| Research Article | ||
Open Vet. J.. 2025; 15(12): 6684-6696 Open Veterinary Journal, (2025), Vol. 15(12): 6684-6696 Research Article The protective effect of baicalin against cyclophosphamide-induced testicular damage in ratsJianqin Li†, Chunlian Song†, Xue Zhang, Yalong Sun, Nan Wang and Xianghua Shu*College of Veterinary Medicine, Yunnan Agricultural University, Kunming, China †Both authors contributed equally to this article *Corresponding Author:Xianghua Shu. College of Veterinary Medicine, Yunnan Agricultural University, Kunming, China. Email: ynndsxh [at] ynau.edu.cn Submitted: 07/09/2025 Revised: 11/11/2025 Accepted: 23/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
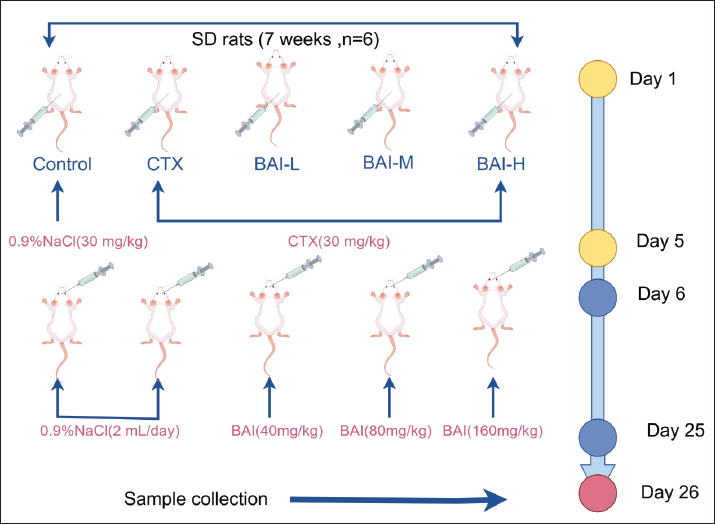
AbstractBackground: Cyclophosphamide (CTX) is a widely used chemotherapeutic agent; however, its clinical application is often limited by significant reproductive toxicity, particularly testicular damage. Baicalin (BAI) has demonstrated broad pharmacological activities, including anti-inflammatory, antioxidant, and anti-apoptotic effects. Aim: To investigate the protective effect of BAI on testicular damage induced by CTX in rats. Methods: Network pharmacology was used to identify the core targets of BAI-testis damage. Animal experiments were conducted using 30 SPF-grade 7-week-old SD rats divided into a control group (Control), a CTX group, a low-dose BAI group (BAI-L), a medium-dose BAI group (BAI-M), and a high-dose BAI group (BAI-H), with six rats in each group. Molecular docking, quantitative polymerase chain reaction, and Western blot were used to validate the core targets of network pharmacology. H&E staining was performed to observe testicular tissue damage. The levels of interleukin-1β, interleukin-10, reactive oxygen species, and malondialdehyde in the testes and the levels of follicle-stimulating hormone, luteinizing hormone, and testosterone in the serum were measured. Immunofluorescence was used to detect germ, supporting, and interstitial cells in the testes. Results: Network pharmacology identified 127 common targets between BAI and testicular damage. BAI acts on Ptgs2, Gsk3b, and Pparg targets. Molecular docking and animal experiments indicated that BAI exhibits strong binding affinity with Ptgs2, Gsk3b, and Pparg. Animal experiments showed that BAI can improve CTX-induced pathological tissue damage, reduce inflammatory responses, and alleviate oxidative stress in rat testes. BAI can repair CTX-induced damage to germ cells, supporting cells, interstitial cells, and repaired reproductive hormones, with the extent of repair showing a dose-dependent relationship. Conclusion: This study demonstrates the beneficial effects of BAI in alleviating CTX-induced reproductive toxicity, and BAI can be used to reduce the side effects of CTX. Keywords: Baicalin, Cyclophosphamide, Network pharmacology, Reproductive hormones, Testicular injury. IntroductionAs vital organs of the male reproductive system, the testes are primarily responsible for spermatogenesis and the secretion of androgens, chiefly testosterone (T). Testicular function integrity is crucial for maintaining male reproductive health, the development of secondary sexual characteristics, and overall metabolic homeostasis (Makela et al., 2019). Owing to its high proliferative rate, the spermatogenic epithelium within the testes exhibits a marked sensitivity to ionizing radiation, elevated temperatures, toxic compounds, and certain drugs. Cyclophosphamide (CTX) is a broad-spectrum anticancer drug that is widely used in the clinical treatment of various malignant tumors and autoimmune diseases. However, its significant therapeutic efficacy is often accompanied by notable reproductive system toxicity side effects (Emadi et al., 2009; Athira et al., 2022). Baicalin (BAI) is a flavonoid glycoside active component isolated and purified from the dried rhizomes of the Lamiaceae family medicinal plant Scutellaria baicalensis (Li et al., 2004). It possesses potent antioxidant, anti-inflammatory, and anti-apoptotic properties and protects tissues and organs from chemical toxins or pathological damage (Liao et al., 2021). In recent years, the potential of BAI in protecting the reproductive system has gradually garnered attention. Studies have shown that S. baicalensis or its extract has a protective effect against testicular damage (Yin et al., 2021). Whether BAI can alleviate CTX-induced testicular damage remains unclear. This study aimed to investigate the protective effects of BAI on CTX-induced testicular damage using a rat testicular damage model. Materials and MethodsExperimental materialsThirty 7-week-old male SPF-grade SD rats weighing 250 ± 10 g were purchased from Kunming University. The rat qualification license number is: SYXK (Yunnan) K2020-0001; BAI (purity ≥98%) was purchased from Nanjing Dousifu Biotechnology Co., Ltd., and CTX was purchased from Beijing Solarbio Technology Co., Ltd. Main reagents and instruments usedRat interleukin-1β (IL-1β), rat interleukin-10 (IL-10), rat reactive oxygen species (ROS), rat malondialdehyde (MDA), rat T, rat follicle-stimulating hormone (FSH), and rat luteinizing hormone (LH) detection kits were all purchased from Jingmei Bioengineering Co., Ltd.; RNA rapid extraction kit was purchased from TaKaRa Company. DDX4, SOX9, HSD3B1, Ptgs2, Gsk3b, and Pparg antibodies were purchased from Jiangsu Qinke Biotechnology Research Center Co., Ltd. A high-speed refrigerated centrifuge was purchased from Eppendorf AG, Germany. A fluorescent quantitative polymerase chain reaction (qPCR) instrument was purchased from Bio-Rad Company. A full-wavelength enzyme-linked immunosorbent assay instrument was purchased from Thermo Fisher Company. An