| Research Article | ||
Open Vet. J.. 2026; 16(1): 303-317 Open Veterinary Journal, (2026), Vol. 16(1): 303-317 Research Article Feasibility and findings of different radiographic methods in diagnosing canine heartworm diseaseLjubica Spasojević Kosić*Department of Veterinary Medicine, Faculty of Agriculture, University of Novi Sad, Novi Sad, Serbia *Corresponding Author: Ljubica Spasojević Kosić. Department of Veterinary Medicine, Faculty of Agriculture, University of Novi Sad, Novi Sad, Serbia. Emails: ljubica.spasojevic [at] gmail.com; ljubicask [at] polj.uns.ac.rs Submitted: 21/08/2025 Revised: 30/11/2025 Accepted: 15/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
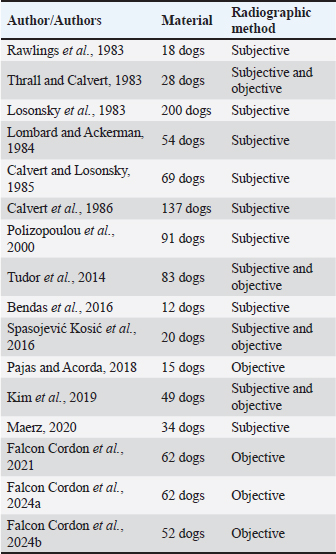
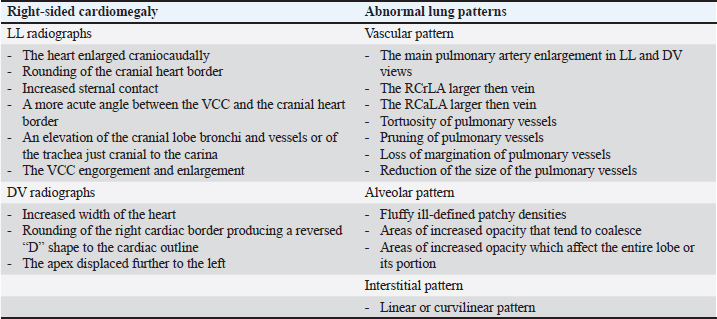
AbstractBackground: Heartworm disease (HWD) represents the pathological consequences of heartworm (HW) (Dirofilaria immitis) infection on different organs and organ systems, primarily the lungs and heart. Radiography can objectively assess the severity of cardiopulmonary disease secondary to HW infection. However, interpreting dogs’ radiographs can be quite challenging with respect to choosing an appropriate radiographic method. Aim: The aim of this article is to review the published literature on radiographic methods, both subjective and objective, emphasizing their advantages and disadvantages. In order to define the feasibility and findings of the used radiographic methods, radiographs (laterolateral-LL and dorsoventral-DV) of dogs (N=40) naturally infected with HWD were retrospectively analysed. Methods: Subjective radiographic assessment included all parameters of abnormal lung patterns and right-sided cardiomegaly in HWD. Objective methods used for the assessment of cardiac size and shape were: modified vertebral heart size (modified VHS), manubrium heart score, sternebral heart size (SHS), thoracic inlet heart size, and cardiac sphericity index (CSI). Blood vessels were measured and expressed in relation to different skeletal structures or the aorta, depending on the blood vessels. Results: While the increased sternal contact, reversed D heart shape, and loss of pulmonary vessel margination were the most feasible subjective parameters, the modified VHS, SHS, and CSI (VHS-LL=10.52 ± 0.97, VHS-DV=10.79 ± 1.12; SHS-LL=9.22 ± 1.04; SHS-DV=9.49 ± 0.96; CSI-LL=0.86 ± 0.08, CSI-DV=0.75 ± 0.07) were the most feasible objective methods. The ratios of the right cranial lobar artery (RCrLA) to the fourth thoracic vertebra (T4) and the fourth rib (4.r) were the most feasible blood vessel measurements (RCrLA/T4=0.27 ± 0.08, RCrLA/4.r=0.89 ± 0.25). In 5% of the dogs, radiographs could only be assessed subjectively. The most prevalent subjective parameters were increased sternal cardiac contact (91.89%), reversed D heart shape (89.65%), RCaLA enlargement (86.67%), and loss of pulmonary vessel margination (84.21%). The main pulmonary artery enlargement in LL, and the areas of increased opacity that tend to coalesce, significantly changed after successful HWD therapy. Conclusion: The most feasible and prevalent subjective parameters were the increased sternal contact, reversed D heart shape, and loss of pulmonary vessel margination. The most feasible objective methods were the modified VHS, SHS, and CSI. Keywords: Heartworm disease, Radiographic methods, Dogs. IntroductionHeartworm disease (HWD) implies heartworm (HW) infection, but also goes beyond this term. Diagnosis of HWD consists of the HW infection detection, and the determination of pathological consequences on relevant organs and organ systems (primarily on the lungs and heart, but also on the kidneys, joints, blood, and immune system) caused by a parasite—HW (Dirofilaria immitis—D. immitis). While the detection of HW infection is based on identifying circulating microfilariae and adult parasite antigen in blood samples, the diagnosis of HWD can require different diagnostic methods depending on the compromised organs or organ systems. The pathological consequences on different organs and organ systems are a result of the pathogenesis of the HWD. Since the parasite resides in the pulmonary arteries, the initial pathological lesion is villous myointimal proliferation (endothelial cell swelling, widening of intracellular junctions, increased endothelial permeability, and periarterial oedema). Endothelial damage further leads to arterial thrombosis, while endothelial permeability and periarterial oedema result in pulmonary infiltrates (neutrophils and eosinophils) and lung consolidation. Pulmonary eosinophilic infiltrates in HWD (eosinophilic bronchopneumopathy, allergic pneumonitis) are considered to be a manifestation of immunologic hypersensitivity caused by the HW (Knight, 1980; Schaub et al., 1981; Keith et al., 1983; Kittleson, 1998; Ware, 2011). When inflammatory pulmonary changes become chronic, their end-stage can be lung fibrosis (Kittleson, 1998). After the parasites die (either naturally or therapeutically), a more intensive host reaction, such as pulmonary thromboembolism (PTE), severe lung tissue damage, shock, and coagulopathy occurs (Kitoh et al., 1994a,b; Kitoh et al., 2001). In addition, severe complications of the HWD may include pulmonary hypertension (PH), right-sided heart failure and caval syndrome. HWD-induced PH is a consequence of increased pulmonary vascular resistance caused by intimal and media thickening of the pulmonary arteries and an obstruction of blood flow in the pulmonary arteries. The aforementioned vascular changes increase the right heart afterload, causing right ventricular hypertrophy and cor pulmonale (Dunn, 2000). Caval syndrome is a severe complication of the HWD, caused by the obstruction of v. cava caudalis (VCC) due to large-scale parasite infestation and their aberrant location towards the right heart (Atkins et al., 1988; Jones, 2016). Besides cardiovascular and pulmonary changes, microfilariae act as circulating antigens in blood, and thus have the potential to stimulate an immunological reaction and form immune complexes, with immune complex depositions in glomeruli or synovial membranes, causing glomerulonephritis or reactive polyarthritis (Dunn, 2000). The complex pathogenesis of the HWD and its pathological changes can be registered by the use of different diagnostic methods. In this article, we will focus on cardiopulmonary manifestations of the HWD, which are by far the most common in clinical practice. Radiography is considered the most objective method of assessing the severity of cardiopulmonary disease secondary to HW infection (AHS, 2025). The aim of the study is to review the literature on the subject of radiographic methods and their features relevant to the HWD, and to retrospectively analyze the feasibility and results of these methods in radiographs of dogs naturally infected with HWD. Literature reviewThoracic radiography is performed in dogs with HWD in order to see the morphology of the lung field and cardiac silhouette, and thus to determine respiratory and cardiovascular pathological changes caused by the parasite. Radiographic changes associated with HWD can be assessed both subjectively and objectively (Table 1). While subjective assessment of radiographs identifies and describes the presence of pulmonary vascular, parenchymal, and interstitial changes, as well as cardiac size and shape, objective radiographic assessment measures and determines cardiac size and shape, as well as the size of blood vessels relevant for HWD. Subjective radiographic assessment shows greater interobserver variability in comparison with objective methods, which are considered to be more reliable for inexperienced veterinarians as well as in questionable cases (Buchanan and Bücheler, 1995; Lamb et al., 2000; Hansson et al., 2005). Table 1. References selected on the basis of radiographic assessment in HW-infected dogs.
