| Research Article | ||
Open Vet. J.. 2026; 16(1): 287-302 Open Veterinary Journal, (2026), Vol. 16(1): 287-302 Research Article Supplementation of nanoencapsulated Peronema canescens jack leaf extract on growth performance, carcass traits, digestive characteristics, and jejunum histomorphometry in broiler chickensBella Putri Maharani1, Hafi Luthfi Sanjaya1, Aji Praba Baskara1, Zuprizal Zuprizal1, Bambang Ariyadi2, Ronny Martien3, Chusnul Hanim1, Rusman Rusman4 and Nanung Danar Dono1*1Department of Animal Nutrition and Feed Science, Faculty of Animal Science, Universitas Gadjah Mada, Yogyakarta, Indonesia 2Department of Animal Production, Faculty of Animal Science, Universitas Gadjah Mada, Yogyakarta, Indonesia 3Department of Pharmaceutics, Faculty of Pharmacy, Universitas Gadjah Mada, Yogyakarta, Indonesia 4Department of Animal Product, Faculty of Animal Science, Universitas Gadjah Mada, Yogyakarta, Indonesia *Corresponding Author: Nanung Danar Dono, Department of Animal Nutrition and Feed Science, Faculty of Animal Science, Universitas Gadjah Mada, Yogyakarta, Indonesia Email: nanungdd [at] ugm.ac.id Submitted: 02/09/2025 Revised: 30/11/2025 Accepted: 10/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
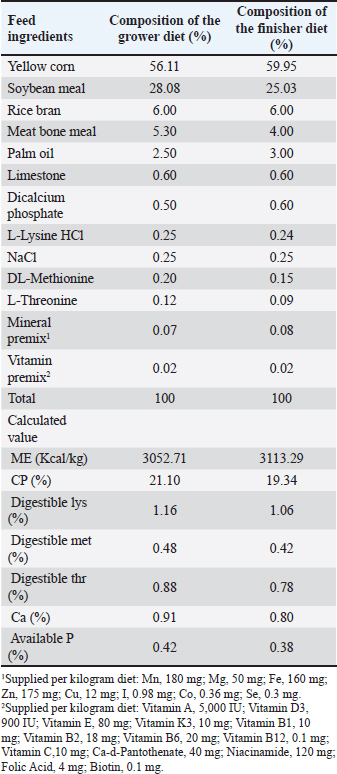
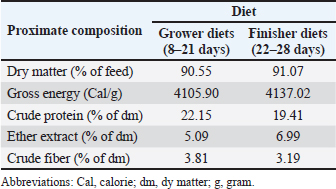
AbstractBackground: Peronema canescens Jack (P. canescens) leaves contain phenols, tannins, flavonoids, and saponins with antibacterial, antioxidant, and growth-promoting activities. However, their lipophilic nature limits their absorption in broilers. Chitosan-based nanoencapsulation is used to enhance the effectiveness of the extract when administered through drinking water. Aim: This study aimed to investigate the effects of nanoencapsulated P. canescens leaf extract (PcNP) administered through drinking water on the growth performance, carcass traits, digestive characteristics, and jejunum histomorphometry of broiler chickens. Methods: This study used a completely randomized design with six treatments, each of which was replicated six times. A total of 288 male Indian River broiler chickens were reared from day 1 to day 28. The treatments were as follows: negative control (no treatment); T1 (8.5 ml/l PcNP); T2 (17 ml/l PcNP); T3 (34 ml/l PcNP); T4 (68 ml/l PcNP); and positive control (25 mg/l zinc bacitracin) in drinking water. Results: PcNP supplementation in broiler chickens consistently improved feed efficiency by 1.42 and carcass percentage at concentrations ranging from 17 to 68 ml/l with 72.74%, 72.54%, and 74.14% (p < 0.05). Feather free weight (1,503.66 g), carcass yield (1,339.00 g), breast yield (520.50 g), upper thigh yield (215.50 g), and ileum length (89.66 cm) were increased in broiler chickens supplemented with 68 ml/l PcNP (p < 0.05). Abdominal fat weight and percentage decreased without PcNP supplementation but increased with supplementation of zinc bacitracin (p < 0.05). A dose of 8.5 ml/l PcNP significantly increased villus height (1570.26 μm) and villus height-to-crypt depth (12.00) (p < 0.005), whereas all doses reduced villus width, apical width, and crypt depth in broiler chickens (p < 0.001). Conclusion: These findings suggest that PcNP may have potential as a natural growth promoter in broiler production due to its beneficial effects on improving feed efficiency, carcass traits, and intestinal health. Keywords: AGPs replacement, Broiler chickens, Drinking water, Nanoencapsulation, Peronema canescens Jack. IntroductionGlobal poultry meat production and consumption have shown a consistent upward trend, serving as a primary driver of global meat production growth. By 2034, poultry meat consumption is projected to reach approximately 173 million tons (ready-to-cook basis), representing approximately 62% of the total increase in global meat consumption (OECD/FAO, 2025). The selection of the most superior genetic traits over the past few years has led to the production of high-quality poultry breeds. One key factor in improving broiler production is the maintenance of a healthy gastrointestinal tract, which directly impacts growth performance (Ducatelle et al., 2023). Traditionally, one of the most common approaches to support gastrointestinal tract health has been the use of antibiotic growth promoters (AGPs). AGPs inhibit pathogen growth through their strong antibacterial effect, thereby preventing subclinical infections (Uehara et al., 2023). The suppression of pathogenic bacteria enhances nutrient availability for the host organism, thereby improving the overall growth efficiency. However, growing global concern over antimicrobial resistance has highlighted the use of AGPs in livestock production as a critical issue, due to their role in fostering a resistant microbial population (Paul et al., 2022). The long-term use of subtherapeutic antibiotic doses can lead to the selection of resistant bacterial strains, which may spread to the environment and humans through the food chain (Zamanizadeh et al., 2021). As a result, many countries have restricted and prohibited the use of AGPs in animal feed (Maria Cardinal et al., 2019). These restrictions have prompted the livestock industry to find new strategies for maintaining gut health in broilers in collaboration with researchers. Extensive research has been conducted to explore alternatives to AGPs, including the use of probiotics (Mohammed et al., 2024; Liu et al., 2025), prebiotics (Shani and Irani, 2024; Al-Baadani et al., 2025), synbiotics (Abdel-Wareth et al., 2019; Hu et al., 2022), organic acids (Khan et al., 2022; Waghmare et al., 2025), and herbal extracts (Pelvan et al., 2022; Maqsood et al., 2025). Alternatives to AGPs function by maintaining gut microbiota balance, improving mucosal integrity, and reducing pathogenic bacteria (Dessalegn et al., 2025). Researchers have increasingly explored natural sources with bioactive properties to achieve these goals. In recent years, herbal plants rich in beneficial secondary metabolites have gained attention as potential substitutes in poultry production (Obianwuna et al., 2024). Peronema canescens Jack (P. canescens), a woody medicinal plant commonly found in Southeast Asia, is a promising candidate. The leaves of P. canescens possess antibacterial (Shalihin et al., 2024), antioxidant (Rizki et al., 2022), immunomodulator (Rahardhian et al., 2025), and anti-inflammatory (Maigoda et al., 2023) properties. Peronema canescens is known to contain various bioactive compounds with notable pharmacological potential, such as polyphenols, tannins, flavonoids, saponins, alkaloids, and diterpenoid clerodanes (peronemins D1, C1, B2, B1, A3, and A2) (Kitagawa et al., 1994; Ahkam et al., 2024). However, the lipophilic nature of many bioactive compounds limits their solubility and bioavailability, leading to rapid degradation in the digestive tract (Mohammadi et al., 2016). Incorporating these compounds into nanoparticle-based delivery systems improves their solubility, stability, and absorption (Erfanian et al., 2014; Aboalnaja et al., 2016), prompting a growing interest in bioactive nano-delivery applications in livestock production. The application of nanoencapsulatiown technology offers significant advantages due to its ability to expand the surface area to volume ratio, thereby improving the bioavailability of encapsulated materials and facilitating their controlled and sustained release at the target site (Dikshit et al., 2021; Osman et al., 2024). Nanoencapsulation has the potential to be a proficient and low-cost strategy to secure bioactive chemicals and improve the functional effects of herbal additives in animal nutrition (Linh et al., 2022). For instance, a comparative study between garlic essential oil and its encapsulated form showed that nanoencapsulated garlic essential oil improved performance and nutrient digestibility in broiler chickens (Amiri et al., 2021). Kusmayadi et al. (2025) also showed that applying nanotechnology to low doses of mangosteen peel extract improved the body weight (BW) and feed conversion ratio in broiler chickens. The application of nanoencapsulation to P. canescens leaf extract is expected to enhance its bioavailability and stability, enabling effective delivery to target organs in broiler chickens. Therefore, this study aimed to assess the impacts of drinking water with nanoencapsulated P. canescens Jack leaf extract on broiler performance, carcass quality, digestive physiology, and jejunum morphology. Materials and MethodsPlant material and leaf extract preparation for P. canescens JackPeronema canescens leaf extract was derived from plants naturally growing in Ogan Komering Ilir, South Sumatra, Indonesia (3.36667°S, 105.36667°E), a lowland region characterized by gentle topography. Fresh P. canescens leaves were sun-dried for 3–7 days until completely dry and then ground into a fine powder. The extraction process was based on a modified maceration method (Samreen et al., 2022). The powdered P. canescens leaves were suspended in 500 mL of 70% ethanol at a ratio of 1:5 for 5 days. The P. cansecens leaf extract was filtered using Whatman No.1 filter paper and centrifuged at 10,000 rpm for 15 minutes using a Centrifuge 5804R (Eppendorf AG, 22331 Hamburg, Germany). The resulting supernatant was then concentrated using a rotavapor® R-300 (BÜCHI Labortechnik AG, Flawil, Switzerland) at 40°C to form a paste. The secondary metabolite compounds of the extract were analyzed to determine total phenolics (Khoddami et al., 2013), tannins (OECD/FAO, 2000), flavonoids (Dashputre and Bandawane, 2021), and saponins (Mora-Ocación et al., 2022). The content of P. canescens leaf extract was as follows: total phenolics, 81.32 g TAE/ml; total tannins, 36.73 g tannic acid equivalent (TAE)/ml; total flavonoids, 7.80% (b/w); and total saponins, 2.69% (b/w). PcNP preparationPcNP was prepared using the ionic gelation method, in which positively charged chitosan interacts with negatively charged sodium tripolyphosphate (STPP), according to the method of Kim et al. (2022) with some modifications. The process began by