| Research Article | ||
Open Vet. J.. 2026; 16(1): 354-362 Open Veterinary Journal, (2026), Vol. 16(1): 354-362 Research Article Proteomic profile of seminal plasma in tropical Saanen bucks under heat stress using 1D-SDS PAGEErni Damayanti1*, Athhar Manabi Diansyah1, Ismah Ulfiyah Azis2, Fahrul Irawan2, Tulus Maulana3, Masturi Muhajir1, Muhammad Ihsan Andi Dagong1 and Nur Amalia Kartika11Department of Animal Production, Faculty of Animal Science, Hasanuddin University, Makassar, Indonesia 2Department of Animal Nutrition, Faculty of Animal Science, Hasanuddin University, Makassar, Indonesia 3Research Center for Applied Zoology, Research Organization for Life Sciences and Environment, National Research and innovation Agency, Bogor, Indonesia *Corresponding Author: Erni Damayanti. Department of Animal Production, Faculty of Animal Science, Submitted: 05/09/2025 Revised: 03/12/2025 Accepted: 17/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
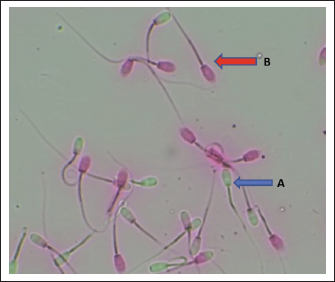
AbstractBackground: Tropical heat and humidity induce oxidative and thermal stress that threaten male fertility. However, the seminal plasma mechanisms that sustain sperm function under these conditions in goats remain poorly understood. Aim: This study aimed to evaluate semen quality and characterize the seminal-plasma proteome of Saanen bucks raised under Indonesia’s tropical conditions to identify molecular adaptations associated with fertility resilience. Methods: Three healthy bucks (five ejaculates each) were examined using computer-assisted sperm analysis (CASA) for sperm motility, viability, abnormality, membrane and acrosome integrity, and kinematics. Seminal-plasma proteins were separated by one-dimensional sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and annotated through Gene Ontology enrichment. Results: While most semen traits and kinematics were comparable among bucks, abnormality and acrosome integrity differed significantly (p < 0.05). A conserved extracellular, vesicle-associated proteome was identified—including BSP1, BSP3, BSP5, ANXA5, ALB, HSPA1A, HSP90AA1, CLU, GPX5, and SOD1—with minor inter-individual variation in LTF, FN1, SPP1, and A2M. Enrichment (FDR < 0.05) highlighted the extracellular region, vesicle, and cytokine-regulatory pathways. Conclusion: This study provides the first proteomic evidence of seminal plasma adaptation to THS in Saanen bucks, revealing a stable core of chaperone, antioxidant, and membrane-coating proteins that safeguard sperm function. These findings establish a novel proteomic basis for heat-resilient fertility and support the development of protein-based sire selection and reproductive management tools in tropical dairy-goat systems. Keywords: Sperm, Oxidative stress defense, Molecular chaperones, Saanen fertility biomarkers, Reproductive adaptability. IntroductionDairy goat production plays a central role in tropical livestock systems, contributing to food security in developing countries by supplying milk as a sustainable source of animal protein. The Saanen goat (Capra hircus), originally bred in Switzerland, has been established in Indonesia for many years and is widely used for dairy production under tropical conditions. Despite this long-term introduction, the fluctuation of semen quality driven by interindividual biological variation and chronic exposure to heat and humidity is a persistent challenge in our region. Under lowland tropical conditions, during hotter periods, bucks frequently exhibit week-to-week variability in semen quality. These instabilities reduce conception rate predictability and complicate AI scheduling and sire selection. In this context, seminal plasma, which is rich in proteins and metabolites that modulate sperm motility, survival, capacitation, and stress resilience, represents an underexplored source of information for understanding and mitigating heat-related declines in fertility (Teissier et al., 2024; Vashisht and Gahlay, 2024; Nalley et al., 2025). Several seminal plasma proteins have been identified as potential fertility markers in domestic livestock over the past two decades. Osteopontin and binder of sperm proteins (BSPs) are linked to sperm survival and capacitation in cattle and swine, whereas proAKAP4 has emerged as a validated biomarker of post-thaw motility and viability (Mukherjee et al., 2023; Peris-Frau et al., 2024; Dordas-Perpinyà et al., 2024). Similarly, proteomic studies in Bali cattle and buffalo have demonstrated correlations between protein expression profiles and fertility outcomes, underlining their potential as molecular indicators for sire selection (Iskandar et al., 2023; Diansyah et al., 2025a). Despite these advances, studies on goats remain scarce. Routine semen evaluation still relies predominantly on conventional measures such as motility, viability, and morphology, which, while useful, fail to capture the molecular determinants underlying reproductive competence. Climatic conditions add further complexity to this issue. Saanen