| Research Article | ||
Open Vet. J.. 2025; 15(12): 6268-6286 Open Veterinary Journal, (2025), Vol. 15(12): 6268-6286 Research Article Clinicobiochemical and gas chromatography-mass spectrometry-based serum metabolomics for determining the therapeutic efficacy of silymarin in pneumonic Barki sheepHany Hassan1*, Ahmed Kamr1, Abdel Nasser El-Gendy2, Ramiro Toribio3, Amira R. Khattab4,5, Walid Mousa6, Hadeer khaled1, Abdelsalam Elkholey1, Mohamed Kasem1 and Ali Arbaga11Department of Animal Medicine and Infectious Diseases (Animal Internal Medicine), Faculty of Veterinary Medicine, University of Sadat City, Sadat City, Egypt 2Medicinal and Aromatic Plants Research Department, Pharmaceutical and Drugs Research Institute, National Research Center, Cairo, Egypt 3Department of Veterinary Clinical Sciences, College of Veterinary Medicine, Ohio State University, Columbus, Ohio 4Pharmacognosy Department, College of Pharmacy, Arab Academy for Science, Technology and Maritime Transport, Alexandria, Egypt 5Graduate School in Alamein, Arab Academy for Science, Technology & Maritime Transport, Alamein, Egypt 6Department of Animal Medicine and Infectious Diseases, Faculty of Veterinary Medicine, University of Sadat City, SadatCity, Egypt *Corresponding Author: Hany Hassan. Department of Animal Medicine and Infectious Diseases (Animal Internal Medicine), Faculty of Veterinary Medicine, University of Sadat City, Sadat City, Egypt. Email: hany.youssef [at] vet.usc.edu.eg Submitted: 18/08/2025 Revised: 05/11/2025 Accepted: 17/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
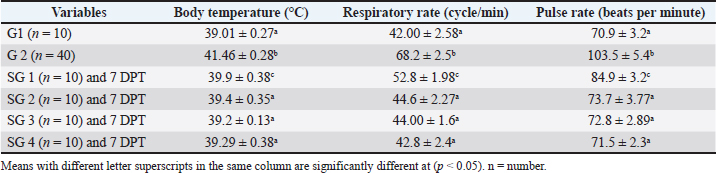
AbstractBackground: Silymarin has anti-inflammatory, anti-oxidative, and antimicrobial properties that are superior to those of traditional treatments. These properties will enhance the further use of this medicinal plant in the veterinary field. Aim: This study aimed to evaluate the therapeutic efficacy of silymarin against sheep bacterial pneumonia using clinical, biochemical, and metabolomics approaches. Methods: Fifty adult male Barki sheep were divided into two groups based on their health status. Group 1 included healthy sheep (n=10); group 2 included sick sheep with clinical evidence of pneumonia (n=40), which were further classified into four subgroups based on treatment protocols: subgroup 1 received traditional treatment; subgroup 2 received traditional treatment plus 280 mg of silymarin orally daily; subgroup 3 (SG3) received 280 mg of silymarin orally daily; and subgroup 4 received 560 mg of silymarin orally daily. Efficacy-directed distinction between therapeutic groups was accomplished based on gas chromatography-mass spectrometry-generated serum metabolite profiles supported by partial least squares regression analysis (PLS). Results: The PLS score plot showed a clear discrimination between the healthy and pneumonic sheep groups that exhibited lower concentrations of total antioxidant capacity (TAC), total cholesterol, high-density lipoprotein (HDL) cholesterol, and glucose, but significantly elevated liver enzyme, urea, creatinine, malondialdehyde, and low-density lipoprotein cholesterol. Clinical evaluations demonstrated that oral administration of 560 mg of silymarin produced the most rapid clinical improvement. However, metabolomic profiling indicated that 280 mg silymarin (SG3) therapy provided the most favorable therapeutic outcome, as it was strongly associated with the upregulation of TAC, glucose, and both total and HDL-cholesterol levels. Conclusion: Pneumonic sheep treated with silymarin (560 and 280 mg) exhibited greater clinical, metabolomic, and biochemical improvement than those treated with traditional treatment alone. Keywords: Silybum marianum, Clinical healing, Sheep, Pneumonia. IntroductionPneumonia is a complex disease that results from a combination of environmental, management, immune, and infectious (bacterial, viral, and mycotic agents) factors in small ruminants, resulting in major economic losses due to the cost of treatment, reduced productivity, and high mortality rates (Thompson, 2019). Antimicrobial drugs are commonly used to treat bacterial pneumonia in sheep (Berge et al., 2006). Anti-inflammatory drugs are often indicated to reduce inflammatory processes and disease severity. Currently, antimicrobial resistance is a global concern that requires multidisciplinary strategies to ensure efficient therapies for human and animal populations (Magstadt et al., 2018). In recent years, herbal therapy has received attention as a novel and alternative therapeutic approach owing to its safety and cultural acceptability (Laudato and Capasso, 2013; Tamminen et al., 2018). Silybum marianum (milk thistle) has been used for centuries as a natural remedy to treat various illnesses, particularly hepatic ones. It contains a flavonolignan complex termed silymarin, which is mainly found in seeds and fruits (Marmouzi et al., 2021). Silymarin, the main active component of which is silibinin, has antimicrobial, antimycotic, anti-inflammatory, antifibrotic, immunomodulating, and anthelmintic properties (Wu et al., 2019). It also reduces the expression of sterol regulatory element binding protein 1 and fatty acid transport protein 5 in hepatic cells to prevent the fat accumulation caused by free fatty acids (Pferschy-Wenzig et al., 2014; Dhami-Shah et al., 2018). The ability of silymarin to scavenge free radicals and boost endogenous antioxidant defenses, such as the glutathione system, is linked to its ability to reduce oxidative stress-induced hepatocellular damage (Ou et al., 2018). It protects kidney cells from medication-induced nephrotoxicity and cyclosporine nephrotoxicity in vitro (Goli et al., 2019). Metabolomics analysis is a contemporary approach for drug development, disease diagnosis, pathophysiology, and prognosis. It provides more biochemical insight and understanding than other systems biology omics techniques (Schrimpe-Rutledge et al., 2016). Furthermore, it enables the measurement of changes in endogenous small molecules within cells, tissues, and biofluids of the body in response to environmental changes or contaminants. Gas chromatography-mass spectrometry (GC-MS) is the most commonly used metabolomics technology for quantitatively analyzing various metabolites in biological materials. It can effectively and rapidly separate and identify large pools of metabolites (Ibrahim et al., 2021). Recently, scientists have focused on the biological and pharmaceutical applications of metabolites obtained from edible plants and foods (Elshamy et al., 2019; Ammar et al., 2022). This study aimed to assess the therapeutic effectiveness of silymarin in pneumonic sheep using clinical examination, metabolomics profiling, and biochemical parameter measurements. Given the pleiotropic actions of silymarin and the clinical importance of respiratory disease in small ruminants, silymarin in sheep with pneumonia could provide valuable information. Materials and MethodsMilk thistle extract (S. marianum)Silymarin powder was provided by Medical Union Pharmaceuticals (Egypt). The powder consists of 50% silymarin with a potency of 104.49% (code number 0111304600). Criteria and experimental designThis study was registered and approved by the Postgraduate Studies and Research Sector, University of Sadat City, Egypt (registration no. 22/29-12-2021). A total of 50 male adult Barki sheep (Ovis aries) at a private farm in Sadat City, Egypt, aged between 1 and 2 years, with a mean body weight of 60 ± 2 kg, were assigned to two groups based on their health condition. Group 1 (G1; n=10) included healthy sheep with no clinical or laboratory evidence of disease, free of external and internal parasites, and served as the control group. Group 2 (G2; n=40) consisted of sheep with bacterial pneumonia and respiratory disease. All diseased sheep showed copious nasal discharge, fever, cough, dyspnea, and abnormal lung sounds upon chest auscultation. This group was further divided into four subgroups according to the