| Research Article | ||
Open Vet. J.. 2026; 16(2): 1178-1192
Open Veterinary Journal, (2026), Vol. 16(2): 1178-1192 Research Article Hepatoprotective effect of dipyridamole against cyclophosphamide-induced hepatotoxicity in ratsAriana Seddiq Rafiq and Hiewa Othman Dyary*Department of Basic Sciences, College of Veterinary Medicine, University of Sulaimani, Sulaymaniyah, Iraq *Corresponding Author: Hiewa Othman Dyary. Department of Basic Sciences, College of Veterinary Medicine, University of Sulaimani, Sulaymaniyah, Iraq. Email: dyary.othman [at] univsul.edu.iq Submitted: 12/09/2025 Revised: 02/01/2026 Accepted: 18/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
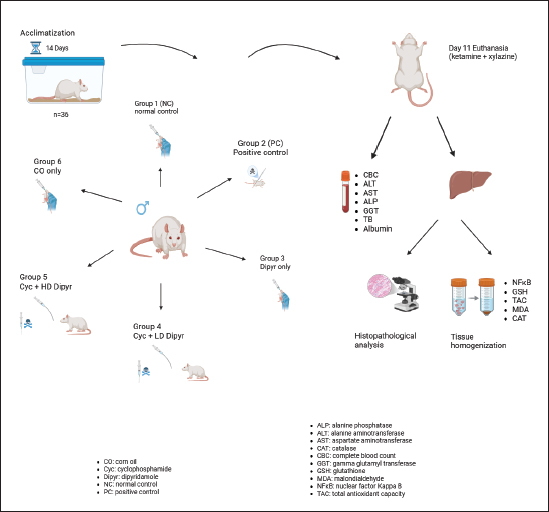
AbstractBackground: Cyclophosphamide (Cyc) is an anticancer and immunomodulatory drug, but its use is associated with toxicity, including hepatotoxicity, mediated by oxidative stress (OS), lipid peroxidation, and inflammation. Hence, coadministration of antioxidants probably mitigates Cyc-induced hepatotoxicity. Aim: This study assessed the hepatoprotective effect of dipyridamole (Dipyr) against Cyc-induced hepatotoxicity in rats. Methods: Thirty-six rats were divided randomly into six equal groups: normal control, Cyc only, Dipyr only (30 mg/kg), Cyc (200 mg/kg) with low-dose Dipyr (15 mg/kg), Cyc with high-dose Dipyr (30 mg/kg), and corn oil (CO) control. Dipyr treatment lasted 10 consecutive days, and Cyc was injected intraperitoneally on day 9. By the end of the study on day 11, body and liver weights, hematological and oxidative stress parameters, liver function biomarkers, NF-κB expression, and histopathology were assessed. Results: Cyc administration caused hepatomegaly, reduced body weight, leukocytopenia, elevated serum ALT, AST, and ALP levels, increased malondialdehyde and NF-κB levels, and reduced glutathione, catalase, and total antioxidant capacity. Histopathological examination revealed severe hepatocellular toxicity. Dipyr coadministration mitigated Cyc-induced hepatotoxicity dose-dependently, lowering lipid peroxidation, restoring antioxidant levels, suppressing hepatic NF-κB levels, and mitigating histopathological damage. However, Dipyr failed to restore leukocyte counts. Conclusion: These findings confirm Dipyr’s protective potential against Cyc-induced hepatotoxicity mainly via antioxidant and anti-inflammatory mechanisms. Dipyr may probably mitigate hepatic injuries when used as an adjuvant in patients undergoing Cyc therapy. Keywords: Antioxidant, Cyclophosphamide, Dipyridamole, Hepatotoxicity, Oxidative stress. IntroductionOxazaphosphorines are a group of bifunctional alkylating agents that have attracted significant research due to their potential to modulate the immune system and fight cancer (Bakar and Kue, 2024). One alkylating agent in the oxazaphosphorine group is cyclophosphamide (Cyc). Since Cyc has been used in clinical settings for over 40 years, a wealth of expertise has developed regarding its use in treating immune-mediated and autoimmune illnesses, as well as cancer. In addition to its effects on the immune system, Cyc possesses immunomodulatory and immunosuppressive qualities (Ahlmann and Hempel, 2016). Cyc is primarily recommended for the treatment of malignant lymphomas in stages III and IV (Ferdinandus et al., 2025). Hodgkin and non-Hodgkin lymphomas (Xu et al., 2013), lymphocytic lymphomas, Burkitt lymphomas, small lymphocytic lymphomas, and multiple myelomas (Mills et al., 2019) are a few examples. The phosphoramide mustard produced when Cyc is metabolized by liver enzymes, such as cytochrome P-450, is primarily responsible for the drug’s antineoplastic actions (Srirangan and Sabina, 2025). Cyc is initially converted to hydroxy Cyc by hepatic enzymes, which is then metabolized to aldophosphamide. Aldophosphamide cleaves to provide acrolein and phosphoramide mustard, the active alkylating agent (Dabbish et al., 2024). The phosphoramide metabolite creates cross-links within and between neighboring DNA strands at the guanine N-7 site. These irreversible changes result in apoptosis (Ahlmann and Hempel, 2016). The primary cause of Cyc cytotoxicity is its hepatic conversion to the toxic metabolite acrolein. This conversion is associated with increased reactive oxygen species (ROS) generation, leading to cell damage, lipid peroxidation, and oxidative stress (OS) (Bryan et al., 2013). It was noted that following a Cyc injection, an imbalance between antioxidant and oxidant states led to the synthesis of pro-inflammatory mediators that aggravated liver injury (Liu et al., 2012). Since the liver plays an essential role in drug and toxin metabolism, it is highly susceptible to damage from medications, including cytotoxic chemotherapy regimens. Furthermore, liver damage brought on by medicine may have a compounding effect, meaning that continued damage can exacerbate drug metabolism issues and lead to higher toxicity (van den Boogaard et al., 2022). In addition, bile duct and blood artery blockage, the creation of toxic compounds, and the infiltration of inflammatory cells into the liver parenchyma can all result in liver injury. These adverse consequences are concerning. Therefore, as new targeted cancer medications are developed, close cooperation between oncologists and hepatologists is necessary to assess the patient’s liver health and determine the most suitable medical intervention to prevent permanent hepatic damage (Choti, 2009; Totzeck et al., 2019). Dipyridamole (Dipyr) is an old drug that was first introduced in 1959 (Allahham et al., 2022). It is an antithrombotic and coronary vasodilator used as secondary stroke prophylaxis and, in combination with warfarin, after mechanical valve replacement (De Schryver et al., 2007). Dipyr is also used off-label in addition to aspirin for secondary prevention of cerebral thromboembolism (Halkes et al., 2006). Dipyr is an antiplatelet and coronary vasodilator agent that inhibits platelet phosphodiesterase and increases interstitial adenosine levels (Allahham et al., 2022). It decreases platelet aggregation by inhibiting platelet phosphodiesterase, thereby increasing levels of cyclic adenosine monophosphate (cAMP) and cyclic guanosine monophosphate (cGMP) (Beura et al., 2023). Dipyr has been shown to possess potent, little-perceived antioxidant activity (Alyasiry et al., 2022), with potential use in several fields where pathophysiological pathways depend on OS, including atherosclerosis, thrombosis, CNS-related diseases, and cancer (Ciacciarelli et al., 2015). Dipyr exerts cardioprotection against doxorubicin, as evidenced by significantly reduced OS, inflammation, and apoptosis in rat serum and cardiac tissue (Alyasiry et al., 2022). With Dipyr having the above-mentioned protective antioxidant properties, we propose that it may alleviate the hepatotoxic effects of Cyc. Materials and MethodsAnimals and housingAlbino rats were used as the experimental model, with an average weight of 172.45 grams. The animals were kept at the Research Center’s Animal House in the College of Veterinary Medicine, University of Sulaimani. The rats were housed in polypropylene cages measuring 40 × 30 × 20 cm3, with water and food provided ad libitum. A bedding of wood shavings was provided and changed every 2–3 days or as needed. The room temperature was 25 ºC with controlled ventilation and following a 12-hour dark/light cycle. At the beginning of the experiment, the rats were given a week of adaptation period. After that, they were marked on the tail with a permanent marker, indicating their number in the group. The scientific. Treatment groupsThirty-six rats were placed into six equal groups. Group 1 was designated the normal control (NC) group and received 0.1 mL of 0.9% NaCl solution by gavage for 10 consecutive days. Group 2 was a positive control (PC) group treated with Cyc (Baxter, USA) at 200 mg/kg intraperitoneally on the ninth day of the experiment (Akamo et al., 2021). Group 3 (Dipyr) received an oral dose of 30 mg/kg Dipyr (manufactured by Hikma Pharmaceuticals, Jordan) dissolved in corn oil (Dik et al., 2023). Group 4 (Cyc + LD Dipyr) received an oral low-dosage Dipyr (15 mg/kg) for 10 consecutive days (Dik et al., 2023) and was injected with Cyc (200 mg/kg) intraperitoneally on the ninth day of the experiment. Group 5 (Cyc + HD Dipyr) received a high dosage of Dipyr (30 mg/kg) and an intraperitoneal injection of Cyc on the ninth day. Group 6 (CO) served as the control group for corn oil (CO), which was used to dissolve Dipyr. The study’s experimental design is illustrated in Figure 1.