goats reared in Indonesian lowlands are consistently exposed to tropical environments where the temperature–humidity index (THI) frequently exceeds 32, a threshold indicative of moderate to severe heat stress (Mamutse et al., 2023). In ruminants, heat stress impairs spermatogenesis, reduces motility, and compromises sperm membrane and acrosome integrity (Capela et al., 2022). Additionally, oxidative stress and disruptions in protein synthesis and folding are thought to alter the seminal plasma proteome, potentially compromising fertility (Udoekong et al., 2025). These findings demonstrate that the seminal plasma protein composition of tropical Saanen bucks may differ markedly from that reported in temperate regions, reflecting adaptive or stress-response mechanisms. Against this backdrop, we hypothesized that chronic exposure to tropical heat conditions promotes specific molecular adaptations in seminal plasma, resulting in a proteomic profile enriched with chaperones, antioxidants, and membrane-coating proteins. These proteins are expected to collectively buffer sperm cells against thermal oxidative stress and maintain the ability to fertilize. Variations in individual protein expression could serve as early molecular indicators of fertility potential. Thus, the findings of this study are intended to establish a scientific foundation for developing protein-based indices to enhance sire selection accuracy and improve reproductive management strategies in tropical dairy-goat production systems. This study aimed to characterize the seminal plasma protein profile of Saanen bucks raised under tropical Indonesian conditions and evaluate its association with semen quality parameters. This research sought to uncover molecular adaptations that enable sperm function and fertility resilience under chronic heat stress by identifying conserved and variable protein components. Materials and MethodsStudy sites and animal managementThe experiment was conducted at the Animal Reproduction Laboratory, Faculty of Animal Science, Hasanuddin University, Makassar, Indonesia, and protein analyses were performed at the Genomic Laboratory, National Research and Innovation Agency (BRIN), Cibinong, Indonesia. The facility is located in a tropical coastal climate (5.13° S, 119.49° E; altitude ~10 m above sea level). During the study, the ambient temperature in the goat barn ranged from 32°C to 34°C, with a relative humidity of 65%–85%, under a near-equatorial photoperiod (~12–12.5 h light/day). Bucks were housed in an open-sided, naturally ventilated barn with natural lighting and elevated slatted floors. Three clinically healthy Saanen bucks, aged approximately 24–36 months (live weight 40–45 kg), were maintained under intensive housing management in individual pens measuring 1.2 m × 1.5 m (1.8 m²) each, equipped with individual feeders and automatic drinkers. Bucks were institutional herd sires routinely collected for on-farm artificial insemination and teaching; animals were selected from the same herd under uniform management and were not preselected for superior semen quality. The diet consisted of 40% Pennisetum purpureum (Napier grass) and 60% concentrate, formulated according to the nutrient requirements of the NRC (2007). Feed was provided twice daily, and clean water was available ad libitum. Semen collection and evaluationSemen was collected twice weekly using an AV designed for small ruminants (liner length ~28 cm; inner diameter ~2.5 cm). Each buck was sampled repeatedly until five distinct batch codes were obtained per individual (total ejaculates analyzed: 15). This schedule provides representative sampling over time and minimizes batch-related variation. Ejaculates were immediately subjected to quality evaluation. Motility parameters were analyzed using a CASA (Program Vision Version 3.7.5 Minitube, Germany), which measured progressive and total motility and immotile spermatozoa, respectively. In addition, sperm kinematics, including curvilinear velocity (VCL), straight-line velocity (VSL), average path velocity (VAP), straightness (STR), linearity (LIN), and wobble (WOB) were assessed (Nirmala et al., 2025). Sperm viability was determined by eosin staining, where viable cells were excluded from the dye and nonviable cells appeared pink to red (Fig. 1) (Diansyah et al., 2025b). From the same slides, at least 200 spermatozoa were examined at 400× magnification to classify head, midpiece, and tail abnormalities. Membrane integrity was evaluated using the hypo-osmotic swelling (HOS) test, in which semen samples were incubated in a hypo-osmotic solution. Spermatozoa with intact membranes showed tail swelling or coiling, whereas those with damaged membranes remained straight (Diansyah et al., 2025c). Formol–saline staining was used to determine acrosome integrity, where spermatozoa with intact acrosomes showed uniform staining of the acrosomal cap, whereas those with absent or irregular staining were classified as acrosome-damaged (Diansyah et al., 2022).
