| Research Article | ||
Open Vet. J.. 2025; 15(12): 6670-6683 Open Veterinary Journal, (2025), Vol. 15(12): 6670-6683 Research Article Integrative analysis of semen quality and metabolomic profiles reveals breed-specific biomarker fertility in Indonesian buffaloSigit Bintara1*, Kurniawan Dwi Prihantoko1, Aeni Nurlatifah1, Tulus Maulana2, Athhar Manabi Diansyah3, Sitti Nurhaliza4, Rahmat Rahmat5 and Muhammad Fajar Amrullah61Faculty of Animal Science, Gadjah Mada University, Depok, Yogyakarta, Indonesia 2Research Center for Applied Zoology, National Research and Innovation Agency, Cibinong Science Center, Bogor, Indonesia 3Faculty of Animal Science, Hasanuddin University , South Sulawesi, Indonesia 4Faculty of Mathematics and Natural Sciences, Tadulako University, Palu, Indonesia. 5Faculty of Agriculture, Lambung Mangkurat University, Banjarbaru, Indonesia 6Doctoral Program of Animal Biomedical Science, School of Veterinary Medicine and Biomedical Sciences, IPB University, Bogor, Indonesia *Corresponding Author: Sigit Bintara, Faculty of Animal Science, Gadjah Mada University, Depok, Yogyakarta, Indonesia. Email: sigitbintara [at] ugm.ac.id Submitted: 05/09/2025 Revised: 28/10/2025 Accepted: 03/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
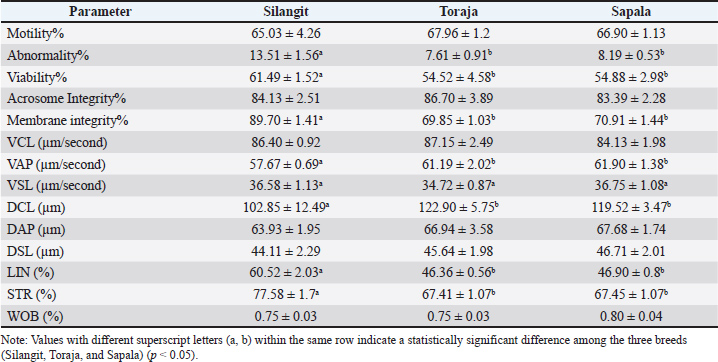
AbstractBackground: The increasing demand for animal protein in Indonesia underscores the importance of improving the productivity of indigenous buffalo breeds, which remain underutilized. Semen quality plays a central role in the success of artificial insemination, and metabolomic profiling provides opportunities to uncover the molecular mechanisms underlying fertility. Aim: This study aimed to investigate the association between post-thawed sperm quality and metabolomic signatures of three Indonesian buffalo breeds (Silangit, Toraja, and Sapala) to identify breed-specific biomarkers associated with fertility potential. Methods: Semen samples were collected from healthy bulls (5–8 years old, 450–750 kg) maintained under standardized management conditions. Using computer-assisted sperm analysis, post-thawed semen quality was evaluated for motility, viability, morph abnormality, acrosome and membrane integrity, and kinematic parameters. Metabolomic profiling was performed using high-resolution liquid chromatography-high-resolution mass spectrometry, and data were analyzed using multivariate statistics, pathway enrichment, and receiver operating characteristic (ROC) curve analysis for biomarker evaluation. Results: Significant breed-dependent differences in semen quality and metabolomic composition were observed. The Silangit bulls showed higher sperm viability and membrane integrity, whereas the Toraja and Sapala bulls exhibited lower morph abnormality rates. Distinct discriminative metabolites were identified across breeds: oxalacetic acid and adenosine in Silangit, dimethylethanolamine and uridine in Toraja, and glutamic acid with succinic acid semialdehyde in Sapala. Pathway enrichment analysis revealed energy metabolism predominance in Silangit, membrane remodeling and DNA stability in Toraja, and antioxidative mechanisms in Sapala. Receiver operating characteristic analysis confirmed the diagnostic potential of these metabolites as candidate biomarkers. Conclusion: Semen quality and metabolomic data reveal breed-specific adaptations to cryopreservation in Indonesian buffalo, guiding fertility prediction, improving cryopreservation, and selective breeding to sustain indigenous genetic resources. Keywords: Cryopreservation, Fertility prediction, Indigenous genetic resources, Seminal plasma, Metabolomic profiles. IntroductionThe global demand for animal protein is projected to increase by 60%–70% by 2050, increasing the pressure on food systems (Makkar, 2018; Henchion et al., 2021). Indonesia’s reliance on cattle, poultry, and fish creates a supply–demand gap that motivates diversification (Anindita et al., 2020; Said 2020; Sari et al., 2021). The indigenous buffaloes, such as Toraja, Sapala, and Silangit, remain underutilized despite local adaptation (resilience, disease tolerance, and marginal-forage use) and potential to contribute to the national protein supply (Suhardi et al., 2022; Tsuji et al., 2022; Azwar et al., 2024). These considerations underscore the urgent need to optimize productivity, particularly reproductive efficiency, in buffalo populations from Toraja, South Kalimantan, and Sumatra (Prihandini et al., 2023). This targeted improvement will diversify protein sources and strengthen national food security through a resilient, sustainable domestic supply. Reproductive efficiency, especially semen quality, underpins AI success (Zuidema et al., 2021). However, local buffalo breeds show variable semen quality and remain understudied (Chen and Huang, 2024). Metabolomics can elucidate pathways underlying semen quality and fertility, offering mechanistic insight (Talluri et al., 2022; Mateo-Otero, 2024). However, integrated studies linking buffalo semen quality with metabolomic profiles are scarce, and implications for food security strategies remain unclear (Martínez et al., 2025). This gap highlights the need for more focused research to optimize reproductive technologies in buffalo, which could have important impacts on livestock productivity and sustainability. Enhancing buffalo productivity can expand animal-protein supply, reduce reliance on a narrow set of species, and strengthen food security (Prihandini et al., 2023). This study aimed to characterize post-thaw sperm quality and metabolomic profiles in local Indonesian buffaloes and identify candidate metabolites correlated with semen quality indices of cryotolerance as hypothesis-generating biomarkers for future validation against fertility outcomes. Materials and MethodsAnimal sources and study locationsThis study was conducted during the dry season between June and August 2025. This study involved three local Indonesian buffalo (Bubalus bubalis) breeds, namely, Toraja buffalo (South Sulawesi), Sapala buffalo, and Silangit buffalo (North Sumatra), with n=3 bulls per breed (total n=9). All bulls were 5–8 years old and weighed between 450 and 750 kg. Animals were maintained under standardized management conditions and monitored by veterinarians to ensure their health status. Feed management was standardized across sites (forage-based ration plus concentrate, mineral–vitamin supplementation, and water ad libitum). The experimental animals were kept at the following institutions: the Technical Implementation Unit for Artificial Insemination and Semen Production, South Sulawesi Provincial Livestock and Animal Health Service (−5.15223, 119.67831); the Artificial Insemination Center, South Kalimantan Provincial Plantation and Livestock Service (−3.44472, 114.81590); and the Siborongborong Superior Livestock Breeding and Forage Center, North Tapanuli Regency, North Sumatra (2.26654, 99.002533). Semen was collected once per week per bull using an artificial vagina (38°C–42°C) under uniform hygiene and handling. The same commercial extender (Andromed®, Minitube, Germany) was used for all ejaculates/breeds. For cryopreservation, the diluted semen was cooled gradually to 5°C, equilibrated for 2–4 hours, loaded into 0.25 ml straws, frozen in liquid nitrogen vapor (~3–4 cm above the surface) for 10–12 minutes, and then