| Research Article | ||
Open Vet. J.. 2026; 16(1): 318-326 Open Veterinary Journal, (2026), Vol. 16(1): 318-326 Research Article Hypofractionated reirradiation for feline nasal epithelial tumors: A retrospective studyHirona Ueno1, Yui Kondo1, Yuta Nishiyama2*, Takayuki Katayama1, Yusuke Banno2, Hideki Kayanuma1 and Takuya Maruo11Laboratory of Veterinary Radiology, College of Veterinary Medicine, Azabu University, Sagamihara, Japan 2Veterinary Teaching Hospital, Azabu University, Sagamihara, Japan *Corresponding Author: Yuta Nishiyama. Veterinary Teaching Hospital, Azabu University, Sagamihara, Japan. Submitted: 18/08/2025 Revised: 27/11/2025 Accepted: 09/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
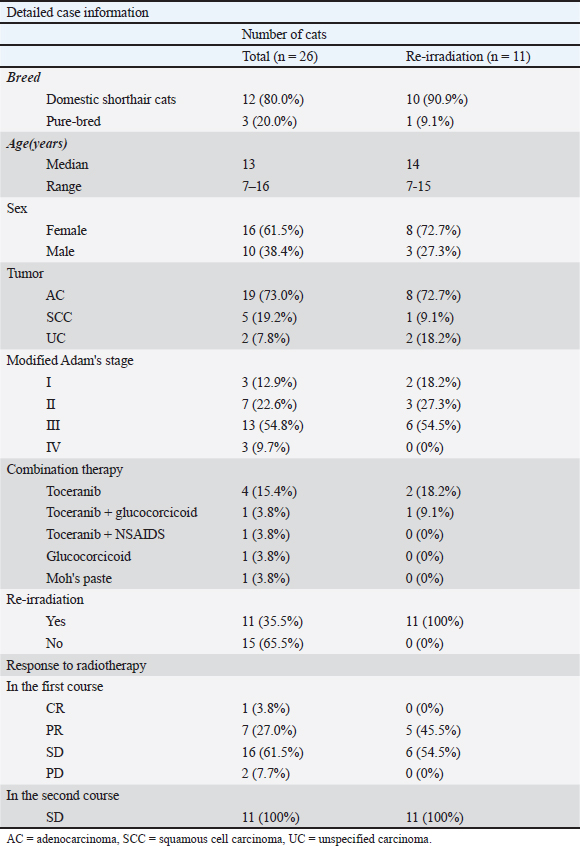
AbstractBackground: Feline nasal epithelial tumors are locally invasive and radiosensitive, and radiotherapy is the standard treatment. However, recurrence is common, and the role of reirradiation in cats remains unclear. Aim: This retrospective study aimed to evaluate the efficacy and safety of hypofractionated reirradiation in cats with recurrent nasal epithelial tumors. Methods: The medical records of cats treated with hypofractionated radiotherapy for intranasal epithelial tumors at Azabu University Veterinary Teaching Hospital between 2014 and 2022 were reviewed. Follow-up continued until August 2023. Results: Twenty-six cats were treated, 11 of which underwent reirradiation after clinical progression. The median interval between treatments was 356 days. In the first course, responses included complete (n=1), partial (n=7), stable (n=16), and progressive (n=2) diseases. In the second course, all cats achieved stable disease. The median overall survival was significantly longer in the reirradiation group than in the nonreirradiation group (562 vs. 58 days, p < 0.001). Although not statistically significant (p=0.08), adenocarcinomas tended to have longer OS than squamous cell carcinomas. Acute adverse events (Veterinary Radiation Therapy Oncology Group grade 1–2), including conjunctivitis, alopecia, and dermatitis, were observed. Late adverse events, such as leukotrichia and hyperpigmentation, did not affect survival. Two cats developed grade 3 late toxicities (cutaneous or oral–cutaneous–nasal fistulas) associated with bone lysis and short inter-treatment intervals. Conclusion: Hypofractionated reirradiation may be a viable salvage option for feline nasal epithelial tumors, particularly in cases with adenocarcinoma or early-stage disease, with acceptable toxicity when patients are carefully selected. Keywords: Adverse event, Feline, Nasal tumor, Recurrence, Reirradiation. IntroductionFeline nasal and paranasal sinus tumors account for approximately 1%–8.4% of all feline tumors (Moulton, 1990; Cox et al., 1991). Among tumors of the nasal cavity, nasal epithelial tumors are the second most common type (Cox et al., 1991). These tumors are locally invasive and can present with clinical signs, such as nasal discharge, epistaxis, dyspnea, anorexia, and facial deformity (Madewell et al., 1976; Moulton, 1990; Henderson et al., 2004). Their anatomical location and invasive nature make complete surgical excision challenging (Weeden and Degner, 2016). Reliable reports on the efficacy of chemotherapy alone for treating feline nasal epithelial tumors are unavailable. Radiotherapy remains the most effective and widely accepted standard treatment for these tumors (Lana and Turek, 2020). Recent advances in radiotherapy, including stereotactic techniques, have also shown promising outcomes in feline nasal and nasopharyngeal tumors (Reczynska et al., 2022). Radiotherapy outcomes for feline nasal tumors show that definitive radiotherapy achieves better results than palliative radiotherapy (Fujiwara-Igarashi et al., 2014; Yoshikawa et al., 2021). Definitive irradiation, however, requires more frequent anesthesia because of the large number of fractions. Palliative radiotherapy is often selected when frequent anesthesia is impractical owing to the overall condition of the patient or the