| Case Report | ||
Open Vet. J.. 2025; 15(12): 6836-6841 Open Veterinary Journal, (2025), Vol. 15(12): 6836-6841 Case Report Aggressive basal cell carcinoma in a non-facial location in a cat: Perineural and lymphatic invasion with metastasisKenichi Maeda1*, Naomi Kawasaki2, Satomi Iwai1 and Shozo Okano11Department of Small Animal Surgery 2, Kitasato University School of Veterinary Medicine, Towada, Japan 2Department of Clinical Laboratory, Kitasato University Veterinary Teaching Hospital, Towada, Japan *Corresponding Author:Kenichi Maeda. Department of Small Animal Surgery 2, Kitasato University School of Veterinary Medicine, Towada, Japan. Email: maeda [at] vmas.kitasato-u.ac.jp Submitted: 15/09/2025 Revised: 11/11/2025 Accepted: 20/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
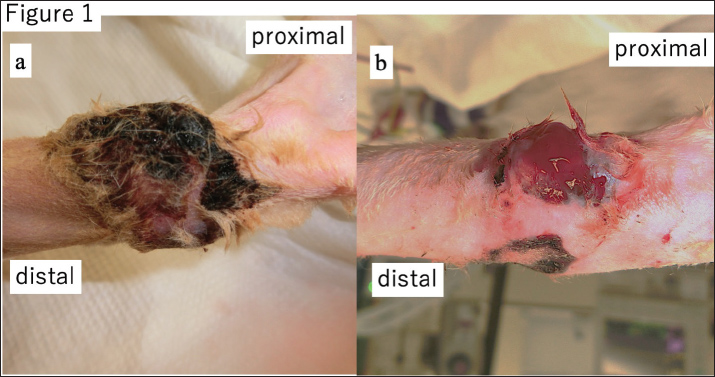
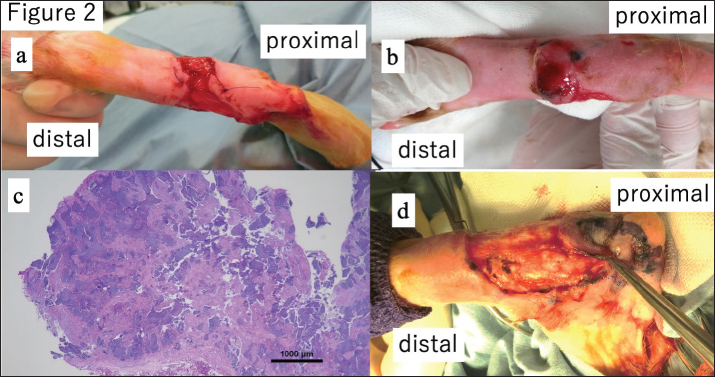
AbstractBackground: Basal cell carcinomas (BCCs) in cats are generally regarded as benign skin tumors with low metastatic potential. However, rare cases of aggressive behavior and metastasis, primarily in the context of facial lesions, have been reported. Case Description: A 12-year-old neutered male Scottish Fold cat presented with a chronic, non-healing ulcer on the left hock. The initial excision lacked a histopathological diagnosis, and the lesion recurred. Biopsy of the lesion confirmed basal cell carcinoma. Despite limb amputation with clear margins and no regional lymph node involvement, distant cutaneous nodules appeared 6 months later and were histologically confirmed as metastatic BCC with lymphatic and perineural invasion. Wide-margin resections and systemic chemotherapy with carboplatin were performed; however, further lesions developed, and the cat succumbed to progressive disease. Conclusion: This case highlights a rare and aggressive variant of feline BCC arising outside the head and neck region. The lesion demonstrated metastatic spread, perineural and lymphatic invasion, and a poor response to treatment. Early histopathological evaluation of chronic skin ulcers is essential, and recognition of aggressive subtypes may facilitate more effective clinical management. Keywords: Basal cell carcinoma, Cat, Metastasis, Perineural invasion, Chemotherapy. IntroductionBasal cell carcinomas (BCCs) are common epithelial skin tumors that originate in the basal layer of the epidermis. In cats, BCCs typically exhibit slow growth, low metastatic potential, and a favorable prognosis following surgical excision (Rubin et al., 2005; Simeonov and Simeonova, 2008). Most feline BCCs are successfully managed with complete resection, and recurrence is uncommon (Moyer et al., 2021). Consequently, the biological behavior of feline BCCs is generally regarded as benign, and limited attention has been paid to their potential systemic spread. Conversely, human BCCs are well characterized by histological subtypes. These include nodular, superficial, infiltrative, and morpheaform variants. The morpheaform subtype is known for its aggressive clinical behavior, including perineural invasion and deep tissue infiltration, which can complicate surgical management and increase recurrence risk (Salasche and Amonette, 1981; Mosterd et al., 2008). Although