dogs share a predisposition to nearly 360 illnesses, including compulsive behaviors, epilepsy, and Alzheimer’s-like syndromes, many of which involve the nervous system (Shearin and Ostrander, 2010; De Risio et al., 2015; Ostrander et al., 2017; Dewey et al., 2019). The shared social and environmental contexts of both species may account for these parallels (Zhang et al., 2020). Alzheimer’s disease is a progressive disorder of the nervous system that results in global cognitive deterioration and gradual memory loss. It is the principal cause of dementia and is increasingly recognized as one of the most lethal and economically demanding illnesses of this century (Scheltens et al., 2021). Although many studies have aimed to identify treatments for AD, therapies that effectively modify disease progression are still lacking, partly because the disease’s underlying biology remains incompletely understood (Klein et al., 2016). Age-related cognitive deterioration is also observed in dogs as part of the normal aging process. However, similar to humans, a proportion of elderly dogs develop a specific dementia-like syndrome known as CCD or canine dementia (Dewey et al., 2019). CCD exhibits many of the same signs seen in human AD dementia (Youssef et al., 2016; Mihevc and Majdič, 2019; Kim et al., 2024). Affected dogs may experience confusion, anxiety, memory impairments, difficulty learning, altered social behaviors, disrupted sleep-wake cycles, and changes in activity, along with structural alterations such as brain atrophy. Neuropathologically, CCD involves intracellular Aβ oligomers, extracellular amyloid plaques, and amyloid accumulation around blood vessels, particularly in the frontal lobes and hippocampus. Neuron loss projecting to the hippocampus, microbleeds, microgliosis, and intraneuronal hyperphosphorylated tau (pTau) have also been observed (Schmidt et al., 2015; Habiba et al., 2021). Some studies have reported that pTau can form neurofibrillary tangles in aged dogs, typically over 10 years of age, although this may vary according to breed, size, and study design (Schmidt et al., 2015; Smolek et al., 2016). Similar to humans, tau pathology in dogs spreads through the entorhinal cortex, hippocampus, and neocortex (Smolek et al., 2016; Davidson et al., 2018; Abey et al., 2021; Yushkevich et al., 2021), contributing to CCD’s cognitive and behavioral impairments (Mihevc and Majdič, 2019). Notably, CCD develops spontaneously in dogs across diverse genetic backgrounds, with all animals carrying the AD risk allele APOE4 in homozygous form (Bunyaluk et al., 2022). This combination of features has fostered growing interest in using CCD as a large-animal model for AD, as it presents a dementia-like condition that mirrors key aspects of human AD (Schütt et al., 2016; Mihevc and Majdič, 2019; Ruple et al., 2022). In this context, we propose the following hypothesis: if the domestication of the dog contributed to the development of CCD, then its wild conspecifics, the gray wolves, should be more protected from the signs of such pathology. To provide a preliminary framework for assessing how domestication may have shaped vulnerability to cognitive decline in canids and given the scarce availability in the scientific literature (both of cerebral neuropathological studies and of behaviors related to the CCD) about wolves, this study employed the approach of evaluating the degree of impact of several key risk factors (shared by AD and CCD) in domestic dogs and gray wolves kept in captivity or semi-captivity. MethodologyThis narrative review adopted an analytical and comparative approach to examine the potential link between dog domestication and increased susceptibility to CCD compared with the gray wolf. A literature search was performed in PubMed, Scopus, and Google Scholar covering 2015–2025, using combinations of terms related to domestication, species, key risk factors (aging, oxidative stress, inflammation, sleep disturbances, and periodontal disease), and cognitive decline. Because comparative neuropathological and behavioral data on gray wolves are scarce, several seminal studies published before 2015 were also included to provide relevant foundational evidence on key mechanisms. Eligible studies addressed at least one of the selected risk factors and reported neurological or behavioral findings in domestic dogs and captive or semi-captive gray wolves. Data were thematically synthesized and qualitatively compared to identify similarities and differences in the expression and impact of risk factors between species. AgingAge is widely recognized as a significant risk factor for Alzheimer’s disease in humans and for CCD in dogs (Azkona et al., 2009; Mihevc and Majdič, 2019; Yarborough et al., 2022; Liu et al., 2024). Interestingly, research suggests that the process of domestication has a stronger impact on brain regions involved in homeostatic regulation—thereby influencing senescence and aging—while exerting comparatively less influence on neural circuits associated with emotions and behaviors typical of ancestral canids (Mazzatenta et al., 2017). CCD is considered one of the leading contributors to morbidity in geriatric dogs. Despite its high prevalence, the syndrome remains markedly underdiagnosed, resulting in a substantial proportion of older adult dogs being affected without recognizing the condition (Mihevc and Majdič, 2019). Similar to the situation in humans with AD, the prevalence of CCD increases exponentially as dogs age (Yarborough et al., 2022). CCD is present in approximately 28% of dogs aged 11–12 years, with a prevalence that increases to up to 68% in dogs aged 15–16 years (Chapagain et al., 2018; MacQuiddy et al., 2022). Recent large-scale studies, such as those from the Dog Aging Project, have confirmed that the prevalence of CCD rises markedly with age, showing a strong age-related effect independent of breed or sex (Urfer et al., 2021; Yarborough et al., 2022; Bray et al., 2023) and that there are no breed-specific differences in the clinical signs or pathology of CCD (Mihevc and Majdič, 2019). Because large-breed dogs generally exhibit shorter lifespans than their smaller counterparts (Turcsán and Kubinyi, 2024), the clinical manifestations of CCD are more frequently identified in small-sized breeds (Schmidt et al., 2015) as well as in purebred populations (Turcsán and Kubinyi, 2024). Purebred dogs appear to have a higher susceptibility to CCD than crossbred animals. One possible explanation is that mixed-breed dogs typically enjoy greater longevity across all body size categories, which has been linked to reduced levels of inbreeding and, consequently, a lower probability of homozygosity for deleterious alleles (Donner et al., 2018; Yordy et al., 2020). This helps mixed-breed dogs maintain their cognitive health for longer periods (Donner et al., 2018). Current evidence linking the domestication of dogs with aging processes remains limited. Nevertheless, Holecková and Chvapil demonstrated that domestication can negatively influence both physiological development and aging in laboratory rats. In comparison to domestic rats (Rattus norvegicus), wild Norway rats displayed several distinct traits: shorter latency of tissue explants, reduced structural stability of collagen fibers, heightened excitability of the central nervous system, prolonged reproductive capacity, improved tolerance to cold exposure, and a lower prevalence of spontaneous tumor formation (Holečková and Chvapil, 1965). In wild canids, such as wolves, senescence is also observed, but some age-related traits may enhance survival and fitness, thus persisting under natural selection (MacNulty et al., 2009; Charruau et al., 2016; Schwarz et al., 2016). In contrast, the manifestation of these ancestral aging patterns within highly modified environments may become maladaptive in domestic dogs and contribute to disease susceptibility (Charruau et al., 2016). Likewise, gene expression patterns that evolved under a hunter-gatherer lifestyle in humans may lead to health problems for individuals with a Western lifestyle and diet (Kopp 2019; Johnson et al., 2021). Consequently, such legacy effects may result in disease in aging individuals for both modern humans and their domestic companions (Charruau et al., 2016). Several ecological studies have indicated that it is unusual for wild gray wolves to reach or surpass 10 years of age (Lawler et al., 2016; Barber-Meyer et al., 2021). However, age-associated pathologies, which are often observed in domestic or protected settings, are rare in natural environments. Factors such as frailty, nutritional stress, and predation dominate mortality patterns, resulting in a considerably shorter average lifespan in free-ranging populations (Tidière et al., 2016). These survival data align with the view that late-life stages are infrequently attained in wild wolves. This is particularly relevant given that the onset of increased risk for CCD and the escalation in symptom severity have been reported to occur from approximately 10 years of age onward (Bray et al., 2023; Turcsán and Kubinyi, 2024). There is considerable evidence that the processes leading to this clinical state begin earlier in life (Dewey et al., 2019). Accordingly, it would be difficult to find signs of CCD in free-roaming wolves because they would not have enough time to develop such a pathology. However, the older wolves expressed a capacity for longevity that exceeded those of similar-sized domestic dogs and breeds (Lawler et al., 2016). In contrast to free-ranging populations, evidence from captive or sanctuary settings indicates that gray wolves can achieve considerably longer lifespans, ranging from approximately 9–16 years (Mech, 1988). For instance, an investigation that assessed scapular glenoids and proximal humeral joints from wolves housed in a sanctuary environment reported mortality at ages ranging from 5 to 19 years (Lawler et al., 2016). The presence of individuals surviving to such advanced ages clearly supports the genetic potential of this species to reach extended longevity. Indeed, gray wolves appear capable of living as long as domestic dogs when not exposed to the challenges of a free-roaming existence. Additionally, aged gray wolves demonstrated a lifespan potential that, in some instances, surpassed that observed in domestic dog breeds of comparable body size (Lawler et al., 2016). Therefore, gray wolves kept in captivity or semi-captivity can be studied from the point of view of the onset and development of canine cognitive dysfunction, in comparison with domestic dogs, according to their longevity, and that certain age-related traits might improve survival and reproductive success (remaining present through natural selection). Oxidative stressAn imbalance between free radical overproduction and the antioxidant defenses of the brain defines oxidative stress. This disturbance is widely recognized as a contributor to the aging process (Iakovou and Kourti, 2022; Maldonado et al., 2023). The brain is especially prone to oxidative injury because it demands about one-fifth of the body’s oxygen supply, is enriched in polyunsaturated fatty acids, and exhibits relatively weak endogenous antioxidant protection when compared to other tissues (Lushchak et al., 2021; Sienes Bailo et al., 2022). Small-breed dogs may generate reactive oxygen species at elevated levels owing to their higher metabolic intensity and rapid developmental pace (Jimenez, 2016). Circulating lipid peroxidation is reported to be more pronounced in small breeds than in large breeds (Middleton et al., 2017; Jimenez and Downs, 2020). By contrast, large-breed dogs remain in a growth phase for a more extended period (Jimenez, 2021). Interestingly, while no measurable elevation of circulating antioxidants has been observed in small breeds (Jimenez and Downs, 2020), they tend to outlive large breeds and display a lower incidence of cancer (Jimenez, 2016; Middleton et al., 2017). Nevertheless, smaller-sized dogs have a higher CCD risk prevalence than larger dogs (Turcsán and Kubinyi, 2024). Naked mole-rats (NMRs, Heterocephalus glaber) are the longest-lived rodents with a maximum life span exceeding 37 years (Oka et al., 2023). As shown in dogs, antioxidant enzyme activities in NMRs do not change with age, unlike mice of similar size, in which some antioxidant enzymes decline with age (Jimenez and Downs, 2020; Buffenstein Amoroso et al., 2022). However, similar to small-breed dogs, NMRs also exhibit higher levels of lipid peroxidation (Jacobs et al., 2023). NMRs exhibit a delayed aging phenotype and resistance to age-related functional decline or diseases (Oka et al., 2023). The artificial selection of domestic dogs seems to have favored traits unrelated to the conservation of antioxidant defenses. Other authors have found that the levels of total antioxidants, such as bilirubin and glutathione metabolites, are significantly lower in small-breed domestic dogs than in large breeds (Jiménez, 2024). Oxidative stress has been strongly linked to brain aging and a higher susceptibility to AD