| Research Article | ||
Open Vet. J.. 2025; 15(12): 6230-6236 Open Veterinary Journal, (2025), Vol. 15(12): 6230-6236 Research Article Molecular detection of Escherichia coli producing Vero cytotoxic toxin in red and white meat available in local Syrian marketsAbdul Aziz Arawneh1, Ghiyath Soliman2 and Ammar Haji Al-Ali1*1Department of Public Health and Preventive Medicine, Faculty of Veterinary Medicine, University of Hama, Hama, Syria 2Faculty of Medicine, Department of Family and Community Medicine, University of Tartous, Tartous, Syria *Corresponding Author: Ammar Haji Al-Ali. Department of Public Health and Preventive Medicine, Faculty of Veterinary Medicine, Hama University, Hama, Syria. Email: ammmmar19994 [at] gmail.com Submitted: 21/07/2025 Revised: 01/11/2025 Accepted: 11/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
AbstractBackground: The importance of this research is to address the significant concern regarding the presence of (Escherichia coli), which is producing cytotoxic Vero O157:H7 in meat products, and the lack of studies that is occurred in our country on the level of meat contamination with E. coli, which is considered dangerous, and it has implications for the health of consumers of red and white meat. Aim: To detect the presence of E. coli, which produces cytotoxin Vero toxin O157:H7 in red and white meat that is consumed in local markets, and the effects of some preservatives. Methods: This study was conducted in the microbiology and polymerase chain reaction at the Faculty of Veterinary Medicine, Hama University. One hundred and twenty random red and white meat samples that were consumed in local markets in Hama were collected to detect E. coli, which produces cytotoxic Vero toxin, as follows: Twenty samples of each beef, goat, sheep, camel, broiler and fish meat, which are available in the markets and widely consumed. Two hundred and fifty grams of each type of meat sample were collected. The samples were placed in sterile nylon bags to prevent contamination and transported to the laboratory in special thermal containers until the necessary tests were conducted. Results: The analysis of broiler meat samples showed the presence of E. coli that produces cytotoxic Vero toxin O157:H7 in 65% of the samples. The fish meat samples revealed that 10% contained E. coli producing the toxic Vero toxin for O157:H7 cells, with 50% of those strains tested for bacterial culture. Red meat samples indicated that 55% of sheep meat contained E. coli producing the toxic Vero toxin for O157:H7 cells, with 10% of the strains isolated. For beef, 10% of the samples contained E. coli producing the toxic Vero toxin for O157:H7 cells, in 15% of the strains tested. Overall, 60% of the samples had E. coli producing Vero toxin for O157:H7, with 10% confirmed by PCR. Additionally, 75% of the samples tested by bacterial culture contained E. coli producing Vero cytotoxic toxin. Conclusion: The presence of this E. coli bacteria and its detection by PCR test in white and red meat carcasses poses a significant risk to public health, which is evidence of the poor health status of meat markets. Keywords: E. coli, White and red meat, PCR, Public Health. IntroductionMeat is defined as the animal tissue of muscle, fat, and connective tissue that can be used in human food. As this term means the muscular system of the animal, including the organs that are fit for human consumption, like internal organs (heart, liver, and kidneys). It must be intact, with a firm texture, an acceptable odor, a natural reaction closer to acidity (6 ± 2), free from pathogens, and coincident with each state's own customs and traditions, as in white meat (poultry - fish) and red meat (sheep - beef - goats - camels). The contamination of meat with germs (Ahmad and Badpa, 2014) depends on the animal health status, the conditions of its breeding, the hygienic methods, and the conditions of dealing with carcasses. There, the meat is exposed to different contamination sources with germs during the process of slaughtering and preparing carcasses, including