| Research Article | ||
Open Vet. J.. 2026; 16(1): 346-353 Open Veterinary Journal, (2026), Vol. 16(1): 346-353 Research Article Histopathological study of the liver and spleen lesions in pigeons experimentally infected with Chlamydia psittaciSuhail Wadi Al-Sultani* and Atheer Abdulrazzaq Al-DooriDepartment of Microbiology, College of Veterinary Medicine, University of Baghdad, Baghdad, Iraq *Corresponding Author: Suhail Wadi Al-Sultani. Department of Microbiology, College of Veterinary Medicine, University of Baghdad, Baghdad, Iraq. Email: sohailalsoltani [at] gmail.com Submitted: 13/08/2025 Revised: 28/11/2025 Accepted: 10/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
AbstractBackground: Chlamydia psittaci, the zoonotic pathogen that causes avian chlamydiosis, is well known to infect domestic pigeons (Columba livia domestica), serving as a primary natural reservoir. Aim: The purpose of this study was to investigate the histopathological effects of the C. psittaci isolate on hepatic and splenic organ tissues in experimentally infected pigeons. Methods: A detailed survey of histopathological abnormalities induced by the genotype B isolate of C. psittaci outer membrane protein A (ompA) in eight experimentally infected (oro-nasal) healthy pigeons was performed. The strain was isolated from infected domestic pigeons in Baghdad, Iraq. The isolate was registered and obtained an accession number (PV243292.1) in GenBank-NCBI. Results: The results showed that the isolate induced hepatitis, splenitis, airsacculitis, conjunctivitis, bronchitis, sinusitis, and enteritis (within 28 days post-infection) in euthanized pigeons. Severe gross and histologic lesions were noted in all tissues of the experimental pigeons, comprising tiny necrosis areas and infiltration of leukocytes in the tissues of the liver and spleen, as well as multifocal hepatitis. The birds with hepatomegaly and splenomegaly had increased numbers of mononuclear phagocytic system cells and loss of normal architecture. Intracytoplasmic inclusions were detected in all tissues of the liver and spleen. Conclusion: The study concluded that C. psittaci is a major disease that causes noticeable gross clinical and histological abnormalities in domestic pigeons. This zoonotic pathogen requires increased attention in the context of domestic pigeons in Iraq. It was considered the first study on the histopathological effects of the C. psittaci isolate on experimentally infected pigeons in Baghdad. Keywords: Chlamydiosis, Experimental pigeons, Hepatomegaly, Intracytoplasmic inclusions, Splenomegaly. IntroductionChlamydia psittaci is a member of the obligatory intracellular bacterial family Chlamydiaceae that maintains its life cycle in the host cell within a nonacidified vacuole called an inclusion (Zaręba-Marchewka et al., 2020). They have deoxyribonucleic acid (DNA), ribonucleic acid (RNA), and a cell wall, which are characteristics of bacteria. In recent years, the Chlamydiaceae family has experienced tremendous expansion (Wu et al., 2013; Al-Jumaa et al., 2024). Currently, the family is classified under the genus Chlamydia and 14 species. Some of these species are C. trachomatis, C. pneumoniae, C. psittaci, C. felis, C. muridarum, C. gallinacean, C. serpentis, and C. poikilotherm (Laroucau et al., 2020). Chlamydia psittaci was thought to be the primary chlamydial species in poultry until 2009. This species is common and has been found in a number of animal species as well as in over 465 bird species, including pigeons (Kaleta and Taday, 2003). In accordance with the host, age, stressors, and chlamydial strain, infections can cause substantial systemic sickness and death in addition to remaining asymptomatic. Furthermore, infections in humans can eventually lead to severe pneumonia due to C. psittaci’s proven zoonotic potential. (Harkinezhad et al., 2009). Genotyping based on the ompA gene confirmed the circulation of Chlamydia psittaci genotype B isolate (PV243292.1) in Iraq, a strain commonly associated with pigeons and known for its zoonotic potential (Al-Sultani, 2025). The strains of C. psittaci that caused high mortality in psittacine were more virulent than the strains of pigeon. Compared to ruminant strains, avian strains were significantly more pathogenic toward chickens. A bovine model of respiratory C. psittaci infection developed respiratory disease due to a ruminant strain (DC15) (Al-Darraji et al., 1992; Reinhold et al., 2012). Fibrinous airsacculitis, hepatomegaly, and splenomegaly may be observed in birds with avian chlamydiosis, but no specific gross lesion is pathognomonic (Balsamo et al., 2017). Remarkably, C. psittaci is especially effective at spreading throughout the host organism, resulting in systemic illness that can occasionally progress to fulminant. At the cellular level, the bacteria have a broad host cell range (from macrophages to epithelial cells), rapid entry and reproduction, and skillful use of intracellular transport channels to mitochondria and the Golgi apparatus (Knittler and Sachse, 2015). Histopathology remains a valuable diagnostic method that aids in our comprehension of how infections proceed. However, it should be carried out in combination with additional verifying diagnostic tests (Hafez et al., 2025). For direct visualization, the Chlamydia can be detected in tissue sections by cytological staining techniques such as Giemsa, H&E, and