therapeutic protocol. SG1 (n=10) included pneumonic sheep that received the traditional antimicrobial treatment for pneumonia using florfenicol (20 mg/kg body weight/IM injection) two doses with 48 hours interval, a non-steroidal anti-inflammatory drug as diclofenac sodium (2.5 mg/kg body weight/IM injection), and an antihistaminic drug as diphenhydramine hydrochloride 20 mg (1 ml/45 kg body weight/IM injection) single dose daily for 72 hours. Subgroup 2 (SG2) (n=10) consisted of pneumonic sheep treated as subgroup 1 (SG1) plus once-daily oral administration of silymarin at a dose of 280 mg dissolved in water every 24 hours for seven consecutive days (Aflatouni et al., 2020). Subgroup 3 (SG3) (n=10) consisted of pneumonic sheep treated with once-daily oral administration of silymarin 280 mg dissolved in water every 24 hours for seven consecutive days (Aflatouni et al., 2020). Subgroup 4 (SG4) (n=10) included pneumonic sheep treated with daily oral administration of silymarin 560 mg dissolved in water every 24 hours for seven consecutive days. General clinical examination resultsComplete clinical examinations were performed on all involved animals according to the protocol of Constable et al. (2016). SamplingBlood samples were collected from healthy and pneumonic sheep in the early morning by jugular venipuncture before starting different treatment protocols (0 day) and 7 days post-treatment (7 DPT) in serum clot tubes and kept at room temperature until coagulation for at least 60 minutes. The clotted blood samples were centrifuged at 2,000 × g for 10 minutes at 4°C, aliquoted into small tubes then stored at −80°C until analysis. Biochemical assays of hepatic and renal function, glucose, lipid profile, malondialdehyde, and total antioxidant capacityColorimetric methods were used to determine serum aspartate aminotransferase (AST), alanine aminotransferase (ALT), gamma-glutamyl transferase (GGT), creatinine, urea, glucose, triglycerides (TG), cholesterol, high-density lipoprotein (HDL)-cholesterol, low-density lipoprotein (LDL)-cholesterol, malondialdehyde (MDA), and total antioxidant capacity (TAC). Preparation of metabolomic samplesFrozen serum samples were thawed on ice, and silymarin metabolites were extracted and silylated using xylitol as an internal standard, as previously described (Ammar et al., 2022). Briefly, 100 µl of serum was combined with cold acetonitrile (100%, 200 µl) and centrifuged at 7,000 × g for 15 minutes. Nitrogen gas was used to evaporate the supernatant until it was completely dry. A 50 µl pyridine solution of methoxyamine (20 mg/ml) was first added to the dried residue and then incubated at 60°C for 1 hour to derivatize the metabolites. A second derivatization phase was performed by adding 100 µl N-methyl-N-(trimethylsilyl) trifluoroacetamide containing 1% trimethylsilyl (TMS) to the mixture and incubating for 1 hour at 60°C. The QC sample was formed by mixing aliquots from all samples into a pooled sample to analyze changes in MS response. Additionally, a standard hydrocarbon mixture (C8–C40) was analyzed. GC–MS-based profiling of serum metabolitesSerum metabolite profiling was performed using gas chromatography (Thermo Scientific Corp., USA) and a thermo-mass spectrometer detector (ISQ Single Quadrupole Mass Spectrometer). The chromatographic separation was performed as previously described (Raish et al., 2017), with a few modifications over Trace Gold- 5M5 columns (30 × 0.25 mm i.d, 0.25 μm film thickness) and helium at a 1 mL/min flow rate as carrier gas, and a 1:10 split ratio, according to the temperature program: 2 minutes at 80°C, rising with 5°C/min up to 300°C and held for 5 minutes. Both the injector and detector were maintained at 280°C. Mass spectral data were acquired via electron ionization at 70 eV, with a spectral range of 35–500 m/z. GC–MS data processing, molecular networking, and multivariate data analysesSerum metabolite identification was accomplished by comparing their retention indices (RIs) relative to n-alkane standards (C8–C40) alongside mass matching to the National Institutes of Standards and Technology library database. AMDIS software (www.amdis.net) was used to deconvolute the peaks before mass spectral matching. Metabolite abundance data were retrieved using MS Dial software with default settings and Pareto scaling in preparation for multivariate data analysis. The limit of detection and limit of quantification of 32 metabolites were also determined, and signal-to-noise ratios of 3:1 and 10:1, respectively, were considered. Statistical analysesA Shapiro-Wilk test was used to determine the normality of the data. Data were normally distributed and expressed as mean with standard deviation. One-way ANOVA was performed to determine the difference between groups using IBM SPSS Statistics 16 (IBM Corporation, Armonk, NY). Comparative analysis of serum metabolic profiles derived from GC-MS of the different therapeutic approaches was performed using both supervised pattern recognition methods, such as partial least-squares regression analysis (PLS), using the SIMCA-P Version 14.0 (Umetrics, Umeå, Sweden). GC-MS derived serum profiles of endogenous metabolites and biochemical parameters MDA, TAC, lipid profile (TG, cholesterol, HDL-cholesterol, and LDL-cholesterol), blood glucose concentrations, liver enzyme activities (AST, ALT, and GGT), and kidney function tests (creatinine and urea) were subjected to PLS for the sake of efficacy discrimination between the therapeutic approaches and to determine the serum metabolites that are positively or negatively correlated to restoration of pneumonic conditions in sheep to the normal status as implied by the concentrations of the different biochemical markers. Serum metabolites responsible for the differentiation among the four therapeutic approaches were identified based on variable influence on projection (VIP) values of the constructed PLS model (VIP > 1.0). Statistical significance for all analyses in this study was set to p < 0.05. All variables were mean-centered and scaled to Pareto variance (Rummun et al., 2023). Ethical approvalThis study was approved by the Animal Ethics Committee at the Faculty of Veterinary Medicine, University of Sadat City, Egypt (Approval code VUSC-028-1-22). All the methods were performed in accordance with the guidelines and regulations of this committee. Written informed consent for participation was obtained from the farm owner, who approved the experimental protocol. ResultsEffect of traditional treatment and/or silymarin on the clinical profileG2 exhibited copious nasal discharge, fever, cough, dyspnea, and abnormal lung sounds upon auscultation. G2 showed significantly higher body temperature, respiratory rate, and pulse rate than the control group (p < 0.05; Table 1). At 7 DPT, SG1 still had significantly higher body temperature, respiratory rate, and pulse rate compared to healthy ones (p value < 0.05; Table 1), whereas SG 2, 3, and 4 were not significantly different from G1 (p > 0.05). Therapeutic improvements in respiratory rate, pulse, and body temperature were evident in SG2, SG3, and SG4 by the 7-day post-treatment, with the most pronounced response observed in SG4 receiving the high oral dose of silymarin (560 mg) (p < 0.05). Table 1. Effect of traditional and/or silymarin on sheep clinical examination.
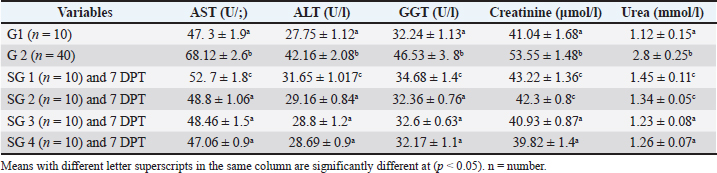
Effect of traditional treatment and/or silymarin on liver and kidney function testsPneumonic sheep had significantly higher AST, ALT, GGT, creatinine, and urea concentrations than healthy sheep (p < 0.05; Table 2). At 7 DPT, SGs 2, 3, and 4 did not differ significantly from healthy sheep (p > 0.05; Table 2), except for SG2, which revealed significantly higher creatinine and urea concentrations than healthy sheep (p < 0.05; Table 2), whereas SG 1 showed increased AST, ALT, creatinine, and urea concentrations than healthy sheep (p < 0.05; Table 2). Table 2. Effect of traditional and/or silymarin on hepatic and renal function tests.