plunged into liquid nitrogen (−196°C) for storage (Said et al., 2025). For metabolomics, five straws per batch from each bull in each breed were pooled and analyzed, as specified in the LC–MS/MS section. Assessment of post-thawed sperm qualityThe frozen semen collected from the three local buffalo breeds was then examined for quality after thawing. The thawing method followed the protocol of Fitriana et al. (2025). Frozen semen straws were removed from the storage container containing liquid nitrogen (−196°C) and thawed at 37°C for 30 seconds. The semen straws were then transferred to microtubes (1.5 ml), and 10 μl was dispensed onto a cover-glass slide for sperm quality assessment. The observed sperm quality parameters were motility (%), morph-abnormality (%), viability (%), acrosome integrity (%), and membrane integrity (%). Sperm viability (%) and morph abnormality (%) were assessed according to the modified method of Alfian et al., (2025). Sperm morphological defects are classified into three groups: head defects, neck and mid-piece defects, and tail defects (Surahman et al., 2021; Diansyah et al., 2023). Membrane integrity was assessed using the hypo-osmotic swelling test. Curled or swollen tails indicate sperm cells with intact plasma membranes, while a straight tail indicates damaged plasma membranes (Diansyah et al., 2025b). The integrity of the spermatozoa acrosome was evaluated, as indicated by the tip of the spermatozoa head, which is thick and black when the semen is exposed to a physiological NaCl solution containing 1% formalin (Nirmala et al., 2025). Assessment of frozen sperm kinematicsThe kinematic assessment of frozen semen sperm was performed using computer-assisted sperm analysis (Vision Version 3.7.5 program Minitube, Germany) following the protocol of Diansyah et al. (2025a). The parameters analyzed were motility, distance curve linear (DCL), distance average path (DAP), distance straight line (DSL), velocity curve linear (VCL), velocity average path (VAP), velocity straight line (VSL), linearity (LIN), straightness (STR), and wobble (WOB) (Maulana et al., 2022). Preparation of metabolomics samplesThe frozen semen samples were removed from the −20°C refrigerator and thawed at 4°C. One hundred microliters of each sample was thoroughly mixed with 400 μl of cold methanol/acetonitrile (1:1, v/v) solution, and then the mixture was followed by centrifugation (14,000 × g, 20 minutes, 4°C) to remove proteins. The supernatant was transferred to new tubes and dried in a vacuum centrifuge. The dried samples were fully dissolved in 100 μl of acetonitrile/water (1:1, v/v) solvent, followed by centrifugation (14,000 × g, 15 minutes, 4°C). The resulting supernatant was filtered 0.2 um and transferred to liquid chromatography glass vials. Metabolomics analysisRobust metabolomics sample preparation is paramount to ensure compatibility with the sensitive downstream liquid chromatography–high-resolution mass spectrometry analysis, as the samples, whether biofluids (e.g., plasma, serum, urine) or tissue/cell extracts, typically undergo a series of critical steps. Initial protein precipitation is essential and is commonly achieved using ice-cold organic solvents such as methanol or acetonitrile, often in combination with centrifugation (e.g., 14,000–16,000 × g, 15–20 minutes, 4°C) to remove proteins and other macromolecules that could foul the ultrahigh-performance liquid chromatography system or suppress ionization. The resulting supernatant must then be carefully handled to avoid metabolite degradation or loss. Crucially, the supernatant is often filtered using 0.1 µm or 0.22 µm pore size filters compatible with organic solvents (e.g., PTFE, nylon) to remove any residual particulates that could clog the intricate fluidics of the Vanquish Utra-High-Performance Liquid Chromatography. Following filtration, samples frequently require concentration or reconstitution to ensure that metabolites are within the Orbitrap Exploris 240 HRMS’s optimal detection range. The final reconstitution solvent must be carefully chosen to be compatible with the starting mobile phase conditions (2% B, aqueous with 0.1% formic acid). This minimizes injection-related band broadening and ensures effective metabolite focus at the column head upon injecting the 5 µl sample volume. Maintaining sample integrity throughout this process is vital for generating data that accurately reflects the true metabolic state. Finally, samples are typically stored at −80°C until analysis and transferred to the UHPLC autosampler, which is kept at a cool temperature (e.g., 4°C–10°C) to maintain stability during the analytical run. Statistical analysis and bioinformaticsData on semen quality and sperm kinematics were analyzed using SPSS v20.0 (IBM, Armonk, NY, USA). The normality and homogeneity of variance were tested using the Shapiro–Wilk and Levene’s tests. Differences between X- and Y-bearing spermatozoa were determined using independent-samples t-tests, reported as mean ± SD. All tests were two-tailed at α=0.05. The peak-intensity matrix from Compound Discoverer 3.3 was processed in MetaboAnalyst 6.0 (www.metaboanalyst.ca) for metabolomics. Intensities were transformed into log2(× + 1) and autodialed. Excessive missing features were removed; remaining gaps were imputed at half the minimum positive value. The unsupervised Principal Component Analysis (PCA) explored global variation. Supervised Partial Least Squares Discriminant Analysis assessed class separation; k-fold cross-validation and permutation testing were used for model reliability (MetaboAnalyst defaults). The VIP scores were calculated, with VIP > 1 deemed discriminatory. Univariate metabolite comparisons were performed using two-tailed t-tests with Benjamini–Hochberg FDR adjustment where applicable. Hierarchical clustering heatmaps were generated from log10 data using the Euclidean distance and Ward’s linkage. Pathway enrichment and topology analyses were performed using KEGG/HMDB sets (hypergeometric enrichment; relative-betweenness centrality), with pathways significant at FDR-adjusted p < 0.05. Biomarker performance was evaluated by receiver operating characteristic (ROC) analysis with Monte Carlo cross-validation (balanced subsampling), reporting AUC. Ethical approvalThis study was approved by the Research Ethics Commission of the National Research and Innovation Agency with the number: 093/KE.02/SK/05/2023, issued on May 12, 2023. ResultsSemen quality and kinematics of post-thawed spermSignificant breed effects were observed in several sperm quality traits (Table 1). The Silangit buffalo exhibited a higher proportion of abnormal spermatozoa (p < 0.05), whereas the Toraja and Sapala showed lower morph abnormality rates. The viability (p < 0.05) and membrane integrity (p < 0.05) were also significantly higher in Silangit than in the other two breeds. In contrast, motility and acrosome integrity did not differ significantly among the different breeds. For sperm kinematics, VAP (p < 0.05) and DCL (p < 0.05) differed significantly among breeds, whereas no statistical differences were observed in VCL, VSL, DAP, DSL, and WOB. The derived parameters revealed that LIN (p < 0.05) and STR (p < 0.05) were significantly higher in Silangit than in Toraja and Sapala. Table 1. Post-thawed sperm quality and kinematic parameters of Indonesian buffalo.