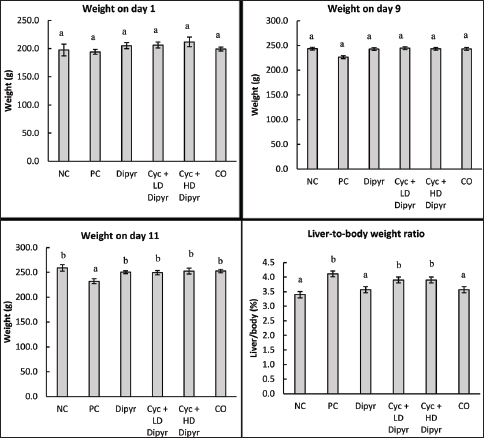
Fig. 1. Experimental design of the study. Group 1 received 0.1 ml of normal saline. Group 2 was treated with Cyc at 200 mg/kg on the ninth day. Group 3 received a dose of 30 mg/kg Dipyr for ten days. Group 4 received an oral dose of 15 mg/kg Dipyr for ten days with Cyc 200 mg/kg injection on the ninth day. Group 5 received an oral dose of 30 mg/kg Dipyr for ten days with Cyc 200 mg/kg injection on the ninth day. Group 6 served as the control group for corn oil.. Animal euthanasia and organ collectionOn the eleventh day of the experiment, the rats were anesthetized by a 0.4 ml combination of ketamine (100 mg/ml manufactured by Doga Ilac, Türkiye) and xylazine (20 mg/ml, manufactured by Beltavetpharma, Türkiye). The final solution of ketamine/xylazine contained ketamine at a rate of 33.3 mg/ml and xylazine at 6.7 mg/ml, and it was injected at a rate of 3 ml/kg. After that, the chest cavity was opened, and the animals were sacrificed by exsanguination. The collected blood was divided into two tubes for complete blood count and biochemical analyses. Then, the peritoneal cavity was opened, and the liver was collected and flushed with 0.9% NaCl solution. The livers were weighed on a sensitive balance, and 2 g of tissue was excised for homogenization by mixing with 18 ml of phosphate-buffered saline (PBS), homogenizing with a mechanical homogenizer, and then centrifuging in a cold centrifuge. The supernatant was then transferred to a separate Eppendorf tube and stored at −80 ºC until used to measure OS, antioxidants, and immunological biomarkers. The remaining liver tissue was placed in 10% formalin for histopathological processing. Body/liver weight measurementsThe rats’ body weights were measured using a digital balance on days 1, 9, and 11 during the experiment. Additionally, the freshly excised livers of sacrificed animals were weighed after washing in 0.9% NaCl solution and drying with medical gauze to determine the liver-to-body weight ratio. Biochemical analysisThe serum samples were centrifuged with a microcentrifuge (Hettich Zentrifugen EBA20, Germany). Then, serum levels of alanine transaminase (ALT), aspartate transaminase (AST), alkaline phosphatase (ALP), gamma-glutamyl transferase (GGT), total bilirubin (TB), and albumin were measured. The above-mentioned parameters were measured with Cobas C 311 (Roche/Hitachi, Switzerland). Assessment of hepatic oxidative stress and antioxidant biomarkersThe MDA (malondialdehyde) ELISA Kit was used to measure MDA levels in liver tissue. Additionally, the Rat TAC ELISA kit was used to measure TAC in liver tissue. This test helps evaluate the liver’s ability to counteract OS by neutralizing free radicals. Moreover, the Rat Glutathione (GSH) ELISA Kit was used to measure GSH levels in the liver. Furthermore, the Rat Catalase ELISA Kit was used to measure catalase enzyme levels. All kits were manufactured by Bioassay Technology Laboratory (China), and the procedures were conducted according to the manufacturer’s instructions. Measuring the nuclear factor kappa B (NF-κB) levels in the liverThe Rat Nuclear Factor Kappa B (NF-κB) ELISA kit, manufactured by Bioassay Technology Laboratory (China), was used to measure NF-κB activation levels in the liver tissue, which play a critical role in inflammation, immune response, OS, and liver disease progression. The procedure was conducted following the manufacturer’s instructions. Hematological analysisThe automated Mindray Auto Hematology Analyzer BC-2800Vet (Shenzhen Mindray Biomedical Electronics Co., Ltd., China) was used to measure hematological parameters, such as total and differential leukocyte counts, erythrocyte and thrombocyte counts, hemoglobin concentration, hematocrit, mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), and mean corpuscular hemoglobin concentration (MCHC). Histopathological procedureThe histopathological protocol used was based on a procedure from a previous study (Arsad et al., 2014). The liver tissue was allowed to fix for more than 48 hours, as was recommended by previous studies (Ming et al., 2021). The liver samples were placed in 10% formalin and then dehydrated through a gradual increase in ethanol concentration (70%, 80%, 90%, and 100%), washed, and cleared in xylol. They were subsequently rehydrated and embedded in paraffin. Then, 5-µm-thick sections were stained with hematoxylin and eosin (H&E), cover-slipped, and examined under a standard light microscope (Motic, Japan) at various magnifications. The hepatic tissue was assessed for changes in architecture, portal triads, hepatocytes, sinusoids, inflammation, and the occurrence of degeneration, necrosis, and fatty change. The alterations were classified into the following categories: Grade 0: Unremarkable histological findings (0%); Grade 1: Mild change involving up to 25%, initiation of change; Grade 2: Moderate change involving 26–50%; Grade 3: Moderate-severe change involving 51–75%; and Grade 4: Severe change involving >75%, widespread changes identified (Hassan et al., 2022). The following lesions were considered during the histopathological examination: Swelling of hepatocytes, hydropic degeneration, congestion of central veins, congestion of portal veins, congestion of sinusoidal capillaries, and the degree of inflammation. Statistical analysisThe data of the results were expressed as the mean ± SEM. A one-way analysis of variance (ANOVA) was used to analyze parametric data, followed by Duncan’s post hoc test; the Kruskal-Wallis test was used for nonparametric data. A probability level of 0.05 was considered to determine significant differences. Ethical approvalThe College of Veterinary Medicine, University of Sulaimani, approved the study protocol through the approval number AUP-2024-32. ResultsAnimal and liver weightsThe weight of rats on day 1 of the study ranged from 194.2 g to 211.8 g, with no significant differences between the groups (Fig. 2). By day 9, their weights increased to between 226.5 g and 244.5 g, yet statistical analysis revealed no significant differences between groups (p < 0.05). However, on day 11, the rat weights in the PC group averaged 232.2 g, which was significantly lower than that of the other groups. This outcome suggests that administering Cyc to the PC group had a toxic and stress-inducing effect on the rats, while the weight loss was less evident in the Dipyr-treated rats. These results demonstrate that Cyc reduces body weight in rats.
