| Research Article | ||
Open Vet. J.. 2026; 16(1): 214-230 Open Veterinary Journal, (2026), Vol. 16(1): 214-230 Research Article Comparative effects of syngeneic graft and xenograft blood for hemolytic anemia modeling in dogsFajar Shodiq Permata*, Herawati Herawati, Dyah Ayu Oktavianie Ardhiana Pratama, Ahmad Fauzi and Dhita Evi AryaniFaculty of Veterinary Medicine, Universitas Brawijaya, Malang, Indonesia *Corresponding Author: Fajar Shodiq Permata. Faculty of Veterinary Medicine, Universitas Brawijaya, Malang, Indonesia. Email: drh.fajar [at] ub.ac.id Submitted: 24/08/2025 Revised: 24/11/2025 Accepted: 07/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
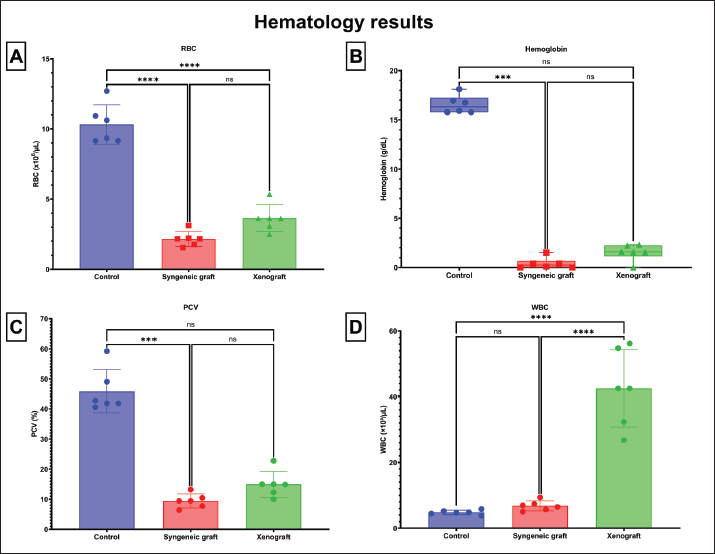
AbstractBackground: Hemolytic anemia (HA) involves premature red blood cell (RBC) destruction and often occurs in dogs as immune-mediated HA (IMHA) and hemolytic transfusion reactions (HTRs). Developing an animal model is crucial for studying its mechanisms. Syngeneic grafts or xenograft blood can be used experimentally to induce hemolytic and autoimmune responses through self-antigen mimicry. Aim: This study aimed to examine the comparative effects of syngeneic graft and xenograft blood on HA induction in mice. Methods: There were three groups (n=18 mice): a negative control, a syngeneic graft group, and a xenograft group. The syngeneic graft blood was obtained from mice of a different seller. Xenograft blood was obtained from a cat. Blood was injected intraperitoneally at 0.2 ml 5 times per week for 7 weeks. Mice were observed for 14 days after the last injection. Effects were evaluated based on complete blood count (RBC, WBC, Hb, and packed cell volume), blood chemistry [TPP, bilirubin, globulin, blood urea nitrogen (BUN), and creatinine], blood smear description, relative splenic CD4+ and CD8+ cells, and spleen, liver, and kidney histopathology. Results: The results showed that the xenograft blood group had higher (p < 0.05) WBC, bilirubin, and BUN levels, as well as a higher number of splenic CD4+ and CD8+ cells, than the syngeneic graft blood group. Additionally, it had lower albumin and creatinine levels (p < 0.05) than the syngeneic graft. Both syngeneic and xenograft blood induced tissue destruction in the spleen, liver, and kidney. Conclusion: The research concluded that syngeneic graft blood induced a HTRs model, and xenograft blood induced an IMHA-like model. Keywords: Blood, Hemolytic anemia, Tissue, Syngeneic graft, Xenograft. IntroductionHemolytic anemia (HA) is a pathological condition characterized by the premature destruction of red blood cells (RBCs), resulting in decreased oxygen-carrying capacity and compensatory erythropoiesis. HA can result from blood transfusion failure or immune-mediated mechanisms, depending on its cause. Dogs are particularly prone to hemolytic transfusion reactions (HTRs) and immune-mediated hemolytic anemia (IMHA), a severe hemolytic disorder in which antibodies target self-erythrocytes, triggering complement activation and hemolysis. The most extensive recent multicenter prospective study found that the incidence of acute hemolytic transfusion reactions (AHTRs) in dogs receiving packed erythrocytes (packed red blood cells) was 2.3% per transfusion event. AHTRs in dogs range from 0% to 6.3% (Steblaj et al., 2023; Hall et al., 2024). IMHA is one of the most common and life-threatening immune-mediated diseases in dogs, accounting for approximately 0.2%–0.5% of referral hospital admissions and showing mortality rates as high as 30%–70% despite therapy (Weinkle et al., 2005; Piek et al., 2008). Clinically, affected dogs exhibit HA, hyperbilirubinemia, and spherocytosis, with a high risk of thromboembolic complications that make management challenging even under aggressive immunosuppression (Swann and Skelly, 2013; Swann and Skelly, 2015). Unpredictable treatment outcomes, frequent relapses, and high fatality rates highlight the need for a better understanding of HA pathogenesis and the development of experimental models that reproduce its clinical and immunopathological features. Murine models offer valuable tools for investigating hemolytic-related disorders because of their genetic uniformity, short reproductive cycles, and well-characterized blood and tissue characteristics. Conventional HA models often rely on transgenic and drugs (Howie and Hudson, 2018; Dei Zotti et al., 2021). Although these models have deepened mechanistic insight, their complexity, cost, and technical requirements restrict their use, particularly in veterinary research settings. As a simpler, more accessible alternative, antigen mimicry approaches can be used to experimentally induce HA. In this method, the repeated administration of syngeneic graft or xenograft blood serves as an antigenic stimulus, eliciting immune recognition of erythrocyte surface antigens as "non-self." This mechanism leads to antibody production, complement activation, and subsequent hemolysis (Garden et al., 2019; Michalak et al., 2020; Loriamini et al., 2024). The choice of mimicry source may influence not only the onset and severity of anemia but also the breadth of immune responses and target-organ pathology—features that could be relevant to the condition of canine HA. In this study, we developed and compared HA induced by syngeneic graft and xenograft blood in murine models. Both approaches successfully reproduced anemic conditions but differed in terms of hematological progression and tissue pathology. By characterizing these differences, this study aims to identify which induction method best approximates the clinicopathological spectrum of canine HA, providing a more practical and translational model for future studies on disease mechanisms and therapeutic interventions. Materials and MethodsAnimal preparationA total of 21 male Balb/c mice (15–20 g, age 4–5 weeks) were obtained from the Animal Laboratory at the Islamic State University of Malang, Indonesia. The experimental mice were randomly allocated into three groups: control, syngeneic graft, and xenograft. Animals were housed in plastic cages (40 × 30 × 12 cm) with 3–4 mice per cage and provided with wood shavings as bedding, feed, and water ad libitum. Based on Federer’s formula, k(n-1)≤15, which is k for group number and n is the murine number per group; therefore, the minimum number of mice per group is 6, with one as a backup. This approach was justified for 3R (reduction, refinement, and replacement) and animal welfare considerations regarding ethical standards. Blood collection and injection of syngeneic graft and xenograftTen additional male Balb/c mice (15–20 g, age 4–5 weeks) obtained from a private breeding facility in Malang were used as syngeneic graft blood donors, while four adult domestic cats were used as xenograft blood donors. Syngeneic graft blood was collected from the retro-orbital vessels of anesthetized mice, yielding 0.5–1 ml per animal, into 1–2 pieces of 0.5 ml Ethylenediaminetetraacetic acid (EDTA)-coated microtubes (Vaculab, Indonesia) and stored at 4°C. Anesthesia was achieved by intraperitoneal injection of ketamine (Ket-A-100, Veterinary use, Agrovet Market, Peru) at 100 mg/kg body weight combined with xylazine (Xyla 2%, Veterinary use, Interchemie, Netherlands) at 50 mg/kg body weight. Xenograft blood was collected from the jugular vein of cats, around 1.5 ml into 1.5 ml of EDTA Vacutainer tubes (OneMed, Indonesia) and stored at 4°C. During blood collection, the veterinary nurse gently handled the cat in a conscious condition. The rationale for choosing cat blood as a xenograft donor is that the responsible protein for HA, glycophorin A (Brain et al., 2002), from the cat (Accession number XP_019684477.1) has a 47.54% identity to that of mice (Accession number AAA37709.1) based on protein blast. Thus, the blood cat would be immunogenic, inducing HA through antigen mimicry. Recipient mice received intraperitoneal injections of 0.2 ml of either syngeneic graft or xenograft blood five times per week for 7 consecutive weeks, followed up for 14 days after the last injection. This method was modified based on the work of Cox and Keast (1973) and Saxena and Chatterjee (2017). Our method used whole blood instead of erythrocyte isolation for induction because some reports of IMHA in humans also showed antibodies against leukocytes (Li et al., 2022), platelets (Fagiolo and E, 1976; Tassies