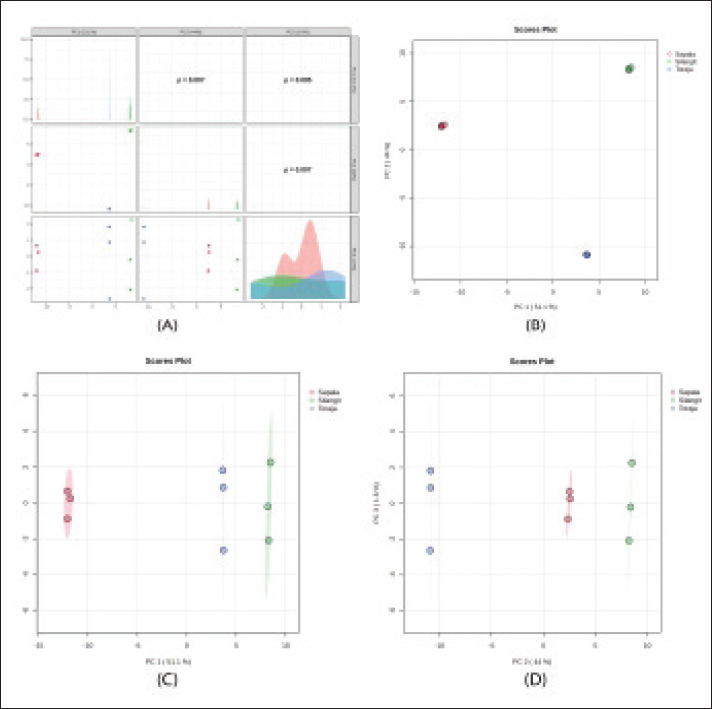
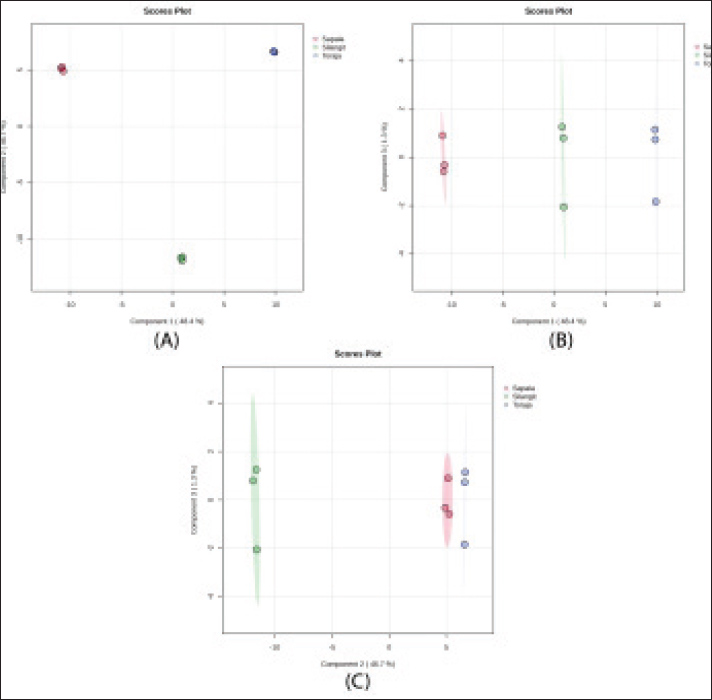
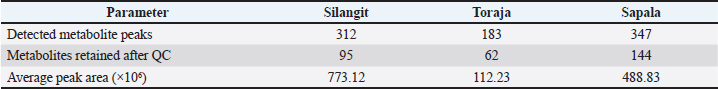
Metabolomic profiling of the spermThe spermatozoa metabolomic profiling revealed clear differences among the three buffalo breeds (Table 2). Kapala showed the highest number of detected metabolite peaks, whereas Toraja had the lowest. After quality control filtering (HMDB ID), the number of retained metabolites also varied markedly across breeds, with Sapala retaining the largest set. The Silangit breed had the highest average peak area, indicating greater overall metabolite abundance than the other breeds. Multivariate analysis was performed to explore and compare sperm metabolomic profiles among buffalo breeds (Fig. 1). PCA revealed a distinct grouping of samples, with minimal overlap between breeds. PLS-DA was performed to evaluate class separation among buffalo breeds based on sperm metabolomic profiles (Fig. 2). The score plots demonstrated clear clustering of samples within each breed, with distinct separation observed among the Silangit, Toraja, and Sapala groups. Non-overlapping clusters indicated consistent differences in metabolomic composition across breeds.
Fig. 1. PCA of sperm metabolomic profiles in Indonesian buffalo breeds. (A) Pairwise metabolite distribution plots, (B) PCA 1 versus. PCA 2, (C) PCA 1 versus. PCA 3, and (D) PCA 2 versus. PCA 3.
Fig. 2. PLS-DA analysis of sperm metabolomic profiles in Indonesian buffalo breeds. (A) Component 1 versus. Component 2, (B) Component 1 versus. Component 3, and (C) Component 2 versus. Component 3. Table 2. Summary of detected and quality-controlled sperm metabolite features in Indonesian buffalo.
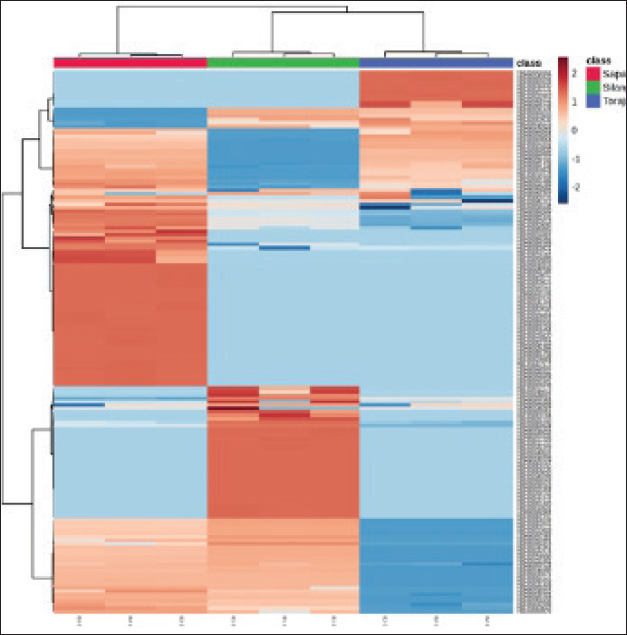
Differential metabolites in the spermHierarchical clustering heatmap analysis showed that sperm metabolomic profiles were distinctly different among Silangit, Toraja, and Sapala buffalo breeds (Fig. 3). The clustering pattern reflected clear breed-specific metabolite abundance, with groups of metabolites displaying higher or lower intensities in one breed compared with the others.