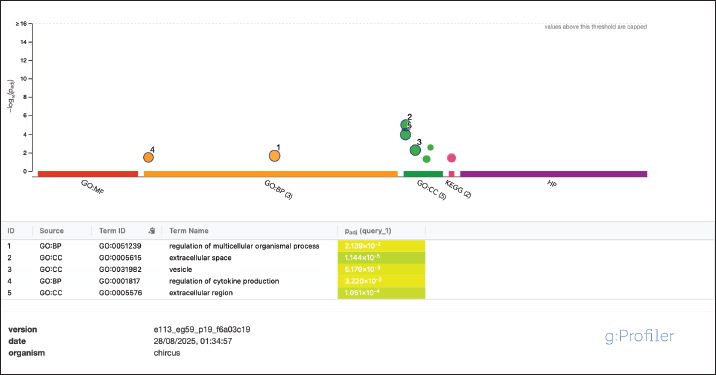
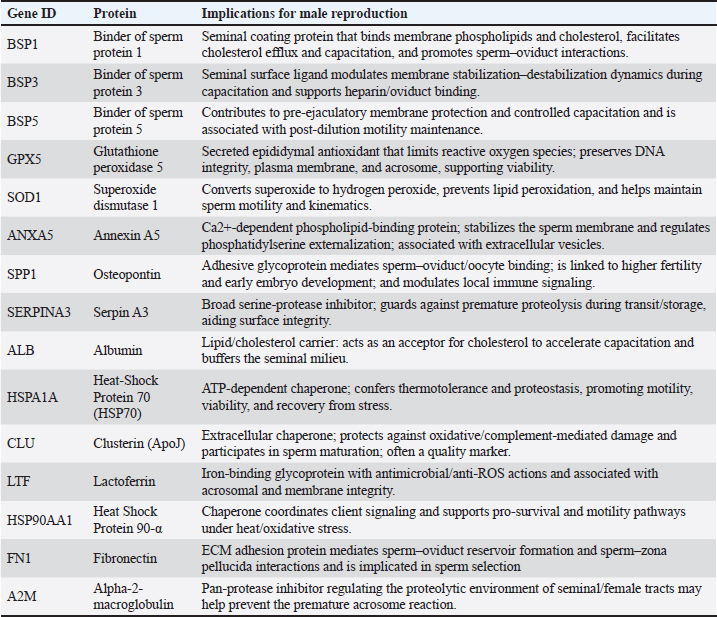
Fig. 1. Sperm viability: a) viable and b) non-viable spermatozoa. Seminal plasma protein profiling by 1D-SDS-PAGESeminal plasma was separated by centrifuging semen at 3,000 × g for 10 min at 4 °C, and protein concentrations were quantified using the Bradford assay with bovine serum albumin as the standard. For proteomic profiling, five ejaculates from each buck were pooled to generate one seminal-plasma sample per individual (three pools total) to reduce temporal variation within the animal. Each pooled sample was mixed with Laemmli buffer (1:1) and heated at 95 °C for 5 min, and 20 µg of total protein was loaded per lane. Protein separation was conducted using 1D-SDS-PAGE with a 12% resolving gel and 4% stacking gel at 60 V (stacking) and 20 mA (resolving) for ~3.5 hours. Gels were stained with Coomassie Brilliant Blue and destained until the bands were visible. A broad-range MW marker (ExcelBand 3-color PM2700, SMOBIO, Taiwan; 5–245 kDa) was included in every gel to enable MW estimation. Each pooled sample was run in duplicate as technical replicates, and two evaluators scored the band presence/absence and relative intensity independently; only matching observations were retained. Molecular-weight identification was inferred by matching band migration to the nearest marker bands using a ±5%–10% tolerance window, and proteins were therefore classified as putative identifications (Diansyah et al., 2023a). Semen collection was performed under natural tropical farm conditions (daily THI 78–84) and standardized to the morning period to minimize environmental variation that could influence seminal-plasma profiles. Bioinformatics and statistical analysesThe protein bands were annotated against the UniProt bovine protein database (http://www.uniprot.org) to assign putative identities based on molecular weights. Gene Ontology (GO) enrichment was performed to classify proteins into biological process, molecular function, and cellular component categories. To construct potential protein–protein interaction networks, pathway enrichment analyses were conducted using g:Profiler (https://biit.cs.ut.ee/gprofiler/gost). Semen quality data derived from multiple ejaculates (15 ejaculates per buck) were first summarized within the animal to avoid pseudoreplication. Batch-level measurements were averaged to generate a single animal-level value for statistical inference, and the buck was treated as the true experimental unit. Data normality was evaluated using the Shapiro–Wilk test, and variance homogeneity was assessed using Levene’s test. Differences among bucks were analyzed using one-way analysis of variance, with significance set at p < 0.05. The results are presented as mean ± SD, reflecting biological dispersion across animals. Band presence/absence and relative intensities were compared descriptively across the three pooled samples for SDS-PAGE. Ethical approvalThis study was conducted in accordance with animal welfare standards and approved by the Animal Ethics Committee of the Faculty of Animal Science, Universitas Hasanuddin (Approval No. 009/UN4.12/EC/V/2025) and conducted from May to August 2025 (≈16 weeks). ResultsSemen quality and kinematicsSperm abnormality differed among bucks (p < 0.05), with Buck 3 having higher sperm abnormality than Bucks 1 and 2 (see row superscripts). Acrosome integrity also differed (p < 0.01), with Buck 1 having lower sperm integrity than Bucks 2 and 3. In contrast, motility, viability, membrane integrity, and all kinematic parameters (VCL, VAP, VSL, LIN, STR, and WOB) were not significantly different between the two groups (all p > 0.05). Seminal plasma protein profilesA conserved core of seminal-plasma proteins was detected across all bucks, including BSP1, BSP3, GPX5, BSP5, SOD1, ANXA5, SERPINA3, ALB, HSPA1A, CLU, and HSP90AA1 (present in 3/3). Variation was confined to a small subset: LTF appeared only in Buck 2, SPP1 (100 kDa) was absent in Buck 3, FN1 (120 kDa) was exclusive to Buck 2, and A2M (150 kDa) was not detected in Buck 3. Consistent with this pattern, Buck 2 exhibited the most comprehensive repertoire, whereas Buck 3 exhibited the fewest detections. Functional enrichmentThe detected