| Review Article | ||
Open Vet. J.. 2026; 16(3): 1468-1478 Open Veterinary Journal, (2026), Vol. 16(3): 1468-1478 Review Article Anticancer power of Artemisia annua: A preclinical systematic reviewMohamed Abdulsamad1,2*, Esmaeil Belead Musa3 and Efaf Miftah31Genetic Engineering Department, Libyan Biotechnology Research Center, Tripoli, Libya 2Libyan Authority of Scientific Research, Tripoli, Libya 3Department of Biomedical Sciences, El Mergib University, Al Khums, Libya *Corresponding Author: Mohamed Abdulsamad. Genetic Engineering Department, Libyan Biotechnology Research Center, Tripoli, Libya. Email: mohamed_abdulsamad [at] lbtrc.edu.ly Submitted: 10/09/2025 Revised: 22/01/2026 Accepted: 06/02/2026 Published: 31/03/2026 © 2026 Open Veterinary Journal
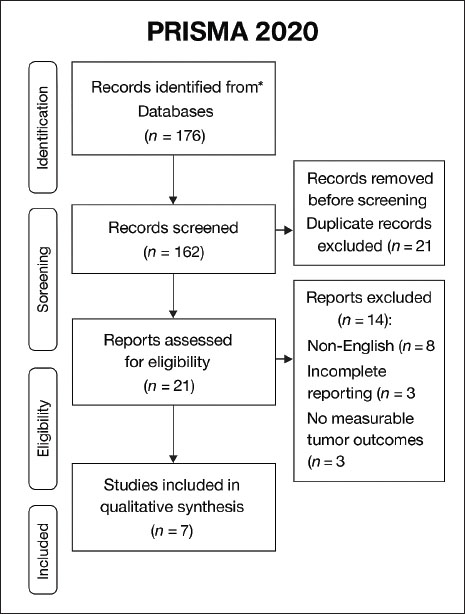
AbstractCancer remains a leading global health challenge, and treatment efficacy is often limited by resistance to conventional therapies and their associated toxicities. These limitations have intensified the exploration of alternative strategies, particularly natural compounds with antitumor potential such as Artemisia annua (AA). This review aimed to systematically evaluate the antitumor efficacy and mechanisms of action of AA extracts and derivatives in preclinical animal models. A structured literature search was conducted in PubMed, Scopus, and Web of Science for studies published between 2019 and August 2025, following PRISMA 2020 guidelines with the protocol prospectively registered in PROSPERO (CRD420251113840). Original in vivo investigations reporting the anticancer activity of AA or its derivatives were considered eligible. Of the 176 records initially identified, 21 underwent full-text review, and seven studies met the inclusion criteria for synthesis. Among the included studies, six of seven eligible studies (86%) reported measurable reductions in tumor size or volume, and four documented apoptosis induction. Additional mechanisms included angiogenesis inhibition and ferroptosis activation. Dihydroartemisinin was the most frequently investigated derivative, while novel compounds such as ZQJ29 also showed promising activity. Importantly, no notable systemic toxicity was reported at the administered doses among the seven included in vivo studies available. This finding reflects only the data available across the seven eligible studies, demonstrates a potentially favorable safety profile. So far, the possibility of dose- or regimen-dependent toxicities reported elsewhere in the literature is not excluded; however, further comprehensive studies are needed to confirm this. This review is the first to consolidate recent preclinical evidence highlighting ferroptosis as one of the key mechanisms of AA derivatives, thereby expanding the understanding of their multifaceted anticancer activity. Collectively, the findings underscore the therapeutic promise of AA derivatives and advocate for further investigation into their pharmacokinetics, safety, and integration into regimens with established therapies. These results demonstrate that AA compounds have significant potential as adjuncts or alternatives in cancer management and warrant rigorous evaluation in standardized clinical trials. Keywords: Angiogenesis, Apoptosis, Artemisia annua, Cancer therapy, Ferroptosis. IntroductionCancer remains a leading global health burden and one of the foremost causes of mortality worldwide, accounting for millions of deaths annually despite substantial advances in diagnostics and therapeutics. Its pathogenesis is multifactorial and shaped by genetic mutations (Moftah Abuhagr et al., 2023). Chronic inflammation, environmental carcinogens, such as tobacco smoke, ionizing radiation, and industrial chemicals, hormonal dysregulation, microbial infections (e.g., Helicobacter pylori and human papillomavirus), lifestyle behaviors, and impaired immune surveillance are all factors that contribute to impaired immune surveillance (Bahrami and Tafrihi, 2023). Although conventional treatment strategies, such as chemotherapy, radiotherapy, surgery, targeted therapies, and immunotherapy, have improved survival for many cancers, they are often compromised by drug resistance, relapse, and serious adverse effects. These limitations highlight the urgent need for safe, effective, and capable of circumventing resistance alternative or adjunctive approaches. Natural products have historically played a pivotal role in drug discovery, serving as a foundation for structurally diverse and biologically active compounds with proven therapeutic relevance in oncology (Efferth, 2017; Kathikeyan and Amaldas, 2020). Within this context, Artemisia annua (AA) L. (sweet wormwood) has emerged as a particularly promising source of novel anticancer agents. The plant is best known as the source of artemisinin, a sesquiterpene lactone that revolutionized malaria treatment and later demonstrated significant anticancer activity (Zhou et al., 2022). Other phytochemicals derived from AA, including Arteannuin B, Casticin, and Chrysosplenol D, have shown cytotoxic and antiproliferative properties in experimental models, particularly against breast cancer and other solid malignancies (Vogel et al., 2023). In vivo studies have increasingly reported that these compounds reduce cancer cell growth, trigger apoptosis, inhibit angiogenesis, and modulate signaling pathways linked to metastasis and immune evasion (Lang, et al., 2020, Ji, et al., 2024, Li, et al., 2024). More recently, a novel mechanism ferroptosis, a regulated form of cell death driven by iron-dependent lipid peroxidation, has been identified as a key pathway through which AA derivatives exert antitumor activity, expanding the mechanistic understanding of their multifaceted