| Research Article | ||
Open Vet. J.. 2026; 16(3): 1808-1822 Open Veterinary Journal, (2026), Vol. 16(3): 1808-1822 Research Article Attenuation of carbon tetrachloride-induced male reproductive damage in albino laboratory rats using varying concentrations of supplemented soybeanZeinab Al-Amgad1, Amany Ahmed Abd El-Aziz2, Fatma A. Madkour3, Hend Al-Amgad4, Asmaa W. Basher5, Heba A. Mohammed6, Asmaa Elnagar7,8, Ibrahim F. Rehan7,8,9,*, František Zigo10<, Martina Zigová11 and Mariam M. Jad121Medical Technical College, Al-Farahidi University, Baghdad, Iraq 2Home Economic Department, Nutrition and Food Science, Faculty of Specific Education, South Valley University, Qena, Egypt 3Department of Anatomy and Embryology, Faculty of Veterinary Medicine, South Valley University, Qena, Egypt 4Human Internal Medicine and Rheumatism, Faculty of Medicine, Kasr Al Ainy, Cairo University Hospitals, Cairo, Egypt 5Department of Pharmacology, Faculty of Veterinary Medicine, South Valley University, Qena, Egypt 6Doctor of Philosophy, Faculty of Science Postgraduation, Qena, Egypt 7Institute of Genetics and Animal Biotechnology, Polish Academy of Sciences, Jastrzebiec, Poland 8Department of Pathobiochemistry, Faculty of Pharmacy, Meijo University, Nagoya-shi, Japan 9Department of Animal Behavior and Husbandry, Faculty of Veterinary Medicine, Menoufia University, Shebin Alkom, Egypt 10Department of Nutrition and Animal Husbandry, University of Veterinary Medicine and Pharmacy, Kosice, Slovakia 11Department of Pharmacology, Faculty of Medicine, Pavol Jozef Safarik University, Kosice, Slovakia 12Department of Zoology, Faculty of Science, Luxor University, Luxor, Egypt *Corresponding Author: : Ibrahim F. Rehan. Institute of Genetics and Animal Biotechnology, Polish Academy of Sciences, Postepu 36A, 05-552 Jastrzebiec, Poland. Email: ibrahim.rehan [at] vet.menofia.edu.eg Submitted: 16/08/2025 Revised: 02/02/2026 Accepted: 15/02/2026 Published: 31/03/2026 © 2025 Open Veterinary Journal
AbstractBackground: Male infertility is closely linked to testicular damage caused by environmental chemicals. Carbon tetrachloride (CCl4), widely used in manufacturing chlorinated chemicals, is highly toxic to the liver, testes, and other organs. Its testicular toxicity involves oxidative stress, apoptosis, and hyperlipidemia. Plant-derived proteins such as soybeans, with phytoestrogenic properties, have gained attention for their potential health benefits. Aim: This study investigated the protective effect of soybean on CCl4-induced testicular damage. Methods: Twenty-eight male albino rats were divided into four groups: control, CCl4-treated (1.0 ml/kg intraperitoneally, thrice weekly for 3 weeks), CCl4-treated plus 10% soybean diet, and CCl4-treated plus 30% soybean diet (as a schematic cartoon in Fig. 1). After 7 weeks, serum lipoproteins, testicular malondialdehyde (MDA), and glutathione peroxidase (GPx) were measured; histopathological, histochemical, and immunohistochemical analyses were performed, including cleaved caspase-3 expression.
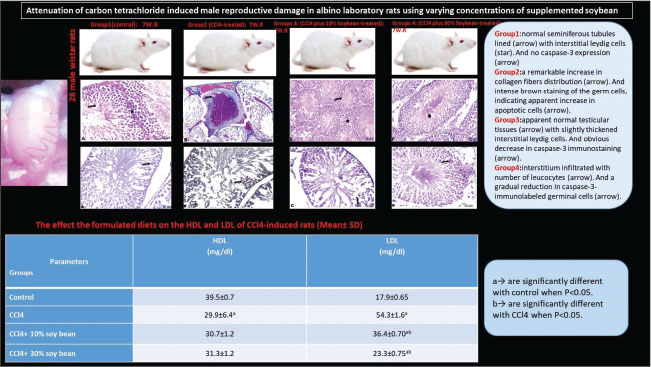
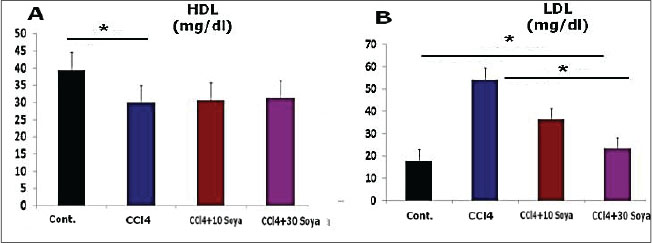
Fig. 1. Graphical abstract illustrates the experimental design of the study. Results: CCl4 exposure significantly reduced high-density lipid-cholesterol, increased low-density lipid-cholesterol, elevated MDA, altered GPx activity, intensified caspase-3 staining, and caused marked testicular structural deterioration. Soybean supplementation ameliorated these effects, restoring lipid profiles, reducing oxidative markers, preserving testicular architecture, improving spermatogenesis, and mitigating caspase-3 expression. Conclusion: Overall, soybean exerts antioxidant and immunoprotective effects against CCl4-induced testicular damage, suggesting a potential dietary strategy to counteract reproductive toxicity. Keywords: CCl4, Soybean, Testes, Histomorphology, GPx. Caspase 3. IntroductionDuring male reproductive life, testes create sperm in equilibrium between the self-renewal and differentiation of the spermatogenic cells (DeFalco et al., 2015). Impaired reproductivity in the male is commonly induced by numerous factors, including alcoholism, drugs, chemicals, hormonal imbalance, aging, smoking, and addiction (Javadi et al., 2011). Earlier studies proved that oxidative tissue damage and alterations in antioxidant mechanisms serve as the two leading issues of the reproductive distortion (Hamza and Diab, 2020). Testicular oxidative stress is the leading cause of infertility in males, consequently inducing dynamic alterations in the endocrine signals, testicular microvascular flow, and germinal cells apoptosis (Turner and Lysiak, 2008). In mammals, spermatozoa membranes consist of huge numbers of polyunsaturated fatty acids, supporting more stimulation to the lipid peroxidation (Adewoyin et al., 2017). Also, spermatogenesis is a continual procedure where mitochondria of the germ epithelium require more oxygen, resulting in reactive oxygen species production. A variety of environmental chemicals and clinically helpful drugs contribute to provoking extensive damage to varying tissues via the metabolic stimulation of the highly reactive free radicals (Unsal et al., 2021). The testicle is one of the major reproductive organs, clearly enclosed in Carbon tetrachloride (CCl4) toxicity (Abdel Moneim, 2016). The toxic effect of CCl4 perhaps occurs owing to an overproduction of free radicals during metabolism. When excessive radicals are produced, the peroxidation of the sperm membrane fatty acids occurs, allowing sperm cells to be more vulnerable to damage (Agarwal et al., 2014). Growing epidemiological concerns refer to the significant predominance of metabolic syndromes, are constellation of increased very-low-density lipoprotein (v-LDL) and low-density lipoprotein cholesterol (LDL) index and is associated with reduced testosterone activity (Salam et al., 2012). Recent experimental studies showed that a rich-fat meal-induced dyslipidemia negatively affects the morphology of Leydig cells, resulting in downregulated steroidogenic proteins (Zhang et al., 2017). The metabolic and hyperlipidemia syndrome is mainly related to male fecundity (Maqdasy et al., 2013). Thereby, lipid metabolic disarray upsets spermatogenesis through blocking testosterone production in Leydig cells. It also documented that lipid/lipoprotein level is negatively related to testosterone in male mice (Amano et al., 2017). Although the mechanisms through which the lipoproteins influence Leydig cell structure and function are nevertheless latent, however, lipid peroxidation, particularly oxidation of low-density lipoprotein, serves as a marker for inflammation and oxidative damage. Circulating oxidized low-density lipoprotein, a modified form of oxidative LDL, is linked with cardiovascular illness and metabolic syndrome in adults (Calcaterra et al., 2017). Clinical evidence showed that fertility insufficiency, together with higher levels of oxidized LDL (Kosola et al., 2013). Oxidative stress and inflammatory responses emitted by significant amounts of LDL contribute to the apoptosis and dysfunction of vascular endothelium (Kim et al., 2013). Leydig cells are clearly prone to oxidative and inflammatory damage in the pathogenesis of male infertility and ultimately impaired testosterone production. Prior reports declared that the elevated plasma oxLDL level is among the independent risk factors of spermatogenesis. It suggested that LDL may confer a crucial task in lipid metabolism disturbance, modifying Leydig cells. A high LDL concentration by a wealthy-cholesterol food in a rat model proved to provoke a higher injurious effect on male genital organs (Khorrami et al., 2015). Accordingly, subsequent CCl4 administration triggered lipid metabolism parameters related with hyperlipidemia process characterized by marked increase in LDL and decrease in high-density lipid-cholesterol (HDL) levels (Hussein and Khan, 2022). Hyperlipidemia related to CCl4, which hinder out flow of liver cytosolic components into the blood stream, resulted in the evoked oxidative stress (Xu et al., 2021). CCl4 modified the level of lipid peroxidation and enhancing the decreased level of antioxidants, such as superoxide dismutase, glutathione peroxidase (GPx), and catalase, in addition to lowered the enzyme activity of reduced glutathione (Rajesh and Latha, 2004). Previous study suggested that oxidative stress aggravated by CCl4 interfere with