preferences of the owner. Given the relatively short progression-free survival associated with palliative treatment (Fujiwara-Igarashi et al., 2014; Yoshikawa et al., 2021), developing an effective strategy for managing recurrence is essential to prolong overall survival (OS). Reirradiation is considered a treatment option for recurrence (Fujiwara-Igarashi et al., 2014; Giuliano and Dobson, 2020; Yoshikawa et al., 2021), although the total dose may exceed the tolerance of normal tissues. Evidence on reirradiation for intranasal tumors in cats remains limited, and its effectiveness is unclear (Fujiwara-Igarashi et al., 2014; Yoshikawa et al., 2021). In contrast, several large studies in dogs with recurrent nasal tumors have demonstrated the efficacy and safety of this approach (Turrel and Théon, 1988; Bommarito et al., 2011; Gieger et al., 2013). Based on these findings, we hypothesized that reirradiation might be effective for feline nasal tumors. This retrospective study aimed to assess the efficacy and safety of reirradiation with hypofractionated radiotherapy for nasal epithelial tumors in cats. Materials and MethodsCasesCats with intranasal epithelial tumors treated with hypofractionated radiotherapy at Azabu University Veterinary Teaching Hospital between 2014 and 2022 were evaluated retrospectively. Follow-up continued until August 2023. Data collected from medical records and follow-up surveys included breed, sex, age, tumor type, tumor stage, radiotherapy protocols (first course: number of fractions, dose per fraction, and total dose; reirradiation and additional reirradiation: number of fractions, dose per fraction, total dose, and interval between courses), cumulative dose from the first irradiation prescribed to the tissue (minimum, maximum, and mean dose of the irradiation volume [IV] and mean dose to the eyeball), calculated using radiotherapy planning software (XiO®; Elekta, Tokyo, Japan), combination therapy, response to radiotherapy, adverse events, cause of death, and OS. General condition and tumor stage were assessed through physical examination, blood testing, radiography, and computed tomography (CT). Cats that did not complete the first course of radiotherapy were excluded from statistical analysis. For reirradiation cases, the reasons that led to reirradiation were investigated. Tumor stage was determined using the modified Adams staging system (Adams et al., 2009). Treatment response was assessed using the Canine Response Evaluation Criteria in Solid Tumors version 1.0 based on CT images (Nguyen et al., 2015). CT images obtained on the last day of the radiotherapy course were compared with those from the first day. Adverse events related to radiotherapy and chemotherapy were evaluated according to the Veterinary Radiation Therapy Oncology Group criteria (Ladue and Klein, 2001) and the Veterinary Cooperative Oncology Group–Common Terminology Criteria for Adverse Events (Veterinary Cooperative Oncology Group, 2016). Routine follow-up was not performed in all cases. OS was defined as the time from the first day of radiotherapy until death. RadiotherapyTreatment began with subcutaneous administration of 0.025 mg/kg atropine sulfate hydrate (Atropine Sulfate Injection; Mitsubishi Tanabe Pharma Company, Osaka, Japan) 15 minutes before induction of anesthesia. Intubation was performed following intravenous administration of 6–8 mg/kg propofol (Rapinovet; Takeda Schering-Plough Animal Health Corporation, Osaka, Japan) or 5 mg/kg alphaxalone (Alfaxan; Meiji Seika Pharma Company Ltd., Tokyo, Japan). General anesthesia was maintained with inhaled isoflurane (Isoflurane for animals; MSD Animal Health, Tokyo, Japan). Cats under general anesthesia were positioned in head-positioning devices (Mori et al., 2009; Maruo et al., 2013; Nemoto et al., 2015). Treatment protocols were planned using CT imaging (Asteon®; Canon, Tokyo, Japan) and radiotherapy planning software (XiO®; Elekta, Tokyo, Japan). Beam shaping was achieved with a multileaf collimator featuring 1 cm leaf width at the isocenter. To correct positioning