effects. Evidence from both animal and human studies indicates that toxicity arises primarily from prolonged systemic exposure rather than short-lived peak concentrations, and the rapid clearance of orally administered artemisinins contributes to their favorable safety profile compared with formulations that delay drug release. This distinction helps clarify why substantial adverse effects have been observed in many animal studies using prolonged exposure, whereas human data generally demonstrate a much lower toxicity risk (Efferth and Kaina 2010; Li, et al., 2016). Together, these findings position AA and its derivatives as candidates with dual advantages: mechanistic versatility and favorable tolerability, both essential attributes for next-generation anticancer therapeutics. Despite encouraging individual reports, the preclinical evidence for AA remains fragmented, with variations in study design, tumor models, and outcome measures hindering clear interpretation. Therefore, a systematic synthesis of in vivo studies is critical for evaluating the consistency across findings, delineating molecular mechanisms, and identifying translational opportunities. Such an effort is scientifically important, as in vivo evidence provides the necessary bridge between in vitro observations and clinical applications, offering insights into pharmacodynamics, safety, and therapeutic feasibility. Accordingly, this systematic review aims to consolidate and critically appraise the available in vivo evidence on the anticancer activity of AA extracts and derivatives. The primary objectives of this study are as follows: (i) to evaluate the antitumor efficacy of the drugs across animal cancer models; (ii) to elucidate mechanistic pathways, including apoptosis, angiogenesis inhibition, and ferroptosis; (iii) to assess reported pharmacological properties and safety profiles; and (iv) to highlight knowledge gaps that may inform translational research and the design of future clinical trials (Lang et al., 2019; Vogel et al., 2023). This review provides a comprehensive preclinical foundation for understanding the therapeutic promise of AA in oncology by addressing these aims. MethodologyThis review was designed and reported in alignment with the PRISMA 2020 guidelines, with the protocol prospectively registered in PROSPERO (CRD420251113840). Eligible studies were restricted to peer-reviewed, English-language publications between January 2019 and August 2025 that investigated the antitumor activity of AA extracts or artemisinin-based derivatives in vivo using preclinical cancer models, reported original experimental data, and provided sufficient methodological quantifiable outcomes such as tumor suppression, apoptosis, angiogenesis inhibition, ferroptosis activation, survival improvement, or mechanistic molecular insights. In vitro studies without animal validation, clinical trials, reviews, meta-analyses, conference abstracts, commentaries, case reports, editorials, non-peer-reviewed work, non-English articles, and studies lacking measurable efficacy endpoints were excluded. A systematic search was conducted in PubMed, Scopus, and Web of Science, supplemented by Google Scholar and manual reference checks, with the last update performed on August 10, 2025. Search strings incorporated both free-text terms and controlled vocabulary (e.g., Artemisia annua, artemisinin, dihydroartemisinin (DHA), cancer, tumor, cytotoxic, preclinical, in vivo, animal model) linked by Boolean operators and limited to the specified publication period; the Supplementary File (1) provides full search strategies. All records were imported into EndNote 20.5 (Clarivate Analytics), duplicates were removed, and titles, abstracts, and full-text articles were independently screened by two reviewers. Discrepancies were resolved through discussion or consultation with a third reviewer. Data extraction was performed using a standardized form, capturing study features (year, country, cancer type, and animal model), intervention details (compound, dose, and administration route), comparators, outcomes (tumor inhibition, apoptosis, angiogenesis, ferroptosis, survival, and toxicity), and mechanistic findings. The SYRCLE risk of bias tool, a validated instrument specifically developed for assessing internal validity in preclinical animal intervention studies, was used to evaluate the methodological quality of the included investigations. This tool was selected because it addresses bias domains, including randomization, allocation concealment, and blinding, which are particularly relevant to in vivo research. Given the heterogeneity across models, interventions, and outcomes, a narrative synthesis was conducted, stratified by cancer type, derivative studied, and mechanistic pathway, while meta-analysis was not feasible due to methodological variability. From the database search, 176 citations were initially identified across all sources, including PubMed (48 records), Scopus (62 records), Web of Science (41 records), and Google Scholar (25 records). This distribution reflects each database’s balanced contribution and supports the comprehensive nature of the search strategy. After removing 21 duplicates, 162 records were subjected to title and abstract screening. Of these, 21 articles were evaluated for eligibility in full text. Fourteen patients were excluded due to language restrictions (n=8), incomplete data reporting (n=3), or lack of quantifiable tumor outcomes (n=3). Seven studies that satisfied the predefined criteria were included in the qualitative synthesis. Figure 1 shows the complete screening and selection process (PRISMA 2020). Fig. 1. PRISMA 2020 flow diagram illustrating the selection process of the studies.
