| Research Article | ||
Open Vet. J.. 2026; 16(2): 1141-1150
Open Veterinary Journal, (2026), Vol. 16(2): 1141-1150 Research Article Prevalence of high zoonotic-importance gastrointestinal helminth parasites and associated risk factors in synanthropic rodents in GreeceIoannis Tsakmakidis1, Androniki Tamvakis2, Ioannis A. Giantsis3 and Anastasia Diakou41School of Agricultural Sciences, Department of Agriculture, University of Western Macedonia, Florina, Greece 2Laboratory of Ecology and System Dynamics, Department of Marine Sciences, University of the Aegean, Mytilene, Greece 3Department of Animal Science, Faculty of Agriculture, Forestry and Natural Environment, Aristotle University of Thessaloniki, Thessaloniki, Greece 4Laboratory of Parasitology and Parasitic Diseases, School of Veterinary Medicine, Faculty of Health Sciences, Aristotle University of Thessaloniki, Thessaloniki, Greece *Corresponding Author: Ioannis Tsakmakidis. School of Agricultural Sciences, Department of Agriculture, University of Western Macedonia, Florina, Greece. Email:gtsakmakidis [at] yahoo.gr Submitted: 21/08/2025 Revised: 11/01/2026 Accepted: 30/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
AbstractBackground: Rodents are a group of animals that are important from a veterinary and public health perspective, as they harbor various pathogens. Among them, gastrointestinal helminths have attracted significant scientific attention in recent years, as some of them can be transmitted to humans, supporting the role of rodents in maintaining and spreading zoonotic pathogens. Information on gastrointestinal helminth infections in rodents in Greece is limited. Aim: The present study aimed to determine the prevalence of gastrointestinal helminthic infection in synanthropic rodent species in Northern Greece, investigate potential factors associated with these infections, and assess the relevance of rodents as hosts of zoonotic parasites. Methods: The investigated areas included urban, semi-urban, and rural habitats. Fecal samples were collected from 100 rodents belonging to three species (68 Mus musculus, 21 Rattus norvegicus, and 11 Rattus rattus) from four regional units in Northern Greece. All fecal samples were examined using standard parasitological methods. Results: Copromicroscopic examination revealed gastrointestinal helminths in 58% of the rodents. The parasites and prevalence were as follows: the nematodes Aspiculuris tetraptera (1%), Capillaria spp. (6%), Heterakis spumosa (20%), Nippostrongylus brasiliensis (20%), Strongyloides ratti (6%), Syphacia spp. (1%), and Trichuris muris (4%); and the zoonotic cestode species Hymenolepis diminuta (21%), and Hymenolepis nana (4%). The prevalence of parasites was significantly associated with habitat, regional unit, season, and body condition. Conclusion: The findings of this study demonstrate the presence of helminth parasitic pathogens in synanthropic rodents in different environments and a strong association with the area of origin, with a particularly high prevalence in an important summer destination. Keywords: Gastrointestinal helminths, Greece, Rodents, Zoonotic. IntroductionRodents represent one of the largest and most successful groups of animals in terms of survival and disease resistance. They are known for their tolerance toward diverse environmental conditions, with approximately 2,550 known species representing nearly 40% of all mammal species worldwide (Burgin et al., 2018). Their global distribution, high reproductive rates, and adaptability to various environments make them a vital component of many terrestrial ecosystems (Zhang et al., 2021). Rodents provide various beneficial services in nature, such as serving as a primary food source for several carnivores and contributing to insect control (Witmer, 2022). Additionally, rodents act as ecological engineers, as their presence at low to moderate densities can positively influence grassland dynamics (Hilal et al., 2024). However, some rodent species are considered agricultural pests due to their negative impact on crop production and their tendency to damage stored food products (Akhtar et al., 2023; Stenseth et al., 2003). These mammalian species have an impact on public health that lies largely in their role as carriers or reservoir hosts for over 60 zoonotic pathogens (Dahmana et al., 2020). These infectious agents can be transmitted to humans through various routes, including direct contact with rodents, food and water contamination by rodent feces or urine, and arthropod vectors (Hamidi, 2018). Several factors influence the prevalence of rodent-born zoonoses, including rodent population, control measures, habitat diversity and quality, human lifestyle, wars, and climate change (Akhtar et al., 2023). Zoonotic diseases caused by various bacteria (e.g., salmonellosis, tuberculosis, plague, leptospirosis, and Lyme disease), viruses (e.g., Hantavirus diseases, tick-borne encephalitis, and Lassa fever), protozoans (e.g., leishmaniosis and toxoplasmosis), and helminths (e.g., hymenolepiasis and trichinellosis) are among the zoonotic diseases of great public health importance (Ibarra-Cerdeña et al., 2024). In particular, gastrointestinal helminthic parasitoses have garnered increasing attention due to the increasing number of reported cases in rodent species across various regions of the world. Such cases include infection with Hymenolepis nana, Hymenolepis diminuta, and Moniliformis spp. (Lima et al., 