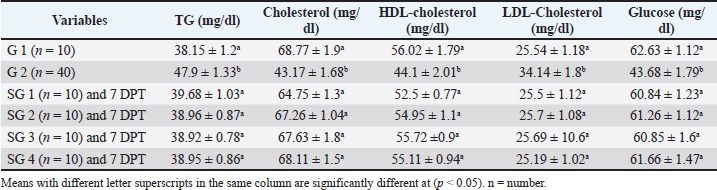
Effect of traditional treatment and/or silymarin on glucose and lipid profilePneumonic sheep had significantly higher triglyceride and LDL-cholesterol and lower cholesterol, HDL-cholesterol, and glucose concentrations than healthy sheep (p < 0.05; Table 3), whereas SG 1, 2, 3, and 4 were not significantly different from healthy sheep at 7 DPT (p > 0.05; Table 3). Table 3. Effect of traditional and/or silymarin treatment on glucose and lipid profiles.
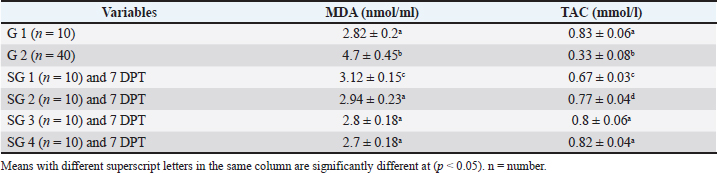
Effect of traditional treatment and/or silymarin on oxidant and antioxidant statusPneumonic sheep had significantly higher MDA and lower TAC concentrations than healthy sheep (p < 0.05; Table 4). For MDA concentrations at 7 DPT, SGs 2, 3, and 4 were not significantly different from healthy sheep (p > 0.05), whereas SG1 showed higher concentrations than healthy sheep (p < 0.05). For TAC concentrations at 7 DPT, SG3 and SG4 were not significantly different from healthy sheep (p > 0.05), whereas SG1 and SG2 had lower TAC concentrations than healthy sheep (p < 0.05; Table 4). Table 4. Effect of traditional treatment and silymarin on oxidative and antioxidant status.
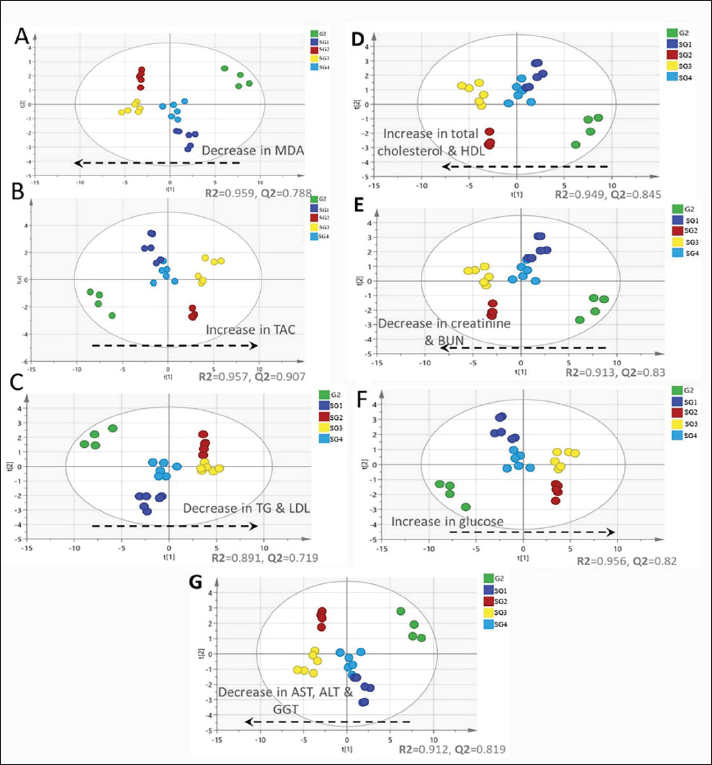
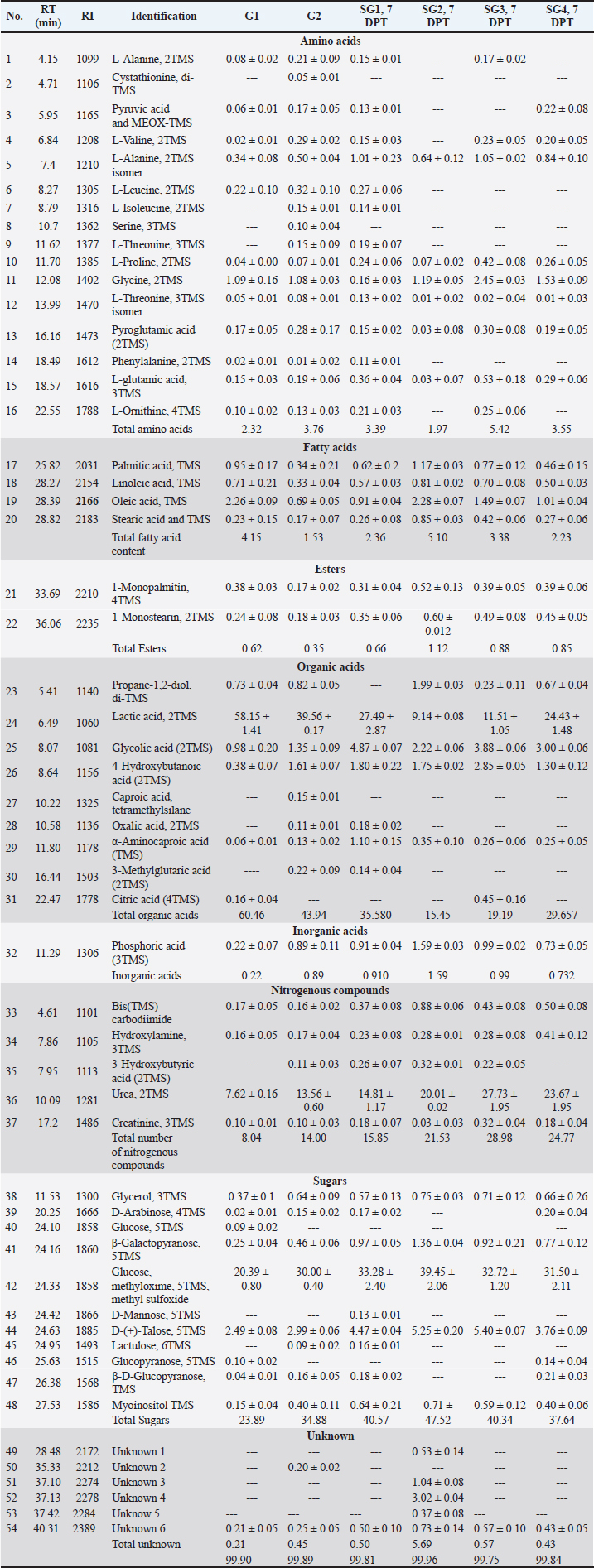
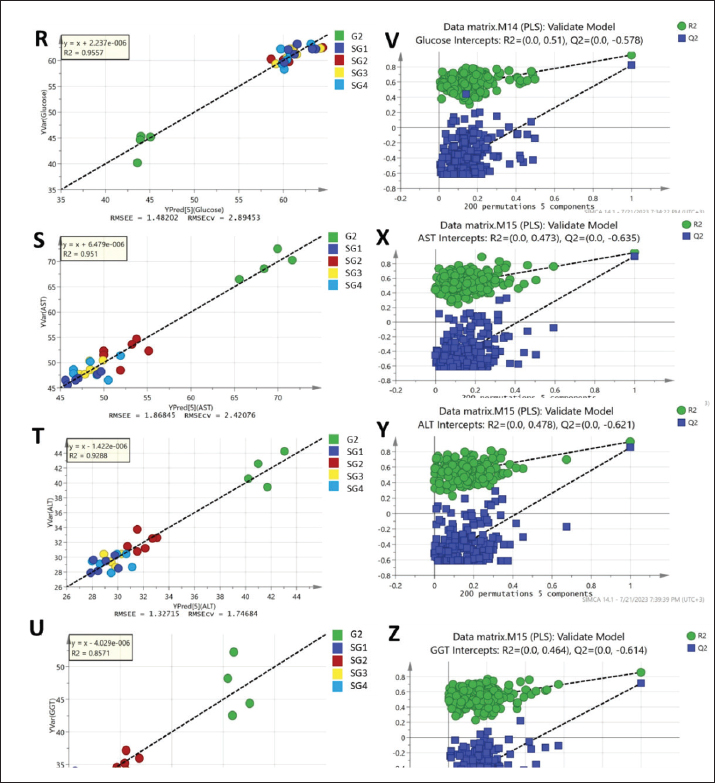
Serum metabolite profiling via GC-MS analysis aided by chemometricsSerum GC/MS-derived metabolite profiles of 49 endogenous metabolites, including organic acids, amino acids, fatty acids, and sugars, were identified in groups 1 and 2 and treated SG1–S4 (Table 5). The supervised pattern recognition method (PLS) was employed to model the serum-based GC-MS-derived metabolite profiles of G1 as X-variables versus G2. The other seven PLS models were attempted to correlate the GC-MS-derived serum metabolite profiles of the four therapeutic approaches (SG1–SG4) along with the pneumonic sheep group (G2) to the 12 tested biochemical parameters (Fig. 1). The validation of the PLS model, represented by the relationship between observed and predicted values and permutation plots, is depicted in Figure S2. The percentage of variation that can be predicted by the models according to a leave-one-out cross-validation procedure ranges from 90.7% to 71.9% [Q2X(cum)].