air, water, processing tools, surfaces, and workers' hands, and during the removal of the skin and intestines from the carcass (Gill, 1998). In addition, the animal's flesh and skin carry millions of germs, as well as its entrails, which are a major source of meat contamination. Also, the contact of the product with unclean surfaces increases its basic load of germs (Marriott and Gravani, 2006). Some pathogens, such as Escherichia coli and Salmonella, are transmitted through eating meat, starting from slaughtering the animal, removing feathers or skin, evisceration, contaminated nearby carcasses, or slaughtering equipment (Yulistiani et al., 2019). The number of infections transmitted through eating meat each year has been estimated at 600 million cases, 420,000 of them die, including 125,000 children under the age of five, according to World Health Organization estimates (World Health Organization, 2015). There is a lot of research on foods that are contaminated with pathogenic germs, and the most famous food-contaminating germs are E. coli, which produces Shiga toxin, a toxic substance classified as a biological agent (Roy, 2016). It is the most important germ that may be found in meat, E. coli, as more than 90% of cases of these diseases can be caused by germs, but the most prominent of them is pathogenic E. coli (Wagner, 2008). Given the lack of studies in our country on the level of meat contamination with E. coli germs that produce the toxic toxin of dangerous Vero cells, which is related to the health of white meat consumers, this study aimed to reveal the presence of E. coli germs that produce the toxic toxin of Vero cells in white meat consumed in local markets. The objectives of the research: 1- Detecting E. coli spores that produce toxic Vero toxin and determining the level of contamination in white meat (broiler meat, fish meat) and red meat (beef meat, sheep and goat meat, camel meat). 2- Detection of the presence of SLT1-SLT2 (VT1-VT2) genes of Vero cytotoxic E. coli bacteria using polymerase chain reaction (PCR) in white and red meat. Materials and MethodsIncubator, discriminating E. coli cultures, sodium lactate, 4% concentration, and 2% concentration, red meat that includes sheep, beef, goat, and camel, and white meat including broiler meat and fish. Samples120 random samples of red and white meat consumed in Hamma markets, including meat, were collected throughout the year, to detect E. coli that produces cytotoxic Vero toxin as follows: White meat, 20 samples of broiler meat, 20 samples of fish meat. Red meat, 20 samples of sheep meat, 20 samples of beef meat, 20 samples of goat meat, and 20 samples of camel meat, at a rate of 250 g of each type of meat for each sample of meat. The samples were transported after being them in clean and sterilized bags to the laboratory of the College of Veterinary Medicine, Hama University, where we conducted the following tests: Apparatus usingIncubator – Electric thermal Oven - Magnetic stirrer - Water bath – Light optical microscope Sensitive balance - Microtome - Thermal mixer - Thermal DNA replicator - Electric mixer - Refrigerator Membrane filter - Micropipettes - Membrane filter Micropipettes - Distiller - Magnetic stirrer (Advantec SR350) - Thermal cycler (TECHNE TC-512) - High-speed Spectra Fuge (LabNet 240). UV reader connected to a camera, computer, and printer (UVIPRO Platinum + Mitsubishi P93D) - Electrophoresis machine (peQlab Bio Technology, GMBH). Sterile chamber (HERolaB) - Eppendorf tubes of different sizes (200 μl PCR tubes, 1.5 ml tubes - KB003 Hi25E biological ruler). After biochemical tests, they revealed the possession of these genes. The stages of implementation were conducted in the PCR laboratory at the College of Veterinary Medicine at the University of Hama. Culture mediaMacConkey agar - Nutritious agar - Brine Heart Infusion Cefixime Tellurite Sorbitol MacConkey agar - Nutrient broth - EMB. Isolation of E. coli secreting Vero cytotoxic toxinIsolation of E. coli spores secreting the toxic toxin of Vero cells from the tested