2021; Hamzavi et al., 2024). Factors potentially contributing to the rise in such reports include human overpopulation, large-scale migration, and habitat modification (Chomel et al., 2007). Furthermore, landscape fragmentation, land use changes, and activities that increase interactions between humans and wildlife, such as ecotourism and hunting, can bring rodents into closer contact with people, thereby facilitating the transmission of parasites to humans (Rodriguez-Morales et al., 2025). Parasitological studies on rodents are a crucial step in preventing the transmission of parasitic diseases to humans and domestic animals, given the medical and veterinary significance of rodents. Such studies also support the development and implementation of effective rodent population control programs (Tijjani et al., 2020). Among the synanthropic rodent species, Mus musculus, Rattus norvegicus, and Rattus rattus inhabit urban, semi-urban, and rural habitats. These species are responsible for most reported cases of zoonotic parasitic infections (Landaeta-Aqueveque et al., 2021). Their impact is related to significant economic losses and considerable risks for human health, and it is largely attributed to characteristics such as high reproductive rates, genetic variability, rapid adaptation to both natural and anthropogenic habitats, and diverse eating behaviors (Ecke et al., 2022). Gastrointestinal helminthic infections are among the most prevalent zoonotic parasitic infections worldwide, causing malnutrition, weight loss, abdominal pain, vomiting, and diarrhea, with more severe clinical manifestations in children, the elderly, and immunocompromised patients (Horton, 2003). However, information on gastrointestinal helminths in rodents is scarce in many countries, including Greece. This study aimed to determine the prevalence of gastrointestinal helminthic infection in synanthropic rodents, investigate the ecological and biological factors that may influence these infections, and assess the relevance of these rodent species as hosts of zoonotic parasites in Greece. Therefore, prevalence rates were estimated and compared in four neighboring regional units (i.e., administrative divisions of Greece) of Northern Greece, including various habitats, along with agricultural, forest, and high residential areas, as well as a particularly high residential density area during summer months, which is less populated from October to May. Materials and MethodsStudy area and rodent collectionBetween December 2020 and November 2022, fecal samples were collected from 100 dead rodents, representing three species (68 M. musculus, 21 R. norvegicus, and 11 R. rattus) and inhabiting three habitat types: urban (areas with more than 10,000 inhabitants), semi-urban (2,000 to 10,000 inhabitants), and rural (fewer than 2,000 inhabitants). The rodents were killed during professional pest control operations. Samples were collected on-site, without necropsy, by pest control companies across four regional units of Northern Greece: Chalkidiki, Imathia, Kilkis, and Thessaloniki (Fig. 1). Identification of rodent species was performed based on the following characteristics: color, body length, tail length, shape of the ear lobes, shape of the eyes, and position and shape of the nose and face (FAO, 1994). Information regarding the date and area of collection, sex, and body condition of each animal was recorded. All animal samples were obtained from pest control application companies. No endangered species that would require a further license were included. Collection and examination of samplesAfter collection, the samples were promptly transported in a polystyrene container at 2°C–4°C to the Laboratory of Parasitology and Parasitic Diseases at the School of Veterinary Medicine, Aristotle University of Thessaloniki. All fecal samples were examined using standard parasitological methods, i.e., the combined sedimentation-zinc sulfate flotation method and the ether-formalin sedimentation method (Faust et al., 1938; Taylor et al., 2016), with minor modifications. For the flotation method, approximately 1 g of fecal material was mixed with tap water and strained through a No. 150 sieve in a centrifuge tube. The sample was centrifuged at 200 × g for 3 minutes, after which the supernatant was decanted until approximately 1 cm liquid remained above the sediment. A 33.2% (w/v) zinc sulfate solution with a specific gravity of 1.18 was then added to the sediment. After thorough mixing, more zinc sulfate solution was added until the tube was filled to the top, and a coverslip was placed over it. The tube was centrifuged again at 150 × g for 1 minute using a swing-out rotor to keep the coverslip in position. The coverslip was then gently lifted, mounted on a glass slide, and examined under a light microscope. For the sedimentation method, approximately 1 g of fecal material was mixed with a formalin/glycerol solution and strained through a No. 150 sieve in a centrifuge tube. Then, 5 ml of ether was added, and the mixture was thoroughly homogenized by vigorous shaking. The sample was centrifuged at 200 × g for 3 minutes, after which all layers except the sediment were carefully discarded. A few drops of the remaining sediment were placed on a microscope slide, covered with a coverslip, and examined.
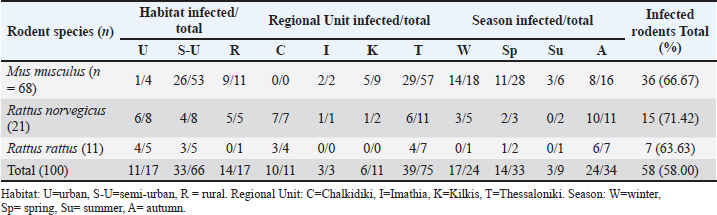
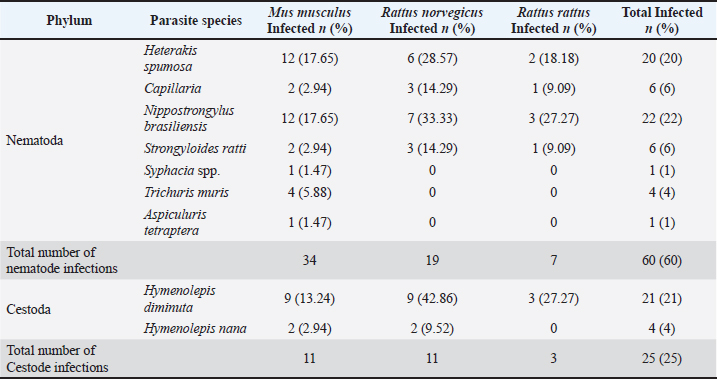
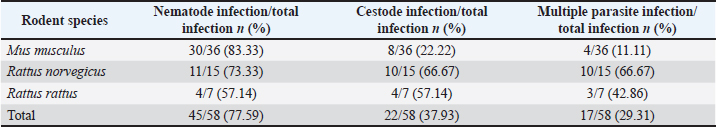
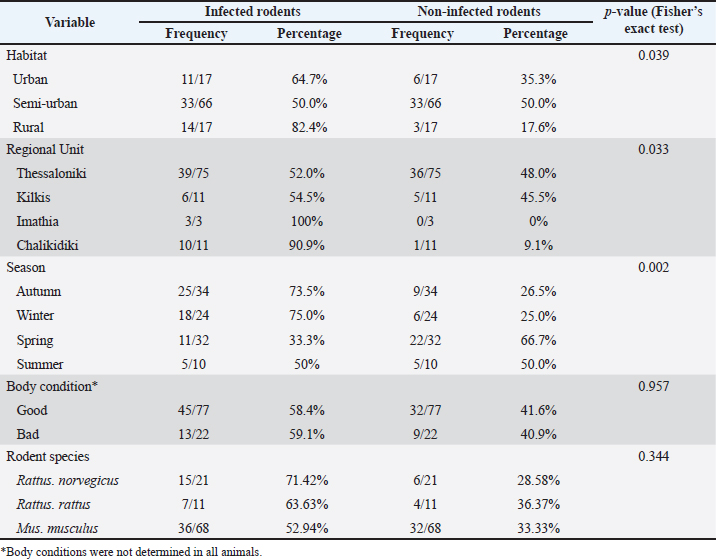
