| Research Article | ||
Open Vet. J.. 2025; 15(12): 6329-6341 Open Veterinary Journal, (2025), Vol. 15(12): 6329-6341 Research Article Assessment of seroprevalence and associated risk factors of Chlamydia abortus among sheep and women with a history of abortion in Northern EgyptHaitham Elaadli1, Yassien Badr2, Magdy A. A. Ayad3*, Asmaa G. Mubarak4, Waleed Younis5, Ehab K. Elmahallawy6,7, Samar A. Eissa8 and Raafat M. Shaapan91Department of Animal Hygiene and Zoonoses, Faculty of Veterinary Medicine, Alexandria University, Alexandria, Egypt 2Department of Infectious Diseases and Epidemics, Faculty of Veterinary Medicine, Damanhour University, Damanhour, Egypt 3Department of Microbiology and Parasitology, Faculty of Veterinary Medicine, Tripoli University, Tripoli, Libya 4Department of Zoonoses, Faculty of Veterinary Medicine, South Valley University, Qena, Egypt 5Department of Microbiology, Faculty of Veterinary Medicine, South Valley University, Qena, Egypt 6Departamento de Sanidad Animal, Grupo de Investigación en Sanidad Animal y Zoonosis, Universidad de Córdoba, Córdoba, Spain 7Department of Zoonoses, Faculty of Veterinary Medicine, Sohag University, Sohag, Egypt 8Department of Medical Microbiology and Immunology, Faculty of Medicine, Kafrelsheikh University, Kafrelsheikh, Egypt 9Department of Zoonotic Diseases, Veterinary Research Institute, National Research Centre, Giza, Egypt *Corresponding Author: Magdy A. A. Ayad. Department of Microbiology and Parasitology, Faculty of Veterinary Medicine, University of Tripoli, Tripoli, Libya. Email: magdyaa73 [at] yahoo.com Submitted: 25/08/2025 Revised: 05/11/2025 Accepted: 16/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
AbstractBackground: Chlamydia abortus (C. abortus) remains a notable zoonotic pathogen that is primarily responsible for abortion in sheep and goats. Despite its veterinary and zoonotic concerns. Aim: This study aimed to assess the prevalence of C. abortus in humans and animals in northern and coastal Egypt and the associated risk factors. Methods: A total of 196 blood samples of apparently healthy and aborted sheep were collected from different herds in three Egyptian governorates. A total of 242 blood samples were collected from women with a history of abortion admitted at El Shatby Hospital in Alexandria, Egypt. The collected samples were serologically tested for specific anti-C. abortus antibodies using an enzyme-linked immunosorbent assay employing the major outer-membrane protein peptide followed by the investigation of various risk factors. Results: Chlamydia abortus Immunoglobulin G (IGg) antibodies could be detected in 12.2% of the sheep examined. Five sociodemographic characteristics were significantly associated with anti-C. abortus IgG antibody positivity in sheep: breed (X2=27.543; p=0.000), governorate (X2=33.564; p=0.000), breeding system (X2=24.298; p=0.000), pregnancy status (p=0.003), and history of abortion (p=0.000). On the other hand, anti-C. abortus IgM antibodies could be detected in the sera of aborted women at a rate of 13.2%. By conducting a Pearson chi-square, a statistically significant association was detected between the infection in aborted women and residence (X2=27.428; p=0.000), contact with sheep (X2=77.744; p=0.000), history of miscarriage (X2=22.044; p=0.000), and time of miscarriage (X2=54.550; p=0.000). Conclusion: To the best of our knowledge, this study is the first to address the seroprevalence of C. abortus in humans and the potential risk factors associated with exposure in northern and coastal Egyptian regions. Furthermore, it provides an updated overview of this zoonotic pathogen’s seroprevalence among sheep in Northern Egypt. Keywords: Chlamydia abortus, Aborted women, Sheep, Risk factors. IntroductionChlamydia abortus (formerly, Chlamydia psittaci serovar 1) is a Gram-negative, obligatory intracellular bacterium. This pathogen has several reservoirs, including sheep, goats, and other livestock. It is one of the most common abortive agents in sheep and the leading cause of enzootic abortion of ewes or ovine enzootic abortion, with a global distribution (Longbottom et al., 2013; Hireche et al., 2016; Tejedor-Junco et al., 2019). Furthermore, it represents a potential zoonotic risk for susceptible veterinarians, workers, farmers, and pregnant women (Longbottom and Colter, 2003; OIE, 2018). This pathogen shares a unique biphasic developmental cycle with all Chlamydiaceae species (Borel et al., 2018a). The placenta, fetuses, and uterine discharge of aborted animals are the primary sources of C. abortus, which tends to transmit the infection orally and nasally to unimmunized sheep, farmers, and veterinarians (Hireche et al., 2016; Mamlouk et al., 2020). Conversely, humans can contract the disease by improperly handling placenta and postpartum secretions, inhaling infective aerosols after contact with infected animals or secondary contaminated environments, or by consuming contaminated food. The clinical profile of C. abortus infection in sheep includes stillbirth, the delivery of weak lambs that die after 48 hours, and abortion, which typically occurs in the last 2–3 weeks of pregnancy. Infected sheep have also exhibited other clinical signs, such as orchitis, pneumonia, encephalomyelitis, enteritis, conjunctivitis, and seminal vasculitis (Öztürk et al., 2016; Arif et al., 2020; Zhang et al., 2020). Although ewes that are aborted due to C. abortus are thought to be immune, this protection does not stop infected ewes from shedding bacteria, and the animals may remain persistently infected. This route represents the most significant source of infection in naive animals. They may also abort in the next lambing season (Essig and Longbottom, 2015). The spread of this enzootic infection to humans represents another critical issue. Although C. abortus infection in humans usually presents as a mild influenza-like illness (Rodolakis and Mohamad, 2010), some health issues have been identified, such as urethritis in men, spontaneous miscarriage, stillbirth, septicemia, or pelvic inflammatory disease in women (Longbottom et al., 2013; Essig and Longbottom, 2015). Chlamydia abortus is found in humans worldwide, with notable prevalence across North America, South America, Europe, Africa, and various regions across Asia and Oceania. According to the Centers for Disease Control and Prevention, 2.8 million people contract the disease annually in the United States alone, with reported infection rates increasing from 35.2 to 332.6 cases per 100,000 people between 1986 and 2005. According to recent reports on disease surveillance and emerging threats in the UK, C. abortus was the primary infectious agent causing fetal illness and reproductive problems in 2021–2022 and the second in 2023 (Nogarol et al., 2024). Increased screening and more accurate diagnosis methods may have contributed to this increase (Barrett and Stanberry, 2009). Regarding the occurrence of C. abortus infection in sheep, it has been recorded in numerous countries in the Middle East and disseminated worldwide, causing severe financial losses to sheep flocks. The seroepidemiological studies revealed prevalence rates of ovine chlamydiosis as 7.5% in Saudi Arabia (Aljumaah and Hussein 2012), 58% in Tunisia (Mamlouk et al., 2020), and 13.7% in Egypt (Selim et al., 2021). The presumptive clinical diagnosis of chlamydiosis is undoubtedly challenging due to the lack of distinct clinical features for C. abortus and the possibility of coinfection with other abortifacient etiological agents (Ababneh et al., 2014). Various serological techniques have been validated as sensitive and specific methods for conducting serosurvey and detecting pathogen exposure. The complement fixation test is commonly used for screening, but its specificity can be compromised due to cross-reactions with Chlamydia pecorum, which may lead to inaccurate results and affect diagnostic accuracy (Everett, 2000; Livingstone et al., 2005). Specific enzyme-linked immunosorbent assay (ELISA) kits that are commercially available have been developed and proven to be highly sensitive and specific for detecting antibodies against various chlamydial antigens. For instance, ID-Vet ELISA employs C. abortus major outer-membrane protein (MOMP) peptide as the coated antigen, whereas IDEXX ELISA uses C. abortus entire elementary bodies as a known antigen. When performing serological tests, it is important to consider the potential cross-reactivity caused by chlamydial lipopolysaccharide (LPS), present in all Chlamydia species, particularly C. pecorum. However, ELISA employing MOMP antigens has been validated to specifically distinguish chlamydial species-specific antibodies in animal sera, making them highly suitable for serological tests (Conlan et al., 1988; Mygind et al., 2000; Hoelzle et al., 2004; Mccauley et al., 2007). Notably, the serological aspects of C. abortus infection in humans have not been investigated, and few studies have been conducted on sheep in Egypt (Selim et al., 2018; Selim et al., 2021). Therefore, this study conducted a survey to assess the seroprevalence of C. abortus among women who had experienced abortions and were admitted to a regional women’s health hospital serving patients from three northern and coastal governorates in northern Egypt: Behera, Alexandria, and Matrouh. Simultaneously, the cross-sectional study determined serological prevalence in samples from apparently healthy sheep collected from the same provinces using the indirect ELISA technique. Risk factors concerning seropositive samples of women and sheep were also evaluated. Materials and MethodsDescription of the study areaA seroepidemiological survey was conducted between May 2021 and February 2022 to determine the seroprevalence of C. abortus in apparently normal sheep and women who had experienced abortions, while also identifying associated risk factors. Sheep samples were randomly collected from small-scale stallholders of different herds in each of the three governorates located in Northern Egypt: EL Beheira (situated in the western Delta of the River Nile, X/Y coordinates: 30.343551/30.848099), Alexandria (X/Y coordinates: 29.924526/31.205753), and Matrouh (X/Y coordinates 27.237316/31.354343). Both Alexandria and Matrouh are on Egypt’s west-north coast. Human samples were collected from El Shatby Hospital for Obstetricians and Gynecologists, which is affiliated with the College of Medicine at Alexandria University, Alexandria (Egypt), and acts as a central hub for medical services in the northern and coastal part of the country, particularly for cases from El Beheira, Alexandria, and Matrouh Governorates. A map of the study area is depicted in Figure 1.
