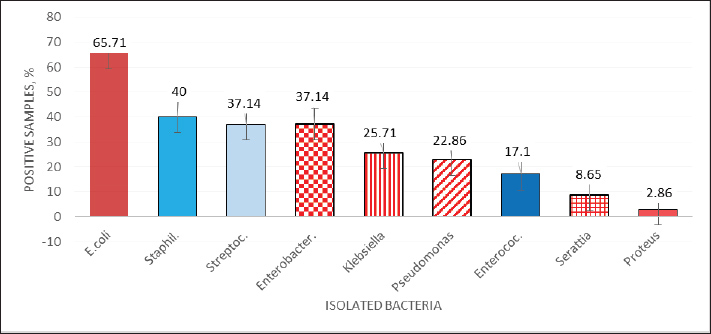
| Research Article | ||
Open Vet. J.. 2025; 15(12): 6351-6359 Open Veterinary Journal, (2025), Vol. 15(12): 6351-6359 Research Article Assessment of surface-associated bacterial contamination in Blatta orientalis from infested pig farms in BulgariaBetina Stefanova Boneva-Marutsova*, Plamen Dimitrov Marutsov and Georgi Zhelev GeorgievDepartment of Veterinary Microbiology, Infectious and Parasitic Diseases, Faculty of Veterinary Medicine, Trakia University, Stara Zagora, Bulgaria *Corresponding Author: Betina Stefanova Boneva-Marutsova. Department of Veterinary Microbiology, Infectious and Parasitic Diseases; Faculty of Veterinary Medicine, Trakia University, Stara Zagora, Bulgaria. Email: betina.boneva [at] trakia-uni.bg Submitted: 08/05/2025 Revised: 30/10/2025 Accepted: 12/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
AbstractBackground: Cockroaches are widely recognized as significant carriers of various infectious and parasitic diseases that affect both animals and humans. They are more than just common pests; they are among the most medically important insects due to their frequent presence in human environments. Understanding the role of cockroaches in disease transmission is crucial, and implementing effective pest control measures is essential. Aim: This study focuses on identifying bacterial contaminants present on the surfaces of Oriental cockroaches (Blatta orientalis) collected from pig farms. Methods: A total of 350 Oriental cockroaches collected from four field populations were examined in this study. Using the Vitek 2 Compact system, a modern platform for rapid and automated phenotypic identification, several bacterial species were isolated. Results: A total of nine bacterial taxa were identified, with Escherichia coli being the predominant species (65.7% of pooled samples), followed by Staphylococcus spp., Streptococcus spp., Enterobacter spp., and Klebsiella pneumoniae. Other less frequent isolates included Pseudomonas spp., Enterococcus spp., Serratia marcescens, and Proteus mirabilis. Conclusion: These results demonstrate that B. orientalis from pig farms harbors diverse bacterial contaminants of sanitary and veterinary concern, underlining the need for strict biosecurity and pest control measures to minimize the risk of disease transmission. Keywords: Bacteria, Blatta orientalis, Cockroaches, Swine, Vitek. IntroductionCockroaches are widely distributed insects of considerable economic, veterinary, and public health importance (Dent and Binks, 2020). Their extensive evolution has endowed them with remarkable adaptability, allowing survival in diverse environments, including extreme conditions, and enabling many species to thrive in human settlements, farms, and food industry facilities (Boneva-Marutsova, 2024). This synanthropic behavior raises critical concerns regarding their role in the transmission of infectious and parasitic (Alesho, 1997). Research has demonstrated that cockroaches are capable of carrying microorganisms and parasites on their surfaces, which positions them as significant vectors in the transmission of various infectious and parasitic diseases affecting both animals and humans (Vatev et al., 2006; Vahabi et al., 2007; Patel et al., 2022; Merad et al., 2023; Geng et al., 2025). Numerous studies identify them as primary carriers of food-borne pathogenic microorganisms (Gwenzi et al., 2021; Turner et al., 2023; Yulianti et al., 2023) and mechanical vectors (Luckyjane Molewa et al., 2022; Alhajeri et al., 2023; Davari et al., 2023; Abdullah et al., 2024). In light of the complex epidemic scenario related to the spread of African swine fever, an increasing number of researchers are raising concerns regarding the potential role of cockroaches and other insects in the dissemination of the disease (Fila and Woźniakowski, 2020; Yoon et al., 2020; Medrano, 2023). Pathogens can remain on the surface of cockroaches or in their digestive systems for over a month after coming into contact with contaminated environments, food, or water. These pathogens may be released into the environment through their chitinous exoskeletons or feces (Moges et al., 2016; Liu et al., 2024). Given their role in disease transmission, assessing surface microbial contamination in cockroach populations cohabiting with humans and animals is crucial, and specialized cultivation and identification methods have been developed for this purpose (Fang et al., 2013; Moges et al., 2016; Solomon et al., 2016; Alikhani et al., 2017; Haile et al., 2018; Guzman and Vilcinskas, 2020; Chen et al., 2023; Davari et al., 2023). Among contemporary techniques enabling rapid and automated phenotypic identification of microorganisms through colorimetric analysis, the Vitek 2 Compact system (BioMérieux, France) is increasingly utilized in clinical practice (Alyas et al., 2021). Our earlier studies on the distribution of cockroaches, conducted in different sites, established their presence in various livestock farms in Bulgaria. The highest prevalence of 100% was observed in industrial pig farms, where the degree of infestation was classified as high or very high. Particularly high