rare, metastatic BCC has also been reported in humans, and it typically involves the lymph nodes, lungs, or bones, and is associated with poor outcomes (Kurian et al., 2014). Metastatic BCC is extremely rare in cats, with only a few cases reported in the literature (Day et al., 1993). These reports primarily involved facial lesions and provided limited insight into the potentially aggressive nature or specific metastatic patterns of the different subtypes. The biological behavior of non-facial feline BCCs remains poorly understood. This case report describes a feline BCC arising on the hock and exhibiting unusually aggressive features, including deep local invasion, perineural and lymphatic infiltration, distant skin metastasis, and a poor response to multiple surgical interventions and systemic chemotherapy. To the best of our knowledge, this is one of the few documented cases of metastatic feline BCC originating outside the head and neck region. This case challenges the prevailing perception that feline BCC is uniformly benign and underscores the importance of early histopathological evaluation of persistent skin ulcers. Recognition of potentially aggressive BCC variants in cats is critical for timely diagnosis, treatment planning, and consideration of adjunctive therapies. Case DetailsA 12-year-old neutered male Scottish Fold cat (4.4 kg) was brought to a local veterinary clinic with a non-healing ulcer on the left hock. The lesion was excised, but histopathological examination was not performed. Despite wound closure, healing was delayed and required prolonged bandaging and antibiotic therapy. Six months postoperatively, the ulcer recurred and gradually enlarged, prompting a referral to a secondary care facility for further evaluation. Upon presentation, the ulcer had extended across the entire hock and had a scabbed surface (Fig. 1a). Removal of the scab revealed a hemorrhagic nodule (Fig. 1b), raising suspicion for neoplasia. Biopsies of the ulcer and nodule were performed, and the wound was managed with wet dressings (Fig. 2a). A new nodule appeared at the biopsy site within 1 week of the biopsy (Fig. 2b), and histopathological analysis confirmed BCC (Fig. 2c). Therefore, a wide-margin excision was recommended.
Fig. 1. (a) Palmar view of the affected limb. A thick scab had formed over a wide area of the hock. (b) After removal and cleansing, a bleeding ulcer was revealed.
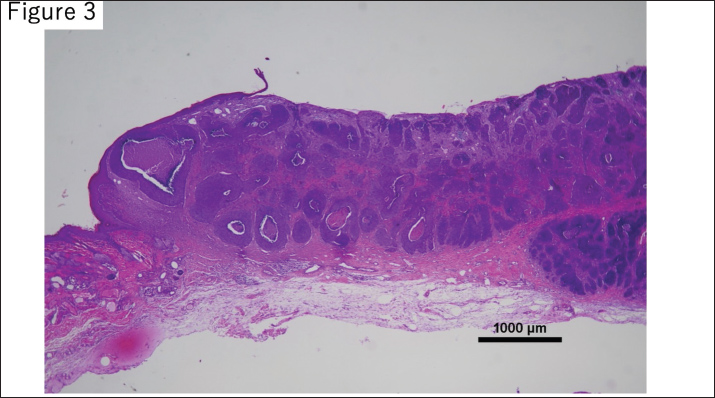
Fig. 2. (a) Following debridement, the ulcer was left open and managed with wet dressings. (b) During follow-up, a new nodule appeared at the wound margin. (c) Histopathological examination confirmed basal cell carcinoma at the debrided margin. (d) Despite plans for maximal local excision under general anaesthesia, multiple white nodules were found within the subcutaneous muscles and tendons, necessitating limb amputation. Deep invasion of the muscles and tendons was observed intraoperatively (Fig. 2d), resulting in insufficient local resection. Therefore, hindlimb amputation was performed. Histological examination confirmed an invasive BCC with clear surgical margins and no regional lymph node involvement (Fig. 3). The patient’s postoperative recovery was uneventful.