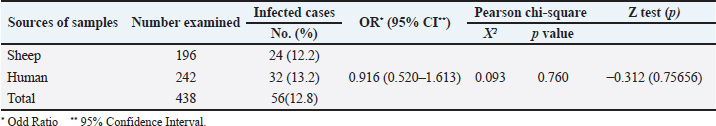
Fig. 1. Map of the studied area, El Beheira, Alexandria, and Matrouh Governorates in the northern and costal part of Egypt. Sample collection and processingA total of 196 blood samples were randomly collected from 18 small-scale sheep farms across three governorates. Six flocks were sampled from each governorate, with 10–11 samples collected per flock. These samples were collected from clinically healthy sheep, many of which had a history of abortion, with their owners providing informed oral consent. During sampling, data were gathered on various factors, including the sheep’s age, breed, pregnancy status, flock size, and household sanitation practices, such as post-parturition isolation of ewes with their lambs and disinfection of the lambing area. Additionally, 242 blood samples were collected from women with a history of abortion, aged 15–40 years, admitted to the labor wards at El Shatby Hospital for Obstetricians and Gynecologists in Alexandria, Egypt. Human samples were obtained with their consent. Relevant data, such as age, residence (by governorate), contact with sheep, abortion history, and miscarriage trimester, were obtained to assess the risk factors influencing C. abortus infection in humans. All blood samples were collected in vacutainer tubes with clotting factor and then sent to the laboratory in an icebox. After centrifugation at 1,000-× g for 15 minutes, sera from each tube were transferred into 1.5 ml Eppendorf tubes marked with the identifier label of each sample and preserved at −20oC until further serological analysis. The sample size was determined to ensure a 95% probability of detecting parasite exposure, assuming a minimum individual prevalence rate of 13.7% (Shaapan et al., 2010). Serological examinationAll sheep serum samples were tested for the presence of specific anti-C. abortus antibodies using a commercially available indirect ELISA kit (ID Screen® Chlamydophila abortus Indirect Multi-species, Product code: CHLMS-MS-2P, Innovative Diagnostic Vet Laboratories, France). The ELISA plates were coated with MOMP specific to C. abortus, thereby reducing the frequency of non-specific reactions. The kit provided positive and negative control animal sera for test validation. The optical densities of the samples were detected using an ELISA reader at 450 nm. The sample-to-positive ratio (S/P) % was calculated for result interpretation, and serum samples with S/P% ≤ 50%, ≥60%, and 50%–60% were considered negative, positive, and doubtful, respectively. According to the manufacturer’s instructions, the assay was considered valid if the average optical density (OD) of the positive controls was > 0.35 and the ratio of the mean OD value of the positive controls to the mean OD value of the negative controls was > 3. The estimated sensitivity and specificity of this kit were 100% and 99.7%, respectively (Pourquier et al., 2007). Three conjugates were used in this study to detect anti-C. abortus-specific antibodies in the serum samples of all women. These conjugates were the anti-multi-species IgG-Horseradish peroxidase (HRP) conjugate of the ELISA kit mentioned above, peroxidase-conjugated anti-human IgG secondary antibody (I2136-1ML, Sigma) at a dilution of 1:1000, and HRP conjugated anti-human IgM secondary antibody (K0211485, Koma Biotech) at a dilution of 1:1000. The three conjugates were used in separate ELISA runs to confirm the serological testing results of serum specimens from aborted women. Checkerboard titration was employed to estimate the antigen, serum, and conjugate concentrations. Negative mean OD values + 2SD were used to deconjugate the cut-off value (Hassanain et al., 2016). Assessment of risk factorsThe correlation between risk variables and the seroprevalence of C. abortus in sheep and women was determined using logistic regression analysis. Age group, breed, habitat, pregnancy status, history of abortion, breeding system, and hygienic measures were the six risk factors examined in sheep. In humans, six risk factors were examined, including age, habitat, place of residency, contact with sheep, previous miscarriages, and trimester of miscarriage were examined. (Abd El Wahab et al., 2018). Statistical analysisThe resulting data were statistically analyzed using SPSS version 22. Chi-square and Fisher’s exact tests were used to investigate the association between each factor and the prevalence rate of C. abortus. Fisher’s exact test was also used when >20% of cells had an expected value ˂5. A logistic regression model was used to assess the potential relationship between variables (Abouelsoued et al., 2020). Differences in the seroprevalence rates were considered statistically significant if the p-value ˂ 0.05. The odds ratios and 95% confidence intervals were calculated for each factor. Ethical approvalThe research protocol was approved by the Ethics Committee of the Faculty of Medicine, Alexandria University (Approval No. 0305821 at June 14, 2024). All participants provided informed consent. For individuals aged 15–20 years, consent was also secured from their parents, who were fully informed about the study’s objectives. Furthermore, the ethics committee of South Valley University in Qena, Egypt, authorized the protocol (No. 76/02. 10.2022). The study was conducted under the Animals in Research: reporting in vivo experiments (ARRIVE) criteria (Du Sert et al., 2020). ResultsSeroprevalence of C. abortus among sheep and humansThis study observed an overall seroprevalence rate of C. abortus of 12.8% (56/438) among the examined cases. The seroprevalence was 12.2% (24/196) in sheep serum samples and slightly higher at 13.2% (32/242) in serum samples from women who had experienced abortions (Table 1). The C. abortus serological assay, utilizing the anti-multi-species IgG-HRP conjugate and peroxidase-conjugated anti-human IgG secondary antibody (I2136-1ML, Sigma), did not detect antibodies in the examined human serum samples. Therefore, it is assumed that IgM antibodies may be circulating due to recent miscarriages in the women who underwent abortion. Accordingly, an ELISA immunoassay was conducted in the final step using HRP-conjugated anti-human IgM secondary antibody (K0211485, Koma Biotech), which detected anti-C. abortus IgM antibodies in the tested human sera samples. The Pearson chi-square test did not indicate a statistically significant difference (χ2=0.093; p=0.760) between the seropositivity of C. abortus in sheep and humans, supporting the null hypothesis. Table 1. Prevalence of Chlamydia abortus infection in the examined sheep and human samples.
