| Review Article | ||
Open Vet. J.. 2026; 16(1): 56-70 Open Veterinary Journal, (2026), Vol. 16(1): 56-70 Review Article A review of rabies in Southern African wildlife: Reservoir hosts, transmission dynamics, and control implicationsUkamaka Uchenna Eze1,2* and Claude Taurai Sabeta21Department of Veterinary Medicine, Faculty of Veterinary Medicine, University of Nigeria, Enugu, Nigeria 2Department of Veterinary Tropical Diseases, Faculty of Veterinary Science, University of Pretoria, Pretoria, South Africa *Corresponding Author: Ukamaka Uchenna Eze. Department of Veterinary Medicine, Faculty of Veterinary Medicine, University of Nigeria, Enugu, Nigeria. Email: ukamakauchenna.eze [at] unn.edu.ng Submitted: 27/08/2025 Revised: 10/12/2025 Accepted: 26/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
AbstractRabies is a fatal zoonotic disease that affects warm-blooded vertebrates and is caused primarily by members of the Lyssavirus genus (family Rhabdoviridae, order Mononegavirales). Three Lyssavirus species, namely Lyssavirus duvenhage and the putative lyssaviruses Lyssavirus matlo and Lyssavirus phyla, were first identified in insectivorous bat species in South Africa through lyssavirus surveillance activities. For the proposed and effective global elimination of dog-mediated human rabies by 2030; it is important to contextualize the pivotal role of wildlife in the maintenance and the likely spillover of rabies virus infections into naive dog populations that could potentially derail the progress toward rabies elimination. This review on rabies in wildlife identifies the current reservoir host species for sylvatic rabies, species predominantly affected by rabies (and non-rabies lyssaviruses) in southern African wildlife, and rabies transmission dynamics between these species. Canid rabies is maintained in a variety of southern African wildlife carnivores, including the black-backed jackal species (Canis mesomelas), side-striped jackals (Canis adustus), bat-eared foxes (Otocyon megalotis), and most recently, aardwolves (Proteles cristatus). In addition, the yellow mongooses (Cynictis penicillata) and slender mongooses (Galerella sanguinea) are reservoirs of the mongoose rabies virus (RABV) biotype (in South Africa and Zimbabwe, respectively). In Namibia, rabies virus infection is associated with wild ruminants, especially the kudu antelope (Tragelaphus strepsiceros), thereby posing a conservation threat to specific wildlife species. Oral rabies vaccination of wild carnivores using baited vaccines has been used as a complementary approach to mass dog vaccinations to mitigate infection spillover into domestic host species. Keywords: Sylvatic rabies, Wild carnivores, Transmission dynamics, Control, Southern Africa. IntroductionRabies is an ancient disease caused by the Lyssavirus rabies (RABV) and upon infection, generally through a bite contact is characterized by progressive encephalomyelitis in both humans and animals (Kumar et al., 2023; Centers for Disease Control and Prevention, 2023). Active rabies surveillance and elimination programs are aimed toward zero dog-mediated human rabies deaths by 2030. Hence, evaluating the current status of rabies in southern Africa with specific reference to animal species affected by the disease and its spread in the region is imperative. This review will enhance our understanding of rabies epidemiology, transmission dynamics, and the implications of sylvatic rabies in the context of rabies control in the sub-region. The African continent sustains a great biological diversity of lyssaviruses considering that six (Lyssavirus rabies, Lyssavirus lagos. Lyssavirus mokola, Lyssavirus duvenhage, Lyssavirus shimoni, and Lyssavirus ikoma) out of the 17 confirmed lyssavirus species in the world were first identified in the continent (Boulger and Porterfield, 1958; Shope et al., 1970; Meredith et al., 1971; Kuzmin et al., 2010; Marston et al., 2012), leading to the hypothesis that Africa is probably the birthplace of lyssaviruses and their subsequent evolution and global spread (Hayman et al., 2016). Lyssaviruses are delineated into three phylogroups (I–III) based on phylogeny, pathogenicity, and serological cross-reactivity (Kuzmin et al., 2005; International Committee on Taxonomy of Viruses, 2024). The Lyssavirus species found in Africa are represented in each of the three phylogroups, showing the continent’s biological diversity. Lyssaviruses originating from Africa include L. rabies and L. duvenhage (Phylogroup I), L. lagos, L. mokola, and L. shimoni (Phylogroup II), and L. ikoma (Phylogroup III) (Kuzmin et al., 2010; International Committee on Taxonomy of Viruses, 2024). Some lyssaviruses on the African continent were identified in the late 1950s, e.g., Lyssavirus lagos was first isolated from a fruit-eating bat (Eidolon helvum) in 1956 on Lagos Island in Nigeria (Boulger and Porterfield, 1958; Shope et al., 1970); L. mokola was originally isolated in 1968 from pooled viscera of three insectivorous shrews (Crocidura spp.) from the Mokola District of Nigeria (Shope et al., 1970); L. duvenhage, was first encountered in South Africa in 1970 following a human fatality that was associated with a contact exposure involving an unidentified bat, most likely an insect-eating bat (Meredith et al., 1971). In 2009, L. shimoni (SHIBV) was isolated from the brain of a dead Commerson’s leaf-nosed bat (Hipposideros commersoni), which was found in a cave in the coastal region of Kenya (Kuzmin et al., 2010). Lyssavirus ikoma (IKOV), a once-off isolate, was recovered from an African civet (Civettictis civetta) that manifested clinical signs consistent with rabies virus infection and was killed by rangers in Ikoma Ward within Serengeti National Park (Tanzania) in 2009 (Marston et al., 2012). Recently, two putative lyssaviruses, Lyssavirus matlo (MBLV) and Lyssavirus phala (PBLV), were identified in two Miniopterus natalensis (Natal long-fingered) bats in 2015 and 2016, and a Nycticeinops schlieffeni (Schlieffen’s) bat in 2021, respectively, in the northern Limpopo Province of South Africa (Viljoen et al., 2023). With the exception of L. mokola and L. ikoma, all other Lyssaviruses originating from Africa were first isolated from bat species (order: Chiroptera), highlighting the importance of bats in the global epidemiology of lyssaviruses (Kuzmin et al., 2011; International Committee on Taxonomy of Viruses, 2024). This observation supports the spillover of bat lyssaviruses into terrestrial mammals, a historical event believed to have occurred approximately four thousand years ago (Bourhy et al., 2008). The recent and new isolations of lyssaviruses from chiroptera support current theories that suggest that lyssaviruses originated in old world bats, the reservoir host species for most of the currently known lyssavirus species (Bourhy et al., 2008; Rupprecht et al., 2011; Banyard., 2014; Hayman et al., 2016; International Committee on Taxonomy of Viruses, 2024). An overview of the history of rabies in Southern AfricaSouthern Africa is made up of 10 countries as shown in Figure 1.
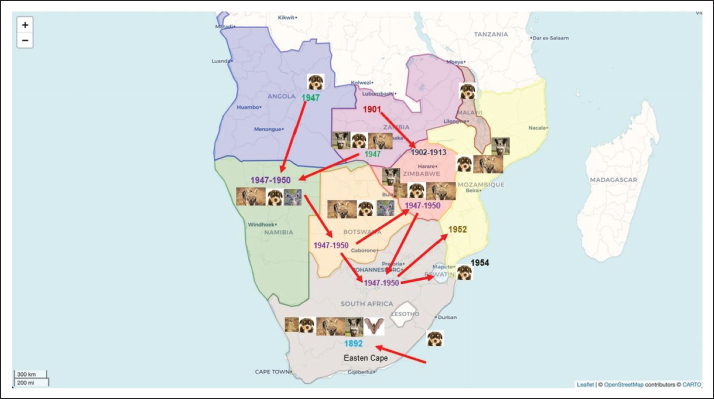
Fig. 1. Map showing the movement and spread of canid rabies in southern Africa. A dog imported from England apparently introduced canid rabies into South Africa’s Eastern Cape Province in 1892. (Swanepoel et al., 1993). This specific outbreak was confined to dogs, cats, and cattle, and no wildlife hosts were involved. This period coincided with the rise of European colonization of the region. The disease was then brought under control through the restriction of dogs and destruction of strays (1894). At the turn of the century, rabies outbreaks in Zambia (then Northern Rhodesia) apparently continued to spread along the major trade routes and initiated outbreak(s) in Zimbabwe (then Southern Rhodesia) in 1902, affecting most of the country before it was eradicated in 1913 (Swanepoel et al., 1993). Rabies was first recorded in Botswana in 1919, but the first confirmed outbreak of animal rabies in the country occurred in 1938 during a dog rabies outbreak in the Ngamiland district (Swanepoel et al., 1993). In the late 1940s, an invasive wave of rabies was spread by dogs from Angola into Namibia and then simultaneously entered northern and eastern Botswana into Zimbabwe and then the Limpopo Province (Northern Province) by 1950 (Fig. 1). This spread continued into Mozambique in 1952, Eswatini in 1954, southern Mozambique, and KwaZulu/Natal (KZN) in 1961, causing a major epidemic before it was brought under control in 1968. Therefore, it took approximately 21 years from 1947 for rabies to spread to the whole of southern Africa (Fig. 1). Rabies outbreaks recurred in Mozambique in 1976 and then spread to KZN, then to Lesotho in 1982, before reaching the eastern Cape province in 1987 (Swanepoel et al., 1993). To date, rabies is still spreading among domestic dogs in different regions of the country, but also specific wildlife canid hosts, notably black-backed jackals, side-striped jackals, bat-eared foxes, and more recently, aardwolves, highlighting the active circulation of the rabies virus in both domestic and wildlife hosts (Swanepoel, 2004; Sabeta et al., 2007; Ngoepe et al., 2022). Although the rabies virus was also maintained in wildlife, spillover events caused outbreaks in domestic and wild ruminants in Namibia, Botswana, Zimbabwe, and South Africa (Swanepoel et al., 1993; Mansfield et al., 2006; Hikufe et al., 2019). In addition to the canid rabies variant maintained in wildlife having spilled over from domestic dogs, current evidence supports that a different rabies biotype, the mongoose rabies biotype, is indigenous to the region (particularly South Africa) and has been estimated to have diverged approximately 200 years ago (1800s), long before canid rabies was introduced (Van Zyl et al., 2010). It was initially thought that the bite of a genet (Genetta genetta), a member of the Viverridae family, causes madness and death (Cluver, 1927; Snyman, 1940); hence, the misnomer of Viverrid rabies. The viverrid rabies (now known and correctly referred to as mongoose rabies) has continued to occur widely in southern African countries, with spillover of infection to cattle and a variety of other domestic and wildlife species. However, the spillover of infection of the mongoose rabies biotype into domestic dogs does not lead to secondary virus infections but to dead-end infections (Ngoepe et al., 2009; Sabeta et al., 2020). MethodologySearch procedureUsing the method previously described by Tran et al. (2020), we searched three public databases, namely, Web of Science, PubMed, and CAB, using the following search terms:
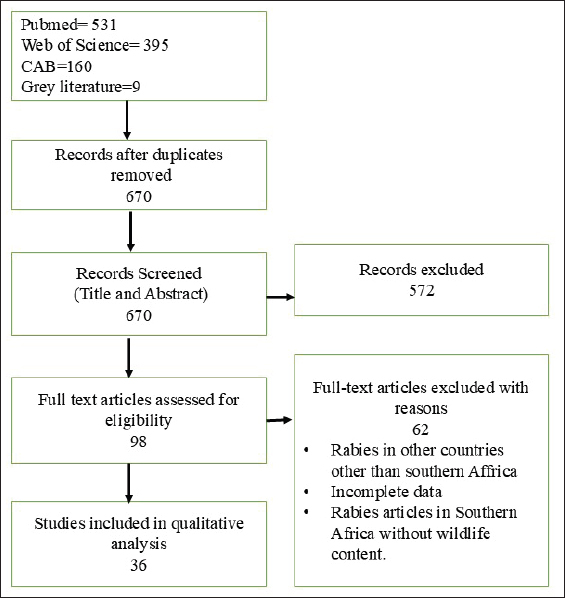
The literature search was conducted between April and October 2024 (Fig. 2). The selected titles, abstracts, and full texts were screened for eligibility, and those that did not meet the inclusion criteria were eliminated. References of related studies were also searched and included if they met the inclusion criteria.