testicular DNA, function of spermatogenesis, and survival of reproductive tissues, accompanied with the male infertility (Abarikwu et al., 2012). Besides this, CCl4 broadly exacerbates histological lesions in testicular tissues (Murali et al., 2004). CCl4 demonstrated necrosis of the seminiferous tubules, collagen fiber accumulation, edema, and damage in the blood vessel walls. The damaging effects are suggested to be because of the excessively produced free radicals exceeding the antioxidant level of the stressed cells (Sönmez et al., 2014). Seminal free radicals disrupt sperm morphology. CCl4 application incite spermatogenic loss of the seminiferous tubules, mitotic stoppage, incomplete atrophy of the interstitial cells, and variations in Sertoli structure (Anatolia, 2014). In CCl4 intoxication, prominent histological abnormalities, including testicular degeneration, necrosis, desquamation, spermatogenesis arrest, decline in germ cells, and significant reduction in the seminiferous tubules, were illuminated. It caused histopathological damage in the testicles and a significant expression of the apoptotic cells in the seminiferous tubules (Maheshwari et al., 2009), by the means of the increased cellular death process by caspase-3 positive cells expression in testicles. Consequently, the extensive oxidative damage promotes caspase 3 immunoactivity and accelerated apoptosis mechanism (Othman et al., 2014). Continual growth of the world population creates challenges with increased consumption of a protein-rich diet, notably meat (Godfray et al., 2018). Nevertheless, the substantial resource demands for meat production and the potential deleterious effects on human health correlated to great meat consumption, and researchers are actually seeking sustainable and healthier substitutes, such as ecologically friendly high-protein plant-based foods that confer potential health benefits (Aschemann-Witzel et al., 2021). Subsequently, a challenge strategy for hyperlipidemia using plant-based diets is being contemplated. Vegetarian and other plant-origin diets established as dietary patterns to improve lipid profiles (Appt et al., 2008). Plant protein origin, notably soy, is full of the required individual lipid-lowering advantages via the specific components, including polyunsaturated lipids, viscous fibers, and plant sterols. Substitution of animal protein with plant source exhibits a preference by extirpation of saturated fatty acids (Azadbakht et al., 2008). The substituting advantages of plant-established protein assessed on the lipid bases for controlling hyperlipidemia parameters involving non-HDL-C, LDL-C, and apolipoprotein B (Cuevas et al., 2003). Soy is an alternative therapy for patients seeking a modest decrease in LDL. Soy can improve serum lipid indices by means of a number of efficient bioactive components illuminated the hypolipidemic effects mechanisms. Soy protein is a rich source of phytoestrogens with multifunction characters (Costa and Summa, 2000). Soybean seeds approximately contained 36% protein and 18% fat (Ogbuewu et al., 2010). Interestingly, clinical investigations suggested that soybean is efficient contributor in the regulation of the regeneration of spermatogenic stem cells (Miura et al., 2003), and male venereal function by estrogen receptors. Soy seeds support the male fertility via increase sperm motility and viability (Oyeyemi and Okediran, 2007). The main phytochemicals in soybean are isoflavones, like 17 β-oestradiol characters. Clinical literatures documented that soybean seeds have estrogenic materials as gonad stimulating components improving male fertility. The soybeans take a part by the antioxidant function of bioactive isoflavones (Ogbuewu et al., 2010). Previous studies demonstrated that soy intake manifests potential protective effects on the oxidative stress by the means of modulating the changes in the activity of the antioxidants signaling (Engelman et al., 2005). However, an ideal dietary supplementation of soybean activated development of testicular tissues. The findings showed that soybean seeds positively influenced gonad development by increased renewal of varying genders of the testicle cells shared in spermatogenesis (Oyeyemi and Okediran, 2007). However, the helpful properties of soybean in modulating the testicular damage triggered by CCl4 remain in dearth. In this context, the current work aimed to elicit the extreme preventative effect of soybean supplementation on the CCl4-induced testicular toxicity through the biochemical assays, oxidative biomarkers, histomorphological approaches, and apoptosis-related gene signaling. Material and MethodsAnimals handlingTwenty-eight male 2–3 months Wistar albino rats weighing 180–200 g were used in the current experimental procedure. The rats were purchased from Laboratory Animal Unit belonging to Sohag University in the Egyptian capital. Once Wistar rats arrived in the housing room, they were harbored under housing conditions and randomly designated into groups. Wistar rats were isolated in clean Stainless-steel cages covered with mesh. Cages bedding was changed twice a week during the treatments, and given commercial pellets and water ad-libitum for two weeks prior to toxicity induction and supplementation. The conditioning system of the housing room was a constant 12-hour light/dark for each, air temperature 22°C, and a relative humidity 50%. Experimental dietsFresh and clean soybean seeds were bought from a reliable market located in Qena Province located in Egypt country. Two feeding treatments were applied in this experiment, consisting of a plant protein-based diet. However, plant sources consisted of increased concentrations of soybeans. Preparation of soybean diets was intended to comprise crude lipid, total protein, and total carbohydrate. Collected soybeans were washed with water, dried in room temperature, grounded into a fine powder, and stored at room temperature for feeding experiments and chemical analysis. Diet in group 3 was incorporated with 10% soybean, but in group 4 was substituted to contain an increased level of soybean up to 30%. Groups 1 and 2 fed normal commercial diet. ChemicalsCCl4 of 100% concentration was purchased from El-Nasr Company in Egypt. A local pure corn oil as a vehicle was gained from the market belonging to Qena Province situated in Egypt. CCl4 dissolved in corn oil and administrated via intraperitoneal injection at a dose of 1 mg/kg body weight of the rat three times weekly. Preparation of the CCl4 solutionEach 50 ml CCl4 was diluted in 100 ml corn oil (0.5:1, v/v). Each rat was injected IP with 1 ml of the prepared CCl4 solution three times weekly as recommended by Khedr and Khedr (2017). Different preparations of soybean diets, as earlier reported by Alada et al. (2004). Experimental modelThe experimental animal's models utilized for this study were locally bred male Wistar albino rats which sourced from the Experimental Animal Unit in Sohag University, Egypt. Animals were adapted under the standard environmental conditions for 2 weeks; allowed suitable food and water ad libitum. To investigate the testicular damage induced by CCl4, rats were randomly set into four main groups during the 7-week feeding period. Group 1 (control group)Seven male albino rats received orally distilled water and fed the standard commercial diet during the experimental period and used as a negative control group. Group 2 (CCl4 group)Seven male albino rats were subjected to i.p. injection with a diluted CCl4 in corn oil in a dose of 1 mg/kg in equal volume 1:1 three time weekly for three weeks and fed standard commercial diet for 7 weeks. Group 3 (CCl4 plus 10% soybean-treated group)Seven male albino rats were quite CCl4 injected and further fed on the formulated diet contained 10% soybean for 7 weeks. Group 4 (CCl4 plus 30% soybean-treated group)Seven male albino rats were quite CCl4 injected and further fed on the formulated diet contained 30% soybean for 7 weeks. Analytical characterization of feed componentsThe formulated diets were analyzed for crude protein content following the descriptive method of Lowry et al., (1951). Meanwhile, crude lipid, carbohydrate, and ash contents were primarily calculated according to the previous report (AOAC, 2000). Biological samplingAt the completion of the exposure time, which extended 7 weeks, all rats were overnight fasted for venous blood samples withdrawal from the eyes by retro-orbital