in humans (Ionescu-Tucker and Cotman, 2021; Tchekalarova and Tzoneva, 2023). Patients with AD undergo pronounced oxidative insults during disease progression, accompanied by a marked decline in endogenous antioxidant defenses compared with individuals of similar age without AD (Isik et al., 2023; Jain et al., 2024). Comparable patterns are seen in CCD, where oxidative lesions are strongly linked to impaired cognition (Romanucci and Della Salda, 2015; Pérez-Montero et al., 2024). The accumulation of oxidative by-products parallels the severity of behavioral impairments in elderly companion dogs (Pan, 2021; Pérez-Montero et al., 2024). Furthermore, reductions in brain and plasma antioxidant reserves are correlated with the progression of cognitive deficits (Mongillo et al., 2015). Therapeutic strategies that include antioxidant diets and behavioral enrichment can result in significant improvements (Mazzatenta et al., 2017; Blanchard et al., 2025). It is important to note that oxidative stress patterns in the blood of wild canids are different from those found in domestic dogs. Recent investigations have shown that lipid peroxidation markers increase with advancing age in domestic dogs, whereas this pattern is not observed in wild canids housed in zoological facilities or under veterinary care. In contrast, wild canids demonstrate an age-related increase in total antioxidant capacity (TAC) and glutathione peroxidase (GPx) activity scales with age and maximum lifespan (Jimenez and Downs, 2020). Such physiological adaptations may contribute to the greater longevity potential of wolves. In addition, larger-bodied wild canids exhibited significantly higher TAC and GPx activity, a trend not evident in domestic dogs, suggesting that selective breeding may have diminished antioxidant defenses in the latter (Jimenez and Downs, 2020). A broader comparative study assessing oxidative stress across 48 mammalian species similarly reported that older individuals often accumulate lipid damage (Jimenez et al., 2019). This outcome parallels findings in small domestic dogs but contrasts with the resilience observed in wild canids (Jimenez and Downs, 2020). The capacity of large wild species, such as the gray wolf, to enhance antioxidant defenses with age could partly explain their extended lifespan relative to domestic breeds of comparable size, representing a distinctive physiological feature not commonly seen in other mammals (Jimenez et al., 2019; Jimenez and Downs, 2020). Comparable results have been noted in NMRs, which maintain stable antioxidant levels throughout aging and exhibit lifespans that far exceed those of rodents of similar body size (Jacobs et al., 2023). The preservation of antioxidant defenses in NMRs is consistent with delayed senescence, as these animals display negligible age-related mortality, rarely develop cancer, and show resistance to Alzheimer’s-like pathologies (Oka et al., 2023). Other aging model organisms typically demonstrate that longer lifespans are associated with higher levels of antioxidants and reduced oxidative damage to most molecules (Jimenez, 2016; Domínguez de Barros et al., 2023; Domínguez-De-Barros et al., 2025). It is important to note that the degree of oxidative stress is also lower in healthy human centenarians than in aged subjects (Belenguer-Varea et al., 2020). Healthy centenarians have a particular profile in which high levels of antioxidants seem to be important for their extreme longevity (Martínez de Toda et al., 2020). The remarkable longevity of human centenarians is linked to enhanced resistance and resilience to Alzheimer’s disease (Andersen, 2020). In contrast, NMRs inhabit subterranean colonies in East Africa, where oxygen availability is limited, and they have evolved a notable tolerance to hypoxic conditions (Park and Reznick, 2022; Oka et al., 2023). In this context, recent comparative work has also highlighted pronounced differences in aerobic performance, hypoxia tolerance, and immune function when gray wolves are compared with domestic dogs (Yang et al., 2018). Aerobic capacity, response to hypoxia endurance training, and immune function were all significantly higher in gray wolves than in domestic dogs (Yang et al., 2018). All of the abovementioned characteristics, which are related to oxidative stress, may confer greater protection against CCD to the gray wolf compared to the domestic dog. InflammationInflammation is a constant biological process that occurs throughout the lifespan and is essential for defending the body against infectious agents. Nevertheless, the same response can harm cells, tissues, and organs and has been identified as a major contributor to the aging process (Li et al., 2023). This association has led to the concept of “inflammaging” which describes a persistent, low-grade inflammatory state that tends to increase with age (Calder et al., 2017; Franceschi et al., 2018; Saavedra et al., 2023). Accumulating evidence indicates that the interplay between aging and inflammation is highly context-dependent and may differ across species and physiological conditions. Thus, a recent study compared exhaustive data on inflammatory molecules from two industrialized populations (Singapore Longitudinal Aging Study-SLAS and Italian-InCHIANTI) and two non-industrialized indigenous populations (Bolivian Amazon’s Tsimane and Peninsular Malaysia’s Orang Asli) and observed clear links between inflammatory aging and chronic diseases in industrialized populations, unlike populations with high infection rates (no industrialized) in which inflammation seems to reflect more infectious diseases than aging itself (Franck et al., 2025). The evidence indicates that inflammatory processes are not uniform but can manifest through diverse mechanisms, showing variable associations with aging depending on the environmental exposures characteristic of a given population. Moreover, current data support the idea of an evolutionary imbalance between the immune system and modern ecological conditions (Franck et al., 2025). Thus, age-related inflammation may represent less of a natural outcome of senescence and more of an adaptive response to industrialized lifestyles and environments. Numerous evidence suggest that the domestic dog has evolved toward high dependence and sensitivity against industrialized human environments due to its history of domestication and adaptation to living with humans. This has led to the dog becoming more vulnerable to stress, diseases, and social changes compared with gray wolves in semi-captivity, which retain more resilient and adaptive behaviors outside human control (Marshall-Pescini et al., 2016; Wynne, 2021; Range and Marshall-Pescini, 2022; Tancredi and Cardinali, 2023). Day (2010) described that in domestic dogs, aging entails the elevation of low-degree systemic inflammation, a decrease in immune function, and greater susceptibility to chronic diseases (Day, 2010). Domestic dogs show increased spontaneous chronic inflammation with age (inflammaging) (Jiménez, 2023; Schmid et al., 2024). In this sense, domestic dogs experience an increase in the expression of chronic inflammation markers, for example, IL-6, TNF -α (Schmid et al., 2024), and IgM, oxidative damage, and C-reactive protein (CRP) (Alexander et al., 2018). Cerebral inflammation plays a crucial and multifactorial role in AD pathogenesis (Si et al., 2023; Thakur et al., 2023; Almutary et al., 2025). Far from being just a consequence of neurodegeneration, today it is recognized as an active process that contributes to the disease’s onset and progression (Twarowski and Herbert, 2023). Neuroinflammation arises from the activity of multiple cellular and molecular components, including astrocytes, microglial populations, and signaling proteins such as chemokines and cytokines. This inflammatory cascade disrupts the neuronal microenvironment, resulting in neuronal injury, oxidative stress, and, in some cases, the programed cell death, processes that have been strongly associated with AD onset (Fakhoury, 2020; Zhang et al., 2024). Chronic neuroinflammation is increasingly recognized as a central feature of age-related cognitive decline in dogs in veterinary medicine. Microglia, the innate immune cells of the brain, exhibit sustained activation in response to neuropathological alterations that occur with aging. Their persistent release of inflammatory mediators promotes neurodegenerative changes, which are closely associated with the clinical presentation of CCD (Pan et al., 2018; Prpar-Mihevc et al., 2020; Hines et al., 2023). Although classical systemic inflammatory markers (e.g., IL-6 or tumor necrosis factor a in plasma) have not been consistently found to be significantly elevated, multiple studies have found that central neuroinflammation (e.g., glial activation and accumulation of Aβ and phosphorylated tau) and increased neurofilament light chain (NfL) levels in the blood are robustly associated with cognitive decline in older dogs. These changes may reflect inflammatory processes in the brain that accompany or precede CCD, similar to that observed in AD (Pan et al., 2018; Vikartovska et al., 2021; Phochantachinda et al., 2021; Hines et al., 2023; Wu et al., 2023; Kim et al., 2024). No comparative studies have directly measured the levels of proinflammatory markers (IL-6, TNF-α, and CRP) in older domestic dogs and semi-captive gray wolves. However, some studies have described how domestic dogs develop greater chronic inflammation compared with captive gray wolves, suggesting that domestication may have influenced differential immunological responses (Marsden et al., 2016; Yang et al., 2018; Barragán-Sánchez et al., 2025). The lower levels of chronic inflammation observed in wolves, relative to domestic dogs, may indicate a lower susceptibility to age-related cognitive decline. However, given the limited data available on cognitive aging in wolves, such an interpretation should be regarded as preliminary. Further comparative studies are needed to determine whether reduced systemic inflammation indeed translates into lower CCD incidence or severity. Sleep disturbancesSleep is a vital physiological function in mammals, including domestic dogs. It contributes to the restoration of energy reserves, supports the stabilization and reorganization of memory processes, counteracts performance deficits resulting from wakefulness, and facilitates the clearance of potentially harmful metabolites that accumulate in the brain during periods of activity (Krueger et al., 2016; Kis et al., 2017; Frank and Craig Heller, 2019; Iotchev et al., 2020; Ji and Yun, 2025). Sleep is essential for preserving cognitive stability, and its relevance increases with aging, as the clearance of Aβ-closely associated with AD pathogenesis and occurs predominantly during sleep (Mondino et al., 2023). In mammals, the sleep-wake cycle is typically divided into three distinct phases: wakefulness (W), non-rapid eye movement sleep (NREM, also referred to as slow-wave sleep), and rapid eye movement sleep, REM (Mondino et al., 2021). Accumulating evidence shows that sleep disturbance contributes to cognitive decline and increases the risk of AD dementia by increasing the brain’s Aβ burden (Cedernaes et al., 2017; Shi et al., 2018; Ungvari et al., 2025). According to meta-analyses, both decreased NREM and REM were significantly associated with the severity of cognitive impairment in patients with AD (Zhang et al., 2022). Similar to humans and other mammals, dogs experience age-related changes in their sleep-wakefulness cycle (Woods et al., 2020; Mondino et al., 2023). CCD has been associated with significant alterations in sleep behavior in dogs (Dewey et al., 2019). Evidence from a longitudinal study indicated that sleep-wake rhythm disruptions were the most frequently observed clinical feature of the disorder (Fast et al., 2013). Consistent with findings in AD, dogs with higher dementia scores and poorer problem-solving performance spent less time in NREM and REM sleep. Recently, a non-invasive polysomnography protocol was applied to record the sleep of captive, extensively socialized young and adult wolves. The control population of companion dogs included both juvenile and geriatric individuals matched to the age categories of the corresponding wolf groups. The analysis indicated that while young dogs and wolves exhibited a comparable distribution of sleep stages, dogs spent a shorter duration in REM sleep. This difference was even more evident in senior animals (Reicher et al., 2022). REM sleep, the stage during which memory consolidation occurs (Mondino et al., 2023), is essential for maintaining neuronal homeostasis in the brain. Disturbances in REM sleep can impair neurogenesis and synaptic pruning, and prolonged reductions or loss of this sleep phase have been associated with the development of neurodegenerative changes (Chauhan and Mallick, 2019; Zhang et al., 2022). Increased REM sleep may contribute to the decreased vulnerability of gray wolves to CCD. However, the relationship between REM sleep duration and CCD remains correlative.