Fig. 1. PLS-derived score plots obtained by correlating GC-MS-derived serum metabolite profiles of the pneumonic sheep group (G2, green dots) and the pneumonic sheep groups treated with: Traditional therapy (SG 1), combined traditional therapy and silymarin 280 mg (SG 2), silymarin 280 mg (SG 3), and silymarin 560 mg (SG 4) to the tested biochemical markers (A) MDA, (B) TAC, (C) TG and LDL-cholesterol, (D) total and HDL-cholesterol, (E) kidney function tests (creatinine and urea), (F) blood glucose concentrations, and (G) liver activities (blood serum AST, ALT, GGT). Table 5. Serum metabolite percentile level based on gas chromatography/MS analysis.
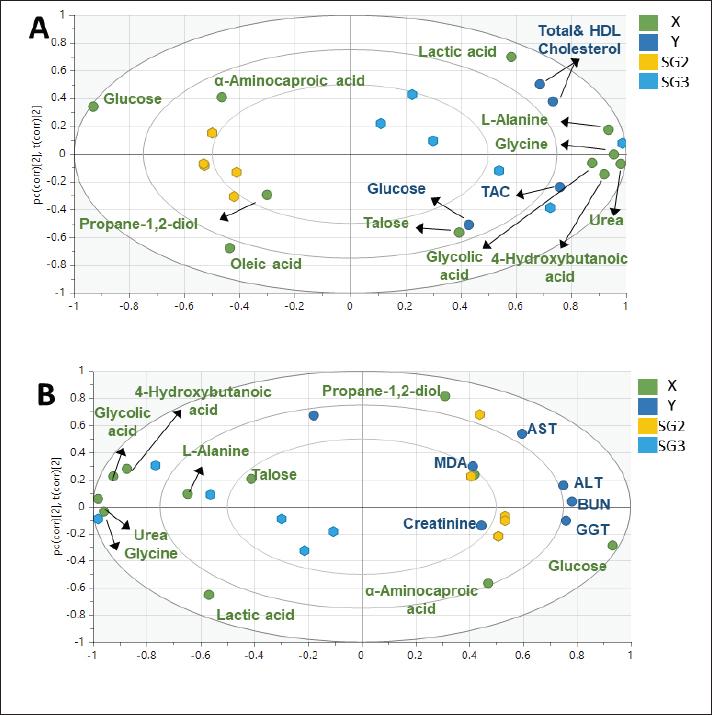
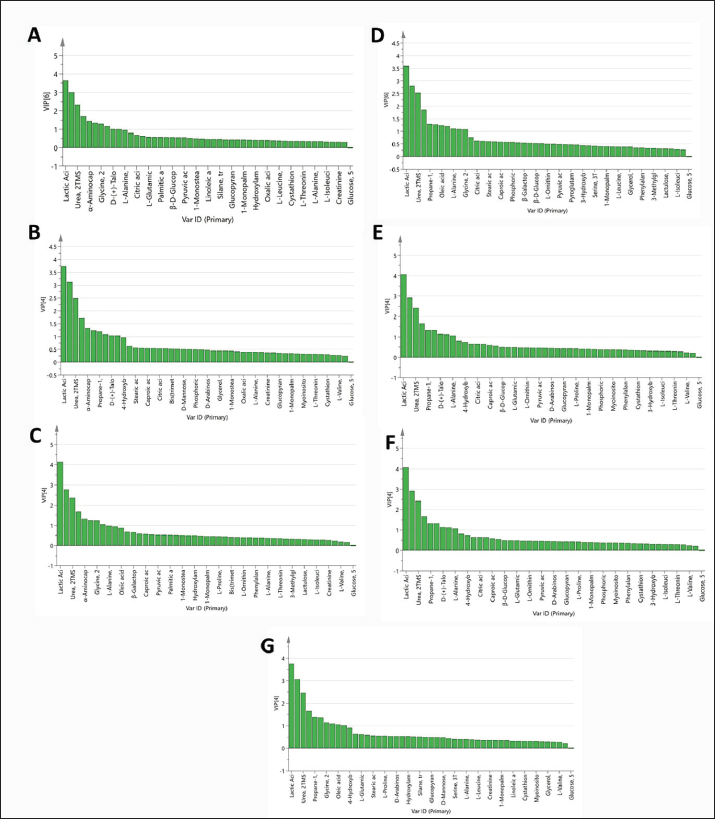
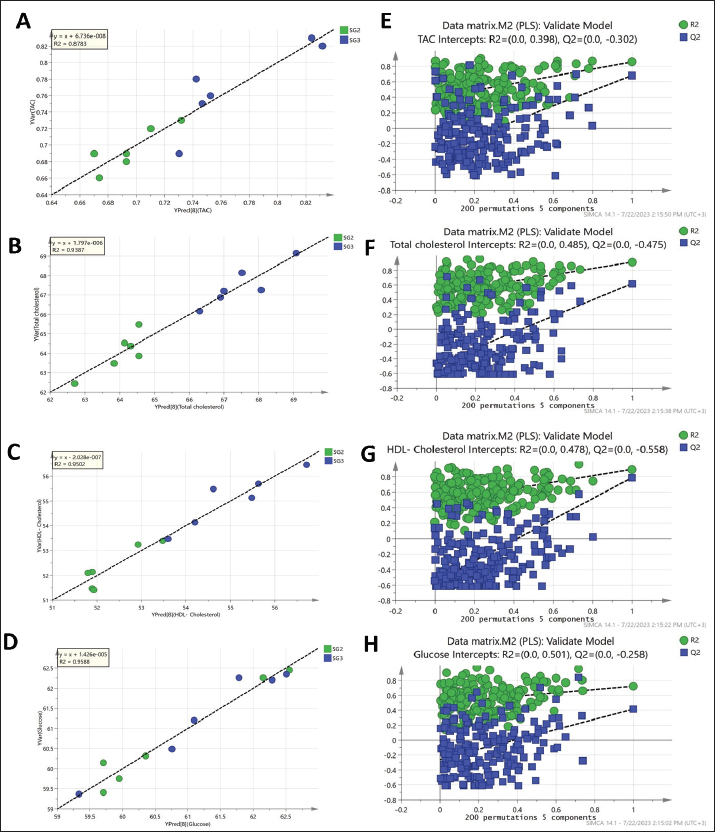
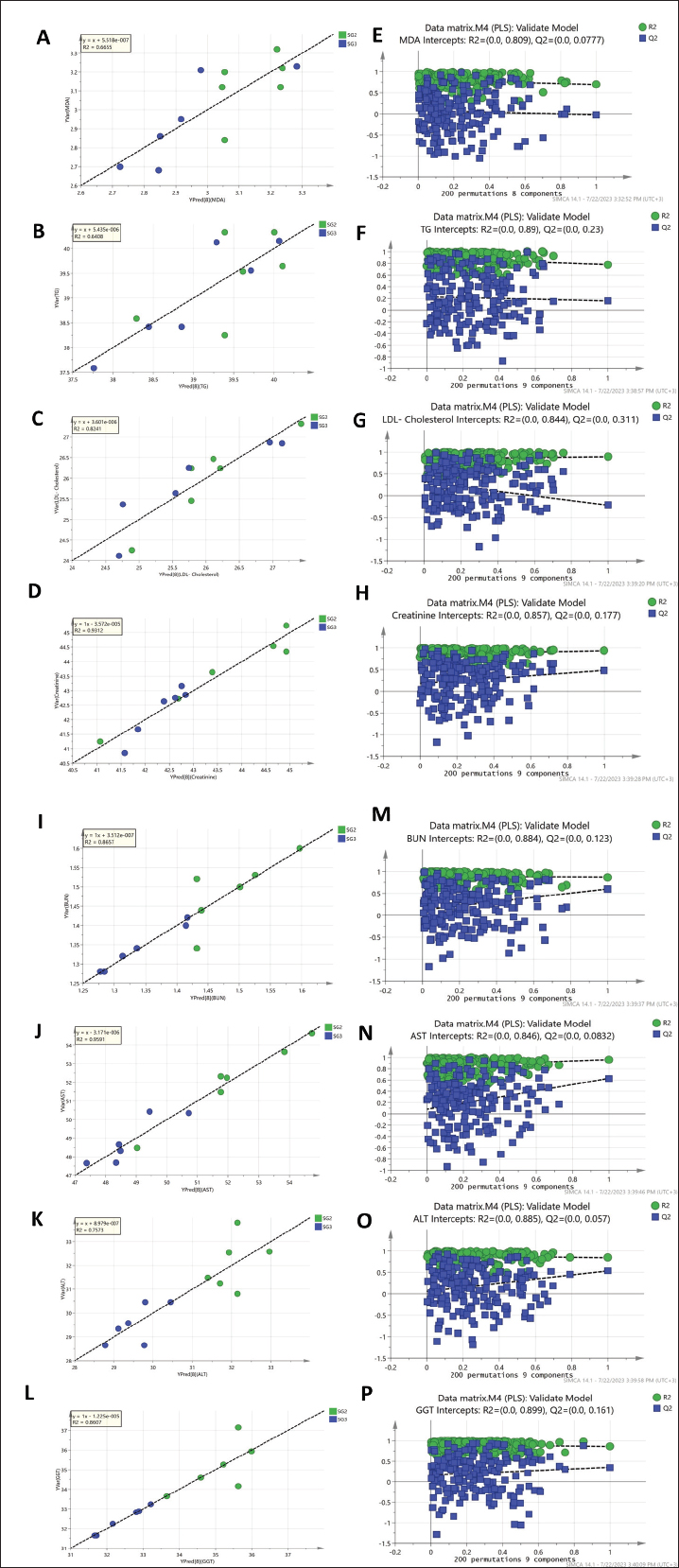
As observed in Figure 1, the PLS models exhibited strong correlations between the GC-MS serum metabolite profiles of the pneumonic sheep treated with the four therapeutic approaches and the pneumonic sheep group (G2) (R2=0.878–0.907), as observed for the regression lines and the significant spread of the samples along the reference lines. Strong relationships exist between the defined and predicted values of the tested biochemical parameters exist. The derived PLS score plots (Fig. 1A) provided a clear distinction between the modeled groups (SG1–SG4), with the pneumonic sheep group (G2) being the most distant from them. Furthermore, all the score plots exhibited trends of either decreasing or increasing order of the samples’ potency in modulating the tested biochemical parameters to restore their values to the normal status. The endogenous metabolites that were deemed crucial in distinguishing between the different therapeutic approaches according to their therapeutic efficacy on restoring the biochemical parameter levels to the normal status were those possessing VIP values > 1 in the VIP plots (Fig. S3). Lactic acid, glycolic acid, urea, glucose, α-aminocaproic acid, propane-1,2-diol, glycine, L-alanine, 4-hydroxybutanoic acid, D-(+)-talose, and oleic acid were considered the most important for the model prediction, which were then used to create two other PLS models to compare the two therapeutic approaches (Fig. 1). Correlations between the two therapeutic approaches (SG2 and SG3) and their respective biochemical parameters were explored using the PLS regression model (Fig. 2). The model was validated using 200 random permutations and showed good model fit (R2=0.956) and predictive power (Q2=0.781). The validation of the PLS model was demonstrated from the regression analysis, permutation plots, and root mean square errors of estimation depicted in Figures S4–S6, respectively.
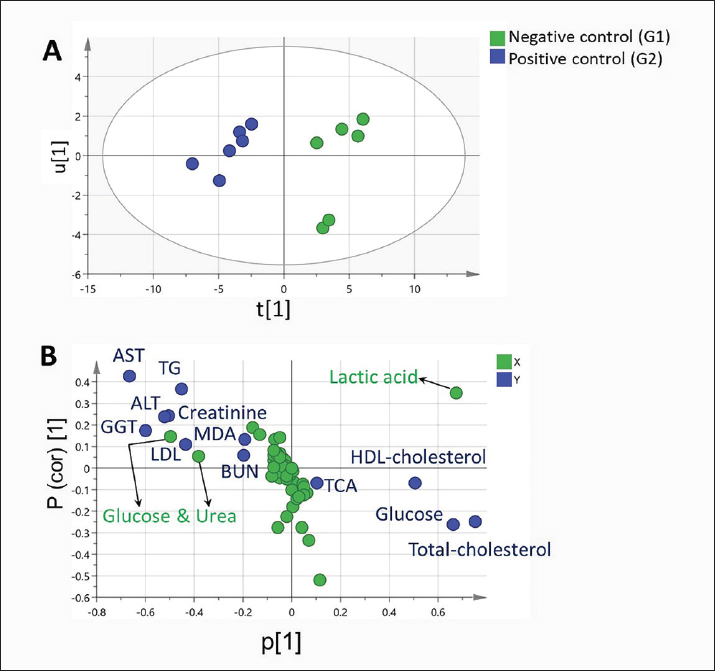
Fig. 2. Two PLS biplots (A and B) correlating therapeutic approaches (SG2, red dots and SG3, violet dots) based on their GC-MS-derived metabolite profiles (X-variables) correlated to the elevated biochemical parameters due to pneumonic condition (TAC, glucose, and total and HDL-cholesterol (Y-variables) and the decreased biochemical parameters due to pneumonic condition as MDA, LDL-cholesterol, creatinine, urea, AST, ALT, and GGT. The derived biplot, (an amalgamation of the information revealed by both the score and loading plots) (Fig. 2) showed that the therapeutic approach relying on a silymarin dose of 280 mg only (SG3) was strongly correlated with the upregulation of TAC, glucose, and total and HDL-cholesterol values as projected close to SG3 samples on the left side of the biplot; however, all of the SG2 samples were remotely distributed on the right side of the biplot. The PLS model (Fig. S1) was initially established by modeling the GC-MS-derived serum metabolite profiles of healthy control (G1) (X-variables) versus pneumonic sheep (G2) to the 12 tested biochemical parameters (Y-variables) to identify endogenous metabolite markers and the biochemical status of each group. The covered variance and prediction power of the model were assessed by R2 and Q2 values, which were computed to be 0.942 and 0.82, respectively, indicating model validity. The PLS score plot (Fig. S1A) showed a clear discrimination between the healthy and pneumonic sheep groups, which was explained by the loading plot (Fig. S1B), which revealed that lactic acid was more elevated in the healthy sheep, whereas urea and creatinine were detected at higher concentrations in the pneumonic sheep. Furthermore, some biochemical parameters were found to be more elevated in the pneumonic sheep group (G2), including liver and kidney functions (AST, ALT, ALT, GGT, creatinine, and urea) as well as oxidative stress, as represented by high MDA values and high LDL-cholesterol. However, TAC, total cholesterol, HDL-cholesterol, and glucose were found at higher concentrations in healthy sheep than in pneumonic sheep. DiscussionThe symptoms observed