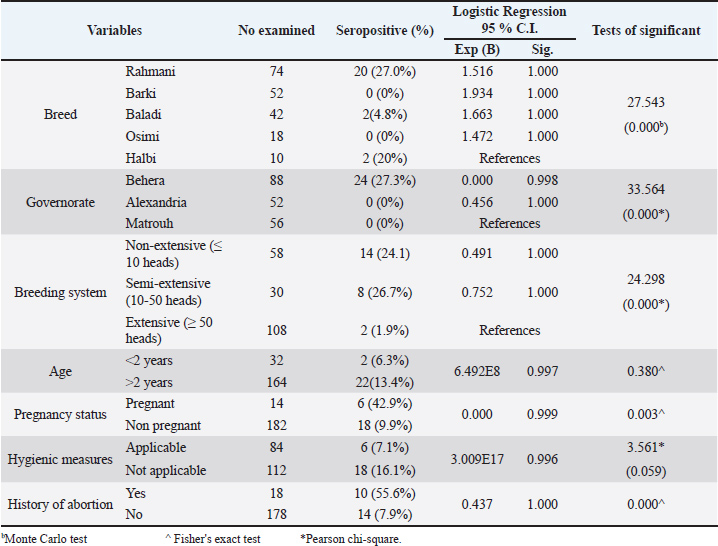
Seroprevalence of anti-C. abortus antibodies in sheep and assessment of risk factorsTable 2 shows the seroprevalence among different categories of sheep used in the current study. The seropositivity rate varied significantly (p=0.000) among sheep breeds, with the highest rate detected in the Rahmani breed (27%; 20/74), followed by Halbi (20%; 2/10) and Baladi (4.8%; 2/42). No antibodies were detected in the sera of the Barki and Osimi breeds. The logistic regression revealed that all breeds were considered as risk factors compared to the Halbi breed, but without statistical significance (p= 1.000). Additionally, antibodies were detected only in sheep sera collected from Behera Governorate (27.3%; 24/88), while they were not detected in samples collected from the other two governorates, Alexandria and Matrouh. Notably, Alexandria can be regarded as a protective factor (Exp=0.456) compared with Matrouh. The breeding system (flock size) additionally significantly (X2=24.298; p=0.000) affected C. abortus seropositivity, with the highest prevalence (26.7%; 8/30) detected in the semi-extensive system and the lowest (1.9%; 2/108) in the extensive system. Table 2. Prevalence of Chlamydia abortus infection in the examined sheep in relation to the associated risk factors.
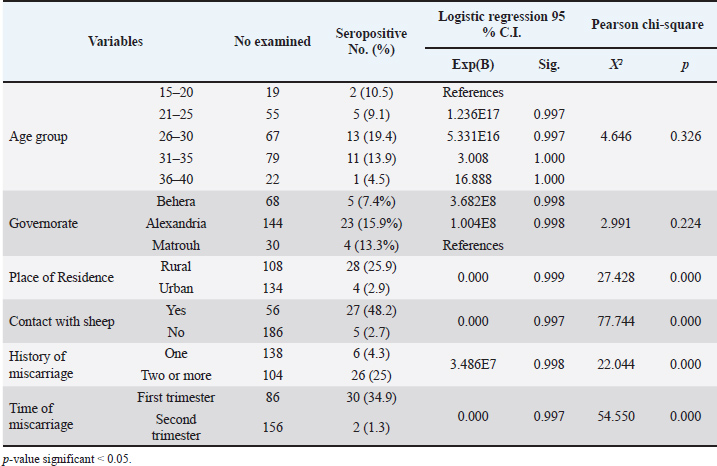
Regarding the sociodemographic characteristics presented in Table 2, the seroprevalence of older animals (>2 years old) was 13.4% (22/164), which was higher than the 6.3% (2/32) observed in younger animals (< 2 years old). However, this difference was not statistically significant (Exp=6.492E8; sig=0.997). Fisher’s exact test revealed no statistical association between age and C. abortus infection (p=0.380). However, pregnancy status was a significant factor associated with C. abortus infection (p=0.003), as pregnant ewes exhibited a higher seroprevalence of 42.9% (6/14) compared to 9.9% (18/182) in non-pregnant ewes. Despite this difference, no statistical significance was found between these groups (Exp=0.000; sig=0.999). Seroprevalence of anti-C. abortus antibodies in women with abortion and risk factor assessmentTable 3 illustrates the associations between C. abortus infection and various sociodemographic characteristics and risk factors among the women who underwent abortion. The highest seroprevalence rate was observed in participants aged 26–30 years (19.4%), followed by those aged 31–35 years (13.9%), 15–20 years (10.5%), and 21–25 years (9.1%). The lowest prevalence (4.5%) was found in the age group of 36–40 years. Although logistic regression indicated that all age groups were associated with an increased risk of infection compared with the reference group (15–20 years), these associations were not statistically significant. The Pearson chi-square test also confirmed that age was not significantly related to C. abortus infection incidence (X²=4.646; p=0.326). Similarly, the prevalence rates across the governorates were highest in Alexandria (15.9%), followed by Matrouh (13.3%) and Behera (7.4%). Table 3. Risk factors associated with C. abortus seropositivity in aborted women.
Among the 242 aborted women surveyed, 108 were from rural areas, showing a significantly higher rate of anti-C. abortus antibodies (25.9%) than the 134 participants from urban areas (2.9%). The prevalence of anti-C. abortus antibodies were notably higher among those who had contact with sheep (48.2%) than those who did not (2.7%), indicating a strong association between sheep contact and C. abortus infection among aborted women (X²=77.744; p=0.000). The logistic regression analysis suggested that both the place of residence and contact with sheep were associated with protective factors, with extremely low significance values (Exp=0.000, p=0.999, and p=0.997, respectively). DiscussionChlamydia abortus infection is a leading cause of abortion in sheep (Essig and Longbottom, 2015; Selim, 2016). In a survey conducted in Egypt examining the seroprevalence of four abortifacient agents, C. abortus ranked second in prevalence, after Brucella species (Selim et al., 2018). In addition, the pathogen holds significant public health importance because of its potential to be transmitted to humans, where it can induce abortion in pregnant women (Turin et al., 2022). This study presents an updated serosurvey of the pathogen in sheep from the northern and coastal regions of the country and, for the first time, provides novel data on the seroprevalence in humans, which might reflect the potential for transmission through contact with infected animals. Furthermore, the study explores various risk factors associated with infection by this pathogen. The overall seroprevalence of C. abortus in sheep in this study (12.2%) is relatively lower than that of Selim et al. (2018) and Selim et al. (2021) in northern Egypt at 14.9% and 13.7%, respectively. To the best of the researchers’ knowledge, previous studies on seroprevalence of C. abortus in Egypt have focused on governorates along the north coast and the Nile Delta regions, with no studies investigating governorates in the southern areas of the country. Comparing the seroprevalence rates from this study with those from other investigations in the Middle East and North Africa, a few studies reported higher seroprevalence rates, such as in Western Iran (26.7%) (Esmaeili et al., 2018) and Jordan (21.8%) (Al-Qudah et al., 2004). In contrast, lower seroprevalence rates were observed in Tunisia (6.6%) (Mamlouk et al., 2020), Saudi Arabia (11.1%) (Fayez et al., 2021), northeastern Iran (10.3%) (Iraninezhad et al., 2020), Syria (11.53%) (Al-Tabbaa et al. (2020), and Algeria (7.2%) (Hireche et al., 2016). The varying seroprevalence observed in different countries can most likely be linked to variations in animal welfare, husbandry practices, climates, and sanitation (Elaadli et al., 2023). This study highlighted that breed significantly influences the risk of pathogen exposure, a factor that was largely overlooked in previous studies in Egypt. Contrary to these findings, Fayez et al. (2021) found no breed-related differences in C. abortus seroprevalence in Saudi Arabia. Considering the limited national-level information on this topic, further research is needed to validate these findings and elucidate the potential relationship between sheep breeding and chlamydiosis. The differences in positive rates among different sheep breeds in the obtained results can be explained by differences in environmental factors such as heat stress, physiological markers, and genetic makeup. These factors can significantly impact immunity and health among sheep breeds and are likely the root cause of varied immune responses (Hadfield et al., 2018). Innate immune response to a LPS challenge varies among sheep breeds; hence, there are differences between breeds in response to infection (Eaton et al., 2007). The Barki and Osimi breeds are highly adapted to arid and hot environments, suggesting a strong physiological and potentially immunological resilience to heat stress. Studies show that the Barki breed is better adapted than the Rahmani breed to withstand heat stress, with less pronounced seasonal changes in some biomarkers. Otherwise, the Rahmani ewe breed shows higher serum total protein and albumin concentrations than other ewe breeds, which might indicate greater productivity but weaker immune response. The Ossimi breed shows higher levels of thyroid hormones and leptin