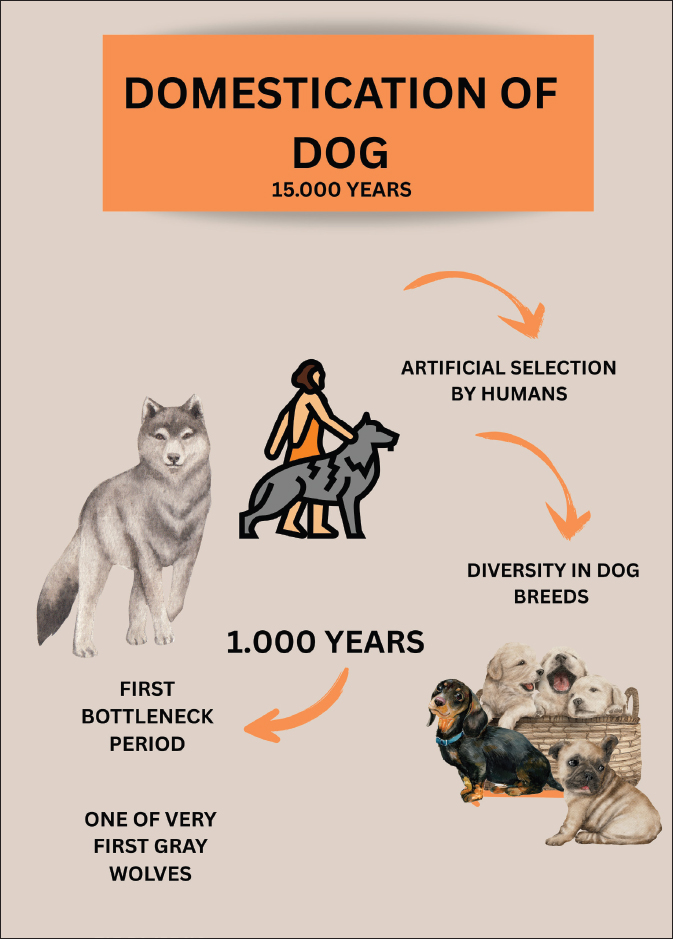
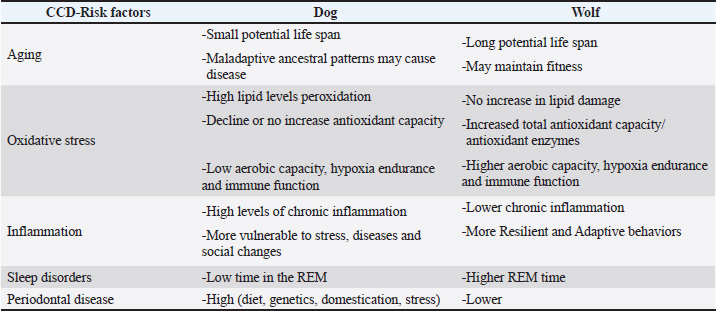
Fig. 1. The process of dog domestication approximately 15,000 years ago: population bottleneck since wolves, human artificial selection, and modern dog breeds diversity. Periodontal disease (PD) is one of the most common oral disorders affecting both dogs and humans. It involves a progressive inflammatory reaction triggered by subgingival colonization with diverse pathogenic microorganisms that damages the connective tissues supporting the teeth, ultimately leading to tooth loss (Kinane et al., 2017; Cunha et al., 2022). The likelihood and severity of PD increase with advancing age (Wallis et al., 2018; Wallis et al., 2019; Wallis and Holcombe, 2020). Interestingly, the progression and age-related aggravation of this condition occur at an even faster rate in dogs than in humans, despite sharing similar underlying causes (Struillou et al., 2010). Despite dietary improvements that have positively influenced the overall health of companion dogs, PD remains a major oral health concern (Cunha et al., 2022). It is considered the most prevalent dental disorder among adult dogs, with reported rates ranging from 44% to nearly 100% (Stella et al., 2018; Wallis et al., 2019; Wallis and Holcombe, 2020). The condition is largely associated with insufficient dental hygiene practices in household animals (Garanayak et al., 2019; Wallis et al., 2019). Although all breeds are susceptible (Di Bello et al., 2014), smaller dogs (under 15 kg), and brachycephalic types tend to be more frequently and severely affected than larger breeds (Wallis et al., 2021). As previously mentioned, are precisely small breeds that have a higher prevalence of CCD (Turcsán and Kubinyi, 2024). Moreover, since PD is more prevalent and more severe in small and brachycephalic breeds, gray wolves, which are larger in size (generally > 25 Kg), would have lesser CCD risk prevalence. Over the last decade, a large body of evidence has demonstrated that PD and its characteristic oral microbial dysbiosis play an important role in the initiation and progression of AD (Dominy et al., 2019; Wan and Fan, 2023; Chaple-Gil et al., 2025), with PD being recognized as a risk factor for the development of AD (Dominy et al., 2019; Hu et al., 2021; Chaple-Gil et al., 2025). Recent experimental research in animal models has provided stronger support for a possible causal association between periodontitis and AD, describing biologically plausible and clinically consistent pathways through which periodontal inflammation might contribute to the onset or progression of AD (Dominy et al., 2019; Hajishengallis and Chavakis, 2021; Kong et al., 2025). To explore the links between PD and CCD, a scoring system was applied to evaluate PD severity in dogs diagnosed with presumptive CCD and in control dogs without any reported signs of cognitive decline. Results showed a significant association between periodontal and cognitive scores, with dogs with more severe PD having higher cognitive impairment scores and vice versa (Dewey and Rishniw, 2021; Templeton et al., 2023). PD is less common in wild carnivores than in domestic dogs (Kapoor et al., 2016; Döring et al., 2018). Several domestication-related elements—including stress, increased longevity, dietary composition, and texture, along with genetic predisposition—may help explain why dogs exhibit a higher frequency of PD than their wild relatives (Pires et al., 2020). Evidence indicates that the physical and nutritional characteristics of food are key determinants of oral health in domestic dogs. Certain types of diet promote inflammation in the gums and teeth. Diets with soft, wet, or paste-like textures (e.g., canned or home-cooked foods) offer less mechanical abrasion, promoting bacterial plaque accumulation at the gingival margin and on tooth surfaces, which in turn provokes gingivitis and facilitates enamel demineralization (Oba et al., 2022; Wallis et al., 2025). Diets rich in fermentable carbohydrates similarly feed acid-producing bacteria, lowering oral pH and promoting gum and dental tissue inflammation (Hiney et al., 2021). Harder, dry, or extruded foods, dental chews, or well-formulated raw meat/bone diets tend to reduce plaque and calculus, maintain a higher salivary pH, and are associated with fewer signs of gingival and dental inflammation, thereby maintaining a healthier oral microbiota (Cunha et al., 2022; Hiney et al., 2024). Additionally, the absence of mechanical cleaning and the presence of dietary sugars and acids can alter plaque composition, increasing the proportion of Gram-negative anaerobes and predisposing to periodontal dysbiosis (Borsa et al., 2021; Cunha et al., 2022). Consequently, feeding practices that incorporate natural chewing activity—such as bones retaining muscle or connective tissues—have been consistently associated with lower prevalence of gingivitis and periodontal disorders compared with predominantly soft-textured diets (Marx et al., 2016; Pinto et al., 2020; Casarin et al., 2023). Research on the trophic behavior of the Iberian wolf in Europe shows that medium-sized ungulates are their principal prey, yet domestic species also make up a notable share of their consumption (Barja et al., 2023). The occasional intake of fish has also been documented (Pires et al., 2020). Where wild prey is scarce due to human disturbance, these predators shift toward anthropogenic sources, relying on livestock and human-related materials, such as waste and carcasses (Newsome et al., 2016). Dental pathologies were examined in predominantly adult Iberian wolf specimens from a Portuguese museum collection. A total of 65 deceased individuals—comprising 61 complete crania and 4 isolated mandibles—collected in Portugal between 1977 and 1995 were assessed. Oral pathology was assessed by combining visual inspection with radiographic techniques, documenting wear, breakage, gum disease, and additional dental problems. The survey revealed that nearly 90% of the teeth exhibited some degree of PD between stages 2–4. However, most findings were classified as stage 2, with a prevalence of 73.9% in the upper jaw and 68% in the lower jaw. Severe manifestations (stage 4) are rare (Pires et al., 2020). The link between nutrition and periodontal changes during aging is still under debate. A possible explanation is that older animals exert less chewing pressure on hard dry feed, which reduces stimulation of the periodontal tissues and diminishes saliva flow, which could accelerate oral disease (Inui 2015). In contrast, according to a 2005 review of biting mammals, the gray wolf has the strongest bite of any canine, at 593 Newtons (Wroe et al., 2005), which may contribute to their developing less severe PD than dogs. Considering the established association between PD and CCD in domestic dogs, this finding suggests a potential protective effect in wolves. However, direct comparative studies assessing cognitive outcomes are required to confirm this. The results of this study indicate that key risk factors for CCD are more pronounced in domestic dogs than in gray wolves maintained in captivity or semi-captivity (Table 1; Fig. 2).
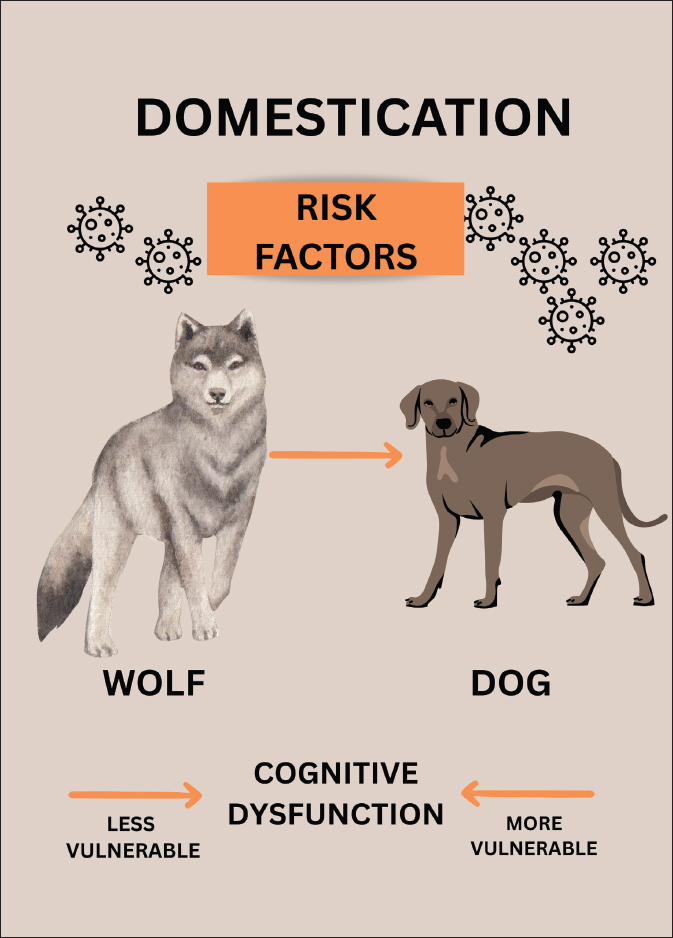
Fig. 2. Domestication may increase the incidence of canine cognitive dysfunction (CCD)-risk factors in dogs compared with gray wolves kept in captivity or semi-captivity; hence, wolves may be less vulnerable to CCD than dogs.). Table 1. Main differences in CCD risk factors between domestic dogs and gray wolves kept in captivity or semi-captivity. Refer to the references in the manuscript.
ConclusionThe results of this literature review suggest that dog domestication may have increased their vulnerability to canine cognitive dysfunction compared with the gray wolf. The risk factors analyzed are more pronounced in domestic dogs than in gray wolves kept in captivity or semi-captivity. This finding suggests that gray wolves may exhibit lower susceptibility to age-associated cognitive decline. However, given the scarcity of neuropathological and behavioral data on aged wolves, these conclusions should be interpreted with caution. Taken together, the results reinforce the need to expand comparative research between dogs and wolves, both at the molecular and behavioral levels, to clarify the role of domestication in brain health and aging. Furthermore, the findings highlight the importance of implementing preventive strategies aimed at mitigating the identified risk factors in domestic dogs, which could help reduce the incidence and progression of CCD. Within the One Health perspective, examining naturally occurring cognitive decline associated with aging in domestic dogs in comparison to wolves can provide valuable knowledge, offering opportunities to deepen our understanding of neurodegenerative diseases across species, including humans. AcknowledgmentsThe authors thank Mr. Carlos Sanz (from the Iberian Wolf Center "Félix Rodríguez de la Fuente", Castilla-León, Spain) for providing important information on the Iberian wolf. Conflict of interestThe authors have no competing interests to declare. FundingThis work did not receive any external funding. Authors’ contributionsRCF: conceptual design, data acquisition, methodology, figures, and original draft preparation. RSP: table, review, and editing. Both authors have approved the final version. Data availabilityAll relevant data are included within the manuscript. ReferencesAbey, A., Davies, D., Goldsbury, C., Buckland, M., Valenzuela, M. and Duncan, T. 2021. Distribution of tau hyperphosphorylation in canine dementia resembles early Alzheimer’s disease and other tauopathies. Brain. Pathol. 