Typical vascular abnormalities include a bulging pulmonary artery segment and enlarged lobar pulmonary arteries, with peripheral pulmonary arteries becoming tortuous and truncated (Ackerman, 1987; Kittleson, 1998; Dunn, 2000; Polizopoulou et al., 2000). Arteritis lies at the root of enlarged arteries, to which PH may also contribute. Severe arteritis produces pulmonary artery tortuosity (Kittleson, 1998). Dead parasites act as pulmonary emboli, interrupting blood flow to lung segments, leading to the appearance of suddenly terminated pulmonary arteries, seen as blunted or pruned arteries (Kittleson, 1998). The enlargement of lobar pulmonary arteries, without concurrent venous distension, is strongly suggestive of HWD or other causes of PH (Ware, 2011). The right caudal lobar artery (RCaLA) is most commonly and heavily infested due to the effect of blood streaming toward this artery. Following the RCaLA, the left caudal lobar artery and subsequently, the cranial lobar arteries are the next commonly affected (Kittleson, 1998). Normally on radiographs, the VCC emerges through the diaphragm and slopes slightly cranioventrally towards cardiac caudal margin. It is similar in size to the aorta (Ao), but may get wider towards the heart (Herrtage and Denis, 1997). Enlargement of the VCC has been reported as a radiographic finding of right-sided cardiomegaly (Suter and Lord, 1984). Pulmonary parenchymal abnormalities vary from increased lung densities, seen in dogs with mild-moderate disease, up to generalized densities throughout the lung field (Kittleson, 1998). Parenchymal abnormalities, apparent especially after adulticide therapy, are seen as patchy alveolar densities (Dunn, 2000). Patchy pulmonary alveolar or interstitial infiltrates are the result of oedema, pneumonia, infarction, or fibrosis. Pulmonary opacities may be perivascular (Ware, 2011). Concerning heart remodeling, right ventricular enlargement is a characteristic radiographic feature (Dunn, 2000). This is a secondary change, arising as a consequence of PH. Right heart enlargement is seen in dogs with moderate to severe PH, and is best assessed on either the dorsoventral (DV) or ventrodorsal (VD) radiographic view (Lombard and Ackerman, 1984). Objective radiographic methods in HWD assess overall cardiac size and shape, and the size of blood vessels relevant to HWD in relation to various skeletal structures (intercostal spaces, vertebrae, sternebrae, thoracic inlet (TI), and etc.). The intercostal space method is based on counting the number of intercostal spaces that cover the heart silhouette (Buchanan, 1968; Buchanan and Bücheler, 1995). Although introduced a long time ago, it is still used, and like all other methods has its advantages and disadvantages. It is well known among clinicians and easy to use, because it is directly performed by counting the number of intercostal spaces while observing a dog’s lateral radiograph (LL). A normal heart silhouette has the size of 2.5 to 3.5 intercostal spaces, for deep-chested or wide-chested dogs respectively (Buchanan, 1968; Kealy, 1979; Owens, 1985). However, false negative and positive findings are possible due to thorax conformation, the position of the heart in the thorax, phase of respiration and position of the diaphragm, superimposition of ribs, and imprecise reference points (Buchanan and Bücheler, 1995). In 1995, the vertebral heart size (VHS) was introduced as an objective method, which expresses heart size through long axis (LA) and short axis (SA) normalized to the midthoracic vertebrae, starting from the fourth thoracic vertebra (T4) (Buchanan and Bücheler, 1995). In LL of dogs, the reference point for the cardiac LA is the ventral border of the left mainstem bronchus and the most distant ventral contour of the cardiac apex. This length reflects the combined size of the left atrium and left ventricle. The measured cardiac LA, made by using an adjustable caliper, is then repositioned over thoracic vertebrae beginning with the cranial edge of the T4. The distance to the caudal caliper point is then estimated to the nearest 0.1 vertebra (v). The maximal cardiac SA is measured in the central third region, perpendicular to the cardiac LA axis, and then also repositioned along thoracic vertebrae starting from the beginning of T4. The sum of LA and SA dimensions represent the overall size of the heart expressed as total units of vertebral length to the nearest 0.1 v. In DV or VD radiographs, the maximum LA and SA of the heart are determined with calipers in a similar fashion and measured against vertebrae in the LL radiograph beginning with T4 (Buchanan and Bücheler, 1995; Buchanan, 2000). The determined VHS in normal dogs is 9.7 ± 0.5 v in LL radiographs, and 10.2 ± 1.45 and 10.2 ± 0.83 v in DV and VD, respectively (Buchanan 2000). By using this method many studies have proposed breed-specific VHS reference values (Lamb et al., 2001; Gulanber et al., 2005; Bavegems et al., 2005; Marin et al., 2007; Castro et al., 2011; Jepsen-Grant et al., 2013). The following factors were found to influence mean VHS: interbreed variations, interobserver differences concerning references points of LA and SA, transformation into vertebral unit (v), and anomalies of thoracic vertebrae and intervertebral disc diseases (Hansson et al., 2005; Jepsen-Grant et al., 2013), cardiac cycle and, to a lesser extent, respiratory cycle (Olive et al., 2015). In order to overcome some of these limitations, modifications of this method, concerning the determination of reference points for SA of the heart and the introduction of the VHS unit (the length of one thoracic vertebra), have been proposed (Hansson et al., 2005; Spasojević Kosić et al., 2007; Costanza et al., 2022). The manubrium heart score (MHS) is proposed as the method that can overcome some limitations of the VHS method (Mostafa and Berry, 2017). In this method, the length of the manubrium (M) is used as a reference value to normalize the measurement of the height and width of the cardiac silhouette in right LL and VD thoracic radiographic views. On the LL view, the cardiac LA is measured from the ventral border of the carina to the caudoventral margin of the cardiac apex. The cardiac SA is measured on a line perpendicular to the LA at the widest point of the cardiac silhouette. On the VD view, the cardiac LA is measured from the distance of the right cranial margin of the heart silhouette to the cardiac apex, and the SA heart axis is measured at the widest line perpendicular to the LA. The M length is measured as the maximum M length on the right LL view. The reason why the M of the sternum is selected is that it is relatively prominent, easily identified, and measured on LL radiographs. At the same time, an evenly elongated, bullet-shaped, rectangular, or camel head-neck-shaped M is accepted for the MHS measurement. However, if the M of dogs is abnormal or its cranial edge cannot be identified, the calculation of the MHS is not possible. Established values of the MHS in normal healthy dogs vary from 4.8 ± 0.5 to 5.3 ± 0.8 in LL view, and from 5.4 ± 0.6 to 5.8 ± 0.9 in VD view in large and small dogs, respectively (Mostafa and Berry, 2017). Furthermore, the MHS is recommended as a useful, objective method for heart disease screening in dogs, capable of distinguishing between left-sided and right-sided cardiomegaly (Mostafa et al., 2020). In addition to MHS, the cardiac sphericity index (CSI) is another measure of the shape of the heart, which can be used to assess the globe-shaped heart. It is calculated by dividing the cardiac SA by the cardiac LA. The value of CSI≥1.00 means a near round figure of the cardiac silhouette (Guglielmini et al., 2012). Recently, the sternebral heart scale was proposed for the assessment of heart size in ferrets (Gutiérrez et al., 2022). Upon determination of cardiac LA and SA in radiograph views, the LA and SA of the heart are measured along the sternum, starting at the cranial edge of the fourth sternebra (S4). The sum of the cardiac LA and SA is expressed in sternebral units (s), estimated to an accuracy of 0.1 seconds. Thus, it determines the sternebral heart size (SHS) in this species. This method overcomes the limitation of the MHS caused by the abnormal shape of the M or the impossibility of cranial margin M line identification (Gutiérrez et al., 2022). Although the correlation between heart size and sternebral length was slightly lower compared to the correlation with the vertebral length in dogs (Buchanan and Bücheler, 1995), to the best of our knowledge, no study on the subject of the normal SHS in dogs has been published yet. First proposed as a reference point to assess tracheal diameter in brachycephalic and non-brachycephalic dogs (Harvey and Fink, 1982; Mostafa and Berry, 2022), the TI has recently been assessed as a unit for measuring thoracic inlet heart size (TIHS) (Marbella Fernández et al., 2023a). In this study, the TIHS has proved to be a simple and accurate method for measuring heart size, which overcomes some limitations seen in previously mentioned methods, such as vertebral or M malformations, and variations due to dog’s