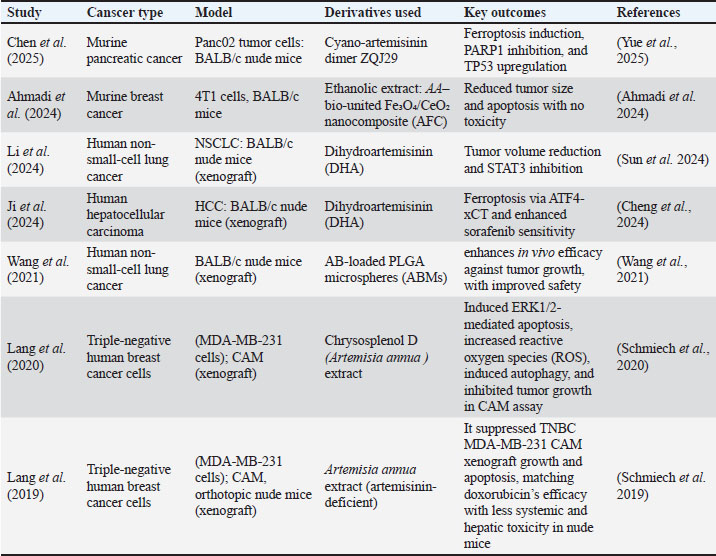
Eligible studies published between 2019 and 2025 investigated a range of malignancies, including breast, lung, liver, and pancreatic cancers, using xenograft and orthotopic murine models. The compounds examined included DHA, ZQJ29. Arteannuin B microspheres, cysosplenol D, casticin, and AFC nanocomposites, each demonstrating measurable antitumor activity, although with variable potency. Table 1 presents a consolidated overview of cancer type, animal model, intervention details, outcomes, and mechanistic findings. Tumor reduction was reported in six studies, apoptosis in four, and ferroptosis in two. Angiogenesis inhibition and autophagy modulation were occasionally described. Table 1. Summary of in vivo preclinical studies on anticancer agents of Artemisia annua and its derivatives.
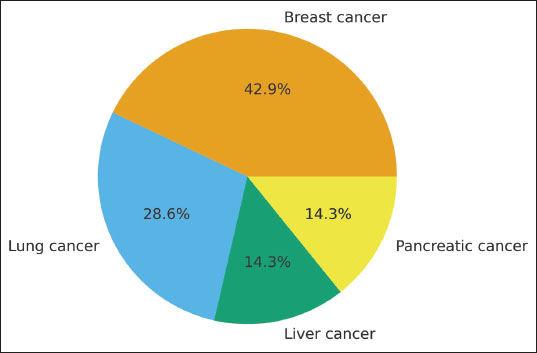
The distribution of cancer models represented in the included preclinical studies is illustrated in Figure 2. Breast cancer accounted for the largest proportion, comprising 43% (3/7) of the studies, followed by lung cancer at 29% (2/7). Hepatocellular carcinoma and pancreatic cancer were less frequently investigated, each contributing 14% (1/7). This distribution highlights a predominant focus on breast and lung malignancies, reflecting their global burden and the priority given to these tumor types in the field of experimental oncology. The limited representation of liver and pancreatic models underscores the need for broader exploration of AA derivatives across diverse cancer settings to better delineate their therapeutic potential. Figure 2. Breast and lung cancers are the most frequently investigated models. Fig. 2. Distribution of cancer types represented across the included preclinical studies.
