| Research Article | ||
Open Vet. J.. 2025; 15(12): 6709-6720 Open Veterinary Journal, (2025), Vol. 15(12): -6720 Research Article Efficacy of tilmicosin and suxamethonium for dog euthanasia: A comparative studyIbraheem Ahmed Zedan1, Mae Thanoon Al-anaaz1, Hiba A. Shekho2, Osamah M. Al Iraqi1, Basima A. Albadrani1 and Layth Mahmoud Alkattan1*1College of Veterinary Medicine, University of Mosul, Mosul, Iraq 2College of Veterinary Medicine, University of Tkirt, Tkirt, Iraq *Corresponding Author: Layth Mahmoud Alkattan. College of Veterinary Medicine, University of Mosul, Mosul, Iraq. Email: laythalkattan [at] uomosul.edu.iq Submitted: 09/09/2025 Revised: 27/10/2025 Accepted: 02/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
AbstractBackground: Euthanasia is a difficult ethical choice with many interrelated components, and a thorough treatment of the topic is outside this document’s purview. Aim: This study aimed to test the possibility of using either tilmicosin or suxamethonium to evaluate the time required for experimentally induced smooth, comfortable euthanasia in dogs. Methods: Six adult dogs of both sexes, their weight (20 ± 5kg) and age (24 ± 6 months) were included in this experiment. The experiment was randomly divided into two equal groups. The general anesthetic regime used in this experiment included a mixture of ketamine and xylazine. The data relied on clinical findings and electrocardiography (ECG) tool to record changes exhibits on experimental dogs to perform comfortable, painless euthanasia Results: The results indicated that the time required to induce euthanasia varied between the two groups. In the Suxamethonium group (SG), it took around 1–12 minutes with clinical findings manifested by the absence of neurological convulsions or muscle contractions and respiratory arrest with cyanosis of the mucous membranes without epilepsy. In the tilmococin group (TG), it took around 1–4 minutes, and the clinical findings were manifested by the presence of severe neurological convulsions for a short period. In the SG group, ECG abnormalities, such as prolonged QT intervals or even arrhythmias, sinus bradycardia, and absence of electrical activity, were observed. In contrast, in the TG group, there were significant concern alterations in ECG, represented by prolonged QT intervals, duration of ventricular depolarization and repolarization, ventricular fibrillation, and sudden death. Conclusion: Both suxamethonium and tilmicosin are viable agents for inducing euthanasia in dogs; however, suxamethonium demonstrated superior efficacy and safety, facilitating euthanasia with minimal complications. Keywords: Tilmicosin, Suxamethonium, Euthanasia, ECG, Canine. IntroductionEuthanasia is a difficult ethical choice with many interrelated components, and a thorough treatment of the topic is outside this document’s purview. According to the World Society for the Protection of Animals, euthanasia is appropriate and required when an animal is in pain due to an incurable illness or injury or when it poses a serious threat to the health and safety of humans or other animals through disease or aggressive behavior (Gates et al., 2023). Veterinary doctors always seek to induce a “good death” to prevent animal discomfort and distress, as well as painless, smooth loss of consciousness prior to death, humane killing to prevent further deterioration of life (Leary et al., 2020). Euthanasia is performed with physical or pharmacological procedures. Pharmacological procedures involve the use of appreciated injectable drugs or inhalant gases that depress cerebral function and inactivate cardiovascular function, resulting in death due to hypoxia and physical brain disruption (Close et al., 1996; Yoshida 2023; Maharani et al., 2024; Verhofstadt et al., 2024). Many chemical agents are commonly used to perform euthanasia (Edney, 1998; Gates et al., 2023). Many agents, such as sedatives, such as xylazine and medetomidine, and preanesthetics, such as acepromazine, are used to prevent fear during euthanasia and reduce severe complications (Pepper et al., 2023). Many drugs and chemical agents are used to perform euthanasia in dogs, but their availability and prices, as well as the extent of harm and the time it takes for euthanasia to occur, are of great importance for ethical considerations (Pepper et al., 2023). Suxamethonium is a muscle relaxant. Profound neuromuscular block and smooth, rapid recovery. It is an anesthetic agent used to cause short-term paralysis (Kogan and Cooney, 2023). This drug was successfully used as a euthanasia agent in dogs (Fawcett, 