and better reproductive performance than the Barki breed, but this may reflect changes in their immune response (El-Malky et al., 2019). This study identified Behera as the only governorate with positive serum samples in sheep, showing a seroprevalence of 27.3%. In contrast, Selim et al. (2018) and Selim et al. (2021) reported higher seroprevalences of C. abortus in Alexandria (29.6% in sheep and 19.2% in goats), with a lower exposure rate in Behera (13.8%). These discrepancies may be attributed to differences in sampling locations within Alexandria or to differences in sample sizes between studies. To confirm these findings, further research is needed to explore various geographical areas with larger sample sizes. Additionally, the desert environment, low population densities, and reliance on nomadic flocks of the Matrouh Governorate may also require investigation due to its distinct conditions. In this study, a significant difference was observed in the seroprevalence among flocks of different sizes, with semi-extensive systems exhibiting the highest infection rate, followed by non-extensive and extensive systems. This highlights the potential role of animal density as a factor associated with infection exposure. This finding is consistent with that of Nogarol et al. (2024), who considered herd size as a proxy for herd management, with a significant association with the C. abortus infection rate. Conversely, these results contradict other studies conducted in Egypt (Selim et al., 2021) and Tunisia (Mamlouk et al., 2020), which observed no significant difference between flocks of different sizes. Additionally, Fayez et al. (2021) reported that sheep flocks larger than 200 heads have higher seropositivity than those with fewer than 200 heads. Other factors, including hygiene practices and management style, could influence prevalence, although the impact may vary depending on the flock size. Regarding the individual risk factors, the study pointed out that animals older than 2 years had a higher prevalence (13.4%), although not statistically significant, than the younger group (6.3%); this difference might be due to a higher likelihood of exposure in older animals and protective immunity in younger ones. These findings are consistent with those of other studies conducted in Egypt (Selim et al., 2021; Elaadli et al., 2023). However, these results contradict the findings reported by Sun et al. (2020), who recorded the highest C. abortus seroprevalence (25.40%) among the youngest age group. In addition, Nogarol et al. (2024) reported that animals aged 1–2 years exhibited a nearly two-fold increase in C. abortus seropositivity compared with those aged over 4 years, which may be attributed to the low levels of antibodies. On the other hand, Mamlouk et al. (2020) and another in Iran (Iraninezhad et al., 2020), detected no difference in the seroprevalence based on age groups older and younger than 3 years. Conversely, in Saudi Arabia, the age range of 1.4–2.8 years was identified as a risk factor compared to the <1.4 year (Fayez et al. (2021). Similarly, another study in Iran (Esmaeili et al., 2018) indicated that seropositivity was highest in the 1-2-year-old group. The discrepancy in the findings across different studies may be attributed to differences in flock management, sampling approaches, or age categorization (Mahmoud et al., 2019). Moreover, the varying incidence across age groups suggests the probability of horizontal transmission in the investigated animals. The role of vertical transmission of C. abortus from ewes to lambs was not sufficiently studied (Rodolakis and Bernard, 1977), and its role in the spreading of C. abortus infection needs to be clarified. Pregnant animals exhibited a higher seroprevalence rate of 42.9%, in contrast to 9.9% in nonpregnant animals. This heightened prevalence among pregnant animals can be attributed to various factors. Pregnancy often represents a stressful phase that can lead to a weakened immune system and increased susceptibility to infections. In pregnant animals, C. abortus invades the placentome, and a higher level of antibodies may be detected during placental infection. Pregnant animals are typically older and more likely to interact with other animals during breeding. In Egypt, traditional sheep management practices may further increase the exposure risk. Some sheep owners send breeding females to other flocks with breeding rams for insemination, whereas others practice ram exchange during specific periods. These practices increase the likelihood of breeding females being exposed to infection. This study revealed that implementing hygienic measures effectively reduced the seroprevalence of chlamydia. The primary source of C. abortus in sheep environments is abortion materials, including placenta, vaginal discharge, abortive dead, and stillbirth lambs (Essig and Longbottom, 2015). Chlamydiae exhibit remarkable resilience to unfavorable conditions when released into the atmosphere. While they may remain contagious for a few days in the spring when temperatures are warm, they can endure lower temperatures for weeks or even months in the winter (Turin et al., 2022). This extended persistence in the environment increases the possibility of contact with susceptible, particularly pregnant, animals. Therefore, hygienic measures, such as isolation of ewes with their kids and environmental disinfection, may reduce the transmission of the agent after abortion or parturition. These results are in accordance with the findings of Mamlouk et al. (2020); Fayez et al. (2021); and Selim et al. (2021), who observed that the application of post-abortion disinfection, the presence of lambing barns, and rapid selling or discarding of animals’ manure can significantly reduce the risk of infection compared with herds lacking such activities. A history of abortion was identified as another significant risk factor for seropositivity in this study. Animals with a history of abortion had a much higher exposure rate (55.6%) than those that had never experienced abortion (7.9%). This may imply that C. abortus plays a substantially greater role as a cause of abortion in sheep than expected. Similarly, some previous studies reported significantly higher seroprevalence in animals with a history of abortion (Esmaeili et al., 2018; Fayez et al., 2021). Nevertheless, the high seroprevalence of C. abortus infection in previously aborted sheep does not exclude the probable role of other abortifacient agents in inducing abortion in sheep. For example, a serological survey of four abortifacient infectious agents among sheep and goats in Egypt found that Brucella species was the most prevalent, followed by C. abortus. Moreover, the interaction between Brucella species and C. abortus had the highest significant effect on the incidence of abortion (Selim et al., 2018). Another crucial aspect of this study is highlighting the seroprevalence of this pathogen in women who underwent abortion. Although C. abortus is not widely recognized as a human pathogen, it is a zoonotic pathogen that can cause various signs in humans, including subclinical infection and severe disease in pregnant women (Nogarol et al., 2024). Studies on miscarriage-related infections with C. abortus in pregnant women are scarce; only case-report studies have been conducted. At the national level, no previous studies have assessed the prevalence of human C. abortus infection and associated risk factors. This current investigation