Gimenez stains, used on histological preparations; these will identify the infectious inclusion bodies (elementary body and reticulate body) of Chlamydia (Campbell, 2015). As far as we are aware, not one study has been done on avian chlamydiosis in this region; thus, the objective of this work was to perform an examination of the gross pathology and the histopathological effects induced by pigeon-derived C. psittaci isolate in an infected (oronasal) healthy pigeon model. It would enable us to investigate the generally held belief that C. psittaci, isolated from domestic pigeons of Iraq, poses a zoonotic risk since it is pathogenic to pigeons. Another goal of this investigation was to collect baseline data on the virulence of C. psittaci in domestic pigeons in Baghdad, Iraq, and help to diagnose this pathogen for better treatment and prevention. Materials and MethodsAnimals and housingEight mature domestic pigeons, aged between 4 and 5 months of age and weighing 200–250 g, were obtained from the bird shop, Baghdad, Iraq. The pigeons were kept in an ideal, controlled environment within the bird shop, where temperature, humidity, and ventilation were provided water and food were provided ad libitum. These pigeon tests do not include Chlamdiaceae; hence, eight pigeon cloacal swabs were obtained. The swabs were immediately transferred into viral transport medium (Liofilchem, USA) under aseptic conditions and transported on ice packs to the laboratory for further analysis. Before the pigeons were delivered, all swabs tested were enzyme-linked immunosorbent assay (ELISA) and polymerase chain reaction (PCR) negative for Chlamydia spp. to ensure that there was no active Chlamydia infection. Prior to the initial inoculation, all pigeons were acclimated for 7 days. Following their arrival, the pigeons were kept in temperature-controlled chambers with ideal humidity and light levels, in groups on sawdust bedding. The temperature levels in the pigeon environment (20°C–30°C) , humidity (40%–60%), air extractors with filters for ventilation, the light levels (12–14 hours) daily, drinking water is provided in irrigation channels protected from droppings, finally, the food was given in special, clean feeders. They have unlimited access (ad libitum) to food and drink. Enzyme-linked immunosorbent assay kitThe samples were tested with an ELISA kit (Pigeons Chlamydia psittaci ELISA Kit, for the qualitative determination of Pigeons C. psittaci, Pigeons Sandwich ELISA Detection, AFG Bioscience, USA). This test is used for the detection of these bacteria; it is more sensitive than other serological tests (Noomi, 2018). To obtain a high concentration of antigen, the samples were centrifuged at 3,000 g for 20 minutes after being left at room temperature for 4 hours. The assay kit as directed by the manufacturer, C. psittaci was used in the sample. Purified antibody was used to coat the microtiter plate, create a solid-phase antibody, and then add the sample to the wells. When combined with C. psittaci antibody that was horseradish peroxidase (HRP) labeled, the result was an antibody-antigen-enzyme-antibody complex. Following thorough washing, Tetramethylbenzidine substrate solution was added, which turned blue. After the HRP enzyme-catalyzed reaction is stopped by adding a 0.1N H2SO4 solution, the color change is measured spectrophotometrically (optical density OD) at a wavelength of 450 nm using an ELISA microplate reader (Expert plus microplate reader, Germany), after the non-combinative antigen and other materials have been cleaned and removed. The OD samples were negative because they were less than the cutoff value (Al-Nuaimy and Al-Jandeel, 2018; Al Sultani et al., 2025). DNA extraction and PCR amplificationSamples were vortexed and centrifuged at 5,000 rpm for 5 minutes. DNA was extracted using the G-spin™ Genomic DNA Kit for Bacteria (Intron, Korea), following the manufacturer’s protocol. PCR detection of C. psittaci was based on amplification of the 16S rRNA gene, using universal primers for the genus Chlamydophila (Sachse et al., 2015). PCR reactions were prepared in 25 μl volumes, containing: 10 μl GoTaq® G2 Green Master Mix (Promega, USA), 4 μl primer mix (forward and reverse), 4 μl template DNA, 7 μl PCR-grade distilled water. Thermal cycling conditions: Initial denaturation: 94°C for 5 minutes, 35 cycles of: Denaturation: 94°C for 30 seconds, Annealing: 54°C for 30 seconds, Extension: 72°C for 1 minute, and Final extension: 72°C for 5 minutes. Electrophoresis was conducted at 90 V for 45 minutes. Gels were visualized using a UV transilluminator (Analytik Jena, UK) at 320 nm. The presence of a 418 bp band was considered positive for C. psittaci (Vilela et al., 2019). All samples tested were PCR negative for Chlamydia spp. Preparation of Chlamydia psittaci isolateChlamydia psittaci isolate (PV243292.1) is an in-house reference isolate of ompA genotype B in the Gene Bank. It was isolated from infected domestic pigeons in Baghdad, Iraq (AL-Sultani, 2025). A 20% yolk sac suspension in sucrose phosphate glutamate was used to preserve the isolate until inoculation at −80°C. The infectious dose was within one log step of the original dose, and the inoculation dose was verified by reverse titration according to Reed and Muench (Muench, 1938). Titration experiments in specific pathogen-free (SPF) embryonated chicken eggs were performed to quantify