errors, CT images were obtained from the opposite side of the linear accelerator across the treatment couch before each treatment session. A new plan was created if the tumor volume showed significant changes. In this study, the clinical target volume was equivalent to the gross tumor volume or mass lesion, as the nasal cavity is surrounded by bone. The gross tumor volume was defined as the soft tissue density mass lesion, including areas of necrosis. When lymphadenopathy or cytologically confirmed lymph node metastases were present, the gross tumor volume also included the affected lymph nodes. For the first course of radiotherapy, the planning target volume was set at a 5 mm isotopic expansion from the clinical target volume. For reirradiation, the planning target volume was set at a 0 mm margin. Hypofractionated radiotherapy was delivered using three-dimensional conformal radiation therapy with a 6 MeV X-ray linear accelerator (Primus; Canon). Any radiotherapy administered after completion of the first course was defined as reirradiation. In all cases, hypofractionated radiotherapy was performed once per week. Statistical analysisSex, age, tumor type, modified Adams stage, radiotherapy protocols (with or without reirradiation), and response to radiotherapy were analyzed to assess their association with OS. For age-related analysis, cases were divided into two groups based on the median age. The modified Adams stage was compared between stages 1–2 and stages 3–4. Median survival time was evaluated using Kaplan–Meier survival curves. During the follow-up period, surviving or lost-to-follow-up cases were treated as censored. Furthermore, a multivariate Cox proportional hazards analysis was performed to assess the independent effects of reirradiation, tumor stage, and tumor type on OS. Statistical significance was determined using the log-rank test, with p < 0.05 considered significant. All analyses were performed using the Statistical Package for the Social Sciences, version 28 (IBM Corp., Armonk, NY). Ethical approvalNot needed for this study. ResultsTwenty-six cats were included in this study (Table 1). Breeds consisted of domestic shorthair (n=22), American Shorthair, Persian, Somali, and Scottish Fold (n=1 each). The cohort included 15 spayed females, one intact female, and 10 neutered males. The median age was 13 years (range, 7–16 years). Tumor types were histopathologically diagnosed as adenocarcinoma (n=19), squamous cell carcinoma (n=5), and unspecified carcinoma (n=2). Eleven cats underwent reirradiation, including eight with adenocarcinoma, one with squamous cell carcinoma, and two with unspecified carcinoma. The modified Adams stage, determined from CT imaging before radiotherapy, was stage I (n=3), stage II (n=7), stage III (n=13), and stage IV (n=3). No cases had distant metastasis. Table 1. Summary of signalments.
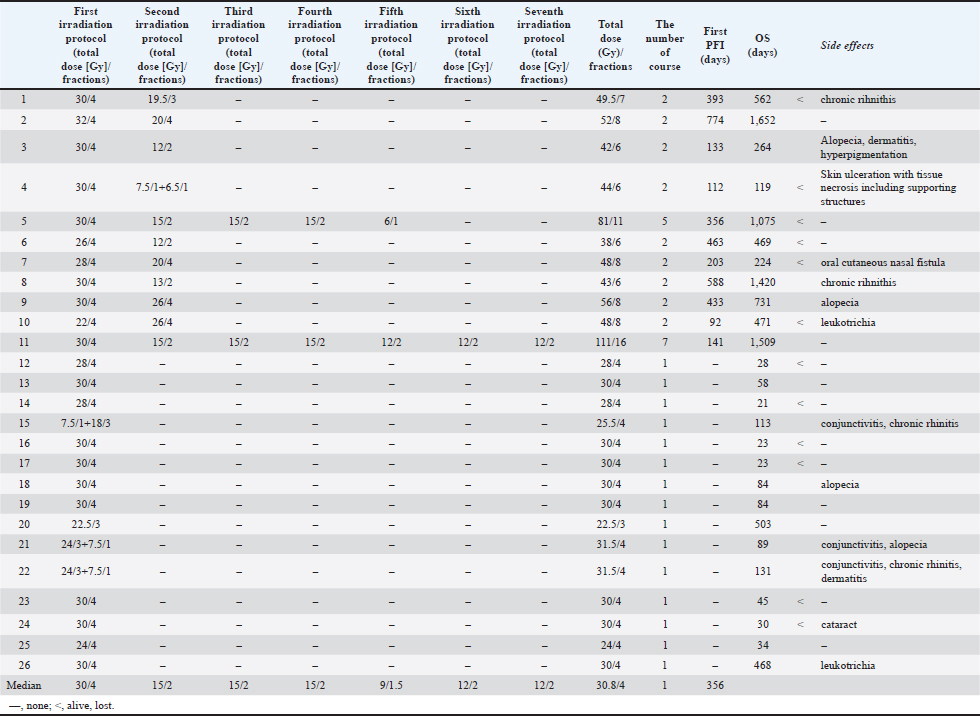
In the first course of radiotherapy, all cats received 4 weekly fractions (Table 2). The median dose per fraction was 7.5 Gy (range, 5.5–8.0 Gy), and the median total dose was 30.0 Gy (range, 22.0–32.0 Gy). Eleven cats underwent reirradiation because of clinical worsening or recurrence after completion of the first course. In the second course, these cats received a median of two fractions (range, 2–4 fractions), with a median dose per fraction of 6.5 Gy (range, 5.0–7.5 Gy) and a median total dose of 15.0 Gy (range, 12.0–26.0 Gy). The median interval between the first and second courses was 356 days (range, 92–774 days). The median OS was 562 days for the reirradiation group and 58 days for the non-reirradiation group. For cases that received reirradiation, the median cumulative total dose from the first to the second course was 45.0 Gy (range, 38.0–56.0 Gy). Two cats received additional reirradiation: Case 5 underwent five courses with a cumulative total dose of 81.0 Gy, and Case 11 underwent seven courses with a cumulative total dose of 111.0 Gy. Details for the eleven reirradiated cases are summarized in Table 2. Table 2. Summary of treatments.