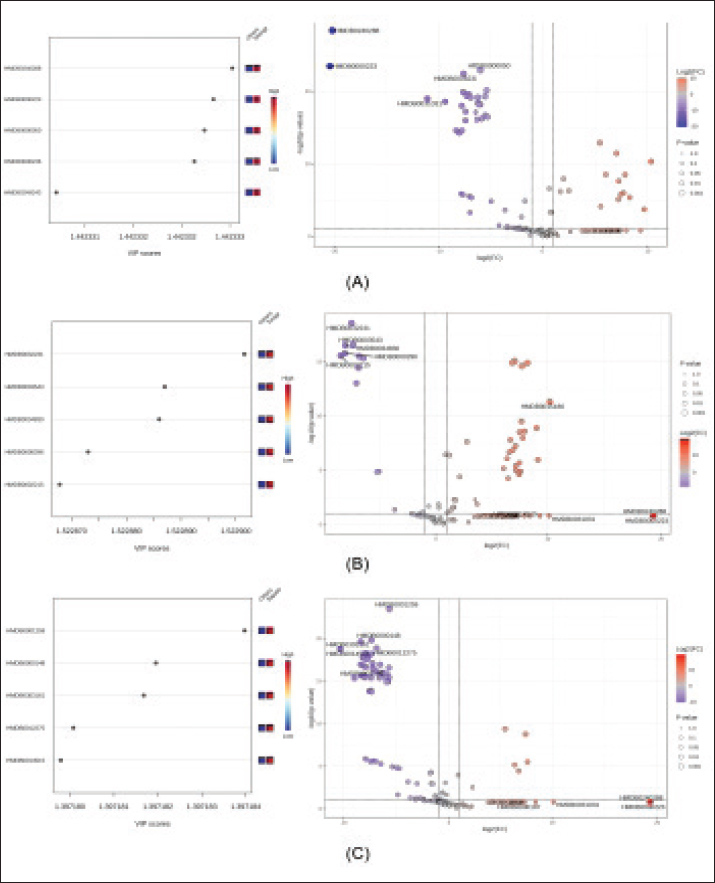
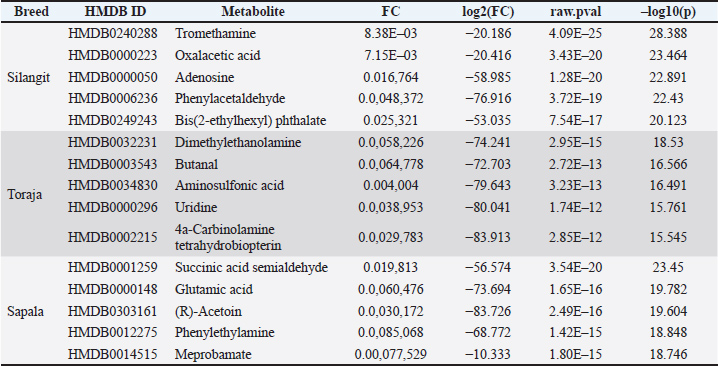
Fig. 3. Hierarchical clustering heatmap of discriminative sperm metabolites in Indonesian buffalo. The top five discriminative metabolites were identified for each breed based on VIP scores and volcano plot analysis (Fig. 4). The key differential metabolites in Silangit were HMDB0240288, HMDB0000223, HMDB0000050, HMDB0006236, and HMDB0249243. Toraja spermatozoa were characterized by HMDB0032231, HMDB0003543, HMDB0034830, HMDB0000296, and HMDB0002215, while Sapala exhibited HMDB0001259, HMDB0000148, HMDB0303161, HMDB0012275, and HMDB0014515. As shown in Table 3, the metabolomic analysis identified distinct sets of the top five differential metabolites across the three buffalo breeds. In Silangit, the most discriminative metabolites were tromethamine, oxalacetic acid, adenosine, phenylacetaldehyde, and bis(2-ethylhexyl) phthalate, all of which showed highly significant differences (p < 1.0 × 10−16). The top metabolites in Toraja were dimethylethanolamine, butanal, aminosulfonic acid, uridine, and 4a-carbinolamine tetrahydrobiopterin, each presenting strong fold changes and statistical significance (p < 1.0 × 10−12). Meanwhile, Sapala was characterized by succinic acid semialdehyde, glutamic acid, (R)-acetoin, phenylethylamine, and meprobamate as the most significantly altered metabolites (p < 1.0 × 10−15). These metabolite profiles highlight breed-specific biochemical signatures, suggesting fundamental differences in sperm metabolic regulation among Silangit, Toraja, and Sapala buffaloes.
Fig. 4. Top 5 differential sperm metabolites in Indonesian buffalo breeds based on Variable Importance in Projection scores and volcano plots. (A) Silangit, (B) Toraja, and (C) Sapala. Table 3. Top 5 differential sperm metabolites in Indonesian buffalo breeds.
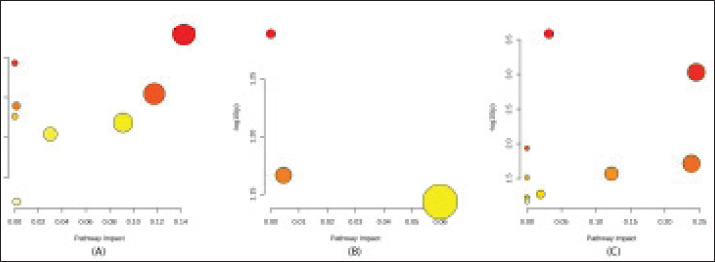
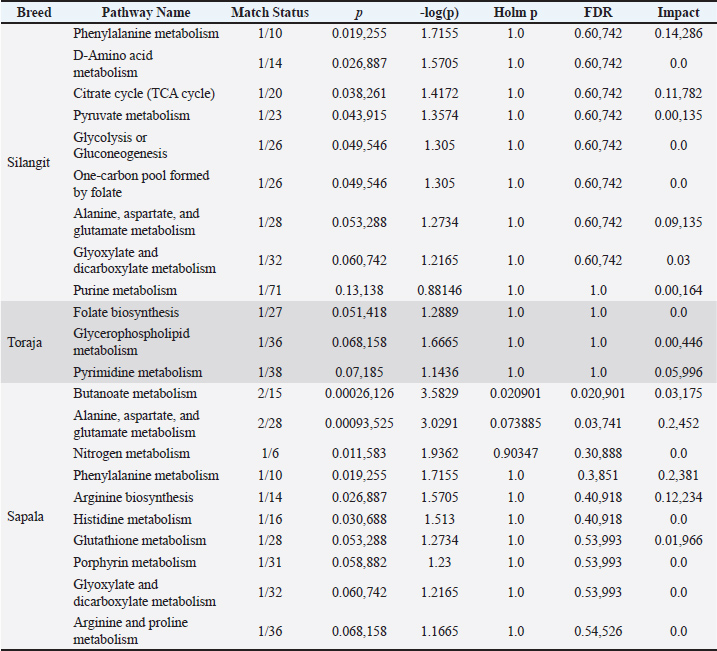
Pathway enrichment (Sperm metabolome)Pathway enrichment analysis revealed distinct metabolic pathways associated with sperm metabolites across the three buffalo breeds (Fig. 5, Table 4). In Silangit, several energy and amino acid–related pathways were highlighted, including phenylalanine metabolism, the citrate cycle (TCA cycle), pyruvate metabolism, and glycolysis/gluconeogenesis, alongside one-carbon pool by folate and alanine, aspartate, and glutamate metabolism. In contrast, Toraja showed enrichment in fewer pathways, with the main contributors being folate biosynthesis, glycerophospholipid metabolism, and pyrimidine metabolism. Kapala demonstrated a broader set of significantly enriched pathways, particularly butanoate, alanine/aspartate/glutamate, nitrogen, phenylalanine, arginine, histidine, and glutathione metabolisms. These results indicate that each breed is characterized by distinct metabolic signatures at the pathway level.