levels of cockroach infestation have been observed in farrowing rooms, where they come into close contact with newborn piglets (Boneva-Marutsova, 2024), who are more susceptible to infectious diseases (Martínez-Boixaderas et al., 2022). The severity of the invasion raises concerns about its health implications regarding the spread of various infectious diseases. Currently, there is no information available about the bacterial carriage of the Oriental cockroach (Blatta orientalis) in intensive pig farms in Bulgaria, nor in many other countries in Eastern Europe. The aim of this study was to investigate bacterial surface carriage in field populations of cockroaches inhabiting pig farms in the Republic of Bulgaria to identify health and epidemic risks, using for the first time the VITEK 2 Compact system for their identification. Materials and MethodsA total of 350 Oriental cockroaches, collected from four industrial pig farms in Bulgaria heavily infested with high cockroach population densities, were examined in this study following the methodology of Solomon et al. (2016) and Davari et al. (2023). Sample collection was conducted between June and July 2023 during a single farm visit, employing live-capture methods. Cockroaches were trapped overnight and collected the next day with a specially adapted entomological bag. The highest population density was observed under the slatted floor of the farrowing house. Each farm yielded between 85 and 243 cockroaches, representing various developmental stages. Specimens were transported to the laboratory in boxes with tightly closing, locking lids, which were placed inside a larger container equipped with coolers to maintain suitable conditions during transit. During pool formation, all cockroaches that died during transport were removed. For each pool, cockroaches were randomly selected, excluding all immature stages. Due to an insufficient number of captured specimens, the number of pools was reduced to eight and seven for farms 2 and 4, respectively. Following euthanasia by supercooling, groups of ten cockroaches were placed in sterile plastic containers containing 5 ml of sterile saline to create pooled samples. Surface contamination was extracted from each pool by vigorous vortexing at low speed (600–800 rpm) for 120 seconds, following the protocols of Alikhani et al. (2017) and Haile et al. (2018). From the supernatant, 1 ml was pipetted into two sterile Eppendorf tubes and transported under refrigeration to a specialized microbiological laboratory. Samples were inoculated onto blood agar (Blood Agar Base, HiMedia Laboratories, India) and MacConkey agar (HiMedia Laboratories, India), and incubated aerobically at 37°C for 24 hours (Dimri et al., 2020; Mariam, 2021). The isolates were identified using the automated densitometric VITEK 2 Compact System (BioMérieux Inc., France), which is increasingly used as a complementary analytical approach for identifying microbes in various samples from humans and animals, including cockroaches (Bizzini et al., 2011; Rettinger et al., 2012; Pérez-Sancho et al., 2015; Singhal et al., 2016; Manukumar and Umesha, 2017; Mehainaoui et al., 2021; Sánchez-Juanes et al., 2022). Upon examination of the Petri dishes, fresh pure monocultures (18–24 hours incubation) were prepared from colonies with distinct morphology. Following Gram staining, the appropriate identification card was selected: GP card for Gram-positive bacteria or GN card for Gram-negative bacteria. From each monoculture (2–3 well-developed colonies), standard suspensions of 0.50–0.63 McFarland were prepared using a DensiChek™ meter. Results were obtained with the VITEK 2 Compact System (BioMérieux, France) and automatically interpreted by the accompanying software. Quality control was ensured using reference strains. Data analysisAll data were analyzed using IBM® SPSS® Statistics version 26.0. Categorical variables were compared among farms using the Chi-square test of independence. The strength of association between farm location and bacterial occurrence was evaluated using Cramér’s V coefficient, with thresholds interpreted as follows: 0.00–0.10 (negligible), 0.10–0.30 (weak), 0.30–0.50 (moderate), and >0.50 (strong association). Statistical significance was set at p < 0.05. Descriptive statistics (frequency, percentage) were used to summarize bacterial occurrence across farms. Ethical approvalNot needed for this study. ResultsMicrobiological analysis aimed at evaluating the epidemiological potential of synanthropic cockroaches as mechanical vectors of pathogens of sanitary-hygienic and veterinary relevance revealed considerable bacterial presence on their external surfaces (Table 1). Table 1. Surface bacterial contamination in Blatta orientalis collected from pig farms.
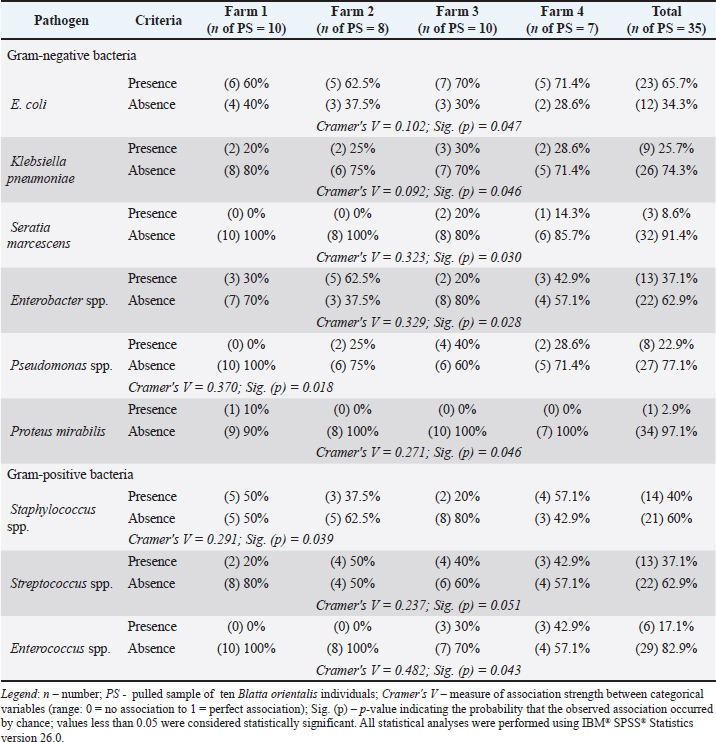
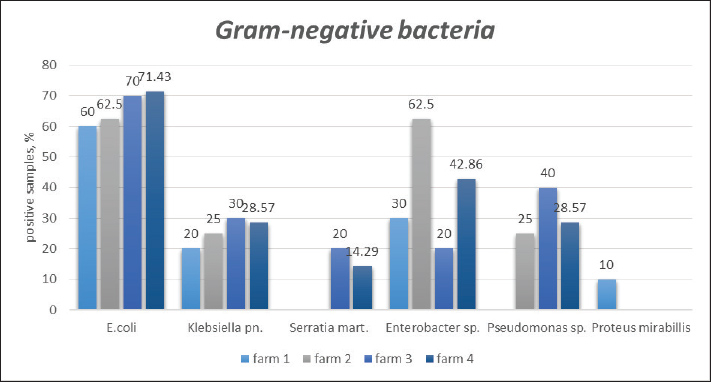
The quantitative distribution of GN and GP microorganisms isolated from the surface of synanthropic cockroaches collected from the surveyed pig farms is shown in Figures 1 and 2. The Cramér's V values, ranging from 0.092 to 0.482, indicate a weak to moderate association between bacterial species variability and farm location.