and Reverter, 2010), and platelet phagocytosis (Basit Momin et al., 2022). Clinical status was monitored daily, and the experimental endpoint was defined as critical illness persistence for at least 8 hours. At the endpoint, mice were anesthetized with ketamine-xylazine and then euthanized by exsanguination via the retro-orbital vessels of the right and left eyes, about 0–3 to 0.5 ml per collection. Blood was also collected from the heart at approximately 0.5 to 0.8 ml. Blood samples were collected into one piece of 0.5 ml EDTA microtubes (Vaculab, Indonesia) for hematological analysis and two pieces of 0.5 ml microtubes (Vaculab, Indonesia) for biochemical analysis. Donor blood was examined by blood film to ensure that there were no blood parasites, as in healthy donors. Hematology examinationA simple blood clotting test was conducted by placing 0.2 ml of blood into a ceramic palette at room temperature for 30 minutes. Clotting and serum formation were observed. A fully automated complete blood count analyzer (Rayto, China) (Farooq et al., 2025) was used in the Veterinary Teaching Hospital of Universitas Brawijaya, Indonesia, to assess hematological parameters from the collected blood samples. The blood variables analyzed included RBCs, hemoglobin, packed cell volume (PCV), and white blood cells (WBCs). Examination of blood chemistryBlood chemistry was assessed using serum obtained from plain microtainer tubes. The serum was separated from the cellular pellet after centrifugation (Centrifugal machine F2C, China) at 3,000 rpm for 15 minutes at room temperature. The collected serum was stored at −20°C before biochemical testing. The biochemical panel included plasma total protein, albumin, globulin, total bilirubin, conjugated bilirubin, unconjugated bilirubin, blood urea nitrogen (BUN), and creatinine. All analyses were performed at the Bioscience Institute, Universitas Brawijaya, Jakarta, Indonesia. Plasma total protein, albumin, total bilirubin, conjugated bilirubin, and creatinine concentrations were measured spectrophotometrically (SmartSpec Plus UV/Vis, Bio-Rad, USA) according to the manufacturer’s instructions (ReiGed Diagnostics, Indonesia). BUN levels were determined using a dedicated assay kit (Evogen, Spain) according to the manufacturer’s protocol. Globulin concentrations were calculated by subtracting albumin from total protein (Hoo et al., 2021), whereas unconjugated bilirubin was obtained by subtracting conjugated bilirubin from total bilirubin (Verran et al., 2024). Similar biochemical approaches have been widely applied in murine models to evaluate the systemic effects of experimental treatments and disease models (Silva-Santana et al., 2020; Song et al., 2024; Wang and Deng, 2024). Tissue histopathological examinationMice were necropsied after euthanasia according to established protocols for small laboratory animals (Treuting and Snyder, 2015). Organs, such as the liver, kidney, and spleen, were collected and examined for gross anatomical pathology. Approximately 1 g of spleen tissue was placed in phosphate-buffered saline (PBS) for quantification of CD4+ and CD8+ lymphocytes, while the remainder of the liver, kidney, and spleen specimens were fixed in neutral buffered formalin with a tissue-to-fixative ratio of at least 1:10 for a minimum of 24 hours, ensuring optimal preservation (Fiette and Slaoui, 2011). Histological samples were processed using standard paraffin embedding, sectioned at 5 µm, and stained with hematoxylin and eosin (H&E). Histopathological evaluation was conducted using an Olympus CX21 microscope (Olympus, Japan) equipped with an Integrated Camera Optilab Advance Plus (Miconos, Indonesia) at 400× magnification (Permata et al., 2025; Marraskuranto et al., 2025). The sections were assessed for characteristic and significant pathological alterations between the experimental groups. Tissue processing was conducted at the Laboratory of Veterinary Histology, Faculty of Veterinary Medicine, Universitas Brawijaya, Indonesia. Splenic CD4+ and CD8+ cell countThe spleens were washed with sterile PBS 2–3 times and mechanically dissociated in PBS using the blunt end of a syringe. The tissue was passed through a wire mesh filter into a 15-ml polypropylene tube, rinsed with 5 ml PBS, and centrifuged at 2,500 rpm for 5 minutes at 4°C. The cell pellet was resuspended in 1 ml of PBS, homogenized, and transferred into Eppendorf tubes. For CD4+ T-cell staining, 70 µl of cell suspension was incubated with 50 µl of fluorescein isothiocyanate-conjugated anti-mouse CD4 monoclonal antibody (BD Biosciences, USA, cat. no 553047) for 15 minutes in the dark on ice. For CD8+ T-cell staining, a separate aliquot was incubated with 50 µl phycoerythrin-conjugated anti-mouse CD8 antibody (cat. no 553033, BD Biosciences, USA) under identical conditions. After incubation, the cells were washed, resuspended in 500 µl PBS, and analyzed using a BD FACSCalibur flow cytometer with BD CellQuest Pro software (BD Biosciences, USA). Relative CD4+ and CD8+ cell populations were determined by quadrant gating based on fluorescence intensity, with CD4+ cells in the bottom right quadrant and CD8+ cells in the upper left quadrant. The measurements were conducted at the Biology Department, Faculty of Mathematics and Natural Sciences, Universitas Brawijaya, Indonesia. This approach for splenocyte isolation and flow cytometric immunophenotyping has been widely applied in murine immunology studies, ensuring reproducible assessment of T-cell subsets (Mincham et al., 2021; Skordos et al., 2021) and confirmation of the immunological status of autoimmune disease (Wu et al., 2014). Data analysisQuantitative data were analyzed using one-way analysis of variance, followed by the Tukey test at the 95% significance level. Statistical analysis and data visualization were conducted using GraphPad Prism ver. 9.5.1. Qualitative data were analyzed and compared with other groups. Because the sample size of each group was limited due to ethical approval, the power of one-way analysis of variance and post hoc results (Tukey test) was determined using the G*power calculator (Faul et al., 2007). Ethical approvalAll experimental procedures were performed in accordance with ethical approval from the Ethics Committee, Faculty of Medicine, Universitas Brawijaya, Indonesia (Approval No. 202/EC/KEPK/07/2019). ResultsIn this study, we observed differences in HA development between the syngeneic and xenograft groups. Syngeneic graft induction produced milder hematological alterations, with no significant elevation in WBC count, whereas xenograft induction was associated with stronger immune responses and greater tissue damage. Thus, to convey our message related to comparative HA in a murine model, we presented the results in two parts: changes in blood parameters and splenic lymphocytes, and pathological changes in major organs. Blood parameters and splenic CD4+ and CD8+ cellsThe hematological and biochemical analyses revealed distinct alterations among the groups. RBC and hemoglobin concentrations were reduced in the syngeneic graft group without a significant increase in WBCs, reflecting HA without leukocyte involvement. In contrast, the xenograft group exhibited elevated WBC counts, indicating a stronger immune response. Both grafted groups showed a decrease in PCV compared with the controls (Fig. 1). However, an insignificantly higher PCV in the xenograft could be correlated with dehydration, as supported by rouleaux formation in its blood film (Fig. 6).
Fig. 1. Comparison of hematological parameters between the experimental groups. The figure shows the hematological profiles of mice in the control, syngeneic graft, and xenograft groups, including RBC count, WBC count, hemoglobin concentration (Hb), and PCV. Data are expressed as mean ± standard deviation (SD). The xenograft group showed a significant increase in WBC counts compared with the control and syngeneic graft groups (p < 0.05), indicating a stronger immune response. The syngeneic graft group showed lower RBC and Hb levels, which were not significantly different from the xenograft UB, with fewer WBC, consistent with HA occurrence that may not involve a leukocyte response. Significant differences between groups are indicated by p < 0.05. The calculated power for RBC, hemoglobin, PCV, and WBC is 90.25%, 74.68%, 78.82%, and 86.69%, respectively.