2019). Studies and clinical reports on the cardiotoxic effects of tilmicosin indicate that it can lead to ventricular arrhythmias in dogs (O’Brien et al., 2018). Increased vagal tone from tilmicosin toxicity may lead to bradycardia (McCarthy, 2019). Increased severe vagal tone can result in sinus arrest. Higher doses and prolonged use are associated with an increased risk of cardiotoxicity (McCarthy, 2019). Interaction with other cardiac-affecting medications can increase the risk of toxicity. Animals with pre-existing heart issues are more susceptible to toxicity (Keating and Johnson, 2020). Symptoms may include lethargy, weakness, syncope, or heart failure (Weller 2017). The drug tilmicosin is frequently used in veterinary medicine. This agent is considered a macrocyclic antibiotic that is frequently used in veterinary clinics, but its high bitterness reduces its use (Yan et al., 2020; et al.,2022). Unintentional injection overdoses may result in mortality by producing cardiotoxicity (Yildiz et al., 2023). Additionally, there is no known antidote for deaths caused by tilmicosin (Er et al., 2014). This study established the perfect suitable humanitarian ways to euthanize dogs to assist researchers who need to collect tissue biopsies and kill experimental animals for research purposes. In this study, the efficacy of both tilmicosin and suxsamethanioum was studied for the painless and confirmable euthanasia of dogs. Materials and MethodsAnimalsSix experimental adult dogs of both sexes, whose ages ranged (24 ± 6) months and their weights ranged (20 ± 5) kg were used in this study. All animals received a regime of general anesthesia with a mixture of 10% ketamine (Rotexmedica, Germany) at a dose of 10 mg/kg IM and 2% xylazine (Interchemie, Holland) at a dose of 3 mg/kg IM (Alkattan and Helal 2013; Mahmood, 2022; Tunahan, 2023). The animals were then divided into 2 equal groups, 3 of each. In the first group (Suxsamethanioum chloride 10% (VUAB Pharma. Czech Republic) used at the dose 100 mg/animal IV, followed by administration of NaCl solution 20% (20 ml/animal). In the second group (Tilmicosin 30%) used at the dose used was 5 ml IV per animal (Interchem, Holland). Examination and assessment of euthanasia patientsThe experimental animals undergoing euthanasia were evaluated based on clinical findings that were exhibited by the euthanized animals during the experiment. These findings were recorded directly following drug administration until all vital signs completely subsided and the animal died. These findings included heartbeat, convulsion, muscle twitching, seizures, coma, unconsciousness, dilated pupil, and finally death. Electrocardiography (ECG) assessmentTo evaluate the clinical findings exhibited by experimental animals undergoing euthanasia. The assessment will include monitoring various vital signs and clinical manifestations, such as Heartbeat, Electrocardiography monitoring, Convulsions, Muscle twitching, seizures, coma, unconsciousness, dilated pupils, and final determination of death. In this study, an electrocardiogram (EDAN Veterinary Electrocardiograph model: VE-100 Germany) was used to record and evaluate the electrocardiogram of the heart from the moment of injection of the drugs used in euthanasia until the heart stopped and the animal died Kistner and Mccarthy (2018). The procedure involved the following stepsLead placementThe electrodes were attached to specific locations on the dog’s body. For standard ECG monitoring in canine patients, the leads were placed as follows (Bistner and McCarthy, 2018): o Right arm: Positioned on the right forelimb; o Left arm: Positioned on the left forelimb; o Left leg: Positioned on the left hind limb. Right leg: Positioned on the right hind limb as a ground reference. Monitoring procedureThe ECG was initiated immediately following the euthanasia drug injection. Continuous monitoring allowed for real-time assessment of the electrical activity of the heart. The recordings captured various metrics, including heart rate and rhythm, enabling detailed analysis. Data analysisThe ECG strips were analyzed according to Kienle and Kienle (2019; Levy and Gunter-Hunt, 2020) to detect any arrhythmias or alterations in heart patterns during the euthanasia process. The analysis focused on significant changes in the waveform and intervals, indicating progression toward cardiac arrest (Fig. 1).
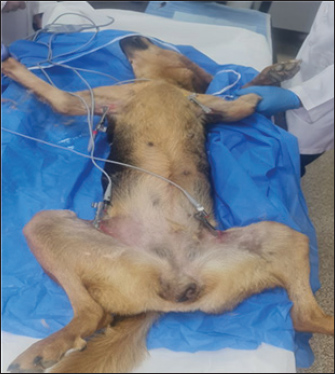
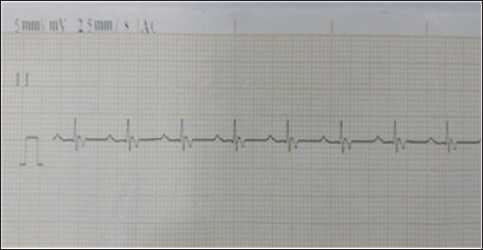
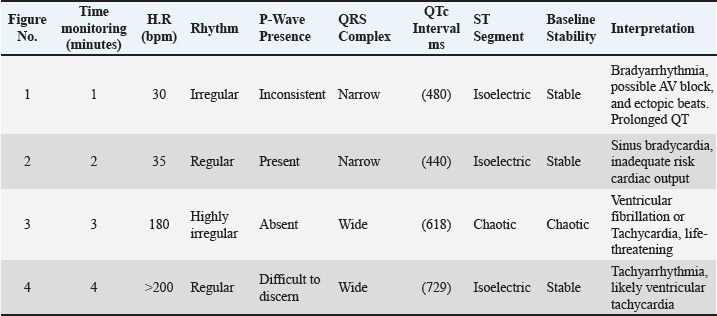
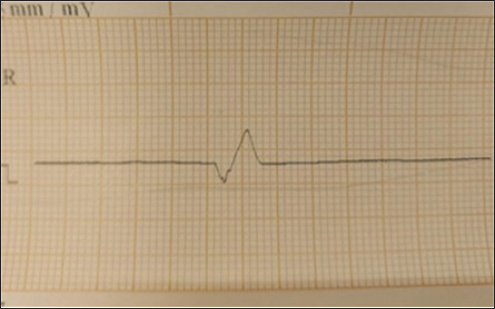