size and breed. Reference points for the cardiac LA and SA in this method are like the ones described for the VHS. Upon being measured the cardiac LA and SA are summed and divided by the corresponding TI length. The TI, defined as the minimum length of the TI, is determined as the distance between the cranio-ventral aspect of the first thoracic vertebra and the cranio-dorsal margin of the M at its highest point. The mean TIHS value for normal dogs is 2.86, and a TIHS value above 3.2 is considered to be an increased heart size in dogs (Marbella Fernández et al., 2023a). Besides methods based on linear determination of cardiac size through its LA and SA, which are then expressed by different skeletal structures, methods based on the estimation of cardiac areas have also been investigated. One of these modalities for cardiac size evaluation is the ratio between cardiac and thoracic areas (Torad and Hassan, 2014). Determination of the cardiothoracic ratio derived from LL thoracic radiographs (Empel and W, 1974) or on both LL and VD radiographs after peak inspiration and expiration (Castro et al., 2011; Torad and Hassan, 2014). Another way of assessing heart size is to compare the heart area to the T4 body area (So et al., 2024). These methods showed not only a correlation with VHS (Azevedo et al., 2016; So et al., 2024), but also, and to a higher extent, with the cardiac volume measured by computed tomography (So et al., 2024). However, these methods are complicated and require specific software, not always available in clinical practice. In addition, thoracic cavity conformation varies in different dog breeds, and individual changes in healthy and diseased animals (Buchanan and Bücheler, 1995; Castro et al., 2011; Torad and Hassan, 2014) make these methods of limited use. Blood vessels relevant to HWD (RCrLA, RCaLA, VCC) can also be assessed objectively, by comparing them with different skeletal structure, or, in case of the VCC, with the Ao. The width of the RCrLA on a LL view in normal dogs should not exceed the narrowest diameter of the 4th rib (4.r). On DV radiographs, the width of the RCaLA is normally not larger than the 9th rib (9.r) at the level of their intersection (Herrtage and Denis, 1997; Ware, 2011). Furthermore, ratios of RCrLA/4.r≥ 1.08, and RCaLA/9.r≥ 1.10 indicate the presence of PH, and thus show potential as a preliminary screening tool for PH in HW-infected dogs (Falcon Cordon 2024a). The measurement of VCC size and its ratio towards other fixed-sized surrounding structures, such as the length of vertebrae at the level of tracheal bifurcation, the descending Ao, and the width of the 4.r can help in the detection of right-side heart disease in dogs (Arya et al., 2021). The diameter of VCC should be considered normal in a dog with a ratio of VCC/Ao < 1.00, VCC/ v < 0.80, or VCC/4.r < 2.25 (Lehmkuhl et al., 1997). Material and MethodsData used in this study related to client-owned dogs in which HW infection was identified. For this purpose, the medical records of these dogs were reviewed with the collection of all data relevant for the retrospective radiographic analyses. The material for this study included radiographs of 40 dogs naturally infected with D. immitis. There were two criteria for this study. The first one was a complete HW infection diagnosis performed in these dogs (detection of antigen of adult parasite D. immitis and modified Knott test). The second criterion was that the dog had either LL or both (LL and DV) radiographs recorded without the use of sedation or anesthesia. While in 4 dogs only LL radiographs were available, in all others, both LL and DV radiographs were used for analyses. The dogs were of different age, gender, and breed. The average age of the dogs was 6.36 ± 3.68 years, and there were 12 females and 28 males belonging to different breeds (German Shepherd, Belgian Shepherd, Labrador Retriever, Golden Retriever, Rottweiler, Boxer, Dobermann, American Staffordshire Terrier, American Pit Bull Terrier, Great Dane, Dogo Argentino, Cane Corso, Hungarian Vizsla, German Hunting Terrier, Breton Spaniel, and mixed-breed dogs of small and medium size). For 8 out of these 40 dogs, radiographs of both projections (LL and DV) were available at the moment of the HWD diagnosis (before therapy) and at the end of successful therapy (after therapy). The alternative therapy (doxycycline and ivermectin) (Spasojević Kosić et al., 2024) was used as the HW infection therapy in all eight dogs. The dog was considered cured of HWD if clinical signs have disappeared and if no D.immitis antigenemia and microfilariaemia have been detected. At that time the post-treatment radiographs had been recorded. Thoracic radiographs were obtained using Eickemeyer Hiray Plus, Porta 100HF, with the exposure settings 46/2,5 kVp/mAs - 80/2,5 kVp/mAs, with a focal film distance of 75 cm (Primax flm RTG - G, 30x40, and DR system - Exam VueDuo). Each radiograph was submitted to subjective and objective assessment. Any difficulties which disabled predefined methodology of subjective parameters identifications and of objective measurements were considered as feasibility limitation criteria. All radiographic assessments were done by an experienced veterinary cardiologist. The radiographic assessments were done after being triple check before the final decision. Parameters relevant for subjective radiographic assessment were classified as radiographic features for right-sided cardiomegaly and abnormal lung patterns (Table 2). Table 2. Specific parameters of cardiomegaly and abnormal lung patterns in the subjective radiographic assessment for the HWD.
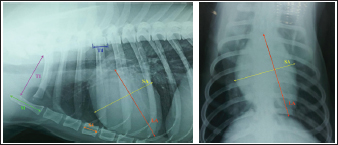
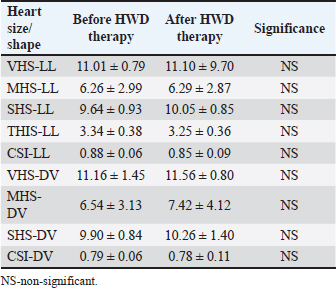
Objective methods for the assessment of cardiac size and shape used in this study were: modified VHS, MHS, SHS, this, and CSI. In each objective methods of cardiac silhouette measurement, the LA and SA were measured in the same way. In LL radiographs, cardiac LA length (mm) was the line extending from the ventral border of the left mainstem bronchus to the most distant ventral contour of the cardiac apex. SA length (mm) was the line drawn perpendicular to the cardiac LA on the widest part of the cardiac silhouette. In DV radiographs, the LA was drawn as the line starting from the dorsal margin of the right atrium projection to the left apex. The SA was drawn perpendicular to the LA at the widest part of the cardiac silhouette (Fig. 1A and B).
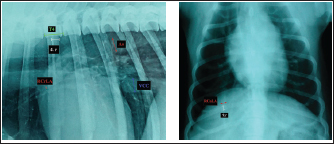
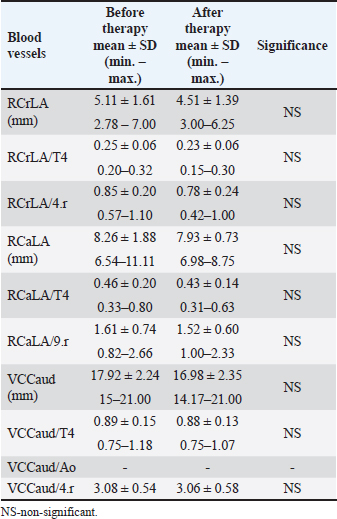
Fig. 1. A and B. Measurements of LA and SA in LL (A) and DV (B) views, with marked T4, S4, M, TI length: LA – long axis, SA – short axis, T4 - the length (mm) of the body and caudal disc of the T4vertebra, S4 - the length (mm) of the S4, M – the length (mm) of the M, TI - the length (mm) of a dog’s TI. Modified vertebral heart sizeThe cardiac LA and SA in LL view were measured as previously described, added up, and then transformed from mm into whole and 0.01 of VHS units (v), by dividing the sum of both axes by the length (mm) of the body and caudal disc of the T4 (VHS-LL) (Spasojević Kosić et al., 2007). The same was done to calculate the cardiac VHS in DV radiographs (VHS-DV). Manubrium heart scoreThe sum of the measured cardiac LA and SA (in mm) was normalized by the corresponding M length (mm) according to the method described by Mostafa and Berry (2017). The same was done for both MHS in LL and in DV radiographs (MHS-LL and MHS-DV). Sternebral heart sizeTo determine SHS, the length of the S4 is used as a unit of measurement. The sum of the measured LA and SA in mm was divided by the length of the S4, both for LL and DV radiographs, in order to get SHS in LL and DV (SHS-LL and SHS-DV). Thoracic inlet heart sizeThe sum of LA and SA (mm) was divided by the length of a dog’s TI. The shortest TI was measured from the craniodorsal M to the cranioventral first thoracic vertebra as described by Marbella Fernández et al. (2023a). For this method, cardiac silhouette measurement was performed only in LL radiographs (TIHS-LL). Cardiac sphericity indexCSI was calculated by dividing the cardiac SA by LA obtained from LL and DV radiographs (CSI-LL and CSI-DV) (Guglielmini et al., 2012). Measurement of blood vesselsObjective methods for assessment of blood vessels relevant to the HWD included several measurements (Fig. 2A and B) and calculations. The diameters of blood vessels relevant for the HWD (RCrLA, RCaLA, and VCC) were first measured and expressed in absolute values (mm). The diameters of the vessels were measured perpendicular to their LA. Relative measurements of the relevant blood vessels were expressed as ratios to the T4 (for all three blood vessels), and to the 4.r (for the RCrLA and VCC), to the 9.r (for the RCaLA), and to the Ao (for the VCC). The T4 was measured as described in modified VHS methods. The measurement of the 4.r was done on its proximal part as the smallest diameter. The measurement of the 9.r was measured at its intersection with the RCaLA. The Ao was measured at the descending part (Herrtage and Denis, 1997; Lehmkuhl et al., 1997; Arya et al., 2021).