the infectious dose. Alethea et al. (2014) found that the Ten-fold serial dilutions of third passage yolk sac cultures of C. psittaci isolate were used to calculate the 50% egg infective dose (EID50). Inoculation was done orally and nasally for C. psittaci; the respiratory and oral methods have been documented by Takahashi et al. (1988). Experimental designThe experiment consisted of two groups. In the first group, six randomly selected pigeons were oro-nasally inoculated with the C. psittaci isolate (0.2 ml of 106.3 EID50/ml). The two pigeons that remained were oro-nasally inoculated with BPS (0.2 ml) and used as a second (control) group. The two groups were kept separate. The groups were transferred to a new site with BSL-3 facilities on day 28 following the initial inoculation. In order to avoid cross-contamination, both groups traveled independently. The inoculations were performed with a syringe 18G × 1 ml × 1.5 (Medicalinject, Germany). Pigeons were first given a single droplet of the inoculum (0.2 ml of 106.3 EID50/ml) in one nostril before the remained suspension was given to them orally as part of the oro-nasal inoculation. Clinical indicators (weight, temperature, appetite, and activity) were documented every day for the duration of the investigation (28 days) using a clinical rating card. Azizpour and Hassani, (2012) revealed that the pigeons were euthanized by cervical dislocation while being anesthetized with an intramuscular injection of a combination of 10 mg/ kg xylazine (Bimeda, Canada) and 30 mg/kg ketamine 10% (Alfasan, Netherlands). NecropsyThe euthanized pigeons were dissected and grossly examined for any abnormalities in the liver and spleen. Carcasses were opened during necropsy on a sterile plastic sheet, which was changed after every necropsy. For each tissue sample (organ), sterile equipment and petri dishes were utilized to avoid cross-contamination. Samples of tissue (liver and spleen) of euthanized pigeons were taken for histopathology. Histopathological examinationAfter at least 48–72 hours of fixation in 10% neutral buffered formalin, the samples were processed by dehydration with ethanol, clearing with xylene, and embedding with paraffin. Utilizing a rotary microtome to section the specimen onto a plane with a thickness of 5 µm. using a rotary microtome, then mounting the tissue sections using cleaned glass slides and staining with H&E, Giemsa, and the special Gimenez stains (Bancroft and Gamble, 2008 Yadav and Sagar, 2022). The histopathological processing steps were performed manually. Histopathological analysis of the tissue sections was examined for histopathological changes using a light microscope with the digital AmScope camera HD182 (AmScope Co., California, USA) for photography (Yin et al., 2015). Ethical approvalThe College of Veterinary Medicine at the University of Baghdad has been granted moral sanction and an endorsement certificate (number P.G./1246) by the committee of scientific morality to conduct this methodical activity. ResultsThe clinical signs observed on the C. psittaci-inoculated pigeons during the experiment were nonspecific. While there were no clinical symptoms or weight variations in the control pigeons. The isolate induced symptoms in all infected pigeons, such as, lethargy, weight loss, conjunctivitis, respiratory signs (dyspnea and nasal discharges), and enteritis within 28 days post-infection, but no mortality was recorded in infected pigeons. The onset of symptoms appeared as early as 1 week post-inoculation, which is an indication of a systemic infection resulting from replication of C. psittaci in the host (Fig. 1). Control birds did not reveal any signs of chlamydiosis. The course of infection with C. psittaci could be monitored until 3 weeks post-inoculation.
Fig. 1. A/ Pre- Pre-inoculated domestic pigeon. B/ One week after experimentally infecting a pigeon with C. psittaci. Lethargy, ruffled feathers, conjunctivitis, and weight loss appear. At necropsy, the liver and spleen of the control birds showed normal appearance without any pathological changes such as congestion, hemorrhage, or necrotic lesions. While in the inoculated pigeon with the C. psittaci isolate, the liver and spleen were observed to be enlarged and congested with hemorrhagic spots. In addition, hepatitis, splenitis, fibrinous polyserositis, and exudative airsacculitis appeared in infected pigeons. All infected groups had diffuse opacity of the abdominal and thoracic air sacs, according to gross pathology. Hepatomegaly, congestion, and petechiae of the liver were noticed at gross pathology; hepatitis was present in all infected pigeons. Fibrinous adhesive pericarditis and thickening of the epicardium were observed. Splenomegaly and the presence of petechiae were indicators of macroscopic lesions in the spleen of an infected pigeon with C. psittaci. The necropsy figures of the control group and the experimentally infected pigeons were illustrated in Figures 2–4.
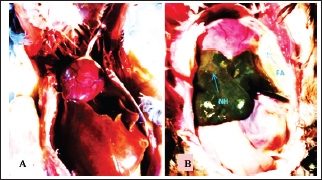
Fig. 2. A). Normal internal organs in a control pigeon. B). Gross lesion of chlamydiosis in a 4-month-old domestic pigeon, Necrotizing hepatitis (NH): Fibrinous airsaculitis (FA) and fibrinous polyserositis (FP).
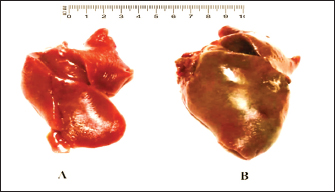
Fig. 3. A). Normal liver of a 4-month-old domestic pigeon (control). B). Hepatomegaly and congestion of the liver in a C. psittaci-infected pigeon.