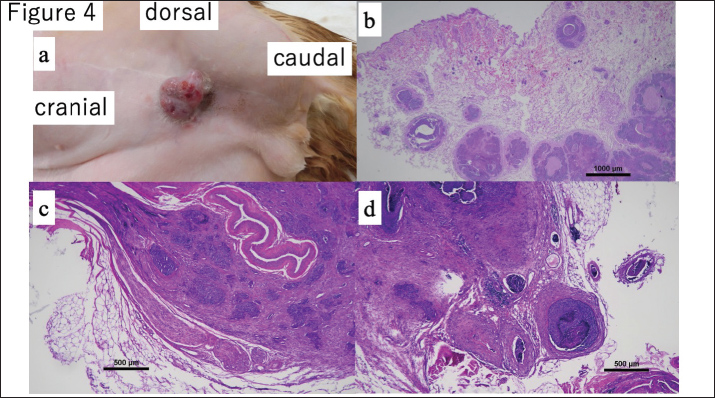
Fig. 3. Histopathological findings from the amputated left hind limb. A subcutaneous tumor with poorly defined borders was observed in the lower leg. The neoplasm was composed of epithelial cells forming alveolar structures with eosinophilic cytoplasm and round nuclei, showing moderate nuclear atypia. No tumor cell infiltration was detected at the surgical margins, bone, or lymph node, indicating complete resection. Six months after the amputation, a bleeding nodule developed in the lower abdominal skin (Fig. 4a). Cytology confirmed BCC, and the lesion was excised with 30 mm lateral margins. Histopathology revealed BCC with lymphatic and perineural invasion (Fig. 4b–d).
Fig. 4. (a) A nodular, ulcerated, and bleeding lesion developed on the lower abdomen following amputation, resembling a nodular type of BCC. (b) Histopathology showed tumor cells with eosinophilic cytoplasm, prominent nucleoli, and moderate pleomorphism. More than 20 mitotic figures per 10 high-power fields were observed. The tumor infiltrated deeply, with evidence of perineural invasion in the caudal direction, though no neoplastic involvement was found in surrounding peripheral tissues. (c) Tumor cells were also identified within lymphatic vessels. (d) Invasion of peripheral nerves by neoplastic cells was confirmed. A month later, the cat developed anorexia and vomiting. Abdominal ultrasonography revealed enlargement of a small intestinal lymph node, raising concern for metastatic disease. Fine-needle aspiration cytology suggested possible lymphoma but was negative for BCC. The enlarged lymph nodes were surgically removed. Histopathological examination confirmed gastrointestinal eosinophilic sclerosing fibroplasia (GESF). Corticosteroid treatment resulted in clinical improvement. Five months after the second BCC excision (16 months after amputation), a new nodule developed caudal to the previous lesion. Surgical excision was performed; however, securing adequate margins was challenging due to penile foreskin involvement. Histological examination revealed no tumor cells at the margins. Adjuvant chemotherapy with carboplatin (200 mg/m² every 3 weeks) was initiated after wound healing. Following initiation of carboplatin chemotherapy, the cat developed recurrent nodular lesions on the perineal skin. Owing to the multifocal and unpredictable nature of the lesions, radiotherapy was considered but not pursued. Instead, topical 5-fluorouracil was selected as a palliative treatment because of its accessibility and ease of application. The owner declined further surgical intervention, and the lesions were managed conservatively with topical 5-fluorouracil ointment. Despite these treatments, the cat’s condition worsened progressively, leading to systemic debilitation. The cat ultimately succumbed to the illness approximately 10 weeks after chemotherapy initiation. Absence of pulmonary metastasis was confirmed via thoracic radiography at every recheck, and systemic screening by CT scan was also performed concurrently with procedures requiring general anesthesia. Ethical approvalThis case report was published in accordance with the ethical standards of our institution. Informed consent was obtained from the owner of the animal for all diagnostic and therapeutic procedures. DiscussionMetastatic BCC is rare in cats, with only a few cases reported in veterinary literature (Day et al., 1993; Moyer et al., 2021). Most documented lesions originate from facial sites, and reports describing biologically aggressive or metastatic variants remain limited. This case provides important clinical and pathological insights, as it describes a non-facial feline BCC with repeated metastatic spread, including perineural and lymphatic invasion, which are features typically associated with aggressive human BCC subtypes. Because the lesion was