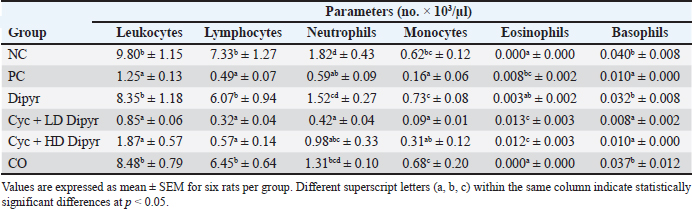
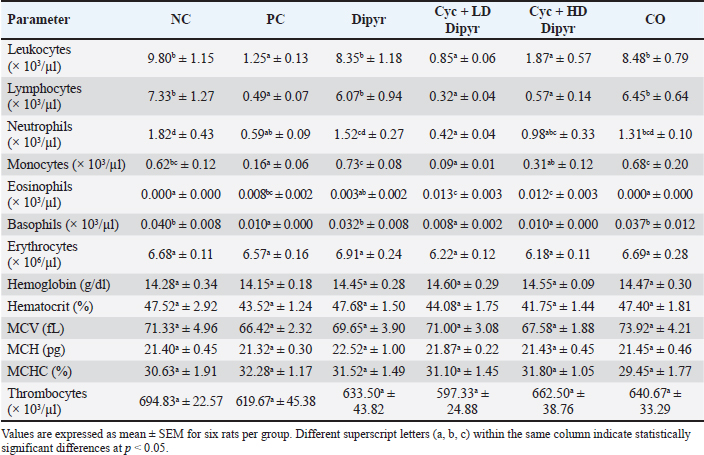
Fig. 2. Body weights on days 1, 9, and 11 of the study, as well as liver-to-body weight ratios across the different experimental groups on day 11 of the study. Data are presented as mean ± SEM (n=6 per group). Error bars labeled with different letters (a, b, c) indicate statistically significant differences at p < 0.05, as determined by one-way ANOVA followed by Duncan’s post hoc test. The liver-to-body weight ratios in the three Cyc-treated groups were significantly higher than those in the NC, Dipyr, and CO groups (Fig. 2). This increase in the liver-to-body weight ratio in the Cyc-treated groups indicates acute hepatotoxicity. However, the liver-to-body weight ratio in the groups treated with Cyc + Dipyr remained significantly higher than in the other NC, Dipyr, and CO groups, suggesting that coadministration of Dipyr with Cyc had only a limited effect on Cyc-induced hepatomegaly. Hematological parametersThe total leukocyte count decreased drastically in the Cyc-treated groups from the baseline of 9.8 × 103 leukocytes/µL in the NC (Table 1). The differential leukocyte count revealed that the leukocytopenia following Cyc treatment was primarily due to a decrease in lymphocyte counts. These findings indicate that Cyc treatment induces marked leukocytopenia, primarily through lymphocyte depletion, whereas Dipyr treatment did not prevent leukocytopenia. Table 1. Total and differential leukocyte profiles across experimental groups.
The erythrocyte concentration in the NC group averaged 6.68 × 106/µl, with no difference from the other groups. Additionally, no statistical differences were observed among the experimental groups for hemoglobin, hematocrit, MCV, MCH, MCHC, and thrombocyte concentrations (Table 2). Table 2. Comparative hematological parameters among experimental groups.
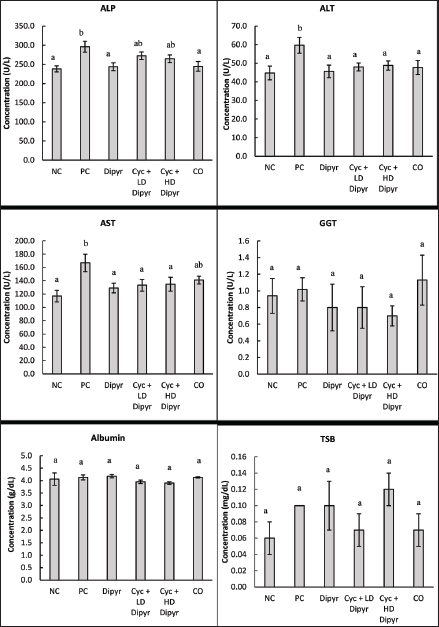
Liver function biomarkers in serumThe serum concentrations of ALP, ALT, AST, GGT, albumin, and TSB were compared statistically among the groups, and the results are shown in Figure 3. Cyc administration in the PC group caused significant hepatic injury, evidenced by elevated serum ALP, ALT, and AST levels compared with the others. On the other hand, coadministration of Dipyr with Cyc in groups 4 and 5 lowered the concentrations of these enzymes to near normal levels observed in the NC group rats. However, GGT, albumin, and total serum bilirubin levels were not affected by the treatments, showing no significant differences among the groups.
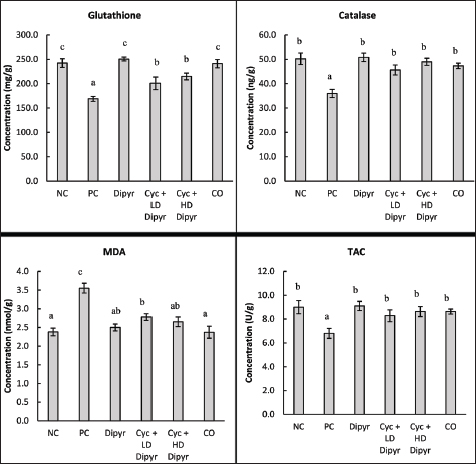
Fig. 3. Serum levels of ALP, ALT, AST, GGT, albumin, and total bilirubin. Values represent the means of six rats per group (columns) ± SEM (error bars). Different letters (a, b) denote significant differences between the groups at p < 0.05, as determined by one-way ANOVA followed by Duncan’s post hoc test. Liver oxidative stress and antioxidant biomarkersGSH levels reduced by more than 30% from the baseline of 242.33 mg/g in the NC to 168.77 mg/g in the PC. Coadministration of Dipyr with Cys in groups 4 and 5 increased GSH levels significantly and dose-dependently compared to the PC, even though the concentrations remained below the baseline values set by the NC group (Fig. 4). These findings indicate that Cyc significantly depletes GSH levels, while coadministration of Dipyr restores GSH dose dependently, though not to baseline levels. Administration of Cyc in the PC reduced catalase levels by 28.3% from the baseline of 50.2 ng/g to 35.85 ng/g (Fig. 4), while coadministration of Cyc with Dipyr elevated the catalase levels significantly close to that of the NC. These results indicated that Cyc significantly reduces catalase activity, whereas coadministration with Dipyr effectively restores catalase levels to near baseline levels, depending on the dosage. The concentration of MDA was 50% higher in the PC group than in the NC, while coadministration of Dipyr with Cyc reduced MDA levels in a dose-dependent manner (Fig. 4). The significantly elevated MDA levels in the PC group compared with the NC and other groups indicate increased lipid peroxidation, confirming pronounced OS and tissue injury. On the other hand, the dose-dependent reduction of MDA levels with Dipyr coadministration demonstrates its protective antioxidant effect, highlighting the relevance of MDA as a sensitive biomarker for evaluating oxidative damage and assessing the efficacy of therapeutic interventions. Treatment with Cyc significantly reduced TAC levels in the PC group, highlighting oxidative damage. Dipyr coadministration ameliorated this effect in a dose-dependent manner, with the higher dosage nearly normalizing TAC levels, suggesting Dipyr’s effectiveness in stimulating antioxidant defenses against Cyc-induced OS (Fig. 4).