Fig. 1. Animal preparation for ECG. Heart rate calculation from ECG/Group 11. Measurement of the R-R Interval: The R-R interval was measured directly from the ECG tracing, identifying the distance between consecutive R waves (Harding and Smith, 2015). 2. Heart Rate Calculation: The heart rate (HR) was calculated using the following formula (Mccarthy, 2019): HR (bpm)=1,500/ R-R interval (mm). QT Interval Calculation from ECG 1. Determination of paper speed: The paper speed of the ECG was confirmed to be 25 mm/second, as indicated on the tracing. 2. Measurement of the QT Interval: The QT interval was measured from the Q wave onset to the T wave termination. The measurement involved visually estimating the number of small squares (1 mm each) encompassed within this interval on the electrocardiogram tracing. 3. Conversion to time: Each small square at a paper speed of 25 mm/s corresponds to 0.04 seconds (40 ms). The total QT interval in seconds was calculated by multiplying the number of small squares by 0.04. 4. QT Correction (QTc): To correct the QT interval for heart rate, Bazett’s formula was employed (Häggström and Guglielmini, 2021): QTc (ms)=QT (ms)/ √R-R interval (s). Statistical analysisTo investigate the effects of tilmicosin or suxsamethanium for euthanasia of dogs on various electrocardiogram parameters by comparing pre-administration values with post-administration metrics in two distinct groups. The heart rate (bpm) was evaluated using analysis of variance (ANOVA) to determine differences across the three measurement points (pre-administration, Group 1, and Group 2). Ethical approvalThis article was approved by the Animal Care and Committee of the University of Mosul, College of Veterinary Medicine (No: UM. Vet.2023) (1582023) No.2023.137. ResultsClinical examinationAnimals in the first group Suxamethonium group (SG), showed clear findings represented by reduced respiratory rate and even respiratory arrest with cyanosis of the mucous membranes. Within 2–3 minutes, there was an increase and acceleration in heart rate. However, during the administration of NaCl 20% solution, the heart rate decreased and there was a complete diminish of pulse. These signs continue for 3–4 minutes after administration of suxamethonium drug. No signs of seizure, convulsions, or nervous convulsions muscle twitching were monitored on animals, and no animal returned to life in this group. Death occurred smoothly without pain and distress. In the second group (TG), 5 ml tilmococin was administered intravenously, and the animals showed clinical findings represented by pulse increase, followed by an irregular heartbeat, apnea, and complete cessation of breathing and pulse, accompanied by rapid bouts of convulsions and epileptic seizures. The vital activity diminished, and animals died for 3–5 minutes, and no animals returned to life. Monitoring of electrocardiographic changesECG changes are recorded in dogs included in this study before euthanasia drug administration. (Fig. 2) showed ECG tracing of a dog under Xylazine-Ketamine sedation pre injection of euthanasia drugs. The dog’s heart rhythm was steady and normal despite being under sedation. The slight delay in the signal traveling from the atria to the ventricles (PR interval) was expected because of the sedative Xylazine, which slows the heart naturally. The addition of ketamine helped maintain a stable heart rate. There are no abnormalities in the electrical activity of the heart.
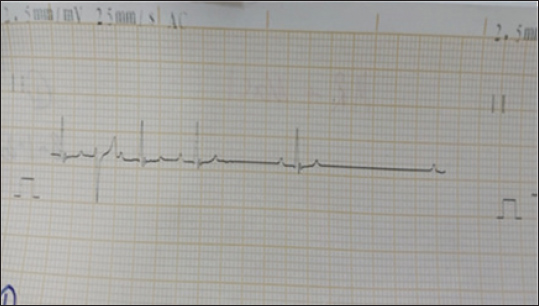
Fig. 2. Electrocardiogram changes before euthanasia under ketamine xylazine anesthesia. In the first group SG, the administration of suxamethonium led to abnormal changes on the ECG record, with prolonged QT intervals (Fig. 3). SG-1) or even arrhythmias. Sinus bradycardia, which is manifested by a decreased heartbeat, or asystole (absence of electrical activity) in severe cases (Fig. 3). SG-2). Several figures indicate bradycardia (Fig. 3 SG-1, 2, and 8). Irregular rhythms noted in the interpreted cases show varying degrees of urgency, particularly in Fig. 3 SG-3 and 8. The key measurements and nterpretations from each ECG tracing of dogs administered suxamethonium are illustrated in Table 1.
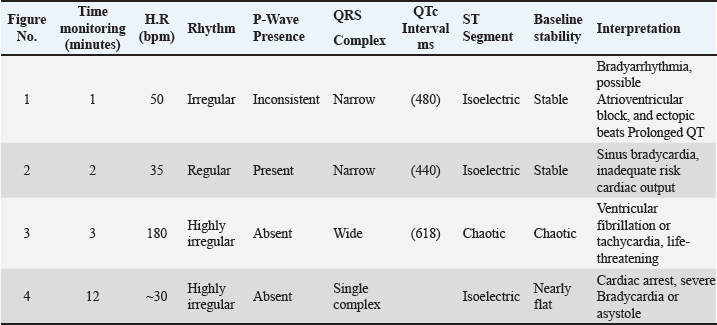
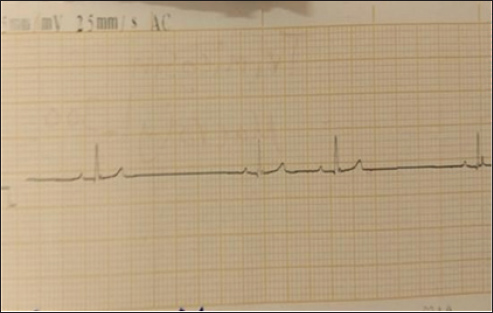
Fig. 3. (SG-1) ECG tracing of the dog after 2 minutes of treatment with suxamethonium. indicates bradyarrhythmia. Heart rate: 50 beats per minute (bpm) with QT prolongation (QTc of 440 ms), with ectopic beats. The QRS complexes are narrow in most instances. Table 1. Key measurements and interpretations from each ECG tracing of dogs after Suxamethonium administration.