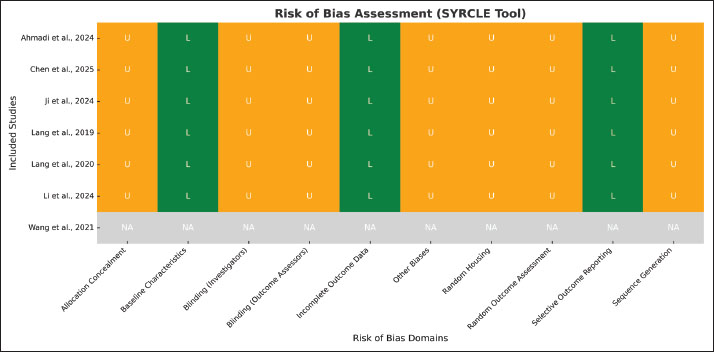
Methodological quality, evaluated with the SYRCLE risk of bias tool, revealed that most studies failed to adequately report domains such as sequence generation, allocation concealment, and blinding of outcome assessment, resulting in frequent “unclear” classifications. Figure 3 summarizes the domain-specific ratings. Notably, some studies, such as Ahmadi et al. (2024), provided greater transparency in allocation and selective reporting, whereas others lacked sufficient methodological detail. Synthesizing across studies, three principal mechanistic themes emerged: induction of ferroptosis, promotion of apoptosis, and inhibition of oncogenic signaling pathways, including STAT3 and angiogenesis-related targets. Additional signals included autophagy modulation and chemosensitization, with DHA showing synergistic potential when combined with sorafenib in hepatocellular carcinoma. Collectively, the evidence indicates that AA derivatives show no valuable systemic toxicity at the administered doses among the seven included in vivo studies, exhibiting consistent preclinical antitumor effects (Schmiech et al., 2019; Wang et al., 2021); however, methodological heterogeneity and small sample sizes limit definitive conclusions, necessitating more rigorous and standardized preclinical investigations before translation to clinical trials. Fig. 3. Risk of bias assessment for the included in vivo studies, evaluated using the SYRCLE Risk of Bias tool.
This systematic review evaluated preclinical evidence on the anticancer activity of AA and its components or derivatives, synthesizing data from diverse cancer models, such as breast, pancreatic, hepatocellular, and non–small cell lung cancers. These compounds exhibited consistent inhibitory effects on tumor growth. Dihydroartemisinin emerged as the most extensively investigated derivative, repeatedly shown to interfere with cancer progression through multiple cellular pathways (Ji et al., 2024; Li et al., 2024). Synthetic analogs, such as the cyano-artemisinin dimer ZQJ29, have expanded the therapeutic scope by exhibiting potent activity against drug-resistant tumor models (Chen et al., 2025). Collectively, these findings emphasize that AA derivatives represent a pharmacologically versatile platform with considerable translational promise and not a single mechanism of action. One of the most striking features of these compounds is their ability to engage alternative modes of programed cell death. Ferroptosis has emerged as a promising and increasingly explored mode of regulated cell death that may contribute to antitumor activity, including in models where apoptotic pathways are impaired. As highlighted in recent literature, ferroptosis is influenced by tumor type, metabolic state, and microenvironmental factors, and its role in therapy resistance is still under investigation (Yang and Stockwell 2016; Li et al., 2024); however, current evidence remains preliminary and highly context-dependent rather than indicative of a dominant or universal mechanism. ZQJ29 has been shown to trigger ferroptotic death in pancreatic cancer by regulating PARP1 and TP53 activity (Chen et al., 2025). Similarly, DHA has been shown to enhance sorafenib responsiveness in hepatocellular carcinoma by modulating the ATF4–xCT axis (Cui et al., 2022; Ji et al., 2024). Ferroptosis emerged as a key mechanistic pathway underlying the anticancer activity of AA derivatives, as consistently reported across multiple in vivo models. In pancreatic cancer, ZQJ29 induced ferroptotic cell death via PARP1 inhibition and TP53 activation (Chen et al., 2025), whereas DHA triggered ferroptosis through ATF4–xCT pathway suppression and enhanced therapeutic responsiveness in hepatocellular carcinoma models (Cui et al., 2022; Ji et al., 2024). The reproducibility of these findings across distinct tumor types and experimental systems supports ferroptosis as a biologically relevant and experimentally validated contributor to the observed antitumor effects. Notably, such mechanistic outcomes are highly model-specific. Although such effects have been demonstrated particularly in hepatocellular carcinoma (HCC) models, they cannot be generalized across all tumor types. Previous studies have shown that responses to artemisinin derivatives vary substantially depending on cancer cell lineage, tumor microenvironment, redox status, and experimental conditions (Wang et al., 2022; Hu et al., 2022). Such activity is therapeutically relevant as it offers a way to circumvent classical resistance mechanisms. Nevertheless, apoptosis remains a consistent hallmark of artemisinin derivatives, with effects mediated by mitochondrial destabilization, caspase cascade activation, and nuclear fragmentation (Du et al., 2013; Lang et al., 2020). These compounds can simultaneously activate both apoptotic and ferroptotic pathways, demonstrating a broad capacity to exploit cancer cell vulnerabilities. In addition to