combining P. canescens leaf extract (1%) with a chitosan solution (0.1%) for 10 minutes under magnetic stirring. Subsequently, an STPP solution (0.1%) was gradually added over 10 min. The resulting nanoencapsulation formulation comprised chitosan (68%), P. canescens leaf extract (18%), and STPP (14%). The PcNP formulation was analyzed to determine particle size, particle distribution, and zeta potential (Liang et al., 2017), phenol and tannin encapsulation efficiency (Chatterjee et al., 2022), and antibacterial inhibition (Govindarajan et al., 2023). The prepared PcNPs exhibited a particle size of 178.73 ± 3.29 nm, a particle distribution of 0.49 ± 0.91, encapsulation efficiency of phenol of 15.61% ± 2.65%, encapsulation efficiency of tannin of 33.29 ±, and a potential zeta of +37.80 ± 1.57 mV. The zone of inhibition test on bacterial inhibition PcNP showed results of 17.92 ± 1.17 mm on Escherichia coli (E. coli), 16.77 ± 1.14 mm on Salmonella typhimurium (S. typhimurium), 17.37 ± 0.86 mm on Staphylococcus aureus (S. aureus), and no inhibition detected on Lactobacillus acidophilus. Birds, diets, and experimental designA total of 288 male chicks of the New Lohmann Indian River (MB 202 Platinum) strain that had been vaccinated against Newcastle disease and Gumboro disease (infectious bursal disease) from the hatchery were placed in rearing cages for 7 days starting on day 1. The chickens were weighed on day 7 and randomly placed in 36 colony cages measuring 1 × 0.75 m with an initial BW of 180 ± 10 g. Treatments began in the morning on day 8 of the rearing period. This study consisted of 6 groups with 6 replicates of each treatment group and 8 chickens per cage. The groups were as follows: negative control (no treatment); T1 (8.5 ml/l PcNP); T2 (17 ml/l PcNP); T3 (34 ml/l PcNP); T4 (68 ml/l PcNP); and positive control (25 mg/l zinc bacitracin). All treatments were supplemented with drinking water. The treatment was applied based on the minimum inhibitory concentration, based on the method of Tamfu et al. (2020). The lowest dose in the treatment was 1/4× the minimum inhibitory dose, and the highest dose was 2× the minimum inhibitory dose against E. coli, S. typhimurium, and S. aureus bacteria. Each treatment was tested for the quantification of phenolic and tannin compounds according to FAO/IAEA (2000). T1 contained 1.17 mg TAE /l of phenol and 0.53 mg TAE/l of tannin, T2 contained 2.35 mg TAE/l of phenol and 1.06 mg TAE/l of tannin, T3 contained 4.70 mg TAE/l of phenol and 2.13 mg TAE/l of tannin, and T4 contained 9.40 mg TAE/l of phenol and 4.25 mg TAE/l of tannin. The starter phase (1–7 days) used a commercial feed (7500, PT. New Hope Indonesia, Cirebon, Indonesia), with a composition of 13% moisture, 23% crude protein, 5% crude fat, 4% crude fiber, 0.80–1.10% calcium (Ca), 0.5% phosphorus, 1.30% lysine, 0.50% methionine, 0.90% methionine + cystine, 0.20% tryptophan, and 0.80% threonine. The grower (8–21 days) and finisher (22–28 days) diets were described according to Aviagen (2022) recommendations, as presented in Tables 1 and 2. Broiler management practices were generally conducted according to the Indian River Broiler Management Handbook (Aviagen, 2025). The broiler house was maintained in a well-ventilated and sanitary condition. During the first three days, the ambient temperature was maintained at 30°C, after which it was gradually reduced by 2.5°C per week until a final temperature of 20°C was achieved. A lighting program of 23 hours of light and 1 hour of darkness was applied during the first week and subsequently adjusted after day 7 to 18–19 hours of light and 5–6 hours of darkness to ensure optimal growth performance and animal welfare. Table 1. Ingredients and composition of the basal diets.
Table 2. Chemical composition of the basal diet.
Growth performance measurementAll chickens were weighed each week collectively using a TM-A 150-kg scale (Altraman, West Jakarta, Indonesia). Feed and drinking water residues were recorded every morning, starting from the treatment period, before feed was added and drinking water was changed. The final body weight (FBW) of broilers was recorded after a 12-hour feed withdrawal, while water remained available for each replicate group. Average daily feed intake (ADFI) was determined by recording the feed consumed per bird each day in grams, and average daily gain (ADG) was calculated as the daily increase in BW (Daniel et al., 2025). The average daily water intake (ADWI) was calculated by dividing the total drinking water consumption from day 8 to day 28 by the number of experimental days. Meanwhile, the water-to-feed ratio (WFR) was determined by dividing the total water consumption by the total feed intake during the rearing period (days 8–28). The survival rate referred to the percentage of animals that remained alive throughout the rearing period. The feed conversion ratio (FCR) was determined using the formula total feed intake (g)/total BW gain (g) (Eid et al., 2025). The chicken performance index (PI) was calculated using the formula by Abd Abd El-Hack et al. (2025).
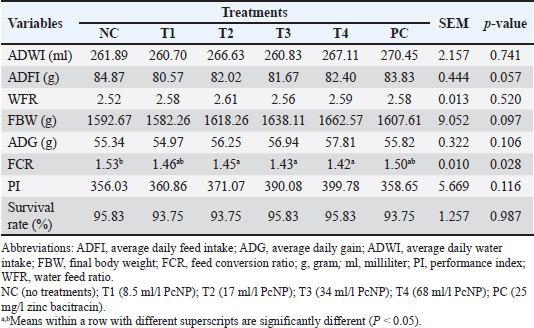
Carcass traitsAt the end of the experiment (28 days), 36 chickens (6 chickens per treatment, 1 chicken per replicate) with FBW close to the median for each group were selected, weighed, and slaughtered using the halal method by decapitation and cutting the neck blood vessels for carcass assessment. Before the slaughter day, the chickens were fasted from feed, but water was still provided. The slaughtered chickens were briefly boiled at 65°C for 1 minute, dried, and then plucked. After the internal organs were plucked and removed, the neck, head, and feet were removed manually. The abdominal fat was collected and weighed. The carcass weight is used to determine the yield percentage. The carcass is then cut into commercial cuts, including the breast, thigh, drumstick, and wing, each of which is weighed. The percentage is defined as the carcass weight relative to the live weight multiplied by 100% (Chaiwang et al., 2023). Digestive characteristicsA total of 36 chickens were used (n=36), with 6 chickens per treatment and 1 chicken per replicate. All digestive organ weights were measured using an electronic balance (Mettler-Toledo GmbH, Greifensee, Switzerland) after being emptied of contents. The esophagus, crop, proventriculus, gizzard, duodenum, jejunum, ileum, colon, cecum, and pancreas were measured. Organ weights were expressed as relative weights, which were calculated as organ weight divided by live body weight. The length of each intestinal segment was measured to the nearest 1 mm using a tape measure (Huang et al., 2022). The length of the small intestine was calculated according to the segments: duodenum (part of the small intestine between the gizzard and the bile duct junction), jejunum (up to Meckel's diverticulum), and ileum (between Meckel᾽s diverticulum and the ileocecal junction) (Farkas et al., 2025). The lengths of the colon and large intestine were also measured. The pH of each digestive part (duodenum, jejunum, and ileum) was determined from fresh intestinal samples using a YY1030 semisolid pH meter (Yinmik, Shandong, China). The acidity was measured at three different points within each organ, and the average of these pH readings was recorded as the acidity value. The pH meter was calibrated with pH 4 and pH 7 buffer standards and rinsed with distilled water before use. Jejunum histomorphometryAfter the experiment, one bird per replicate was randomly selected for jejunum sampling. Samples were fixed in 10% formalin–saline solution (Abdel-Moneim et al., 2025). Sections measuring 4–5 μm in thickness were prepared using a Leica RM2255 rotary microtome (Leica Biosystems, Nussloch, Germany) and stained with hematoxylin and eosin for morphometric evaluation under light microscopy (Daneshmand et al., 2017). Villus height, villus width, apical width, and crypt depth were measured using Optilab Viewer 4 image analysis software (Miconos, Yogyakarta, Indonesia). Five well-oriented villi and their associated crypts were selected from five intestinal cross-sections for each sample. Images of the villi were captured at 40x magnification and analyzed using Image Raster 3 software (Miconos, Yogyakarta, Indonesia). The experiment used 36 chickens (n=36), distributed into treatments at a rate of 6 birds per treatment, with one bird representing each replicate. The average of the measurements was used as the basis for further analysis (Abdelhady et al., 2021). Statistical analysisThe collected data were analyzed using analysis of variance in a completely randomized design with SPSS Statistics software version 29.0. One-way analysis of variance was used to evaluate treatment effects, and differences between group means were assessed using Duncan’s post hoc test, with significance set at P ≤ 0.05. The statistical model applied was as follows: Yij=π + Ti + eij Yij represents the value of the concerned treatment, π is the mean for the concerned treatment, Ti is the fixed effect of the treatments, and eij is the random error. Ethical approvalThis study was conducted at the Faculty of Animal Science, Universitas Gadjah Mada, Yogyakarta, Indonesia, from July to October 2024. All experimental procedures involving animals were reviewed and approved by the Research Ethics Committee of the Faculty of Veterinary Medicine, Universitas Gadjah Mada (Approval No. 67/EC-FKH/int./2024) and were performed in strict accordance with the Guidelines for the Care and Use of Laboratory Animals. ResultsGrowth performanceThe effects of PcNP supplementation on broiler growth performance are presented in Table 3. PcNP supplementation at a dose of 17 ml/l provided the best FCR value in broiler chickens compared with the control group (P < 0.05). However, ADWI, ADFI, WFR, ADG, PI, and survival rate were not significantly affected by the inclusion of PcNP in drinking water. Table 3. Growth performance of broiler chickens supplemented with nanoencapsulated Peronema canescens Jack leaf extract in drinking water.