Fig. 1. The four Regional Units in Central Macedonia, Greece, where sampling was conducted. The morphological and morphometric characteristics of helminth eggs were identified under light microscopy (Taylor et al., 2016). Statistical analysisThe association between parasite infection and various factors—including rodent species, season, region, habitat, and body condition—was assessed using Fisher’s exact test (for small sample sizes). The same statistical test was used to examine the relationship between specific parasite species and the aforementioned parameters. A p-value of < 0.05 was considered statistically significant. All statistical analyses were performed using the R statistical software (R Core Team, 2021). Ethical approvalNot needed for this study. ResultsSample examinationOf the total number (100), 66 rodents were sampled from semi-urban, 17 from urban, and 17 from rural habitats. Seventy-seven were in good body condition (body condition scores 3 and 4), 22 were in poor (body condition scores 1 and 2), and in 1 animal, the body condition was not determined according to the species parameters (Ullman-Culleré and Foltz, 1999). Male-to-female analogies were 38/30, 6/13, and 5/5 for M. musculus, R. norvegicus, and R. rattus, respectively. Overall, 58 (58%) of the 100 examined rodents were infected with gastrointestinal helminths. At the rodent species level, 36 (52.94%) of the 68 M. musculus, 15 (71.42%) of the 21 R. norvegicus, and 7 (63.63%) of the 11 R. rattus were infected. Regarding the collection habitat, 33 out of 66, 14 out of 17, and 11 out of 17 were infected in semi-urban, rural, and urban habitats, respectively (Table 1). The helminth parasites included seven nematodes (Aspiculuris tetraptera, Capillaria spp., Heterakis spumosa, Nippostrongylus brasiliensis, Strongyloides ratti, Syphacia spp., and Trichuris muris) and two cestode species (H. diminuta and H. nana). The most prevalent species were H. diminuta (21%), H. spumosa (20%), and N. brasiliensis (20%) (Table 2). Of the 58 infected animals, 45 (77.59%) were parasitized by nematodes and 22 (37.93%) by cestode species. More specifically, 30 (83.33%) and 8 (22.22%) of 36 M. musculus were infected with nematodes and with cestodes, respectively, whereas 11 (73.33%) and 10 (66.67%) of 15 R. norvegicus were infected with nematodes and with cestodes, respectively, and 4 (57.14%) and 4 (57.14%) of 7 R. rattus were infected with nematodes and with cestodes, respectively. Multiple parasitic infections occurred in 17/58 (29.31%) rodents, with up to 4 species per animal in different combinations (Table 3). Table 1. Infected rodents in relation to species, habitat, regional unit, and season.
Table 2. Infection rates of helminth parasites in examined rodents.
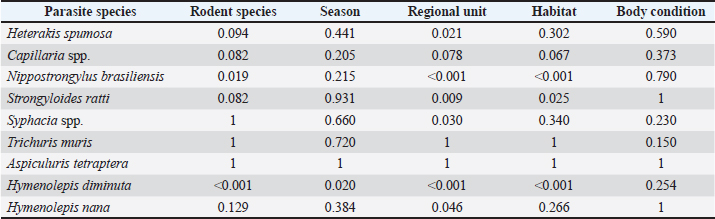
Statistical analysisTable 4 presents the independence analysis between parasite prevalence and various measured factors. Sex was not included in the statistical analysis because the sample sizes per sex and per rodent species were insufficient for reliable conclusions. The most significant factor affecting the prevalence of parasites was the Regional Unit. The prevalence of H. spumosa, N. brasiliensis, S. ratti, Syphacia spp., H. diminuta, and H. nana was statistically associated with the geographical region in which the animals were found (p-value < 0.05). A significant association was also observed between habitat type and the prevalence of N. brasiliensis, S. ratti, and H. diminuta (p-value < 0.05), indicating that these parasite species are more likely to infect rodents from urban areas. Moreover, the prevalence of N. brasiliensis and H. diminuta significantly varied across rodent species (p-value < 0.05), with R. norvegicus showing the highest infection rates (33.33% and 42.86%, respectively), followed by R. rattus (27.27% for both parasites) and M. musculus (17.65% and 13.24%). Finally, the prevalence of H. diminuta exhibited significant seasonal variation (p-value < 0.05). Statistical analysis revealed that several variables were significantly associated with the prevalence of parasite infection among rodents (Table 5). Habitat, regional unit, and season showed statistically significant correlations with infection status (p < 0.05). Rodents from rural habitats exhibited the highest infection rate (82.4%), followed by urban (64.7%) and semi-urban (50.0%) habitats (p=0.039). Significant geographical variation was also observed. Infection rates were markedly higher in rats from Imathia (100%) and Chalkidiki (90.9%) than in those from Thessaloniki (52.0%) and Kilkis (54.5%) (p=0.033). Seasonal differences were evident, with the highest prevalence recorded during autumn (73.5%) and winter (75.0%), while infection rates dropped considerably in spring (33.3%) and were moderate in summer (50.0%) (p=0.002). On the other hand, body condition was not associated with infection (p=0.957), with rodents in both "good" and "bad" condition showing similar infection prevalence (58.4% and 59.1%, respectively). No statistically significant association was found with rodent species (p=0.344); however, R. norvegicus had a slightly higher prevalence (71.42%) than M. musculus (52.94%) and R. rattus (63.63%). Table 3. Occurrence of nematodes, cestodes, and multiple parasite infections in infected rodents.
Table 4. Results (p-values) of Fisher’s exact test showing the independence of the parasites’ species prevalence against the factors measured in the rodent samples.