Fig. 2. Prisma flow diagram showing study selection for systematic review of rabies in southern Africa, literature screening and selection process. Screening criteriaThe inclusion criteria for an article in the study included the following: originality, peer-review, and inclusion of rabies in wildlife in any of the southern African countries. There were very few articles that were solely on rabies in wildlife in southern Africa; thus, we included those articles that had other animal species, such as domestic dogs and humans, provided that the content included wildlife rabies. A total of 36 articles met the inclusion criteria (Fig. 2). All articles we came across from 1927 to 2024 on rabies in wildlife in southern African countries were included in the study. Exclusion criteriaArticles were excluded if they did not focus on rabies in wildlife or if rabies in wildlife was not mentioned in the content. Studies on rabies in wildlife conducted outside the southern African region, review papers without primary data, conference abstracts, editorials, and non-peer-reviewed materials were also excluded from this study. Duplicate publications, articles without accessible full text, or articles published in languages other than English were excluded from the analysis. Sylvatic rabies in Southern AfricaSouthern Africa is endowed with a wide variety of eco-regions, including grassland, bushveld, karoo, savannah, and riparian zones (Marks, 2024). Southern Africa’s climate (both sub-tropical and temperate) and geographical terrain (forests, grasslands, and deserts) enable it to support the life of diverse fauna, including a wide variety of wild carnivores (Cowling et al., 2004). The occurrence and geographical distribution of diverse sylvatic reservoir species that can maintain and transmit rabies complicate the epidemiology of rabies in southern Africa. Furthermore, the close proximity of human habitats to wildlife conservation areas enhances transmission from domestic dogs to wildlife species and vice versa. Since the first recognition of rabies in southern Africa, its occurrence has been primarily associated with specific wildlife species. At the outset, a few species were recognized as being involved and are still important in wildlife rabies. However, over the years, many other species have been identified to play a role in rabies transmission in the wild, either as reservoir, incidental, or dead-end hosts. In this paper, we look at the species that have played some roles in the evolving rabies situation in southern Africa and the factors influencing that situation. Rabies in JackalsDomestic dogs were believed to be solely responsible for the rabies epizootic that originated in Angola and Zambia in the mid-1940s. However, during that period, jackals became an important reservoir in the following countries: Zimbabwe, Botswana, and Namibia and remain so till date (Snyman, 1940; Meredith, 1982). Furthermore, in South Africa, there were recorded cases of rabies in the jackal, Canis mesomelas, from 1933 onwards (Meredith, 1982). In southern Africa, two jackal species, the black-backed jackal and side-striped jackal, are found in the region. The jackal species mostly inhabits the bushveld areas of southern Africa, particularly northern South Africa, Limpopo Province, Zimbabwe, and Namibia, which are dominated by subsistence, commercial farmlands, and ranches, thus giving them access to domestic ruminants. The black-backed jackal is the main maintenance host of the canid rabies biotype, particularly in farming areas where conditions are suitable and promote the proliferation of jackals to densities optimal enough to maintain rabies outbreaks (Sabeta et al., 2007; Zulu et al., 2009), independent of other host species (Bingham et al., 1999). Black-backed jackal species maintain the canid rabies biotype in several southern African countries, namely, South Africa, Zimbabwe, and Namibia, whereas Canis adustus in Malawi, Zambia, and Zimbabwe (Hussein et al., 1984; Everard and Everard, 1988; Everard and Everard, 1992; Von Teichman et al., 1995; Rhodes et al., 1998; Bingham et al., 1999; Swanepoel, 2004; Nel et al., 2005; Sabeta et al., 2007; Munang’Andu et al., 2011; World Health Organization, 2013). Contrary to C. mesomelas, C. adustus may not be able to sustain and successfully transmit rabies to dogs, as the latter does not reach threshold densities to maintain rabies cycles (Sabeta et al., 2003; Bingham et al., 1999). In the northern regions of South Africa, the black-backed jackal population has been shown to sustain rabies cycles (Cumming, 1982; Bingham et al., 1999), and spillover of infection to other species, such as domestic dogs, cattle, and other ruminants, is common (Brückner et al., 1978; Barnard, 1979). In 1997, rabies in black-backed jackals almost exterminated a pack of African wild dogs in the Madikwe game reserve in South Africa (Hofmeyr et al., 2000). Between 1997 and 1998, rabies in jackals was reported to be 6.5% of the total rabies cases (1071) in South Africa (Bishop et al., 2002). In 2012, a rabies outbreak in the black-backed jackal population in the uThukela area of KZN (South Africa) was observed for the first time outside its normal range in the northern and north-western regions of the country. This specific outbreak highlighted that a wildlife host species could sustain a rabies outbreak independent of domestic dogs in KZN, a region historically associated with dog rabies since the late 1970s. In this outbreak, 62% of the positive rabies cases originated from jackals, which is similar to the observations made in 2016, where 53.8% of the laboratory-confirmed lyssaviruses were attributed to this wildlife host species in Gauteng Province (Ngoepe et al., 2022). Jackals were also found to be the most commonly affected wildlife species between 1998 and 2017 and accounted for 46% (n=255) of 560 confirmed rabies-positive cases in wildlife species (Mogano et al., 2022). Wildlife (jackals) and domestic animals (dogs) frequently come into contact in both Zimbabwe and South Africa, creating ideal opportunities for rabies transmission between the two host species. The highly opportunistic nature of the canid rabies variant drives these interactions (Ngoepe et al., 2022). In Zimbabwe, domestic dogs were recognized as potential sources of rabies during wildlife rabies outbreaks in jackals, and these were always preceded by outbreaks in domestic dog populations (Bingham et al., 1999). However, there are reports of jackals initiating outbreaks in dogs when the front of the epidemic reached an area with a large dog population, as seen in the final stage of a large epidemic in 1982 in Zimbabwe (Bingham and Foggin, 1993). In a genetic analysis of rabies viruses from a variety of host species in Zambia, Zimbabwe, and South Africa (Muleya et al., 2019), rabies viruses that occurred in the different hosts and regions were indistinguishable. Similarly, in Zimbabwe, rabies viruses from both dogs and jackals showed similar reactivity patterns on monoclonal antibody panels (King et al., 2004) and belonged to the same genetic clusters, demonstrating that rabies viruses from both wildlife and domestic animals are indistinguishable. In addition, these findings support the common historical origin of dog and jackal rabies viruses in southern Africa. In Zimbabwe, African civet rabies cases were often diagnosed during jackal rabies outbreaks, indicating spillover of infection among wildlife carnivore species (Sabeta et al., 2020). In a retrospective study of wildlife rabies conducted in Zimbabwe between 1992 and 2003, 78.9% of 1,540 rabies cases were from jackals (C. mesomelas and C. adustus) (Pfukenyi et al., 2009). Between 1986 and 1992, jackals accounted for 2.2% of the reported rabies cases in Malawi (Nel and Rupprecht, 2007). Records from veterinary laboratories (Blantyre, Mzuzu, and Central Veterinary Laboratories) in Malawi showed that the total number of rabies-positive jackal cases recorded between 1991 and 2000 was 1% (n=15) out of 1,488 rabies-positive animal cases (Chimera and Chikungwa, 2001). This low number could be due to the lack of rabies surveillance in wildlife species at the time. However, data from another study conducted in Malawi from 2008 to 2021 showed that of the 26 confirmed rabies-positive cases, the side-stripped jackals were responsible for 30.8% (Kainga et al., 2023), indicating increased involvement of jackals in the maintenance and transmission of rabies in the wild or increased surveillance. According to data on confirmed cases of wildlife rabies at the National Veterinary Laboratory Botswana, between 1991 and 1998, 64.1% (%) of 233 cases were from jackals (World Health Organization, 1999). Furthermore, an 18-year retrospective study (1989–2006) conducted in Botswana using a cohort of 2,419 rabies cases revealed that 14.2% (%) of the cases originated from wild carnivore species and 67% (%) were of jackal origin (Moagabo et al., 2009). A study conducted in Namibia between 2011 and 2017 revealed that of the 419 wildlife species that tested positive for rabies antigen, 41 (9.8%) were from jackals (Hifuke et al., 2019). Jackal rabies has been