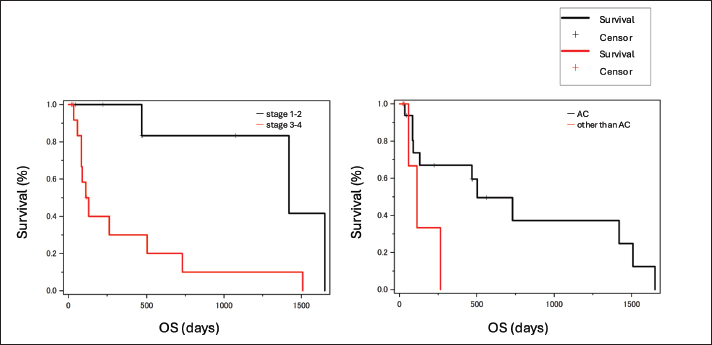
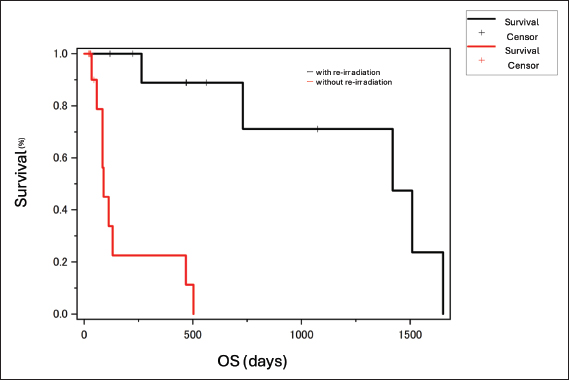
The median cumulative dose to the tissue was calculated using radiotherapy planning software. For the first course (n=31), the minimum, maximum, and mean doses to the IV of the tumor were 11.2 Gy (range, 4.2–21.3 Gy), 30.4 Gy (range, 22.9–32.1 Gy), and 27.7 Gy (19.9–28.6 Gy), respectively. The mean dose to the eyeball on the affected side was 22.9 Gy (range, 17.0–28.8 Gy), and to the contralateral eyeball was 18.1 Gy (range, 8.8–26.4 Gy). From the first to the second course (n=11), the minimum, maximum, and mean doses to the IV of the tumor were 17.2 Gy (range, 4.8–21.6 Gy), 47.5 Gy (range, 13.9–57.1 Gy), and 41.1 Gy (range, 10.0–50.6 Gy), respectively. The mean dose to the eyeball on the affected side was 29.2 Gy (range, 4.5–40.9 Gy), and to the contralateral eyeball was 23.3 Gy (range, 30.6–30.3 Gy). From the first to the fifth course (Case 5), the minimum, maximum, and mean doses to the IV of the tumor were 31.1, 100.1, and 86.4 Gy, respectively. The mean dose to the eyeball on the affected side was 57.8 Gy, and to the contralateral eyeball was 45.3 Gy. From the first to the seventh course (Case 11), the minimum, maximum, and mean doses to the IV of the tumor were 37.6, 82.6, and 70.6 Gy, respectively. The mean dose to the eyeball on the affected side was 33.5 Gy, and to the contralateral eyeball was 15.9 Gy. Responses to the first course of radiotherapy were assessed as complete response (n=1), partial response (n=7), stable disease (n=16), and progressive disease (n=2). In the second course, all responses were stable disease (n=11). Acute adverse events classified as Veterinary Radiation Therapy Oncology Group grade 1–2 occurred in four of 15 cats without reirradiation (conjunctivitis, n=3; alopecia, n=2; dermatitis, n=3) and in two of 11 cats with reirradiation (alopecia, n=2; dermatitis, n=1). Late adverse events classified as grade 1–2 were observed in two of 15 cats without reirradiation (cataract, n=1; leukotrichia, n=1) and in two of 11 cats with reirradiation (leukotrichia, n=1; hyperpigmentation, n=1). These events were tolerable and did not affect survival. However, severe late adverse events (grade 3) occurred in two of 11 cats with reirradiation: one developed a cutaneous fistula, and one developed an oral–cutaneous–nasal fistula. One cat (Case 4), diagnosed with unspecified carcinoma classified as modified Adams stage III with facial deformity and bone lysis, developed a cutaneous fistula shortly after reirradiation. Partial response was achieved in the first course of 30 Gy in four fractions. Epistaxis was subsequently observed, and at the request of the owner, the cat underwent reirradiation with 15 Gy in two fractions, 112 days after completing the first course. The overall total dose was 44.0 Gy. The minimum, maximum, and mean cumulative doses to the IV of the tumor were 12.5, 45.5, and 39.6 Gy, respectively. The mean cumulative dose to the eyeball on the affected side was 31.3 Gy, and to the contralateral eyeball was 25.4 Gy. One cat (Case 7), diagnosed with adenocarcinoma classified as modified Adams stage II, developed an oral–cutaneous–nasal fistula. Partial response was achieved in the first course of 28 Gy in four fractions. Although the nasal cavity lesion was controlled, a new lesion with partial nasal bone lysis developed subcutaneously dorsal to the initial site. Reirradiation was therefore performed with 20 Gy in four fractions, 203 days after completing the first course, with a bolus placed over the lesion, as it was primarily located in the superficial layer. The overall total dose was 48.0 Gy. The minimum, maximum, and mean cumulative doses to the IV of the tumor were 18.1, 49.0, and 43.1 Gy, respectively. The mean cumulative dose to the eyeball on the affected side was 19.7 Gy, and to the contralateral eyelid was 11.4 Gy. Although the exact onset was unclear, the primary care veterinarian reported that the oral–cutaneous–nasal fistula appeared several months after reirradiation. During the observation period, 14 cats (53.8%) died, and one cat (3.8%) was confirmed alive at the end of follow-up. Eleven cases were lost to follow-up. No necropsies were performed. Causes of death included primary tumor-related disease