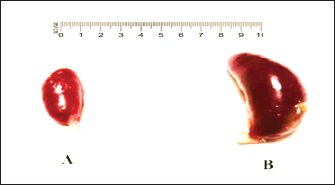
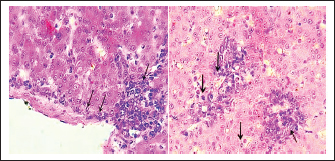
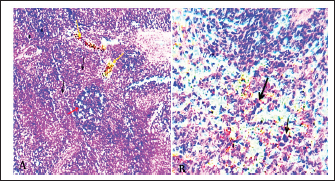
Fig. 4. A). Normal spleen (control), B). splenomegaly and congestion of the spleen in an infected pigeon with C. psittaci. The microscopic examination of the liver and spleen sections revealed moderate to severe pathologic changes compared with the normal histological view of the sections from the control group. The histopathological figure of the infected liver showed hepatitis with multiple necrosis of hepatocytes and marked aggregation of the mononuclear leukocytes, periportal aggregation of leukocytes, with dilation of portal veins (Fig. 5). Histopathology of infected liver reveals chlamydial inclusion bodies (IB) within the cytoplasm of macrophages, as shown in Figures 6–8.
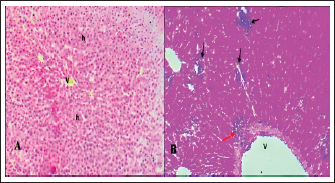
Fig. 5. A). Section of liver (Control) shows a normal central vein (V), a normal hepatic cords (h). H&E.100x. B). Section of liver (infected) shows hepatitis with multiple necroses (black arrows), periportal aggregation of leukocytes with dilation of portal veins (red arrow). H&E.100x.
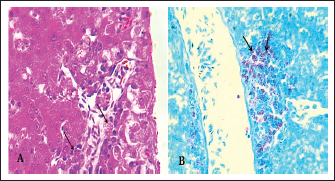
Fig. 6. Section of infected liver with C. psittaci shows marked aggregation of the mononuclear leukocytes with chlamydial reticulate inclusion bodies (IB) within the cytoplasm of macrophages (Arrow), H&E stain, 400x.
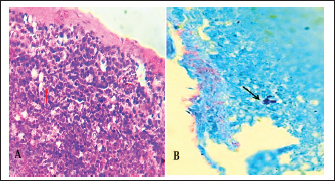
Fig. 7. A). Section of chlamydial-infected liver shows chlamydial reticulate inclusion bodies (IB) within the cytoplasm of hepatocytes (Arrow), H&E stain, 400x. B). Section of infected liver shows inclusion bodies (IB) stained purple within the cytoplasm of macrophages (Arrows) with periportal aggregation of leukocytes and dilation of portal veins, Giemsa stain, 400x.
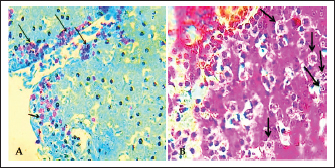
Fig. 8. Section of liver (infected) shows chlamydial inclusion bodies (IB) within the cytoplasm of macrophages (Arrows), 400x, A). Giemsa stain, B). Gimenez stain. The normal histological view of the spleen sections showed a normal lymphoid follicles and normal splenic sinusoidal red pulp with normal population of megakaryocytes (Fig. 9), compared with the histopathological figures of the infected spleen that showed marked atrophy of lymphoid follicles with multiple focal cellular depletion and mild congestion of splenic sinusoid and focal hemorrhage with presence of fibrin thrombi (Fig. 10), other figures showed sub capsular degeneration of lymphoid tissue with infiltration of leukocytes. The magnification of these figures of spleen shows chlamydial inclusion bodies of C. psttaci within the cytoplasm of macrophages (Fig. 11).
Fig. 9. A). Section of spleen (control) shows: normal lymphoid follicles (black arrow) and normal splenic sinusoidal red pulp (red arrow). H&E stain.100x. B). Section of spleen (control) shows: normal lymphoid follicles and normal splenic sinusoid with normal population of megakaryocytes and splenic cords. H&E stain.400x.
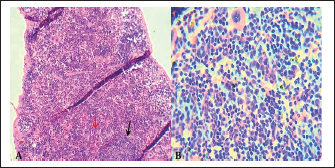
Fig. 10. A). Section of chlamydial-infected spleen shows: atrophy of lymphoid follicles with multiple focal cellular depletion (red arrows) and congestion with distention of splenic sinusoid (black arrow) and focal hemorrhage (yellow arrow). H&E stain 100x. B). Section of spleen (infected) shows chlamydial inclusion bodies (IB) within the cytoplasm of macrophages (black arrows). H&E stain 400x.