31(1), 144–162; doi:10.1111/bpa.12893 Alexander, J.E., Colyer, A., Haydock, R.M., Hayek, M.G. and Park, J. 2018. Understanding how dogs Age: longitudinal analysis of markers of inflammation, immune function, and oxidative Stress. J. Gerontol. A. Biol. Sci. Med. Sci. 73(6), 720–728; doi:10.1093/gerona/glx182 Almutary, A.G., Begum, M.Y., Kyada, A.K., Gupta, S., Jyothi, S.R., Chaudhary, K., Sharma, S., Sinha, A., Abomughaid, M.M., Imran, M., Lakhanpal, S., Babalghith, A.O., Abu-Seer, E.A., Avinash, D., Alzahrani, H.A., Alhindi, A.A., Iqbal, D., Kumar, S., Jha, N.K. and Alghamdi, S. 2025. Inflammatory signaling pathways in Alzheimer’s disease: mechanistic insights and possible therapeutic interventions. Ageing. Res. Rev. 104, 102548; doi:10.1016/j.arr.2024.102548 Andersen, S.L. 2020. Centenarians as models of resistance and resilience to Alzheimer's disease and related dementias. Adv. Geriatr. Med. Res. 2(3), e200018; doi:10.20900/agmr20200018 Axelsson, E., Ljungvall, I., Bhoumik, P., Conn, L.B., Muren, E., Ohlsson, A., Olsen, L.H., Engdahl, K., Hagman, R., Hanson, J., Kryvokhyzha, D., Pettersson, M., Grenet, O., Moggs, J., Del Rio-espinola, A., Epe, C., Taillon, B., Tawari, N., Mane, S., Hawkins, T., Hedhammar, A., Gruet, P., Häggström, J. and Lindblad-Toh, K. 2021. The genetic consequences of dog breed formation-Accumulation of deleterious genetic variation and fixation of mutations associated with myxomatous mitral valve disease in cavalier King Charles spaniels. PLos Genet. 17(9), e1009726; doi:10.1371/journal.pgen.1009726 Azkona, G., García-Belenguer, S., Chacón, G., Rosado, B., León, M. and Palacio, J. 2009. Prevalence and risk factors of behavioural changes associated with age-related cognitive impairment in geriatric dogs. J. Small Anim. Pract. 50(2), 87–91; doi: 10.1111/j.1748-5827.2008.00718.x Barber-Meyer, S.M., Wheeldon, T.J. and Mech, L.D. 2021. The importance of wilderness to wolf (Canis lupus) survival and cause-specific mortality over 50 years. Biol. Conserv. 258, 109145; doi:10.1016/j.biocon.2021.109145 Barja, I., Navarro-Castilla., Ortiz-Jiménez, L., España., Hinojosa, R., Sánchez-Sotomayor, D., Iglesias., España, J., Rubio-Sánchez, S., Martín-Romero, S., Vielva, J. and Horcajada-Sánchez, F. 2023. Wild ungulates constitute the basis of the diet of the iberian wolf in a recently recolonized area: wild boar and roe deer as key species for its conservation. Animals. (Basel). 13(21), 3364; doi:10.3390/ani13213364 Barragán-Sánchez, P., Balastegui, M.T., Marín-García, P.J. and Llobat, L. 2025. Genetic regulation of immune response in dogs. Genes. (Basel). 16(7), 764; doi:10.3390/genes16070764 Belenguer-Varea, A., Tarazona-Santabalbina, F.J., Avellana-Zaragoza, J.A., Martínez-Reig, M., Mas-Bargues, C. and Inglés, M. 2020. Oxidative stress and exceptional human longevity: systematic review. Free Radic. Biol. Med. 149, 51–63; doi:10.1016/j.freeradbiomed.2019.09.019 Benítez-Burraco, A., Uriagereka, J. and Nataf, S. 2023. The genomic landscape of mammal domestication might be orchestrated by selected transcription factors regulating brain and craniofacial development. Dev. Genes. Evol. 233(2), 123–135; doi:10.1007/s00427-023-00709-7 Bergström, A., Stanton, D.W.G., Taron, U.H., Frantz, L., Sinding, M.S., Ersmark, E., Pfrengle, S., Cassatt-Johnstone, M., Lebrasseur, O., Girdland-Flink, L., Fernandes, D.M., Ollivier, M., Speidel, L., Gopalakrishnan, S., Westbury, M.V., Ramos-Madrigal, J., Feuerborn, T.R., Reiter, E., Gretzinger, J., Münzel, S.C., Swali, P., Conard, N.J., Carøe, C., Haile, J., Linderholm, A., Androsov, S., Barnes, I., Baumann, C., Benecke, N., Bocherens, H., Brace, S., Carden, R.F., Drucker, D.G., Fedorov, S., Gasparik, M., Germonpré, M., Grigoriev, S., Groves, P., Hertwig, S.T., Ivanova, V.V., Janssens, L., Jennings, R.P., Kasparov, A.K., Kirillova, I.V., Kurmaniyazov, I., Kuzmin, Y.V., Kosintsev, P.A., Lázničková-Galetová, M., Leduc, C., Nikolskiy, P., Nussbaumer, M., O'Drisceoil, C., Orlando, L., Outram, A., Pavlova, E.Y., Perri, A.R., Pilot, M., Pitulko, V.V., Plotnikov, V.V., Protopopov, A.V., Rehazek, A., Sablin, M., Seguin-Orlando, A., Storå, J., Verjux, C., Zaibert, V.F., Zazula, G., Crombé, P., Hansen, A.J., Willerslev, E., Leonard, J.A., Götherström, A., Pinhasi, R., Schuenemann, V.J., Hofreiter, M., Gilbert, M.T.P., Shapiro, B., Larson, G., Krause, J., Dalén, L. and Skoglund, P. 2022. Grey wolf genomic history reveals a dual ancestry of dogs. Nature 78(7918), 313–320; doi: 10.1038/s41586-022-04824-9 Blanchard, T., Eppe, J., Mugnier, A., Delfour, F. and Meynadier, A. 2025. Enhancing cognitive functions in aged dogs and cats: a systematic review of enriched diets and nutraceuticals. GeroScience 47(3), 2925–2947; doi:10.1007/s11357-025-01521-z Borsa, L., Dubois, M., Sacco, G. and Lupi, L. 2021. Analysis the link between periodontal disease and Alzheimer’s disease: a systematic review. Int. J. Environ. Res. Public Health 18(17), 9312; doi:10.3390/ijerph18179312 Bosse, M., Megens, H.J., Derks, M.F.L., De Cara, A.M.R. and Groenen, M.A.M. 2018. Deleterious alleles in the context of domestication, inbreeding, and selection. Evol. Appl. 12(1), 6–17; doi:10.1111/eva.12691 Bray, E.E., Raichlen, D.A., Forsyth, K.K., Promislow, D.E.L., Alexander, G.E., Maclean, E.L., Akey, J.M., Benton, B., Borenstein, E., Castelhano, M.G., Coleman, A.E., Creevy, K.E., Crowder, K., Dunbar, M.D., Fajt, V.R., Fitzpatrick, A.L., Jeffrey, U., Jonlin, E.C., Kaeberlein, M., Karlsson, E.K., Kerr, K.F., Levine, J.M., Ma, J., Mcclelland, R.L., Ruple, A., Schwartz, S.M., Shrager, S., Snyder-Mackler, N., Tolbert, M.K., Urfer, S.R. and Wilfond, B.S. 2023. Associations between physical activity and cognitive dysfunction in older companion dogs: results from the Dog Aging Project. GeroScience 45(2), 645–661; doi:10.1007/s11357-022-00655-8 Buffenstein, R., Amoroso, V., Andziak, B., Avdieiev, S., Azpurua, J., Barker, A.J., Bennett, N.C., Brieño-Enríquez, M.A., Bronner, G.N., Coen, C., Delaney, M.A., Dengler-Crish, C.M., Edrey, Y.H., Faulkes, C.G., Frankel, D., Friedlander, G., Gibney, P.A., Gorbunova, V., Hine, C., Holmes, M.M., Jarvis, J.U.M., Kawamura, Y., Kutsukake, N., Kenyon, C., Khaled, W.T., Kikusui, T., Kissil, J., Lagestee, S., Larson, J., Lauer, A., Lavrenchenko, L.A., Lee, A., Levitt, J.B., Lewin, G.R., Lewis Hardell, K.N., Lin, T.D., Mason, M.J., McCloskey, D., McMahon, M., Miura, K., Mogi, K., Narayan, V., O'Connor, T.P., Okanoya, K., O'Riain, M.J., Park, T.J., Place, N.J., Podshivalova, K., Pamenter, M.E., Pyott, S.J., Reznick, J., Ruby, J.G., Salmon, A.B., Santos-Sacchi, J., Sarko, D.K., Seluanov, A., Shepard, A., Smith, M., Storey, K.B., Tian, X., Vice, E.N., Viltard, M., Watarai, A., Wywial, E., Yamakawa, M., Zemlemerova, E.D., Zions, M. and Smith, E.S.J. 2022. The naked truth: a comprehensive clarification and classification of current 'myths' in naked mole-rat biology. Biol. Rev. 97(1), 115–140. Bunyaluk, D., Srisanyong, W. and Amporn, C. 2022. Apolipoprotein E4 prevalence in different canine breeds. Int. J. Vet. Sci. 11(3), 367–372; doi:10.47278/journal.ijvs/2021.123. Cagan, A. and Blass, T. 2016. Identification of genomic variants putatively targeted by selection during dog domestication. BMC. Evol. Biol. 16, 10; doi:10.1186/s12862-015-0579-7 Calder, P.C., Bosco, N., Bourdet-Sicard, R., Capuron, L., Delzenne, N., Doré, J., Franceschi, C., Lehtinen, M.J., Recker, T., Salvioli, S. and Visioli, F. 2017. Health relevance of the modification of low-grade inflammation in ageing (inflammageing) and the role of nutrition. Ageing Res. Rev. 40, 95–119; doi:10.1016/j.arr.2017.09.001 Calderon-Garciduenas, L., Maronpot, R.R., Torres-Jardon, R., Henriquez-Roldan, C., Schoonhoven, R., Acuna-Ayala, H., Villarreal-Calderon, A., Nakamura, J., Fernando, R., Reed, W., Azzarelli, B. and Swenberg, J.A. 2003. DNA damage in nasal and brain tissues of canines exposed to air pollutants is associated with evidence of chronic brain inflammation and neurodegeneration. Toxicol. Pathol. 31(5), 524–538; doi:10.1080/01926230390226645 Casarin, M., Da Silveira, T.M., Bezerra, B., Pirih, F.Q. and Pola, N.M. 2023. Association between different dietary patterns and eating disorders and periodontal diseases. Front. Oral Health 4, 1152031; doi:10.3389/froh.2023.1152031 Cedernaes, J., Osorio, R.S., Varga, A.W., Kam, K., Schiöth, H.B. and Benedict, C. 2017. Candidate mechanisms underlying the association between sleep-wake disruptions and Alzheimer’s disease. Sleep Med. Rev. 31, 102–111; doi:10.1016/j.smrv.2016.02.002 Chapagain, D., Range, F., Huber, L. and Virányi, Z. 2018. Cognitive aging in dogs. Gerontology 64(2), 165–171; doi:10.1159/000481621 Chaple-Gil, A.M., Santiesteban-Velázquez, M. and Urbizo-Vélez, J.J. 2025. Association between oral microbiota dysbiosis and the risk of dementia: a systematic review. Dent. J. (Basel). 13(6), 227; doi:10.3390/dj13060227 Charruau, P., Johnston, R.A., Stahler, D.R., Lea, A., Snyder-Mackler, N., Smith, D.W., Vonholdt, B.M., Cole, S.W., Tung, J. and Wayne, R.K. 2016. Pervasive effects of aging on gene expression in wild wolves. Mol. Biol. Evol. 33(8), 1967–1978; doi:10.1093/molbev/msw072 Chauhan, A.K. and Mallick, B.N. 2019. Association between autophagy and rapid eye movement sleep loss-associated neurodegenerative and patho-physio-behavioral changes. Sleep Med. 63, 29–37; doi:10.1016/j.sleep.2019.04.019 Clutton-Brock. 1999. A natural history of domesticated mammals. Cambridge: cambridge University Press. Cambridge: Cambridge University Press. Craun, K., Luethcke, K.R., Shafer, M., Stanton, N., Zhang, C., Schauer, J., Faulkes, J., Sundling, K.E., Kurtycz, D., Malecki, K. and Trepanier, L. 2020. Environmental chemical exposures in the urine of dogs and people sharing the same households. J. Clin. Transl. Sci. 5(1), 54; doi:10.1017/cts.2020.548 Cruz, F., Vila, C. and Webster, M.T. 2008. The legacy of domestication: accumulation of deleterious mutations in the dog genome. Mol. Biol. Evol. 25(11), 2331–2336; doi:10.1093/molbev/msn177 Cunha, E., Tavares. and Oliveira, M. 2022. Revisiting periodontal disease in dogs: how to manage this new old problem?. Antibiotics. (Basel). 11(12), 1729; doi:10.3390/antibiotics11121729 Davidson, Y.S., Robinson, A., Prasher, V.P. and Mann, D.M.A. 2018. The age of onset And evolution of Braak tangle stage and Thal amyloid pathology of Alzheimer’s disease in individuals with Down syndrome. Acta Neuropathol. Commun. 6(1), 56; doi:10.1186/s40478-018-0559-4 Day, M.J. 2010. Ageing, immunosenescence and inflammageing in the dog and cat. J. Comp. Pathol. 