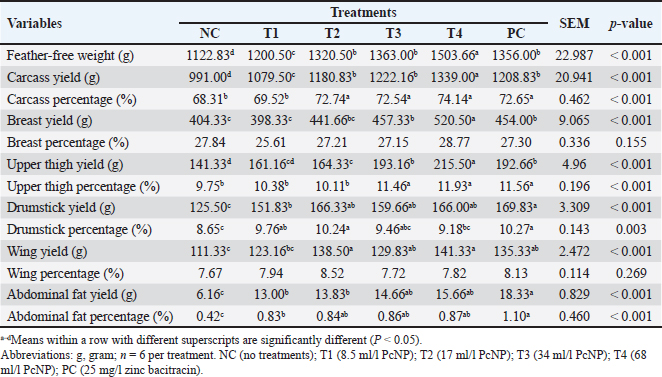
Carcass and carcass percentageResults in Table 4 showed the impacts of PcNP supplementation on carcass yield and percentage. Adding PcNP to drinking water at 68 ml/l resulted in the greatest feather-free weight, carcass yield, breast yield, and upper thigh yield compared to other groups (P < 0.001). Wing yield increased with PcNP supplementation at 17 and 68 ml/l of PcNP (P < 0.001). Supplementation of 34 and 68 ml/l PcNP showed increased upper thigh yield similar to the administration of 25 mg/l zinc bacitracin (P < 0.001). A different trend was observed in carcass percentage, which was higher at a dosage of 25 mg/l and PcNP at a dosage of 17 ml/l, and higher than that in chickens that were not treated and received the lowest dosage of PcNP (P < 0.001). Administration of 17 and 68 ml/l PcNP increased wing yield compared with other treatments (P < 0.001). Drumstick per increased with PcNP supplementation at 17 ml/l and 25 mg/l zinc bacitracin (P < 0.001). Administration of antibiotics (25 mg/l zinc bacitracin) resulted in higher drumstick yield, abdominal fat yield, and abdominal fat percentage (P < 0.001). In contrast, the breast and wing percentages were not affected by PcNP treatment. Table 4. Carcass weight and yield of broiler chickens supplemented with nanoencapsulated Peronema canescens Jack leaf extract in drinking water.
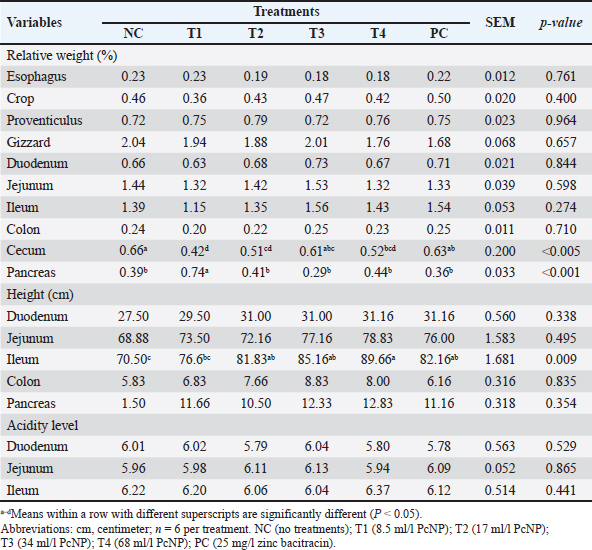
Digestive tract characteristicsNo significant differences were found in the relative weights of the esophagus, crop, proventriculus, gizzard, duodenum, jejunum, ileum, and colon, as well as in the lengths of the duodenum, jejunum, colon, and pancreas, or the acidity of the duodenum, jejunum, and ileum, between the PcNP supplementation and control groups (Table 5). However, the ileum length increased with the supplementation of 68 ml/l PcNP compared with the other groups (P < 0.001). The cecum yield was greater on chickens without treatment (P < 0.05), whereas the pancreas yield increased with the administration of 8.5 ml/l PcNP in broiler chicken drinking water (P < 0.05). Table 5. Digestive tract characteristics of broiler chickens supplemented with nanoencapsulated Peronema canescens Jack leaf extract in drinking water.
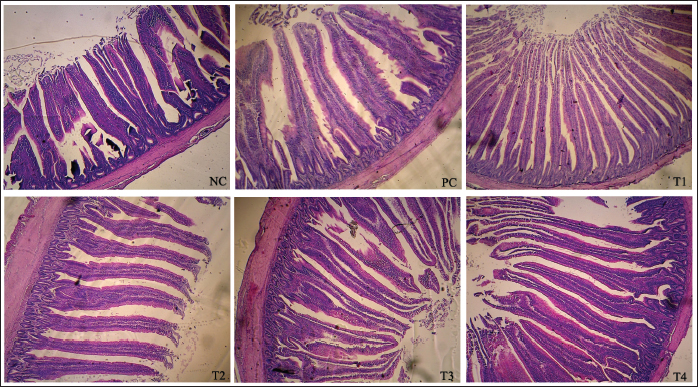
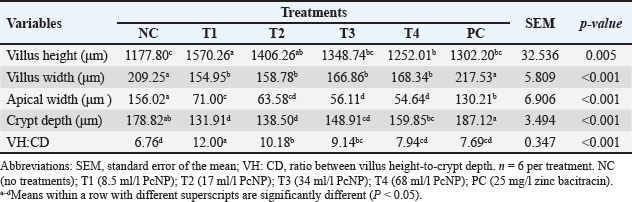
Jejunum morphometricsTable 6 showed the results of the morphometrics of the jejunum of broiler chickens, which were also confirmed in Figure 1. An increased villus height and villus height-to-crypt depth were observed by adding 8.5 ml/l PcNP to the drinking water of broiler chickens compared to other treatments (P < 0.005). The application of the entire dose of PcNP decreased the villus width, apical width, and crypt depth of broiler chickens compared with the control (P < 0.001).
Fig. 1. Microstructure of the intestinal villi in the broiler chicken jejunum supplemented with nanoencapsulated Peronema canescens Jack extract in drinking water. NC (no treatments); T1 (8.5 ml/l PcNP); T2 (17 ml/l PcNP); T3 (34 ml/l PcNP); T4 (68 ml/l PcNP); PC (25 mg/l zinc bacitracin) Table 6. Jejunum morphometrics of broiler chickens supplemented with nanoencapsulated Peronema canescens Jack leaf extract in drinking water.