detected anti-C. abortus IgM antibodies in 13.22% of the examined aborted women, indicating the presence of a recent infection. Various authors have reported that humans, particularly pregnant women, are susceptible to C. abortus infection (Longbottom and Coulter, 2003; Aitken and Longbottom, 2007; Essig and Longbottom, 2015; OIE, 2018; Sing, 2023). For instance, Walder et al. (2005) recorded a case of C. abortus infection in a 32-year-old woman who was infected during the 16th week of pregnancy, and fetal death occurred after 3 days of fever. In addition, a case of severe C. abortus infection, including stillbirth and a sepsis-like disease, was observed in pregnant women with a history of contact with infected goats (Meijer et al., 2004). On the other hand, Walder et al. (2003) detected C. abortus in non-pregnant women and confirmed a conclusive case of C. abortus infection in a 39-year-old woman suffering from increased vaginal discharge, abnormal menstruation, and chronic abdominal pain using C. abortus-specific Polymerase Chain Reaction (PCR) and the presence of anti-chlamydia antibodies. Furthermore, Borel et al. (2018b) revealed the presence of that microorganism in the colon of an 81-year-old female patient via Deoxyribonucleic acid (DNA) microarray analysis of an intestinal biopsy. Recently, Burgener et al. (2022) emphasized that C. abortus is a zoonotic pathogen that causes severe illness during pregnancy and can be life-threatening to both the mother and her fetus. Furthermore, men are additionally at risk; Gong et al. (2023) demonstrated C. abortus infection in a 51-year-old man who had shortness of breath and weakness in the extremities with irregular, high-density shadows observed in the right lung by computed tomography scan of the chest. The spread of the disease to humans is largely driven by environmental contamination caused by bacteria from abortion materials or infected animals. Abortion-related materials, including vaginal fluids, placentas, aborted lambs, and feces, are particularly contaminated with bacteria. These substances pose a significant threat to both humans and vulnerable animals, as they can effectively spread the infection (Essig and Longbottom, 2015). Additionally, a spore-like cell wall in C. abortus enhances its durability, enabling it to persist in the environment even under adverse conditions. In this study, various risk factors associated with C. abortus seroprevalence in women were assessed. The results demonstrated a non-significant increased rate of anti-C. abortus IgM antibodies in the age group of 26–30 years (19.4%), followed by the 31–35 age group (13.9%). Pichon et al. (2020) reported a case of C. abortus in a 27-year-old pregnant woman in rural France who then suffered fetal loss. Additionally, Burgener et al. (2022) detected C. abortus in a 33-year-old pregnant woman who lived on a farm with 200 sheep at 19 weeks of her second pregnancy. Notably, there was no direct correlation between age and the C. abortus infection rate in these women. However, the higher seropositivity rates observed in these two age groups (26–30 and 31–35) may be attributed to the higher likelihood of women in these age ranges being involved in animal handling and care. The highest seroprevalence rates were observed in Alexandria and Matrouh (15.9% and 13.3%, respectively), which are located in a humid region with similar climate conditions, whereas a lower seroprevalence rate was detected in Behera (7.4%), but without statistical disparity. Mamlouk et al. (2020) and Selim et al. (2021) reported that climate might play a significant role in the prevalence of C. abortus infection and shedding. Similarly, Essig and Longbottom (2015) explained that lower temperatures and higher humidity contribute to the organism’s prolonged survival in the environment. The significant association between the residence of aborted women (p=0.000) and the presence of anti-C. abortus IgM antibodies in this study underscores the potential risk of infection from exposed animals, particularly sheep and goats, as key sources of transmission. This study observed that rural residents had a higher risk of being C. abortus seropositive than urban residents at 25.9% and 2.9%, respectively. There are numerous reports of pregnant women contracting severe infections after exposure to animals infected with C. abortus, leading to spontaneous abortion, stillbirth, and septicemia, especially during the lambing or kidding season. This exposure can occur through direct contact with animals or their products or by inhaling contaminated dust and aerosols (Walder et al., 2003; Meijer et al., 2004; Walder et al., 2005). The data obtained from the present study revealed that the rate of exposure to C. abortus was higher among sheep keepers than among those who did not come into contact with sheep. These results were consistent with those of Burgener et al. (2022), who confirmed evidence of zoonotic transmission of C. abortus causing stillbirth in a 33-year-old pregnant farmer during the 19th week of her gestation after assisting in the expulsion of a deceased fetus at a sheep farm in Switzerland. Additionally, Pospischil et al. (2022) reported a case of spontaneous abortion in a pregnant female during the 19th week of her pregnancy after close contact with aborted does following an abortion outbreak at a goat farm in Switzerland. Similarly, Walder et al. (2005) recorded a case of intrauterine fetal death in a 32-year-old pregnant woman during the 16th week of pregnancy who presented with escalating septicemia after prolonged exposure to aborted material from her goat herd in Sterzing City, northern Italy. This finding can be attributed to unhygienic handling of infected placentas, unsanitary manipulation of aborting fetuses, and direct contact with infectious post-abortion secretions, vaginal discharges, fetal membranes, and infective aerosols, particularly during lambing and kidding seasons (Essig and Longbottom, 2015). Additionally, this contact can be indirect, such as coming into contact with infected fomites, wearing and handling contaminated footwear and clothes (Turin et al., 2022). Staying on or residing next to a farm where enzootic ovine abortion occurred (Meijer et al., 2004; Cheong et al., 2019; Pichon et al., 2020), and working with animals that have recently had the live attenuated C. abortus vaccine or have experienced an inoculation event via self-injection during administration of the vaccine mentioned above (Longbottom et al., 2018). Moreover, substantial amounts of C. abortus are released in the vaginal secretions of infected animals within 2 weeks before and after abortion. Even in the absence of abortion, C. abortus bacteria can spread via the infective placentas and vaginal discharges of naturally infected animals at the time of parturition. Besides, it can additionally be detected in trace levels in milk, feces, and urine after an abortion (Pichon et al., 2020; Turin et al., 2022). Furthermore, a significant correlation was noted between the seropositivity to C. abortus and a history of miscarriage in the examined aborted women, as reflected by a higher seroprevalence rate in females with recurrent spontaneous abortions. Interestingly, no prior research, case studies, or brief communications in the scientific literature indicating that C. abortus can cause repeated miscarriages in pregnant females are available. Meanwhile, healthcare professionals must receive education on the importance