Fig. 5. Pathway enrichment analysis of differential sperm metabolites in Indonesian buffalo breeds: Silangit (A), Toraja (B), and Sapala (C). Table 4. Pathway enrichment analysis of differentially expressed sperm metabolites in Indonesian buffalo.
Evaluation of biomarker candidatesReceiver operating characteristic curve analysis was conducted to assess the discriminatory ability of the top differential sperm metabolites as biomarker candidates in each buffalo breed (Fig. 6). In Silangit (Panel A), HMDB0000590, HMDB0000223, HMDB0006236, HMDB0240288, and HMDB0249243 were identified as strong candidates. The biomarker candidates for Toraja (Panel B) included HMDB0000296, HMDB0002215, HMDB0003543, HMDB0032231, and HMDB0034830. In Sapala (Panel C), the ROC curves of HMDB0000548, HMDB0001259, HMDB0012275, HMDB0014515, and HMDB0033261 showed good classification potential. These findings suggest that distinct sets of metabolites may serve as breed-specific biomarker candidates for sperm metabolomic profiling.
Fig. 6. ROC curve analysis of the top differential sperm metabolites in Indonesian buffalo breeds. (A) Silangit, (B) Toraja, and (C) Sapala. DiscussionThis study revealed clear breed-dependent variations in post-thawed sperm quality and metabolomic composition among Silangit, Toraja, and Sapala buffaloes. These phenotypic and biochemical differences reinforce the concept that sperm freezability and fertility potential are not uniform traits but are shaped by intrinsic breed-specific factors, including membrane composition, mitochondrial activity, and Reduction-Oidation Reactions balance. Previous reports in cattle and buffalo have emphasized that the degree of lipid unsaturation, antioxidant reserves, and energy metabolic capacity influence sperm tolerance to cryopreservation (Inyawilert et al., 2021; Nain et al., 2023). Our findings that Silangit sperm exhibited superior viability and membrane integrity, while Toraja and Sapala sperm showed lower morph-abnormality rates, suggest that each breed may employ distinct molecular strategies to withstand cryo-induced stress, thereby justifying the integration of metabolomic analysis to delineate these mechanisms. Metabolomic profiling identified five discriminative metabolites per breed, highlighting biochemical pathways that are potentially linked to sperm function (Said et al., 2025). In Silangit, tromethamine, oxalacetic acid, adenosine, phenylacetaldehyde, and bis(2-ethylhexyl) phthalate levels were significantly altered. Oxalacetic acid, a central TCA intermediate, is pivotal for sustaining ATP production in spermatozoa, which directly fuels dynein ATPases for flagellar motion (Du Plessis et al., 2015; Sengupta et al., 2020). Adenosine has been implicated in purinergic signaling, capacitation regulation, acrosome exocytosis, and sperm hyperactivation via cAMP-mediated pathways (Wertheimer et al., 2013; Bellezza and Minelli 2017). The presence of phenylacetaldehyde may be linked to aromatic amino acid metabolism, which influences REDOX balance and sperm lifespan (Akbarinejad et al., 2018; Saad et al., 2019). Interestingly, bis(2-ethylhexyl) phthalate, an environmental contaminant, has been consistently associated with oxidative damage and impaired semen quality in livestock and humans (Sedha et al., 2015; Xuexia et al., 2023), raising the possibility of exogenous influences on Silangit sperm metabolome. Toraja sperm were characterized using dimethylethanolamine, butanal, aminosulfonic acid, uridine, and 4a-carbinolamine tetrahydrobiopterin. Dimethylethanolamine is an essential precursor of phosphatidylcholine, a key determinant of sperm membrane fluidity and coresistance (Castro et al., 2025). Uridine supports nucleotide metabolism and RNA stability, both of which are crucial for chromatin integrity during fertilization (Sharma et al., 2016). Kapala sperm displayed glutamic acid, succinic acid semialdehyde, (R)-acetoin, phenylethylamine, and meprobamate. Glutamic acid is a major excitatory amino acid that is critical for nitrogen balance and mitochondrial metabolism and has been positively associated with sperm motility and fertilization capacity in bovine and human studies (Velho et al., 2018). Succinic acid semialdehyde reflects Gamma-aminobutyric acid shunt activity, which has been implicated in sperm hyperactivation regulation (Menezes et al., 2019). The identification of phenylethylamine, a biogenic amine, points to potential neuromodulatory influences on sperm physiology, which requires further validation. Pathway enrichment analysis provided additional mechanistic insights into breed-specific metabolic programming. In Silangit, the enrichment of glycolysis, pyruvate metabolism, and the TCA cycle highlights a strong reliance on energy metabolism to maintain motility, consistent with reports in bull sperm linking Adenosine triphosphate production to linear velocity and motility persistence (Abruzzese et al., 2024). Phenylalanine metabolism, which is also enriched in Silangit, may be associated with antioxidant defense because aromatic amino acids contribute to REDOX buffering (Egbujor et al., 2024). In contrast, Toraja showed enrichment in folate biosynthesis, glycerophospholipid metabolism, and pyrimidine metabolism. Folate pathways are essential for nucleotide synthesis and methylation reactions, which safeguard sperm DNA integrity (Socha et al., 2024). Glycerophospholipid metabolism contributes to membrane fluidity, a trait fundamental for surviving freeze–thaw stress (Su et al., 2025). Kapala demonstrated the enrichment of butanoate metabolism, glutathione metabolism, and arginine biosynthesis. These pathways are strongly linked to REDOX regulation and ROS scavenging: glutathione metabolism is a central antioxidant pathway that prevents sperm membrane lipid peroxidation (Ribeiro et al., 2022), whereas arginine serves as a substrate for nitric oxide, which modulates motility and capacitation (Maciel et al., 2018). Thus, while Silangit sperm emphasize energy-intensive pathways to sustain