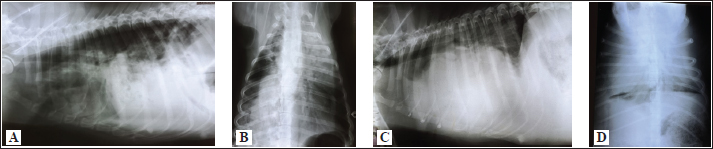
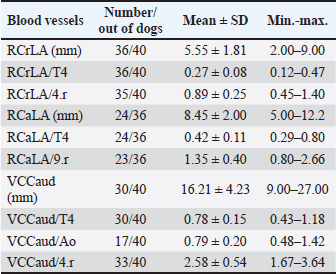
Fig. 2. A and B. Measurements of blood vessels relevant to the HWD in LL (A) and DV (B) radiographs: RCrLA – the width (mm) of the RCrLA, VCC – the width (mm) of the VCC, Ao – the width (mm) of the Ao, T4 – the length (mm) of the body and caudal disc of the T4 vertebra, 4.r – the width (mm) of the 4.r, RCaLA – the width (mm) of the right caudal lobar artery, 9.r – the width (mm) of the ninth rib. Ethical approvalAccording to the national law, an ethical approval for this research was not needed. However, a written consent was obtained from the owners. This study was carried out in compliance with the existing animal welfare laws in Europe. Statistical analysesA commercial software package TIBCO Statistica was used for the following statistical analyses: descriptive statistics, the correlation coefficient (r) and coefficient of determination (R2), the Fisher probability test, and the Wilcoxon matched pairs test. The prevalence of subjective radiographic parameters was expressed in absolute and relative numbers (percentages). The correlations between skeletal units (T4, M, S4, TI), as well as between LA, SA, the sum of LA and SA with skeletal units of heart size, were assessed. The correlation was classified as negligible (r=0.00–0.09), weak (r=0.10–0.39), moderate (r=0.40–0.69), strong (r=0.70–0.89), and very strong (r=0.90–1.00). In order to assess the occurrence of subjective parameters of cardiomegaly and abnormal lung patterns in the dogs with HWD before and after therapy the Fisher probability test was used. The Fisher probability test was followed by the post hoc false discovery rate (FDR) test (Benjamini and Hockberg, 1995). The objective parameters of radiographic assessments (cardiac size and shape, and blood vessels measurements) were expressed as the mean ± SD, as well as the minimum and the maximum. The same parameters in the dogs with HWD before and after therapy were assessed by using the Wilcoxon matched pairs test. A probability value of p < 0.05 was considered as significant. ResultsIn 2 out of 40 dogs (2/40 dogs or 5% of dogs), radiographs could only be assessed subjectively. These two dogs had the most severe radiographic features in the areas of lung fields, which made it impossible to assess their cardiac silhouette and vascular pattern, either subjectively or objectively (Fig. 3A–D).
Fig. 3. Radiographs of two dogs with the most severe changes. 3A and B - LL and DV views of a 9-year-old female Rottweiler. Massive opacities can be seen in the ventral areas of the cranial, middle and accessory lobes (in LL view), and in the left cranial, right cranial and caudal and accessory lobes (in DV view). These changes cover the heart silhouette. Also, reduced flows to the left caudal and right middle lobes can be seen. In the caudal lobes in LL view, and in the right caudal lobes in DV view, air alveolograms can be seen. 3C and D - LL and DV views of a 5-year-old female mix-breed dog: Massive opacification in the ventral areas of lung lobes in LL view made it impossible to define the heart silhouette. The trachea lies almost parallel to the spine, with a mixed interstitial-alveolar pattern of the caudal lung lobes. On the DV view diffuse opacities in lung lobes prevent cardiac silhouette and vascular pattern assessment. The radiographs of all other dogs were considered suitable for assessment both subjectively and objectively. Aside from 4 dogs, in which only LL were available, there were also two dogs in which opacity in their lung field in their DV radiographs enabled only the assessment of some subjective and objective parameters. The most prevalent features of subjective radiographic assessment (Table 3) of the HWD in these dogs were the increased sternal contact (91.89%), the rounding of the right cardiac border (89.65%), and the RCaLA larger than the vein (86.67%). Radiographs of almost all the dogs (37/38 dogs or 97.37%) or all of them (38/38 dogs or 100%) were eligible for assessment and detection of the more prevalent parameters of cardiomegaly (increased sternal contact, rounding of the cranial heart border) and abnormal lung field (loss of margination of pulmonary vessels). However, not each of the subjective assessment parameters could be analyzed in all the dogs. In 55.88% of the dogs (19/34 dogs), it was not possible to compare the width of the RCaLA and the corresponding vein. The most common reasons were one or several of these: unclear vein margins, opacity of the surrounding lung field, incorrect dog position. In 55.26% of the dogs (21/38 dogs), it was not possible to define whether the VCC is larger than the Ao due to the dogs’ respiratory cycle (position of the diaphragm) at the time of radiographic recordings, the opacity of surrounding tissues and not clearly visible margins of the Ao. In one dog (5.88% of dogs), VCC was wider than the Ao. Table 3. Prevalence and feasibility of subjective radiographic features in the dogs with HWD.
When we statistically analyzed the subjective radiographic parameters in the dogs with HWD before and after therapy, no statistically significant difference was registered in the occurrence of cardiomegaly parameters. However, among the subjective parameters of abnormal lung patterns, the enlargement of the main pulmonary artery in LL (p=0.0256), the loss of margination of pulmonary vessels (p= 0.0203), the areas of increased opacity that tend to coalesce (p= 0.0385), have shown significant differences before and after therapy in the dogs with HWD. The FDR post hoc analysis showed that the enlargement of the main pulmonary artery in LL (p ≤ 0.033, FDR), and the areas of increased opacity that tend to coalesce (p ≤ 0.05, FDR) had the significant lower occurrence after therapy. Objective measurements of cardiac size and shape were done in different number of dogs depending on the methods used (Table 4). VHS could not be determined in 2 and 4 dogs in LL and DV, respectively. The reason for this was lung opacity, which prevented heart silhouette determination. The same reason for the feasibility of SHS and CSI in LL and DV views was noted in an equal number of dogs. However, the number of dogs in which it was not possible to determine MHS and TIHS was higher, mainly due to incorrect positioning of the dogs and the impossibility of cranial M margin determination. Table 4. Cardiac size and shape in the dogs with HWD measured by different objective methods.
Measured values of the T4, the M and the S4 varied among the dogs, but significant correlations were determined among them. The strongest correlation existed between the T4 and the S4 (r=0.91, p < 0.05; 0.95CI; R2 =0.83), but the correlation between the T4 and the M (r=0.86, p < 0.05; 0.95CI; R2 =0.74), and the M and the S4 were also significant (r=0.84, p < 0.05; 0.95CI; R2 =0.70). However, the correlations between the T4, the M, the S4, and the length of TI have not been found. Significant correlations (p < 0.05) between LA, SA, the sum of LA and SA, and each skeletal unit (T4, S4, M, TI) were detected both in LL and DV radiographs. The strongest correlations were found between LA, SA, the sum of LA and SA, and T4 (very strong and strong, p < 0.05) and S4 (very strong and strong, p < 0.05). There were strong correlations between LA, SA, the sum of LA and SA, and TI (r > 0.7, p < 0.05). Moderate correlations existed between LA, SA, the sum of LA and SA, and M (r > 0.6, p < 0.05). Blood vessel (RCrLA, RCaLA, and VCC) measurements, as absolute and relative values, are shown in Table 5. The RCrLA could be measured in the majority of dogs (36 out of 40). In four dogs, this vessel could not be assessed because of areas of increased densities in the cranial lung lobes and perivascular opacities. The RCaLA could not be defined in 12 dogs, due to perivascular opacities, increased densities in lung lobes, incorrect positioning, or the dog’s respiratory phase. It was not possible to assess the VCC in 10 dogs. The reasons for this were: respiratory phase, perivascular opacities, increased opacity in that area of the lung field, superposition with ribs or bronchi. Table 5. The measurements of the relevant blood vessels in the dogs with HWD.
Cardiac size and shape assessed by calculated modified VHS, MHS, SHS, TIHS, and CSI, both in LL and DV radiographs, did not differ significantly before and after HWD therapy (Table 6). Table 6. Objective heart size/shape measurements before and after therapy in the dogs with HWD.
Dimensions of the RCrLA, the RCaLA, and the VCC were lower after therapy for HWD (Table 7). However, the dimensions of the blood vessels (RCrLA, RCaLA, VCC) in those eight dogs before and after therapy did not show a statistically significant difference, regardless of the form of width expressions (absolutely in mm or relatively as a ratio to different skeletal structures). The VCC/Ao ratio could only be measured in one dog before and after therapy, which made this parameter irrelevant for statistical analysis. Table 7. The blood vessel measurements before and after therapy in the dogs with HWD.