in the nasal cavity (n=3), gastrointestinal tumors (n=1), renal failure (n=1), and cluster seizures (n=1). Reirradiation was performed due to recurrence of symptoms or changes in appearance in 11 cats, including some that underwent more than one reirradiation course. No significance differences in OS were observed when sex, age, or response to radiotherapy were analyzed. Survival time did not differ significantly between tumor types (p=0.08; Fig. 1, left), although adenocarcinomas tended to have longer survival than squamous cell carcinomas. Cats with stages 1–2 disease had significantly longer OS than those with stages 3–4 (p=0.039; Fig. 1, right). OS was also significantly longer in cats treated with reirradiation compared with those that did not receive reirradiation (p < 0.001; Fig. 2). In a multivariate Cox proportional hazards analysis including reirradiation, tumor stage, and tumor type, reirradiation remained an independent prognostic factor for OS (HR=0.07, 95% CI: 0.01–0.56, p=0.013). Neither tumor stage (p=0.20) nor tumor type (p=0.15) was significantly associated with survival.
Fig. 1. The median OS for stages 1–2 (black) and stages 3–4 (red) was 469 and 89 days, respectively (left). OS for stages 1–2 was significantly longer than for stages 3–4 (p=0.039). The median OS for AC (red) and non-AC tumors (black) was 346 and 86 days, respectively. Although the difference was not statistically significant (p=0.08), adenocarcinomas tended to have a longer survival time than squamous cell carcinomas. AC, adenocarcinoma; OS, overall survival.
Fig. 2. The median OS for cats without reirradiation (red) and with reirradiation (black) was 58 and 562 days, respectively. OS for cats with reirradiation was significantly longer than for those without reirradiation (p < 0.001). OS, overall survival. DiscussionReirradiation is a treatment option for recurrent tumors (Stewart, 1999). Yoshikawa et al. (2021) reported longer survival in cats receiving definitive-intent reirradiation (median survival time 824 days) compared with non-reirradiated cats (434 days, p=0.028), whereas palliative reirradiation showed unclear benefit (Yoshikawa et al., 2021). Fujiwara-Igarashi et al. (2014) found no significant survival difference between cats that underwent reirradiation and those that did not (median survival time 450 vs. 432 days, p=0.910), although their outcomes were better than in earlier reports, suggesting possible benefit in selected cases (Fujiwara-Igarashi et al., 2014). In dogs, hypofractionated reirradiation is a viable option for nasal tumors (Gieger et al., 2013). The discrepancy in reported outcomes between dogs and cats is likely related to differences in case numbers. In the present study, OS was significantly longer in the reirradiation group than in the non-reirradiation group. Furthermore, multivariate Cox proportional hazards analysis including reirradiation, tumor stage, and tumor type demonstrated that reirradiation remained the only significant independent prognostic factor for OS (HR=0.07, 95% CI: 0.01–0.56, p=0.013), whereas tumor stage and type were not significant. However, published data on reirradiation for feline nasal tumors remain limited. Variations in case numbers, total dose, and fractionation schedules may account for differences between our results and those of previous studies. When lesions progress after completion of hypofractionated radiotherapy for feline nasal epithelial tumors, reirradiation may help suppress further progression and extend survival. In general, tumor type correlates with prognosis (Rasotto et al., 2017). In dogs, carcinomas typically have poorer outcomes than sarcomas (Adams et al., 2009). Several previous studies have reported that tumor type does not influence survival after radiotherapy for feline nasal tumors (Fujiwara-Igarashi et al., 2014; Giuliano and Dobson, 2020). In the present study, adenocarcinomas tended to have a longer OS than squamous cell carcinomas (p=0.08), suggesting that adenocarcinomas may have a more favorable prognosis. Although this trend was not statistically significant in the multivariate analysis (p=0.15), the same direction of association was observed, implying that tumor type could still influence survival outcomes. Further studies with a larger number of