motility, Sapala sperm rely on antioxidative and amino acid metabolism to maintain functionality, and Toraja sperm prioritize DNA stability and membrane resilience. The ROC curve analysis further refined the discriminatory power of the identified metabolites, revealing potential breed-specific biomarker candidates. In Silangit, oxalacetic acid and adenosine were found to be directly correlated with fertility outcomes in previous reports in bovine sperm metabolomics, where purine nucleotides and TCA intermediates were directly correlated with fertility outcomes (Talluri et al., 2022). In Toraja, uridine and dimethylethanolamine are of particular interest; while uridine has been linked to genomic stability, dimethylethanolamine represents a relatively novel discovery in sperm metabolomics as a marker of membrane remodeling. Glutamic acid was validated as a fertility-associated metabolite in Sapala, consistent with studies in human and bovine sperm (Kostyunina et al., 2025). Several limitations warrant consideration. Putative phenethylamine and succinic acid semialdehyde signals are novel and require targeted confirmation (authentic standards, MS/MS, retention time, and independent cohorts). The likely exogenous compounds bis(2-ethylhexyl)phthalate and meprobamate are reported but not interpreted as intrinsic biomarkers. The small sample size (n=3 per breed; total n=9) limits the power/generalizability and may inflate the cross-validated multivariate/ROC metrics. Pooling five straws per batch per bull can mask inter-ejaculate variability. To verify markers and establish robust cut-offs, larger, independent, unpooled cohorts are needed. ConclusionThis study demonstrates that post-thawed sperm quality and metabolomic profiles differed among Silangit, Toraja, and Sapala buffalo breeds, yielding concise breed-specific metabolite panels with supportive ROC metrics that link semen quality phenotypes to key pathways. Semen quality and metabolomic data reveal breed-specific adaptations to cryopreservation in Indonesian buffalo, thereby guiding fertility prediction, improving cryopreservation, and selective breeding to sustain indigenous genetic resources. AcknowledgmentsAll authors express their sincere gratitude to the Institute for Research and Community Service and the entire team at the Faculty of Animal Science, Universitas Gadjah Mada. FundingThis research was supported by Regular Fundamental Research Funding, Research and Community Service Program, Directorate of Research and Community Service, Ministry of Higher Education, Science, and Technology of the Republic of Indonesia (Grant Number: 2428/UN1/DITLIT/Dit-Lit/PT.01.03/2025). Authors' contributionSB, KDP, and TM contributed to supervising the experiment and the conception and design of the study. SB, KDP, and AN contributed to the conception and design of the study and to the improvement of the manuscript. AMD was responsible for data collection and drafting of the manuscript. RR and MFA contributed to data analysis and drafting of the manuscript. RR and SN provided critical review, suggestions, and further improvement of the manuscript. Conflict of interestThe authors declare no conflict of interest with any financial, personal, or other relationships with other people or organizations related to the material discussed in the manuscript. Data availabilityAll data supporting this study’s findings are available within the manuscript. ReferencesAbruzzese, G.A., Sanchez-Rodriguez, A. and Roldan, E.R.S. 2024. Sperm metabolism. Mol. Reprod. Develop. 91(10), e23772. Akbarinejad, V., Fathi, R., Shahverdi, A., Esmaeili, V., Rezagholizadeh, A. and Ghaleno, L. 2018. Dehydrogenase (Alda-1) Could enhance quality of equine cooled semen by ameliorating loss of mitochondrial function over time. J. Equine. Vet. Sci. 70, 63–70; doi:10.1016/j.jevs.2018.08.004 Alfian, A.M., Yusuf, M., Ako, A., Toleng, A.L., Diansyah, A.M., Amrullah, M.F., Rahmat, R., Hasrin, H. 2025. The effects of mineral mix supplementation on reproductive performance of Bali bulls. Egyptian. J. Vet. Sci. 1, 1-8. doi: 10.21608/ejvs.2025.335655.2502. Anindita, R., Sadiyah, A.A., Khoiriyah, N., and Nendyssa, D.R. 2020. The demand for beef in Indonesian urban. IOP Conference Series: Earth and Environmental Science, Volume 411, Second International Conference on Food and Agriculture 2019 2–3 November 2019, Bali Nusa Dua Convention Center, Bali, Indonesia. doi: 10.1088/1755-1315/411/1/012057 Azwar, F., Premono, B.T. and Tata, H.L. 2024. Understanding Farmers’ Livelihoods in Managing Swamp Buffalo in Adapting to Climate Change in South Sumatra. In: Lestari, S., et al. Proceedings of the International Conference on Radioscience, Equatorial Atmospheric Science and Environment and Humanosphere Science. INCREASE 2023. Springer Proceedings in Physics, vol 305. Springer, Singapore. https://doi.org/10.1007/978-981-97-0740-9_61 Bellezza, I. and Minelli, A. 2017. Adenosine in sperm physiology. Mol. Aspects Med. 55, 102–109; doi: 10.1016/j.mam.2016.11.009 Castro, M., Leal, K., Pezo, F. and Contreras, M.J. 2025. Sperm membrane: molecular implications and strategies for cryopreservation in productive species. Animals 15(12), 1808. Chen, X. and Huang, S. 2024. Optimization of reproductive technologies in water buffalo: a review of current practices. Int. J. Mol. Zool. 14 305–314. doi: 10.5376/ijmz.2024.14.0027. Diansyah, A.M., Santoso, S., Herdis, H., Yusuf, M., Priyatno, T.P., Maulana, T., Toleng, A.L., Dagong, M.I.A., Said, S., Iskandar, H., Nurlatifah, A., Lestari, P., Affandy, L. and Baharun, A. 2025. Identification of reproductive performance in Bali-polled bulls using computer-assisted semen analysis and plasma seminal proteomics. Vet. World. 