initially misdiagnosed as a chronic ulcer, histopathological examination was not performed on the excised tissue. The delay in diagnosis of neoplasia subsequently led to multiple unsuccessful surgical interventions. Certain BCC subtypes are known to mimic benign skin conditions; nodular types may resemble dermal cysts, superficial variants can mimic eczema, and morpheaform or infiltrative types may present as indurated or ulcerated lesions, complicating early diagnosis (Kefel et al., 2012). Such diagnostic challenges may have contributed to disease progression prior to definitive treatment in this case. Although BCCs are not typically associated with malignant transformation, studies on other tumor types, such as gliomas, have suggested that incomplete resection may promote tumor aggressiveness (Swetter et al., 2003; Chaichana et al., 2010). Although this concept, sometimes referred to as “malignant degeneration,” has not been established in feline BCC, the absence of initial tumor samples in this case precludes firm conclusions. Despite limb amputation with tumor-free margins and no regional lymph node involvement, the patient subsequently developed distant cutaneous metastases. These lesions appeared distant from the primary site and exhibited characteristic features of aggressive tumor behavior, including deep infiltration, perineural invasion, and intralymphatic spread. Collectively, these findings supported true metastatic dissemination rather than local recurrence. Additional supporting factors include the rapid appearance of distant lesions, histological confirmation of lymph vascular and perineural invasion, and the anatomical distribution of new nodules. Surgical excision remains the standard treatment for feline BCC. However, this case illustrates that even wide-margin excision (> 30 mm) may be insufficient for biologically aggressive tumors. In human BCC, low-risk tumors typically require 2–3 mm margins, whereas high-risk subtypes, particularly the morpheaform subtype, require margins exceeding 13 mm (Breuninger and Dietz, 1991; Ito et al., 2015; Nakamura et al., 2018). The development of further metastases despite aggressive surgery in this case underscores the need for adjunctive therapies in selected patients. Although radiotherapy was considered, it was not pursued due to the absence of localized disease post-amputation and the unpredictable distribution of recurrent lesions. Histopathological evidence of lymphatic and perineural invasion further complicated the delineation of an appropriate radiation field. Carboplatin (200 mg/m2 every 3 weeks) was used as the chemotherapy regimen as described in detail in the Case Description section. However, its implementation was complicated, and systemic chemotherapy was subsequently initiated. However, treatment was delayed due to a gastrointestinal illness caused by GESF, which required immunosuppressive treatment. This delay may have contributed to tumor progression and subsequent lesion development during chemotherapy. Interestingly, histopathological evaluation of the metastatic lesions revealed lesions that resembled the nodular subtype, which is generally associated with indolent behavior. However, the clinical course more closely paralleled that of the morphoform variant of human BCC. This discrepancy highlights the potential limitations of applying human histological classifications directly to feline tumors and emphasizes the need for a more detailed pathological and molecular characterization of feline BCC. In human dermatopathology, mixed-type BCCs—comprising nodular, infiltrative, and superficial components—are commonly associated with larger tumor sizes, deeper invasion, necrosis, and occurrence at non-facial sites (Ghanadan et al., 2014). The morphological and clinical features observed in this case may correspond to a similar mixed or histologically