puncture. Blood was allowed to coagulate, centrifugation at 3,000 rpm for 15 minutes for serum separation. The serum was thoroughly collected into clean dry tubes using a Pasteur pipette and frozen at −20°C till determination of the lipoproteins index. Testes samplesThe abdomen region was accurately incised via midline abdominal surgery under anesthetized conditions to disclose the reproductive tissue. The testes of each rat were cautiously exercised and removed. Then they were trimmed free of the fat and adjoining tissues. Testes samples were rapidly split into two halves; one washed by the normal saline and directly soaked up in the buffered formalin solution for histopathological assay. While other half well rinsed in ice-cold saline for oxidant biomarkers assay. Measurement of serum lipoproteinsMeasurement of serum concentrations of HDL and LDL were executed by standard diagnostic kits using calorimetric ways. Each of HDL and LDL was measured according to methods of Lopes-Virella et al. (1977) and Lee and Nieman (1996), respectively. Measurement of the oxidant biomarkersWhile testicles were guardedly homogenized using an effectual homogenizer, then cold centrifuged at 4°C for 10 minutes, and the consequent supernatants were stocked in Eppendorf tubes at −80°C for an estimation of Malondialdehyde (MDA) levels, and GPx activities. MDA values as well as and GPx activities were determined using diagnostic kit according to the previous reports (Ohkawa et al., 1979) and (Paglia and Valentine, 1967), respectively. Histopathological preparationsFresh testes specimens of about 1.5 cm diameter were extracted from all existing groups fixation in the freshly prepared buffered formalin before tissue processing. Histological procedure using automated histological processor included the specimen's dehydrated using increasing level of alcohol concentration (70%, 90%, and 100%, respectively, 2 hours for each), the dehydrated specimens were cleared into changes of xylene solution 1 hour for each at room temperature, then softened in two changes of molten paraffin wax at 58°C. The blocked paraffin tissues were mounted for sectioning on the rotary microtome. The serial sections of 4 μm thicknesses gained and stained with the following histological stains involving Hematoxylin-eosin for histological lesions, Masson’s trichrome for collagen fibers and fibrous tissues, Bromophenol Blue (BPB) for protein elements. Moreover, Toluidine blue techniques performed to appreciate the morphological features of the treated testes. The testis slides for each group viewed and the photomicrographs using the light Leica microscope (Bancroft and Gamble, 2008). Nominal semi-quantitative approach was done using testes sections selected from three rats (n=9 for each group) for assessment of the histological scoring was absent, mild, moderate, and severe lesions (0, +, ++, and +++), respectively, as investigated by Gibson-Corley et al. (2013). Testicular immunohistochemical stainingImmunohistochemical staining of testes sections was done following the experimental protocol described by Magaki et al. (2019). In summary, deparaffinized and rehydrated testes sections were circumstantially done through a series of alcohol washes. For the sections prepared to caspase-3, the slides were washed with 3% H2O2 in absolute methanol for half hour at 4°C and then flushed with phosphate-buffered saline (PBS). Subsequently, testes sections treated with 10% normal blocking serum at 25°C for an hour. The primary antibody, termed anti-caspase-3 (monoclonal anti-cleaved caspase-3 at dilution 1:200), was applied to the sections and allowed to incubate. Afterward, the slides were rinsed for an hour on PBS. The further step was addition streptavidin-peroxidase conjugate to the sections slides for 30 minutes incubation period. Thereafter, testes sections were ready for the immunohistochemical readings and micrographs. Evaluation of apoptotic cells index was performed in random 10 sections of testes per group using ImageJ version. Statistical analysisTriplicates analysis (n=3) per group was performed for each parameter. The uniformity within groups was done using SPSS. ANOVA accompanied by post-Hoc Scheffe test run to compare between the four independent experimental groups when p less than 0.05 value as statistically significant. Data descriptive analysis was the means and standard deviation of data (M ± SD). Ethical approvalEthical statement of this experimentation was approached with stringent guides to keep animals without any endurance or stress in accordance with Ethical Guidelines and Policies granted by the Institutional Animal Care Committee, Faculty of Science, South Valley University, Qena, Egypt (011/02/25). ResultsFeed compositionThe proximate ingredients in diets are mainly incorporated with soybean protein sources. Correspondingly, crude lipid level (g/kg) was 8.635 and 3.362 in 10%, and 30% soybean, respectively. Total carbohydrate concentration (g/kg) was 124.21 and 78.104, respectively. Total protein (g/kg) of the formulated diets in diets with 10% and 30% soybean was 55.406 and 106.324, respectively. Eventually, Kcal content (g/kg) was 796.179 and 767.97, respectively. Biochemical lipoproteins findingsEffect on HDL-cholesterol level (mg/dl)Serum HDL, as illustrated in Figure 2 implicit that the mean values in the control, CCl4, CCl4+ 10% soybean, and CCl4+ 30% soybean groups were 39.5 ± 0.7, 29.9 ± 6.4, 30.7 ± 1.2, and 31.3 ± 1.2 mg/dl, respectively. HDL level was significantly decreased (p < 0.05) in CCl4 (p < 0.05) when compared with the control. Effect on LDL-cholesterol level (mg/dl)Serum LDL level in Figure 2 displayed the mean values of LDL levels in the control, CCl4, CCl4+ 10% soybean, and CCl4+ 30% soybean groups were 17.9 ± 0.65, 54.3 ± 1.6, 36.4 ± 0.70, and 23.3 ± 0.75 mg/dl, respectively. Accordingly, LDL level showed significant increase (p < 0.05) among all treated groups in comparison with control, contrary to significant improvement (p < 0.05) was recorded in LDL level of both two soybean-treated concentrations when compared with CCl4.
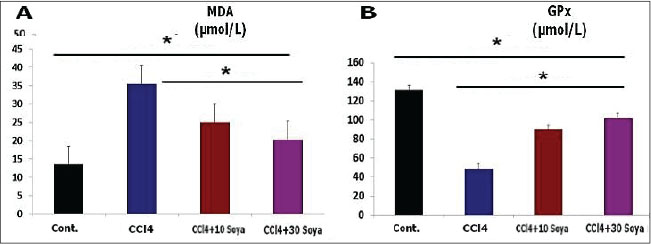
Fig. 2. The effect the soybean diets on the lipoprotein index of CCl4-treated rats, HDL (A) and LDL (B). (Mean+ SEM), p-value*, comparing significant difference between groups (p < 0.05) using one-way ANOVA. Effect on the oxidative profile (µmol/l)The results exhibited that CCl4 increased significantly (p < 0.05) the testicular MDA level (35.5 ± 1.1 µmol/l), but it decreased (p < 0.05) the GPx activity (49.03 ± 2.9 µmol/l) compared to the control group values (13.5 ± 1.7 and 131.26 ± 4.1 µmol/l), respectively. Opposite results were obtained in the MDA level of the testicles of CCl4+ 10% soybean and CCl4+ 30% soybean-induced rats. MDA level was significantly decreased (p < 0.05) (25.0 ± 1.6 and 20.3 ± 2.6 µmol/l) than those in the CCl4 group. Simultaneously with GPx activity, which expressed significant increases (p < 0.05) in both soybean treated groups (90.03 ± 3.4 and 102.13 ± 3.7 µmol/l), respectively, when compared with CCl4, as in Figure 3.
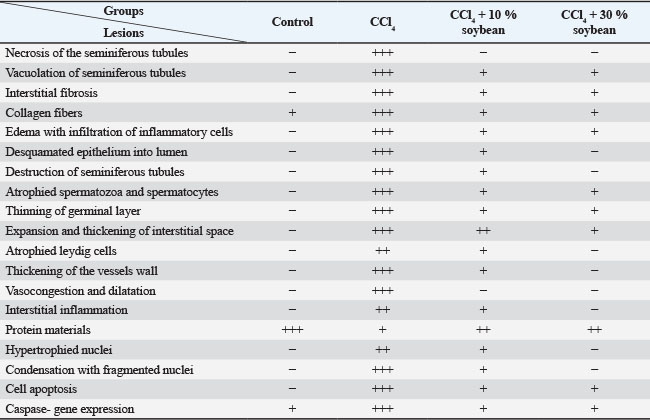
Fig. 3. The effect the soybean diets on the oxidative indicator of CCl4-treated rats, MDA (A) and GPx (B). (Mean+ SEM), P-value*, comparing significant difference between groups (p < 0.05) using one-way ANOVA Gross appearanceTestes of the control male rats showed normal color, consistency, and texture. However, this trial research discovered that CCl4 damaged the testes, inducing them to be congested and more friable. Fortunately, treatment with different concentrations of the soybean positively restored the normal appearance of the testicle, this indicate that soybean has potential corrective and protective effect. Microscopic featuresThe CCl4-exposed rats revealed extensive testicular damage in male rats with marked disturbance in the structure of seminiferous tubules. The parenchyma of the testes appeared disorganized, disintegrated, and distorted. Loss of the regular testicular architecture was observed. Most of the spermatocytes and spermatozoa were damaged with a significant increase in collagen contents compared to the testicular histology of the control rats. CCl4 plus soybean groups exhibited a partial restoration of the damaged tissues, a dose and time dependent (Table 1). Table 1. The semi-quantitative histological approach of the testes differentiated depending on the degree of histological lesions.