DiscussionThoracic radiography is a widely available diagnostic method used for the evaluation of cardiovascular and respiratory organ systems in small animal practice. In spite of its limitations, it is still used to assess the heart, blood vessels, and lung field. Interpreting dog radiographs can be challenging not only because of the wide variety of thoracic shapes and sizes in different breeds, but also due to positioning and technique. Furthermore, the numerous described radiographic methods for assessing heart size and shape [intercostal spaces, VHS, heart to single vertebra ratio (HSVR), MHS, SHS, TIHS, CSI, cardiothoracic ratio, tracheal-bifurcation angle, vertebral left atrial size, modified vertebral left atrial size, radiographic left atrial dimension, radiographic left atrial dimension to spine, bronchus to spine] seem to contribute to interpretation complexity. In the present study, radiographic methods relevant for the assessment of HWD characteristics were applied on radiographs of dogs with HWD in order to test their feasibility and determine their results. In clinical practice, diagnostic methods that are accurate, easy to perform, and no time consuming are the preferable ones. By comparing different methods on the same radiograph, it is possible to find out which diagnostic methods have higher potential over others in terms of feasibility. However, as this study shows, it is possible that in some patients more accurate and objective radiographic methods are inferior to others. Therefore, a holistic approach that includes both subjective and objective methods is required in interpreting dog radiographs. Subjective radiographic methods could be used in all dogs, while none of the applied objective methods could be used in two dogs, which had the most severe radiographic changes. Abnormal lung field, in the sense of massive opacities throughout several lung lobes, was the reason that prevented the assessment of the cardiac silhouette and blood vessels in these two dogs. Such changes raised suspicions about the existence of PTE, one of the most severe HWD complications. Subjective parameters of cardiomegaly, and even more so of abnormal lung patterns, could be assessed in the majority of dogs. However, the impossibility of assessing certain subjective parameters, even the most prevalent ones in the dogs with HWD, such as the size of the RCaLA in comparison to that of the corresponding vein, was observed. Pathological changes in the lung field, and incorrect positioning of the patient were mostly the reasons. Among the objective methods for the determination of cardiac size and shape, the most feasible ones were modified VHS, SHS, and CSI (in 38 out of 40 dogs for LL view, and 32 out of 36 dogs for DV view). The determination of cardiac size by MHS and TIHS was possible in only a smaller number of the dogs (in 23 and 26 dogs, respectively). While pathological lung changes prevented the determination of modified VHS, SHS, and CSI, particularly in DV radiographic view, the incorrect positioning of the dog and unsuccessful determination of the cranial M margin were the reasons for the impossibility of MHS and TIHS measurements. The assessment of the blood vessels relevant for HWD was most feasible for the RCrLA, followed by the VCC and the RCaLA, both for subjective and objective methods. In the case of the RCrLA, detection was possible in 36 dogs, both for the subjective method, in which it was compared with the corresponding vein, and for the objective methods, in which its width was expressed in mm, or as a ratio to the T4. It was possible to assess the VCC in comparison to the Ao, either subjectively or objectively, only in 17 dogs. However, it was more feasible to assess the VCC by direct measurement or indirectly as a ratio to the T4 (in 30 dogs) or to the 4.r (in 33 dogs). In the present study, objective methods of cardiac size and shape measurements were done as previously described (Guglielmini et al., 2012; Mostafa and Berry, 2017; Marbella Fernández et al., 2023a), with the exception of VHS, in which case the modified VHS was used. We have previously proved that no significant differences exist between heart size measured by the original VHS and the modified VHS methods in healthy dogs of the same breed (Spasojević Kosić et al., 2007), and in dogs with HWD of different breeds (Spasojević Kosić and Lalošević, 2025). Recently, the comparison of the cardiac axes with a single thoracic vertebra was also introduced as an objective method of evaluation of the cardiac silhouette (Costanza et al., 2022). The most accurate way of measurement in this, so-called, heart to single vertebra ratio (HSVR) is when the cardiac axes sum is compared with the length of the seventh thoracic vertebra (T7). Besides T7, other thoracic vertebrae can be used in the following order of preference T8, T5, T6, and, at the end T4, as it shows the lowest, although still acceptable level of agreement with VHS (Costanza et al, 2022). In our study, we used T4, not just because of the lack of significant difference from the original VHS method, but because of the uniformity of the measurements, since the blood vessels dimensions were also presented in this manner. We have found a very strong correlation between T4 and S4 (r=0.91, p < 0.05), and also significant but lower correlation between T4 and M (0.86, p < 0.05), and M and S4 (0.84, p < 0.05). However, there were no significant correlations between any of these skeletal structures and TI. This is opposite to the significant correlation found between TI and T4 in the study by Marbella Fernández et al. (2023a). In the study of Buchanan and Bücheler (1995), good correlations were detected between the LA dimension, SA dimension, and the sum of LA and SA dimensions and the 10 v reference length, as well as between heart size and the length of 3 or 4 sternebrae. Similarly, significant correlations were detected between cardiac LA, SA, the sum of the LA and SA, and the M length (Mostafa and Berry, 2017). These findings are in accordance with significant correlations, from very strong to moderate, detected in the present study between LA, SA, the sum of LA and SA, and each of the skeletal units (T4, S4, TI, M). In the study of Buchanan and Bücheler (1995), the M was excluded because of its variability, and the sternal measurements extended from the cranial edge of the second sternebra to the caudal edge of the fourth and fifth sternebrae were done. The purpose of the sternal measurement was to determine whether sternal length is more reliable than vertebral length as an index correlate for the heart size. As sternebral correlations were slightly less than vertebral correlations, no advantages of such measurement were seen, except in dogs with vertebral anomalies. In spite of these findings in dogs, and similar ones in cats (Litster and Buchanan, 2000; Ghadiri et al., 2008), sternebral heart score was not introduced as a method for heart size assessment until the recent study in ferrets (Gutiérrez et al., 2022). In accordance with the modified VHS method, the original SHS method (Gutiérrez et al., 2022) is modified in the present study, and a single sternebra, the S4 is used as a skeletal unit. In addition, a pronounced sternum arch in certain dogs due to chest conformation made it impossible to perform the measurement along the sternum. It was not difficult to perform this method, and it was equally feasible in the dogs with HWD as the modified VHS. Also, there were very strong and strong correlations between the S4 and the T4, and the S4 and heart size. Thus, the SHS method might be a useful method for evaluating heart size in dogs. However, the accuracy of each novel method of measurement should be estimated in comparison with gold standard methods, and its reproducibility should be tested so that an operator’s experience has a minimum impact on the results. Finally, reference values of measurement in healthy dogs should be defined. The subjective parameters of cardiomegaly, such as the increased sternal contact (91.89%), reversed “D” shape of the heart (89.65%), vascular patterns, such as the RCaLA enlargement (86.67%), and the loss of margination of pulmonary vessels (84.21%) were the most prevalent subjective radiographic parameters in the dogs with HWD. Similarly to our study, the reverse D shape of the heart, main pulmonary artery, and peripheral pulmonary artery dilation, as well as evidence of right heart failure were noted in the study by Kim et al. (2019). In