proteins were significantly enriched for extracellular space/region and vesicle-related components, with biological-process terms including regulation of multicellular organismal process and regulation of cytokine production. The mapped set predominantly comprised BSPs, ANXA5, SPP1, SERPINA3, ALB, CLU, LTF, FN1, A2M, HSPA1A, and HSP90AA1. Several detected proteins are classically linked to male fertility. SPP1 and FN1 support sperm adhesion within the female tract and engagement with the oviduct and the zona pellucida, which are processes associated with successful fertilization. The BSP family (BSP1, BSP3, and BSP5) and ANXA5 facilitate cholesterol efflux and controlled membrane remodeling that enable capacitation, whereas ALB supports this transition by acting as a cholesterol acceptor. These factors are associated with membrane stability, acrosomal competence, and effective sperm interactions with the female tract. In parallel, the profile also contains markers consistent with thermal and oxidative load typical of tropical conditions. HSPA1A and HSP90AA1 provide chaperone protection during heat and oxidative stress, with CLU contributing to extracellular chaperoning and damage control; SOD1 and GPX5 further limit reactive oxygen species to preserve motility and structural integrity. Immune and protease balance is reflected by LTF (antimicrobial, iron chelation), SERPINA3 (serine protease inhibition), and A2M (broad protease trapping), which together may curb premature acrosomal events and maintain a supportive seminal milieu. Taken together, the panel indicates a dual emphasis on fertility-supporting mechanisms and stress adaptation safeguards, which are features that are consistent with reproduction under tropical heat exposure. DiscussionThis study profiled the semen quality and seminal plasma proteins of Saanen bucks raised under tropical Indonesian conditions and interpreted the findings in the context of reproductive function. Most conventional semen traits and kinematic parameters were comparable among bucks, whereas differences in abnormality and acrosome integrity were observed, indicating that cellular integrity endpoints were more sensitive than motility metrics in this cohort. These outcomes support the idea that head and membrane structures in small ruminants capture subtle variation under field conditions (Diansyah et al., 2023a; Rahmat et al., 2024). Kinematic parameters such as VCL, VSL, and LIN, although not significantly different among bucks, remain critical reproductive success determinants because they influence sperm transport through the female tract and the probability of reaching and penetrating the oocyte. Although CASA-derived values did not differ significantly, the maintenance of motility and trajectory indices within normal ranges indicates that tropical Saanen bucks can sustain sperm function under heat load. Together with the conserved seminal plasma proteome, this stability indicates that molecular chaperones and antioxidants may buffer sperm motility and integrity under thermal stress, thereby maintaining reproductive potential. The protein profile revealed a conserved set that was detected in all bucks, including BSP1, BSP3, BSP5, GPX5, SOD1, ANXA5, SERPINA3, ALB, HSPA1A, CLU, and HSP90AA1. This repertoire is consistent with processes that support cholesterol efflux and controlled membrane remodeling for capacitation, together with antioxidant and chaperone defenses that sustain sperm structure and function (Diansyah et al., 2025a). Functional enrichment of the seminal-plasma protein in Saanen bucks presented in Table 3 and Figure 2. Functional enrichment highlighted extracellular region and extracellular space together with vesicle-related terms, which align with a secreted and vesicle-rich seminal milieu that delivers regulatory cargo to sperm and agrees with recent reports on seminal extracellular vesicles and fertility-associated proteins (Dlamini et al., 2025).
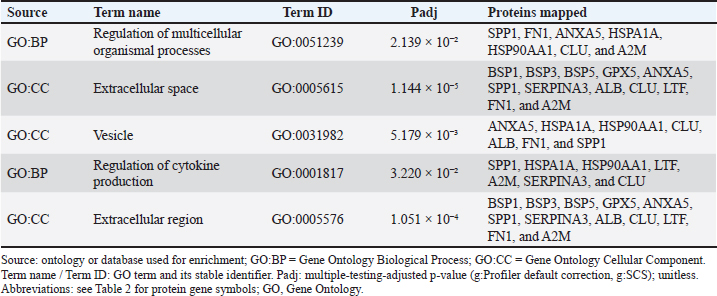
Fig. 2. Functional enrichment of the seminal-plasma protein in Saanen bucks. adjusted P values (Padj) are FDR-corrected (g:Profiler; species Capra hircus; default background). Table 3. Functional enrichment of the seminal-plasma protein in Saanen bucks.