DiscussionIn this study, a high prevalence of helminth infection was recorded in two zoonotic cestode species in rodents from Northern Greece. These findings highlight the potential role of commensal rodents in spreading zoonotic parasitoses in synanthropic environments. Similar investigations have been carried out across the world, including Bangladesh (Fuehrer et al., 2012), Brazil (Lima et al., 2021), Cambodia (Chaisiri et al., 2017), France (Desvars-Larrive et al., 2017), Hungary (Juhász et al., 2024), Iran (Mohtasebi et al., 2020), Italy (Milazzo et al., 2010), Malaysia (Tijjani et al., 2020), Nigeria (Egbunu and Dada, 2016), Serbia (Kataranovski et al., 2011), Spain (Galán-Puchades et al., 2018), Thailand (Ribas et al., 2016), the Netherlands (Franssen et al., 2016), and the United Kingdom (McGarry et al., 2015). Reported infection levels differed substantially, likely reflecting variation in rodent species, environmental conditions, and geographic settings. A high prevalence of R. norvegicus has been consistently documented. Rates of 85% in Spain (Galán-Puchades et al., 2018), 98.6% in Italy (Milazzo et al., 2010), 87.5% in France (Desvars-Larrive et al., 2017), and 100% in the United Kingdom (McGarry et al., 2015) mirror the elevated prevalence observed in the present study (71.42%). Similarly, multispecies surveys show substantial variation: Brazil reported 73.6% infection across five rodent species (Lima et al., 2021), while Thailand recorded 89.8% infection in R. norvegicus and R. rattus from wet markets (Ribas et al., 2016). In contrast, Malaysia reported a lower prevalence of 17.1% in the same two species (Tijjani et al., 2020), indicating the strong effects of local habitat and host composition. A previous investigation in Greece (Founda, 1992) identified several of the same gastrointestinal helminths recorded in the present study, along with Mathevotaenia symmetrica, Mastophorus muris, and Moniliformis moniliformis. Infection levels did not always align with those found in this study, likely due to differences in diagnostic approach, sample size, and host composition. Table 5. Infection of examined rodents in correlation with variable factors.
Among the species examined, R. norvegicus showed the highest infection rate (71.42%), likely reflecting several features of this synanthropic murid: its high reproductive capacity, omnivorous feeding habits, broad environmental tolerance, and tendency to occur at high population densities that promote close contact among individuals (Battersby et al., 2008; Munshi-South et al., 2024). The results of this study showed a positive correlation between the presence of parasites in both urban and rural habitats. A possible explanation is that garbage accumulates in urban habitats, and these animals usually utilize kitchen waste and sewage, a factor that possibly favors their proliferation and survival in large populations and high densities, thereby enhancing the transmission and prevalence of parasites (Battersby et al., 2008). Infection with helminths that have a life cycle with one or more intermediate hosts, usually an arthropod (i.e., H. diminuta), is probably favored in rural habitats (Anders et al., 2019). Moreover, food crops attract rodents in rural habitats, and people engaged in agricultural activities have an elevated risk of close contact with rodents near or in their houses, which could promote parasite transmission (Suwannarong and Chapman, 2015). Analysis by the Regional Unit revealed higher parasite infection rates in Chalkidiki and Imathia than in Kilkis and Thessaloniki. Such regional differences in infection rates have also been observed in previous studies, often linked to variations in climatic factors, such as temperature and humidity, as well as landscape features, including vegetation. Rainfall, moderate temperatures, resource-rich landscapes (crop fragments), and rich vegetation have been reported to favor helminth infections (Froeschke and Matthee, 2014; Hamzavi et al., 2024). Furthermore, landscapes closely associated with humans (livestock and urban landscapes) pose a greater risk for zoonotic species (Froeschke and Matthee, 2014). Interestingly, the Regional Unit of Chalkidiki showed a significant correlation with high prevalence rates. Chalkidiki, although composed mostly of semi-urban and rural habitats, presents some unique characteristics as a tourist destination, such as overcrowding during summer (Krabokoukis et al., 2021). Consequently, garbage accumulation is more intense during tourist periods than in classic urban environments, a condition that could attract rodents and favor high rodent population densities. This study revealed a seasonal variation in the prevalence of H. diminuta, with higher rates during autumn. Season is a factor that influences parasitic infection in various hosts (Altizer et al., 2006). Seasonal changes in rainfall patterns, environmental temperature, and humidity can influence the intensity and prevalence of parasitic infections in various ways, including altering host susceptibility, host exposure, and parasite development and survival (Islam et al., 2017; Shearer and Ezenwa, 2020). Seasonal variation in H. diminuta prevalence was also reported in a study in Pakistan, where higher rates were recorded in summer. According to the researchers, this was because various foodstuffs were stored during the summer, attracting rodents that roam in search of food. As a result, the risk of parasitic infections was relatively higher in this season than in other seasons (Ahmad et al., 2014). In contrast, in a study in India, season did not have any statistically significant