direct cytotoxicity, AA derivatives modulate the tumor microenvironment. DHA suppresses angiogenesis by blocking ATF4-driven vascularization, thereby reducing tumor proliferation (Ji et al., 2024). The inhibition of STAT3 signaling in non-small cell lung cancer models further demonstrates their ability to impair metastatic progression (Li et al., 2024). In addition to their direct cytotoxic effects, AA derivatives modulate the tumor immune microenvironment by suppressing pro-tumorigenic signaling pathways, such as STAT3, and reducing angiogenesis-associated immune evasion. These immunomodulatory effects may indirectly enhance antitumor immunity by altering cytokine signaling, tumor–stroma interactions, and vascular support within the tumor milieu (Ji et al., 2024; Li et al., 2024). Although autophagy was reported only infrequently among the included studies, its mechanistic role warrants attention because the available evidence shows context-dependent effects, functioning either as a cytoprotective response or as a contributor to cell death as it served as a survival pathway, while in others it contributed to tumor cell death (Du et al., 2013; Lang et al., 2020; Zhu et al., 2021). This dual role underscores the complexity of autophagy as a therapeutic target and highlights the need for further mechanistic clarity. Taken together, these observations point to the pleiotropic nature of AA derivatives, which may represent an advantage in addressing cancer’s multifaceted biology. From a therapeutic perspective, the preclinical evidence supports the use of AA derivatives in combination regimens. The ability of DHA to enhance the efficacy of sorafenib in resistant hepatocellular carcinoma exemplifies its potential role in overcoming treatment resistance. Equally, the seven selected studies suggest that these compounds exhibit relatively no notable systemic toxicity compared with conventional chemotherapies, as demonstrated by findings showing the absence of detectable toxicity in murine models (Ahmadi et al., 2024), as in vivo evidence across the included studies frequently suggests a higher selectivity and a better therapeutic window for these compounds compared to standard chemotherapeutics, often demonstrating effective cancer cell cytotoxicity at concentrations minimally toxic to normal cells. and reduced systemic and hepatic toxicity relative to doxorubicin in preclinical comparisons (Schmiech et al., 2019). Although these seven eligible studies did not report notable systemic toxicity, this observation should be interpreted cautiously. Other preclinical investigations have shown that artemisinin derivatives induce neurotoxic lesions in brainstem regions and exhibit embryotoxicity at certain doses (Gordi and Lepist, 2004; Efferth and Kaina 2010). Therefore, the absence of toxicity in our selected studies likely reflects differences in dosing, formulation, treatment duration, and animal model characteristics rather than a universally favorable safety profile, a property that could expand their clinical applicability. The combination of mechanistic diversity, capacity to resensitize resistant tumors, and clear tolerability constitutes a compelling rationale for advancing these agents into early-phase clinical evaluation. Despite this promise, several caveats must be acknowledged. The methodological rigor was frequently limited. The risk of bias assessments using the SYRCLE tool highlighted recurring shortcomings in randomization, allocation concealment, and blinding. Several reports lacked sufficient methodological transparency, leading to “unclear” judgments across multiple domains (Wang et al., 2021). Although a few studies have reported stronger adherence to good practices, including allocation concealment and selective reporting (Ahmadi et al., 2024), the overall evidence base remains vulnerable to potential bias. Such deficiencies limit reproducibility and risk overestimating therapeutic effects. Beyond the methodological issues, the structural limitations of the literature constrain the strength of the conclusions. Heterogeneity in cancer types, experimental designs, and formulations complicates synthesis and limits cross-comparability. The predominance of small animal cohorts raises concerns regarding reproducibility and statistical robustness. Notably, while these methodological limitations weaken the quantitative comparability of the data, they do not invalidate the qualitative apparent finding of consistent antitumor activity observed across all eligible studies, rather than uniformity in study quality or design. Although severe systemic toxicity was not reported in the included studies, the overall scarcity of pharmacokinetic and dedicated long-term toxicity data necessitates caution in defining a definitive therapeutic window. Most importantly, this review excluded human trials, restricting interpretation to preclinical translation. Without clinical validation, the promise of these compounds remains