cases are needed to clarify this relationship. The differences observed in this study may reflect the narrow margin setting, variations in tumor invasiveness, and the substantial impact of reirradiation. (Al-Mamgani et al., 2021) Tumor stage correlates with prognosis (Adams et al., 2009). In this study, OS was significantly shorter in cats diagnosed with modified Adams stage III or IV disease. CT staging appears to be more accurate than radiographic staging for prognostication in dogs (Kondo et al., 2008; Adams et al., 2009). Some reports on feline nasal tumors indicate that cats classified as modified Adams stage I have a better prognosis with radiation therapy or chemotherapy (Sfiligoi et al., 2007), whereas other studies found no significant prognostic difference (Fujiwara-Igarashi et al., 2014; Yoshikawa et al., 2021). The absence of a significant difference in some reports may be related to the fact that this staging system was not specifically developed for cats and to the small number of cases analyzed. Therefore, increasing the number of cases and developing a staging system specific to cats would be desirable. Reirradiation increases the total radiation dose and the risk of radiation-related adverse events (Nieder, 2019). Although most adverse events observed in this study were tolerable, two of the 11 reirradiated cases developed fistulas. In human medicine, understanding the recovery from acute or occult radiation damage to critical organs is essential for safely implementing reirradiation, and organ-specific recovery is being investigated (Nieder et al., 2000). In general, acutely responding tissues recover from radiation injury within a few months and can therefore tolerate another full course of radiation. In contrast, the heart, bladder, and kidney show no long-term recovery. The skin, mucosa, lung, and spinal cord can partially recover from subclinical injury, with the extent of recovery depending on the organ type, the initial dose, and, to a lesser extent, the interval between radiation courses (Nieder et al., 2000). However, comparable research is lacking in the veterinary field. A previous study evaluating reirradiation in dogs and cats reported a significantly higher risk of complications in cases with squamous cell carcinoma (p=0.015) and in those with a reirradiation field size greater than 30 cm2 (p=0.056). The risk of complications was significantly lower when the interval between irradiation courses exceeded 5 months (p=0.022) (Turrel and Théon, 1988). In the present study, the two cats that developed fistulas had similar total doses, tissue doses, and irradiated target volumes compared with other reirradiated cases. However, their intervals between courses were shorter than the median (112 and 201 days vs. 356 days). The tumor in the cat with a cutaneous fistula showed subcutaneous infiltration with bone lysis and facial deformation, suggesting that preexisting osteolysis from tumor infiltration may have contributed to fistula formation. In cases with subcutaneous infiltration, bone lysis, and facial deformation, careful consideration should be given to the timing of reirradiation, with sufficient intervals between courses to minimize the risk of severe complications. The limitations of this study include its retrospective design, variable methods for assessing adverse events, unclear causes of death in some cases, inconsistent follow-up, small sample size, and lack of a standardized treatment protocol. In addition, reirradiation was carried out at the owner's request; clear criteria for reirradiation have not yet been established. Due to the owner's wishes and in cases with long survival times, multiple reirradiations were performed, resulting in variations in the cumulative dose. ConclusionReirradiation with hypofractionated radiotherapy for feline nasal adenocarcinoma may be an effective option when the tumor recurs and may help prolong OS. However, severe late effects, such as fistula formation, can occur in cases with bone lysis. AcknowledgmentsNone. Conflict of interestThe authors declare no conflict of interest. FundingThis research received no specific grant. Authors' contributionsH.U., Y.K., and T.M. designed the study; H.U., Y.K., Y.N., T.K., and Y.B. collected the data; H.U., Y.K., and T.K. analyzed and interpreted the data; Y.N. and T.M. drafted the manuscript; T.M. and H.K. supervised and directed the study; all authors critically revised the manuscript for important intellectual content and approved the final version for publication. Data availabilityAll data supporting the findings of this study are available within the manuscript. ReferencesAdams, W.M., Kleiter, M.M., Thrall, D.E., Klauer, J.M., Forrest, L.J., La Due, T.A. and Havighurst, T.C. 2009. Prognostic significance of tumor histology and computed tomographic staging for radiation treatment response of canine nasal tumors. Vet. Radiol. Ultrasound. 