effect on H. diminuta infections in rodents (Brar et al., 2021). Body condition was not associated with parasitism in this study. Previous research has reported both positive and negative associations, suggesting that factors such as infection intensity (number of parasites), parasite species, and individual host traits likely play a decisive role in this relationship (Grandón-Ojeda et al., 2022). In the present study, infection with multiple helminthic species was recorded, an observation that has also been reported in other studies, confirming that high parasite species richness in hosts is the rule and not an exception (Porta et al., 2014; Desvars-Larrive et al., 2017; Islam et al., 2020; Dini et al., 2024). Polyparasitism is a common phenomenon in wild animals and emphasizes the ability of rodents to endure and survive invasion by various parasitic species (Bordes and Morand, 2011; Porta et al., 2014). The cestodes H. nana and H. diminuta found in the present study have been previously reported among the frequent zoonotic helminths in rodents, in various rates of infection and in different areas of the world, such as Brazil, Cambodia, France, Guatemala, Hungary, Iran, Italy, Malaysia, Mexico, the Netherlands, Serbia, and Spain (Milazzo et al., 2010; Kataranovski et al., 2011; Meshkekar et al., 2014; Chaisiri et al., 2017; Desvars-Larrive et al., 2017; Galán-Puchades et al., 2018; Hernández et al., 2020; Islam et al., 2020; Panti-May et al., 2020a; Lima et al., 2021; Mohd-Qawiem et al., 2022; Hamzavi et al., 2024; Juhász et al., 2024). In Greece, so far, there are no reported clinical cases of Hymenolepis spp. infection in humans (Mijatović et al., 2024). However, the parasite has been found in an epidemiological study conducted on children, and there is also a report of H. diminuta infection in a dog in the Greek islands (Diakou et al., 2019; Topouzis, 2022). Hymenolepiasis, caused by the rodent cestode species H. nana and H. diminuta, is probably considered the most prevalent human cestodiasis worldwide, as it is estimated that 50–75 million humans suffer from this parasitic disease (Aguilar-Marcelino et al., 2023). This infection is more common in children in poor and crowded communities and areas with poor sanitation and poor health, structural, and socio-environmental conditions, where rodents are in close contact with people (Sungkar et al., 2017). In fact, H. nana, also known as the "dwarf tapeworm," is an exception among cestodes, as it can complete a direct life cycle and be directly transmitted from rodents to humans via the oral-fecal route. Hymenolepis diminuta, known as the “rat tapeworm,” has an indirect life cycle, with an arthropod serving as an intermediate host. Most of these infections are typically asymptomatic (Aguilar-Marcelino et al., 2023). However, clinical manifestations have been reported, including fever, diarrhea, abdominal pain, loss of appetite, pruritus, and reduced growth (Panti-May et al., 2020b; Coello Peralta et al., 2023). This study provides valuable insights into the occurrence of gastrointestinal helminths in commensal rodents; however, certain limitations should be acknowledged. Although the overall number of rodents examined was adequate for a preliminary parasitological investigation, the distribution of samples among species, seasons, and RUs was not entirely balanced. The relatively small number of R. rattus specimens limits the robustness of interspecific comparisons. In addition, rodents were examined only through fecal analysis, and necropsy was not performed; incorporating necropsy would likely have allowed the detection of a greater diversity of parasite species. These aspects may have reduced the statistical power of some analyses and should be considered when interpreting the results. Nevertheless, the total sample size and the findings provide important preliminary parasitological data and highlight patterns that may guide future research on the role of rodents as zoonotic parasite reservoirs. Because these parasites can be transmitted to humans via contaminated soil, water, food, or poor sanitation, high rodent densities and close proximity to humans may increase the risk of transmission. This highlights the public health relevance of the present findings, particularly as these helminth species were detected in rodents inhabiting a major summer tourist destination (Aguilar-Marcelino et al., 2023). Human activities have modified natural environments, creating conditions that may bring rodents into closer contact with humans. These species can act as ‟bridge hosts,” transferring parasites and other pathogens across habitats, especially in densely populated urban areas, and often represent dominant mammals in human-altered environments. ConclusionParasitism by nine gastrointestinal helminth taxa was documented in the rodent species R. rattus, R. norvegicus, and M. musculus collected from Northern Greece. The prevalence rates and their potential associations with ecological and biological risk factors were assessed. The findings confirm that synanthropic rodent populations in the study area host a range of gastrointestinal helminths and reveal significant statistical correlations with several epidemiological factors, including a strong association between infection rates and sampling in a major tourist destination. Although the small sample size of R. rattus limits the statistical power of analyses for this species, the overall results underscore the importance of gastrointestinal helminths in rodents, some of which are transmissible to humans. These findings highlight the role of rodents in maintaining and disseminating