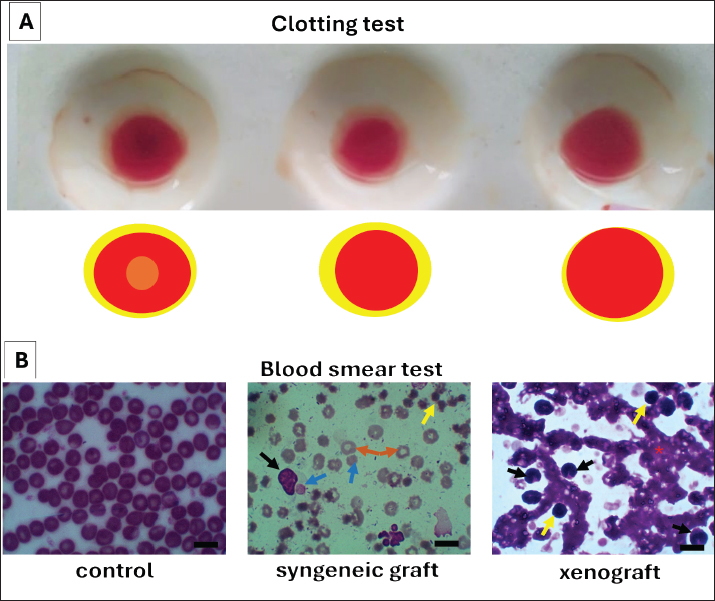
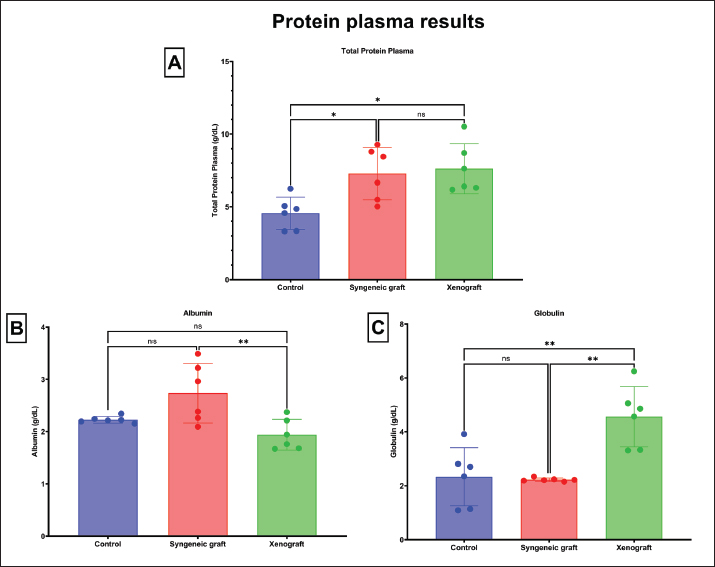
Fig. 6. Clotting test and blood smear findings of the experimental groups (A) Macroscopic clotting test of blood left at room temperature for 30 minutes. A visible central clot was formed in the control group, surrounded by a yellowish area interpreted as serum. In contrast, blood from the syngeneic graft and xenograft groups failed to form a clot and remained in a liquid state. (B) Representative blood smear images. Left: control group with normal erythrocyte morphology. Middle: Syngeneic graft group displaying numerous lymphocytes (yellow arrow), spherocytes (blue arrow), polychromatic erythrocytes with central pallor (orange arrow, indicative of hypochromic erythrocytes), and monocytes (black arrow) exhibiting phagocytosis of spherocytes. Right: xenograft group demonstrating rouleaux formation (red asterisk) of erythrocytes, with abundant monocytes (black arrow) and lymphocytes (yellow arrow). Giemsa staining 1,000× magnification. Scale bars=20 μm. Plasma protein analysis revealed reduced albumin and total plasma protein in the syngeneic graft group, whereas the xenograft group showed elevated globulin and markedly lower albumin, suggesting enhanced immunoglobulin production with impaired protein balance (Fig. 2).
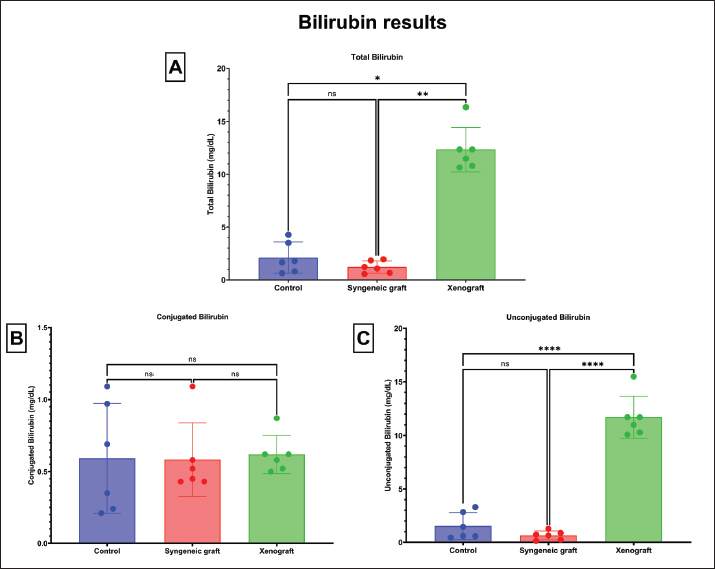
Fig. 2. Comparison of plasma protein parameters between the experimental groups The figure presents the plasma biochemistry results of mice in the control, syngeneic graft, and xenograft groups, including total plasma protein (TPP), albumin, and globulin levels. Data are presented as mean ± standard deviation (SD). The xenograft group exhibited significantly higher globulin levels than the control and syngeneic graft groups (p < 0.05), suggesting enhanced immunoglobulin production. In contrast, albumin concentrations were markedly lower in the xenograft group, indicating impaired protein balance. The total plasma protein levels were significantly altered in both grafted groups compared with the control, reflecting the systemic effects of HA induction. Significant differences between groups are indicated by p < 0.05. The calculated power for TPP is 35.36%, albumin is 36.43%, and globulin is 57.47% Bilirubin assessment confirmed HA, with syngeneic groups exhibiting lower total bilirubin (1.24 ± 0.58 mg/dl) and unconjugated bilirubin (0.65 ± 0.42 mg/dl) levels than the control group (insignificant) and the xenograft group (significantly different, p < 0.05). The conjugated bilirubin score (0.5–1.1 mg/dl) among groups highlighted insignificant differences (p ≥ 0.05), indicating the limitations of liver capacity to conjugate free bilirubin. Thus, in the syngeneic graft group, conjugated bilirubin could be transferred into the stool to regulate bilirubin homeostasis (Fig. 7). In contrast, the xenograft group exhibited the highest levels of total bilirubin (12.33 ± 2.10 mg/dl) and unconjugated bilirubin (11.71 ± 1.97 mg/dl), indicating more severe hemolysis and greater hepatic capacity loss than the syngeneic graft group. This loss is related to pathological injury in the xenograft group’s liver tissue, which impairs the clearance of conjugated bilirubin into the intestine and stool.