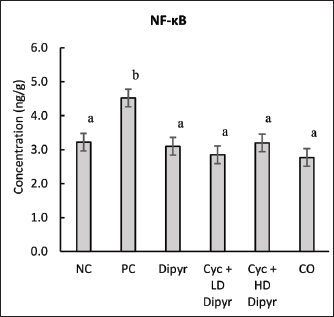
Fig. 4. Levels of glutathione, catalase, MDA, and TAC in the hepatic tissue. Values represent the means of six rats per group (columns) ± SEM (error bars). Different letters (a, b, c) denote significant differences between the groups at p < 0.05, as determined by one-way ANOVA followed by Duncan’s post hoc test. Dipyr alone maintained GSH, CAT, TAC, and MDA at levels comparable to the NC, suggesting intrinsic antioxidant activity without inducing OS. Moreover, the CO group showed no significant differences from NC in any parameter, confirming that the vehicle had no adverse impact. Overall, the results demonstrated that Cyc administration enhanced marked OS, evidenced by decreased GSH, CAT, and TAC levels, alongside elevated MDA levels. Dipyr coadministration counteracted these changes in a dose-dependent manner, underscoring its antioxidant efficacy in palliating Cyc-induced oxidative damage. NF-κB levels in the hepatic tissueNF-κB levels in the PC group were 40% higher than baseline (3.22 ng/g) in the NC group, reflecting enhanced OS-driven inflammation. On the other hand, Dipyr coadministration significantly reduced NF-κB activation in a dose-dependent manner, restoring values to near-normal levels (Fig. 5). These outcomes imply that Dipyr exerts anti-inflammatory capabilities against Cyc-induced tissue damage, possibly by modulating redox-sensitive transcription pathways, such as NF-κB.
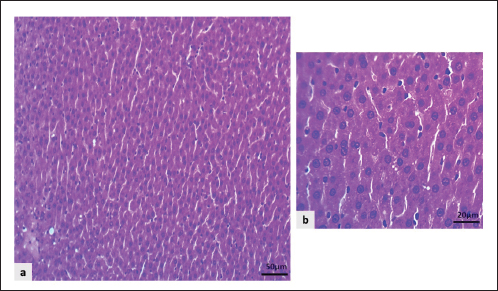
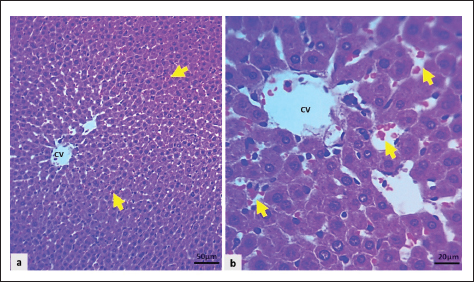
Fig. 5. NF-κB levels in the hepatic tissue. Values represent the means of six rats per group (columns) ± SEM (error bars). Different letters (a, b) denote significant differences between the groups at p < 0.05, as determined by one-way ANOVA followed by Duncan’s post hoc test. Histopathological changes of the hepatic tissueThe microscopic liver section presented normal histologic organization and features in the NC group. It characterized by well-organized hepatic lobules with the central vein surrounded by plates of intact hepatocytes, in which rows of liver cells separated by sinusoidal capillary, at each hepatic periphery, contain typical structures of the portal area including branches of the hepatic artery, portal vein, and hepatic bile duct without any signs of circulatory disturbance or inflammation (Fig. 6a, b).
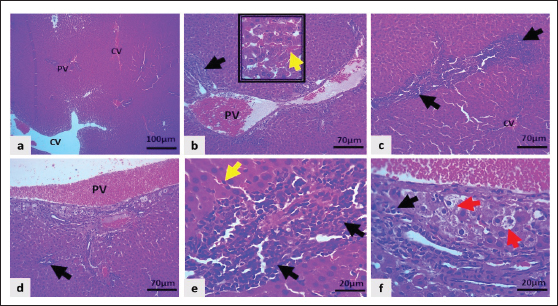
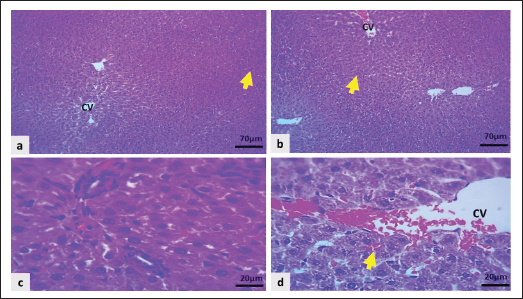
Fig. 6. Light microscopic sections of the liver in the NC group revealed: a and b: The regular hepatocyte arrangement and intact structures of the sinusoidal capillaries (H&E stain). The liver sections from PC group rats showed a higher score and more severe hepatic changes than those from the NC group. They revealed marked swelling of hepatocytes, enlargement of hepatocytes with a centrally located nucleus that leads to narrowing of sinusoidal capillaries with hydropic degeneration of hepatocytes, enlarged, pale vacuole cells with eccentrically located nuclei and marked congestion of the central vein, portal vein, and sinusoidal capillaries, along with noticeable infiltration of neutrophils, particularly surrounding the central vein, periportal area, and even within liver parenchyma (Fig. 7a–f).
Fig. 7. Light microscopic section of the liver in the PC group showed: a–e: Severe congestion of the central vein (CV), portal vein (PV), and sinusoidal capillaries (inset and yellow arrows), marked infiltration of neutrophils in peri-centrilobular regions and periportal area (black arrows). f: Marked hepatocytic swelling and hydropic degeneration indicated by red arrows (H&E stain).
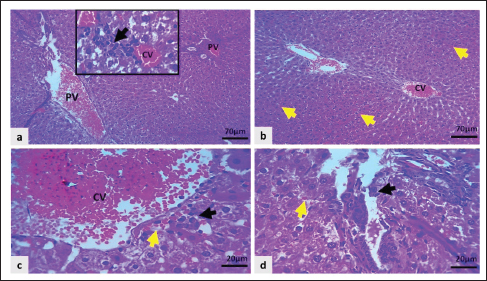
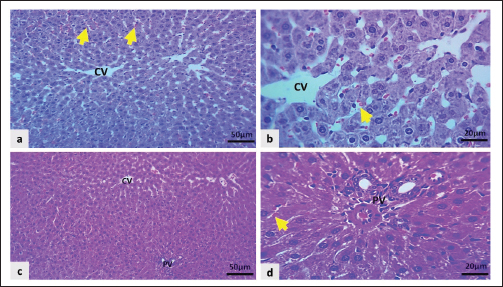
Fig. 8. Light microscopic section of the liver in the CO group showed: a and b: Uncongested central vein (CV) with mild sinusoidal congestion (black arrows), and hepatocytes swollen mildly (H&E stain). Rats in the CO showed mild hepatocyte swelling with a mild degree of congestion in the sinusoid, while the central vein and portal vein remained normal (Fig. 8a and b). The Cyc + HD Dipyr revealed focal congestion of the central vein. At the same time, the portal area remained normal, with mild congestion of sinusoidal capillaries and moderate swelling of hepatocytes. The hepatocytes were swollen and pale, looking like a cloud (Fig. 9a–d).