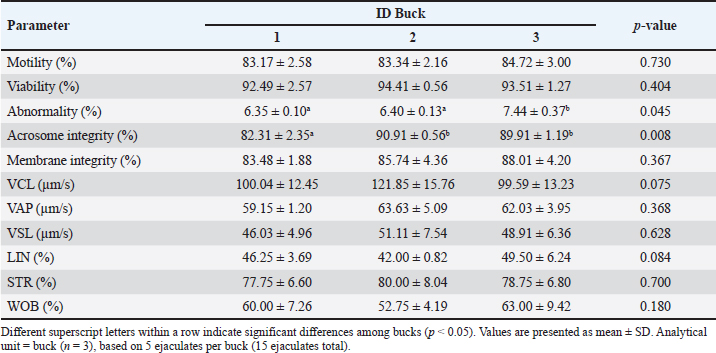
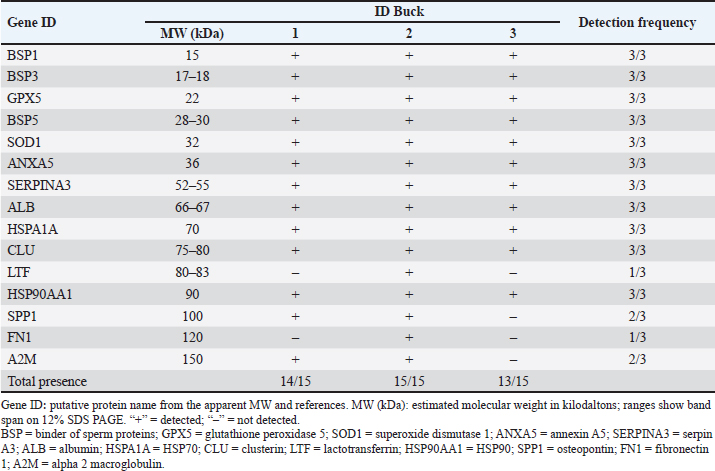
The variation across individuals was limited but biologically informative. LTF and FN1 at 120 kDa were detected only in Buck 2, whereas A2M and SPP1 at 100 kDa were not detected in Buck 3 (Table 2). LTF is linked to antimicrobial action and iron chelation with implications for membrane and acrosomal protection, FN1 contributes to adhesion within the female tract, and A2M modulates extracellular proteolysis that can influence premature acrosomal events (Verma, 2022). Although these associations are descriptive in a three-buck design, the pattern is coherent with the selective differences observed for abnormality and acrosome integrity in Table 1 and provides candidates for targeted validation. Table 1. Semen quality and kinematics of Saanen bucks.
Table 2. Seminal plasma protein bands detected using 1D-SDS-PAGE in Saanen bucks.
The tropical context helps explain the prominence of stress-buffering proteins. The consistent detection of HSPA1A, HSP90AA1, CLU, GPX5, and SOD1 under tropical heat conditions demonstrates molecular protection against persistent oxidative and thermal challenges typical of humid climates. These proteins collectively help preserve sperm motility, sperm viability, anti-stress mechanisms, membrane stability, and acrosomal competence despite heat stress in the environment. Chronic exposure to elevated temperature–humidity indices likely induces continuous cellular heat-shock responses, which stimulate the upregulation of these protective proteins to maintain sperm function and structural integrity under tropical heat stress. Simultaneously, BSP family members, together with ANXA5, ALB, SPP1, and FN1, facilitate capacitation and adhesion within the female tract—processes essential for maintaining fertility under tropical heat exposure (Diansyah et al., 2023a; Diansyah et al., 2025a). These results have clear implications for fertility management under tropical conditions from an applied perspective. Functional implications of seminal-plasma protein in Saanen bucks presented in Table 4. The conserved extracellular and vesicle-oriented core provides a molecular baseline for monitoring semen quality and selecting donors in tropical herds. The few variable proteins suggest candidate markers, such as SPP1, FN1, BSPs, HSP70, HSP90, CLU, GPX5, SOD1, and LTF, that could serve as a simple proteomic checklist alongside routine semen testing. Implementing such protein-informed screening may enhance sire selection accuracy, optimize semen preservation strategies, and improve fertility performance in heat-stressed environments. Such a panel could help flag ejaculates with stronger integrity and adhesion capacity and may inform cooling strategies and extender formulation during semen handling (Diansyah et al., 2023b; Diansyah et al., 2025a). Table 4. Functional implications of seminal-plasma protein in Saanen bucks.
Our results concur with recent tropical goat studies showing that, under sustained heat and humidity, motility indices often remain within functional ranges while structural endpoints—membrane and acrosomal integrity and morphology—are more labile across individuals. A year-round field study in Thailand documented season-linked fluctuations in semen quality and seminal fluid across a long dry period in a tropical lowland setting (Kimsakulvech et al., 2022). In Egypt, Baladi bucks exposed to a very high temperature–humidity index exhibited compromised testicular hemodynamics and semen traits, underscoring individual variability in heat resilience (El-Sherbiny et al., 2023). Furthermore, broader syntheses concluded that heat stress impairs male reproductive performance in goats despite their general thermotolerance (Danso et al., 2024). At the molecular level, integrative proteo-metabolomic analyses in goats show that management and handling choices alter proteomic pathways tied to oxidative phosphorylation and ROS balance, with downstream effects on membrane and acrosomal integrity (Li et al., 2024). Complementarily, emerging livestock literature highlights seminal extracellular vesicles as a vesicle-rich regulatory milieu for sperm function and a source of fertility biomarkers relevant to preservation and field performance (Martínez-Díaz et al., 2024; Rana et al., 2024). Finally, interventional work in dairy goats demonstrates that mitigating heat-driven oxidative and inflammatory damage can preserve spermatogenesis and semen quality, reinforcing our interpretation that stress-buffering mechanisms help sustain function under tropical heat loads (Guo et al., 2024). These