in Group 2 were previously recorded by Hassan et al. (2019). Microbial resistance to antibiotic administration could be responsible for the higher body temperature and respiratory and pulse rates observed in SG1 (Hassan et al., 2024), whereas SG2–SG4 did not significantly differ from G1. The rapid clinical response to treatment with a high dose of oral silymarin (560 mg) in SG4 could be attributed to its broad-spectrum activity as an anti-inflammatory, anti-oxidative, and antimicrobial agent against Pseudomonas aeruginosa, Staphylococcus aureus, Escherichia coli, and Mycoplasma ovipneumoniae (Ou et al., 2018; Hassan et al., 2024). The higher AST, ALT, GGT, creatinine, and urea concentrations recorded in pneumonic sheep have been previously reported (Hassan et al., 2019; Saeed et al., 2021; Arbaga et al., 2022). Although these biochemical parameters are not specific markers for pneumonia, their increase likely reflects the systemic impact of infection, oxidative stress, and inflammatory mediators that can impair hepatic and renal function (Aytekin et al., 2011). Higher creatinine and urea concentrations could be related to renal damage caused by the excessive release of free radicals exhibited during the inflammatory progression and enhanced protein catabolism (Saleh and Allam, 2014). Interestingly, silymarin treatment improved liver and kidney functions in pneumonic sheep compared with traditional treatment. Our findings were similar to those of previous studies (Saller et al., 2007). It is plausible that silymarin has anti-inflammatory and antioxidant properties that protect against cellular injury (Habib-ur-Rehman et al., 2009; Metwally et al., 2009; Andrade and Tulkens, 2011). Pneumonic sheep had significantly higher triglyceride and LDL-cholesterol levels and lower cholesterol, HDL-cholesterol, and glucose concentrations than healthy sheep. Previous studies have reported similar findings (El-Deeb and Tharwat, 2015). Inflammation can cause hypertriglyceridemia in both humans and animals (Phetteplace et al., 2000). The lower serum cholesterol concentrations in the sheep in this study could have been due to liver injury with subsequent changes to lipoprotein metabolism (El-Deeb and Elmoslemany, 2016). Pneumonic sheep that received silymarin had significantly lower triglyceride and LDL-cholesterol and higher cholesterol, HDL cholesterol, and glucose concentrations. Potential explanations include that silymarin could decrease blood cholesterol concentration by slowing down liver cell cholesterol synthesis and speeding up cholesterol conversion to other molecules ( Huseini et al., 2004). The binding of low-density LDL is also normalized by silymarin (Khazaei et al., 2022). The significant increase in HDL-cholesterol may be due to reduced cholesterol absorption in pneumonic sheep receiving silymarin in the treatment protocol (Sobolová et al., 2006). Furthermore, the differential therapeutic efficacy of the experimental therapeutic approaches on restoring the normal biochemical parameters in pneumonic sheep was demonstrated by the employed supervised pattern recognition approach. One potential explanation is that silymarin could decrease blood cholesterol concentration by slowing down liver cell cholesterol synthesis and speeding up the conversion of cholesterol to other molecules (Tamminen et al., 2018). In addition, silymarin normalizes the binding of LDL (Sobolová et al., 2006). The significant increase in HDL-C may be due to reduced cholesterol absorption in pneumonic sheep receiving silymarin in the treatment protocol (Khazaei et al., 2022). In the current study, pneumonic sheep had significantly higher MDA and lower TAC concentrations than healthy sheep, similar to the results reported by Hassan et al. (2019). The increase in serum MDA concentrations could be due to excessive lipid peroxidation, while the reduction of TAC in pneumonic sheep may be due to its sequestration in lung tissue during the inflammatory process (Del Rio et al., 2005). Pneumonic sheep treated with silymarin showed a significant decrease in MDA and an increase in TAC concentrations compared with sheep treated with traditional treatment. It is possible that silymarin has antioxidant and neutralizing effects on free radicals or toxins (Khazaei et al., 2022). As revealed by the biplot (Fig. 2), the SG3 treatment was more effective in increasing the TAC, glucose, and total and HDL-cholesterol values than the SG2 treatment. Hence, SG2 was more effective in suppressing oxidative stress and restoring the lipid profile and blood sugar levels to their normal levels. An important limitation is the treatment duration with silymarin; the fact that the sheep received silymarin for 7 days may have affected the magnitude and timing of the therapeutic response. Future studies should evaluate longer and shorter regimens to determine whether treatment time modulates