another study (Maerz, 2020), enlarged peripheral pulmonary arteries were detected in the majority of dogs (65%), followed by parenchymal lesions (41%) and bulging of the main pulmonary artery (23.5%). In the study by Tudor et al. (2014), vascular radiographic changes (72.29%), pulmonary parenchymal changes (61.44%), and heart shape and silhouette changes (46.99%) dominated in the radiographs of dogs with HWD. Thrall and Calvert (1983) detected an enlarged right ventricle and RCaLA in every dog with HWD. These radiographic features were followed by pathological pulmonary abnormalities, enlarged RCrLA, main pulmonary artery, and VCC (Thrall and Calvert, 1983). Similar findings in dogs with HWD, such as the detection of right ventricular, main pulmonary, and RCrLA enlargement, were found by other authors (Losonsky et al., 1983). In the present study, pleural effusion was not detected in the dogs with HWD. The same result was reported in the study by Thrall and Calvert (1983), while Tudor et al. (2014) identified pleural effusions in 2.41% of dogs. Even though the parameters of cardiomegaly were more prevalent than the lung abnormality parameters, when radiographs before and after the HWD therapy were analyzed, no significant difference was found in the occurrence of the parameters of cardiomegaly, but among abnormal lung pattern parameters. After HWD therapy, the occurrence of main pulmonary artery enlargement in LL (p=0.0256; p ≤ 0.033, FDR), and the areas of increased opacity that tend to coalesce (p=0.0385; p ≤ 0.05, FDR) were significantly lower than before therapy. Thus, subjective radiographic assessment in the present study was able to register an improvement of lung pattern abnormalities, but not changes in cardiac size. In another study, radiographic features, such as increased pulmonary trunk, right atrial and ventricular enlargement, diffuse densification, and reticular interstitial lung patterning of the diaphragmatic lobes, became less frequent in the radiographs of the dogs after the therapy for HWD (Bendas et al., 2016). The only radiographic characteristic that was more frequent in dogs after the HWD therapy was increased diameter of the pulmonary arteries (Bendas et al., 2016). However, in the study of Bendas et al. (2016), the frequency of the radiographic features before and after the therapy, was expressed in absolute numbers without any statistical analysis. Cardiac size showed higher values in DV radiographs in all methods of objective measurement. In LL view, a heart size of 10.52 ± 0.97 v was measured by the modified VHS method, and in DV, the measured heart size was 10.79 ± 1.12 v. A similar value of VHS (10.59 ± 0.87 v in LL view) was measured in dogs of different breeds with HWD (Tudor et al., 2014). The value of VHS≤10.5 v is suggested as the clinically upper limit for normal heart size in most breeds (Buchanan and Bücheler, 1995). Despite the fact that thoracic radiography can underestimate the severity of HWD in small dogs, no significant difference was found between VHS in small and large breed dogs (Kim et al., 2019). Also, when values of VHS in LL and DV views were compared between dogs in different classes of HWD (classes 1–3), no significant difference was observed (Pajas and Acorda, 2018). When the M is used as a skeletal unit of cardiac size measurement, the mean value of 5.19 ± 1.80 was measured in LL radiographs in the present study. This value is similar to the value of MHS (5.2 ± 0.4) in large dogs with right-sided heart disease reported by other authors (Mostafa et al., 2020). More than overall MHS, the value of short MHS (the ratio of cardiac SA and the M length) is suggestive of right-sided heart disease (Mostafa et al., 2020). Known as a measure of cardiac shape, the CSI values of 0.86 ± 0.08 and 0.75 ± 0.07, in LL and DV views, respectively, were measured in the dogs with HWD in the present study. These values are lower than the CSI values of 88 ± 6.0 and 92 ± 8.0 measured in large and small dogs with right-sided heart disease (Mostafa et al., 2020), and in dogs with dirofilariasis belonging to various classes of HWD (Pajas and Acorda, 2018). By using TI as a skeletal unit, the heart size of 3.03 ± 0.41 was measured in LL radiographs of the dogs with HWD in the present study. This value is higher than the mean TIHS value of normal dogs (2.86 ± 0.27), but lower than the upper limit (3.2) of normal heart size in healthy dogs (Marbella Fernandez et al., 2023a). Although it is shown that a value of TIHS ≥ 3.3 can identify left heart enlargement in dogs with MMVD (Marbella Fernandez et al., 2023b), no studies on the subject of TIHS in right heart enlargement, right-sided heart disease, or HWD have been published yet. Values of 9.22 ± 1.04 and 9.49 ± 0.96 were measured by SHS in LL, and DV views, respectively. Since no values of SHS in healthy and diseased dogs have been published yet, it is not possible to compare this result with other studies. Similarly to the results of subjective radiographic assessment, no significant differences were detected between objective parameters of heart size and shape before and after therapy in the present study. However, another study shows that VHS has a tendency to increase after therapy for HWD in dogs with or without PH (Falcon Cordon et al., 2024b). In the study by Falcón Cordón et al. (2021), the RCrLA and the RCaLA expressed as a ratio to the ribs in dogs with HWD-induced PH were 1.4 ± 0.4 and 1.6 ± 0.6, respectively, while lower values (0.89 ± 0.25 for the RCrLA and 1.35 ± 0.40 for the RCaLA) were measured in the present study. There is a scarcity of reports about measurements of the pulmonary lobar arteries in HWD. On the other hand, several studies reported measurements of VCC. In dogs with dirofilariasis, classified in classes from 1 to 3, the VCC was measured as a ratio to Ao, T4, and 4.r (Pajas and Acorda, 2018). Our measurement of VCC/Ao (0.79 ± 0.20) was lower compared to the values in dogs in different classes of dirofilariasis (0.84 ± 0.11 – 1.02 ± 0.11) published by Pajas and Acorda (2018). The value of the VCC/T4 in our study (0.78 ± 0.15) was in the range of measured values in dogs in different stages of the disease (0.74 ± 0.07–0.81 ± 0.09) (Pajas and Acorda, 2018). However, the VCC/4.r in our study (2.58 ± 0.54) was higher than values reported in dogs in different classes of dirofilariasis (1.96 ± 0.21–2.25 ± 0.28) (Pajas and Acorda, 2018). Nonetheless, our measurements of the VCC (VCC/Ao, VCC/T4, VCC/4.r) were much lower than the values (VCC/Ao >1.50, VCC/T4 >1.30, VCC/4.r>3.50) reported for right sided heart abnormality by Lehmkuhl et al. (1997). Although observed, the diminishment of the relevant blood vessel diameters was not of statistical significance in the present study. The same findings for the lobar arteries were detected after HWD therapy in dogs without PH, but not in dogs with PH, where a significant increase in RCrLA/4.r and RCaLA/9.r ratios was observed after HWD therapy, indicating the persistence and worsening of the arterial damage (Falcon Cordon et al., 2024b). In this sense, it was concluded that RCrLA/4.r and RCaLA/9.r ratios, together with VHS, may be useful screening tools with which clinicians can decide whether to perform further diagnostic procedures, primarily echocardiography, to determine the presence of PH (Falcon Cordon et al., 2024a,b). Some limitations of the present study should also be addressed. The study design did not allow considerations of interobserver variability and interbreed variations for different radiographic methods. Due to the fact that SHS in normal healthy dogs has not been reported yet, it was not possible to compare the calculated value of SHS in the dogs with HWD. In addition, other diseases, mainly of cardiovascular and respiratory systems, which could influence or potentiate the radiographic features, were not ruled out by echocardiography. When HW infection is diagnosed, in order to establish the HWD diagnosis, both radiographic and echocardiographic examinations should be performed, as the two are considered complementary and successful in the classification of the disease and its risks. In addition, more sophisticated methods, like echocardiography and computed tomography, can definitely evaluate the utility and reliability of radiographic methods for HWD diagnosis. ConclusionOnly subjective radiographic assessment was possible in 5% of the dogs with HWD due to the severity of their lung abnormality pattern. Among the most prevalent