speculative. Therefore, future research must prioritize methodological transparency and standardization. Studies should adopt rigorous randomization and blinding protocols, comprehensively report experimental details, and employ adequately powered animal cohorts. The inclusion of systematic pharmacokinetic and toxicological evaluations is equally critical, as these parameters will determine the feasibility of translating AA derivatives into clinical use. Early-phase clinical trials are urgently needed to establish whether the encouraging preclinical findings can be reproduced in human populations. These agents could significantly enrich the therapeutic landscape of oncology by providing novel mechanisms of action with favorable safety profiles if validated. In summary, AA and its derivatives demonstrated consistent and mechanistically diverse antitumor effects across multiple malignancies in preclinical models. Their ability to activate apoptosis and ferroptosis, suppress angiogenesis, and modulate autophagy illustrates a breadth of activity that is rare among natural product–derived agents. Although methodological shortcomings and the absence of clinical evidence limit definitive conclusions, the overall body of data provides strong justification for advancing these compounds into carefully designed translational studies. The future of AA in oncology will depend not only on continued mechanistic exploration but also on rigorous clinical validation to confirm its true therapeutic potential. ConclusionThis systematic review indicates that AA–derived compounds, particularly DHA and newly engineered dimers, exert consistent anticancer effects through multiple mechanisms, including apoptosis, ferroptosis, angiogenesis inhibition, and oncogenic signaling pathway suppression. Their pleiotropic activity highlights their potential as standalone agents and synergistic partners in combination regimens. Nonetheless, the current evidence base is limited by heterogeneous methodologies, inconsistent dosing strategies, and inadequate pharmacokinetic and toxicity profiling. These shortcomings restrict direct clinical application and underscore the need for more rigorous experimental frameworks. Future investigations should prioritize standardized preclinical models, advanced delivery systems to enhance bioavailability, and rationally design more stable derivatives. The conclusion highlights the need for improved Artemisia research delivery systems, standardized methodologies, and combination strategies. This finding can be strongly supported by grouping studies on nanotechnology and bioengineering. For example, Fekry et al. (2021) compared magnetite nanoparticles synthesized by different methods and evaluated their biological effects, Salem et al. (2025) reported mesenchymal stem cell-loaded 3D-printed scaffolds enhancing bone regeneration, and Aglan et al. (2024) developed nanofibrous scaffolds for cardiomyocyte generation in myocardial infarction. Hence, the ability of AA derivatives to modulate oncogenic signaling aligns with a broader body of evidence demonstrating that natural compounds and nano formulations exert anticancer effects through similar regulatory pathways. Some studies have reported that pharmaceutical bioactives attenuate inflammation by modulating PI3K/AKT signaling (Abd Rabou et al. 2025). In a subsequent study, it was shown that viramidine-loaded nanoparticles suppress hepatocellular carcinoma by targeting the CDC25A/p53/PI3K axis (Abd Rabou et al. 2022). Consistently, nano-encapsulated epigallocatechin-3-gallate induces apoptosis in breast cancer cells (Abd-Rabou et al., 2024), confirming the capacity of phytochemical-based nano-systems to trigger programed cell death. Collectively, these findings support the mechanistic breadth described for AA derivatives, including their roles in apoptosis, ferroptosis, and tumor-associated signaling network modulation. Collectively, these studies emphasize the role of innovative delivery and tissue engineering strategies in enhancing efficacy, reducing toxicity, and promoting the translation of natural compounds, such as AA derivatives. Systematic testing along with established chemotherapy regimens or immunotherapies may also reveal therapeutic synergies. Importantly, the carefully designed early-phase clinical trials are warranted, particularly in refractory cancers where ferroptosis induction offers a promising therapeutic avenue. In summary, AA derivatives represent a scientifically credible bridge between traditional medicine and modern oncology. With careful translational development, they can progress from promising experimental agents to clinically relevant components of next-generation cancer therapy. AcknowledgmentsThe authors gratefully acknowledge the support provided by the Libyan Biotechnology Research Center and the Libyan Authority of Scientific Research, whose facilities and guidance contributed to the successful completion of this work. Conflicts