inverted microscope was purchased from Chongqing Optoelectronic Instrument Co., Ltd. An electronic balance was purchased from Shanghai Tianping Jingke Company. Network pharmacology and molecular dockingScreening of the active components and targets of BAIThe SMILES number was obtained from the PubChem database using “BAI” as the keyword. Subsequently, the SMILES number was imported into the Swiss ADME database to screen for the active components of BAI. Subsequently, the SMILES number was imported into the Swiss Target Prediction database to predict the potential targets of BAI. The two bioinformatics databases were merged and duplicates were removed to obtain the target sites of BAI. Acquisition of target testicular injuryUsing “testicular injury” as the keyword, searches were conducted in the GeneCards and OMIM databases. The two bioinformatics databases were merged, and duplicates were removed to obtain the targets associated with testicular injury. Scutellarin-testis injury Venn diagram, PPI network construction, and core target screeningTo identify common intersection targets, the putative targets of BAI and known targets associated with testicular injury were imported into the Venny 2.1.0 platform, and a Venn diagram was constructed. Subsequently, these overlapping targets were submitted to the STRING database to construct a PPI network. Subsequently, the resulting PPI network was visualized and analyzed using Cytoscape 3.9.1. GO function and Kyoto Encyclopedia of Genes and Genomes pathway enrichment analysisThe intersection target points of BAI action targets and testicular injury targets were imported into the DAVID website to obtain the GO function and KEGG pathway enrichment analysis of BAI treatment for testicular injury. Perform plotting using the Wei Sheng Xin website. Molecular docking and visualizationCytoscape was used to screen the core targets and perform molecular docking with BAL. First, the 3D structure of BAI was downloaded from PubChem, then hydrogenation and dehydration were performed using Autodock Vina, and the file was saved in pbat format. The crystal structure of the core protein was downloaded from the RCSB Protein Database (PDB). Water molecules and the original ligand were removed from the target protein using Pymol and saved in pdb format. Next, the pdbqt format of the receptor (protein) was obtained using Autodock Vina. Finally, Autodock Vina was used for molecular docking. The results were visualized using Pymol and Discovery Studio software. Grouping and treatment of animalsAfter 7 days of acclimatization, the rats were randomly divided into 5 groups: Control group (Control), CTX group (30 mg/kg, CTX), low-dose BAI group (40 mg/kg, BAI-L), medium-dose BAI group (80 mg/kg, BAI-M), and high-dose BAI group (160 mg/kg, BAI-H), with six rats in each group. The control group received intraperitoneal injection of 0.9% normal saline for five consecutive days, the other groups received intraperitoneal injection of CTX (30 mg/kg) for five consecutive days, and the control and CTX groups, Control group and CTX group received gastric perfusion of 0.9% normal saline for 19 consecutive days from the sixth day. Rats in the BAI group were orally administered with 2 ml of BAI solution of different concentrations every day and euthanized on the 20th day after 19 days. Serum, epididymis, and testicular tissue samples were then collected. To express the experiment process more intuitively, we created an experiment flow chart (Fig. 1).
Fig. 1. The experimental flow timeline. Sample collectionAt the end of the experiment, blood was collected after anesthesia, and euthanasia was performed by cervical dislocation. Blood was collected and centrifuged to obtain serum, and the testicular tissue was washed with Phosphate-Buffered Saline. One side of the testis was fixed in 4% paraformaldehyde solution, and the other side was stored at −80°C until further use. Measurement and methods of indicatorsMorphological examination of testicular tissueFormalin-fixed testicles were rinsed under running water for 10 minutes, dehydrated with alcohol, and cleared with xylene. The tissue was then embedded in paraffin, and the wax was trimmed after the paraffin block had cooled and solidified. The sections were placed in a constant-temperature water bath for spreading. Sections were baked in a constant-temperature oven at 60°C for 6 hours. After cooling, sections were stained with hematoxylin and eosin, air-dried at room temperature, mounted with neutral binder, and examined under a microscope. Enzyme-linked immunosorbent assay (ELISA) measurementThe collected testicular tissue was ground and centrifuged at 12,000 rpm for 10 minutes to separate the supernatant, and the levels of IL-1β, IL-10, Superoxide Dismutase, and MDA in the testicular tissue were measured. An ELISA reader was used to measure the levels of T, LSH, and LH in the serum, following the specific steps outlined in the relevant assay kit instructions. Detection of sperm countThe tail of the epididymis was placed in a culture dish and rinsed with 1 ml of physiological saline at 37°C to collect semen. The epididymis rinse solution and epididymis were collected together in a 1.5 ml centrifuge tube, and the sperm were counted using a cell counting chamber. DNA/RNA extraction and real-time quantitative fluorescence polymerase chain reaction (PCR) detectionTotal RNA was extracted from homogenized testis tissues in liquid nitrogen using a commercial DNA/RNA extraction kit according to the manufacturer’s instructions. The extracted RNA was subsequently used as a template for real-time qPCR analysis. Table 1 lists the sequences of the specific primers employed. Table 1. Relevant primers.