meat was performed by following the steps as per sample preparation (Al-Sharjabi, 2015): According to (Al-Sharjabi, 2015), 25 g of each sample were placed in a sterile bag containing 225 ml of Brine Heart Infusion Broth and homogenized by Stomacher, then incubated at 41.5°C for 6 hours. After that, they were transferred to Cefixime Tellurite Sorbitol MacConkey Agar (CT-SMAC) and incubated at 37°C for 18–24 hours. Bacterial isolationA loopful of homogenized samples was transferred to CT-SMAC and incubated at 37°C for 18–24 hours. Non-sorbitol-fermented colonies were cultured again on Eosin-Methylene Blue agar (EMB) and incubated at 37°C for 24 hours. Colonies with a green metallic sheen were then selected for biochemical tests. A microbial smear was prepared and stained with Gram stain using a dye from Hi Media, following the manufacturer's instructions. The Hi20™ *Enterobacteriaceae* Identification Kit (KB003) was used for biochemical tests following the manufacturer's instructions. Purification and preservation of isolatesSome of the growing isolates were transferred from the culture media to the nutrient agar medium using a sterile conveyor in a streaking vector, and then incubated at 37°C for 24 hours, where the purification stage was carried out by transferring the purified isolates three consecutive times. After purification of the colonies, the bacterial isolates were preserved in the nutrient broth, where they were incubated at a temperature of 37°C for 24 hours, after which they were stored in the refrigerator until tests were conducted on them, taking into account the renewal of the culture every (1–3) months (Collee et al., 1996). Study of microscopic and chemical propertiesA bacterial smear was prepared and stained with Gram stain using a dye by Hi Media®, following the instructions of the dye manufacturer. The Hi20™ *Enterobacteriaceae* Identification Kit (KB003) was also used for biochemical testing following the manufacturer's instructions. PCR testThis technique was used to detect the presence of genes from the toxin (SLT1–SLT2) (VT1–VT2). These genes are considered the most important virulence factors possessed by pathogenic E. coli in humans. The germline progenies that tested positive for E. coli were subjected to bacterial culture and biochemical tests to this test to confirm the presence of these genes. This test was performed in the PCR laboratory at the Faculty of Veterinary Medicine at Hama University. Reagents used in the PCR test1 primer: The sequence of these primers was chosen to amplify the genes encoding both vt2 and vt1 (Oie, 2008), as shown in the following Table 1. Table 1. The sequence of these primers was chosen to amplify the genes encoding both vt2 and vt1 (Oie, 2008).
-It was obtained from TaKaRa Company of Japan (Cod No. RR01AM). This kit contains DNA polymerase TaKaRa EX, TaqTM 10X, EX TaqTM Polymerase Reaction Buffer mg+2 free, 2525M MgCl2 salt, and a mixture of the four nitrogenous bases (dNTP: A/C/G/T). -Sterile DNA-free sterile distilled water: Used in the DNA-Extraction stage as well as in the PCR reaction mix preparation stage. -Agarose: It is obtained from PeqGold Universal Company (lot N O2) and used to prepare a 1.5% agarose gel to detect reaction products amplified in PCR by electrophoresis as shown in Table 2. Table 2. Components of the TBE gel electrophoresis buffer. Tris-borate-EDTA (TBE-1X) TBE.
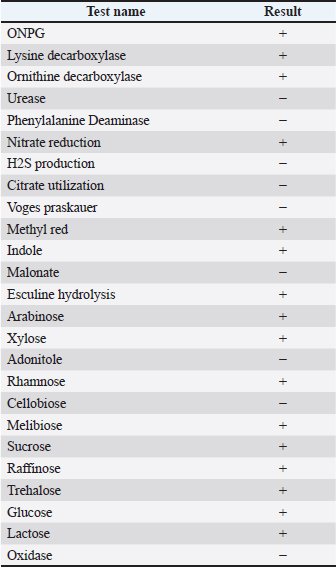
Statistical studyPearson's chi-square method was used for statistical analysis in SPSS 20. Ethical approvalEthical approval was provided by the Faculty of Veterinary Medicine, Hama University, Syria. ResultsResults of the bioassaysThe results of the confirmatory biochemical tests were read using the KB003 Hi25 Enterobacteriaceae diagnostic system for E. coli, where the results were read with reference to the standard tables for each test to confirm the identity of the bacterial isolate, and Table 3 shows the results of the confirmatory biochemical tests for E. coli isolates. Table 3. Shows the results of the biochemical tests.