reported to occur during epidemics. For instance, a large epidemic that occurred in jackals in the Mashonaland area of Zimbabwe from 1979 to 1982 moved up to 20 km/month and lasted for 12 months before dying out (Foggin, 1988). The main victims of jackal rabies are cattle, as there is a significant association between cattle and jackal cases. In addition, rabies outbreaks in jackals have been associated with a high incidence of rabies in other wildlife species, such as honey badgers, civets, mongooses, and antelope species (Bingham and Foggin, 1993). In general, reports have shown that jackal rabies peaks in winter and recedes during summer. This cycle could be associated with territory establishment and increased contact rates due to scarcity of food and water in winter and care of the young in summer (Bingham and Foggin, 1993). Rabid jackals lose their fear of humans and approach their homesteads during broad daylight to attack humans and animals; in these circumstances, they are often killed by domestic dogs. The domestic dog, if not vaccinated, will contact rabies in the process, and the spillover of infection continues (Foggin, 1988). In conclusion, rabies in jackals is a key factor in the maintenance and transmission of the virus within wildlife and between wildlife and domestic animals in southern Africa. The black-backed jackal serves as the main maintenance host of the canid rabies biotype, capable of sustaining independent transmission cycles. Jackals can maintain rabies epidemics and share genetically similar virus strains with domestic dogs, indicating ongoing interspecies transmission. Controlling rabies in jackals should therefore be an integral component of rabies elimination strategies in southern Africa. Rabies in FoxesBat-eared foxes inhabit the arid grasslands and savannas of East and Southern Africa. They can dig and live in dens that are dug by themselves or other animals, such as aardwolves. Cape foxes prefer the sub-Saharan African deserts of South Africa, Namibia, and Botswana (Kruger National Park, 2024). Bat-eared foxes (Otocyon megalotis) and the Cape fox (Vulpes chama) are pivotal in rabies transmission dynamics, with the former being a major maintenance host of canid rabies in wildlife in South Africa, Zambia, Zimbabwe, Botswana, Namibia, and Angola (Swanepoel, 2004; Sabeta et al., 2007; Mogano et al., 2022). The latter could be an incidental host in Botswana, Eswatini, and Namibia. Rabies was first diagnosed in bat-eared foxes in the mid-1950s in Limpopo province, South Africa (Swanepoel et al., 1993). Since then, this species has played a significant role in the maintenance and spread of RABV infection (Sabeta et al., 2007). A study conducted in Botswana for 18 years (1989–2006) showed that 11 (3.21%) of 343 cases from wild carnivore species were from foxes (Moagabo et al., 2009). In comparison to the Republic of South Africa, it appears that the bat-eared fox plays a minor role in the sylvatic rabies cycle in Zimbabwe, as only 0.13% (2/1,574) of laboratory-confirmed wildlife cases were reported in the country for the 36-year period of 1950 to 1986 (Foggin, 1988), and only 0.06% (1/1,540) was confirmed between 1992 and 2003 (Pfukenyi et al., 2009). Of the 419 wildlife-positive rabies cases obtained in Namibia between 2011 and 2017, six (1.4%) were from bat-eared foxes. A report on rabies in South Africa between 1997 and 1998 showed that rabies in bat-eared foxes was 4.2% (n=45) and cape foxes was 0.4% (n=4) out of 1,071 animal rabies cases (Bishop et al., 2002). Furthermore, a retrospective study of rabies in wildlife species from 1998 to 2017 showed that 2.1% (n=12) out of the 560 confirmed rabies cases were from bat-eared foxes. This species also accounted for 1.9 (n=12) of 621 confirmed rabies-positive wildlife cases between 1998 and 2022 (Mogano et al., 2024). The bat-eared foxes are key maintenance hosts of rabies in southern Africa, sustaining transmission among wildlife, particularly in South Africa. In contrast, the Cape fox mainly acts as an incidental host with limited transmission potential. The epidemiological role of these species varies regionally and is influenced by ecological conditions and population density. Rabies in African wild dogsThe African wild dog is endangered as its population has declined drastically in the past few years. The population decline and local extinction have been linked to rabies outbreaks in different packs (Gascoyne et al., 1993; Hofmeyr et al., 2000; Davies-Mostert et al., 2016). The death of 20 in a park of 24 African wild dogs that were established through conservation efforts at the Madikwe game reserve was attributed to a rabies outbreak in the pack (Hofmeyr et al., 2000). In early 2000, a second outbreak of rabies occurred in a pack in the same Madikwe Game Reserve, which resulted in the death or disappearance of 10 out of 12 8-month-old pups (Hofmeyr et al., 2006). These outbreaks were believed to have originated from black-backed jackals, as the molecular characterization of the viruses showed that they were of the canid rabies biotype. Another rabies outbreak in Madikwe Game Reserve, South Africa, occurred in 2014–2015, where the last surviving pack of 6 and the last of a larger park of 18 were confirmed to be rabies positive. The antigenic typing of the viruses showed that they belonged to the canid rabies biotype, and phylogenetic analysis confirmed that they belonged to this biotype (Sabeta et al., 2018). In late 2014 and early 2015, a rabies outbreak in Limpopo-Lipadi Private Game and Wilderness Reserve in the Tuli region of south-eastern Botswana resulted in the death of 29 out of 35 African wild dogs (Canning et al., 2019). In conclusion, rabies outbreaks have caused severe population declines and local extinctions of African wild dogs in southern Africa. Multiple incidents in reserves such as Madikwe and Limpopo-Lipadi have wiped out entire packs, with molecular evidence linking the outbreaks to the canid rabies biotype transmitted from black-backed jackals. These events underscore the high susceptibility of the species to rabies and the urgent need for effective disease control and vaccination measures in conservation areas. Rabies in the AardwolfThe aardwolf (Proteles cristatus) is an insect-eating and burrow-dwelling carnivore that inhabits the semi-arid, open plains, savannas, and grasslands of Southern and Eastern Africa (Kruger National Park, 2024). This mammalian species has been known to maintain a rabies variant of the bat-eared fox origin in the Northern, Eastern, and Western Cape of South Africa, where both species co-habit. The aardwolf was initially thought to be an incidental host of the rabies virus; however, over time, a host switch from the bat-eared fox appears to have occurred. This resulted in the frequent transmission of the canid rabies variant from aardwolf to aardwolf and aardwolf to bat-eared fox, an observation supported by trend analyses of rabies cases (2011–2016) and genetic studies (Koeppel et al., 2022; Ngoepe et al., 2024). The exchange of rabies virus between the two host species is particularly marked during winter, when food supplies are scarce. The aardwolf, generally a nocturnal, switches to diurnal feeding on a different termite species, Hodotermes mossambicus, which is the primary food source of the bat-eared fox, resulting in fights and ultimately rabies transmission (Sillero-Zuberi, 2009). Furthermore, the use of the same size of dens by the two species leads to fighting, thereby creating an opportunity for exchange of RABV canid variant between the two species (Richardson, 1987; Anderson, 1994; Ngoepe et al., 2024). The aardwolf is now considered a maintenance host for canid rabies in South Africa because the number of positive cases in aardwolves between 2011 and 2016 surpassed that of the bat-eared fox (Koeppel et al., 2022; Ngoepe et al., 2024). In Zimbabwe, aardwolf rabies cases accounted for 0.5% (%) (n=7) of the 1,540 confirmed rabies cases in wild animals between 1992 and 2003 (Pfukenyi et al., 2009). In conclusion, the aardwolf has transitioned from being an incidental host to a maintenance host of the canid rabies variant in South Africa, likely originating from the bat-eared fox. Transmission between aardwolves and bat-eared foxes occurs mainly during winter due to food and den competition. Genetic and epidemiological evidence confirms sustained aardwolf-to-aardwolf transmission, making the species an emerging reservoir of rabies in southern Africa. Rabies in the Herpestidae and Viverridae generaIn the early 1940s in South Africa, rabies was confirmed in yellow mongoose (Cynictis penicillata), genet (G. genetta), meerkats (Suricata suricatta), and Cape gray mongoose (Herpestes pulverulentus), although there is evidence of rabies in genets as far back as 1916 (Cluver, 1927; Meredith, 1982). Mongooses and meerkats inhabit the grassveld region of the inland plateau, the Highveld of Middleburg, to the eastern Cape cost between Gqeberha and East London, up to the eastern Karoo scrub bushveld, while the genets prefer the bushveld zone of the northern Cape and southern Botswana, extending into Namibia (Meredith, 