zoonotic pathogens, which may pose a public health risk. Regular monitoring of rodent populations and population control measures, such as improved waste management, enhanced hygiene practices, and appropriate use of biocide in human settlements, are essential for preventing zoonotic diseases. AcknowledgmentsNone. Conflict of interestThe authors declare that the research was conducted without any commercial or financial relationships that could be understood as a potential conflict of interest. FundingThis research received no specific grant. Authors' contributionsConceptualization: I. T. and A. D. Methodology: I. T., A. D, A. T., and I. A. G. Formal analysis and investigation: I. T. and I. A. G. Writing—preparation of the original draft: I. T. Writing, review, and editing: A. D, A.T., I.A.G. Resources: A. D. Supervision: A. D. Data availabilityAll data supporting this study’s findings are available within the manuscript. ReferencesAguilar-Marcelino, L., Aguilar-Figueroa, B.R., Oropeza-Guzmán, G., Mendoza-Galvez, B., Bautista-Garfias, C.R. and Viladomat, G.R.C. 2023. Hymenolepiasis.In One Health Triad. Aguilar-Marcelino, L., Younus, M., Khan, A., Saeed, N.M. and Abbas, R.Z Unique Scientific Publishers, Faisalabad, Pakistan, pp: 122–7. Ahmad, M., Maqbool, A., Anjum, A.A., Ahmad, N., Khan, M.R., Sultana, R. and Ali, M.A. 2014. Occurrence of Hymenolepis diminuta in rats and mice captured from urban localities of Lahore, Pakistan. J. Anim. Plant. Sci. 24, 392–396. Akhtar, N., Hayee, S., Idnan, M., Nawaz, F. and BiBi, S. 2023. Human zoonotic pathogen transmission in rats: historical background and future prospects.In Rodents and Their Role in Ecology, Medicine and Agriculture. Ed.. and Manjur Shah, M. London, England, IntechOpen Publisher, pp: 1–8. Altizer, S., Dobson, A., Hosseini, P., Hudson, P., Pascual, M. and Rohani, P. 2006. Seasonality and infectious disease dynamics. Ecological. Lett. 9, 467–484. Anders, J.L., Nakao, M., Uchida, K., Ayer, C.G., Asakawa, M. and Koizumi, I. 2019. Comparison of the intestinal helminth community of the large Japanese field mouse (Apodemus speciosus) between urban, rural, and natural sites in Hokkaido, Japan. Parasitology Int. 70, 51–57. Battersby, S., Hirschorn, R.B. and Amman, B.R. 2008. Commensal rodents.In Public health significance of urban pest. Bonnefoy, X., Kampen, H. and Sweeney, K World Health Organization, Geneva, Switzerland, pp: 387–419. Bordes. and Morand. 2011. Impact of multiple infections on wild animal hosts: a review. Infect. Ecol. Epidemiol. 1, 1. Brar, S.K., Singla, N. and Singla, L.D. 2021. A Comparative Comprehensive Analysis of Natural Infections of Hymenolepis Diminuta and Hymenolepis Nana in Commensal Rodents. Helminthologic 58, 248–262. Burgin, C.J., Colella, J.P., Kahn, P.L. and Upham, N.S. 2018. How many species of mammals are there?. J. Mammal. 99, 1–14. Chaisiri, K., Chou, M., Siew, C.C., Morand, S. and Ribas, A. 2017. Gastrointestinal helminth fauna of rodents from Cambodia: emphasizing the community ecology of host–parasite associations. J. Helminthol. 91, 726–738. Chomel, B.B., Belotto, A. and Meslin, F.X. 2007. Wildlife, exotic pets, and emerging zoonoses. Infect. Dis. 13, 6–11. Coello Peralta, R.D., Salazar Mazamba, M.D.L., Pazmiño Gómez, B.J., Cushicóndor Collaguazo, D.M., Gómez Landires, E.A. and Ramallo, G. 2023. Hymenolepiasis Caused by Hymenolepis nana in Humans and Natural Infection in Rodents in a Marginal Urban Sector of Guayaquil, Ecuador. Am. J. Case Rep. 24, 939476; e939476 Dahmana, H., Granjon, L., Diagne, C., Davoust, B., Fenollar, F. and Mediannikov, O. 2020. Rodents as host of pathogens and related zoonotic disease risk. Pathogens 9, 202. Desvars-Larrive, A., Pascal, M., Gasqui, P., Cosson, J.F., Benoît, E., Lattard, V., Crespin, L., Lorvelec, O., Pisanu, B., Teynié, A., Vayssier-Taussat, M., Bonnet, S., Marianneau, P., Lacôte, S., Bourhy, P., Berny, P., Pavio, N., Le Poder, S., Gilot-Fromont, E., Jourdain, E., Hammed, A., Fourel, I., Chikh, F. and Vourc’H, G. 2017. Population genetics, community of parasites, and resistance to rodenticides in an urban brown rat (Rattus norvegicus) population. PLos One 12, e0184015; doi:10.1371/journal.pone.0184015 Diakou, A., Di Cesare, A., Morelli, S., Colombo, M., Halos, L., Simonato, G., Tamvakis, A., Beugnet, F., Paoletti, B. and Traversa, D. 2019. Endoparasites and vector-borne pathogens in dogs from Greek islands: pathogen distribution and zoonotic implications. PLos Negl. Trop. Dis. 13, 7003. Dini, F.M., Mazzoni Tondi, C. and Galuppi, R. 2024. Helminthofauna Diversity in Synanthropic Rodents of the Emilia-Romagna Region (Italy): implications for Public Health and Rodent Control. Vet. Sci. 11, 585. Ecke, F., Han, B.A., Hörnfeldt, B., Khalil, H., Magnusson, M., Singh, N.J. and Ostfeld, R.S. 2022. Population fluctuations and synanthropy explain transmission risk in rodent-borne zoonoses. Nat. Commun. 13, 7532. Egbunu, A.A. and Dada, E.O. 2016. Prevalence of Intestinal Helminth Parasites of Domestic Rats in Selected Sites Around Students Hall of Residence in the Federal University of Technology, Akure, Nigeria. Int. J. Curr. Microbiol. App. Sci. 5, 918–923. Fao. 1994. Rodent species of importance post-harvest.In Grain Storage Techniques. Proctor, D.L FAO, Rome, Italy. Faust, E.C., D’Antoni, J.S., Odom, V., Miller, M.J., Peres, C., Sawitz, W., Thomen, L.F., Tobie, L.E. and Walker, J.H. 1938. Clinical laboratory techniques for diagnosing protozoan cysts and helminth eggs in feces: a critical study. Am. J. Trop. Med. Hyg. 18, 169–183. Founda, I 1992. Contribution to the study of the metazoan parasites of rats and mice and their public health importance. Ph.D. thesis, Aristotle University of Thessaloniki. Franssen, F., Swart, A., Van Knapen, F. and Van Der Giessen, J. 2016. Helminth parasites in black rats (Rattus rattus) and brown rats (Rattus norvegicus) from different environments in the Netherlands. Infect. Ecol. Epidemiol. 