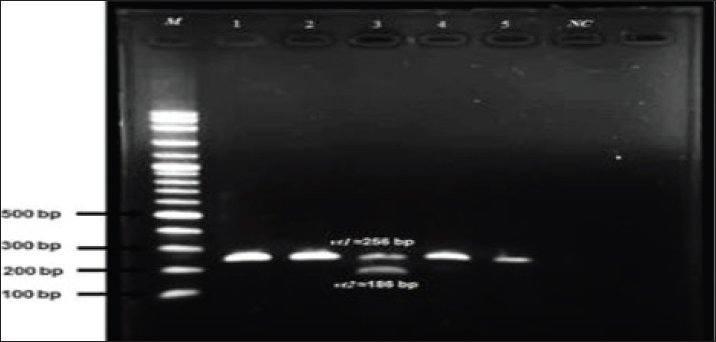
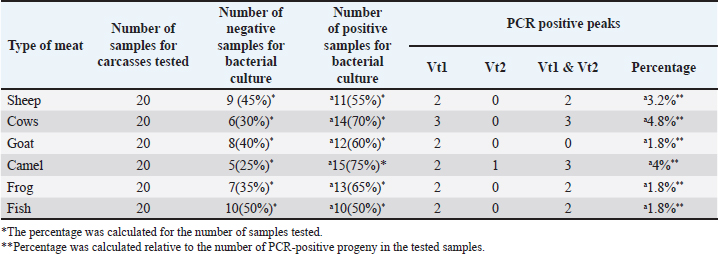
The results of culture and microbial characterization tests for the detection of E. coli that produce Vero cell toxin for E. coli progeny in meat samples in PCR, as seen in Table 4 and Figure 1.
Fig. 1. Shows the results of multiple PCRs. PCR amplification product for E. coli isolates for Vt1 gene (1,2,3,4,5) at 256 bp Vt2 genes (3) at 185 bp, M=ladar, (6) –Ve results. Table 4. The results of culture and microbial characterization tests for the detection of E. coli producing Vero cell toxin for E. coli progeny in meat samples in PCR.
DiscussionAfter reviewing the results of this research conducted on E. coli in general and specifically on E. coli producing Vero toxin in animal products in Syria, Hamma governorate, which agrees with what other studies have relied on, where it targeted E. coli serotype O157:H7, we find that the results of the examination of sheep meat showed the presence of E. coli producing Vero Cytotoxin O157:H7 in 55% of the samples studied for bacterial culture: H7 by 55% with the studied samples for bacterial culture and 10% for PCR, which has both the gene encoding the Vero-1 and Vero-2 cytotoxic toxin. This percentage constitutes a significant increase and clear evidence of the great weakness and low health security procedures followed in the meat markets from which samples were taken, and this constitutes a very serious issue for the health of consumers of this meat. These results are consistent with (Zweifel and Stephan, 2003), where it was found that the prevalence of E. coli in Switzerland is 36.6% in sheep carcasses. The results are also consistent with (Lenahan et al., 2007), has mentioned in their study on sheep carcasses in Ireland, where the prevalence of E. coli producing VT2 was 1%. Also, they are consistent with the study of (Al-Safi and Al-Raouf, 2003), where the prevalence of E. coli producing VT2 in sheep meat was 4%, and all isolated progeny were producing VT2 during the test procedure, and consistent with a study in (Tarawneh et al., 2009) for the detection of VTEC-producing E. coli in sheep, where the results showed that VTEC-producing E. coli progeny constituted 6. In Iran, his study (Shekar Forush et al., 2008) on sheep carcasses, he found the presence of E. coli producing Vero-toxic toxin in 3.92%. The presence of Vero-cytotoxic E. coli in sheep meat is attributed to several factors, including the poor hygienic measures followed in slaughterhouses, sheep meat markets, the transportation of carcasses for cooling and distribution, and the sites where the meat samples were taken from. These factors play an important role in the presence of these bacteria. Also, the conditions of the carcass play an important role in determining the rate of spread, as the percentage of these bacteria increases (Duffy et al., 2003). In addition, the season of the year plays an important role. Especially, the summer season increases the rate of E. coli producing the toxin in Vero cells. The techniques, quality, and sensitivity play a crucial role in the presence of contamination with these bacteria. Furthermore, various geographical areas are consistent with the study of (Shekar Forush et al., 2008), compatible with the study of (Rahimi et al., 2012), and with the study of (Hassan et al., 2016). Considering the results for bovine meat, E. coli producing Vero O157 cytotoxic E. coli was found to be present at 70% with the studied samples for bacterial