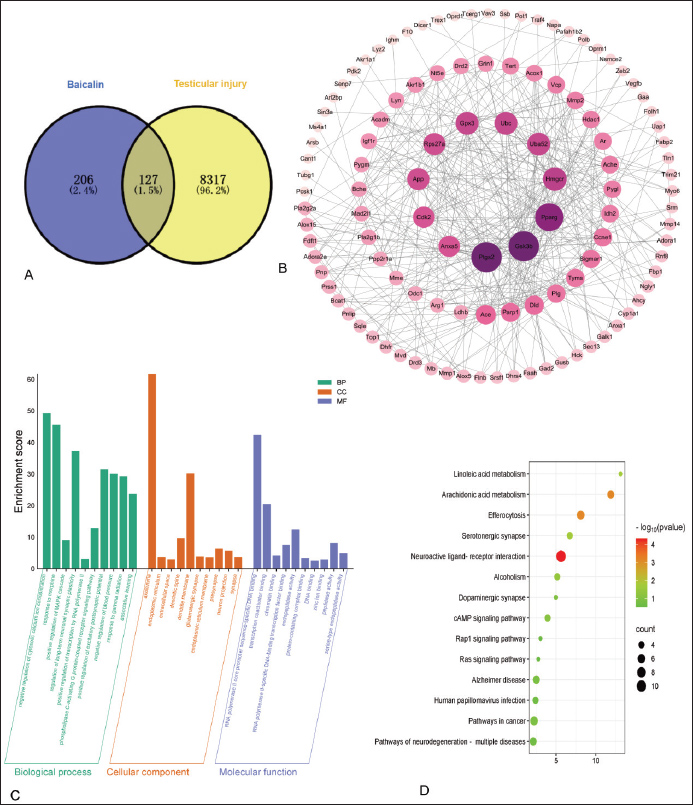
Western blot analysisTotal protein was extracted from approximately 0.1 g of testicular tissue using RIPA lysis buffer. The lysate was cleared by centrifugation at 12,000 × g for 15 minutes at 4°C. The protein concentration was quantified using a BCA kit, and the samples were denatured in loading buffer. Proteins were separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis, transferred to a PVDF membrane, and blocked. The membrane was sequentially incubated with a specific rabbit primary antibody overnight at 4°C and an HRP-conjugated goat anti-rabbit secondary antibody for 2 hours at room temperature, followed by thorough TBST washes between and after incubation. The signal was detected using an ECL reagent, and the band intensities were quantified by normalizing the gray value of the target protein to that of GAPDH. ImmunofluorescenceParaffin section of testicular tissue was dewaxed and rehydrated. Sections were blocked for 1 hour at room temperature using a blocking solution containing 5% normal serum and 1% bovine serum albumin. The primary antibody was incubated in a wet chamber at 4°C overnight, followed by three washes with Phosphate-Buffered Saline with Tween® 20 for 5 minutes each. The HRP-labeled secondary antibody was added and incubated at room temperature for 1 hour, followed by three washes with PBST for 5 minutes each. DAPI solution was added and incubated at room temperature in the dark for 10 minutes. The slides were sealed with an antifade mounting medium (to fix the sample) and stored in the dark. Statistical methodsAll data were analyzed using one-way analysis of variance in GraphPad Prism 10.1.2 software. Results are expressed as “mean ± standard deviation,” and comparison between groups was performed using t-test. Ethical approvalThe Institutional Animal Care and Use Committee of Yunnan Agricultural University approved all animal experiments, and all procedures were performed in accordance with relevant guidelines and regulations. ResultsResults of network pharmacologyA total of 333 drug activity targets for BAI and 8,444 targets related to testicular injury were screened from the database, with 127 overlapping targets (Fig. 2C). As shown in the core target diagram, the icons for targets such as Ptgs2, Gsk3b, and Pparg were relatively larger and darker in color, indicating their potential therapeutic effects on testicular injury (Fig. 2B). Biological process analysis primarily includes negative regulation of cytoplasmic calcium ion concentration, response to morphine, and positive regulation of the MAPK cascade (Fig. 2C). KEGG pathway enrichment analysis primarily includes neuroactive ligand-receptor interactions, linoleic acid metabolism, and serotonergic synaptic signaling pathways (Fig. 2D).
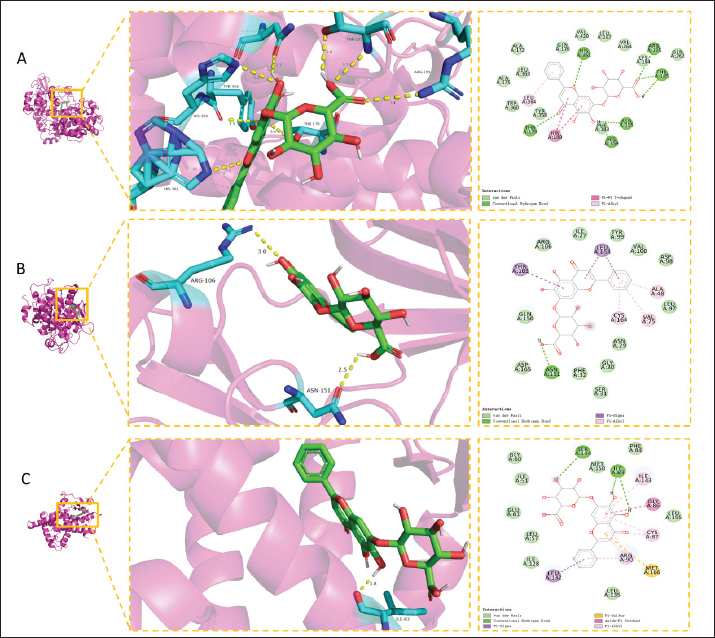
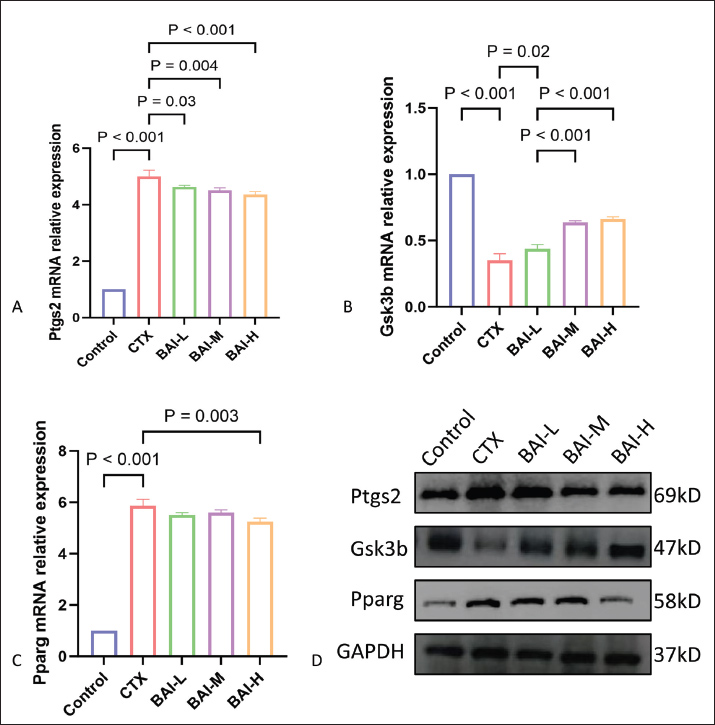
Fig. 2. Network pharmacology results of BAI and testicular injury. (A) Venn diagram of BAI and testicular injury. (B) Core target screening. Note: The thickness of the line represents the connection score, and the thicker the line, the closer the relationship; the larger the target icon, the darker the color, the greater the degree value, and the greater the correlation. (C) GO enrichment analysis bar graph. GO enrichment analysis bar graph. Note: Biological process (BP), cellular component (CC) 1, and molecular function (MF). The different items are arranged according to the size of the enrichment score. The greater the enrichment score, the more significant the enrichment items. The greater the enrichment score, the more significant the enrichment items. (D)Bubble map for KEGG signaling pathway enrichment analysis. Note: P-value: represents the degree of enrichment, expressed by the bubble color; the darker the color, the higher the degree of enrichment; count value: represents the number of genes enriched, expressed by the bubble size; the larger the bubble, the more