Fig. 11. A). Section of spleen (infected) shows subcapsular degeneration of lymphoid tissue with infiltration of leukocytes and chlamydial inclusion bodies (Red arrow). H&E stain 400x. B). Individual chlamydial inclusion bodies (IB) of C. psittaci stained purple within the cytoplasm of macrophages (Black arrow). Giemsa stain 400x. DiscussionThe obtained results demonstrated that experimental infections of domestic pigeons with pigeon-derived C. psittaci isolate induced a clinical course of systemic infection with complications of the respiratory tract. Takahashi et al. (1988) demonstrated that, given that infections with C. psittaci are more effective when administered orally, the infected birds received an oro-nasal injection that resulted in a more systemic infection, especially in the liver and spleen. In previous studies, researchers used SPF chickens for experimental infection with C. psittaci (Suwa and Itakura, 1992; Yin et al., 2013a; Yin et al., 2015). In this study, we used pigeons for the first time as a model for experimental infection with C. psittaci because it is a primary target for this pathogen. However, until recently, it was widely believed that pigeons were less susceptible to the disease caused by C. psittaci than ducks, turkeys, and Psittacine. (Reinhold et al., 2012). Perhaps for this reason, avian chlamydiosis in pigeons is no longer of interest to study in this region. The reason for this limitation of the small sample size (eight pigeons) used in this study was that the goal was simply to obtain positive experimental infection and observe the histopathological changes, regardless of the number of infected pigeons, in order to prove thesuccess of the experiment. The difficulty of obtaining pathogen-free pigeons to conduct the experiment was another cause for the small sample size. It is appropriate to mention that the use of pigeons in this study as an experimental model is the first of its kind, at least in this region. It should be noted that it is very difficult to obtain disease-free birds for testing, and many tests must be carried out to ensure they are disease-free, as is the case in this study. In this study, we confirmed that domestic pigeons were more susceptible to the disease through the experimental infection with C. psittaci that caused severe histopathological effects in the internal organs particularly the liver and spleen. However, some studies demonstrated that C. psittaci is the most pathogenic agent found in the pigeons’ environment, and it has a high prevalence (Sariya et al., 2015). Therefore, when infected birds exhibit gross clinical and histological lesions as described here, a thorough C. psittaci evaluation of the internal organs in pigeons is justified. The results of this study showed that the normal histological structure of the liver of a control pigeon represents the lobular pattern of the central vein, from which radiate the hepatic cords, divided by blood sinusoids. The normal histological structure of the spleen of a control pigeon represents normal lymphoid follicles and normal splenic sinusoidal red pulp; this was pointed out by Yin et al. (2015) in their research on chickens. The present study’s gross pathology was either the same as or less severe than that reported for naturally infected birds, which are frequently thought to be especially vulnerable to developing severe pathology after contracting C. psittaci. Furthermore, the gross lesions varied greatly and were not pathognomonic. A previous similar study on a psittacine bird by Billington (2005) found that the most typical findings in parrots include fibrinous airsacculitis, pericarditis, splenomegaly, and hepatomegaly. Generally, lesions resulting from systemic chlamydiosis in infected birds are commonly reported to affect the gastrointestinal tract, liver, and spleen (Walsh and Shivaprasad, 2023). A similar study on the chicken by Yin et al. (2015) confirmed that experimental infections with C. psittaci strains induced moderate to severe clinical signs of disease. Guo et al. (2016) reported that the experimental chlamydial infections of the chicken with C. gallinacea may not cause clinical symptoms; they may cause production loss, such as decreased weight gain. We conclude from the difference in the results of the two previous studies that C. psittaci are more virulent to domestic birds, and thus confirms its virulence to the pigeons, which this consistent with the results of this study. The experiment of the present study was an inoculation of C. psittaci genotype B isolate, which induced hepatitis, splenomegaly, and fibrinous airsacculitis, and no mortality rate was recorded in the infected pigeons. This agrees with an earlier study by Yin et al. (2013b), who trialed different genotypes of C. psittaci in chickens and found that the genotype B strains cause severe systemic diseases in the infected birds, while the infections with genotype D caused more severe clinical illness and a high mortality rate in infected birds (Yin et al., 2013b). The