1982). Members of the Herpestidae family, particularly the yellow mongoose (C. penicillata) and slender mongoose (Galerella sanguinea) in southern Africa, maintain and transmit the mongoose rabies biotype (Kissi et al., 1995; Nel et al., 2005). However, not all Herpestidae species can maintain and transmit this variant. For instance, Selous’s mongoose (Paracynictis selousi) has been ruled out as a wildlife reservoir for mongoose rabies in Zambia (Röttcher and Sawchuk, 1978). Mongooses and other Herpestidae species accounted for 8.9% (n=17) of 190 confirmed rabies-positive cases in 2016 in Gauteng province, South Africa (Ngoepe et al., 2022). Approximately 43.2% (n=242) of the 560 infected mongoose cases in South Africa were recorded between 1998 and 2017 (Mogano et al., 2022). In a more recent study (1998–2022), mongooses accounted for 8.29% (n=259) rabies-positive cases out of 2,902 positive wildlife cases in South Africa (Mogano et al., 2024). An 18-year retrospective study (1989–2006) conducted in Botswana revealed almost equal prevalence of rabies in genets and mongooses [8.45% (n=29) and 7% (n=24)], respectively (Moagabo et al., 2009). Mongooses have also been implicated in wildlife rabies epizootiology in Malawi, although a low prevalence of 3.8% of the 26 rabies-positive cases was confirmed from mongooses between 2008 and 2021 (Kainga et al., 2023). Other studies on wildlife rabies in Zimbabwe have shown that the African civet (C. civetta), genets, and mongooses accounted for a very low prevalence of 4% (n=62) between 1992 and 2003 (Pfukenyi et al., 2009). African civets accounted for 3.2% of all rabies cases from 2010 to 2017 in South Africa, indicating that they are crucial to rabies epidemiology in Southern Africa (Horton et al., 2014; Sabeta et al., 2020). In conclusion, yellow and slender mongooses are the primary maintenance hosts of the mongoose rabies biotype in southern Africa, whereas other Herpestidae and Viverridae species (e.g., genets, civets, and meerkats) mostly serve as incidental hosts. Regional data indicate that mongooses significantly contribute to rabies cases in wildlife, highlighting the key role of this wildlife carnivore species in sustaining the rabies cycle in the region. Rabies in wild herbivoresAn unusual and persistent rabies cycle has been documented in the greater kudu antelope (Tragelaphus strepsiceros) in Namibia. Although the kudu is generally regarded as a dead-end host, evidence suggests that the rabies virus is maintained within the kudu population through intraspecific transmission, resulting in a localized and sustained infection cycle (Hübschle, 1988; Scott et al., 2012; Müller et al., 2022). Rabies was first observed in this species in 1975 near the capital of Windhoek, and the first epizootic disease began in Okahandja district in 1977. This was later confirmed during the same year as the outbreak in the Etosha National Park (Barnard and Hassel, 1981). Since then, rabies has increasingly spread among the kudu antelope population, and during the 2002–2003 period, an estimated 2,500 animals succumbed to the disease in this country (Müller et al., 2022). In Botswana, most rabies cases are in domestic ruminants and account for over 70% of all rabies cases in the desert country (Moagabo et al., 2009). This could have resulted from a consistent spillover of infection from black-backed jackals, domestic dogs, the yellow mongoose, and small-spotted genets (Swanepoel et al., 1993). The social behavior and feeding habits of kudu make the horizontal spread of RABV from kudu to kudu through a non-bite route potent and easy to transmit (Barnard and Hassel, 1981). Kudu are social leaf eaters that graze together on thorny acacia plants, leaving several inconspicuous lesions in their oral cavities, which are suitable routes for facilitating entry via infectious saliva left on the plant (Shaw, 1980; Hübschle, 1988). Mansfield et al. (2006)reported that jackals and kudus share the same rabies epidemiological cycle in Namibian wildlife. The genetic analysis (including a very limited panel of rabies viruses) from the Mansfield study showed that the canid rabies biotype, most probably the black-backed jackal or the domestic dog, was the source of the 2002 epizootic. The study further reported that jackal and kudu may form part of the same epidemiological cycle of rabies, which explains the close relationships between rabies virus strains that circulate within Namibia and those that circulate between Namibia and neighboring countries such as Botswana and South Africa (Mansfield et al., 2006). In a retrospective study conducted in Namibia between 2011 and 2017, the prevalence rate of rabies in kudu antelope was 77.3% out of 419 cases, indicating that rabies in Namibian wildlife is mainly in Kudu (Hikufe et al., 2019). However, this observation may reflect the researchers’ surveillance focus and may not accurately represent the broader epidemiological situation of rabies in Namibian wildlife. Other herbivore species, such as eland antelope (Taurotragus oryx), accounted for 8.4%, whereas waterbuck (Kobus ellipsiprymnus), blue wildebeest (Connochaetes taurinus), oryx antelope (Oryx gazelle), and Kirk’s dik dik (Madoqua kirkii) each accounted for less than 1% of cases in the same cohort (Hikufe et al., 2019). In Botswana, most rabies cases are in domestic ruminants and account for over 70% of all rabies cases in the desert country (Moagabo et al., 2009). However, in Zimbabwe and South Africa, rabies rates in wild ruminants were much lower than in Namibia. This could be a result of the population density of kudu in Namibia, the historical colonial heritage, and kudu farming, which is part of the economic utilization of natural species richness (Barnard and Hassel, 1981). In a cohort of 1,540 rabies-positive cases collected in Zimbabwe from 1992 to 2003, the total number of rabies-positive wild ruminants, including kudu antelope, eland antelope (T. oryx), and duiker (Sylicapra grimmia), was 0.4% (n=6). Some wild ruminants that were found to be positive in a rabies outbreak in Gauteng province, South Africa, in 2016 included kudu (T. strepsiceros), impala (Aepyceros melampus), and sable antelope (Hippotragus niger) (Ngoepe et al., 2022). Therefore, rabies infections in noncarnivorous species typically result in a rapid dead end for the virus. However, the ongoing epizootics observed in the kudu population in Namibia represent a unique exception. The remarkable scale of these outbreaks, combined with high population densities and specific social behaviors, such as mutual grooming and feeding on thorny vegetation that can cause oral lesions, facilitates a high level of horizontal, non-bite transmission within the kudu population. Collectively, these factors enable the sustained circulation of the rabies virus and the persistence of the epidemic over extended periods. Rabies in BatsThe role of bats (Order: Chiroptera) in the epidemiology of rabies in Southern Africa is not fully understood, as rabies surveillance in these species is generally limited, apart from the work done by a few independent researchers and bat interest groups. However, the involvement of bats in two human fatalities in South Africa shows the importance of chiroptera in the maintenance, transmission, and spillover of lyssaviruses to human and animal populations (Paweska et al., 2006). More importantly, some bat species identified as vectors of lyssaviruses co-roost and switch roost with other bat species, thereby posing the risk of infecting other bat species (AfricanBats NPC, 2024). Different Lyssavirus spp. have been detected in both insectivorous and frugivorous bats in South Africa and Zimbabwe. Lyssavirus duvenhage has been detected in insectivorous bats, the Miniopterus schreibersi, and the Egyptian slit-faced bat (Nycteris thebaica) in South Africa and Zimbabwe (Schneider et al., 1985; Moagabo et al., 2009; Coertse et al., 2020). Lagos bat lyssavirus has been confirmed in frugivorous bats (Epomophorus wahlbergi) in Pine town and Durban, South Africa (Markotter et al., 2006). In addition, two putative lyssaviruses, L. matlo and L. phala, have been identified in a Natal long-fingered bat (M. natalensis) and a Schlieffen’s bat (N. schlieffeni) (Viljoen et al., 2023). Rabies has also been confirmed in some unidentified bat species in South Africa (Ngoepe et al., 2022). Bats in southern Africa are important but understudied reservoirs of lyssaviruses, including rabies. Their roosting behaviors facilitate virus transmission among bat species, and multiple lyssaviruses have been detected in both insectivorous and frugivorous bats, highlighting their role in human and animal spillover. Incidental wildlife hostsRabies occurs in different incidental and dead-end hosts. These mammalian carnivores contribute to the overall number of confirmed cases of rabies in wildlife. The numbers of these hosts are rather low but are crucial to the dynamics of rabies transmission in the region. Rabies in the HyenaAlthough hyenas are wild canids, they appear to be incidental hosts of rabies rather than reservoir hosts, as their involvement in rabies outbreaks is very low compared with