50, 330–335. Al-Mamgani, A., Kessels, R., Navran, A., Hamming-Vrieze, O., Zuur, C.L., Paul De Boer, J., Jonker, M.C.J., Janssen, T., Sonke, J.J. and Marijnen, C.A.M. 2021. Reduction of GTV to high-risk CTV radiation margin in head and neck squamous cell carcinoma significantly reduced acute and late radiation-related toxicity with comparable outcomes. Radiother. Oncol. 162, 170–177. Bommarito, D.A., Kent, M.S., Selting, K.A., Henry, C.J. and Lattimer, J.C. 2011. Reirradiation of recurrent canine nasal tumors. Vet. Radiol. Ultrasound. 52, 207–212. Cox, N.R., Brawner, W.R., Powers, R.D. and Wright, J.C. 1991. Tumors of the nose and paranasal sinuses in cats: 32 cases with comparison to a national database (1977–1987). J. Am. Anim. Hosp. Assoc. 27, 339–347. Fujiwara-Igarashi, A., Fujimori, T., Oka, M., Nishimura, Y., Hamamoto, Y., Kazato, Y., Sawada, H., Yayoshi, N., Hasegawa, D. and Fujita, M. 2014. Evaluation of outcomes and radiation complications in 65 cats with nasal tumours treated with palliative hypofractionated radiotherapy. Vet. J. 202, 455–461. Gieger, T., Siegel, S., Rosen, K., Jackson, D., Ware, K., Kiselow, M. and Shiomitsu, K. 2013. Reirradiation of canine nasal carcinomas treated with coarsely fractionated radiation protocols: 37 cases. J. Am. Anim. Hosp. Assoc. 49, 318–324. Giuliano, A. and Dobson, J. 2020. Clinical response and survival time of cats with carcinoma of the nasal cavity treated with palliative coarse fractionated radiotherapy. J. Feline Med. Surg. 22, 922–927. Henderson, S.M., Bradley, K., Day, M.J., Tasker, S., Caney, S.M., Hotston Moore, A. and Gruffydd-Jones, T.J. 2004. Investigation of nasal disease in the cat--a retrospective study of 77 cases. J. Feline. Med. Surg. 6, 245–257. Kondo, Y., Matsunaga, S., Mochizuki, M., Kadosawa, T., Nakagawa, T., Nishimura, R., and Sasaki, N. 2008. Prognosis of canine patients with nasal tumors according to modified clinical stages based on computed tomography : a retrospective study. J. Vet. Med. Sci. 70, 207–212. Ladue, T. and Klein, M.K. 2001. Toxicity criteria of the veterinary radiation therapy oncology group. Vet. Radiol. Ultrasound. 42, 475–476. Lana, S.E. and Turek, M.M. 2020. Nasal cavity and sinus tumors. In Small animal clinical oncology, 6th ed. Eds., Vail, D.M., Thamm, D.H. and Liptak, J.M. St. Louis, MI: Saunders Elsevier, pp: 494–523. Madewell, B.R., Priester, W.A., Gillette, E.L. and Snyder, S.P. 1976. Neoplasms of the nasal passages and paranasal sinuses in domesticated animals as reported by 13 veterinary colleges. Am. J. Vet. Res. 37, 851–856. Maruo, T., Nakamura, S., Fukuyama, Y. and Kawarai, S. 2013. Validation of new bite block-type head-immobilization devices for radiotherapy in dogs. Vet. Radiol. Ultrasound 54, 674–679. Mori, A., Shida, T., Maruo, T., Fukuyama, Y., Imai, R., Ito, T., Kayanuma, H. and Suganuma, T. 2009. Examination of the utility of a bite block-type head immobilization device in dogs and cats. J. Vet. Med. Sci. 71, 453–456. Moulton, J.E. 1990. Tumors of respiratory tract. In Tumors in domestic animals, 3rd ed. Berkeley, CA: University of California Press, pp: 308–13. Nemoto, Y., Maruo, T., Fukuyama, Y., Kawarai, S., Shida, T. and Nakayama, T. 2015. A novel support device for head immobilization during radiation therapy that is applicable to both cats and dogs. Vet. Radiol. Ultrasound. 56, 680–686. Nguyen, S.M., Thamm, D.H., Vail, D.M. and London, C.A. 2015. Response evaluation criteria for solid tumours in dogs (v1.0): a Veterinary Cooperative Oncology Group (VCOG) consensus document. Vet. Comp. Oncol. 