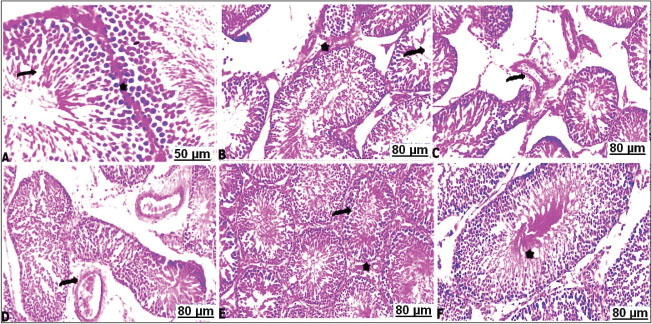
Hematoxylin and Eosin (H&E)In the control testes, the light sections of the seminiferous tubules with H&E stain appeared well-defined architecture lined dense epithelial layers of Sertoli cells and spermatogonia, and separated by interstitial cells of the Leydig and normal spermatogenesis. The seminiferous tubules were composed of spermatogenic and Sertoli cells. Spermatogenic cells contained primary spermatogonia, spermatocytes and spermatid (Fig. 4A). Whereas administration of CCl4 resulted in severe damage on the testicular tissue with arrest of spermatogenesis characterized by marked necrosis and degenerative changes of the spermatogenic cells, disruption of the seminiferous tubules with random absence of the germ cells, wideness of interstitial space, and loss of Leydig cells (Fig. 4B–D). Sections of testes tissues of the CCl4 fed 10 % or 30 % soybean are likely to improve the function of testes with well-enhanced spermatogenesis, inducing normal appearance of the testicular parenchyma (Fig. 4E and F).
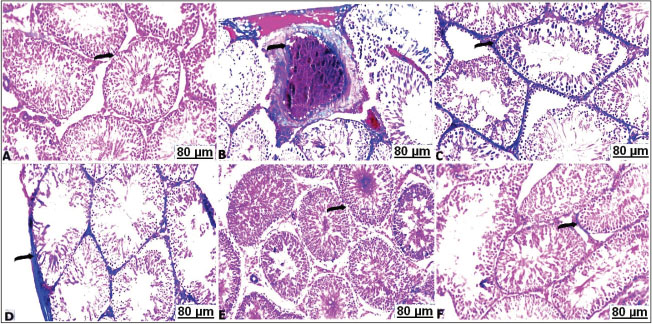
Fig. 4. Transverse sections of testes with H&E of the control and treated groups: (A) control testes showing normal seminiferous tubules (arrow) separated by basement membrane from interstitial Leydig cells (star). (B) CCl4-exposed testes showing necrosis and arrest of the spermatozoa (arrow), besides thickening of interstitial tissues (star). (C) CCl4-exposed testes showing perivascular edema with thickening of the blood vessels wall (arrow). (D) CCl4-exposed testes showing sharp congestion and dilatation of the vasculature (arrow). (E) CCl4-exposed testes+10% soybean showing apparent normal testicular tissues (arrow) with slightly thickened interstitial Leydig cells (star). (F) CCl4-exposed testes+30% soybean showing apparent normal seminiferous tubules suffered mild vacuolations (star). Histochemical observationsMasson's trichrome stain resultsThe light sections of the control testes stained with Masson's trichrome revealed thin layer of the collagen fibers (Fig. 5A). Although, CCl4 injected group manifested a significant degree of the perivascular or intertubular fibrosis was detectable by a distinct layer of the collagen infiltrated neighboring blood vessels and at the interstitium (Fig. 5B–D). Thus, CCl4 animals after treatment with a low concentration of soybean demonstrated mild distribution of collagen fibers around vasculature along the testicular tissues (Fig. 5E). As well as high concentration of dietary soybean showed minimal collagen tissues deposition (Fig. 5F).
Fig. 5. Transverse sections of testes with Masson's trichrome of the control and treated groups: A) control testes showing hard detectable collagen fibers (arrow). B,C,D) CCl4-exposed testes showing thickened interstitial tissues with collagen (arrow). E) CCl4-exposed testes+10% soybean showing unequal distribution of stained collagen fibers (arrow). F) CCl4-exposed testes+30% soybean showing apparent a decrease of the faint-stained collagen fibers (arrow). BPB stain resultsA positive BPB reactivity was noticed with a dark bluish discoloration in the control testes, accordingly all the seminiferous tubules were characterized by a sharp content of total protein (Fig. 6A). CCl4 treated rats; most of the seminiferous tubules showed a marked depletion with a lower content of total protein (Fig. 6B). In the CCl4 treated rats with soybean, moderate to marked reaction was observed in the seminiferous tubules in a dose-dependent protective impact, the activity of the testicle cells showed a moderate improvement with amount of total protein (Fig. 6C and D).
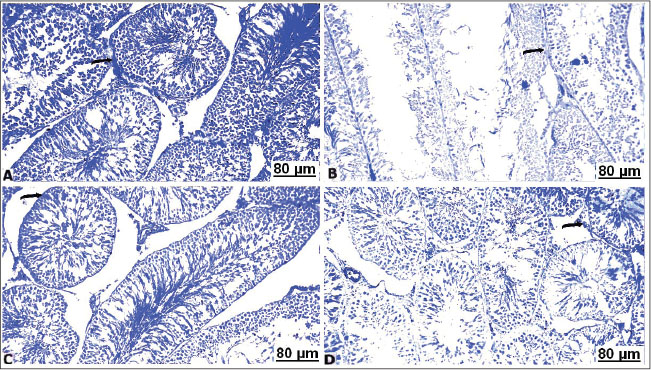
Fig. 6. Transverse sections of testes with BPB of the control and treated groups: (A) control testes showing strongly reacted protein contents (arrow). (B) CCl4-exposed testes showing pale protein staining (arrow). (C) CCl4-exposed testes+10% soybean showing well-organized protein contents (arrow). (D) CCl4-exposed testes+30% soybean showing detectable stained protein elements (arrow). Toluidine blue stain resultsMicroscopic semi-thin section in the control testis of male albino rats showed seminiferous tubules lined with normal germinal cells, intact interstitial septa, and normal basement membrane (Fig. 7A). The semi-thin section in testes of the treated rats with CCl4 displayed seminiferous tubules lined with thin layer of germinal cells consisting mainly of spermatogonia having vacuoles and enclosed by thickened interstitium (Fig. 7B). The testis of male rats of the CCl4+ 10% soybean showed normal seminiferous tubules with normal germinal lining and less prominent interstitium (Fig. 7C). Furthermore, testes of the CCl4+ 30% soybean detected healthy spermatozoa with intact germinal lining, and basement membrane (Fig. 7D).