ECG changes associated with Tilmicosin phosphate in dogs group 2 (TG)Several ECG alterations have been documented in dogs receiving tilmicosin phosphate, with prolonged QT intervals being the most prevalent. The QT interval represents the duration of ventricular depolarization and repolarization. Prolongation of the QT interval increases the risk of ventricular tachyarrhythmia, including ventricular fibrillation, and sudden cardiac death (Fig. 3). TG- 1). This ECG alteration is of significant concern and warrants close monitoring of dogs undergoing TP treatment. Tilmicosin phosphate is associated with ST-segment depression, T-wave inversion, and reduced R-wave amplitude. ST-segment depression often occurs in conjunction with QT interval prolongation and signifies MI. T-wave inversion reflects abnormalities in ventricular repolarization, whereas reduced R-wave amplitude indicates impaired ventricular depolarization (Fig. 3 TG-2). Also, in the tilmocosine group, the dog’s ECG showed absence of P-waves with myocardial infarction (MI), and Q-waves were wider (0.04) and deeper than normal (Fig. 3 TG-3). Features of sinus tachycardia that may indicate myocardial ischemia, including deep S waves and T-wave inversion. (Fig. 3 TG-4). The ECG changes associated with tilmicosin phosphate in dogs are summarized in Table 2. Table 2. ECG changes associated with tilmicosin phosphate levels in dogs.
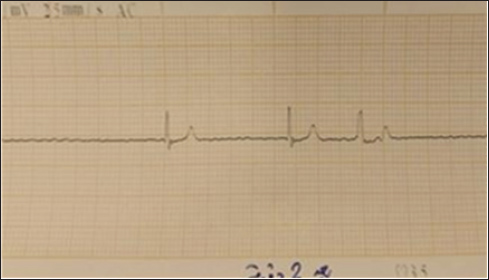
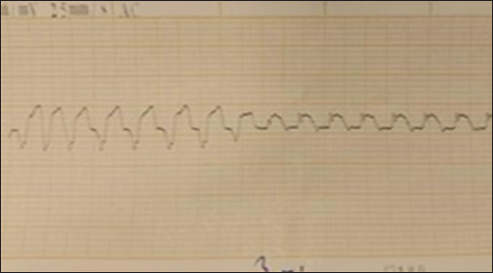
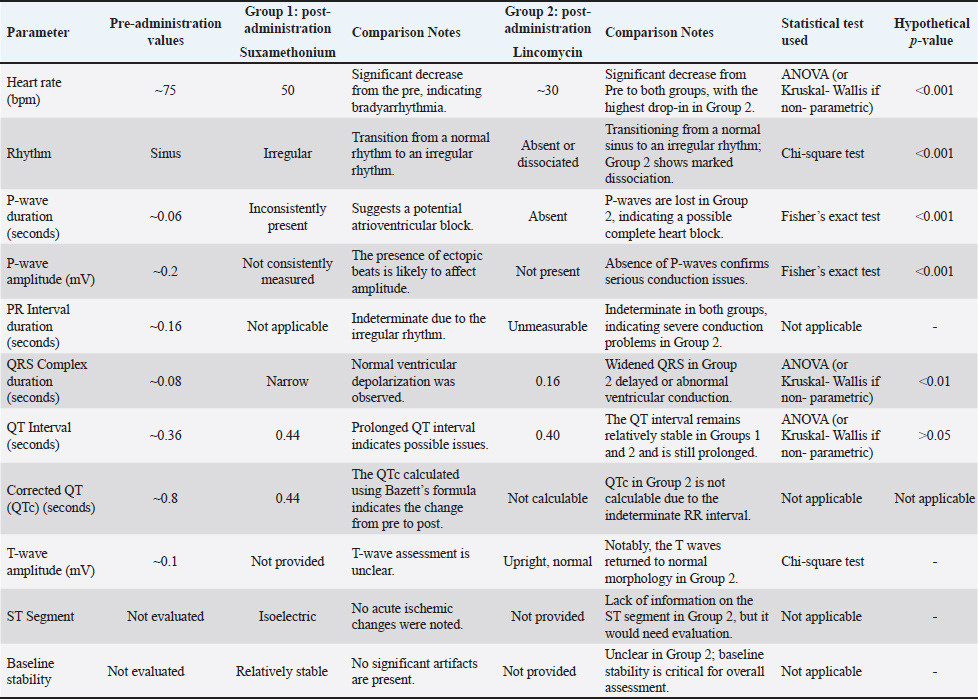
The comparison of cardiac effects between Suxsamethonium (Group 1) and Tilmicosin (Group 2) in dogs revealed significant differences in heart rate, rhythm, and conduction disturbances (Figs. 4–6). The mean heart rate was 75 bpm before treatment; however, it dropped to 50 bpm after Suxsamethonium administration, indicating severe bradycardia. In contrast, tilmicosin resulted in a mean heart rate of 30 bpm, which, while low, remained close to the normal range before treatment. ANOVA demonstrated a significant reduction in heart rate across groups (p < 0.01), with the greatest decrease in Group 2. Pre-treatment rhythms were normal sinus rhythms, but suxamethonium administration led to irregular rhythms and notable bradyarrhythmia, while some dogs receiving tilmicosin exhibited absent or dissociated rhythms. Chi-square tests confirmed significant deterioration in rhythm (p < 0.05), highlighting the critical nature of consistent P waves. Pre-treatment P waves were consistently present, but in Group 1, they became inconsistently present, indicating conduction issues, and were absent in Group 2, suggesting significant conduction block (p < 0.01). The QRS complex, which was normal pretreatment, narrowed into Group 1 and widened in Group 2, with one-way ANOVA revealing significant changes across groups (p < 0.01). The QT interval showed relative stability in both groups post-administration, with T-tests indicating no significant changes but clinically relevant values (p > 0.05). T wave data were unavailable for Group 1; however, T waves were upright in Group 2, suggesting recovery potential. Overall, the findings underline significant cardiac dysfunction following drug administration, with Suxsamethanioum showing severe bradyarrhythmia and conduction disturbances compared to Tilmicosin, warranting careful ECG monitoring in these scenarios. Table 3 summarizes the comparison of ECG parameters between the two groups (Figs. 7–10).