18(1), 102–109; doi:10.14202/vetworld.2025.102-109 Diansyah, A.M., Yusuf, M., Rahmat, R. and Alfian, A.M. 2025. Enhanced sperm sexing efficiency and quality preservation in Bali bulls using freeze-dried albumin separation media. Open Vet. J. 15(6), 2416–2426; doi:10.5455/OVJ.2025.v15.i6.14 Diansyah, A.M., Yusuf, M., Toleng, A.L., Dagong, M.I.A., Maulana, T. and Hasrin, B.A. 2023. The sperm post-thawing quality and proteomic seminal plasma on fertility performance of Bali-polled bull. Adv. Anim. Vet. Sci. 11(4), 517–525; doi:10.17582/journal.aavs/2023/11.4.517.525 Du Plessis, S., Agarwal, A., Mohanty, G. and Van Der Linde, M. 2015. Oxidative phosphorylation versus glycolysis: what fuel do spermatozoa use?. Asian. J. Andrology. 17(2), 230–235; doi: 10.4103/1008-682X.135123 Egbujor, M.C., Olaniyan, O.T., Emeruwa, C.N., Saha, S., Saso, L. and Tucci, P. 2024. An insight into role of amino acids as antioxidants via NRF2 activation. Amino Acids 56(1), 23. Fitriana, S.B., Maghfiroh, N.A., Baity, A.N., Diatmono, D.F.F., Prihantoko, K.D., Bintara, S. and Widayati, D.T. 2025. Effect of different thawing methods on frozen semen characteristics and DNA damage of Indonesian Simmental bull. Pak. J. Agricult. Res. 38(1), 08–18; doi:10.17582/journal.pjar/2025/38.1.8.18 Henchion, M., Moloney, A.P., Hyland, J., Zimmermann, J. and Mccarthy, S. 2021. Review: trends for meat, milk and egg consumption for the next decades and the role played by livestock systems in the global production of proteins. Animal 15, 100287; doi:10.1016/j.animal.2021.100287 Inyawilert, W., Rungruangsak, J., Liao, Y.J., Tang, P.C. and Paungsukpaibool, V. 2021. Melatonin supplementation improved cryopreserved Thai swamp buffalo semen. Reprod. Domestic. Animals. 56(1), 83–88; doi:10.1111/rda.13851 Kostyunina, O., Ermilov, A., Traspov, A., Bakoev, F., Chesnokov, D., Panova, A. and Kuznetsov, A. Investigation of the seminal plasma metabolome in Holstein bulls. In BIO Web of Conferences, Moscow, Rusian, 2025 179, p 1002. doi: 10.1051/bioconf/202517901002 Maciel, V.L., Caldas-Bussiere, M.C., Silveira, V., Reis, R.S., Rios, A.F.L. and Paes De Carvalho, C.S. 2018. L-arginine alters the proteome of frozen-thawed bovine sperm during in vitro capacitation. Theriogenology 119, 1–9. Makkar, H.P.S. 2018. Review: feed demand landscape and implications of food-not feed strategy for food security and climate change. Animal 12(8), 1744–1754; doi:10.1017/S175173111700324X Martínez, A., Abanto, M., Días, N.B., Olate, P., Pérez Nuñez, I., Díaz, R., Sepúlveda, N., Paz, E.A. and Quiñones, J. 2025. Recent trends in food quality and authentication: the role of omics technologies in dairy and meat production. Int. J. Mol. Sci. 26(9), 4405. Mateo-Otero, Y. 2024. Integrating metabolomics into reproduction: sperm metabolism and fertility enhancement in pigs. Anim. Reprod. Sci. 269, 107539. Maulana, T., Agung, P.P., Gunawan, M. and Said, S. 2022. Computer aided semen analysis (CASA) to determine the quality and fertility of frozen thawed sumba ongole sperm supplemented with amino acids. Livestock. Anim. Res. 20(2), 194–201; doi:10.20961/lar.v20i2.58754 Menezes, E.B., Velho, A.L.C., Santos, F., Dinh, T., Kaya, A., Topper, E., Moura, A.A. and Memili, E. 2019. Uncovering sperm metabolome to discover biomarkers for bull fertility. BMC. Genomics. 20(1), 714. Nain, D., Mohanty, T.K., Dewry, R.K., Bhakat, M., Nath, S., Gupta, V.K. and Parray, M.A. 2023. Butylated Hydroxytoluene (Bht) improves the post-thaw semen quality in low-dose sperm cryopreservation in Murrah buffalo bull. Cryo-Letters 44(1), 57–65; doi:10.54680/Fr23110110612 Nirmala, A., Toleng, A.L., Yusuf, M., Herdis, H., Diansyah, A.M., Amrullah, M.F., Rahmat, R., Rajamuddin, R., Alfian, A.M. and Hasrin, H. 2025. Effect of adding Kasumba Turate (Carthamus tinctorius L.) Extract in tris-egg yolk diluent on sperm quality in Bali bulls. J. Anim. Health. Prod. 13(1), 154–159; doi:10.17582/journal.jahp/2025/13.1.154.159 Prihandini, P.W., Tribudi, Y.A., Hariyono, D.N.H., Sari, A.P.Z.N.L., Praharani, L., Handiwirawan, E., Tiesnamurti, B., Romjali, E., Matitaputty, P.R. and Wiyono, A. 2023. Biodiversity of Indonesian indigenous buffalo: first review of the status, challenges, and development opportunities. Vet. World 16(11), 2217–2229; doi:10.14202/vetworld.2023.2217-2229 Ribeiro, J.C., Nogueira-Ferreira, R., Amado, F., Alves, M.G., Ferreira, R. and Oliveira, P.F. 2022. Exploring the role of oxidative stress in sperm motility: a proteomic network approach. Antioxidants &. Redox Signaling 37(7-9), 501–520. Saad, A.A., Hussein, T., El-Sikaily, A., Abdel-Mohsen, M.A., Mokhamer, E.H., Youssef, A.I. and Mohammed, J. 2019. Effect of polycyclic aromatic hydrocarbons exposure on sperm DNA in idiopathic male infertility. J. Health. Pollut. 9(21), 190309; doi:10.5696/2156-9614-9.21.190309 Said, S., Diansyah, A.M., Rahayu, J.D., Maulana, T., Gunawan, M. and Kaiin, E.M. 2025. A comparative analysis of semen quality traits and sperm kinematic parameters in relation to fertility prediction in Murrah buffaloes. J. Adv. Vet. Anim. Res. 12(3), 751–759; doi:10.5455/javar.2025.l938_ Said, S. 2020. Integrated livestock business and industry in Indonesia. In IOP Conference Series: Earth and Environmental Science, in International Conference: Improving Tropical Animal Production for Food Security 22-24 November 2019, South East Sulawesi, Indonesia. 465(1), p 12. doi:10.1088/1755-1315/465/1/012003 Sari, Y.W., Widyarani., Sanders, J.P.M. and Heeres, H.J. 2021. The protein challenge: matching future demand and supply in Indonesia. Biofuels. Bioproducts Biorefining 15(2), 341–356; doi:10.1002/bbb.2176 Sedha, S., Kumar, S. and Shukla, S. 2015. Role of oxidative stress in male reproductive dysfunctions with reference to phthalate compounds. Urology. J. 12(5), 2304–2316. Sengupta, P., Durairajanayagam, D., Agarwal, A. 2020. Fuel/Energy Sources of Spermatozoa. Male Infertility: Contemporary Clinical Approaches, Andrology, ART and Antioxidants: Second Edition, Springer International Publishing, pp 323-335. DOI: 10.1007/978-3-030-32300-4_26. Sharma, U., Conine, C.C., Shea, J.M., Boskovic, A., Derr, A.G., Bing, X.Y., Belleannee, C., Kucukural, A., Serra, R.W., Sun, F., Song, L., Carone, B.R., Ricci, E.P., Li, X.Z., Fauquier, L., Moore, M.J., Sullivan, R., Mello, C.C., Garber, M. and Rando, O.J. 2016. Biogenesis and function