genotype or strain of C. psittaci and the stage of infection may influence the degree of pathogenesis. Following the inoculation, C. psittaci was found in organs like the liver and spleen, indicating that either no immune response was produced or that infection could not be prevented. Neutralizing antibodies against the variable domain of major outer membrane protien were crucial in avoiding infection in a mouse model, according to a C. trachomatis vaccine study by Olsen et al. (2017). However, this impact was also specific. Therefore, more in vitro research is required to determine whether an infection with C. psittaci causes an immune response in the form of neutralizing antibodies and whether these antibodies have a neutralizing effect on C. psittaci. Suwa et al. (1990) recorded that the acute chlamydial septicemic lesions were induced by the inoculation into the air sac and trachea, while no lesions were produced by the pharyngeal inoculation. The birds in the previously mentioned tests were not infected by aerosol inhalation or oral contact, which is the normal route. Rather, the trachea or air sac received a direct injection of Chlamydia. Different approaches could be the cause of these differences in the results. The present histopathological study focused on the investigation of histopathological lesions in the liver and spleen of infected pigeons. These lesions were severe and obvious as a result of the high replication of C. psittaci. However, a previous study on chickens revealed that replication of C. psittaci is the highest in the air sacs and, to a lesser extent, in the lung, liver, and spleen tissues of infected birds (Yin et al., 2015). The strain virulence of C. psittaci and the type of animal model may be the reason for this difference. Most previous studies on the pathogenesis and histopathological effects of C. psittaci isolates are not recent. They have been performed in different species of birds and were intended to elucidate both the pathogenesis and virulence of isolates in one experimental design. A study conducted by Heijne et al. (2021) on the chicken demonstrated that throughout the entire investigation, the C. psittaci quantitative PCR was negative in tissue samples, pharyngeal swabs, and cloacal swabs. We lack a clear explanation for this observation in this part of the study, which contradicts the results of the present study. We speculate that it may be due to biological variance or heterogeneity in illness susceptibility or outcome between species, despite the fact that they have a common ancestor. The severity of lesions and histopathological changes by the Chlamydia isolate in infected pigeons indicates how virulent and dangerous to people that contact. Therefore, the presence of this pathogen in Baghdad’s urban pigeon population highlights a public health concern, especially for individuals with occupational or recreational bird exposure. Often, conclusions have been made on low numbers of experimental animals, with heterogeneous birds (not SPF or otherwise defined) and without appropriate controls. Histopathological examination alone cannot be relied upon to diagnose chlamydiosis in pigeons, and PCR testing must be used to confirm the presence of the disease in the birds because it is a specific, accurate, and sensitive test. Molecular biological analysis is important to detect C. psittaci and differentiate between the chlamydial strains found in the numerous bird species, to predict potential risks of avian strains to humans and to other animals. This study contributes to the pathogenic epidemiological understanding of C. psittaci in Iraq. ConclusionPigeons were successfully infected with the C. psittaci isolate in the experimental domestic pigeons, whereas the controls did not have any pathological changes. Chlamydia is detected in systemic organs such as the spleen and liver. During the experimental period, there was a clear appearance of clinical indicators of chlamydial infection in the pigeons. As with other bird species, C. psittaci is a pathogen that causes noticeable gross pathological and histological lesions in pigeons. Therefore, C. psittaci is significant as a risk in the domestic pigeon environment, and it may pose a zoonotic risk to humans. All stains used in this study (Hematoxylin–Eosin, Giemsa, and Gimenez) were successful in clearly showing the inclusion bodies of C. psttaci in tissue sections of infected pigeons. The Giemsa stain is considered the most elucidating of the histological changes among the stains. AcknowledgmentsWe gratefully thank the staff of the Department of Pathology, College of Veterinary Medicine, University of Baghdad, for their technical assistance. Conflict of interestThe writers confirm that they have no conflicts of interest to declare. FundingThe authors have not received any funds for this work. Data availabilityAll data is available in this manuscript. ReferencesAl Sultani, S.W., Al-Doori, A.A. and Samawi, F.T. 2025. Prevalence and Zoonotic Potential of Chlamydia psittaci