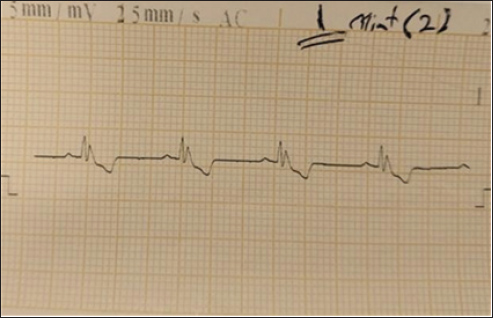
Fig. 4. (SG-2) This tracing of dog post 2 minutes Suxsamethanioum injection shows sinus bradycardia (heart rate of 35 bpa), which is an irregular but abnormally slow heart rhythm.
Fig. 5. (SG-3) This tracing appears to show a chaotic, wide, and irregular pattern with no discernible P-waves, as the ventricular arrhythmia overwhelms the atrial activity and then abruptly terminates into asystole.
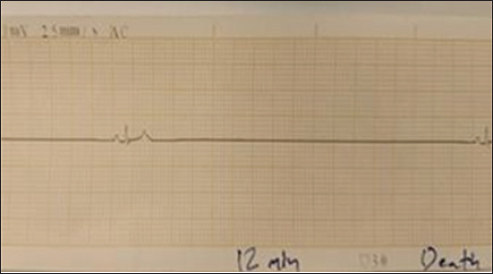
Fig. 6. (SG-4) This ECG tracing of dog shows significant findings consistent with cardiac arrest HR of 30 bpm 12 minutes after suxamethonium administration.
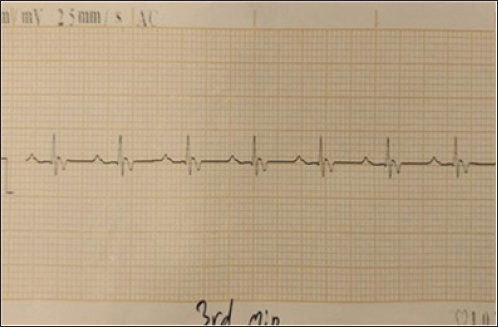
Fig. 7. (TG-1) ECG of a dog with a heart rate of approximately 38 bam. The QT interval was approximately 320 ms, and the corrected QT (QTc) was 254 ms. These findings, combined with bradycardia and irregularity, suggest that cardiac conduction disturbances were likely intensified by the cardiotoxic effects of tilmicosin.
Fig. 8. TG-2 This ECG tracing appears to demonstrate VT based on the rapid rhythm and morphology. The R wave in the lateral leads may be either “M-shaped”, notched, monophasic, or RS complex.
Fig. 9. (TG-3) ECG of the dog showed absence of P waves with myocardial infarction (MI). The Q-waves were wider (0.04) and deeper than normal.
Fig. 10. (TG-4) This tracing of the dog shows sinus tachycardia with features, including deep S waves and T-wave inversion, that may myocardial ischemia. Table 3. Comparison of ECG parameters between suxamethonium and tilcomycin for euthanasia in dogs with statistical tests of hypothetical p-values.
DiscussionThis comprehensive ECG monitoring from the moment of drug administration until the cessation of heart activity provided valuable insights into the physiological effects of euthanasia on canine patients. The clinical signs accompanying euthanasia in group SG animals are consistent with the principles and ethics of euthanasia (Maharani et al., 2024; Verhofstadt et al., 2024). In terms of the smoothness of the events and the absence of a return to life, with no convulsions or seizures occurring, the animals of the second group TG2, suffered from these symptoms accompanying euthanasia, which gives an advantage to the first group. Euthanasia procedures typically result in death within 2–4 minutes, allowing for a quick and painless transition for the dog. Using suxsamethanioum along with sodium chloride (NaCl), along with xylazine and ketamine premedication, provides a comprehensive approach to this sensitive procedure. Xylazine is an alpha-2 adrenergic agonist that produces sedation and analgesia, while ketamine is a dissociative anesthetic that provides pain relief and anesthesia (O’Brien, 2018). Together, they create a balanced premedication protocol that enhances the euthanasia process. Administering these drugs before euthanasia minimizes distress for the animal, ensuring that they are relaxed and unaware of the euthanasia agent (Mahmood, 2022). After adequate sedation and analgesia have been achieved through the premedication, suxamethonium, combined with sodium chloride, can be administered. Suxsamethanioum induces rapid paralysis, which is crucial for achieving a swift and humane euthanasia. The addition of sodium chloride serves as a vehicle for intravenous administration, facilitating effective delivery (McCarthy, 2019). Although suxamethonium effectively causes paralysis, it does not provide pain relief. Therefore, the pre-medication phase with xylazine and ketamine is essential to ensure that the euthanasia process is completely humane and devoid of suffering Monahan (2023). Moreover, dogs receiving suxamethonium may experience alterations in potassium levels because the drug can release potassium from the muscles Kollias et al. (2023). Changes in potassium levels can lead to abnormalities in the ECG, such as prolonged QT intervals or even arrhythmia. QT prolongation predisposes the heart to arrhythmias, including torsade de pointes and ventricular fibrillation, especially if accompanied by electrolyte imbalances (e.g., hyperkalemia, hypocalcemia) (Kienle and Kienle, 2019). The addition of sodium chloride to the equation can intensify the cardiac toxicity of suxamethonium. This element is an essential electrolyte that is responsible for various physiological activities, including the maintenance of proper heart function. Sodium imbalances caused by excessive sodium chloride administration can disrupt the electrical impulses of the heart, leading to various cardiac dysfunctions (Keatingn et al., 2020). The current study reinforces and expands on previous research regarding the cardiotoxic effects of TC in dogs. Consistent with the findings of O’Brien et