DiscussionOur research showed that adding PcNP to the drinking water of broiler chickens improved FCR, with an initial dose of 17–68 ml/l. There have been no previous comparisons regarding the use of P. canescens leaves, either in extract or nanoencapsulated form, in animal husbandry. Therefore, we compared the results of this study with those of plants from the same family that contain bioactive compounds similar to those in P. canescens leaves. Similar findings have been reported by Minhas et al. (2025) that the application of 1% Vitex negundo leaf extract can improve FCR in broiler chickens. Another study reported that supplementation with microencapsulated essential oil of Ocimum basilicum Linn. (a species belonging to the same family as P. canescens) was more effective in improving the FCR in broiler chickens compared with the non-microencapsulated form (Thuekeaw et al., 2022). P. canescens leaves contain various important bioactive compounds, notably tannins, which have long been recognized for their antimicrobial activity (Huang et al., 2024). Tannins inhibit bacterial growth by binding to iron, thereby preventing its utilization by bacteria and causing iron deficiency within the cells (Farha et al., 2020). Therefore, PcNP may contribute to better control of pathogenic bacterial growth in the intestine of broilers. Minimizing pathogenic bacterial growth supports microbial balance in the digestive tract, thereby optimizing feed use and improving growth performance (Jie et al., 2024). However, PcNP treatment did not significantly improve other growth performance variables, which may be influenced by genetics (Prakash et al., 2020), feed composition, and environmental conditions (Kyriazakis et al., 2025). This study showed an increase in carcass and feather-free yield in broiler chickens supplemented with 68 ml/l PcNP in drinking water (Table 4). However, the bioactive compounds in the leaf extract (Deeb et al., 2024; Farag et al., 2024; Wang et al., 2024) have been associated with growth enhancement and other beneficial effects in poultry. The improvement in carcass and feather-free weight may be associated with the bioactive compounds in PcNP that could help support gut microflora balance, thereby increasing the efficiency of feed conversion into muscle tissue. Phenolics in plants modulate higher levels of Lactobacillus and Enterococcus spp. in the small intestine, whereas Clostridium levels are lower in broiler chickens (Mahfuz et al., 2021). The mode of action of phenolic compounds is known to be due to the hydroxyl group (–OH) inhibiting the activity of these bacteria (Lobiuc et al., 2023). Our results indicate that the administration of 68 ml/l PcNP in broiler chickens also improved carcass weight in the breast and upper thigh. On the other hand, wing weight increased with PcNP administration at doses of 17 and 68 ml/l, and drumstick weight was higher in the zinc bacitracin group than in the PcNP and groups without PcNP. Aligned with the findings of this study, Nouri (2019) reported an increase in breast meat yield when mint, thyme, and cinnamon essential oils were nanoencapsulated compared to those that were not nanoencapsulated. In contrast to our findings, Elmeligy et al. (2025) reported that dietary supplementation with basil oil in broiler chickens showed no improvement in carcass percentage or weights of major carcass cuts (breast and thigh). The increase in carcass cuts observed with PcNP supplementation in drinking water may be caused by nanoencapsulation technology, which reduces particle size and facilitates the delivery of its bioactive compounds to the digestive tract. The small particle size and enhanced solubility of nanoparticles enable more efficient penetration of nutrients through gastrointestinal epithelial cells, increasing intestinal membrane permeability and promoting greater nutrient absorption throughout the intestinal lining (Ognik et al., 2016; Kumar et al., 2022). Thus, a consistent correlation was found between low FCR and increased weights of these body parts, which has positive implications for the production performance of high economic value cuts. Our study reports that zinc bacitracin supplementation increased abdominal fat yield and percentage compared to chickens treated with and without PcNP. These findings are similar to those reported by Martínez et al. (2022), who found that zinc bacitracin use led to an increase in abdominal fat in broiler chickens. The reduction in abdominal fat observed with PcNP supplementation in the present study may be attributed to saponins, a bioactive compound identified in P. canescens leaf extract. Saponins act as lipase inhibitors, and inhibiting lipase activity after ingestion is a viable strategy for reducing fat absorption (Marrelli et al., 2016). The encapsulation of P. canescens leaf extract into nanosized particles, which includes saponin compounds (Shalihin et al., 2024), may enhance the bioactivity of saponins as lipase inhibitors. Sanneur et al. (2023) found that Pouteria cambodiana bark saponin-rich extracts and their hydrolysates demonstrated pancreatic lipase inhibitory activity. These results are consistent with those of Navarro del Hierro et al. (2021), who also reported that fenugreek and quinoa extracts, both high in saponins, inhibited pancreatic lipase. Lipase activity inhibition reduces fat breakdown and absorption, thereby decreasing the amount of fat entering the systemic circulation and ultimately lowering abdominal fat deposition in broiler chickens. The relative weights of most digestive organs, including the esophagus, proventriculus, gizzard, duodenum, jejunum, ileum, and colon, showed no difference with PcNP supplementation in drinking water. In line with our results, the relative weight of the proventriculus and gizzard (Azzam et al., 2020) and intestines (Pereira Farias et al., 2021) was not influenced by the supplementation of plant extracts with polyphenol contents. In contrast, another study using polyphenol-rich plant sources found only negligible gains in gizzard and intestinal weights (Omar et al., 2020). Our results showed that the relative weight of the cecum decreased with PcNP supplementation. The decrease in cecum yield with PcNP administration may be related to improved digestive efficiency and nutrient absorption in the small intestine, resulting in a reduction in substrate reaching the cecum (Yadav and Jha, 2019). Conversely, the relative weight of the pancreas increased with the application of 8.5 ml/l PcNP in the drinking water of broiler chickens. The increase in pancreas relative weight at the lowest dose of PcNP is probably due to an initial adaptive response to exposure to bioactive compounds, while at higher doses, more energy and nutrients are allocated to overall body growth rather than to the development of digestive organs (Obianwuna et al., 2024). The dimensions of most digestive organs were not affected by PcNP supplementation, except for ileum length, which increased with the highest PcNP concentration (68 ml/l). Yasmin et al. (2021) observed no effect on the overall digestive tract length in broilers supplemented with Catharanthus roseus (containing various phenolic compounds). The increase in ileum length at the highest PcNP dose may be influential because the ileum plays an important role in the absorption of various complex nutrients in the small intestine. The controlled release of bioactive compounds from PcNP may specifically target the ileum, where enhanced activity stimulates tissue growth, resulting in ileal elongation. In addition, flavonoids, which have been confirmed to be present in P. canescens extracts, exhibit antibiofilm activity by disrupting the extracellular matrix. The biofilm matrix of pathogenic bacteria, such as Staphylococcus aureus, consists of various molecules, including EPS, proteins, extracellular DNA, and amyloid fibrils, which interact to ensure its structural integrity (Karygianni et al., 2020). Flavonoids can reduce pathogenic bacterial colonization by damaging bacterial membranes and disrupting biofilm structures (Buiatte et al., 2024), thereby promoting intestinal elongation and improving the overall health of the digestive tract. This is supported by Miao et al. (2020), who reported that flavonoid-rich jujube fruit extracts reduce the thickness of S. aureus biofilms by disrupting their 3D structure and inhibiting biofilm matrix maturation. Our research reports that there was no difference in pH in any small intestine segment of broiler chickens treated with PcNP. The observed pH value (approximately 4.5) is likely attributed to the combined effect of P. canescens leaf extract (pH 4–5), chitosan (pH ~4), and STPP (pH ~9) in the PcNP formulation. This pH level is necessary for nanoencapsulation performance, particularly for chitosan, which serves as the encapsulating agent. The primary amino groups of chitosan undergo deprotonation at higher pH, causing the matrix to shrink and slowing the release of encapsulated compounds (Picos-Corrales et al., 2023). Conversely, at an excessively low pH, protonation leads to polymer matrix swelling (Popat et al., 2012). A pH of approximately 4.5 is categorized as mildly acidic, and it tends to become neutralized when added to drinking water (neutral pH) at low concentrations (Table S1). Supplementation