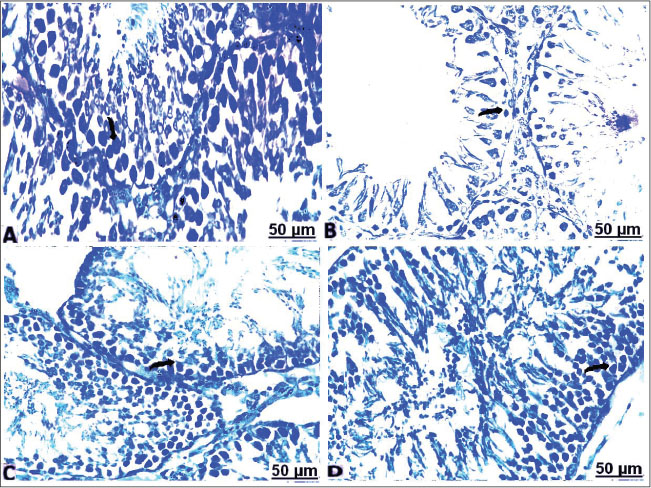
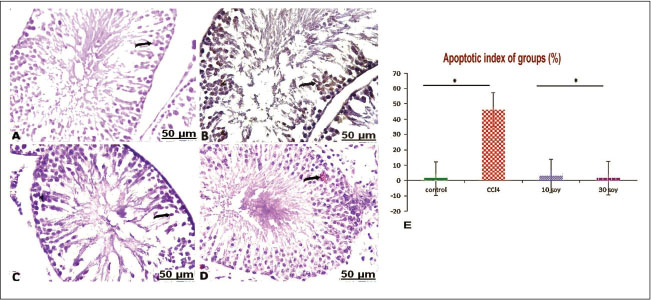
Fig. 7. Transverse semi-sections of testis with Toluidine blue of the control and treated groups: (A) control testes showing seminiferous tubules normally lined with normal germinal cells (arrow). (B) CCl4-exposed testes showing seminiferous tubules lined with thin epithelial layer of Sertoli cells and germinal cells (arrow). (C) CCl4-exposed testes+10% soybean normal germinal lining and less prominent interstitium (arrow). (D) CCl4-exposed testes+30% soybean showing healthy germinal cells, and basement membrane (arrow). Immunohistochemical resultsIn the testes of the non-treated rats, caspase-3-immunolabeled cells showed hard expression in the semiserious tubules (Fig. 8A). In CCl4 group, intense brown staining observed clearly in the germ cells, indicating increase in the caspase-3 immunolabeled germ cells, and a marked increase in apoptotic cells (Fig. 8B). In CCl4+10% soybean group, there was a distinct decrease in caspase-3-immunolabeled cells, suggesting a significant improvement in the testes section (Fig. 8C). In CCl4+30% soybean group, an evident decrease in caspase-3-immunolabeled spermatogonia was detected, referring that testes section was almost normal near to the control group (Fig. 8D).
Fig. 8. Representative sections of the immunostaining reaction of CCl4-induced testes apoptosis and testicular injury in caspase-3 male rats. (A) control testes showing weak expression of caspase-3 in the semiserious tubules (arrow). B) CCl4-exposed testes showing positive increase in the caspase-3 immunolabeled spermatogonia (arrow). C) CCl4-exposed testes+10% soybean showing a distinct decrease in caspase-3-immunolabeled cells (arrow). D) CCl4-exposed testes+30% soybean showing evident decrease in caspase-3-immunolabeled germ cells (arrow). (E) Apoptotic testicular index in groups. (Mean+ SEM), P-value*, comparing significant difference between groups (P<0.05) using one-way ANOVA. Statistically, the apoptotic index of groups by immunohistochemical analysis indicated that the increase of caspase activity is in a correlation with increase of apoptotic cells death in the current model. Whereas the shown findings exhibited a significant increase (p < 0.05) in the number of apoptotic cells associated with increased expressed caspase-3 activity in CCl4 group, which indicate marked testicles apoptosis comparable with control. However, significant decreases (p < 0.05) in the number of apoptotic cells were more detectable in CCl4+10% & 30% soybean-induced groups comparison with CCl4 group, as evidence to dramatically decline in caspase-3 activity, as illustrated. DiscussionThe present results showed that CCl4 triggered lipid metabolism led to a significant increase in the levels of lipid parameters. Significant elevation in LDL-c concentrations with pronounced lowering effect on HDL was discussed previously in CCl4-treated rats (Hosseinzadeh et al., 2007). Intraperitoneal CCl4 toxicity with 1 ml/kg induced disturbance in lipoprotein indices, resulting in lowered HDL with elevated LDL, and vLDL values (Emmanuel et al., 2021) associated with hyperlipidemia activity (Dutta et al., 2018). CCl4 increased oxidants concentrations and accelerate peroxidation of polyunsaturated fatty acids (ElMazoudy et al., 2011). The administration of CCl4 led to severe destruction of the seminiferous tubules, expanded interstitial cells, and damaged spermatogonia, and similar effects after CCl4 administration were in accordance with Keshtmand et al. (2021). In the current CCl4-induced histopathological challenges, sections of the studied testes revealed vascular congestion and edema, and interstitial homogenous eosinophilic infiltration. Certainly with multifocal numbers of sharp degeneration of epithelial lining seminiferous tubules evident by swollen areas of the discrete vacuoles, collapse of the degenerated tubules guided by lessened spermatogenesis, and absent spermatozoa in constistent of Hashem (2021). Repeated administration of CCl4 for 4 weeks led to marked disturbance of seminiferous tubule structure in form of pronounced degeneration with reduction in spermatogenic cells, sertoli cells, and primary spermatocytes also reduced numbers of the spermatid in the lumen of seminiferous tubules, with the disappearance of the interstitial cells was detected (Eljaafari et al., 2024). The disruption of the morphology of the testes might due to increased LDL level and a decrease in HDL, which affected negatively the testis through promoting the resultant lipid peroxidation and oxidative stress (Ibrahim et al., 2012), thereby increasing the germ cell apoptosis and promoting hypospermatogenesis (Yousef and Salama, 2009). Histochemical observations confirmed that CCl4 elicited cell complications in their protein contents. The damaging mechanism of CCl4 within tissues is explained by the initiation of the lipid peroxidation products, which are very reactive and display significant selective deteriorations in cell signaling, cytotoxicity, and damage to protein (Ayala et al., 2014). Additionally, oxidation of the free radicals of proteins induced changes in the dimensional structure of proteins, as well as the cleavage and crosslinking of the proteins could occur (Davies, 2016). Immunohistochemical results indicated that CCl4 alone encourages the activity of caspase-3 in the testicles. Conversely, co-treatment of different level of the soy food with CCl4 repressed and upregulated the activity of testicular caspase-3 thereby reverse apoptotic process in the rats testes (Tijani et al., 2024). Apoptosis is programmed phenomenon of the cell death by genomic and morphological modifications induced. Apoptotic process is mitochondrial dependence via pro-apoptotic and anti-apoptotic pathways correlated proteins genes (Sinha et al., 2013). Disequilibrium between pro-and anti-apoptotic genes caused apoptosis (Opferman and Kothari, 2018). The increase of Bax within cells unfavorably altered the mitochondrial permeability and ultimately release of cytochrome-c into cytosol (Abdeen et al., 2020), which vulnerable to binding with other apoptotic proteins and hence activate caspase-3, executioner caspase, and accordingly cell death occurred (Kaur et al., 2020). Chemical and synthetic drugs applied to solve the problem, but the residual effects of such drugs almost exceeded their limits owing to being costly and producing fatal adverse impacts, and thus, traditional medicines have been suggested by several researches to improve fertility in the male and female gender. Plant-based proteins are proteins concentrated or isolated from plant-based origin like cereals, legumes, algae, and others that possess valuable functional activities and potential biological characters (Nasrabadi et al., 2021). The active ingredients make the plants to have both estrogenic and androgenic properties (Ekere et al., 2013). Herein, dietary administration of soybean could suppress the apparent alterations of the lipoprotein indices promoted in the CCl4-induced male rats. Soya succeeded lipid-reducing and weight-lowering impacts (Li et al., 2017). The results of vivo investigations proved that soya effectively improved blood lipoproteins, consequently relieved risk of cardiovascular ailments (Wagner et al., 2001). The beneficial mechanism of soya on the serum lipids by which some potential