in Domestic Pigeons from Baghdad: eLISA and Rapid Test Insights. Dijlah. J. Med. Sci. 1(3), 189–192. Alethea, M.F., Sandra, K.C. and Peg, A.P., 2014. Supplemental assay method for titration of Chlamydophila felis (formly feline Chlamydia psittaci) in embryonated chicken eggs. United States of America, Department of Agriculture. Center for Veterinary Biologics Testing Protocol, pp.1–16. Al-Jumaa, Z.M., Jaber, M.T. and Al-Doori, A.A. 2024. Molecular detection of Chlamydophila felis from conjunctiva of cats infected with conjunctivitis and upper respiratory disease. Open. Vet. J. 14(12), 3289; doi:10.5455/OVJ.2024.v14.i12.13 Al-Nuaimy, W.A. and Al-Jandeel, T.J., 2018. Immunological and molecular study of Chlamydia trachomatis as causative agent of abortion in Al-Muthanna province. Iraqi J. Vet. Med. 42(1), 99–104. Al Sultani, S.W. 2025. Molecular detection and genotyping of Chlamydia psittaci in domestic pigeons and human contacts in Baghdad city. Kerbala J. Vet. Med. Sci. 1(Supplement I), 165–171. Azizpour, A. and Hassani, Y. 2012. Clinical evaluation of general anaesthesia in pigeons using a combination of ketamine and diazepam. J. South Afr. Vet. Assoc. 83(1), 1–4; doi:10.4102/jsava.v83i1.12 Balsamo, G., Maxted, A.M., Midla, J.W., Murphy, J.M., Wohrle, R., Edling, T.M., Fish, P.H., Flammer, K., Hyde, D., Kutty, P.K. and Kobayashi, M., 2017. Compendium of measures to control Chlamydia psittaci infection among humans (psittacosis) and pet birds (avian chlamydiosis), 2017. J. Avian Med. Surg. 31(3), 262–282. Bancroft, J.D. and Gamble, M. eds., 2008. Theory and practice of histological techniques. Elsevier health sciences. Billington, S. 2005. Clinical and zoonotic aspects of psittacosis. Pract 27(5), 256–263; doi:10.1136/inpract.27.5.256 Campbell, T.W. 2015. Evaluation of the blood film. Vet. Clinics. Exotic Anim. Pract. 18(1), 117–135; doi:10.1016/j.cvex.2014.09.001 Guo, W., Li, J., Kaltenboeck, B., Gong, J., Fan, W. and Wang, C. 2016. Chlamydia gallinacea, not C. psittaci, is the endemic chlamydial species in chicken (Gallus gallus). Scientific Rep. 6(1), 19638; doi:10.1038/srep19638 Hafez, R.M., Behour, T.S., Amer, H.M., Abdel-Maogood, S.Z. and Mousa, W.M.A. 2025. Molecular identification and histopathological findings of Neospora caninum as a cause of bovine abortion in some Egyptian dairy farms. J. Appl. Vet. Sci. 10(1), 46–56; doi:10.21608/javs.2024.327431.1446 Harkinezhad, T., Geens, T. and Vanrompay, D. 2009. Chlamydophila psittaci infections in birds: a review with emphasis on zoonotic consequences. Vet. Microbiol. 135(1-2), 68–77; doi:10.1016/j.vetmic.2008.09.046 Heijne, M., Van Der Goot, J., Buys, H., Dinkla, A., Roest, H.J., Van Keulen, L. and Koets, A. 2021. Experimental Chlamydia gallinacea infection in chickens does not protect against a subsequent experimental Chlamydia psittaci infection. Vet. Res. 52(1), 141; doi:10.1186/s13567-021-01011-y Jafaar Al-jandeel, T. 2018. Immunological and molecular study of Chlamydia trachomatis as causative agent of abortion in Al-Muthanna province. Iraqi. J. Vet. Med. 42(1), 99–104. Kaleta, E.F. and Taday, E.M.A. 2003. Avian host range of Chlamydophila spp. based on isolation, antigen detection and serology. Avian. Pathol. 32(5), 435–462; doi:10.1080/03079450310001593613 Knittler, M.R. and Sachse, K. 2015. Chlamydia psittaci: update on an underestimated zoonotic agent. Pathogens Dis. 73(1), 1–15; doi:10.1093/femspd/ftu007 Laroucau, K., Ortega, N., Vorimore, F., Aaziz, R., Mitura, A., Szymanska-Czerwinska, M., Cicerol, M., Salinas, J., Sachse, K. and Caro, M.R. 2020. Detection of a novel Chlamydia species in captive spur-thighed tortoises (Testudo graeca) in southeastern Spain and proposal of Candidatus Chlamydia testudinis. Systematic. Appl. Microbiol. 43(2), 126071; doi:10.1016/j.syapm.2020.126071 M. Al-darraji, A., H. Dhahir, S. and J. Alwan, M. 1992. Aetiological and pathological study on ovine and capring aborted foeti. Iraqi. J. Vet. Med. 16(1), 59–71; doi:10.30539/ijvm.v16i1.1614 Muench, H.R. 1938. A simple method of estimating 50 percent end points. Am. J. Hyg. 27, 493–497. Noomi, B.S. 2018. Comparative diagnostic study of avian salmonellosis in Salahaldeen province. Iraqi J. Agricult. Sci. 49(5), 49. Olsen, A.W., Lorenzen, E.K., Rosenkrands, I., Follmann, F. and Andersen, P. 2017. Protective effect of vaccine promoted neutralizing antibodies against the intracellular pathogen Chlamydia trachomatis. Front. Immunol. 8, 1652; doi:10.3389/fimmu.2017.01652 Reinhold, P., Ostermann, C., Liebler-Tenorio, E., Berndt, A., Vogel, A., Lambertz, J., Rothe, M., Rüttger, A., Schubert, E. and Sachse, K. 2012. A bovine model of respiratory Chlamydia psittaci infection: challenge dose titration. PLos One 7(1), e30125; doi:10.1371/journal.pone.0030125 Sachse, K., Laroucau, K. and Vanrompay, D. 2015. Avian chlamydiosis. Curr. Clin. Microbiol. Rep. 2(1), 10–21; doi:10.1007/s40588-014-0010-y Sariya, L., Prompiram, P., Tangsudjai, S., Poltep, K., Chamsai, T., Mongkolphan, C., Rattanavibul, K. and Sakdajivachareon, V. 2015. Detection and characterization of Chlamydophila psittaci in asymptomatic feral pigeons (Columba livia domestica) in central Thailand. Asian Pacific J. Trop. Med. 8(2), 94–97; doi:10.1016/S1995-7645(14)60195-4 Suwa, T. and Itakura, C. 1992. Ultrastructural studies of Chlamydia-infected air sacs of chicks. Avian Pathol. 