with encapsulated Averrhoa carambola L leaf extract did not significantly alter the pH across the small intestinal segments of broiler chickens (Ma'rifah et al., 2025). Rahma et al. (2025) reported that supplementation with encapsulated noni fruit extract (rich in tannins and saponins), together with copper and zinc, led to a dose-dependent decrease in the gastrointestinal pH of broiler chickens. The intestinal villi are the main sites for nutrient absorption, and the integrity and structure of the intestinal epithelium play an important role in the digestive system, impacting absorption and overall gut health (Khanghahi et al., 2024). Increases in villus length and the ratio of villus height-to-crypt depth were observed in broiler chickens given PcNP at 8.5 ml/l compared with other PcNP doses and the control group. Administration of herbal plants containing bioactive compounds, such as tannins, also increased the villus height of broiler chickens (Gul et al., 2024; Liu et al., 2024). In contrast, a previous study reported a decrease in villus height in broilers supplemented with Salvia hispanica seed (family Lamiaceae) at both 5% and 10% supplementation (Khatun et al., 2025). The growth of intestinal villi increases the total absorption area of the villi lumen and the activity of intestinal brush-border enzymes (Prakatur et al., 2019). This may be attributed to the presence of bioactive compounds that support a more balanced small-intestinal microflora, which could, in turn, facilitate nutrient absorption and potentially contribute to increased villus height. The antimicrobial properties of various bioactive compounds can promote the dominance of lactic acid bacteria in the small intestine, thereby improving nutrient absorption (Apajalahti and Vienola, 2016) and stimulating the growth of intestinal villi associated with increased absorption surface area (Sabry and El-Ghany, 2021). This mechanism is inseparable from the role of various bioactive compounds in plants that cause the lysis of pathogenic bacteria (Clavijo and Flórez, 2018). Nanoencapsulation of P. canescens leaf extract in chitosan further enhances this effect, as chitosan, which forms the coating layer, has antibacterial properties. Positively charged amino groups (NH3+) in chitosan interact with negatively charged microbial cell membranes at pH levels below 6.3, causing intracellular component leakage and ultimately inactivating microorganisms (Park and Kim, 2020). The VH:CD ratio is also a valuable indicator of digestive tract health (Wawrzyniak et al., 2017). Consistent with our research findings, Acalypha australis L leaf extract encapsulated in chitosan increases the ratio of villus height to crypt depth (Sugiharto et al., 2024). Nanoencapsulation of bioactive compounds helps inhibit the growth of various pathogenic bacteria in the digestive tract, particularly in the small intestine. The smaller the size of the nanoparticle, the greater its surface area available for interaction with the external environment (Ozdal and Gurkok, 2022). Intermolecular interactions may occur between the core material and the wall material during the nanoencapsulation process. The intensity of these interactions may vary depending on the types of molecules involved, thus affecting the diffusion rate of the encapsulated compound into the external environment, which in turn directly impacts its antimicrobial activity (Lelis et al., 2021). The width of villi and apical villi decreased with PcNP administration compared with the control group. The results of this study are in line with those of a previous study that reported a decrease in ileal villus width in broilers supplemented with 3.5% Coriandrum sativum L seed, a source of flavonoids and phenolic compounds (Khubeiz et al., 2024). The decrease in villus and apical width observed after PcNP administration is possibly an adaptive response to the gradual release of bioactive compounds. The nanoencapsulated P. canescens leaf extract may stimulate intestinal epithelial remodeling, resulting in thinner yet fully functional villi. At optimal concentrations, the release of the compounds begins rapidly, followed by a slow and sustained phase that maintains their availability in the intestine (Bayer, 2023). The accompanying gut elongation may represent a physiological compensation to maintain the total absorptive surface area. Since nutrient absorption capacity is positively correlated with both intestinal length and villus height (Belote et al., 2023). This morphological adaptation may enhance nutrient absorption and metabolic utilization efficiency. Our study recorded a decrease in crypt depth with PcNP supplementation in drinking water. Adil et al. (2024) also reported a decrease in crypt depth in broiler chickens supplemented with rosemary essential oil encapsulated in chitosan shells. Nanodelivery may contribute to this outcome, as low crypts represent longer-lasting villi without the need for renewal. Crypt depth is a key indicator of optimal growth (Ashayerizadeh et al., 2025). Crypts play an important role in generating new epithelial cells, and their depth reflects the rate of tissue replacement required to sustain continuous villous renewal (Shang et al., 2018; Rahmani et al., 2025). Excessive crypt depth indicates an inflammatory process. The structure and health of the intestinal villi are closely reflected in the overall performance of birds (Rysman et al., 2023). Nanoencapsulation of P. canescens leaf extract provides a more stable and efficient delivery into the gastrointestinal tract, allowing its bioactive compounds to act more effectively than when administered in crude extract form. The smaller particle size facilitates the drug delivery system as the initial goal of nanoencapsulation. ConclusionIn conclusion, the addition of PcNP to drinking water improves intestinal morphology at low concentrations (8.5 ml/l) and increases feed efficiency and meat yield at higher concentrations (up to 68 ml/l). These findings suggest that PcNP can be recommended as a natural growth promoter to improve the performance and quality of broiler chicken meat. Further studies are recommended to investigate the diversity of gut microbiota to confirm the antibacterial potential of PcNP and its role in supporting digestive health. In addition, a comprehensive evaluation of the release profile of the bioactive compounds encapsulated within PcNP under simulated gastrointestinal conditions should be included in future research. AcknowledgmentsThe authors would like to express their gratitude to the Laboratory of Tropical Animal Research Center, Faculty of Animal Science, Universitas Gadjah Mada, for the assistance and facilities provided in this study. Conflict of interestThe authors declare that they have no financial interests or personal relationships that could have influenced the work reported in this manuscript. FundingThis research was funded by the Directorate-General for Science, Technology, and Higher Education Resources, Ministry of Research and Technology/National Agency for Research and Innovation of the Republic of Indonesia (008/E5/PG.02.00/PL.PMDSU/2024; 2085/UN1/DITLIT/PT.01.03/2024). Authors' contributionsAll authors read and approved the final draft of the manuscript. BPM collected and analyzed the data and drafted the entire initial version of the manuscript. HLS verified and interpreted the experimental results. APB supervised the experiment. BPM and NDD conceptualized the manuscript. ZZ developed the study design. BA, RM, CH, and RR validated the experimental design and contributed to the revision of the manuscript. Data availabilityAll data are shown in the table and manuscript file. ReferencesAbd El-hack, M.E., Aldhalmi, A.K., Ashour, E.A., Kamal, M., Khan, M.M.H. and Swelum, A.A. 2025. The effects of formic acid or herbal mixture on growth performance, carcass quality, blood chemistry, and gut microbial load in broiler chickens: formic acid & herbal mixture in broiler diets. Poult. Sci. 104(6), 105085. Abdelhady, A.Y., El-Safty, S.A., Hashim, M., Ibrahim, M.A., Mohammed, F.F., Elbaz, A.M. and Abdel-Moneim, A.M.E. 2021. Comparative evaluation of single or combined anticoccidials on performance, antioxidant status, immune response, and intestinal architecture of broiler chickens challenged with mixed Eimeria species. Poult. Sci. 100(6), 101162. Abdel-Moneim, A.M.E., Mesalam, N.M., Yang, B. and Elsadek, M.F. 2025. Dietary incorporation of biological curcumin nanoparticles improved growth performance, ileal architecture, antioxidative status, serum lipid profile, and humoral immune response of heat-stressed broiler chickens. Poult. Sci. 104(2), 104740. Abdel-Wareth, A.A.A., Hammad, S., Khalaphallah, R., Salem, W.M. and Lohakare, J. 2019. Synbiotic as eco-friendly feed additive in diets of chickens under hot climatic conditions. Poult. Sci. 98(10), 4575–4583. Aboalnaja, K.O., Yaghmoor, S., Kumosani, T.A. and Mcclements, D.J. 2016. Utilization of nanoemulsions to enhance bioactivity of pharmaceuticals, supplements, and nutraceuticals: nanoemulsion delivery systems and nanoemulsion excipient systems. Expert. Opinion. Drug. Del. 13, 1327–1336. Adil, S., Banday, M.T., Hussain, S.A., Wani, M.A., Al-Olayan, E., Patra, A.K., Rasool, S., Gani, A., Sheikh, I.U., Khan, A.A. and Muzamil, S. 2024. Impact of nanoencapsulated rosemary essential oil as a novel feed additive on growth performance, nutrient utilization, carcass traits, meat quality and gene expression of broiler chicken. Foods 13(10), 1515. Ahkam, A.H., Susilawati, Y. and Sumiwi, S.A. 2024. Peronema canescens as a source of immunomodulatory agents: a new opportunity and perspective. Biology 13(9), 744. Al-Baadani, H.H., Aboragah, A.A., Alharthi, A.S. and Alhotan, R.A. 2025. Interaction effect of prebiotic products as feed additives and rearing systems on performance, slaughter yield and intestinal health of broiler chickens. J. Appl. Poult. Res. 34(2), 100535. Amiri, N., Afsharmanesh, M., Salarmoini, M., Meimandipour, A., Hosseini, S.A. and Ebrahimnejad, H. 2021. Nanoencapsulation (in vitro and in vivo) as an efficient technology to boost the potential of garlic essential oil as alternatives for antibiotics in broiler nutrition. Animals 15(1), 100022. Apajalahti, J. and Vienola, K. 2016. Interaction between chicken intestinal microbiota and protein digestion. Anim. Feed Sci. Technol. 