culture, 15% for PCR: H7 was found in 70% of the studied samples for microbial culture, and 15% for PCR for isolated offspring that possessed both the gene encoding the Vero-1 and Vero-2 cytotoxic toxin 2. This is clear evidence of the presence of contamination with these bacteria. This percentage constitutes a significant increase and clear evidence of the great weakness and low health security procedures followed in meat markets and slaughterhouses spread in the governorate, and these results are consistent with several studies and research, such as the study of (Al-Safi and Al-Raouf, 2003) in Egypt, where VTEC O157 E. coli was found to be 5%. In a study in Mosul conducted by (Shaker et al., 2022) in Mosul, the prevalence of E. coli VTEC O157 in veal meat was found to be 9.4%, and in a study conducted by (Sahar et al., 2016) in Basra Governorate, the percentage of E. coli VTEC O157 was 5%. Another study conducted by (Beneduce et al., 2008) in Morocco, the prevalence of E. coli was 9% in the studied stores. By comparing the results with previous studies, we find a notable difference in the percentage of VTEC O157 contamination is attributed in the first level to the health security followed in bovine slaughterhouses, and this is consistent with the study of (Zahra, 2010). The second level is attributed to the annual season in which the samples were collected. As many studies indicated that E. coli VTEC O157 increases in the summer, such as the study of (Shekar Forush et al., 2008) and (Rahimi et al., 2012). In contrast to the study of (Murphy et al., 2005), his results were negative for the presence of genes encoding the toxic toxins of Vero toxin cells (VT1and VT2). As for the results of goat meat, they showed the presence of E. coli producing Vero O157:H7 is present in 60% of the studied samples based on microbial culture and 10% for isolated offspring based on PCR, which possess both the gene encoding the Vero-1 and Vero-2 cytotoxic toxin. This is consistent with the study that conducted by (Zahra, 2010). It was found that the prevalence of evidence of its presence within this meat and this E. coli producing Vero cytotoxic O157:H7 by 8%, as PCR results showed that 100% of the offspring possessed the gene encoding the toxin for Vero-2 cells, which is the main factor causing diseases in humans, and this is great evidence of a defect in health procedures in these markets and slaughterhouses. As for camel meat, we found that the presence of E. coli producing Vero cytotoxic toxin is 75% with the studied samples for microbial culture and 10% for PCR for isolated offspring that have both the genes encoding Vero cytotoxic toxin-1 and -2.1 and 2. We have not received any studies on the presence of E. coli producing Vero toxin in camel meat, but there are studies, such as a study conducted by (Abdullah et al., 2023) on camel milk, where they found a prevalence of E. coli of 10% and in a study conducted by (Altee and Yousif, 2023), where he revealed the presence of E. coli in camel feces, he found only four isolates that were diagnosed as E. coli O157:H7. Coli O157:H7 out of 117 samples. This, anyway, is evidence of the deteriorating reality of slaughterhouses, and places where these meats are sold. This is a serious matter that must be addressed. Considering the results of the broiler meat samples, the presence of E. coli producing O157:H7 was 65% with the studied samples for microbial culture and 10% for PCR for the isolated progeny examined on the PCR machine, which is consistent with the results of a study conducted by (Shimaa et al., 2021) where the prevalence of E. coli was 16%17.