al. (2018), which identified ventricular arrhythmias as a significant risk, this study further emphasizes the importance of monitoring cardiac function in canine patients receiving this medication. Additionally, the current findings align with those of McCarthy (2019). Tilcomycin toxicity symptoms, such as lethargy, weakness, syncope, and signs of heart failure, were also observed in this study, reinforcing the recognition of these clinical signs as indicators of serious adverse effects that warrant prompt attention. The rapid onset of death during euthanasia, typically within 2–4 minutes, highlights the importance of selecting appropriate euthanasia agents that prioritize the welfare of the animal and the owner’s emotional well-being. The cardiotoxic risks associated with TC, as indicated by prior research, reinforce the need for a cautious and informed approach to euthanasia protocols. Tilmicosin induces myocardial necrosis (Weller 2017). Tilmicosin can cause ventricular arrhythmias in dogs. Such risks raise concerns about using this drug in euthanasia, where rapid and humane death is critical. Research by McCarthy (2019). Cardiotoxicity risk may increase with higher doses and prolonged exposure (McCarthy, 2019). This underscores the importance of precise dosing during euthanasia—a crucial factor for ensuring that the procedure achieves its goal swiftly and with minimal suffering. Veterinarians must critically assess any concurrent treatments to mitigate the risks. Dogs with pre-existing heart issues are particularly susceptible to toxicity (Mazón-Ruiz et al., 2024). Alternative agents, such as pentobarbital, which are well-documented for their swift and humane effects, should be prioritized to ensure that dogs pass quietly and without fear or pain. The decision to euthanize a dog is a profoundly sensitive one, aimed at relieving suffering and ensuring a compassionate end to life. Given the critical importance of a swift and humane process, the use of tilmicosin for euthanasia is supported due to its rapid onset of death, which typically occurs within 2–4 minutes. This quick action allows for an immediate and painless transition for dogs with terminal illness, which is a significant consideration in veterinary practice. By prioritizing the welfare of the animal and ensuring a peaceful and swift passing, tilmicosin can serve as an appropriate option for veterinarians seeking to ease suffering in their patients during their final moments. In this study, we aimed to investigate the effects of tilmicosin or suxamethonium for euthanasia of dogs on various ECG parameters by comparing pre-administration values with post-administration metrics in two distinct groups. Finally, the ST segment and baseline stability were assessed qualitatively because the data were not sufficiently evaluable for quantitative analysis. Comparison between the Suxamethonium and Tilmicosin as euthanasia in dogs according to the following criteria: Bradycardia, Rhythm Stability, Conduction Issues, Ventricular Activation, Atrial Activity, and QT Interval. Tilcomycin causes severe bradycardia, whereas Suxsamethanioum produces moderate bradycardia, indicating that the effect of tilcomycin on heart rate is more profound and potentially dangerous. Both drugs result in prolonged RR intervals, with Sux demonstrating a potentially more stable rhythm than Tilcomycin. Both drugs cause severe conduction issues, as indicated by the inability to measure the PR interval. This indicates a complete AV block, which is critical in both cases. The QRS duration is prolonged in tilmicosin, indicating significant ventricular compromise, whereas Suxamethonium initially maintains a narrower QRS duration, less disturbance in ventricular conduction. Suxamethonium may preserve P waves longer than tilmicosin, initially reflecting better atrial function, although both drugs can lead to dissociation. The QT interval remained normal for both drugs based on the bradycardic state; however, continuous monitoring is warranted to ensure that QT prolongation does not occur later in Suxamethonium (Mazón-Ruiz et al., 2024). The clinical signs accompanying euthanasia in group 1 animals are consistent with the principles and ethics of euthanasia (Al-Qadhi et al., 2022; Maharani et al., 2024) in terms of the smoothness of the events and the absence of a return to life, with no convulsions or seizures occurring, while the animals of the second group suffered from these symptoms accompanying euthanasia, which gives an advantage to the first group. The prolongation of QT and depression of ST-segment and development of myocardial ischemia after tilmocosine administration. These changes collectively indicate the cardiac toxicity potential of tilmicosin phosphate and