interpretations should be viewed in light of several limitations: the study did not include a control group under thermoneutral conditions, the sample size was small with three bucks, which limits statistical power, protein identities were inferred from 1D SDS PAGE with emphasis on the presence or absence rather than quantitative abundance, repeated ejaculates were summarized within animals rather than modeled as repeated measures in a mixed model with buck as a random factor, and field fertility outcomes were not assessed. Future work should increase the cohort size, include explicit comparison groups under cooler or actively cooled conditions, and apply mixed-effects models with repeated sampling across weeks that incorporate buck as a random factor. Proteome characterization should employ LC MS MS for confident identification and quantification, followed by targeted validation of key proteins, such as BSPs, HSP70, HSP90, CLU, GPX5, SOD1, SPP1, FN1, and LTF, using western blot or ELISA. Correlation and regression analyses should link protein band intensities or LC MS MS abundances to semen quality metrics, including motility kinematics, morphology, membrane integrity, and acrosome status, and, where possible, conception rates, so that mechanistic associations can be tested and translated into selection and semen handling strategies for tropical herds. ConclusionOverall, the seminal plasma of Saanen bucks raised under tropical heat conditions is characterized by an extracellular, vesicle-oriented proteome that supports both capacitation-related membrane remodeling and thermal–oxidative stress protection, thereby sustaining sperm functional competence. The consistent presence of membrane-coating proteins, chaperones, and antioxidants provides a mechanistic basis for male fertility resilience in hot–humid environments. Moreover, the limited interindividual variation observed in specific proteins—such as BSPs, HSP70, HSP90, CLU, GPX5, SOD1, LTF, and FN1—represents promising fertility performance biomarker candidates. These findings highlight the practical potential of developing protein-based screening indices for sire selection, semen preservation, and reproductive management in tropical dairy-goat systems, contributing directly to improved fertility efficiency and herd productivity under heat-stress conditions. AcknowledgmentsThe authors gratefully acknowledge the Thematic Research Group of Local Livestock Genetic Resources for their valuable support and collaboration throughout this study. The authors also extend their sincere appreciation to all colleagues at the Faculty of Animal Science, Hasanuddin University, for their technical assistance, constructive input, and encouragement. FundingThis research was funded by Hasanuddin University through the 2025 PTN-BH Early Career Faculty Research Grant (PDPU) (Number 01260/UN-4.22/PT.01.03/2025) and the Local Livestock Genetic Resources Thematic Research Group. Author’s contributionsED, AMD, IUA, and FI conceived and designed the study. MM and NAK performed the analysis and data collection. TM and MIAD contributed data or analysis tools. All authors equally contributed to the study design and approved the final version of the manuscript. Conflict of interestThe authors declared that there is no conflict of interests. Data availabilityAll data supporting this study’s findings are available within the manuscript. ReferenceCandenas, L. and Chianese, R. 2020. Exosome composition and seminal plasma proteome: a promising source of biomarkers of male infertility. Int. J. Mol. Sci. 21(19), 7022. Capela, L., Leites, I., Romão, R., Lopes-Da-Costa, L. and Pereira, R.M.L.N. 2022. Impact of heat stress on bovine sperm quality and competence. Animals 12(8), 975. Danso, F., Iddrisu, L., Lungu, S.E., Zhou, G. and Ju, X. 2024. Effects of Heat Stress on Goat Production and Mitigating Strategies: a Review. Animals 14(12), 1793. Diansyah, A.M., Santoso, S., Herdis, H., Yusuf, M., Priyatno, T.P., Maulana, T., Toleng, A.L., Dagong, M.I.A., Said, S., Iskandar, H., Nurlatifah, A., Lestari, P., Affandy, L. and Baharun, A. 2025. Identification of reproductive performance in Bali-polled bulls using computer-assisted semen analysis and plasma seminal proteomics. Vet. World 18(1), 102. Diansyah, A.M., Yusuf, M., Rahmat, R. and Alfian, A.M. 2025. Enhanced sperm sexing efficiency and quality preservation in Bali bulls using freeze-dried albumin separation media. Open Vet. J. 15(6), 2416. Diansyah, A.M., Yusuf, M., Toleng, A.L. and Dagong, M.I.A. 2023a. The effect of thawing duration on the sperms quality of Bali polled bull. In Proceedings Of The 4th