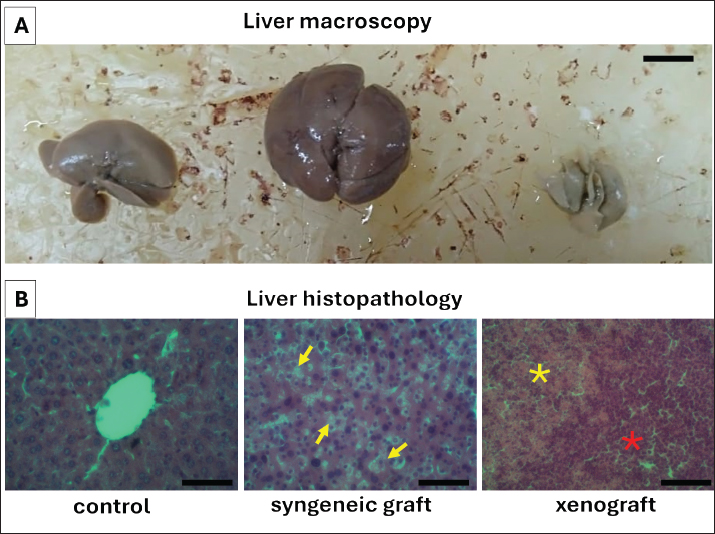
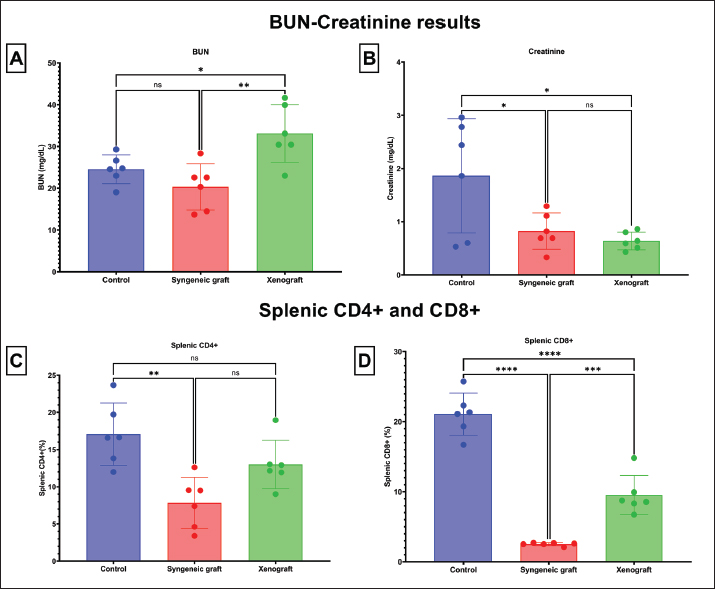
Fig. 7. Macroscopic pathology and histopathological changes of the liver in the experimental groups. (A) Representative macroscopic appearance of the liver in the control, syngeneic graft, and xenograft groups, showing apparent alterations in size and color morphology in the treated groups compared with the control. (B) Histopathological examination of the liver with H&E staining at 400× magnification. Left: Control group with normal hepatic architecture and hepatocyte morphology. Middle: Hepatocellular degeneration characterized by nuclear pyknosis and hepatocyte atrophy in the syngeneic graft group (yellow arrows). Right: The xenograft group showed severe inflammatory infiltrates (red asterisk) accompanied by exudative changes (yellow asterisk), consistent with extensive hepatic injury. A. Scale bars=10 mm, B. Scale bars=100 μm. Renal function markers indicated increased BUN in the xenograft group, whereas creatinine was higher in the syngeneic graft group. In addition, both syngeneic graft and xenograft groups showed significant reductions in splenic CD4+ and CD8+ lymphocytes, consistent with splenic injury and lymphocyte redistribution (Fig. 4). The lower percentage of CD4+ and CD8+ lymphocytes in the syngeneic group than in the xenograft group (Fig. 5) could indicate that HA involved fewer leukocytes or delayed the immune response due to syngeneic blood antigens.
Fig. 4. Comparison of renal function markers and splenic lymphocyte counts between the experimental groups BUN, creatinine levels, and splenic CD4+ and CD8+ lymphocyte counts in the control, syngeneic graft, and xenograft groups are presented. Data are expressed as mean ± SD. The xenograft group exhibited significantly elevated BUN compared with the other groups (p < 0.05), indicating renal stress, whereas the syngeneic graft group had lower creatinine levels. Both syngeneic graft and xenograft groups exhibited a significant reduction in splenic CD4+ and CD8+ lymphocytes compared with controls (p < 0.05), consistent with splenic tissue damage and lymphocyte migration into the peripheral blood. Significant differences between groups are indicated by p < 0.05. The calculated power for BUN is 42.99%, creatinine is 34.19%, splenic CD4+ is 47.43%, and splenic CD8+ is 89.65%. The clotting test demonstrated that only the control group formed a stable clot, whereas both grafted groups failed to clot (Fig. 6A). Blood smear examination revealed spherocytes, hypochromic erythrocytes, and monocytes phagocytosing spherocytes in the syngeneic graft group, whereas rouleaux formation and abundant inflammatory cells were evident in the xenograft group (Fig. 6B). This formation also indicates dehydration in the xenograft group and correlates with a slightly higher PCV score (Fig. 1). Changes in the tissue pathologyMacroscopic and histopathological evaluation revealed organ-specific alterations. In the liver, the syngeneic graft group exhibited hepatocellular degeneration with nuclear pyknosis and atrophy, whereas the xenograft group exhibited severe inflammatory infiltrates and exudation (Fig. 7). The more severe histopathology of the liver in the xenograft group correlates with an incapable liver of conjugation of unconjugated bilirubin; thus, the xenograft group had a higher level of unconjugated bilirubin in the blood than the syngeneic group (Fig. 3).
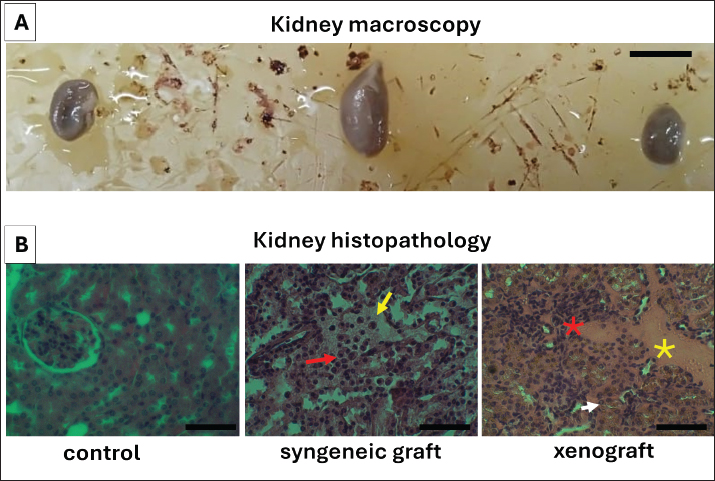
Fig. 3. Bilirubin levels among the experimental groups. The figure illustrates serum bilirubin concentrations —total, conjugated, and unconjugated —across the control, syngeneic graft, and xenograft groups. Data are expressed as mean ± SD. Both the syngeneic graft and xenograft groups exhibited significant elevations in total bilirubin (approximately) and unconjugated bilirubin compared with the control (p < 0.05), consistent with the diagnosis of HA. The xenograft group exhibited the highest bilirubin levels, particularly unconjugated bilirubin, indicating more pronounced hemolysis and a greater burden of hepatic processing. Significant differences between groups are indicated by p < 0.05. The calculated power for total bilirubin is 59.94%, conjugated bilirubin is 5.18%, and unconjugated bilirubin is 90.68%. The syngeneic graft group demonstrated interstitial edema and inflammatory cell infiltration in the kidney, whereas the xenograft group presented with more severe damage, including liquefactive necrosis, eosinophilic exudates, and hemosiderin accumulation in tubular epithelial cells (Fig. 8).
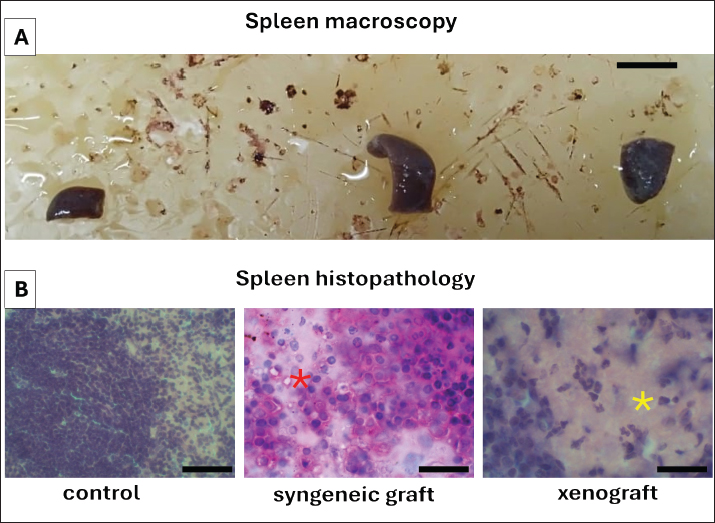
Fig. 8. Macroscopic pathology and histopathological changes of the kidney in the experimental groups. (A) Representative macroscopic appearance of kidneys in the control, syngeneic graft, and xenograft groups, showing differences in size and morphology compared with the controls.(B) Histopathological examination of the kidney with H&E staining at 400× magnification. In the spleen, coagulative necrosis resembling infarction was observed in the syngeneic graft group, whereas liquefactive necrosis with extensive tissue destruction was observed in the xenograft group (Fig. 9). The histopathological features of the spleen in both groups supported the observed decrease in CD4+ and CD8+ lymphocytes (Fig. 5). However, the lymphocytes remaining in the spleen could contribute to the bloodstream.