mechanisms, such as direct phytoestrogenic characters, antioxidant properties, modified liver metabolism, increased bile acid excretion, and increased arterial compliance (Costa and Summa, 2000). Estrogen plays a pivotal role in many spermatogenesis pathways (Shiraishi and Matsuyama, 2017). Since, soybean serves as the major sources of bioactive plant estrogens origin (Chen et al., 2019). The main bioactive compound discovered in soybean subsequently imitates function and/or structure of steroidal estrogens of the mammalian species (Křížová et al., 2019). The histopathological analysis of the testicular sections confirmed recovery of seminiferous tubules against CCl4 associated sperms abnormalities in rats after the soy supplementation. Results confirmed that the prospective uptake of soya protected from an incidence of testicular stress, reflected by considerable suppression in MDA level with successful improvement in GPx activity and subsequently increased the viability of healthy sperms within seminiferous tubules. Scientific reports have proved that soy seeds make the possibility to neutralize the free radicals, diminished the ability of DNA to oxidation, reducing MDA content and rise the antioxidant activities (Alipour and Karimi-Sales, 2020). Testis diameter and seminiferous tubules lumens were significantly larger and well-developed in the rich phytoestrogen-fed rats (Assinder et al., 2007). The phytochemical in soybean is mainly of the phytoestrogenic features (Ogbuewu et al., 2010). The functional and structural similar to estrogens in soybean aids in provoking affect the estrogen-regulated systems mainly the reproductive system (Rietjens et al., 2017). Soya seeds exert overwhelming physiological measures on the development and functioning action of male reproductivity (Patisaul, 2017). Isoflavones found in soya significantly improved the genes and proteins expression related to testosterone synthesis in male rat (Li et al., 2017). ConclusionIt was evident that exposure of male albino rats to CCl4 exert considerable toxic effect on the testes through assorted mechanisms. The results indicated that CCl4 had significant alterations in the lipoprotein indexes through altered lipid content, and subsequently, severe testicular damage occurred. The toxic role by CCl4 is a result of a detrimental impact of oxidative stress on sperm morphology exacerbated by high LDL. The present study indicated that CCl4 could increase caspase 3 gene-related apoptosis in CCl4-treated albino rats. Conversely, soybean fosters a helpful part in trials as clinical treatment toward CCl4 toxicity. Dietary supplementation of soybean has ameliorated the testicular damage, reduced oxidative injury and downregulated by caspase-3 gene expression through antioxidant capacity in the testes. AcknowledgmentsThe authors are grateful for technicians at Faculty of medicine, South Valley University for their assistants to complete this study. Conflict of interestThe authors ensured no conflict of interest. FundingThis work was supported by the Cultural and Educational Grant Agency of the Ministry of Education, Research, Development and Youth of the Slovak Republic under the project KEGA no. 011UVLF 4/2024. Also, this study was supported by the Slovak Research and Development Agency under the Contract no. APVV-22-0457. Authors’ contributionsAll authors contributed to the study conception and design. Conceptualization: ZA-A, FAM, and HA-A; methodology: AAAE-A, ZA-A, FAM, HA-A, and AWB; software: ZA-A, FAM, HA-A, AWB, and MMJ; validation: AAAE-A, ZA-A, FAM, HA-A, and AWB; formal analysis: AAAE-A, ZA-A, FAM, HA-A, AWB, MMJ, AE, IFR, FZ, and MZ; resources: ZA-A, FAM, HA-A, AWB, and MMJ.; data curation: AAAE-A, ZA-A, FAM, HA-A, AWB, MMJ, AE, IFR, FZ, and MZ; writing— original draft preparation: AAAE-A, ZA-A, FAM, HA-A, AWB, MMJ, AE, IFR, FZ, and MZ; writing— review and editing: AAAE-A, ZA-A, FAM, HA-A, AWB, MMJ, AE, and IFR; visualization: AAAE-A, HA-A, AWB, MMJ, AE, IFR, FZ, and MZ; supervision: ZA-A, FAM, and MMJ; funding acquisition: MZ and FZ. All authors have read and agreed to the published version of the manuscript. Data availabilityExtra data can be provided upon request from the corresponding author. ReferencesAbarikwu, S.O., Pant, A.B. and Farombi, E.O. 2012. The protective effects of quercetin on the cytotoxicity of atrazine on rat Sertoli-germ cell co-culture. Int. J. Androl. 35(4), 590–600; doi:10.1111/j.1365-2605.2011.01239.x Abdeen, A., Abdelkader, A., Elgazzar, D., Aboubakr, M., Abdulah, O.A., Shoghy, K., Abdel-Daim, M., El-Serehy, H.A., Najda, A. and El-Mleeh, A. 2020. Coenzyme Q10 supplementation mitigates piroxicam-induced oxidative injury and apoptotic pathways in the stomach, liver, and kidney. Biomedicine Pharmacotherapy 130, 110627. Abdel Moneim, A.E. 2016. Prevention of carbon tetrachloride (CCl₄)-induced toxicity in testes of rats treated with Physalis peruviana L. fruit. Toxicol. Ind. Health. 32(6), 1064–1073; doi:10.1177/0748233714545502 Adewoyin, M., Ibrahim, M., Roszaman, R., Isa, M., Alewi, N., Rafa, A. and Anuar, M. 2017. Male infertility: the effect of natural antioxidants and phytocompounds on seminal oxidative stress. Diseases 5(1), 1–16; doi:10.3390/diseases5010009 Agarwal, A., Virk, G., Ong, C. and Du Plessis, S.S. 2014. Effect of oxidative stress on male reproduction. World J. Men's Health 32(1), 1. Alada, A., Akande, O. and Ajayt, F. 2004. Effect of soya bean diet preparations on some haematological and biochemical indices in the rat. Afr. J. Biomed. Res. 7(2), 71–75; doi:10.4314/ajbr.v7i2.54072 Alipour, M.R. and Karimi-Sales, E. 2020. Molecular mechanisms of protective roles of isoflavones against chemicals-induced liver injuries. Chem. Biol. Interact. 329, 109213; doi:10.1016/j.cbi.2020.109213 Amano, A., Kondo, Y., Noda, Y., Ohta, M., Kawanishi, N., Machida, S., Mitsuhashi, K., Senmaru, T., Fukui, M., Takaoka, O. and Mori, T. 2017. Abnormal lipid/lipoprotein metabolism and high plasma testosterone levels in male but not female aromatase-knockout mice. Arch. Biochem. Biophys. 622, 47–58. AOAC. 2000. Official Methods of Analysis, 17th ed. Washington DC, USA: Journal of AOAC International. Appt, S.E., Törmälä, R., Franke, A.A., Mikkola, T.S., Tikkanen, M.J., Ylikorkala, O. and Clarkson, T.B. 2008. Soy-tibolone combination: effect on lipids in postmenopausal monkeys and women. Maturitas 60(3–4), 216–222; doi:10.1016/j.maturitas.2008.06.003 Anatolia AM. 2014. The effect of heparin on the carbon tetrachloride induced changes in rat testis. Acta Med. Anatol. 2(2), 56–9. Aschemann-Witzel, J., Gantriis, R.F., Fraga, P. and Perez-Cueto, F.J. 2021. Plant-based food and protein trend from a business perspective: markets, consumers, and the challenges and opportunities in the future. Crit. Rev. Food Sci. Nutri. 61(18), 3119–3128. Assinder, S., Davis, R., Fenwick, M. and Glover, A. 2007. Adult-only exposure of male rats to a diet of high phytoestrogen content increases apoptosis of meiotic and post-meiotic germ cells. Reproduction 133(1), 11–19. Ayala, A., Muñoz, M.F. and Argüelles, S. 2014. Lipid peroxidation: production, metabolism, and signaling mechanisms of malondialdehyde and 4-hydroxy-2-nonenal. Oxid. Med. Cell. Longev. 