Fig. 9. Light microscopic sections of the liver in the Cyc + HD Dipyr group displayed: a, b: Normal central vein (CV) and portal vein (PV) structures with mild congestion of the sinusoidal capillaries (yellow arrows), and moderate cloudy swelling of hepatocytes. c and d: Mild focal central vein and sinusoidal congestion (yellow arrows), and moderate cellular swelling (H&E stain).
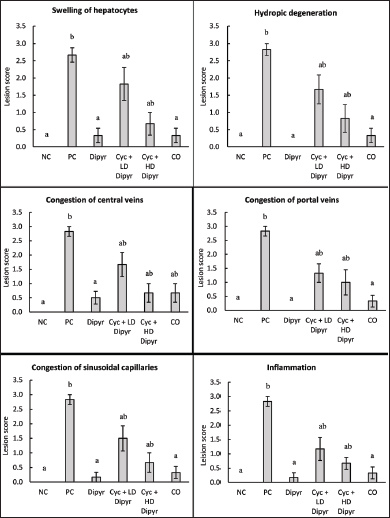
Fig. 10. Light microscopic sections of the liver in the Cyc + LD Dipyr group exhibited: a–d: Severe congestion of the central and portal veins, moderate congestion of sinusoidal capillaries (yellow arrows), moderate cellular swelling, and mild infiltration of neutrophils in centrilobular and perilobular regions, as in the inset and indicated by black arrows H&E stain). The administration of Dipyr with Cyc improved the liver lesions by reducing the pathologic scores. For example, the Cyc + LD Dipyr group was characterized by marked congestion of the hepatic vasculature with moderate hepatocytic swelling, with only a mild degree of inflammatory reaction (Fig. 10a–d). The combination of Cyc + HD Dipyr attenuated the alteration to moderate congestion of the central vein. At the same time, the portal vasculature remained intact, with mild congestion of sinusoidal capillaries and moderate cloudy swelling of liver cells without any evidence of inflammatory reactions (Fig. 11a–d). The histopathological lesion scores are illustrated in Figure 12.
Fig. 11. Light microscopic sections of the liver in the Cyc + HD Dipyr group showed: a and b: Moderate congestion of the central veins (CV) with mild congestion in the sinusoidal capillaries (yellow arrows) and moderate cloudy hepatocytic swelling (H&E stain).
Fig. 12 Grading of lesions across the experimental groups. Scores are shown as columns, while the error bars indicate standard errors of the means. Different letters indicate statistical differences between the groups at p < 0.05, as determined by the Kruskal-Wallis test. DiscussionCyc is an anticancer and immunomodulating agent known for causing hepatotoxicity (Srirangan and Sabina, 2025). This study assessed the protective potential of Dipyr against Cyc-induced hepatotoxicity in rats, with a special focus on changes in body and liver weights, hematological parameters, liver function biomarkers, OS indices, inflammatory mediators, and histopathological alterations. The findings confirmed Cyc’s well-established hepatotoxic and immunosuppressive effects. On the other hand, the results also highlighted Dipyr’s dose-dependent ameliorative effects on OS, inflammation, and hepatic tissue injury. Body weight changes are indicators of the general health status, and fluctuations often indicate chemical-induced systemic toxicity in animal models (Nevzorova et al., 2020). In this study, no significant differences in body weight were observed from day 1 to day 9, when Cyc was not yet administered to the rats. However, the weights of the rats in the PC group were significantly lower than those of the other groups. This decline in body weight highlights Cyc’s toxic and stress-inducing effects, which may have resulted from reduced appetite, nutrient absorption, or tissue metabolism. A significant reduction in body weight following Cyc administration was also reported by other researchers (Zhang et al., 2021). In contrast, Dipyr-treated rats exhibited less pronounced weight loss, suggesting that Dipyr had a protective effect against Cyc-induced systemic toxicity, likely through its antioxidant and cytoprotective properties. The liver-to-body weight ratio serves as a valuable measure of hepatomegaly and hepatic injury (Hussein et al., 2024; El Fatimi et al., 2025). Cyc treatment in our study resulted in a significant increase in liver-to-body weight ratios, consistent with acute hepatotoxicity, as reported by other researchers (Alam et al., 2023). The hepatomegaly in the PC and the other two Cyc-treated groups is probably attributed to hepatocellular degeneration and swelling, and vascular congestion (Ozturk et al., 2023), as confirmed by the histopathological findings. Although Dipyr partially reduced the hepatomegaly induced by Cyc, the ratios remained higher than those in the NC, CO, and Dipyr-only groups. This outcome implies that, although Dipyr mitigates some aspects of hepatic injury, its effect on organ enlargement was limited within the study’s short timeframe. Hematological analysis revealed significant leukocytopenia in Cyc-treated groups, accompanied by profound lymphocyte depletion. This result coincides with the well-documented myelosuppressive effects of Cyc, which poses toxic impacts on proliferating lymphoid cells (Kang et al., 2022; Basu et al., 2025). However, treatment with Dipyr at low and high doses did not reverse the myelosuppressive effect of Cyc. This outcome reinforced the fact that Cyc is an immunosuppressive and myelosuppressive agent, as reported by other studies (Basu et al., 2025), while showing that Dipyr does not protect against Cyc-induced bone marrow suppression. The results confirmed that Dipyr had hepatoprotective effects, without a significant impact on bone marrow tissue. On the other hand, Cyc treatment did not significantly affect erythrocyte counts, hemoglobin, hematocrit, and platelet indices. This is because the rats were sacrificed 2 days after Cyc treatment, a time interval insufficient to cause erythrocytopenia, as erythrocytes have a long lifespan. This outcome was expected given the short duration of the study relative to the erythrocytes’ long lifespan. The liver biochemical markers further supported the hepatotoxic effects of Cyc, as evidenced by elevated serum ALT, AST, and ALP levels. These results are consistent with earlier reports of Cyc’s hepatotoxic effects, as evidenced by elevated levels of liver function biomarkers (Mostafa et al., 2022; Qian et al., 2022; Alam et al., 2023). Dipyr coadministration reduced the levels of these enzymes toward baseline, especially at the higher dosage, showing its hepatoprotective effect. However, serum GGT and albumin concentrations were not altered across groups. These results demonstrated that the Cyc-induced liver injury was acute and had not progressed to protein synthesis impairment and did not alter bile metabolism within the study timeframe. Cyc-induced hepatotoxicity is primarily mediated by OS induction (Turedi, 2023), as evidenced by the current study. GSH and TAC concentrations decreased, and catalase activity was significantly suppressed, while MDA levels increased in the PC. These