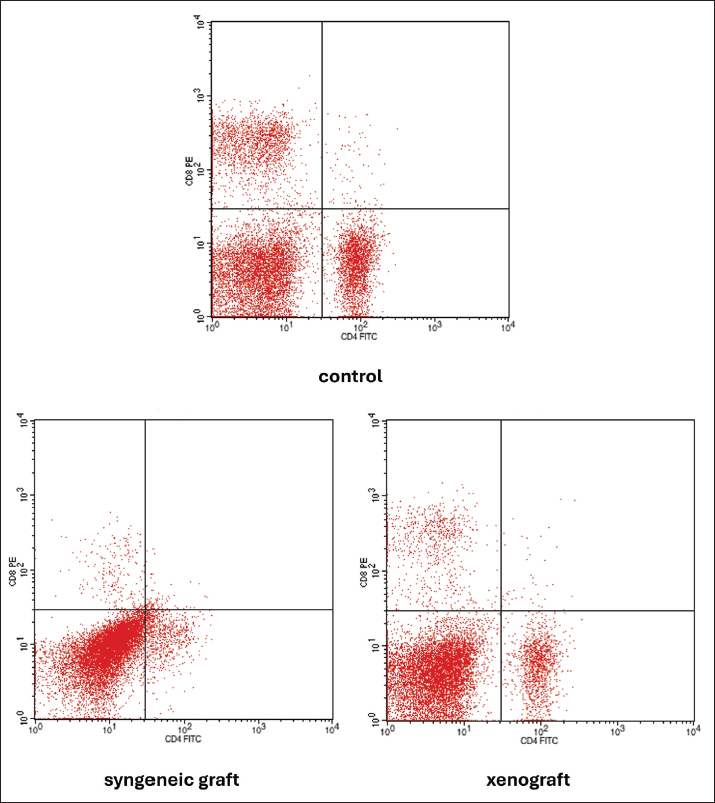
Fig. 5. Flow cytometric analysis of splenic CD4+ and CD8+ T lymphocytes in the control, syngeneic graft, and xenograft groups Dotplots show that syngeneic grafts and xenografts have lower CD8+ (upper left) and CD4+ (bottom right) lymphocytes than the control. However, the xenograft group had more dot plots in CD4+ and CD8+ lymphocytes than the syngeneic graft group.
Fig. 9. Macroscopic pathology and histopathological changes in the spleen of the experimental groups. Representative macroscopic appearance of the spleen in the control, syngeneic graft, and xenograft groups, showing differences in size between the treated and control groups. B. Histopathological examination of the spleen with H&E staining at 400× magnification. Left: Control group with normal splenic architecture and preserved lymphoid follicles. Middle: Syngeneic graft group demonstrating coagulative necrosis resembling infarction (red asterisk). Right: xenograft group exhibiting liquefactive necrosis characterized by a liquefied necrotic mass (yellow asterisk), indicative of severe splenic tissue destruction. A. Scale bars=10 mm, B. Scale bars=100 μm. These findings are summarized in Table 1, which highlights the comparative hematological, biochemical, macroscopic, and histopathological changes observed among the control, syngeneic graft, and xenograft groups. Table 1. Hematological, biochemical, and tissue changes in the control, syngeneic graft, and xenograft groups.
DiscussionIn this study, we compared the effects of syngeneic graft and xenograft blood induction on HA development in mice and observed that the two models produced distinct disease patterns. We found that syngeneic graft blood caused hemolysis, and fewer leukocytes were involved, likely because the blood donor was less immunogenic. In contrast, xenograft blood induced a stronger immune response, as reflected in increased WBC counts, elevated globulin levels, higher splenic CD8+, and more severe tissue pathology. These differences highlight how the source of antigenic blood shapes both the kinetics and severity of human leukocyte antigen (HLA), making it a valuable animal model. Based on power calculations for our statistical results, the findings for hematology profile, unconjugated bilirubin, and splenic CD8+ showed high power (>70%), whereas the other variables showed lower power (< 70%). However, the globulin variable had moderate power (>50%). The results observed in the syngeneic graft group, showing minimal leukocyte involvement in hemolysis, are consistent with those of Nazimek et al. (2019), who reported similar mechanisms associated with delayed-type hypersensitivity. The mice do not have a specific blood type; however, they possess immunogenic antigens, such as Hbb variants (Hbb single or Hbb diffuse) (Randelia and Naik, 1979). We predicted that the mechanism of syngeneic blood-induced HA involves type 2 hypersensitivity type 2 after repeated blood administration. We assumed that the antibody from a blood donor bound to the recipient erythrocyte, initiating complement activation without elevating globulin levels. These findings are associated with delayed HTRs, even in patients with lower IgM levels (Salama and Mueller-Eckhardt, 1984). Thus, our syngeneic blood model could serve as a transfusion model to induce HA, leading to HTRs. HTRs are characterized by cytokine storms that actively activate complement production to eliminate the mimicked antigen (Hod et al., 2008). Interestingly, the delayed HTRs induce HA associated with sickle cell disease (Anwar S Prakash et al., 2025). Blood transfusion in dogs also carries a risk of causing HA (Holm et al., 2025). Although many reports have indicated that HA is due to HTRs or type 2 hypersensitivity (Adjei et al., 2024), we acknowledge the limitations of our endpoint (14 days after the last intraperitoneal blood administration), during which immune responses to syngeneic blood may occur after 120 hours (Nazimek et al., 2019). However, our study strengthens the evidence for the systemic effects of repeated syngeneic blood transfusions on tissue injury profiles (liver, kidney, and spleen). Notably, the liver of this group sustained less injury than that of the xenograft blood group. Multiorgan injury caused by complement-HA has also been reported by Peliconi et al. (2024). Inappropriate blood storage increases the antigenicity of the blood donor, thereby inducing HTRs (Patterson et al., 2011). On the other hand, xenograft blood showed HA-induced leukocytosis, as evidenced by higher WBC count, globulin level, and splenic CD8+ cell count compared with the syngeneic group. In murine transfusion models, antibody formation accelerates the destruction of donor xenograft RBCs (Saxena and Chatterjee, 2017; Michalak et al., 2020). Interestingly, this pattern of hemolysis parallels IMHA in dogs, in which decreases in RBCs and hemoglobin are often the first diagnostic indicators, and hyperbilirubinemia is associated with poorer outcomes and higher mortality (Duclos et al., 2024). However, our xenograft blood-induced HA model suffered dehydration, as evidenced by reduced rouleaux formation and a non-significantly higher PCV than that of the syngeneic group. According to Duclos et al. (2024), dehydration associated with canine IMHA is correlated with vomiting (7%) and diarrhea (21%). These alterations resemble more severe IMHA cases, in which systemic immune dynamics, such as CD8+, are evident. Although CD8+ T cells are cytotoxic lymphocytes that target intracellular microorganisms within infected cells, recent studies in human AIHA and related autoimmune cytopenias indicate that CD8+ T cell dysregulation, including increased clonal expansions, plays a role in disease pathogenesis. These T cells may contribute to the immune-mediated destruction of RBCs, either directly or by influencing other immune cells (Smirnova et al., 2016; Michalak et al., 2020). However, direct evidence in dogs is limited, and most veterinary research focuses on hematological markers rather than specific T cell subsets (Duclos et al., 2025). Therefore, the pronounced rise in globulin and bilirubin in our xenograft model suggests not only persistent hemolysis but also an adaptive and humoral response, which has been highlighted as a driver of complex pathophysiology in IMHA. A report of canine IMHA in the UK and Ireland from 222 dogs showed that 11.25% of the dogs had hyperglobulinemia and 49% had hyperbilirubinemia (Morrison et al., 2025). The reduction in splenic CD4+ and CD8+ lymphocytes observed in both experimental groups underscores the pivotal role of the spleen in HA. Beyond its function in erythrocyte clearance and T-cell activation, hemolysis induces splenic immune remodeling: heme stress impairs dendritic cell differentiation and compromises antigen presentation, leading to further CD4+ T-cell dysfunction (Richards et al., 2021; Vallelian et al., 2022). The AIHA consensus guidelines further emphasize the spleen as a diagnostic and pathological hub, with erythrophagocytosis being a key feature in canine disease (Kim et al., 2020). Clinically, splenectomy has been shown