the number of genes enriched. Represents the number of enriched genes, expressed by the bubble size. The larger the bubble, the more the number of enriched genes. Molecular docking results and experimental validationMolecular docking analysis revealed that BAI exhibits strong binding affinity with three core target proteins (Ptgs2, Gsk3b, and Pparg). The binding energy between BAI and Ptgs2 was −10.5 kcal/mol (Fig. 3A), the binding energy between BAI and Gsk3b was −8.6 kcal/mol (Fig. 3B), and the binding energy between BAI and Pparg was −8.6 kcal/mol (Fig. 3C); all binding energies are less than −7 kcal/mol. Real-time fluorescent quantitative PCR was used to detect the mRNA expression levels of Ptgs2, Gsk3b, and Pparg in rat testes. The gene expression of Ptgs2, Gsk3b, and Pparg was upregulated in the CTX group compared with the control group. Different concentrations of BAI could downregulate the upregulated genes (Fig. 4A–C). The protein expression levels were detected by Western blot. Relative to the control group, CTX exposure significantly upregulated Ptgs2 and Pparg expression while downregulating Gsk3b expression. Baicalin treatment effectively reversed these alterations in a dose-dependent manner: the elevated levels of Ptgs2 and Pparg were significantly reduced, and the suppressed level of Gsk3b was increased (Fig. 4D).
Fig. 3. Molecular docking and animal experiment verification result diagram. (A) Molecular docking model of Baicalin-Ptgs2. (B) Molecular docking model of Baicalin-Gsk3b. (C) Molecular docking model of Baicalin-Pparg.
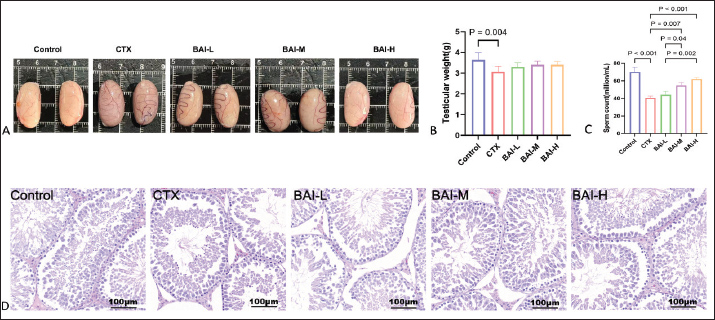
Fig. 4. Ptgs2, Gsk3b, and Pparg mRNA and protein expression levels (A) Relative expression of Ptgs2 mRNA. (B) Relative expression of Gsk3b mRNA. (C) Relative expression of Pparg mRNA. (D) Ptgs2, Gsk3b, and Pparg protein expression levels. Effects of BAI on testicular damage induced by CTX in ratsIn the Control group, the seminiferous tubules maintained their intact tubular structure, with various types of germ cells in the seminiferous epithelium arranged in a polarized manner according to their developmental gradient, forming neat and orderly regular layers. The testicular interstitium had densely distributed interstitial cells with normal interstitial spaces, and no pathological changes were observed in the testicular tissue. The CTX group showed a reduction in interstitial cells, with significantly enlarged interstitial spaces and the appearance of vacuolization and germ cell depletion, compared with the control group. The interstitial spaces in the testicular interstitium of all BAI groups were larger than those in the Control group, but smaller than those in the CTX group. The BAI-H group showed significant improvement in lesions (Fig. 5D). No significant differences in testicular weight changes were observed (Fig. 5A and B). Compared with the Control group, sperm count was extremely significantly reduced in the CTX group (p < 0.001); compared with the CTX group, sperm count was significantly increased in the BAI-L group (p < 0.05), extremely significantly increased in the BAI-M group (p < 0.01), and extremely significantly increased in the BAI-H group (p < 0.001) (Fig. 5C).
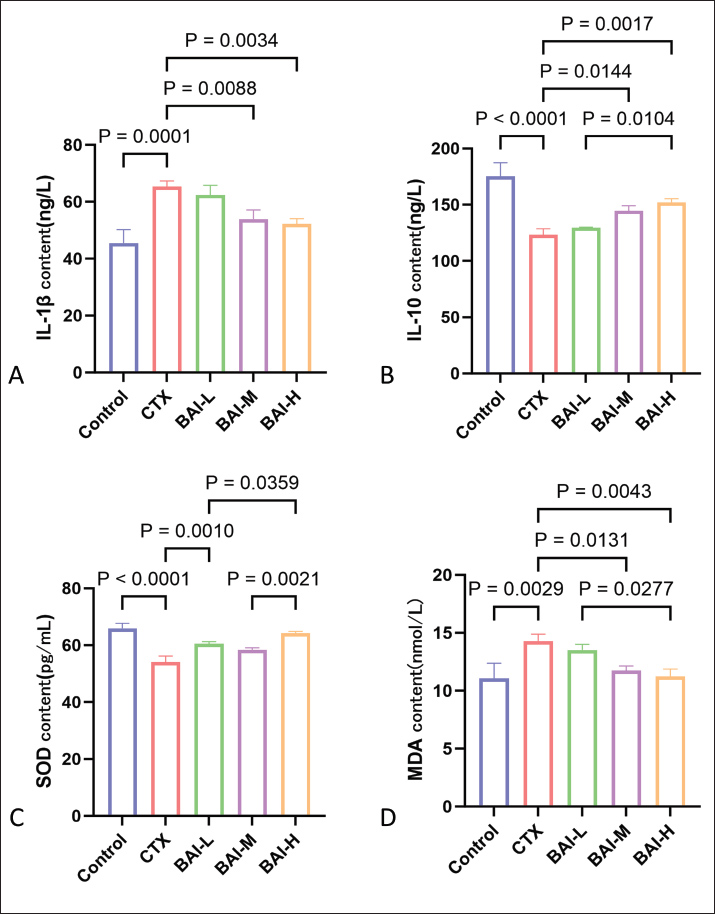
Fig. 5. Effects of CTX and different doses of BAI on testes. (A) Images of testicular tissue. (B) Testicular weight and (C) Sperm counts. (D) H&E sections 200x. Effects of BAI on inflammatory factors and antioxidant parameters in CTX-induced testes of ratsCompared with the Control group, the CTX group showed significantly increased levels of IL-1β and MDA (p < 0.05) and significantly decreased levels of IL-10 and SOD (p < 0.05); Compared with the CTX group, the BAI-H group showed significantly lower levels of IL-1β and MDA (p < 0.01) and significantly higher levels of IL-10 and SOD (p < 0.01), indicating that BAI exerts anti-inflammatory and antioxidant effects in CTX-induced testicular damage in rats (Fig. 6A–D).