of interestThe authors have no conflicts of interest relevant to this work. FundingNo financial support was obtained. Authors’ contributionsThe concept and overall design of the review were developed by Dr. Mohamed Abdulsamad. Literature searching, screening, and data extraction were performed by Dr. Esmaeil and Dr. Mohamed Abdulsamad. Data analysis and interpretation were also conducted. The initial draft of the manuscript was prepared by Dr. Efaf, and critical revisions and intellectual input were provided by Dr. Esmaeil. All authors have reviewed the content, contributed to improvements, and approved the final version of the manuscript for submission. Data availabilityData are available from the corresponding author upon reasonable request. ReferencesAbd-Rabou, A., Melegy, W., Kishta, M. and El-Ganzuri, M. 2024. Nano-Encapsulation and Apoptotic Impact of Green Tea Polyphenol and Epigallocatechin-3-Gallate on Breast Cancer Cells In vitro. Egypt. J. Chem. 67(3), 139–150. Abd Rabou, A.A., Ahmed, H.H. and Kishta, M.S. 2022. Implication of extrinsic and intrinsic apoptotic pathways in the targeted therapy of hepatocellular carcinoma using aptamer labeled viramidine nanoparticles. BMC Cancer 22, 1106; doi: 10.1186/s12885 022 10201 6 Abd Rabou, A.A., Kamal, M., Alharbi, H.Y., Aljohani, M.S., El Atawy, M.A. and Kishta, M.S., 2025. Modulation of PI3K/AKT signaling and DFT modeling via selected pharmaceutical compounds attenuates carrageenan induced inflammation and oxidative stress in rats. Naunyn Schmiedeberg’s Arch. Pharmacol. 398(6), 7583–7603; doi: 10.1007/s00210-024-03689-1 Aglan, H.A., Ahmed, H.H., Beherei, H.H., Abdel-Hady, B.M., Ekram, B. and Kishta, M.S. 2024. Generation of cardiomyocytes from stem cells cultured on nanofibrous scaffold: experimental approach for attenuation of myocardial infarction. Tissue Cell 89, 102461. Ahmadi, Z., Bayrami, A., Zahri, S., Rahim Pouran, S. and Habibi Yangjeh, A. 2024. Antineoplastic activity of Artemisia annua bio united Fe₃O₄/CeO₂ on 4T1 breast cancer cells: in vivo and in vitro. Cancer Nanotechnol. 15(1), 53; doi: 10.1186/s12645 024 00294 y Bahrami, H. and Tafrihi, M. 2023. Global trends of cancer: the role of diet, lifestyle, and environmental factors. Cancer. Innov. 2(4), 290–301. Chen, J., Yue, L., Pan, Y., Jiang, B., Wan, J., Lin, H., Guo, F., Li H Li. and Zhao. 2025. Novel Cyano-Artemisinin Dimer ZQJ29 Targets PARP1 to induce ferroptosis in pancreatic cancer treatment. Adv. Sci. (Weinh). 12(29), e01935. Cui, Z., Wang, H., Li, S., Qin, T., Shi, H., Ma, J., Li, L., Yu, G., Jiang, T. and Li, C. 2022. Dihydroartemisinin enhances the inhibitory effect of sorafenib on HepG2 cells by inducing ferroptosis and inhibiting energy metabolism. J. Pharmacol. Sci. 148, 73–85; doi:10.1016/j.jps.2013.01.003 Du, X.X., Li, Y.J., Wu, C.L., Zhou, J.H., Han, Y., Sui, H., Wei, X.L., Liu, L., Huang, P., Yuan, H.H., Zhang, T.T., Zhang, W.J., Xie, R., Lang, X.H., Jia, D.X. and Bai, Y.X., 2013. Initiation of apoptosis, cell cycle arrest and autophagy of esophageal cancer cells by dihydroartemisinin. Biomed. and Pharmacother. 67(5), 417–424; doi: 10.1016/j.biopha.2013.01.013 Efferth, T. 2017. From ancient herb to modern drug: Artemisia annua and artemisinin for cancer therapy. Semin. Cancer Biol. 46, 65–83. Efferth, T. and B. Kaina, K. 2010. Toxicity of the antimalarial artemisinin and its derivatives. Crit. Rev. Toxicol. 40(5), 405–421. Fekry, M., Mazrouaa, A.M., Mohamed, M.G., Kishta, M.S. and Mansour, N.A. 2021. The comparison between magnetite nanoparticles co-precipitated by different bases and their effects on human cells. Int. J. Nanosci. 20(03), 2150021. Gordi, T. and Lepist, E.I. 2004. Artemisinin derivatives: toxic for laboratory animals, safe for humans?. Toxicol. Lett. 147(2), 99–107. Hu N Guo., Yang J Yan. and Wang , X. Li. 2022. The potential mechanisms by which Artemisinin and its derivatives induce ferroptosis for treating cancer. Oxid. Med. Cell. Longev. 2022, 1458143; doi:10.1016/j.omcl.2022.04.0843 Ji, J., Cheng J Zhang., Wu X Xu. and Guo , J. Feng. 2024. Dihydroartemisinin induces ferroptosis of hepatocellular carcinoma via inhibiting ATF4-xCT pathway. J. Cell. Mol. Med. 28(8), e18335; doi:10.1016/j.jcm.2018.01.015 Karthikeyan, R. and Amaldas, J. 2020. GC-MS analysis of bioactive compounds of Artemesia annua and assessment of its anti-proliferative activity against human cancer cell lines. J. 11(2), 1840–1843; doi: 10.26452/ijrps.v11i2.2089 Lang, S.J., Schmiech, M., Hafner, S., Paetz, C., Steinborn, C., Huber, R., Gaafary, M.E., Werner, K., Schmidt, C.Q., Syrovets, T. and Simmet, T. 2019. Antitumor activity of an Artemisia annua herbal preparation and identification of active ingredients. Phytomedicine 62, 152962. Lang, S.J., Schmiech, M., Hafner, S., Paetz, C., Werner, K., El Gaafary, M., Schmidt, C.Q., Syrovets, T. and Simmet, T. 2020. Chrysosplenol d, a flavonol from Artemisia annua, induces ERK1/2-mediated apoptosis in triple negative human breast cancer cells. Int. J. Mol. Sci. 21(11), 4090. Li, Y., Sun, H., Bai, C., Hu, Y., Tang, J., Zhang, Y., Chen, J., Zhong, Z., He, Y., Hu, K. and Yang, J., 2024. Dihydroartemisinin inhibits tumor progress via blocking ROR1-induced STAT3-activation in non-small cell lung cancer. Int. Immunopharmacol. 