ambiguous subtype with increased biological aggressiveness in cats. Most previously reported metastatic feline BCCs demonstrated lymphatic dissemination (Chaichana et al., 2010; Moyer et al., 2021). Specifically, PNI has been identified as a critical predictor of local recurrence in feline squamous cell carcinoma, where PNI-positive cases are associated with a poor prognosis (Santos et al., 2024). This histopathological feature, along with the aggressive clinical behavior observed in our case, supports earlier reports, such as Day et al. (1993), suggesting the existence of a biologically aggressive feline BCC subtype with true metastatic potential. ConclusionThis case highlights a rare but clinically significant presentation of metastatic BCC in a cat, originating from a non-facial site and demonstrating aggressive behavior despite appropriate surgical management. Unlike previously reported cases, the tumor in this case did not metastasize to visceral organs such as the lungs. Instead, it exhibited distant cutaneous spread along with confirmed lymphatic and perineural invasion. These findings challenge the conventional view of feline BCC as a uniformly benign neoplasm and underscore the importance of early histological evaluation of chronic or atypical skin lesions. Clinicians should be aware that certain subtypes may possess metastatic potential and cannot be adequately managed with surgery alone. In such cases, adjunctive therapies, including systemic chemotherapy, should be considered. AcknowledgmentsThe authors thank the NORTH LaB team and Drs. Chie Usui, Souichiro Inamoto, and Yumiko Kagawa for their contributions to the histopathological diagnosis. Conflicts of interestThe authors declare no conflicts of interest. FundingNo funding was received to assist with the preparation of this manuscript. Author contributionsKenichi Maeda: Conceptualization, Surgery, Writing—original draft. Naomi Kawasaki: Histopathological evaluation, Data interpretation. Satomi Iwai and Shozo Okano: Clinical management, Review, and editing. All authors read and approved the final manuscript. Data availabilityAll data were provided in the manuscript. ReferencesGhanadan A, Abbasi A, Rabet M, Abdollahi P, Abbasi M. 2014. Characteristics of Mixed Type Basal Cell Carcinoma in Comparison to Other BCC Subtypes. Indian J. Dermatol. 59, 57–61. Breuninger, H. and Dietz, K. 1991. Prediction of subclinical tumor infiltration in basal cell carcinoma. J. Dermatol. Surg. Oncol. 17, 574–578. Chaichana, K.L., McGirt, M.J., Laterra, J., Olivi, A. and Quiñones-Hinojosa, A. 2010. Recurrence and malignant degeneration after resection of adult hemispheric low-grade gliomas. J. Neurosurg. 112, 10–17. Day, D.G., Couto, C.G., Weisbrode, S.E. and Smeak, D.D. 1993. Basal cell carcinoma in two cats. Vet. Pathol. 30, 392–395. Dos Santos, F.F.Q., Linhares, L.C.M., Rocha, M.D.C.P., De Oliveira, K.D., Rangel, M.M.M. and De Nardi, A.B. 2024. Perineural invasion as a predictor of local recurrence in cats with squamous cell carcinoma treated with electrochemotherapy. Front. Vet. Sci. 11, 1408260. Ito, T., Inatomi, Y., Nagae, K., Nakano-Nakamura, M., Nakahara, T., Furue, M. and Uchi, H. 2015. Narrow-margin excision is a safe, reliable treatment for well-defined, primary pigmented basal cell carcinoma: an analysis of 288 lesions in Japan. J. Eur. Acad. Dermatol. Venereol. 29, 1828–1831. Kefel, S., Guvenc, P., Leander, R., Stricklin, S.M. and Stoecker, W.V. 2012. Discrimination of basal cell carcinoma from benign lesions based on extraction of ulcer features in polarized-light dermoscopy images. Skin. Res. Technol. 18, 471–475. Kurian, R.R., Di Palma, S. and Barrett, A.W. 2014. Basal cell carcinoma metastatic to parotid gland. Head Neck Pathol. 