142(1), S60–S69. De Risio, L., Bhatti, S., Muñana, K., Penderis, J., Stein, V., Tipold, A., Berendt, M., Farqhuar, R., Fischer, A., Long, S., Mandigers, P.J., Matiasek, K., Packer, R.M., Pakozdy, A., Patterson, N., Platt, S., Podell, M., Potschka, H., Batlle, M.P., Rusbridge, C. and Volk, H.A. 2015. International veterinary epilepsy task force consensus proposal: diagnostic approach to epilepsy in dogs. BMC Vet. Res. 11, 148; doi:10.1186/s12917-015-0462-1 Dewey, C. and Rishniw, M. 2021. Periodontal disease is associated with cognitive dysfunction in aging dogs: a blinded prospective comparison of visual periodontal and cognitive questionnaire scores. Open Vet. J. 11(2), 210–216; doi:10.5455/OVJ.2021.v11.i2.4 Dewey, C.W., Davies, E.S., Xie, H. and Wakshlag, J.J. 2019. Canine cognitive dysfunction: pathophysiology, diagnosis, and treatment. Vet. Clin. Small. Anim. 49(3), 477–499; doi:10.1016/j.cvsm.2019.01.013 Di Bello, A., Buonavoglia, A., Franchini, D., Valastro, C., Ventrella, G., Greco, M.F. and Corrente, M. 2014. Periodontal disease associated with red complex bacteria in dogs. J. Small Anim. Pract. 55(3), 160–163. Domínguez-de-Barros, A., Sifaoui, I., Borecka, Z., Dorta-Guerra, R., Lorenzo-Morales, J., Castro-Fuentes, R. and Córdoba-Lanús, E. 2023. An approach to the effects of longevity, sexual maturity, and reproduction on telomere length and oxidative stress in different Psittacidae species. Front. Genet. 14, 1156730; doi:10.3389/fgene.2023.1156730 Domínguez-De-Barros, A., Sifaoui, I., Dorta-Guerra, R., Lorenzo-Morales, J., Castro-Fuentes, R. and Córdoba-Lanús, E. 2025. Telomere- and oxidative stress dynamics in Psittacidae species with different longevity trajectories. GeroScience 47(1), 121–134; doi:10.1007/s11357-024-01397-5 Dominy, S.S., Lynch, C., Ermini, F., Benedyk, M., Marczyk, A., Konradi, A., Nguyen, M., Haditsch, U., Raha, D., Griffin, C., Holsinger, L.J., Arastu-Kapur, S., Kaba, S., Lee, A., Ryder, M.I., Potempa, B., Mydel, P., Hellvard, A., Adamowicz, K., Hasturk, H., Walker, G.D., Reynolds, E.C., Faull, R.L.M., Curtis, M.A., Dragunow, M. and Potempa, J. 2019. Porphyromonas gingivalis in Alzheimer's disease brains: evidence for disease causation and treatment with small-molecule inhibitors. Sci. Adv. 5(1), eaau3333; doi:10.1126/sciadv.aau3333 Donner, J., Anderson, H., Davison, S., Hughes, A.M., Bouirmane, J., Lindqvist, J., Lytle, K.M., Ganesan, B., Ottka, C., Ruotanen, P., Kaukonen, M., Forman, O.P., Fretwell, N., Cole, C.A. and Lohi, H. 2018. Frequency and distribution of 152 genetic disease variants in over 100000 mixed breed and purebred dogs. PLos Genet. 14(4), e1007361; doi:10.1371/journal.pgen.1007361 Döring, S., Arzi, B., Winer, J.N., Kass, P.H. and Verstraete, F.J.M. 2018. Dental and temporomandibular joint pathology of the grey wolf (Canis lupus). J. Comp. Pathol. 60, 56–70; doi:10.1016/j.jcpa.2018.03.001 Dupré, G. and Heidenreich, D. 2016. Brachycephalic syndrome. Vet. Clin. North Am. Small Anim. Pract. 46(4), 691–707; doi:10.1016/j.cvsm.2016.02.002 Fakhoury, M. 2020. Inflammation in Alzheimer's disease. Curr. Alzheimer Res. 17(11), 959–961; doi:10.2174/156720501711210101110513 Fast, R., Schütt, T., Toft, N., Møller, A. and Berendt, M. 2013. An observational study with long-term follow-up of canine cognitive dysfunction: clinical characteristics, survival, and risk factors. J. Vet. Intern. Med. 27(4), 822–829; doi:10.1111/jvim.12109 Franceschi, C., Garagnani, P., Parini, P., Giuliani, C. and Santoro, A. 2018. Inflammaging: a new immune-metabolic viewpoint for age-related diseases. Nat. Rev. Endocrinol. 14(10), 576–590; doi:10.1038/s41574-018-0059-4 Franck, M., Tanner, K.T., Tennyson, R.L., Daunizeau, C., Ferrucci, L., Bandinelli, S., Trumble, B.C., Kaplan, H.S., Aronoff, J.E., Stieglitz, J., Kraft, T.S., Lea, A.J., Venkataraman, V.V., Wallace, I.J., Lim, Y.A.L., Ng, K.S., Yeong, J.P.S., Ho, R., Lim, X., Mehrjerd, A., Charalambous, E.G., Aiello, A.E., Pawelec, G., Franceschi, C., Hertel, J., Fülöp, T., Lemoine, M., Gurven, M. and Cohen, A.A. 2025. Nonuniversality of inflammaging across human populations. Nat. Aging 5(8), 1471–1480; doi:10.1038/s43587-025-00888-0 Frank, M.G. and Craig Heller, H. 2019. The function(s) of sleep. Handb. Exp. Pharmacol. 253, 3-34. doi: 10.1007/164_2018_140 Freedman, A.H. and Wayne, R.K. 2017. Deciphering the origin of dogs: from fossils to genomes. Ann. Rev. Anim. Biosci. 5, 281–307; doi:10.1146/annurev-animal-022114-110937 Freedman, A.H., Lohmueller, K.E. and Wayne, R.K. 2016. Evolutionary history, selective sweeps, and deleterious variation in the dog. Annu. Rev. Ecol. Evol. Syst. 47, 73–96; doi:10.1146/annurev-ecolsys-121415-032155 Garanayak, N., Das, M., Patra, R.C., Biswal, S. and Panda, S.K. 2019. Effect of age on dental plaque deposition and its control by ultrasonic scaling, dental hygiene chew, and chlorhexidine (0.2%w/v) in dogs. Vet. World. 12(11), 1872–1876; doi:10.14202/vetworld.2019.1872-1876 Gray, M.M., Granka, J.M., Bustamante, C.D., Sutter, N.B., Boyko, A.R., Zhu, L., Ostrander, E.A. and Wayne, R.K. 2009. Linkage disequilibrium and demographic history of wild and domestic canids. Genetics 181, 1493–1505; doi:10.1534/genetics.108.098830 Habiba, U., Ozawa, M., Chambers, J.K., Uchida, K., Descallar, J., Nakayama, H., Summers, B.A., Morley, J.W. and Tayebi, M. 2021. Neuronal deposition of amyloid-β oligomers and hyperphosphorylated Tau is closely connected with cognitive dysfunction in aged dogs. J. Alzheimers. Dis. Rep. 5(1), 749–760; doi:10.3233/ADR-210035 Hajishengallis, G. and Chavakis, T. 2021. Local and systemic mechanisms linking periodontal disease and inflammatory comorbidities. Nat. Rev. Immunol. 21(7), 426–440; doi:10.1038/s41577-020-00488-6 Henn, B.M., Botigué, L.R., Bustamante, C.D., Clark, A.G. and Gravel, S. 2015. Estimating the mutation load in human genomes. Nat. Rev. Genet. 16(6), 333–343; doi:10.1038/nrg3931 Hines, A.D., Mcgrath, S., Latham, A.S., Kusick, B., Mulligan, L., Richards, M.L. and Moreno, J.A. 2023. Activated gliosis, accumulation of amyloid β, and hyperphosphorylation of tau in aging canines with and without cognitive decline. Front. Aging. Neurosci. 15, 1128521; doi:10.3389/fnagi.2023.1128521 Hiney, K., Sypniewski, L., Desilva, U., Pezeshki, A., Rudra, P., Goodarzi, P., Willis, E. and Mcfarlane, D. 2024. Fecal microbiota composition, serum metabolomics, and markers of inflammation in dogs fed a raw meat-based diet compared to those on a kibble diet. Front. Vet. Sci. 11, 328513; doi:10.3389/fvets.2024.1328513 Hiney, K., Sypniewski, L., Rudra, P., Pezeshki, A. and Mcfarlane, D. 2021. Clinical health markers in dogs fed raw meat-based or commercial extruded kibble diets. J. Anim. Sci. 99(6), 133; doi:10.1093/jas/skab133 Hoffman, J.M., Creevy, K.E., Franks, A., O'Neill, D.G. and Promislow, D.E.L. 2018. The companion dog as a model for human aging and mortality. Aging Cell 17(3), e12737. Holečková, E. and Chvapil, M. 1965. The effect of intermittent feeding and fasting and of domestication on biological age in the rat. Gerontologia 11(1), 96–119; doi:10.1159/000211483 Hou, Y., Qi, F., Bai, X., Ren, T., Shen, X., Chu, Q., Zhang, X. and Lu, X. 2020. Genome-wide analysis reveals molecular convergence underlying domestication in 7 bird and mammals. BMC. Genomics. 21(1), 204; doi:10.1186/s12864-020-6613-1 Hu, X., Zhang, J., Qiu, Y. and Liu, Z. 2021. Periodontal disease and the risk of Alzheimer’s disease and mild cognitive impairment: a systematic review and meta-analysis. Psychogeriatrics 21(5), 813–825; doi:10.1111/psyg.12743 Huang, Z., Pan, Z., Yang, R., Bi, Y. and Xiong, X. 2020. The canine gastrointestinal microbiota: early studies and research frontiers. Gut Microbes 11(4), 635–654; doi:10.1080/19490976.2019.1704142 Hytönen, M.K. and Lohi, H. 2016. Canine models of human rare disorders. Rare Dis. 4(1), e1241362; doi:10.1080/21675511.2016.1241362 Iakovou, E. and Kourti, M. 2022. A comprehensive overview of the complex role of oxidative stress in aging, the contributing environmental stressors and emerging antioxidant therapeutic interventions. Front. Aging Neurosci. 14, 827900; doi:10.3389/fnagi.2022.827900 Inui, T. 2015. The beneficial effects of regular chewing.In Dry Mouth: a guide to clinical causes, effects and treatments. Carpenter. and G Berlin, Germany: Springer-Verlag, pp: 175–94. Ionescu-Tucker, A. and Cotman, C.W. 2021. Emerging roles of oxidative stress in brain aging and Alzheimer's disease. Neurobiol. Aging 107, 86–95; doi:10.1016/j.neurobiolaging.2021.07.014 Iotchev, I.B., Reicher, V., Kovács, E., Kovács, T., Kis, A., Gácsi, M. and Kubinyi, E. 2020. Averaging sleep spindle occurrence in dogs predicts learning performance better than single measures. Sci. Rep. 10(1), 22461; doi:10.1038/s41598-020-80417-8 Işık, M., Tunç, A., Duran, H.E., Naldan, M.E., Yılmaz, A., Koçak, M.N. and Beydemir. 2023. Evaluation of the relationship among gene expressions and enzyme activities with antioxidant role and presenilin 1 expression in Alzheimer's disease. J. Cell. Mol. Med. 27(21), 3388–3394; doi:10.1111/jcmm.17953 Jacobs, P.J., Hart, D.W., Merchant, H.N., Voigt, C. and Bennett, N.C. 2023. The evolution and ecology of oxidative and antioxidant status: a comparative approach in African mole-rats. Antioxidants (Basel). 12(8), 1486; doi:10.3390/antiox12081486 Jain, S.K., Stevens, C.M., Margret, J.J. and Levine, S.N. 2024. Alzheimer’s disease: a review of pathology, current treatments, and the potential therapeutic effect of decreasing oxidative stress by combined vitamin D and l-cysteine supplementation. Antioxid. Redox. Signal. 40(10-12), 663–678; doi:10.1089/ars.2023.0245 Ji, K.H. and Yun, C.H. 2025. Brain health in sleep disorders. Sleep Med. Clin. 20(1), 57–72; doi:10.1016/j.jsmc.2024.10.004 Jimenez, A.G. 2016. Physiological underpinnings in life-history trade-offs in man’s most popular selection experiment: the dog. J. Comp. Physiol. B. 186(7), 813–827; doi:10.1007/s00360-016-1002-4 Jimenez, A.G. 2021. The physiological conundrum that is the domestic dog. Integr. Comp. Biol. 61(1), 140–153; doi:10.1093/icb/icab005 Jiménez, A.G. 2023. Inflammaging in domestic dogs: basal level concentrations of IL-6, IL-1β, and TNF-α in serum of healthy dogs of different body sizes and ages. Biogerontology 24(4), 593–602; doi:10.1007/s10522-023-10037-y Jiménez, A.G. 2024. A revisiting of "the hallmarks of aging" in domestic dogs: current status of the literature. Geroscience 46(1), 241–255. doi: 10.1007/s11357-023-00911-5 Jimenez, A.G. and Downs, C.J. 2020. Untangling life span and body mass discrepancies in canids: phylogenetic comparison of oxidative stress in blood from domestic dogs and wild canids. Am. J. Physiol. Regul. Integr. Comp. Physiol. 