to induce remission or improve outcomes in dogs with IMHA by removing the primary site of hemolysis and immune reaction (Bestwick et al., 2022). Splenic pathology, including splenomegaly, lymphoid depletion, and histiocytic hyperplasia, is frequently reported in dogs with IMHA, which reflects both erythrophagocytosis and immune response dynamics (Tucker et al., 2023). The more profound depletion of splenic lymphocytes in xenograft animals suggests a more substantial immunological challenge, consistent with the mechanisms of xenograft rejection. Multi-organ injuries were also observed in the xenograft group, with more severe histopathological features in the liver, kidney, and spleen, including inflammation and liquefaction as exudative necrosis. These results are consistent with transcriptomic analysis, which showed that HLA induces organ damage and inflammation (Peliconi et al., 2024). Hemolysis burdens hepatic function and can cause decreased hepatic bilirubin uptake, hypoxic injury, or even thromboembolic events, leading to hepatic necrosis (Bianco et al., 2021). Thus, the toxic effects of free bilirubin on renal tissue damage the kidney (Barton et al., 2025; Lantzaki et al., 2025). Therefore, it increased the BUN level. In summary, our murine model of HA demonstrated two distinct pathological patterns: syngeneic graft blood-induced HTRs and xenograft blood-induced stronger immune activation, resulting in an IMHA-like model. These outcomes parallel the clinical presentation of canine HA, in which some patients present with red cell destruction and others with more protracted courses complicated by systemic inflammation and organ injury. Therefore, the model provides a translational bridge to better understand the mechanisms underlying canine HA from two perspectives. Nevertheless, this study has limitations. We did not directly quantify syn- or xenoreactive antibodies, complement activity, or cytokine responses, all of which are critical for thoroughly dissecting the immunological mechanisms. We only provided splenic CD4+ and CD8+ comparisons from each perspective. We also acknowledge that EDTA-preserved whole blood was used instead of purified erythrocytes alone, which may have elicited immune responses against red blood cells and other blood cell components. However, some reports have shown that autoimmune HA in humans has antibodies against leukocytes and platelets (Fagiolo and E, 1976; Li et al., 2022; Amit et al., 2024), which is called Evans syndrome, and there is limited information on canine IMHA. Given the limited sample sizes per group that complied with ethical standards, we calculated G power for our statistical results to better inform our models. The control groups could use naïve status as a placebo group with PBS treatment, and the model could be further compared across different sexes. Furthermore, the relatively short observation window precluded the differentiation between acute and chronic disease processes. Future studies should expand immunophenotyping, include autoantibodies against RBC, WBC, or platelets, examine cytokine profiles, and extend longitudinal observations. Such refinements will strengthen the applicability of this model not only for investigating the pathogenesis of HTRs and IMHA in dogs but also for evaluating targeted therapeutic interventions and predicting clinical outcomes. ConclusionThis study demonstrates that syngeneic grafts and xenografts induce HA in mice, which follows distinct pathological trajectories. Syngeneic graft blood represented HTRs, and xenograft blood represented an IMHA-like model, even though xenograft blood induction highlighted more severe HA and its systemic effects than syngeneic graft blood. The respective model could be a simpler model for dogs with HA disorders, with further studies on the pathomechanism and treatment. AcknowledgmentWe would like to thank the Animal Laboratory at Islamic State University Malang and the Molecular Biology Department for their support of our animal facility and laboratory. Additionally, we thank the Biology Department, Faculty of Mathematics and Natural Sciences, Universitas Brawijaya, for providing the flow cytometer facility, the Veterinary Teaching Hospital, Universitas Brawijaya for the automatic hematology analyzer facility, the Institut Biosains Universitas Brawijaya for the blood chemistry examination facility, and the Laboratory of Veterinary Histology, Faculty of Veterinary Medicine, Universitas Brawijaya, for providing the tissue processing facility. We also acknowledge Dr. Mark Duncan for his assistance and support in collecting the blood samples of the cats. Conflict of interestThe authors declare that they have no known competing financial interests or personal relationships that could have influenced the work reported in this paper. FundingThis research was funded by the Faculty of Veterinary Medicine, Universitas Brawijaya, with the DPP SPP scheme in 2019. Author's contributionFSP: Conceptualization, Methodology, Investigation, Data Curation, Formal Analysis, Original Draft Writing. HRW: Methodology, Validation, Resources, Supervision, Writing, Review, and Editing. DAO: Histological/Immunological analysis, data interpretation, writing, review, and editing. AF: Laboratory work, data collection, visualization, writing, review, and editing. DEA: Methodology, Validation, Resources, Supervision, Writing, Review, and Editing. Data availabilityThe data supporting the findings of this study are available from the corresponding author upon reasonable request. All relevant data generated or analyzed during this study have been included in this article. ReferencesAdjei, S.K., Lartey, E.T., Mensah, H.A. and Bryony Brookman-Eshun. 2024. Acute transfusion reactions in a district hospital: a 2-year retrospective review. Int. J. Sci. Res. Arch. 11(1), 2587–2594; doi:10.30574/ijsra.2024.11.1.0328 Amit , K., Ayush, M., Upendra, K., Abhishek, K., Abhishek, A. and Mithul, V. 2024. Evans syndrome: a rare case of concurrent autoimmune hemolytic anemia and immune thrombocytopenia. HIJ 8(1), 00253; doi: 10.23880/hij-16000253 Anwar, T., Prakash, S., Sahu, A., Mukherjee, S. and Mishra, D. 2025. Alloimmunization and consequential delayed hemolytic transfusion reactions in patients with sickle cell disease: a case series. Asian J. Transfusion Sci. 19, 167–172; doi:10.4103/ajts.ajts_191_23 Barton, J.C., German, A.J. and O'Connell, E.M. 2025. Prevalence of proteinuria in dogs with immune-mediated disease. J. Vet. Intern. Med. 39, e70162; doi:10.1111/jvim.70162 Basit Momin, M.A., Aluri, A. and Bhaskar, P.M.B. 2022. Donath-landsteiner hemolytic anemia with erythrophagocytosis and platelet phagocytosis in peripheral smear: a rare case report. Med. J. Dr. DY. Patil. Vidyapeeth. 15, 601–604; doi:10.4103/mjdrdypu.mjdrdypu_373_20 Bestwick, J.P., Skelly, B.J., Swann, J.W., Glanemann, B., Bexfield, N., Gkoka, Z., Walker, D.J., Silvestrini, P., Adamantos, S., Seth, M. and Warland, J. 2022. Splenectomy in the management of primary immune-mediated hemolytic anemia and primary immune-mediated thrombocytopenia in dogs. Vet. Internal. Med. 36, 1267–1280; doi:10.1111/jvim.16469 Bianco, C., Coluccio, E., Prati, D. and Valenti, L. 2021. Diagnosis and management of autoimmune hemolytic anemia in patients with liver and bowel disorders. J. Clin. Med. 10(3), 423; doi: 10.3390/jcm10030423 Brain, M.C., Prevost, J.M., Pihl, C.E. and Brown, C.B. 2002. Glycophorin A-mediated hemolysis of normal human erythrocytes: evidence for antigen aggregation in the pathogenesis of immune hemolysis. Br. J. Haematol. 