221, 323–330. Ashayerizadeh, A., Shirazi, M.R.J., Moradi, H.R., Kazemi, K., Akbarabadi, Z.K. and Jazi, V. 2025. Effects of drinking water supplemented with apple vinegar, essential oils, or colistin sulfate on growth performance, blood lipids, antioxidant status, intestinal morphology, and gut microflora of broiler chickens. Poult. Sci. 104(2), 104801. Aviagen. 2025. Aviagen IR broiler handbook. Available via https://aviagen.com/assets/Tech_Center/LIR_Broiler/Aviagen-IR-Broiler-Handbook-EN.pdf Aviagen. 2025. Aviagen IR broiler handbook. Available via https://aviagen.com/assets/Tech_Center/LIR_Broiler/Aviagen-IR-Broiler-Handbook-EN.pdf Azzam, M.M., Qaid, M.M., Al-Mufarrej, S.I., Al-Garadi, M.A., Albaadani, H.H. and Alhidary, I.A. 2020. Rumex nervosus leaves meal improves body weight gain, duodenal morphology, serum thyroid hormones, and cecal microflora of broiler chickens during the starter period. Poult. Sci. 99(11), 5572–5581. Bayer, I.S. 2023. Controlled drug release from nanoengineered polysaccharides. Pharmaceutics 15(5), 1364. Belote, B.L., Soares, I., Sanches, A.W.D., De Souza, C., Scott-Delaunay, R., Lahaye, L., Kogut, M.H. and Santin, E. 2023. Applying different morphometric intestinal mucosa methods and the correlation with broilers performance under Eimeria challenge. Poult. Sci. 102(9), 102849. Buiatte, V., Proszkowiec-Weglarz, M., Miska, K., Dominguez, D., Mahmoud, M., Lesko, T., Panek, B.P., Chopra, S., Jenkins, M. and Lorenzoni, A.G. 2024. The effects of a high-flavonoid corn cultivar on the gastrointestinal tract microbiota in chickens undergoing necrotic enteritis. PLos One. 19(9), 307333. Chaiwang, N., Marupanthorn, K., Krutthai, N., Wattanakul, W., Jaturasitha, S., Arjin, C., Sringarm, K. and Setthaya, P. 2023. Assessment of nucleic acid content, amino acid profile, carcass, and meat quality of Thai native chicken. Poult. Sci. 102(11), 103067. Chatterjee, N.S., Sukumaran, H.G., Dara, P.K., Ganesan, B., Ashraf, M., Anandan, R., Mathew, S. and Nagarajarao, R.C. 2022. Nano-encapsulation of curcumin in fish collagen grafted succinyl chitosan hydrogel accelerates wound healing process in experiment rats. Food. Hydrocolloids. Health. 2, 1–10. Clavijo, V. and Flórez, M.J.V. 2018. The gastrointestinal microbiome and its association with the control of pathogens in broiler chicken production: a review. Poult. Sci. 97(3), 1006–1021. Daneshmand, A., Kermanshahi, H., Danesh Mesgaran, M., King, A.J. and Ibrahim, S.A. 2017. Effects of pyrimidine nucleosides on growth performance, gut morphology, digestive enzymes, serum biochemical indices and immune response in broiler chickens. Livest. Sci. 204, 1–6. Daniel, S., Zeid, S., Jinghong, L. and Hang, S. 2025. Exploring the effect of feeding broiler chickens a diet incorporating unfermented or fermented palm kernel cake: growth performance, digestibility, biochemical indices, digestive enzyme activity, and mRNA gene expression of nutrient transporters. Ital. J. Anim. Sci. 24(1), 324–335. Dashputre, N.L. and Bandawane, D.D. 2021. Effect of Abelmoschus ficulneus (L.) Wight & Arn. on immunomodulation: in vivo experimental animal models. Futur. J. Pharm. Sci. 7(1), 149. Deeb, S., Ashour, E.A., Abd El-hack, M.E., El-Maaty, M.A., Youssef, I.M., Adil, S., Elolimy, A.A. and Swelum, A.A. 2024. Impacts of dietary different levels of thyme leaf powder as a natural growth promoter on growth performance, carcass characteristics, and blood indices of broilers. Poult. Sci. 103(12), 104396. Dessalegn, B., Debelo, M., Hess, M. and Awad, W.A. 2025. Gut microbiota—Campylobacter jejuni crosstalk in broiler chickens: a comprehensive review. Poultry. Basel. 4(4), 44. Dikshit, P., Kumar, J., Das, A., Sadhu, S., Sharma, S., Singh, S., Gupta, P. and Kim, B. 2021. Green synthesis of metallic nanoparticles: applications and limitations. Catalysts 11(8), 902. Ducatelle, R., Goossens, E., Eeckhaut, V. and Van Immerseel, F. 2023. Poultry gut health and beyond. Anim. Nutr. 13, 240–248. Eid, Y., El-Soud, S.A., Gamel, M.Z., El-Kassas, S., Azzam, M.M., Di Cerbo, A., Elolimy, A.A., Alagawany, M. and Kirrella, A.A. 2025. Dietary lysophospholipids enhance broiler performance, immune response, meat quality, and mitigate oxidative stress. Front. Immunol. 16, 1572314. Elmeligy, E.H., Attallah, S.T., Sallam, E.A. and Mohammed, L.S. 2025. Impact of Yucca extract and basil oil supplementation on carcass characteristics, quality of meat, and the cecal microbiota in broiler chickens. J. Adv. Vet. Res. 15(1), 20–27. Erfanian, A., Mirhosseini, H., Manap, M.Y.A., Rasti, B. and Bejo, M.H. 2014. Influence of nano-size reduction on absorption and bioavailability of calcium from fortified milk powder in rats. Food Res. Int. 66, 1–11. FAO/IAEA. 2000. Quantification of tannins in tree foliage. Joint FAO/IAEA Division of Nuclear Techniques in Food and Agriculture. Farag, S.A., El-Keredy, A., Abd El Gawad, S.A., Swelum, A.A., Tellez-Isaias, G. and Abouzeid, A.E. 2024. Impacts of willow (Salix babylonica L.) leaf extract on growth, cecal microbial population, and blood biochemical parameters of broilers. Poult. Sci. 103(3), 103386. Farha, A.K., Yang, Q.Q., Kim, G., Zhang, D., Mavumengwana, V., Habimana, O., Li, H.B., Corke, H. and Gan, R.Y. 2020. Inhibition of multidrug-resistant foodborne Staphylococcus aureus biofilms by a natural terpenoid (+)-nootkatone and related molecular mechanism. Food. Control. 112, 107154. Farkas, V., Mayer, A., Poór, J., Farkas, E.P., Tewelde, K.G., Kiss, B., Such, N., Pál, L., Csitári, G. and Dublecz, K. 2025. Even low amounts of amorphous lignocellulose affect some upper gut parameters, but they do not modify ileal microbiota in young broiler chickens. Animals 15(6), 851. Govindarajan, D.K., Selvaraj, V., Selvaraj, A.S.J.M., Shahul Hameed, S., Pandiarajan, J. and Veluswamy, A. 2023. Green synthesis of silver micro-and nano-particles using phytochemical extracts of Cymbopogon citratus exhibits antibacterial properties. Mater. Today. Proc. 76, 103–108. Gul, S., Hussain, F., Taj, R. and Ullah, A. 2024. Effect of dietary Moringa oleifera on production performance and gut health in broilers. J. Adv. Vet. Anim. Res. 11(2), 339–348. Hu, J.Y., Mohammed, A.A., Murugesan, G.R. and Cheng, H.W. 2022. Effect of a synbiotic supplement as an antibiotic alternative on broiler skeletal, physiological, and oxidative parameters under heat stress. Poult. Sci. 101(4), 101769. Huang, J., Zaynab, M., Sharif, Y., Khan, J., Al-Yahyai, R., Sadder, M., Ali, M., Alarab, S.R. and Li, S. 2024. Tannins as antimicrobial agents: understanding toxic effects on pathogens. Toxicon 247, 107812. Huang, Z., Jin, S. and Lv, Z. 2022. Dietary genistein supplementation alters mRNA expression profile and alternative splicing signature in the thymus of chicks with lipopolysaccharide challenge. Poult. Sci. 101(2), 101561. Jie, Y., Wen, C., Huang, Q., Gu, S., Sun, C., Li, G., Yan, Y., Wu, G. and Yang, N. 2024. Distinct patterns of feed intake and their association with growth performance in broilers. Poult. Sci. 103(9), 103974. Karygianni, L., Ren, Z., Koo, H. and Thurnheer, T. 2020. Biofilm matrixome: extracellular components in structured microbial communities. Trends. Microbiol. 28(8), 668–681. Khan, R.U., Naz, S., Raziq, F., Qudratullah, Q., Khan, N.A., Laudadio, V., Tufarelli, V. and Ragni, M. 2022. Prospects of organic acids as safe alternative to antibiotics in broiler chickens diet. Environ. Sci. Pollut. Res. Int. 29(22), 32594–32604. Khanghahi, M.R.R., Jafari, M.A., Khorshidi, K.J. and Gharahveysi, S. 2024. Effect of feed containing cinnamon and turmeric extract with peroxide oils on the growth performance, antibody titers, cecal microbial population, and jejunum morphology of broiler chickens. Braz. J. Poult. Sci. 26(04), eRBCA-2024. Khatun, P., Sachi, S., Islam, R., Rafiq, K., Islam, P., Dey, B., Hossain, M.T. and Islam, M.Z. 2025. Impact of dietary chia seed supplementation on body weight, breast meat proximate composition, hematological and biochemical parameters, and gut health in broiler chickens. Discov. Appl. Sci. 7(5), 486. Khoddami, A., Wilkes, M. and Roberts, T. 2013. Techniques for analysis of plant phenolic compounds. Molecules 18(2), 2328–2375. Khubeiz, M.M., Algriany, O.A., Elmghirbi, W.M., Bilkhayr, G.R. and Shirif, A.M. 2024. Effect of dietary supplementation of coriander seed (Coriandrum sativum L.) on gut morphology in broiler chickens. Online J. Anim. Feed Res. 14(3), 204–210. Kim, E.S., Baek, Y., Yoo, H.J., Lee, J.S. and Lee, H.G. 2022. Chitosan-tripolyphosphate nanoparticles prepared by ionic gelation improve the antioxidant activities of astaxanthin in the in vitro and in vivo model. Antioxidants. 