of thorough anamnesis. Additionally, pregnant women in rural communities should be made aware of the risks associated with severe zoonotic infections (Pichon et al., 2020). In addition, obstetricians and gynecologists should consider that C. abortus can be a potential zoonotic etiological agent in cases of recurrent miscarriage or pregnancy loss, especially in women who are close to small ruminants throughout their delivery and/or abortion. In addition, women who experienced early miscarriages in the first trimester were more susceptible to infection than those who had late miscarriages in the second trimester. This result contradicts the findings of Burgener et al. (2022). Pospischil et al. (2022) and Walder et al. (2005) described the clinical manifestations of C. abortus infection during second-trimester miscarriages. As anticipated, this finding aligns with Quenby et al. (2021) Selim et al. (2021) finding that almost 80% of clinical miscarriage cases occur in the first 12 weeks of gestation. This result underscores the need to enhance the awareness of physicians regarding C. abortus as a potential zoonotic pathogen. Clinicians should include C. abortus along with other infectious abortive agents in the panel of laboratory investigations following a preliminary diagnosis of spontaneous abortion, particularly during the first trimester. This study has a few limitations that should be considered when interpreting the findings. First, despite the high sensitivity and specificity of the ELISA, determining the presence of C. abortus IgG antibodies in serum samples from ewes could not confirm the presence of active infection with living and replicating C. abortus. Due to financial constraints, this study employed a single serological technique, and it provides a snapshot at one point in time, without follow-up to assess whether infections were temporary, recurrent, or chronic—a key area for future exploration. Furthermore, molecular characterization of C. abortus from vaginal swabs from aborted ewes and women will be useful in identifying genetic relatedness between different isolates. Despite these limitations, our study is the first to examine the seroprevalence of C. abortus in humans and the associated risk factors in the northern and coastal regions of Egypt. This study underscores the alarming exposure rates among women and offers an updated overview of the seroprevalence of the pathogen in sheep in Northern Egypt. ConclusionDespite the zoonotic importance of C. abortus, only a few studies have supported evolutionary and epidemiological investigations. The findings of this study confirm that C. abortus is a significant cause of miscarriages in women, as indicated by the presence of anti-C. abortus IgM antibodies. The study highlighted key risk factors associated with the exposure to C. abortus and clearly demonstrated the spread of the disease among sheep and aborted women in the study area. Further research is necessary to explore this issue more comprehensively in previous animal species, with an emphasis on developing and implementing risk maps for both humans and infected animals. Preventing C. abortus infection is the most effective strategy; therefore, pregnant women living in farming areas should be informed about the zoonotic risks and avoid contact with sheep, particularly during the lambing season. In addition, veterinarians and veterinary laboratory staff should be vigilant in their practices. AcknowledgmentsThe authors would like to express their gratitude to Prof. Dr. Wafaa Abo El-Enein. Dr. Ahmed Esmat and all members of the nursing staff The authors would like to thank the Department of Obstetrics and Gynecology, College of Medicine, Alexandria University, for their allowance and help in blood sampling from the studied aborted women admitted to the labor ward at El Shatby Hospital for Obstetricians and Gynecologists. We also thank all the veterinary practitioners for their assistance in collecting sheep samples. Conflict of interestThe author declares no conflict of interest. FundingThis study received no specific grant. Authors' contributionsH.E., R.M.S., and A.G.M. performed numerous activities in the existing study, including conceptualization, formal analysis, investigation, methodology development, software implementation, and validation. M.A.A.A., W.Y., Y.B., E.K.E., and S.A.E. wrote the original draft of the manuscript. The final version of the manuscript was read, reviewed, and approved by all authors. Data availabilityThe data supporting the findings of this study are available on request from the corresponding author, and no additional data sources are required. ReferencesAbabneh, H.S., Ababneh, M.M.K., Hananeh, W.M., Alsheyab, F.M., Jawasreh, K.I., Al-Gharaibeh, M.A. and Ababneh, M.M. 2014. Molecular identification of chlamydial cause of abortion in small ruminants in Jordan. Trop. Anim. Health. Prod. 46(8), 1407–1412; doi:10.1007/s11250-014-0654-x Abd El Wahab, W.M., Shaapan, R.M., Abd El Haf, M.E.D., Elfadaly, H.A. and Ahmed Hamd, D. 2018. Toxoplasma Gondii Infection and Associated Sociodemographic and Behavioral Risk Factors among Blood Donors. Asian. J. Epidemiol. 11(2), 52–58; doi: 10.3923/aje.2018.52.58 Abouelsoued, D., Shaapan, R., Elkhateeb, R.M., Elnattat, W., Abd Elhameed, M., Hammam, A.M.M. and Hammam, A.M. 2020. Therapeutic Efficacy of Ginger (Zingiber officinale), Ginseng (Panax ginseng) and Sage (Salvia officinalis) Against Cryptosporidium parvum in Experimentally Infected Mice. Egypt. J. Vet. Sci. 51(2), 241–151; doi:10.21608/EJVS.2020.24183.1152 Aitken, I.D. and Longbottom, D. 2007. Chlamydial abortion. Dis. Sheep 4(16), 105–112; https://wiley.com/doi/pdf/10.1002/9780470753316 Aljumaah, R.S. and Hussein, M.F., 2012. Serological prevalence of ovine and caprine chlamydophilosis in Riyadh region, Saudi Arabia. Serological prevalence of ovine and caprine chlamydophilosis in Riyadh region, Saudi Arabia. Afr. J. Microbiol. Res. 6(11), 2654–2658; doi:10.5897/AJMR11.1056 Al-Qudah, K.M., Sharif, L.A., Raouf, R.Y., Hailat, N.Q. and Al-Domy, F.M. 2004. Seroprevalence of antibodies to Chlamydophila abortus shown in Awassi sheep and local goats in Jordan. Vet. Med. 49(12), 460; doi:10.17221/5740-VETMED Al-Tabbaa, M.F., Allouz, A.K. and Alomar, Y. 2020. Seroprevalence and risk factors of Chlamydia abortus infection in awasi sheep in hama governorate in the middle region of Syria. Assiut Vet. Med. J. 66(167), 66; doi:10.21608/avmj.2020.168657 Arif, E.D., Saeed, N.M. and Rachid, S.K. 2020. Isolation and identification of Chlamydia abortus from aborted ewes in Sulaimani Province, Northern Iraq. Pol. J. Microbiol. 69(1), 65; doi:10.33073/pjm-2020-009 Barrett, A.D. and Stanberry, L.R. 2009. Vaccines for biodefense and emerging and neglected diseases. London: Academic Press. Available via https://elsevier.com/books/vaccines-for-biodefense-and-emerging-and-neglected-diseases/barrett/978-0-12-369408-9 Borel, N., Marti, H., Pospischil, A., Pesch, T., Prähauser, B., Wunderlin, S., Seth-Smith, H.M., Low, N. and Flury, R. 2018. Chlamydiae in human intestinal biopsy samples. Pathog. Dis. 76(8), fty081. Borel, N., Polkinghorne, A. and Pospischil, A. 2018. A review on chlamydial diseases in animals: still a challenge for pathologists?. Vet. Pathol. 