118, 899–908; doi:10.1046/j.1365-2141.2002.03657.x Cox, K.O. and Keast, D. 1973. Erythrocyte autoantibodies induced in rat erythrocyte-immunized mice. Immunology 25, 531–539. Dei Zotti, F., Qiu, A., La Carpia, F., Moriconi, C. and Hudson, K.E. 2021. A new murine model of primary autoimmune hemolytic anemia. Front. Immunol. 12, 752330; doi:10.3389/fimmu.2021.752330 Duclos, A.A., Bailén, E.L., Barr, K., Le Boedec, K. and Cuq, B. 2024. Clinical presentation, outcome, and prognostic factors in dogs with immune-mediated hemolytic anemia: a single-center retrospective study of 104 cases in Ireland (2002–2020). Ir. Vet. J. 77, 16; doi:10.1186/s13620-024-00277-w Duclos, A.A., O’Sullivan, L., McPhedran, C., Hocker, S., Le Boedec, K., Blois, S. and Cuq, B. 2025. Retrospective evaluation of hematological ratios in dogs with nonassociative immune-mediated hemolytic anemia: 206 cases. Vet. Internal Med. 39, e70101; doi:10.1111/jvim.70101 Fagiolo, E. 1976. Platelet and leukocyte antibodies in patients with autoimmune hemolytic anemia. Acta Haematol. 56, 97–106; doi:10.1159/000207925 Farooq, U., Lashari, M.H., Rehman, Z.U., Idris, M., Rashid, H., Nasreen, S., Laraib, F., Ameer, R., Chauhdary, M. and Fatima, I. 2025. Performance evaluation of Rayto RT-7600Vet hematology analyzer in side-by-side comparison with manual hematological methods for apparently healthy Cholistani cattle blood. PLos One 20(3), e0302617; doi:10.1371/journal.pone.0302617 Faul, F., Erdfelder, E., Lang, A., -G.. and Buchner, A. 2007. G*Power 3: a flexible statistical power analysis program for social, behavioral, and biomedical sciences. Behav. Res. Methods 39, 175–191; doi:10.3758/BF03193146 Fiette, L. and Slaoui, M. 2011. Necropsy and sampling procedures in rodents. In Drug safety evaluation, methods in molecular biology. Eds., Gautier, C. Totowa, NJ: Humana Press, pp: 39–67. doi: 10.1007/978-1-60761-849-2_3 Garden, O.A., Kidd, L., Mexas, A.M., Chang, Y., Jeffery, U., Blois, S.L., Fogle, J.E., MacNeill, A.L., Lubas, G., Birkenheuer, A., Buoncompagni, S., Dandrieux, J.R.S., Di Loria, A., Fellman, C.L., Glanemann, B., Goggs, R., Granick, J.L., LeVine, D.N., Sharp, C.R., Smith., Carr, S., Swann, J.W. and Szladovits, B. 2019. ACVIM consensus statement on the diagnosis of immune-mediated hemolytic anemia in dogs and cats. Vet. Internal. Med. 33, 313–334; doi:10.1111/jvim.15441 Hall, G.B.F., Birkbeck, R., Brainard, B.M., Camacho, F., Davidow, E.B., LeVine, D.N., Mackin, A., Moss, T., Nash, K.J., Stanzani, G., Starybrat, D., Stoye, D.Q., Tai, C., Thomason, J., Walker, J.M., Wardrop, K.J., Wilson, H., Wurlod, V.A. and Humm, K. 2024. A prospective multicenter observational study assessing the incidence and risk factors of acute blood transfusion reactions in dogs. Vet. Internal Med. 38, 2495–2506; doi:10.1111/jvim.17175 Hod, E.A., Zimring, J.C. and Spitalnik, S.L. 2008. Lessons learned from mouse models of hemolytic transfusion reactions. Curr. Opinion. Hematol. 15, 601–605; doi:10.1097/MOH.0b013e328311f40a Holm, N.G., Nielsen, L.N. and Langhorn, R. 2025. Retrospective evaluation of blood product transfusion outcomes and risk factors for transfusion reactions in dogs at a veterinary teaching hospital with an established blood bank: 137 dogs (2018–2022). J. Vet. Emergen. Crit. Care. 70034, 70034; doi:10.1111/vec.70034 Hoo, T., Lim, E.M., John, M., D’Orsogna, L. and McLean-Tooke, A. 2021. Calculated globulin levels as a screening tool for hypogammaglobulinemia or paraproteins in hospitalized patients. Ann. Clin. Biochem. 58, 236–243; doi:10.1177/0004563221989737 Howie, H.L. and Hudson, K.E. 2018. Murine models of autoimmune hemolytic anemia. Curr. Opinion Hematol. 25, 473–481; doi:10.1097/MOH.0000000000000459 Kim, S.M., Kim, G.N., Jeong, S.W. and Kim, J.H. 2020. Multiple splenic infarctions in a dog with immune-mediated hemolytic anemia: therapeutic implications. Iran. J. Vet. Res. 21, 65–69. Lantzaki, V., Fulton, E.A., Mclaughlin, M., Bennet, E.D., Conway, E.A. and Ridyard, A.E. 2025. Urine neutrophil gelatinase-associated lipocalin in non-associative immune mediated hemolytic anemia: a prospective controlled study in 22 dogs. J. Vet. Intern. Med. 39, e70002; doi:10.1111/jvim.70002 Li, P., Chen, Z., Wang, R., Shao, C., Liu, Y., Ju, Q., Zong, P. and Wang, H. 2022. Pancytopenia in a newborn due to maternal piperacillin and HLA antibodies. Transfusion Apheresis Sci. 61, 103324; doi:10.1016/j.transci.2021.103324 Loriamini, M., Cserti-Gazdewich, C. and Branch, D.R. 2024. Autoimmune hemolytic anemias: classifications, pathophysiology, diagnoses and management. IJMS 25, 4296; doi:10.3390/ijms25084296 Marraskuranto, E., Permata, F.S., Chasanah, E., Martosuyono, P. and Ariyani, F. 2025. Immunostimulatory activity of clarias catfish protein hydrolysate without pathological effects. J. Pharm. Innov. 20, 149; doi:10.1007/s12247-025-10049-w Michalak, S.S., Olewicz-Gawlik, A., Rupa-Matysek, J., Wolny-Rokicka, E., Nowakowska, E. and Gil, L. 2020. Autoimmune hemolytic anemia: current knowledge and perspectives. Immunoaging 17, 38; doi:10.1186/s12979-020-00208-7 Mincham, K.T., Young, J.D. and Strickland, D.H. 2021. OMIP 076: high-dimensional immunophenotyping of murine T-cell, B-cell, and antibody secreting cell subsets. Cytometry. Part A J. Int. Soc. Anal. Cytol. 99(9), 888–892; doi: 10.1002/cyto.a.24474 Morrison, T., Oikonomidis, I.L., Walker, H.K., Santos, N.S., Conway, S.A., Cuq, B., Albuquerque, C.S.C., Gil., Morales, C. and Woods, G. 2025. Multicenter, retrospective determination of the clinical utility of screening tests in dogs with immune-mediated hemolytic anemia in the United Kingdom and Ireland. Vet. Internal Med. 39, e70226. Nazimek, K., Bustos-Morán, E., Blas-Rus, N., Nowak, B., Ptak, W., Askenase, P.W., Sánchez-Madrid, F. and Bryniarski, K. 2019. Syngeneic red blood cell–induced extracellular vesicles suppress delayed-type hypersensitivity to self-antigens in mice. Clin. Exp. Allergy 49, 1487–1499; doi:10.1111/cea.13475 Patterson, J., Rousseau, A., Kessler, R.J. and Giger, U. 2011. In Vitro lysis and acute transfusion reactions with hemolysis caused by inappropriate storage of canine red blood cell products. Vet. Internal. Med. 25, 927–933; doi:10.1111/j.1939-1676.2011.0737.x Peliconi, J., Lavergne, J., Brochier, C., Guinot, F., David, T., Roumenina, L.T. and Grunenwald, A. 2024. Transcriptomic analysis shows a common multi-organ inflammatory/complement signature in patients with hemolysis-induced damage. Blood 144, 2644; doi:10.1182/blood-2024-201529 Permata, F.S., Amri, I.A., Nurmaningdyah, A.A., Mahardika, F., Budiarto, S., Vera, A.K.E. and Yulian, B.D. 2025. Antigen removal enhances host tolerance in sheep-derived xenogeneic myocardial scaffold: modulating acute and chronic inflammatory responses in