jackals and bat-eared foxes. Most importantly, hyenas have not been involved in any epidemics in the wild to date. In an 18-year (1989–2006) retrospective study in Botswana, hyenas were responsible for 2.91% (10) of 343 recorded rabies cases (Moagabo et al., 2009). In Zimbabwe, a retrospective study of rabies in wildlife (1992–2003), hyenas accounted for 0.4% (n=6) of the 1,540 positive cases. In Malawi, a study showed that the total number of rabies-positive wildlife cases between 2008 and 2021 was 26 (34.7%) out of 75 submitted cases, of which 9 (34.6%) were from hyenas (Kainga et al., 2023). In conclusion, hyenas are incidental hosts of rabies, minimally contributing to outbreaks and showing no evidence of sustaining epidemics. Rabies in the Honey BadgerAlthough honey badgers (Mellivora capensis) are believed to be incidental hosts for rabies, they are pivotal in rabies transmission. Of the 343 cases of wildlife rabies from 1989 to 2006 in Botswana, 7 (2.04%) were from the honey badger (Moagabo et al., 2009). The honey badger was responsible for approximately 1.8% (n=27) of the 1,540 confirmed rabies-positive cases in Zimbabwe between 1992 and 2003 (Pfukenyi et al., 2009). In July 2021, a rabid honey badger from the Kromdraai area, Gauteng Province, South Africa, bit a dog on a small farm and attacked three humans in the same vicinity. The honey badger was euthanized, and brain tissue was submitted to the Rabies Reference Laboratory for a rabies diagnosis, where it was subjected to phylogenetic analysis and subsequently shown to be a dog rabies variant (Mohale et al., 2023). In Malawi, honey badgers account for 3.8% of the 26 confirmed rabies-positive cases from 2008 to 2021 (Kainga et al., 2023). In conclusion, honey badgers are incidental rabies hosts but can transmit the virus to other animals and humans, contributing to a small yet notable proportion of wildlife rabies cases in southern Africa. Rabies in wild catsCases of rabies have been reported in wild felines, and they are considered either incidental or dead-end hosts. In an 18-year study conducted in Botswana, wild cats accounted for 5.3% (n=18) of rabies cases in a cohort of 343 rabies-positive wildlife cases (Moagabo et al., 2009). In Zimbabwe, wild felines accounted for slightly less prevalence of 1.6% (n=25) of rabies cases out of 1,540 confirmed positive cases in wildlife as follows: sarval (Felis serval; n=12); wildcat (Felis lybica; n=8); cheetah (Actinonyx jubatus; n=2); leopard (Panthera panlus; n=2); and lion (Panthera leo; n=1) (Pfukenyi et al., 2009). In South Africa, rabies has been confirmed in different wild feline species, including wild cats and lions (Mogano et al., 2022; Mogano et al., 2024). Wild felines are incidental or dead-end rabies hosts, contributing minimally to wildlife cases across southern Africa. Other incidental hostsA retrospective study conducted in Malawi from 2008 to 2021 showed that of the 26 positive cases recorded, only 1 (3.8%) was recovered from a monkey (Simiiformes spps), 3 from wild pigs (11.5%), and 2 (7.7%) from zebras (Equus quagga) (Kainga et al., 2023). Rabies has also been confirmed in elephants (loxodonta africana), monkeys, shrews (Sorex spps), and squirrels (Sciurus spps) in Zimbabwe (Pfukenyi et al., 2009) and in squirrels and wild pigs in Botswana (Moagabo et al., 2009). In South Africa, rabies has been confirmed in zebras, baboons (Papio saps), rabbits (Oryctolagus cuniculus), warthogs (Phacochoerus africanus), and rats (Rattus rattus) (Ngoepe et al., 2022; Mogano et al., 2024). The low numbers observed in this study could be attributed to the limited contacts between the canine rabies reservoirs and the incidental hosts due to their different habitats and feeding habits. Rabies seasonality in wildlife in Southern AfricaRabies cases in domestic dogs and wildlife species were reported throughout the year, with the majority occurring in the mid-dry season, most frequently in the northern and eastern parts of Limpopo and Mpumalanga provinces. This seasonality of rabies coincides with the scarcity period of resources, promoting animals to converge where resources are available. This creates an opportunity for the interaction of several animal hosts, thereby facilitating the spread of rabies (Okell et al., 2013; Lachica et al., 2020). The geographical areas associated with the occurrence of rabies have evolved over the years, with thicket bush and grasslands between 1998 and 2002 and cultivated commercial crops and water bodies between 2008 and 2012; currently, rabies cases are most commonly found in plantations and woodlands (Mogano et al., 2024). Way forwardThe data and information presented here are not uniform across the southern African countries and probably reflect the research efforts toward rabies epidemiology in the respective countries. Improved rabies surveillance and decentralized rabies confirmatory diagnostic centers situated in strategic places are probably the first step toward achieving control of sylvatic rabies in southern Africa (Banyard et al., 2013). A study conducted by Rossouw et al. (2021)on the serum samples of 168 carnivores [African wild dog (Lycaon pictus), spotted hyena (Crocuta crocuta), lion (P. leo), leopard (Panthera pardus), and banded mongoose (Mungos mungo)] in Kruger Park, South Africa, showed that only 4.8% had low levels (0.00 IU/ml–0.22 IU/ml) of neutralizing rabies antibodies. This shows a population at risk of contracting rabies if an outbreak occurs, and this is a highly likely scenario. Continuous integrated surveillance and control measures should be put in place for effective sylvatic rabies elimination in southern Africa and the continent to a larger extent. Information and education campaigns should be carried out in human populations living close to farms, and the wild (human/wildlife interfaces), and professional training should be conducted periodically in monitoring, diagnosis, and evaluation of rabies in wildlife (Holanda Duarte et al., 2021). The control and elimination of wildlife-mediated rabies through oral rabies vaccination campaigns in wild carnivores have been successful in Europe and America (Müller and Freuling, 2018; Fehlner-Gardiner and C, 2018). Several trials on oral rabies vaccination of jackals and wild dogs have been undertaken by different and independent research groups in southern Africa and have shown great promise. It will be more effective and sustainable if the Southern African governments and the African Union (AU) financially support a consistent strategic mass oral vaccination of wildlife throughout the continent. For instance, the SAD (Berne) vaccine was shown to be efficacious in both black-backed jackals and side-striped jackals for a period of 12 months post-oral vaccination (Bingham et al., 1995) and subsequently survived challenges with a field strain of rabies virus (Bingham et al., 1995). However, the pathogenic effect of the SAD (Berne) vaccine on chacma baboons and rodents renders it unsuitable for use in southern African wildlife (Bingham et al., 1992). The SAD Avirulent Gif-2 vaccine showed great promise in field trials in Ethiopian wolves and demonstrated adequate immunity in Ethiopian wolves for up to 12 months post-vaccination (Sillero-Zubiri et al., 2016). Raboral V-RG® vaccine was used in captive and free-range jackal species and elicited humoral antibodies for up to 48 weeks after oral vaccination (Koeppel et al., 2020; Koeppel et al., 2022). Koeppel et al. (2020) reported that black-backed jackals preferred chicken head bait type, particularly at dusk during summer, to minimize its uptake by non-target species (Koeppel et al., 2020). Moving forward, a standardized vaccine and bait system, supported by robust scientific data, should be adopted for use in wildlife rabies control programs across southern Africa. The Southern African Development Community (SADC), functioning as a Regional Economic Community, collaborates with the AU and international partners such as the World Health Organization, the World Organization for Animal Health, and the Food and Agriculture Organization to implement coordinated rabies control strategies within its member states. These efforts aim to strengthen the collaboration between veterinary and public health authorities under the One Health framework, enhance political commitment, and promote the elimination of dog-mediated rabies in the SADC region (World Organization for Animal Health, 2018). The African Union Pan-African Veterinary Vaccine Center (AU-PANVAC) plays a critical role in ensuring the quality control of rabies vaccines produced or imported into Africa (World Organization for Animal Health, 2025). To achieve sustainable control and eventual elimination of wildlife-associated rabies in southern Africa, concerted regional and national efforts are essential. Governments within the region, through the SADC and the AU-PANVAC, should continue to provide financial and logistical support to ensure the sustainable production of high-quality, efficacious vaccines, a consistent supply of oral baits, and efficient vaccine delivery systems. Equally important is dog owners’ education, particularly those living