55(3), 374–390; doi:10.1177/0300985817751218 Burgener, A.V., Seth-Smith, H.M.B., Kern-Baumann, S., Durovic, A., Blaich, A., Menter, T., Bruder, E., Roloff, T., Martinez, A., Borel, N., Albini, S., Hösli, I., Egli, A., Weisser, M. and Hinić, V. 2022. A case study of zoonotic Chlamydia abortus infection: diagnostic challenges from clinical and microbiological perspectives. Open Forum Infect. Dis. 9(10), 524; doi:10.1093/ofid/ofac524 Cheong, H.C., Lee, C.Y.Q., Cheok, Y.Y., Tan, G.M.Y., Looi, C.Y. and Wong, W.F. 2019. Chlamydiaceae: diseases in primary hosts and zoonosis. Microorganisms 7(5), 146; doi:10.3390/microorganisms7050146 Conlan, J.W., Clarke, I.N. and Ward, M.E. 1988. Epitope mapping with solid-phase peptides: identification of type–, subspecies–, species–and genus-reactive antibody binding domains on the major outer membrane protein of Chlamydia trachomatis. Mol. Microbiol. 2(5), 673–679; doi:10.1111/j.1365-2958.1988.tb00076.x Du Sert, N.P., Ahluwalia, A., Alam, S., Avey, M.T., Baker, M., Browne, W.J., Clark, A., Cuthill, I.C., Dirnagl, U., Emerson, M. and Garner, P. 2020. Reporting animal research: explanation and elaboration for the ARRIVE guidelines 2.0. PLos Biol. 18(7), e3000411; doi:10.1371/journal.pbio.3000411 Eaton, S.L., Rocchi, M., González, L., Hamilton, S., Finlayson, J., Sales, J., Jeffrey, M., Steele, P.J., Dagleish, M.P., Rodger, S.M., Reid, H.W. and Chianini, F. 2007. Immunological differences between susceptible and resistant sheep during the preclinical phase of scrapie infection. J. Gen. Virol. 88(4), 1384–1391; doi:10.1099/vir.0.82197-0 Elaadli, H., Abo El-makarem, H., Abd Elrahma, A.H., Shaapan, R.M. and Bessat, M. 2023. Prevalence and associated risk factors of Toxoplasma gondii infection in sheep and aborted women in Egypt. Iraqi. J. Vet. Sci. 37(2), 437–445; doi:10.33899/ijvs.2022.135777.2518 El-Malky, O.M., Mostafa, T.H., Ibrahim, N.H., Younis, F.E., Abd El-salaam, A.M. and Tag El-din, H.A. 2019. Comparison between productive and reproductive performance of Barki and Ossimi ewes under Egyptian conditions. Egypt. J. Sheep &. Goats Sci. 14(1), 61–82; doi:10.21608/ejsgs.2019.32573 Esmaeili, H., Bolourchi, M. and Mokhber-Dezfouli, M.R. 2018. Seroprevalence of Chlamydia abortus infection in sheep and goats in Iran. Iran. J. Vet. Med. 9(2), 73–77. Essig, A. and Longbottom, D. 2015. Chlamydia abortus: new aspects of infectious abortion in sheep and potential risk for pregnant women. Curr. Clin. Microbiol. Rep. 1, 22–34; doi:10.1007/s40588-015-0014-2 Everett, K.D.E. 2000. Chlamydia and Chlamydiales: more than meets the eye. Vet. Microbiol. 75(2), 109–126; doi:10.1016/S0378-1135(00)00213-3 Fayez, M., Elmoslemany, A., Alorabi, M., Alkafafy, M., Qasim, I., Al-Marri, T. and Elsohaby, I. 2021. Seroprevalence and risk factors associated with Chlamydia abortus infection in sheep and goats in Eastern Saudi Arabia. Pathog 10(4), 489; doi:10.3390/pathogens10040489 Gong, F., Chen, Z., Chang, J., Liu, J., Wang, X., Mo, W., Tan, M. and Jiang, T. 2023. Metagenomic next-generation sequencing contributes to the diagnosis of pneumonia caused by Chlamydia abortus in a male patient: case report and literature review. Infect. Drug. Resist. 16, 3463–3468; doi:10.2147/IDR.S407741 Hadfield, J.M., Bowdridge, E.C., Holásková, I., Elsasser, T.H. and Dailey, R.A. 2018. Breed-specific differences in the immune response to lipopolysaccharide in ewes. J. Anim. Sci. 96(10), 4220–4228; doi:10.1093/jas/sky288 Hassanain, M.A., Shaapan, R.M. and Khalil, F.A.M. 2016. Sero-epidemiological value of some hydatid cyst antigen in diagnosis of human cystic echinococcosis. J. Parasit. Dis. 40(1), 52–56; doi:10.1007/s12639-014-0443-5 Hireche, S., Ababneh, M.M.K., Bouaziz, O. and Boussena, S. 2016. Seroprevalence and molecular characterization of Chlamydia abortus in frozen fetal and placental tissues of aborting ewes in northeastern Algeria. Trop. Anim. Health Prod. 48(2), 255–262; doi:10.1007/s11250-015-0944-y Hoelzle, L.E., Hoelzle, K. and Wittenbrink, M.M. 2004. Recombinant major outer membrane protein (MOMP) of Chlamydophila abortus, Chlamydophila pecorum, and Chlamydia suis as antigens to distinguish chlamydial species-specific antibodies in animal sera. Vet. Microbiol. 103(1-2), 85–90; doi:10.1016/j.vetmic.2004.07.016 Iraninezhad, Z., Azizzadeh, M., Razavizadeh, A.T., Mehrzad, J. and Rashtibaf, M. 2020. Seroepidemiological feature of Chlamydia abortus in sheep and goat population located in northeastern Iran. Vet. Res. Forum. 11(4), 423; doi:10.30466/vrf.2019.101946.2429 Livingstone, M., Entrican, G., Wattegedera, S., Buxton, D., Mckendrick, I.J. and Longbottom, D. 2005. Antibody responses to recombinant protein fragments of the major outer membrane protein and polymorphic outer membrane protein POMP90 in Chlamydophila abortus-infected pregnant sheep. Clin. Vaccine Immunol. 12(6), 770–777; doi:10.1128/CDLI.12.6.770-777.2005 Longbottom, D. and Coulter, L.J. 2003. Animal chlamydioses and zoonotic implications. J. Comp. Pathol. 128(4), 217–244; doi:10.1053/jcpa.2002.0629 Longbottom, D., Livingstone, M., Maley, S., Van Der Zon, A., Rocchi, M., Wilson, K., Wheelhouse, N., Dagleish, M., Aitchison, K., Wattegedera, S., Nath, M., Entrican, G. and Buxton, D. 2013. Intranasal infection with Chlamydia abortus induces dose-dependent latency and abortion in sheep. PLos One 8(2), e57950; doi:10.1371/journal.pone.0057950 Longbottom, D., Sait, M., Livingstone, M., Laroucau, K., Sachse, K., Harris, S.R., Thomson, N.R. and Seth-Smith, H.M.B. 2018. Genomic evidence that the live Chlamydia abortus vaccine strain 1B is not attenuated and has the potential to cause disease. Vaccine 36(25), 3593–3598; doi:10.1016/j.vaccine.2018.05.042 Mahmoud, M.A.E.F., Ghazy, A.A. and Shaapan, R.M. 2019. Diagnosis and Control of Foot and Mouth Disease (FMD) in Dairy Small Ruminants; Sheep and Goats. Int. J. Dairy Sci. 14, 45–52; doi:10.3923/IJDS.2019.45.52 Mamlouk, A., Guesmi, K., Ouertani, I., Kalthoum, S., Selmi, R., Ben Aicha, E., Bel Haj Mohamed, B., Gharbi, R., Lachtar, M., Dhaouadi, A., Seghaier, C. and Messadi, L. 2020. Seroprevalence and associated risk factors of Chlamydia abortus infection in ewes in Tunisia. Comp. Immunol. Microbiol. Infect. Dis. 71, 101500; doi:10.1016/j.cimid.2020.101500 Mccauley, L., Lancaster, M., Young, P., Butler, K. and Ainsworth, C. 2007. Comparison of ELISA and CFT assays for Chlamydophila abortus antibodies in ovine sera. Aust. Vet. J. 85(8), 325–328; doi:10.1111/j.1751-0813.2007.00189.x Meijer, A., Brandenburg, A., De Vries, J., Beentjes, J., Roholl, P. and Dercksen, D. 2004. Chlamydophila abortus infection in a pregnant woman associated with indirect contact with infected goats. Eur. J. Clin. Microbiol. Infect. Dis. 23(6), 487–490; doi:10.1007/s10096-004-1139-z Mygind, P., Christiansen, G., Persson, K. and Birkelund, S. 2000. Detection of Chlamydia trachomatis-specific antibodies in human sera by recombinant major outer-membrane protein polyantigens. J. Med. Microbiol. 