Fig. 1. Quantitative distribution of Gram-negative microorganisms isolated from Blatta orientalis across different pig farms.
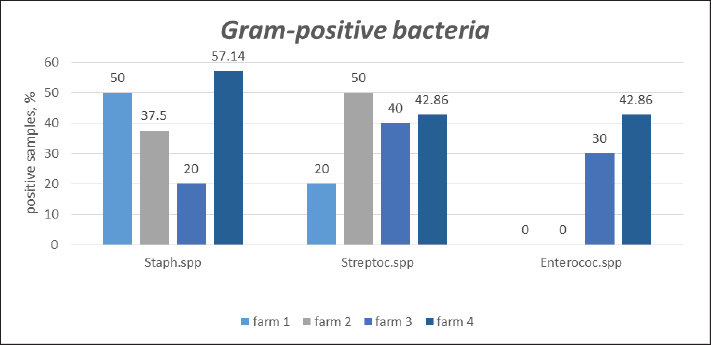
Fig. 2. Quantitative distribution of Gram-positive microorganisms isolated from Blatta orientalis across different pig farms. As shown in the summarized data in Figure 3, Escherichia coli was the predominant microbial isolate recovered from synanthropic cockroaches across all pig farms, followed by Staphylococcus spp., Streptococcus spp., and Enterobacter spp.
Fig. 3. Summary of the quantitative distribution of microorganisms isolated from Blatta orientalis across all pig farms. DiscussionCockroaches are recognized as sanitary and hygienic pests of high epidemiological importance, acting as vectors for numerous pathogenic agents. When inhabiting contaminated environments or consuming infected food and water, various viruses, bacteria, fungi, and protozoa may persist on their exoskeletons or within their digestive tracts for extended periods and be subsequently released into the environment (Moges et al., 2016; Liu et al., 2024; Crespo et al., 2025). Fang et al. (2013) and Chen et al. (2023) emphasize the critical need to assess microbial contamination in cockroach populations, particularly those cohabiting with humans and animals. Globally, most studies in this area have focused on German cockroaches and other synanthropic or exotic species. In Bulgaria, one of the few studies was conducted by Popova et al. (2010), also on an exotic cockroach species. However, there remains a substantial knowledge gap regarding the vectorial role of the black oriental cockroach, especially considering its frequent and severe infestations in pig farming facilities documented in our field observations. In our investigation, significant surface bacterial contamination was detected in oriental cockroaches from pig farms (Table 1). The isolates included nine microbial taxa: Escherichia coli, Staphylococcus spp., Streptococcus spp., Enterobacter spp., Klebsiella pneumoniae, Pseudomonas spp., Enterococcus spp., Serratia marcescens, and Proteus mirabilis. These microorganisms are considered conditionally pathogenic, forming part of the normal or transient flora of the skin, mucous membranes, and intestines of mammals and birds, yet capable of causing disease under specific conditions in humans and animals. Our findings are consistent with previous studies, such as those by Zarchi and Vatani (2009), who identified 19 bacterial species from cockroach surfaces and digestive tracts, including E. coli, Group D Streptococcus, Bacillus spp., Klebsiella pneumoniae, and Proteus vulgaris. Similarly, Khodabandeh et al. (2020) reported a dominant presence of Klebsiella spp., Pseudomonas spp., Proteus spp., Citrobacter spp., Enterobacter spp., and Serratia spp. in German cockroaches. A pronounced prevalence of Gram-negative bacteria, particularly from the Enterobacteriaceae family, was observed in our samples. This aligns with previous studies reporting similar bacterial profiles in German and American cockroach species and supports the potential role of cockroaches in spreading enteropathogens (Fotedar et al., 1991; Cloarec et al., 1992; Fotedar et al., 1992; Kobayashi et al., 1999; Pai et al., 2003; Magdy et al., 2023; Nirwan et al., 2024; Zainab Adesewa et al., 2024). Experimental studies by Zurek and Schal (2004) demonstrated that cockroaches shed large amounts of viable, virulent E. coli in their feces after initial exposure to contaminated material. Similarly, our study identified E. coli as the predominant contaminant on the surface of oriental cockroaches across all sampled farms. Pathogenic strains of E. coli are among the most frequently isolated pathogens in swine in veterinary practice (Fairbrother, 1999; Jacobson, 2022). Enterotoxigenic and verotoxigenic strains of E. coli are the main cause of colienteritis and colienterotoxicosis in newborn and weaned pigs, as well as edematous disease in postweaned pigs, often with high mortality (Bertschinger, 1999; Moon et al., 1999; Bassi et al., 2023). Waldvogel et al. (1999) reported the persistence of viable E. coli F18 strains in cockroach feces for up to 8 days’ post-exposure, with high bacterial loads (4.4 × 105 CFU g−1) comparable to those found in infected pigs (1.9 × 106 CFU g−1). This underscores the vector potential of cockroaches for these pathogens. Therefore, targeted cockroach density control is recommended as an integral part of disease prevention and control programs in pig farming (Waldvogel et al., 1999; Boneva et al., 2023). Staphylococcus spp. and Streptococcus spp. were the next most frequently isolated genera in our study. Numerous investigations have highlighted the potential role of cockroaches as vectors of bacterial pathogens, particularly