International Conference Of Animal Science And Technology (ICAST 2021), AIP Publishing. Makassar, Indonesia, 2628(1), pp 70–007. Diansyah, A.M., Yusuf, M., Toleng, A.L., Dagong, M.I.A., Maulana, T., Hasrin, H. and Baharun, A. 2023b. The sperms post-thawing quality and proteomic seminal plasma on fertility performance of Bali-Polled Bull. Adv. Anim. Vet. Sci. 11(4), 517–525; doi: 10.17582/journal.aavs/2023/11.4.517.525 Diansyah, A.M., Yusuf, M., Toleng, A.L., Dagong, M.I.A. and Maulana, T. 2022. The expression of plasma protein in Bali-polled bulls using 1D-SDS-PAGE. World’s. Vet. J. 12(3), 316–322. Dlamini, N.H., Bridi, A., Da Silveira, J.C. and Feugang, J.M. 2025. Unlocking gamete quality through extracellular vesicles: emerging perspectives. Biology 14(2), 198. Dordas-Perpinyà, M., Yánez-Ortiz, I., Sergeant, N., Mevel, V., Catalán, J., Bruyas, J.F., Miró, J. and Briand-Amirat, L. 2024. ProAKAP4 as indicator of long-lasting motility marker in post-thaw conditions in stallions. Animals 14(9), 1264. El-Sherbiny, H.R., Hashem, N.M. and Abdelnaby, E.A. 2023. Coat color affects the resilience against heat stress impacts on testicular hemodynamics, reproductive hormones, and semen quality in Baladi goats. BMC Vet. Res. 19, 107. Guo, X., Xu, J., Zhao, Y., Wang, J., Fu, T., Richard, M.L., Sokol, H., Wang, M., Li, Y., Liu, Y., Wang, H., Wang, C., Wang, X., He, H., Wang, Y., Ma, B. and Peng, S. 2024. Melatonin alleviates heat stress-induced spermatogenesis dysfunction in male dairy goats by regulating arachidonic acid metabolism mediated by remodeling the gut microbiota. Microbiome 12, 233. Iskandar, H., Andersson, G., Sonjaya, H., Arifiantini, R., Said, S., Hasbi, H., Maulana, T. and Baharun, A. 2023. Protein identification of seminal plasma in Bali bull (Bos javanicus). Animals 13(3), 514. Kimsakulvech, S., Nakthong, C., Lanamtiang, Y. and Wongtawan, T. 2022. The influence of a tropical climate with a long dry season on goat semen quality and seminal fluid change in the Kanchanaburi Province, Thailand. Trop. Anim. Sci. J. 45(3), 291–298. Li, C., Liang, J., Allai, L., Badaoui, B., Shao, Q., Ouyang, Y., Wu, G., Quan, G. and Lv, C. 2024. Integrating proteomics and metabolomics to evaluate impact of semen collection techniques on the quality and cryotolerance of goat semen. Scientific Rep. 14, 29489. Mamutse, J., Susanto, A., Purwantini, D., Sumaryadi, M.Y., Subagyo, Y. and Sodiq, A. 2023. Exploring the non-genetic factors that affect reproduction traits of Saanen Goats in Indonesia. J. Agr. Rur. Dev. Trop. Subtrop. 124(2), 129–135. Manabi Diansyah, A., Yusuf, M., Latief Toleng, A., Ihsan Andi Dagong, M., Maulana, T., Hasrin, H. and Baharun, A. 2023. The sperms post-thawing quality and proteomic seminal plasma on fertility performance of Bali-polled bull. Adv. Anim. Vet. Sci. 11(4), 517–525. Martínez-Díaz, P., Parra, A., Montesdeoca, M., Barranco, I. and Roca, J. 2024. Updating Research on Extracellular Vesicles of the Male Reproductive Tract in Farm Animals: a Systematic Review. Animals 14(21), 3135. Mukherjee, A., Gali, J., Kar, I., Datta, S., Roy, M., Acharya, A.P. and Patra, A.K. 2023. Candidate genes and proteins regulating bull semen quality: a review. Trop. Anim. Health. Prod. 55(3), 212. Nalley, W.M.M., Hine, T.M., Marawali, A., Makandolu, S.M., Kota, M.M.R., Diansyah, A.M., Rahmi, A. and Baharun, A. 2025. Identification of reproductive performance in Landrace boar: the sperm quality and molecular weight of protein. Adv. Anim. Vet. Sci. 13(2), 289–296. National Research Council (NRC). 2007. Nutrient requirements of small ruminants: sheep, goats, cervids, and new world camelids. Washington, DC: National Academies Press. Nirmala, A., Toleng, A.L., Yusuf, M., Herdis, H., Diansyah, A.M., Amrullah, M.F., Rahmat, R., Rajamuddin, R., Alfian, A.M. and Hasrin, H. 2025. Effect of adding kasumba turate (Carthamus tinctorius L.) extract in tris-egg yolk diluent on sperm quality in bali bulls. J. Anim. Health Prod. 13(1), 154–159. Diansyah, A.M., Nurlatifah, A., Damaris, C., Rahmat, R., Saili, T., Amrullah, M.F., Alfian, A.M., Adam, A.A.S., Kaiin, E.M. and Damaynti, E., 2025. Evaluation of sperm sexing technique using BSA column medium in Garut rams: enhancing sperm quality, kinematics, and fertility performance for improved breeding efficiency. Open. Vet. J.15(8), 3727. Peris-Frau, P., Soler, A.J., Iniesta-Cuerda, M., Martín-Maestro, A., Sánchez-Ajofrín, I., Medina-Chávez, D.A., Fernández-Santos, M.R., García-Álvarez, O., Maroto-Morales, A., Montoro, V. and Garde, J.J. 2020. Sperm cryodamage in ruminants: understanding the molecular changes induced by the cryopreservation process to