133, 112157; doi: 10.1016/j.intimp.2024.112157 Li, D., Zhang, Z. and Wang, L. 2024. Emerging role of tumor microenvironmental nutrients and metabolic molecules in ferroptosis: mechanisms and clinical implications. Biomed. Pharmacother. 179, 117406; doi: 10.1016/j.biopha.2024.117406 Li, X., Zhou, Y., Liu, Y., Zhang, X., Chen, T., Chen, K., Ba, Q., Li, J., Liu, H. and Wang, H. 2016. Preclinical Efficacy and Safety Assessment of Artemisinin-Chemotherapeutic Agent Conjugates for Ovarian Cancer. EBioMedicine 14, 44–54. Li, Y., Sun, H., Bai, C., Hu, Y., Tang, J., Zhang, Y., Chen, J., Zhong, Z., He, Y., Hu, K. and Yang, J. 2015. Dihydroartemisinin inhibits tumor progress via blocking ROR1-induced STAT3-activation in non-small-cell lung cancer. Int. Immunopharmacology 133, 112157. Moftah Abuhagr, A., Belead Musa, E., Ali Attir, O. and Alati, R. 2023. Targeting P53 gene in breast cancer. J. Adv. Zool. 44(S-7), 295–309. Salem, N.A., Elshebiney, S.A., Mabrouk, M., Kishta, M.S., Galal, A.F., Osama, L. and Beherei, H.H. 2025. Enhanced bone regeneration using mesenchymal stem cell-loaded 3D-printed alginate-calcium titanate scaffolds: a calvarial defect model study. Int. J. Biol. Macromolecules. 302, 140516. Vogel, D., Loots, E., Oladimeji, O., Gouws, C. and van der Kooy, F., 2023. The anti-neoplastic activity of Artemisia afra in breast cancer cell lines. South Afr. J. Botany 157, 115–121; doi: 10.1016/j.sajb.2023.03.049 Wang, Y., Huang, W., Wang, N., Ouyang, D., Xiao, L., Zhang, S., Ou, X., He, T., Yu, R. and Song, L., 2021. Development of arteannuin B sustained-release microspheres for anti-tumor therapy by integrated experimental and molecular modeling approaches. Pharmaceutics 13(8), 1236; doi: 10.3390/pharmaceutics13081236 Wang, M., Chen, H., He, X., Zhao, X., Zhang, H., Wang, Y. and Wang, H., 2022. Artemisinin inhibits the development of esophageal cancer by targeting HIF-1α to reduce glycolysis levels. J. Gastrointestinal Oncol. 13(5), 2144–2153; doi: 10.21037/jgo-22-877 Yang, W.S. and Stockwell, B.R., 2016. Ferroptosis: death by lipid peroxidation. Trends Cell Biol. 26(3), 165–176; doi: 10.1016/j.tcb.2015.10.014 Zhou, X., Suo, F., Haslinger, K. and Quax, W.J., 2022. Artemisinin-type drugs in tumor cell death: mechanisms, combination treatment with biologics and nanoparticle delivery. Pharmaceutics 14(2), 395; doi: 10.3390/pharmaceutics14020395 Zhu, S., Yu, Q., Huo, C., Li, Y., He, L., Ran, B., Chen, J., Li, Y. and Liu, W., 2020. Ferroptosis: a novel mechanism of artemisinin and its derivatives in cancer therapy. Curr. Med. Chem. 28(2), 329–345; doi: 10.2174/0929867327666200121124404 Supplementary File 1. Full electronic search strategies.Systematic Review Title:
| ||
| How to Cite this Article |
| Pubmed Style Abdulsamad M, Musa EB, Miftah E. Anticancer power of Artemisia annua: A preclinical systematic review. Open Vet. J.. 2026; 16(3): 1468-1478. doi:10.5455/OVJ.2026.v16.i3.6 Web Style Abdulsamad M, Musa EB, Miftah E. Anticancer power of Artemisia annua: A preclinical systematic review. https://www.openveterinaryjournal.com/?mno=283055 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.6 AMA (American Medical Association) Style Abdulsamad M, Musa EB, Miftah E. Anticancer power of Artemisia annua: A preclinical systematic review. Open Vet. J.. 2026; 16(3): 1468-1478. doi:10.5455/OVJ.2026.v16.i3.6 Vancouver/ICMJE Style Abdulsamad M, Musa EB, Miftah E. Anticancer power of Artemisia annua: A preclinical systematic review. Open Vet. J.. (2026), [cited March 31, 2026]; 16(3): 1468-1478. doi:10.5455/OVJ.2026.v16.i3.6 Harvard Style Abdulsamad, M., Musa, . E. B. & Miftah, . E. (2026) Anticancer power of Artemisia annua: A preclinical systematic review. Open Vet. J., 16 (3), 1468-1478. doi:10.5455/OVJ.2026.v16.i3.6 Turabian Style Abdulsamad, Mohamed, Esmaeil Belead Musa, and Efaf Miftah. 2026. Anticancer power of Artemisia annua: A preclinical systematic review. Open Veterinary Journal, 16 (3), 1468-1478. doi:10.5455/OVJ.2026.v16.i3.6 Chicago Style Abdulsamad, Mohamed, Esmaeil Belead Musa, and Efaf Miftah. "Anticancer power of Artemisia annua: A preclinical systematic review." Open Veterinary Journal 16 (2026), 1468-1478. doi:10.5455/OVJ.2026.v16.i3.6 MLA (The Modern Language Association) Style Abdulsamad, Mohamed, Esmaeil Belead Musa, and Efaf Miftah. "Anticancer power of Artemisia annua: A preclinical systematic review." Open Veterinary Journal 16.3 (2026), 1468-1478. Print. doi:10.5455/OVJ.2026.v16.i3.6 APA (American Psychological Association) Style Abdulsamad, M., Musa, . E. B. & Miftah, . E. (2026) Anticancer power of Artemisia annua: A preclinical systematic review. Open Veterinary Journal, 16 (3), 1468-1478. doi:10.5455/OVJ.2026.v16.i3.6 |