319(2), R203–R210; doi:10.1152/ajpregu.00067.2020 Jimenez, A.G., O’Connor, E.S., Tobin, K.J., Anderson, K.N., Winward, J.D., Fleming, A., Winner, C., Chinchilli, E., Maya, A., Carlson, K. and Downs, C.J. 2019. Does cellular metabolism from primary fibroblasts and oxidative stress in blood differ between mammals and birds? The (lack-thereof) scaling of oxidative stress. Integr. Comp. Biol. 59(4), 953–969; doi:10.1093/icb/icz017 Johnson, C.S., Shively, C.A., Michalson, K.T., Lea, A.J., Debo, R.J., Howard, T.D., Hawkins, G.A., Appt, S.E., Liu, Y., Mccall, C.E., Herrington, D.M., Ip, E.H., Register, T.C. and Snyder-Mackler, N. 2021. Contrasting effects of Western vs Mediterranean diets on monocyte inflammatory gene expression and social behavior in a primate model. eLife 10, e68293. Kapoor, V., Antonelli, T., Parkinson, J.A. and Hartstone-Rose, A. 2016. Oral health correlates of captivity. Res. Vet. Sci. 107, 213–219; doi:10.1016/j.rvsc.2016.06.009 Kim, C.Y., Kim, J., Yoon, S., Yi, I.J., Lee, H., Seo, S., Kim, D.W., Ko, S., Kim, S.A., Kwon, C. and Yi, S.S. 2024. Advancing the early detection of canine cognitive dysfunction syndrome with machine learning-enhanced blood-based biomarkers. Front. Vet. Sci. 11, 1390296; doi:10.3389/fvets.2024.1390296 Kinane, D.F., Stathopoulou, P.G. and Papapanou, P.N. 2017. Periodontal diseases. Nat. Rev. Dis. Primers 3, 17038; doi:10.1038/nrdp.2017.38 Kis, A., Szakadát, S., Gácsi, M., Kovács, E., Simor, P., Török, C., Gombos, F., Bódizs, R. and Topál, J. 2017. The interrelated effect of sleep and learning in dogs (Canis familiaris); an EEG and behavioural study. Sci. Rep. 7, 41873; doi:10.1038/srep41873 Klein, H.U., Bennett, D.A. and De Jager, P.L. 2016. The epigenome in Alzheimer’s disease: current state and approaches for a new path to gene discovery and understanding disease mechanism. Acta Neuropathol. 132(4), 503–514; doi:10.1007/s00401-016-1612-7 Kong, L., Li, J., Gao, L., Zhao, Y., Chen, W., Wang, X., Wang, S. and Wang, F. 2025. Periodontitis-induced neuroinflammation triggers IFITM3-Abeta axis to cause Alzheimer's disease-like pathology and cognitive decline. Alzheimers. Res. Ther. 17(1), 166; doi:10.1186/s13195-025-01818-3 Kopp, W. 2019. How western diet and lifestyle drive the pandemic of obesity and civilization diseases. Diabetes. Metab. Syndr. Obes. 12, 2221–2236; doi:10.2147/DMSO.S216791 Krueger, J.M., Frank, M.G., Wisor, J.P. and Roy, S. 2016. Sleep function: toward elucidating an enigma. Sleep Med. Rev. 28, 46–54; doi:10.1016/j.smrv.2015.08.005 Lawler, D., Becker, J., Reetz, J., Goodmann, P., Evans, R., Rubin, D., Tangredi, B., Widga, C., Sackman, J., Martin, T., Kohn, L. and Smith, G. 2016. Pathology of gray wolf shoulders: lessons in species and aging. Anat. Rec. 299(10), 1338–1347; doi:10.1002/ar.23380 Li, X., Li, C., Zhang, W., Wang, Y., Qian, P. and Huang, H. 2023. Inflammation and aging: signaling pathways and intervention therapies. Signal Transduct. Target. Ther. 8(1), 239; doi:10.1038/s41392-023-01502-8 Li, Y., Vonholdt, B.M., Reynolds, A., Boyko, A.R., Wayne, R.K., Wu, D.D. and Zhang, Y.P. 2013. Artificial selection on brain-expressed genes during the domestication of dog. Mol. Biol. Evol. 30(8), 1867–1876; doi:10.1093/molbev/mst088 Lindblad-Toh, K., Wade, C.M., Mikkelsen, T.S., Karlsson, E.K., Jaffe, D.B., Kamal, M., Clamp, M., Chang, J.L., Kulbokas, E.J., Zody, M.C., Mauceli, E., Xie, X., Breen, M., Wayne, R.K., Ostrander, E.A., Ponting, C.P., Galibert, F., Smith, D.R., Dejong, P.J., Kirkness, E., Alvarez, P., Biagi, T., Brockman, W., Butler, J., Chin, C.W., Cook, A., Cuff, J., Daly, M.J., Decaprio, D., Gnerre, S., Grabherr, M., Kellis, M., Kleber, M., Bardeleben, C., Goodstadt, L., Heger, A., Hitte, C., Kim, L., Koepfli, K.P., Parker, H.G., Pollinger, J.P., Searle, S.M., Sutter, N.B., Thomas, R., Webber, C., Baldwin, J., Abebe, A., Abouelleil, A., Aftuck, L., Ait-Zahra, M., Aldredge, T., Allen, N., An, P., Anderson, S., Antoine, C., Arachchi, H., Aslam, A., Ayotte, L., Bachantsang, P., Barry, A., Bayul, T., Benamara, M., Berlin, A., Bessette, D., Blitshteyn, B., Bloom, T., Blye, J., Boguslavskiy, L., Bonnet, C., Boukhgalter, B., Brown, A., Cahill, P., Calixte, N., Camarata, J., Cheshatsang, Y., Chu, J., Citroen, M., Collymore, A., Cooke, P., Dawoe, T., Daza, R., Decktor, K., Degray, S., Dhargay, N., Dooley, K., Dooley, K., Dorje, P., Dorjee, K., Dorris, L., Duffey, N., Dupes, A., Egbiremolen, O., Elong, R., Falk, J., Farina, A., Faro, S., Ferguson, D., Ferreira, P., Fisher, S., Fitzgerald, M., Foley, K., Foley, C., Franke, A., Friedrich, D., Gage, D., Garber, M., Gearin, G., Giannoukos, G., Goode, T., Goyette, A., Graham, J., Grandbois, E., Gyaltsen, K., Hafez, N., Hagopian, D., Hagos, B., Hall, J., Healy, C., Hegarty, R., Honan, T., Horn, A., Houde, N., Hughes, L., Hunnicutt, L., Husby, M., Jester, B., Jones, C., Kamat, A., Kanga, B., Kells, C., Khazanovich, D., Kieu, A.C., Kisner, P., Kumar, M., Lance, K., Landers, T., Lara, M., Lee, W., Leger, J.P., Lennon, N., Leuper, L., Levine, S., Liu, J., Liu, X., Lokyitsang, Y., Lokyitsang, T., Lui, A., Macdonald, J., Major, J., Marabella, R., Maru, K., Matthews, C., Mcdonough, S., Mehta, T., Meldrim, J., Melnikov, A., Meneus, L., Mihalev, A., Mihova, T., Miller, K., Mittelman, R., Mlenga, V., Mulrain, L., Munson, G., Navidi, A., Naylor, J., Nguyen, T., Nguyen, N., Nguyen, C., Nguyen, T., Nicol, R., Norbu, N., Norbu, C., Novod, N., Nyima, T., Olandt, P., O'Neill, B., O'Neill, K., Osman, S., Oyono, L., Patti, C., Perrin, D., Phunkhang, P., Pierre, F., Priest, M., Rachupka, A., Raghuraman, S., Rameau, R., Ray, V., Raymond, C., Rege, F., Rise, C., Rogers, J., Rogov, P., Sahalie, J., Settipalli, S., Sharpe, T., Shea, T., Sheehan, M., Sherpa, N., Shi, J., Shih, D., Sloan, J., Smith, C., Sparrow, T., Stalker, J., Stange-Thomann, N., Stavropoulos, S., Stone, C., Stone, S., Sykes, S., Tchuinga, P., Tenzing, P., Tesfaye, S., Thoulutsang, D., Thoulutsang, Y., Topham, K., Topping, I., Tsamla, T., Vassiliev, H., Venkataraman, V., Vo, A., Wangchuk, T., Wangdi, T., Weiand, M., Wilkinson, J., Wilson, A., Yadav, S., Yang, S., Yang, X., Young, G., Yu, Q., Zainoun, J., Zembek, L., Zimmer, A. and Lander, E.S. 2005. Genome sequence, comparative analysis and haplotype structure of the domestic dog. Nature 438(7069), 803–819; doi:10.1038/nature04338 Liu, Y., Tan, Y., Zhang, Z., Yi, M., Zhu, L. and Peng, W. 2024. The interaction between ageing and Alzheimer’s disease: insights from the hallmarks of ageing. Transl. Neurodegener. 13(1), 7; doi:10.1186/s40035-024-00397-x Lushchak, V.I., Duszenko, M., Gospodaryov, D.V. and Garaschuk, O. 2021. Oxidative stress and energy metabolism in the brain: midlife as a turning point. Antioxidants. (Basel). 10(11), 1715; doi:10.3390/antiox10111715 Macnulty, D.R., Smith, D.W., Mech, L.D. and Eberly, L.E. 2009. Body size and predatory performance in wolves: is bigger better?. J. Anim. Ecol. 78(3), 532–539; doi:10.1111/j.1365-2656.2008.01517.x Macquiddy, B., Moreno, J.A., Kusick, B. and Mcgrath, S. 2022. Assessment of risk factors in dogs with presumptive advanced canine cognitive dysfunction. Front. Vet. Sci. 9, 958488; doi:10.3389/fvets.2022.958488 Makino, T., Rubin, C.J., Carneiro, M., Axelsson, E., Andersson, L. and Webster, M.T. 2018. Elevated proportions of deleterious genetic variation in domestic animals and plants. Genome Biol. Evol. 10(1), 276–290; doi:10.1093/gbe/evy004 Maldonado, E., Morales-Pison, S., Urbina, F. and Solari, A. 2023. Aging hallmarks and the role of oxidative stress. Antioxidants (Basel). 12(3), 651; doi:10.3390/antiox12030651 Marsden, C.D., Ortega-del Vecchyo, D., O'Brien, D.P., Taylor, J.F., Ramirez, O., Vilà, C., Marques-Bonet, T., Schnabel, R.D., Wayne, R.K. and Lohmueller, K.E. 2016. Bottlenecks and selective sweeps during domestication have increased deleterious genetic variation in dogs. Proc. Natl. Acad. Sci. U. S. A. 113(1), 152–157; doi:10.1073/pnas.1512501113 Marshall-Pescini, S., Besserdich, I., Kratz, C. and Range, F. 2016. Exploring differences in dogs' and wolves' preference for risk in a foraging task. Front. Psychol. 7, 1241; doi:10.3389/fpsyg.2016.01241 Martínez De Toda, I., Vida, C., Garrido, A. and De La Fuente, M. 2020. Redox parameters as markers of the rate of aging and predictors of life span. J. Gerontol. A. Biol. Sci. Med. Sci. 75(4), 613–620; doi:10.1093/gerona/glz033 Marx, F., Machado, G., Pezzali, J., Marcolla, C., Kessler, A., Ahlstrøm, Ø. and Trevizan, L. 2016. Raw beef bones as chewing items to reduce dental calculus in Beagle dogs. Aust. Vet. J. 94(1-2), 18–23; doi:10.1111/avj.12394 Mazzatenta, A., Carluccio, A., Robbe, D., Giulio, C.D. and Cellerino, A. 2017. The companion dog as a unique translational model for aging. Seminars. Cell. &. Develop. Biol. 70, 141–153; doi:10.1016/j.semcdb.2017.08.024 Meadows, J.R.S., Kidd, J.M., Wang, G.D., Parker, H.G., Schall, P.Z., Bianchi, M., Christmas, M.J., Bougiouri, K., Buckley, R.M., Hitte, C., Nguyen, A.K., Wang, C., Jagannathan, V., Niskanen, J.E., Frantz, L.A.F., Arumilli, M., Hundi, S., Lindblad-Toh, K., Ginja, C., Agustina, K.K., André, C., Boyko, A.R., Davis, B.W., Drögemüller, M., Feng, X.Y., Gkagkavouzis, K., Iliopoulos, G., Harris, A.C., Hytönen, M.K., Kalthoff, D.C., Liu, Y.H., Lymberakis, P., Poulakakis, N., Pires, A.E., Racimo, F., Ramos-Almodovar, F., Savolainen, P., Venetsani, S., Tammen, I., Triantafyllidis, A., Vonholdt, B., Wayne, R.K., Larson, G., Nicholas, F.W., Lohi, H., Leeb, T., Zhang, Y.P. and Ostrander, E.A. 2023. Genome sequencing of 2000 canids by the Dog10K consortium advances the understanding of demography, genome function and architecture. Genome. Biol. 24(1), 187; doi:10.1186/s13059-023-03023-7 Mech, L.D. 1988. Longevity in wild wolves. J. Mamm. 69(1), 197–198; doi:10.2307/1381776 Middleton, R.P., Lacroix, S., Scott-Boyer, M., -P.., Dordevic, N., Kennedy, A.D., Slusky, A.R., Carayol, J., Petzinger-Germain, C., Beloshapka, A. and Kaput, J. 2017. Metabolic differences between dogs of different body sizes. J. Nutr. Metab. 2017, 4535710; doi:10.1155/2017/4535710 Mihevc, S.P. and Majdič, G. 2019. Canine cognitive dysfunction and Alzheimer’s disease– Two Facets of the Same Disease?. Front. Neurosci. 