17, 31413. Froeschke, G. and Matthee, S. 2014. Landscape characteristics influence helminth infestations in a peri-domestic rodent: implications for possible zoonotic disease. Parasites. &. Vectors. 7, 393. Fuehrer, H.P., Baumann, T.A., Riedl, J., Treiber, M., Igel, P., Swoboda, P., Joachim, A. and Noedl, H. 2012. Rodent endoparasites from the Chittagong Hill Tracts in southeastern Bangladesh. Wien. Klin. Wochenschr. 3, 27–30. Galán-Puchades, M.T., Sanxis-Furió, J., Pascual, J., Bueno-Marí, R., Franco, S., Peracho, V., Montalvo, T. and Fuentes, M.V. 2018. First survey on zoonotic helminthosis in urban brown rats (Rattus norvegicus) in Spain and associated public health considerations. Parasitol 15, 49–52. Grandón-Ojeda, A., Moreno, L., Garcés-Tapia, C., Figueroa-Sandoval, F., Beltrán-Venegas, J., Serrano-Reyes, J., Bustamante-Garrido, B., Lobos-Chávez, F., Espinoza-Rojas, H., Silva-de La Fuente, M.C., Henríquez, A. and Landaeta-Aqueveque, C. 2022. Gastrointestinal Helminth Infection Patterns in Rattus rattus, Rattus norvegicus, and Mus musculus in Chile. Front. Vet. Sci. 9, 929208. Hamidi. and K. 2018. How do Rodents Play Role in the Transmission of Foodborne Diseases?. Nutr. Food. Sci. Int. J. 6, 1–4. Hamzavi, Y., Khodayari A Davari., Shiee SA Karamati., Raeghi H Jabarmanesh., Bashiri. and Bozorgomid. 2024. A systematic review and meta-analysis on the prevalence of gastrointestinal helminthic infections in Iranian rodents: an emphasis on zoonotic aspects. Helicon 10, e31955. Hernández, W.C., Morán, D., Villatoro, F., Rodríguez, M. and Álvarez, D. 2020. Zoonotic Gastrointestinal Helminths in Rodent Communities in Southern Guatemala. J. Parasitol. 106, 341–345. Hilal, M.G., Ji, C., Li, Y., Tang, K., Li, H., Liu, X., Lin, K. and Wang, D. 2024. Deciphering the role of rodents in grassland degradation: a review. J. Environ. Manage. 370, 122618. Horton. and J. 2003. Human gastrointestinal helminth infections: are they now neglected diseases?. Trends. Parasitol. 19, 527–531. Ibarra-Cerdeña, C.N., Rodríguez-Luna, C.R., Palomo-Arjona, E.E., Ibarra-López, M.P., Barber, M.F. and Dirzo, R. 2024. Rodents as key hosts of zoonotic pathogens and parasites in the neotropics.In Ecology of Wildlife Diseases in the Neotropics. Acosta-Jamett, G. and Chaves, A Springer International Publishing, pp: 143–84. Islam, M., Hossain, M., Dey, A., Alim, M., Akter, S. and Alam, M. 2017. Epidemiology of gastrointestinal parasites of small ruminants in Mymensingh, Bangladesh. J. A. V. A. R. 4, 356–362. Islam, M.M., Farag, E., Hassan, M.M., Bansal, D., Awaidy, S.A., Abubakar, A., Al-Romaihi, H. and Mkhize-Kwitshana, Z. 2020. Helminth Parasites among Rodents in Middle Eastern Countries: a Systematic Review and Meta-Analysis. Animals 10, 2342. Juhász A, Tóth T, Eldridge, C.J.L. and Majoros, G. 2024 The first survey of endoparasite infection in the brown rat (Rattus norvegicus) from a synanthropic environment in Hungary. Vet. 2024; 58: 69-79. Parasitol. Reg. Stud. Reports. 56, 101141. Kataranovski, M., Mirkov, I., Belij, S., Popov, A., Petrovic, Z., Gaci, Z. and Kataranovski, D. 2011. Intestinal helminth infection of rats (Ratus norvegicus) in the Belgrade area: the effect of sex, age, and habitat. Parasite 18, 189–196. Krabokoukis, T., Tsiotas, D. and Polyzos, S. Examining the Relationship Between Tourism Seasonality and Saturation in Greek Prefectures: a Combined Operational and Theoretical Approach. In Proceedings of 2021 in Business and Economics Conference, Cham, Switzerland, 2021, pp 171–184. Landaeta-Aqueveque, C., Moreno Salas, L., Henríquez, A., Silva-de La Fuente, M.C. and González-Acuña, D. 2021. Parasites of Native and Invasive Rodents in Chile: ecological and Human Health Needs. Front. Vet. Sci. 11, 643742. Lima, V.F.S., Ramos, R.A.N., Giannelli, A., Andrade, W.W.A., López, I.Y.T., Ramos, I.C.D.N., Rinaldi, L., Cringoli, G. and Alves, L.C. 2021. Occurrence of zoonotic gastrointestinal parasites of rodents and the risk of human infection in different biomes of Brazil. Braz. J. Vet. Med. 43, 113820; e113820 Mcgarry, J.W., Higgins, A., White, N.G., Pounder, K.C. and Hetzel, U. 2015. Zoonotic helminths of urban brown rats (Rattus norvegicus) in the UK: neglected public health considerations?. Zoonoses. Public. Health. 62, 44–52; doi:10.1016/j.zoph.2016.09.010 Meshkekar, M., Sadraei, J., Mahmoodzadeh, A. and Mobedi, I. 2014. Helminth Infections in Rattus ratus and Rattus norvegicus in Tehran, Iran. Iran. J. Parasitol. 9, 548–552. Mijatović, S., Štajner, T., Čalovski, I.C., Dubljanin, E., Bobić, B., Leković, Z., Barać, A. and Džamić, A.M. 2024. Human infections by Hymenolepis diminuta in Europe: a case report and literature review. Trans. Roy. Soc. Trop. Med. Hygiene. 118, 580–588. Milazzo, C., Ribas, A., Casanova, J.C., Cagnin, M., Geraci, F. and Di Bella, C. 2010. The brown rat (Rattus norvegicus) (Berkenhout, 1769) helminths in the city of Palermo, Italy. Helminthologia 47, 238–240. Mohd-Qawiem, F., Nur-Fazila, S.H., Ain-Fatin, R., Yong, Q.H., Nur-Mahiza, M.I. and Yasmin, A.R. 2022. Detection of zoonotic-borne parasites in Rattus spp. in Klang Valley, Malaysia. World 15, 1006–1014. Mohtasebi, S., Teimouri, A., Mobedi, I., Mohtasebi, A., Abbasian, H. and Abbaszadeh Afshar, M.J. 2020. Intestinal helminthic parasites of rodents in the central region of Iran: first report of a capillariid nematode from Dryomys nitedula. BMC. Res. Notes. 