2014, 360438; doi:10.1155/2014/360438 Azadbakht, L., Atabak, S. and Esmaillzadeh, A. 2008. Soy protein intake, cardiorenal indices, and C-reactive protein in type 2 diabetes with nephropathy. Diabetes. Care. 31(4), 648–654; doi:10.2337/dc07-2065 Bancroft J, Gamble M. 2008. Theory and practice of histological techniques 5th ed. Philadelphia, PA: Churchill Livingstone; 125–138. Calcaterra, V., De Giuseppe, R., Biino, G., Mantelli, M., Marchini, S., Bendotti, G., Madè, A., Avanzini, M.A., Montalbano, C., Cossellu, G. and Larizza, D 2017. Relation between circulating oxidized-LDL and metabolic syndrome in children with obesity: the role of hypertriglyceridemic waist phenotype. J. Pediatr. Endocrinol. Metabol. 30(12), 1257–1263. Chen, L.R., Ko, N.Y. and Chen, K.H. 2019. Isoflavone supplements for menopausal women: a systematic review. Nutrients 11(11), 2649; doi:10.3390/nu11112649 Costa, R.L. and Summa, M.A. 2000. Soy protein in the management of hyperlipidemia. Ann. Pharmacother. 34(7–8), 931–935; doi:10.1345/aph.19371 Cuevas, A.M., Irribarra, V.L., Castillo, O.A., Yañez, M.D. and Germain, A.M. 2003. Isolated soy protein improves endothelial function in postmenopausal hypercholesterolemic women. Eur. J. Clin. Nutr. 57(8), 889–894; doi:10.1038/sj.ejcn.1601622 Davies, M.J. 2016. Protein oxidation and peroxidation. Biochem. J. 473(7), 805–825; doi:10.1042/BJ20151227 Defalco, T., Potter, S.J., Williams, A.V., Waller, B., Kan, M.J. and Capel, B. 2015. Macrophages contribute to the spermatogonial niche in the adult testis. Cell. Rep. 12(7), 1107–1119; doi:10.1016/j.celrep.2015.07.015 Dutta, S., Chakraborty, A.K., Dey, P., Kar, P., Guha, P., Sen, S., Kumar, A., Sen, A. and Chaudhuri, T.K. 2018. Amelioration of CCl4 induced liver injury in swiss albino mice by antioxidant rich leaf extract of Croton bonplandianus Baill. PLoS One 13(4), e0196411. Ekere, S., Okoye, C. and Udoumoh, A. 2013. Fertility-enhancing effects of Dracaena arborea leaf extract in albino rats. Czech. J. Anim. Sci. 58(11), 520–524; doi:10.17221/7047-CJAS. Eljaafari, H., Mabrouk, Z.E., Mohamed, F., Tunsi, H. and Sasi, S.M. 2024. Carbon tetrachloride-induced testicular toxicity and histopathological alteration in Male Swiss Albino Mice. AlQalam J. Med. Appl. Sci. 7(1), 36–43. ElMazoudy, R.H., Attia, A.A. and El-Shenawy, N.S. 2011. Protective role of propolis against chlorpyrifos-induced reproductive toxicity in male rats. Pestic. Biochem. Physiol. 101(3), 175–181; doi:10.1016/j.pestbp.2011.09.003 Emmanuel, O., Okezie, U.M., Iweala, E.J. and Ugbogu, E.A. 2021. Pretreatment of red palm oil extracted from palm fruit (Elaeis guineensis) attenuates carbon tetrachloride induced toxicity in Wistar rats. Phytomedicine Plus 1(4), 100079. Engelman, H.M., Alekel, D.L., Hanson, L.N., Kanthasamy, A.G. and Reddy, M.B. 2005. Blood lipid and oxidative stress responses to soy protein with isoflavones and phytic acid in postmenopausal women. Am. J. Clin. Nutr. 81(3), 590 -596. Gibson-Corley, K.N., Olivier, A.K. and Meyerholz, D.K. 2013. Principles for valid histopathologic scoring in research. Vet. Pathol. 50(6), 1007–1015; doi:10.1177/0300985813485099 Godfray, H.C.J., Aveyard, P., Garnett, T., Hall, J.W., Key, T.J., Lorimer, J., Pierrehumbert, R.T., Scarborough, P., Springmann, M. and Jebb, S.A. 2018. Meat consumption, health, and the environment. Science 361(6399), eaam5324. Hamza, R.Z. and Diab, A.E.A.A. 2020. Testicular protective and antioxidant effects of selenium nanoparticles on monosodium glutamate-induced testicular structure alterations in male mice. Toxicol. Rep. 7, 254–260; doi:10.1016/j.toxrep.2020.01.012 Hashem, A.S. 2021. Defensive impact of propolis against CCl₄-actuated testicular damage in rats. J. Adv. Vet. Anim. Res. 8(1), 70–77; doi:10.5455/javar.2021.h487 Hosseinzadeh, H., Parvardeh, S., Asl, M.N., Sadeghnia, H.R. and Ziaee, T. 2007. Effect of thymoquinone and Nigella sativa seeds oil on lipid peroxidation level during global cerebral ischemia-reperfusion injury in rat hippocampus. Phytomedicine 14(9), 621 -627. Hussein, M. and Khan, R. 2022. CCl₄-induced hepatotoxicity: study in rats treated with camel milk and urine. J. Chem. Stud. 1(1), 7–11; doi:10.32996/jcs.2022.1.1.2 Ibrahim, H.A., Zhu, Y., Wu, C., Lu, C., Ezekwe, M.O., Liao, S.F. and Haung, K. 2012. Selenium-enriched probiotics improves murine male fertility compromised by high fat diet. Biol. Trace Element Res. 147(1), 251 -260. Javadi, L., Farzadi, L., Fathiazad, F. and Nouri, M. 2011. Anti-oxidative effects of citro flavonoids on spermatogenesis in rat. Afr. J. Pharm. Pharmacol. 5(6), 721 -725. Kaur, P., Dhandayuthapani, S., Venkatesan, T., Gantor, M. and Rathinavelu, A. 2020. Molecular mechanism of C-phycocyanin-induced apoptosis in LNCaP cells. Bioorganic. MedChem. 28(3), 115272; doi:10.1016/j.bmc.2019.115272 Keshtmand, Z., Akbaribazm, M., Bagheri, Y. and Oliaei, R. 2021. The ameliorative effects of Lactobacillus coagulans and Lactobacillus casei probiotics on CCl4‐induced testicular toxicity based on biochemical, histological and molecular analyses in rat. Andrologia, 53(1), e13908. Khedr, N.F. and Khedr, E.G. 2017. Branched-chain amino acid supplementation modulates TGF-β1/Smad signaling in CCl₄-induced liver fibrosis. Fundam. Clin. Pharmacol. 31(5), 534–545; doi:10.1111/fcp.12297 Khorrami, A., Ghanbarzadeh, S., Ziaee, M., Arami, S., Vajdi, R. and Garjani, A. 2015. Dietary cholesterol and oxidised cholesterol: effects on sperm characteristics, antioxidant status and hormonal profile in rats. Andrologia 47(3), 310 -317. Kim, M., Choi, S.H., Jin, Y.B., Lee, H.J., Ji, Y.H., Kim, J., Lee, Y.S. and Lee, Y.J. 2013. The effect of oxidized low-density lipoprotein (ox-LDL) on radiation-induced endothelial-to-mesenchymal transition. Int. J. Radiat. Biol. 89(5), 356 -363. Kosola, J., Ahotupa, M., Kyröläinen, H., Santtila, M. and Vasankari, T. 2013. Young men with poor cardiorespiratory fitness combined with lower testosterone have high levels of oxidized LDL lipids --being fit alters this relationship. Int. J. Sport Nutr. Exerc. Metabol. 23(6), 629 -637. Křížová, L., Dadáková, K., Kašparovská, J. and Kašparovský, T. 2019. Isoflavones. Molecules 24(6), 1076. Lee, R.D. and Nieman, D.C. 1996. Nutritional assessment, 2nd ed. Mosby, Missouri, USA. Li, L., Chen, X., Luo, Q., Huang, C., Liu, W. and Chen, Z. 2017. Effects of soy isoflavones on testosterone synthetase in diet-induced obesity male rats. Int. J. Clin. Exp. Pathol. 10(9), 9202. Lopes-Virella, M.F., Stone, P., Ellis, S. and Colwell, J.A. 1977. Cholesterol determination in high-density lipoproteins separated by three different methods. Clin. Chem. 23(5), 882 -884. Lowry, O., Rosebrough, N., Farr, A.L. and Randall, R. 1951. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193(1), 265–275; doi:10.1016/S0021-9258(19)52451-6 Magaki, S., Hojat, S.A., Wei, B., So, A. and Yong, W.H. 2018. An introduction to the performance of immunohistochemistry. Biobanking: methods and protocols. New York, NY: Springer New York; 289–298. Maheshwari, A., Misro, M.M., Aggarwal, A., Sharma, R.K. and Nandan, D. 2009. Pathways involved in testicular germ cell apoptosis induced by H2O2 in vitro. The FEBS J. 276(3), 870 -881. Maqdasy, S., Baptissart, M., Vega, A., Baron, S., Lobaccaro, J.M.A. and Volle, D.H. 2013. Cholesterol and male fertility: what about orphans and adopted?. Mol. Cell. Endocrinol. 