the efficacy of the measured outcome. ConclusionDiscriminatory analysis of the metabolomics profile revealed that pneumonic sheep treated with 280 mg oral silymarin had improved health status and metabolomics profile. Silymarin administration, either alone or in combination with traditional treatment, exhibited greater therapeutic improvement than traditional treatment alone. AcknowledgmentsThe authors would like to thank the postgraduate studies and research sector of the University of Sadat City, Egypt, for funding this study , Grant number (Code no. 22 in 29/12/2021). Conflict of interestThe authors declare no conflict of interest. FundingThis study was funded by postgraduate studies and research sector - University of Sadat City, Egypt, under grant number: 22 in (XXX). Authors’ contributionsHany Hassan: Conceptualization, project administration, supervision, and manuscript editing review. Ahmed Kamr: Writing, original draft, review, and editing. Abdel Nasser El-Gendy: Metabolomic analysis, validation resources, original draft writing, review, and editing. Ramiro Toribio: Writing, original draft, review, and editing. Amira R. Khattab: Metabolomic analysis, writing, original draft, review, and editing. Walid Mousa: Investigation and Methodology Hadeer Khaled: Investigation and Methodology Abdelsalam Elkholey: Investigation and Methodology; Mohamed Kasem: Investigation and Methodology Ali Arbaga: Investigation, methodology, writing, original draft, review, and editing. Data availabilityAll data generated and analyzed during this study are included in this manuscript. The raw data are available upon reasonable request from the corresponding author. ReferencesAflatouni, M., Panahi, N., Mortazavi, P., Shemshadi, B. and Kakoolaki, S. 2020. Hepatoprotective Activity of Silymarin in Combination with Clorsulon Against Fasciola hepatica in Naturally Infected Sheep. Kafkas Univ. Vet. Fak. Derg. 26(2), 279–285. Ammar, N., Hassan, H., Abdallah, H., Afifi, S., Elgamal, A., Farrag, A., El-Gendy, A., Farag, M. and Elshamy, A. 2022. Protective effects of naringenin from Citrus sinensis (var. Valencia) peels against CCl4-induced hepatic and renal injuries in rats assessed by metabolomics, histological and biochemical analyses. Nutrients 14(4), 841. Andrade, R.J. and Tulkens, P.M. 2011. Hepatic safety of antibiotics used in primary care. J. Antimicrob. Chemother. 66(7), 1431–1446. Arbaga, A., Hassan, H., Anis, A., Osthman, N. and Kamr, A. 2022. Hematological changes and serum minerals concentrations in pneumonic sheep. BVMJ 42(2), 143–146. Aytekin, I., Mamak, N., Ulucan, A. and Kalinbacak, A. 2011. Clinical, hematological, biochemical, and pathological findings in lambs with peste des petits ruminants. Kafkas. Universitesi. Veteriner. Fakultesi. Dergisi. 17(3), 349–355. Berge, A.C.B., Sischo, W.M. and Craigmill, A.L. 2006. Antimicrobial susceptibility patterns of respiratory tract pathogens from sheep and goats. J. Am. Vet. Med. Assoc. 229(8), 1279–1281. Constable, P. D., Hinchcliff, K. W., Done, S. H., & Grünberg, W. (2016). Veterinary medicine: a textbook of the diseases of cattle, horses, sheep, pigs, and goats Elsevier Health Sciences, Inc. Del Rio, D., Stewart, A.J. and Pellegrini, N. 2005. A review of recent studies on malondialdehyde as toxic molecule and biological marker of oxidative stress. Nutr. Metab. Cardiovasc. Dis. 15(4), 316–328. Dhami-Shah, H., Vaidya, R., Udipi, S., Raghavan, S., Abhijit, S., Mohan, V., Balasubramanyam, M. and Vaidya, A. 2018. Picroside II attenuates fatty acid accumulation in HepG2 cells via modulation of fatty acid uptake and synthesis. Clin. Mol. Hepatol. 24(1), 77. El-Deeb, W.M. and Elmoslemany, A.M. 2016. The diagnostic accuracy of acute phase proteins and proinflammatory cytokines in sheep with pneumonic pasteurellosis. Peer J. , e2161. El-Deeb, W.M. and Tharwat, M. 2015. Lipoproteins profile, acute phase proteins, proinflammatory cytokines and oxidative stress biomarkers in sheep with pneumonic pasteurellosis. Comp. Clin. Path. 24, 581–588. Elshamy, A.I., Abdallah, H.M.I., El Gendy, A.E.N.G., El-Kashak, W., Muscatello, B., De Leo, M. and Pistelli, L. 2019. Evaluation of anti-inflammatory, antinociceptive, and antipyretic activities of Prunus persica var. nucipersica (nectarine) kernel. Planta Med. 85(11/12), 1016–1023. Goli, F., Karimi, J., Khodadadi, I., Tayebinia, H., Kheiripour, N., Hashemnia, M. and Rahimi, R. 2019. Silymarin attenuates ELMO-1 and KIM-1 expression and oxidative stress in the kidney of rats with type 2 diabetes. Indian. J. Clin. Biochem. 34, 172–179. Habib-ur-Rehman, M., Mahmood, T., Salim, T., Afzal, N., Ali, N., Iqbal, J., Tahir, M. and Khan, A. 2009. Effect of silymarin on serum levels of ALT and GGT in ethanol induced hepatotoxicity in albino rats. J. Ayub. Med. Coll. Abbottabad. 