of tRNA fragments during sperm maturation and fertilization in mammals. Science 351(6271), 391–396. Socha, M.W., Flis, W. and Wartęga, M. 2024. Epigenetic genome modifications during pregnancy: the impact of essential nutritional supplements on DNA methylation. Nutrients 16(5), 678. Su, X., Yao, L., Wang, X., Zhang, Y., Zhang, G. and Li, X. 2025. Mechanisms for cell survival during abiotic stress: focusing on plasma membrane. Stress. Biol. 5(1), 1. Surahman, S., M Yusuf, M. Y., S Garantjang, S. G. and AL Toleng, A. L. 2021. Sperms motility, viability, and abnormality of the frozen semen at different bull breeds. Sperms motility, viability, and abnormality of the frozen semen at different bull breeds. InIOP Conference Series: Earth and Environmental Science (Vol. 788, No. 1, p. 012140). IOP Publishing. https://doi.org/10.1088/1755-1315/788/1/012140 Suhardi, S., Summpunn, P., Duangjinda, M., Wuthisuthimethavee, S. 2022. Buffalo (Bubalus bubalis) phenotypic diversity characterization reveals the need for improved performance based on quantitative and qualitative characteristics: a comparison of Kalang buffalo in Kalimantan, Indonesia and Thale Noi buffalo in Phatthalung, Thailand. Buffalo Bull. 41(3), 373–390. Talluri, T.R., Kumaresan, A., Sinha, M.K., Paul, N., Ebenezer Samuel King, J.P. and Datta, T.K. 2022. Integrated multi-omics analyses reveals molecules governing sperm metabolism potentially influence bull fertility. Scientific. Rep. 12(1), 10692. Tsuji, T., Febriany, D.S., Widiastuti, I. and Yazid, M. 2022. Uses of domestic water buffalo milk in South Sumatra, Indonesia. In IOP Conference Series: Earth and Environmental Science, in Sriwijaya Conference on Sustainable Environment, Agriculture and Farming System 29th September 2021, Palembang, Indonesia. 995(1), p 12018. doi: 10.1088/1755-1315/995/1/012018 Velho, A.L.C., Menezes, E., Dinh, T., Kaya, A., Topper, E., Moura, A.A. and Memili, E. 2018. Metabolomic markers of fertility in bull seminal plasma. PLos One 13(4), 195279. Wertheimer, E., Krapf, D., De La Vega-beltran, J.L., Sánchez-Cárdenas, C., Navarrete, F., Haddad, D., Escoffier, J., Salicioni, A.M., Levin, L.R., Buck, J., Mager, J., Darszon, A. and Visconti, P.E. 2013. Compartmentalization of distinct cAMP signaling pathways in mammalian sperm. J. Biol. Chem. 288(49), 35307–35320. XueXia, L., Yanan, L., Zi, T., Yusheng, Z., Zelin, W., Peng, Z., Meina, X. and Fujun, L. 2023. Di-2-ethylhexyl phthalate (DEHP) exposure induces sperm quality and functional defects in mice. Chemosphere 312, 137216. Zuidema, D., Kerns, K. and Sutovsky, P. 2021. An exploration of current and prospective semen analysis and sperm selection for livestock artificial insemination. Animals 11(12), 3563. | ||
| How to Cite this Article |
| Pubmed Style Bintara S, Prihantoko KD, Nurlatifah A, Maulana T, Diansyah AM, Nurhaliza S, Rahmat R, Amrullah MF. Integrative analysis of semen quality and metabolomic profiles reveals breed-specific biomarker fertility in Indonesian buffalo. Open Vet. J.. 2025; 15(12): 6670-6683. doi:10.5455/OVJ.2025.v15.i12.48 Web Style Bintara S, Prihantoko KD, Nurlatifah A, Maulana T, Diansyah AM, Nurhaliza S, Rahmat R, Amrullah MF. Integrative analysis of semen quality and metabolomic profiles reveals breed-specific biomarker fertility in Indonesian buffalo. https://www.openveterinaryjournal.com/?mno=282106 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.48 AMA (American Medical Association) Style Bintara S, Prihantoko KD, Nurlatifah A, Maulana T, Diansyah AM, Nurhaliza S, Rahmat R, Amrullah MF. Integrative analysis of semen quality and metabolomic profiles reveals breed-specific biomarker fertility in Indonesian buffalo. Open Vet. J.. 2025; 15(12): 6670-6683. doi:10.5455/OVJ.2025.v15.i12.48 Vancouver/ICMJE Style Bintara S, Prihantoko KD, Nurlatifah A, Maulana T, Diansyah AM, Nurhaliza S, Rahmat R, Amrullah MF. Integrative analysis of semen quality and metabolomic profiles reveals breed-specific biomarker fertility in Indonesian buffalo. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6670-6683. doi:10.5455/OVJ.2025.v15.i12.48 Harvard Style Bintara, S., Prihantoko, . K. D., Nurlatifah, . A., Maulana, . T., Diansyah, . A. M., Nurhaliza, . S., Rahmat, . R. & Amrullah, . M. F. (2025) Integrative analysis of semen quality and metabolomic profiles reveals breed-specific biomarker fertility in Indonesian buffalo. Open Vet. J., 15 (12), 6670-6683. doi:10.5455/OVJ.2025.v15.i12.48 Turabian Style Bintara, Sigit, Kurniawan Dwi Prihantoko, Aeni Nurlatifah, Tulus Maulana, Athhar Manabi Diansyah, Sitti Nurhaliza, Rahmat Rahmat, and Muhammad Fajar Amrullah. 2025. Integrative analysis of semen quality and metabolomic profiles reveals breed-specific biomarker fertility in Indonesian buffalo. Open Veterinary Journal, 15 (12), 6670-6683. doi:10.5455/OVJ.2025.v15.i12.48 Chicago Style Bintara, Sigit, Kurniawan Dwi Prihantoko, Aeni Nurlatifah, Tulus Maulana, Athhar Manabi Diansyah, Sitti Nurhaliza, Rahmat Rahmat, and Muhammad Fajar Amrullah. "Integrative analysis of semen quality and metabolomic profiles reveals breed-specific biomarker fertility in Indonesian buffalo." Open Veterinary Journal 15 (2025), 6670-6683. doi:10.5455/OVJ.2025.v15.i12.48 MLA (The Modern Language Association) Style Bintara, Sigit, Kurniawan Dwi Prihantoko, Aeni Nurlatifah, Tulus Maulana, Athhar Manabi Diansyah, Sitti Nurhaliza, Rahmat Rahmat, and Muhammad Fajar Amrullah. "Integrative analysis of semen quality and metabolomic profiles reveals breed-specific biomarker fertility in Indonesian buffalo." Open Veterinary Journal 15.12 (2025), 6670-6683. Print. doi:10.5455/OVJ.2025.v15.i12.48 APA (American Psychological Association) Style Bintara, S., Prihantoko, . K. D., Nurlatifah, . A., Maulana, . T., Diansyah, . A. M., Nurhaliza, . S., Rahmat, . R. & Amrullah, . M. F. (2025) Integrative analysis of semen quality and metabolomic profiles reveals breed-specific biomarker fertility in Indonesian buffalo. Open Veterinary Journal, 15 (12), 6670-6683. doi:10.5455/OVJ.2025.v15.i12.48 |