368(1-2), 30 -46. Miura, T., Ohta, T., Miura, C.I. and Yamauchi, K. 2003. Complementary deoxyribonucleic acid cloning of spermatogonial stem cell renewal factor. Endocrinology 144(12), 5504 -5510. Murali, B., Korrapati, M.C., Warbritton, A., Latendresse, J.R. and Mehendale, H.M. 2004. Tolerance of aged Fischer 344 rats against chlordecone-amplified carbon tetrachloride toxicity. Mech. Ageing Dev. 125(6), 421 -435. Nasrabadi, M.N., Doost, A.S. and Mezzenga, R. 2021. Modification approaches of plant-based proteins. Food. Hydrocoll. 118, 106789; doi:10.1016/j.foodhyd.2021.106789 Ogbuewu, I.P., Omede, A.A., Chukwuka, O.K., Iheshiulor, O.O.M., Uchegbu, M.C., Udebuani, A.C., Ekenyem, B.C., Okoli, I.C. and Iloeje, M.U. 2010. The overview of the chemistry, health benefits and the potential threats associated with prolonged exposure to dietary soy isoflavones. Int. J. Agri. Res. 5(12), 1084 -1099. Ohkawa, H., Ohishi, W. and Yagi, K. 1979. Colorimetric method for malondialdehyde determination. Anal. Biochem. 95(2), 351–358; doi:10.1016/0003-2697(79)90738-3 Opferman, J.T. and Kothari, A. 2018. Anti-apoptotic BCL-2 family members in development. Cell. Death. Differentiation. 25(1), 37–45; doi:10.1038/cdd.2017.170 Othman, M.S., Nada, A., Zaki, H.S. and Abdel Moneim, A.E. 2014. Effect of Physalis peruviana L. on cadmium-induced testicular toxicity in rats. Biol. Trace Element Res. 159(1), 278 -287. Oyeyemi, M.O. and Okediran, B.S. 2007. Testicular parameters and sperm morphology of chinchilla rabbit fed with different planes of soymeal. Int. J. Morphol. 139–44; doi: 10.4067/S0717-95022007000100021 Paglia, D.E. and Valentine, W.N. 1967. Characterization of erythrocyte glutathione peroxidase. J. Lab. Clin. Med. 70(1), 158–169. Patisaul, H.B. 2017. Endocrine disruption by dietary phyto-oestrogens. Proc. Nutr. Soc. 76(2), 130–144; doi:10.1017/S0029665116000677 Rajesh, M. and Latha, M. 2004. Protective activity of Glycyrrhiza glabra on CCl₄-induced damage. Indian J. Pharmacol. 36(5), 284–287. Rietjens, I., Louisse, J. and Beekmann, K. 2017. Potential health effects of dietary phytoestrogens. Br. J. Pharmacol. 174(11), 1263–1280; doi:10.1111/bph.13622 Salam, R., Kshetrimayum, A.S. and Keisam, R. 2012. Testosterone and metabolic syndrome. Indian. J. Endocrinol. Metab. 16(Suppl 1), S12–S19; doi:10.4103/2230-8210.94248 Shiraishi, K. and Matsuyama, H. 2017. Gonadotropin actions on spermatogenesis. Endocr. J. 64(2), 123–131; doi:10.1507/endocrj.EJ17-0001 Sinha, K., Das, J., Pal, P.B. and Sil, P.C. 2013. Oxidative stress: the mitochondria-dependent and mitochondria-independent pathways of apoptosis. Arch. Toxicol. 87(7), 1157 -1180. Sönmez, M., Türk, G., Çeribaşı, S., Çiftçi, M., Yüce, A., Güvenç, M.E.H.M.E.T., Özer Kaya, Ş., Çay, M. and Aksakal, M. 2014. Quercetin attenuates carbon tetrachloride‐induced testicular damage in rats. Andrologia 46(8), 848–858. Tijani, A.S., Daba, T.M., Ubong, I.A., Olufunke, O., Ani, E.J. and Farombi, E.O. 2024. Rutin attenuated hexachlorobenzene-induced testicular injury via regulation of oxidative stress, steroidogenic enzymes and apoptotic process in male rats. Eur. J. Med. Chem. Rep. 10, 100121. Turner, T.T. and Lysiak, J.J. 2008. Oxidative stress in testicular dysfunction. J. Androl. 29(5), 488–498; doi:10.2164/jandrol.108.005132 Unsal, V., Cicek, M. and Sabancilar, I. 2021. Toxicity of carbon tetrachloride and antioxidants. Rev. Environ. Health 36(2), 279–295; doi:10.1515/reveh-2020-0048 Wagner, J.D., Anthony, M.S. and Cline, J.M. 2001. Soy phytoestrogens: benefits and risks. Clin. Obstet. Gynecol. 44(4), 843–852; doi:10.1097/00003081-200112000-00022 Xu, Y., Xie, L., Tang, J., He, X., Zhang, Z., Chen, Y., Zhou, J., Gan, B. and Peng, W. 2021. Morchella importuna polysaccharides alleviate carbon tetrachloride-induced hepatic oxidative injury in mice. Front. Physiol. 12, 669331. Yousef, M.I. and Salama, A.F. 2009. Propolis protects against aluminium chloride reproductive toxicity. Food Chem. Toxicol. 47(6), 1168–1175; doi:10.1016/j.fct.2009.02.006 Zhang, Z., Yu, Y., Xu, H., Wang, C., Ji, M., Gu, J., Yang, L., Zhu, J., Dong, H., Wang, S.L. 2017. High-fat diet aggravates 2, 2′, 4, 4′-tetrabromodiphenyl ether-inhibited testosterone production via DAX-1 in Leydig cells in rats. Toxicol. Appl. Pharmacol. 323, 1 -8. | ||
| How to Cite this Article |
| Pubmed Style Al-amgad Z, El-aziz AAA, Madkour FA, Al-amgad H, Basher AW, Mohammed HA, Elnagar A, Rehan IF, Zigo F, Zigová M, Jad MM. Attenuation of carbon tetrachloride-induced male reproductive damage in albino laboratory rats using varying concentrations of supplemented soybean. doi:10.5455/OVJ.2026.v16.i3.37 Web Style Al-amgad Z, El-aziz AAA, Madkour FA, Al-amgad H, Basher AW, Mohammed HA, Elnagar A, Rehan IF, Zigo F, Zigová M, Jad MM. Attenuation of carbon tetrachloride-induced male reproductive damage in albino laboratory rats using varying concentrations of supplemented soybean. https://www.openveterinaryjournal.com/?mno=277757 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.37 AMA (American Medical Association) Style Al-amgad Z, El-aziz AAA, Madkour FA, Al-amgad H, Basher AW, Mohammed HA, Elnagar A, Rehan IF, Zigo F, Zigová M, Jad MM. Attenuation of carbon tetrachloride-induced male reproductive damage in albino laboratory rats using varying concentrations of supplemented soybean. doi:10.5455/OVJ.2026.v16.i3.37 Vancouver/ICMJE Style Al-amgad Z, El-aziz AAA, Madkour FA, Al-amgad H, Basher AW, Mohammed HA, Elnagar A, Rehan IF, Zigo F, Zigová M, Jad MM. Attenuation of carbon tetrachloride-induced male reproductive damage in albino laboratory rats using varying concentrations of supplemented soybean. doi:10.5455/OVJ.2026.v16.i3.37 Harvard Style Al-amgad, Z., El-aziz, . A. A. A., Madkour, . F. A., Al-amgad, . H., Basher, . A. W., Mohammed, . H. A., Elnagar, . A., Rehan, . I. F., Zigo, . F., Zigová, . M. & Jad, . M. M. (2026) Attenuation of carbon tetrachloride-induced male reproductive damage in albino laboratory rats using varying concentrations of supplemented soybean. doi:10.5455/OVJ.2026.v16.i3.37 Turabian Style Al-amgad, Zeinab, Amany Ahmed Abd El-aziz, Fatma A. Madkour, Hend Al-amgad, Asmaa W. Basher, Heba A. Mohammed, Asmaa Elnagar, Ibrahim F. Rehan, František Zigo, Martina Zigová, and Mariam M. Jad. 2026. Attenuation of carbon tetrachloride-induced male reproductive damage in albino laboratory rats using varying concentrations of supplemented soybean. doi:10.5455/OVJ.2026.v16.i3.37 Chicago Style Al-amgad, Zeinab, Amany Ahmed Abd El-aziz, Fatma A. Madkour, Hend Al-amgad, Asmaa W. Basher, Heba A. Mohammed, Asmaa Elnagar, Ibrahim F. Rehan, František Zigo, Martina Zigová, and Mariam M. Jad. "Attenuation of carbon tetrachloride-induced male reproductive damage in albino laboratory rats using varying concentrations of supplemented soybean." doi:10.5455/OVJ.2026.v16.i3.37 MLA (The Modern Language Association) Style Al-amgad, Zeinab, Amany Ahmed Abd El-aziz, Fatma A. Madkour, Hend Al-amgad, Asmaa W. Basher, Heba A. Mohammed, Asmaa Elnagar, Ibrahim F. Rehan, František Zigo, Martina Zigová, and Mariam M. Jad. "Attenuation of carbon tetrachloride-induced male reproductive damage in albino laboratory rats using varying concentrations of supplemented soybean." doi:10.5455/OVJ.2026.v16.i3.37 APA (American Psychological Association) Style Al-amgad, Z., El-aziz, . A. A. A., Madkour, . F. A., Al-amgad, . H., Basher, . A. W., Mohammed, . H. A., Elnagar, . A., Rehan, . I. F., Zigo, . F., Zigová, . M. & Jad, . M. M. (2026) Attenuation of carbon tetrachloride-induced male reproductive damage in albino laboratory rats using varying concentrations of supplemented soybean. doi:10.5455/OVJ.2026.v16.i3.37 |