4.4, 13–461. Munshi-South, J., Garcia, J.A., Orton, D. and Phifer-Rixey, M. 2024. Evolutionary history of wild and domestic brown rats (Rattus norvegicus). Science 385, 1292–1297. Panti-May, J.A., Palomo-Arjona, E.E., Gurubel-González, Y.M., Barrientos-Medina, R.C., Digiani, M.C., Robles, M.R., Hernández-Betancourt, S.F. and Machain-Williams, C. 2020a. Patterns of helminth infections in Rattus rattus and Mus musculus from two Mayan communities in Mexico. J. Helminthol. 94, 30; doi:10.1016/j.jhelm.2013.09.010 Panti-May, J.A., Rodríguez-Vivas, R.I., García-Prieto, L., Servián, A. and Costa, F. 2020b. Worldwide overview of human infections with Hymenolepis diminuta. Parasitol. Res. 119, 1997–2004. R Core Team, 2021. R: A language and environment for statistical computation. Vienna, Austria: The R Foundation for Statistical Computing. Ribas, A., Saijuntha, W., Agatsuma, T., Thongjun, C., Lamsan, K. and Poonlaphdecha, S. 2016. Helminths in rodents from Thailand’s wet markets. Helminthologic 53, 326–330. Rodriguez-Morales, A.J., Shehata, A.A., Parvin, R., Tasnim, S., Duarte, P.M. and Basiouni, S. 2025. Rodent Borne Parasites and Human Disease: a Growing Public Health Concern. Animals 15. 15, 2681. Shearer, C.L. and Ezenwa, V.O. 2020. Rainfall as a driver of seasonality in parasitism. Int. J. Parasitol. Parasites Wild. 12, 8–12. Stenseth, N.C., Leirs, H., Skonhoft, A., Davis, S.A., Pech, R.P., Andreassen, H.P., Singleton, G.R., Lima, M., Machang'u, R.S., Makundi, R.H., Zhang, Z., Brown, P.R., Shi, D. and Wan, X. 2003. Mice, rats, and people: the bioeconomics of agricultural rodent pests. Front. Ecol. Environ. 1, 367–375. Sungkar, S., Sianturi, I. and Kusumowidagdo, G. 2017. Human Infection with Hymenolepis Spp.: case reports from East Indonesia. Arch. Parasitol. 1, 104; doi:10.1016/j.archparasitol.2011.01.010 Suwannarong, K. and Chapman, R.S. 2015. Characteristics associated with rodent contact in, around, and outside homes in Khon Kaen Province, Thailand. Am. J. Trop. Med. Hyg. 92, 784–790. Taylor, M.A., Coop, R.L. and Wall, R.L. 2016. Veterinary Parasitology, 4th ed. Wiley Blackwell. Tijjani, M., Abdullahi, M.D., Roslaini, A.M. and Ngah, Z.U. 2020. A review on rodent-borne parasitic zoonosis: public health risk to humans. SAJP 2, 263–277. Topouzis, M. 2022. Comparative study of the frequency of intestinal parasites in children of different social groups. Thesis, Democritus University of Thrace. Ullman-Culleré, M.H. and Foltz, C.J. 1999. Body condition scoring: a rapid and accurate method for assessing the health status of mice. Comp. Med. 49, 319–323. Witmer, G. 2022. Rodents in agriculture: a broad perspective. Agron 12, 1458. Zhang, K., Fu, Y., Li, J. and Zhang, L. 2021. Public health and ecological significance of rodents in the treatment of Cryptosporidium infections. One. Health. 14, 100364. | ||
| How to Cite this Article |
| Pubmed Style Tsakmakidis I, Tamvakis A, Giantsis IA, Diakou A. Prevalence of high zoonotic-importance gastrointestinal helminth parasites and associated risk factors in synanthropic rodents in Greece. Open Vet. J.. 2026; 16(2): 1141-1150. doi:10.5455/OVJ.2026.v16.i2.32 Web Style Tsakmakidis I, Tamvakis A, Giantsis IA, Diakou A. Prevalence of high zoonotic-importance gastrointestinal helminth parasites and associated risk factors in synanthropic rodents in Greece. https://www.openveterinaryjournal.com/?mno=278822 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.32 AMA (American Medical Association) Style Tsakmakidis I, Tamvakis A, Giantsis IA, Diakou A. Prevalence of high zoonotic-importance gastrointestinal helminth parasites and associated risk factors in synanthropic rodents in Greece. Open Vet. J.. 2026; 16(2): 1141-1150. doi:10.5455/OVJ.2026.v16.i2.32 Vancouver/ICMJE Style Tsakmakidis I, Tamvakis A, Giantsis IA, Diakou A. Prevalence of high zoonotic-importance gastrointestinal helminth parasites and associated risk factors in synanthropic rodents in Greece. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 1141-1150. doi:10.5455/OVJ.2026.v16.i2.32 Harvard Style Tsakmakidis, I., Tamvakis, . A., Giantsis, . I. A. & Diakou, . A. (2026) Prevalence of high zoonotic-importance gastrointestinal helminth parasites and associated risk factors in synanthropic rodents in Greece. Open Vet. J., 16 (2), 1141-1150. doi:10.5455/OVJ.2026.v16.i2.32 Turabian Style Tsakmakidis, Ioannis, Androniki Tamvakis, Ioannis A. Giantsis, and Anastasia Diakou. 2026. Prevalence of high zoonotic-importance gastrointestinal helminth parasites and associated risk factors in synanthropic rodents in Greece. Open Veterinary Journal, 16 (2), 1141-1150. doi:10.5455/OVJ.2026.v16.i2.32 Chicago Style Tsakmakidis, Ioannis, Androniki Tamvakis, Ioannis A. Giantsis, and Anastasia Diakou. "Prevalence of high zoonotic-importance gastrointestinal helminth parasites and associated risk factors in synanthropic rodents in Greece." Open Veterinary Journal 16 (2026), 1141-1150. doi:10.5455/OVJ.2026.v16.i2.32 MLA (The Modern Language Association) Style Tsakmakidis, Ioannis, Androniki Tamvakis, Ioannis A. Giantsis, and Anastasia Diakou. "Prevalence of high zoonotic-importance gastrointestinal helminth parasites and associated risk factors in synanthropic rodents in Greece." Open Veterinary Journal 16.2 (2026), 1141-1150. Print. doi:10.5455/OVJ.2026.v16.i2.32 APA (American Psychological Association) Style Tsakmakidis, I., Tamvakis, . A., Giantsis, . I. A. & Diakou, . A. (2026) Prevalence of high zoonotic-importance gastrointestinal helminth parasites and associated risk factors in synanthropic rodents in Greece. Open Veterinary Journal, 16 (2), 1141-1150. doi:10.5455/OVJ.2026.v16.i2.32 |