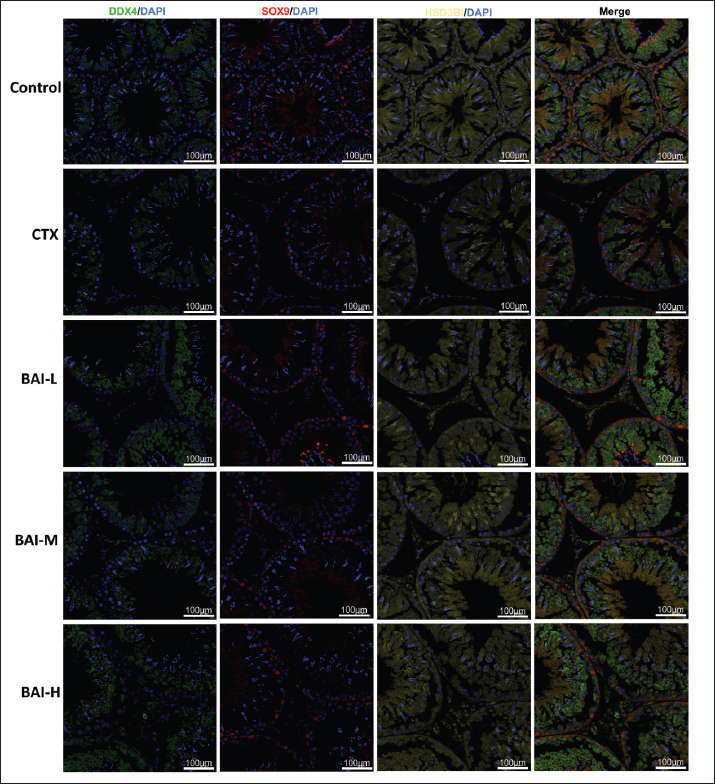
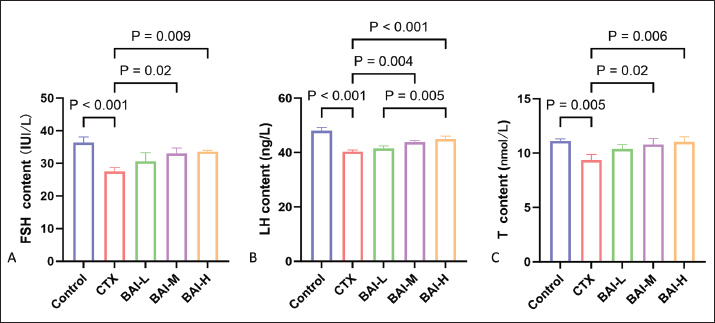
Fig. 6. The effects of CTX and different doses of BAI on inflammatory factors and antioxidant parameters. (A) IL-1β content. (B) IL-10, (C) SOD, and (D) MDA contents. Effects of CTX-induced BAI on testicular germ cells and reproductive hormones in ratsDDX4 (testicular germ cell marker), SOX9 (testicular supporting cell marker), and HSD3B (testicular interstitial cell marker) exhibited typical distribution and expression patterns in the Control group. The CTX group showed a significant reduction in the number of DDX4-positive germ cells, disorganized structures of SOX9-positive supporting cells, and a marked decrease in the number or fluorescence intensity of HSD3B-positive interstitial cells compared with the Control group. The damage to testicular tissue was improved to varying degrees after intervention with different doses of BAI. The number and morphological characteristics of the three marker cells were partially restored in the BAI-L group compared to the CTX group, but the improvement was limited. The protective effect of the BAI-M group was more pronounced, characterized by significantly enhanced positive signals for DDX4, SOX9, and HSD3B, approaching the levels of the control group. The BAI-H group exhibited the strongest protective effect, with the fluorescence signal intensity and distribution patterns of germ cells, supporting cells, and interstitial cells significantly restored, approaching or even reaching the levels of the Control group (Fig. 7). The levels of reproductive hormones FSH, LH, and T in the CTX group showed a significant decrease, whereas the reproductive hormone levels in the BAI groups showed a significant dose-dependent increase (Fig. 8A–C).
Fig. 7. Expression of germ, Sertoli, and stromal cells in the testes.