49(5), 457–465; doi:10.1099/0022-1317-49-5-457 Nogarol, C., Marchino, M., Scala, S., Belvedere, M., Renna, G., Vitale, N. and Mandola, M.L. 2024. Seroprevalence and risk factors associated with Chlamydia abortus infection in sheep and goats in North-Western Italy. Anim 14(2), 291; doi:10.3390/ani14020291 OIE. 2018. Enzootic abortion of ewes (ovine chlamydiosis). Manual of diagnostic tests and vaccines for terrestrial animals. Office International des Epizooties, pp: 1456–65. Available Via: https://www.oie.int/fleadmin/Home/eng/Health_standards/tahm/3.07.05_ENZ_ABOR.pdf Öztürk, D., Türütoğlu, H. and Kaya, M. 2016. Seroprevalence of Chlamydophila abortus infection in sheep in Burdur Province. Vet. J. Mehmet Akif Ersoy Univ. 1(2), 17–20. Pichon, N., Guindre, L., Laroucau, K., Cantaloube, M., Nallatamby, A. and Parreau, S. 2020. Chlamydia abortus in pregnant woman with acute respiratory distress syndrome. Emerg. Infect. Dis. 26(3), 628; doi:10.3201/eid2603.191417 Pospischil, A., Thoma, R., Hilbe, M. and Grest, P. 2002. Abortion in woman caused by caprine Chlamydophila abortus (Chlamydia psittaci serovar 1). Swiss. Med. Wkly. 132(0506), 64; https://smw.ch/index.php/smw/article/134/131/256 Pourquier, P., Rodalakis, A. and Mohamad, K.Y. Preliminary validation of a new commercial ELISA Kit for the detection of antibodies directed against C. abortus. In 13th Conference of the World Association of Veterinary laboratory Diagnosticians (WAVLD),2007 , p 11. Quenby, S., Gallos, I.D., Dhillon-Smith, R.K., Podesek, M., Stephenson, M.D., Fisher, J., Brosens, J.J., Brewin, J., Ramhorst, R., Lucas, E.S., Mccoy, R.C., Anderson, R., Daher, S., Regan, L., Al-Memar, M., Bourne, T., Macintyre, D.A., Rai, R., Christiansen, O.B., Sugiura-Ogasawara, M., Odendaal, J., Devall, A.J., Bennett, P.R., Petrou, S. and Coomarasamy, A. 2021. Miscarriage matters: the epidemiological, physical, psychological, and economic costs of early pregnancy loss. Lancet 397(10285), 1658–1667; doi:10.1016/s0140-6736(21)00682-6 Rodolakis, A. and Bernard, K. 1977. Isolation of Chlamydia ovis from genital organs of rams with epididymitis. Bull. Acad. Vet. Fr. 50(1), 65–69; https://www.org/doi/full/10.5555/19772297176 Rodolakis, A. and Mohamad, Y. K. 2010. Zoonotic potential of Chlamydophila. Vet. Microbiol. 140(3-4), 382–391; doi:10.1016/j.vetmic.2009.03.014 Selim, A. 2016. Chlamydophila abortus infection in small ruminants: a review. Asian J. Anim. Vet. Adv. 11(10), 587–593; doi:10.3923/ajava.2016.587.593 Selim, A., Elhaig, M. and Moawed, S. 2018. A serological survey of four abortifacient infectious agents among small ruminant in Egypt. Asian J. Anim. Vet. Adv. 13(2), 114–121; doi:10.3923/ajava.2018.114.121 Selim, A., Manaa, E.A., Waheed, R.M. and Alanazi, A.D. 2021. Seroprevalence, associated risk factors analysis and first molecular characterization of Chlamydia abortus among Egyptian sheep. Comp. Immunol. Microbiol. Infect. Dis. 74, 101600; doi:10.1016/j.cimid.2020.101600 Shaapan, R.M., Hassanain, M.A. and Khalil, F.A.M. 2010. Modified agglutination test for serologic survey of Toxoplasma gondii infection in goats and water buffaloes in Egypt. Res. J. Parasitol. 5, 13–17; https://scialert.net/abstract/?doi=jp.2010.13.17 Sing, A. ed., 2023. Zoonoses: Infections Affecting Humans and Animals. Berlin, Germany: Springer. https://10.1007/978-94-017-9457-2.pdf Sun, L.X., Liang, Q.L., Hu, X.H., Li, Z., Yang, J.F., Zou, F.C. and Zhu, X.Q. 2020. First report of chlamydia seroprevalence and risk factors in domestic black-boned sheep and goats in China. Front. Vet. Sci. 363, 363.; doi: 10.3389/fvets.2020.00363 Tejedor-Junco, M.T., González-Martín, M., Corbera, J.A., Santana, A., Hernández, C.N. and Gutiérrez, C. 2019. Preliminary evidence of the seroprevalence and risk factors associated with Chlamydia abortus infection in goats on the Canary Islands, Spain. Trop. Anim. Health. Prod. 51(1), 257–260; doi:10.1007/s11250-018-1654-z Turin, L., Surini, S., Wheelhouse, N. and Rocchi, M.S. 2022. Recent advances and public health implications for environmental exposure to Chlamydia abortus: from enzootic to zoonotic disease. Vet. Res. 53(1), 37; doi:10.1186/s13567-022-01052-x Walder, G., Hotzel, H., Brezinka, C., Gritsch, W., Tauber, R., Würzner, R. and Ploner, F. 2005. An unusual cause of sepsis during pregnancy: recognizing infection with: Chlamydophila abortus. Obstet. Gynecol. 106(2), 1215–1217; doi:10.1097/01.AOG.0000161060.69470.9c Walder, G., Meusburger, H., Hotzel, H., Oehme, A., Neunteufel, W., Dierich, M.P. and Würzner, R. 2003. Chlamydophila abortus pelvic inflammatory disease. Emerg. Infec. Dis. 9(12), 1642; doi:10.3201/eid0912.020566 Zhang, H., Deng, X., Cui, B., Shao, Z., Zhao, X., Yang, Q., Song, S., Wang, Z., Wang, Y., Wang, Y., Liu, Z., Sheng, J. and Chen, C. 2020. Abortion and various associated risk factors in dairy cow and sheep in Ili, China. PLos One 15(10), e0232568; doi:10.1371/journal.pone.0232568 | ||
| How to Cite this Article |
| Pubmed Style Elaadli H, Badr Y, Ayad MAA, Mubarak AG, Younis W, Elmahallawy EK, Eissa SA, Shaapan RM. Assessment of seroprevalence and associated risk factors of Chlamydia abortus among sheep and women with a history of abortion in Northern Egypt. Open Vet. J.. 2025; 15(12): 6329-6341. doi:10.5455/OVJ.2025.v15.i12.16 Web Style Elaadli H, Badr Y, Ayad MAA, Mubarak AG, Younis W, Elmahallawy EK, Eissa SA, Shaapan RM. Assessment of seroprevalence and associated risk factors of Chlamydia abortus among sheep and women with a history of abortion in Northern Egypt. https://www.openveterinaryjournal.com/?mno=279662 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.16 AMA (American Medical Association) Style Elaadli H, Badr Y, Ayad MAA, Mubarak AG, Younis W, Elmahallawy EK, Eissa SA, Shaapan RM. Assessment of seroprevalence and associated risk factors of Chlamydia abortus among sheep and women with a history of abortion in Northern Egypt. Open Vet. J.. 2025; 15(12): 6329-6341. doi:10.5455/OVJ.2025.v15.i12.16 Vancouver/ICMJE Style Elaadli H, Badr Y, Ayad MAA, Mubarak AG, Younis W, Elmahallawy EK, Eissa SA, Shaapan RM. Assessment of seroprevalence and associated risk factors of Chlamydia abortus among sheep and women with a history of abortion in Northern Egypt. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6329-6341. doi:10.5455/OVJ.2025.v15.i12.16 Harvard Style Elaadli, H., Badr, . Y., Ayad, . M. A. A., Mubarak, . A. G., Younis, . W., Elmahallawy, . E. K., Eissa, . S. A. & Shaapan, . R. M. (2025) Assessment of seroprevalence and associated risk factors of Chlamydia abortus among sheep and women with a history of abortion in Northern Egypt. Open Vet. J., 15 (12), 6329-6341. doi:10.5455/OVJ.2025.v15.i12.16 Turabian Style Elaadli, Haitham, Yassien Badr, Magdy A. A. Ayad, Asmaa G. Mubarak, Waleed Younis, Ehab K. Elmahallawy, Samar A. Eissa, and Raafat M. Shaapan. 2025. Assessment of seroprevalence and associated risk factors of Chlamydia abortus among sheep and women with a history of abortion in Northern Egypt. Open Veterinary Journal, 15 (12), 6329-6341. doi:10.5455/OVJ.2025.v15.i12.16 Chicago Style Elaadli, Haitham, Yassien Badr, Magdy A. A. Ayad, Asmaa G. Mubarak, Waleed Younis, Ehab K. Elmahallawy, Samar A. Eissa, and Raafat M. Shaapan. "Assessment of seroprevalence and associated risk factors of Chlamydia abortus among sheep and women with a history of abortion in Northern Egypt." Open Veterinary Journal 15 (2025), 6329-6341. doi:10.5455/OVJ.2025.v15.i12.16 MLA (The Modern Language Association) Style Elaadli, Haitham, Yassien Badr, Magdy A. A. Ayad, Asmaa G. Mubarak, Waleed Younis, Ehab K. Elmahallawy, Samar A. Eissa, and Raafat M. Shaapan. "Assessment of seroprevalence and associated risk factors of Chlamydia abortus among sheep and women with a history of abortion in Northern Egypt." Open Veterinary Journal 15.12 (2025), 6329-6341. Print. doi:10.5455/OVJ.2025.v15.i12.16 APA (American Psychological Association) Style Elaadli, H., Badr, . Y., Ayad, . M. A. A., Mubarak, . A. G., Younis, . W., Elmahallawy, . E. K., Eissa, . S. A. & Shaapan, . R. M. (2025) Assessment of seroprevalence and associated risk factors of Chlamydia abortus among sheep and women with a history of abortion in Northern Egypt. Open Veterinary Journal, 15 (12), 6329-6341. doi:10.5455/OVJ.2025.v15.i12.16 |