11(3), 479. Kitagawa, I., Simanjuntak, P., Hori, K., Nagami, N., Mahmud, T., Shibuya, H. and Kobayashi, M. 1994. Indonesian medicinal plants. vii. seven new clerodane-type diterpenoids, peronemins A2, A3, B1, B2, B3, C1, and D1, from the leaves of Peronema canescens (verbenaceae). Chem. Pharm. Bull. 42, 1050–1055. Kumar, R., Thakur, A.K., Chaudhari, P. and Banerjee, N. 2022. Particle size reduction techniques of pharmaceutical compounds for the enhancement of their dissolution rate and bioavailability. J. Pharm. Innov. 17(2), 333–352. Kusmayadi, A., Mardianingrum, R., Wijayanti, D. and Yanti, Y. 2025. The effect of mangosteen rind extract nanocapsule as a feed additive on the growth performance and blood lipid profile of broiler chicken. Open Vet. J. 15(6), 2762. Kyriazakis, I., Dokou, S., Taylor, J., Giannenas, I. and Murphy, E. 2025. A meta-analysis of the sources of variation in the environmental impacts of different broiler production systems. Br. Poult. Sci. 66(2), 193–205. Lelis, C.A., De Carvalho, A.P.A. and Conte Junior, C.A. 2021. A systematic review on nanoencapsulation natural antimicrobials in foods: in vitro versus in situ evaluation, mechanisms of action and implications on physical-chemical quality. Int. J. Mol. Sci. 22(21), 12055. Liang, J., Yan, H., Wang, X., Zhou, Y., Gao, X., Puligundla, P. and Wan, X. 2017. Encapsulation of epigallocatechin gallate in zein/chitosan nanoparticles for controlled applications in food systems. Food Chem. 231, 19–24. Linh, N.T., Qui, N.H. and Triatmojo, A. 2022. The effect of nano-encapsulated herbal essential oils on poultry’s health. Arch. Razi Inst. 77(6), 2013–2021. Liu, F., Yu, J., Chen, Z., Zhang, S., Zhang, Y., Zhang, L., Zhang, Y., Li, J., Ding, L. and Wu, J. 2025. Isolation of Bacillus cereus and its probiotic effect on growth performance, antioxidant capacity, and intestinal barrier protection of broilers. Poult. Sci. 104(4), 104944. Liu, M., Huang, G., Lin, Y., Huang, Y., Xuan, Z., Lun, J., He, S., Zhou, J., Chen, X., Qu, Q., Lv, W. and Guo, S. 2024. Effects of dietary Callicarpa nudiflora aqueous extract supplementation on growth performance, growth hormone, antioxidant and immune function, and intestinal health of broilers. Antioxidants 13(5), 572. Lobiuc, A., Pavăl, N.E., Mangalagiu, I.I., Gheorghiță, R., Teliban, G.C., Amăriucăi-Mantu, D. and Stoleru, V. 2023. Future antimicrobials: natural and functionalized phenolics. Molecules 28(3), 1114. Mahfuz, S., Shang, Q. and Piao, X. 2021. Phenolic compounds as natural feed additives in poultry and swine diets: a review. J. Anim. Sci. Biotechnol. 12(1), 48. Maigoda, T.C., Judiono, J., Purkon, D.B. and Haerussana, A.N.E.M. 2023. Peronema canescens ethanol extract attenuates inflammatory biomarkers and lung damage in ARDS rats animal models induced by LPS. J. Appl. Pharm. Sci. 13, 123–131. Maqsood, I., Sareer, S., Rehman, S.U., Hidayat, A., Shah, M., Awaz, S. and Ke, R. 2025. Dietary supplementation with Jasminum sambac leaf powder: effects on growth, hematology, and immune parameters in broiler chickens. Poult. Sci. 104(1), 104645. Maria Cardinal, K., Kipper, M., Andretta, I. and Machado Leal Ribeiro, A. 2019. Withdrawal of antibiotic growth promoters from broiler diets: performance indexes and economic impact. Poult. Sci. 98(12), 6659–6667. Ma’rifah, B., Suprijatna, E., Sunarti, D., Mahfudz, L.D., Kismiati, S., Sarjana, T.A., Muryani, R., Shihah, H.D., Wahyuni, N.M. and Sugiharto, S. 2025. Effect of encapsulated Averrhoa carambola L. leaves extract on growth performance, haemato-biochemical parameters, gut health, and antioxidant activity of broiler chicken. Vet. Anim. Sci. 30, 100504. Marrelli, M., Conforti, F., Araniti, F. and Statti, G. 2016. Effects of saponins on lipid metabolism: a review of potential health benefits in the treatment of obesity. Molecules 21(10), 1404. Martínez, Y., Paredes, J., Avellaneda, M., Botello, A. and Valdivié, M. 2022. Diets with Ganoderma lucidum mushroom powder and zinc-bacitracin on growth performance, carcass traits, lymphoid organ weights, and intestinal characteristics in broilers. Braz. J. Poult. Sci. 24(01), 2021. Miao, W., Sheng, L., Yang, T., Wu, G., Zhang, M., Sun, J. and Ainiwaer, A. 2020. The impact of flavonoids-rich Ziziphus jujuba Mill. Extract on Staphylococcus aureus biofilm formation. BMC Complement. Med. Ther. 20(1), 187. Minhas, S., Sharma, A. and Chahota, R. 2025. Effects of Chaste tree (Vitex negundo) leaves as an antibiotic growth promoter alternative on growth performance, digestibility, carcass traits, immunity and antioxidant status of broiler birds. Trop. Anim. Health Prod. 57(6), 281. Mohammadi, A., Jafari, S.M., Assadpour, E. and Faridi Esfanjani, A. 2016. Nano-encapsulation of olive leaf phenolic compounds through WPC-pectin complexes and evaluating their release rate. Int. J. Biol. Macromol. 82, 816–822. Mohammed, A.A., Mahmoud, M.A., Zaki, R.S. and Cheng, H.W. 2024. Effect of a probiotic supplement (Bacillus subtilis) on struggling behavior, immune response, and meat quality of shackled broiler chickens exposed to preslaughter stress. Poult. Sci. 103(10), 104051. Mora-Ocación, M.S., Morillo-Coronado, A.C. and Manjarres-Hernández, E.H. 2022. Extraction and quantification of saponins in quinoa (Chenopodium quinoa Willd.) genotypes from Colombia. Int. J. Food. Sci. 1(1), 7287487. Navarro Del Hierro, J., Casado-Hidalgo, G., Reglero, G. and Martin, D. 2021. The hydrolysis of saponin-rich extracts from fenugreek and quinoa improves their pancreatic lipase inhibitory activity and hypocholesterolemic effect. Food Chem. 338, 128113. Nouri, A. 2019. Chitosan nano-encapsulation improves the effects of mint, thyme, and cinnamon essential oils in broiler chickens. Br. Poult. Sci. 60(5), 530–538. Obianwuna, U.E., Chang, X., Oleforuh-Okoleh, V.U., Onu, P.N., Zhang, H., Qiu, K. and Wu, S. 2024. Phytobiotics in poultry: revolutionizing broiler chicken nutrition with plant-derived gut health enhancers. J. Anim. Sci. Biotechnol. 15(1), 169. OECD/FAO. 2025. OECD–FAO Agricultural Outlook 2025–2034. Paris, France: OECD Publishing, ; Rome, Italy: FAO. Ognik, K., Stępniowska, A., Cholewińska, E. and Kozłowski, K. 2016. The effect of administration of copper nanoparticles to chickens in drinking water on estimated intestinal absorption of iron, zinc, and calcium. Poult. Sci. 95(9), 2045–2051. Omar, A.E., Al-Khalaifah, H.S., Mohamed, W.A.M., Gharib, H.S.A., Osman, A., Al-Gabri, N.A. and Amer, S.A. 2020. Effects of phenolic-rich onion (Allium cepa L.) extract on the growth performance, behavior, intestinal histology, amino acid digestibility, antioxidant activity, and the immune status of broiler chickens. Front. Vet. Sci. 7, 582612. Osman, A.I., Zhang, Y., Farghali, M., Rashwan, A.K., Eltaweil, A.S., Abd El-monaem, E.M., Mohamed, I.M.A., Badr, M.M., Ihara, I., Rooney, D.W. and Yap, P.S. 2024. Synthesis of green nanoparticles for energy, biomedical, environmental, agricultural, and food applications: a review. Environ. Chem. Lett. 22(2), 841–887. Ozdal, M. and Gurkok, S. 2022. Recent advances in nanoparticles as antibacterial agent. ADMET DMPK 10(2), 115–129. Park, J.H. and Kim, I.H. 2020. Effects of dietary Achyranthes japonica extract supplementation on the growth performance, total tract digestibility, cecal microflora, excreta noxious gas emission, and meat quality of broiler chickens. Poult. Sci. 99(1), 463–470. Paul, S.S., Rama, R.S.V., Hegde, N., Williams, N.J., Chatterjee, R.N., Raju, M.V.L.N., Reddy, G.N., Kumar, V., Kumar, P.S.P., Mallick, I.S. and Gargi, M. 2022. Effects of dietary antimicrobial growth promoters on performance parameters and abundance and diversity of broiler chicken gut microbiome and selection of antibiotic resistance genes. Front. Microbiol. 13, 905050. Pelvan, E., Karaoğlu., Fırat, E., Kalyon, K.B., Ros, E. and Alasalvar, C. 2022. Immunomodulatory effects of selected medicinal herbs and their essential oils: a comprehensive review. J. Funct. Foods. 94, 105–108. Pereira Farias, N.N., Freitas, E.R., Nepomuceno, R.C., Marques Gomes, H., Herik Souza, D., Costa, M.K.D.O., Soares Da Costa, H., Fernandes, D.R., Santos Araújo, L.R., Jerônimo Do Nascimento, G.A., Abreu De Melo, M.C. and Watanabe, P.H. 2021. Ethanolic extract of mango seed in broiler feed: effect on productive performance, segments of the digestive tract and blood parameters. Anim. Feed. Sci. Technol. 