8, 349–353. Mosterd, K., Krekels, G.A., Nieman, F.H., Ostertag, J.U., Essers, B.A., Dirksen, C.D. and Nelemans, P.J. 2008. Surgical excision versus Mohs’ micrographic surgery for primary and recurrent basal-cell carcinoma of the face: a prospective randomised controlled trial with 5-years’ follow-up. Lancet. Oncol. 9, 1149–1156. Moyer, A., Vinayak, A., Heo, J., Moller, C. and Ehrhart, E.J. 2021. Metastatic basal cell carcinoma in a cat with brief review of the human and veterinary literature. Vet. Rec. Case Rep. 9, 18. Nakamura, Y., Tanese, K., Hirai, I., Kameyama, K., Kawakami, Y., Amagai, M. and Funakoshi, T. 2018. Evaluation of the appropriate surgical margin for pigmented basal cell carcinoma according to the risk factors for recurrence: a single-institute retrospective study in Japan. J. Eur. Acad. Dermatol. Venereol. 32, e453–e455. Rubin, A.I., Chen, E.H. and Ratner, D. 2005. Basal-cell carcinoma. N. Engl. J. Med. 353, 2262–2269. Salasche, S.J. and Amonette, R.A. 1981. Morpheaform basal-cell epitheliomas: a study of subclinical extensions in a series of 51 cases. J. Dermatol. Surg. Oncol. 7, 387–394. Simeonov, R. and Simeonova, G. 2008. Nucleomorphometric analysis of feline basal cell carcinomas. Res. Vet. Sci. 84, 440–443. Swetter, S.M., Boldrick, J.C., Pierre, P., Wong, P. and Egbert, B.M. 2003. Effects of biopsy-induced wound healing on residual basal cell and squamous cell carcinomas: rate of tumor regression in excisional specimens. J. Cutan. Pathol. 30, 139–146. | ||
| How to Cite this Article |
| Pubmed Style Maeda K, Kawasaki N, Iwai S, Okano S. Aggressive basal cell carcinoma in a non-facial location in a cat: Perineural and lymphatic invasion with metastasis. Open Vet. J.. 2025; 15(12): 6836-6841. doi:10.5455/OVJ.2025.v15.i12.63 Web Style Maeda K, Kawasaki N, Iwai S, Okano S. Aggressive basal cell carcinoma in a non-facial location in a cat: Perineural and lymphatic invasion with metastasis. https://www.openveterinaryjournal.com/?mno=283856 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.63 AMA (American Medical Association) Style Maeda K, Kawasaki N, Iwai S, Okano S. Aggressive basal cell carcinoma in a non-facial location in a cat: Perineural and lymphatic invasion with metastasis. Open Vet. J.. 2025; 15(12): 6836-6841. doi:10.5455/OVJ.2025.v15.i12.63 Vancouver/ICMJE Style Maeda K, Kawasaki N, Iwai S, Okano S. Aggressive basal cell carcinoma in a non-facial location in a cat: Perineural and lymphatic invasion with metastasis. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6836-6841. doi:10.5455/OVJ.2025.v15.i12.63 Harvard Style Maeda, K., Kawasaki, . N., Iwai, . S. & Okano, . S. (2025) Aggressive basal cell carcinoma in a non-facial location in a cat: Perineural and lymphatic invasion with metastasis. Open Vet. J., 15 (12), 6836-6841. doi:10.5455/OVJ.2025.v15.i12.63 Turabian Style Maeda, Kenichi, Naomi Kawasaki, Satomi Iwai, and Shozo Okano. 2025. Aggressive basal cell carcinoma in a non-facial location in a cat: Perineural and lymphatic invasion with metastasis. Open Veterinary Journal, 15 (12), 6836-6841. doi:10.5455/OVJ.2025.v15.i12.63 Chicago Style Maeda, Kenichi, Naomi Kawasaki, Satomi Iwai, and Shozo Okano. "Aggressive basal cell carcinoma in a non-facial location in a cat: Perineural and lymphatic invasion with metastasis." Open Veterinary Journal 15 (2025), 6836-6841. doi:10.5455/OVJ.2025.v15.i12.63 MLA (The Modern Language Association) Style Maeda, Kenichi, Naomi Kawasaki, Satomi Iwai, and Shozo Okano. "Aggressive basal cell carcinoma in a non-facial location in a cat: Perineural and lymphatic invasion with metastasis." Open Veterinary Journal 15.12 (2025), 6836-6841. Print. doi:10.5455/OVJ.2025.v15.i12.63 APA (American Psychological Association) Style Maeda, K., Kawasaki, . N., Iwai, . S. & Okano, . S. (2025) Aggressive basal cell carcinoma in a non-facial location in a cat: Perineural and lymphatic invasion with metastasis. Open Veterinary Journal, 15 (12), 6836-6841. doi:10.5455/OVJ.2025.v15.i12.63 |