parameters of cardiomegaly and abnormal lung patterns, increased sternal contact, reversed D cardiac shape, and loss of pulmonary vessel margination were available for the assessment in the highest number of dogs, while the RCaLA was possible to assess in the lowest number of dogs. The most feasible objective methods for the assessment of cardiac size and shape were the modified VHS, SHS, and CSI, while the most feasible ones for blood vessels were RCrLA/T4 and RCrLA/4.r. Heart sizes of 10.52 ± 0.97 and 10.79 ± 1.12 measured by the modified VHS method in LL and DV view, respectively, were slightly above the value for the normal dogs. In the present study, the values of the RCrLA in the dogs with HWD were 0.27 ± 0.08 for RCrLA/T4, and 0.89 ± 0.25 for RCrLA/4.r. Finally, the present study revealed subjective parameters as superior to objective parameters in defining an improvement in the radiographs after therapy. Conflict of interest The author declares that there is no conflict of interest. Funding This work was partly supported by the Ministry of Science, Technological Development and Innovation, Republic of Serbia (contract number 451-03-137/2025-03/200117). Author’s contributions Lj. Spasojević Kosić performed all measurements and analyses, wrote the manuscript and created all tables and figures. Data availability All data supporting the findings of this study are available within the manuscript. ReferencesAckerman, N. 1987. Radiographic aspects of heartworm disease. Semin. Vet. Med. Surg. 2, 15–27. AHS. 2025. American heartworm society canine guidelines for the prevention, diagnosis, and management of heartworm (Dirofilaria immitis) infection in dogs. Available via https://d3ft8sckhnqim2.cloudfront.net/images/AHS_Canine_Guidelines_WEB_19JUN2025.pdf? Arya, M., Tyagi, S.P., Kumar, A., Kumar, A. and Kumar, S. 2021. Radiographic mensuration of Caudal Vena Cava, Aorta, vertebral length and fourth rib width and ascertaining their ratios in healthy dogs. Indian J. Anim. Res. B-4676, 1–4. Atkins, C.E., Keene, B.W. and Mcguirk, S.M. 1988. Pathophysiologic mechanism of cardiac dysfunction in experimentally induced heartworm caval syndrome in dogs: an echocardiographic study. Am. J. Vet. Res. 49, 403–410. Azevedo, G.M., Pessoa, G.T., Moura, L.D.S., Sousa, F.D.C.A., Rodrigues, R.P.D.S., Sanches, M.P., Fontenele, R.D., Barbosa, M.A.P.D.S., Neves, W.C., Sousa, J.M.D. and Alves, F.R. 2016. Comparative study of the Vertebral Heart Scale (VHS) and the cardiothoracic ratio (CTR) in healthy poodle breed dogs. Acta. Sci. Vet. 44, 1387. Bavegems, V., Van Caelenberg, A., Duchateau, L., Sys, S.U., Van Bree, H. and De Rick, A. 2005. Vertebral heart size ranges specific for Whippets. Vet. Radiol. Ultrasound 46, 400–403. Bendas, A., Rodrigues, A.C.M., Soares, D.D.V., Labarthe, N.V. and Mendes-de-Almeida, F. Radiographic findings of Dirofilaria immitis in naturally infected dogs submitted to slow-kill adulticide treatment. In Proceedings of the 2016 WSAVA Congress, 2016. Available via https://www.vin.com/members/cms/project/defaultadv1.aspx?pId=19840&catId=105918&id=8250044&ind=165&objTypeID=17 Benjamini, Y. and Hochberg, Y. 1995. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Soc. B Method. 57(1), 289–300. Buchanan, J.W. 1968. Radiology of the heart. In Proceedings of the 35th Annual Meeting of the American Animal Hospital Association 1968, American Animal Hospital Association, South Bend, Indiana, pp 34–45. Buchanan, J. 2000. Vertebral scale system to measure heart size in radiographs. Vet. Clin. North. Am. Small. Anim. Prac. 30(2), 379–393. Buchanan, J.W. and Bücheler, J. 1995. Vertebral scale system to measure canine heart size in radiographs. J. Am. Vet. Med. Assoc. 206, 194–199. Calvert, C.A. and Losonsky, J.M. 1985. Pneumonitis associated with occult heartworm disease in dogs. J. Am. Vet. Med. Assoc. 186, 10, 1097–1098. Calvert, C.A., Losonsky, J.M., Brown, J., and Lewis, R.E. 1986. Comparisons of radiographic and electrocardiographic abnormalities in canine heartworm disease. Vet. Radiol. 27, 2–7. Castro, M.G., Tôrres, R.C.S., Araújo, R.B., Muzzi, R.A.L. and Silva, E.F. 2011. Radiographic evalu-ation of the cardiac silhouette in clinically normal Yorkshire Terrier dogs through the vertebral heart size method. Arq. Bras. Med. Vet. Zootec. 63, 850–857. Costanza, D., Greco, A., Piantedosi, D., Bruzzese, D., Pasolini, M.P., Coluccia, P., Castiello, E., Baptista, C.S. and Meomartino, L. 2022. The heart to single vertebra ratio: a new objective method for radiographic assessment of cardiac silhouette size in dogs. Vet. Radiol. Ultrasound 64(3), 378–384. Dunn, J.K. 2000. Diseases of the cardiovascular system. In Textbook of small animal medicine. Ed., Dunn, J.K. London, UK: WB Saunders, pp: 255–344. Empel, W. 1974. Beurteilung der Grosse und Gestalt des Herzschattens des Deutschen Schäferhundes. Kleintierprax 19, 7–13. Falcón Cordón, S., Carretón Gómez, E., Rivero, J.I.M., Costa Rodríguez, N., Rodríguez, S.N.G. and Alonso, J.A.M. Utility of thoracic radiology as clinical indicator of pulmonary hypertension in dogs with heartworm disease (Dirofilaria immitis). In Proceedings of the 28th International Conference of the World Association for the Advancement of Veterinary Parasitology, 2021. Available via https://www.researchgate.net/publication/353435749_Utility_of_thoracic_radiology_as_clinical_indicator_of_pulmonary_hypertension_in_dogs_with_heartworm_disease_Dirofilaria_immitis Falcón-Cordón, S., Falcón-Cordón, Y., Caro-Vadillo, A., Costa-Rodríguez, N., Montoya-Alonso, J.A. and Carretón, E. 2024b. Association between thoracic radiographic changes and indicators of pulmonary hypertension in dogs with heartworm disease. Animals 14(13), 1900; doi:10.3390/ani14131900 Falcón-Cordón, S., Falcón-Cordón, Y., Costa-Rodríguez, N., Matos, J.I., Montoya-Alonso, J.A. and Carretón, E. 2024a. Assessment of thoracic radiographic alterations in dogs with heartworm and their correlation with pulmonary hypertension, pre- and post-adulticide treatment. Animals 14, 2551; doi:10.3390/ani14172551 Ghadiri, A., Avizel, A., Resekh, A. and Yadegari, A. 2008. Radiographic measurement of vertebral heart size in healthy stray cats. J. Feline Med. Surg. 10, 61–65. Guglielmini, C., Diana, A., Santarelli, G., Torbidone, A., Di Tommaso, M., Baron Toaldo, M. and Cipone, M. 2012. Accuracy of radiographic vertebral heart score and sphericity index in the detection of pericardial effusion in dogs. J. Am. Vet. Med. Assoc. 241, 1048–1055. Gulanber, E.G., Gonenci, R., Kaya, U., Aksoy, O. and Biricik, H.S. 2005. Vertebral scale system to measure heart size in thoracic radiographs of Turkish Shepherd (Kangal) dogs. Turk. J. Vet. Anim. Sci. 29, 723–726. Gutiérrez, A., Ezquerra, L.J., Rodríguez, P.L. and Jiménez, J. 2022. Cardiac radiographic measurements in ferrets using the OsiriX MD programme. Front. Vet. Sci. 8, 795947; doi:10.3389/fvets.2021.795947 Hansson, K., Haggstrom, J., Kvart, C. and Lord, P. 2005. Interobserver variability of vertebral heart size measurement in dogs with normal and enlarged hearts. Vet. Radiol. Ultrasound 46, 122–130. Harvey, C.E. and Fink, E.A. 1982. Tracheal diameter: analysis of radiographic measurements in brachycephalic and nonbrachycephalic dogs. J. Am. Anim. Hosp. Assoc. 18, 570–576. Herrtage, M. and Denis, R. 1997. The thorax. In Manual of small animal diagnostic imaging, 2nd ed. Ed., Lee, R. Quedgeley, UK: BSAVA, pp: 43–67. Jepsen-Grant, K., Pollard, R.E. and Johnson, L.R. 2013. Vertebral heart scores in eight dog breeds. Vet. Radiol. Ultrasound. 