these two genera. Their frequent isolation from cockroach populations suggests a possible route for transmission to humans and animals. Studies by Fotedar et al. (1991) in India, Salehzadeh et al. (2007) in Iran, and Vazirianzadeh et al. (2009) have reported significant associations between cockroach infestations and the presence of pathogenic bacteria. Additionally, Schauer et al. (2012, 2014), Lampert et al. (2019), and Petrovskiy et al. (2024) identified high concentrations of staphylococci in various cockroach species, especially in the hindgut of Shelfordella lateralis. Of particular concern is S. aureus, a bacterium colonizing the skin and mucosal surfaces of approximately 30% of the human population, known for its ability to cause serious infections (Gorwitz et al., 2008). The increasing detection of antibiotic-resistant S. aureus strains in cockroaches, as documented by Menasria et al. (2014); Islam et al. (2016); Abdolmaleki et al. (2019); Akter et al. (2023); Yun et al. (2024), underscores their growing importance as a public health threat. Our investigation revealed concerning levels of Klebsiella pneumoniae contamination across all surveyed pig farms. These findings are consistent with those of Cotton et al. (2000), who reported a high prevalence of Klebsiella in German cockroaches. Their work highlights the critical role of cockroaches as vectors in the transmission of nosocomial infections, emphasizing the urgent need for comprehensive pest control strategies in both agricultural and healthcare environments. In one of the studied farms, Proteus mirabilis was detected on the surfaces of examined cockroaches. This bacterium has also been identified in the gastrointestinal tracts of German and American cockroaches (Robertson, 2007; Kundera et al., 2020), as well as in exotic species (Popova et al., 2010). Additionally, the presence of Pseudomonas aeruginosa was confirmed an opportunistic pathogen frequently isolated from various cockroach species, including those implicated in hospital infestations (Saitou et al., 2009). Pseudomonas aeruginosa is particularly notable for its high resistance to disinfectants and its prominent role in healthcare-associated infections. The data unequivocally highlight cockroach monitoring and population control as crucial components of biosecurity programs, essential both for preserving animal health and for reducing the direct health risks to farm workers from potential exposure to cockroach-borne pathogens. The implementation of integrated pest management—encompassing physical, chemical, and organizational measures, alongside the maintenance of strict sanitary hygiene—is strongly recommended. A potential limitation of this study is the lack of direct evidence demonstrating the transmission of pathogens between cockroaches and pigs on intensive farms, such as through molecular genetic analyses. Future research could address this gap by examining the impact of cockroach infestations on pig health across farms with varying levels of infestation (low, medium, high), focusing particularly on the incidence of neonatal diarrhea in farrowing facilities. The health risks associated with cockroaches, especially their role in maintaining and transmitting pathogens, become more pronounced during disease outbreaks. In such situations, control measures—like removing animals, cleaning feeders and waterers, and performing intensive disinfection with strong-smelling agents—often lead to the mass migration of cockroaches to nearby, unaffected facilities (Koehler et al., 2022). Therefore, implementing effective cockroach control should be an integral part of herd health management programs on pig farms. Conducting future in-depth studies would provide a more comprehensive understanding of the epidemiological significance of cockroaches and help inform targeted biosecurity and pest management strategies. ConclusionThe findings of this study highlight significant sanitary and epidemiological risks associated with cockroach infestations in livestock production and food processing environments. In particular, the detection of Blatta orientalis in swine facilities was strongly associated with high levels of bacterial surface contamination. Escherichia coli was identified as the predominant microbial isolate, followed by Staphylococcus spp., Streptococcus spp., Enterobacter spp., Klebsiella pneumoniae, Pseudomonas spp., Enterococcus spp., Serratia marcescens, and Proteus mirabilis, reflecting a diverse and complex microbial profile. These results underscore the necessity of implementing strict hygiene and biosecurity measures to mitigate the risk of pathogen transmission in such settings. Conflicts of interestThe authors declare no conflicts of interest. FundingThis research received no external