outcomes confirmed that Cyc alters the cellular redox balance, enhancing lipid peroxidation and depleting endogenous antioxidants. The reduced endogenous antioxidants and increased lipid peroxides ultimately cause membrane damage and cell death (Abdulrahman et al., 2024; Mohammed-Rashid and Dyary, 2025). Dipyr coadministration restored GSH and catalase concentrations, improved TAC, and reduced MDA levels. The antioxidant effect of Dipyr in this study was consistent with previous reports about its antioxidant properties (Alyasiry et al., 2022). The MDA-reducing ability of Dipyr demonstrates its capacity to inhibit lipid peroxidation, a critical factor in Cyc-induced liver toxicity (Cengiz et al., 2022). The activation of NF-κB by Cyc also played a significant role in the pathogenesis of Cyc-induced hepatotoxicity, as the level of the inflammatory marker was significantly higher in the PC group than in the NC group. NF-κB activation reflects an increased inflammatory response induced by OS. In contrast, coadministration of Dipyr with Cyc inhibited NF-κB expression in a dose-dependent manner, restoring its levels to near baseline values. This outcome highlights Dipyr’s hepatoprotective effect through redox-sensitive transcription factors, such as NF-κB (Pantano et al., 2006). In this study, NF-κB levels were quantified using a total NF-κB ELISA kit, which measures overall NF-κB protein concentration in tissue homogenates rather than distinguishing between cytosolic and nuclear fractions. Therefore, the results reflect total NF-κB content rather than nuclear translocation or transcriptionally active NF-κB. Histopathological examination of hepatic tissues further confirmed the biochemical findings and provided insight into the cellular toxicities caused by Cyc. The livers of Cyc-only-treated rats exhibited classical features of hepatocellular toxicity, evidenced by hydropic degeneration, vascular congestion, narrowing of sinusoids, and infiltration of inflammatory cells, especially neutrophils. On the other hand, concomitant treatment with Cyc and Dipyr significantly ameliorated these pathological changes to mild congestion, moderate hepatocyte swelling, and minimal infiltration of inflammatory cells. Interestingly, the ameliorative effect of Dipyr was more evident at the higher drug dosage. The Dipyr-only and CO groups showed the typical hepatic tissue architecture, further confirming that the changes observed in the PC were specifically due to Cyc treatment. In the context of previously evaluated antioxidant agents, Dipyr appears particularly promising due to its multimodal pharmacodynamic profile and its ability to target several key mechanisms simultaneously (Guo et al., 2023; Martynov et al., 2025), a characteristic that many conventional antioxidants lack. Most antioxidants assessed in earlier Cyc-hepatotoxicity models, such as vitamin E (Ben Amara et al., 2011), N-acetylcysteine (Lasram et al., 2014), quercetin (Gupta et al., 2010), or silymarin (El-Gazayerly et al., 2014), primarily exert their effects through radical scavenging or replenishment of intracellular thiol pools. While effective to a certain extent, these agents typically provide narrow-spectrum protection, focusing predominantly on oxidative endpoints without meaningfully modulating downstream inflammatory transcription factors or microvascular dysfunction. The aforementioned results provide compelling evidence that Cyc induces hepatotoxicity via oxidative stress, lipid peroxidation, and inflammation. Although Dipyr could not prevent leukocytopenia, its coadministration ameliorated these harmful effects principally by stimulating antioxidant defenses and suppressing NF-κB. These results highlight Dipyr’s potential to reduce hepatocellular injury during Cyc therapy. The findings of this study hold potential translational relevance for both veterinary and human clinical practice. Cyc remains widely used across species as an antineoplastic and immunomodulatory drug, yet its dose-limiting hepatotoxicity continues to challenge treatment tolerance and therapeutic continuity. Dipyr is an already licensed and well-characterized drug with a favorable safety profile in humans and animals that may offer a practical hepatoprotective adjunct during Cyc therapy. Its multimodal antioxidant, anti-inflammatory, and microcirculatory effects could help mitigate chemotherapy-associated hepatic injury, improve treatment tolerability, and potentially allow safer dosing regimens. Despite this study’s strengths, several limitations should be acknowledged. The experimental duration was relatively short, capturing only acute hepatotoxic and nephrotoxic changes, which may limit the clinical relevance of the findings. Such a short timeframe does not reflect the cumulative or chronic nature of doxorubicin-induced toxicity, nor does it allow evaluation of dipyridamole’s long-term protective capacity. In addition, only two doses of dipyridamole were tested, which restricts the ability to establish a full dose–response relationship or determine the minimally effective dose. Key functional assessments, such as bile flow measurement, mitochondrial respiration assays, and genomic markers of oxidative stress (e.g., Nrf2/HO-1 pathway activation), were also not performed, limiting the mechanistic depth of the study. Future research employing longer experimental timelines, broader dose ranges, and detailed molecular analyses in clinically relevant models will be essential to fully elucidate dipyridamole’s protective potential. ConclusionThis study provided compelling evidence that Cyc induces hepatotoxicity through OS, lipid peroxidation, and inflammation. Even though Dipyr could not ameliorate Cyc-induced leukocytopenia, it effectively reduced hepatic OS, attenuated inflammation, and preserved liver architecture. These findings indicate Dipyr’s potential as a supportive agent during Cyc therapy in patients to reduce the drug’s side effects on the liver. AcknowledgmentsThe University of Sulaimani provided the necessary laboratories to conduct this study. FundingThis research received no funds. Authors’ contributionsA.S.R. contributed to the conceptualization and design of the study, conducted the research, and drafted the manuscript. H.O.D. co-conceptualized the study, analyzed data, and reviewed and finalized the manuscript. Conflicts of interestThe authors declare no conflict of interest. Data availabilityAll data supporting the findings of this study are available within the manuscript, and no additional data sources are required. ReferencesAbdulrahman, H., Dyary, H., Mohammed, R., Hamad, D., Star, F. and Saeed, N. 2024. Preventing free radical damage: the significance of including antioxidants in diet to strengthen immunity. Open. Vet. J. 14, 1526; doi:10.5455/OVJ.2024.v14.i7.2 Ahlmann, M. and Hempel, G. 2016. The effect of cyclophosphamide on the immune system: implications for clinical cancer therapy. Cancer Chemother. Pharmacol. 