optimize sperm quality. Int. J. Mol. Sci. 21(8), 2781. Raafi, M., Yusuf, M., Toleng, A.L. and Diansyah, A.M. 2021. Movement patterns of sperms at different bull breeds using computer-assisted sperm analysis (CASA). In IOP Conference Series: Earth and Environmental Science, IOP Publishing, Makassar, Indonesia, 788(1), pp 12–137. Rahmat, R., Yusuf, M., Toleng, A.L., Herdis, H., Diansyah, A.M. and Hasrin, H. 2024. The quality of Bali bull sexed sperms using dreeze dry albumin at different concentrations of sexing medium. Adv. Anim. Vet. 12(2), 249–258. Rana, S., Lone, F.A., Souza-Junior, J.B.F. and Bhat, G.R. 2024. The potential role of seminal extracellular vesicles as biomarkers of male fertility and sperm cryotolerance in livestock species. Discover. Appl. Sci. 6, 619. Teissier, M., Brito, L.F., Schenkel, F.S., Bruni, G., Fresi, P., Bapst, B., Robert-Granie, C. and Larroque, H. 2024. Genetic parameters for milk production and type traits in North American and European Alpine and Saanen dairy goat populations. JDS. Commun. 5(1), 28–32. Udoekong, E.C., Ramirez-Lopez, C.J., Silva Okano, D., Barros, E., Pereira Vidigal, P.M., Ribeiro, I.M., Rodrigues Carvalho, R.P., Machado-Neves, M., Guimarães, J.D. and Facioni Guimarães, S.E. 2025. Proteomic Alterations and Oxidative Stress in Seminal Plasma of Nellore Bulls Under Sexual Rest. Int. J. Mol. Sci. 26(6), 2457. Vashisht, A. and Gahlay, G.K. 2024. Understanding seminal plasma in male infertility: emerging markers and their implications. Andrology 12(5), 1058–1077. Verma, A. 2022. Impact of oxidative stress on bull sperm proteome and functional attributes. Thesis, National Dairy Research Institute, Karnal, India. | ||
| How to Cite this Article |
| Pubmed Style Damayanti E, Diansyah AM, Azis IU, Irawan F, Maulana T, Muhajir M, Dagong MIA, Kartika NA. Proteomic profile of seminal plasma in tropical Saanen bucks under heat stress using 1D-SDS PAGE. Open Vet. J.. 2026; 16(1): 354-362. doi:10.5455/OVJ.2026.v16.i1.32 Web Style Damayanti E, Diansyah AM, Azis IU, Irawan F, Maulana T, Muhajir M, Dagong MIA, Kartika NA. Proteomic profile of seminal plasma in tropical Saanen bucks under heat stress using 1D-SDS PAGE. https://www.openveterinaryjournal.com/?mno=282170 [Access: January 31, 2026]. doi:10.5455/OVJ.2026.v16.i1.32 AMA (American Medical Association) Style Damayanti E, Diansyah AM, Azis IU, Irawan F, Maulana T, Muhajir M, Dagong MIA, Kartika NA. Proteomic profile of seminal plasma in tropical Saanen bucks under heat stress using 1D-SDS PAGE. Open Vet. J.. 2026; 16(1): 354-362. doi:10.5455/OVJ.2026.v16.i1.32 Vancouver/ICMJE Style Damayanti E, Diansyah AM, Azis IU, Irawan F, Maulana T, Muhajir M, Dagong MIA, Kartika NA. Proteomic profile of seminal plasma in tropical Saanen bucks under heat stress using 1D-SDS PAGE. Open Vet. J.. (2026), [cited January 31, 2026]; 16(1): 354-362. doi:10.5455/OVJ.2026.v16.i1.32 Harvard Style Damayanti, E., Diansyah, . A. M., Azis, . I. U., Irawan, . F., Maulana, . T., Muhajir, . M., Dagong, . M. I. A. & Kartika, . N. A. (2026) Proteomic profile of seminal plasma in tropical Saanen bucks under heat stress using 1D-SDS PAGE. Open Vet. J., 16 (1), 354-362. doi:10.5455/OVJ.2026.v16.i1.32 Turabian Style Damayanti, Erni, Athhar Manabi Diansyah, Ismah Ulfiyah Azis, Fahrul Irawan, Tulus Maulana, Masturi Muhajir, Muhammad Ihsan Andi Dagong, and Nur Amalia Kartika. 2026. Proteomic profile of seminal plasma in tropical Saanen bucks under heat stress using 1D-SDS PAGE. Open Veterinary Journal, 16 (1), 354-362. doi:10.5455/OVJ.2026.v16.i1.32 Chicago Style Damayanti, Erni, Athhar Manabi Diansyah, Ismah Ulfiyah Azis, Fahrul Irawan, Tulus Maulana, Masturi Muhajir, Muhammad Ihsan Andi Dagong, and Nur Amalia Kartika. "Proteomic profile of seminal plasma in tropical Saanen bucks under heat stress using 1D-SDS PAGE." Open Veterinary Journal 16 (2026), 354-362. doi:10.5455/OVJ.2026.v16.i1.32 MLA (The Modern Language Association) Style Damayanti, Erni, Athhar Manabi Diansyah, Ismah Ulfiyah Azis, Fahrul Irawan, Tulus Maulana, Masturi Muhajir, Muhammad Ihsan Andi Dagong, and Nur Amalia Kartika. "Proteomic profile of seminal plasma in tropical Saanen bucks under heat stress using 1D-SDS PAGE." Open Veterinary Journal 16.1 (2026), 354-362. Print. doi:10.5455/OVJ.2026.v16.i1.32 APA (American Psychological Association) Style Damayanti, E., Diansyah, . A. M., Azis, . I. U., Irawan, . F., Maulana, . T., Muhajir, . M., Dagong, . M. I. A. & Kartika, . N. A. (2026) Proteomic profile of seminal plasma in tropical Saanen bucks under heat stress using 1D-SDS PAGE. Open Veterinary Journal, 16 (1), 354-362. doi:10.5455/OVJ.2026.v16.i1.32 |