21(4), 73–75. Hassan, H., Kamr, A., Mousa, W., Toribio, R., El-Gendy, A.N., Khaled, H., Elkholey, A. and Arbaga, A. 2024. Silymarin Antibacterial Efficacy against some Isolated Bacterial Strains from Pneumonic Sheep-Vitro Study. Pak. Vet. J. 44(2), 280–285. Hassan, H.Y., Kamr, A., Abdelazeim, A. and Khaled, H. 2019. Hemato-Biochemical Response with Highlights on the Role of Oxidants and Antioxidants in Pneumonic Sheep. J. Vet. Mar. Sci. 1(1), 7–14. Huseini, H., Zaree, A.B., Zarch, A. and Heshmat, R. 2004. The effect of herbal medicine Silybum marianum (L.) Gaertn. seed extract on galactose induced cataract formation in rat. J. Med. Plants 3(12), 58–62. Ibrahim, N., Taleb, M., Heiss, A.G., Kropf, M. and Farag, M.A. 2021. GC-MS based metabolites profiling of nutrients and anti-nutrients in 10 Lathyrus seed genotypes: a prospect for phyto-equivalency and chemotaxonomy. Food Biosci. 42, 101183. Khazaei, R., Seidavi, A. and Bouyeh, M. 2022. A review on the mechanisms of the effect of silymarin in milk thistle (Silybum marianum) on some laboratory animals. Vet. Med. Sci. 8(1), 289–301. Laudato, M. and Capasso, R. 2013. Useful plants for animal therapy. OA. Alternative Med. 1(1), 1. Magstadt, D.R., Schuler, A.M., Coetzee, J.F., Krull, A.C., O’Connor, A.M., Cooper, V.L. and Engelken, T.J. 2018. Treatment history and antimicrobial susceptibility results for Mannheimia haemolytica, Pasteurella multocida, and Histophilus somni isolates from bovine respiratory disease cases submitted to the Iowa State University Veterinary Diagnostic Laboratory from 2013 to 2015. J. Vet. Diagn. Invest. 30(1), 99–104. Marmouzi, I., Bouyahya, A., Ezzat, S.M., El Jemli, M. and Kharbach, M. 2021. The food plant Silybum marianum (L.) Gaertn.: phytochemistry, Ethnopharmacology and clinical evidence. J. Ethnopharmacol. 265, 113303. Metwally, M.A.A., El-Gellal, A.M. and El-Sawaisi, S.M. 2006. Effects of silymarin on lipid metabolism in rats. World. Appl. Sci. J. 6, 1634–1637. Ou Q, Weng Y, Wang S, Zhao Y, Zhang F, Zhou J, Wu Z (2018) Silybin alleviates hepatic steatosis and fibrosis in NASH mice by inhibiting oxidative stress and involvement with the Nf-κB pathway. Dig Dis Sci 63:3398–3408, 2010. Pferschy-Wenzig, E.M., Atanasov, A.G., Malainer, C., Noha, S.M., Kunert, O., Schuster, D., Heiss, E.H., Oberlies, N.H., Wagner, H., Bauer, R. and Dirsch, V.M. 2014. Identification of isosilybin a from milk thistle seeds as an agonist of peroxisome proliferator-activated receptor gamma. J. Nat. Prod. 77(4), 842–847. Phetteplace, H.W., Sedkova, N., Hirano, K., Davidson, N.O., Lanza. and Jacoby, S.P. 2000. Escherichia coli sepsis increases hepatic apolipoprotein B secretion by inhibiting degradation. Lipids 35(10), 1079–1086. Raish, M., Ahmad, A., Jan, B.L., Alkharfy, K.M., Mohsin, K., Ahamad, S.R. and Ansari, M.A. 2017. GC-MS-based metabolomic profiling of thymoquinone in streptozotocin-induced diabetic nephropathy in rats. Nat. Prod. Comput. 12(4), 553–558. Rummun, N., Khattab, A.R., Bahorun, T., Farag, M.A. and Neergheen, V.S. 2023. Biochemometric approach to reveal Terminalia bentzoë cytotoxic effect against HepG2 cells in relation to its different organs and extraction solvents as analysed via UPLC-MS. S. Afr. J. Bot. 159, 507–518. Saleh, N.S. and Allam, T.S. 2014. Pneumonia in Sheep: bacteriological and Clinicopathological Studies. Am. J. Res. Commun. 2(211), 73–88. Saller, R., Melzer, J., Reichling, J., Brignoli, R. and Meier, R. 2007. An updated systematic review of the pharmacology of silymarin. CAM 14(2), 70–80. Schrimpe-Rutledge, A.C., Codreanu, S.G., Sherrod, S.D. and McLean, J.A. 2016. Untargeted metabolomics strategies—challenges and emerging directions. J. Am. Soc. Mass Spectrom. 27(12), 1897–1905. Sobolová, L., Škottová, N., Večeřa, R. and Urbánek, K. 2006. Effect of silymarin and its polyphenolic fraction on cholesterol absorption in rats. Pharmacol. Res. 53, 104–112. Tamminen, L.M., Emanuelson, U. and Blanco-Penedo, I. 2018. Systematic review of phytotherapeutic treatments for different farm animals under European conditions. Front. Vet. Sci. 5(22), 1–10. Thompson. 2019. Respiratory diseases in sheep. Vet. Pract. Today 7(4), 1–2. Wu, S.C., Han, F., Song, M.R., Chen, S., Li, Q., Zhang, Q., Zhu, K. and Shen, J.Z. 2019. Natural flavones from Morus alba against methicillin-resistant Staphylococcus aureus via targeting the proton motive force and membrane permeability. J. Agri. Food Chem. 67(36), 10222–10234. Supplementary Material
Fig. S1. PLS model correlating GC/MS-metabolite profiles of the serum of healthy control group (G 1) and pneumonic control group (G2) to the tested biochemical parameters as MDA and TAC, lipid profile (TG, cholesterol, HDL-cholesterol, LDL-cholesterol), blood glucose concentrations, liver activities (blood serum AST, ALT, GGT, and kidney function tests (creatinine and urea). (A) PLS score plot. (B) Loading plot showing the covariance p[1] against the correlation p(cor) [1] of the discriminating component variables of the PLS model.