funding. Authors' contributionsConceptualization, B.B-M., P.M. and G.Z.; methodology, B.B-M., P.M. and G.Z.; resources, B.B-M., P.M. and G.Z; writing—original draft preparation, B.B-M., P.M. and G.Z.; writing—review and editing, B.B-M., P.M. and G.Z.; visualization, B.B-M., P.M. and G.Z. All authors have read and agreed to the published version of the manuscript. Data availabilityThe original contributions presented in this manuscript are included in the article. Further inquiries can be directed to the corresponding author. ReferencesAbdolmaleki, Z., Mashak, Z. and Safarpoor Dehkordi, F. 2019. Phenotypic and genotypic characterization of antibiotic resistance in the methicillin-resistant Staphylococcus aureus strains isolated from hospital cockroaches. Antimicrob. Resist. Infect. Control. 8(1), 1–14. Abdullah, A.M., Merza, A.S. and Meerkhan, A.A. 2024. Cockroaches as carriers of human medically important parasites. Parasitol. Res. 123(2), 119. Akter, M.S., Islam, M.S., Mohanta, M.K. and Parvin, S. 2023. Antibiogram and MDR pattern of the bacterial isolates from German cockroaches (Blattella germanica L.) at RMCH, Rajshahi, Bangladesh. J. Sci. Res. 15(2), 559–570. Alesho, N.A. 1997. Synanthropic cockroaches of Russia. Proc. Int. Colloq. Social Insects. 3, 45–50. Alhajeri, N., Alharbi, J. and El-Azazy, O. 2023. The potential role of the American cockroach (Periplaneta americana) as a vector of Enterobacteriaceae in Kuwait. Egypt. Acad. J. Biol. Sci. E. Med. Entomol. Parasitol. 15(1), 11–20. Alikhani, M.Y., Parsavash, S., Arabestani, M.R. and Hosseini, S.M. 2017. Prevalence of antibiotic resistance and class 1 integrons in clinical and environmental isolates of Pseudomonas aeruginosa. Avicenna J. Clin. Microbiol. Infect. 4(4), 12086. Alyas, S.S., Ibrahim, R.K. and Kareem, A.A. 2021. Study the pathogenicity of the bacteria associated with Periplaneta americana cockroach on Galleria mellonella L. worm larva. J Phys Conf Ser, IOP Publishing, 1999(1), 12032. Bassi, P., Bosco, C., Bonilauri, P., Luppi, A., Fontana, M.C., Fiorentini, L. and Rugna, G. 2023. Antimicrobial resistance and virulence factors assessment in Escherichia coli isolated from swine in Italy from 2017 to 2021. Pathogens 12(1), 112. Bertschinger, H.U. 1999. Postweaning Escherichia coli diarrhea and edema disease.In Diseases of Swine. Straw, B.E., D’Allaire, S., Mengeling, W.L. and Taylor, D.J Ames: Iowa State Univ Press, pp: 441–54. Bizzini, A., Jaton, K., Romo, D., Bille, J., Prod'Hom, G. and Greub, G. 2011. Matrix-assisted laser desorption ionization–time of flight mass spectrometry as an alternative to 16S rRNA gene sequencing for identification of difficult-to-identify bacterial strains. J. Clin. Microbiol. 49, 693–696. Boneva, B., Marutsov, P. and Zhelev, G. 2023. A survey of the distribution of synanthropic cockroaches in animal farms and food processing plants in Bulgaria. Trakia J. Sci. 21(3), 217. Boneva-Marutsova, B. 2024. Study on the distribution and evaluation of the efficiency of the control methods of synanthropic cockroach species in animal farms and food processing plants in Bulgaria. Ph. D. thesis, Trakia University, Stara Zagora, Bulgaria. Chen, Z., Wen, S., Shen, J., Wang, J., Liu, W. and Jin, X. 2023. Composition and diversity of the gut microbiota across different life stages of American cockroach (Periplaneta americana). Bull. Entomol. Res. 113(6), 787–793. Cloarec, A., Rivault, C., Fontaine, F. and Le Guyader, A. 1992. Cockroaches as carriers of bacteria in multi-family dwellings. Epidemiol. Infect. 109(3), 483–490. Cotton, M.F., Wasserman, E., Pieper, C.H., Theron, D.C., Tubbergh, D.V., Campbell, G., Fang, F.C. and Barnes, J. 2000. Invasive disease due to extended spectrum beta-lactamase-producing Klebsiella pneumoniae in a neonatal unit: the possible role of cockroaches. J. Hosp. Infect. 44(1), 13–17. Crespo, A., De Armas, Y., Capó, V., Iglesias, E., Palomares-Marín, J., Fonte, L., Plascencia-Hernández, A., Cueto-Aragón, C.L., Calderón, E.J. and Pérez-Gómez, H.R. 2025. Pathogens of medical importance identified in hospital-collected cockroaches: a systematic review. Microorganisms 13(2), 337. Davari, B., Hassanvand, A.E., Salehzadeh, A., Alikhani, M.Y. and Hosseini, S.M. 2023. Bacterial contamination of collected cockroaches and determination of their antibiotic susceptibility in Khorramabad City, Iran. J. Arthropod. Borne. Dis. 17(1), 63–71. Dent, D. and Binks, R.H. 2020. Insect Pest Management. London, UK: CABI. Dimri, A.G., Chaudhary, S., Singh, D., Chauhan, A. and Aggarwal, M.L. 2020. Morphological and biochemical characterization of food borne gram-positive and gram-negative bacteria. Sci. Arch. 1(1), 16–23. Fairbrother, J.M. 1999. Neonatal Escherichia coli diarrhea.In Diseases of Swine. Straw, B.E., D’Allaire, S., Mengeling, W.L. and Taylor, D.J Ames: Iowa State Univ Press, pp: 433–41. Fang, W., Fang, Z., Liu, Z., Yuan, J., Zhang, X., Peng, H., Hong, Y. and Xiao, Y. 2013. Phylogenetic analysis of bacterial community in the gut of American cockroach (Periplaneta americana). Wei. Sheng. Wu. Xue. Bao.