demand cautious management to minimize adverse effects (Yildiz et al., 2023). ConclusionThe combination of xylazine and ketamine for premedication, followed by the administration of suxsamethanioum with sodium chloride, offers a comprehensive and humane approach to euthanasia in dogs. This method prioritizes the safety and welfare of the animals by ensuring that they are calm, unaware, and free from pain throughout the euthanasia technique. The comparative analysis indicates that tilmicosin may lead to more severe cardiac effects, characterized by profound bradycardia, prolonged QRS duration, and significant atrial dissociation, potentially posing higher risks during euthanasia procedures. In contrast, suxamethonium seems to initially maintain better heart rhythm stability and conduction, suggesting a comparatively lesser impact on cardiac function. AcknowledgmentWe are grateful to our College of Veterinary Medicine, the University of Mosul, Mosul, Iraq, for supporting this research. Conflict of interestThe authors declare no conflict of interest. Funding: The authors funded this study. FundingThe research was self-funded by the researchers. Authors’ contributionsAll authors contributed in this manuscript Data availabilityAll data are available in the revised research. ReferencesAlkattan, L.M. and Helal. 2013. Effects of ketamine-xylazine and propofol-halothane anesthetic protocols on blood gases and some anesthetic parameters in dogs. World 6(2), 95–99; doi:10.5455/vetworld.2013.95-99 Al-Qadhi, A.S., Zedan, I.A., Al-Ajeli, R.R., Al-Badrany, M.S. and Ali, A.K. 2022. The effect of therankeron D6 on the healing of intestinal anastomosis in dogs. Iraq J. Vet. Scie. 36(1), 261–265. Bistner, S.I. and Mccarthy, R.N. 2018.Clinical Application of Electrocardiography in Dogs and Cats. Vet. Clin. N. Am. Small Ani. Pract. 48(1), 1 -20. Close, B., Banister, K., Baumans, V., Bernoth, E.M., Bromage, N., Bunyan, J., Erhardt, W., Flecknell, P., Gregory, N., Hackbarth, H., Morton, D. and Warwick, C. 1996. Recommendations for euthanasia of experimental animals: part 1. Lab. Anim. 30(4), 293–316. Edney, A.T.B. 1998. Reasons for the euthanasia of dogs and cats. Vet. Rec. 143(4), 114; doi:10.1136/vr.143.4.114 Er, A., Tras, B., Cetin, G. and Dik, B. 2014. Amiodarone may prevent the tilmicosin-caused lethal toxicity. Acta Scie. Veter. 42(1), 1–4. Fawcett, W.J. 2019. Suxamethonium or rocuronium for rapid sequence induction of anesthesia. BJA. Educ. 19(12), 380e382; doi:10.1016/j.bjae.2019.09.001 Gates, M., Kells, N., Kongara, K. and Littlewood, K. 2023. Euthanasia of dogs and cats by veterinarians in New Zealand: protocols, procedures and experiences. New. Zealand. VetJ. 71(4), 172–185; doi:10.1080/00480169.2023.2194687 Häggström, J. and Guglielmini, C. 2021. Canine and Feline Electrocardiography: a Review. J. Vet. Inter. Med. 35(3), 1254–1271. Harding, J.R. and Smith, R.A. 2015. An introduction to electrocardiography in small animal practice. Vet. Record 177(2), 36–41. Keating, J.H. and Johnson, L.R. 2020. Cardiac Considerations in Veterinary Practice. J. Americ. Vet. Med. Assoc. , p67. Kienle, R.D. and Kienle, G.S. 2019. Electrocardiography for the veterinary technician. London: Wiley-Blackwell. p. 89 Kistner, S.I. and Mccarthy, R.N. 2018. Clinical Application of Electrocardiography in Dogs and Cats. Vet. Clinics. North. Amer. Small. AnimPract. 48(1), 1–20. Kogan, L.R. and Cooney, K.A. 2023. Defining a “Good Death”: exploring Veterinarians’ Perceptions of Companion Animal Euthanasia. Animals 13, 2117; doi:10.3390/ani13132117 Kollias, N.S., Hess, W.J., Johnson, C.L., Murphy, M. and Golab, G.A. 2023. Literature review on current practices, knowledge, and viewpoints on pentobarbital euthanasia performed by veterinarians and animal remains disposal in the United States. J. Am. Vet. Med. Assoc. 261, 733–738; doi:10.2460/javma.22.08.0373 Leary, S., Underwood,W., Anthony, R. and Cartner, S. 2020. Guidelines for the euthanasia of animals. J. Am. Vet. Med. Assoc. Schaumberg, IL, USA.p:69 Levy, M.A. and Gunter-hunt, G. 2020. ECG interpretation: the basics. Vet. Med. 115 (12), 376–384. Maharani, N.P.R., Fadlyah, N.A., Setyaningrum, D.A.W. and Hairunisa, N. 2024. Effects of euthanasia on animal research. Vet. Biomed. Clin. J. 6(1), 41–46. Mahmood, B.M. 2022. A Comparison between ketamine-xylazine and ketamine-midazolam or all of them to induce balance anesthesia in rabbits. Iraqi. JVet. Scie. 36(2), 499–506; doi:10.33899/ijvs.2021.130618.1852 Mazón-Ruizj, G., Romero-González, E., Sánchez, J., Banegas-derase, M., Salgado-Barquinero, L.A., Gutiérrez-de, R., Alcázar, J.J., Bande-Fernández, M., Gorostidia, R., Vargal, J.J., Bande-Fernández, M. and Gorostidia, R. 2024. Hypertonic saline and heart failure: “sodium-centric” or “chlorine-centric”? Nefrologia, 44(3), 338–343. doi: 10.1016/j.nefroe.2024.06.009 Mccarthy, J.R. 2019. Monitoring techniques in veterinary cardiology. Vet. Clinics North Amer. Small Anim. Pract. 