54, 3–8. Jones , S.L. 2016. Canine caval syndrome series part 2: a practical approach to diagnosing caval syndrome. TVP. 2016, 63–67. Kealy, J.K. 1979. Diagnostic radiology of the dog and cat. Philadelphia, PA: WB Saunders. Keith, J.C., Schaub, R.G. and Ralings, C. 1983. Early arterial injury induces myointimal proliferation in canine pulmonary artery. Am. J. Vet. Res. 44, 181–186. Kim, S.Y., Park, H.Y., Lee, J.Y., Lee, Y.W. and Choi, H.J. 2019. Comparison of radiographic and echocardiographic features between small and large dogs with heartworm disease. J. Vet. Clin. 36(4), 207–211. Kitoh, K., Katoh, H., Kitagawa, H., Nagase, M., Sasaki, N. and Sasaki, Y. 2001. Role of histamine in heartworm extract-induced shock in dogs. Am. J. Vet. Res. 62(5), 770–774. Kitoh, K., Watoh, K., Chaya, K., Kitagawa, H. and Sasaki, Y. 1994b. Clinical, hematological, and biochemical findings in dogs after induction of shock by injection of heartworm extract. Am. J. Vet. Res. 55, 1535–1541. Kitoh, K., Watoh, K., Kitagawa, H. and Sasaki, Y. 1994a. Blood coagulopathy in dogs with shock induced by injection of heartworm extract. Am. J. Vet. Res. 55(11), 1542–1547. Kittleson, M.D. 1998. Heartworm infestation and disease (Dirofilariasis). In Small animal cardiovascular disease. Eds., Kittleson, M.D. and Kienle, R.D. Mosby, St. Louis, MI, pp: 370–401. Knight, D.H. 1980. Evolution of pulmonary artery disease in canine dirofilariasis: evaluation by blood pressure measurements and angiography. In the Proceedings of the 1980 Heartworm Symposium, Veterinary Medicine Publishing Co, Edwardsville, Kansas, pp: 55–62. Lamb, C.R., Tyler, M., Boswood, A., Skelly, B.J. and Cain, M. 2000. Assessment of the value of the vertebral heart scale in the radiographic diagnosis of cardiac disease in dogs. Vet. Rec. 146, 687–690. Lamb, C.R., Wikeley, H., Boswood, A. and Pfeiffer, D.U. 2001. Use of breed specific ranges for the vertebral heart scale as an aid to the radiographic diagnosis in dogs. Vet. Rec. 148, 707–711. Lehmkuhl, L.B., Bonagura, J.D., Biller, D.S. and Hartman, W.M. 1997. Radiographic evaluation of caudal vena cava size in dogs. Vet. Radiol. Ultrasound. 38(2), 94–100. Litster, A. and Buchanan, J. 2000. Vertebral scale system to measure heart size in radiographs of cats. J. Am. Vet. Med. Assoc. 216, 210–214. Lombard, C.W. and Ackerman, N. 1984. Right heart enlargement in heartworm-infected dogs: a radiohraphic, electrocardiographic, and echocardiographic correlation. Vet. Radiol. 25(5), 210–217. Losonsky, J.M., Thrall, D.E. and Lewis, R.E. 1983. Thoracic radiographic abnormalities in 200 dogs with spontaneous heartworm infestation. Vet. Radiol. 24(3), 120–123. Maerz, I. 2020. Clinical and diagnostic imaging findings in 37 rescued dogs with heartworm disease in Germany. Vet. Parasitol. 283, 109156; doi:10.1016/jvetpar.2020.109156 Marbella Fernández, D., García, V., Santana, A.J. and Montoya-Alonso, J.A. 2023a. The Thoracic inlet heart size, a new approach to radiographic cardiac measurement. Animals 13, 389; doi:10.3390/ani13030389 Marbella Fernández, D., García, V., Santana, A.J. and Montoya-Alonso, J.A. 2023b. The thoracic inlet length as a reference point to radiographically assess cardiac enlargement in dogs with myxomatous mitral valve disease. Animals 13, 2666; doi:10.3390/ani13162666 Marin, L.M., Brown, J., Mcbrien, C., Baumwart, R., Samii, V.F. and Couto, C.G. 2007. Vertebral heart size in retired racing greyhounds. Vet. Radiol. Ultrasound. 48, 332–334. Mostafa, A.A. and Berry, C.R. 2017. Radiographic assessment of the cardiac silhouette in clinically normal large- and small-breed dogs. Am. J. Vet. Res. 78(2), 168–177. Mostafa, A.A. and Berry, C.R. 2022. Radiographic vertical tracheal diameter assessment at different levels along the trachea as an alternative method for the evaluation of the tracheal diameter in non-brachycephalic small breed dogs. BMC. Vet. Res. 18, 61; doi:10.1186/s12917-022-03160-4 Mostafa, A.A., Peper, K.E. and Berry, C.R. 2020. Use of cardiac sphericity index and manubrium heart scores to assess radiographic cardiac silhouettes in large-and small-breed dogs with and without cardiac disease. J. Am. Vet. Med. Assoc. 256(8), 888–898. Olive, J., Javard, R., Specchi, S., Belanger, M., -C.., Belanger, C., Beauchamp, G. and Alexander, K. 2015. Effect of cardiac and respiratory cycles on vertebral heart score measured on fluoroscopic images of healthy dogs. J. Am. Vet. Med. Assoc. 246, 1091–1097. Owens, J.M. 1985. Radiology of the heart. In Manual of small animal cardiology, Eds. Tilley, L.P. and J.M. Owens. New York, NY: Churchill Livingstone, pp: 25–54. Pajas, A.M.G. and Acorda, J.A. 2018. Echocardiographic, electrocardiographic and thoracic radiographic findings in dogs with dirofilarioasis. Philipp. J. Vet. Med. 55(2), 71–84. Polizopoulou, Z.S., Koutinas, A.F., Saridomichelakis, M.N., Patsikas, M.N., Desiris, A.K., Roubies, N.A. and Leontidis, L.S. 2000. Clinical and laboratory observations in 91 dogs infected with Dirofilaria immitis in northern Greece. Vet. Rec. 146(16), 466–469. Rawlings, C.A., Keith, J.C., Lewis, R.E., Losonsky, J.M., McCall, J.W. 1983. Aspirin and prednisolone modification of radiographic changes caused by adulticide treatment in dogs with heartworm disease. J. Am. Vet. Med. Assoc. 182, 131–136. Schaub, R.G., Rawlings, C.A. and Keith, J.C. 1981. Platelet adhesion and intimal proliferation in canine pulmonary arteries. Am. J. Pathol. 104, 13–22. So, J., Chung, J., Je, M., Kang, K., Choi, J. and Yoon, J. 2024. Development and feasibility of new cardiac measurement method using vertebral heart area ratio in dogs. J. Vet. Med. Sci. 86(1), 28–34. Spasojević Kosić, Lj, Krstić, N., Trailović, D.D. 2007. Comparison of three methods of measuring vertebral heart size in German shepherd dogs. Acta Vet. 57(2-3), 133–141. Spasojević Kosić, Lj, Lalošević, L., Kozoderović, G., Vračar, V., Simin, S. and Potkonjak, A. 2024. Effectiveness of doxycycline/ivermectin therapy for heartworm disease in regard to Wolbachia status in dogs. J. Helenic. Vet. Med. Soc. 75(1), 7173–7180. Spasojević Kosić, Lj, and Lalošević, V. 2025. Radiographic diagnostic of the canine heartworm disease: old and new methods. In Proceedings of the 8th ESDA Days and 2025 Annual EVPC Scientific meeting, European Society of Dirofilariosis and Angiostrongylosis (ESDA), European Veterinary Parasitology College (EVPC), Thessaloniki, Greece, p 77. Spasojević Kosić, Lj, Lalošević, V., Naglić, A., Simin, S., Lj, K. and Spasović, A. 2016. Subjective and objective assessment of radiographic findings in dogs with heartworm disease. Parasit. Vectors 10(Suppl 1), 5. Suter, P.F. and Lord, P.F. 1984. Cardiac diseases. a text atlas of thoracic diseases of the dog and cat. Wettswil, Switzerland. Thrall, D.E. and Calvert, C.A. 1983. Radiographic evaluation of canine heartworm disease coexisting with right heart failure. Vet. Radiol. 24(3), 124–126. Torad, F.A. and Hassan, E.A. 2014. Two-dimensional cardiothoracic ratio for evaluation of cardiac size in German Shepherd Dogs. J. Vet. Cardiol. 16, 237–244. Tudor, N., Ionita, L., Tapaloaga, D., Tudor, P., Ionita, C. and Vlagioiu, C. 2014. Radiographic cardiopulmonary changes in dogs with heartworm disease. Rom. Biotechnol. Lett. 19(6), 9918–9924. Ware, W. 2011. Heartworm disease. In Cardiovascular disease in small animal disease. Ed., Ware, W London, Uk: Manson Publishing/The Veterinary Press, pp: 351–71. | ||
| How to Cite this Article |
| Pubmed Style Ljubica Spasojević Kosić. Feasibility and findings of different radiographic methods in diagnosing canine heartworm disease. Open Vet. J.. 2026; 16(1): 303-317. doi:10.5455/OVJ.2026.v16.i1.27 Web Style Ljubica Spasojević Kosić. Feasibility and findings of different radiographic methods in diagnosing canine heartworm disease. https://www.openveterinaryjournal.com/?mno=278894 [Access: January 31, 2026]. doi:10.5455/OVJ.2026.v16.i1.27 AMA (American Medical Association) Style Ljubica Spasojević Kosić. Feasibility and findings of different radiographic methods in diagnosing canine heartworm disease. Open Vet. J.. 2026; 16(1): 303-317. doi:10.5455/OVJ.2026.v16.i1.27 Vancouver/ICMJE Style Ljubica Spasojević Kosić. Feasibility and findings of different radiographic methods in diagnosing canine heartworm disease. Open Vet. J.. (2026), [cited January 31, 2026]; 16(1): 303-317. doi:10.5455/OVJ.2026.v16.i1.27 Harvard Style Ljubica Spasojević Kosić (2026) Feasibility and findings of different radiographic methods in diagnosing canine heartworm disease. Open Vet. J., 16 (1), 303-317. doi:10.5455/OVJ.2026.v16.i1.27 Turabian Style Ljubica Spasojević Kosić. 2026. Feasibility and findings of different radiographic methods in diagnosing canine heartworm disease. Open Veterinary Journal, 16 (1), 303-317. doi:10.5455/OVJ.2026.v16.i1.27 Chicago Style Ljubica Spasojević Kosić. "Feasibility and findings of different radiographic methods in diagnosing canine heartworm disease." Open Veterinary Journal 16 (2026), 303-317. doi:10.5455/OVJ.2026.v16.i1.27 MLA (The Modern Language Association) Style Ljubica Spasojević Kosić. "Feasibility and findings of different radiographic methods in diagnosing canine heartworm disease." Open Veterinary Journal 16.1 (2026), 303-317. Print. doi:10.5455/OVJ.2026.v16.i1.27 APA (American Psychological Association) Style Ljubica Spasojević Kosić (2026) Feasibility and findings of different radiographic methods in diagnosing canine heartworm disease. Open Veterinary Journal, 16 (1), 303-317. doi:10.5455/OVJ.2026.v16.i1.27 |