13, 176–183. Nieder, C. 2019. Second re-irradiation: a delicate balance between safety and efficacy. Phys. Med. 58, 155–158. Nieder, C., Milas, L. and Ang, K.K. 2000. Tissue tolerance to reirradiation. Semin. Radiat. Oncol. 10, 200–209. Rasotto, R., Berlato, D., Goldschmidt, M.H. and Zappulli, V. 2017. Prognostic significance of canine mammary tumor histologic subtypes: an observational cohort study of 229 cases. Vet. Pathol. 54, 571–578. Reczynska, A.I., Larue, S.M., Boss, M.K., Lee, B.I., Leary, D., Pohlmann, K., Griffin, L., Lana, S. and Wormhoudt Martin, T. 2022. Outcome of stereotactic body radiation for treatment of nasal and nasopharyngeal lymphoma in 32 cats. J. Vet. Intern. Med. 36, 733–742. Sfiligoi, G., Théon, A.P. and Kent, M.S. 2007. Response of nineteen cats with nasal lymphoma to radiation therapy and chemotherapy. Vet. Radiol. Ultrasound. 48, 388–393. Stewart, F.A. 1999. Re-treatment after full-course radiotherapy: is it a viable option?. Acta Oncol. 38, 855–862. Turrel, J. and Théon, A.P. 1988. Reirradiation of tumors in cats and dogs. J. Am. Vet. Med. Assoc. 193, 465–469. Veterinary Cooperative Oncology Group. 2016. Veterinary Cooperative Oncology Group - common terminology criteria for adverse events (VCOG-CTCAE) following chemotherapy or biological antineoplastic therapy in dogs and cats v1.1. 2016. Vet. Comp. Oncol. 14, 417–446. Weeden, A.M. and Degner, D.A. 2016. Surgical approaches to the nasal cavity and sinuses. Vet. Clin. North. Am. Small. Anim. Pract. 46, 719–733. Yoshikawa, H., Gieger, T.L., Saba, C.F., Fredrickson, K., Kubicek, L., Haney, S., Ruslander, D., Kelsey, K.L., Mcentee, M.C. and Nolan, M.W. 2021. Retrospective evaluation of intranasal carcinomas in cats treated with external-beam radiotherapy: 42 cases. J. Vet. Intern. Med. 35, 1018–1030. | ||
| How to Cite this Article |
| Pubmed Style Ueno H, Kondo Y, Nishiyama Y, Katayama T, Banno Y, Kayanuma H, Maruo T. Hypofractionated reirradiation for feline nasal epithelial tumors: A retrospective study. Open Vet. J.. 2026; 16(1): 318-326. doi:10.5455/OVJ.2026.v16.i1.28 Web Style Ueno H, Kondo Y, Nishiyama Y, Katayama T, Banno Y, Kayanuma H, Maruo T. Hypofractionated reirradiation for feline nasal epithelial tumors: A retrospective study. https://www.openveterinaryjournal.com/?mno=277956 [Access: January 31, 2026]. doi:10.5455/OVJ.2026.v16.i1.28 AMA (American Medical Association) Style Ueno H, Kondo Y, Nishiyama Y, Katayama T, Banno Y, Kayanuma H, Maruo T. Hypofractionated reirradiation for feline nasal epithelial tumors: A retrospective study. Open Vet. J.. 2026; 16(1): 318-326. doi:10.5455/OVJ.2026.v16.i1.28 Vancouver/ICMJE Style Ueno H, Kondo Y, Nishiyama Y, Katayama T, Banno Y, Kayanuma H, Maruo T. Hypofractionated reirradiation for feline nasal epithelial tumors: A retrospective study. Open Vet. J.. (2026), [cited January 31, 2026]; 16(1): 318-326. doi:10.5455/OVJ.2026.v16.i1.28 Harvard Style Ueno, H., Kondo, . Y., Nishiyama, . Y., Katayama, . T., Banno, . Y., Kayanuma, . H. & Maruo, . T. (2026) Hypofractionated reirradiation for feline nasal epithelial tumors: A retrospective study. Open Vet. J., 16 (1), 318-326. doi:10.5455/OVJ.2026.v16.i1.28 Turabian Style Ueno, Hirona, Yui Kondo, Yuta Nishiyama, Takayuki Katayama, Yusuke Banno, Hideki Kayanuma, and Takuya Maruo. 2026. Hypofractionated reirradiation for feline nasal epithelial tumors: A retrospective study. Open Veterinary Journal, 16 (1), 318-326. doi:10.5455/OVJ.2026.v16.i1.28 Chicago Style Ueno, Hirona, Yui Kondo, Yuta Nishiyama, Takayuki Katayama, Yusuke Banno, Hideki Kayanuma, and Takuya Maruo. "Hypofractionated reirradiation for feline nasal epithelial tumors: A retrospective study." Open Veterinary Journal 16 (2026), 318-326. doi:10.5455/OVJ.2026.v16.i1.28 MLA (The Modern Language Association) Style Ueno, Hirona, Yui Kondo, Yuta Nishiyama, Takayuki Katayama, Yusuke Banno, Hideki Kayanuma, and Takuya Maruo. "Hypofractionated reirradiation for feline nasal epithelial tumors: A retrospective study." Open Veterinary Journal 16.1 (2026), 318-326. Print. doi:10.5455/OVJ.2026.v16.i1.28 APA (American Psychological Association) Style Ueno, H., Kondo, . Y., Nishiyama, . Y., Katayama, . T., Banno, . Y., Kayanuma, . H. & Maruo, . T. (2026) Hypofractionated reirradiation for feline nasal epithelial tumors: A retrospective study. Open Veterinary Journal, 16 (1), 318-326. doi:10.5455/OVJ.2026.v16.i1.28 |