Fig. 8. Effects of BAI on reproductive hormones in CTX-induced rats. (A) FSH content. (B) LH content. (C) T content. DiscussionWhile chemotherapy for malignant tumors saves lives, its severe side effects pose a significant threat to patients’ quality of life. CTX, a widely used alkylating agent and immunosuppressant, has significant cytotoxic effects that effectively kill tumor cells and cause significant damage to rapidly proliferating normal tissues, especially the reproductive system (Ghobadi et al., 2017). Male testicular tissue is highly sensitive to CTX toxicity, with damage primarily manifesting as spermatogenic epithelial structure disruption, a sharp reduction or even apoptosis of spermatogenic cells at all stages, impaired spermatogenesis, and a significant decline in sperm quality (Elangovan et al., 2006). In severe cases, this can lead to irreversible spermatogenic dysfunction and infertility. The core pathological mechanism of this damage involves the explosive production of ROS, which triggers oxidative stress, excessive activation of inflammatory cascades, and widespread apoptosis of germ and supporting cells (Ceribasi et al., 2010). In recent years, natural products have demonstrated great potential in the prevention and treatment of drug-induced organ damage due to their multi-targeted and low-toxicity properties. Baicalin, a flavonoid compound, has been extensively studied and proven to possess outstanding antioxidant, anti-inflammatory, anti-apoptotic, and immunomodulatory activities (Dinda et al., 2017; Wang et al., 2024). Therefore, exploring safe and effective strategies to antagonize CTX-induced testicular damage holds significant clinical implications for preserving fertility and reproductive health in male patients with cancer. The results show that network pharmacology identified 127 target genes at the intersection of BAI and testicular injury, with Ptgs2, Gsk3b, and Pparg as core target genes. These target genes’ functions are highly consistent with the pathological mechanisms of testicular injury. Prostaglandin (PG)-endoperoxide synthase 2 (Ptgs2) encodes a key rate-limiting enzyme in PG biosynthesis. It plays a central role in mediating inflammatory processes and associated tissue repair mechanisms (Kosaka et al., 1994). Ptgs2 expression is upregulated in inflammatory diseases such as orchitis and varicocele, promoting PG production, worsening inflammatory responses, or participating in the repair process. However, excessive inflammatory responses may also impair the spermatogenic microenvironment. PGs can indirectly regulate T synthesis, thereby affecting interstitial cell function (Xiao et al., 2024). The results showed that Ptgs2 expression was upregulated in rat testicular tissue after CTX induction, with intensified inflammatory responses and oxidative stress. However, BAI treatment helped downregulate Ptgs2 expression, alleviate inflammatory responses, and reduce oxidative stress, confirming the protective effect of S. baicalensis extract. Glycogen synthase kinase-3β (Gsk3b) is a multifunctional serine/threonine kinase that plays a pivotal role in regulating diverse cellular processes, including cell proliferation, apoptosis, metabolism, and inflammatory responses (Lin et al., 2020). Gsk3b serves as a key node in the inflammatory amplification process in testicular injury. Inhibition of NF-B activity leads to excessive activation of NF-κB, which is the core mechanism driving destructive inflammatory responses. In addition, its pro-apoptotic function may be involved. This leads to atrophy of the seminiferous tubules in testicular tissue and intensifies inflammatory responses, impairing testicular spermatogenic function and increasing sperm abnormalities (Carmely et al., 2009). The results of this study show that following CTX induction, Gsk3b expression is downregulated in rat testicular tissue, while BAI treatment helps to upregulate Gsk3b expression, suggesting that BAI may help normalize CTX-disrupted Gsk3b expression. Peroxisome proliferator activated receptor gamma(Pparg) exerts a protective role in testicular injury by regulating inflammation and oxidative stress and supporting cell function. It can mitigate testicular inflammatory damage by regulating the inflammatory response and inhibiting the release of pro-inflammatory factors (Li et al., 2025). Additionally, Pparg can regulate cellular metabolism, maintain energy balance in testicular cells, and protect testicular spermatogenic function. Furthermore, it participates in the regulation of apoptosis, inhibiting excessive apoptosis of testicular cells, and exerts certain repair and protective effects when the testis is damaged (Regueira et al., 2018). CTX induction significantly upregulated Pparg expression in the rat testis, concurrent with heightened inflammation and oxidative stress. This likely signifies a failed compensatory response, where the body attempts to leverage the protective potential of Pparg against severe injury. Baicalin treatment subsequently lowered Pparg levels. We hypothesize that this effect is indirect: by directly reducing testicular damage through its robust anti-inflammatory and antioxidant properties, BAI weakens the primary insult that initially drove Pparg upregulation. As the pathological drive diminishes, Pparg expression returns to a physiological baseline. Thus, the action of BAI reflects a fundamental resolution of tissue stress rather than an inhibition of the protective pathway per se. This study did not evaluate Pparg transcriptional activity, which is a limitation. The enrichment of pathways related to neuroactive ligand-receptor interaction and serotonergic synapse in KEGG analysis points to a potential mechanism by which BAI influences testicular function by regulating the neuroendocrine network, such as the hypothalamic-pituitary-gonadal axis (HPG axis). However, this putative mechanism remains speculative due to the lack of direct validation, a key limitation that defines an important direction for future research. CTX can cause spermatogenic cells to undergo vacuolization, disorganization, and detachment into the lumen, with the formation of multinucleated giant cells in the lumen. Reduced numbers, smaller volumes, and cytoplasmic vacuolization of interstitial cells. Cells exhibit vacuolization, morphological abnormalities, and disruption of tight junction structures (Mu et