%, so it was concluded that E. coli is present in broiler meat. The study of (Haillehizeb et al., 2024) found that E. coli O157:H7, which was imported from chicken meat in Addis Ababa slaughterhouses, was isolated and identified, and the percentage was 5.2%. Regarding the results for fish meat, we found that E. coli O157:H7 was present in 50% of the samples based on microbial culture, and 10% based on PCR of isolated colonies or cultures examined on the PCR device. Oluwafunmilayo et al. (2020) reported that fish meat is contaminated with E. coli producing toxins in Vero cells. Additionally, Ayalew et al. (2019) found the presence of E. coli at a rate of 9.4%. These findings highlight the worsening situation in meat markets. Strict measures, including monitoring, periodic inspections of meat sales locations, and renewed food control, are necessary. ConclusionThe study revealed the presence of E. coli bacteria, which produces Vero toxins. This is clear evidence of the significant weakness in health security measures in the meat preparation sites and markets where the samples were taken from, which could pose a potential health risk to consumers. AcknowledgmentDr. Murshid Kassoha for his assistance in the PCR laboratory and Dr. Abdul Karim Hallaq for completing the statistical study. FundingHama University is the main contributor to this research, and Ibn Al-Haytham Veterinary Medicines Company also assisted with this research. Authors' contributionsAmmar Haji Al-Ali is the principal investigator, implementer, and author. This research is part of a doctoral dissertation currently under preparation. Dr. Abdul Aziz Arwaneh is supervising this research, serving as scientific supervisor and assistant in guidance and writing the dissertation. Dr. Ghiyath Soliman is the co-supervisor, having contributed to the success of the dissertation. Conflict of interestThe authors declare that they have no conflict of interest. Data availabilityAll data are included in this manuscript. ReferencesAhmad , S. and Badpa, A.G. 2014. Meat products and byproducts for value addition. In Food processing strategies for quality assessment. Eds., Erginkaya, A.M.Z., Ahmad, S. and Erten, H. Springer Science + Business Media, New York, pp: 124 Alsayeqh, A.F., Mohamed, A.S., Mohamed, R.E., Ibrahim, N.A., Hamdy, E. and Alnakip, M.E. 2023. Prevalence of multidrug-resistant Shiga toxin-producing E. coli in the milk of cattle, buffaloes, and camels. Slov. Vet. Res. 60(Suppl 25), 291. Al-Sharjabi. and Hamid, F.A. 2015. Food microbiology. Yemen: Taiz University Publications. Altee, A.K. and Yousif, A.A. Occurrences of E. coli in dromedary camels in some provinces in Iraq with a molecular study of E. coli O157: h7. In Proceedings of the 14th (4th International) Scientific Conference, College of Veterinary Medicine, University of Baghdad,2023 37(Supplement II), pp 49–54. Assefa, A., Regassa, F., Ayana, D., Amenu, K. and Abunna, F. 2019. Prevalence and antibiotic susceptibility pattern of Escherichia coli O157:H7 isolated from harvested fish at Lake Hayq and Tekeze dam, Northern Ethiopia, December 4, 1–6. Ayodele, O.A., Deji-Agboola, A.M., Faneye, A.O. and Akinduti, P.A. 2020. Characterization and antibiotic susceptibility of E. coli O157: h7 in meat and fish sold in major Ibadan Markets, Nigeria. JBiomedSci 12(2), 99–106; doi:10.5099/aj200200099 Beneduce, L., Spano, G., Nabi, A.Q., Lamacchia, F., Massa, S., Aouni, R. and Hamama, A. 2008. Occurrence and characterization of Escherichia coli O157:H7 and other serotypes in raw meat products in Morocco. J. Food Prot. 71(10):2082–2086. Collee, J.G., Fraser, A.G., Marmion, B.P. and Simmons, A. 1996. Practical medical microbiology. 