279, 114999. Picos-Corrales, L.A., Morales-Burgos, A.M., Ruelas-Leyva, J.P., Crini, G., García-Armenta, E., Jimenez-Lam, S.A., Ayón-Reyna, L.E., Rocha-Alonzo, F., Calderón-Zamora, L., Osuna-Martínez, U., Calderón-Castro, A., De-Paz-Arroyo, G. and Inzunza-Camacho, L.N. 2023. Chitosan as an outstanding polysaccharide improving health-commodities of humans and environmental protection. Polymers 15(3), 526. Popat, A., Liu, J., Lu, G.Q. and Qiao, S.Z. 2012. A pH-responsive drug delivery system based on chitosan coated mesoporous silica nanoparticles. J. Mater. Chem. 22, 11173–11178. Prakash, A., Saxena, V.K. and Singh, M.K. 2020. Genetic analysis of residual feed intake, feed conversion ratio and related growth parameters in broiler chicken: a review. World's. Poultry. Sci. J. 76(2), 304–317. Prakatur, I., Miskulin, M., Pavic, M., Marjanovic, K., Blazicevic, V., Miskulin, I. and Domacinovic, M. 2019. Intestinal morphology in broiler chickens supplemented with propolis and bee pollen. Animals 9(6), 301. Rahardhian, M.R.R., Sumiwi, S.A., Susilawati, Y. and Muchtaridi, M. 2025. Immunomodulatory potential of kaempferol isolated from Peronema canescens Jack. leaves through inhibition of IL-6 expression. Int. J. Mol. Sci. 26(7), 3068. Rahma, S.A., Mulyono, M., Yunianto, V.D. and Krismiyanto, L. 2025. Synergistic effects of noni fruit extract, copper, and zinc encapsulated on immunity, protein digestibility, and growth performance in broiler. J. Adv. Vet. Res. 15(6), 750–754. Rahmani, M.M., Ding, W., Wei, Q., Sun, J., Hou, L., Elsaid, S.H., Ali, I., Zhou, W. and Shi, F. 2025. Impact of fermented bamboo powder on the morphology and physiology of the gastrointestinal tract in yellow-feather broiler chickens. Poult. Sci. 104(2), 104793. Rizki, M., Pratama, F., Sutomo, S., Arnida, A., Yulistati, F.R. and Normaidah, N. 2022. Pharmacognostic study and antioxidant activity of sungkai (Peronema canescens Jack.) methanol extract from Indonesia. Bull. Pharm. Sci. Assiut. Univ. 45(2), 655–665. Rysman, K., Eeckhaut, V., Ducatelle, R., Goossens, E. and Van Immerseel, F. 2023. Broiler performance correlates with gut morphology and intestinal inflammation under field conditions. Avian. Pathol. 52(4), 232–241. Sabry, M.I.E. and El-Ghany, W.A.A. 2021. A mathematical model for calculating intestinal villus surface area in broiler chickens. Int. J. Vet. Sci. 10(4), 317–322. Samreen., Qais, F.A. and Ahmad, I. 2022. Anti-quorum sensing and biofilm inhibitory effect of some medicinal plants against gram-negative bacterial pathogens: in vitro and in silico investigations. Heliyon 8(10), e11113. Sanneur, K., Leksawasdi, N., Sumonsiri, N., Techapun, C., Taesuwan, S., Nunta, R. and Khemacheewakul, J. 2023. Inhibitory effects of saponin-rich extracts from Pouteria cambodiana against digestive enzymes α-Glucosidase and pancreatic lipase. Foods 12(20), 3738. Shalihin, M.I., Khatib, A., Yusnaidar, Y., Tarigan, I.L. and Latief, M. 2024. An in-vogue plant, Peronema canescens Jack: traditional uses and scientific evidence of its bioactivities. Discover. Plants. 1(1), 58. Shang, Y., Kumar, S., Oakley, B. and Kim, W.K. 2018. Chicken gut microbiota: importance and detection technology. Front. Vet. Sci. 5, 254. Shani, M.A. and Irani, M. 2024. Feeding strategy and prebiotic supplementation: effects on immune responses and gut health in the early life stage of broiler chickens. Res. Vet. Sci. 171, 105226. Sugiharto, S., Zulpa, Y., Agusetyaningsih, I., Widiastuti, E., Wahyuni, H.I., Yudiarti, T. and Sartono, T.A. 2024. Physiological responses and intestinal conditions of broiler chickens treated with encapsulated Acalypha australis L. leaf extract and chitosan. Vet. World. 17(5), 994–1000. Tamfu, A.N., Ceylan, O., Kucukaydin, S., Ozturk, M., Duru, M.E. and Dinica, R.M. 2020. Antibiofilm and enzyme inhibitory potentials of two annonaceous food spices, African pepper (Xylopia aethiopica) and African nutmeg (Monodora myristica). Foods 9(12), 1768. Thuekeaw, S., Angkanaporn, K. and Nuengjamnong, C. 2022. Microencapsulated basil oil (Ocimum basilicum Linn.) enhances growth performance, intestinal morphology, and antioxidant capacity of broiler chickens in the tropics. Anim. Bioscience. 35(5), 752. Uehara, A., Maekawa, M. and Nakagawa, K. 2023. Enhanced intestinal barrier function as the mechanism of antibiotic growth promoters in feed additives. Biosci. Biotechnol. Biochem. 87, 1381–1392. Waghmare, S., Gupta, M., Bahiram, K.B., Korde, J.P., Bhat, R., Datar, Y., Rajora, P., Kadam, M.M., Kaore, M. and Kurkure, N.V. 2025. Effects of organic acid blends on the growth performance, intestinal morphology, microbiota, and serum lipid parameters of broiler chickens. Poult. Sci. 104(1), 104546. Wang, C., Chen, D., Wu, S., Zhou, W., Chen, X., Zhang, Q. and Wang, L. 2024. Dietary supplementation with Neolamarckia cadamba leaf extract improves broiler meat quality by enhancing antioxidant capacity and regulating metabolites. Anim. Nutr. 17, 358–372. Wawrzyniak, A., Kapica, M., Stępień-Pyśniak, D., Łuszczewska-Sierakowska, I., Szewerniak, R. and Jarosz, Ł. 2017. The effect of dietary supplementation of Transcarpathian zeolite on intestinal morphology in female broiler chickens. J. Appl. Poult. Res. 26(3), 421–430. Yadav, S. and Jha, R. 2019. Strategies to modulate the intestinal microbiota and their effects on nutrient utilization, performance, and health of poultry. J. Anim. Sci. Biotechnol. 10(1), 2. Yasmin, S., Yousaf, M.S., Majeed, K.A., Rashid, M.A., Tahir, S.K., Numan, M., Mustafa, R., Nagra, S.I., Zaneb, H. and Rehman, H. 2021. Dietary Catharanthus roseus modulates intestinal microarchitecture in broilers. S. Afr. J. Anim. Sci. 51(4), 488–496. Zamanizadeh, A., Mirakzehi, M.T., Agah, M.J., Saleh, H. and Baranzehi, T. 2021. A comparison of two probiotics Aspergillus oryzae and Saccharomyces cerevisiae on productive performance, egg quality, small intestinal morphology, and gene expression in laying Japanese quail. Italian J. Anim. Sci. 20(1), 232–242. Table S1. pH of the PcNP components, PcNP stock, and drinking water.
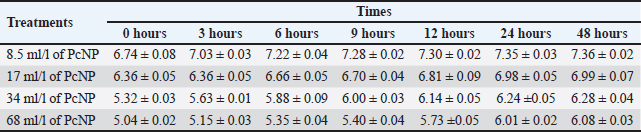
Table S2. pH dose of PcNP.
| ||
| How to Cite this Article |
| Pubmed Style Maharani BP, Sanjaya HL, Baskara AP, Zuprizal Z, Ariyadi B, Martien R, Hanim C, Rusman R, Dono ND. Supplementation of nanoencapsulated Peronema canescens jack leaf extract on growth performance, carcass traits, digestive characteristics, and jejunum histomorphometry in broiler chickens. Open Vet. J.. 2026; 16(1): 287-302. doi:10.5455/OVJ.2026.v16.i1.26 Web Style Maharani BP, Sanjaya HL, Baskara AP, Zuprizal Z, Ariyadi B, Martien R, Hanim C, Rusman R, Dono ND. Supplementation of nanoencapsulated Peronema canescens jack leaf extract on growth performance, carcass traits, digestive characteristics, and jejunum histomorphometry in broiler chickens. https://www.openveterinaryjournal.com/?mno=281429 [Access: January 31, 2026]. doi:10.5455/OVJ.2026.v16.i1.26 AMA (American Medical Association) Style Maharani BP, Sanjaya HL, Baskara AP, Zuprizal Z, Ariyadi B, Martien R, Hanim C, Rusman R, Dono ND. Supplementation of nanoencapsulated Peronema canescens jack leaf extract on growth performance, carcass traits, digestive characteristics, and jejunum histomorphometry in broiler chickens. Open Vet. J.. 2026; 16(1): 287-302. doi:10.5455/OVJ.2026.v16.i1.26 Vancouver/ICMJE Style Maharani BP, Sanjaya HL, Baskara AP, Zuprizal Z, Ariyadi B, Martien R, Hanim C, Rusman R, Dono ND. Supplementation of nanoencapsulated Peronema canescens jack leaf extract on growth performance, carcass traits, digestive characteristics, and jejunum histomorphometry in broiler chickens. Open Vet. J.. (2026), [cited January 31, 2026]; 16(1): 287-302. doi:10.5455/OVJ.2026.v16.i1.26 Harvard Style Maharani, B. P., Sanjaya, . H. L., Baskara, . A. P., Zuprizal, . Z., Ariyadi, . B., Martien, . R., Hanim, . C., Rusman, . R. & Dono, . N. D. (2026) Supplementation of nanoencapsulated Peronema canescens jack leaf extract on growth performance, carcass traits, digestive characteristics, and jejunum histomorphometry in broiler chickens. Open Vet. J., 16 (1), 287-302. doi:10.5455/OVJ.2026.v16.i1.26 Turabian Style Maharani, Bella Putri, Hafi Luthfi Sanjaya, Aji Praba Baskara, Zuprizal Zuprizal, Bambang Ariyadi, Ronny Martien, Chusnul Hanim, Rusman Rusman, and Nanung Danar Dono. 2026. Supplementation of nanoencapsulated Peronema canescens jack leaf extract on growth performance, carcass traits, digestive characteristics, and jejunum histomorphometry in broiler chickens. Open Veterinary Journal, 16 (1), 287-302. doi:10.5455/OVJ.2026.v16.i1.26 Chicago Style Maharani, Bella Putri, Hafi Luthfi Sanjaya, Aji Praba Baskara, Zuprizal Zuprizal, Bambang Ariyadi, Ronny Martien, Chusnul Hanim, Rusman Rusman, and Nanung Danar Dono. "Supplementation of nanoencapsulated Peronema canescens jack leaf extract on growth performance, carcass traits, digestive characteristics, and jejunum histomorphometry in broiler chickens." Open Veterinary Journal 16 (2026), 287-302. doi:10.5455/OVJ.2026.v16.i1.26 MLA (The Modern Language Association) Style Maharani, Bella Putri, Hafi Luthfi Sanjaya, Aji Praba Baskara, Zuprizal Zuprizal, Bambang Ariyadi, Ronny Martien, Chusnul Hanim, Rusman Rusman, and Nanung Danar Dono. "Supplementation of nanoencapsulated Peronema canescens jack leaf extract on growth performance, carcass traits, digestive characteristics, and jejunum histomorphometry in broiler chickens." Open Veterinary Journal 16.1 (2026), 287-302. Print. doi:10.5455/OVJ.2026.v16.i1.26 APA (American Psychological Association) Style Maharani, B. P., Sanjaya, . H. L., Baskara, . A. P., Zuprizal, . Z., Ariyadi, . B., Martien, . R., Hanim, . C., Rusman, . R. & Dono, . N. D. (2026) Supplementation of nanoencapsulated Peronema canescens jack leaf extract on growth performance, carcass traits, digestive characteristics, and jejunum histomorphometry in broiler chickens. Open Veterinary Journal, 16 (1), 287-302. doi:10.5455/OVJ.2026.v16.i1.26 |