78, 661–671; doi:10.1007/s00280-016-3152-1 Akamo, A.J., Rotimi, S.O., Akinloye, D.I., Ugbaja, R.N., Adeleye, O.O., Dosumu, O.A., Eteng, O.E., Amah, G., Obijeku, A. and Cole, O.E. 2021. Naringin prevents cyclophosphamide-induced hepatotoxicity in rats by attenuating oxidative stress, fibrosis, and inflammation. Food Chem. Toxicol. 153, 112266; doi:10.1016/j.fct.2021.112266 Alam, M.F., Ajeibi, A.O., Safhi, M.H., Alabdly, A.J.A., Alshahrani, S., Rashid, H., Qadri, M., Jali, A.M., Alqahtani, S., Nomier, Y., Moni, S.S., Khalid, M. and Anwer, T. 2023. Therapeutic potential of capsaicin against cyclophosphamide-induced liver damage. J. Clin. Med. 12, 911; doi:10.3390/jcm12030911 Allahham, M., Lerman, A., Atar, D. and Birnbaum, Y. 2022. Why not dipyridamole: a review of current guidelines and re-evaluation of utility in the modern era. Cardiovasc. Drugs. Ther. 36, 535–532; doi:10.1007/s10557-021-07224-9 Alyasiry, E., Janabi, A. and Hadi, N. 2022. Dipyridamole ameliorates doxorubicin-induced cardiotoxicity. J. Med. Life 15, 1184–1190; doi:10.25122/jml-2021-0199 Arsad, S., Esa, N. and Hamzah, H. 2014. Histopathologic changes in liver and kidney tissues from male Sprague Dawley rats treated with Rhaphidophora decursiva (Roxb.) Schott extract. J. Cytol. Histol. S4, 1–6; doi:10.4172/2157-7099.S4-001 Bakar, S.N. and Kue, C.S. 2024. A combination therapy of cyclophosphamide and immunomodulating agents in cancer. Curr. Cancer. Drug. Targets. 1187, 1187–1199; doi:10.2174/0115680096314791240830111909 Basu, S., Mukherjee, S., Chakraborty, S. and Dey, A. 2025. Role of traditional Indian plants on treating myelosuppression caused by chemotherapy with special reference to cyclophosphamide. Nucleus 68, 195–211; doi:10.1007/s13237-023-00461-3 Ben Amara, I., Soudani, N., Troudi, A., Bouaziz, H., Boudawara, T. and Zeghal, N. 2011. Antioxidant effect of vitamin E and selenium on hepatotoxicity induced by dimethoate in female adult rats. Ecotoxicol. Environ. Saf. 74, 811–819; doi:10.1016/j.ecoenv.2010.11.007 Beura, S.K., Dhapola, R., Panigrahi, A.R., Yadav, P., Kumar, R., Reddy, D.H. and Singh, S.K. 2023. Antiplatelet drugs: potential therapeutic options for the management of neurodegenerative diseases. Med. Res. Rev. 43, 1835–1877; doi:10.1002/med.21965 Bryan, H.K., Olayanju, A., Goldring, C.E. and Park, B.K. 2013. The Nrf2 cell defence pathway: keap1-dependent and -independent mechanisms of regulation. Biochem. Pharmacol. 85, 705–717; doi:10.1016/j.bcp.2012.11.016 Cengiz, M., Kutlu, H.M., Peker Cengiz, B. and Ayhancı, A. 2022. Escin attenuates oxidative damage, apoptosis and lipid peroxidation in a model of cyclophosphamide-induced liver damage. Drug. Chem. Toxicol. 45, 1180–1187; doi:10.1080/01480545.2020.1810262 Choti, M.A. 2009. Chemotherapy-associated hepatotoxicity: do we need to be concerned?. Ann. Surg. Oncol. 16, 2391–2394; doi:10.1245/s10434-009-0512-7 Ciacciarelli, M., Zerbinati, C., Violi, F. and Iuliano, L. 2015. Dipyridamole: a drug with unrecognized antioxidant activity. Curr. Top. Med. Chem. 15, 822–829; doi:10.2174/1568026615666150220111942 Dabbish, E., Scoditti, S., Shehata, M.N., Ritacco, I., Ibrahim, M.A., Shoeib, T. and Sicilia, E. 2024. Insights on cyclophosphamide metabolism and anticancer mechanism of action: a computational study. J. Comput. Chem. 45, 663–670; doi:10.1002/jcc.27280 De Schryver, E., Algra, A. and Van Gijn, J. 2007. Dipyridamole for preventing stroke and other vascular events in patients with vascular disease. Cochrane Database Syst. Rev. 3, CD001820; doi:10.1002/14651858.CD001820.pub3 Dik, B., Parlak, T.M., Tufan, O., Ozgur, F.B. and Er, A. 2023. Comparative evaluation of single and combined efficacy of dipyridamole, ketotifen and quercetin on cyclosporine induced hepatorenal toxicity. Pol. J. Vet. Sci. 26, 549–557; doi:10.24425/pjvs.2023.148275 El Fatimi, H., El-Mansoury, B., Fdil, N., Ait Tastift, M., Ouhaddou, S., Lafhal, K., Tamegart, L., Es-said, S., Ezoubeiri, A. and Gamrani, H. 2025. Therapeutic approaches using natural substances on the thioacetamide-induced animal model of acute liver failure: uHPLC analysis, Biochemical and Histopathological studies. PRENAP 100345, 100345; doi:10.1016/j.prenap.2025.100345 El-Gazayerly, O., Makhlouf, A., Soelm, A. and Mohmoud, M. 2014. Antioxidant and hepatoprotective effects of silymarin phytosomes compared to milk thistle extract in CCl4 induced hepatotoxicity in rats. J. Microencapsul. 31, 23–30; doi:10.3109/02652048.2013.805836 Ferdinandus, J., Kaul, H., Fosså, A., Hüttmann, A., Keil, F., Ko, Y.D., Hitz, F., Schwarz, M., Trenker, C., Kerkhoff, A., Staib, P., Wille, K., Dresel, I., Hahn, D., Hertenstein, B., Moosmann, P., Mey, U., Balabanov, S., Armytage, T., Roncolato, F., Hellmuth, J.C., Hertzberg, M., Kobe, C., Klapper, W., Baues, C., Eich, H.T., Kreissl, S., Fuchs, M., Jablonski, J., Schneider, G., Tharmaseelan, H., Eichenauer, D.A., Von Tresckow, B., Borchmann, P. and Bröckelmann, P.J. 2025. Positron emission tomography–guided brentuximab vedotin, etoposide, cyclophosphamide, doxorubicin, dacarbazine, and dexamethasone in older patients with advanced-stage classic Hodgkin lymphoma: a prospective, multicenter, single-arm, phase II cohort of the German Hodgkin Study Group HD21 Trial. J. Clin. Oncol. 43, 2974–2985; doi:10.1200/JCO-25-00439 Guo, W., Dong, X., Li, Y., Li, C., Tian, Y., Gao, H., Li, T., Zhu, H., Wang, J. and Yang, C. 2023. Co-amorphous formulation of dipyridamole with p-hydroxybenzoic acid: underlying molecular mechanisms, physical stability, dissolution behavior and pharmacokinetic study. Eur. J. Pharm. Biopharm. 184, 139–149; doi:10.1016/j.ejpb.2023.01.018 Gupta, C., Vikram, A., Tripathi, D.N., Ramarao, P. and Jena, G.B. 2010. Antioxidant and antimutagenic effect of quercetin against DEN induced hepatotoxicity in rat. Phytotherapy Res. 24, 119–128; doi:10.1002/ptr.2883 Halkes, P., Van Gijn, J., Kappelle, L., Koudstaal, P. and Algra, A. 2006. Aspirin plus dipyridamole versus aspirin alone after cerebral ischaemia of arterial origin (ESPRIT): randomised controlled trial. Lancet 367, 1665–1673; doi:10.1016/s0140-6736(06)68734-5 Hassan, S.M.A., Saeed, A.K., Rahim, O.O. and Mahmood, S.A.F. 2022. Alleviation of cisplatin-induced hepatotoxicity and nephrotoxicity by L-carnitine. Iran. J. Basic Med. Sci. 25, 897–903; doi:10.22038/IJBMS.2022.65427.14395 Hussein, H.K., Dyary, H.O. and Ibrahim, S.H. 2024. Effect of ellagic acid on tamoxifen-induced hepatotoxicity in rats. Int. J. Pharmacol. 