14th ed. New York, USA: Churchill Livingstone, Inc. Duffy, G., Cagney, C., Crowley, H. and Sheridan, J. 2003. A nationwide surveillance study on E. coli 0157:H7 and enterobacteriaceae in Irish minced beef products. Teagasc. El-Safey, E.M. and Abdul-Raouf, U.M. 2003. Detection of Escherichia coli o157: h7 in some Egyptian foods. J. Agricult. Sci. 34(6). George, S.S., Al-Shatti, S.M.H. and Hussein, N.A. 2016. Isolation and identification of bacteria O157: h7 and study of the numbers of total coliform bacteria and E. coli in fresh beef in Basra city. J. Thi. Qar. Univ. For. Agricult. Res. 5. Gill, C.O. 1998. Microbiological contamination of meat during slaughter and butchering of cattle, sheep, and pigs. The Microbiology of Meat and Poultry, London: Blackie Academic & Professional, pp: 118–157. Hassan, M.A., Heikal, G.I. and Barhoma, R.M. 2016. Traceability of enteropathogenic E. coli in cattle and camel carcasses. Benha. Vet. Med. J. 31(1), 50–55; Available via http://www.bvmj.bu.edu.eg Lenahan, M., O'Brien, S., Kinsella, K., Sweeney, T. and Sheridan, J.J. 2007. Prevalence and molecular characterization of Escherichia coli O157: h7 on Irish lamb carcasses, fleece, and in feces samples. J. Appl. Microbiol. 103(6), 2401–2409. Marriott, N.G. and Gravani, R.B. 2006. Food contamination sources. In Princ. Food Sanit. pp: 76–82. Mohammed, Z..2010. Investigation of Escherichia coli producing cytotoxic toxin Vero in some animal products. Scientific research for a master's degree in microbiology, Faculty of Veterinary Medicine, Al-Baath University. Murphy, M., Carroll, A., Whyte, P., O'Mahony, M., Anderson, W., Mcnamara, E. and Fanning, S. 2005. Prevalence and characterization of Escherichia coli O26 and O111 in retail minced beef in Ireland. Foodborne Pathog. Dis. 2(4), 357–360. OIE. 2008. Manual of diagnostic tests and vaccines for terrestrial animals. Rahimi E, Kazemeini HR, Salajegheh M. 2012. Escherichia coli O157:H7/NM prevalence in raw beef, camel, sheep, goat, and water buffalo meat in Fars and Khuzestan provinces, Iran. Vet. Res. Forum. Int. Q. J. 3, 15–17. Roy AL, 2016. Reducing risk of foodborne illness in older adults: interventions targeting at-home and foodservice handling behaviors. Iowa: Iowa State University. Sheet, O. Sheet, O. Othman, S. and Alsanjary, R. 2022. Isolation and identification of Escherichia coli O157: h7 isolated from Veal Meats and Butchers' Shops in Mosul city, Iraq. J. Appl. Vet. Sci. 7(4), 55–61. Shekarforo, S., Tahamtan, Y. and Pourbakhsh, A. 2008. Detection and frequency of Stx2 gene in Escherichia coli O157 and O157: h7 strains isolated from sheep carcasses in Shiraz-Iran. Pak. J. Biol. Sci. 11(8), 1085–1092. Sherif, S., Saad, S., Hassanin, F. and Nassif, M. 2021. Enteropathogenic Escherichia coli contaminating chicken meat cuts. Benha Vet. Med. J. 40, 99–102. Tarawneh, K.A., Al-Tawarah, N.M., Abdel-Ghani, A.H., Al-Majali, A.M. and Khleifat, K.M. 2009. Characterization of verotoxigenic Escherichia coli (VTEC) isolates from feces of small ruminants and environmental samples in southern Jordan. J. Basic Microbiol. 