90. Monahan, S. 2023. The effects of repeated doses of xylazine-ketamine and medetomidine-ketamine anesthesia on DNA damage in the liver and kidney. J. Clin. Med. 111, 123; doi:10.1590/acb385723 O’Brien, J., Wright, B.M. and Mccarthy, J.R. 2018. Cardiac arrhythmias in dogs receiving antibiotic therapy. Vet. Cardio. 20(6), 453–460; doi:10.1016/j.jvc.2018.05.005 O’Brien. 2018. Supportive Care in Veterinary Emergency Medicine. J. Vet. Mere. Crit. Care. , 34. Pepper, B.M., Chan, H., Ward, M. and Quain, P. 2023. Euthanasia of dogs by Australian veterinarians: a survey of clinical practice. Vet. Sci. 27, 317; doi:10.3390/vetsci10050317 Tunahan, S.2023. The effects of repeated doses of xylazine-ketamine and medetomidine- ketamine anesthesia on DNA damage in the liver and kidney. Acta Cir Bras. 38, e385723. doi: 10.1590/acb385723 Verhofstadt, M., Moureau, L., Pardon, K. and Liégeois, A. 2024. Ethical perspectives regarding euthanasia, including in the context of adult psychiatry: a qualitative interview study among healthcare workers in Belgium. BMC. Med. Ethics 25, 60. Weller, R. 2017. Clinical management of heart disease in dogs. Vet. J. 224, 38–45; doi:10.1016/j.tvjl.2016.12.011 Yan, G., Liang, Q., Wen, X., Peng, J., Deng, R., Lv, L., Ji, M., Deng, X., Wu, L., Feng, X. and He, J. 2020. Preparation, characterization, and pharmacokinetics of tilmicosin taste-masked formulation via hot-melt extrusion technology. Colloids Surf. B. Biointerfaces 196, 111293; doi:10.1016/j.colsurfb.2020.111293 Yildiz, R., Durna Corum, D., Corum, O., Ider, M., Atik, O., Ok, M. and Uney, K. 2023. The effect of tilmicosin and diclofenac sodium combination on cardiac biomarkers in sheep. J. Vet. Scie. 26(1), 5–12. Yoshida, K. 2023. Comparison of low and high doses of pentobarbital or thiopental for euthanasia of isoflurane-anesthetized pigs. J. Am. Assoc. Lab. Anim. Sci. 62(2), 185–188; doi:10.30802/AALAS-JAALAS-22-000093/ | ||
| How to Cite this Article |
| Pubmed Style Zedan IA, Al-anaaz MT, Shekho HA, Iraqi OMA, Albadrani BA, Alkattan LM. Efficacy of tilmicosin and suxamethonium for dog euthanasia: A comparative study. Open Vet. J.. 2025; 15(12): 6709-6720. doi:10.5455/OVJ.2025.v15.i12.51 Web Style Zedan IA, Al-anaaz MT, Shekho HA, Iraqi OMA, Albadrani BA, Alkattan LM. Efficacy of tilmicosin and suxamethonium for dog euthanasia: A comparative study. https://www.openveterinaryjournal.com/?mno=282826 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.51 AMA (American Medical Association) Style Zedan IA, Al-anaaz MT, Shekho HA, Iraqi OMA, Albadrani BA, Alkattan LM. Efficacy of tilmicosin and suxamethonium for dog euthanasia: A comparative study. Open Vet. J.. 2025; 15(12): 6709-6720. doi:10.5455/OVJ.2025.v15.i12.51 Vancouver/ICMJE Style Zedan IA, Al-anaaz MT, Shekho HA, Iraqi OMA, Albadrani BA, Alkattan LM. Efficacy of tilmicosin and suxamethonium for dog euthanasia: A comparative study. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6709-6720. doi:10.5455/OVJ.2025.v15.i12.51 Harvard Style Zedan, I. A., Al-anaaz, . M. T., Shekho, . H. A., Iraqi, . O. M. A., Albadrani, . B. A. & Alkattan, . L. M. (2025) Efficacy of tilmicosin and suxamethonium for dog euthanasia: A comparative study. Open Vet. J., 15 (12), 6709-6720. doi:10.5455/OVJ.2025.v15.i12.51 Turabian Style Zedan, Ibraheem Ahmed, Mae Thanoon Al-anaaz, Hiba A. Shekho, Osamah M. Al Iraqi, Basima A. Albadrani, and Layth Mahmoud Alkattan. 2025. Efficacy of tilmicosin and suxamethonium for dog euthanasia: A comparative study. Open Veterinary Journal, 15 (12), 6709-6720. doi:10.5455/OVJ.2025.v15.i12.51 Chicago Style Zedan, Ibraheem Ahmed, Mae Thanoon Al-anaaz, Hiba A. Shekho, Osamah M. Al Iraqi, Basima A. Albadrani, and Layth Mahmoud Alkattan. "Efficacy of tilmicosin and suxamethonium for dog euthanasia: A comparative study." Open Veterinary Journal 15 (2025), 6709-6720. doi:10.5455/OVJ.2025.v15.i12.51 MLA (The Modern Language Association) Style Zedan, Ibraheem Ahmed, Mae Thanoon Al-anaaz, Hiba A. Shekho, Osamah M. Al Iraqi, Basima A. Albadrani, and Layth Mahmoud Alkattan. "Efficacy of tilmicosin and suxamethonium for dog euthanasia: A comparative study." Open Veterinary Journal 15.12 (2025), 6709-6720. Print. doi:10.5455/OVJ.2025.v15.i12.51 APA (American Psychological Association) Style Zedan, I. A., Al-anaaz, . M. T., Shekho, . H. A., Iraqi, . O. M. A., Albadrani, . B. A. & Alkattan, . L. M. (2025) Efficacy of tilmicosin and suxamethonium for dog euthanasia: A comparative study. Open Veterinary Journal, 15 (12), 6709-6720. doi:10.5455/OVJ.2025.v15.i12.51 |