in sympatric zones, on the importance of regular dog vaccination and responsible dog ownership. With these measures effectively implemented and maintained, wildlife-associated rabies control and eventual elimination in southern Africa is an attainable goal. ConclusionIn southern Africa, rabies persists through a complex cycle maintained by both domestic and wild carnivores. Although wild ruminants, such as kudus, are not typical reservoir hosts, their demonstrated ability to transmit the virus among themselves represents a unique and significant epidemiological feature. Strengthening active surveillance and diagnostic capacity is essential, as improved monitoring may reveal additional reservoir species, particularly in regions with limited lyssavirus surveillance. Sylvatic reservoirs, especially black-backed jackals, pose a persistent risk for the reintroduction of rabies into naïve domestic dog populations, potentially undermining global eradication efforts if current control strategies remain unchanged. The domestic dog remains the critical bridge between wildlife and human rabies transmission; thus, the ongoing viral exchange between black-backed jackals and domestic dogs constitutes a serious public and veterinary health threat. Overall, jackals, aardwolves, foxes, mongooses, and bats serve as the principal sylvatic reservoirs of rabies in southern Africa, while species such as honey badgers, hyenas, wild dogs, and kudu antelopes act primarily as incidental hosts. A sustained, multisectoral One Health approach that integrates wildlife management, domestic dog vaccination, and robust surveillance is imperative to break this transmission cycle and move closer to rabies elimination. LimitationsThere is a paucity of information on rabies in some southern African countries. For instance, in Lesotho, rabies surveillance is constrained by inadequate sample submissions for routine surveillance at the only laboratory in the country capable of diagnosing rabies, the Central Veterinary Laboratory in Maseru. Although rabies was first confirmed in Angola in 1928, little or no information is available about the current epidemiology of the disease in the country, which could be a sequel to the prolonged civil war (1975–2002) (Nel and Rupprecht, 2007). Other limitations encountered in accessing relevant literature for this study included resource constraints, such as paywalled journals and restricted database access, as well as the limited availability of digitized or indexed studies. In addition, gray literature and older publications were often difficult to locate, further constraining the comprehensiveness and completeness of the literature search. The paucity of data from some countries within the region may have implications for accurately characterizing the epidemiology of rabies in southern Africa. Consequently, the conclusions drawn from this study may not fully capture the true dynamics and distribution of the disease across the region. AcknowledgmentEze U.U is currently a research fellow in the Department of Veterinary Tropical Diseases of the University of Pretoria (Faculty of Veterinary Science, South Africa). Conflict of interestThe authors declare no conflict of interest. FundingThis work was supported by the Africa Research Excellence Fund (AREF) through the 2023 Research Development Fellowship Programme, which provides funding for early-career African researchers to undertake research training and capacity-building placements (AREF-312-EZE-F-C0953). Authors’ contributionConceptualization: Sabeta CT: Writing—original draft: Eze U.U; Writing—review and editing: Eze U.U and Sabeta CT. Data availabilityAll data supporting this study’s findings are available within the manuscript. All data were provided in the manuscript. ReferencesAfricanBats NPC. 2019. African Chiroptera report 2019. Pretoria, South Africa: AfricanBats NPC. Available via https://africanbats. org/publication/african-chiroptera-report-2019/ Anderson, M.D. 1994. The influence of seasonality and quality of diet on the metabolism of the aardwolf, Proteles cristatus (Sparrman, 1783). Unpublished master’s thesis, University of Pretoria, Pretoria, South Africa. Banyard, A., Evans, J., Luo, T. and Fooks, A. 2014. Lyssaviruses and bats: emergence and zoonotic threat. Viruses 6, 2974–2990; doi:10.3390/v6082974 Banyard, A.C., Horton, D.L., Freuling, C., Müller, T. and Fooks, A.R. 2013. Control and prevention of canine rabies: the need for building laboratory-based surveillance capacity. Antiviral Res. 98(3), 357–364; doi:10.1016/j.antiviral.2013.04.004 Barnard, B.J. 1979. The role played by wildlife in the epizootiology of rabies in South Africa and South West Africa. Onderstepoort J. Vet. Res. 46, 155–163. Barnard, B.J. and Hassel, R.H. 1981. Rabies in kudus (Tragelaphus strepsiceros) in South West Africa/Namibia. J. South Afr. Vet. Assoc. 52, 309–314. Bingham, J. and Foggin, C.M. 1993. Jackal rabies in Zimbabwe. Onderstepoort. J. Vet. Res. 60, 365–366. Bingham, J., Foggin, C.M., Gerber, H., Hill, F.W., Kappeler, A., King, A.A., Perry, B.D. and Wandeler, A.I. 1992. Pathogenicity of SAD rabies vaccine given orally in chacma baboons (Papio ursinus). Vet. Record 131(3), 55–56; doi: 10.1136/vr.131.3.55 Bingham, J., Foggin, C.M., Wandeler, A.I. and Hill, F.W.G. 1999. The epidemiology of rabies in Zimbabwe. II. Rabies in jackals (Canis adustus and Canis mesomelas). Onderstepoort. J. Vet. Res. 66, 11–23. Bingham, J., Kappeler, A., Hill, F.W.G., King, A.A., Perry, B.D. and Foggin, C.M. 1995. Efficacy of SAD (Berne) rabies vaccine given by the oral route in two species of jackal (Canis mesomelas and Canis adustus). J. Wildlife. Dis. 31(3), 416–419; doi:10.7589/0090-3558-31.3.416 Bishop, G.C., Durrheim, D.N., Kloeck, P.E., Godlonton, J.D., Bingham, J. and Speare, R., 2002. Rabies guide for the medical, veterinary and allied professions. Rabies Advisory Group, South African Department of Agriculture and Health, Pretoria, South Africa. pp: 2–16. Boulger, L.R. and Porterfield, J.S. 1958. Isolation of a virus from Nigerian fruit bats. Trans. Roy. Soc. Trop. Med. Hygiene 52, 421–424. Bourhy, H., Reynes, J.M., Dunham, E.J., Dacheux, L., Larrous, F., Huong, V.T.Q., Xu, G., Yan, J., Miranda, M.E.G. and Holmes, E.C. 2008. The origin and phylogeography of dog rabies virus. J. Gen. Virol. 89(11), 2673–2681; doi:10.1099/vir.0.2008/003913-0 Brückner, G.K., Hurter, L.R. and Boshoff, J.N. 1978. Field observations on the occurrence of rabies in cattle in the magisterial districts of Soutpansberg and Messina. J. South. Afr. Vet. Assoc. 49(1), 33–36. Canning, G., Camphor, H. and Schroder, B. 2019. Rabies outbreak in African wild dogs (Lycaon pictus) in the Tuli region, Botswana: interventions and management mitigation recommendations. J. For Nat. Conserv. 48, 71–76; doi:10.1016/j.jnc.2019.02.001 Centers for Disease Control and Prevention. 2023. Rabies. In: CDC Yellow Book 2024: travel-associated infections and diseases. Eds., Wallace, R., Petersen, B. and Shlim, D. Available via https://wwwnc.cdc.gov/travel/yellowbook/2024/infections-diseases/rabies Chimera, B.A.R. and Chikungwa, P.B. 2001. Rabies in Malawi: country report. Lilongwe, Malawi: Sixth SEARG Meeting. Cluver. 1927. Rabies in South Africa. J. Med. Assoc. South. Afr. 1, 247–253. Coertse, J., Grobler, C.S., Sabeta, C.T., Seamark, E.C.J., Kearney, T., Paweska, J.T. and Markotter, W. 2020. Lyssaviruses in insectivorous bats, South Africa, 2003–2018. Emerg. Infect. Dis. 26(12), 3056–3060; doi:10.3201/eid2612.203592 Cowling, R.M., Richardson, D.M. and Pierce, S.M. 1997. Vegetation of Southern Africa. Cambridge University Press, Edinburgh, Scotland: p: 278; doi: 10.1017/CBO9780511482876 Cumming, D.H.M. 1982. A case history of the spread of rabies in an African country. South Afr. J. Sci. 78, 443–447. Davies-Mostert, H.T., Page-Nicholson, S., Marneweck, D.G., Marnewick, K., Cilliers, D., Whittington-Jones, B., Killian, H., Mills, M.G.L., Parker, D., Power, J., Rehse, T. and Child, M.F. 2016. A conservation assessment of Lycaon pictus. In The red list of mammals of South Africa, Swaziland and Lesotho. Eds., Child, M.F., Roxburgh, L., Do Linh San, E., Raimondo, D. and Davies-Mostert, H.T. South African National Biodiversity Institute and Endangered Wildlife Trust. South Africa. Everard, C.O. and Everard, J.D. 1988. Mongoose rabies. Rev. Infect. Dis. 10(Suppl 4), S610–S614; doi: 10.1093/clinids/10.supplement_4.s610 Everard, C.O.R. and Everard, J.D. 1992. Mongoose rabies in the Caribbean. Ann. New York Acad. Sci. 653, 356–366; doi:10.1111/j.1749-6632.1992.tb19662.x Fehlner-Gardiner, C. 2018. Rabies control in North America: past, present and future. Revue. Scientifique. Et. Tech. 37(2), 421–437; doi:10.20506/rst.37.2.2812 Foggin, C.M. 1988. Rabies and rabies-related viruses in Zimbabwe: historical, virological and ecological aspects. Doctoral dissertation, University of Zimbabwe, Harare, Zimbabwe. Gascoyne, S.C., King, A.A., Laurenson, M.K., Borner, M., Schildger, B. and Barrat, J. 1993. Aspects of rabies infection and control in the conservation of the African wild dog (Lycaon pictus) in the Serengeti region, Tanzania. Onderstepoort J. Vet. Res. 60(4), 415–420. Hayman, D.T.S., Fooks, A.R., Marston, D.A. and Garcia-R, J.C. 2016. The global phylogeography of lyssaviruses: challenging the ‘Out of Africa’ hypothesis. PLos Neglected. Trop. Dis. 10(12), 5266; doi: 10.1371/journal.pntd.0005266 Hikufe, E.H., Freuling, C.M., Athingo, R., Shilongo, A., Ndevaetela, E.E., Helao, M., Shiindi, M., Hassel, R., Bishi, A., Khaiseb, S., Kabajani, J., Van Der Westhuizen, J., Torres, G., Britton, A., Letshwenyo, M., Schwabenbauer, K., Mettenleiter, T.C., Denzin, N., Amler, S., Conraths, F.J., Müller, T. and Maseke, A. 2019. Ecology and epidemiology of rabies in humans, domestic animals and wildlife in Namibia, 2011–2017. PLos Neglected. Trop. Dis. 13(4), e0007355; doi:10.1371/journal.pntd.0007355 Hofmeyr, M., Bingham, J., Lane, E.P., Ide, A. and Nel, L. 2000. Rabies in African wild dogs (Lycaon pictus) in the Madikwe Game Reserve, South Africa. Vet. Rec. 146(2), 50–52. Hofmeyr, M., Hofmeyr, D., Nel, L. and Bingham, J. 2006. A second outbreak of rabies in African wild dogs (Lycaon pictus) in Madikwe Game Reserve, South Africa, demonstrating the efficacy of vaccination against natural rabies challenge. Anim. Conservation. 