49(3), 310–317. Tegegne, H., Filie, K., Tolosa, T., Debelo, M. and Ejigu, E. 2024. Isolation and identification of Escherichia coli O157:H7 recovered from chicken meat at Addis Ababa Slaughterhouses (17) 851-863- 851. Wagner, A.B. 2008. Bacterial Food Poisoning. [online] Texas A&M AgriLife Extension. Available via https://aggie-horticulture.tamu.edu/food-technology/bacterial-food-poisoning/ World Health Organization. 2015. WHO estimates of the global burden of foodborne diseases: Foodborne disease burden epidemiology reference group, 2007-2015. World Health Organization. Yulistiani, R., Praseptiangga, D. and Supyani, S. 2019. Occurrences of Salmonella spp. and Escherichia coli in chicken meat, intestinal contents, and rinse water at the slaughtering place from the traditional market in Surabaya, Indonesia. In IOP Conf. Series: Materials Science and Engineering, 2019, 633, p 012007. Zweifel, C. and Stephan, R. 2003. Microbiological monitoring of sheep carcass contamination in three Swiss abattoirs. J. Food Prot. 66(6), 946–952. | ||
| How to Cite this Article |
| Pubmed Style Arawneh AA, Soliman G, Al-ali AH. Molecular detection of Escherichia coli producing Vero cytotoxic toxin in red and white meat available in local Syrian markets. Open Vet. J.. 2025; 15(12): 6230-6236. doi:10.5455/OVJ.2025.v15.i12.7 Web Style Arawneh AA, Soliman G, Al-ali AH. Molecular detection of Escherichia coli producing Vero cytotoxic toxin in red and white meat available in local Syrian markets. https://www.openveterinaryjournal.com/?mno=278821 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.7 AMA (American Medical Association) Style Arawneh AA, Soliman G, Al-ali AH. Molecular detection of Escherichia coli producing Vero cytotoxic toxin in red and white meat available in local Syrian markets. Open Vet. J.. 2025; 15(12): 6230-6236. doi:10.5455/OVJ.2025.v15.i12.7 Vancouver/ICMJE Style Arawneh AA, Soliman G, Al-ali AH. Molecular detection of Escherichia coli producing Vero cytotoxic toxin in red and white meat available in local Syrian markets. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6230-6236. doi:10.5455/OVJ.2025.v15.i12.7 Harvard Style Arawneh, A. A., Soliman, . G. & Al-ali, . A. H. (2025) Molecular detection of Escherichia coli producing Vero cytotoxic toxin in red and white meat available in local Syrian markets. Open Vet. J., 15 (12), 6230-6236. doi:10.5455/OVJ.2025.v15.i12.7 Turabian Style Arawneh, Abdul Aziz, Ghiyath Soliman, and Ammar Haji Al-ali. 2025. Molecular detection of Escherichia coli producing Vero cytotoxic toxin in red and white meat available in local Syrian markets. Open Veterinary Journal, 15 (12), 6230-6236. doi:10.5455/OVJ.2025.v15.i12.7 Chicago Style Arawneh, Abdul Aziz, Ghiyath Soliman, and Ammar Haji Al-ali. "Molecular detection of Escherichia coli producing Vero cytotoxic toxin in red and white meat available in local Syrian markets." Open Veterinary Journal 15 (2025), 6230-6236. doi:10.5455/OVJ.2025.v15.i12.7 MLA (The Modern Language Association) Style Arawneh, Abdul Aziz, Ghiyath Soliman, and Ammar Haji Al-ali. "Molecular detection of Escherichia coli producing Vero cytotoxic toxin in red and white meat available in local Syrian markets." Open Veterinary Journal 15.12 (2025), 6230-6236. Print. doi:10.5455/OVJ.2025.v15.i12.7 APA (American Psychological Association) Style Arawneh, A. A., Soliman, . G. & Al-ali, . A. H. (2025) Molecular detection of Escherichia coli producing Vero cytotoxic toxin in red and white meat available in local Syrian markets. Open Veterinary Journal, 15 (12), 6230-6236. doi:10.5455/OVJ.2025.v15.i12.7 |