al., 2023). Baicalin significantly improves sperm count, motility, and deformity rate, reduces spermatogenic epithelium damage, and alleviates oxidative stress, inflammation, and apoptosis (Sui et al., 2019). In a cadmium-induced reproductive toxicity model, scutellarin reduces testicular tissue congestion and edema, as well as damage to the structure of seminiferous tubules; reduces cadmium accumulation; significantly enhances antioxidant enzyme activity; and lowers MDA levels (Wen et al., 2013; Ali et al., 2022). Baicalin attenuated CTX-induced testicular injury and restored the decreased sperm count in rats, corroborating its reparative effect on testicular tissue injury. The use of Bouin’s fixative in future studies would provide superior morphological details. As a highly sensitive immune-privileged organ, the testis relies on the antioxidant system and the homeostasis of inflammatory factors to maintain its function. CTX also increases the release of pro-inflammatory factors such as IL-1β and IL-6, exacerbating oxidative stress, cell apoptosis, and fibrosis (Sarkar et al., 2021). The metabolism of CTX produces ROS, and excessive ROS attacks lipid membranes, causing damage and enzyme inactivation, thereby worsening cellular damage and apoptosis (Cengiz et al., 2020). The results of this experiment showed that after CTX induction, the pro-inflammatory factor IL-1β was abnormally elevated in testicular tissue, whereas BAI significantly inhibited its elevation, promoted the release of the anti-inflammatory factor IL-10, and alleviated the inflammatory response. CTX treatment caused a decrease in SOD activity and an increase in MDA levels in testicular tissue, indicating exacerbated oxidative stress, whereas BAI intervention restored antioxidant enzyme activity. Support cells, as key cells constituting the blood-testis barrier and providing nutritional and microenvironmental support for spermatogenic cells, have impaired structural and functional integrity, further worsening spermatogenic process disruptions. In addition, CTX can directly damage testicular interstitial cells, significantly reducing their ability to synthesize and secrete T (Sancho et al., 2017). The reduction in T levels not only directly affects spermatogenesis but also disrupts the HPG axis through negative feedback regulation, manifested as compensatory increases in serum FSH and LH levels, forming a vicious cycle (McCoskey and Vernon, 2024). CTX causes degeneration of the spermatogenic epithelium, apoptosis of spermatogenic cells, dysfunction of supporting cells and interstitial cells, and interstitial fibrosis. However, studies have shown that BAI can protect supporting cells and the blood-testis barrier (Oyagbemi et al., 2016). The results showed that CTX infection destroyed spermatogonia at all stages and reduced the number of supporting and interstitial cells. The destruction of spermatogonia at all stages was reversed after BAI intervention, the number of supporting cells and interstitial cells increased, and reproductive hormone levels were restored. Testicular cell recovery is beneficial for maintaining spermatogenesis. The experimental results suggest that BAI has a protective effect on testicular integrity. This study focuses on the protective effects of BAI against CTX-induced testicular damage and its potential mechanisms. The expression of Ptgs2, Gsk3b, and Pparg under CTX challenge implicates these factors in the testicular protective mechanism of BAI. We propose that BAI alleviates oxidative stress and inflammation by modulating these targets, thereby preserving testicular integrity and hormonal balance. Nevertheless, future studies should directly validate the exact upstream and downstream relationships within this pathway. ConclusionBaicalin inhibits inflammatory responses and enhances antioxidant defense by regulating Ptgs2, Gsk3b, and Pparg expression. It repairs CTX-induced testicular tissue damage in a dose-dependent manner, significantly increases sperm count, and restores the functions of germ cells, supporting cells, and interstitial cells. Reversing the decline in FSH, LH, and T reproductive hormones maintains the local microenvironment balance in the testicles. AcknowledgmentsThe author would like to express his gratitude to his supervisor, Professor Shu Xianghua, for providing the experimental platform and assistance. Conflict of interestThe authors declare no conflicts of interest. FundingThe Yunnan Key Laboratory of Veterinary Etiological Biology (Grant No: 202449CE340019), Yunnan Province Weixin County Recycling Agriculture Science and Technology Mission (202304BI090011), supported this project. Authors' contributionsJianqin Li, Chunlian Song, and Xue Zhang: Original draft, methodology, visualization, investigation, and data curation. Yalong Sun and Nan Wang: Methodology and visualization. Chunlian Song, Jianqin Li: Writing original draft, Writing Review. Chunlian Song and Xue Zhang: Writing Review Xianghua Shu: Project administration. Data availabilityData availability all data supporting the findings of this study can be found in the manuscript. ReferencesAli, W., Ma, Y., Zhu, J., Zou, H. and Liu, Z. 2022. Mechanisms of Cadmium-Induced Testicular Injury: a Risk to Male Fertility. Cells-Basel 11, 3601. Athira, V.R., Shivanandappa, T. and Yajurvedi, H.N. 2022. Cyclophosphamide, a cancer chemotherapy drug-induced early onset of reproductive senescence and alterations in reproductive performance and their prevention in mice. Drug. Chem. Toxicol. 45, 760–766. Carmely, A., Meirow, D., Peretz, A., Albeck, M., Bartoov, B. and Sredni, B. 2009. Protective effect of the immunomodulator AS101 against cyclophosphamide-induced testicular damage in mice. Hum. Reprod. 24, 1322–1329. Cengiz, M., Sahinturk, V., Yildiz, S.C., Sahin, I.K., Bilici, N., Yaman, S.O., Altuner, Y., Appak-Baskoy, S. and Ayhanci, A. 2020. Cyclophosphamide induced oxidative stress, lipid peroxidation, apoptosis and histopathological changes in rats: protective role of boron. J. Trace Elem. Med. Bio. 62, 126574. Ceribasi, A.O., Turk, G., Sonmez, M., Sakin, F. and Atessahin, A. 2010. Toxic effect of cyclophosphamide on sperm morphology, testicular histology and blood oxidant-antioxidant balance, and protective roles of lycopene and ellagic acid. Basic Clin. Pharmacol. 