7(2), 193–198; doi:10.1017/S1367943004229 Holanda Duarte, N.F., Alencar, C.H., Pires Neto, R.D.J., Moreno, J.D.O., Araújo Melo, I.M.L., Duarte, B.H. and Heukelbach, J. 2021. Integration of human rabies surveillance and preventive measures in the State of Ceará, Northeast Brazil. One. Health. Implement. Res. 1(1), 17–30; doi:10.20517/ohir.2021.02 Horton, D.L., Banyard, A.C., Marston, D.A., Wise, E., Selden, D., Nunez, A., Hicks, D., Lembo, T., Cleaveland, S., Peel, A.J., Kuzmin, I.V., Rupprecht, C.E. and Fooks, A.R. 2014. Antigenic and genetic characterization of a divergent African virus, Ikoma lyssavirus. J. Gen. Virol. 95(5), 1025–1032; doi:10.1099/vir.0.061952-0 Hübschle, O.J. 1988. Rabies in the kudu antelope (Tragelaphus strepsiceros). Rev. Infect. Dis. 10(Suppl. 4), S629–S633; doi: 10.1093/clinids/10.supplement_4.s629 Hussein, N.A., Sharma, R.N., Ando, R. and Chizyuka, H.G.B. 1984. Further review of the epidemiology of rabies in Zambia (1975–1982). Revue. Scientifique. Et. Tech. (OIE). 3(1), 125–135; doi:10.20506/rst.3.1.149 International Committee on Taxonomy of Viruses. 2024. Virus taxonomy: classification and nomenclature of viruses. Online report of the International Committee on Taxonomy of Viruses (2024 release). Available via https://ictv.global/report Kainga, H., Chatanga, E., Phonera, M.C., Kothowa, J.P., Dzimbiri, P., Kamwendo, G., Mulavu, M., Khumalo, C.S., Changula, K., Chambaro, H., Harima, H., Kajihara, M., Mkandawire, K., Chikungwa, P., Chulu, J., Njunga, G., Chitanga, S., Mubemba, B., Sasaki, M., Orba, Y., Qiu, Y., Yamagishi, J., Simulundu, E., Takada, A., Namangala, B., Sawa, H. and Muleya, W. 2023. Current status and molecular epidemiology of rabies virus from different hosts and regions in Malawi. Arch. Virol. 168(2), 61; doi:10.1007/s00705-022-05635-z King, A.A., Fooks, A.R., Aubert, M. and Wandeler, A.I. 2004. Historical perspective of rabies in Europe and the Mediterranean Basin. Paris, France: OIE. Kissi, B., Tordo, N. and Bourhy, H. 1995. Genetic polymorphism in the rabies virus nucleoprotein gene. Virology 209(2), 526–537; doi:10.1006/viro.1995.1285 Koeppel, K., Kuhn, B. and Thompson, P. 2020. Oral bait preferences for rabies vaccination in free-ranging black-backed jackal (Canis mesomelas) and non-target species in a multi-site field study in a peri-urban protected area in South Africa. Prev. Vet. Med. 175, 104867; doi: 10.1016/j.prevetmed.2019.104867 Koeppel, K.N., Geertsma, P., Kuhn, B.F., Van Schalkwyk, O.L. and Thompson, P.N. 2022. Antibody response to Raboral VR-G® oral rabies vaccine in captive and free-ranging black-backed jackals (Canis mesomelas). Onderstepoort J. Vet. Res. 89(1), e1–e9; doi:10.4102/ojvr.v89i1.1975 Kruger National Park. 2024. Aardwolf occurs throughout eastern and southern Africa. South Africa: Siyabona Africa (Pty) Ltd. Available via https://www.krugerpark.co.za/africa_aardwolf.html#:~:text=Aardwolf%20occurs%20throughout%20eastern%20and,in%20burrows%20in%20the%20ground Kumar, A., Bhatt, S., Kumar, A. and Rana, T. 2023. Canine rabies: an epidemiological significance, pathogenesis, diagnosis, prevention, and public health issues. Comparative Immunol. Microbiol. Infect. Dis. 97, 101992; doi: 10.1016/j.cimid.2023.101992 Kuzmin, I.V., Bozick, B., Guagliardo, S.A., Kunkel, R., Shak, J.R., Tong, S. and Rupprecht, C.E. 2011. Bats, emerging infectious diseases, and the rabies paradigm revisited. Emerg. Health Threats J. 4, 7159; doi:10.3402/ehtj.v4i0.7159 Kuzmin, I.V., Hughes, G.J., Botvinkin, A.D., Orciari, L.A. and Rupprecht, C.E. 2005. Phylogenetic relationships of Irkut and West Caucasian bat viruses within the Lyssavirus genus and suggested quantitative criteria based on the N gene sequence for Lyssavirus genotype definition. Virus. Res. 111(1), 28–43. Kuzmin, I.V., Mayer, A.E., Niezgoda, M., Markotter, W., Agwanda, B., Breiman, R.F. and Rupprecht, C.E. 2010. Shimoni bat virus, a new representative of the Lyssavirus genus. Virus. Res. 149(2), 197–210. Lachica, Z.P.T., Peralta, J.M., Diamante, E.O., Murao, L.A.E., Mata, M.A.E. and Alviola Iv, P.A. 2020. A cointegration analysis of rabies cases and weather components in Davao City, Philippines from 2006 to 2017. PLos One 15(8), e0236278; doi:10.1371/journal.pone.0236278 Mansfield, K., Mcelhinney, L., Hübschle, O., Mettler, F., Sabeta, C., Nel, L.H. and Fooks, A.R. 2006. A molecular epidemiological study of rabies epizootics in kudu (Tragelaphus strepsiceros) in Namibia. BMC. Vet. Res. 2(2), 2; doi:10.1186/1746-6148-2-2 Markotter, W., Randles, J., Rupprecht, C.E., Sabeta, C.T., Taylor, P.J., Wandeler, A.I. and Nel, L.H. 2006. Lagos bat virus, South Africa. Emerg. Infect. Dis. 12(3), 504–506; doi:10.3201/eid1203.051306 Marks, S.E. 2024. Southern Africa. Chicago, IL: Encyclopedia Britannica. Available via https://www.britannica.com/place/Southern-Africa (Accessed April 27, 2024). Marston, D.A., Ellis, R.J., Horton, D.L., Kuzmin, I.V., Wise, E.L., Mcelhinney, L.M., Banyard, A.C., Ngeleja, C., Keyyu, J., Cleaveland, S., Lembo, T., Rupprecht, C.E. and Fooks, A.R. 2012. Complete genome sequence of Ikoma lyssavirus. J. Virol. 86(18), 10242–10243. Meredith, C.D. 1982. Wildlife rabies: past and future in South Africa. South Afr. J. Sci. 78, 411–415. Meredith, C.D., Rossouw, A.P. and Van Praag Koch, H. 1971. An unusual case of human rabies thought to be of chiropteran origin. South Afr. Med. J. 45, 767–769. Moagabo, K.T., Monyame, K.B., Baipoledi, E.K., Letshwenyo, M., Mapitse, N. and Hyera, J.M.K. 2009. A retrospective longitudinal study of animal and human rabies in Botswana, 1989–2006. Onderstepoort. J. Vet. Res. 76(4), 399–407; doi:10.4102/ojvr.v76i4.24 Mogano, K., Sabeta, C.T., Suzuki, T., Makita, K. and Chirima, G.J. 2024. Patterns of animal rabies prevalence in northern South Africa between 1998 and 2022. Trop. Med. Infect. Dis. 9(1), 27; doi:10.3390/tropicalmed9010027 Mogano, K., Suzuki, T., Mohale, D., Phahladira, B., Ngoepe, E., Kamata, Y., Chirima, G., Sabeta, C. and Makita, K. 2022. Spatio-temporal epidemiology of animal and human rabies in northern South Africa between 1998 and 2017. PLos Neglected. Trop. Dis. 16(7), 10464; doi:10.1371/journal.pntd.0010464 Mohale, D.K., Ngoepe, E., Mparamoto, M., Blumberg, L. and Sabeta, C.T. 2023. A case report on a human bite contact with a rabid honey badger (Mellivora capensis) (Kromdraai Area, Cradle of Humankind, South Africa). Trop. Med. Infect. Dis. 8(4), 186; doi:10.3390/tropicalmed8040186 Muleya, W., Chambaro, H.M., Sasaki, M., Gwenhure, L.F., Mwenechanya, R., Kajihara, M., Saasa, N., Mupila, Z., Mori-Kajihara, A., Qiu, Y., Kangwa, E., Mweene, A., Namangala, B., Takada, A. and Sawa, H. 2019. Genetic diversity of rabies virus in different host species and geographic regions of Zambia and Zimbabwe. Virus. Genes. 55, 713–719; doi:10.1007/s11262-019-01682-y Müller, F.T. and Freuling, C.M. 2018. Rabies control in Europe: an overview of past, current and future strategies. Revue. Scientifique. Et. Tech. 37(2), 409–419; doi:10.20506/rst.37.2.2811 Müller, T., Hassel, R., Jago, M., Khaiseb, S., Van Der Westhuizen, J., Vos, A., Calvelage, S., Fischer, S., Marston, D.A., Fooks, A.R., Höper, D. and Freuling, C.M. 2022. Rabies in kudu: revisited. Adv. Virus Res. 112, 115–173; doi:10.1016/bs.aivir.2022.04.001 Munang’Andu, H.M., Mweene, A.S., Siamudaala, V., Muma, J.B. and Matandiko, W. 2011. Rabies status in Zambia for the period 1985–2004. Zoonoses. Public. Health. 58(1), 21–27; doi:10.1111/j.1863-2378.2010.01368.x Nel, L.H. and Rupprecht, C.E. 2007. Emergence of lyssaviruses in the Old World: the case of Africa. Curr. Topics Microbiol. Immunol. 315, 161–193; doi:10.1007/978-3-540-70962-6_8 Nel, L.H., Sabeta, C.T., Teichman, B.V., Jaftha, J.B., Rupprecht, C.E. and Bingham, J. 2005. Mongoose rabies in southern Africa: a re-evaluation based on molecular epidemiology. Virus. Res. 109(2), 165–173; doi:10.1016/j.virusres.2004.12.003 Ngoepe, C., Shumba, W. and Sabeta, C. 2024. Evidence for a host switching in the maintenance of canid rabies variant in two wild carnivore species in the Northern Cape Province, South Africa. J. South Afr. Vet. Assoc. 