=. Acta Microbiol. Sin. 53(9), 984–994. Fila, M. and Woźniakowski, G. 2020. African swine fever virus – the possible role of flies and other insects in virus transmission. J. Vet. Res. 64, 1–7. Fotedar, R., Banerjee, U. and Samantray, J.C. 1992. Vector potential of hospital houseflies with special reference to Klebsiella species. Epidemiol. Infect. 109(1), 143. Fotedar, R., Shriniwas, U.B. and Verma, A. 1991. Cockroaches (Blattella germanica) as carriers of microorganisms of medical importance in hospitals. Epidemiol. Infect. 107(1), 181–187. Geng, D., Yu, H., Zhao, T. and Li, C. 2025. The medical importance of cockroaches as vectors of pathogens: implications for public health. Zoonoses 5(1), 982. Gorwitz, R.J., Kruszon-Moran, D., McAllister, S.K., McQuillan, G., McDougal, L.K., Fosheim, G.E., Jensen, B.J., Killgore, G., Tenover, F.C. and Kuehnitz, M.J. 2008. Changes in the prevalence of nasal colonization with Staphylococcus aureus in the United States, 2001–2004. J. Infect. Dis. 197(9), 1226–1234. Guzman, J. and Vilcinskas, A. 2020. Bacteria associated with cockroaches: health risk or biotechnological opportunity?. Appl. Microbiol. Biotechnol. 104(24), 10369–10387. Gwenzi, W., Chaukura, N., Muisa-Zikali, N., Teta, C., Musvuugwa, T., Rzymski, P. and Abia, A.L.K. 2021. Insects, rodents, and pets as reservoirs, vectors, and sentinels of antimicrobial resistance. Antibiotics 10(1), 68. Haile, T., Mariam, A.T., Kiros, S. and Teffera, Z. 2018. Cockroaches as carriers of human gastrointestinal parasites in Wolkite Town, southwestern Ethiopia. J. Parasitol. Vector Biol. 10(2), 33–38. Islam, A., Nath, A., Islam, K., Islam, S., Chakma, S., Hossain, M., Faruq, A. and Hassan, M. 2016. Isolation, identification and antimicrobial resistance profile of Staphylococcus aureus in cockroaches (Periplaneta americana). J. Adv. Vet. Anim. Res. 3(3), 221–228. Jacobson, M. 2022. On the infectious causes of neonatal piglet diarrhoea—a review. Vet. Sci. 9(8), 422. Khodabandeh, M., Shirani-Bidabadi, L., Madani, M. and Zahraei-Ramazani, A. 2020. Study on Periplaneta americana (Blattodea: blattidae) fungal infections in hospital sewer system, Esfahan City, Iran, 2017. J. Pathog. 1(1), 4296720. Kobayashi, M., Sasaki, T., Saito, N., Tamura, K., Suzuki, K., Watanabe, H. and Agui, N. 1999. Houseflies: not simple mechanical vectors of enterohemorrhagic Escherichia coli O157:H7. Am. J. Trop. Med. Hyg. 61(4), 625–629. Koehler, P.G., Bayer, B.E. and Branscome, D. 2022. Cockroaches and their management. Gainesville: University of Florida IFAS extension. Kundera, I.N., Sapu, E.H. and Bialangi, M. 2020. Identification of bacteria on cockroach feet (Periplaneta americana) in Resident Bay of Palu Permai and sensitivity test against antibiotics. Techno. J. Penelitian. 9(1), 353–362. Lampert, N., Mikaelyan, A. and Brune, A. 2019. Diet is not the primary driver of bacterial community structure in the gut of litter-feeding cockroaches. BMC. Microbiol. 19(1), 238. Liu, J., Yuan, Y., Feng, L., Lin, C., Ye, C., Liu, J. and Liu, H. 2024. Intestinal pathogens detected in cockroach species within different food-related environments in Pudong, China. Sci. Rep. 14(1), 1–10. Luckyjane Molewa, M., Barnard, T. and Naicker, N. 2022. A potential role of cockroaches in the transmission of pathogenic bacteria with antibiotic resistance: a scoping review. J. Infect. Dev. Ctries. 16(11), 1671–1678. Magdy, H., Rady, M.H., Salama, M.S., Sayed, H.A., Hamza, D., Azzam, M. and Essa, E.E. 2023. Isolation of multidrug-resistant Helicobacter pylori from wild houseflies Musca domestica with a new perspective for the treatment. Vector. Borne. Zoonotic. Dis. 23(2), 63–74. Manukumar, H.M. and Umesha, S. 2017. MALDI-TOF-MS based identification and molecular characterization of food associated methicillin-resistant Staphylococcus aureus. Sci. Rep. 7, 1–16. Mariam, S.H. 2021. Isolation and characterization of Gram-negative bacterial species from pasteurized dairy products: potential risk to consumer health. J. Food. Qual. 17(1), 1–10. Martínez-Boixaderas, N., Garza-Moreno, L., Sibila, M. and Segalés, J. 2022. Impact of maternally derived immunity on immune responses elicited by piglet early vaccination against the most common pathogens involved in porcine respiratory disease complex. Porcine Health Manag. 8(1), 11. Medrano, M. 2023. A literature review to gather the scientific evidence for an African swine fever virus (ASFV) exposure assessment of US domestic pigs raised in total confinement and/or with outdoor access to ASFV-infected feral swine. Minnesota: University of Minnesota. Mehainaoui, A., Menasria, T., Benouagueni, S., Benhadj, M., Lalaoui, R. and Gacemi-Kirane, D. 2021. Rapid screening and characterization of bacteria associated with hospital cockroaches (Blattella germanica L.) using MALDI-TOF mass spectrometry. J. Appl. Microbiol. 