107, 730–736. Dinda, B., Dinda, S., DasSharma, S., Banik, R., Chakraborty, A., Dinda, M., 2017. Therapeutic potentials of baicalinBAI and its aglycone, baicalein against inflammatory disorders. Eur. J. Med. Chem. 131, 68-80. Elangovan, N., Chiou, T.J., Tzeng, W.F. and Chu, S.T. 2006. Cyclophosphamide treatment causes impairment of sperm and its fertilizing ability in mice. Toxicology 222, 60–70. Emadi, A., Jones, R.J. and Brodsky, R.A. 2009. Cyclophosphamide and cancer: golden anniversary. Nat. Rev. Clin. Oncol. 6, 638–647. Ghobadi, E., Moloudizargari, M., Asghari, M.H. and Abdollahi, M. 2017. The mechanisms of cyclophosphamide-induced testicular toxicity and the protective agents. Expert Opin. Drug Met. 13, 525–536. Kosaka, T., Miyata, A., Ihara, H., Hara, S., Sugimoto, T., Takeda, O., Takahashi, E.I. and Tanabe, T. 1994. Characterization of the human gene (PTGS2) encoding prostaglandin-endoperoxide synthase 2. Eur. J. Biochem. 221, 889–897. Li, H., Jiang, Y. and Chen, F. 2004. Separation methods used for Scutellaria baicalensis active components. J. Chromatogr. B 812, 277–290. Liao, H., Ye, J., Gao, L. and Liu, Y. 2021. The main bioactive compounds of Scutellaria baicalensis Georgi. for alleviation of inflammatory cytokines: a comprehensive review. Biomed. Pharmacother. 133, 110917. Lin, J., Song, T., Li, C. and Mao, W. 2020. GSK-3beta in DNA repair, apoptosis, and resistance of chemotherapy, radiotherapy of cancer. Bba-Mol. Cell. Res. 1867, 118659. Mäkelä, J.A., Koskenniemi, J.J., Virtanen, H.E. and Toppari, J. 2019. Testis Development. Endocr. Rev. 40, 857–905. Mccoskey, M. and Vernon, N. 2024. Male reproductive endocrine disorders. Primary Care 51, 455–466. Mu, Y., Che, B., Tang, K., Zhang, W., Xu, S., Li, W., He, J., Liu, M., Chen, P., Zhong, S. and Li, G. 2023. Dendrobium officinale polysaccharides improved reproductive oxidative stress injury in male mice treated with cyclophosphamide. Environ. Sci. Pollut. Res. 30, 106431–106441. Oyagbemi, A.A., Omobowale, T.O., Saba, A.B., Adedara, I.A., Olowu, E.R., Akinrinde, A.S. and Dada, R.O. 2016. Gallic acid protects against cyclophosphamide-induced toxicity in testis and epididymis of rats. Andrologia 48, 393–401. Regueira, M., Gorga, A., Rindone, G.M., Pellizzari, E.H., Cigorraga, S., Galardo, M.N., Riera, M.F. and Meroni, S.B. 2018. Apoptotic germ cells regulate Sertoli cell lipid storage and fatty acid oxidation. Reproduction 156, 515–525. Sancho, M., Triguero, D., Lafuente-Sanchis, A. and Garcia-Pascual, A. 2017. Proliferation of Interstitial Cells in the Cyclophosphamide-Induced Cystitis and the Preventive Effect of Imatinib. Biomed. Res. Int. 2017, 3457093. Sarkar, A.A., Allyn, D.M., Delay, R.J. and Delay, E.R. 2021. Cyclophosphamide-Induced Inflammation of Taste Buds and Cytoprotection by Amifostine. Chem. Senses. 62(46),126574. Sui, J., Feng, Y., Li, H., Cao, R., Tian, W. and Jiang, Z. 2019. Baicalin protects mouse testis from injury induced by heat stress. J. Therm. Biol. 82, 63–69. Wang, R., Wang, C., Lu, L., Yuan, F. and He, F. 2024. Baicalin and baicalein in modulating tumor microenvironment for cancer treatment: a comprehensive review with future perspectives. Pharmacol. Res. 199, 107032. Wen, Y.F., Zhao, J.Q., Bhadauria, M. and Nirala, S.K. 2013. Baicalin prevents cadmium induced hepatic cytotoxicity, oxidative stress and histomorphometric alterations. Exp. Toxicol. Pathol. 65, 189–196. Xiao, F., Cheng, W.J., Yuan, G.X., Cheng, J.Q. and Liu, P.Y. 2024. Improving effect of selenium on spermatogenesis in mice with cyclophosphamide-induced spermatogenic impairment and its underlying mechanism. Zhonghua Nan Ke Xue 30, 291–299. Yin, B., Li, W., Qin, H., Yun, J. and Sun, X. 2021. The Use of Chinese Skullcap (Scutellaria baicalensis) and Its Extracts for Sustainable Animal Production. Animals-Basel 11, 1039. Zhou, L., Yi-Jian, X., Zhi-Chuan, Z., Yu-Ming, F., Hui, W., Wei-Qing, C., Xie, G., Jin-Zhao, M., Jun, J. and Bing, Y. 2025. Multi-omics analysis and experimental verification reveal testicular fatty acid metabolism disorder in non-obstructive azoospermia. Zool. Res. 46, 177–192. | ||
| How to Cite this Article |
| Pubmed Style Li J, Song C, Zhang X, Sun Y, Wang N, Shu X. The protective effect of baicalin against cyclophosphamideinduced testicular damage in rats. Open Vet. J.. 2025; 15(12): 6684-6696. doi:10.5455/OVJ.2025.v15.i12.49 Web Style Li J, Song C, Zhang X, Sun Y, Wang N, Shu X. The protective effect of baicalin against cyclophosphamideinduced testicular damage in rats. https://www.openveterinaryjournal.com/?mno=282359 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.49 AMA (American Medical Association) Style Li J, Song C, Zhang X, Sun Y, Wang N, Shu X. The protective effect of baicalin against cyclophosphamideinduced testicular damage in rats. Open Vet. J.. 2025; 15(12): 6684-6696. doi:10.5455/OVJ.2025.v15.i12.49 Vancouver/ICMJE Style Li J, Song C, Zhang X, Sun Y, Wang N, Shu X. The protective effect of baicalin against cyclophosphamideinduced testicular damage in rats. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6684-6696. doi:10.5455/OVJ.2025.v15.i12.49 Harvard Style Li, J., Song, . C., Zhang, . X., Sun, . Y., Wang, . N. & Shu, . X. (2025) The protective effect of baicalin against cyclophosphamideinduced testicular damage in rats. Open Vet. J., 15 (12), 6684-6696. doi:10.5455/OVJ.2025.v15.i12.49 Turabian Style Li, Jianqin, Chunlian Song, Xue Zhang, Yalong Sun, Nan Wang, and Xianghua Shu. 2025. The protective effect of baicalin against cyclophosphamideinduced testicular damage in rats. Open Veterinary Journal, 15 (12), 6684-6696. doi:10.5455/OVJ.2025.v15.i12.49 Chicago Style Li, Jianqin, Chunlian Song, Xue Zhang, Yalong Sun, Nan Wang, and Xianghua Shu. "The protective effect of baicalin against cyclophosphamideinduced testicular damage in rats." Open Veterinary Journal 15 (2025), 6684-6696. doi:10.5455/OVJ.2025.v15.i12.49 MLA (The Modern Language Association) Style Li, Jianqin, Chunlian Song, Xue Zhang, Yalong Sun, Nan Wang, and Xianghua Shu. "The protective effect of baicalin against cyclophosphamideinduced testicular damage in rats." Open Veterinary Journal 15.12 (2025), 6684-6696. Print. doi:10.5455/OVJ.2025.v15.i12.49 APA (American Psychological Association) Style Li, J., Song, . C., Zhang, . X., Sun, . Y., Wang, . N. & Shu, . X. (2025) The protective effect of baicalin against cyclophosphamideinduced testicular damage in rats. Open Veterinary Journal, 15 (12), 6684-6696. doi:10.5455/OVJ.2025.v15.i12.49 |