13, 604; doi:10.3389/fnins.2019.00604 Mondino, A., Catanzariti, M., Mateos, D.M., Khan, M., Ludwig, C., Kis, A., Gruen, M.E. and Olby, N.J. 2023. Sleep and cognition in aging dogs. A polysomnographic study. Front. Vet. Sci. 10, 1151266. Mondino, A., Delucchi, L., Moeser, A., Cerdá-González, S. and Vanini, G. 2021. Sleep disorders in dogs: a pathophysiological and clinical review. Top. Companion Anim. Med. 43, 100516; doi:10.1016/j.tcam.2021.100516 Mongillo, P., Bertotto, D., Pitteri, E., Stefani, A., Marinelli, L. and Gabai, G. 2015. Peripheral leukocyte populations and oxidative stress biomarkers in aged dogs showing impaired cognitive abilities. Age. (Dordr). 37(3), 9778; doi:10.1007/s11357-015-9778-9 Newsome, T.M., Boitani, L., Chapron, G., Ciucci, P., Dickman, C.R., Dellinger, J.A., López-Bao, J.V., Peterson, R.O., Shores, C.R., Wirsing, A.J. and Ripple, W.J. 2016. Food habits of the world's grey wolves. Mammal Rev. 46, 255–269; doi:10.1111/mam.12067 O’Rourke, T. and Boeckx, C. 2020. Glutamate receptors in domestication and modern human evolution. Neurosci. Biobehav. Rev. 108, 341–357; doi:10.1016/j.neubiorev.2019.10.004 Oba, P.M., Sieja, K.M., Keating, S.C.J., Hristova, T., Somrak, A.J. and Swanson, K.S. 2022. Oral microbiota populations of adult dogs consuming wet or dry foods. J. Anim. Sci. 100(8), skac200; doi:10.1093/jas/skac200 Oka, K., Yamakawa, M., Kawamura, Y., Kutsukake, N. and Miura, K. 2023. The naked mole-rat as a model for healthy aging. Annu. Rev. Anim. Biosci. 11, 207–226; doi:10.1146/annurev-animal-050322-074744 Ostrander, E.A., Wayne, R.K., Freedman, A.H. and Davis, B.W. 2017. Demographic history, selection and functional diversity of the canine genome. Nat. Rev. Genet. 18(12), 705–720; doi:10.1038/nrg.2017.67 Pan, Y. 2021. Nutrients, cognitive function, and brain aging: what we have learned from dogs?. Med. Sci. (Basel). 9(4), 72; doi:10.3390/medsci9040072 Pan, Y., Landsberg, G., Mougeot, I., Kelly, S., Xu, H., Bhatnagar, S., Gardner, C.L. and Milgram, N.W. 2018. Efficacy of a therapeutic diet on dogs with signs of cognitive dysfunction syndrome: (CDS): A prospective double blinded placebo controlled clinical study. Front. Nutr. 5, 127; doi:10.3389/fnut.2018.00127 Park, T.J. and Reznick, J. 2022. Extreme physiology extreme tolerance to hypoxia, hypercapnia, and pain in the naked mole-rat. J. Muscle. Res. Cell. Motil. 44(2), 61–72; doi:10.1007/s10974-022-09623-3 Parker, H.G., Dreger, D.L., Rimbault, M., Davis, B.W., Mullen, A.B., Carpintero-Ramirez, G. and Ostrander, E.A. 2017. Genomic analyses reveal the influence of geographic origin, migration, and hybridization on modern dog breed development. Cell Rep. 19(4), 697–708; doi:10.1016/j.celrep.2017.03.079 Pendleton, A.L., Shen, F., Taravella, A.M., Emery, S., Veeramah, K.R., Boyko, A.R. and Kidd, J.M. 2018. Comparison of village dog and wolf genomes highlights the role of the neural crest in dog domestication. BMC. Biol. 16(1), 64; doi:10.1186/s12915-018-0535-2 Pérez-Montero, B., Fermín-Rodríguez, M.L., Guadalupe, M. and Cruz-López, F. 2024. Oxidative stress in canine diseases: a comprehensive review. Antioxidants. (Basel). 13(11), 1396; doi:10.3390/antiox13111396 Phochantachinda, S., Chantong, B., Reamtong, O. and Chatchaisak, D. 2021. Change in the plasma proteome associated with canine cognitive dysfunction syndrome (CCDS) in Thailand. BMC. Vet. Res. 17(1), 60; doi:10.1186/s12917-021-02744-w Pinto, C.F.D., Lehr, W., Pignone, V.N., Chain, C.P. and Trevizan, L. 2020. Evaluation of teeth injuries in Beagle dogs caused by autoclaved beef bones used as a chewing item to remove dental calculus. PLos One. 15(2), 228146; doi:10.1371/journal.pone.0228146 Pires, A., Caldeira, I., Petrucci-Fonseca, F., Viegas, I., Viegas, C., Bastos-Silveira, C. and Requicha, J. 2020. Dental pathology of the wild Iberian wolf (Canis lupus signatus): the study of a 20th century Portuguese museum collection. Vet. Anim. Sci. 9, 100100; doi:10.1016/j.vas.2020.100100 Plassais, J., Kim, J., Davis, B.W., Karyadi, D.M., Hogan, A.N., Harris, A.C., Decker, B., Parker, H.G. and Ostrander, E.A. 2019. Whole genome sequencing of canids reveals genomic regions under selection and variants influencing morphology. Nat. Commun. 10(1), 1489; doi:10.1038/s41467-019-09373-w Prpar Mihevc, S., Zakošek Pipan, M., Štrbenc, M., Rogelj, B. and Majdič, G. 2020. Nitrosative stress in the frontal cortex from dogs with canine cognitive dysfunction. Front. Vet. Sci. 7, 573155; doi: 10.3389/fvets.2020.573155 Range, F. and Marshall-Pescini, S. 2022. Comparing wolves and dogs: current status and implications for human 'self-domestication'. Trends. Cognit. Sci. 26(4), 337–349; doi:10.1016/j.tics.2022.01.003 Reicher, V., Bálint, A., Újváry, D. and Gácsi, M. 2022. Non-invasive sleep EEG measurement in hand raised wolves. Sci. Rep. 12(1), 9792; doi:10.1038/s41598-022-13643-x Romanucci, M. and Della Salda, L. 2015. Oxidative stress and protein quality control systems in the aged canine brain as a model for human neurodegenerative disorders. Oxid. Med. Cell. Longev. 2015, 940131. doi: 10.1155/2015/940131. Ruple, A., Maclean, E., Snyder-Mackler, N., Creevy, K.E. and Promislow, D. 2022. Dog models of aging. Annu. Rev. Anim. Biosci. 10, 419–439; doi:10.1146/annurev-animal-051021-080937 Saavedra, D., Añé-Kourí, A.L., Barzilai, N., Caruso, C., Cho, K.H., Fontana, L., Franceschi, C., Frasca, D., Ledón, N., Niedernhofer, L.J., Pereira, K., Robbins, P.D., Silva, A., Suarez, G.M., Berghe, W.V., Von Zglinicki, T., Pawelec, G. and Lage, A. 2023. Aging and chronic inflammation: highlights from a multidisciplinary workshop. Immun. Ageing. 20(1), 25; doi:10.1186/s12979-023-00352-w Sánchez-Villagra, M.R., Geiger, M. and Schneider, R.A. 2016. The taming of the neural crest: a developmental perspective on the origins of morphological covariation in domesticated mammals. R. Soc. Open Sci. 3(6), 160107; doi:10.1098/rsos.160107 Scheltens, P., De Strooper, B., Kivipelto, M., Holstege, H., Chételat, G., Teunissen, C.E., Cummings, J. and Van Der Flier, W.M. 2021. Alzheimer’s disease. Lancet 397(10284), 1577–1590; doi:10.1016/S0140-6736(20)32205-4 Schmid, S.M., Hoffman, J.M., Prescott, J., Ernst, H., Promislow, D.E.L., Akey, J.M., Benton, B., Borenstein, E., Castelhano, M.G., Coleman, A.E., Creevy, K.E., Crowder, K., Dunbar, M.D., Fajt, V.R., Fitzpatrick, A.L., Jeffery, U., Jonlin, E.C., Kaeberlein, M., Karlsson, E.K., Kerr, K.F., Levine, J.M., Ma, J., Mcclelland, R.L., Ruple, A., Schwartz, S.M., Shrager, S., Snyder-Mackler, N., Tolbert, K., Urfer, S.R., Wilfond, B.S. and Creevy, K.E. 2024. The companion dog as a model for inflammaging: a cross-sectional pilot study. GeroScience 46(6), 5395–5407; doi:10.1007/s11357-024-01217-w Schmidt, F., Boltze, J., Jäger, C., Hofmann, S., Willems, N., Seeger, J., Härtig, W. and Stolzing, A. 2015. Detection and quantification of β-amyloid, pyroglutamil Aβ, and tau in aged canines. J. Neuropathol. Exp. Neurol. 74(9), 912–923; doi:10.1097/NEN.0000000000000230 Schütt, T., Helboe, L., Pedersen, L.Ø., Waldemar, G., Berendt, M. and Pedersen, J.T. 2016. Dogs with cognitive dysfunction as a spontaneous model for early Alzheimer's disease: a translational study of neuropathological and inflammatory markers. J. Alzheimers. Dis. 52(2), 433–449; doi:10.3233/JAD-151085 Schwarz, F., Springer, S.A., Altheide, T.K., Varki, N.M., Gagneux, P. and Varki, A. 2016. Human-specific derived alleles of CD33 and other genes protect against postreproductive cognitive decline. Proc. Natl. Acad. Sci. USA. 113(1), 74–79; doi:10.1073/pnas.1517951112 Shearin, A.L. and Ostrander, E.A. 2010. Leading the way: canine models of genomics and disease. Dis. Mod. Mech. 3(1−2), 27–34; doi:10.1242/dmm.004358 Shi, L., Chen, S.J., Ma, M.Y., Bao, Y.P., Han, Y., Wang, Y.M., Shi, J., Vitiello, M.V. and Lu, L. 2018. Sleep disturbances increase the risk of dementia: a systematic review and meta-analysis. Sleep Med. Rev. 40, 4–16; doi:10.1016/j.smrv.2017.06.010 Shpigler, H.Y., Saul, M.C., Corona, F., Block, L., Ahmed, A.C., Zhao, S.D. and Robinson, G.E. 2017. Deep evolutionary conservation of autism-related genes. Proc. Natl. Acad. Sci. 114(36), 9653–9658; doi:10.1073/pnas.1708127114 Si, Z.Z., Zou, C.J., Mei, X., Li, X.F., Luo, H., Shen, Y., Hu, J., Li, X.X., Wu, L. and Liu, Y. 2023. Targeting neuroinflammation in Alzheimer’s disease: from mechanisms to clinical aplications. Neural. Regen. Res. 18(4), 708–715; doi:10.4103/1673-5374.353484 Sienes Bailo, P., Llorente Martín, E., Calmarza, P., Montolio Breva, S., Bravo Gómez, A., Pozo Giráldez, A., Sánchez-pascuala Callau, J.J., Vaquer Santamaría, J.M., Dayaldasani Khialani, A., Cerdá Micó, C., Camps Andreu, J., Sáez Tormo, G. and Fort Gallifa, I. 2022. The role of oxidative stress in neurodegenerative diseases and potential antioxidant therapies. Adv. Lab. Med. 3(4), 342–360; doi:10.1515/almed-2022-0111 Smolek, T., Madari, A., Farbakova, J., Kandrac, O., Jadhav, S., Cente, M., Brezovakova, V., Novak, M. and Zilka, N. 2016. Tau hyperphosphorylation in synaptosomes and neuroinflammation are associated with canine cognitive impairment. J. Comp. Neurol. 524(4), 874–895; doi:10.1002/cne.23877 Stella, J.L., Bauer, A.E. and Croney, C.C. 2018. A cross-sectional study to estimate prevalence of periodontal disease in a population of dogs (Canis familiaris) in commercial breeding facilities in Indiana and Illinois. PLos One. 13(1), 191395; doi:10.1371/journal.pone.0191395 Struillou, X., Boutigny, H., Soueidan, A. and Layrolle, P. 2010. Experimental animal models in periodontology: a review. Open Dent. J. 4, 37–47. Sundman, A.S., Pértille, F., Lehmann Coutinho, L., Jazin, E., Guerrero-Bosagna, C. and Jensen, P. 2020. DNA methylation in canine brains is related to domestication and dog-breed formation. PLos One. 15(10), 240787; doi:10.1371/journal.pone.0240787 Tancredi, D. and Cardinali, I. 2023. Being a Dog: a Review of the domestication process. Genes (Basel). 14(5), 992; doi:10.3390/genes14050992 Tchekalarova, J. and Tzoneva, R. 2023. Oxidative stress and aging as risk factors for Alzheimer's disease and Parkinson's disease: the role of the antioxidant melatonin. Int. J. Mol. Sci. 24(3), 3022; doi:10.3390/ijms24033022 Templeton, G.B., Fefer, G., Case, B.C., Roach, J., Azcarate-Peril, M.A., Gruen, M.E., Callahan, B.J. and Olby, N.J. 2023. Longitudinal analysis of canine oral microbiome using whole genome sequencing in aging companion dogs. Animals. (Basel). 