Fig. S2. Regression analysis of the PLS predictive model for each y variable modeled in the PLS models shown in Figure 1 and their respective permutation plots. (A and E) MDA, (B and F) TAC, (C and G) TG, (D and H) LDL-cholesterol, (J and N) total cholesterol, (L and O) HDL-cholesterol, (L and P) creatinine, (M and Q) urea, (R and V) blood glucose concentrations, (S and X) blood serum AST, (T and Y) ALT, and (U and Z) GGT.
Fig. S3. VIP plots (A–G) obtained from the PLS models in Figure 1A–G.
Fig. S4. Regression analysis of the PLS predictive model for each y variable modeled in the PLS model shown in Figure 2 and their respective permutation plots. (A and E) TAC, (B and F) total cholesterol, (C and G) HDL-cholesterol, and (D and H) blood glucose concentrations.
Fig. S5. Regression analysis of the PLS predictive model for each y variable modeled in the PLS model shown in Figure 2 and their respective permutation plots. (A and E) MDA, (B and F) TG, (C and G) LDL cholesterol, (D and H) creatinine, (I and M) urea, (J and N) blood serum AST, (K and O) ALT, and (L and P) GGT, respectively.
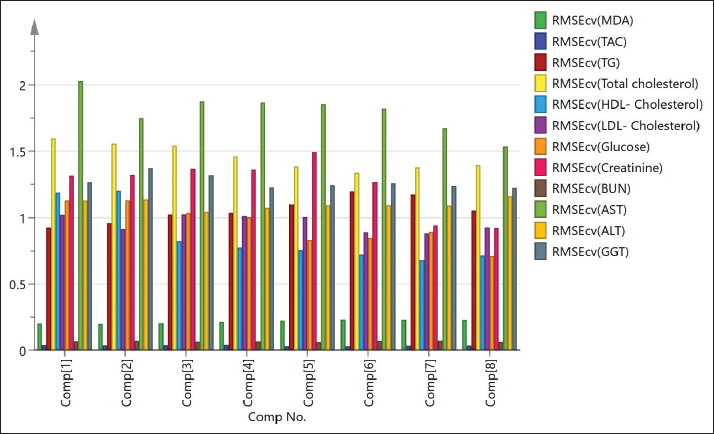
Fig. S6. Root mean square errors of cross-validation (RMSEcv) of the PLS models shown in Figures S4 and S5 for all the tested biochemical parameters as TAC, total cholesterol, HDL-cholesterol, blood glucose concentrations, MDA, TG, LDL-cholesterol, creatinine, urea, blood serum AST, ALT, and GGT. | ||
| How to Cite this Article |
| Pubmed Style Hassan H, Kamr A, El-gendy AN, Toribio R, Khattab AR, Mousa W, Khaled H, Elkholey A, Kasem M, Arbaga A. Clinicobiochemical and gas chromatography-mass spectrometry-based serum metabolomics for determining the therapeutic efficacy of silymarin in pneumonic Barki sheep. Open Vet. J.. 2025; 15(12): 6268-6286. doi:10.5455/OVJ.2025.v15.i12.11 Web Style Hassan H, Kamr A, El-gendy AN, Toribio R, Khattab AR, Mousa W, Khaled H, Elkholey A, Kasem M, Arbaga A. Clinicobiochemical and gas chromatography-mass spectrometry-based serum metabolomics for determining the therapeutic efficacy of silymarin in pneumonic Barki sheep. https://www.openveterinaryjournal.com/?mno=278183 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.11 AMA (American Medical Association) Style Hassan H, Kamr A, El-gendy AN, Toribio R, Khattab AR, Mousa W, Khaled H, Elkholey A, Kasem M, Arbaga A. Clinicobiochemical and gas chromatography-mass spectrometry-based serum metabolomics for determining the therapeutic efficacy of silymarin in pneumonic Barki sheep. Open Vet. J.. 2025; 15(12): 6268-6286. doi:10.5455/OVJ.2025.v15.i12.11 Vancouver/ICMJE Style Hassan H, Kamr A, El-gendy AN, Toribio R, Khattab AR, Mousa W, Khaled H, Elkholey A, Kasem M, Arbaga A. Clinicobiochemical and gas chromatography-mass spectrometry-based serum metabolomics for determining the therapeutic efficacy of silymarin in pneumonic Barki sheep. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6268-6286. doi:10.5455/OVJ.2025.v15.i12.11 Harvard Style Hassan, H., Kamr, . A., El-gendy, . A. N., Toribio, . R., Khattab, . A. R., Mousa, . W., Khaled, . H., Elkholey, . A., Kasem, . M. & Arbaga, . A. (2025) Clinicobiochemical and gas chromatography-mass spectrometry-based serum metabolomics for determining the therapeutic efficacy of silymarin in pneumonic Barki sheep. Open Vet. J., 15 (12), 6268-6286. doi:10.5455/OVJ.2025.v15.i12.11 Turabian Style Hassan, Hany, Ahmed Kamr, Abdel Nasser El-gendy, Ramiro Toribio, Amira R. Khattab, Walid Mousa, Hadeer Khaled, Abdelsalam Elkholey, Mohamed Kasem, and Ali Arbaga. 2025. Clinicobiochemical and gas chromatography-mass spectrometry-based serum metabolomics for determining the therapeutic efficacy of silymarin in pneumonic Barki sheep. Open Veterinary Journal, 15 (12), 6268-6286. doi:10.5455/OVJ.2025.v15.i12.11 Chicago Style Hassan, Hany, Ahmed Kamr, Abdel Nasser El-gendy, Ramiro Toribio, Amira R. Khattab, Walid Mousa, Hadeer Khaled, Abdelsalam Elkholey, Mohamed Kasem, and Ali Arbaga. "Clinicobiochemical and gas chromatography-mass spectrometry-based serum metabolomics for determining the therapeutic efficacy of silymarin in pneumonic Barki sheep." Open Veterinary Journal 15 (2025), 6268-6286. doi:10.5455/OVJ.2025.v15.i12.11 MLA (The Modern Language Association) Style Hassan, Hany, Ahmed Kamr, Abdel Nasser El-gendy, Ramiro Toribio, Amira R. Khattab, Walid Mousa, Hadeer Khaled, Abdelsalam Elkholey, Mohamed Kasem, and Ali Arbaga. "Clinicobiochemical and gas chromatography-mass spectrometry-based serum metabolomics for determining the therapeutic efficacy of silymarin in pneumonic Barki sheep." Open Veterinary Journal 15.12 (2025), 6268-6286. Print. doi:10.5455/OVJ.2025.v15.i12.11 APA (American Psychological Association) Style Hassan, H., Kamr, . A., El-gendy, . A. N., Toribio, . R., Khattab, . A. R., Mousa, . W., Khaled, . H., Elkholey, . A., Kasem, . M. & Arbaga, . A. (2025) Clinicobiochemical and gas chromatography-mass spectrometry-based serum metabolomics for determining the therapeutic efficacy of silymarin in pneumonic Barki sheep. Open Veterinary Journal, 15 (12), 6268-6286. doi:10.5455/OVJ.2025.v15.i12.11 |