mice. Regen. Eng. Transl. Med; doi:10.1007/s40883-025-00417-w Available via https://link.springer.com/article/10.1007/s40883-025-00417-w Piek, C.J., Junius, G., Dekker, A., Schrauwen, E., Slappendel, R.J. and Teske, E. 2008. Idiopathic immune-mediated hemolytic anemia: treatment outcome and prognostic factors in 149 dogs. Vet. Internal. Med. 22, 366–373; doi:10.1111/j.1939-1676.2008.0060.x Randelia, H.P. and Naik, S.N. 1979. HBV variants in mice. Lab. Anim. 13, 11–13; doi:10.1258/002367779781071285 Richards, A.L., Qiu, A., Zotti, F.D., Sheldon, K., Usaneerungrueng, C., Gruber, D.R. and Hudson, K.E. 2021. Autoantigen presentation by splenic dendritic cells is required for RBC -specific autoimmunity. Transfusion 61, 225–235; doi:10.1111/trf.16191 Salama, A. and Mueller-Eckhardt,, C. 1984. Delayed hemolytic transfusion reactions complement activation involving allogeneic and autologous red cells. Transfusion 24, 188–193; doi:10.1046/j.1537-2995.1984.24384225018.x Saxena, R.K. and Chatterjee, S. 2017. Binding of autoantibodies and apoptotic response in erythroid cells in the mouse model of autoimmune hemolytic anemia. HTIJ 5(2), 211–217; doi: 10.15406/htij.2017.05.00115 Silva-Santana, G., Bax, J.C., Fernandes, D.C.S., Bacellar, D.T.L., Hooper, C., Dias, A.A.S.O., Silva, C.B., De Souza, A.M., Ramos, S., Santos, R.A., Pinto, T.R., Ramão, M.A. and Mattos-Guaraldi, A.L. 2020. Clinical hematological and biochemical parameters in Swiss, BALB/c, C57BL/6, and B6D2F1 musculus. Anim. Model Exp. Med. 3, 304–315; doi:10.1002/ame2.12139 Skordos, I., Demeyer, A. and Beyaert, R. 2021. Analysis of T cells in mouse lymphoid tissue and blood with flow cytometry. STAR Protocols 2(1), 100351; doi: 10.1016/j.xpro.2021.100351 Smirnova, S.J., Sidorova, J.V., Tsvetaeva, N.V., Nikulina, O.F., Biderman, B.V., Nikulina, E.E., Kulikov, S.M. and Sudarikov, A.B. 2016. CD8+ cell expansion in autoimmune hemolytic anemia. Autoimmunity 49, 147–154; doi:10.3109/08916934.2016.1138219 Song, Y., Li, J. and Wu, Y. 2024. Understanding autoimmune mechanisms and new therapeutic strategies for autoimmune disorders. Sig. Transduct. Target. Ther. 9, 263; doi:10.1038/s41392-024-01952-8 Steblaj, B., Galli, J., Torgerson, P. and Kutter, A. 2023. Evaluation of leukocyte depletion of packed red blood cell units and impact on clinically observed transfusion reactions. Front. Vet. Sci. 10, 1217575; doi:10.3389/fvets.2023.1217575 Swann, J.W. and Skelly, B.J. 2013. Systematic review of evidence relating to the treatment of immune-mediated hemolytic anemia in dogs. Vet. Internal. Med. 27(1), 1–9; doi:10.1111/jvim.12028 Swann, J.W. and Skelly, B.J. 2015. Systematic review of prognostic factors for mortality in dogs with immune-mediated hemolytic anemia. Vet. Internal Med. 29(7), 7–13; doi:10.1111/jvim.12514 Tassies, D. and Reverter, J.-C. 2010. Hematologic abnormalities in the antiphospholipid syndrome. Curr. Rheumatol. Rev. 6(1), 55–63; doi: 10.2174/157339710790827740 Treuting, P.M. and Snyder, J.M. 2015. Mouse necropsy. CP. Mouse Biol. 5, 223–233; doi:10.1002/9780470942390.mo140296 Tucker, S.M., Baja, A.J., Rasche, B.L., Negrão Watanabe, T.T. and Harrell, K.A. 2023. Immune-mediated hemolytic anemia with severe thrombocytopenia in a pembroke Welsh corgi. Vet. Rec. Case. Rep. 11, e727; doi:10.1002/vrc2.727 Vallelian, F., Buzzi, R.M., Pfefferlé, M., Yalamanoglu, A., Dubach, I.L., Wassmer, A., Gentinetta, T., Hansen, K., Humar, R., Schulthess, N., Schaer, C.A. and Schaer, D.J. 2022. Heme stress-activated NRF2 skews the fate trajectories of bone marrow cells from dendritic cells toward red pulp-like macrophages in patients with hemolytic anemia. Cell Death Differ. 29, 1450–1465; doi:10.1038/s41418-022-00932-1 Verran, C., Shipman, A.R. and Shipman, K.E. 2024. Investigative algorithms for disorders affecting plasma bilirubin levels: a narrative review. J. Lab. Precis. Med. 9(6), 6; doi:10.21037/jlpm-23-24 Wang, X. and Deng, G.M. 2024. Animal models for studying the pathogenesis of multi-organ tissue damage in lupus. Clin. Immunol. 263, 110231; doi:10.1016/j.clim.2024.110231 Weinkle, T.K., Center, S.A., Randolph, J.F., Warner, K.L., Barr, S.C. and Erb, H.N. 2005. Evaluation of prognostic factors, survival rates, and treatment protocols for immune-mediated hemolytic anemia in dogs: 151 cases (1993–2002). JAMA 226, 1869–1880; doi:10.2460/javma.2005.226.1869 Wu, Y., Cai, B., Feng, W., Yang, B., Huang, Z., Zuo, C. and Wang, L. 2014. Double-positive CD4+CD8+ T cells: key suppressive role in autoantibody production in systemic lupus erythematosus. Indian J. Med. Res. 140, 513–519. | ||
| How to Cite this Article |
| Pubmed Style Permata FS, Herawati H, Pratama DAOA, Fauzi A, Aryani DE. Comparative effects of syngeneic graft and xenograft blood for hemolytic anemia modeling in dogs. Open Vet. J.. 2026; 16(1): 214-230. doi:10.5455/OVJ.2026.v16.i1.20 Web Style Permata FS, Herawati H, Pratama DAOA, Fauzi A, Aryani DE. Comparative effects of syngeneic graft and xenograft blood for hemolytic anemia modeling in dogs. https://www.openveterinaryjournal.com/?mno=279335 [Access: January 31, 2026]. doi:10.5455/OVJ.2026.v16.i1.20 AMA (American Medical Association) Style Permata FS, Herawati H, Pratama DAOA, Fauzi A, Aryani DE. Comparative effects of syngeneic graft and xenograft blood for hemolytic anemia modeling in dogs. Open Vet. J.. 2026; 16(1): 214-230. doi:10.5455/OVJ.2026.v16.i1.20 Vancouver/ICMJE Style Permata FS, Herawati H, Pratama DAOA, Fauzi A, Aryani DE. Comparative effects of syngeneic graft and xenograft blood for hemolytic anemia modeling in dogs. Open Vet. J.. (2026), [cited January 31, 2026]; 16(1): 214-230. doi:10.5455/OVJ.2026.v16.i1.20 Harvard Style Permata, F. S., Herawati, . H., Pratama, . D. A. O. A., Fauzi, . A. & Aryani, . D. E. (2026) Comparative effects of syngeneic graft and xenograft blood for hemolytic anemia modeling in dogs. Open Vet. J., 16 (1), 214-230. doi:10.5455/OVJ.2026.v16.i1.20 Turabian Style Permata, Fajar Shodiq, Herawati Herawati, Dyah Ayu Oktavianie Ardhiana Pratama, Ahmad Fauzi, and Dhita Evi Aryani. 2026. Comparative effects of syngeneic graft and xenograft blood for hemolytic anemia modeling in dogs. Open Veterinary Journal, 16 (1), 214-230. doi:10.5455/OVJ.2026.v16.i1.20 Chicago Style Permata, Fajar Shodiq, Herawati Herawati, Dyah Ayu Oktavianie Ardhiana Pratama, Ahmad Fauzi, and Dhita Evi Aryani. "Comparative effects of syngeneic graft and xenograft blood for hemolytic anemia modeling in dogs." Open Veterinary Journal 16 (2026), 214-230. doi:10.5455/OVJ.2026.v16.i1.20 MLA (The Modern Language Association) Style Permata, Fajar Shodiq, Herawati Herawati, Dyah Ayu Oktavianie Ardhiana Pratama, Ahmad Fauzi, and Dhita Evi Aryani. "Comparative effects of syngeneic graft and xenograft blood for hemolytic anemia modeling in dogs." Open Veterinary Journal 16.1 (2026), 214-230. Print. doi:10.5455/OVJ.2026.v16.i1.20 APA (American Psychological Association) Style Permata, F. S., Herawati, . H., Pratama, . D. A. O. A., Fauzi, . A. & Aryani, . D. E. (2026) Comparative effects of syngeneic graft and xenograft blood for hemolytic anemia modeling in dogs. Open Veterinary Journal, 16 (1), 214-230. doi:10.5455/OVJ.2026.v16.i1.20 |