95, 1; doi:10.36303/JSAVA.527 Ngoepe, C.E., Sabeta, C. and Nel, L. 2009. The spread of canine rabies into Free State province of South Africa: a molecular epidemiological characterization. Virus. Res. 142(1–2), 175–180; doi:10.1016/j.virusres.2009.02.012 Ngoepe, E., Chirima, J.G., Mohale, D., Mogano, K., Suzuki, T., Makita, K. and Sabeta, C.T. 2022. Rabies outbreak in black-backed jackals (Canis mesomelas), South Africa, 2016. Epidemiol. Infect. 150, e137; doi: 10.1017/S0950268821002685 Okell, C.N., Pinchbeck, G.P., Stringer, A.P., Tefera, G. and Christley, R.M. 2013. A community-based participatory study investigating the epidemiology and effects of rabies to livestock owners in rural Ethiopia. Prev. Vet. Med. 108(1), 1–9; doi:10.1016/j.prevetmed.2012.07.003 Paweska, J.T., Blumberg, L.H., Liebenberg, C., Hewlett, R.H., Grobbelaar, A.A., Leman, P.A., Croft, J.E., Nel, L.H., Nutt, L. and Swanepoel, R. 2006. Fatal human infection with rabies-related Duvenhage virus, South Africa. Emerg. Infect. Dis. 12(12), 1965–1967; doi: 10.3201/eid1212.060764 Pfukenyi, D.M., Pawandiwa, D., Makaya, P.V. and Ushewokunze-Obatolu, U. 2009. A retrospective study of wildlife rabies in Zimbabwe, between 1992 and 2003. Trop. Anim. Health. Prod. 41(4), 565–572; doi:10.1007/s11250-008-9224-4 Rhodes, C.J., Atkinson, R.P.D., Anderson, R.M. and Macdonald, D.W. 1998. Rabies in Zimbabwe: reservoir dogs and the implications for disease control. Phil. Trans. Roy. Soc. London. Ser. B. Biol. Sci. 353, 999–1010; doi:10.1098/rstb.1998.0263 Richardson, P.R.K. 1987. Food consumption and seasonal variation in the diet of the aardwolf Proteles cristatus in southern Africa. Zeitschrift Für Säugetierkunde 52(5), 307–325. Rossouw, L., Boshoff, C., Sabeta, C. and Kotzé, J. 2021. A preliminary investigation of exposure to rabies virus in selected wildlife in the Kruger National Park, South Africa. Koedoe 63(1), 1651; doi:10.4102/koedoe.v63i1.1651 Röttcher, D. and Sawchuk, A.M. 1978. Wildlife rabies in Zambia. J. Wildlife Dis. 14(4), 513–517; doi:10.7589/0090-3558-14.4.513 Rupprecht, C.E., Turmelle, A. and Kuzmin, I.V. 2011. A perspective on lyssavirus emergence and perpetuation. Curr. Opinion Virol. 1, 662–670; doi:10.1016/j.coviro.2011.10.014 Sabeta, C.T., Bingham, J. and Nel, L.H. 2003. Molecular epidemiology of canid rabies in Zimbabwe and South Africa. Virus Res. 91(2), 203–211; doi:10.1016/S0168-1702(02)00272-1 Sabeta, C.T., Janse Van Rensburg, D., Phahladira, B., Mohale, D., Harrison-White, R.F., Esterhuyzen, C. and Williams, J.H. 2018. Rabies of canid biotype in wild dog (Lycaon pictus) and spotted hyaena (Crocuta crocuta) in Madikwe Game Reserve, South Africa in 2014–2015: diagnosis, possible origins and implications for control. J. South. Afr. Vet. Assoc. 89, e1–e13; doi:10.4102/jsava.v89i0.1517 Sabeta, C.T., Mansfield, K.L., Mcelhinney, L.M., Fooks, A.R. and Nel, L.H. 2007. Molecular epidemiology of rabies in bat-eared foxes (Otocyon megalotis) in South Africa. Virus. Res. 129, 1–10. Sabeta, C.T., Marston, D.A., Mcelhinney, L.M., Horton, D.L., Phahladira, B.M.N. and Fooks, A.R. 2020. Rabies in the African civet: an incidental host for lyssaviruses?. Viruses 12(4), 368; doi:10.3390/v12040368 Schneider, L.G., Barnard, B.J.H. and Schneider, H.P. 1985. Application of monoclonal antibodies for epidemiological investigations and oral vaccination studies: i. African viruses. In: Rabies in the tropics. Eds., Kuwert, E., Merieux, C., Koprowski, H. and Bagel, K. Springer Berlin Heidelberg, Germany: Springer-Verlag, pp: 47–59. Scott, T., Hasse, R. and Nel, L. 2012. Rabies in kudu (Tragelaphus strepsiceros). Berliner. Und. Münchener. Tierärztliche. Wochenschrift. 125(5–6), 236–241. Shaw, J.J.H. 1980. Hondsdolheid by koedoes. J. South Afr. Vet. Assoc. 51, 209. Shope, R.E., Murphy, F.A., Harrison, A.K., Causey, O.R., Kemp, G.E., Simpson, D.I.H. and Moore, D.L. 1970. Two African viruses serologically and morphologically related to rabies virus. J. Virol. 6, 690–692. Sillero-Zuberi, C. 2009. Bat-eared fox (Otocyon megalotis). In: Handbook of the mammals of the world: volume 1. Carnivores. Eds., Wilson, D.E. and Mittermeier, R.A. Barcelona, Spain: Lynx Edicions, vol. 1, pp: 435–6. Sillero-Zubiri, C., Marino, J., Gordon, C.H., Bedin, E., Hussein, A., Regassa, F., Banyard, A. and Fooks, A.R. 2016. Feasibility and efficacy of oral rabies vaccine SAG2 in endangered Ethiopian wolves. Vaccine 34(40), 4792–4798; doi:10.1016/j.vaccine.2016.08.021 Snyman, P.S. 1940. The study and control of the vectors of rabies in South Africa. Onderstepoort. J. Vet. Sci. Anim. Ind. 15, 9–140. Swanepoel, R. 2004. Rabies, 2nd ed. In Infectious diseases of livestock: with special reference to southern Africa. Eds. Coetzer, J.A.W. and Tustin, R.C. Cape Town, South Africa: Oxford University Press, pp: 1123–1182. Swanepoel, R., Barnard, B.J., Meredith, C.D., Bishop, G.C., Brückner, G.K., Foggin, C.M. and Hübschle, O.J. 1993. Rabies in southern Africa. Onderstepoort. J. Vet. Res. 60(4), 325–346. Tran, H., Mcconville, M. and Loukopoulos, P. 2020. Metabolomics in the study of spontaneous animal diseases. J. Vet. Diagnostic. Invest. 32(5), 635–647; doi:10.1177/1040638720948505 Van Zyl, N., Markotter, W. and Nel, L.H. 2010. Evolutionary history of African mongoose rabies. Virus Res. 150(1–2), 93–102; doi:10.1016/j.virusres.2010.02.018 Viljoen, N., Weyer, J., Coertse, J. and Markotter, W. 2023. Evaluation of taxonomic characteristics of Matlo and Phala bat rabies-related lyssaviruses identified in South Africa. Viruses 15, 2047; doi:10.3390/v15102047 Von Teichman, B.F., Thomson, G.R., Meredith, C.D. and Nel, L.H. 1995. Molecular epidemiology of rabies virus in South Africa: evidence for two distinct virus groups. J. Gen. Virol. 76(1), 73–82; doi:10.1099/0022-1317-76-1-73 World Health Organization. 1999. Proceedings of the Southern and Eastern African Rabies Group/World Health Organization meeting. Editions Fondation Marcel Mérieux. World Health Organization. 2013. Rabies country profile: Zambia. Available via https://www.who.int/rabies/epidemiology/Rabies_CP_Zambia_09_2013.pdf World Organization for Anima Health. 2025. 26th Conference of the Regional Commission for Africa- Final Report. Addis Ababa, Ethiopia, 4 to 7 February 2025. Available via https://rr-africa.woah.org/app/uploads/2025/04/EN_26-Conference-regional_Africa.pdf . World Organization for Animal Health. 2018. SADC region reaffirms its commitment to eliminate dog-mediated rabies by 2030. Available via https://rr-africa.woah.org/en/news/sadc-region-reaffirms-its-commitment-to-eliminate-dog-mediated-rabies-by-2030/ (Accessed 29 October 2025) Zulu, G.C., Sabeta, C.T. and Nel, L.H. 2009. Molecular epidemiology of rabies: focus on domestic dogs (Canis familiaris) and black-backed jackals (Canis mesomelas) from northern South Africa. Virus. Res. 140(1–2), 71–78; doi:10.1016/j.virusres.2008.11.004 | ||
| How to Cite this Article |
| Pubmed Style Eze UU, Sabeta CT. A review of rabies in Southern African wildlife: Reservoir hosts, transmission dynamics, and control implications. Open Vet. J.. 2026; 16(1): 56-70. doi:10.5455/OVJ.2026.v16.i1.5 Web Style Eze UU, Sabeta CT. A review of rabies in Southern African wildlife: Reservoir hosts, transmission dynamics, and control implications. https://www.openveterinaryjournal.com/?mno=280154 [Access: January 31, 2026]. doi:10.5455/OVJ.2026.v16.i1.5 AMA (American Medical Association) Style Eze UU, Sabeta CT. A review of rabies in Southern African wildlife: Reservoir hosts, transmission dynamics, and control implications. Open Vet. J.. 2026; 16(1): 56-70. doi:10.5455/OVJ.2026.v16.i1.5 Vancouver/ICMJE Style Eze UU, Sabeta CT. A review of rabies in Southern African wildlife: Reservoir hosts, transmission dynamics, and control implications. Open Vet. J.. (2026), [cited January 31, 2026]; 16(1): 56-70. doi:10.5455/OVJ.2026.v16.i1.5 Harvard Style Eze, U. U. & Sabeta, . C. T. (2026) A review of rabies in Southern African wildlife: Reservoir hosts, transmission dynamics, and control implications. Open Vet. J., 16 (1), 56-70. doi:10.5455/OVJ.2026.v16.i1.5 Turabian Style Eze, Ukamaka Uchenna, and Claude Taurai Sabeta. 2026. A review of rabies in Southern African wildlife: Reservoir hosts, transmission dynamics, and control implications. Open Veterinary Journal, 16 (1), 56-70. doi:10.5455/OVJ.2026.v16.i1.5 Chicago Style Eze, Ukamaka Uchenna, and Claude Taurai Sabeta. "A review of rabies in Southern African wildlife: Reservoir hosts, transmission dynamics, and control implications." Open Veterinary Journal 16 (2026), 56-70. doi:10.5455/OVJ.2026.v16.i1.5 MLA (The Modern Language Association) Style Eze, Ukamaka Uchenna, and Claude Taurai Sabeta. "A review of rabies in Southern African wildlife: Reservoir hosts, transmission dynamics, and control implications." Open Veterinary Journal 16.1 (2026), 56-70. Print. doi:10.5455/OVJ.2026.v16.i1.5 APA (American Psychological Association) Style Eze, U. U. & Sabeta, . C. T. (2026) A review of rabies in Southern African wildlife: Reservoir hosts, transmission dynamics, and control implications. Open Veterinary Journal, 16 (1), 56-70. doi:10.5455/OVJ.2026.v16.i1.5 |