130(3), 960–970. Menasria, T., Tine, S., Souad, E., Mahcene, D., Moussa, F., Benammar, L. and Mekahlia, M.N. 2014. A survey of the possible role of German cockroaches as a source for bacterial pathogens. J. Adv. Sci. Appl. Eng. 1(1), 67–70. Merad, Y., Belkacemi, M., Merad, Z., Bassaid, A., Benmansour, Z., Matmour, D. and Belmokhtar, Z. 2023. Fungal carriage of hospital trapped cockroaches: a prospective study. New Microbes New Infect. 52, 101086. Moges, F., Eshetie, S., Endris, M., Huruy, K., Muluye, D., Feleke, T., G/Silassie, F., Ayalew, G. and Nagappan, R. 2016. Cockroaches as a source of high bacterial pathogens with multidrug resistant strains in Gondar Town, Ethiopia. Biomed. Res. Int. 2016, 2825056. Moon, H.W., Hoffman, L.J., Cornick, N.A., Booher, S.L. and Bosworth, B.T. 1999. Prevalence of some virulence genes among Escherichia coli isolates from swine presented to a diagnostic laboratory in Iowa. J. Vet. Diagn. Invest. 11, 557–560. Nirwan, M., Rafiuddin, A.T. and Amraeni, Y. 2024. Cockroach biodiversity and the application of biosecurity in regional and private public hospitals in Kendari City, Southeast Sulawesi. ASPIRATOR. J. Vector. Borne. Dis. Stud. 15(2), 37–52. Pai, H.H., Chen, W.C. and Peng, C.F. 2003. Isolation of non-tuberculous mycobacteria from hospital cockroaches (Periplaneta americana). J. Hosp. Infect. 53(3), 224–228. Patel, A., Jenkins, M., Rhoden, K. and Barnes, A.N. 2022. A systematic review of zoonotic enteric parasites carried by flies, cockroaches, and dung beetles. Pathogens 11, 90. Pérez-Sancho, M., Vela, A.I., García-Seco, T., Gottschalk, M., Domínguez, L. and Fernández-Garayzábal, J.F. 2015. Assessment of MALDI-TOF MS as alternative tool for Streptococcus suis identification. Front. Public Health 3, 202. Petrovskiy, A.B., Raldugina, A.O. and Reshetnikov, A.N. 2024. Turkestan cockroach Periplaneta lateralis Walker, 1868—A new alien species in Russia. Russ. J. Biol. Invasions 15(4), 606–613. Popova, T., Trencheva, K. and Tomov, R. 2010. Investigation on the microflora of the Brazilian cockroach Blaberus giganteus (L.) (Blattodea: blaberidae) with a view to assess its epizootiological significance. Ecol. Future J. Agric. Sci. Forest. Sci. 9(3), 30–33. Rettinger, A., Krupka, I., Grünwald, K., Dyachenko, V., Fingerle, V., Konrad, R., Raschel, H., Busch, U., Sing, A., Straubinger, R.K. and Huber, I. 2012. Leptospira spp. strain identification by MALDI TOF MS is an equivalent tool to 16S rRNA gene sequencing and multilocus sequence typing (MLST). BMC. Microbiol. 12, 185. Robertson, A.R. 2007. The isolation and characterization of the microbial flora in the alimentary canal of Gromphadorhina portentosa based on rDNA sequences. M.S. thesis, East Tennessee State Univ., Johnson City, TN. Saitou, K., Furuhata, K., Kawakami, Y. and Fukuyama, M. 2009. Biofilm formation abilities and disinfectant-resistance of Pseudomonas aeruginosa isolated from cockroaches captured in hospitals. Biocontrol. Sci. 14(2), 65–68. Salehzadeh, A., Tavacol, P. and Mahjub, H. 2007. Bacterial, fungal and parasitic contamination of cockroaches in public hospitals of Hamadan, Iran. J. Vector. Borne. Dis. 44, 105–110. Sánchez-Juanes, F., Calvo Sánchez, N., Belhassen García, M., Vieira Lista, C., Román, R.M., Álamo Sanz, R., Muro Álvarez, A. and Muñoz Bellido, J.L. 2022. Applications of MALDI-TOF mass spectrometry to the identification of parasites and arthropod vectors of human diseases. Microorganisms 10(11), 2300. Schauer, C., Thompson, C. and Brune, A. 2014. Pyrotag sequencing of the gut microbiota of the cockroach Shelfordella lateralis reveals a highly dynamic core but only limited effects of diet on community structure. PLos One 9(1), e85861. Schauer, C., Thompson, C.L. and Brune, A. 2012. The bacterial community in the gut of the cockroach Shelfordella lateralis reflects the close evolutionary relatedness of cockroaches and termites. Appl. Environ. Microbiol. 78(8), 2758–2767. Singhal, N., Kumar, M. and Virdi, J.S. 2016. MALDI-TOF MS in clinical parasitology: applications, constraints and prospects. Parasitology 143, 1491–1500. Solomon, F., Belayneh, F., Kibru, G. and Ali, S. 2016. Vector potential of Blattella germanica (L.) (Dictyoptera: blattidae) for medically important bacteria at food handling establishments in Jimma town, Southwest Ethiopia. Biomed Res. Int. 2016, 3490906. Turner, M., Van Hulzen, L., Peta, V. and Pietri, J.E. 2023. Survival of Salmonella Typhimurium in the hemolymph of the German cockroach vector is limited by both humoral immune factors and hemocytes but not by trehalose metabolism. J. Med. Entomol. 