20, 383–393; doi:10.3923/ijp.2024.383.393 Kang, M., Park, S., Chung, Y., Lim, J.O., Kang, J.S. and Park, J.H. 2022. Hematopoietic effects of Angelica gigas Nakai extract on cyclophosphamide-induced myelosuppression. Plants 11, 3476; doi:10.3390/plants11243476 Lasram, M.M., Lamine, A.J., Dhouib, I.B., Bouzid, K., Annabi, A., Belhadjhmida, N., Ahmed, M.B., El Fazaa, S., Abdelmoula, J. and Gharbi, N. 2014. Antioxidant and anti-inflammatory effects of N-acetylcysteine against malathion-induced liver damages and immunotoxicity in rats. Life. Sci. 107, 50–58; doi:10.1016/j.lfs.2014.04.033 Liu, S., Hou, W., Yao, P., Li, N., Zhang, B., Hao, L., Nüssler, A.K. and Liu, L. 2012. Heme oxygenase-1 mediates the protective role of quercetin against ethanol-induced rat hepatocytes oxidative damage. Toxicol. Vitro. 26, 74–80; doi:10.1016/j.tiv.2011.10.013 Martynov, A., Farber, B. and Katz, A. 2025. Phosphodiesterase inhibition and immunotropic activity of dipyridamole dynamic derivatives. Curr. Issues. Mol. Biol. 47, 214; doi:10.3390/cimb47040214 Mills, K.A., Chess-Williams, R. and McDermott, C. 2019. Novel insights into the mechanism of cyclophosphamide-induced bladder toxicity: chloroacetaldehyde’s contribution to urothelial dysfunction in vitro. Arch. Toxicol. 93, 3291–3303; doi:10.1007/s00204-019-02589-1 Ming, L., Qi, B., Hao, S. and Ji, R. 2021. Camel milk ameliorates inflammatory mechanisms in an alcohol-induced liver injury mouse model. Sci. Rep. 11, 22811; doi:10.1038/s41598-021-02357-1 Mohammed-Rashid, S.S. and Dyary, H.O. 2025. Effect of catechin on hepatotoxicity induced by combined doxorubicin and paclitaxel treatment. J. Toxicol. 2025, 6775839; doi:10.1155/jt/6775839 Mostafa, R.E., Morsi, A.H. and Asaad, G.F. 2022. Piracetam attenuates cyclophosphamide-induced hepatotoxicity in rats: amelioration of necroptosis, pyroptosis and caspase-dependent apoptosis. Life Sci. 303, 120671; doi:10.1016/j.lfs.2022.120671 Nevzorova, Y.A., Boyer-Diaz, Z., Cubero, F.J. and Gracia-Sancho, J. 2020. Animal models for liver disease–a practical approach for translational research. J. Hepatol. 73, 423–440; doi:10.1016/j.jhep.2020.04.011 Ozturk, N.B., Herdan, E., Saner, F.H. and Gurakar, A. 2023. A comprehensive review of the diagnosis and management of acute liver failure. J. Clin. Med. 12, 7451; doi:10.3390/jcm12237451 Pantano, C., Reynaert, N.L., Vliet, A.V.D. and Janssen–Heininger, Y.M.W. 2006. Redox-sensitive kinases of the nuclear factor-κB signaling pathway. Antioxid. Redox. Signal. 8, 1791–1806; doi:10.1089/ars.2006.8.179 Qian, L., Yang, F., Lin, X., Jiang, S., Zhang, Y. and Tang, Y. 2022. Pyrroloquinoline quinone ameliorates liver injury in mice induced by cyclophosphamide. Environ. Sci. Pollut. Res. 29, 30383–30393; doi:10.1007/s11356-021-17990-6 Srirangan, P. and Sabina, E.P. 2025. Protective effects of herbal compounds against cyclophosphamideinduced organ toxicity: a pathway-centered approach. Drug. Chem. Toxicol. 48, 972–1014; doi: 10.108 0/01480545.2025.2455442 Totzeck, M., Schuler, M., Stuschke, M., Heusch, G. and Rassaf, T. 2019. Cardio-oncology-strategies for management of cancer-therapy related cardiovascular disease. Int. J. Cardiol. 280, 163–175; doi:10.1016/j.ijcard.2019.01.038 Turedi, S. 2023. Protective/preventive effects of quercetin against cyclophosphamide-induced hepatic inflammation, apoptosis and fibrosis in rats. Hepatol. Forum 4, 135–141; doi:10.14744/hf.2023.2023.0026 Van Den Boogaard, W.M.C., Komninos, D.S.J. and Vermeij, W.P. 2022. Chemotherapy side-effects: not all DNA damage is equal. Cancers 14, 627; doi:10.3390/cancers14030627 Xu, Y., Wang, H., Zhou, S., Yu, M., Wang, X., Fu, K., Qian, Z., Zhang, H., Qiu, L. and Liu, X. 2013. Risk of second malignant neoplasms after cyclophosphamide-based chemotherapy with or without radiotherapy for non-Hodgkin lymphoma. Leuk. Lymphoma. 54, 1396–1404; doi:10.3109/10428194.2012.743657 Zhang, Z., Pan, T., Liu, C., Shan, X., Xu, Z., Hong, H., Lin, H., Chen, J. and Sun, H. 2021. Cyclophosphamide induced physiological and biochemical changes in mice with an emphasis on sensitivity analysis. Ecotoxicol. Environ. Saf. 211, 111889; doi:10.1016/j.ecoenv.2020.111889 | ||
| How to Cite this Article |
| Pubmed Style Rafiq AS, Dyary HO. Hepatoprotective effect of dipyridamole against cyclophosphamide-induced hepatotoxicity in rats. Open Vet. J.. 2026; 16(2): 1178-1192. doi:10.5455/OVJ.2026.v16.i2.36 Web Style Rafiq AS, Dyary HO. Hepatoprotective effect of dipyridamole against cyclophosphamide-induced hepatotoxicity in rats. https://www.openveterinaryjournal.com/?mno=283521 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.36 AMA (American Medical Association) Style Rafiq AS, Dyary HO. Hepatoprotective effect of dipyridamole against cyclophosphamide-induced hepatotoxicity in rats. Open Vet. J.. 2026; 16(2): 1178-1192. doi:10.5455/OVJ.2026.v16.i2.36 Vancouver/ICMJE Style Rafiq AS, Dyary HO. Hepatoprotective effect of dipyridamole against cyclophosphamide-induced hepatotoxicity in rats. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 1178-1192. doi:10.5455/OVJ.2026.v16.i2.36 Harvard Style Rafiq, A. S. & Dyary, . H. O. (2026) Hepatoprotective effect of dipyridamole against cyclophosphamide-induced hepatotoxicity in rats. Open Vet. J., 16 (2), 1178-1192. doi:10.5455/OVJ.2026.v16.i2.36 Turabian Style Rafiq, Ariana Seddiq, and Hiewa Othman Dyary. 2026. Hepatoprotective effect of dipyridamole against cyclophosphamide-induced hepatotoxicity in rats. Open Veterinary Journal, 16 (2), 1178-1192. doi:10.5455/OVJ.2026.v16.i2.36 Chicago Style Rafiq, Ariana Seddiq, and Hiewa Othman Dyary. "Hepatoprotective effect of dipyridamole against cyclophosphamide-induced hepatotoxicity in rats." Open Veterinary Journal 16 (2026), 1178-1192. doi:10.5455/OVJ.2026.v16.i2.36 MLA (The Modern Language Association) Style Rafiq, Ariana Seddiq, and Hiewa Othman Dyary. "Hepatoprotective effect of dipyridamole against cyclophosphamide-induced hepatotoxicity in rats." Open Veterinary Journal 16.2 (2026), 1178-1192. Print. doi:10.5455/OVJ.2026.v16.i2.36 APA (American Psychological Association) Style Rafiq, A. S. & Dyary, . H. O. (2026) Hepatoprotective effect of dipyridamole against cyclophosphamide-induced hepatotoxicity in rats. Open Veterinary Journal, 16 (2), 1178-1192. doi:10.5455/OVJ.2026.v16.i2.36 |