13(24), 3846; doi:10.3390/ani13243846 Thakur, S., Dhapola, R., Sarma, P., Medhi, B. and Reddy, D.H. 2023. Neuroinflammation in Alzheimer's Disease: current progress in molecular signaling and therapeutics. Inflammation 46(1), 1–17; doi:10.1007/s10753-022-01721-1 Tidière, M., Gaillard, J.M., Berger, V., Müller, D.W.H., Bingaman Lackey, L., Gimenez, O., Clauss, M. and Lemaître, J.F. 2016. Comparative analyses of longevity and senescence reveal variable survival benefits of living in zoos across mammals. Sci. Rep. 6, 36361; doi:10.1038/srep36361 Tkaczyk-Wlizło, A., Kowal, K. and Ślaska, B. 2022. Mitochondrial DNA alterations in the domestic dog (Canis lupus familiaris) and their association with development of diseases: a review. Mitochondrion 63, 72–84. Turcsán, B. and Kubinyi, E. 2024. Differential behavioral aging trajectories according to body size, expected lifespan, and head shape in dogs. GeroScience 46(2), 1731–1754; doi:10.1007/s11357-023-00945-9 Twarowski, B. and Herbet, M. 2023. Inflammatory processes in Alzheimer’s disease-pathomechanism, diagnosis and treatment: a Review. Int. J. Mol. Sci. 24(7), 6518; doi:10.3390/ijms24076518 Ungvari, Z., Fekete, M., Lehoczki, A., Munkácsy, G., Fekete, J.T., Zábó, V., Purebl, G., Varga, P., Ungvari, A. and Győrffy, B. 2025. Sleep disorders increase the risk of dementia, Alzheimer's disease, and cognitive decline: a meta-analysis. GeroScience 47(3), 4899–4920; doi:10.1007/s11357-025-01637-2 Urfer, S.R., Darvas, M., Czeibert, K., Sándor, S., Promislow, D.E.L., Creevy, K.E., Kubinyi, E. and Kaeberlein, M. 2021. Canine Cognitive Dysfunction (CCD) scores correlate with amyloid beta 42 levels in dog brain tissue. Geroscience 43(5), 2379–2386; doi:10.1007/s11357-021-00422-1 Vaz, R.L., Outeiro, T.F. and Ferreira, J.J. 2018. Zebrafish as an animal model for drug discovery in Parkinson’s disease and other movement disorders: a systematic review. Front. Neurol. 9, 347; doi:10.3389/fneur.2018.00347 Vikartovska, Z., Farbakova, J., Smolek, T., Hanes, J., Zilka, N., Hornakova, L., Humenik, F., Maloveska, M., Hudakova, N. and Cizkova, D. 2021. Novel diagnostic tools for identifying cognitive impairment in dogs: behavior, biomarkers, and pathology. Front. Vet. Sci. 7, 551895; doi:10.3389/fvets.2020.551895 Wallis, C. and Holcombe, L.J. 2020. A review of the frequency and impact of periodontal disease in dogs. J. Small. Anim. Pract. 61(9), 529–540; doi:10.1111/jsap.13218 Wallis, C., Ellerby, Z., Amos, G. and Holcombe, L.J. 2025. Influence of wet and dry commercial diets on the oral microbiota of Yorkshire terriers. BMC. Vet. Res. 21(1), 290; doi:10.1186/s12917-025-04533-1 Wallis, C., Patel, K.V., Marshall, M., Staunton, R., Milella, L., Harris, S. and Holcombe, L.J. 2018. A longitudinal assessment of periodontal health status in 53 Labrador retrievers. J. Small. Anim. Pract. 59(9), 560–569; doi:10.1111/jsap.12870 Wallis, C., Pesci, I., Colyer, A., Milella, L., Southerden, P., Holcombe, L.J. and Desforges, N. 2019. A longitudinal assessment of periodontal disease in Yorkshire terriers. BMC. Vet. Res. 15(207), 1–11; doi:10.1186/s12917-019-1923-8 Wallis, C., Saito, E.K., Salt, C., Holcombe, L.J. and Desforges, N.G. 2021. Association of periodontal disease with breed size, breed, weight, and age in pure-bred client-owned dogs in the United States. Vet. J. 275, 105717; doi:10.1016/j.tvjl.2021.105717 Wan, J. and Fan, H. 2023. Oral microbiome and Alzheimer’s disease. Microorganisms 11(10), 2550; doi:10.3390/microorganisms11102550 Wang, G.D., Zhai, W., Yang, H.C., Fan, R.X., Cao, X., Zhong, L., Wang, L., Liu, F., Wu, H., Cheng, L.G., Poyarkov, A.D., Poyarkov Jr, N.A., Tang, S.S., Zhao, W.M., Gao, Y., Lv, X.M., Irwin, D.M., Savolainen, P., Wu, C.I. and Zhang, Y.P. 2013. The genomics of selection in dogs and the parallel evolution between dogs and humans. Nat. Commun. 17, 1860; doi:10.1038/ncomms2814 Webb, A.A. and Cullen, C.L. 2010. Coat color and coat color pattern-related neurologic and neuro-ophthalmic diseases. Canad. Vet. J. 51(6), 653–657. Wilkins, A.S., Wrangham, R.W. and Fitch, W.T. 2014. The “Domestication Syndrome” in Mammals: a Unified Explanation Based on Neural Crest Cell Behavior and Genetics. Genetics 197(3), 795–808; doi:10.1534/genetics.114.165423 Wise, C.F., Hammel, S.C., Herkert, N., Ma, J., Motsinger-Reif, A., Stapleton, H.M. and Breen, M. 2020. Comparative exposure assessment using silicone passive samplers indicates that domestic dogs are sentinels to support human health research. Environ. Sci. Technol. 54(12), 7409–7419; doi:10.1021/acs.est.9b06605 Woods, H.J., Li, M.F., Patel, U.A., Lascelles, B.D.X., Samson, D.R. and Gruen, M.E. 2020. A functional linear modeling approach to sleep-wake cycles in dogs. Sci. Rep. 10(1), 22233; doi:10.1038/s41598-020-79274-2 Wroe, S., Mchenry, C. and Thomason, J. 2005. Bite club: comparative bite force in big biting mammals and the prediction of predatory behaviour in fossil taxa. Proc. Biol. Sci. 272(1563), 619–625; doi:10.1098/rspb.2004.2986 Wu, C.H., Pan, X.S., Su, L.Y. and Yang, S.Y. 2023. Plasma neurofilament light chains as blood-based biomarkers for early diagnosis of canine cognitive dysfunction syndrome. Int. J. Mol. Sci. 24(18), 13771; doi:10.3390/ijms241813771 Wynne, C.D.L. 2021. Dogs’ (Canis lupus familiaris) behavioral adaptations to a human-dominated niche: A review and novel hypothesis. Adv. Study Behav. 53, 97–162. doi: 10.1016/bs.asb.2021.03.004. Yang, X., Zhang, H., Shang, J., Liu, G., Xia, T., Zhao, C., Sun, G. and Dou, H. 2018. Comparative analysis of the blood transcriptomes between wolves and dogs. Anim. Genet. 49(4), 291–302; doi:10.1111/age.12675 Yarborough, S., Fitzpatrick, A., Schwartz, S.M., Creevy, K.E., Ruple, A., Wilkins, V., Kaeberlein, M., Promislow, D., Akey, J.M., Benton, B., Borenstein, E., Castelhano, M.G., Coleman, A.E., Creevy, K.E., Crowder, K., Dunbar, M.D., Fajt, V.R., Fitzpatrick, A.L., Jeffery, U., Jonlin, E.C., Kaeberlein, M., Karlsson, E.K., Kerr, K.F., Levine, J.M., Ma, J., Mcclelland, R.L., Promislow, D.E.L., Ruple, A., Schwartz, S.M., Shrager, S., Snyder-Mackler, N., Tolbert, M.K., Urfer, S.R. and Wilfond, B.S. 2022. Evaluation of cognitive function in the Dog Aging Project: associations with baseline canine characteristics. Sci. Rep. 12(1), 13316; doi:10.1038/s41598-022-15837-9 Yordy, J., Kraus, C., Hayward, J.J., White, M.E., Shannon, L.M., Creevy, K.E., Promislow, D.E.L. and Boyko, A.R. 2020. Body size, inbreeding, and lifespan in domestic dogs. Conserv. Genet. 21(1), 137–148; doi:10.1007/s10592-019-01240-x Youssef, S.A., Capucchio, M.T., Rofina, J.E., Chambers, J.K., Uchida, K., Nakayama, H. and Head, E. 2016. Pathology of the aging brain in domestic and laboratory animals, and animal models of human neurodegenerative diseases. Vet. Pathol. 53(2), 327–348; doi:10.1177/0300985815623997 Yushkevich, P.A., Muñoz López, M., Iñiguez De Onzoño Martin, M.M., Ittyerah, R., Lim, S., Ravikumar, S., Bedard, M.L., Pickup, S., Liu, W., Wang, J., Hung, L.Y., Lasserve, J., Vergnet, N., Xie, L., Dong, M., Cui, S., Mccollum, L., Robinson, J.L., Schuck, T., De Flores, R., Grossman, M., Tisdall, M.D., Prabhakaran, K., Mizsei, G., Das, S.R., Artacho-Pérula, E., Arroyo Jiménez, M.D.M., Marcos Raba, M.P., Molina Romero, F.J., Cebada Sánchez, S., Delgado González, J.C., De La Rosa-prieto, C., Córcoles Parada, M., Lee, E.B., Trojanowski, J.Q., Ohm, D.T., Wisse, L.E.M., Wolk, D.A., Irwin, D.J. and Insausti, R. 2021. Three-dimensional mapping of neurofibrillary tangle burden in the human medial temporal lobe. Brain 144(9), 2784–2797; doi:10.1093/brain/awab262 Zhang, Q., Yang, G., Luo, Y., Jiang, L., Chi, H. and Tian, G. 2024. Neuroinflammation in Alzheimer's disease: insights from peripheral immune cells. Immun. Ageing 21(1), 38; doi:10.1186/s12979-024-00445-0 Zhang, Y., Ren, R., Yang, L., Zhang, H., Shi, Y., Okhravi, H.R., Vitiello, M.V., Sanford, L.D. and Tang, X. 2022. Sleep in Alzheimer's disease: a systematic review and meta-analysis of polysomnographic findings. Transl. Psychiatry 12(1), 136; doi:10.1038/s41398-022-01897-y Zhang, Z., Khederzadeh, S. and Li, Y. 2020. Deciphering the puzzles of dog domestication. Zool. Res. 41(2), 97–104; doi:10.24272/j.issn.2095-8137.2020.002 | ||
| How to Cite this Article |
| Pubmed Style Castro-fuentes R, Socas-pérez R. Unveiling the significance of dog domestication in cognitive dysfunction: Are wolves protected?. Open Vet. J.. 2025; 15(12): 6126-6145. doi:10.5455/OVJ.2025.v15.i12.1 Web Style Castro-fuentes R, Socas-pérez R. Unveiling the significance of dog domestication in cognitive dysfunction: Are wolves protected?. https://www.openveterinaryjournal.com/?mno=284254 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.1 AMA (American Medical Association) Style Castro-fuentes R, Socas-pérez R. Unveiling the significance of dog domestication in cognitive dysfunction: Are wolves protected?. Open Vet. J.. 2025; 15(12): 6126-6145. doi:10.5455/OVJ.2025.v15.i12.1 Vancouver/ICMJE Style Castro-fuentes R, Socas-pérez R. Unveiling the significance of dog domestication in cognitive dysfunction: Are wolves protected?. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6126-6145. doi:10.5455/OVJ.2025.v15.i12.1 Harvard Style Castro-fuentes, R. & Socas-pérez, . R. (2025) Unveiling the significance of dog domestication in cognitive dysfunction: Are wolves protected?. Open Vet. J., 15 (12), 6126-6145. doi:10.5455/OVJ.2025.v15.i12.1 Turabian Style Castro-fuentes, Rafael, and Rosy Socas-pérez. 2025. Unveiling the significance of dog domestication in cognitive dysfunction: Are wolves protected?. Open Veterinary Journal, 15 (12), 6126-6145. doi:10.5455/OVJ.2025.v15.i12.1 Chicago Style Castro-fuentes, Rafael, and Rosy Socas-pérez. "Unveiling the significance of dog domestication in cognitive dysfunction: Are wolves protected?." Open Veterinary Journal 15 (2025), 6126-6145. doi:10.5455/OVJ.2025.v15.i12.1 MLA (The Modern Language Association) Style Castro-fuentes, Rafael, and Rosy Socas-pérez. "Unveiling the significance of dog domestication in cognitive dysfunction: Are wolves protected?." Open Veterinary Journal 15.12 (2025), 6126-6145. Print. doi:10.5455/OVJ.2025.v15.i12.1 APA (American Psychological Association) Style Castro-fuentes, R. & Socas-pérez, . R. (2025) Unveiling the significance of dog domestication in cognitive dysfunction: Are wolves protected?. Open Veterinary Journal, 15 (12), 6126-6145. doi:10.5455/OVJ.2025.v15.i12.1 |