21(3), 443–452; doi:10.1080/03079459208418862 Suwa, T., Ando, S., Hashimoto, N. and Itakura, C. 1990. Pathology of experimental chlamydiosis in chicks. Nihon. Juigaku. Zasshi. Japanese J. Vet. Sci. 52(2), 275–283; doi:10.1292/jvms1939.52.275 Takahashi, T., Takashima, I. and Hashimoto, N. 1988. A chicken model of systemic infection with Chlamydia psittaci: comparison of the virulence among avian and mammalian strains. Japanese J. Vet. Sci. 50(3), 622–631; doi:10.1292/jvms1939.50.622 Vilela, D.A.R., Marin, S.Y., Resende, M., Coelho, H.L.G., Resende, J.S., Ferreira-Junior, F.C., Ortiz, M.C., Araujo, A.V., Raso, T.D.F. and Martins, N.R.S. 2019. Phylogenetic analyses of Chlamydia psittaci ompA gene sequences from captive blue-fronted Amazon parrots (Amazona aestiva) with hepatic disease in Brazil. Rev. Sci. Tech. 38, 711–719. Walsh, A.L. and Shivaprasad, H.L. 2023. Unusual cases of chlamydiosis in psittacine birds. Vet. Pathol. 60(3), 360–368; doi:10.1177/03009858231162 Wu, S.M., Huang, S.Y., Xu, M.J., Zhou, D.H., Song, H.Q. and Zhu, X.Q. 2013. Chlamydia felis exposure in companion dogs and cats in Lanzhou, China: a public health concern. BMC Vet. Res. 9(1), 104; doi:10.1186/1746-6148-9-104 Yadav, R. and Sagar, M. 2022. Comparison of Different Histological Staining Methods for Detection of Helicobacter pylori Infection in Gastric Biopsy. Cureus 14(7), doi: 10.7759/cureus.27316 Yin, L., Kalmar, I., Chiers, K., Debyser, I. and Vanrompay, D. 2015. Chlamydia psittaci strains from broiler chickens induce histopathological lesions and mortality in SPF chickens. J. Vet. Res. 59(1), 43–50; doi:10.1515/bvip-2015-0007 Yin, L., Kalmar, I.D., Lagae, S., Vandendriessche, S., Vanderhaeghen, W., Butaye, P., Cox, E. and Vanrompay, D. 2013b. Emerging Chlamydia psittaci infections in the chicken industry and pathology of Chlamydia psittaci genotype B and D strains in specific pathogen free chickens. Vet. Microbiol. 162(2-4), 740–749; doi:10.1016/j.vetmic.2012.09.026 Yin, L., Lagae, S., Kalmar, I., Borel, N., Pospischil, A. and Vanrompay, D. 2013a. Pathogenicity of low and highly virulent Chlamydia psittaci isolates for specific-pathogen-free chickens. Avian. Dis. 57(2), 242–247; doi:10.1637/10439-102612-Reg.1 Zaręba-Marchewka, K., Szymańska-Czerwińska, M. and Niemczuk, K. 2020. Chlamydiae–what’s new?. J. Vet. Res. 64(4), 461. | ||
| How to Cite this Article |
| Pubmed Style Al-sultani SW, Al-doori AA. Histopathological study of the liver and spleen lesions in pigeons experimentally infected with Chlamydia psittaci. Open Vet. J.. 2026; 16(1): 346-353. doi:10.5455/OVJ.2026.v16.i1.31 Web Style Al-sultani SW, Al-doori AA. Histopathological study of the liver and spleen lesions in pigeons experimentally infected with Chlamydia psittaci. https://www.openveterinaryjournal.com/?mno=277064 [Access: January 31, 2026]. doi:10.5455/OVJ.2026.v16.i1.31 AMA (American Medical Association) Style Al-sultani SW, Al-doori AA. Histopathological study of the liver and spleen lesions in pigeons experimentally infected with Chlamydia psittaci. Open Vet. J.. 2026; 16(1): 346-353. doi:10.5455/OVJ.2026.v16.i1.31 Vancouver/ICMJE Style Al-sultani SW, Al-doori AA. Histopathological study of the liver and spleen lesions in pigeons experimentally infected with Chlamydia psittaci. Open Vet. J.. (2026), [cited January 31, 2026]; 16(1): 346-353. doi:10.5455/OVJ.2026.v16.i1.31 Harvard Style Al-sultani, S. W. & Al-doori, . A. A. (2026) Histopathological study of the liver and spleen lesions in pigeons experimentally infected with Chlamydia psittaci. Open Vet. J., 16 (1), 346-353. doi:10.5455/OVJ.2026.v16.i1.31 Turabian Style Al-sultani, Suhail Wadi, and Atheer Abdulrazzaq Al-doori. 2026. Histopathological study of the liver and spleen lesions in pigeons experimentally infected with Chlamydia psittaci. Open Veterinary Journal, 16 (1), 346-353. doi:10.5455/OVJ.2026.v16.i1.31 Chicago Style Al-sultani, Suhail Wadi, and Atheer Abdulrazzaq Al-doori. "Histopathological study of the liver and spleen lesions in pigeons experimentally infected with Chlamydia psittaci." Open Veterinary Journal 16 (2026), 346-353. doi:10.5455/OVJ.2026.v16.i1.31 MLA (The Modern Language Association) Style Al-sultani, Suhail Wadi, and Atheer Abdulrazzaq Al-doori. "Histopathological study of the liver and spleen lesions in pigeons experimentally infected with Chlamydia psittaci." Open Veterinary Journal 16.1 (2026), 346-353. Print. doi:10.5455/OVJ.2026.v16.i1.31 APA (American Psychological Association) Style Al-sultani, S. W. & Al-doori, . A. A. (2026) Histopathological study of the liver and spleen lesions in pigeons experimentally infected with Chlamydia psittaci. Open Veterinary Journal, 16 (1), 346-353. doi:10.5455/OVJ.2026.v16.i1.31 |