60(5), 875–883. Vahabi, A., Rafinejad, J., Mohammadi, P. and Biglarian, F. 2007. Regional evaluation of bacterial contamination in hospital environment cockroaches. J. Environ. Health Sci. Eng. 4, 57–60. Vatev, N.T., Kevorkyan, A.K., Rakadzhieva, T.A. and Stoilova, Y.D. 2006. Manual for Practical Exercises in the Epidemiology of Infectious Diseases. Raikov Publishing, pp. 26–27. Vazirianzadeh, B., Mehdinejad, M. and Dehghani, R. 2009. Identification of bacteria which possible transmitted by Polyphaga aegyptica (Blattodea: blattidae) in the region of Ahvaz, SW Iran. Jundishapur J. Microbiol. 2(1), 36–40. Waldvogel, M.G., Moore, C.B., Nalyanya, G.W., Stringham, S.M., Watson, D.W. and Schal, C.O.B.Y. Integrated cockroach (Dictyoptera: blattellidae) management in confined swine production. In Proc 3rd Int Conf Urban Pests,1999 Graficke Zavody Hronov, Prague, pp 183–188. Yoon, H., Hong, S.K., Lee, I. and Lee, E.S. 2020. Insects as potential vectors of African swine fever virus in the Republic of Korea. Authorea. Yulianti, D.M., Hikam, A.R., Ambarningrum, T.B. and Satwika, T.D. 2023. Detection of pathogen foodborne disease bacteria Staphylococcus aureus from German cockroach (Blattella germanica) in the hospital area. IOP Conf Ser Earth Environ Sci, 1230(1), 12–85. Yun, S., Choi, J.H., Oh, S., Kim, M., Yi, M.H., Kang, D., Jang, Y.S., Lee, I.Y., Yong, T.S., Kim, J., Kim, H.C., Lee, J.R. and Kim, J.Y. 2024. Microbiome of laboratory-reared and environmentally collected cockroaches. Entomol. Res. 54(4), e12727. Zainab Adesewa, A., Fatima, O., Ganiu Dauda, O. and Kolawole Aremu, H. 2024. Bacterial communities of the Musca domestica housefly in different urban habitats and their antimicrobial susceptibility profiles. Afr. J. Pure. Appl. Sci. 5(1) 142–160. Zarchi, A.A.K. and Vatani, H. 2009. A survey on species and prevalence rate of bacterial agents isolated from cockroaches in three hospitals. Vector. Borne. Zoonotic. Dis. 9(2), 197–200. Zurek, L. and Schal, C. 2004. Evaluation of the German cockroach (Blattella germanica) as a vector for verotoxigenic Escherichia coli F18 in confined swine production. Vet. Microbiol. 101(4), 263–267. | ||
| How to Cite this Article |
| Pubmed Style Boneva-marutsova BS, Marutsov PD, Georgiev GZ. Assessment of surface-associated bacterial contamination in Blatta orientalis from infested pig farms in Bulgaria. Open Vet. J.. 2025; 15(12): 6351-6359. doi:10.5455/OVJ.2025.v15.i12.18 Web Style Boneva-marutsova BS, Marutsov PD, Georgiev GZ. Assessment of surface-associated bacterial contamination in Blatta orientalis from infested pig farms in Bulgaria. https://www.openveterinaryjournal.com/?mno=275507 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.18 AMA (American Medical Association) Style Boneva-marutsova BS, Marutsov PD, Georgiev GZ. Assessment of surface-associated bacterial contamination in Blatta orientalis from infested pig farms in Bulgaria. Open Vet. J.. 2025; 15(12): 6351-6359. doi:10.5455/OVJ.2025.v15.i12.18 Vancouver/ICMJE Style Boneva-marutsova BS, Marutsov PD, Georgiev GZ. Assessment of surface-associated bacterial contamination in Blatta orientalis from infested pig farms in Bulgaria. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6351-6359. doi:10.5455/OVJ.2025.v15.i12.18 Harvard Style Boneva-marutsova, B. S., Marutsov, . P. D. & Georgiev, . G. Z. (2025) Assessment of surface-associated bacterial contamination in Blatta orientalis from infested pig farms in Bulgaria. Open Vet. J., 15 (12), 6351-6359. doi:10.5455/OVJ.2025.v15.i12.18 Turabian Style Boneva-marutsova, Betina Stefanova, Plamen Dimitrov Marutsov, and Georgi Zhelev Georgiev. 2025. Assessment of surface-associated bacterial contamination in Blatta orientalis from infested pig farms in Bulgaria. Open Veterinary Journal, 15 (12), 6351-6359. doi:10.5455/OVJ.2025.v15.i12.18 Chicago Style Boneva-marutsova, Betina Stefanova, Plamen Dimitrov Marutsov, and Georgi Zhelev Georgiev. "Assessment of surface-associated bacterial contamination in Blatta orientalis from infested pig farms in Bulgaria." Open Veterinary Journal 15 (2025), 6351-6359. doi:10.5455/OVJ.2025.v15.i12.18 MLA (The Modern Language Association) Style Boneva-marutsova, Betina Stefanova, Plamen Dimitrov Marutsov, and Georgi Zhelev Georgiev. "Assessment of surface-associated bacterial contamination in Blatta orientalis from infested pig farms in Bulgaria." Open Veterinary Journal 15.12 (2025), 6351-6359. Print. doi:10.5455/OVJ.2025.v15.i12.18 APA (American Psychological Association) Style Boneva-marutsova, B. S., Marutsov, . P. D. & Georgiev, . G. Z. (2025) Assessment of surface-associated bacterial contamination in Blatta orientalis from infested pig farms in Bulgaria. Open Veterinary Journal, 15 (12), 6351-6359. doi:10.5455/OVJ.2025.v15.i12.18 |